Short-Term Physiological Effects of a Very Low-Calorie Ketogenic Diet: Effects on Adiponectin Levels and Inflammatory States
Abstract
1. Introduction
2. Results
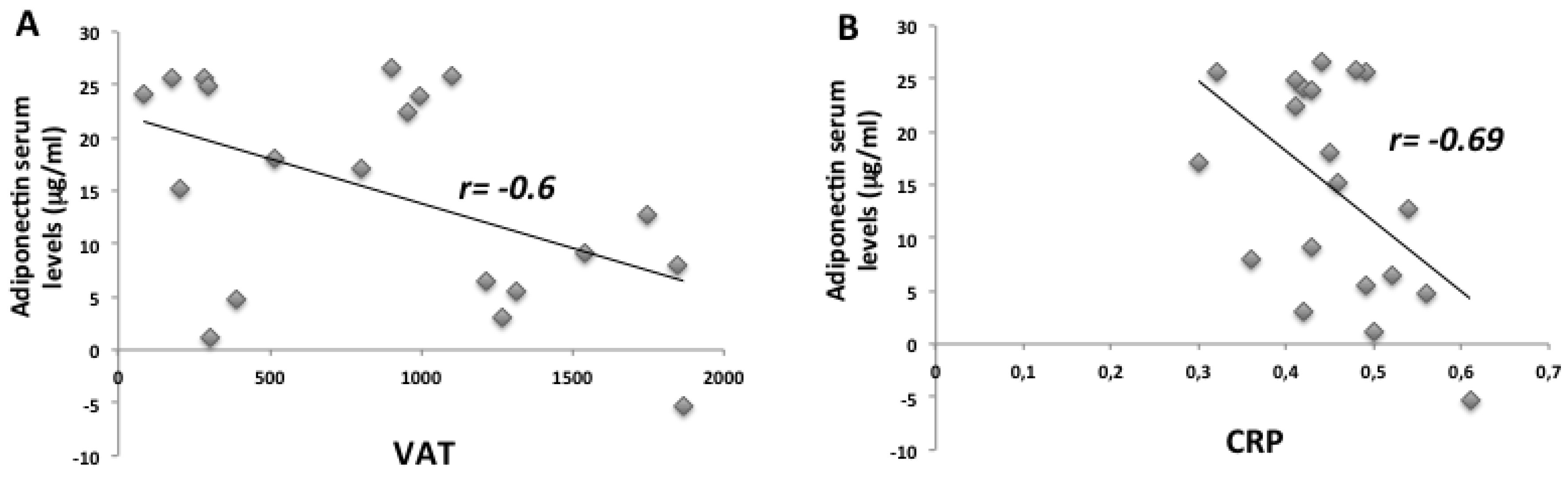
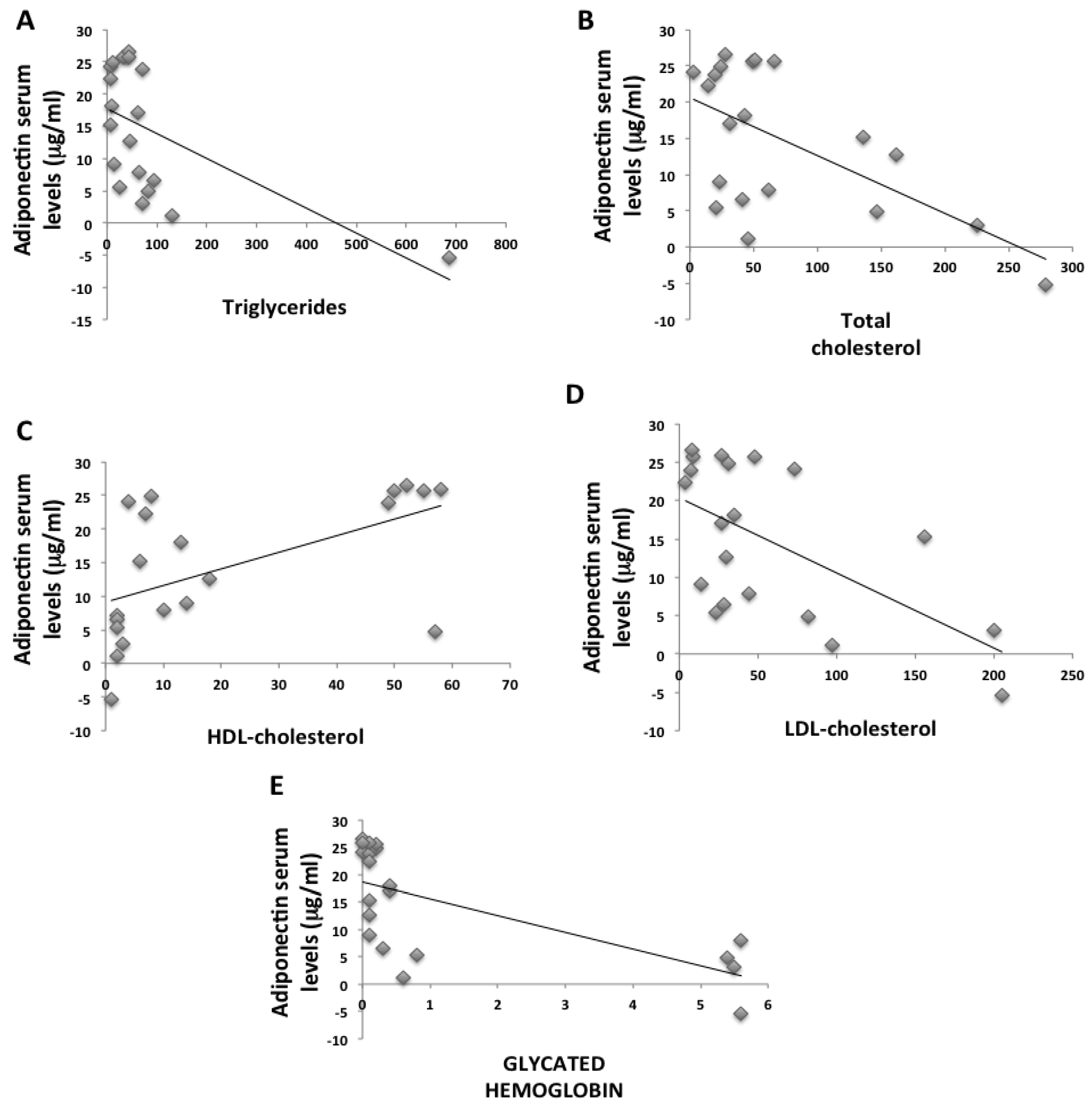
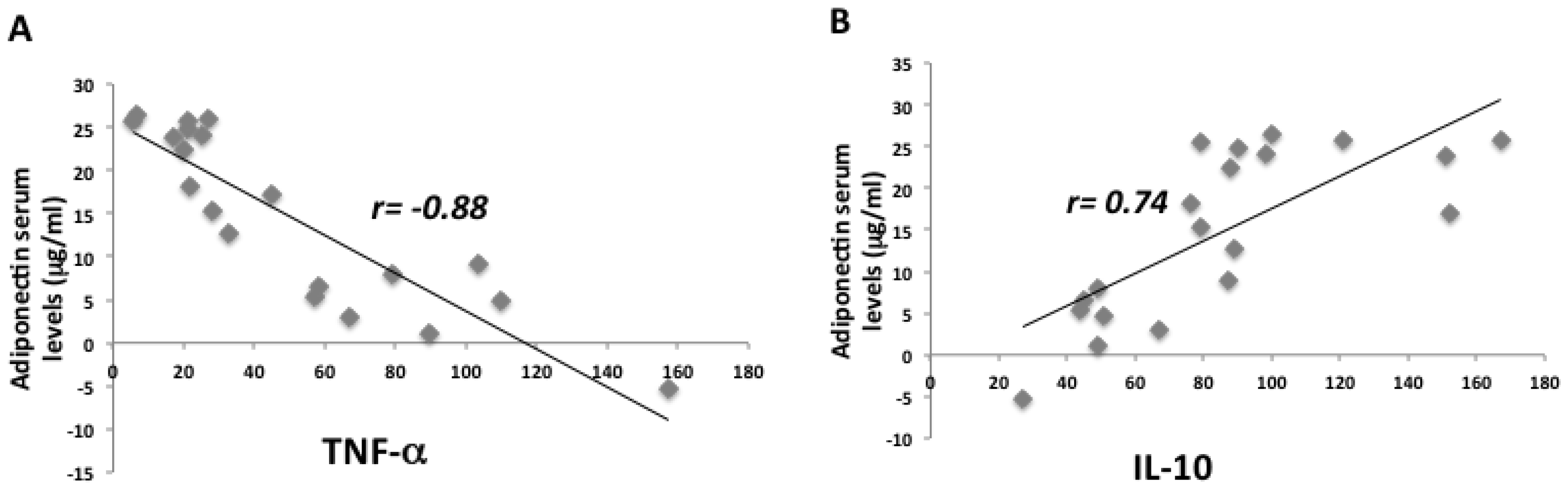
2.1. Anthropometric and Biochemical Futures of VLCKD Obese Patients
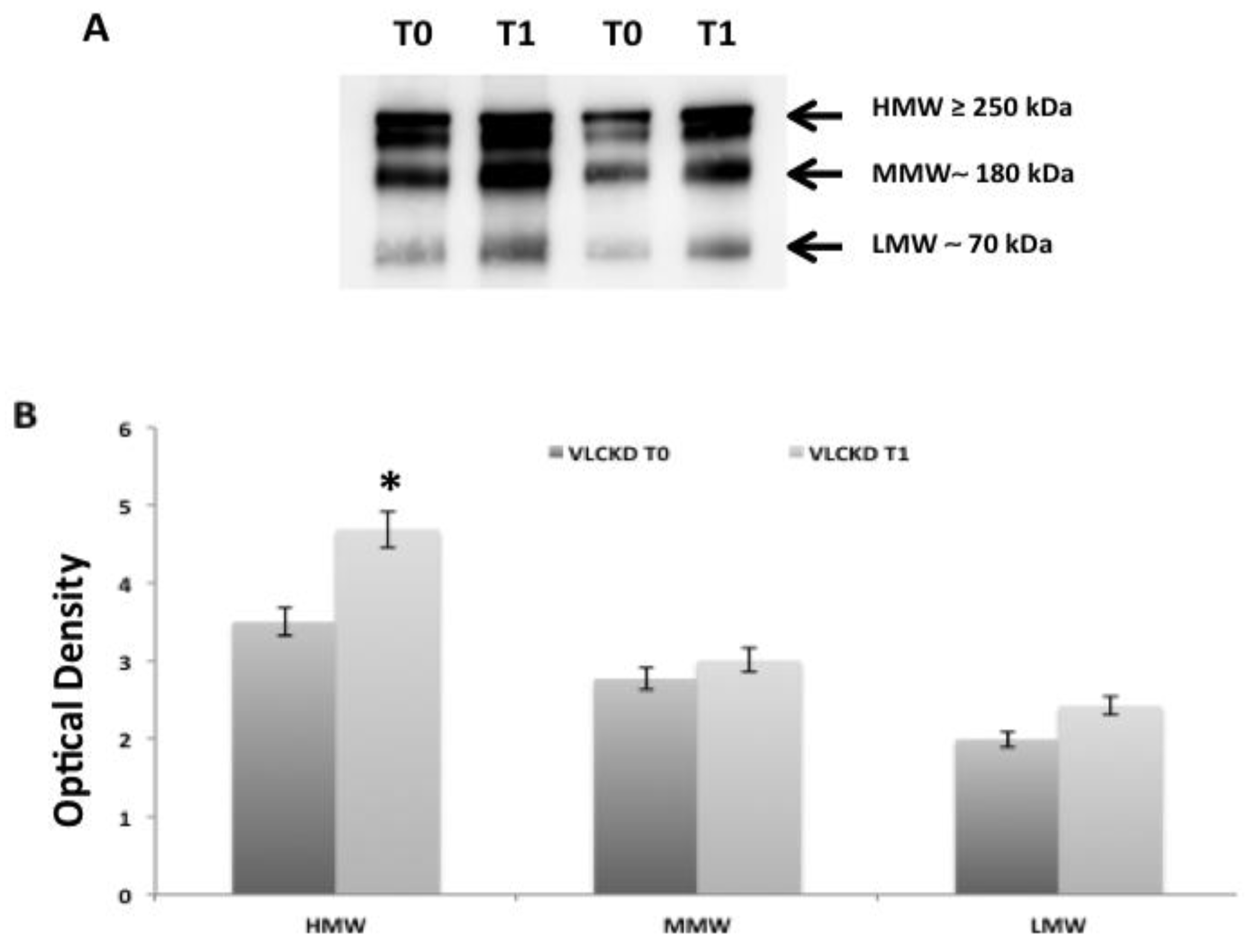
2.2. Adiponectin Oligomerization State Analysis in Serum by Western Blotting Ting
3. Discussion
4. Materials and Methods
4.1. Participants
4.2. Study Protocol
4.3. Anthropometric Measurements
4.4. Biochemical Parameters
4.5. Western Blotting Ting Analysis
4.6. Diet Intervention
4.7. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Versini, M.; Jeandel, P.-Y.; Rosenthal, E.; Shoenfeld, Y. Obesity in autoimmune diseases: Not a passive bystander. Autoimmun. Rev. 2014, 13, 981–1000. [Google Scholar] [CrossRef] [PubMed]
- Manzel, A.; Müller, D.N.; Hafler, D.A.; Erdman, S.E.; Linker, R.A.; Kleinewietfeld, M. Role of “Western diet” in inflammatory autoimmune diseases. Curr. Allergy Asthma Rep. 2014, 14, 404. [Google Scholar] [CrossRef] [PubMed]
- Nigro, E.; Scudiero, O.; Monaco, M.L.; Palmieri, A.; Mazzarella, G.; Costagliola, C.; Bianco, A.; Daniele, A. New Insight into Adiponectin Role in Obesity and Obesity-Related Diseases. BioMed Res. Int. 2014, 2014, 1–14. [Google Scholar] [CrossRef]
- Fernández-Real, J.M.; Botas-Cervero, P.; López-Bermano, A.; Casamitjana, R.; Funahashi, T.; Delgado, E.; Kihara, S.; Ricart-Engel, W. Adiponectin is independently associated with glycosylated haemoglobin. Eur. J. Endocrinol. 2004, 150, 201–205. [Google Scholar] [CrossRef][Green Version]
- Yamauchi, T.; Kamon, J.; Ito, Y.; Tsuchida, A.; Yokomizo, T.; Kita, S.; Sugiyama, T.; Miyagishi, M.; Hara, K.; Tsunoda, M.; et al. Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature 2003, 423, 762–769. [Google Scholar] [CrossRef]
- Abraham, P.A.; Attipoe, S.; Kazman, J.B.; Zeno, S.A.; Poth, M.; Deuster, P.A. Role of plasma adiponectin /C-reactive protein ratio in obesity and type 2 diabetes among African Americans. Afr. Health Sci. 2017, 17, 99–107. [Google Scholar] [CrossRef][Green Version]
- Smekal, A.; Vaclavik, J. Adipokines and cardiovascular disease: A comprehensive review. Biomed. Pap. 2017, 161, 31–40. [Google Scholar] [CrossRef]
- Scheja, L.; Heeren, J. The endocrine function of adipose tissues in health and cardiometabolic disease. Nat. Rev. Endocrinol. 2019, 15, 507–524. [Google Scholar] [CrossRef]
- Singh, M.; Benencia, F. Inflammatory processes in obesity: Focus on endothelial dysfunction and the role of adipokines as inflammatory mediators. Int. Rev. Immunol. 2019, 38, 157–171. [Google Scholar] [CrossRef]
- Polito, R.; Costabile, G.; Nigro, E.; Giacco, R.; Vetrani, C.; Anniballi, G.; Luongo, D.; Riccardi, G.; Daniele, A.; Annuzzi, G. Nutritional factors influencing plasma adiponectin levels: Results from a randomised controlled study with whole-grain cereals. Int. J. Food Sci. Nutr. 2019, 22, 1–7. [Google Scholar] [CrossRef]
- Kosinski, C.; Jornayvaz, F.R. Effects of Ketogenic Diets on Cardiovascular Risk Factors: Evidence from Animal and Human Studies. Nutrients 2017, 9, 517. [Google Scholar] [CrossRef] [PubMed]
- Boison, D. New insights into the mechanisms of the ketogenic diet. Curr. Opin. Neurol. 2017, 30, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Veech, R. The therapeutic implications of ketone bodies: The effects of ketone bodies in pathological conditions: Ketosis, ketogenic diet, redox states, insulin resistance, and mitochondrial metabolism. Prostaglandins Leukot. Essent. Fat. Acids 2004, 70, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Benlloch, M.; López-Rodríguez, M.M.; Cuerda-Ballester, M.; Drehmer, E.; Carrera, S.; Cerón, J.J.; Tvarijonaviciute, A.; Chirivella, J.; Fernandez-Garcia, D.; Ortí, J.E.D.L.R. Satiating Effect of a Ketogenic Diet and Its Impact on Muscle Improvement and Oxidation State in Multiple Sclerosis Patients. Nutrients 2019, 11, 1156. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.D.; Aminzadeh-Gohari, S.; Tulipan, J.; Catalano, L.; Feichtinger, R.G.; Kofler, B. Ketogenic diet in the treatment of cancer—Where do we stand? Mol. Metab. 2020, 33, 102–121. [Google Scholar] [CrossRef]
- Dąbek, A.; Wojtala, M.; Pirola, L.; Balcerczyk, A. Modulation of Cellular Biochemistry, Epigenetics and Metabolomics by Ketone Bodies. Implications of the Ketogenic Diet in the Physiology of the Organism and Pathological States. Nutrients 2020, 12, 788. [Google Scholar] [CrossRef]
- O’Neill, B.; Raggi, P. The ketogenic diet: Pros and cons. Atherosclerosis 2019, 292, 119–126. [Google Scholar] [CrossRef]
- Gershuni, V.M.; Yan, S.L.; Medici, V. Nutritional Ketosis for Weight Management and Reversal of Metabolic Syndrome. Curr. Nutr. Rep. 2018, 7, 97–106. [Google Scholar] [CrossRef]
- Puchalska, P.; Crawford, P. Multi-dimensional Roles of Ketone Bodies in Fuel Metabolism, Signaling, and Therapeutics. Cell Metab. 2017, 25, 262–284. [Google Scholar] [CrossRef]
- Castellana, M.; Conte, E.; Cignarelli, A.; Perrini, S.; Giustina, A.; Giovanella, L.; Giorgino, F.; Trimboli, P. Efficacy and safety of very low calorie ketogenic diet (VLCKD) in patients with overweight and obesity: A systematic review and meta-analysis. Rev. Endocr. Metab. Disord. 2019, 21, 5–16. [Google Scholar] [CrossRef]
- Zhang, Q.; Xu, L.; Xia, J.; Wang, N.; Qian, M.; Ding, S. Treatment of Diabetic Mice with a Combination of Ketogenic Diet and Aerobic Exercise via Modulations of PPARs Gene Programs. PPAR Res. 2018, 2018, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A. Ketogenic Diet for Obesity: Friend or Foe? Int. J. Environ. Res. Public Health 2014, 11, 2092–2107. [Google Scholar] [CrossRef] [PubMed]
- Ellenbroek, J.H.; Van Dijck, L.; Töns, H.A.; Rabelink, T.J.; Carlotti, F.; Ballieux, B.E.P.B.; De Koning, E.J.P. Long-term ketogenic diet causes glucose intolerance and reduced β- and α-cell mass but no weight loss in mice. Am. J. Physiol. Metab. 2014, 306, E552–E558. [Google Scholar] [CrossRef] [PubMed]
- Steven, S.; Hollingsworth, K.G.; Al-Mrabeh, A.; Avery, L.; Aribisala, B.; Caslake, M.; Taylor, R. Very Low-Calorie Diet and 6 Months of Weight Stability in Type 2 Diabetes: Pathophysiological Changes in Responders and Nonresponders. Diabetes Care 2016, 39, 808–815. [Google Scholar] [CrossRef]
- Gomez-Arbelaez, D.; Crujeiras, A.B.; Castro, A.I.; Goday, A.; Mas-Lorenzo, A.; Bellon, A.; Tejera, C.; Bellido, D.; Galban, C.; Sajoux, I.; et al. Acid–base safety during the course of a very low-calorie-ketogenic diet. Endocrine 2017, 58, 81–90. [Google Scholar] [CrossRef]
- Bergman, R.N.; Kim, S.P.; Catalano, K.J.; Hsu, I.R.; Chiu, J.D.; Kabir, M.; Hücking, K.; Ader, M. Why Visceral Fat is Bad: Mechanisms of the Metabolic Syndrome. Obesity 2006, 14, 16S–19S. [Google Scholar] [CrossRef]
- Gustafson, B.; Hammarstedt, A.; Andersson, C.X.; Smith, U. Inflamed Adipose Tissue. Arter. Thromb. Vasc. Boil. 2007, 27, 2276–2283. [Google Scholar] [CrossRef]
- Nigro, E.; Scudiero, O.; Monaco, M.L.; Polito, R.; Schettino, P.; Grandone, A.; Perrone, L.; Del Giudice, E.M.; Daniele, A. Adiponectin profile and Irisin expression in Italian obese children: Association with insulin-resistance. Cytokine 2017, 94, 8–13. [Google Scholar] [CrossRef]
- Okoro, N.; Okpara, H.; Azinge, E.; Builders, M. Serum Total Adiponectin and its Relationship with Glycemic Control ad Markers of Lipoprotein Metabolism in Nigerians with Type 2 Diabetes Mellitus. Ann. Med. Health Sci. Res. 2019, 9, 478–483. [Google Scholar]
- Berg, A.H.; Combs, T.P.; DU, X.; Brownlee, M.; Scherer, P.E. The adypocyte-secreted protein Acrp30 enhances hepatic insulin action. Nat. Med. 2001, 7, 947–953. [Google Scholar] [CrossRef]
- Yamauchi, T.; Kamon, J.; Waki, H.; Terauchi, Y.; Kubota, N.; Hara, K.; Mori, Y.; Ide, T.; Murakami, K.; Tsuboyama-Kasaoka, N.; et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat. Med. 2001, 7, 941–946. [Google Scholar] [CrossRef] [PubMed]
- Bacha, F.; Saad, R.; Gungor, N.; A Arslanian, S. Adiponectin in youth: Relationship to visceral adiposity, insulin sensitivity, and beta-cell function. Diabetes Care 2004, 27, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Mohorko, N.; Černelič-Bizjak, M.; Vatovec, T.P.; Grom, G.; Kenig, S.; Petelin, A.; Jenko-Pražnikar, Z. Weight loss, improved physical performance, cognitive function, eating behavior, and metabolic profile in a 12-week ketogenic diet in obese adults. Nutr. Res. 2018, 62, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Miller, V.; Villamena, F.A.; Volek, J.S. Nutritional Ketosis and Mitohormesis: Potential Implications for Mitochondrial Function and Human Health. J. Nutr. Metab. 2018, 2018, 1–27. [Google Scholar] [CrossRef]
- Sherrier, M.; Li, H. The impact of keto-adaptation on exercise performance and the role of metabolic-regulating cytokines. Am. J. Clin. Nutr. 2019, 110, 562–573. [Google Scholar] [CrossRef]
- Makki, K.; Froguel, P.; Wolowczuk, I. Adipose Tissue in Obesity-Related Inflammation and Insulin Resistance: Cells, Cytokines, and Chemokines. ISRN Inflamm. 2013, 2013, 1–12. [Google Scholar] [CrossRef]
- Garaulet, M.; Viguerie, N.; Porubsky, S.; Klimcakova, E.; Clément, K.; Langin, D.; Štich, V. Adiponectin Gene Expression and Plasma Values in Obese Women during Very-Low-Calorie Diet. Relationship with Cardiovascular Risk Factors and Insulin Resistance. J. Clin. Endocrinol. Metab. 2004, 89, 756–760. [Google Scholar] [CrossRef]
- Awazawa, M.; Ueki, K.; Inabe, K.; Yamauchi, T.; Kubota, N.; Kaneko, K.; Kobayashi, M.; Iwane, A.; Sasako, T.; Okazaki, Y.; et al. Adiponectin Enhances Insulin Sensitivity by Increasing Hepatic IRS-2 Expression via a Macrophage-Derived IL-6-Dependent Pathway. Cell Metab. 2011, 13, 401–412. [Google Scholar] [CrossRef]
- Nigro, E.; Scudiero, O.; Sarnataro, D.; Mazzarella, G.; Sofia, M.; Bianco, A.; Daniele, A. Adiponectin affects lung epithelial A549 cell viability counteracting TNFa and IL-1ß toxicity through AdipoR1. Int. J. Biochem. Cell Boil. 2013, 45, 1145–1153. [Google Scholar] [CrossRef]
- Chinetti, G.; Zawadski, C.; Fruchart, J.; Staels, B. Expression of adiponectin receptors in human macrophages and regulation by agonists of the nuclear receptors PPARα, PPARγ, and LXR. Biochem. Biophys. Res. Commun. 2004, 314, 151–158. [Google Scholar] [CrossRef]
- Shih, R.-H.; Wang, C.-Y.; Yang, C.-M. NF-kappaB Signaling Pathways in Neurological Inflammation: A Mini Review. Front. Mol. Neurosci. 2015, 8, 3244. [Google Scholar] [CrossRef] [PubMed]
- Hui, X.; Gu, P.; Zhang, J.; Nie, T.; Pan, Y.; Wu, N.; Feng, T.; Zhong, C.; Wang, Y.; Lam, K.S.; et al. Adiponectin Enhances Cold-Induced Browning of Subcutaneous Adipose Tissue via Promoting M2 Macrophage Proliferation. Cell Metab. 2015, 22, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Polito, R.; Nigro, E.; Messina, A.; Monaco, M.L.; Monda, V.; Scudiero, O.; Cibelli, G.; Valenzano, A.; Picciocchi, E.; Zammit, C.; et al. Adiponectin and Orexin-A as a Potential Immunity Link Between Adipose Tissue and Central Nervous System. Front. Physiol. 2018, 9, 982. [Google Scholar] [CrossRef] [PubMed]
- Micklesfield, L.; Goedecke, J.H.; Punyanitya, M.; Wilson, K.E.; Kelly, T.L. Dual-Energy X-Ray Performs as Well as Clinical Computed Tomography for the Measurement of Visceral Fat. Obesity 2012, 20, 1109–1114. [Google Scholar] [CrossRef]
- Tsuchida, A.; Yamauchi, T.; Takekawa, S.; Hada, Y.; Ito, Y.; Maki, T.; Kadowaki, T. Peroxisome Proliferator-Activated Receptor (PPAR) Activation Increases Adiponectin Receptors and Reduces Obesity-Related Inflammation in Adipose Tissue: Comparison of Activation of PPAR, PPAR, and Their Combination. Diabetes 2005, 54, 3358–3370. [Google Scholar] [CrossRef]
- Available online: http://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi (accessed on 1 December 2019).
- Valenzano, A.; Polito, R.; Trimigno, V.; Di Palma, A.; Moscatelli, F.; Corso, G.; Sessa, F.; Salerno, M.; Montana, A.; Di Nunno, N.; et al. Effects of Very Low Calorie Ketogenic Diet on the Orexinergic System, Visceral Adipose Tissue, and ROS Production. Antioxidants 2019, 8, 643. [Google Scholar] [CrossRef]
- SCOOP-VLCD Task 7.3 Reports on Tasks for Scientific Cooperation. Collection of data on products intended for use in very-low calorie- diets. In Proceedings of the Report Brussels European Commission, Brussels, Belgium, 13 September 2002.
- Daniele, A.; De Rosa, A.; Nigro, E.; Scudiero, O.; Capasso, M.; Masullo, M.; De Laurentiis, G.; Oriani, G.; Sofia, M.; Bianco, A. Adiponectin oligomerization state and adiponectin receptors airway expression in chronic obstructive pulmonary disease. Int. J. Biochem. Cell Boil. 2012, 44, 563–569. [Google Scholar] [CrossRef]
| VLCKD Obese Participants | p-Value | ||
|---|---|---|---|
| T0 | T1 | ||
| Sex male/female | 10/10 | 10/10 | |
| Age | 48 ± 8.2 | - | ns |
| Height (m) | 1.67 ± 0.1 | ns | |
| Weight (kg) | 91.33 ± 17.1 | 78.73 ± 13.3 | <0.001 |
| BMI (kg/m²) | 32.19 ± 4.78 | 27.76 ± 3.6 | <0.001 |
| VAT (g) | 1541.55 ± 141.6 | 927.79 ± 104.9 | <0.001 |
| FM (g) | 39,208.77 ± 1432.5 | 27,377.03 ± 1217.4 | <0.001 |
| FFM (g) | 48,789.57 ± 1712.3 | 48,093.68 ± 1670.6 | ns |
| BMD | 1225.57 ± 21.2 | 1229.31 ± 21.4 | ns |
| Total cholesterol (mg/dL) | 220.13 ± 50.7 | 173.91 ± 32.9 | <0.05 |
| HDL (mg/dL) | 55.13 ± 11.1 | 47.76 ± 9.1 | ns |
| LDL (mg/dL) | 141.83 ± 36.4 | 107.57 ± 27.7 | <0.05 |
| Triglycerides (mg/dL) | 135.54 ± 125.2 | 83.25 ± 26.1 | <0.05 |
| Glycemia (mg/dL) | 96.68 ± 4.6 | 93.09 ± 3.3 | <0.05 |
| HGB (g/dL) | 14.13 ± 1,3 | 13.83 ± 0.9 | ns |
| Hba1c (%) | 5.65 ± 0.3 | 5.38 ± 0.3 | ns |
| Insulinemia (μU/mL) | 10.53 ± 7.1 | 5.37 ± 3.7 | <0.05 |
| Uric acid (mg/dL) | 4.86 ± 1.0 | 5.27 ± 1.1 | ns |
| Total protein (g/dL) | 7.30 ± 0.4 | 7.13 ± 0.4 | ns |
| AST-GOT (U/L) | 21.27 ± 5.9 | 23.31 ± 11.4 | <0.05 |
| ALT-GPT (U/L) | 26.51 ± 14.8 | 26.06 ± 16.2 | <0.05 |
| Gamma GT (U/L) | 31.19 ± 19.8 | 15.31 ± 5.4 | <0.05 |
| CRP (mg/mL) | 0.89 ± 0.1 | 0.48 ± 0.1 | <0.05 |
| Adiponectin (μg/mL) | 10.8 ± 1.2 | 25.55 ± 1.3 | <0.001 |
| TNF-α (pg/mL) | 345 ± 6.5 | 278 ± 9.2 | <0.05 |
| IL-10 (pg/mL) | 117 ± 7 | 168 ± 8.8 | <0.001 |
| IL-6 (pg/mL) | 236 ± 4.4 | 232 ± 5 | ns |
| Adiponectin Levels (μg/mL) | p-Value | ||
|---|---|---|---|
| Male | Female | ||
| VLCKD obese participants T0 | 9.23 ± 0.7 | 12.44 ± 1.07 | <0.05 |
| VLCKD obese participants T1 | 23.67 ± 1.6 | 27.3 ± 1.33 | <0.05 |
| Fats (%) | 43 |
| Proteins (%) | 43 |
| Carbohydrates | 14 |
| Carbohydrates from vegetables (g/day) | <50 |
| Total Kcal/day | 700–900 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monda, V.; Polito, R.; Lovino, A.; Finaldi, A.; Valenzano, A.; Nigro, E.; Corso, G.; Sessa, F.; Asmundo, A.; Di Nunno, N.; et al. Short-Term Physiological Effects of a Very Low-Calorie Ketogenic Diet: Effects on Adiponectin Levels and Inflammatory States. Int. J. Mol. Sci. 2020, 21, 3228. https://doi.org/10.3390/ijms21093228
Monda V, Polito R, Lovino A, Finaldi A, Valenzano A, Nigro E, Corso G, Sessa F, Asmundo A, Di Nunno N, et al. Short-Term Physiological Effects of a Very Low-Calorie Ketogenic Diet: Effects on Adiponectin Levels and Inflammatory States. International Journal of Molecular Sciences. 2020; 21(9):3228. https://doi.org/10.3390/ijms21093228
Chicago/Turabian StyleMonda, Vincenzo, Rita Polito, Annarita Lovino, Antonio Finaldi, Anna Valenzano, Ersilia Nigro, Gaetano Corso, Francesco Sessa, Alessio Asmundo, Nunzio Di Nunno, and et al. 2020. "Short-Term Physiological Effects of a Very Low-Calorie Ketogenic Diet: Effects on Adiponectin Levels and Inflammatory States" International Journal of Molecular Sciences 21, no. 9: 3228. https://doi.org/10.3390/ijms21093228
APA StyleMonda, V., Polito, R., Lovino, A., Finaldi, A., Valenzano, A., Nigro, E., Corso, G., Sessa, F., Asmundo, A., Di Nunno, N., Cibelli, G., & Messina, G. (2020). Short-Term Physiological Effects of a Very Low-Calorie Ketogenic Diet: Effects on Adiponectin Levels and Inflammatory States. International Journal of Molecular Sciences, 21(9), 3228. https://doi.org/10.3390/ijms21093228










