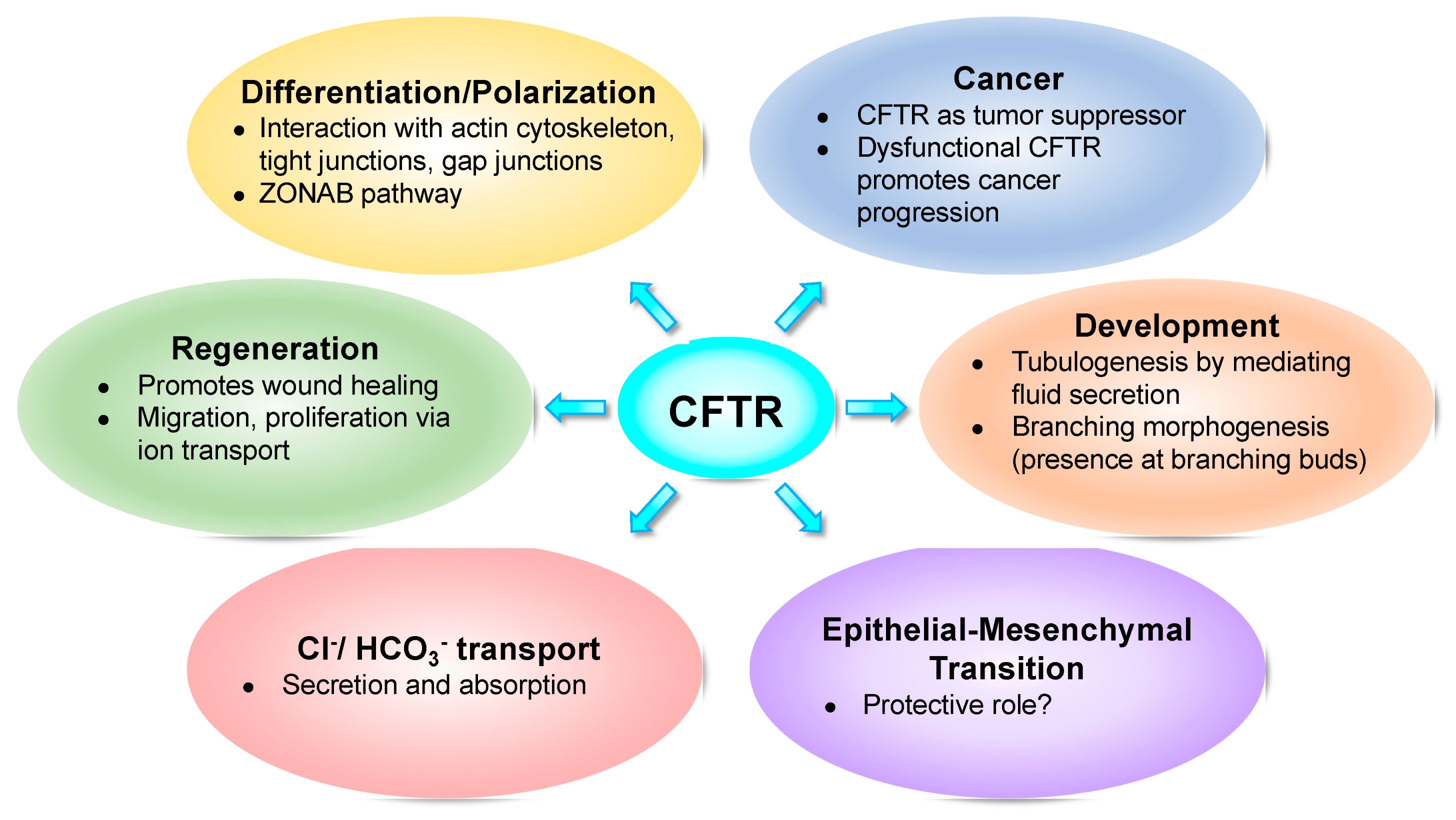
What Role Does CFTR Play in Development, Differentiation, Regeneration and Cancer?
Abstract
1. Introduction
2. CFTR and Development
2.1. CFTR Expression is Highly Regulated During Development
2.2. Individuals with CF Display Lung Malformations and CBAVD
2.3. What Is the Major Role of CFTR in Development?
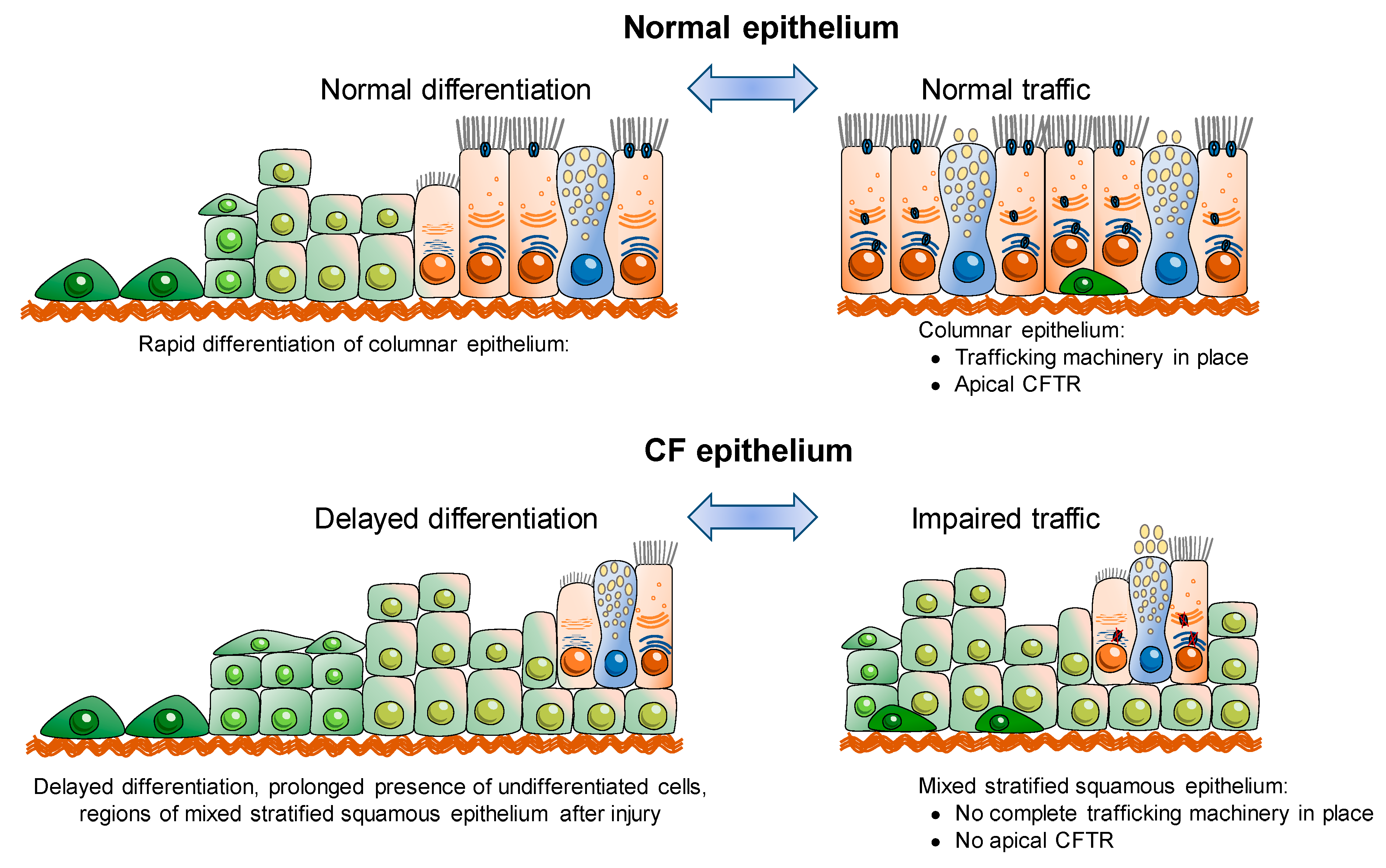
3. CFTR and Epithelial Differentiation
3.1. Is CFTR Essential for Epithelial Differentiation?
3.2. CFTR, Actin Cytoskeleton, Tight Junctions and Differentiation
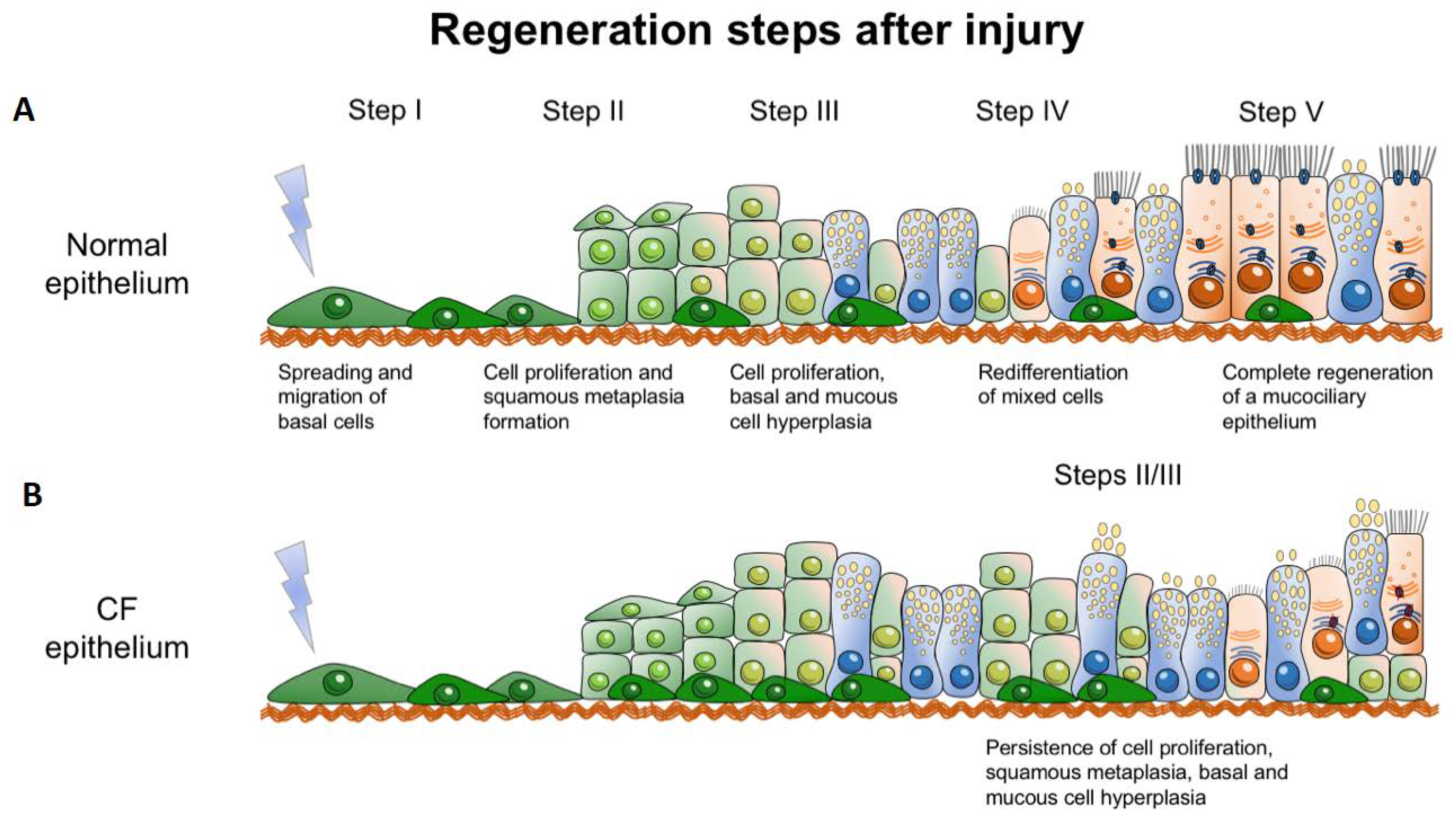
4. CFTR and Regeneration
4.1. CFTR is Needed for Proper Epithelial Regeneration
4.2. CFTR and Wound Healing as a Model of Physiological Regeneration
5. CFTR and Cancer
5.1. CF as a Disease of Increased Cancer Risk
5.2. CF Carrier Status and Cancer Risk
5.3. Is CFTR a Tumour Suppressor Protein?
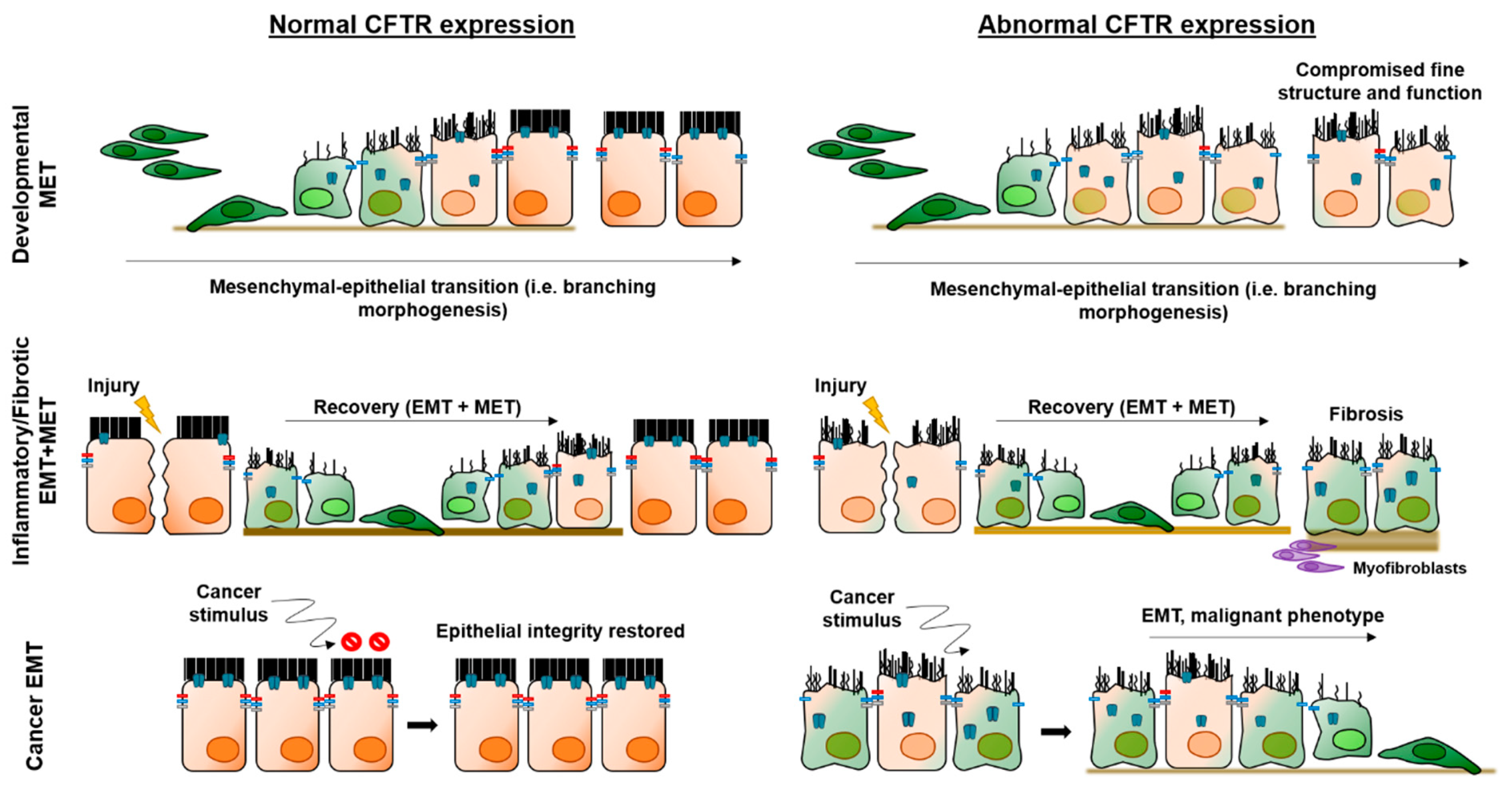
6. CFTR and Epithelial–Mesenchymal Transition (EMT)
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Neglia, J.P.; Fitzsimmons, S.C.; Maisonneuve, P.; Schöni, M.H.; Schöni-Affolter, F.; Corey, M.; Lowenfels, A.B. The risk of cancer among patients with cystic fibrosis. N. Engl. J. Med. 1995, 332, 494–499. [Google Scholar] [CrossRef] [PubMed]
- Maisonneuve, P.; Marshall, B.C.; Knapp, E.A.; Lowenfels, A.B. Cancer Risk in Cystic Fibrosis: A 20-Year Nationwide Study From the United States. JNCI J. Natl. Cancer Inst. 2013, 105, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.C.; Comellas, A.P.; Hornick, D.B.; Stoltz, D.A.; Cavanaugh, J.E.; Gerke, A.K.; Welsh, M.J.; Zabner, J.; Polgreen, P.M. Cystic fibrosis carriers are at increased risk for a wide range of cystic fibrosis-related conditions. Proc. Natl. Acad. Sci. USA 2020, 117, 1621–1627. [Google Scholar] [CrossRef] [PubMed]
- Larson, J.E.; Cohen, J.C. Developmental paradigm for early features of cystic fibrosis. Pediatr. Pulmonol. 2005, 40, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Craig Cohen, J.; Larson, J.E. Cystic fibrosis transmembrane conductance regulator (CFTR) dependent cytoskeletal tension during lung organogenesis. Dev. Dyn. 2006, 235, 2736–2748. [Google Scholar] [CrossRef]
- Meyerholz, D.K.; Stoltz, D.A.; Namati, E.; Ramachandran, S.; Pezzulo, A.A.; Smith, A.R.; Rector, M.V.; Suter, M.J.; Kao, S.; McLennan, G.; et al. Loss of Cystic Fibrosis Transmembrane Conductance Regulator Function Produces Abnormalities in Tracheal Development in Neonatal Pigs and Young Children. Am. J. Respir Crit. Care Med. 2010, 182, 1251–1261. [Google Scholar] [CrossRef]
- Bagnat, M.; Navis, A.; Marjoram, L. Cftr controls lumen expansion and function of Kupffer’s vesicle in zebrafish. Development 2013, 140, 1703–1712. [Google Scholar]
- Liu, Z.; Guo, J.; Wang, Y.; Weng, Z.; Huang, B.; Yu, M.K.; Zhang, X.; Yuan, P.; Zhao, H.; Chan, W.Y.; et al. CFTR-β-catenin interaction regulates mouse embryonic stem cell differentiation and embryonic development. Cell Death Differ. 2017, 24, 98–110. [Google Scholar] [CrossRef]
- de Souza, D.A.S.; Faucz, F.R.; Pereira-Ferrari, L.; Sotomaior, V.S.; Raskin, S. Congenital bilateral absence of the vas deferens as an atypical form of cystic fibrosis: Reproductive implications and genetic counseling. Andrology 2018, 6, 127–135. [Google Scholar] [CrossRef]
- Puchelle, E.; Gaillard, D.; Ploton, D.; Hinnrasky, J.; Fuchey, C.; Boutterin, M.C.; Jacquot, J.; Dreyer, D.; Pavirani, A.; Dalemans, W. Differential localization of the cystic fibrosis transmembrane conductance regulator in normal and cystic fibrosis airway epithelium. Am. J. Respir Cell Mol. Biol. 1992, 7, 485–491. [Google Scholar] [CrossRef]
- Dupuit, F.; Kälin, N.; Brézillon, S.; Hinnrasky, J.; Tümmler, B.; Puchelle, E. CFTR and differentiation markers expression in non-CF and delta F 508 homozygous CF nasal epithelium. J. Clin. Invest. 1995, 96, 1601–1611. [Google Scholar] [CrossRef] [PubMed]
- Brezillon, S.; Dupuit, F.; Hinnrasky, J.; Marchand, V.; Kälin, N.; Tümmler, B.; Puchelle, E. Decreased expression of the CFTR protein in remodeled human nasal epithelium from non-cystic fibrosis patients. Lab. Investig. 1995, 72, 191–200. [Google Scholar] [PubMed]
- Moyer, B.D.; Denton, J.; Karlson, K.H.; Reynolds, D.; Wang, S.; Mickle, J.E.; Milewski, M.; Cutting, G.R.; Guggino, W.B.; Li, M.; et al. A PDZ-interacting domain in CFTR is an apical membrane polarization signal. J. Clin. Investig. 1999, 104, 1353–1361. [Google Scholar] [CrossRef] [PubMed]
- LeSimple, P.; Liao, J.; Robert, R.; Gruenert, D.C.; Hanrahan, J.W. Cystic fibrosis transmembrane conductance regulator trafficking modulates the barrier function of airway epithelial cell monolayers. J. Physiol. 2010, 588, 1195–1209. [Google Scholar] [CrossRef] [PubMed]
- Coraux, C.; Hajj, R.; Lesimple, P.; Puchelle, E. In vivo models of human airway epithelium repair and regeneration. Eur. Respir. Rev. 2005, 14, 131–136. [Google Scholar] [CrossRef]
- Hajj, R.; Lesimple, P.; Nawrocki-Raby, B.; Birembaut, P.; Puchelle, E.; Coraux, C. Human airway surface epithelial regeneration is delayed and abnormal in cystic fibrosis. J. Pathol. 2007, 211, 340–350. [Google Scholar] [CrossRef]
- Schiller, K.R.; Maniak, P.J.; O’Grady, S.M. Cystic fibrosis transmembrane conductance regulator is involved in airway epithelial wound repair. Am. J. Physiol. 2010, 299, C912–C921. [Google Scholar] [CrossRef]
- Zhang, J.T.; Jiang, X.H.; Xie, C.; Cheng, H.; Da Dong, J.; Wang, Y.; Fok, K.L.; Zhang, X.H.; Sun, T.T.; Tsang, L.L.; et al. Downregulation of CFTR promotes epithelial-to-mesenchymal transition and is associated with poor prognosis of breast cancer. Biochim. Biophys. Acta - Mol. Cell Res. 2013, 1833, 2961–2969. [Google Scholar] [CrossRef]
- Clarke, L.A.; Botelho, H.M.; Sousa, L.; Falcao, A.O.; Amaral, M.D. Transcriptome meta-analysis reveals common differential and global gene expression profiles in cystic fibrosis and other respiratory disorders and identifies CFTR regulators. Genomics 2015, 106, 268–277. [Google Scholar] [CrossRef]
- Nyabam, S.; Wang, Z.; Thibault, T.; Oluseyi, A.; Basar, R.; Marshall, L.; Griffin, M. A novel regulatory role for tissue transglutaminase in epithelial-mesenchymal transition in cystic fibrosis. Biochim. Biophys. Acta Mol. Cell Res. 2016, 1863, 2234–2244. [Google Scholar] [CrossRef]
- Rout-Pitt, N.; Farrow, N.; Parsons, D.; Donnelley, M. Epithelial mesenchymal transition (EMT): A universal process in lung diseases with implications for cystic fibrosis pathophysiology. Respir. Res. 2018, 19, 136. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, V.; Schappell, L.E.; Mayer, C.R.; Duke, A.A.; Armiger, T.J.; Arsenovic, P.T.; Mohan, A.; Dahl, K.N.; Gleghorn, J.P.; Conway, D.E. Osmotic Gradients in Epithelial Acini Increase Mechanical Tension across E-cadherin, Drive Morphogenesis, and Maintain Homeostasis. Curr. Biol. 2020, 30, 624–633.e4. [Google Scholar] [CrossRef] [PubMed]
- Son, J.W.; Kim, Y.J.; Cho, H.M.; Lee, S.Y.; Lee, S.M.; Kang, J.-K.; Lee, J.U.; Lee, Y.M.; Kwon, S.J.; Choi, E.; et al. Promoter hypermethylation of the CFTR gene and clinical/pathological features associated with non-small cell lung cancer. Respirology 2011, 16, 1203–1209. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.T.; Wang, Y.; Cheng, H.; Zhang, X.H.; Xiang, J.J.; Zhang, J.T.; Yu, S.B.S.; Martin, T.A.; Ye, L.; Tsang, L.L.; et al. Disrupted interaction between CFTR and AF-6/afadin aggravates malignant phenotypes of colon cancer. Biochim. Biophys. Acta - Mol. Cell Res. 2014, 1843, 618–628. [Google Scholar] [CrossRef] [PubMed]
- Tu, Z.; Chen, Q.; Zhang, J.T.; Jiang, X.; Xia, Y.; Chan, H.C. CFTR is a potential marker for nasopharyngeal carcinoma prognosis and metastasis. Oncotarget 2016, 7, 76955–76965. [Google Scholar] [CrossRef][Green Version]
- Zhang, J.; Wang, Y.; Jiang, X.; Chan, H.C. Cystic fibrosis transmembrane conductance regulator—Emerging regulator of cancer. Cell Mol. Life Sci 2018. [Google Scholar] [CrossRef]
- Trezise, A.E.O.; Chambers, J.A.; Wardle, C.J.; Gould, S.; Harris, A. Expression of the cystic fibrosis gene in human foetal tissues. Hum. Mol. Genet. 1993, 2, 213–218. [Google Scholar] [CrossRef]
- Tizzano, E.F.; O’Brodovich, H.; Chitayat, D.; Bènichou, J.C.; Buchwald, M. Regional expression of CFTR in developing human respiratory tissues. Am. J. Respir Cell Mol. Biol 1994, 10, 355–362. [Google Scholar] [CrossRef]
- Gaillard, D.; Ruocco, S.; Lallemand, A.; Dalemans, W.; Hinnrasky, J.; Puchelle, E. Immunohistochemical Localization of Cystic Fibrosis Transmembrane Conductance Regulator in Human Fetal Airway and Digestive Mucosa. Pediatr. Res. 1994, 36, 137–143. [Google Scholar] [CrossRef]
- Harris, A.; Chalkley, G.; Goodman, S.; Coleman, L. Expression of the cystic fibrosis gene in human development. Development 1991, 113, 305–310. [Google Scholar]
- Tizzano, E.F.; Chitayat, D.; Buchwald, M. Cell-specific localization of CFTR mRNA shows developmentally regulated expression in human fetal tissues. Hum. Mol. Genet. 1993, 2, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Hyde, K.; Reid, C.J.; Tebbutt, S.J.; Weide, L.; Hollingsworth, M.A.; Harris, A. The cystic fibrosis transmembrane conductance regulator as a marker of human pancreatic duct development. Gastroenterology 1997, 113, 914–919. [Google Scholar] [CrossRef]
- Devuyst, O.; Burrow, C.R.; Schwiebert, E.M.; Guggino, W.B.; Wilson, P.D. Developmental regulation of CFTR expression during human nephrogenesis. Am. J. Physiol. 1996, 271, F723–F735. [Google Scholar] [CrossRef] [PubMed]
- Broackes-Carter, F.C. Temporal regulation of CFTR expression during ovine lung development: Implications for CF gene therapy. Hum. Mol. Genet. 2002, 11, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Marcorelles, P.; Montier, T.; Gillet, D.; Lagarde, N.; Ferec, C. Evolution of CFTR protein distribution in lung tissue from normal and CF human fetuses. Pediatr. Pulmonol. 2007, 42, 1032–1040. [Google Scholar] [CrossRef] [PubMed]
- McGrath, S.A.; Basu, A.; Zeitlin, P.L. Cystic Fibrosis Gene and Protein Expression during Fetal Lung Development. Am. J. Respir. Cell Mol. Biol 1993, 8, 201–208. [Google Scholar] [CrossRef]
- Gosden, C.; Gosden, J. Fetal Abnormalities in Cystic Fibrosis Suggest a Deficiency in Proteolysis of Cholecystokinin. Lancet 1984, 324, 541–546. [Google Scholar] [CrossRef]
- Ornoy, A.; Arnon, J.; Katznelson, D.; Granat, M.; Caspi, B.; Chemke, J.; Opitz, J.M.; Reynolds, J.F. Pathological confirmation of cystic fibrosis in the fetus following prenatal diagnosis. Am. J. Med. Genet. 1987, 28, 935–947. [Google Scholar] [CrossRef]
- Bonvin, E.; Le Rouzic, P.; Bernaudin, J.-F.; Cottart, C.-H.; Vandebrouck, C.; Crié, A.; Leal, T.; Clement, A.; Bonora, M. Congenital tracheal malformation in cystic fibrosis transmembrane conductance regulator-deficient mice. J. Physiol. 2008, 586, 3231–3243. [Google Scholar] [CrossRef]
- Tuggle, K.L.; Birket, S.E.; Cui, X.; Hong, J.; Warren, J.; Reid, L.; Chambers, A.; Ji, D.; Gamber, K.; Chu, K.K.; et al. Characterization of defects in ion transport and tissue development in Cystic Fibrosis Transmembrane Conductance Regulator (CFTR)-knockout rats. PLoS ONE 2014, 9, 1–14. [Google Scholar] [CrossRef]
- Meyerholz, D.K.; Stoltz, D.A.; Gansemer, N.D.; Ernst, S.E.; Cook, D.P.; Strub, M.D.; Leclair, E.N.; Barker, C.K.; Adam, R.J.; Leidinger, M.R.; et al. Lack of cystic fibrosis transmembrane conductance regulator disrupts fetal airway development in pigs. Lab. Investig. 2018, 98, 825–838. [Google Scholar] [CrossRef]
- Cohen, J.C.; Larson, J.E. Pathophysiologic consequences following inhibition of a CFTR-dependent developmental cascade in the lung. BMC Dev. Biol. 2005, 5, 1–13. [Google Scholar] [CrossRef]
- Cohen, J.C.; Morrow, S.L.; Cork, R.J.; Delcarpio, J.B.; Larson, J.E. Molecular Pathophysiology of Cystic Fibrosis Based on the Rescued Knockout Mouse Model. Mol. Genet. Metab. 1998, 64, 108–118. [Google Scholar] [CrossRef]
- Larson, J.E.; Delcarpio, J.B.; Farberman, M.M.; Morrow, S.L.; Cohen, J.C. CFTR modulates lung secretory cell proliferation and differentiation. Am. J. Physiol. - Lung Cell Mol. Physiol. 2000, 279, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Morrow, S.L.; Larson, J.E.; Nelson, S.; Sekhon, H.S.; Ren, T.; Cohen, J.C. Modification of Development by the CFTR Genein Utero. Mol. Genet. Metab. 1998, 65, 203–212. [Google Scholar] [CrossRef]
- Larson, J.E.; Morrow, S.L.; Delcarpio, J.B.; Bohm, R.P.; Ratterree, M.S.; Blanchard, J.L.; Cohen, J.C. Gene transfer into the fetal primate: Evidence for the secretion of transgene product. Mol. Ther. 2000, 2, 631–639. [Google Scholar] [CrossRef]
- Quinzii, C.; Castellani, C. The cystic fibrosis transmembrane regulator gene and male infertility. J. Endocrinol. Invest. 2000, 23, 684–689. [Google Scholar] [CrossRef]
- Chen, H.; Ruan, Y.C.; Xu, W.M.; Chen, J.; Chan, H.C. Regulation of male fertility by CFTR and implications in male infertility. Hum. Reprod. Update 2012, 18, 703–713. [Google Scholar] [CrossRef]
- Yefimova, M.; Bourmeyster, N.; Becq, F.; Burel, A.; Lavault, M.-T.; Jouve, G.; Veau, S.; Pimentel, C.; Jégou, B.; Ravel, C. Update on the cellular and molecular aspects of cystic fibrosis transmembrane conductance regulator (CFTR) and male fertility. Morphologie 2019, 103, 4–10. [Google Scholar] [CrossRef]
- Kaplan, E.; Shwachman, H.; Perlmutter, A.D.; Rule, A.; Khaw, K.-T.; Holsclaw, D.S. Reproductive Failure in Males with Cystic Fibrosis. N. Engl. J. Med. 1968, 279, 65–69. [Google Scholar] [CrossRef]
- Anguiano, A.; Oates, R.D.; Amos, J.A.; Dean, M.; Gerrard, B.; Stewart, C.; Maher, T.A.; White, M.B.; Milunsky, A. Congenital Bilateral Absence of the Vas Deferens. JAMA 1992, 267, 1794–1797. [Google Scholar] [CrossRef]
- Gaillard, D.A.; Carre-Pigeon, F.; Lallemand, A. Normal vas deferens in fetuses with cystic fibrosis. J. Urol. 1997, 158, 1549–1552. [Google Scholar] [CrossRef]
- Marcorelles, P.; Gillet, D.; Friocourt, G.; Ledé, F.; Samaison, L.; Huguen, G.; Ferec, C. Cystic fibrosis transmembrane conductance regulator protein expression in the male excretory duct system during development. Hum. Pathol. 2012, 43, 390–397. [Google Scholar] [CrossRef]
- Dumur, V.; Gervais, R.; Rigot, J.-M.; Lafitte, J.-J.; Manouvrier, S.; Biserte, J.; Mazeman, E.; Roussel, P. Abnormal distribution of CF ΔF508 allele in azoospermic men with congenital aplasia of epididymis and vas deferens. Lancet 1990, 336, 512. [Google Scholar] [CrossRef]
- Gervais, R.; Dumur, V.; Rigot, J.-M.; Lafitte, J.-J.; Roussel, P.; Claustres, M.; Demaille, J. High Frequency of the R117H Cystic Fibrosis Mutation in Patients with Congenital Absence of the Vas Deferens. N. Engl. J. Med. 1993, 328, 446–447. [Google Scholar] [CrossRef]
- Mercier, B.; Verlingue, C.; Lissens, W.; Silber, S.J.; Novelli, G.; Bonduelle, M.; Audrézet, M.P.; Férec, C. Is congenital bilateral absence of vas deferens a primary form of cystic fibrosis? Analyses of the CFTR gene in 67 patients. Am. J. Hum. Genet. 1995, 56, 272–277. [Google Scholar]
- Claustres, M.; Guittard, C.; Bozon, D.; Chevalier, F.; Verlingue, C.; Ferec, C.; Girodon, E.; Cazeneuve, C.; Bienvenu, T.; Lalau, G.; et al. Spectrum of CFTR mutations in cystic fibrosis and in congenital absence of the vas deferens in France. Hum. Mutat. 2000, 16, 143–156. [Google Scholar] [CrossRef]
- Noone, P.G.; Knowles, M.R. ‘CFTR-opathies’: Disease phenotypes associated with cystic fibrosis transmembrane regulator gene mutations. Respir. Res. 2001, 2, 328–332. [Google Scholar] [CrossRef]
- Yu, J.; Chen, Z.; Ni, Y.; Li, Z. CFTR mutations in men with congenital bilateral absence of the vas deferens (CBAVD): A systemic review and meta-analysis. Hum. Reprod. 2012, 27, 25–35. [Google Scholar] [CrossRef]
- Chillón, M.; Casals, T.; Mercier, B.; Bassas, L.; Lissens, W.; Silber, S.; Romey, M.C.; Ruiz-Romero, J.; Verlingue, C.; Claustres, M.; et al. Mutations in the cystic fibrosis gene in patients with congenital absence of the vas deferens. N. Engl. J. Med. 1995, 332, 1475–1480. [Google Scholar] [CrossRef]
- Knowles, M.R.; Drumm, M. The influence of genetics on cystic fibrosis phenotypes. Cold Spring Harb. Perspect. Med. 2012, 2, 1–14. [Google Scholar] [CrossRef]
- De Boeck, K.; Amaral, M.D. Progress in therapies for cystic fibrosis. Lancet Respir. Med. 2016, 4, 662–674. [Google Scholar] [CrossRef]
- Vanscoy, L.L.; Blackman, S.M.; Collaco, J.M.; Bowers, A.; Lai, T.; Naughton, K.; Algire, M.; McWilliams, R.; Beck, S.; Hoover-Fong, J.; et al. Heritability of lung disease severity in cystic fibrosis. Am. J. Respi.r Crit. Care Med. 2007, 175, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- Cuppens, H.; Cassiman, J.J. CFTR mutations and polymorphisms in male infertility. Int. J. Androl. 2004, 27, 251–256. [Google Scholar] [CrossRef]
- Ben-Chetrit, A. Expression of cystic fibrosis transmembrane conductance regulator during early human embryo development. Mol. Hum. Reprod. 2002, 8, 758–764. [Google Scholar] [CrossRef][Green Version]
- Lu, Y.C.; Chen, H.; Fok, K.L.; Tsang, L.L.; Yu, M.K.; Zhang, X.H.; Chen, J.; Jiang, X.; Chung, Y.W.; Ma, A.C.H.; et al. CFTR mediates bicarbonate-dependent activation of miR-125b in preimplantation embryo development. Cell Res. 2012, 22, 1453–1466. [Google Scholar] [CrossRef]
- Chen, M.H.; Chen, H.; Zhou, Z.; Ruan, Y.C.; Wong, H.Y.; Lu, Y.C.; Guo, J.H.; Chung, Y.W.; Huang, P.B.; Huang, H.F.; et al. Involvement of CFTR in oviductal HCO-3 secretion and its effect on soluble adenylate cyclase-dependent early embryo development. Hum. Reprod. 2010, 25, 1744–1754. [Google Scholar] [CrossRef]
- Lu, Y.C.; Yang, J.; Fok, K.L.; Ye, Y.H.; Jin, L.; Chen, Z.Y.; Zhang, X.M.; Huang, H.F.; Chan, H.C. Involvement of Cl−/HCO3− exchanger SLC26A3 and SLC26A6 in preimplantation embryo cleavage. Sci. Rep. 2016, 6, 28402. [Google Scholar] [CrossRef]
- Pankow, S.; Bamberger, C.; Calzolari, D.; Martínez-Bartolomé, S.; Lavallée-Adam, M.; Balch, W.E.; Yates, J.R. Δf508 CFTR interactome remodelling promotes rescue of cystic fibrosis. Nature 2015, 528, 510–516. [Google Scholar] [CrossRef]
- Li, X.; Fok, K.L.; Guo, J.; Wang, Y.; Liu, Z.; Chen, Z.; Wang, C.; Ruan, Y.C.; Yu, S.S.; Zhao, H.; et al. Retinoic acid promotes stem cell differentiation and embryonic development by transcriptionally activating CFTR. Biochim. Biophys. Acta - Mol. Cell Res. 2018, 1865, 605–615. [Google Scholar] [CrossRef]
- Liao, H.; Chen, Y.; Li, Y.; Xue, S.; Liu, M.; Lin, Z.; Liu, Y.; Chan, H.C.; Zhang, X.; Sun, H. CFTR is required for the migration of primordial germ cells during zebrafish early embryogenesis. Reproduction 2018, 156, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Mammoto, T.; Ingber, D.E. Mechanical control of tissue and organ development. Development 2010, 137, 1407–1420. [Google Scholar] [CrossRef] [PubMed]
- Nedvetsky, P.I.; Emmerson, E.; Finley, J.K.; Ettinger, A.; Cruz-Pacheco, N.; Prochazka, J.; Haddox, C.L.; Northrup, E.; Hodges, C.; Mostov, K.E.; et al. Parasympathetic Innervation Regulates Tubulogenesis in the Developing Salivary Gland. Dev. Cell 2014, 30, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Navis, A.; Bagnat, M. Developing pressures: Fluid forces driving morphogenesis. Curr. Opin. Genet. Dev. 2015, 32, 24–30. [Google Scholar] [CrossRef]
- Varner, V.D.; Nelson, C.M. Cellular and physical mechanisms of branching morphogenesis. Development 2014, 141, 2750–2759. [Google Scholar] [CrossRef]
- Nelson, C.M.; Gleghorn, J.P.; Pang, M.F.; Jaslove, J.M.; Goodwin, K.; Varner, V.D.; Miller, E.; Radisky, D.C.; Stone, H.A. Microfluidic chest cavities reveal that transmural pressure controls the rate of lung development. Development 2017, 144, 4328–4335. [Google Scholar] [CrossRef]
- Moore, K.A.; Polte, T.; Huang, S.; Shi, B.; Alsberg, E.; Sunday, M.E.; Ingber, D.E. Control of basement membrane remodeling and epithelial branching morphogenesis in embryonic lung by Rho and cytoskeletal tension. Dev. Dyn. 2005, 232, 268–281. [Google Scholar] [CrossRef]
- Ruan, Y.C.; Wang, Y.; Da Silva, N.; Kim, B.; Diao, R.Y.; Hill, E.; Brown, D.; Chan, H.C.; Breton, S. CFTR interacts with ZO-1 to regulate tight junction assembly and epithelial differentiation through the ZONAB pathway. J. Cell Sci. 2014, 127, 4396–4408. [Google Scholar] [CrossRef]
- Roxo-Rosa, M.; Jacinto, R.; Sampaio, P.; Lopes, S.S. The zebrafish Kupffer’s vesicle as a model system for the molecular mechanisms by which the lack of Polycystin-2 leads to stimulation of CFTR. Biol. Open 2015, 4, 1356–1366. [Google Scholar] [CrossRef]
- Bagnat, M.; Navis, A.; Herbstreith, S.; Brand-Arzamendi, K.; Curado, S.; Gabriel, S.; Mostov, K.; Huisken, J.; Stainier, D.Y.R. Cse1l is a negative regulator of CFTR-dependent fluid secretion. Curr. Biol. 2010, 20, 1840–1845. [Google Scholar] [CrossRef][Green Version]
- Cohen, J.C.; Larson, J.E.; Killeen, E.; Love, D.; Takemaru, K.I. CFTR and Wnt/beta-catenin signaling in lung development. BMC Dev. Biol. 2008, 8, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Navis, A.; Bagnat, M. Loss of cftr function leads to pancreatic destruction in larval zebrafish. Dev. Biol. 2015, 399, 237–248. [Google Scholar] [CrossRef] [PubMed]
- McCray, P.B.; Reenstra, W.W.; Louie, E.; Johnson, J.; Bettencourt, J.D.; Bastacky, J. Expression of CFTR and presence of cAMP-mediated fluid secretion in human fetal lung. Am. J. Physiol. Cell Mol. Physiol. 1992, 262, L472–L481. [Google Scholar] [CrossRef] [PubMed]
- Buchwald, M.; Sood, R.; Auerbach, W. Regulation of expression of CFTR in human intestinal epithelial cells. Adv. Exp. Med. Biol. 1991, 290, 241–252. [Google Scholar] [PubMed]
- Montrose-Rafizadeh, C.; Guggino, W.B.; Montrose, M.H. Cellular differentiation regulates expression of Cl- transport and cystic fibrosis transmembrane conductance regulator mRNA in human intestinal cells. J. Biol. Chem. 1991, 266, 4495–4499. [Google Scholar]
- Sood, R.; Bear, C.; Auerbach, W.; Reyes, E.; Jensen, T.; Kartner, N.; Riordan, J.R.; Buchwald, M. Regulation of CFTR expression and function during differentiation of intestinal epithelial cells. EMBO J. 1992, 11, 2487–2494. [Google Scholar] [CrossRef]
- Montrose-Rafizadeh, C.; Blackmon, D.L.; Hamosh, A.; Oliva, M.M.; Hawkins, A.L.; Curristin, S.M.; Griffin, C.A.; Yang, V.W.; Guggino, W.B.; Cutting, G.R.; et al. Regulation of cystic fibrosis transmembrane conductance regulator (CFTR) gene transcription and alternative RNA splicing in a model of developing intestinal epithelium. J. Biol. Chem. 1992, 267, 19299–19305. [Google Scholar]
- Hollande, E.; Fanjul, M.; Chemin-Thomas, C.; Devaux, C.; Demolombe, S.; Van Rietschoten, J.; Guy-Crotte, O.; Figarella, C. Targeting of CFTR protein is linked to the polarization of human pancreatic duct cells in culture. Eur. J. Cell Biol. 1998, 76, 220–227. [Google Scholar] [CrossRef]
- Kälin, N.; Claass, A.; Sommer, M.; Puchelle, E.; Tümmler, B. DeltaF508 CFTR protein expression in tissues from patients with cystic fibrosis. J. Clin. Invest. 1999, 103, 1379–1389. [Google Scholar] [CrossRef]
- Penque, D.; Mendes, F.; Beck, S.; Farinha, C.; Pacheco, P.; Nogueira, P.; Lavinha, J.; Malhó, R.; Amaral, M.D. Cystic fibrosis F508del patients have apically localized CFTR in a reduced number of airway cells. Lab. Invest. 2000, 80, 857–868. [Google Scholar] [CrossRef]
- Brézillon, S.; Hamm, H.; Heilmann, M.; Schäfers, H.J.; Hinnrasky, J.; Wagner, T.O.F.; Puchelle, E.; Tümmler, B. Decreased expression of the cystic fibrosis transmembrane conductance regulator protein in remodeled airway epithelium from lung transplanted patients. Hum. Pathol. 1997, 28, 944–952. [Google Scholar] [CrossRef]
- Bebök, Z.; Venglarik, C.J.; Pánczél, Z.; Jilling, T.; Kirk, K.L.; Sorscher, E.J. Activation of DeltaF508 CFTR in an epithelial monolayer. Am. J. Physiol. 1998, 275, C599–C607. [Google Scholar] [CrossRef] [PubMed]
- Moniz, S.; Sousa, M.; Moraes, B.J.; Mendes, A.I.; Palma, M.; Barreto, C.; Fragata, J.I.; Amaral, M.D.; Matos, P. HGF stimulation of Rac1 signaling enhances pharmacological correction of the most prevalent cystic fibrosis mutant F508del-CFTR. ACS Chem. Biol. 2013, 8, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Jun, E.S.; Kim, Y.S.; Yoo, M.A.; Roh, H.J.; Jung, J.S. Changes in expression of ion channels and aquaporins mRNA during differentiation in normal human nasal epithelial cells. Life Sci. 2001, 68, 827–840. [Google Scholar] [CrossRef]
- Clarke, L.A.; Sousa, L.; Barreto, C.; Amaral, M.D. Changes in transcriptome of native nasal epithelium expressing F508del-CFTR and intersecting data from comparable studies. Respir. Res. 2013, 14, 1. [Google Scholar] [CrossRef]
- Chifflet, S.; Hernández, J.A. The Plasma Membrane Potential and the Organization of the Actin Cytoskeleton of Epithelial Cells. Int. J. Cell Biol. 2012, 2012, 121424. [Google Scholar] [CrossRef]
- Hartsock, A.; Nelson, W.J. Adherens and tight junctions: Structure, function and connections to the actin cytoskeleton. Biochim. Biophys. Acta 2008, 1778, 660–669. [Google Scholar] [CrossRef]
- Olk, S.; Zoidl, G.; Dermietzel, R. Connexins, cell motility, and the cytoskeleton. Cell Motil. Cytoskeleton 2009, 66, 1000–1016. [Google Scholar] [CrossRef]
- Prat, A.G.; Xiao, Y.F.; Ausiello, D.A.; Cantiello, H.F. cAMP-independent regulation of CFTR by the actin cytoskeleton. Am. J. Physiol. Physiol. 1995, 268, C1552–C1561. [Google Scholar] [CrossRef]
- Prat, A.G.; Cunningham, C.C.; Jackson, G.R.; Borkan, S.C.; Wang, Y.; Ausiello, D.A.; Cantiello, H.F. Actin filament organization is required for proper cAMP-dependent activation of CFTR. Am. J. Physiol. Physiol. 1999, 277, C1160–C1169. [Google Scholar] [CrossRef]
- Chasan, B.; Geisse, N.A.; Pedatella, K.; Wooster, D.G.; Teintze, M.; Carattino, M.D.; Goldmann, W.H.; Cantiello, H.F. Evidence for direct interaction between actin and the cystic fibrosis transmembrane conductance regulator. Eur. Biophys. J. 2002, 30, 617–624. [Google Scholar] [PubMed]
- Stanke, F.; Hedtfeld, S.; Becker, T.; Tümmler, B. An association study on contrasting cystic fibrosis endophenotypes recognizes KRT8 but not KRT18 as a modifier of cystic fibrosis disease severity and CFTR mediated residual chloride secretion. BMC Med. Genet. 2011, 12, 62. [Google Scholar] [CrossRef] [PubMed]
- Favia, M.; Guerra, L.; Fanelli, T.; Cardone, R.A.; Monterisi, S.; Di Sole, F.; Castellani, S.; Chen, M.; Seidler, U.; Reshkin, S.J.; et al. Na+/H+ exchanger regulatory factor 1 overexpression-dependent increase of cytoskeleton organization is fundamental in the rescue of F508del cystic fibrosis transmembrane conductance regulator in human airway CFBE41o- cells. Mol. Biol. Cell 2010, 21, 73–86. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, H.E.; Dragomir, A.; Lazorova, L.; Johannesson, M.; Roomans, G.M. CFTR and tight junctions in cultured bronchial epithelial cells. Exp. Mol. Pathol. 2010, 88, 118–127. [Google Scholar] [CrossRef]
- Lasalvia, M.; Castellani, S.; D’Antonio, P.; Perna, G.; Carbone, A.; Colia, A.L.; Maffione, A.B.; Capozzi, V.; Conese, M. Human airway epithelial cells investigated by atomic force microscopy: A hint to cystic fibrosis epithelial pathology. Exp. Cell Res. 2016, 348, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Monterisi, S.; Favia, M.; Guerra, L.; Cardone, R.A.; Marzulli, D.; Reshkin, S.J.; Casavola, V.; Zaccolo, M. CFTR regulation in human airway epithelial cells requires integrity of the actin cytoskeleton and compartmentalized cAMP and PKA activity. J. Cell Sci. 2012, 125, 1106–1117. [Google Scholar] [CrossRef]
- Dransfield, D.T.; Bradford, A.J.; Smith, J.; Martin, M.; Roy, C.; Mangeat, P.H.; Goldenring, J.R. Ezrin is a cyclic AMP-dependent protein kinase anchoring protein. EMBO J. 1997, 16, 35–43. [Google Scholar] [CrossRef]
- Short, D.B.; Trotter, K.W.; Reczek, D.; Kreda, S.M.; Bretscher, A.; Boucher, R.C.; Stutts, M.J.; Milgram, S.L. An apical PDZ protein anchors the cystic fibrosis transmembrane conductance regulator to the cytoskeleton. J. Biol. Chem. 1998, 273, 19797–19801. [Google Scholar] [CrossRef]
- Guerra, L.; Fanelli, T.; Favia, M.; Riccardi, S.M.; Busco, G.; Cardone, R.A.; Carrabino, S.; Weinman, E.J.; Reshkin, S.J.; Conese, M.; et al. Na+/H+ exchanger regulatory factor isoform 1 overexpression modulates cystic fibrosis transmembrane conductance regulator (CFTR) expression and activity in human airway 16HBE14o- cells and rescues DeltaF508 CFTR functional expression in cystic fibrosis c. J. Biol. Chem. 2005, 280, 40925–40933. [Google Scholar] [CrossRef]
- Castellani, S.; Guerra, L.; Favia, M.; Di Gioia, S.; Casavola, V.; Conese, M. NHERF1 and CFTR restore tight junction organisation and function in cystic fibrosis airway epithelial cells: Role of ezrin and the RhoA/ROCK pathway. Lab. Investig. 2012, 92, 1527–1540. [Google Scholar] [CrossRef]
- Powell, D.W. Barrier function of epithelia. Am. J. Physiol. 1981, 241, G275–G288. [Google Scholar] [CrossRef] [PubMed]
- Weiser, N.; Molenda, N.; Urbanova, K.; Bähler, M.; Pieper, U.; Oberleithner, H.; Schillers, H. Paracellular permeability of bronchial epithelium is controlled by CFTR. Cell Physiol. Biochem. 2011, 28, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Molina, S.A.; Stauffer, B.; Moriarty, H.K.; Kim, A.H.; McCarty, N.A.; Koval, M. Junctional abnormalities in human airway epithelial cells expressing F508del CFTR. Am. J. Physiol. Lung Cell Mol. Physiol. 2015, 309, L475–L487. [Google Scholar] [CrossRef] [PubMed]
- Breton, S.; Ruan, Y.C.; Park, Y.-J.; Kim, B. Regulation of epithelial function, differentiation, and remodeling in the epididymis. Asian J. Androl. 2016, 18, 3–9. [Google Scholar] [CrossRef]
- Itoh, M.; Furuse, M.; Morita, K.; Kubota, K.; Saitou, M.; Tsukita, S. Direct binding of three tight junction-associated MAGUKs, ZO-1, ZO-2, and ZO-3, with the COOH termini of claudins. J. Cell Biol. 1999, 147, 1351–1363. [Google Scholar] [CrossRef] [PubMed]
- De Lisle, R.C. Disrupted tight junctions in the small intestine of cystic fibrosis mice. Cell Tissue Res. 2014, 355, 131–142. [Google Scholar] [CrossRef]
- Chanson, M.; Suter, S. Regulation of gap junctional communication in CFTR-expressing pancreatic epithelial cells. Pflugers Arch. 2001, 443, S81–S84. [Google Scholar] [CrossRef]
- Barbry, P.; Cavard, A.; Chanson, M.; Jaffe, A.B.; Plasschaert, L.W. Regeneration of airway epithelial cells to study rare cell states in cystic fibrosis. J. Cyst Fibros 2020, 19, S42–S46. [Google Scholar] [CrossRef]
- Crosby, L.M.; Waters, C.M. Epithelial repair mechanisms in the lung. Am. J. Physiol. Lung Cell Mol. Physiol. 2010, 298, L715–L731. [Google Scholar] [CrossRef] [PubMed]
- Beers, M.F.; Morrisey, E.E. The three R’s of lung health and disease: Repair, remodeling, and regeneration. J. Clin. Investig. 2011, 121, 2065–2073. [Google Scholar] [CrossRef]
- Tran-Paterson, R.; Davin, D.; Krauss, R.D.; Rado, T.A.; Miller, D.M. Expression and regulation of the cystic fibrosis gene during rat liver regeneration. Am. J. Physiol. 1992, 263, C55–C60. [Google Scholar] [CrossRef] [PubMed]
- Castillon, N.; Hinnrasky, J.; Zahm, J.-M.; Kaplan, H.; Bonnet, N.; Corlieu, P.; Klossek, J.-M.; Taouil, K.; Avril-Delplanque, A.; Péault, B.; et al. Polarized expression of cystic fibrosis transmembrane conductance regulator and associated epithelial proteins during the regeneration of human airway surface epithelium in three-dimensional culture. Lab. Investig. 2002, 82, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Jorissen, M.; Van der Schueren, B.; Van den Berghe, H.; Cassiman, J.J. The preservation and regeneration of cilia on human nasal epithelial cells cultured in vitro. Arch. Otorhinolaryngol. 1989, 246, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Castillon, N.; Avril-Delplanque, A.; Coraux, C.; Delenda, C.; Péault, B.; Danos, O.; Puchelle, E. Regeneration of a well-differentiated human airway surface epithelium by spheroid and lentivirus vector-transduced airway cells. J. Gene Med. 2004, 6, 846–856. [Google Scholar] [CrossRef] [PubMed]
- Leigh, M.W.; Kylander, J.E.; Yankaskas, J.R.; Boucher, R.C. Cell proliferation in bronchial epithelium and submucosal glands of cystic fibrosis patients. Am. J. Respir. Cell Mol. Biol. 1995, 12, 605–612. [Google Scholar] [CrossRef]
- Voynow, J.A.; Fischer, B.M.; Roberts, B.C.; Proia, A.D. Basal-like Cells Constitute the Proliferating Cell Population in Cystic Fibrosis Airways. Am. J. Respir Crit. Care Med. 2005, 172, 1013–1018. [Google Scholar] [CrossRef]
- Crespin, S.; Bacchetta, M.; Bou Saab, J.; Tantilipikorn, P.; Bellec, J.; Dudez, T.; Nguyen, T.H.; Kwak, B.R.; Lacroix, J.S.; Huang, S.; et al. Cx26 regulates proliferation of repairing basal airway epithelial cells. Int. J. Biochem. Cell Biol. 2014, 52, 152–160. [Google Scholar] [CrossRef]
- Adam, D.; Roux-Delrieu, J.; Luczka, E.; Bonnomet, A.; Lesage, J.; Mérol, J.-C.; Polette, M.; Abély, M.; Coraux, C. Cystic fibrosis airway epithelium remodelling: Involvement of inflammation. J. Pathol. 2015, 235, 408–419. [Google Scholar] [CrossRef]
- Adam, D.; Bilodeau, C.; Sognigbé, L.; Maillé, É.; Ruffin, M.; Brochiero, E. CFTR rescue with VX-809 and VX-770 favors the repair of primary airway epithelial cell cultures from patients with class II mutations in the presence of Pseudomonas aeruginosa exoproducts. J. Cyst. Fibros. 2018, 17, 705–714. [Google Scholar] [CrossRef]
- Coraux, C.; Hajj, R.; Lesimple, P.; Puchelle, E. [Repair and regeneration of the airway epithelium]. Med. Sci. (Paris) 2005, 21, 1063–1069. [Google Scholar] [CrossRef][Green Version]
- Fischer, B.M.; Wong, J.K.; Degan, S.; Kummarapurugu, A.B.; Zheng, S.; Haridass, P.; Voynow, J.A. Increased expression of senescence markers in cystic fibrosis airways. Am. J. Physiol. Lung Cell Mol. Physiol. 2013, 304, L394–L400. [Google Scholar] [CrossRef] [PubMed]
- Zoso, A.; Sofoluwe, A.; Bacchetta, M.; Chanson, M. Transcriptomic profile of cystic fibrosis airway epithelial cells undergoing repair. Sci. Data 2019, 6, 240. [Google Scholar] [CrossRef] [PubMed]
- Stamm, A.; Reimers, K.; Strauß, S.; Vogt, P.; Scheper, T.; Pepelanova, I. In vitro wound healing assays – state of the art. BioNanoMaterials 2016, 17, 79–87. [Google Scholar] [CrossRef]
- Maillé, E.; Trinh, N.T.N.; Privé, A.; Bilodeau, C.; Bissonnette, E.; Grandvaux, N.; Brochiero, E. Regulation of normal and cystic fibrosis airway epithelial repair processes by TNF-α after injury. Am. J. Physiol. Lung Cell Mol. Physiol. 2011, 301, L945–L955. [Google Scholar] [CrossRef]
- Kirk, K.L. CFTR channels and wound healing. Focus on ‘Cystic fibrosis transmembrane conductance regulator is involved in airway epithelial wound repair’. Am. J. Physiol. Cell Physiol. 2010, 299, C888–C890. [Google Scholar] [CrossRef]
- Trinh, N.T.N.; Bardou, O.; Privé, A.; Maillé, E.; Adam, D.; Lingée, S.; Ferraro, P.; Desrosiers, M.-Y.; Coraux, C.; Brochiero, E. Improvement of defective cystic fibrosis airway epithelial wound repair after CFTR rescue. Eur. Respir. J. 2012, 40, 1390–1400. [Google Scholar] [CrossRef]
- Itokazu, Y.; Pagano, R.E.; Schroeder, A.S.; O’Grady, S.M.; Limper, A.H.; Marks, D.L. Reduced GM1 ganglioside in CFTR-deficient human airway cells results in decreased β1-integrin signaling and delayed wound repair. Am. J. Physiol. Physiol. 2014, 306, C819–C830. [Google Scholar] [CrossRef]
- Marino, G.I.; Kotsias, B.A. Cystic fibrosis transmembrane regulator (CFTR) in human trophoblast BeWo cells and its relation to cell migration. Placenta 2014, 35, 92–98. [Google Scholar] [CrossRef]
- Dong, J.; Jiang, X.; Zhang, X.; Liu, K.S.; Zhang, J.; Chen, J.; Yu, M.K.; Tsang, L.L.; Chung, Y.W.; Wang, Y.; et al. Dynamically Regulated CFTR Expression and Its Functional Role in Cutaneous Wound Healing. J. Cell Physiol. 2015, 230, 2049–2058. [Google Scholar] [CrossRef]
- Molenda, N.; Urbanova, K.; Weiser, N.; Kusche-Vihrog, K.; Günzel, D.; Schillers, H.; Günzel, D.; Schillers, H. Paracellular transport through healthy and cystic fibrosis bronchial epithelial cell lines--do we have a proper model? PLoS ONE 2014, 9, e100621. [Google Scholar] [CrossRef]
- Dodge, J.A.; Lewis, P.A.; Stanton, M.; Wilsher, J. Cystic fibrosis mortality and survival in the UK: 1947-2003. Eur. Respir. J. 2007, 29, 522–526. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.W. Childhood Cancer and Congenital Defects A Study of U.S. Death Certificates During the Period 1960–1966. Pediatr. Res. 1969, 3, 389–397. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Abdul-Karim, F.W.; King, T.A.; Dahms, B.B.; Gauderer, M.W.L.; Boat, T.F. Carcinoma of extrahepatic biliary system in an adult with cystic fibrosis. Gastroenterology 1982, 82, 758–762. [Google Scholar] [CrossRef]
- Davis, T.M.E.; Sawicka, E.H. Adenocarcinoma in cystic fibrosis. Thorax 1985, 40, 199–200. [Google Scholar] [CrossRef] [PubMed][Green Version]
- McIntosh, J.C.; Schoumacher, R.A.; Tiller, R.E. Pancreatic adenocarcinoma in a patient with cystic fibrosis. Am. J. Med. 1988, 85, 592. [Google Scholar] [CrossRef]
- Siraganian, P.; Miller, R.; Swender, P. Cystic Fibrosis and Ileal Carcinoma. Lancet 1987, 330, 1158. [Google Scholar] [CrossRef]
- Roberts, J.A.; Tullett, W.M.; Thomas, J.S.; Galloway, D.; Stack, B.H.R. Bowel Adenocarcinoma in a Patient with Cystic Fibrosis. Scott. Med. J. 1986, 31, 109. [Google Scholar] [CrossRef]
- Biggs, B.G.; Vaughan, W.; Colombo, J.L.; Sanger, W.; Purtilo, D.T. Cystic fibrosis complicated by acute leukemia. Cancer 1986, 57, 2441–2443. [Google Scholar] [CrossRef]
- Neglia, J.P.; Wielinski, C.L.; Warwick, W.J. Cancer risk among patients with cystic fibrosis. J. Pediatr. 1991, 119, 764–766. [Google Scholar] [CrossRef]
- Sheldon, C.D.; Hodson, M.E.; Carpenter, L.M.; Swerdlow, A.J. A cohort study of cystic fibrosis and malignancy. Br. J. Cancer 1993, 68, 1025. [Google Scholar] [CrossRef]
- Hadjiliadis, D.; Khoruts, A.; Zauber, A.G.; Hempstead, S.E.; Maisonneuve, P.; Lowenfels, A.B.; Braid, A.L.; Cullina, J.; Daggett, A.; Fink, A.; et al. Cystic Fibrosis Colorectal Cancer Screening Consensus Recommendations. Gastroenterology 2018, 154, 736–745.e14. [Google Scholar] [CrossRef] [PubMed]
- Vekens, K.; Vincken, S.; Hanon, S.; Demuynck, K.; Stylemans, D.; Vanderhelst, E. Lung cancer in a CF patient: Combination of bad luck or is there more to say? Acta Clin. Belg. 2020, 00, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Warren, N.; Holmes, J.A.; Al-Jader, L.; West, R.R.; Lewis, D.C.; Padua, R.A. Frequency of carriers of cystic fibrosis gene among patients with myeloid malignancy and melanoma. BMJ 1991, 302, 760–761. [Google Scholar] [CrossRef] [PubMed]
- Padua, R.A.; Warren, N.; Grimshaw, D.; Smith, M.; Lewis, C.; Whittaker, J.; Laidler, P.; Wright, P.; Douglas-Jones, A.; Fenaux, P.; et al. The cystic fibrosis ΔF508 gene mutation and cancer. Hum. Mutat. 1997, 10, 45–48. [Google Scholar] [CrossRef]
- Li, Y.; Sun, Z.; Wu, Y.; Babovic-Vuksanovic, D.; Li, Y.; Cunningham, J.M.; Pankratz, V.S.; Yang, P. Cystic fibrosis transmembrane conductance regulator gene mutation and lung cancer risk. Lung Cancer 2010, 70, 14–21. [Google Scholar] [CrossRef]
- Qiao, D.; Yi, L.; Hua, L.; Xu, Z.; Ding, Y.; Shi, D.; Ni, L.; Song, N.; Wang, Y.; Wu, H. Cystic fibrosis transmembrane conductance regulator (CFTR) gene 5T allele may protect against prostate cancer: A case-control study in Chinese Han population. J. Cyst. Fibros 2008, 7, 210–214. [Google Scholar] [CrossRef][Green Version]
- McWilliams, R. Cystic fibrosis transmembrane regulator gene carrier status is a risk factor for young onset pancreatic adenocarcinoma. Gut 2005, 54, 1661–1662. [Google Scholar] [CrossRef]
- McWilliams, R.R.; Petersen, G.M.; Rabe, K.G.; Holtegaard, L.M.; Lynch, P.J.; Bishop, M.D.; Highsmith, W.E. Cystic fibrosis transmembrane conductance regulator (CFTR) gene mutations and risk for pancreatic adenocarcinoma. Cancer 2010, 116, 203–209. [Google Scholar] [CrossRef]
- Southey, M.C.; Batten, L.; Andersen, C.R.; Mccredie, M.R.E.; Giles, G.G.; Dite, G.; Hopper, J.L.; Venter, D.J. CFTR ΔF508 carrier status, risk of breast cancer before the age of 40 and histological grading in a population-based case-control study. Int. J. Cancer 1998, 79, 487–489. [Google Scholar] [CrossRef]
- Johannesson, M.; Askling, J.; Montgomery, S.M.; Ekbom, A.; Bahmanyar, S. Cancer risk among patients with cystic fibrosis and their first-degree relatives. Int J. Cancer 2009, 125, 2953–2956. [Google Scholar] [CrossRef]
- Abraham, E.H.; Vos, P.; Kahn, J.; Grubman, S.A.; Jefferson, D.M.; Ding, I.; Okunieff, P. Cystic fibrosis hetero-and homozygosity is associated with inhibition of breast cancer growth. Nat. Med. 1996, 2, 593–596. [Google Scholar] [CrossRef] [PubMed]
- Moribe, T.; Iizuka, N.; Miura, T.; Kimura, N.; Tamatsukuri, S.; Ishitsuka, H.; Hamamoto, Y.; Sakamoto, K.; Tamesa, T.; Oka, M. Methylation of multiple genes as molecular markers for diagnosis of a small, well-differentiated hepatocellular carcinoma. Int J. Cancer 2009, 125, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zhu, T.; Wang, Z.; Zhang, H.; Qian, Z.; Xu, H.; Gao, B.; Wang, W.; Gu, L.; Meng, J.; et al. A novel set of DNA methylation markers in urine sediments for sensitive/specific detection of bladder cancer. Clin. Cancer Res. 2007, 13, 7296–7304. [Google Scholar] [CrossRef] [PubMed]
- Ashour, N.; Angulo, J.C.; Andrés, G.; Alelú, R.; González-Corpas, A.; Toledo, M.V.; Rodríguez-Barbero, J.M.; López, J.I.; Sánchez-Chapado, M.; Ropero, S. A DNA hypermethylation profile reveals new potential biomarkers for prostate cancer diagnosis and prognosis. Prostate 2014, 74, 1171–1182. [Google Scholar] [CrossRef]
- Liu, K.; Dong, F.; Gao, H.; Guo, Y.; Li, H.; Yang, F.; Zhao, P.; Dai, Y.; Wang, J.; Zhou, W.; et al. Promoter hypermethylation of the CFTR gene as a novel diagnostic and prognostic marker of breast cancer. Cell Biol. Int. 2020, 44, 603–609. [Google Scholar] [CrossRef]
- Shin, Y.; Kim, M.; Won, J.; Kim, J.; Oh, S.B.; Lee, J.-H.; Park, K. Epigenetic Modification of CFTR in Head and Neck Cancer. J. Clin. Med. 2020, 9, 734. [Google Scholar] [CrossRef]
- Cheng, S.; Li, C.; Xie, W.; Miao, Y.; Guo, J.; Wang, J.; Zhang, Y. Integrated analysis of DNA methylation and mRNA expression profiles to identify key genes involved in the regrowth of clinically non-functioning pituitary adenoma. Aging (Albany NY) 2020, 12, 2408–2427. [Google Scholar] [CrossRef]
- Singh, A.P.; Chauhan, S.C.; Andrianifahanana, M.; Moniaux, N.; Meza, J.L.; Copin, M.C.; Van Seuningen, I.; Hollingsworth, M.A.; Aubert, J.P.; Batra, S.K. MUC4 expression is regulated by cystic fibrosis transmembrane conductance regulator in pancreatic adenocarcinoma cells via transcriptional and post-translational mechanisms. Oncogene 2007, 26, 30–41. [Google Scholar] [CrossRef]
- Li, J.; Zhang, J.T.; Jiang, X.; Shi, X.; Shen, J.; Feng, F.; Chen, J.; Liu, G.; He, P.; Jiang, J.; et al. The cystic fibrosis transmembrane conductance regulator as a biomarker in non-small cell lung cancer. Int J. Oncol. 2015, 46, 2107–2115. [Google Scholar] [CrossRef]
- Tian, F.; Zhao, J.; Fan, X.; Kang, Z. Weighted gene co-expression network analysis in identification of metastasis-related genes of lung squamous cell carcinoma based on the Cancer Genome Atlas database. J. Thorac Dis 2017, 9, 42–53. [Google Scholar] [CrossRef]
- Li, W.; Wang, C.; Peng, X.; Zhang, H.; Huang, H.; Liu, H. CFTR inhibits the invasion and growth of esophageal cancer cells by inhibiting the expression of NF-κB. Cell Biol. Int. 2018, 42, 1680–1687. [Google Scholar] [CrossRef] [PubMed]
- Than, B.L.N.; Linnekamp, J.F.; Starr, T.K.; Largaespada, D.A.; Rod, A.; Zhang, Y.; Bruner, V.; Abrahante, J.; Schumann, A.; Luczak, T.; et al. CFTR is a tumor suppressor gene in murine and human intestinal cancer. Oncogene 2016, 35, 4191–4199. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Chen, H.Q.; Yang, X.L.; Wang, Q.; Chen, W.; Li, C. CFTR activation suppresses glioblastoma cell proliferation, migration and invasion. Biochem. Biophys. Res. Commun. 2019, 508, 1279–1285. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Jiang, X.H.; Zhang, J.T.; Sun, T.T.; Dong, J.D.; Sanders, A.J.; Diao, R.Y.; Wang, Y.; Fok, K.L.; Tsang, L.L.; et al. CFTR suppresses tumor progression through miR-193b targeting urokinase plasminogen activator (uPA) in prostate cancer. Oncogene 2013, 32, 2282–2291. [Google Scholar] [CrossRef]
- Peng, X.; Wu, Z.; Yu, L.; Li, J.; Xu, W.; Chan, H.C.; Zhang, Y.; Hu, L. Overexpression of cystic fibrosis transmembrane conductance regulator (CFTR) is associated with human cervical cancer malignancy, progression and prognosis. Gynecol. Oncol. 2012, 125, 470–476. [Google Scholar] [CrossRef]
- Wu, Z.; Peng, X.; Li, J.; Zhang, Y.; Hu, L. Constitutive Activation of Nuclear Factor κB Contributes to Cystic Fibrosis Transmembrane Conductance Regulator Expression and Promotes Human Cervical Cancer Progression and Poor Prognosis. Int. J. Gynecol. Cancer 2013, 23, 906–915. [Google Scholar] [CrossRef]
- Xu, J.; Yong, M.; Li, J.; Dong, X.; Yu, T.; Fu, X.; Hu, L. High level of CFTR expression is associated with tumor aggression and knockdown of CFTR suppresses proliferation of ovarian cancer in vitro and in vivo. Oncol. Rep. 2015, 33, 2227–2234. [Google Scholar] [CrossRef]
- Rochwerger, L.; Buchwald, M. Stimulation of the cystic fibrosis transmembrane regulator expression by estrogen in vivo. Endocrinology 1993, 133, 921–930. [Google Scholar] [CrossRef]
- Verhaeghe, C.; Tabruyn, S.P.; Oury, C.; Bours, V.; Griffioen, A.W. Intrinsic pro-angiogenic status of cystic fibrosis airway epithelial cells. Biochem. Biophys. Res. Commun. 2007, 356, 745–749. [Google Scholar] [CrossRef]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.J.; Nieto, M.A. Epithelial-Mesenchymal Transitions in Development and Disease. Cell 2009, 139, 871–890. [Google Scholar] [CrossRef]
- Lamouille, S.; Xu, J.; Derynck, R. Molecular mechanisms of epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2014, 15, 178–196. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 2009, 119, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Snodgrass, S.M.; Cihil, K.M.; Cornuet, P.K.; Myerburg, M.M.; Swiatecka-Urban, A. Tgf-β1 Inhibits Cftr Biogenesis and Prevents Functional Rescue of ΔF508-Cftr in Primary Differentiated Human Bronchial Epithelial Cells. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Sulaiman, A.; Yao, Z.; Wang, L. Re-evaluating the role of epithelial-mesenchymal-transition in cancer progression. J. Biomed. Res. 2018, 32, 81–90. [Google Scholar]
- Fingerlin, T.E.; Murphy, E.; Zhang, W.; Peljto, A.L.; Brown, K.K.; Steele, M.P.; Loyd, J.E.; Cosgrove, G.P.; Lynch, D.; Groshong, S.; et al. Genome-wide association study identifies multiple susceptibility loci for pulmonary fibrosis. Nat. Genet. 2013, 45, 613–620. [Google Scholar] [CrossRef]
- van Moorsel, C.H.M. Trade-offs in aging lung diseases. Curr. Opin. Pulm. Med. 2018, 24, 309–317. [Google Scholar] [CrossRef]
- Corvol, H.; Rousselet, N.; Thompson, K.E.; Berdah, L.; Cottin, G.; Foussigniere, T.; Longchampt, E.; Fiette, L.; Sage, E.; Prunier, C.; et al. FAM13A is a modifier gene of cystic fibrosis lung phenotype regulating rhoa activity, actin cytoskeleton dynamics and epithelial-mesenchymal transition. J. Cyst. Fibros. 2018, 17, 190–203. [Google Scholar] [CrossRef]
- Jolly, M.K.; Ward, C.; Eapen, M.S.; Myers, S.; Hallgren, O.; Levine, H.; Sohal, S.S. Epithelial-mesenchymal transition, a spectrum of states: Role in lung development, homeostasis, and disease. Dev. Dyn. 2018, 247, 346–358. [Google Scholar] [CrossRef]
- Harris, W.T.; Muhlebach, M.S.; Oster, R.A.; Knowles, M.R.; Clancy, J.P.; Noah, T.L. Plasma TGF-β1 in pediatric cystic fibrosis: Potential biomarker of lung disease and response to therapy. Pediatr. Pulmonol. 2011, 46, 688–695. [Google Scholar] [CrossRef]
- Zhang, J.T.; Wang, Y.; Chen, J.J.; Zhang, X.H.; Dong, J.D.; Tsang, L.L.; Huang, X.R.; Cai, Z.; Lan, H.Y.; Jiang, X.H.; et al. Defective CFTR leads to aberrant β-catenin activation and kidney fibrosis. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef]
- Li, P.; Singh, J.; Sun, Y.; Ma, X.; Yuan, P. CFTR constrains the differentiation from mouse embryonic stem cells to intestine lineage cells. Biochem. Biophys. Res. Commun. 2019, 510, 322–328. [Google Scholar] [CrossRef] [PubMed]
| Tissues | 1st Trimester | 2nd Trimester | 3rd Trimester | References |
|---|---|---|---|---|
| Pancreas | +++ | +++ | +++ | [27,30,31,32] |
| Liver | ++ | ++ | ++ | [27,31] |
| Kidneys | ++ | +++ | ++ | [33] |
| Colon and small intestine | +++ | +++ | +++ | [10,27,31] |
| Large airways | + d | + | + | [27,28,29,31] |
| Small airways | + | ++ | + | [27,28,31] |
| Submucosal glands | ND | ND | ND | [28] |
| Epididymis Vas deferens | + | + | + | [30,31] |
| Uterus Fallopian tubes | ND | ND | + | [31] |
| Protein/ Marker | Reported Difference in CF | Cells/Tissue | Assay | Reference |
|---|---|---|---|---|
| Actin stress fibers | Disorganized | CF (CFBE41o-) vs. non-CF (16HBE14o-) cells | Immunostaining | [103,104] |
| Actin stress fibers | Not present in CFBE41o- compared to 16HBE14o- Some present in CFBE41o-/wt-CFTR | CF (CFBE41o-) vs. 1 non-CF (6HBE14o-) vs. isogenic non-CF (CFBE41o-/wt-CFTR) cells | Immunostaining | [105] |
| KRT5 KRT14 EGFR | Increased | Airway sections from human CF and non-CF explanted lungs | Immunohisto-chemistry | [126] |
| ZO-1 | Increased when cells cultured at 29 °C compared to 37 °C | CF (CFBE41o-) cells | Immunostaining | [14] |
| ZO-1 Occludin Cldn1 JAM-1 | Not expressed | CF (CFBE41o-) vs. non-CF (16HBE14o-) cells | Immunostaining | [110] |
| Cldn1 Cldn7 Cldn8 Pmp22 | Decreased | CF and non-CF mouse intestine | qRT-PCR Immunohisto-chemistry | [116] |
| Cldn2 | Increased | CF and non-CF mouse intestine | qRT-PCR Western Blot Immunohisto-chemistry | [116] |
| Cldn3 | Decreased in CFBE41o-, wt-CFTR, F508del-CFTR compared to 16HBE | CF (CFBE41o-), isogenic non-CF (CFBE wt-CFTR) or CF (CFBE F508del-CFTR) vs. non-CF (16HBE14o-) cells | Immunostaining | [140] |
| Connexin-43 | Mislocalized | CF (CuFi-5) vs. non-CF (NuLi-1) cells | Immunostaining | [113] |
| TEER | Decreased in GFP-F508del-CFTR expressing cells vs. GFP-wt-CFTR | CF (CFBE41o-) cells | Volt-Ohm Meter | [14] |
| TEER | Decreased | CF (CFBE41o- vs. non-CF (16HBE14o-) cells | Volt-Ohm Meter | [112] |
| TEER | Lower | CF (CuFi-5) vs. non-CF (NuLi-1) cells | Volt-Ohm Meter | [113] |
| Epithelium height | Increased | CF and control human airway tissue | Histological examination | [16] |
| Ki-67 KRT13 | Increased | CF and control human airway tissue | Histological examination | [16] |
| MUC5B | Reduced | CF and control human airway tissue | Histological examination | [16] |
| IL-8 MMP-7 MMP-9 TIMP-1 | Increased | CF and control human airway tissue | RT-qPCR | [16] |
| PCNA | Increased | CF and control human airway tissue | Immunostaining | [125] |
| Ki-67 | Increased | CF and control lung sections | Immunohisto-chemistry | [126] |
| KLF4 KLF2 | No change during repair of CF HAEC | CF HAEC and non-CF HAEC | RT-qPCR | [127] |
| Ki-67 Connexin-26 | Elevated in CF HAEC during repair | CF HAEC and non-CF HAEC | Immunostaining | [127] |
| Cilia biology related genes | Downregulated | CF Nasal Epithelia | Microarray | [95] |
| Proliferation related genes | Upregulated | CF and non-CF Nasal Epithelia | Microarray | [95] |
| β-Tubulin | Lower | CF and non-CF human airway epithelial cells | Immunostaining | [128] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amaral, M.D.; Quaresma, M.C.; Pankonien, I. What Role Does CFTR Play in Development, Differentiation, Regeneration and Cancer? Int. J. Mol. Sci. 2020, 21, 3133. https://doi.org/10.3390/ijms21093133
Amaral MD, Quaresma MC, Pankonien I. What Role Does CFTR Play in Development, Differentiation, Regeneration and Cancer? International Journal of Molecular Sciences. 2020; 21(9):3133. https://doi.org/10.3390/ijms21093133
Chicago/Turabian StyleAmaral, Margarida D., Margarida C. Quaresma, and Ines Pankonien. 2020. "What Role Does CFTR Play in Development, Differentiation, Regeneration and Cancer?" International Journal of Molecular Sciences 21, no. 9: 3133. https://doi.org/10.3390/ijms21093133
APA StyleAmaral, M. D., Quaresma, M. C., & Pankonien, I. (2020). What Role Does CFTR Play in Development, Differentiation, Regeneration and Cancer? International Journal of Molecular Sciences, 21(9), 3133. https://doi.org/10.3390/ijms21093133





