18F-FDG PET-Based Imaging of Myocardial Inflammation Following Acute Myocardial Infarction in a Mouse Model
Abstract
1. Introduction
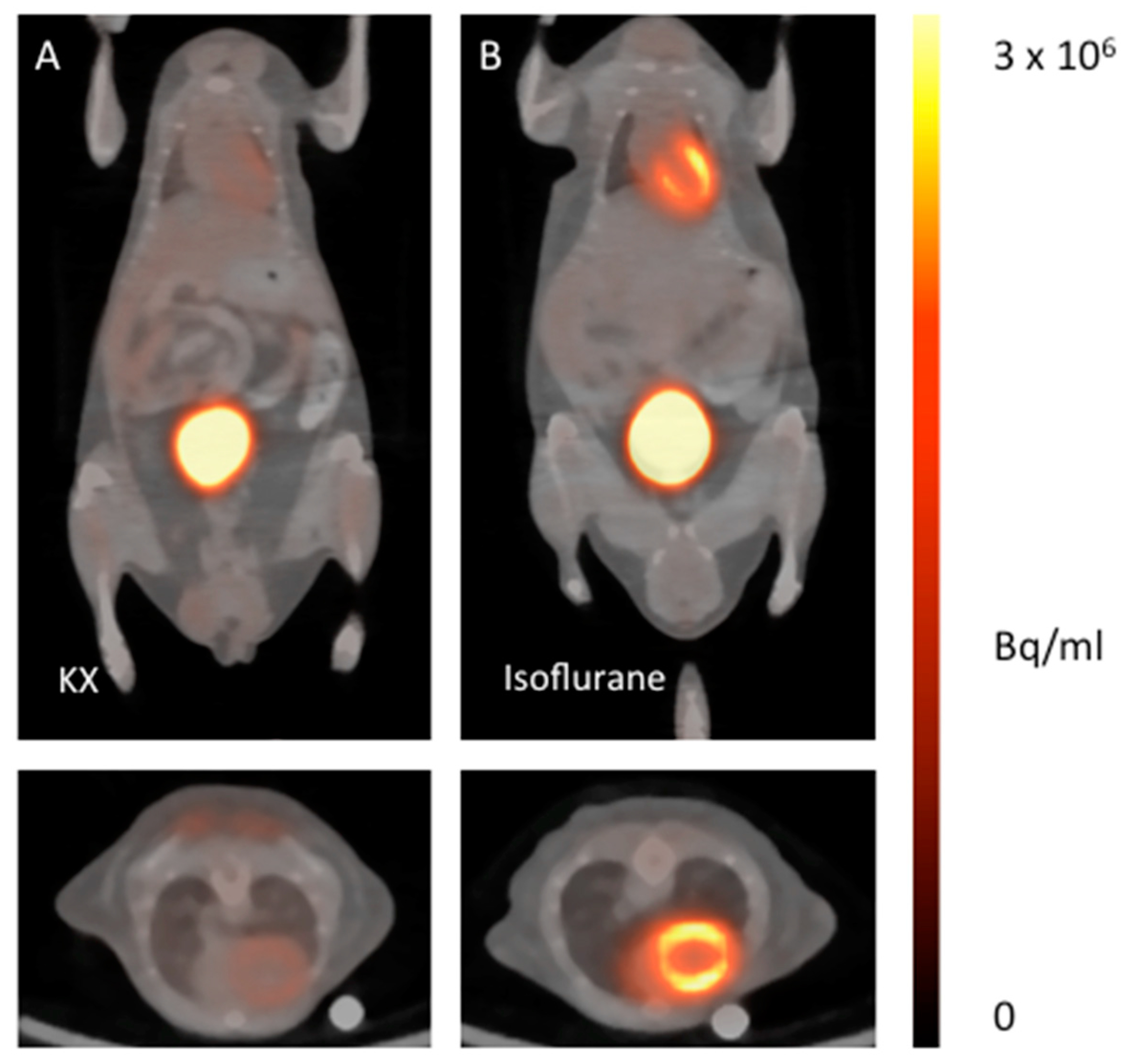
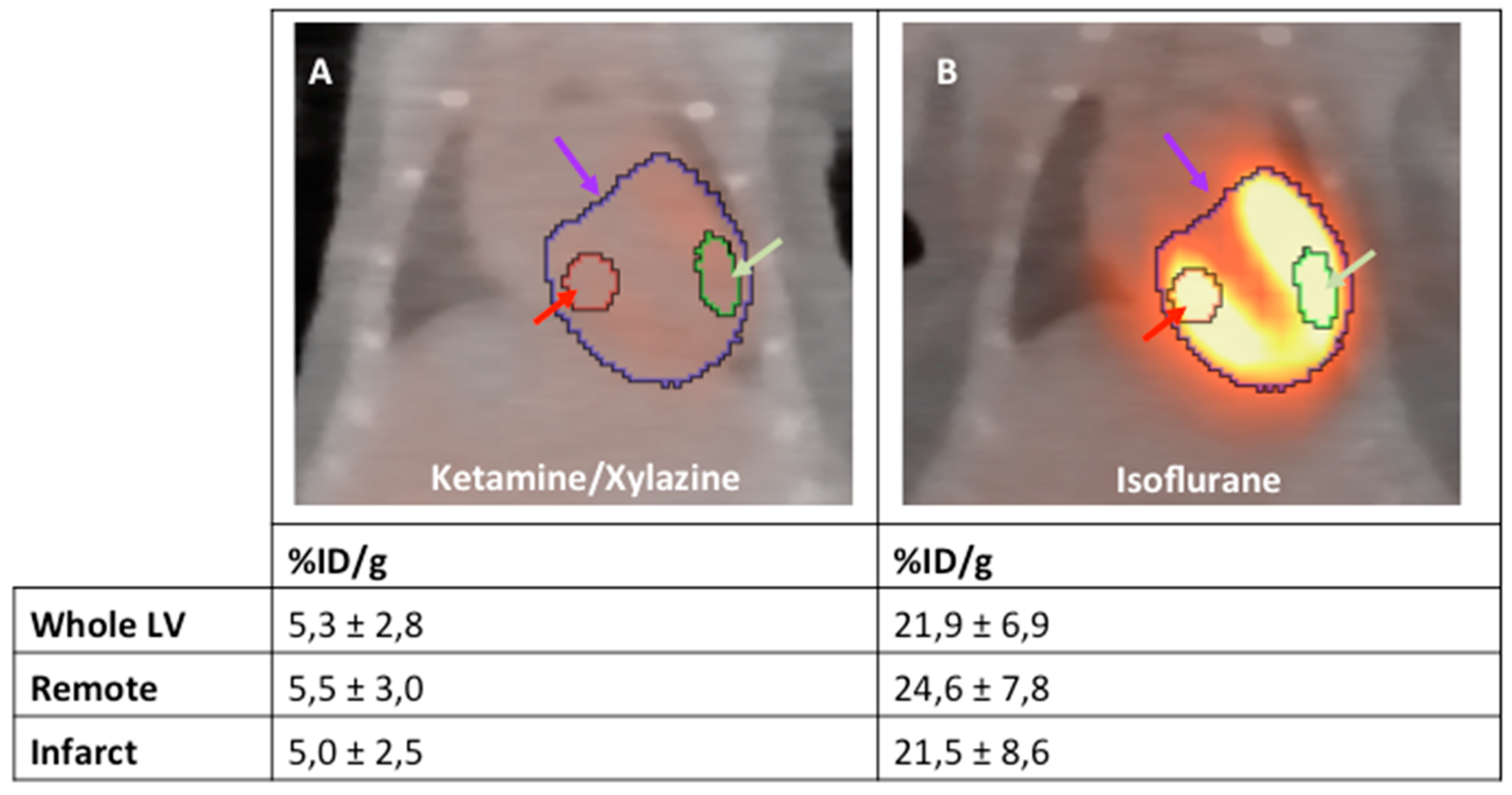
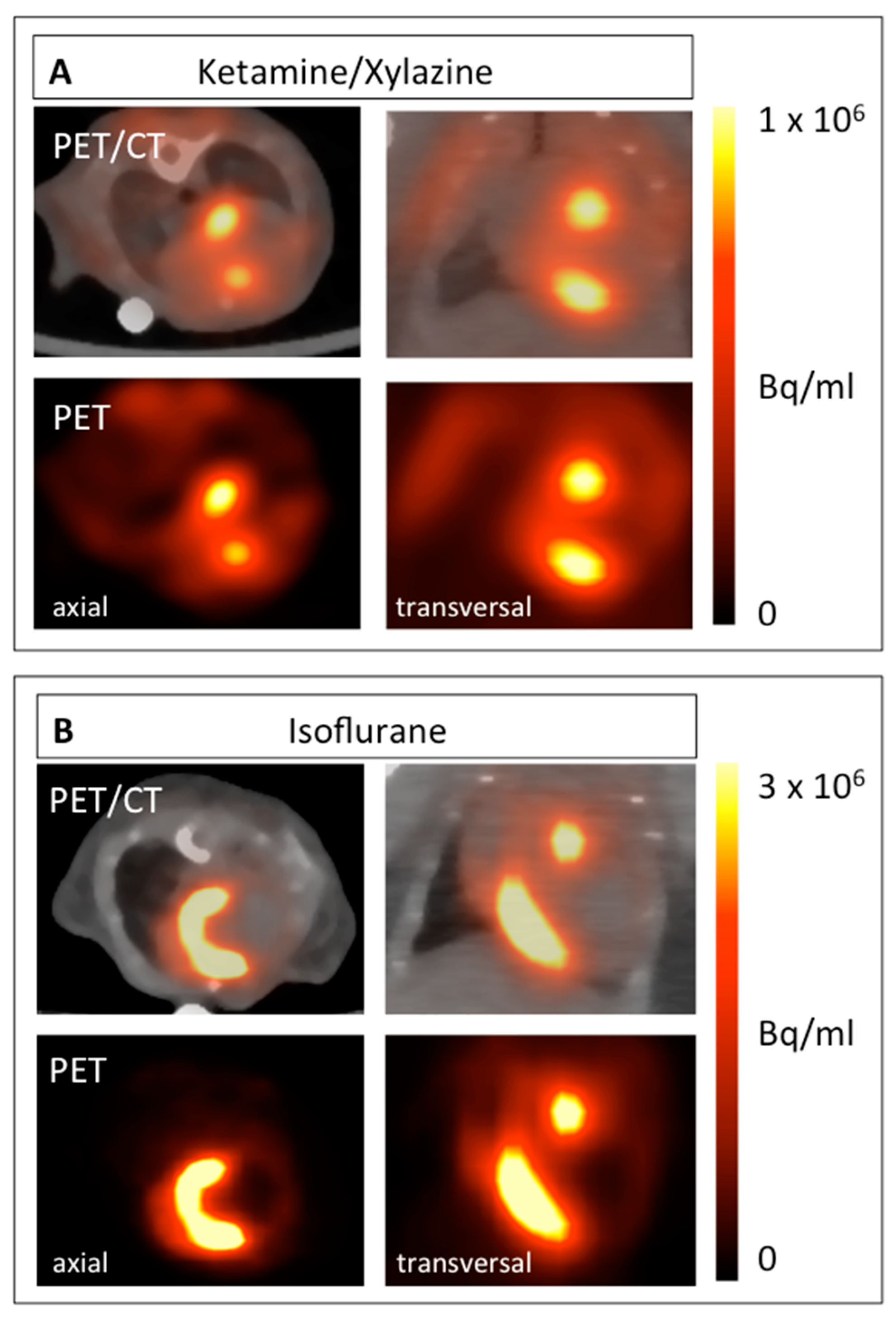
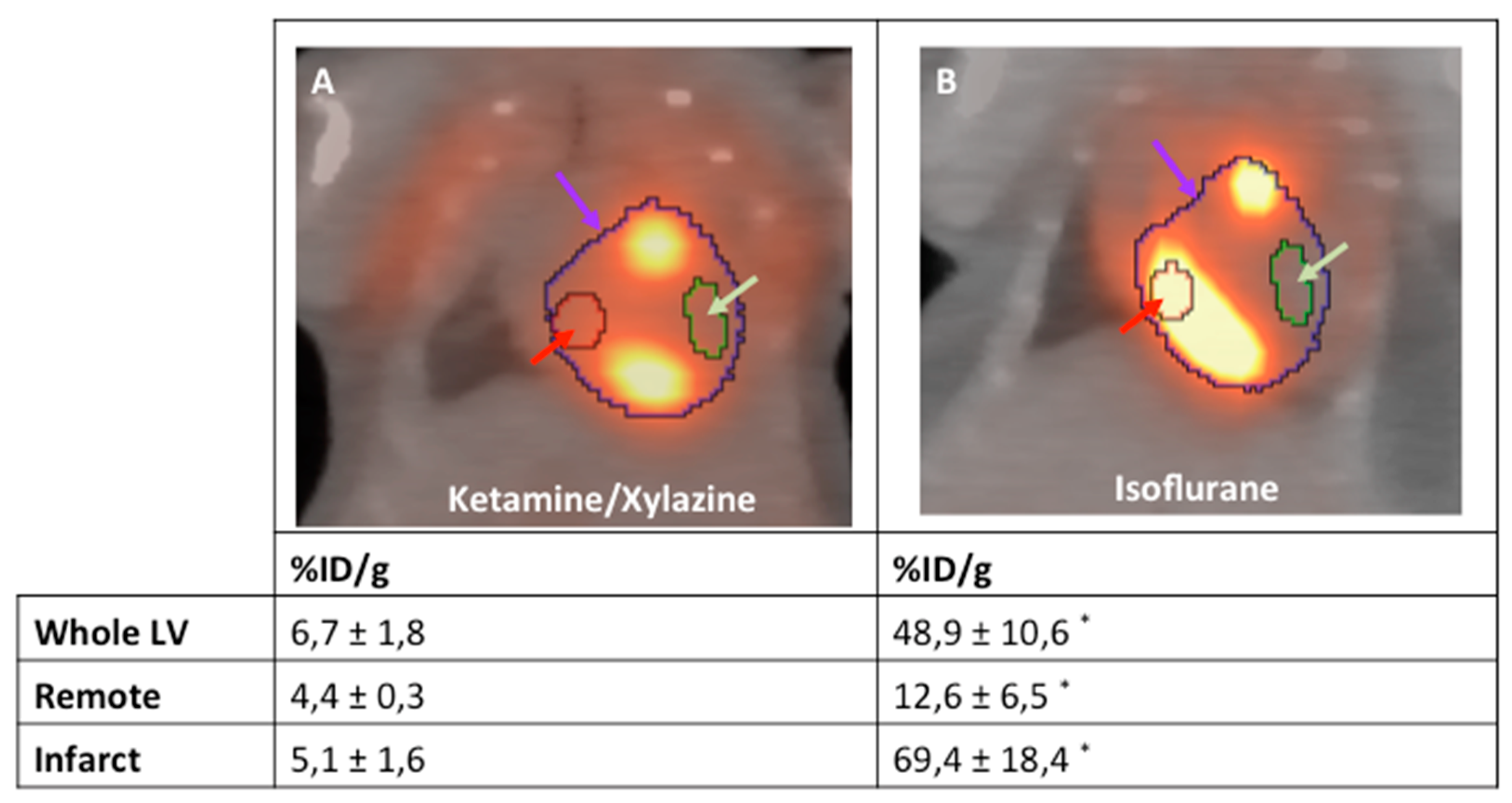
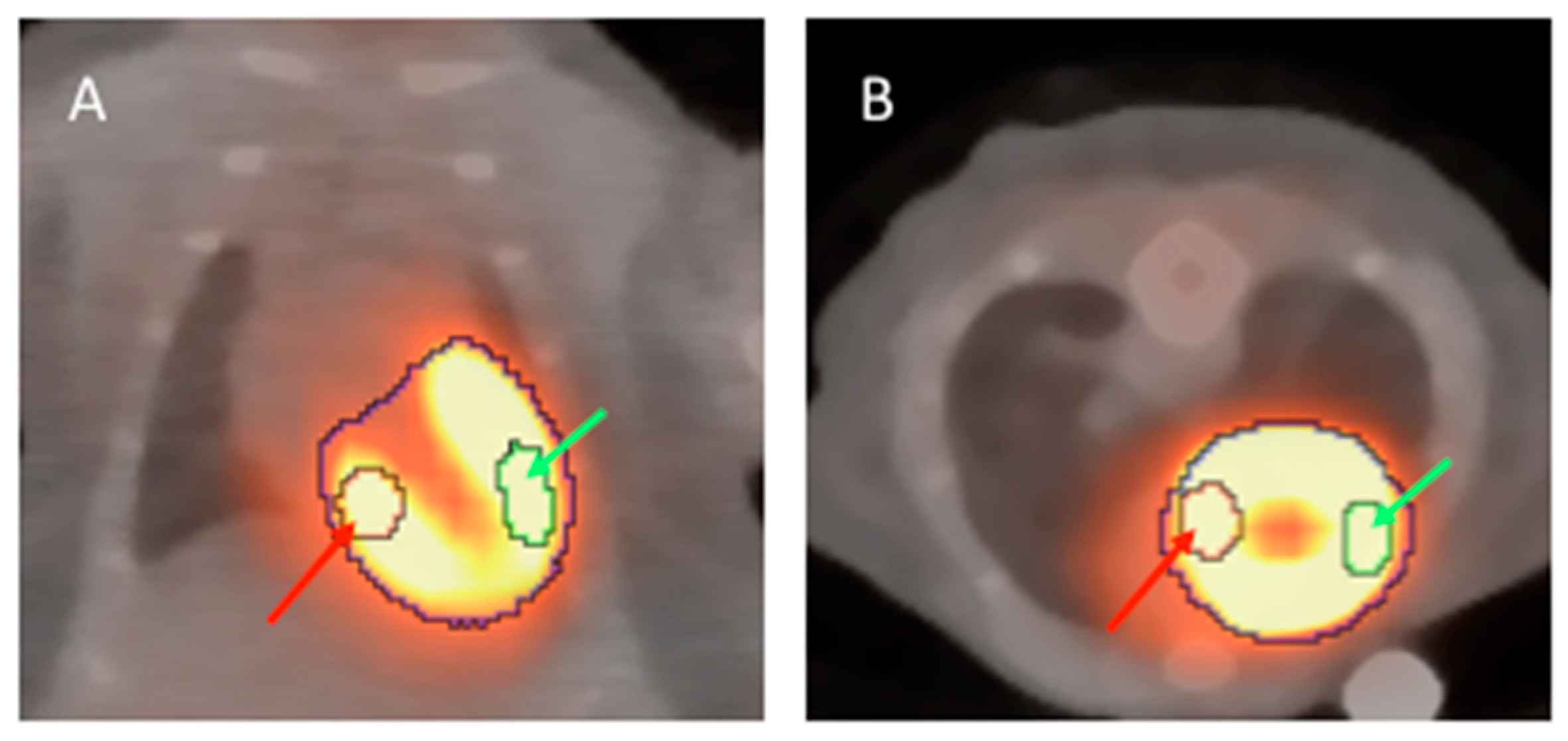
2. Results
3. Discussion
4. Limitations
5. Materials and Methods
5.1. Animal Model
5.2. PET Imaging
5.3. PET Image Analysis
5.4. Statistics
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| FDG | fluorodeoxyglucose |
| PET | positron emission tomography |
| KX | ketamine/xylazine |
| CT | computer tomography |
| VOI | volume of interest |
| AMI | acute myocardial infarction |
| MI | myocardial infarction |
| %ID/g | % of injected dose per gram |
| SD | standard deviation |
| LV | left ventricle |
| GLUT | glucose transporter |
References
- Knuuti, J.; Wijns, W.; Saraste, A.; Capodanno, D.; Barbato, E.; Funck-Brentano, C.; Prescott, E.; Storey, R.F.; Deaton, C.; Cuisset, T.; et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur. Heart J. 2020, 41, 407–477. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Mensah, G.A.; Croft, J.B.; Keenan, N.L. Heart failure-related hospitalization in the U.S., 1979 to 2004. J. Am. Coll. Cardiol. 2008, 52, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Seropian, I.M.; Toldo, S.; Van Tassell, B.W.; Abbate, A. Anti-inflammatory strategies for ventricular remodeling following ST-segment elevation acute myocardial infarction. J. Am. Coll. Cardiol. 2014, 63, 1593–1603. [Google Scholar] [CrossRef] [PubMed]
- Van Hout, G.P.; Jansen of Lorkeers, S.J.; Wever, K.E.; Sena, E.S.; Kouwenberg, L.H.; van Solinge, W.W.; Macleod, M.R.; Doevendans, P.A.; Pasterkamp, G.; Chamuleau, S.A.; et al. Translational failure of anti-inflammatory compounds for myocardial infarction: A meta-analysis of large animal models. Cardiovasc. Res. 2016, 109, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.W.; Marinelli, B.; van der Laan, A.M.; Sena, B.F.; Gorbatov, R.; Leuschner, F.; Dutta, P.; Iwamoto, Y.; Ueno, T.; Begieneman, M.P.; et al. PET/MRI of inflammation in myocardial infarction. J. Am. Coll. Cardiol. 2012, 59, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Nahrendorf, M.; Swirski, F.K.; Aikawa, E.; Stangenberg, L.; Wurdinger, T.; Figueiredo, J.L.; Libby, P.; Weissleder, R.; Pittet, M.J. The healing myocardium sequentially mobilizes two monocyte subsets with divergent and complementary functions. J. Exp. Med. 2007, 204, 3037–3047. [Google Scholar] [CrossRef] [PubMed]
- Love, C.; Tomas, M.B.; Tronco, G.G.; Palestro, C.J. FDG PET of infection and inflammation. Radiographics 2005, 25, 1357–1368. [Google Scholar] [CrossRef] [PubMed]
- Rischpler, C.; Dirschinger, R.J.; Nekolla, S.G.; Kossmann, H.; Nicolosi, S.; Hanus, F.; van Marwick, S.; Kunze, K.P.; Meinicke, A.; Gotze, K.; et al. Prospective Evaluation of 18F-Fluorodeoxyglucose Uptake in Postischemic Myocardium by Simultaneous Positron Emission Tomography/Magnetic Resonance Imaging as a Prognostic Marker of Functional Outcome. Circ. Cardiovasc. Imaging 2016, 9, e004316. [Google Scholar] [CrossRef] [PubMed]
- Thackeray, J.T.; Bankstahl, J.P.; Wang, Y.; Wollert, K.C.; Bengel, F.M. Clinically relevant strategies for lowering cardiomyocyte glucose uptake for 18F-FDG imaging of myocardial inflammation in mice. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Thackeray, J.T.; Bankstahl, J.P.; Wang, Y.; Korf-Klingebiel, M.; Walte, A.; Wittneben, A.; Wollert, K.C.; Bengel, F.M. Targeting post-infarct inflammation by PET imaging: Comparison of (68)Ga-citrate and (68)Ga-DOTATATE with (18)F-FDG in a mouse model. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Vasudevan, P.; Gaebel, R.; Doering, P.; Mueller, P.; Lemcke, H.; Stenzel, J.; Lindner, T.; Kurth, J.; Steinhoff, G.; Vollmar, B.; et al. 18F-FDG PET-Based Imaging of Myocardial Inflammation Predicts a Functional Outcome Following Transplantation of mESC-Derived Cardiac Induced Cells in a Mouse Model of Myocardial Infarction. Cells 2019, 8, 1613. [Google Scholar] [CrossRef] [PubMed]
- Saha, J.K.; Xia, J.; Grondin, J.M.; Engle, S.K.; Jakubowski, J.A. Acute hyperglycemia induced by ketamine/xylazine anesthesia in rats: Mechanisms and implications for preclinical models. Exp. Biol. Med. (Maywood) 2005, 230, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Kansara, M.; Berridge, M.V. Acute regulation of glucose transport in a monocyte-macrophage cell line: Glut-3 affinity for glucose is enhanced during the respiratory burst. Biochem. J. 1997, 327 Pt 2, 369–375. [Google Scholar] [CrossRef]
- Todica, A.; Beetz, N.L.; Gunther, L.; Zacherl, M.J.; Grabmaier, U.; Huber, B.; Bartenstein, P.; Brunner, S.; Lehner, S. Monitoring of Cardiac Remodeling in a Mouse Model of Pressure-Overload Left Ventricular Hypertrophy with [(18)F]FDG MicroPET. Mol. Imaging Biol. 2018, 20, 268–274. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasudevan, P.; Gäbel, R.; Stenzel, J.; Förster, J.; Kurth, J.; Vollmar, B.; Krause, B.J.; Ince, H.; David, R.; Lang, C.I. 18F-FDG PET-Based Imaging of Myocardial Inflammation Following Acute Myocardial Infarction in a Mouse Model. Int. J. Mol. Sci. 2020, 21, 3340. https://doi.org/10.3390/ijms21093340
Vasudevan P, Gäbel R, Stenzel J, Förster J, Kurth J, Vollmar B, Krause BJ, Ince H, David R, Lang CI. 18F-FDG PET-Based Imaging of Myocardial Inflammation Following Acute Myocardial Infarction in a Mouse Model. International Journal of Molecular Sciences. 2020; 21(9):3340. https://doi.org/10.3390/ijms21093340
Chicago/Turabian StyleVasudevan, Praveen, Ralf Gäbel, Jan Stenzel, Joanna Förster, Jens Kurth, Brigitte Vollmar, Bernd Joachim Krause, Hüseyin Ince, Robert David, and Cajetan Immanuel Lang. 2020. "18F-FDG PET-Based Imaging of Myocardial Inflammation Following Acute Myocardial Infarction in a Mouse Model" International Journal of Molecular Sciences 21, no. 9: 3340. https://doi.org/10.3390/ijms21093340
APA StyleVasudevan, P., Gäbel, R., Stenzel, J., Förster, J., Kurth, J., Vollmar, B., Krause, B. J., Ince, H., David, R., & Lang, C. I. (2020). 18F-FDG PET-Based Imaging of Myocardial Inflammation Following Acute Myocardial Infarction in a Mouse Model. International Journal of Molecular Sciences, 21(9), 3340. https://doi.org/10.3390/ijms21093340




