The Delta-Specific Opioid Glycopeptide BBI-11008: CNS Penetration and Behavioral Analysis in a Preclinical Model of Levodopa-Induced Dyskinesia
Abstract
:1. Introduction
2. Results
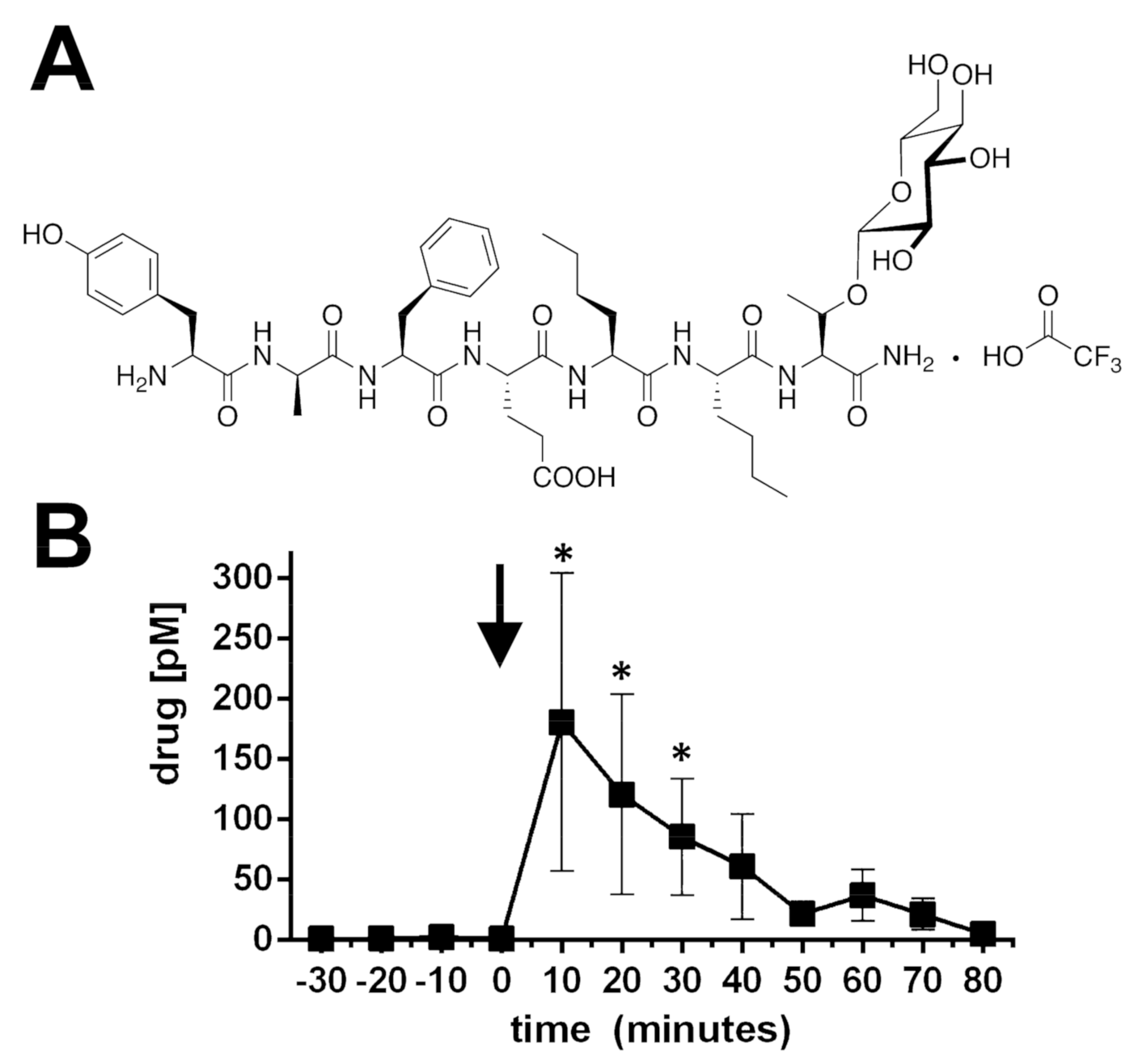
2.1. BBI-11008 Reached High Concentrations in the Rodent Dorsolateral Striatum after Systemic Injection
2.2. Verification of the 6-Hydroxydopamine-Lesion
2.3. Establishment of Limb, Axial, Orolingual and Locomotor Abnormal Involuntary Movements
2.4. BBI-11008 Reduced L-DOPA-Induced Locomotor Abnormal Involuntary Movements
2.5. BBI-11008 Did Not Affect Either the Anti-Dyskinetic Nor the Pro-Parkinsonian Activity of MK-801
3. Discussion
4. Materials and Methods
4.1. Synthesis of BBI-11008
4.2. Animals for Microdialysis
4.3. Animals for L-DOPA-Induced Dyskinesia Model
4.4. In Vivo Microdialysis
4.5. Unilateral 6-Hydroxydopamine-Lesion Rat Model of Parkinson’s Disease
4.6. Amphetamine-Induced Rotation Test
4.7. Induction of L-DOPA-Induced Dyskinesia in Unilateral 6-OHDA-Lesioned Rats
4.8. Behavioral Analysis in the L-DOPA-Induced Dyskinesia Rat Model
4.9. Measurement of Striatal Dopamine Content
4.10. Western Analysis of Striatal Tyrosine Hydroxylase Content
4.11. Data Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| [3H] | Tritium |
| 6-OHDA | 6-hydroxydopamine |
| AIMs | Abnormal involuntary movements |
| ANOVA | Analysis of variance |
| AIR | Amphetamine-induced rotation |
| BBB | Blood-brain barrier |
| BBI-11008 | glycosylated δ-opioid receptor agonist |
| βA | Beta Actin |
| CHO | Chinese hamster ovary |
| CNS | Central nervous system |
| DA | Dopamine |
| DMF | Dimethylformamide |
| DMSO | dimethylsulfoxide |
| DCM | Dichloromethane |
| DIC | N,N′-Diisopropylcarbodiimide |
| DOR | δ opioid receptor |
| DLS | Dorsolateral striatum |
| Fmoc | fluorenylmethoxycarbonyl |
| GABA | gamma-aminobutyric acid |
| Gal | Galactose |
| Glc | Glucose |
| HPLC-EC | High performance liquid chromatography with electrochemical detection |
| HOBT | Hydroxybenzotriazole |
| HRP | Horseradish peroxidase |
| I | Intact hemisphere |
| i.p. | Intraperitoneal |
| LAO | Limb, Axial, and Orolingual |
| LC-MS | Liquid chromatography with mass spectrometry detection |
| L-DOPA | Levodopa, L-3,4-dihydroxyphenylalanine |
| LID | L-DOPA-Induced Dyskinesia |
| KOR | κ Opioid Receptor |
| LX | Lesioned hemisphere |
| Man | Mannose |
| MSNs | Medium spiny neurons |
| MK-801 | Dizocilpine, N-methyl-D-aspartate receptor antagonist |
| MMP-2200 | Lactomorphin, glycosylated mixed μ/δ-opioid receptor agonist |
| MOR | μ opioid receptor |
| NIH | National Institute of Health |
| NIDA | National Institute on Drug Abuse |
| NMDA | N-methyl-D-aspartate |
| NMP | N-methylpyrrolidinone |
| Nle | Nor-Leucine |
| Nva | Nor-Valine |
| PD | Parkinson’s disease |
| PPE-A, Penk | Preproenkephalin-A |
| PPE-B, Pdyn | Preproenkephalin-B |
| TH | Tyrosine Hydroxylase |
References
- Fox, S.H.; Brotchie, J.M. Non-Dopaminergic Approaches to the Treatment of Parkinson’s Disease. In Parkinson’s Disease—Non-Motor and Non-Dopaminergic Features; Olanow, W.C., Stocchi, F., Lang, A.E., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2011; pp. 443–444. [Google Scholar]
- Olanow, C.W.; Stern, M.B.; Sethi, K. The scientific and clinical basis for the treatment of Parkinson disease. Neurology 2009, 72, S1–S136. [Google Scholar]
- Huot, P.; Johnston, T.H.; Koprich, J.B.; Fox, S.H.; Brotchie, J.M. The Pharmacology of l-DOPA-Induced Dyskinesia in Parkinson’s Disease. Pharmacol. Rev. 2013, 65, 171–222. [Google Scholar]
- Cuello, A.C.; Paxinos, G. Evidence for a long Leu-enkephalin striopallidal pathway in rat brain. Nature 1978, 271, 178–180. [Google Scholar]
- Gerfen, C.R.; Engber, T.M.; Mahan, L.C.; Susel, Z.; Chase, T.N.; Monsma, F.J.; Sibley, D.R. D1 and D2 dopamine receptor regulated gene expression of striatonigral and striatopallidal neurons. Science 1990, 250, 1429–1432. [Google Scholar]
- Seizinger, B.R.; Grimm, C.; Höllt, V.; Herz, A. Evidence for a selective processing of proenkephalin B into different opioid peptide forms in particular regions of rat brain and pituitary. J. Neurochem. 1984, 42, 447–457. [Google Scholar]
- Breslin, M.B.; Lindberg, I.; Benjannet, S.; Mathis, J.P.; Lazure, C.; Seidah, N.G. Differential processing of proenkephalin by prohormone convertases 1(3) and 2 and furin. J. Biol. Chem. 1993, 268, 27084–27093. [Google Scholar]
- Sgroi, S.; Tonini, R. Opioidergic Modulation of Striatal Circuits, Implications in Parkinson’s Disease and Levodopa Induced Dyskinesia. Front. Neurol. 2018, 9, 524. [Google Scholar]
- Bezard, E.; Li, Q.; Hulme, H.; Fridjonsdottir, E.; Nilsson, A.; Pioli, E.; Andren, P.E.; Crossman, A.R. µ Opioid Receptor Agonism for L-DOPA-Induced Dyskinesia in Parkinson’s Disease. J. Neurosci. 2020, 40, 6812–6819. [Google Scholar]
- Herrero, M.T.; Augood, S.J.; Hirsch, E.C.; Javoy-Agid, F.; Luquin, M.R.; Agid, Y.; Obeso, J.A.; Emson, P.C. Effects of l-DOPA on preproenkephalin and preprotachykinin gene expression in the MPTP-treated monkey striatum. Neuroscience 1995, 68, 1189–1198. [Google Scholar]
- Nisbet, A.P.; Foster, O.J.; Kingsbury, A.; Eve, D.J.; Daniel, S.E.; Marsden, C.D.; Lees, A.J. Preproenkephalin and preprotachykinin messenger RNA expression in normal human basal ganglia and in Parkinson’s disease. Neuroscience 1995, 66, 361–376. [Google Scholar]
- Cenci, M.A.; Lee, C.S.; Björklund, A. L-DOPA-induced dyskinesia in the rat is associated with striatal overexpression of prodynorphin- and glutamic acid decarboxylase mRNA. Eur. J. Neurosci. 1998, 10, 2694–2706. [Google Scholar]
- Sgroi, S.; Capper-Loup, C.; Paganetti, P.; Kaelin-Lang, A. Enkephalin and dynorphin neuropeptides are differently correlated with locomotor hypersensitivity and levodopa-induced dyskinesia in Parkinsonian rats. Exp. Neurol. 2016, 280, 80–88. [Google Scholar]
- Tel, B.C.; Zeng, B.Y.; Cannizzaro, C.; Pearce, R.K.B.; Rose, S.; Jenner, P. Alterations in striatal neuropeptide mRNA produced by repeated administration of L-DOPA, ropinirole or bromocriptine correlate with dyskinesia induction in MPTP-treated common marmosets. Neuroscience 2002, 115, 1047–1058. [Google Scholar]
- Aubert, I.; Guigoni, C.; Li, Q.; Dovero, S.; Bioulac, B.H.; Gross, C.E.; Crossman, A.R.; Bloch, B.; Bezard, E. Enhanced Preproenkephalin-B-Derived Opioid Transmission in Striatum and Subthalamic Nucleus Converges Upon Globus Pallidus Internalis in L-3,4-dihydroxyphenylalanine-Induced Dyskinesia. Biol. Psychiatry 2007, 61, 836–844. [Google Scholar]
- Hille, C.J.; Fox, S.H.; Maneuf, Y.P.; Crossman, A.R.; Brotchie, J.M. AntiParkinsonian action of a delta opioid agonist in rodent and primate models of Parkinson’s disease. Exp. Neurol. 2001, 172, 189–198. [Google Scholar]
- Hudzik, T.J.; Howell, A.; Payza, K.; Cross, A.J. Antiparkinson potential of delta-opioid receptor agonists. Eur. J. Pharmacol. 2000, 396, 101–107. [Google Scholar]
- Westin, J.E.; Andersson, M.; Lundblad, M.; Cenci, M.A. Persistent changes in striatal gene expression induced by long-term L-DOPA treatment in a rat model of Parkinson’s disease. Eur. J. Neurosci. 2001, 14, 1171–1176. [Google Scholar]
- Calon, F.; Rajput, A.H.; Hornykiewicz, O.; Bédard, P.J.; Di Paolo, T. Levodopa-induced motor complications are associated with alterations of glutamate receptors in Parkinson’s disease. Neurobiol. Dis. 2003, 14, 404–416. [Google Scholar]
- Tamim, M.K.; Samadi, P.; Morissette, M.; Grégoire, L.; Ouattara, B.; Lévesque, D.; Rouillard, C.; Di Paolo, T. Effect of non-dopaminergic drug treatment on Levodopa induced dyskinesias in MPTP monkeys: Common implication of striatal neuropeptides. Neuropharmacology 2010, 58, 286–296. [Google Scholar]
- Piccini, P.; Weeks, R.A.; Brooks, D.J. Alterations in opioid receptor binding in Parkinson’s disease patients with levodopa-induced dyskinesias. Ann. Neurol. 1997, 42, 720–726. [Google Scholar]
- Rascol, O.; Fabre, N.; Blin, O.; Poulik, J.; Sabatini, U.; Senard, J.M.; Ane, M.; Montastruc, J.L.; Rascol, A. Naltrexone, an opiate antagonist, fails to modify motor symptoms in patients with Parkinson’s disease. Mov. Disord. 1994, 9, 437–440. [Google Scholar]
- Fox, S.H.; Silverdale, M.; Kellett, M.; Davies, R.; Steiger, M.; Fletcher, N.; Crossman, A.; Brotchie, J. Non-subtype-selective opioid receptor antagonism in treatment of levodopa-induced motor complications in Parkinson’s disease. Mov. Disord. 2004, 19, 554–560. [Google Scholar]
- Portoghese, P.S.; Lipkowski, A.W.; Takemori, A.E. Binaltorphimine and nor-binaltorphimine, potent and selective k-opioid receptor antagonists. Life Sci. 1987, 40, 1287–1292. [Google Scholar]
- Bilsky, E.J.; Egleton, R.D.; Mitchell, S.A.; Palian, M.M.; Davis, P.; Huber, J.D.; Jones, H.; Yamamura, H.I.; Janders, J.; Davis, T.P.; et al. Enkephalin glycopeptide analogues produce analgesia with reduced dependence liability. J. Med. Chem. 2000, 43, 2586–2590. [Google Scholar]
- Gengo, P.J.; Chang, K.-J. Mixed opioid receptor agonists as a new class of agents for the treatment of moderate to severe pain. In The Delta Receptor; CRC Press: Boca Raton, FL, USA, 2003; pp. 231–244. [Google Scholar]
- Mabrouk, O.S.; Falk, T.; Sherman, S.J.; Kennedy, R.T.; Polt, R. CNS penetration of the opioid glycopeptide MMP-2200: A microdialysis study. Neurosci. Lett. 2012, 531, 99–103. [Google Scholar]
- Yue, X.; Falk, T.; Zuniga, L.A.; Szabò, L.; Porreca, F.; Polt, R.; Sherman, S.J. Effects of the novel glycopeptide opioid agonist MMP-2200 in preclinical models of Parkinson’s disease. Brain Res. 2011, 1413, 72–83. [Google Scholar]
- Flores, A.J.; Bartlett, M.J.; Root, B.K.; Parent, K.L.; Heien, M.L.; Porreca, F.; Polt, R.; Sherman, S.J.; Falk, T. The combination of the opioid glycopeptide MMP-2200 and a NMDA receptor antagonist reduced l-DOPA-induced dyskinesia and MMP-2200 by itself reduced dopamine receptor 2-like agonist-induced dyskinesia. Neuropharmacology 2018, 141, 260–271. [Google Scholar]
- Paquette, M.A.; Anderson, A.M.; Lewis, J.R.; Meshul, C.K.; Johnson, S.W.; Paul Berger, S. MK-801 inhibits L-DOPA-induced abnormal involuntary movements only at doses that worsen parkinsonism. Neuropharmacology 2010, 58, 1002–1008. [Google Scholar]
- Flores, A.J.; Bartlett, M.J.; So, L.Y.; Laude, N.D.; Parent, K.L.; Heien, M.L.; Sherman, S.J.; Falk, T. Differential effects of the NMDA receptor antagonist MK-801 on dopamine receptor D1- and D2-induced abnormal involuntary movements in a preclinical model. Neurosci. Lett. 2014, 564, 48–52. [Google Scholar]
- Negri, L.; Melchiorri, P.; Lattanzi, R. Pharmacology of amphibian opiate peptides. Peptides 2000, 21, 1639–1647. [Google Scholar]
- Stevenson, G.W.; Giuvelis, D.; Cormier, J.; Cone, K.; Atherton, P.; Krivitsky, R.; Warner, E.; St Laurent, B.; Dutra, J.; Bidlack, J.M.; et al. Behavioral pharmacology of the mixed-action delta-selective opioid receptor agonist BBI-11008: Studies on acute, inflammatory and neuropathic pain, respiration, and drug self-administration. Psychopharmacology 2020, 237, 1195–1208. [Google Scholar]
- Cenci, M.A.; Crossman, A.R. Animal models of l-dopa-induced dyskinesia in Parkinson’s disease. Mov. Disord. 2018, 33, 889–899. [Google Scholar]
- Manson, A.J.; Katzenschlager, R.; Hobart, J.; Lees, A.J. High dose naltrexone for dyskinesias induced by levodopa. J. Neurol. Neurosurg. Psychiatry 2001, 70, 554–556. [Google Scholar]
- Lowery, J.J.; Yeomans, L.; Keyari, C.M.; Davis, P.; Porreca, F.; Knapp, B.I.; Bidlack, J.M.; Bilsky, E.J.; Polt, R. Glycosylation improves the central effects of DAMGO. Chem. Biol. Drug Des. 2007, 69, 41–47. [Google Scholar]
- Lowery, J.J.; Raymond, T.J.; Giuvelis, D.; Bidlack, J.M.; Polt, R.; Bilsky, E.J. In vivo characterization of MMP-2200, a mixed δ/μ opioid agonist, in mice. J. Pharmacol. Exp. Ther. 2011, 336, 767–778. [Google Scholar]
- Hay, M.; Polt, R.; Heien, M.L.; Vanderah, T.W.; Largent-Milnes, T.M.; Rodgers, K.; Falk, T.; Bartlett, M.J.; Doyle, K.P.; Konhilas, J.P. A Novel Angiotensin-(1-7) Glycosylated Mas Receptor Agonist for Treating Vascular Cognitive Impairment and Inflammation-Related Memory Dysfunction. J. Pharmacol. Exp. Ther. 2019, 369, 9–25. [Google Scholar]
- Allen, D.D.; Crooks, P.A.; Yokel, R.A. 4-Trimethylammonium antipyrine: A quaternary ammonium nonradionuclide marker for blood–brain barrier integrity during in vivo microdialysis. J. Pharmacol. Toxicol. Meth. 1992, 28, 129–135. [Google Scholar]
- De Lange, E.C.; Danhof, M.; de Boer, A.G.; Breimer, D.D. Methodological considerations of intracerebral microdialysis in pharmacokinetic studies on drug transport across the blood–brain barrier. Brain Res. Brain Res. Rev. 1997, 25, 27–49. [Google Scholar]
- Tetsuya, T.; Yoshiharu, D.; Yuko, K.; Pardridge, W.M.; Akira, T. Determination of in vivo steady-state unbound drug concentration in the brain interstitial fluid by microdialysis. Int. J. Pharmaceutics 1992, 81, 143–152. [Google Scholar]
- Morgan, M.E.; Singhal, D.; Anderson, B.D. Quantitative assessment of blood–brain barrier damage during microdialysis. J. Pharmacol. Exp. Ther. 1996, 277, 1167–1176. [Google Scholar]
- Lundblad, M.; Andersson, M.; Winkler, C.; Kirik, D.; Wierup, N.; Cenci, M.A. Pharmacological validation of behavioural measures of akinesia and dyskinesia in a rat model of Parkinson’s disease. Eur. J. Neurosci. 2002, 15, 120–132. [Google Scholar]
- Johnston, T.H.; Versi, E.; Howson, P.A.; Ravenscroft, P.; Fox, S.H.; Hill, M.P.; Reidenberg, B.E.; Corey, R.; Brotchie, J.M. DPI-289, a novel mixed delta opioid agonist/mu opioid antagonist (DAMA), has L-DOPA-sparing potential in Parkinson’s disease. Neuropharmacology 2018, 131, 116–127. [Google Scholar]
- Potts, L.F.; Park, E.S.; Woo, J.M.; Dyavar Shetty, B.L.; Singh, A.; Braithwaite, S.P.; Voronkov, M.; Papa, S.M.; Mouradian, M.M. Dual κ-agonist/μ-antagonist opioid receptor modulation reduces levodopa-induced dyskinesia and corrects dysregulated striatal changes in the nonhuman primate model of Parkinson’s disease. Neurology 2015, 77, 930–941. [Google Scholar]
- Bartlett, M.J.; So, L.Y.; Szabò, L.; Skinner, D.P.; Parent, K.L.; Heien, M.L.; Vanderah, T.W.; Polt, R.; Sherman, S.J.; Falk, T. Highly-selective µ-opioid receptor antagonism does not block L-DOPA-induced dyskinesia in a rodent model. BMC Res. Notes 2020, 13, 149. [Google Scholar]
- Koprich, J.B.; Fox, S.H.; Johnston, T.H.; Goodman, A.; Bourdonnec, B.; Dolle, R.E.; DeHaven, R.N.; DeHaven-Hudkins, D.L.; Little, P.J.; Brotchie, J.M. The selective mu-opioid receptor antagonist ADL5510 reduces levodopa-induced dyskinesia without affecting antiparkinsonian action in MPTP-lesioned macaque model of Parkinson’s disease. Mov. Disord. 2011, 26, 1225–1233. [Google Scholar]
- Bartlett, M.J.; Joseph, R.M.; LePoidevin, L.M.; Parent, K.L.; Laude, N.D.; Lazarus, L.B.; Heien, M.L.; Estevez, M.; Sherman, S.J.; Falk, T. Long-term effect of sub-anesthetic ketamine in reducing L-DOPA-induced dyskinesias in a preclinical model. Neurosci. Lett. 2016, 612, 121–125. [Google Scholar]
- Bartlett, M.J.; Flores, A.J.; Ye, T.; Smidt, S.I.; Dollish, H.K.; Stancati, J.A.; Farrell, D.C.; Parent, K.L.; Doyle, K.P.; Besselsen, D.G.; et al. Preclinical evidence in support of repurposing sub-anesthetic ketamine as a treatment for L-DOPA-induced dyskinesia. Exp. Neurol. 2020, 333, 113413. [Google Scholar]
- Finck, A.D.; Ngai, S.H. Opiate receptor mediation of ketamine analgesia. Anesthesiology 1982, 56, 291–297. [Google Scholar]
- Pacheco, D.F.; Romero, T.R.; Duarte, I.D. Central antinociception induced by ketamine is mediated by endogenous opioids and μ- and δ-opioid receptors. Brain Res. 2014, 1562, 69–75. [Google Scholar]
- Cenci, M.A.; Riggare, S.; Pahwa, R.; Eidelberg, D.; Hauser, R.A. Dyskinesia matters. Mov. Disord. 2020, 35, 392–396. [Google Scholar]
- Mitchell, S.A.; Pratt, M.R.; Hruby, V.J.; Polt, R. Solid-phase synthesis of O-linked glycopeptide analogues of enkephalin. J. Org. Chem. 2001, 66, 2327–2342. [Google Scholar]
- Apostol, C.R.; Hay, M.; Polt, R. Glycopeptide drugs: A pharmacological dimension between “Small Molecules” and “Biologics”. Peptides 2020, 131, 170369. [Google Scholar]
- Mignogna, G.; Severini, C.; Simmaco, M.; Negri, L.; Erspamer, G.F.; Kreil, G.; Barra, D. Identification and characterization of two dermorphins from skin extracts of the Amazonian frog Phyllomedusa bicolor. FEBS Lett. 1992, 302, 151–154. [Google Scholar]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates, 6th ed.; Academic Press: Cambridge, MA, USA, 2007. [Google Scholar]
- Dekundy, A.; Lundblad, M.; Danysz, W.; Cenci, M.A. Modulation of L-DOPA-induced abnormal involuntary movements by clinically tested compounds: Further validation of the rat dyskinesia model. Behav. Brain Res. 2007, 179, 76–89. [Google Scholar]



| Sequence | % Inhibition at DOR 100 nM, 1 µM | % Inhibition at MOR 100 nM, 1 µM | Ki DOR (nM) | Ki MOR (nM) | Ki KOR (nM) | |
|---|---|---|---|---|---|---|
| DPDPE | 72, 84 | 23, 62 | 4.3 | 180 | ||
| H2N-YaFDVVG-S(β-D-Glc)-G | LSZ-27 | 51, 67 | 6, 48 | 33 | 570 | |
| H2N-YmFHLM-S(β-D-Glc) | BBI-11001 | 72, 84 | 63, 89 | |||
| H2N-YaFHLA-S(β-D-Glc) | BBI-11002 | 55, 77 | 82, 97 | |||
| H2N-YmFHLMT-(β-D-Glc) | BBI-11003 | 75, 83 | 57, 89 | |||
| H2N-YaFHLAT-(β-D-Glc) | BBI-11004 | 61, 76 | 88, 98 | |||
| H2N-YaFE-Nva-Nva-S-(β-D-Glc) | BBI-11005 | 65, 80 | 11, 44 | 20 | 710 | 5.3% |
| H2N-YaFE-Nle-Nle-S-(β-D-Glc) | BBI-11006 | 66, 81 | 7, 42 | 11 | 820 | 38% |
| H2N-YaFE-Nva-Nva-T-(β-D-Glc) | BBI-11007 | 64, 80 | 13, 36 | 16 | 1300 | 0% |
| H2N-YaFE-Nle-Nle-T-(β-D-Glc) | BBI-11008 | 74, 84 | 7, 37 | 14 | 1100 | 2.9% |
| H2N-YaFE-Nle-Nle-T | BBI-11009 | 11 | 510 | 3.7% | ||
| H2N-YaFEII-T-(β-D-Glc) | BBI-11014 | 80 | 2600 | 36% | ||
| H2N-YaFEVV-S-(β-D-Glc) | BBI-11015 | 320 | 1500 | 0% | ||
| H2N-YaFE-Nle-Nle-S-(βGlc-βGal) | BBI-11021 | 17 | 1500 | 23% | ||
| H2N-YaFEVV-S-(βGlc-βGal) | BBI-11022 | 3600 | 1000 | 71% | ||
| H2N-YaFE-Nle-Nle-S-(α-D-Man) | BBI-11023 | 8.2 | 840 | 4.7% | ||
| H2N-YaFE-Nva-Nva-S-(α-D-Man) | BBI-11024 | 13 | 990 | 0% | ||
| H2N-YaFE-Nle-Nle-S-(βGlc-βGlc) | BBI-11025 | 13 | 1200 | 46% | ||
| H2N-YaFE-Ile-Ile-S-(βGlc-βGlc) | BBI-11026 | 61 | 3000 | 2.7% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bartlett, M.J.; Mabrouk, O.S.; Szabò, L.; Flores, A.J.; Parent, K.L.; Bidlack, J.M.; Heien, M.L.; Kennedy, R.T.; Polt, R.; Sherman, S.J.; et al. The Delta-Specific Opioid Glycopeptide BBI-11008: CNS Penetration and Behavioral Analysis in a Preclinical Model of Levodopa-Induced Dyskinesia. Int. J. Mol. Sci. 2021, 22, 20. https://doi.org/10.3390/ijms22010020
Bartlett MJ, Mabrouk OS, Szabò L, Flores AJ, Parent KL, Bidlack JM, Heien ML, Kennedy RT, Polt R, Sherman SJ, et al. The Delta-Specific Opioid Glycopeptide BBI-11008: CNS Penetration and Behavioral Analysis in a Preclinical Model of Levodopa-Induced Dyskinesia. International Journal of Molecular Sciences. 2021; 22(1):20. https://doi.org/10.3390/ijms22010020
Chicago/Turabian StyleBartlett, Mitchell J., Omar S. Mabrouk, Lajos Szabò, Andrew J. Flores, Kate L. Parent, Jean M. Bidlack, Michael L. Heien, Robert T. Kennedy, Robin Polt, Scott J. Sherman, and et al. 2021. "The Delta-Specific Opioid Glycopeptide BBI-11008: CNS Penetration and Behavioral Analysis in a Preclinical Model of Levodopa-Induced Dyskinesia" International Journal of Molecular Sciences 22, no. 1: 20. https://doi.org/10.3390/ijms22010020
APA StyleBartlett, M. J., Mabrouk, O. S., Szabò, L., Flores, A. J., Parent, K. L., Bidlack, J. M., Heien, M. L., Kennedy, R. T., Polt, R., Sherman, S. J., & Falk, T. (2021). The Delta-Specific Opioid Glycopeptide BBI-11008: CNS Penetration and Behavioral Analysis in a Preclinical Model of Levodopa-Induced Dyskinesia. International Journal of Molecular Sciences, 22(1), 20. https://doi.org/10.3390/ijms22010020





