Molecular Interactions between Dietary Lipids and Bone Tissue during Aging
Abstract
1. Introduction
2. Bone Biology
2.1. Bone Structure
2.2. Bone Remodeling Processes
2.3. Regulation of Bone Remodeling
3. Aging at the Bone
3.1. Age-Related Changes in Bone Structure
3.2. Age-Related Changes in Bone Remodeling Processes
4. Dietary Lipids and Bone Health
| Population; Age | Intervention vs. Control Diet/Placebo; Duration | Main Changes vs. Control Diet or Placebo | Ref. |
|---|---|---|---|
| 42 women and 23 men; 51.3 ± 7.1 y | HFD (61%E fat) vs. SD (30%E fat); 12 m |
| [78] |
| 208 women and 99 men; 45.5 + 9.7 y | HFD (>45%E fat) vs. SD (30%E fat); 24 m |
| [79] |
| 242 women and 182 men; 51.8 ± 8.9 y | HFD (40%E fat) vs. LFD (20%E fat); 24 m |
| [80] |
| 236 women; 44–50 y | SD (32%E fat) vs. LFD (24%E fat); 18 m |
| [94] |
| 230 women; 27.3 + 1.1 y | LFD dc (<28%E fat) + vs. SD dc (30%E fat); 108 m |
| [81] |
| n-3 PUFA supplementation | |||
| 3 women and 20 men; 49.3 + 1.6 y | n-3 PUFA rich HFD (37.6%E fat; 6.5%E ALA; n-6/n-3 ratio: 1.6/1) vs. HFD (34.5%E fat; 0.8%E ALA; n-6/n-3 ratio: 9.5/1); 6 wks |
| [83] |
| n-3 PUFA rich HFD (37.6%E fat; 6.5%E ALA; n-6/n-3 ratio: 1.6/1) vs. n-6 PUFA rich HFD (37.1%E fat; 3.6%E ALA; n-6/n-3 ratio: 3.5/1); 6 wks |
| [83] | |
| 43 women and 2 men with RA; 57.9 ± 10.8 | NCD + n-3 PUFA supplement (2.4 g of n-3 PUFA/d; 1.1 g ALA+ 0.7 g EPA + 0.1 g DPA + 0.4 g DHA) vs. NCD + dairy supplement (2.4 g of SFA/d); 3 m |
| [87] |
| 87 woman and 26 men; 18–67 y | NCD + n-3 PUFA supplement (1.48 g EPA + DHA/d) vs. NCD + placebo (NA g olive oil); 12 wks |
| [85] |
| 75 women and 6 men with RA; 49.24 ± 10.46 y | NCD + n-3 PUFA supplement (2.090 g of EPA and 1.165 g of DHA/d) vs. NCD + placebo (NA g of high-oleic-acid sunflower oil); 16 wks |
| [84] |
| 126 women; 75 ± 7 y | NCD + n-3 PUFA supplement (1.2 g EPA + DHA/d) vs. NCD + placebo (NA g olive oil); 6 m |
| [86] |
| 60 women and 15 men; 35–65 y | NCD + n-3 PUFA/MUFA enriched dairy supplement (23.7 g saturated fat + 5.17 g oleic acid + 0.14 g DHA + 0.20 g EPA/d) vs. NCD + semiskimmed milk (70g saturated fat + 2.05 g oleic acid); 12 m |
| [88] |
| n-6 PUFA supplementation | |||
| 3 women and 20 men; 49.3 + 1.6 y | n-6 PUFA rich HFD (37.1%E fat; 12.6%E LA; n-6/n-3 ratio: 3.5/1) vs. HFD (34.5%E fat; 7.7%E LA; n-6/n-3 ratio: 9.5/1); 6 wks |
| [83] |
| n-6 PUFA rich HFD (37.1%E fat; 12.6%E LA; n-6/n-3 ratio: 3.5/1) n-3 PUFA rich HFD (37.6%E fat; 10.5%E LA; n-6/n-3 ratio: 1.6/1); 6 wks |
| [83] | |
| 38 women and 6 men with RA; 46.2 ± 13.1 y | NCD + n-6 PUFA supplement (2.5 g CLA/d) vs. NCD + placebo (2.5 g of high-oleic-acid sunflower oil); 3 m |
| [90] |
| 60 men; 49.1+ 6.2 y | NCD + n-6 PUFA supplement (3 g CLA/d) vs. NCD + placebo (NA g palm and bean oil blend/d); 8 wks |
| [89] |
| MUFA supplementation | |||
| 127 men; 67.9 ± 6.9 | CD + EVOO supplementation (>50 mL EVOO/d) vs. CD + nuts supplementation (30 g of mixed walnuts, almonds and hazelnuts); 2 y |
| [93] |
| 98 women and 104 men; 67.8 ± 6.5 | CD + EVOO supplementation (15L EVOO/3 m) vs. CD + nuts supplementation (1350 g of mixed walnuts, almonds and hazelnuts/3 m); 1 y |
| [91] |
| 104 women and 7 men severely obese; 18–40 y | CD + EVOO supplementation (52 mL EVOO/d) vs. CD; 12 wks |
| [92] |
5. Molecular Mechanisms Operating under the Observed Effects of Dietary Lipids on Bone Health of Interest in Relation to Aging
5.1. Mitochondrial Dysfunction and Oxidative Stress
5.2. Apoptosis Dysregulation of Bone Cells
5.3. Dietary Lipids and Genomic Instability at Bone
5.4. Dietary Lipids and Inflammation
5.5. Autophagy Alteration and Bone Cell Differentiation
5.6. Altered Levels of Hormones Involved in Bone Biology
5.6.1. Growth Hormone Axis
5.6.2. Insulin
5.6.3. Thyroid Hormones
5.6.4. Parathyroid Hormone
5.6.5. Sexual Hormones
6. Future Perspectives
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Roberts, S.; Colombier, P.; Sowman, A.; Mennan, C.; Rölfing, J.H.D.; Guicheux, J.; Edwards, J.R. Ageing in the Musculoskeletal System: Cellular Function and Dysfunction throughout Life. Acta Orthop. 2016, 87, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Harvey, N.; Dennison, E.; Cooper, C. Osteoporosis: Impact on Health and Economics. Nat. Rev. Rheumatol. 2010, 6, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.; Wu, B.; Fan, F.; Xie, H.; Xue, W.; Zhu, H.; Chen, Y. Dietary Patterns and the Risk of Hip Fractures in Elderly Chinese: A Matched Case-Control Study. J. Clin. Endocrinol. Metab. 2013, 98, 2347–2355. [Google Scholar] [CrossRef] [PubMed]
- Florencio-Silva, R.; Sasso, G.R.d.S.; Sasso-Cerri, E.; Simões, M.J.; Cerri, P.S. Biology of Bone Tissue: Structure, Function, and Factors That Influence Bone Cells. BioMed Res. Int. 2015, 2015, e421746. [Google Scholar] [CrossRef]
- Capulli, M.; Paone, R.; Rucci, N. Osteoblast and Osteocyte: Games without Frontiers. Arch. Biochem. Biophys. 2014, 561, 3–12. [Google Scholar] [CrossRef]
- Cappariello, A.; Ponzetti, M.; Rucci, N. The “Soft” Side of the Bone: Unveiling Its Endocrine Functions. Horm. Mol. Biol. Clin. Investig. 2016, 28, 5–20. [Google Scholar] [CrossRef]
- Crockett, J.C.; Rogers, M.J.; Coxon, F.P.; Hocking, L.J.; Helfrich, M.H. Bone Remodelling at a Glance. J. Cell Sci. 2011, 124, 991–998. [Google Scholar] [CrossRef]
- Mohamad, N.V.; Soelaiman, I.N.; Chin, K.Y. A Concise Review of Testosterone and Bone Health; Dove Medical Press Ltd.: Macclesfield, UK, 2016; Volume 11. [Google Scholar]
- Nakashima, T.; Kobayashi, Y.; Yamasaki, S.; Kawakami, A.; Eguchi, K.; Sasaki, H.; Sakai, H. Protein Expression and Functional Difference of Membrane-Bound and Soluble Receptor Activator of NF-κB Ligand: Modulation of the Expression by Osteotropic Factors and Cytokines. Biochem. Biophys. Res. Commun. 2000, 275, 768–775. [Google Scholar] [CrossRef]
- Theill, L.E.; Boyle, W.J.; Penninger, J.M. RANK-L and RANK: T Cells, Bone Loss, and Mammalian Evolution. Annu. Rev. Immunol. 2002, 20, 795–823. [Google Scholar] [CrossRef]
- Robling, A.G.; Castillo, A.B.; Turner, C.H. Biomechanical and Molecular Regulation of Bone Remodeling. Annu. Rev. Biomed. Eng. 2006, 8, 455–498. [Google Scholar] [CrossRef]
- Riggs, B.L.; Khosla, S.; Melton, L.J. A Unitary Model for Involutional Osteoporosis: Estrogen Deficiency Causes Both Type I and Type II Osteoporosis in Postmenopausal Women and Contributes to Bone Loss in Aging Men. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 1998, 13, 763–773. [Google Scholar] [CrossRef]
- Almeida, M.; Han, L.; Martin-Millan, M.; O’Brien, C.A.; Manolagas, S.C. Oxidative Stress Antagonizes Wnt Signaling in Osteoblast Precursors by Diverting β-Catenin from T Cell Factor- to Forkhead Box O-Mediated Transcription. J. Biol. Chem. 2007, 282, 27298–27305. [Google Scholar] [CrossRef]
- Zebaze, R.M.; Ghasem-Zadeh, A.; Bohte, A.; Iuliano-Burns, S.; Mirams, M.; Price, R.I.; Mackie, E.J.; Seeman, E. Intracortical Remodelling and Porosity in the Distal Radius and Post-Mortem Femurs of Women: A Cross-Sectional Study. Lancet 2010, 375, 1729–1736. [Google Scholar] [CrossRef]
- Rosen, C.J.; Bouxsein, M.L. Mechanisms of Disease: Is Osteoporosis the Obesity of Bone? Nat. Clin. Pract. Rheumatol. 2006, 2, 35–43. [Google Scholar] [CrossRef]
- Scheller, E.L.; Khoury, B.; Moller, K.L.; Wee, N.K.Y.; Khandaker, S.; Kozloff, K.M.; Abrishami, S.H.; Zamarron, B.F.; Singer, K. Changes in Skeletal Integrity and Marrow Adiposity during High-Fat Diet and after Weight Loss. Front. Endocrinol. 2016, 7. [Google Scholar] [CrossRef]
- Tuljapurkar, S.R.; McGuire, T.R.; Brusnahan, S.K.; Jackson, J.D.; Garvin, K.L.; Kessinger, M.A.; Lane, J.T.; O’Kane, B.J.; Sharp, J.G. Changes in Human Bone Marrow Fat Content Associated with Changes in Hematopoietic Stem Cell Numbers and Cytokine Levels with Aging. J. Anat. 2011, 219, 574–581. [Google Scholar] [CrossRef]
- Patel, V.S.; Ete Chan, M.; Rubin, J.; Rubin, C.T. Marrow Adiposity and Hematopoiesis in Aging and Obesity: Exercise as an Intervention. Curr. Osteropos. Rep. 2018, 16, 105–115. [Google Scholar] [CrossRef]
- Gimble, J.M.; Zvonic, S.; Floyd, Z.E.; Kassem, M.; Nuttall, M.E. Playing with Bone and Fat. J. Cell Biochem. 2006, 98, 251266. [Google Scholar] [CrossRef]
- Lecka-Czernik, B. PPARs and Bone Metabolism. PPAR Res. 2006, 2006, 18089. [Google Scholar] [CrossRef]
- Zhou, S.; Greenberger, J.S.; Epperly, M.W.; Goff, J.P.; Adler, C.; Leboff, M.S.; Glowacki, J. Age-Related Intrinsic Changes in Human Bone-Marrow-Derived Mesenchymal Stem Cells and Their Differentiation to Osteoblasts. Aging Cell 2008, 7, 335–343. [Google Scholar] [CrossRef]
- Rosen, C.J.; Ackert-Bicknell, C.; Rodriguez, J.P.; Pino, A.M. Marrow Fat and the Bone Microenvironment: Developmental, Functional, and Pathological Implications. Crit. Rev. Eukaryot. Gene Expr. 2009, 19, 109–124. [Google Scholar] [CrossRef]
- Justesen, J.; Stenderup, K.; Ebbesen, E.N.; Mosekilde, L.; Steiniche, T.; Kassem, M. Adipocyte Tissue Volume in Bone Marrow Is Increased with Aging and in Patients with Osteoporosis. Biogerontology 2001, 2, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Cremers, S.; Farooki, A. Biochemical Markers of Bone Turnover in Osteonecrosis of the Jaw in Patients with Osteoporosis and Advanced Cancer Involving the Bone. Ann. N. Y. Acad. Sci. 2011, 1218, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Venton, L.; Sakata, T.; Halloran, B.P. Expression of RANKL and OPG Correlates with Age-Related Bone Loss in Male C57BL/6 Mice. J. Bone Miner. Res. 2003, 18, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.C.I.; Fletcher, M.D.; Tarantal, A.F. Effect of Age on the Frequency, Cell Cycle, and Lineage Maturation of Rhesus Monkey (Macaca Mulatta) CD34+ and Hematopoietic Progenitor Cells. Pediatr. Res. 2005, 58, 315–322. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jiang, Y.; Mishima, H.; Sakai, S.; Liu, Y.K.; Ohyabu, Y.; Uemura, T. Gene Expression Analysis of Major Lineage-Defining Factors in Human Bone Marrow Cells: Effect of Aging, Gender, and Age-Related Disorders. J. Orthop. Res. 2008, 26, 910–917. [Google Scholar] [CrossRef] [PubMed]
- Szulc, P.; Seeman, E. Thinking inside and Outside the Envelopes of Bone: DDDedicated to PDD. Osteoporos. Int. 2009, 20, 1281–1288. [Google Scholar] [CrossRef]
- Michaëlsson, K.; Holmberg, L.; Mallmin, H.; Wolk, A.; Bergström, R.; Ljunghall, S. Diet, Bone Mass, and Osteocalcin: A Cross-Sectional Study. Calcif. Tissue Int. 1995, 57, 86–93. [Google Scholar] [CrossRef]
- Cooper, C.; Atkinson, E.J.; Hensrud, D.D.; Wahner, H.W.; O’Fallon, W.M.; Riggs, B.L.; Melton, L.J. Dietary Protein Intake and Bone Mass in Women. Calcif. Tissue Int. 1996, 58, 320–325. [Google Scholar] [CrossRef]
- Corwin, R.L. Effects of Dietary Fats on Bone Health in Advanced Age. Prostaglandins Leukot. Essent. Fatty Acids 2003, 68, 379–386. [Google Scholar] [CrossRef]
- Kato, I.; Toniolo, P.; Zeleniuch-Jacquotte, A.; Shore, R.E.; Koenig, K.L.; Akhmedkhanov, A.; Riboli, E. Diet, Smoking and Anthropometric Indices and Postmenopausal Bone Fractures: A Prospective Study. Int. J. Epidemiol. 2000, 29, 85–92. [Google Scholar] [CrossRef]
- Wang, Y.; Dellatore, P.; Douard, V.; Qin, L.; Watford, M.; Ferraris, R.P.; Lin, T.; Shapses, S.A. High Fat Diet Enriched with Saturated, but Not Monounsaturated Fatty Acids Adversely Affects Femur, and Both Diets Increase Calcium Absorption in Older Female Mice. Nutr. Res. 2016, 36, 742–750. [Google Scholar] [CrossRef]
- Sanderson, J.P.; Binkley, N.; Roecker, E.B.; Champ, J.E.; Pugh, T.D.; Aspnes, L.; Weindruch, R. Influence of Fat Intake and Caloric Restriction on Bone in Aging Male Rats. J. Gerontol. Ser. A 1997, 52, B20–B25. [Google Scholar] [CrossRef]
- Schmiedl, A.; Schwille, P.O.; Bonucci, E.; Erben, R.G.; Grayczyk, A.; Sharma, V. Nephrocalcinosis and Hyperlipidemia in Rats Fed a Cholesterol- and Fat-Rich Diet: Association with Hyperoxaluria, Altered Kidney and Bone Minerals, and Renal Tissue Phospholipid-Calcium Interaction. Urol. Res. 2000, 28, 404–415. [Google Scholar] [CrossRef]
- Parhami, F.; Tintut, Y.; Beamer, W.G.; Gharavi, N.; Goodman, W.; Demer, L.L. Atherogenic High-Fat Diet Reduces Bone Mineralization in Mice. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2001, 16, 182–188. [Google Scholar] [CrossRef]
- Parhami, F.; Jackson, S.M.; Tintut, Y.; Le, V.; Balucan, J.P.; Territo, M.; Demer, L.L. Atherogenic Diet and Minimally Oxidized Low Density Lipoprotein Inhibit Osteogenic and Promote Adipogenic Differentiation of Marrow Stromal Cells. J. Bone Miner. Res. 1999, 14, 2067–2078. [Google Scholar] [CrossRef]
- Macri, E.V.; Gonzales Chaves, M.M.; Rodriguez, P.N.; Mandalunis, P.; Zeni, S.; Lifshitz, F.; Friedman, S.M. High-Fat Diets Affect Energy and Bone Metabolism in Growing Rats. Eur. J. Nutr. 2012, 51, 399–406. [Google Scholar] [CrossRef]
- Lukas, R.; Gigliotti, J.C.; Smith, B.J.; Altman, S.; Tou, J.C. Consumption of Different Sources of Omega-3 Polyunsaturated Fatty Acids by Growing Female Rats Affects Long Bone Mass and Microarchitecture. Bone 2011, 49, 455–462. [Google Scholar] [CrossRef]
- Beier, E.E.; Inzana, J.A.; Sheu, T.J.; Shu, L.; Edward Puzas, J.; Mooney, R.A. Effects of Combined Exposure to Lead and High-Fat Diet on Bone Quality in Juvenile Male Mice. Environ. Health Perspect. 2015, 123, 935–943. [Google Scholar] [CrossRef]
- Picke, A.K.; Sylow, L.; Møller, L.L.V.; Kjøbsted, R.; Schmidt, F.N.; Steejn, M.W.; Salbach-Hirsch, J.; Hofbauer, C.; Blüher, M.; Saalbach, A.; et al. Differential Effects of High-Fat Diet and Exercise Training on Bone and Energy Metabolism. Bone 2018, 116, 120–134. [Google Scholar] [CrossRef]
- Jatkar, A.; Kurland, I.J.; Judex, S. Diets High in Fat or Fructose Differentially Modulate Bone Health and Lipid Metabolism. Calcif. Tissue Int. 2017, 100, 20–28. [Google Scholar] [CrossRef]
- Lorincz, C.; Reimer, R.A.; Boyd, S.K.; Zernicke, R.F. High-Fat, Sucrose Diet Impairs Geometrical and Mechanical Properties of Cortical Bone in Mice. Br. J. Nutr. 2010, 103, 1302–1308. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.-L.; Yeh, J.K.; Rasty, J.; Chyu, M.-C.; Dunn, D.M.; Li, Y.; Watkins, B.A. Improvement of Bone Quality in Gonad-Intact Middle-Aged Male Rats by Long-Chain n-3 Polyunsaturated Fatty Acid. Calcif. Tissue Int. 2007, 80, 286–293. [Google Scholar] [CrossRef]
- Wohl, G.R.; Loehrke, L.; Watkins, B.A.; Zernicke, R.F. Effects of High-Fat Diet on Mature Bone Mineral Content, Structure, and Mechanical Properties. Calcif. Tissue Int. 1998, 63, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Varela-López, A.; Ochoa, J.J.; Llamas-Elvira, J.M.; López-Frías, M.; Planells, E.; Speranza, L.; Battino, M.; Quiles, J.L. Loss of Bone Mineral Density Associated with Age in Male Rats Fed on Sunflower Oil Is Avoided by Virgin Olive Oil Intake or Coenzyme Q Supplementation. Int. J. Mol. Sci. 2017, 18, 1397. [Google Scholar] [CrossRef] [PubMed]
- Bullon, P.; Battino, M.; Varela-Lopez, A.; Perez-Lopez, P.; Granados-Principal, S.; Ramirez-Tortosa, M.C.; Ochoa, J.J.; Cordero, M.D.; Gonzalez-Alonso, A.; Ramirez-Tortosa, C.L.; et al. Diets Based on Virgin Olive Oil or Fish Oil but Not on Sunflower Oil Prevent Age-Related Alveolar Bone Resorption by Mitochondrial-Related Mechanisms. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Watkins, B.A.; Li, Y.; Allen, K.G.D.; Hoffmann, W.E.; Seifert, M.F. Dietary Ratio of (n-6)/(n-3) Polyunsaturated Fatty Acids Alters the Fatty Acid Composition of Bone Compartments and Biomarkers of Bone Formation in Rats. J. Nutr. 2000, 130, 2274–2284. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Cui, J.; Li, Y.X.; Shi, Y.H.; Le, G.W. Expression of Genes Associated with Bone Resorption Is Increased and Bone Formation Is Decreased in Mice Fed a High-Fat Diet. Lipids 2010, 45, 345–355. [Google Scholar] [CrossRef]
- Malvi, P.; Piprode, V.; Chaube, B.; Pote, S.T.; Mittal, M.; Chattopadhyay, N.; Wani, M.R.; Bhat, M.K. High Fat Diet Promotes Achievement of Peak Bone Mass in Young Rats. Biochem. Biophys. Res. Commun. 2014, 455, 133–138. [Google Scholar] [CrossRef]
- Lau, B.Y.L.Y.; Fajardo, V.A.F.A.; McMeekin, L.M.; Sacco, S.M.S.M.; Ward, W.E.W.E.; Roy, B.D.R.D.; Peters, S.J.P.J.; LeBlanc, P.J.L.J. Influence of High-Fat Diet from Differential Dietary Sources on Bone Mineral Density, Bone Strength, and Bone Fatty Acid Composition in Rats. Appl. Physiol. Nutr. Metab. 2010. [Google Scholar] [CrossRef]
- Watkins, B.A.; Shen, C.L.; Allen, K.G.; Seifert, M.F. Dietary (n-3) and (n-6) Polyunsaturates and Acetylsalicylic Acid Alter Ex Vivo PGE2 Biosynthesis, Tissue IGF-I Levels, and Bone Morphometry in Chicks. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 1996, 11, 1321–1332. [Google Scholar] [CrossRef]
- Shen, C.-L.; Yeh, J.K.; Rasty, J.; Li, Y.; Watkins, B.A. Protective Effect of Dietary Long-Chain n -3 Polyunsaturated Fatty Acids on Bone Loss in Gonad-Intact Middle-Aged Male Rats. Br. J. Nutr. 2006, 95, 462–468. [Google Scholar] [CrossRef]
- Tencerova, M.; Figeac, F.; Ditzel, N.; Taipaleenmäki, H.; Nielsen, T.K.; Kassem, M. High-Fat Diet–Induced Obesity Promotes Expansion of Bone Marrow Adipose Tissue and Impairs Skeletal Stem Cell Functions in Mice. J. Bone Miner. Res. 2018, 33, 1154–1165. [Google Scholar] [CrossRef]
- Lecka-Czernik, B.; Stechschulte, L.A.; Czernik, P.J.; Dowling, A.R. High Bone Mass in Adult Mice with Diet-Induced Obesity Results from a Combination of Initial Increase in Bone Mass Followed by Attenuation in Bone Formation; Implications for High Bone Mass and Decreased Bone Quality in Obesity. Mol. Cell. Endocrinol. 2015, 410, 35–41. [Google Scholar] [CrossRef]
- Lindenmaier, L.B.; Philbrick, K.A.; Branscum, A.J.; Kalra, S.P.; Turner, R.T.; Iwaniec, U.T. Hypothalamic Leptin Gene Therapy Reduces Bone Marrow Adiposity in Ob/Ob Mice Fed Regular and High-Fat Diets. Front. Endocrinol. 2016, 7. [Google Scholar] [CrossRef]
- Halade, G.V.; Rahman, M.M.; Williams, P.J.; Fernandes, G. Combination of Conjugated Linoleic Acid with Fish Oil Prevents Age-Associated Bone Marrow Adiposity in C57Bl/6J Mice. J. Nutr. Biochem. 2011, 22, 459–469. [Google Scholar] [CrossRef]
- Watkins, B.A.; Li, Y.; Seifert, M.F. Dietary Ratio of N-6/n-3 PUFAs and Docosahexaenoic Acid: Actions on Bone Mineral and Serum Biomarkers in Ovariectomized Rats. J. Nutr. Biochem. 2006, 17, 282–289. [Google Scholar] [CrossRef]
- Tagliaferri, C.; Davicco, M.J.; Lebecque, P.; Georgé, S.; Amiot, M.J.; Mercier, S.; Dhaussy, A.; Huertas, A.; Walrand, S.; Wittrant, Y.; et al. Olive Oil and Vitamin D Synergistically Prevent Bone Loss in Mice. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Hagan, C. When Are Mice Considered Old? Available online: https://www.jax.org/news-and-insights/jax-blog/2017/november/when-are-mice-considered-old (accessed on 2 June 2021).
- Sengupta, P. The Laboratory Rat: Relating Its Age With Human’s. Int. J. Prev. Med. 2013, 4, 624–630. [Google Scholar]
- Cao, J.J.; Gregoire, B.R.; Gao, H. High-Fat Diet Decreases Cancellous Bone Mass but Has No Effect on Cortical Bone Mass in the Tibia in Mice. Bone 2009, 44, 1097–1104. [Google Scholar] [CrossRef]
- Xiao, Y.; Cui, J.; Li, Y.X.; Shi, Y.H.; Wang, B.; Le, G.W.; Wang, Z.P. Dyslipidemic High-Fat Diet Affects Adversely Bone Metabolism in Mice Associated with Impaired Antioxidant Capacity. Nutrition 2011, 27, 214–220. [Google Scholar] [CrossRef]
- Nuttall, M.E.; Patton, A.J.; Olivera, D.L.; Nadeau, D.P.; Gowen, M. Human Trabecular Bone Cells Are Able to Express Both Osteoblastic and Adipocytic Phenotype: Implications for Osteopenic Disorders. J. Bone Miner. Res. 1998, 13, 371–382. [Google Scholar] [CrossRef]
- Halade, G.V.; El Jamali, A.; Williams, P.J.; Fajardo, R.J.; Fernandes, G. Obesity-Mediated Inflammatory Microenvironment Stimulates Osteoclastogenesis and Bone Loss in Mice. Exp. Gerontol. 2011, 46, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Kruger, M.C.; Coetzer, H.; De Winter, R.; Gericke, G.; Van Papendorp, D.H. Calcium, Gamma-Linolenic Acid and Eicosapentaenoic Acid Supplementation in Senile Osteoporosis. Aging Clin. Exp. Res. 1998, 10, 385–394. [Google Scholar] [CrossRef]
- Bassey, E.J.; Littlewood, J.J.; Rothwell, M.C.; Pye, D.W. Lack of Effect of Supplementation with Essential Fatty Acids on Bone Mineral Density in Healthy Pre- and Postmenopausal Women: Two Randomized Controlled Trials of Efacal® v. Calcium Alone. Br. J. Nutr. 2000, 83, 629–635. [Google Scholar] [CrossRef]
- Beresford, J.N.; Bennett, J.H.; Devlin, C.; Leboy, P.S.; Owen, M.E. Evidence for an Inverse Relationship between the Differentiation of Adipocytic and Osteogenic Cells in Rat Marrow Stromal Cell Cultures. J. Cell Sci. 1992, 102, 341–351. [Google Scholar] [CrossRef]
- Nuttall, M.E.; Gimble, J.M. Is There a Therapeutic Opportunity to Either Prevent or Treat Osteopenic Disorders by Inhibiting Marrow Adipogenesis? Bone 2000, 27, 177–184. [Google Scholar] [CrossRef]
- Ogawa, S.; Urano, T.; Hosoi, T.; Miyao, M.; Hoshino, S.; Fujita, M.; Shiraki, M.; Orimo, H.; Ouchi, Y.; Inoue, S. Association of Bone Mineral Density with a Polymorphism of the Peroxisome Proliferator-Activated Receptor γ Gene: PPARγ Expression in Osteoblasts. Biochem. Biophys. Res. Commun. 1999, 260, 122–126. [Google Scholar] [CrossRef]
- Diascro, D.D.; Vogel, R.L.; Johnson, T.E.; Witherup, K.M.; Pitzenberger, S.M.; Rutledge, S.J.; Prescott, D.J.; Rodan, G.A.; Schmidt, A. High Fatty Acid Content in Rabbit Serum Is Responsible for the Differentiation of Osteoblasts Into Adipocyte-like Cells. J. Bone Miner. Res. 1998, 13, 96–106. [Google Scholar] [CrossRef]
- Lecka-Czernik, B.; Moerman, E.J.; Grant, D.F.; Lehmann, J.M.; Manolagas, S.C.; Jilka, R.L. Divergent Effects of Selective Peroxisome Proliferator-Activated Receptor-Γ2 Ligands on Adipocyte versus Osteoblast Differentiation. Endocrinology 2002, 143, 2376–2384. [Google Scholar] [CrossRef] [PubMed]
- Weiler, H.A. Dietary Supplementation of Arachidonic Acid Is Associated with Higher Whole Body Weight and Bone Mineral Density in Growing Pigs. Pediatr. Res. 2000, 47, 692–697. [Google Scholar] [CrossRef] [PubMed]
- Judex, S.; Wohl, G.R.; Wolff, R.B.; Leng, W.; Gillis, A.M.; Zernicke, R.F. Dietary Fish Oil Supplementation Adversely Affects Cortical Bone Morphology and Biomechanics in Growing Rabbits. Calcif. Tissue Int. 2000, 66, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.J.; Gregoire, B.R.; Michelsen, K.G.; Picklo, M.J. Increasing Dietary Fish Oil Reduces Adiposity and Mitigates Bone Deterioration in Growing C57BL/6 Mice Fed a High-Fat Diet. J. Nutr. 2020, 150, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Claassen, N.; Coetzer, H.; Steinmann, C.M.L.; Kruger, M.C. The Effect of Different N-6 n-3 Essential Fatty Acid Ratios on Calcium Balance and Bone in Rats. Prostaglandins Leukot. Essent. Fatty Acids 1995, 53, 13–19. [Google Scholar] [CrossRef]
- Umegaki, K.; Hashimoto, M.; Yamasaki, H.; Fujii, Y.; Yoshimura, M.; Sugisawa, A.; Shinozuka, K. Docosahexaenoic Acid Supplementation-Increased Oxidative Damage in Bone Marrow DNA in Aged Rats and Its Relation to Antioxidant Vitamins. Free Radic. Res. 2001, 34, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Brinkworth, G.D.; Wycherley, T.P.; Noakes, M.; Buckley, J.D.; Clifton, P.M. Long-Term Effects of a Very-Low-Carbohydrate Weight-Loss Diet and an Isocaloric Low-Fat Diet on Bone Health in Obese Adults. Nutrition 2016, 32, 1033–1036. [Google Scholar] [CrossRef] [PubMed]
- Foster, G.D.; Wyatt, H.R.; Hill, J.O.; Makris, A.P.; Rosenbaum, D.L.; Brill, C.; Stein, R.I.; Mohammed, B.S.; Miller, B.; Rader, D.J.; et al. Weight and Metabolic Outcomes After 2 Years on a Low-Carbohydrate Versus Low-Fat Diet. Ann. Intern. Med. 2010, 153, 147–157. [Google Scholar] [CrossRef]
- Tirosh, A.; de Souza, R.J.; Sacks, F.; Bray, G.A.; Smith, S.R.; LeBoff, M.S. Sex Differences in the Effects of Weight Loss Diets on Bone Mineral Density and Body Composition: POUNDS LOST Trial. J. Clin. Endocrinol. Metab. 2015, 100, 2463–2471. [Google Scholar] [CrossRef]
- Dorgan, J.F.; Liu, L.; Klifa, C.; Hylton, N.; Shepherd, J.A.; Stanczyk, F.Z.; Snetselaar, L.G.; Van Horn, L.; Stevens, V.J.; Robson, A.; et al. Adolescent Diet and Subsequent Serum Hormones, Breast Density and Bone Mineral Density in Young Women: Results of the Dietary Intervention Study in Children (DISC) Follow-Up Study. Cancer Epidemiol. Biomark. Prev. Publ. Am. Assoc. Cancer Res. Cosponsored Am. Soc. Prev. Oncol. 2010, 19, 1545–1556. [Google Scholar] [CrossRef]
- Villareal, D.T.; Fontana, L.; Das, S.K.; Redman, L.; Smith, S.R.; Saltzman, E.; Bales, C.; Rochon, J.; Pieper, C.; Huang, M.; et al. Effect of Two-Year Caloric Restriction on Bone Metabolism and Bone Mineral Density in Non-Obese Younger Adults: A Randomized Clinical Trial. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2016, 31, 40–51. [Google Scholar] [CrossRef]
- Griel, A.E.; Kris-Etherton, P.M.; Hilpert, K.F.; Zhao, G.; West, S.G.; Corwin, R.L. An Increase in Dietary N-3 Fatty Acids Decreases a Marker of Bone Resorption in Humans. Nutr. J. 2007, 6, 2. [Google Scholar] [CrossRef]
- Park, Y.; Lee, A.; Shim, S.-C.; Lee, J.H.; Choe, J.-Y.; Ahn, H.; Choi, C.B.; Sung, Y.K.; Bae, S.C. Effect of N-3 Polyunsaturated Fatty Acid Supplementation in Patients with Rheumatoid Arthritis: A 16-Week Randomized, Double-Blind, Placebo-Controlled, Parallel-Design Multicenter Study in Korea. J. Nutr. Biochem. 2013, 24, 1367–1372. [Google Scholar] [CrossRef]
- Appleton, K.M.; Fraser, W.D.; Rogers, P.J.; Ness, A.R.; Tobias, J.H. Supplementation with a Low–Moderate Dose of n-3 Long-Chain PUFA Has No Short-Term Effect on Bone Resorption in Human Adults. Br. J. Nutr. 2011, 105, 1145–1149. [Google Scholar] [CrossRef]
- Dong, H.; Hutchins-Wiese, H.; Kleppinger, A.; Annis, K.; Liva, E.; Lammi-Keefe, C.; Durham, H.; Feinn, R.; Kenny, A.M. Effects of Omega-3 Polyunsaturated Fatty Acid Supplementation on Bone Turnover in Older Women. Int. J. Vitam. Nutr. Res. 2014, 84, 124–132. [Google Scholar] [CrossRef]
- Dawczynski, C.; Schubert, R.; Hein, G.; Müller, A.; Eidner, T.; Vogelsang, H.; Basu, S.; Jahreis, G. Long-Term Moderate Intervention with n-3 Long-Chain PUFA-Supplemented Dairy Products: Effects on Pathophysiological Biomarkers in Patients with Rheumatoid Arthritis. Br. J. Nutr. 2009, 101, 1517–1526. [Google Scholar] [CrossRef]
- Martin-Bautista, E.; Muñoz-Torres, M.; Fonolla, J.; Quesada, M.; Poyatos, A.; Lopez-Huertas, E. Improvement of Bone Formation Biomarkers after 1-Year Consumption with Milk Fortified with Eicosapentaenoic Acid, Docosahexaenoic Acid, Oleic Acid, and Selected Vitamins. Nutr. Res. 2010, 30, 320–326. [Google Scholar] [CrossRef]
- Doyle, L.; Jewell, C.; Mullen, A.; Nugent, A.P.; Roche, H.M.; Cashman, K.D. Effect of Dietary Supplementation with Conjugated Linoleic Acid on Markers of Calcium and Bone Metabolism in Healthy Adult Men. Eur. J. Clin. Nutr. 2005, 59, 432–440. [Google Scholar] [CrossRef]
- Aryaeian, N.; Shahram, F.; Djalali, M. CLA Has a Useful Effect on Bone Markers in Patients with Rheumatoid Arthritis. Lipids 2016, 51, 1397–1405. [Google Scholar] [CrossRef]
- Bulló, M.; Amigó-Correig, P.; Márquez-Sandoval, F.; Babio, N.; Martínez-González, M.A.; Estruch, R.; Basora, J.; Solà, R.; Salas-Salvadó, J. Mediterranean Diet and High Dietary Acid Load Associated with Mixed Nuts: Effect on Bone Metabolism in Elderly Subjects. J. Am. Geriatr. Soc. 2009, 57, 1789–1798. [Google Scholar] [CrossRef]
- Cardoso, C.K.d.S.; Santos, A.S.e.A.d.C.; Rosa, L.P.d.S.; Mendonça, C.R.; Vitorino, P.V.d.O.; Peixoto, M.d.R.G.; Silveira, É.A. Effect of Extra Virgin Olive Oil and Traditional Brazilian Diet on the Bone Health Parameters of Severely Obese Adults: A Randomized Controlled Trial. Nutrients 2020, 12, 403. [Google Scholar] [CrossRef]
- Fernández-Real, J.M.; Bulló, M.; Moreno-Navarrete, J.M.; Ricart, W.; Ros, E.; Estruch, R.; Salas-Salvadó, J. A Mediterranean Diet Enriched with Olive Oil Is Associated with Higher Serum Total Osteocalcin Levels in Elderly Men at High Cardiovascular Risk. J. Clin. Endocrinol. Metab. 2012, 97, 3792–3798. [Google Scholar] [CrossRef]
- Salamone, L.M.; Cauley, J.A.; Black, D.M.; Simkin-Silverman, L.; Lang, W.; Gregg, E.; Palermo, L.; Epstein, R.S.; Kuller, L.H.; Wing, R. Effect of a Lifestyle Intervention on Bone Mineral Density in Premenopausal Women: A Randomized Trial. Am. J. Clin. Nutr. 1999, 70, 97–103. [Google Scholar] [CrossRef] [PubMed]
- McMurray, F.; Patten, D.A.; Harper, M.-E. Reactive Oxygen Species and Oxidative Stress in Obesity—Recent Findings and Empirical Approaches. Obesity 2016, 24, 2301–2310. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.L.; Norhaizan, M.E. Effect of High-Fat Diets on Oxidative Stress, Cellular Inflammatory Response and Cognitive Function. Nutrients 2019, 11, 2579. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.L.; Yuan, N.; Sun, T.; Dun, A.S.; Hou, H.F. Effects of Selenium Supplement on Atherogenesis of ApoE-Knockout Mice Fed High Fat Diet. Zhonghua Xin Xue Guan Bing Za Zhi 2016, 44, 244–249. [Google Scholar] [CrossRef]
- Xie, H.; Wang, Q.; Zhang, X.; Wang, T.; Hu, W.; Manicum, T.; Chen, H.; Sun, L. Possible Therapeutic Potential of Berberine in the Treatment of STZ plus HFD-Induced Diabetic Osteoporosis. Biomed. Pharmacother. 2018, 108, 280–287. [Google Scholar] [CrossRef]
- Higashimoto, M.; Isoyama, N.; Ishibashi, S.; Inoue, M.; Takiguchi, M.; Suzuki, S.; Ohnishi, Y.; Sato, M. Tissue-Dependent Preventive Effect of Metallothionein against DNA Damage in Dyslipidemic Mice under Repeated Stresses of Fasting or Restraint. Life Sci. 2009, 84, 569–575. [Google Scholar] [CrossRef]
- Coetzer, H.; Claassen, N.; van Papendorp, D.H.; Kruger, M.C. Calcium Transport by Isolated Brush Border and Basolateral Membrane Vesicles: Role of Essential Fatty Acid Supplementation. Prostaglandins Leukot. Essent. Fatty Acids 1994, 50, 257–266. [Google Scholar] [CrossRef]
- Wauquier, F.; Leotoing, L.; Coxam, V.; Guicheux, J.; Wittrant, Y. Oxidative Stress in Bone Remodelling and Disease. Trends Mol. Med. 2009, 15, 468–477. [Google Scholar] [CrossRef]
- Bai, X.; Lu, D.; Liu, A.; Zhang, Z.; Li, X.; Zou, Z.; Zeng, W.; Cheng, B.; Luo, S. Reactive Oxygen Species Stimulates Receptor Activator of NF-KappaB Ligand Expression in Osteoblast. J. Biol. Chem. 2005, 280, 17497–17506. [Google Scholar] [CrossRef]
- Baek, K.H.; Oh, K.W.; Lee, W.Y.; Lee, S.S.; Kim, M.K.; Kwon, H.S.; Rhee, E.J.; Han, J.H.; Song, K.H.; Cha, B.Y.; et al. Association of Oxidative Stress with Postmenopausal Osteoporosis and the Effects of Hydrogen Peroxide on Osteoclast Formation in Human Bone Marrow Cell Cultures. Calcif. Tissue Int. 2010, 87, 226–235. [Google Scholar] [CrossRef]
- Bai, B.; Vanhoutte, P.M.; Wang, Y. Loss-of-SIRT1 Function during Vascular Ageing: Hyperphosphorylation Mediated by Cyclin-Dependent Kinase 5. Trends Cardiovasc. Med. 2014, 24, 81–84. [Google Scholar] [CrossRef]
- Liu, J.M.; Zhao, H.Y.; Ning, G.; Zhao, Y.J.; Chen, Y.; Zhang, Z.; Sun, L.H.; Xu, M.-Y.; Chen, J.L. Relationships between the Changes of Serum Levels of OPG and RANKL with Age, Menopause, Bone Biochemical Markers and Bone Mineral Density in Chinese Women Aged 20-75. Calcif. Tissue Int. 2005, 76, 1–6. [Google Scholar] [CrossRef]
- Liu, A.L.; Zhang, Z.M.; Zhu, B.F.; Liao, Z.H.; Liu, Z. Metallothionein Protects Bone Marrow Stromal Cells against Hydrogen Peroxide-Induced Inhibition of Osteoblastic Differentiation. Cell Biol. Int. 2004, 28, 905–911. [Google Scholar] [CrossRef]
- Iyer, S.; Ambrogini, E.; Bartell, S.M.; Han, L.; Roberson, P.K.; De Cabo, R.; Jilka, R.L.; Weinstein, R.S.; O’Brien, C.A.; Manolagas, S.C.; et al. FOXOs Attenuate Bone Formation by Suppressing Wnt Signaling. J. Clin. Investig. 2013, 123, 3409–3419. [Google Scholar] [CrossRef]
- Manolagas, S.C.; Almeida, M. Gone with the Wnts: β-Catenin, T-Cell Factor, Forkhead Box O, and Oxidative Stress in Age-Dependent Diseases of Bone, Lipid, and Glucose Metabolism. Mol. Endocrinol. 2007, 21, 2605–2614. [Google Scholar] [CrossRef]
- Almeida, M.; Ambrogini, E.; Han, L.; Manolagas, S.C.; Jilka, R.L. Increased Lipid Oxidation Causes Oxidative Stress, Increased Peroxisome Proliferator-Activated Receptor-γ Expression, and Diminished Pro-Osteogenic Wnt Signaling in the Skeleton. J. Biol. Chem. 2009, 284, 27438–27448. [Google Scholar] [CrossRef]
- Rochefort, G.Y.; Pallu, S.; Benhamou, C.L. Osteocyte: The Unrecognized Side of Bone Tissue. Osteoporos. Int. J. Establ. Result Coop. Eur. Found. Osteoporos. Natl. Osteoporos. Found. USA 2010, 21, 1457–1469. [Google Scholar] [CrossRef]
- Jilka, R.L.; Noble, B.; Weinstein, R.S. Osteocyte Apoptosis. Bone 2013, 54, 264–271. [Google Scholar] [CrossRef]
- Kliewer, S.A.; Sundseth, S.S.; Jones, S.A.; Brown, P.J.; Wisely, G.B.; Koble, C.S.; Devchand, P.; Wahli, W.; Willson, T.M.; Lenhard, J.M.; et al. Fatty Acids and Eicosanoids Regulate Gene Expression through Direct Interactions with Peroxisome Proliferator-Activated Receptors α and γ. Proc. Natl. Acad. Sci. USA 1997, 94, 4318–4323. [Google Scholar] [CrossRef]
- Nagy, L.; Tontonoz, P.; Alvarez, J.G.A.; Chen, H.; Evans, R.M. Oxidized LDL Regulates Macrophage Gene Expression through Ligand Activation of PPARγ. Cell 1998, 93, 229–240. [Google Scholar] [CrossRef]
- Jules, J.; Ashley, J.W.; Feng, X. Selective Targeting of RANK Signaling Pathways as New Therapeutic Strategies for Osteoporosis. Expert Opin. Ther. Targets 2010, 14, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Farr, J.N.; Fraser, D.G.; Wang, H.; Jaehn, K.; Ogrodnik, M.B.; Weivoda, M.M.; Drake, M.T.; Tchkonia, T.; LeBrasseur, N.K.; Kirkland, J.L.; et al. Identification of Senescent Cells in the Bone Microenvironment. J. Bone Miner. Res. 2016, 31, 1920–1929. [Google Scholar] [CrossRef] [PubMed]
- Fehrendt, H.; Linn, T.; Hartmann, S.; Szalay, G.; Heiss, C.; Schnettler, R.; Lips, K.S. Negative Influence of a Long-Term High-Fat Diet on Murine Bone Architecture. Int. J. Endocrinol. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Al-Dujaili, S.A.; Lau, E.; Al-Dujaili, H.; Tsang, K.; Guenther, A.; You, L. Apoptotic Osteocytes Regulate Osteoclast Precursor Recruitment and Differentiation in Vitro. J. Cell. Biochem. 2011, 112, 2412–2423. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, O.D.; Laudier, D.M.; Majeska, R.J.; Sun, H.B.; Schaffler, M.B. Osteocyte Apoptosis Is Required for Production of Osteoclastogenic Signals Following Bone Fatigue in Vivo. Bone 2014, 64, 132–137. [Google Scholar] [CrossRef]
- Carrington, J.L. Aging Bone and Cartilage: Cross-Cutting Issues. Biochem. Biophys. Res. Commun. 2005, 328, 700–708. [Google Scholar] [CrossRef]
- Jilka, R.L.; Weinstein, R.S.; Parfitt, A.M.; Manolagas, S.C. Quantifying Osteoblast and Osteocyte Apoptosis: Challenges and Rewards. J. Bone Miner. Res. 2007, 22, 1492–1501. [Google Scholar] [CrossRef]
- Feng, W.; Liu, B.; Liu, D.; Hasegawa, T.; Wang, W.; Han, X.; Cui, J.; Yimin Oda, K.; Amizuka, N. Long-Term Administration of High-Fat Diet Corrects Abnormal Bone Remodeling in the Tibiae of Interleukin-6-Deficient Mice. J. Histochem. Cytochem. 2016, 64, 42–53. [Google Scholar] [CrossRef]
- Liu, H.; Huang, H.; Li, B.; Wu, D.; Wang, F.; Zheng, X.h.; Chen, Q.; Wu, B.; Fan, X. Olive Oil in the Prevention and Treatment of Osteoporosis after Artificial Menopause. Clin. Interv. Aging 2014, 9, 2087–2095. [Google Scholar] [CrossRef]
- Zhang, Q.; Riddle, R.C.; Yang, Q.; Rosen, C.R.; Guttridge, D.C.; Dirckx, N.; Faugere, M.C.; Farber, C.R.; Clemens, T.L. The RNA Demethylase FTO Is Required for Maintenance of Bone Mass and Functions to Protect Osteoblasts from Genotoxic Damage. Proc. Natl. Acad. Sci. USA 2019, 116, 17980–17989. [Google Scholar] [CrossRef]
- Hanawalt, P.C. Subpathways of Nucleotide Excision Repair and Their Regulation. Oncogene 2002, 21, 8949–8956. [Google Scholar] [CrossRef]
- Hoeijmakers, J.H.J. DNA Damage, Aging, and Cancer. N. Engl. J. Med. 2009, 361, 1475–1485. [Google Scholar] [CrossRef]
- Chandra, A.; Lin, T.; Young, T.; Tong, W.; Ma, X.; Tseng, W.J.; Kramer, I.; Kneissel, M.; Levine, M.A.; Zhang, Y.; et al. Suppression of Sclerostin Alleviates Radiation-Induced Bone Loss by Protecting Bone-Forming Cells and Their Progenitors Through Distinct Mechanisms. J. Bone Miner. Res. 2017, 32, 360–372. [Google Scholar] [CrossRef]
- Chen, Q.; Liu, K.; Robinson, A.R.; Clauson, C.L.; Blair, H.C.; Robbins, P.D.; Niedernhofer, L.J.; Ouyang, H. DNA Damage Drives Accelerated Bone Aging via an NF-κB-Dependent Mechanism. J. Bone Miner. Res. 2013, 28, 1214–1228. [Google Scholar] [CrossRef]
- Wijnhoven, S.W.P.; Beems, R.B.; Roodbergen, M.; Van Den Berg, J.; Lohman, P.H.M.; Diderich, K.; Van Der Horst, G.T.J.; Vijg, J.; Hoeijmakers, J.H.J.; Van Steeg, H. Accelerated Aging Pathology in Ad Libitum Fed XpdTTD Mice Is Accompanied by Features Suggestive of Caloric Restriction. DNA Repair 2005, 4, 1314–1324. [Google Scholar] [CrossRef]
- Diderich, K.E.M.; Nicolaije, C.; Priemel, M.; Waarsing, J.H.; Day, J.S.; Brandt, R.M.C.; Schilling, A.F.; Botter, S.M.; Weinans, H.; Van Der Horst, G.T.J.; et al. Bone Fragility and Decline in Stem Cells in Prematurely Aging DNA Repair Deficient Trichothiodystrophy Mice. Age 2012, 34, 845–861. [Google Scholar] [CrossRef]
- Carrero, D.; Soria-Valles, C.; López-Otín, C. Hallmarks of Progeroid Syndromes: Lessons from Mice and Reprogrammed Cells. Dis. Models Mech. 2016, 9, 719–735. [Google Scholar] [CrossRef]
- Burtner, C.R.; Kennedy, B.K. Progeria Syndromes and Ageing: What Is the Connection? Nat. Rev. Mol. Cell Biol. 2010, 11, 567–578. [Google Scholar] [CrossRef]
- Niedernhofer, L.J.; Garinis, G.A.; Raams, A.; Lalai, A.S.; Robinson, A.R.; Appeldoorn, E.; Odijk, H.; Oostendorp, R.; Ahmad, A.; Van Leeuwen, W.; et al. A New Progeroid Syndrome Reveals That Genotoxic Stress Suppresses the Somatotroph Axis. Nature 2006, 444, 1038–1043. [Google Scholar] [CrossRef]
- Itin, P.H.; Sarasin, A.; Pittelkow, M.R. Trichothiodystrophy: Update on the Sulfur Deficient Brittle Hair Syndromes. J. Am. Acad. Dermatol. 2001, 44, 891–924. [Google Scholar] [CrossRef]
- Leffa, D.D.; Da Silva, J.; Daumann, F.; Dajori, A.L.F.; Longaretti, L.M.; Damiani, A.P.; de Lira, F.; Campos, F.; Ferraz, A.d.B.F.; CÔrrea, D.S.; et al. Corrective Effects of Acerola (Malpighia Emarginata DC.) Juice Intake on Biochemical and Genotoxical Parameters in Mice Fed on a High-Fat Diet. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2014, 770, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Quiles, J.L.; Ochoa, J.J.; Ramirez-Tortosa, C.; Battino, M.; Huertas, J.R.; Martín, Y.; Mataix, J. Dietary Fat Type (Virgin Olive vs. Sunflower Oils) Affects Age-Related Changes in DNA Double-Strand-Breaks, Antioxidant Capacity and Blood Lipids in Rats. Exp. Gerontol. 2004, 39, 1189–1198. [Google Scholar] [CrossRef] [PubMed]
- Quiles, J.L.; Ochoa, J.J.; Ramirez-Tortosa, M.C.; Huertas, J.R.; Mataix, J. Age-Related Mitochondrial DNA Deletion in Rat Liver Depends on Dietary Fat Unsaturation. J. Gerontol. A. Biol. Sci. Med. Sci. 2006, 61, 107–114. [Google Scholar] [CrossRef]
- Ramirez-Tortosa, C.L.; Varela-López, A.; Navarro-Hortal, M.D.; Ramos-Pleguezuelos, F.M.; Márquez-Lobo, B.; Ramirez-Tortosa, M.C.; Ochoa, J.J.; Battino, M.; Quiles, J.L. Longevity and Cause of Death in Male Wistar Rats Fed Lifelong Diets Based on Virgin Olive Oil, Sunflower Oil or Fish Oil. J. Gerontol. A. Biol. Sci. Med. Sci. 2019. [Google Scholar] [CrossRef]
- Lam, Y.Y.; Ha, C.W.Y.; Hoffmann, J.M.A.; Oscarsson, J.; Dinudom, A.; Mather, T.J.; Cook, D.I.; Hunt, N.H.; Caterson, I.D.; Holmes, A.J.; et al. Effects of Dietary Fat Profile on Gut Permeability and Microbiota and Their Relationships with Metabolic Changes in Mice. Obesity 2015, 23, 1429–1439. [Google Scholar] [CrossRef] [PubMed]
- Hafner, H.; Chang, E.; Carlson, Z.; Zhu, A.; Varghese, M.; Clemente, J.; Abrishami, S.; Bagchi, D.P.; MacDougald, O.A.; Singer, K.; et al. Lactational High-Fat Diet Exposure Programs Metabolic Inflammation and Bone Marrow Adiposity in Male Offspring. Nutrients 2019, 11, 1393. [Google Scholar] [CrossRef] [PubMed]
- Schwarzer, M.; Makki, K.; Storelli, G.; Machuca-Gayet, I.; Srutkova, D.; Hermanova, P.; Martino, M.E.; Balmand, S.; Hudcovic, T.; Heddi, A.; et al. Lactobacillus Plantarum Strain Maintains Growth of Infant Mice during Chronic Undernutrition. Science 2016, 351, 854–857. [Google Scholar] [CrossRef]
- Yamasaki, M.; Hasegawa, S.; Imai, M.; Takahashi, N.; Fukui, T. High-Fat Diet-Induced Obesity Stimulates Ketone Body Utilization in Osteoclasts of the Mouse Bone. Biochem. Biophys. Res. Commun. 2016, 473, 654–661. [Google Scholar] [CrossRef]
- Carmo, L.S.; Rogero, M.M.; Cortez, M.; Yamada, M.; Jacob, P.S.; Bastos, D.H.M.; Borelli, P.; Ambrósio Fock, R. The Effects of Yerba Maté (Ilex Paraguariensis) Consumption on IL-1, IL-6, TNF-α and IL-10 Production by Bone Marrow Cells in Wistar Rats Fed a High-Fat Diet. Int. J. Vitam. Nutr. Res. 2013, 83, 26–35. [Google Scholar] [CrossRef]
- Cortez, M.; Carmo, L.S.; Rogero, M.M.; Borelli, P.; Fock, R.A. A High-Fat Diet Increases IL-1, IL-6, and TNF-α Production by Increasing NF-κB and Attenuating PPAR-γ Expression in Bone Marrow Mesenchymal Stem Cells. Inflammation 2013, 36, 379–386. [Google Scholar] [CrossRef]
- Zhou, Z.; Pan, C.; Wang, N.; Zhou, L.; Shan, H.; Gao, Y.; Yu, X. A High-Fat Diet Aggravates Osteonecrosis through a Macrophage-Derived IL-6 Pathway. Int. Immunol. 2019, 31, 263–273. [Google Scholar] [CrossRef]
- Zhang, K.; Wang, C.; Chen, Y.; Ji, X.; Chen, X.; Tian, L.; Yu, X. Preservation of High-Fat Diet-Induced Femoral Trabecular Bone Loss through Genetic Target of TNF-α. Endocrine 2015, 50, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Kitaura, H.; Zhou, P.; Kim, H.J.; Novack, D.V.; Ross, F.P.; Teitelbaum, S.L. M-CSF Mediates TNF-Induced Inflammatory Osteolysis. J. Clin. Investig. 2005, 115, 3418–3427. [Google Scholar] [CrossRef]
- Wei, S.; Kitaura, H.; Zhou, P.; Ross, F.P.; Teitelbaum, S.L. IL-1 Mediates TNF-Induced Osteoclastogenesis. J. Clin. Investig. 2005, 115, 282–290. [Google Scholar] [CrossRef]
- Ding, J.; Ghali, O.; Lencel, P.; Broux, O.; Chauveau, C.; Devedjian, J.C.; Hardouin, P.; Magne, D. TNF-α and IL-1β Inhibit RUNX2 and Collagen Expression but Increase Alkaline Phosphatase Activity and Mineralization in Human Mesenchymal Stem Cells. Life Sci. 2009, 84, 499–504. [Google Scholar] [CrossRef]
- Zhang, X.; Li, X.; Sheng, Z.; Wang, S.; Li, B.; Tao, S.; Zhang, Z. Effects of Combined Exposure to Cadmium and High-Fat Diet on Bone Quality in Male Mice. Biol. Trace Elem. Res. 2020, 193, 434–444. [Google Scholar] [CrossRef]
- Narayanan, S.A.; Metzger, C.E.; Bloomfield, S.A.; Zawieja, D.C. Inflammation-Induced Lymphatic Architecture and Bone Turnover Changes Are Ameliorated by Irisin Treatment in Chronic Inflammatory Bowel Disease. FASEB J. 2018, 32, 4848–4861. [Google Scholar] [CrossRef]
- Liu, X.-H.; Kirschenbaum, A.; Yao, S.; Levine, A.C. Androgens Promote Preosteoblast Differentiation via Activation of the Canonical Wnt Signaling Pathway. Ann. N. Y. Acad. Sci. 2007, 1116, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Nanes, M.S.; Rubin, J.; Titus, L.; Hendy, G.N.; Catherwood, B. Tumor Necrosis Factor-α Inhibits 1,25-Dihydroxyvitamin D3-Stimulated Bone Gla Protein Synthesis in Rat Osteosarcoma Cells (ROS 17/2.8) by a Pretranslational Mechanism. Endocrinology 1991, 128, 2577–2582. [Google Scholar] [CrossRef]
- Gilbert, L.; He, X.; Farmer, P.; Rubin, J.; Drissi, H.; Van Wijnen, A.J.; Lian, J.B.; Stein, G.S.; Nanes, M.S. Expression of the Osteoblast Differentiation Factor RUNX2 (Cbfa1/AML3/Pebp2αA) Is Inhibited by Tumor Necrosis Factor-α. J. Biol. Chem. 2002, 277, 2695–2701. [Google Scholar] [CrossRef]
- Abbas, S.; Clohisy, J.C.; Abu-Amer, Y. Mitogen-Activated Protein (MAP) Kinases Mediate PMMA-Induction of Osteoclasts. J. Orthop. Res. 2003, 21, 1041–1048. [Google Scholar] [CrossRef]
- Zwerina, J.; Redlich, K.; Polzer, K.; Joosten, L.; Kroenke, G.; Distler, J.; Hess, A.; Pundt, N.; Pap, T.; Hoffmann, O.; et al. TNF-Induced Structural Joint Damage Is Mediated by IL-1. Proc. Natl. Acad. Sci. USA 2007, 104, 11742–11747. [Google Scholar] [CrossRef] [PubMed]
- Polzer, K.; Joosten, L.; Gasser, J.; Distler, J.H.; Ruiz, G.; Baum, W.; Redlich, K.; Bobacz, K.; Smolen, J.S.; Van Den Berg, W.; et al. Interleukin-1 Is Essential for Systemic Inflammatory Bone Loss. Ann. Rheum. Dis. 2010, 69, 284–290. [Google Scholar] [CrossRef]
- Kim, J.-H.; Kim, A.R.; Choi, Y.H.; Jang, S.; Woo, G.-H.; Cha, J.-H.; Bak, E.-J.; Yoo, Y.-J. Tumor Necrosis Factor-α Antagonist Diminishes Osteocytic RANKL and Sclerostin Expression in Diabetes Rats with Periodontitis. PLoS ONE 2017, 12, e0189702. [Google Scholar] [CrossRef]
- Heiland, G.R.; Zwerina, K.; Baum, W.; Kireva, T.; Distler, J.H.; Grisanti, M.; Asuncion, F.; Li, X.; Ominsky, M.; Richards, W.; et al. Neutralisation of Dkk-1 Protects from Systemic Bone Loss during Inflammation and Reduces Sclerostin Expression. Ann. Rheum. Dis. 2010, 69, 2152–2159. [Google Scholar] [CrossRef]
- Watkins, B.A.; Li, Y.; Lippman, H.E.; Seifert, M.F. Omega-3 Polyunsaturated Fatty Acids and Skeletal Health1. Exp. Biol. Med. 2001, 226, 485–497. [Google Scholar] [CrossRef]
- Calder, P.C.; Zurier, R.B. Polyunsaturated Fatty Acids and Rheumatoid Arthritis. Curr. Opin. Clin. Nutr. Metab. Care 2001, 4, 115–121. [Google Scholar] [CrossRef]
- Darlington, L.G.; Stone, T.W. Antioxidants and Fatty Acids in the Amelioration of Rheumatoid Arthritis and Related Disorders. Br. J. Nutr. 2001, 85, 251–269. [Google Scholar] [CrossRef]
- Fan, Y.-Y.; Chapkin, R.S. Importance of Dietary γ-Linolenic Acid in Human Health and Nutrition. J. Nutr. 1998, 128, 1411–1414. [Google Scholar] [CrossRef]
- Kremer, J.M. Effects of Modulation of Inflammatory and Immune Parameters in Patients with Rheumatic and Inflammatory Disease Receiving Dietary Supplementation of N-3 and n-6 Fatty Acids. Lipids 1996, 31, S243–S247. [Google Scholar] [CrossRef]
- Little, C.; Parsons, T. Herbal Therapy for Treating Rheumatoid Arthritis. Cochrane Database Syst. Rev. 2001, 4, 3–26. [Google Scholar] [CrossRef]
- Rothman, D.; DeLuca, P.; Zurier, R.B. Botanical Lipids: Effects on Inflammation, Immune Responses, and Rheumatoid Arthritis. Semin. Arthritis Rheum. 1995, 25, 87–96. [Google Scholar] [CrossRef]
- Zurier, R.B. Gammalinolenic acid treatment of rheumatoid arthritis. In Medicinal Fatty Acids in Inflammation; Kremer, J.M., Ed.; Birkhäuser: Basel, Swizerland, 1998; pp. 29–43. ISBN 978-3-0348-8825-7. [Google Scholar]
- Marks, S.C.; Miller, S. Local Infusion of Prostaglandin E1 Stimulates Mandibular Bone Formation in Vivo. J. Oral Pathol. Med. 1988, 17, 500–505. [Google Scholar] [CrossRef]
- Johnson, M.M.; Swan, D.D.; Surette, M.E.; Stegner, J.; Chilton, T.; Fonteh, A.N.; Chilton, F.H. Dietary Supplementation with γ-Linolenic Acid Alters Fatty Acid Content and Eicosanoid Production in Healthy Humans. J. Nutr. 1997, 127, 1435–1444. [Google Scholar] [CrossRef]
- Barham, J.B.; Edens, M.B.; Fonteh, A.N.; Johnson, M.M.; Easter, L.; Chilton, F.H. Addition of Eicosapentaenoic Acid to γ-Linolenic Acid-Supplemented Diets Prevents Serum Arachidonic Acid Accumulation in Humans. J. Nutr. 2000, 130, 1925–1931. [Google Scholar] [CrossRef]
- Giampieri, F.; Afrin, S.; Forbes-Hernandez, T.Y.; Gasparrini, M.; Cianciosi, D.; Reboredo-Rodriguez, P.; Varela-Lopez, A.; Quiles, J.L.; Battino, M. Autophagy in Human Health and Disease: Novel Therapeutic Opportunities. Antioxid. Redox Signal. 2019, 30, 577–634. [Google Scholar] [CrossRef]
- Rendina-Ruedy, E.; Graef, J.L.; Lightfoot, S.A.; Ritchey, J.W.; Clarke, S.L.; Lucas, E.A.; Smith, B.J. Impaired Glucose Tolerance Attenuates Bone Accrual by Promoting the Maturation of Osteoblasts: Role of Beclin1-Mediated Autophagy. Bone Rep. 2016, 5, 199–207. [Google Scholar] [CrossRef]
- Piemontese, M.; Onal, M.; Xiong, J.; Han, L.; Thostenson, J.D.; Almeida, M.; O’Brien, C.A. Low Bone Mass and Changes in the Osteocyte Network in Mice Lacking Autophagy in the Osteoblast Lineage. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Liu, K.; Zhao, E.; Ilyas, G.; Lalazar, G.; Lin, Y.; Haseeb, M.; Tanaka, K.E.; Czaja, M.J. Impaired Macrophage Autophagy Increases the Immune Response in Obese Mice by Promoting Proinflammatory Macrophage Polarization. Autophagy 2015, 11, 271–284. [Google Scholar] [CrossRef] [PubMed]
- Singer, K.; Maley, N.; Mergian, T.; DelProposto, J.; Cho, K.W.; Zamarron, B.F.; Martinez-Santibanez, G.; Geletka, L.; Muir, L.; Wachowiak, P.; et al. Differences in Hematopoietic Stem Cells Contribute to Sexually Dimorphic Inflammatory Responses to High Fat Diet-Induced Obesity. J. Biol. Chem. 2015, 290, 13250–13262. [Google Scholar] [CrossRef] [PubMed]
- Shirakawa, K.; Yano, W.; Inoue, K.; Katsumata, Y.; Endo, J.; Sano, M. Influence of Long Term Administration of Tofogliflozin on Chronic Inflammation of Visceral Adipose Tissue in Mice with Obesity Induced by a High-Fat Diet. PLoS ONE 2019, 14, e0211387. [Google Scholar] [CrossRef]
- Rodda, S.J.; McMahon, A.P. Distinct Roles for Hedgehog and Caronical Wnt Signaling in Specification, Differentiation and Maintenance of Osteoblast Progenitors. Development 2006, 133, 3231–3244. [Google Scholar] [CrossRef]
- Kopchick, J.J.; Berryman, D.E.; Puri, V.; Lee, K.Y.; Jorgensen, J.O.L. The Effects of Growth Hormone on Adipose Tissue: Old Observations, New Mechanisms. Nat. Rev. Endocrinol. 2020, 16, 135–146. [Google Scholar] [CrossRef]
- Deshpande, N.; Hulbert, A.J. Dietary ω6 Fatty Acids and the Effects of Hyperthyroidism in Mice. J. Endocrinol. 1995, 144, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Souza, L.L.; Cordeiro, A.; Oliveira, L.S.; De Paula, G.S.M.; Faustino, L.C.; Maria, T.O.C.; Oliveira, K.J.; Cabanelas, C.P.M. Thyroid Hormone Contributes to the Hypolipidemic Effect of Polyunsaturated Fatty Acids from Fish Oil: In Vivo Evidence for Cross Talking Mechanisms. J. Endocrinol. 2011, 211, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Seck, T.; Scheppach, B.; Scharla, S.; Diel, I.; Blum, W.F.; Bismar, H.; Schmid, G.; Krempien, B.; Ziegler, R.; Pfeilschifter, J. Concentration of Insulin-Like Growth Factor (IGF)-I and -II in Iliac Crest Bone Matrix from Pre- and Postmenopausal Women: Relationship to Age, Menopause, Bone Turnover, Bone Volume, and Circulating IGFs1. J. Clin. Endocrinol. Metab. 1998, 83, 2331–2337. [Google Scholar] [CrossRef] [PubMed]
- Lips, P.; Duong, T.; Oleksik, A.; Black, D.; Cummings, S.; Cox, D.; Nickelsen, T. A Global Study of Vitamin D Status and Parathyroid Function in Postmenopausal Women with Osteoporosis: Baseline Data from the Multiple Outcomes of Raloxifene Evaluation Clinical Trial. J. Clin. Endocrinol. Metab. 2001, 86, 1212–1221. [Google Scholar] [CrossRef] [PubMed]
- Sigurdsson, G.; Franzson, L.; Steingrimsdottir, L.; Sigvaldason, H. The Association between Parathyroid Hormone, Vitamin D and Bone Mineral Density in 70-Year-Old Icelandic Women. Osteoporos. Int. 2000, 11, 1031–1035. [Google Scholar] [CrossRef]
- Boonen, S.; Cheng, X.G.; Nijs, J.; Nicholson, P.H.F.; Verbeke, G.; Lesaffre, E.; Aerssens, J.; Dequeker, J. Factors Associated with Cortical and Trabecular Bone Loss as Quantified by Peripheral Computed Tomography (PQCT) at the Ultradistal Radius in Aging Women. Calcif. Tissue Int. 1997, 60, 164–170. [Google Scholar] [CrossRef]
- Garnero, P.; Tsouderos, Y.; Marton, I.; Pelissier, C.; Varin, C.; Delmas, P.D. Effects of Intranasal 17 β-Estradiol on Bone Turnover and Serum Insulin-like Growth Factor I in Postmenopausal Women. J. Clin. Endocrinol. Metab. 1999, 84, 2390–2397. [Google Scholar] [CrossRef]
- Cappon, J.P.; Ipp, E.; Brasel, J.A.; Cooper, D.M. Acute Effects of High Fat and High Glucose Meals on the Growth Hormone Response to Exercise. J. Clin. Endocrinol. Metab. 1993, 76, 1418–1422. [Google Scholar] [CrossRef]
- Whitley, H.A.; Humphreys, S.M.; Campbell, I.T.; Keegan, M.A.; Jayanetti, T.D.; Sperry, D.A.; MacLaren, D.P.; Reilly, T.; Frayn, K.N. Metabolic and Performance Responses during Endurance Exercise after High-Fat and High-Carbohydrate Meals. J. Appl. Physiol. 1998. [Google Scholar] [CrossRef]
- Bhathena, S.J.; Berlin, E.; Judd, J.T.; Kim, Y.C.; Law, J.S.; Bhagavan, H.N.; Ballard-Barbash, R.; Nair, P.P. Effects of Ω3 Fatty Acids and Vitamin E on Hormones Involved in Carbohydrate and Lipid Metabolism in Men. Am. J. Clin. Nutr. 1991, 54, 684–688. [Google Scholar] [CrossRef]
- Volkan, B.; Fettah, A.; Lek, A.; Kara, S.S.; Kurt, N.; Cayr, A. Bone Mineral Density and Vitamin K Status in Children with Celiac Disease: Is There a Relation? Turk. J. Gastroenterol. 2018, 29, 215–220. [Google Scholar] [CrossRef]
- Lai, P.; Song, Q.; Yang, C.; Li, Z.; Liu, S.; Liu, B.; Li, M.; Deng, H.; Cai, D.; Jin, D.; et al. Loss of Rictor with Aging in Osteoblasts Promotes Age-Related Bone Loss. Cell Death Dis. 2016, 7. [Google Scholar] [CrossRef]
- Mendes, M.M.; Hart, K.H.; Lanham-New, S.A.; Botelho, P.B. Association between 25-Hydroxyvitamin D, Parathyroid Hormone, Vitamin D and Calcium Intake, and Bone Density in Healthy Adult Women: A Cross-Sectional Analysis from the D-SOL Study. Nutrients 2019, 11, 1267. [Google Scholar] [CrossRef]
- Li, M.; Lv, F.; Zhang, Z.; Deng, W.; Li, Y.; Deng, Z.; Jiang, Y.; Wang, O.; Xing, X.; Xu, L.; et al. Establishment of a Normal Reference Value of Parathyroid Hormone in a Large Healthy Chinese Population and Evaluation of Its Relation to Bone Turnover and Bone Mineral Density. Osteoporos. Int. 2016, 27, 1907–1916. [Google Scholar] [CrossRef]
- Cipriani, C.; Colangelo, L.; Santori, R.; Renella, M.; Mastrantonio, M.; Minisola, S.; Pepe, J. The Interplay Between Bone and Glucose Metabolism. Front. Endocrinol. 2020, 11, 122. [Google Scholar] [CrossRef]
- Geloneze, B.; De Oliveira, M.D.S.; Vasques, A.C.J.; Novaes, F.S.; Pareja, J.C.; Tambascia, M.A. Impaired Incretin Secretion and Pancreatic Dysfunction with Older Age and Diabetes. Metabolism 2014, 63, 922–929. [Google Scholar] [CrossRef]
- Napoli, N.; Schwartz, A.V.; Schafer, A.L.; Vittinghoff, E.; Cawthon, P.M.; Parimi, N.; Orwoll, E.; Strotmeyer, E.S.; Hoffman, A.R.; Barrett-Connor, E.; et al. Vertebral Fracture Risk in Diabetic Elderly Men: The MrOS Study. J. Bone Miner. Res. 2018, 33, 63–69. [Google Scholar] [CrossRef]
- Patsch, J.M.; Burghardt, A.J.; Yap, S.P.; Baum, T.; Schwartz, A.V.; Joseph, G.B.; Link, T.M. Increased Cortical Porosity in Type 2 Diabetic Postmenopausal Women with Fragility Fractures. J. Bone Miner. Res. 2013, 28, 313–324. [Google Scholar] [CrossRef]
- Burghardt, A.J.; Issever, A.S.; Schwartz, A.V.; Davis, K.A.; Masharani, U.; Majumdar, S.; Link, T.M. High-Resolution Peripheral Quantitative Computed Tomographic Imaging of Cortical and Trabecular Bone Microarchitecture in Patients with Type 2 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2010, 95, 5045–5055. [Google Scholar] [CrossRef] [PubMed]
- Poundarik, A.A.; Wu, P.C.; Evis, Z.; Sroga, G.E.; Ural, A.; Rubin, M.; Vashishth, D. A Direct Role of Collagen Glycation in Bone Fracture. J. Mech. Behav. Biomed. Mater. 2015, 52, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Briggs, D.I.; Enriori, P.J.; Lemus, M.B.; Cowley, M.A.; Andrews, Z.B. Diet-Induced Obesity Causes Ghrelin Resistance in Arcuate NPY/AgRP Neurons. Endocrinology 2010, 151, 4745–4755. [Google Scholar] [CrossRef] [PubMed]
- Numao, S.; Kawano, H.; Endo, N.; Yamada, Y.; Konishi, M.; Takahashi, M.; Sakamoto, S. Short-Term Low Carbohydrate/High-Fat Diet Intake Increases Postprandial Plasma Glucose and Glucagon-like Peptide-1 Levels during an Oral Glucose Tolerance Test in Healthy Men. Eur. J. Clin. Nutr. 2012, 66, 926–931. [Google Scholar] [CrossRef]
- Van Herpen, N.A.; Schrauwen-Hinderling, V.B.; Schaart, G.; Mensink, R.P.; Schrauwen, P. Three Weeks on a High-Fat Diet Increases Intrahepatic Lipid Accumulation and Decreases Metabolic Flexibility in Healthy Overweight Men. J. Clin. Endocrinol. Metab. 2011, 96, e691–e695. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, J.; Wu, G.; Sun, J.; Wang, Y.; Guo, H.; Shi, Y.; Cheng, X.; Tang, X.; Le, G. Dietary Methionine Restriction Regulated Energy and Protein Homeostasis by Improving Thyroid Function in High Fat Diet Mice. Food Funct. 2018, 9, 3718–3731. [Google Scholar] [CrossRef]
- Shao, S.S.; Zhao, Y.F.; Song, Y.F.; Xu, C.; Yang, J.M.; Xuan, S.M.; Yan, H.L.; Yu, C.X.; Zhao, M.; Xu, J.; et al. Dietary High-Fat Lard Intake Induces Thyroid Dysfunction and Abnormal Morphology in Rats. Acta Pharmacol. Sin. 2014, 35, 1411–1420. [Google Scholar] [CrossRef]
- Xia, S.F.; Qiu, Y.Y.; Chen, L.M.; Jiang, Y.Y.; Huang, W.; Xie, Z.X.; Tang, X.; Sun, J. Myricetin Alleviated Hepatic Steatosis by Acting on MicroRNA-146b/Thyroid Hormone Receptor b Pathway in High-Fat Diet Fed C57BL/6J Mice. Food Funct. 2019, 10, 1465–1477. [Google Scholar] [CrossRef]
- Hollowell, J.G.; Staehling, N.W.; Dana Flanders, W.; Harry Hannon, W.; Gunter, E.W.; Spencer, C.A.; Braverman, L.E. Serum TSH, T4, and Thyroid Antibodies in the United States Population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J. Clin. Endocrinol. Metab. 2002, 87, 489–499. [Google Scholar] [CrossRef]
- Waring, A.C.; Arnold, A.M.; Newman, A.B.; Bùžková, P.; Hirsch, C.; Cappola, A.R. Longitudinal Changes in Thyroid Function in the Oldest Old and Survival: The Cardiovascular Health Study All-Stars Study. J. Clin. Endocrinol. Metab. 2012, 97, 3944–3950. [Google Scholar] [CrossRef]
- Bremner, A.P.; Feddema, P.; Leedman, P.J.; Brown, S.J.; Beilby, J.P.; Lim, E.M.; Wilson, S.G.; O’Leary, P.C.; Walsh, J.P. Age-Related Changes In Thyroid Function: A Longitudinal Study of a Community-Based Cohort. J. Clin. Endocrinol. Metab. 2012, 97, 1554–1562. [Google Scholar] [CrossRef]
- Calsolaro, V.; Niccolai, F.; Pasqualetti, G.; Maria Calabrese, A.; Polini, A.; Okoye, C.; Magno, S.; Caraccio, N.; Monzani, F. Overt and Subclinical Hypothyroidism in the Elderly: When to Treat? Front. Endocrinol. 2019, 10. [Google Scholar] [CrossRef]
- Fade, J.V.; Franklyn, J.A.; Cross, K.W.; Jones, S.C.; Sheppard, M.C. Prevalence and Follow-up of Abnormal Thyrotrophin (TSH) Concentrations in the Elderly in the United Kingdom. Clin. Endocrinol. 1991, 34, 77–84. [Google Scholar] [CrossRef]
- Meyerovitch, J.; Rotman-Pikielny, P.; Sherf, M.; Battat, E.; Levy, Y.; Surks, M.I. Serum Thyrotropin Measurements in the Community: Five-Year Follow-up in a Large Network of Primary Care Physicians. Arch. Intern. Med. 2007, 167, 1533–1538. [Google Scholar] [CrossRef] [PubMed]
- Nagata, M.; Suzuki, A.; Sekiguchi, S.; Ono, Y.; Nishiwaki-Yasuda, K.; Itoi, T.; Yamamoto, S.; Imamura, S.; Katoh, T.; Hayakawa, N.; et al. Subclinical Hypothyroidism Is Related to Lower Heel QUS in Postmenopausal Women. Endocr. J. 2007, 54, 625–630. [Google Scholar] [CrossRef]
- Lee, J.S.; Bůžková, P.; Fink, H.A.; Vu, J.; Carbone, L.; Chen, Z.; Cauley, J.; Bauer, D.C.; Cappola, A.R.; Robbins, J. Subclinical Thyroid Dysfunction and Incident Hip Fracture in Older Adults. Arch. Intern. Med. 2010, 170, 1876–1883. [Google Scholar] [CrossRef]
- Waring, A.C.; Harrison, S.; Fink, H.A.; Samuels, M.H.; Cawthon, P.M.; Zmuda, J.M.; Orwoll, E.S.; Bauer, D.C. A Prospective Study of Thyroid Function, Bone Loss, and Fractures in Older Men: The MrOS Study. J. Bone Miner. Res. 2013, 28, 472–479. [Google Scholar] [CrossRef]
- Bauer, D.C.; Ettinger, B.; Nevitt, M.C.; Stone, K.L. Risk for Fracture in Women with Low Serum Levels of Thyroid-Stimulating Hormone. Ann. Intern. Med. 2001, 134, 561–568. [Google Scholar] [CrossRef]
- Bauer, D.C.; Nevitt, M.C.; Ettinger, B.; Stone, K. Low Thyrotropin Levels Are Not Associated with Bone Loss in Older Women: A Prospective Study*. J. Clin. Endocrinol. Metab. 1997, 82, 2931–2936. [Google Scholar] [CrossRef] [PubMed]
- McKane, W.R.; Khosla, S.; Ristela, J.; Robins, S.P.; Muhs, J.M.; Riggs, B.L. Role of Estrogen Deficiency in Pathogenesis of Secondary Hyperparathyroidism and Increased Bone Resorption in Elderly Women. Obst. Gynecol. Survey 1997, 52, 429–431. [Google Scholar] [CrossRef]
- Dobnig, H.; Turner, R.T. The Effects of Programmed Administration of Human Parathyroid Hormone Fragment (1–34) on Bone Histomorphometry and Serum Chemistry in Rats. Endocrinology 1997, 138, 4607–4612. [Google Scholar] [CrossRef] [PubMed]
- Ledger, G.A.; Burritt, M.F.; Kao, P.C.; O’fallon, W.M.; Riggs, B.L.; Khosla, S. Abnormalities of Parathyroid Hormone Secretion in Elderly Women That Are Reversible by Short Term Therapy with 1,25-Dihydroxyvitamin D3. J. Clin. Endocrinol. Metab. 1994, 79, 211–216. [Google Scholar] [CrossRef]
- Haden, S.T.; Brown, E.M.; Hurwitz, S.; Scott, J.; Fuleihan, G.E.H. The Effects of Age and Gender on Parathyroid Hormone Dynamics. Clin. Endocrinol. 2000, 52, 329–338. [Google Scholar] [CrossRef]
- Need, A.G.; O’Loughlin, P.D.; Morris, H.A.; Horowitz, M.; Nordin, B.E.C. The Effects of Age and Other Variables on Serum Parathyroid Hormone in Postmenopausal Women Attending an Osteoporosis Center. J. Clin. Endocrinol. Metab. 2004, 89, 1646–1649. [Google Scholar] [CrossRef]
- Yuen, N.K.; Ananthakrishnan, S.; Campbell, M.J. Hyperparathyroidism of Renal Disease. Perm. J. 2016, 20. [Google Scholar] [CrossRef]
- Liu, B.Y.; Wu, P.W.; Hsu, Y.T.; Leu, J.S.; Wang, J.T. Estrogen Blocks Parathyroid Hormone-Stimulated Osteoclast-like Cell Formation in Modulating Differentiation of Mouse Marrow Stromal Cells in Vitro. J. Formos. Med. Assoc. 2002, 101, 24–33. [Google Scholar]
- Martins, J.S.; Palhares, M.D.O.; Teixeira, O.C.M.; Gontijo Ramos, M. Vitamin D Status and Its Association with Parathyroid Hormone Concentration in Brazilians. J. Nutr. Metab. 2017, 2017. [Google Scholar] [CrossRef]
- Corapi, K.M.; McMahon, G.M.; Wenger, J.B.; Seifter, J.L.; Bhan, I. Association of Loop Diuretic Use with Higher Parathyroid Hormone Levels in Patients with Normal Renal Function. JAMA Int. Med. 2015, 175, 137–138. [Google Scholar] [CrossRef][Green Version]
- Ledger, G.A.; Burritt, M.F.; Kao, P.C.; O’Fallon, W.M.; Riggs, B.L.; Khosla, S. Role of Parathyroid Hormone in Mediating Nocturnal and Age-Related Increases in Bone Resorption. J. Clin. Endocrinol. Metab. 1995, 80, 3304–3310. [Google Scholar] [CrossRef]
- Delmez, J.A.; Tindira, C.; Grooms, P.; Dusso, A.; Windus, D.W.; Slatopolsky, E. Parathyroid Hormone Suppression by Intravenous 1,25-Dihydroxyvitamin D. A Role for Increased Sensitivity to Calcium. J. Clin. Investig. 1989, 83, 1349–1355. [Google Scholar] [CrossRef] [PubMed]
- Khosla, S. Update in Male Osteoporosis. J. Clin. Endocrinol. Metab. 2010, 95, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Chen, Z.; Wang, H.; Ding, K. Tetrahydropalmatine Prevents High-Fat Diet-Induced Hyperlipidemia in Golden Hamsters (Mesocricetus Auratus). Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2018, 24, 6564–6572. [Google Scholar] [CrossRef] [PubMed]
- Park, J.M.; Park, C.Y.; Han, S.N. High Fat Diet-Induced Obesity Alters Vitamin D Metabolizing Enzyme Expression in Mice. BioFactors 2015, 41, 175–182. [Google Scholar] [CrossRef]
- Song, Q.; Sergeev, I.N. High Vitamin D and Calcium Intakes Increase Bone Mineral (Ca and P) Content in High-Fat Diet-Induced Obese Mice. Nutr. Res. 2015, 35, 146–154. [Google Scholar] [CrossRef]
- Jung, Y.S.; Wu, D.; Smith, D.; Meydani, S.N.; Han, S.N. Dysregulated 1,25-Dihydroxyvitamin D Levels in High-Fat Diet–Induced Obesity Can Be Restored by Changing to a Lower-Fat Diet in Mice. Nutr. Res. 2018, 53, 51–60. [Google Scholar] [CrossRef]
- Moseley, K.F.; Weaver, C.M.; Appel, L.; Sebastian, A.; Sellmeyer, D.E. Potassium Citrate Supplementation Results in Sustained Improvement in Calcium Balance in Older Men and Women. J. Bone Miner. Res. 2013, 28, 497–504. [Google Scholar] [CrossRef]
- Maurer, M.; Riesen, W.; Muser, J.; Hulter, H.N.; Krapf, R. Neutralization of Western Diet Inhibits Bone Resorption Independently of K Intake and Reduces Cortisol Secretion in Humans. Am. J. Physiol. Ren. Physiol. 2003, 284, 32–40. [Google Scholar] [CrossRef]
- Sage, A.P.; Lu, J.; Atti, E.; Tetradis, S.; Ascenzi, M.G.; Adams, D.J.; Demer, L.L.; Tintut, Y. Hyperlipidemia Induces Resistance to PTH Bone Anabolism in Mice via Oxidized Lipids. J. Bone Miner. Res. 2011, 26, 1197–1206. [Google Scholar] [CrossRef]
- Deftos, L.J.; Miller, M.M.; Burton, D.W. A High-Fat Diet Increases Calcitonin Secretion in the Rat. Bone Miner. 1989, 5, 303–308. [Google Scholar] [CrossRef]
- Khosla, S.; Atkinson, E.J.; Melton, L.J.; Riggs, B.L. Effects of Age and Estrogen Status on Serum Parathyroid Hormone Levels and Biochemical Markers of Bone Turnover in Women: A Population-Based Study 1. J. Clin. Endocrinol. Metab. 1997, 82, 1522–1527. [Google Scholar] [CrossRef][Green Version]
- Cauley, J.A. Estrogen and Bone Health in Men and Women; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 99. [Google Scholar]
- Noh, D.; Lim, Y.; Lee, H.; Kim, H.; Kwon, O. Soybean-Hop Alleviates Estrogen Deficiency-Related Bone Loss and Metabolic Dysfunction in Ovariectomized Rats Fed a High-Fat Diet. Molecules 2018, 23, 1205. [Google Scholar] [CrossRef]
- Ikegami, H.; Kawawa, R.; Ichi, I.; Ishikawa, T.; Koike, T.; Aoki, Y.; Fujiwara, Y. Excessive Vitamin E Intake Does Not Cause Bone Loss in Male or Ovariectomized Female Mice Fed Normal or High-Fat Diets. J. Nutr. 2017, 147, 1932–1937. [Google Scholar] [CrossRef]
- Dong, X.-L.; Li, C.-M.; Cao, S.-S.; Zhou, L.-P.; Wong, M.-S. A High-Saturated-Fat, High-Sucrose Diet Aggravates Bone Loss in Ovariectomized Female Rats. J. Nutr. 2016, 146, 1172–1179. [Google Scholar] [CrossRef]
- Cao, J.J.; Gregoire, B.R. A High-Fat Diet Increases Body Weight and Circulating Estradiol Concentrations but Does Not Improve Bone Structural Properties in Ovariectomized Mice. Nutr. Res. 2016, 36, 320–327. [Google Scholar] [CrossRef]
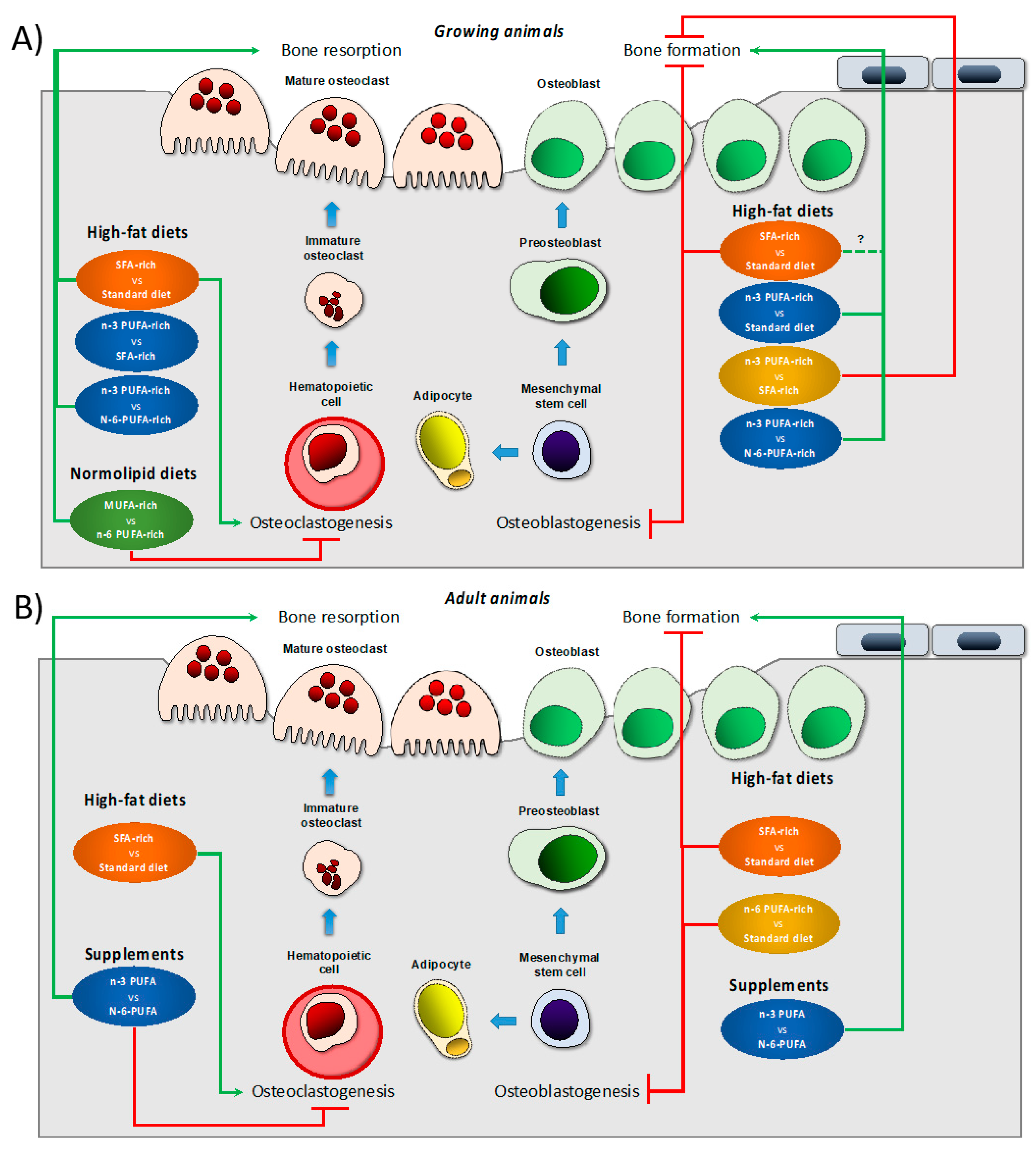
| Model; Age | Experimental Diet vs. Control Diet;Duration | Main Changes vs. Control Diet | Ref. |
|---|---|---|---|
| Growing animals | |||
| Male Wistar rat; 3 wks | SFA-HFD al (40%E beef tallow) vs. SD al (AIN-93G with soybean oil asa fat source); 8 wks |
| [38] |
| n-6 PUFA-rich HFD al (40%E soybean oil) vs. SFA-HFD al (40%E beef tallow); 8 wks |
| [38] | |
| n-6 PUFA-rich HFD al (40%E corn oil) vs. SFA-HFD al (40%E beef tallow); 8 wks |
| [38] | |
| n-3 PUFA- rich HFD al (40%E linseed oil) vs. SFA-HFD al (40%E beef tallow); 8 wks |
| [38] | |
| n-3 PUFA- rich HFD al (40%E linseed oil) vs. SD al (AIN-93G with soybean oil as fat source); 8 wks |
| [38] | |
| Male Wistar rats; Weanling | MUFA-rich SD al (AIN-93 diet with 9.5%E extra virgin olive oil) vs. n-6 PUFA-rich SD al (AIN-93 diet 9.5%E sunflower oil); 6 m |
| [46] |
| MUFA-rich SD al (AIN-93 diet with 9.5%E extra virgin olive oil) vs. n-6 PUFA-rich SD al (AIN-93 diet 9.5%E sunflower oil); 6 m |
| [46] | |
| MUFA-rich SD al (AIN-93 diet with 9.5%E extra virgin olive oil) vs. n-6 PUFA-rich SD al (AIN-93 diet 9.5%E sunflower oil); 24 m |
| [47] | |
| n-3 PUFA-rich SD al (AIN-93 diet 9.5%E fish oil); vs. n-6 PUFA-rich SD al (AIN-93 diet 9.5%E sunflower oil); 24 m |
| [47] | |
| MUFA-rich SD al (AIN-93 diet with 9.5%E extra virgin olive oil) vs. n-3 PUFA-rich SD al (AIN-93 diet 9.5%E fish oil); 24 m |
| [47] | |
| Male Sprague-Dawley rats; Weanling | HFD al (AIN93G with added 70 g/kg of safflower oil + menhaden oil) with n-6/n-3 PUFA ratio = 23.8, 9.8 2.6 or 1.2; 42 d |
| [48] |
| Female Sprague Dawley rats; 4 wks | HFD al (12wt% tuna oil) vs. HFD al (12wt% corn oil); 8 wks |
| [39] |
| HFD al (12wt% flaxseed oil) vs. HFD al (12wt% corn oil); 8 wks |
| [39] | |
| HFD al (12wt% menhaden oil) vs. HFD al (12wt% corn oil); 8 wks |
| [39] | |
| Male C57BL/6J mice; 5 wks | SFA-HFD al (60%E lard) vs. SD al (D12450B chow with 10%E fat); 12 wks |
| [40] |
| Male C57BL/6J mice; 6 wks | SFA-HFD al (60%E lard) vs. SD al (LabDiet 5LOD with 13.5% E lard); 20 wks |
| [16] |
| Male C57BL/6J mice; 6 wks | SFA-HFD al (60%E lard) vs. SD al (12%E fat); 12 wks |
| [41] |
| Male C57BL/6 mice; 4 wks | SFA-HFD al (21.2%E lard) vs. SD al (4.8%E fat); 13 wks |
| [49] |
| Male BALB/cByJ mice; 7 wks | SFA-HFD al (45%E lard) vs. SD al (13.5%E fat); 15 wks |
| [42] |
| Male Wistar rats; 9 wks | SFA-HFD al (24% fat with 100 g/kg of bw per day of ground nut and 50 g/kg of bw per day dried coconut) vs. SD al; 38 wks |
| [50] |
| Male Sprague-Dawley rat; 40 d | SFA-HFD al (20wt% coconut oil) vs. n-3 PUFA-HFD al (20wt% flaxseed oil) or n-6 PUFA-HFD al (20wt% safflower oil); 65 d |
| [51] |
| Newly hatched chicks; 4 d | n-3-rich PUFA diet al (menhaden oil + safflower oil at 90 g/kg) vs. n-6-rich PUFA diet al (soybean oil + safflower oil at 90 g/kg); 17 d |
| [52] |
| Adult animals | |||
| Male Sprague-Dawley rat; 200 g | Cholesterol-enriched HFD al (10.0 g cholesterol, 20.0 g sodium-cholate, and 112.0 g crude fat %per kg dry matter) vs. SD al (50.83 g crude fat %per kg dry matter); 114 d |
| [35] |
| Male F344 × BNF1 rats; 12 m | n-3 PUFA-rich diet (167 g safflower oil + 33 g menhaden oil) vs. n-6 PUFA-rich diet (200 g safflower oil) or n-3 + n-6 PUFA-rich diet (190 menhaden oil + 10 g corn oil); 20 wks |
| [53] |
| Male F344 × BNF1 rats; 12 m | n-3 PUFA-rich diet (167 g safflower oil + 33 g menhaden oil) vs. n-6 PUFA-rich diet (200 g safflower oil) or n-3 + n-6 PUFA-rich diet (190 menhaden oil + 10 g corn oil); 20 wks |
| [44] |
| Male C57BL/6J mice; 8 wks | SFA-HFD al (35wt% lard) vs. SD al (6wt% fat); 20 wks |
| [54] |
| Male C57BL/6 mice; 3 m | HFD al (45%E) vs. SD al (12%E fat); 11 wks |
| [55] |
| Male C57BL/6J; 4 m | HFD al (45%E) vs. SD al (11%E fat); 8 wks |
| [56] |
| Female C57BL/6J mice; 8 m | SFA-HFD al (45%E lard) vs. SD al (10%E fat); 8 wks |
| [33] |
| MUFA-rich HFD al (45%E olive oil) vs. SFA-HFD al (45%E lard); 8 wks |
| [33] | |
| Female C57BL/6J mice; 13 m | n-6 PUFA-rich HFD al (19.5%E corn oil) al vs. SD al (9.5%E fat); 26 wks |
| [57] |
| n-3 PUFA-rich HFD al (19.5%E fish oil) vs. n-6 PUFA-rich HFD al (19.5%E corn oil); 26 wks |
| [57] | |
| Roosters; 40 wks | HFD al (8% palmitic acid) vs. LFD al (8% cellulose); 20 wks |
| [45] |
| C57BL/6 mice | Cholesterol-enriched HFD al (15.8wt% fat + 1.25wt% cholesterol) vs. SD al (6wt% fat), 7 m |
| [36] |
| C3H/HeJ mice | Cholesterol-enriched HFD al (15.8wt% fat + 1.25wt% cholesterol) vs. SD al (6wt% fat), 7 m |
| [36] |
| Aged animals | |||
| OVX female Sprague Dawley; 3 m | DHA-rich diets (HP5 and LP5) High-PUFA diet vs. low-PUFA diet with a ratio of n-6/n-3 PUFAs of 5:1 or 10:1 (110.4 g/kg of fat from safflower oil (110.4 g/kg of high-oleate safflower oil blended with n-3 PUFAs); 12 wks | Fatty acid analyses confirmed that the dietary ratio of 5:1 significantly elevated the amount of DHA in the periosteum, marrow and cortical and trabecular bones of the femur.
Regardless of the dietary PUFA content, DHA in the 5:1 diets (HP5 and LP5) preserved rat femur BMC in the absence of estrogen | [58] |
| OVX female C57BL/6J mice; 8 wks | AIN-93 diet al (10%E virgin olive oil) vs. AIN-93 diet al (10%E refined olive oil); 4 wks |
| [59] |
| Marrow stromal cells from C57BL/6 mice fed a high fat, atherogenic diet failed to undergo osteogenic differentiation in vitro | [37] | ||
| Female Wistar rats; 56 m | HFD al (31%E peanut + canola seed oil); 19 wks |
| [59] |
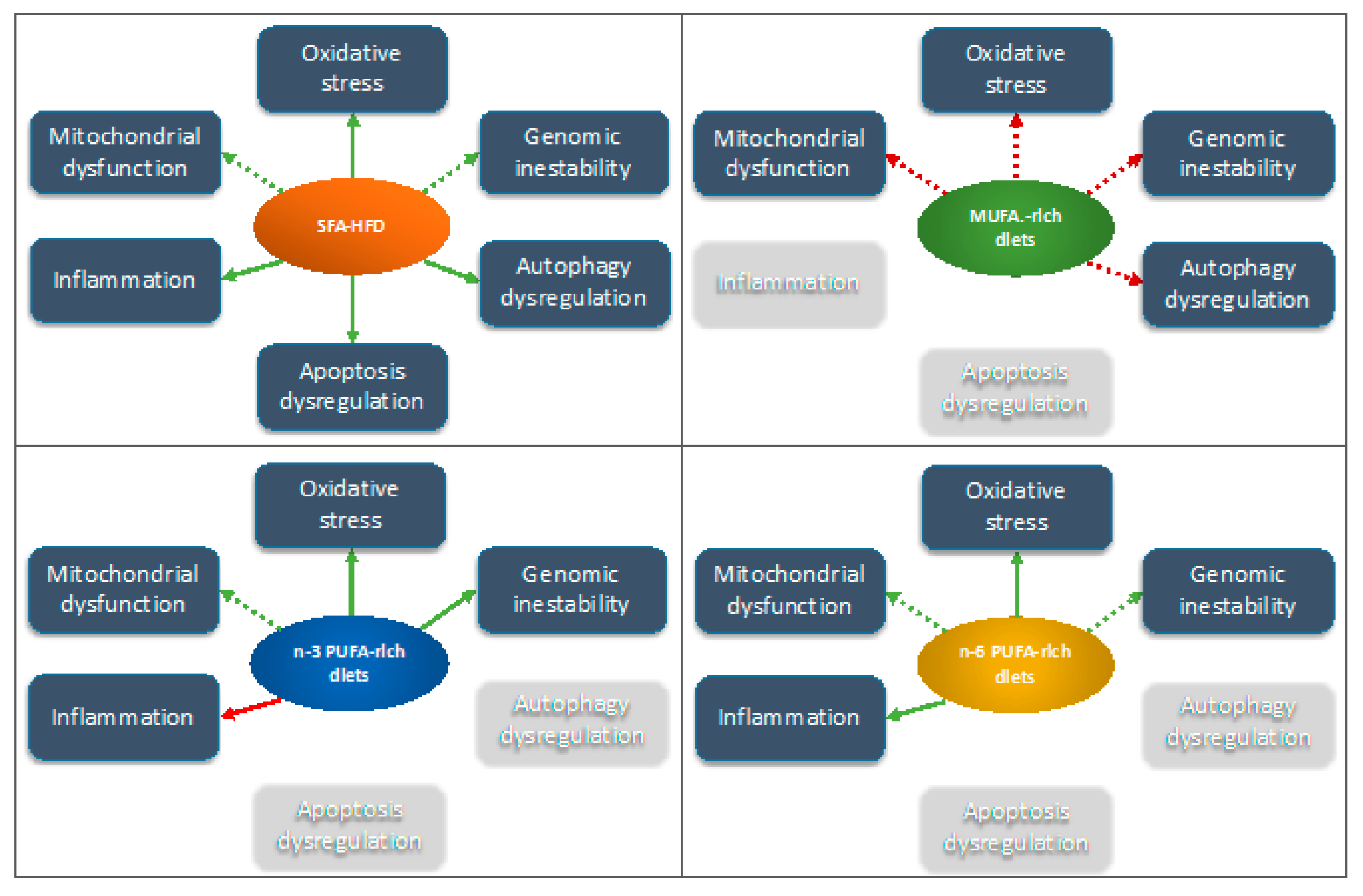
| Diet | Possible Effect | Ref. | |
|---|---|---|---|
| Oxidative stress | SFA-rich HFD |
| [59,63,97,98,99] |
| MUFA-rich diet |
| [46,47,136] | |
| n-6 PUFA-rich diet |
| [46,47,136] | |
| Apoptotis | SFA-rich HFD |
| [116,121] |
| Genomic inestability | SFA-rich HFD |
| [99,134] |
| n-3 PUFA-rich HFD |
| [100] | |
| MUFA-rich diet |
| [46,136] | |
| n-6 PUFA-rich diet |
| [46,136] | |
| Inflammation | SFA-rich HFD |
| [62,141,142,143] |
| n-3 PUFA-rich HFD |
| [62] | |
| Autophagy dysregulation | SFA-rich HFD |
| [173] |
| MUFA-rich diet |
| [47] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romero-Márquez, J.M.; Varela-López, A.; Navarro-Hortal, M.D.; Badillo-Carrasco, A.; Forbes-Hernández, T.Y.; Giampieri, F.; Domínguez, I.; Madrigal, L.; Battino, M.; Quiles, J.L. Molecular Interactions between Dietary Lipids and Bone Tissue during Aging. Int. J. Mol. Sci. 2021, 22, 6473. https://doi.org/10.3390/ijms22126473
Romero-Márquez JM, Varela-López A, Navarro-Hortal MD, Badillo-Carrasco A, Forbes-Hernández TY, Giampieri F, Domínguez I, Madrigal L, Battino M, Quiles JL. Molecular Interactions between Dietary Lipids and Bone Tissue during Aging. International Journal of Molecular Sciences. 2021; 22(12):6473. https://doi.org/10.3390/ijms22126473
Chicago/Turabian StyleRomero-Márquez, Jose M., Alfonso Varela-López, María D. Navarro-Hortal, Alberto Badillo-Carrasco, Tamara Y. Forbes-Hernández, Francesca Giampieri, Irma Domínguez, Lorena Madrigal, Maurizio Battino, and José L. Quiles. 2021. "Molecular Interactions between Dietary Lipids and Bone Tissue during Aging" International Journal of Molecular Sciences 22, no. 12: 6473. https://doi.org/10.3390/ijms22126473
APA StyleRomero-Márquez, J. M., Varela-López, A., Navarro-Hortal, M. D., Badillo-Carrasco, A., Forbes-Hernández, T. Y., Giampieri, F., Domínguez, I., Madrigal, L., Battino, M., & Quiles, J. L. (2021). Molecular Interactions between Dietary Lipids and Bone Tissue during Aging. International Journal of Molecular Sciences, 22(12), 6473. https://doi.org/10.3390/ijms22126473












