Engineering of a Biomimetic Interface between a Native Dental Tissue and Restorative Composite and Its Study Using Synchrotron FTIR Microscopic Mapping
Abstract
1. Introduction
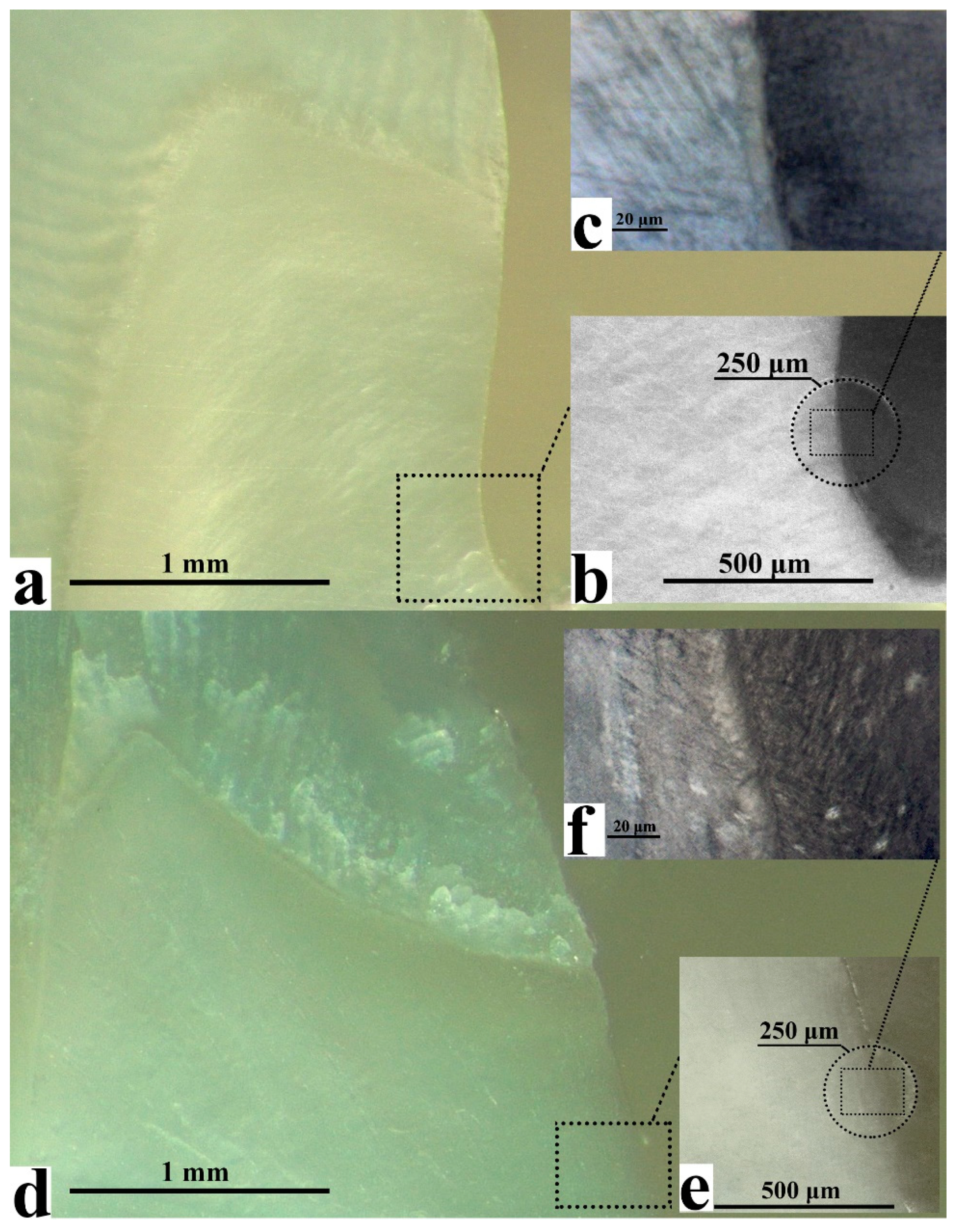
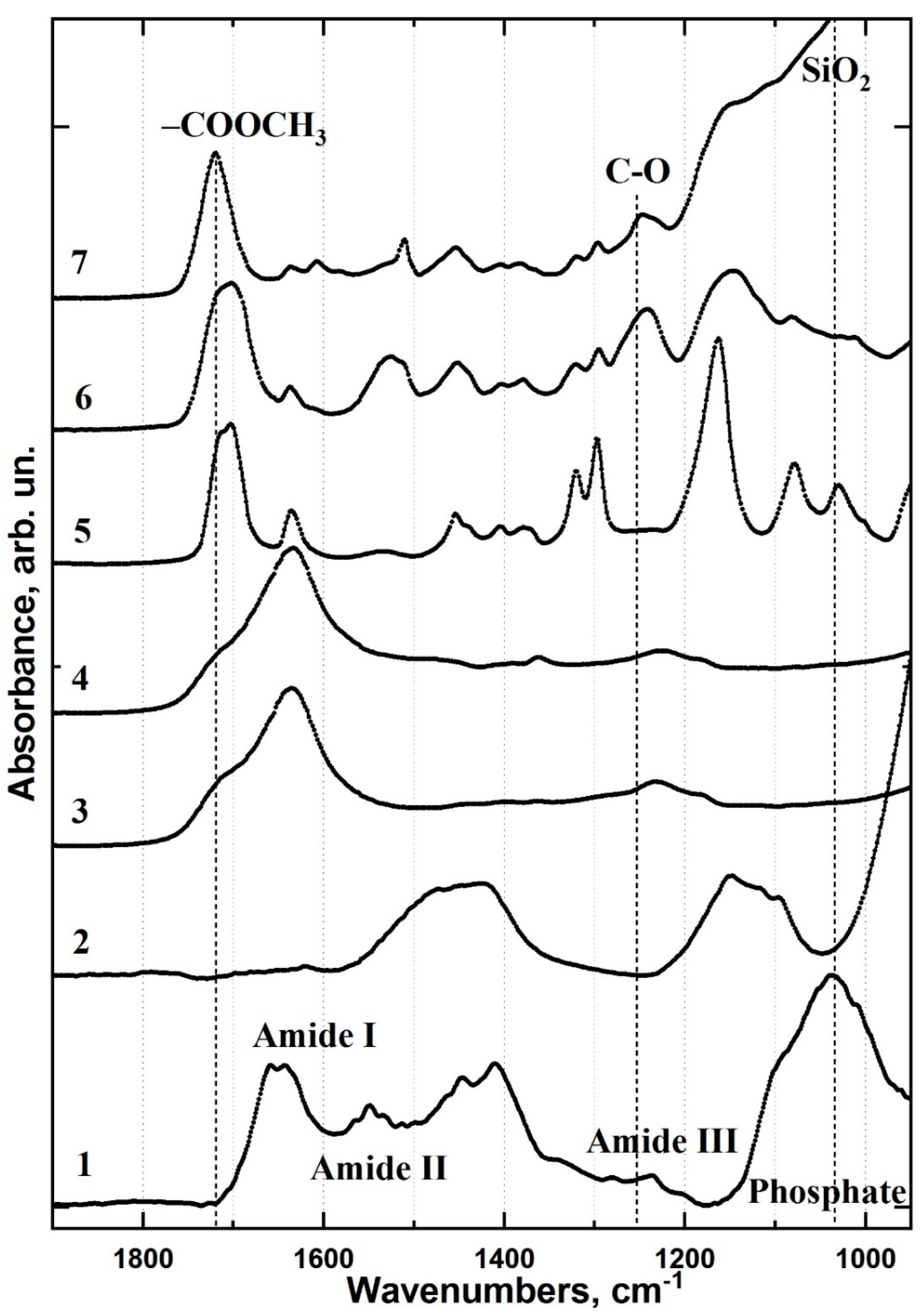
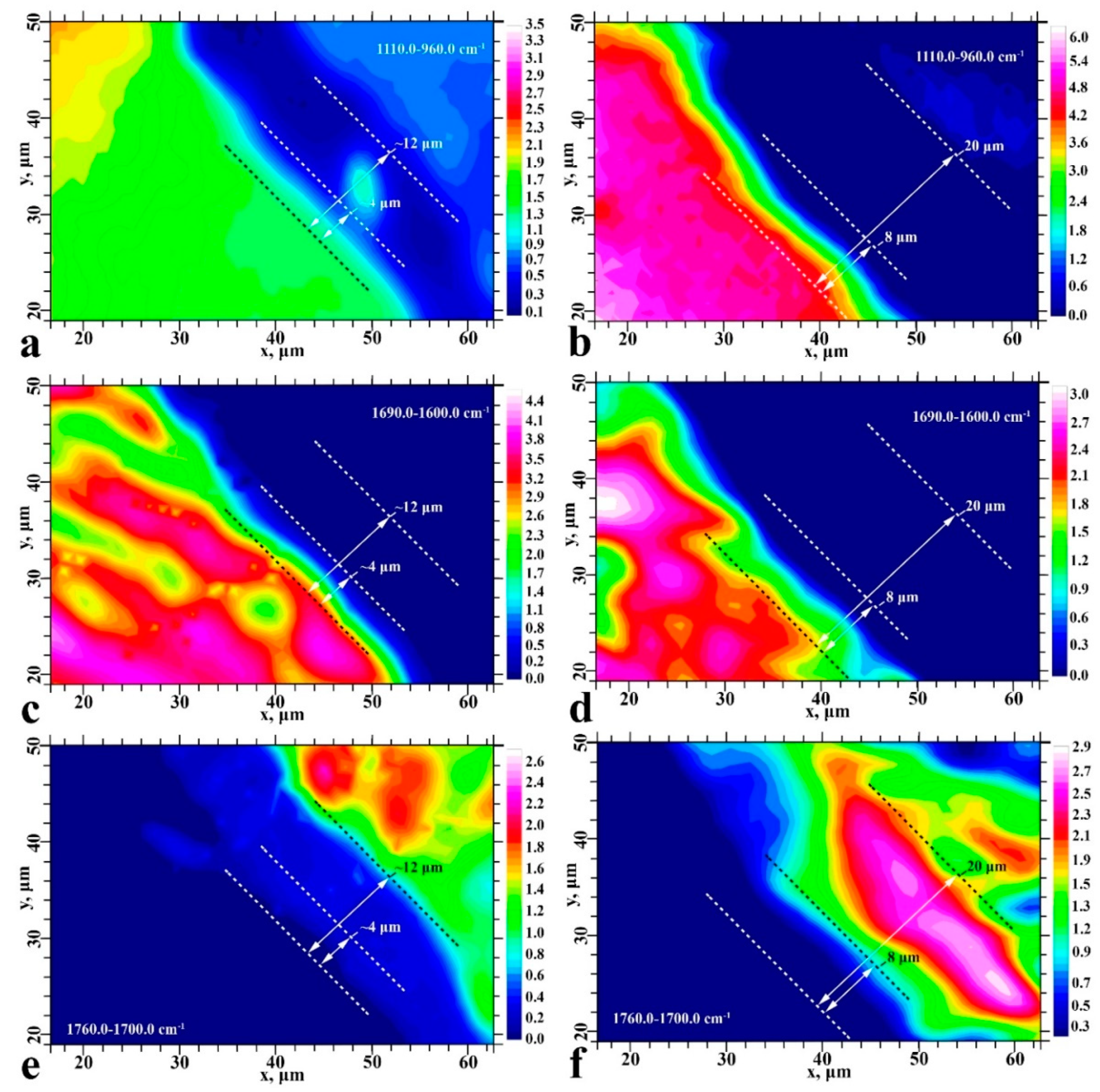
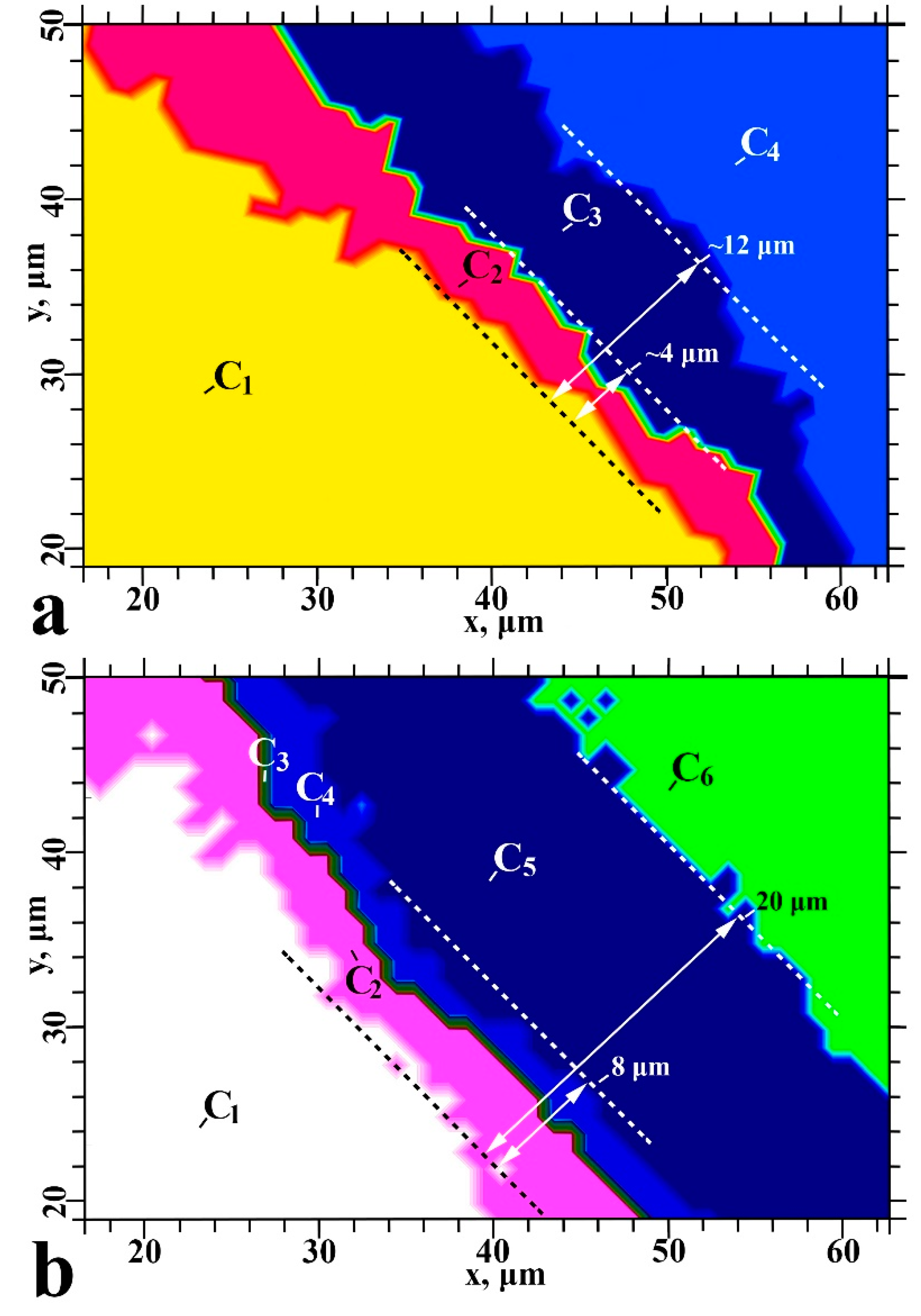
2. Results
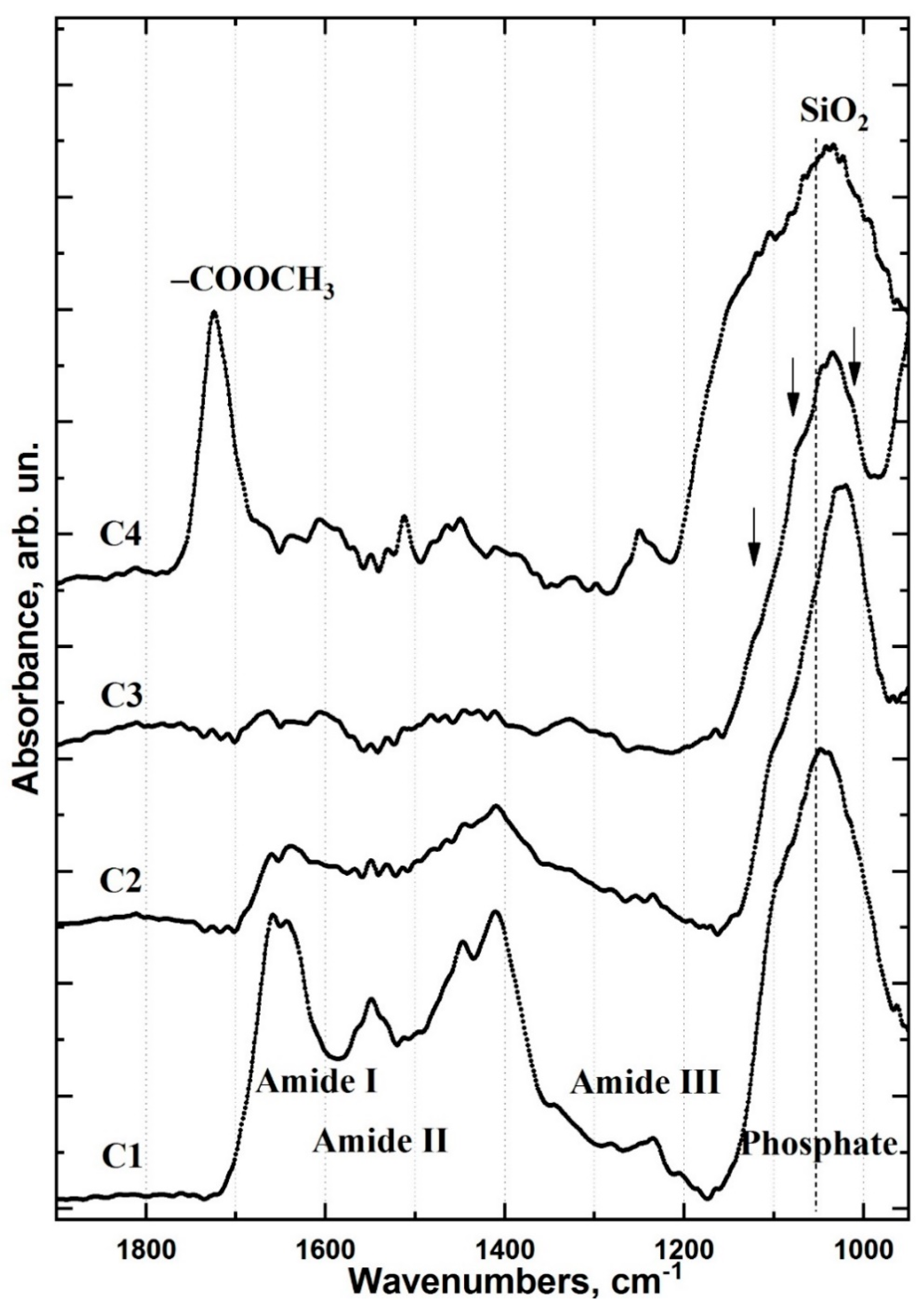
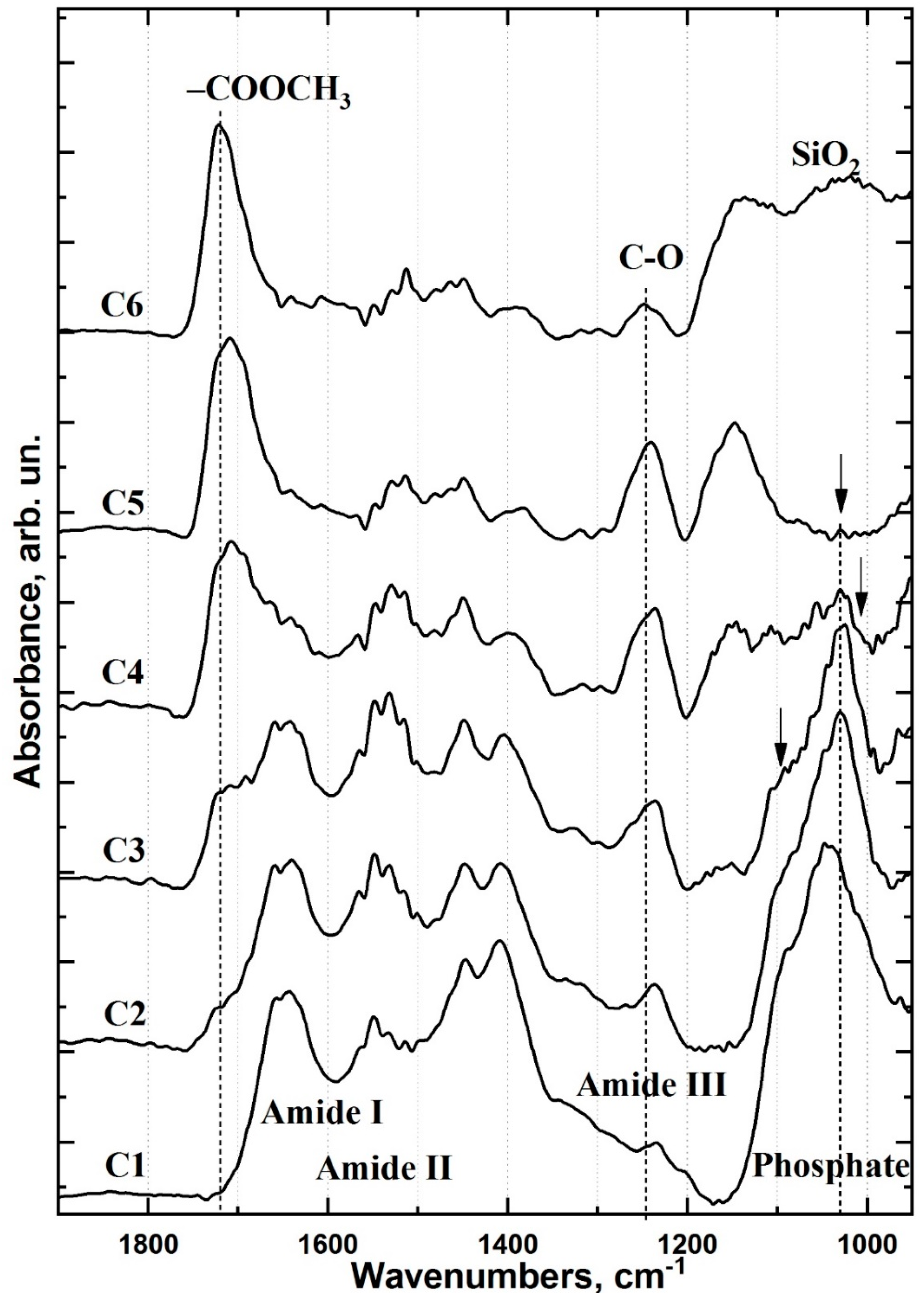
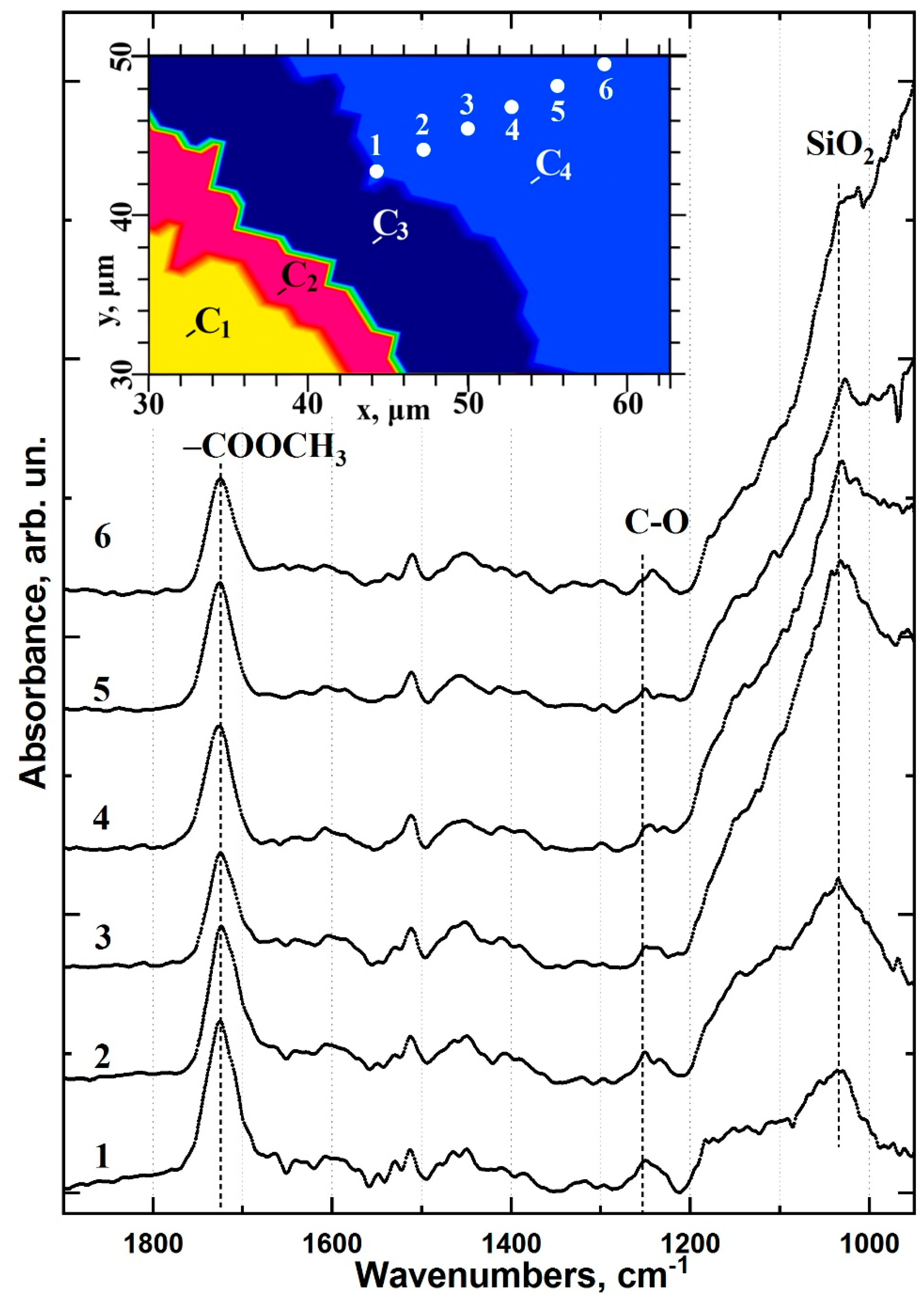
2.1. FTIR Chemical Imaging
2.2. Cluster Analysis
3. Discussion
4. Materials and Methods
4.1. Tooth Preparation and Treatment
4.1.1. Tooth Samples
4.1.2. Treatment of Tooth Samples
4.1.3. Sectionalisation
4.2. Materials
4.2.1. Dentine Conditioner
4.2.2. Modified Dentine Conditioner
4.2.3. Bioprimer
4.2.4. Modified Adhesive
4.2.5. Nanocrystalline Calcium Carbonate-Substituted Hydroxyapatite
4.3. Synchrotron FTIR Microspectroscopy
4.4. Hierarchical Cluster Analysis
5. Conclusions
6. Ethics Declarations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhou, W.; Liu, S.; Zhou, X.; Hannig, M.; Rupf, S.; Feng, J.; Peng, X.; Cheng, L. Modifying Adhesive Materials to Improve the Longevity of Resinous Restorations. Int. J. Mol. Sci. 2019, 20, 723. [Google Scholar] [CrossRef] [PubMed]
- Takamizawa, T.; Imai, A.; Hirokane, E.; Tsujimoto, A.; Barkmeier, W.W.; Erickson, R.L.; Latta, M.A.; Miyazaki, M. SEM Observation of Novel Characteristic of the Dentin Bond Interfaces of Universal Adhesives. Dent. Mater. 2019, 35, 1791–1804. [Google Scholar] [CrossRef]
- Perdigão, J. Current Perspectives on Dental Adhesion: (1) Dentin Adhesion—Not There Yet. Jpn. Dent. Sci. Rev. 2020, 56, 190–207. [Google Scholar] [CrossRef]
- Spencer, P.; Ye, Q.; Park, J.; Topp, E.M.; Misra, A.; Marangos, O.; Wang, Y.; Bohaty, B.S.; Singh, V.; Sene, F.; et al. Adhesive/Dentin Interface: The Weak Link in the Composite Restoration. Ann. Biomed. Eng. 2010, 38, 1989–2003. [Google Scholar] [CrossRef] [PubMed]
- Mota, A.L.M.; Macedo, F.A.A.; Lemos, M.V.S.; Mendes, T.A.D.; Lourenço, G.A.; Albuquerque, N.L.G.; Feitosa, V.P.; Santiago, S.L. Evaluation of Dentinal Conditioning with Natural Acids in Dentin-Resin Interface. Dent. Mater. 2018, 34, e82. [Google Scholar] [CrossRef]
- Carvalho, A.O.; Bacelar-Sá, R.; Wodevotzky, O.; Ambrosano, G.M.B.; Magne, P.C.; Giannini, M. Bond Strength and Micromorphology of Resin-Dentin Interface of Etch-and-Rinse Dentin Bonding Agents after 1-Year of Water Storage. Appl. Adhes. Sci. 2016, 4, 16. [Google Scholar] [CrossRef]
- Ye, Q.; Spencer, P. Analyses of material-tissue interfaces by Fourier transform infrared, Raman spectroscopy, and chemometrics. In Material-Tissue Interfacial Phenomena; Elsevier: Amsterdam, The Netherlands, 2017; pp. 231–251. ISBN 978-0-08-100330-5. [Google Scholar]
- Marghalani, H.Y. Resin-Based Dental Composite Materials. In Handbook of Bioceramics and Biocomposites; Antoniac, I.V., Ed.; Springer International Publishing: Cham, Switzerland, 2016; pp. 357–405. ISBN 978-3-319-12460-5. [Google Scholar]
- Chinelatti, M.A.; do Amaral, T.H.A.; Borsatto, M.C.; Palma-Dibb, R.G.; Corona, S.A.M. Adhesive Interfaces of Enamel and Dentin Prepared by Air-Abrasion at Different Distances. Appl. Surf. Sci. 2007, 253, 4866–4871. [Google Scholar] [CrossRef]
- Stape, T.H.S.; Seseogullari-Dirihan, R.; Tjäderhane, L.; Abuna, G.; Martins, L.R.M.; Tezvergil-Mutluay, A. A Novel Dry-Bonding Approach to Reduce Collagen Degradation and Optimize Resin-Dentin Interfaces. Sci. Rep. 2018, 8, 16890. [Google Scholar] [CrossRef] [PubMed]
- Delgado, A.H.; Young, A.M. Modelling ATR-FTIR Spectra of Dental Bonding Systems to Investigate Composition and Polymerisation Kinetics. Materials 2021, 14, 760. [Google Scholar] [CrossRef]
- Zhang, J.; Zhao, Y.; Tian, Z.; Zhu, J.; Shi, Z.; Cui, Z.; Zhu, S. Enhancement Performance of Application Mussel-Biomimetic Adhesive Primer for Dentin Adhesives. RSC Adv. 2020, 10, 12035–12046. [Google Scholar] [CrossRef]
- Delaviz, Y.; Finer, Y.; Santerre, J.P. Biodegradation of Resin Composites and Adhesives by Oral Bacteria and Saliva: A Rationale for New Material Designs That Consider the Clinical Environment and Treatment Challenges. Dent. Mater. 2014, 30, 16–32. [Google Scholar] [CrossRef] [PubMed]
- El Gezawi, M.; Wölfle, U.C.; Haridy, R.; Fliefel, R.; Kaisarly, D. Remineralization, Regeneration, and Repair of Natural Tooth Structure: Influences on the Future of Restorative Dentistry Practice. ACS Biomater. Sci. Eng. 2019, 5, 4899–4919. [Google Scholar] [CrossRef]
- Tosco, V.; Vitiello, F.; Furlani, M.; Gatto, M.L.; Monterubbianesi, R.; Giuliani, A.; Orsini, G.; Putignano, A. Microleakage Analysis of Different Bulk-Filling Techniques for Class II Restorations: Μ-CT, SEM and EDS Evaluations. Materials 2021, 14, 31. [Google Scholar] [CrossRef]
- Nonoyama, T. Robust Hydrogel–Bioceramics Composite and Its Osteoconductive Properties. Polym. J. 2020, 52, 709–716. [Google Scholar] [CrossRef]
- Barandehfard, F.; Kianpour Rad, M.; Hosseinnia, A.; Khoshroo, K.; Tahriri, M.; Jazayeri, H.E.; Moharamzadeh, K.; Tayebi, L. The Addition of Synthesized Hydroxyapatite and Fluorapatite Nanoparticles to a Glass-Ionomer Cement for Dental Restoration and Its Effects on Mechanical Properties. Ceram. Int. 2016, 42, 17866–17875. [Google Scholar] [CrossRef]
- Uskoković, V. Biomineralization and biomimicry of tooth enamel. In Non-Metallic Biomaterials for Tooth Repair and Replacement; Elsevier: Amsterdam, The Netherlands, 2013; pp. 20–44. ISBN 978-0-85709-244-1. [Google Scholar]
- Han, M.; Li, Q.-L.; Cao, Y.; Fang, H.; Xia, R.; Zhang, Z.-H. In Vivo Remineralization of Dentin Using an Agarose Hydrogel Biomimetic Mineralization System. Sci. Rep. 2017, 7, 41955. [Google Scholar] [CrossRef] [PubMed]
- Provenzi, C.; Leitune, V.C.; Collares, F.M.; Trommer, R.; Bergmann, C.P.; Samuel, S.M. Interface Evaluation of Experimental Dental Adhesives with Nanostructured Hydroxyapatite Incorporation. Appl. Adhes. Sci. 2014, 2, 2. [Google Scholar] [CrossRef]
- Thomas, B.; Chandak, M.; Deosarkar, B. Comparison of Acidic versus Alkaline Environment for Furcation Perforation Repair among Calcium Silicate Based Materials: An in Vitro Comparative Study. J. Adv. Med. Med Res. 2017, 1–8. [Google Scholar] [CrossRef]
- Spencer, P.; Wang, Y.; Katz, J.L.; Misra, A. Physicochemical Interactions at the Dentin/Adhesive Interface Using FTIR Chemical Imaging. J. Biomed. Opt. 2005, 10, 031104. [Google Scholar] [CrossRef]
- Wang, Y.; Yao, X.; Parthasarathy, R. Characterization of Interfacial Chemistry of Adhesive/Dentin Bond Using FTIR Chemical Imaging with Univariate and Multivariate Data Processing. J. Biomed. Mater. Res. A 2009, 91, 251–262. [Google Scholar] [CrossRef]
- Oinas, J.; Rieppo, L.; Finnilä, M.A.J.; Valkealahti, M.; Lehenkari, P.; Saarakkala, S. Imaging of Osteoarthritic Human Articular Cartilage Using Fourier Transform Infrared Microspectroscopy Combined with Multivariate and Univariate Analysis. Sci. Rep. 2016, 6, 30008. [Google Scholar] [CrossRef]
- Seredin, P.V.; Goloshchapov, D.L.; Prutskij, T.; Ippolitov, Y.A. Fabrication and Characterisation of Composites Materials Similar Optically and in Composition to Native Dental Tissues. Results Phys. 2017, 7, 1086–1094. [Google Scholar] [CrossRef]
- Sharma, V.; Rastogi, S.; Bhati, K.; Srinivasan, A.; Roychoudhury, A.; Nikolajeff, F.; Kumar, S. Mapping the Inorganic and Proteomic Differences among Different Types of Human Teeth: A Preliminary Compositional Insight. Biomolecules 2020, 10, 1540. [Google Scholar] [CrossRef]
- Honda, R.; Ryu, M.; Balčytis, A.; Vongsvivut, J.; Tobin, M.J.; Juodkazis, S.; Morikawa, J. Paracetamol Micro-Structure Analysis by Optical Mapping. Appl. Surf. Sci. 2019, 473, 127–132. [Google Scholar] [CrossRef]
- Seredin, P.V.; Goloshchapov, D.L.; Gushchin, M.S.; Ippolitov, Y.A.; Prutskij, T. The Importance of the Biomimetic Composites Components for Recreating the Optical Properties and Molecular Composition of Intact Dental Tissues. J. Phys. Conf. Ser. 2017, 917, 042019. [Google Scholar] [CrossRef]
- Hędzelek, W.; Marcinkowska, A.; Domka, L.; Wachowiak, R. Infrared Spectroscopic Identification of Chosen Dental Materials and Natural Teeth. Acta Phys. Pol. A 2008, 114, 471–484. [Google Scholar] [CrossRef]
- Seredin, P.; Goloshchapov, D.; Ippolitov, Y. Pimm Vongsvivut Pathology-Specific Molecular Profiles of Saliva in Patients with Multiple Dental Caries—Potential Application for Predictive, Preventive and Personalised Medical Services. EPMA J. 2018, 9, 195–203. [Google Scholar] [CrossRef]
- Khan, A.S.; Khalid, H.; Sarfraz, Z.; Khan, M.; Iqbal, J.; Muhammad, N.; Fareed, M.A.; Rehman, I.U. Vibrational Spectroscopy of Selective Dental Restorative Materials. Appl. Spectrosc. Rev. 2017, 52, 507–540. [Google Scholar] [CrossRef]
- Sharma, V.; Srinivasan, A.; Roychoudhury, A.; Rani, K.; Tyagi, M.; Dev, K.; Nikolajeff, F.; Kumar, S. Characterization of Protein Extracts from Different Types of Human Teeth and Insight in Biomineralization. Sci. Rep. 2019, 9, 9314. [Google Scholar] [CrossRef]
- Kobrina, Y.; Rieppo, L.; Saarakkala, S.; Pulkkinen, H.J.; Tiitu, V.; Valonen, P.; Kiviranta, I.; Jurvelin, J.S.; Isaksson, H. Cluster Analysis of Infrared Spectra Can Differentiate Intact and Repaired Articular Cartilage. Osteoarthr. Cartil. 2013, 21, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Le, Q.-T.; Bertrand, C.; Vilar, R. Structural Modifications Induced in Dentin by Femtosecond Laser. J. Biomed. Opt 2016, 21, 125007. [Google Scholar] [CrossRef]
- Spevak, L.; Flach, C.R.; Hunter, T.; Mendelsohn, R.; Boskey, A. Fourier Transform Infrared Spectroscopic Imaging Parameters Describing Acid Phosphate Substitution in Biologic Hydroxyapatite. Calcif. Tissue Int. 2013, 92, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Daood, U.; Swee Heng, C.; Neo Chiew Lian, J.; Fawzy, A.S. In Vitro Analysis of Riboflavin-Modified, Experimental, Two-Step Etch-and-Rinse Dentin Adhesive: Fourier Transform Infrared Spectroscopy and Micro-Raman Studies. Int. J. Oral. Sci. 2015, 7, 110–124. [Google Scholar] [CrossRef]
- Van Landuyt, K.; Munck, J.; Coutinho, E.; Peumans, M.; Lambrechts, P.; Van Meerbeek, B. Bonding to Dentin: Smear Layer and the Process of Hybridization. Dent. Hard Tissues Bond. Interfacial Phenom. Relat. Prop. 2005, 89–122. [Google Scholar] [CrossRef]
- Drouet, C. Apatite Formation: Why It May Not Work as Planned, and How to Conclusively Identify Apatite Compounds. BioMed Res. Int. 2013, 2013, 490946. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-I.; Kim, D.-A.; Patel, K.D.; Shin, U.S.; Kim, H.-W.; Lee, J.-H.; Lee, H.-H. Carbon Nanotube Incorporation in PMMA to Prevent Microbial Adhesion. Sci. Rep. 2019, 9, 4921. [Google Scholar] [CrossRef] [PubMed]
- Goloshchapov, D.L.; Ippolitov, Y.A.; Seredin, P.V. Mechanism of Interaction among Nanocrystalline Carbonate-Substituted Hydroxyapatite and Polar Amino-Acids for the Biomimetic Composite Technology: Spectroscopic and Structural Study. Results Phys. 2020, 18, 103277. [Google Scholar] [CrossRef]
- Tekçe, N.; Tuncer, S.; Demirci, M.; Serim, M.E.; Baydemir, C. The Effect of Different Drinks on the Color Stability of Different Restorative Materials after One Month. Restor. Dent. Endod. 2015, 40, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Seredin, P.; Goloshchapov, D.; Prutskij, T.; Ippolitov, Y. Phase Transformations in a Human Tooth Tissue at the Initial Stage of Caries. PLoS ONE 2015, 10, e0124008. [Google Scholar] [CrossRef] [PubMed]
- Goloshchapov, D.L.; Kashkarov, V.M.; Ippolitov, Y.A.; Prutskij, T.; Seredin, P.V. Early Screening of Dentin Caries Using the Methods of Micro-Raman and Laser-Induced Fluorescence Spectroscopy. Results Phys. 2018, 10, 346–347. [Google Scholar] [CrossRef]
- Fugolin, A.P.; Sundfeld, D.; Ferracane, J.L.; Pfeifer, C.S. Toughening of Dental Composites with Thiourethane-Modified Filler Interfaces. Sci. Rep. 2019, 9, 2286. [Google Scholar] [CrossRef]
- Goloshchapov, D.L.; Kashkarov, V.M.; Rumyantseva, N.A.; Seredin, P.V.; Lenshin, A.S.; Agapov, B.L.; Domashevskaya, E.P. Synthesis of Nanocrystalline Hydroxyapatite by Precipitation Using Hen’s Eggshell. Ceram. Int. 2013, 39, 4539–4549. [Google Scholar] [CrossRef]
- Goloshchapov, D.L.; Lenshin, A.S.; Savchenko, D.V.; Seredin, P.V. Importance of Defect Nanocrystalline Calcium Hydroxyapatite Characteristics for Developing the Dental Biomimetic Composites. Results Phys. 2019, 13, 102158. [Google Scholar] [CrossRef]
- Liu, Q.; Huang, S.; Matinlinna, J.P.; Chen, Z.; Pan, H. Insight into Biological Apatite: Physicochemical Properties and Preparation Approaches. BioMed Res. Int. 2013, 2013, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Nanci, A. Ten Cate’s Oral Histology: Development, Structure, and Function, 8th ed.; Elsevier Health Sciences: Amsterdam, The Netherlands, 2013; ISBN 978-0-323-07846-7. [Google Scholar]
- Seredin, P.; Goloshchapov, D.; Ippolitov, Y.; Vongsvivut, J. Development of a New Approach to Diagnosis of the Early Fluorosis Forms by Means of FTIR and Raman Microspectroscopy. Sci. Rep. 2020, 10, 20891. [Google Scholar] [CrossRef] [PubMed]
- Vongsvivut, J.; Pérez-Guaita, D.; Wood, B.R.; Heraud, P.; Khambatta, K.; Hartnell, D.; Hackett, M.J.; Tobin, M.J. Synchrotron Macro ATR-FTIR Microspectroscopy for High-Resolution Chemical Mapping of Single Cells. Analyst 2019, 144, 3226–3238. [Google Scholar] [CrossRef]
- Savić, D.; Joković, N.; Topisirović, L. Multivariate Statistical Methods for Discrimination of Lactobacilli Based on Their FTIR Spectra. Dairy Sci. Technol. 2008, 88, 273–290. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seredin, P.; Goloshchapov, D.; Ippolitov, Y.; Vongsvivut, J. Engineering of a Biomimetic Interface between a Native Dental Tissue and Restorative Composite and Its Study Using Synchrotron FTIR Microscopic Mapping. Int. J. Mol. Sci. 2021, 22, 6510. https://doi.org/10.3390/ijms22126510
Seredin P, Goloshchapov D, Ippolitov Y, Vongsvivut J. Engineering of a Biomimetic Interface between a Native Dental Tissue and Restorative Composite and Its Study Using Synchrotron FTIR Microscopic Mapping. International Journal of Molecular Sciences. 2021; 22(12):6510. https://doi.org/10.3390/ijms22126510
Chicago/Turabian StyleSeredin, Pavel, Dmitry Goloshchapov, Yuri Ippolitov, and Jitraporn Vongsvivut. 2021. "Engineering of a Biomimetic Interface between a Native Dental Tissue and Restorative Composite and Its Study Using Synchrotron FTIR Microscopic Mapping" International Journal of Molecular Sciences 22, no. 12: 6510. https://doi.org/10.3390/ijms22126510
APA StyleSeredin, P., Goloshchapov, D., Ippolitov, Y., & Vongsvivut, J. (2021). Engineering of a Biomimetic Interface between a Native Dental Tissue and Restorative Composite and Its Study Using Synchrotron FTIR Microscopic Mapping. International Journal of Molecular Sciences, 22(12), 6510. https://doi.org/10.3390/ijms22126510







