Biochemistry of Terpenes and Recent Advances in Plant Protection
Abstract
1. Introduction
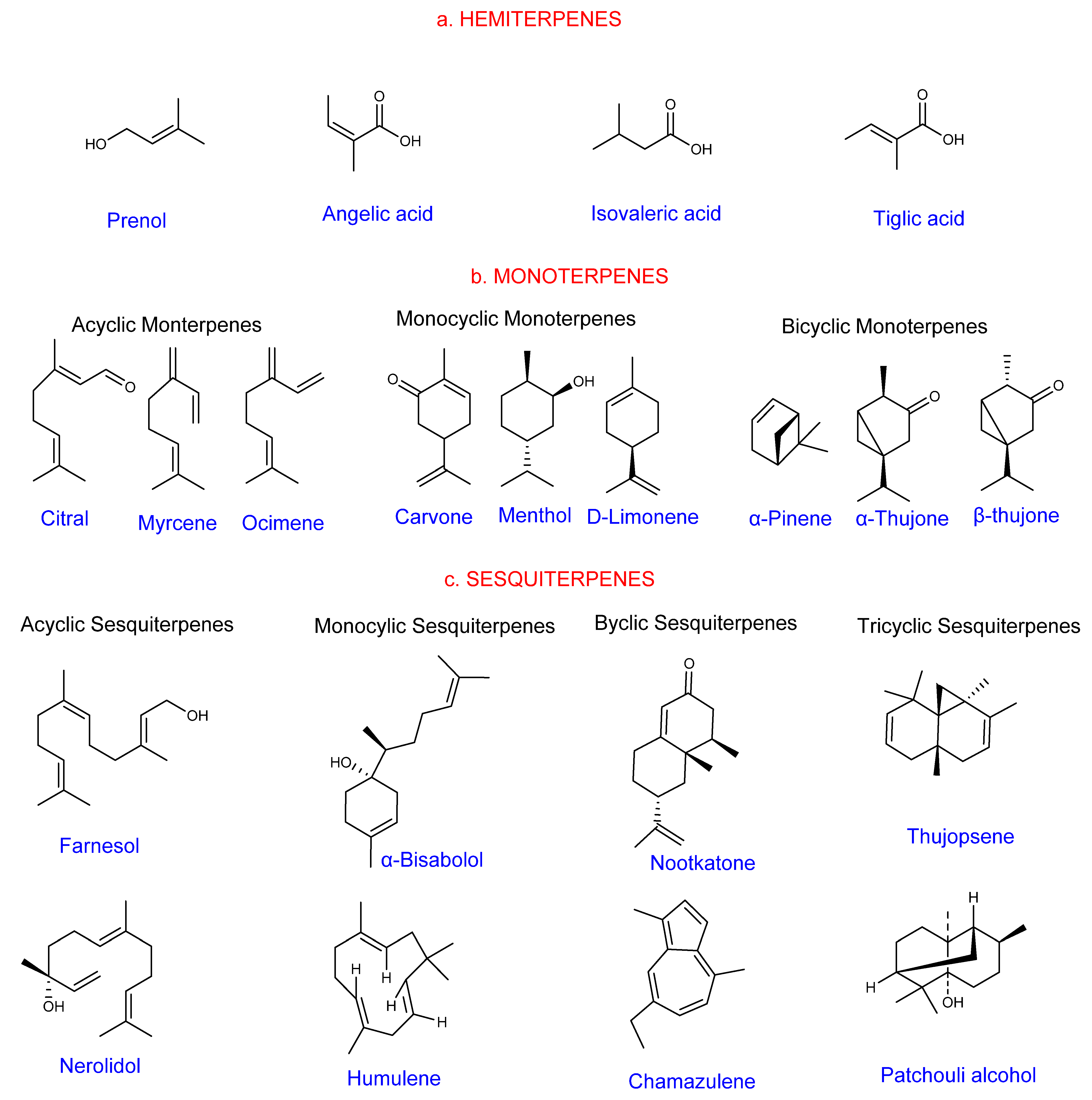
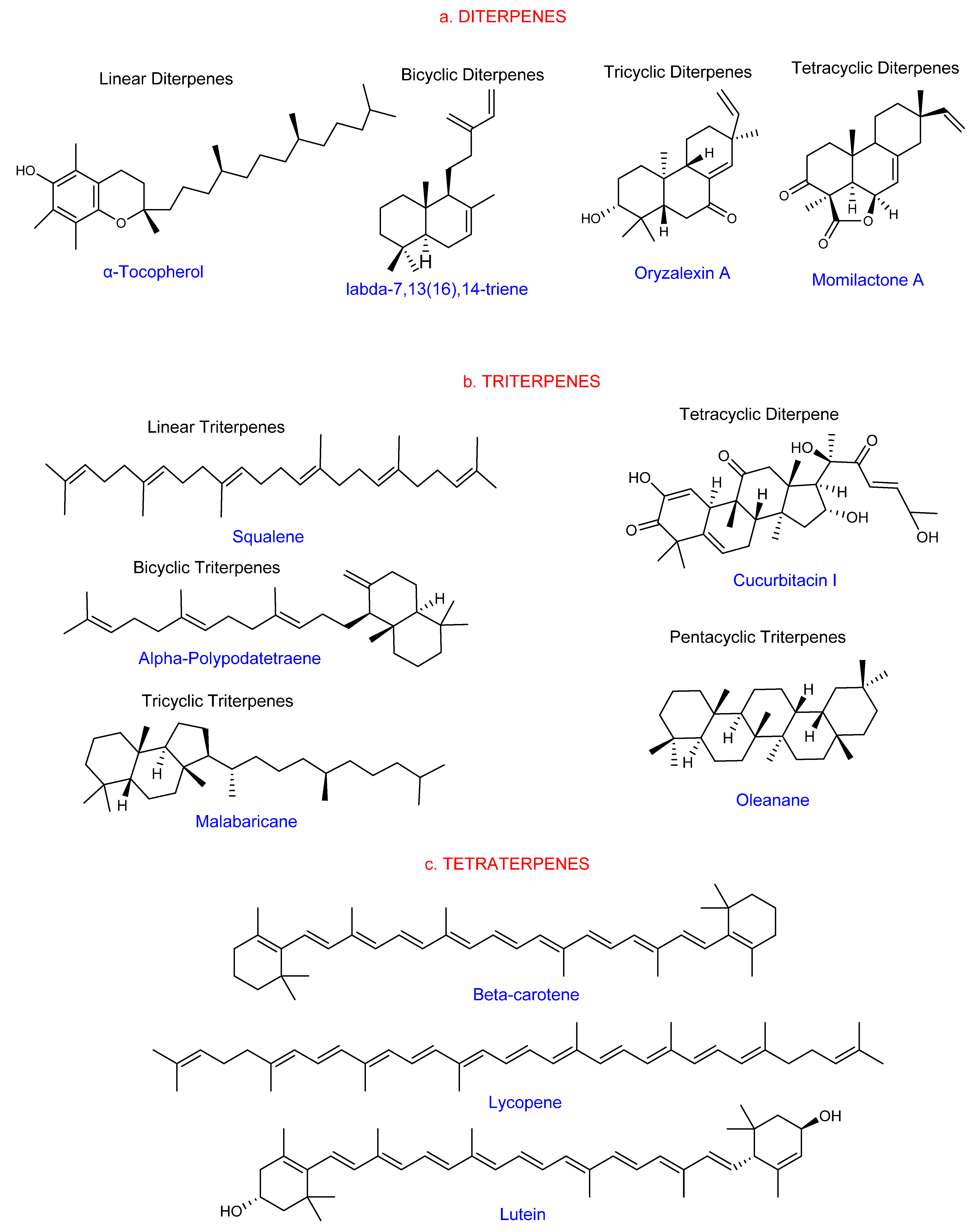
1.1. General Overview of Terpenes
1.2. Classification of Terpenes
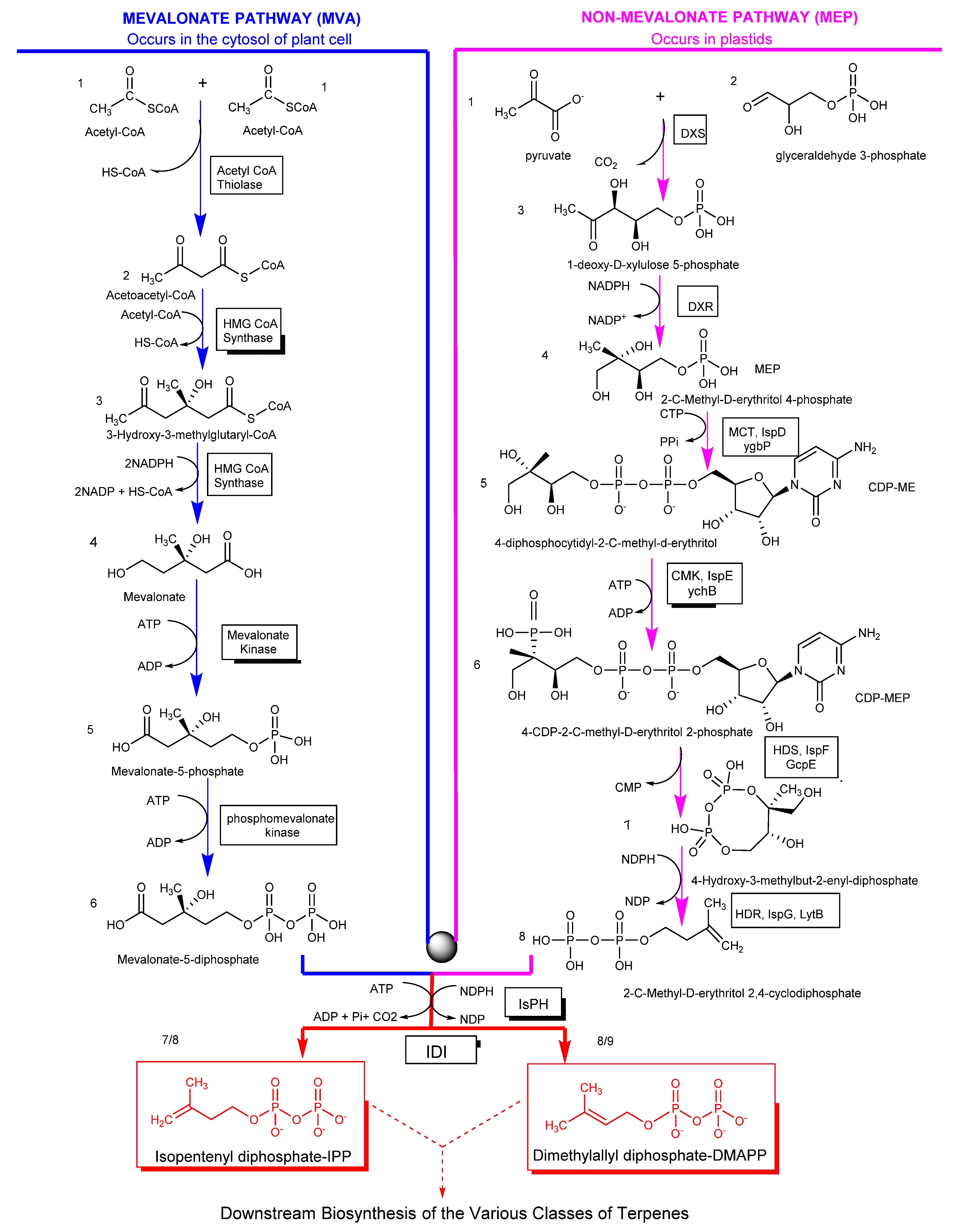
2. Biosynthesis of Terpenes
2.1. Terpene Synthases (TPSs), Classes, and Terpenes Encoded
2.2. Elicitor-Induced Terpenes Biosynthesis
3. Role of Terpenes in Plant Protection
3.1. Role of Terpenes in Insect Modulation
3.2. Antimicrobial (Phytoalexin) Activities of Terpenes
3.3. Allelopathic Activities of Terpenes in Agriculture
| Name of Terpene | Plant | Activity | Reference |
|---|---|---|---|
| Eucalyptol | Oak | Attracts cockchafer larva | [121] |
| Rhizathalene A | Arabidopsis | Resistance of the roots to the herbivore dark-winged fungus gnat (Bradysia spp) | [122] |
| Eugenol, caryophyllene oxide, α-pinene, α-humulene, and α-phellandrene | Cinnamon and clove | Secret toxic terpenes to deter the adult pest of Sitophilus granaries (grain weevil) | [123] |
| β-Pinene | Citrus paradisix, Poncirus trifoliata | Attracts entomopathogenic nematodes, e.g., Steinernema diaprepesi | [124] |
| S-methyl methionine | Lecythidaceae | Deters oviposition sites-seeking beetle | [125] |
| 1,8-cineole | Brassica Tropical orchids | Attracts egg-laying parasitoids to the caterpillars of feeding herbivores. Furthermore, attracts and reward pollinators | [126] |
| β-trans-ocimene, (+)-R-limonene | Lavender | Deters pests, e.g., aphids | [127] |
| β-costic acid | Zea mays | Inhibits the growth of Diabrotica balteata (cucumber beetle) | [128] |
| β-ocimene | Tomato and tobacco | Defense against pests, e.g., Macrosiphum euphorbiae (potato aphid) | [129] |
| Decanal | Zea mays | Enhanced resistance against Ostrinia nubilalis | [130] |
| Name of Terpene | Plant | Activity | Reference |
|---|---|---|---|
| α-Terpinene Terpinen-4-ol α-Thujene | Oryza sativa | Antibacterial activity on Xoo | [131,132] |
| Linalool | Oryza sativa | Antibacterial activity on Xoo | [133] |
| epoxydolabranol | Zea mays | Defense against F. verticillioides and F. graminearum | [101] |
| Kauralexin A3 and B3 | Zea mays | Antifungal activity against R. microsporus and C. graminicola | [134] |
| 9β-Pimara-7,15-diene-3β,6β,19-triol | Rice leaves | Weak antimicrobial | [99] |
| Marneral | A. thaliana | Pathogenesis activities | [135] |
| Cucurbitadieno | Cucumis sativus | Pathogenesis and insecticidal activities | [136] |
| Oryzalexin A–F | Oryza sativa Leersia perrier | Antimicrobial (Inhibits spore germination and the growth of the germ tube of O. oryzae) | [24,99,136] |
| Phytocassane A–F | Oryza sativa | Antifungal activities M. oryzae and R. Solani | [99] |
| Oryzalexin S | Oryza sativa | Antifungal activity | [137] |
| Momilactone A, momilactone B, | Oryza sativa | Antimicrobial activities | [138] |
| Tirucalla-7, 24-dien-3b-ol | A. thaliana | Pathogenesis-related activities | [139] |
| Name of Terpene | Plant | Activity | Reference |
|---|---|---|---|
| Momilactone A | Oryza sativa, Arabidopsis, alfalfa, lettuce, cress, timothy, barnyard grass, E. colonum, crabgrass | Momilactones are generally not toxic to rice plants but mainly inhibit germination and growth of other seeds, e.g., barnyard grass Enhances plants to outcompete other field crops by roots exudates serving as inhabitants, especially Momilactones A and B | [120,140,141,142] |
| Momilactone B | |||
| Momilactone C | |||
| Momilactone D | |||
| Momilactone E | |||
| Carvone, Betulin | Amaranthus retroflexu, Sinapis arvensis | Exhibits high inhibition against common weeds at a lower concentration | [143] |
| Limonene and (+)-citronellal | Transgenic Arabidopsis thaliana | Exhibits vigorous antimicrotubule activity in transgenic Arabidopsis thaliana | [144] |
| Lanast-7,9(11)-dien-3α, 15α-diol-3α-D-glucofuranoside | Oryza sativa | Growth inhibition | [145] |
4. Other Applications of Terpenes
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PSM | Plants Secondary Metabolite |
| IPP | Isopentenyl diphosphate |
| DMAPP | Dimethylallyl diphosphate |
| MVA | Mevalonate pathway |
| MEP | 2C-methyl-d-erythritol-4-phosphate |
| GPP | Geranyl pyrophosphate |
| FGPP | Farnesyl geranyl pyrophosphate |
| GGPP | Geranylgeranyl pyrophosphate |
| GPPS | Geranyl pyrophosphate synthase |
| FPPS | Farnesyl pyrophosphate synthase |
| TPS | terpenes synthase |
| NGS | Next-Generation Sequencing |
| IPPC | International Plant Protection Convention |
| Xoo | Xanthomonas oryzae pv. oryzae |
| EOs | Essential oils |
| PPAR pathway | Peroxisome proliferator-activated receptor pathway |
| DXP | 1-deoxy-d-xylulose 5-phosphate |
| CDP-ME | 4-diphosphocytidyl-2-C-methyl-d-erythritol |
| CDP-MEP | 4-diphosphocytidyl-2-C-methyl-d-erythritol-2-phosphate |
| MEcPP | 2-C-methyl-d-erythritol-2,4-cyclodiphosphate |
| HMBPP | 4-hydroxy-3-methyl-butenyl 1-diphosphate |
| CTP | cytidine 5’-triphosphate |
| IDI | isopentenyl-pyrophosphate delta isomerase |
| HMG-CoA | 3-Hydroxy-3-methylglutaryl-coenzyme A |
| CTP | Cytidine triphosphate |
References
- Han, G. Origin and evolution of the plant immune system. New Phytol. 2018, 222, 70–83. [Google Scholar] [CrossRef]
- Willis, K.J. State of the World’s Plants Report-2017; Royal Botanic Gardens: Richmond, UK, 2017. [Google Scholar]
- Yousaf, H.K.; Shan, T.; Chen, X.; Ma, K.; Shi, X.; Desneux, N.; Biondi, A.; Gao, X. Impact of the secondary plant metabolite cucurbitacin B on the demographical traits of the melon aphid, Aphis gossypii. Sci. Rep. 2018, 8, 16473. [Google Scholar] [CrossRef]
- Isah, T. Stress and defense responses in plant secondary metabolites production. Biol. Res. 2019, 52, 1–25. [Google Scholar] [CrossRef]
- UDSA. Medicinal Botany: How Long Have People Been Using Medicinal Plants? 2008. Available online: https://www.fs.fed.us/ (accessed on 15 February 2021).
- Kumar, R.; Tewari, A.K. Isolation of medicinally important constituents from rare and exotic medicinal plants. In Synthesis of Medicinal Agents from Plants; Elsevier: Amsterdam, The Netherlands, 2018; pp. 229–256. [Google Scholar]
- Silpa, P.; Roopa, K.; Thomas, T.D. Production of Plant Secondary Metabolites: Current Status and Future Prospects. In Biotechnological Approaches for Medicinal and Aromatic Plants; Springer Science and Business Media LLC: Cham, Switzerland, 2018; pp. 3–25. [Google Scholar]
- Silva, L.N.; Zimmer, K.R.; Macedo, A.J.; Trentin, D.S. Plant Natural products targeting bacterial virulence factors. Chem. Rev. 2016, 116, 9162–9236. [Google Scholar] [CrossRef]
- Fürstenberg-Hägg, J.; Zagrobelny, M.; Bak, S. Plant Defense against Insect Herbivores. Int. J. Mol. Sci. 2013, 14, 10242–10297. [Google Scholar] [CrossRef]
- Gao, F.; Zhu, S.-R.; Sun, Y.-C.; Du, L.; Parajulee, M.; Kang, L.; Ge, F. Interactive effects of elevated CO2 and cotton cultivar on tri-trophic interaction of Gossypium hirsutum, Aphis gossyppii, and Propylaea japonica. Environ. Entomol. 2014, 37, 29–37. [Google Scholar] [CrossRef]
- Ashour, M.; Wink, M.; Gershenzon, J.; Wink, M. Biochemistry of plant secondary metabolism. Ann. Plant Rev. 2010, 40, 258–303. [Google Scholar]
- Cox-Georgian, D.; Ramadoss, N.; Dona, C.; Basu, C. Therapeutic and medicinal uses of terpenes. In Medicinal Plants; Springer: Berlin/Heidelberg, Germany, 2019; pp. 333–359. [Google Scholar]
- Li, R.; Tee, C.S.; Jiang, Y.L.; Jiang, X.Y.; Venkatesh, P.N.; Sarojam, R.; Ye, J. A terpenoid phytoalexin plays a role in basal defense of Nicotiana benthamiana against potato virus, X. Sci. Rep. 2015, 5, 9682. [Google Scholar] [CrossRef] [PubMed]
- Thimmappa, R.; Geisler, K.; Louveau, T.; O’Maille, P.; Osbourn, A. Triterpene Biosynthesis in Plants. Annu. Rev. Plant Biol. 2014, 65, 225–257. [Google Scholar] [CrossRef]
- Mahdavi, A.; Moradi, P.; Mastinu, A. Variation in Terpene Profiles of Thymus vulgaris in Water Deficit Stress Response. Molecules 2020, 25, 1091. [Google Scholar] [CrossRef] [PubMed]
- Chapman, P.M. Is the regulatory regime for the registration of plant protection products in the EU potentially compromising food security? Food Energy Secur. 2014, 3, 1–6. [Google Scholar] [CrossRef]
- Sparks, T.C.; Nauen, R. IRAC: Mode of action classification and insecticide resistance management. Pesticide Biochem. Physiol. 2015, 121, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, I.I.; Quax, W.J. A glimpse into the biosynthesis of terpenoids. KnE Life Sci. 2017, 3, 81. [Google Scholar] [CrossRef]
- Reynolds, W.F.; Enriquez, R.G. Chapter 7 Terpenes: Mono-, sesqui-, and higher terpenes, in modern NMR approaches to the structure elucidation of natural products: Volume 2: Data acquisition and applications to compound classes. R. Soc. Chem. 2017, 251–274. [Google Scholar] [CrossRef]
- Brahmkshatriya, P.P.; Brahmkshatriya, P.S. Terpenes: Chemistry, biological role, and therapeutic applications. In Natural Products: Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes; Ramawat, K.G., Mérillon, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 2665–2691. [Google Scholar]
- Trost, B.M.; Min, C. Total synthesis of terpenes via palladium-catalyzed cyclization strategy. Nat. Chem. 2020, 12, 568–573. [Google Scholar] [CrossRef] [PubMed]
- Loreto, F.; Pollastri, S.; Fineschi, S.; Velikova, V. Volatile isoprenoids and their importance for protection against environmental constraints in the Mediterranean area. Environ. Exp. Bot. 2014, 103, 99–106. [Google Scholar] [CrossRef]
- Magnard, J.L.; Roccia, A.; Caissard, J.C.; Vergne, P.; Sun, P.; Hecquet, R.; Dubois, A.; Hibrand-Saint Oyant, L.; Jullien, F.; Nicolè, F.; et al. Plant volatiles. Biosynthesis of monoterpene scent compounds in roses. Science 2015, 349, 81–83. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Li, Y.; Dang, P.; Zhao, S.; Lai, D.; Zhou, L. Rice Secondary Metabolites: Structures, Roles, Biosynthesis, and Metabolic Regulation. Molecules 2018, 23, 3098. [Google Scholar] [CrossRef]
- Kang, A.; Lee, T.S. Chapter 2—Secondary metabolism for isoprenoid-based biofuels. In Biotechnology for Biofuel Production and Optimization; Eckert, C.A., Trinh, C.T., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 35–71. [Google Scholar]
- Ashour, M.; Wink, M.; Gershenzon, J. Biochemistry of terpenoids: Monoterpenes, sesquiterpenes, and diterpenes. Annu. Plant Rev. 2018, 15, 258–303. [Google Scholar]
- Mander, L.; Liu, H.-W. Comprehensive natural products II. In Chemistry and Biology, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2010. [Google Scholar]
- Lorigooini, Z.; Jamshidi-Kia, F.; Dodman, S. Chapter 8—Analysis of sesquiterpenes and sesquiterpenoids, in Recent Advances. In Natural Products Analysis; Sanches, S.A., Nabavi, S.F., Saeedi, M., Nabavi, S.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 289–312. [Google Scholar]
- Li, Z.; Howell, K.; Fang, Z.; Zhang, P. Sesquiterpenes in grapes and wines: Occurrence, biosynthesis, functionality, and influence of winemaking processes. Compr. Rev. Food Sci. Food Saf. 2019, 19, 247–281. [Google Scholar] [CrossRef] [PubMed]
- Yadava, N.; Yadava, R.; Goyalb, A. Chemistry of terpenoids. Int. J. Pharm. Sci. Rev. 2014, 27, 272–278. [Google Scholar]
- De Sousa, I.P.; Sousa Teixeira, M.V.; Jacometti Cardoso Furtado, N.A. An overview of biotransformation and toxicity of diterpenes. Molecules 2018, 23, 1387. [Google Scholar] [CrossRef]
- Ludwiczuk, A.; Skalicka-Woźniak, K.; Georgiev, M.I. Chapter 11—Terpenoids. In Pharmacognosy; Badal, S., Delgoda, R., Eds.; Academic Press: Boston, MA, USA, 2017; pp. 233–266. [Google Scholar]
- Takaichi, S. Tetraterpenes: Carotenoids. In Natural Products: Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes; Ramawat, K.G., Mérillon, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 3251–3283. [Google Scholar]
- Maoka, T. Carotenoids as natural functional pigments. J. Nat. Med. 2020, 74, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Sozer, O.; Komenda, J.; Ughy, B.; Domonkos, I.; Laczkó-Dobos, H.; Malec, P.; Gombos, Z.; Kis, M. Involvement of Carotenoids in the Synthesis and Assembly of Protein Subunits of Photosynthetic Reaction Centers of Synechocystis sp. PCC 6803. Plant Cell Physiol. 2010, 51, 823–835. [Google Scholar] [CrossRef]
- Domonkos, I.; Kis, M.; Gombos, Z.; Ughy, B. Carotenoids, versatile components of oxygenic photosynthesis. Prog. Lipid Res. 2013, 52, 539–561. [Google Scholar] [CrossRef] [PubMed]
- Block, A.K.; Vaughan, M.M.; Schmelz, E.A.; Christensen, S.A. Biosynthesis and function of terpenoid defense compounds in maize (Zea mays). Planta 2019, 249, 21–30. [Google Scholar] [CrossRef]
- Tholl, D. Biosynthesis and Biological Functions of Terpenoids in Plants. Blue Biotechnol. 2015, 148, 63–106. [Google Scholar] [CrossRef]
- Zhao, L.; Chang, W.-C.; Xiao, Y.; Liu, H.-W.; Liu, P. Methylerythritol Phosphate Pathway of Isoprenoid Biosynthesis. Annu. Rev. Biochem. 2013, 82, 497–530. [Google Scholar] [CrossRef]
- Wang, Q.; Quan, S.; Xiao, H. Towards efficient terpenoid biosynthesis: Manipulating IPP and DMAPP supply. Bioresour. Bioprocess. 2019, 6, 6. [Google Scholar] [CrossRef]
- Völler, J.-S. Shortcut for isoprenoid biosynthesis. Nat. Catal. 2019, 2, 104. [Google Scholar] [CrossRef]
- He, H.; Bian, G.; Herbst-Gervasoni, C.J.; Mori, T.; Shinsky, S.A.; Hou, A.; Mu, X.; Huang, M.; Cheng, S.; Deng, Z.; et al. Discovery of the cryptic function of terpene cyclases as aromatic prenyltransferases. Nat. Commun. 2020, 11, 1–13. [Google Scholar] [CrossRef]
- Chen, C.-L.; Paul, L.N.; Mermoud, J.C.; Steussy, C.N.; Stauffacher, C.V. Visualizing the enzyme mechanism of mevalonate diphosphate decarboxylase. Nat. Commun. 2020, 11, 3969. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.-C.; Song, H.; Liu, H.-W.; Liu, P. Current development in isoprenoid precursor biosynthesis and regulation. Curr. Opin. Chem. Biol. 2013, 17, 571–579. [Google Scholar] [CrossRef]
- Chatzivasileiou, A.O.; Ward, V.; Edgar, S.M.; Stephanopoulos, G. Two-step pathway for isoprenoid synthesis. Proc. Natl. Acad. Sci. USA 2019, 116, 506–511. [Google Scholar] [CrossRef]
- Major, D.T. Complex terpenes in one pot. Nat. Catal. 2018, 1, 567–568. [Google Scholar] [CrossRef]
- Christianson, D.W. Structural and chemical biology of terpenoid cyclases. Chem. Rev. 2017, 117, 11570–11648. [Google Scholar] [CrossRef]
- Rudolf, J.D.; Chang, C.Y. Terpene synthases in disguise: Enzymology, structure, and opportunities of non-canonical terpene synthases. Nat. Product Rep. 2020, 37, 425–463. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.-Y.; Jin, J.; Sarojam, R.; Ramachandran, S. A Comprehensive Survey on the Terpene Synthase Gene Family Provides New Insight into Its Evolutionary Patterns. Genome Biol. Evol. 2019, 11, 2078–2098. [Google Scholar] [CrossRef]
- Tholl, D. Terpene synthases and the regulation, diversity and biological roles of terpene metabolism. Curr. Opin. Plant Biol. 2006, 9, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Bathe, U.; Tissier, A. Cytochrome P450 enzymes: A driving force of plant diterpene diversity. Phytochemistry 2019, 161, 149–162. [Google Scholar] [CrossRef]
- Chen, F.; Tholl, D.; Bohlmann, J.; Pichersky, E. The family of terpene synthases in plants: A mid-size family of genes for specialized metabolism that is highly diversified throughout the kingdom. Plant J. 2011, 66, 212–229. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.M.; Aubourg, S.; Schouwey, M.B.; Daviet, L.; Schalk, M.; Toub, O.; Lund, S.T.; Bohlmann, J. Functional Annotation, Genome Organization and Phylogeny of the Grapevine (Vitis vinifera) Terpene Synthase Gene Family Based on Genome Assembly, FLcDNA Cloning, and Enzyme Assays. BMC Plant Biol. 2010, 10, 226. [Google Scholar] [CrossRef]
- Falara, V.; Akhtar, T.A.; Nguyen, T.T.H.; Spyropoulou, E.A.; Bleeker, P.M.; Schauvinhold, I.; Matsuba, Y.; Bonini, M.E.; Schilmiller, A.L.; Last, R.L.; et al. The tomato terpene synthase gene family. Plant Physiol. 2011, 157, 770. [Google Scholar] [CrossRef]
- Dudareva, N.; Klempien, A.; Muhlemann, J.K.; Kaplan, I. Biosynthesis, function and metabolic engineering of plant volatile organic compounds. New Phytol. 2013, 198, 16–32. [Google Scholar] [CrossRef]
- Du, F.; Wang, T.; Fan, J.-M.; Liu, Z.-Z.; Zong, J.-X.; Fan, W.-X.; Han, Y.-H.; Grierson, D. Volatile composition and classification of Lilium flower aroma types and identification, polymorphisms, and alternative splicing of their monoterpene synthase genes. Hortic. Res. 2019, 6, 1–15. [Google Scholar] [CrossRef]
- Dodds, P.N.; Rathjen, J.P. Plant immunity: Towards an integrated view of plant-pathogen interactions. Nat. Rev. Genet. 2010, 11, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.W.; Wu, J.Y. Effective elicitors and process strategies for enhancing secondary metabolite production in hairy root cultures. Biotechnol. Hairy Root Syst. 2013, 55–89. [Google Scholar] [CrossRef]
- Namdeo, A. Plant cell elicitation for production of secondary metabolites: A review. Pharmacogn. Rev. 2007, 1, 69–79. [Google Scholar]
- Halder, M.; Sarkar, S.; Jha, S. Elicitation: A biotechnological tool for enhanced production of secondary metabolites in hairy root cultures. Eng. Life Sci. 2019, 19, 880–895. [Google Scholar] [CrossRef] [PubMed]
- Rutledge, P.J.; Challis, G.L. Discovery of microbial natural products by activation of silent biosynthetic gene clusters. Nat. Rev. Microbiol. 2015, 13, 509–523. [Google Scholar] [CrossRef]
- Ma, Y.; Yuan, L.; Wu, B.; Li, X.; Chen, S.; Lu, S. Genome-wide identification and characterization of novel genes involved in terpenoid biosynthesis in Salvia miltiorrhiza. J. Exp. Bot. 2012, 63, 2809–2823. [Google Scholar] [CrossRef]
- Lv, S.; Wang, Z.; Yang, X.; Guo, L.; Qiu, D.; Zeng, H. Transcriptional Profiling of Rice Treated with MoHrip1 Reveal the Function of Protein Elicitor in Enhancement of Disease Resistance and Plant Growth. Front. Plant Sci. 2016, 7, 181. [Google Scholar] [CrossRef]
- Farag, M.A.; Al-Mahdy, D.A.; Meyer, A.; Westphal, H.; Wessjohann, L.A. Metabolomics reveals biotic and abiotic elicitor effects on the soft coral Sarcophyton ehrenbergi terpenoid content. Sci. Rep. 2017, 7, 648. [Google Scholar] [CrossRef]
- Lau, W.; Sattely, E.S. Six enzymes from mayapple that complete the biosynthetic pathway to the etoposide aglycone. Science 2015, 349, 1224–1228. [Google Scholar] [CrossRef]
- Pateraki, I.; Andersen-Ranberg, J.; Jensen, N.B.; Wubshet, S.G.; Heskes, A.M.; Forman, V.; Hallström, B.; Hamberger, B.; Motawia, M.S.; Olsen, C.E.; et al. Total biosynthesis of the cyclic AMP booster forskolin from Coleus forskohlii. eLife 2017, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Caputi, L.; Franke, J.; Farrow, S.C.; Chung, K.; Payne, R.M.E.; Nguyen, T.-D.; Dang, T.-T.T.; Carqueijeiro, I.S.T.; Koudounas, K.; De Bernonville, T.D.; et al. Missing enzymes in the biosynthesis of the anticancer drug vinblastine in Madagascar periwinkle. Science 2018, 360, 1235–1239. [Google Scholar] [CrossRef]
- De La Peña, R.; Sattely, E.S. Rerouting plant terpene biosynthesis enables momilactone pathway elucidation. Nat. Chem. Biol. 2021, 17, 205–212. [Google Scholar] [CrossRef]
- Gao, F.-K.; Yong, Y.-H.; Dai, C.-C. Effects of endophytic fungal elicitor on two kinds of terpenoids production and physiological indexes in Euphorbia pekinensis suspension cells. J. Med. Plants Res. 2011, 5, 4418–4425. [Google Scholar]
- Zhang, J.; Gong, S.; Guo, Z. Effects of different elicitors on 10-deacetylbaccatin III-10-O-acetyltransferase activity and cytochromeP450 monooxygenase content in suspension cultures ofTaxus cuspidatacells. Cell Biol. Int. Rep. 2011, 18, 7–13. [Google Scholar] [CrossRef]
- Badi, H.; Abdoosi, V.; Farzin, N. New Approach to Improve Taxol Biosynthetic. Trakia J. Sci. 2015, 13, 115–124. [Google Scholar] [CrossRef]
- Wyckhuys, K.G.; Lu, Y.; Zhou, W.; Cock MJ, W.; Naranjo, S.E.; Fereti, A.; Williams, F.E.; Furlong, M.J. Ecological pest control fortifies agricultural growth in Asia–Pacific economies. Nat. Ecol. Evolut. 2020, 4, 1522–1530. [Google Scholar] [CrossRef] [PubMed]
- Glas, J.J.; Schimmel, B.C.; Alba, J.M.; Escobar-Bravo, R.; Schuurink, R.C.; Kant, M.R. Plant glandular trichomes as targets for breeding or engineering resistance to herbivores. Int. J. Mol. Sci. 2012, 13, 17077–17103. [Google Scholar] [CrossRef] [PubMed]
- Park, K.S.; Kim, B.H.; Chang, I.-M. Inhibitory potencies of several iridoids on cyclooxygenase-1, cyclooxygenase-2 enzyme activities, tumor necrosis factor-α, and nitric oxide production in vitro. Evid. Based Complement. Altern. Med. eCAM 2010, 7, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Duplais, C.; Papon, N.; Courdavault, V. Tracking the Origin and Evolution of Plant Metabolites. Trends Plant Sci. 2020, 25, 1182–1184. [Google Scholar] [CrossRef]
- War, A.R.; Taggar, G.K.; Hussain, B.; Taggar, M.S.; Nair, R.M.; Sharma, H.C. Plant defense against herbivory and insect adaptations. AoB Plants 2018, 10. [Google Scholar] [CrossRef]
- Olayemi, R.F. The role of monoterpenoids and sesquiterpenoids as defense chemicals in plants—A review. Nigerian Res. J. Chem. Sci. 2017, 3, 1–15. [Google Scholar]
- Lybrand, D.B.; Xu, H.; Last, R.L.; Pichersky, E. How Plants Synthesize Pyrethrins: Safe and Biodegradable Insecticides. Trends Plant Sci. 2020, 25, 1240–1251. [Google Scholar] [CrossRef]
- Chen, M.; Du, Y.; Zhu, G.; Takamatsu, G.; Ihara, M.; Matsuda, K.; Zhorov, B.S.; Dong, K. Action of six pyrethrins purified from the botanical insecticide pyrethrum on cockroach sodium channels expressed in Xenopus oocytes. Pestic. Biochem. Physiol. 2018, 151, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Dhifi, W.; Bellili, S.; Jazi, S.; Bahloul, N.; Mnif, W. Essential Oils’ Chemical Characterization and Investigation of Some Biological Activities: A Critical Review. Medicines 2016, 3, 25. [Google Scholar] [CrossRef] [PubMed]
- Kanno, H.; Hasegawa, H.K.; Kodama, O. Accumulation of salicylic acid, jasmonic acid and phytoalexins in rice, Oryza sativa, infested by the white-backed planthopper, Sogatella furcifera (Hemiptera: Delphacidae). Appl. Ѐntomol. Zool. 2011, 47, 27–34. [Google Scholar] [CrossRef]
- Zhao, M.; Cheng, J.; Guo, B.; Duan, J.; Che, C.-T. Momilactone and Related Diterpenoids as Potential Agricultural Chemicals. J. Agric. Food Chem. 2018, 66, 7859–7872. [Google Scholar] [CrossRef]
- Chiriboga, M.X.; Campos-Herrera, R.; Jaffuel, G.; Röder, G.; Turlings, T.C.J. Diffusion of the maize root signal (E)-β-caryophyllene in soils of different textures and the effects on the migration of the entomopathogenic nematode Heterorhabditis megidis. Rhizosphere 2017, 3, 53–59. [Google Scholar] [CrossRef]
- Tamiru, A.; Bruce, T.J.A.; Richter, A.; Woodcock, C.M.; Midega, C.O.; Degenhardt, J.; Kelemu, S.; Pickett, J.A.; Khan, Z.R. A maize landrace that emits defense volatiles in response to herbivore eggs possess a strongly inducible terpene synthase gene. Ecol. Evolut. 2017, 7, 2835–2845. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.X.; Xiang, C.Y.; Li, J.X.; Yang, C.Q.; Hu, W.L.; Wang, L.J.; Lou, Y.G.; Chen, X.Y. The rice (E)-beta-caryophyllene synthase (OsTPS3) accounts for the major inducible volatile sesquiterpenes. Phytochemistry 2007, 68, 1632–1641. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, V.; Maisnam, J.; Jain, A.; Sharma, K.K.; Bhattacharya, R. Aphid-repellent pheromone E-β-farnesene is generated in transgenic Arabidopsis thaliana over-expressing farnesyl diphosphate synthase2. Ann. Bot. 2014, 115, 581–591. [Google Scholar] [CrossRef]
- Xin, Z.; Cai, X.; Chen, S.; Luo, Z.; Bian, L.; Li, Z.; Ge, L.; Chen, Z. A Disease Resistance Elicitor Laminarin Enhances Tea Defense against a Piercing Herbivore Empoasca (Matsumurasca) onukii Matsuda. Sci. Rep. 2019, 9, 814. [Google Scholar] [CrossRef] [PubMed]
- Winnacker, M.; Rieger, B. Recent Progress in Sustainable Polymers Obtained from Cyclic Terpenes: Synthesis, Properties, and Application Potential. ChemSusChem 2015, 8, 2455–2471. [Google Scholar] [CrossRef]
- Sanchez-Vallet, A.; Ramos, B.; Bednarek, P.; López, G.; Piślewska-Bednarek, M.; Schulze-Lefert, P.; Molina, A. Tryptophan-derived secondary metabolites in Arabidopsis thaliana confer non-host resistance to necrotrophic Plectosphaerella cucumerina fungi. Plant J. 2010, 63, 115–127. [Google Scholar] [CrossRef]
- Ahuja, I.; Kissen, R.; Bones, A.M. Phytoalexins in defense against pathogens. Trends Plant Sci. 2012, 17, 73–90. [Google Scholar] [CrossRef]
- Huang, M.; Sanchez-Moreiras, A.M.; Abel, C.; Sohrabi, R.; Lee, S.; Gershenzon, J.; Tholl, D. The major volatile organic compound emitted from Arabidopsis thaliana flowers, the sesquiterpene (E)-β-caryophyllene, is a defense against a bacterial pathogen. New Phytol. 2012, 193, 997–1008. [Google Scholar] [CrossRef]
- Shibata, Y.; Kawakita, K.; Takemoto, D. Age-Related Resistance of Nicotiana benthamiana Against Hemibiotrophic Pathogen Phytophthora infestans Requires Both Ethylene- and Salicylic Acid–Mediated Signaling Pathways. Mol. Plant Microbe Interact. 2010, 23, 1130–1142. [Google Scholar] [CrossRef] [PubMed]
- Song, N.; Ma, L.; Wang, W.; Sun, H.; Wang, L.; Baldwin, I.T.; Wu, J. An ERF2-like transcription factor regulates production of the defense sesquiterpene capsidiol upon Alternaria alternata infection. J. Exp. Bot. 2019, 70, 5895–5908. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Wang, Q.; Hillwig, M.L.; Peters, R.J. Picking sides: Distinct roles for CYP76M6 and CYP76M8 in rice oryzalexin biosynthesis. Biochem. J. 2013, 454, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Zhan, C.; Lei, L.; Liu, Z.; Zhou, S.; Yang, C.; Zhu, X.; Guo, H.; Zhang, F.; Peng, M.; Li, Y.; et al. Selection of a subspecies-specific diterpene gene cluster implicated in rice disease resistance. Nat. Plants 2020, 6, 1447–1454. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Zhang, J.; Brown, B.; Li, R.; Rodríguez-Romero, J.; Berasategui, A.; Liu, B.; Xu, M.; Luo, D.; Pan, Z.; et al. Inferring Roles in Defense from Metabolic Allocation of Rice Diterpenoids. Plant Cell 2018, 30, 1119–1131. [Google Scholar] [CrossRef]
- Horie, K.; Inoue, Y.; Sakai, M.; Yao, Q.; Tanimoto, Y.; Koga, J.; Toshima, H.; Hasegawa, M. Identification of UV-Induced Diterpenes Including a New Diterpene Phytoalexin, Phytocassane F, from Rice Leaves by Complementary GC/MS and LC/MS Approaches. J. Agric. Food Chem. 2015, 63, 4050–4059. [Google Scholar] [CrossRef]
- Kuljanabhagavad, T.; Wink, M. Biological activities and chemistry of saponins from Chenopodium quinoa Willd. Phytochem. Rev. 2009, 8, 473–490. [Google Scholar] [CrossRef]
- Umemura, K.; Ogawa, N.; Shimura, M.; Koga, J.; Usami, H.; Kono, T. Possible Role of Phytocassane, Rice Phytoalexin, in Disease Resistance of Rice against the Blast Fungus Magnaporthe grisea. Biosci. Biotechnol. Biochem. 2003, 67, 899–902. [Google Scholar] [CrossRef]
- Seo, S.; Gomi, K.; Kaku, H.; Abe, H.; Seto, H.; Nakatsu, S.; Neya, M.; Kobayashi, M.; Nakaho, K.; Ichinose, Y.; et al. Identification of Natural Diterpenes that Inhibit Bacterial Wilt Disease in Tobacco, Tomato and Arabidopsis. Plant Cell Physiol. 2012, 53, 1432–1444. [Google Scholar] [CrossRef]
- Mafu, S.; Ding, Y.; Murphy, K.M.; Yaacoobi, O.; Addison, B.; Wang, Q.; Shen, Z.; Briggs, S.P.; Bohlmann, J.; Castro-Falcon, G.; et al. Discovery, Biosynthesis and Stress-Related Accumulation of Dolabradiene-Derived Defenses in Maize. Plant Physiol. 2018, 176, 2677–2690. [Google Scholar] [CrossRef]
- Christensen, S.A.; Huffaker, A.; Sims, J.; Hunter, C.T.; Block, A.; Vaughan, M.M.; Willett, D.; Romero, M.; Mylroie, J.E.; Williams, W.P. Fungal and herbivore elicitation of the novel maize sesquiterpenoid, zealexin A4, is attenuated by elevated CO2. Planta 2018, 247, 863–873. [Google Scholar] [CrossRef]
- Huffaker, A.; Kaplan, F.; Vaughan, M.M.; Dafoe, N.J.; Ni, X.; Rocca, J.R.; Alborn, H.T.; Teal, P.E.A.; Schmelz, E.A. Novel acidic sesquiterpenoids constitute a dominant class of pathogen-induced phytoalexins in maize. Plant Physiol. 2011, 156, 2082–2097. [Google Scholar] [CrossRef] [PubMed]
- Saad, M.M.G.; Gouda, N.A.; Abdelgaleil, S.M. Bioherbicidal activity of terpenes and phenylpropenes against Echinochloa crus-galli. J. Environ. Sci. Health B 2019, 54, 954–963. [Google Scholar] [CrossRef]
- Heuzé, V.; Thiollet, H.; Tran, G.; Lebas, F. Cockspur Grass (Echinochloa crus-galli) Forage; Feedipedia, A programme by INRA, CIRAD, AFZ and FAO; FAO: Rome, Italy, 2017. [Google Scholar]
- Mahaut, L.; Cheptou, P.-O.; Fried, G.; Munoz, F.; Storkey, J.; Vasseur, F.; Violle, C.; Bretagnolle, F. Weeds: Against the Rules? Trends Plant Sci. 2020, 25, 1107–1116. [Google Scholar] [CrossRef] [PubMed]
- Fahad, S.; Saud, S.; Akhter, A.; Bajwa, A.A.; Hassan, S.; Battaglia, M.; Adnan, M.; Wahid, F.; Datta, R.; Babur, E.; et al. Bio-based integrated pest management in rice: An agro-ecosystems friendly approach for agricultural sustainability. J. Saudi Soc. Agric. Sci. 2021, 20, 94–102. [Google Scholar] [CrossRef]
- Guo, L.; Qiu, J.; Ye, C.; Jin, G.; Mao, L.; Zhang, H.; Yang, X.; Peng, Q.; Wang, Y.; Longjiang, F.; et al. Echinochloa crus-galli genome analysis provides insight into its adaptation and invasiveness as a weed. Nat. Commun. 2017, 8, 1–10. [Google Scholar] [CrossRef]
- Tolaini, V.; Del Fiore, A.; Nobili, C.; Presenti, O.; De Rossi, P.; Procacci, S.; Vitali, F.; Brunori, A. Exploitation of Tartary Buckwheat as Sustainable Ingredient for Healthy Foods Production. Agric. Agric. Sci. Procedia 2016, 8, 455–460. [Google Scholar] [CrossRef][Green Version]
- Cheng, F.; Cheng, Z. Corrigendum: Research Progress on the use of Plant Allelopathy in Agriculture and the Physiological and Ecological Mechanisms of Allelopathy. Front. Plant Sci. 2016, 7, 1697. [Google Scholar] [CrossRef]
- Scognamiglio, M.; D’Abrosca, B.; Esposito, A.; Pacifico, S.; Monaco, P.; Fiorentino, A. Plant growth inhibitors: Allelopathic role or phytotoxic effects? Focus on Mediterranean biomes. Phytochem. Rev. 2013, 12, 803–830. [Google Scholar] [CrossRef]
- Abd-Elgawad, A.M.; El Gendy, A.E.-N.G.; Assaeed, A.M.; Al-Rowaily, S.L.; Alharthi, A.S.; Mohamed, T.A.; Nassar, M.I.; Dewir, Y.H.; Elshamy, A.I. Phytotoxic effects of plant essential oils: A systematic review and structure-activity relationship based on chemometric analyses. Plants 2021, 10, 36. [Google Scholar] [CrossRef]
- De Martino, L.; Mancini, E.; De Almeida, L.F.; De Feo, V. The antigerminative activity of twenty-seven monoterpenes. Molecules 2010, 15, 6630–6637. [Google Scholar] [CrossRef] [PubMed]
- Chon, S.U.; Nelson, C.J. Allelopathy in compositae plants. A review. Agron. Sustain. Dev. 2010, 30, 349–358. [Google Scholar] [CrossRef]
- Santonja, M.; Bousquet-Mélou, A.; Greff, S.; Ormeño, E.; Fernandez, C. Allelopathic effects of volatile organic compounds released from Pinus halepensis needles and roots. Ecol. Evol. 2019, 9, 8201–8213. [Google Scholar] [CrossRef]
- Toyomasu, T.; Usui, M.; Sugawara, C.; Otomo, K.; Hirose, Y.; Miyao, A.; Hirochika, H.; Okada, K.; Shimizu, T.; Koga, J.; et al. Reverse-genetic approach to verify physiological roles of rice phytoalexins: Characterization of a knockdown mutant of OsCPS4 phytoalexin biosynthetic gene in rice. Physiol. Plant 2013, 150, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Galhano, R.; Wiemann, P.; Bueno, E.; Tiernan, M.; Wu, W.; Chung, I.; Gershenzon, J.; Tudzynski, B.; Sesma, A.; et al. Genetic evidence for natural product-mediated plant–plant allelopathy in rice (Oryza sativa). New Phytol. 2012, 193, 570–575. [Google Scholar] [CrossRef] [PubMed]
- Kato-Noguchi, H.; Ota, K.; Kujime, H.; Ogawa, M. Effects of momilactone on the protein expression in Arabidopsis germination. Weed Biol. Manag. 2013, 13, 19–23. [Google Scholar] [CrossRef]
- Kato-Noguchi, H.; Ota, K.; Kujime, H. Absorption of momilactone A and B by Arabidopsis thaliana L. and the growth inhibitory effects. J. Plant Physiol. 2012, 169, 1471–1476. [Google Scholar] [CrossRef]
- Noguchi, H.K.; Ota, K. Biological Activities of Rice Allelochemicals Momilactone A and B. Rice Res. Open Access 2013, 1. [Google Scholar] [CrossRef]
- Weissteiner, S.; Huetteroth, W.; Kollmann, M.; Weißbecker, B.; Romani, R.; Schachtner, J.; Schütz, S. Cockchafer Larvae Smell Host Root Scents in Soil. PLoS ONE 2012, 7, e45827. [Google Scholar] [CrossRef]
- Vaughan, M.M.; Wang, Q.; Webster, F.X.; Kiemle, D.; Hong, Y.J.; Tantillo, D.J.; Coates, R.M.; Wray, A.T.; Askew, W.; O’Donnell, C. Formation of the unusual semi-volatile diterpene rhizathalene by the Arabidopsis class I terpene synthase TPS08 in the root stele is involved in defense against below ground herbivory. Plant Cell 2013, 25, 1108–1125. [Google Scholar] [CrossRef]
- Plata-Rueda, A.; Campos, J.M.; Da Silva Rolim, G.; Martínez, L.C.; Dos Santos, M.H.; Fernandes, F.L.; Serrão, J.E.; Zanuncio, J.C. Terpenoid constituents of cinnamon and clove essential oils cause toxic effects and behavior repellency response on granary weevil, Sitophilus granarius. Ecotoxicol. Environ. Saf. 2018, 156, 263–270. [Google Scholar] [CrossRef]
- Ali, J.G.; Alborn, H.T.; Stelinski, L.L. Constitutive and induced subterranean plant volatiles attract both entomopathogenic and plant-parasitic nematodes. J. Ecol. 2011, 99, 26–35. [Google Scholar] [CrossRef]
- Bian, G.; Han, Y.; Hou, A.; Yuan, Y.; Liu, X.; Deng, Z.; Liu, T. Releasing the potential power of terpene synthases by a robust precursor supply platform. Metab. Eng. 2017, 42, 1–8. [Google Scholar] [CrossRef]
- Byers, K.J.; Bradshaw, H.; Riffell, J.A. Three floral volatiles contribute to differential pollinator attraction in monkeyflowers (Mimulus). J. Exp. Biol. 2014, 217, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Liu, F.; Davis, R.W. Engineering Escherichia coli for the production of terpene mixture enriched in caryophyllene and caryophyllene alcohol as potential aviation fuel compounds. Metab. Eng. Commun. 2018, 6, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Huffaker, A.; Köllner, T.G.; Weckwerth, P.; Robert, C.A.; Spencer, J.L.; Lipka, A.E.; Schmelz, E.A. Selinene volatiles are essential precursors for maize defense promoting fungal pathogen resistance. Plant Physiol. 2017, 175, 1455–1468. [Google Scholar] [CrossRef]
- Cascone, P.; Iodice, L.; Maffei, M.E.; Bossi, S.; Arimura, G.-I.; Guerrieri, E. Tobacco overexpressing β-ocimene induces direct and indirect responses against aphids in receiver tomato plants. J. Plant Physiol. 2015, 173, 28–32. [Google Scholar] [CrossRef]
- Solé, J.; Sans, A.; Riba, M.; Guerrero, A. Behavioural and electrophysiological responses of the European corn borer Ostrinia nubilalis to host-plant volatiles and related chemicals. Physiol. Ѐntomol. 2010, 35, 354–363. [Google Scholar] [CrossRef]
- Yoshitomi, K.; Taniguchi, S.; Tanaka, K.; Uji, Y.; Akimitsu, K.; Gomi, K. Rice terpene synthase 24 (OsTPS24) encodes a jasmonate-responsive monoterpene synthase that produces an antibacterial γ-terpinene against rice pathogen. J. Plant Physiol. 2016, 191, 120–126. [Google Scholar] [CrossRef]
- Chumpolsri, W.; Wijit, N.; Boontakham, P.; Nimmanpipug, P.; Sookwong, P.; Luangkamin, S.; Wongpornchai, S. Variation of terpenoid flavor odorants in the bran of some black and white rice varieties analyzed by GCxGC-MS. J. Food Nutr. Res. 2015, 3, 114–120. [Google Scholar] [CrossRef]
- Lee, G.W.; Chung, M.-S.; Kang, M.; Chung, B.Y.; Lee, S. Direct suppression of a rice bacterial blight (Xanthomonas oryzae pv. oryzae) by monoterpene (S)-limonene. Protoplasma 2015, 253, 683–690. [Google Scholar] [CrossRef]
- Schmelz, E.A.; Kaplan, F.; Huffaker, A.; Dafoe, N.J.; Vaughan, M.M.; Ni, X.; Rocca, J.R.; Alborn, H.T.; Teal, P.E. Identity, regulation, and activity of inducible diterpenoid phytoalexins in maize. Proc. Natl. Acad. Sci. USA 2011, 108, 5455–5460. [Google Scholar] [CrossRef]
- Almeida, A.; Dong, L.; Appendino, G.; Bak, S. Plant triterpenoids with bond-missing skeletons: Biogenesis, distribution and bioactivity. Nat. Prod. Rep. 2020, 37, 1207–1228. [Google Scholar] [CrossRef]
- Miyamoto, K.; Fujita, M.; Shenton, M.R.; Akashi, S.; Sugawara, C.; Sakai, A.; Horie, K.; Hasegawa, M.; Kawaide, H.; Mitsuhashi, W.; et al. Evolutionary trajectory of phytoalexin biosynthetic gene clusters in rice. Plant J. 2016, 87, 293–304. [Google Scholar] [CrossRef]
- Pusztahelyi, T.; Holb, I.J.; Pócsi, I. Secondary metabolites in fungus-plant interactions. Front. Plant Sci. 2015, 6, 573. [Google Scholar] [CrossRef]
- Hasegawa, M.; Mitsuhara, I.; Seo, S.; Imai, T.; Koga, J.; Okada, K.; Yamane, H.; Ohashi, Y. Phytoalexin Accumulation in the Interaction Between Rice and the Blast Fungus. Mol. Plant-Microbe Interact. 2010, 23, 1000–1011. [Google Scholar] [CrossRef] [PubMed]
- Boutanaev, A.M.; Moses, T.; Zi, J.; Nelson, D.R.; Mugford, S.T.; Peters, R.J.; Osbourn, A. Investigation of terpene diversification across multiple sequenced plant genomes. Proc. Natl. Acad. Sci. USA 2015, 112, E81–E88. [Google Scholar] [CrossRef] [PubMed]
- Kato-Noguchi, H.; Peters, R.J. The role of momilactones in rice allelopathy. J. Chem. Ecol. 2013, 39, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Long, J.; Lin, X.; Chang, Z.; Baerson, S.R.; Ding, C.; Wu, X.; Pan, Z.; Song, Y.; Zeng, R. Momilactone B inhibits Arabidopsis growth and development via disruption of ABA and auxin signaling. BioRxiv 2020. [Google Scholar] [CrossRef]
- Quan, N.V.; Xuan, T.D.; Tran, H.-D.; Dieu Thuy, N.T. Inhibitory activities of momilactones A, B, E, and 7-ketostigmasterol isolated from rice husk on paddy and invasive weeds. Plants 2019, 8, 159. [Google Scholar] [CrossRef]
- Mirmostafaee, S.; Azizi, M.; Fujii, Y. Study of Allelopathic Interaction of Essential Oils from Medicinal and Aromatic Plants on Seed Germination and Seedling Growth of Lettuce. Agronomy 2020, 10, 163. [Google Scholar] [CrossRef]
- Chaimovitsh, D.; Shachter, A.; Abu-Abied, M.; Rubin, B.; Sadot, E.; Dudai, N. Herbicidal Activity of Monoterpenes Is Associated with Disruption of Microtubule Functionality and Membrane Integrity. Weed Sci. 2016, 65, 19–30. [Google Scholar] [CrossRef]
- Shrestha, S. Template B v3. 0 (beta): Created by J. Nail 06/2015. Ph.D. Thesis, Mississippi State University, Starkville, MS, USA, 2018. [Google Scholar]
- Ogundipe, A.A.; Okwara, C.; Ogundipe, O.M. CO2 Emissions and environmental implications in Nigeria. Int. J. Energy Econom. Policy 2020, 10, 317. [Google Scholar] [CrossRef]
- Harvey, B.G.; Meylemans, H.A.; Gough, R.V.; Quintana, R.L.; Garrison, M.D.; Bruno, T.J. High-density biosynthetic fuels: The intersection of heterogeneous catalysis and metabolic engineering. Phys. Chem. Chem. Phys. 2014, 16, 9448–9457. [Google Scholar] [CrossRef]
- Hellier, P.; Al-Haj, L.; Talibi, M.; Purton, S.; Ladommatos, N. Combustion and emissions characterization of terpenes with a view to their biological production in cyanobacteria. Fuel 2013, 111, 670–688. [Google Scholar] [CrossRef]
- Mewalal, R.; Rai, D.K.; Kainer, D.; Chen, F.; Külheim, C.; Peter, G.F.; Tuskan, G.A. Plant-derived terpenes: A feedstock for specialty biofuels. Trends Biotechnol. 2017, 35, 227–240. [Google Scholar] [CrossRef] [PubMed]
- Caputi, L.; Aprea, E. Use of terpenoids as natural flavoring compounds in the food industry. Recent Patents Food Nutr. Agric. 2011, 3, 9–16. [Google Scholar] [CrossRef]
- Bergner, E.J.; Ebel, K.; Johann, T.; Löber, O. Method for the Production of Menthol. U.S. Patent No: US7709688B2, 4 May 2010. [Google Scholar]
- Witkewitz, D.L.; Mcgrew, G.N. Combinations of Cooling Agents for Use in Confections. U.S. Patent No: US7767243B2, 3 August 2010. [Google Scholar]
- Zhao, D.-D.; Jiang, L.-L.; Li, H.-Y.; Yan, P.-F.; Zhang, Y.-L. Chemical Components and Pharmacological Activities of Terpene Natural Products from the Genus Paeonia. Molecules 2016, 21, 1362. [Google Scholar] [CrossRef] [PubMed]
- Kokkiripati, P.K.; Kamsala, R.V.; Bashyam, L.; Manthapuram, N.; Bitla, P.; Peddada, V.; Raghavendra, A.S.; Tetali, S. Stem-bark of Terminalia arjuna attenuates human monocytic (THP-1) and aortic endothelial cell activation. J. Ethnopharmacol. 2013, 146, 456–464. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ninkuu, V.; Zhang, L.; Yan, J.; Fu, Z.; Yang, T.; Zeng, H. Biochemistry of Terpenes and Recent Advances in Plant Protection. Int. J. Mol. Sci. 2021, 22, 5710. https://doi.org/10.3390/ijms22115710
Ninkuu V, Zhang L, Yan J, Fu Z, Yang T, Zeng H. Biochemistry of Terpenes and Recent Advances in Plant Protection. International Journal of Molecular Sciences. 2021; 22(11):5710. https://doi.org/10.3390/ijms22115710
Chicago/Turabian StyleNinkuu, Vincent, Lin Zhang, Jianpei Yan, Zhenchao Fu, Tengfeng Yang, and Hongmei Zeng. 2021. "Biochemistry of Terpenes and Recent Advances in Plant Protection" International Journal of Molecular Sciences 22, no. 11: 5710. https://doi.org/10.3390/ijms22115710
APA StyleNinkuu, V., Zhang, L., Yan, J., Fu, Z., Yang, T., & Zeng, H. (2021). Biochemistry of Terpenes and Recent Advances in Plant Protection. International Journal of Molecular Sciences, 22(11), 5710. https://doi.org/10.3390/ijms22115710






