Trospium Chloride Transport by Mouse Drug Carriers of the Slc22 and Slc47 Families
Abstract
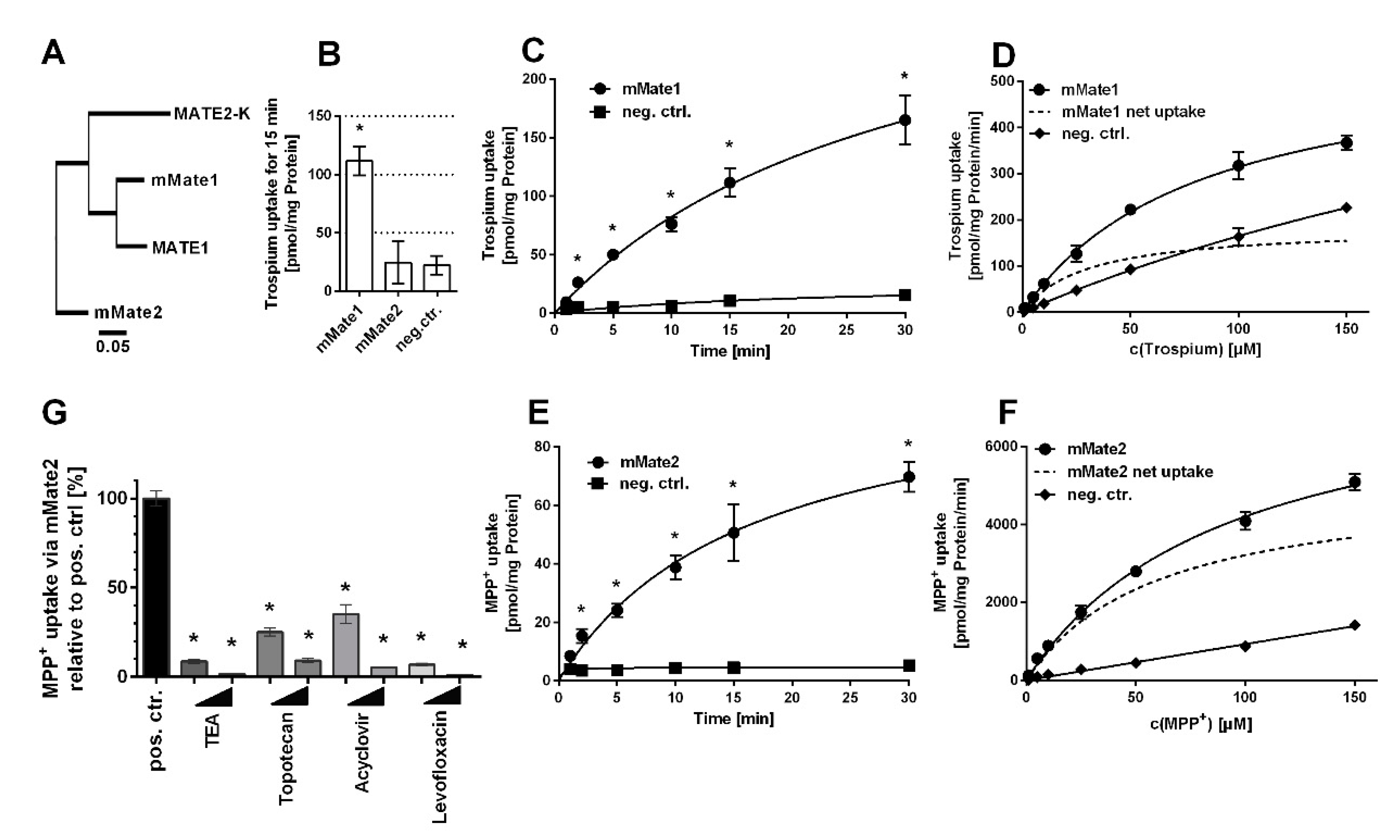
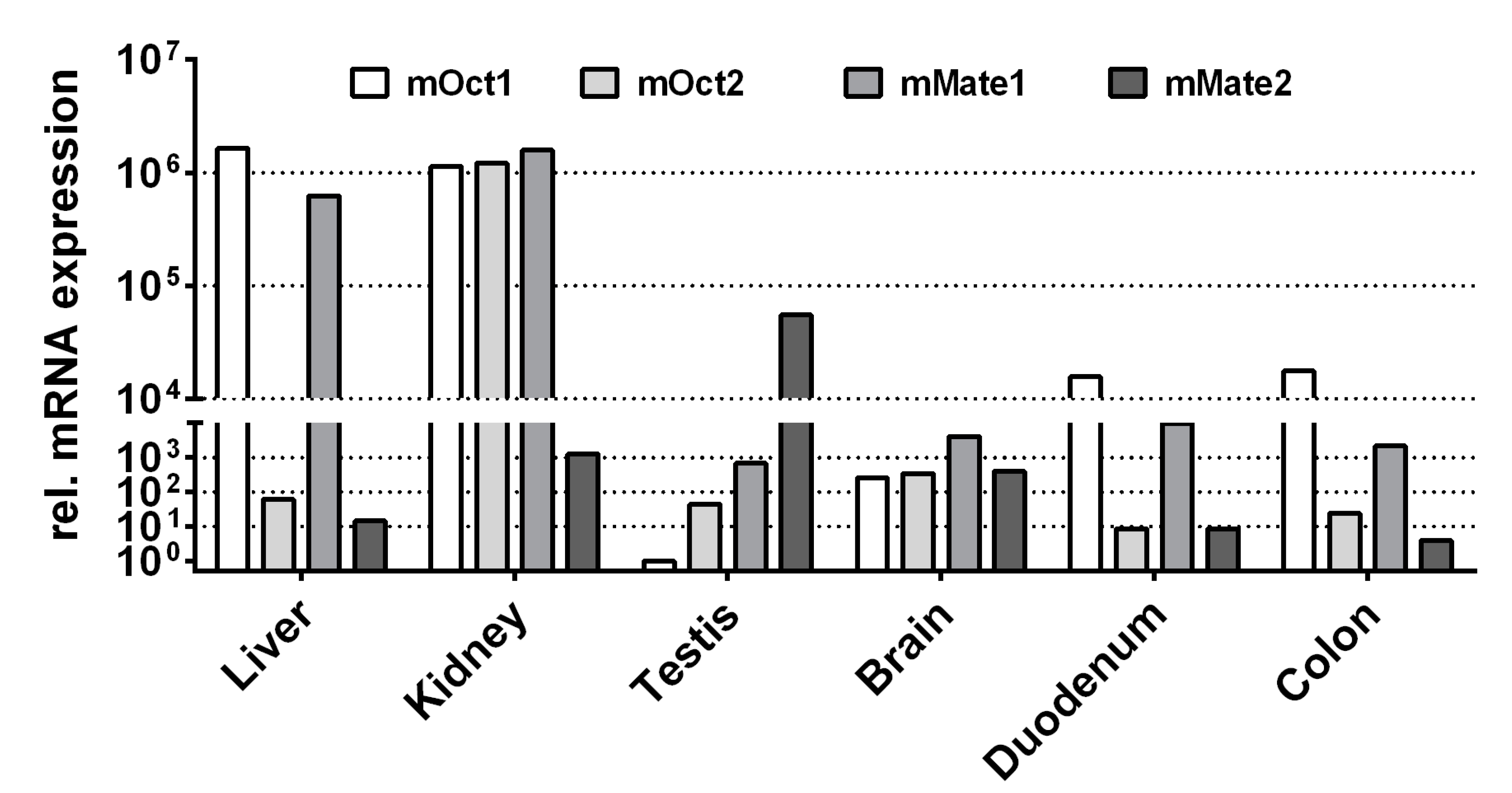
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Materials and Chemicals
4.2. Cloning of mOct1, mOct2, mMate1, and mMate2
4.3. Generation of Stably Transfected HEK293 Cell Lines
4.4. Transport Measurements with MPP+ and Trospium
4.5. RNA Preparation and Quantitative Real-Time PCR Expression Analysis
4.6. Graphical and Statistical Analysis of Data
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| OCT | Organic cation transporter |
| MATE | Multidrug and toxin extrusion |
| SLC | Solute carrier |
| ABCB1 | ATP-binding cassette transporter B1 |
References
- Abrams, P.; Andersson, K.E. Muscarinic receptor antagonists for overactive bladder. BJU Int. 2007, 100, 987–1006. [Google Scholar] [CrossRef] [PubMed]
- Wiedemann, A.; Schwantes, P.A. Antimuscarinic drugs for the treatment of overactive bladder: Are they really all the same?—A comparative review of data pertaining to pharmacological and physiological aspects. Eur. J. Geriat. 2001, 9 (Suppl. 1), 29–42. [Google Scholar]
- Abebe, B.T.; Weiss, M.; Modess, C.; Tadken, T.; Wegner, D.; Meyer, M.J.; Schwantes, U.; Neumeister, C.; Scheuch, E.; Schulz, H.U.; et al. Pharmacokinetic drug-drug interactions between trospium chloride and ranitidine substrates of organic cation transporters in healthy human subjects. J. Clin. Pharmacol. 2020, 60, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Callegari, E.; Malhotra, B.; Bungay, P.J.; Webster, R.; Fenner, K.S.; Kempshall, S.; LaPerle, J.L.; Michel, M.C.; Kay, G.G. A comprehensive non-clinical evaluation of the CNS penetration potential of antimuscarinic agents for the treatment of overactive bladder. Br. J. Clin. Pharmacol. 2011, 72, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Staskin, D.; Kay, G.; Tannenbaum, C.; Goldman, H.B.; Bhashi, K.; Ling, J.; Oefelein, M.G. Trospium chloride has no effect on memory testing and is assay undetectable in the central nervous system of older patients with overactive bladder. Int. J. Clin. Pract. 2010, 64, 1294–1300. [Google Scholar] [CrossRef] [PubMed]
- Chancellor, M.B.; Staskin, D.R.; Kay, G.G.; Sandage, B.W.; Oefelein, M.G.; Tsao, J.W. Blood-brain barrier permeation and efflux exclusion of anticholinergics used in the treatment of overactive bladder. Drugs Aging 2012, 2, 259–273. [Google Scholar] [CrossRef]
- Geyer, J.; Gavrilova, O.; Petzinger, E. The role of P-glycoprotein in limiting brain penetration of the peripherally acting anticholinergic overactive bladder drug trospium chloride. Drug Metab. Dispos. 2009, 37, 1371–1374. [Google Scholar] [CrossRef]
- Wenge, B.; Geyer, J.; Bönisch, H. Oxybutynin and trospium are substrates of the human organic cation transporters. Naunyn Schmiedebergs Arch. Pharmacol. 2011, 383, 203–208. [Google Scholar] [CrossRef]
- Bexten, M.; Oswald, S.; Grube, M.; Jia, J.; Graf, T.; Zimmermann, U.; Rodewald, K.; Zolk, O.; Schwantes, U.; Siegmund, W.; et al. Expression of drug transporters and drug metabolizing enzymes in the bladder urothelium in man and affinity of the bladder spasmolytic trospium chloride to transporters likely involved in its pharmacokinetics. Mol. Pharm. 2015, 12, 171–178. [Google Scholar] [CrossRef]
- Chen, J.; Brockmöller, J.; Seitz, T.; König, J.; Chen, X.; Tzvetkov, M.V. Tropane alkaloids as substrates and inhibitors of human organic cation transporters of the SLC22 (OCT) and the SLC47 (MATE) families. Biol. Chem. 2017, 398, 237–249. [Google Scholar] [CrossRef]
- Deutsch, B.; Neumeister, C.; Schwantes, U.; Fromm, M.F.; König, J. Interplay of the organic cation transporters OCT1 and OCT2 with the apically localized export protein MATE1 for the polarized transport of trospium. Mol. Pharm. 2019, 1, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Koepsell, H.; Lips, K.; Volk, C. Polyspecific organic cation transporters: Structure, function, physiological roles, and biopharmaceutical implications. Pharm. Res. 2007, 24, 1227–1251. [Google Scholar] [CrossRef]
- Terada, T.; Inui, K. Physiological and pharmacokinetic roles of H+/organic cation antiporters (MATE/SLC47A). Biochem. Pharmacol. 2008, 75, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Samodelov, S.L.; Kullak-Ublick, G.A.; Gai, Z.; Visentin, M. Organic cation transporters in human physiology, pharmacology, and toxicology. Int. J. Mol. Sci. 2020, 21, 7890. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Masuda, S.; Yonezawa, A.; Tanihara, Y.; Katsura, T.; Inui, K. Transcellular transport of organic cations in double-transfected MDCK cells expressing human organic cation transporters hOCT1/hMATE1 and hOCT2/hMATE1. Biochem. Pharmacol. 2008, 76, 894–903. [Google Scholar] [CrossRef]
- König, J.; Zolk, O.; Singer, K.; Hoffmann, C.; Fromm, M.F. Double-transfected MDCK cells expressing human OCT1/MATE1 or OCT2/MATE1: Determinants of uptake and transcellular translocation of organic cations. Br. J. Pharmacol. 2011, 163, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Jonker, J.W.; Wagenaar, E.; Van Eijl, S.; Schinkel, A.H. Deficiency in the organic cation transporters 1 and 2 (Oct1/Oct2 [Slc22a1/Slc22a2]) in mice abolishes renal secretion of organic cations. Mol. Cell. Biol. 2003, 23, 7902–7908. [Google Scholar] [CrossRef]
- Tsuda, M.; Terada, T.; Mizuno, T.; Katsura, T.; Shimakura, J.; Inui, K. Targeted disruption of the multidrug and toxin extrusion 1 (mate1) gene in mice reduces renal secretion of metformin. Mol. Pharmacol. 2009, 75, 1280–1286. [Google Scholar] [CrossRef]
- Watanabe, S.; Tsuda, M.; Terada, T.; Katsura, T.; Inui, K. Reduced renal clearance of a zwitterionic substrate cephalexin in MATE1-deficient mice. J. Pharmacol. Exp. Ther. 2010, 334, 651–656. [Google Scholar] [CrossRef]
- Nies, A.T.; Damme, K.; Kruck, S.; Schaeffeler, E.; Schwab, M. Structure and function of multidrug and toxin extrusion proteins (MATEs) and their relevance to drug therapy and personalized medicine. Arch. Toxicol. 2016, 90, 1555–1584. [Google Scholar] [CrossRef]
- Doroshyenko, O.; Jetter, A.; Odenthal, K.P.; Fuhr, U. Clinical pharmacokinetics of trospium chloride. Clin. Pharmacokinet. 2005, 44, 701–720. [Google Scholar] [CrossRef] [PubMed]
- Tadken, T.; Weiss, M.; Modess, C.; Wegner, D.; Roustom, T.; Neumeister, C.; Schwantes, U.; Schulz, H.U.; Weitschies, W.; Siegmund, W. Trospium chloride is absorbed from two intestinal “absorption windows” with different permeability in healthy subjects. Int. J. Pharm. 2016, 515, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Jonker, J.W.; Wagenaar, E.; Mol, C.A.; Buitelaar, M.; Koepsell, H.; Smit, J.W.; Schinkel, A.H. Reduced hepatic uptake and intestinal excretion of organic cations in mice with a targeted disruption of the organic cation transporter 1 (Oct1 [Slc22a1]) gene. Mol. Cell. Biol. 2001, 21, 5471–5477. [Google Scholar] [CrossRef] [PubMed]
- Hiasa, M.; Matsumoto, T.; Komatsu, T.; Moriyama, Y. Wide variety of locations for rodent MATE1, a transporter protein that mediates the final excretion step for toxic organic cations. Am. J. Physiol. Cell Physiol. 2006, 291, C678–C686. [Google Scholar] [CrossRef]
- Hiasa, M.; Matsumoto, T.; Komatsu, T.; Omote, H.; Moriyama, Y. Functional characterization of testis-specific rodent multidrug and toxic compound extrusion 2, a class III MATE-type polyspecific H+/organic cation exporter. Am. J. Physiol. Cell Physiol. 2007, 293, C1437–C1444. [Google Scholar] [CrossRef]
- Otsuka, M.; Matsumoto, T.; Morimoto, R.; Arioka, S.; Omote, H.; Moriyama, Y. A human transporter protein that mediates the final excretion step for toxic organic cations. Proc. Natl. Acad. Sci. USA 2005, 102, 17923–17928. [Google Scholar] [CrossRef]
- Lips, K.S.; Wunsch, J.; Zarghooni, S.; Bschleipfer, T.; Schukowski, K.; Weidner, W.; Wessler, I.; Schwantes, U.; Koepsell, H.; Kummer, W. Acetylcholine and molecular components of its synthesis and release machinery in the urothelium. Eur. Urol. 2007, 51, 1042–1053. [Google Scholar] [CrossRef]
- Geyer, J.; Döring, B.; Meerkamp, K.; Ugele, B.; Bakhiya, N.; Fernandes, C.F.; Godoy, J.R.; Glatt, H.; Petzinger, E. Cloning and functional characterization of human sodium-dependent organic anion transporter (SLC10A6). J. Bio. Chem. 2007, 282, 19728–19741. [Google Scholar] [CrossRef]

| Human Carrier | Km (µM) | Vmax (pmol/mg protein/min) | Mouse Carrier | Km (µM) | Vmax (pmol/mg protein/min) |
|---|---|---|---|---|---|
| OCT1 | 17 ± 5 [8] | 93 ± 26 [8] | mOct1 | 58.7 ± 15.5 | 352.9 ± 39.4 |
| 106 ± 16 [9] | 269 ± 18 [9] | ||||
| 15 ± 3 [10] | 1142 ± 157 [10] | ||||
| OCT2 | 8 ± 1 [8] | 92 ± 11 [8] | mOct2 | 78.5 ± 25.9 | 899.3 ± 139.7 |
| 0.6 ± 0.1 [10] | 98 ± 22 [10] | ||||
| MATE1 | 15 ± 2 [10] | 1083 ± 143 [10] | mMate1 | 29.3 ± 6.7 | 184.7 ± 14.0 |
| MATE2-K | 8 ± 2 [10] | 297 ± 6 [10] | mMate2 | No transport | No transport |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gorecki, M.; Müller, S.F.; Leidolf, R.; Geyer, J. Trospium Chloride Transport by Mouse Drug Carriers of the Slc22 and Slc47 Families. Int. J. Mol. Sci. 2021, 22, 22. https://doi.org/10.3390/ijms22010022
Gorecki M, Müller SF, Leidolf R, Geyer J. Trospium Chloride Transport by Mouse Drug Carriers of the Slc22 and Slc47 Families. International Journal of Molecular Sciences. 2021; 22(1):22. https://doi.org/10.3390/ijms22010022
Chicago/Turabian StyleGorecki, Matthias, Simon F. Müller, Regina Leidolf, and Joachim Geyer. 2021. "Trospium Chloride Transport by Mouse Drug Carriers of the Slc22 and Slc47 Families" International Journal of Molecular Sciences 22, no. 1: 22. https://doi.org/10.3390/ijms22010022
APA StyleGorecki, M., Müller, S. F., Leidolf, R., & Geyer, J. (2021). Trospium Chloride Transport by Mouse Drug Carriers of the Slc22 and Slc47 Families. International Journal of Molecular Sciences, 22(1), 22. https://doi.org/10.3390/ijms22010022






