Bruton’s Tyrosine Kinase Targeting in Multiple Myeloma
Abstract
1. Introduction
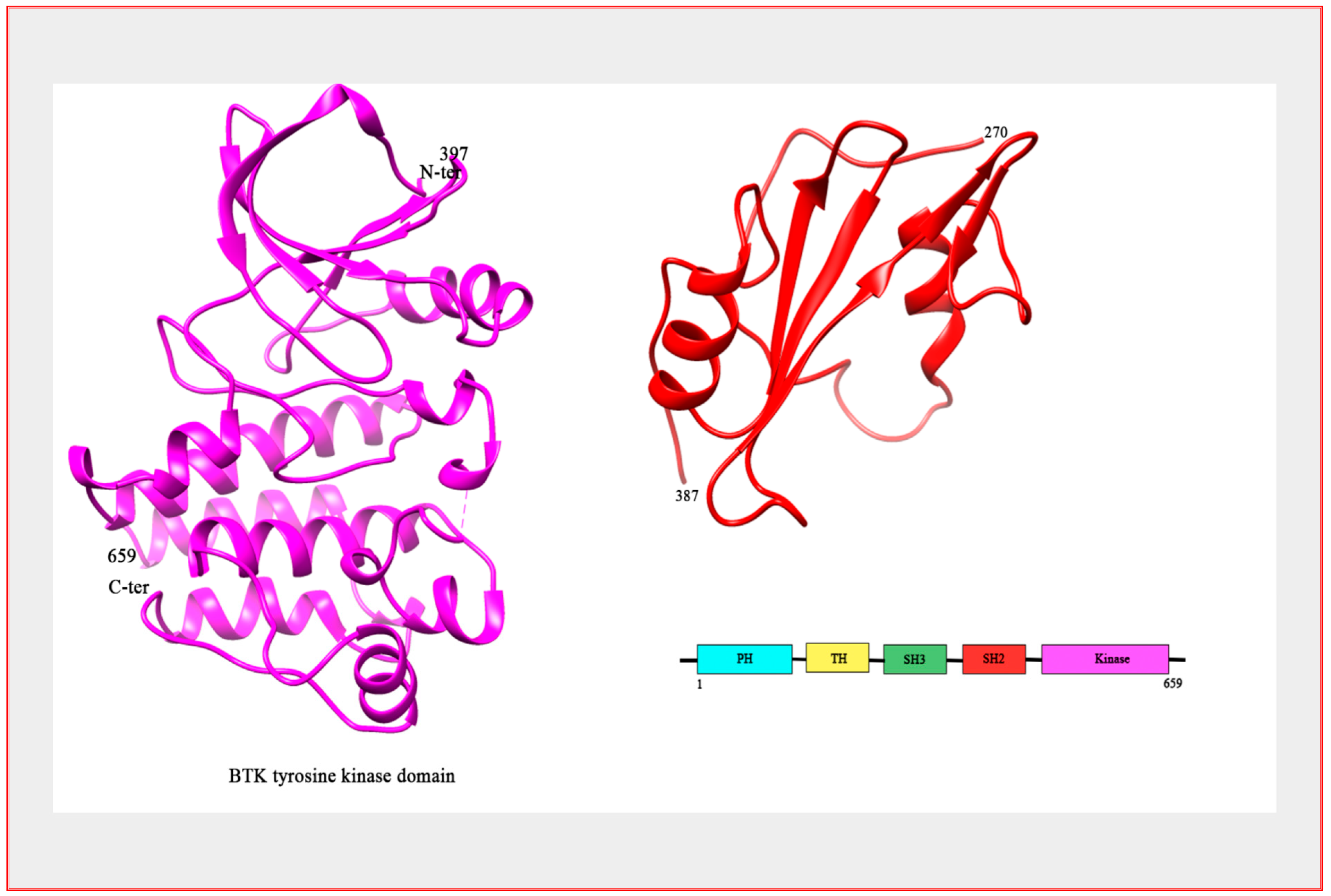
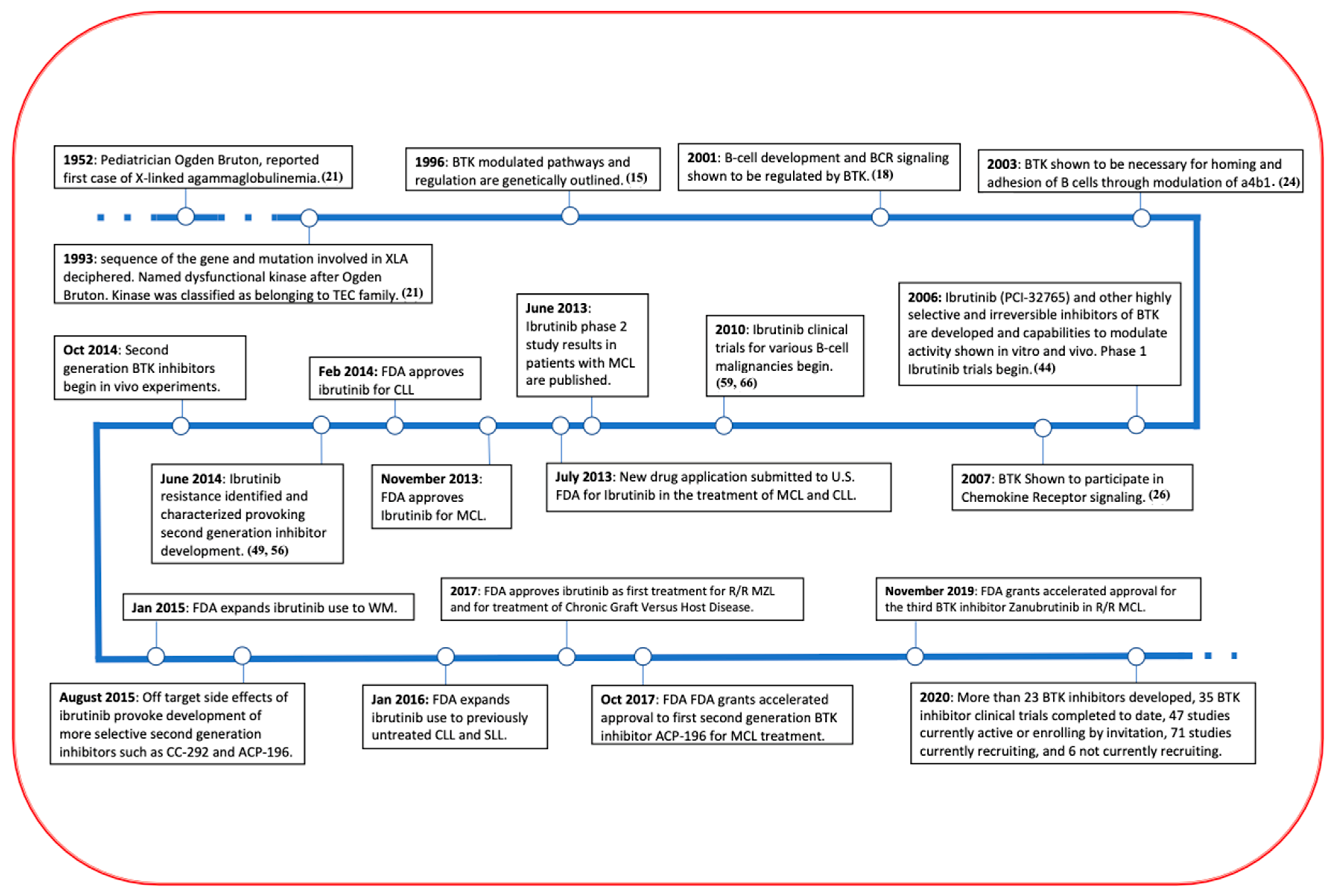
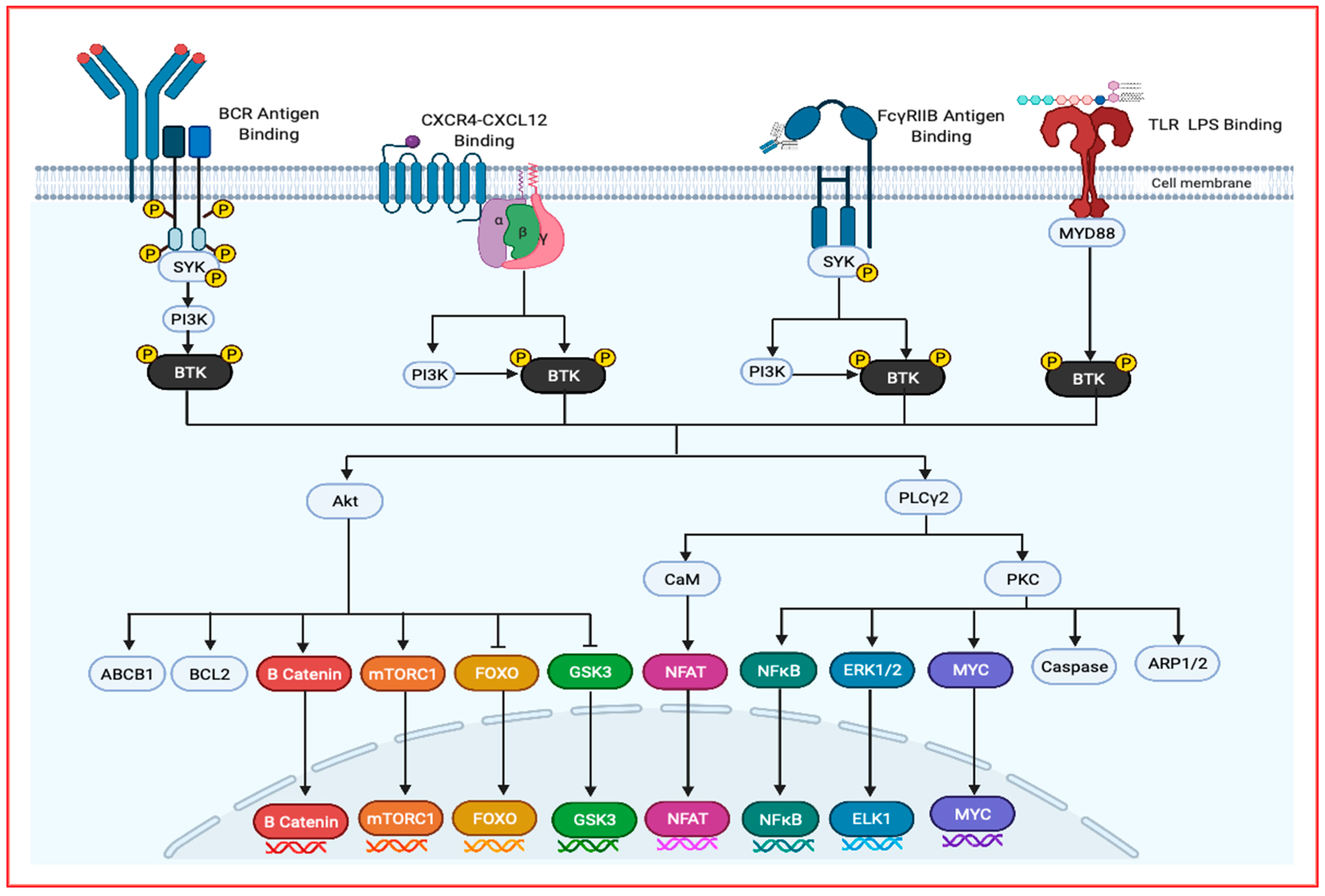
2. Bruton’s Tyrosine Kinase, Structure and Regulation
3. BTK as a Potential Target in Malignancies
4. Role of BTK in Bone Marrow Microenvironment
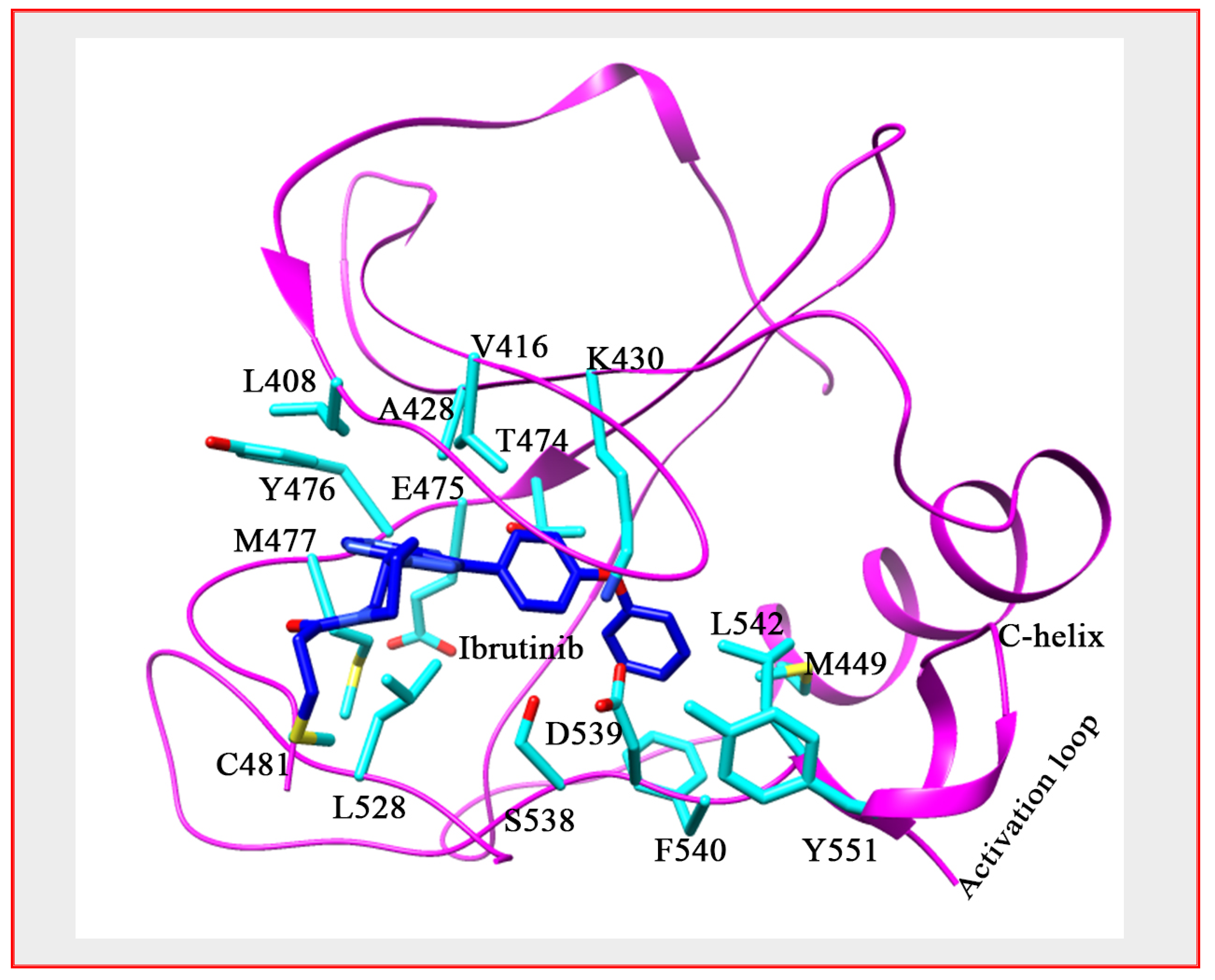
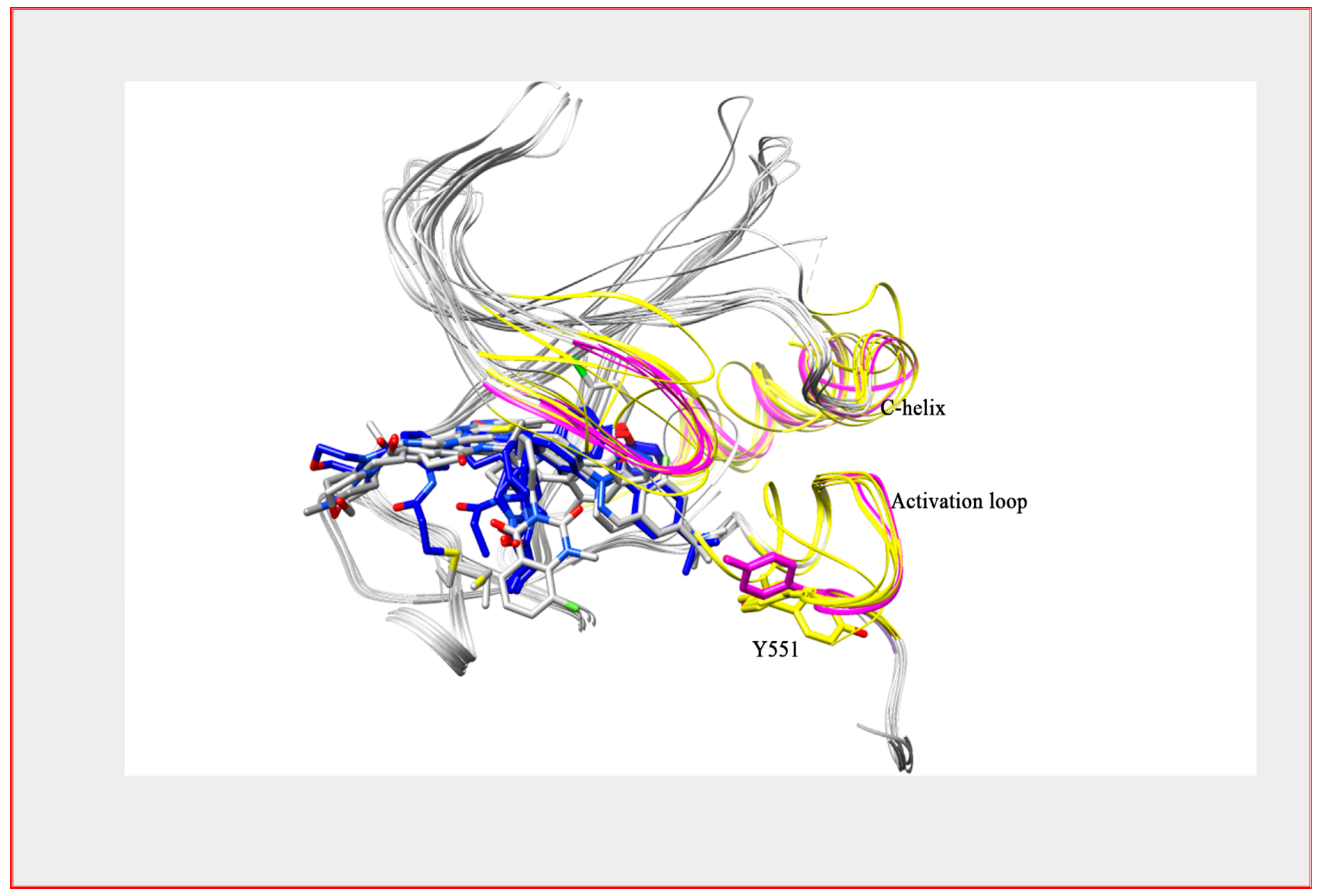
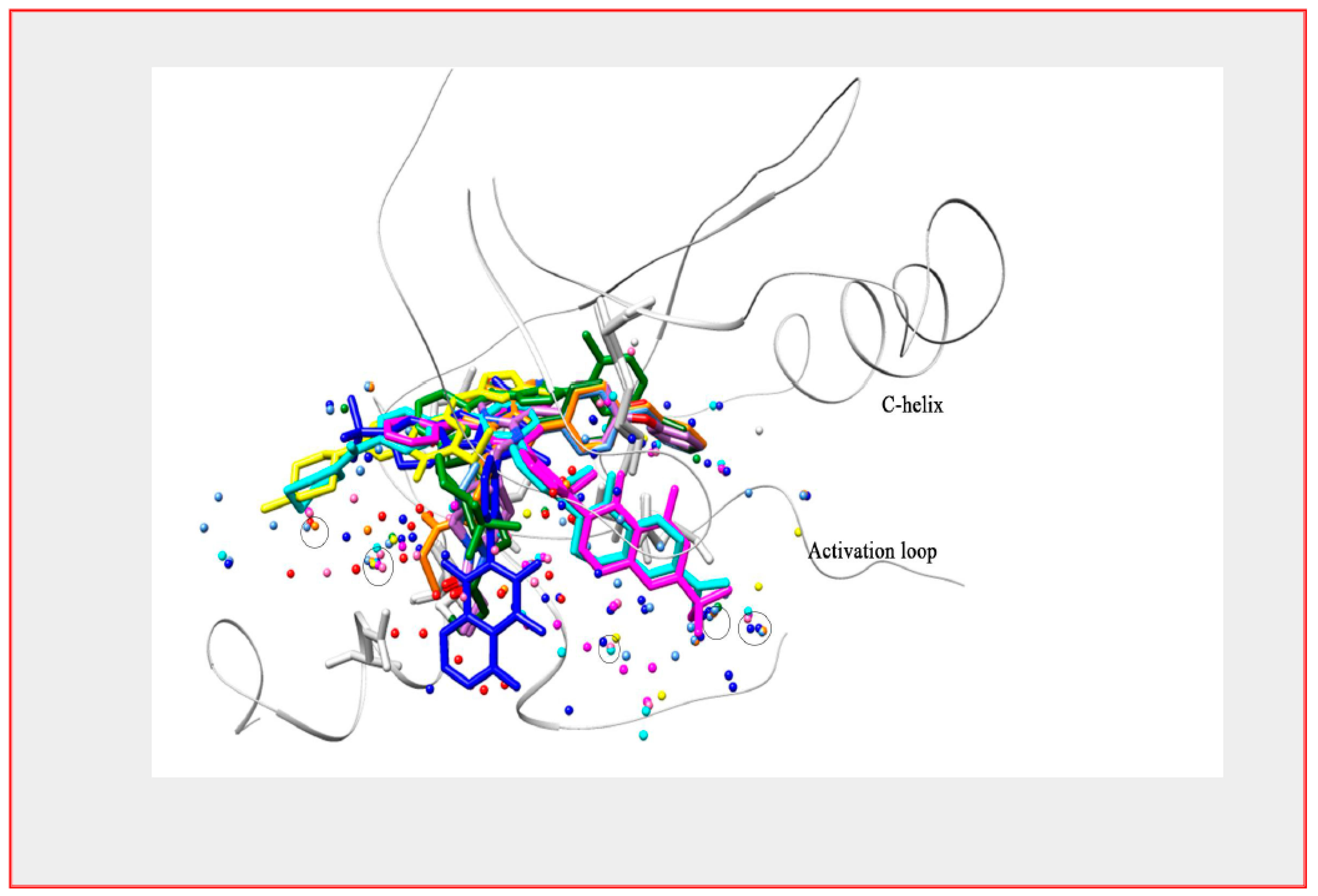
5. Development of BTK Inhibitors
6. How Good a Target Is BTK for Myeloma Therapy?
7. Future Directions
8. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kazandjian, D. Multiple myeloma epidemiology and survival: A unique malignancy. Semin. Oncol. 2016, 43, 676–681. [Google Scholar] [CrossRef]
- Pinto, V.; Bergantim, R.; Caires, H.R.; Seca, H.; Guimarães, J.E.; Vasconcelos, M.H. Multiple Myeloma: Available Therapies and Causes of Drug Resistance. Cancers 2020, 12, 407. [Google Scholar] [CrossRef] [PubMed]
- Moehler, T.; Goldschmidt, H. Multiple Myeloma, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Worsley, C.M.; Mayne, E.S.; Veale, R.B. Clone wars: The evolution of therapeutic resistance in cancer. Evol. Med. Public Health 2016, 2016, 180–181. [Google Scholar] [CrossRef][Green Version]
- Palumbo, A.; Anderson, K. Multiple Myeloma. N. Engl. J. Med. 2011, 364, 1046–1060. [Google Scholar] [CrossRef] [PubMed]
- Solimando, G.A.; Da Vià, C.M.; Cicco, S.; Leone, P.; Di Lernia, G.; Desantis, V.; Frassanito, A.M.; Morizio, A.; Delgado Tascon, J.; Melaccio, A.; et al. High-Risk Multiple Myeloma: Integrated Clinical and Omics Approach Dissects the Neoplastic Clone and the Tumor Microenvironment. J. Clin. Med. 2019, 8, 997. [Google Scholar] [CrossRef] [PubMed]
- Pal Singh, S.; Dammeijer, F.; Hendriks, R.W. Role of Bruton’s tyrosine kinase in B cells and malignancies. Mol. Cancer 2018, 17, 57. [Google Scholar] [CrossRef] [PubMed]
- Amatya, N.; Lin, D.Y.-w.; Andreotti, A.H. Dynamic regulatory features of the protein tyrosine kinases. Biochem. Soc. Trans. 2019, 47, 1101–1116. [Google Scholar] [CrossRef]
- Mano, H. Tec family of protein-tyrosine kinases: An overview of their structure and function. Cytokine Growth Factor Rev. 1999, 10, 267–280. [Google Scholar] [CrossRef]
- Šedivá, A.; Smith, C.I.E.; Asplund, A.C.; Hadač, J.; Zeman, J.; Hansíková, H.; Dvořáková, L.; Mrázová, L.; Velbri, S.; Koehler, C.; et al. Contiguous X-chromosome Deletion Syndrome Encompassing the BTK, TIMM8A, TAF7L, and DRP2 Genes. J. Clin. Immunol. 2007, 27, 640–646. [Google Scholar] [CrossRef]
- Mohamed, A.J.; Yu, L.; Bäckesjö, C.M.; Vargas, L.; Faryal, R.; Aints, A.; Christensson, B.; Berglöf, A.; Vihinen, M.; Nore, B.F.; et al. Bruton’s tyrosine kinase (Btk): Function, regulation, and transformation with special emphasis on the PH domain. Immunol. Rev. 2009, 228, 58–73. [Google Scholar] [CrossRef] [PubMed]
- Rawlings, D.J.; Scharenberg, A.M.; Park, H.; Wahl, M.I.; Lin, S.; Kato, R.M.; Fluckiger, A.C.; Witte, O.N.; Kinet, J.P. Activation of BTK by a Phosphorylation Mechanism Initiated by SRC Family Kinases. Science 1996, 271, 822–825. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Wahl, M.I.; Afar, D.E.H.; Turck, C.W.; Rawlings, D.J.; Tam, C.; Scharenberg, A.M.; Kinet, J.P.; Witte, O.N. Regulation of Btk Function by a Major Autophosphorylation Site within the SH3 Domain. Immunity 1996, 4, 515–525. [Google Scholar] [CrossRef]
- Wang, Q.; Pechersky, Y.; Sagawa, S.; Pan, A.C.; Shaw, D.E. Structural mechanism for Bruton’s tyrosine kinase activation at the cell membrane. Proc. Natl. Acad. Sci. USA 2019, 116, 9390–9399. [Google Scholar] [CrossRef] [PubMed]
- Afar, D.E.; Park, H.; Howell, B.W.; Rawlings, D.J.; Cooper, J.; Witte, O.N. Regulation of Btk by Src family tyrosine kinases. Mol. Cell. Biol. 1996, 16, 3465–3471. [Google Scholar] [CrossRef][Green Version]
- Campbell, R.; Chong, G.; Hawkes, E.A. Novel Indications for Bruton’s Tyrosine Kinase Inhibitors, beyond Hematological Malignancies. J. Clin. Med. 2018, 7, 62. [Google Scholar] [CrossRef]
- Gross, S.; Rahal, R.; Stransky, N.; Lengauer, C.; Hoeflich, K.P. Targeting cancer with kinase inhibitors. J. Clin. Investig. 2015, 125, 1780–1789. [Google Scholar] [CrossRef]
- Rip, J.; de Bruijn, M.J.W.; Appelman, M.K.; Pal Singh, S.; Hendriks, R.W.; Corneth, O.B.J. Toll-Like Receptor Signaling Drives Btk-Mediated Autoimmune Disease. Front. Immunol. 2019, 10, 95. [Google Scholar] [CrossRef]
- Khan, W.N. Regulation of B lymphocyte development and activation by Bruton’s tyrosine kinase. Immunol. Res. 2001, 23, 147–156. [Google Scholar] [CrossRef]
- Suri, D.; Rawat, A.; Singh, S. X-linked Agammaglobulinemia. Indian, J. Pediatrics 2016, 83, 331–337. [Google Scholar] [CrossRef]
- Ponader, S.; Burger, J.A. Bruton’s tyrosine kinase: From X-linked agammaglobulinemia toward targeted therapy for B-cell malignancies. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2014, 32, 1830–1839. [Google Scholar] [CrossRef]
- Petty, J.M.; Lenox, C.C.; Weiss, D.J.; Poynter, M.E.; Suratt, B.T. Crosstalk between CXCR4/stromal derived factor-1 and VLA-4/VCAM-1 pathways regulates neutrophil retention in the bone marrow. J. Immunol. 2009, 182, 604–612. [Google Scholar] [CrossRef]
- Weber, M.; Treanor, B.; Depoil, D.; Shinohara, H.; Harwood, N.E.; Hikida, M.; Kurosaki, T.; Batista, F.D. Phospholipase C-gamma2 and Vav cooperate within signaling microclusters to propagate B cell spreading in response to membrane-bound antigen. J. Exp. Med. 2008, 205, 853–868. [Google Scholar] [CrossRef]
- Spaargaren, M.; Beuling, E.A.; Rurup, M.L.; Meijer, H.P.; Klok, M.D.; Middendorp, S.; Hendriks, R.W.; Pals, S.T. The B Cell Antigen Receptor Controls Integrin Activity through Btk and PLCγ2. J. Exp. Med. 2003, 198, 1539–1550. [Google Scholar] [CrossRef]
- Chatterjee, S.; Azad, B.B.; Nimmagadda, S. The intricate role of CXCR4 in cancer. Adv. Cancer Res. 2014, 124, 31–82. [Google Scholar] [PubMed]
- de Gorter, D.J.; Beuling, E.A.; Kersseboom, R.; Middendorp, S.; van Gils, J.M.; Hendriks, R.W.; Pals, S.T.; Spaargaren, M. Bruton’s tyrosine kinase and phospholipase Cgamma2 mediate chemokine-controlled B cell migration and homing. Immunity 2007, 26, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-S.; Chang, B.Y.; Chang, S.; Tong, T.; Zou, Y.R.; Sherry, B.; Buggy, J.J.; Burger, J.A.; Rai, K.R.; Chiorazzi, N. In Vivo Evidence That Ibrutininb Deregulates Chemokine Receptor CXCR4 Surface Membrane Expression and Signaling, Along with Inhibiting B Cell Antigen Receptor Signaling, As Causes for Defective Homing and Impaired Retention of CLL Cells in Tissues. Blood 2014, 124, 1948. [Google Scholar] [CrossRef]
- Bence, K.; Ma, W.; Kozasa, T.; Huang, X.-Y. Direct stimulation of Bruton’s tyrosine kinase by Gq-protein α-subunit. Nature 1997, 389, 296–299. [Google Scholar] [CrossRef] [PubMed]
- Botos, I.; Segal, D.M.; Davies, D.R. The Structural Biology of Toll-like Receptors. Structure 2011, 19, 447–459. [Google Scholar] [CrossRef]
- Ping, L.; Ding, N.; Shi, Y.; Feng, L.; Li, J.; Liu, Y.; Lin, Y.; Shi, C.; Wang, X.; Pan, Z.; et al. The Bruton’s tyrosine kinase inhibitor ibrutinib exerts immunomodulatory effects through regulation of tumor-infiltrating macrophages. Oncotarget 2017, 8, 39218–39229. [Google Scholar] [CrossRef] [PubMed]
- Kenny, E.F.; Quinn, S.R.; Doyle, S.L.; Vink, P.M.; van Eenennaam, H.; O’Neill, L.A.J. Bruton’s Tyrosine Kinase Mediates the Synergistic Signalling between TLR9 and the B Cell Receptor by Regulating Calcium and Calmodulin. PLoS ONE 2013, 8, e74103. [Google Scholar] [CrossRef]
- Nimmerjahn, F.; Ravetch, J.V. Fcγ receptors as regulators of immune responses. Nat. Rev. Immunol. 2008, 8, 34–47. [Google Scholar] [CrossRef]
- Burger, J.A. Nurture versus Nature: The Microenvironment in Chronic Lymphocytic Leukemia. Hematology 2011, 2011, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Lemaire, M.; Deleu, S.; De Bruyne, E.; Van Valckenborgh, E.; Menu, E.; Vanderkerken, K. The Microenvironment and Molecular Biology of the Multiple Myeloma Tumor. In Advances in Cancer Research; Klein, G., Ed.; Academic Press: Cambridge, MA, USA, 2011; pp. 19–42. [Google Scholar]
- Kawano, Y.; Moschetta, M.; Manier, S.; Glavey, S.; Görgün, G.T.; Roccaro, A.M.; Anderson, K.C.; Ghobrial, I.M. Targeting the bone marrow microenvironment in multiple myeloma. Immunol. Rev. 2015, 263, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Dehghanifard, A.; Kaviani, S.; Abroun, S.; Mehdizadeh, M.; Saiedi, S.; Maali, A.; Ghaffari, S.; Azad, M. Various Signaling Pathways in Multiple Myeloma Cells and Effects of Treatment on These Pathways. Clin. Lymphomamyeloma Leuk. 2018, 18, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Fruman, D.A.; Chiu, H.; Hopkins, B.D.; Bagrodia, S.; Cantley, L.C.; Abraham, R.T. The PI3K Pathway in Human Disease. Cell 2017, 170, 605–635. [Google Scholar] [CrossRef]
- Eda, H.; Santo, L.; Cirstea, D.D.; Yee, A.J.; Scullen, T.A.; Nemani, N.; Mishima, Y.; Waterman, P.R.; Arastu-Kapur, S.; Evans, E.; et al. A novel Bruton’s tyrosine kinase inhibitor CC-292 in combination with the proteasome inhibitor carfilzomib impacts the bone microenvironment in a multiple myeloma model with resultant antimyeloma activity. Leukemia 2014, 28, 1892–1901. [Google Scholar] [CrossRef] [PubMed]
- Tai, Y.A.K. Bruton’s tyrosine kinase: Oncotarget in myeloma. Oncotarget 2012, 3, 913–914. [Google Scholar] [CrossRef]
- Weber, A.; Bittner, Z.; Liu, X.; Dang, T.M.; Radsak, M.P.; Brunner, C. Bruton’s Tyrosine Kinase: An Emerging Key Player in Innate Imunity. Front. Immunol. 2017, 8, 1454. [Google Scholar] [CrossRef]
- Good, L.; Benner, B.; Carson, W.E. Bruton’s tyorinse kinase: An emerging targeted therapy in myeloid cells within the tumor microenvironment. Cancer Immunol. Immunother 2021, 1–13. [Google Scholar]
- Bam, R.; Ling, W.; Khan, S.; Pennisi, A.; Venkateshaiah, S.U.; Li, X.; van Rhee, F.; Usmani, S.; Barlogie, B.; Shaughnessy, J.; et al. Role of Bruton’s tyrosine kinase in myeloma cell migration and induction of bone disease. Am. J. Hematol. 2013, 88, 463–471. [Google Scholar] [CrossRef]
- Yang, Y.; Gu, Z.; Xu, H.; Huang, J.; He, J.; Tao, Y.; Tricot, G.J.; Zhan, F. Targeting BTK As a Treatment For Multiple Myeloma Stem Cells. Blood 2013, 122, 271. [Google Scholar] [CrossRef]
- Yang, Y.; Shi, J.; Gu, Z.; Salama, M.E.; Das, S.; Wendlandt, E.; Xu, H.; Huang, J.; Tao, Y.; Hao, M.; et al. Bruton Tyrosine Kinase Is a Therapeutic Target in Stem-like Cells from Multiple Myeloma. Cancerresearch 2015, 75, 594–604. [Google Scholar] [CrossRef]
- Pan, Z.; Scheerens, H.; Li, S.-J.; Schultz, B.E.; Sprengeler, P.A.; Burrill, L.C.; Mendonca, R.V.; Sweeney, M.D.; Scott, K.C.K.; Grothaus, P.G.; et al. Discovery of Selective Irreversible Inhibitors for Bruton’s Tyrosine Kinase. ChemMedChem 2007, 2, 58–61. [Google Scholar] [CrossRef]
- Richardson, P.G.; Bensinger, W.I.; Huff, C.A.; Costello, C.L.; Lendvai, N.; Berdeja, J.G.; Anderson, L.D.; Siegel, D.S.; Lebovic, D.; Jagannath, S.; et al. Ibrutinib alone or with dexamethasone for relapsed or relapsed and refractory multiple myeloma: Phase 2 trial results. Br. J. Haematol. 2018, 180, 821–830. [Google Scholar] [CrossRef] [PubMed]
- Bender, A.T.; Gardberg, A.; Pereira, A.; Johnson, T.; Wu, Y.; Grenningloh, R.; Head, J.; Morandi, F.; Haselmayer, P.; Liu-Bujalski, L. Ability of Bruton’s Tyrosine Kinase Inhibitors to Sequester Y551 and Prevent Phosphorylation Determines Potency for Inhibition of Fc Receptor but not B-Cell Receptor Signaling. Mol. Pharmacol. 2017, 91, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Barf, T.; Covey, T.; Izumi, R.; van de Kar, B.; Gulrajani, M.; van Lith, B.; van Hoek, M.; de Zwart, E.; Mittag, D.; Demont, D.; et al. Acalabrutinib (ACP-196): A Covalent Bruton Tyrosine Kinase Inhibitor with a Differentiated Selectivity and In Vivo Potency Profile. J. Pharmacol. Exp. Ther. 2017, 363, 240. [Google Scholar] [CrossRef]
- Wu, J.; Zhang, M.; Liu, D. Acalabrutinib (ACP-196): A selective second-generation BTK inhibitor. J. Hematol. Oncol. 2016, 9, 21. [Google Scholar] [CrossRef]
- Vidal-Crespo, A.; Rodríguez, V.; Matas-Céspedes, A.; Campos, E.; López-Guillermo, A.; Roué, G.; Colomer, D.; Pérez-Galán, P. Abstract 1757: The novel BTK inhibitor CC-292 exerts in vitro and in vivo antitumor activity, interferes with adhesion, cell migration, and synergizes with lenalidomide in MCL models. Cancer Res. 2014, 74 (Suppl. 19), 1757. [Google Scholar]
- Jain, M.; Chatterjee, A.; Mohapatra, J.; Bandhyopadhyay, D.; Ghoshdostidar, K.; Bhatnagar, U.; Patel, H.; Bahekar, R.; Ramanathan, V.; Patel, P.; et al. 326 A novel Bruton’s tyrosine kinase (BTK) inhibitor with anticancer and anti-inflammatory activities. Eur. J. Cancer 2015, 51, S63. [Google Scholar] [CrossRef]
- De Cesco, S.; Kurian, J.; Dufresne, C.; Mittermaier, A.K.; Moitessier, N. Covalent inhibitors design and discovery. Eur. J. Med. Chem. 2017, 138, 96–114. [Google Scholar] [CrossRef]
- Molina-Cerrillo, J.; Alonso-Gordoa, T.; Gajate, P.; Grande, E. Bruton’s tyrosine kinase (BTK) as a promising target in solid tumors. Cancer Treat. Rev. 2017, 58, 41–50. [Google Scholar] [CrossRef]
- Li, N.; Sun, Z.; Liu, Y.; Guo, M.; Zhang, Y.; Zhou, D.; Zhang, B.; Su, D.; Zhang, S.; Han, J.; et al. Abstract 2597: BGB-3111 is a novel and highly selective Bruton’s tyrosine kinase (BTK) inhibitor. Cancer Res. 2015, 75, 2597. [Google Scholar]
- Munakata, W.; Ando, K.; Hatake, K.; Fukuhara, N.; Kinoshita, T.; Fukuhara, S.; Shirasugi, Y.; Yokoyama, M.; Ichikawa, S.; Ohmachi, K.; et al. Phase I study of tirabrutinib (ONO-4059/GS-4059) in patients with relapsed or refractory B-cell malignancies in Japan. Cancer Sci. 2019, 110, 1686–1694. [Google Scholar] [CrossRef]
- Rule, S.; Chen, R.W. New and emerging Bruton tyrosine kinase inhibitors for treating mantle cell lymphoma—where do they fit in? Expert Rev. Hematol. 2018, 11, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.P.S.; McMullen, J.; Tam, C. Cardiac side effects of bruton tyrosine kinase (BTK) inhibitors. Leukemia & lymphoma 2018, 59, 1554–1564. [Google Scholar]
- de Weerdt, I.; Koopmans, S.M.; Kater, A.P.; van Gelder, M. Incidence and management of toxicity associated with ibrutinib and idelalisib: A practical approach. Haematologica 2017, 102, 1629–1639. [Google Scholar] [CrossRef]
- Byrd, J.C.; Furman, R.R.; Coutre, S.E.; Flinn, I.W.; Burger, J.A.; Blum, K.A.; Grant, B.; Sharman, J.P.; Coleman, M.; Wierda, W.G.; et al. Targeting BTK with ibrutinib in relapsed chronic lymphocytic leukemia. N. Engl. J. Med. 2013, 369, 32–42. [Google Scholar] [CrossRef]
- Liang, C.; Tian, D.; Ren, X.; Ding, S.; Jia, M.; Xin, M.; Thareja, S. The development of Bruton’s tyrosine kinase (BTK) inhibitors from 2012 to 2017: A mini-review. Eur. J. Med. Chem. 2018, 151, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Akinleye, A.; Chen, Y.; Mukhi, N.; Song, Y.; Liu, D. Ibrutinib and novel BTK inhibitors in clinical development. J. Hematol. Oncol. 2013, 6, 59. [Google Scholar] [CrossRef]
- Woyach, J.A.; Furman, R.R.; Liu, T.M.; Ozer, H.G.; Zapatka, M.; Ruppert, A.S.; Xue, L.; Li, D.H.; Steggerda, S.M.; Versele, M.; et al. Resistance Mechanisms for the Bruton’s Tyrosine Kinase Inhibitor Ibrutinib. N. Engl. J. Med. 2014, 370, 2286–2294. [Google Scholar] [CrossRef]
- Sedlarikova, L.; Petrackova, A.; Papajik, T.; Turcsanyi, P.; Kriegova, E. Resistance-Associated Mutations in Chronic Lymphocytic Leukemia Patients Treated With Novel Agents. Front. Oncol. 2020, 10, 894. [Google Scholar] [CrossRef]
- Sharma, S.; Galanina, N.; Guo, A.; Lee, J.; Kadri, S.; Van Slambrouck, C.; Long, B.; Wang, W.; Ming, M.; Furtado, L.V.; et al. Identification of a structurally novel BTK mutation that drives ibrutinib resistance in CLL. Oncotarget 2016, 7, 68833–68841. [Google Scholar] [CrossRef]
- Edwards, C.M. BTK inhibition in myeloma: Targeting the seed and the soil. Blood 2012, 120, 1757–1759. [Google Scholar] [CrossRef]
- Murray, M.Y.; Zaitseva, L.; Auger, M.J.; Craig, J.I.O.; MacEwan, D.J.; Rushworth, S.A.; Bowles, K.M. Ibrutinib inhibits BTK-driven NF-κB p65 activity to overcome bortezomib-resistance in multiple myeloma. Cell Cycle 2015, 14, 2367–2375. [Google Scholar] [CrossRef] [PubMed]
- Hajek, R.; Pour, L.; Ozcan, M.; Martin Sánchez, J.; García Sanz, R.; Anagnostopoulos, A.; Oriol, A.; Cascavilla, N.; Terjung, A.; Lee, Y.; et al. A phase 2 study of ibrutinib in combination with bortezomib and dexamethasone in patients with relapsed/refractory multiple myeloma. Eur. J. Haematol. 2020, 104, 435–442. [Google Scholar] [CrossRef]
- Chari, A.; Larson, S.; Holkova, B.; Cornell, R.F.; Gasparetto, C.; Karanes, C.; Matous, J.V.; Niesvizky, R.; Valent, J.; Lunning, M.; et al. Phase 1 trial of ibrutinib and carfilzomib combination therapy for relapsed or relapsed and refractory multiple myeloma. Leuk. Lymphoma 2018, 59, 2588–2594. [Google Scholar] [CrossRef] [PubMed]
- Abramson, H.N. The Multiple Myeloma Drug Pipeline—2018: A Review of Small Molecules and Their Therapeutic Targets. Clin. Lymphoma Myeloma Leuk. 2018, 18, 611–627. [Google Scholar] [CrossRef]
- Kyle, R.A.; Rajkumar, S.V. Treatment of Multiple Myeloma: A Comprehensive Review. Clin. Lymphoma Myeloma 2009, 9, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Advani, R.H.; Buggy, J.J.; Sharman, J.P.; Smith, S.M.; Boyd, T.E.; Grant, B.; Kolibaba, K.S.; Furman, R.R.; Rodriguez, S.; Chang, B.Y.; et al. Bruton tyrosine kinase inhibitor ibrutinib (PCI-32765) has significant activity in patients with relapsed/refractory B-cell malignancies. J. Clin. Oncol. 2013, 31, 88–94. [Google Scholar] [CrossRef]
- Uckun, F.M.; Qazi, S.; Demirer, T.; Champlin, R.E. Contemporary patient-tailored treatment strategies against high risk and relapsed or refractory multiple myeloma. EBioMedicine 2019, 39, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Hideshima, T.; Mitsiades, C.; Tonon, G.; Richardson, P.G.; Anderson, K.C. Understanding multiple myeloma pathogenesis in the bone marrow to identify new therapeutic targets. Nat. Rev. Cancer 2007, 7, 585–598. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Von Suskil, M.; Sultana, K.N.; Elbezanti, W.O.; Al-Odat, O.S.; Chitren, R.; Tiwari, A.K.; Challagundla, K.B.; Srivastava, S.K.; Jonnalagadda, S.C.; Budak-Alpdogan, T.; et al. Bruton’s Tyrosine Kinase Targeting in Multiple Myeloma. Int. J. Mol. Sci. 2021, 22, 5707. https://doi.org/10.3390/ijms22115707
Von Suskil M, Sultana KN, Elbezanti WO, Al-Odat OS, Chitren R, Tiwari AK, Challagundla KB, Srivastava SK, Jonnalagadda SC, Budak-Alpdogan T, et al. Bruton’s Tyrosine Kinase Targeting in Multiple Myeloma. International Journal of Molecular Sciences. 2021; 22(11):5707. https://doi.org/10.3390/ijms22115707
Chicago/Turabian StyleVon Suskil, Max, Kazi Nasrin Sultana, Weam Othman Elbezanti, Omar S. Al-Odat, Robert Chitren, Amit K. Tiwari, Kishore B. Challagundla, Sandeep Kumar Srivastava, Subash C. Jonnalagadda, Tulin Budak-Alpdogan, and et al. 2021. "Bruton’s Tyrosine Kinase Targeting in Multiple Myeloma" International Journal of Molecular Sciences 22, no. 11: 5707. https://doi.org/10.3390/ijms22115707
APA StyleVon Suskil, M., Sultana, K. N., Elbezanti, W. O., Al-Odat, O. S., Chitren, R., Tiwari, A. K., Challagundla, K. B., Srivastava, S. K., Jonnalagadda, S. C., Budak-Alpdogan, T., & Pandey, M. K. (2021). Bruton’s Tyrosine Kinase Targeting in Multiple Myeloma. International Journal of Molecular Sciences, 22(11), 5707. https://doi.org/10.3390/ijms22115707






