The Influence of Food Intake Specificity in Children with Autism on Gut Microbiota
Abstract
1. Introduction
2. Results and Discussion
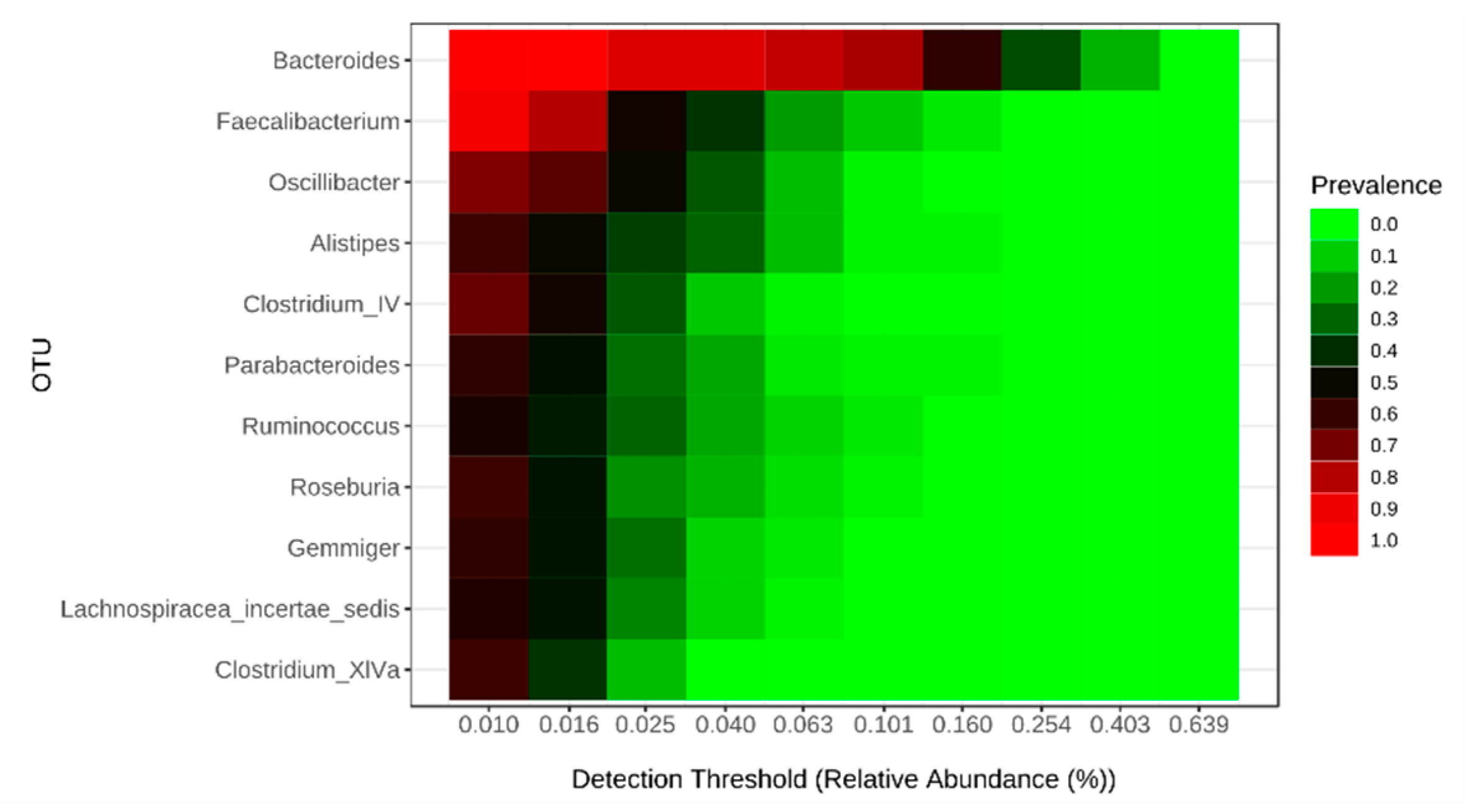
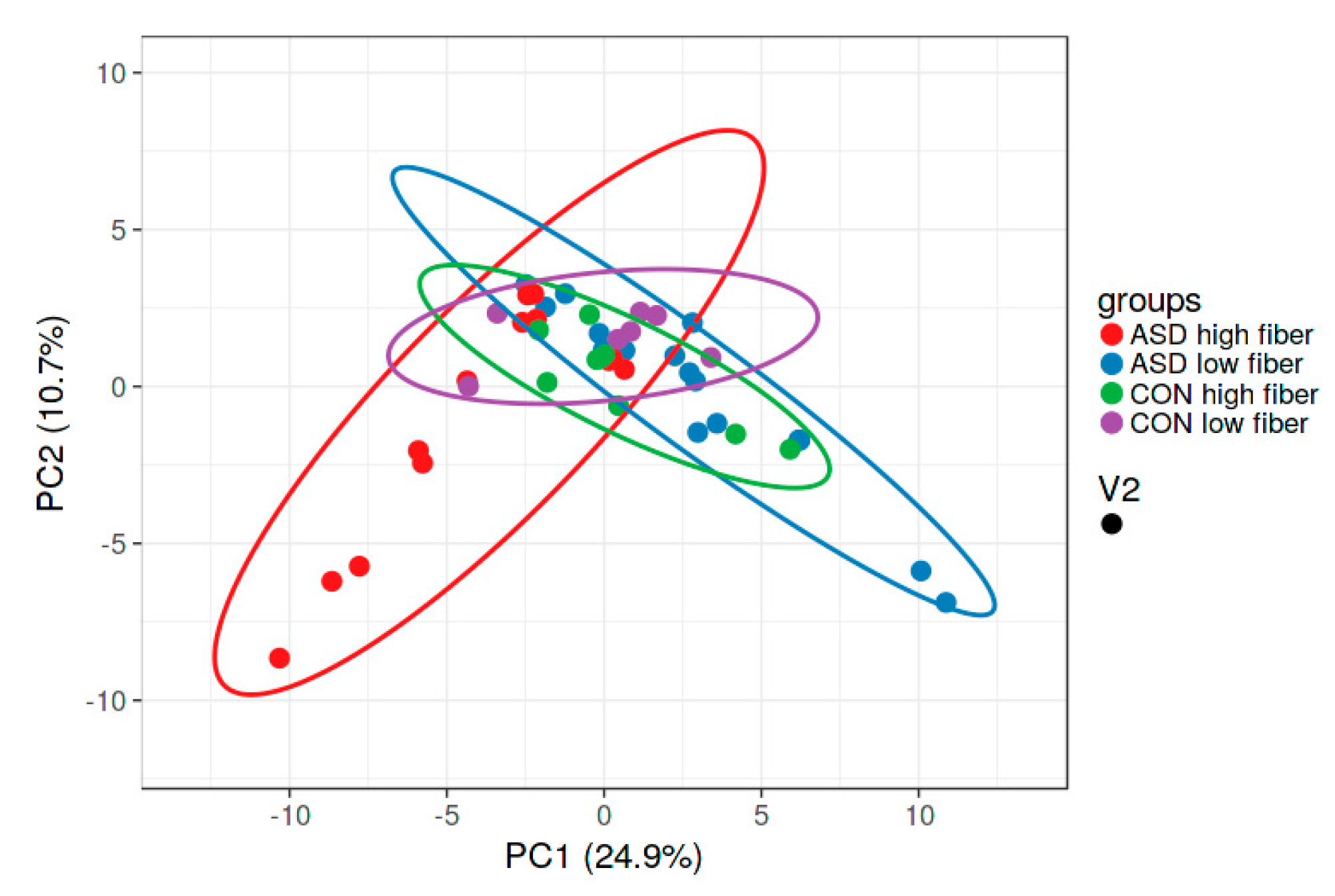
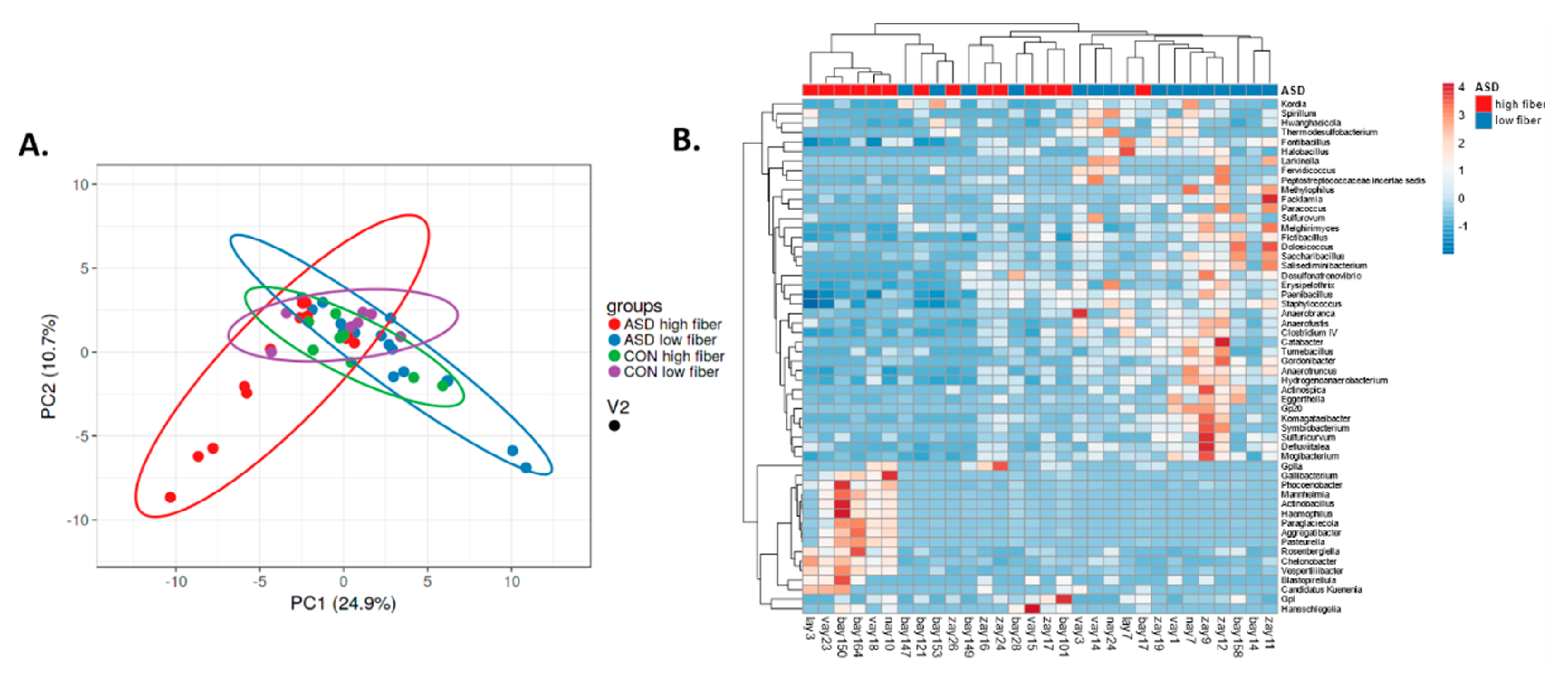
2.1. Comparison of Feeding Habits and Intestinal Microbiota of Children with and without ASD
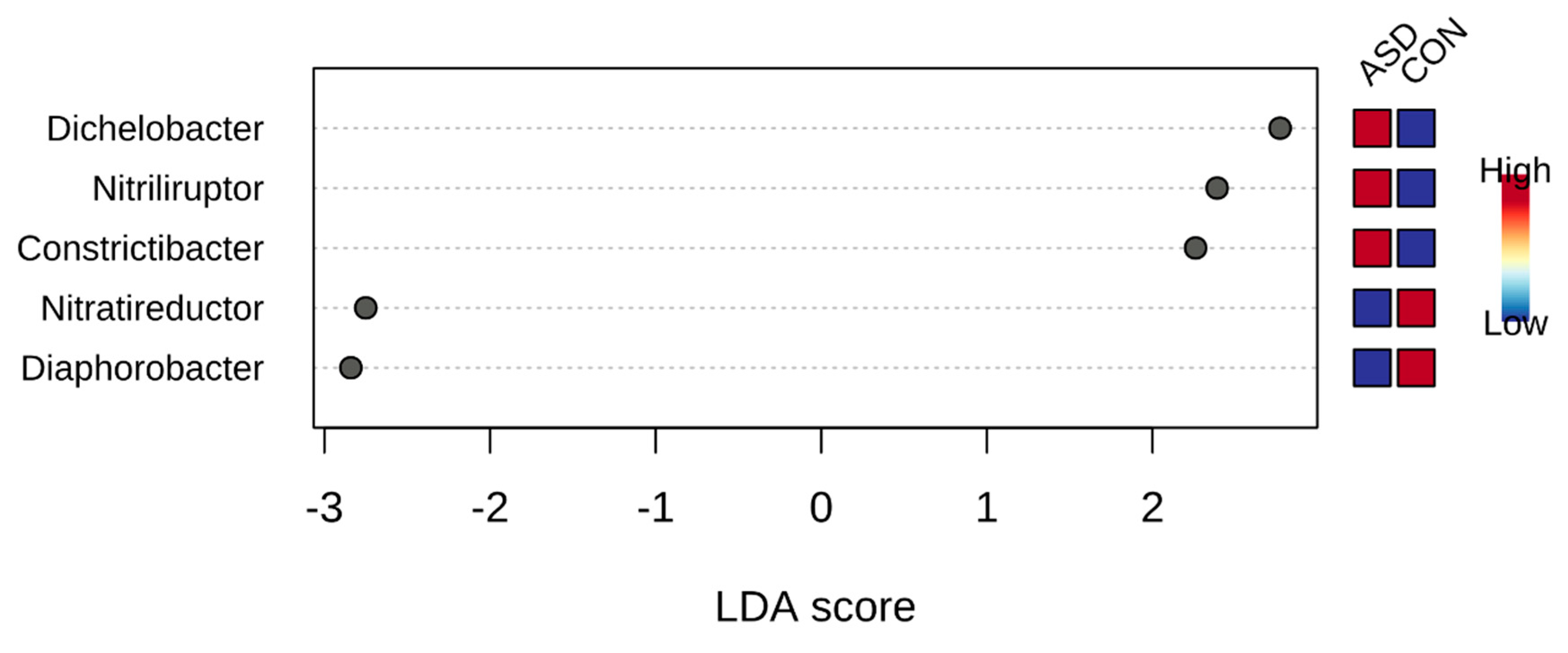
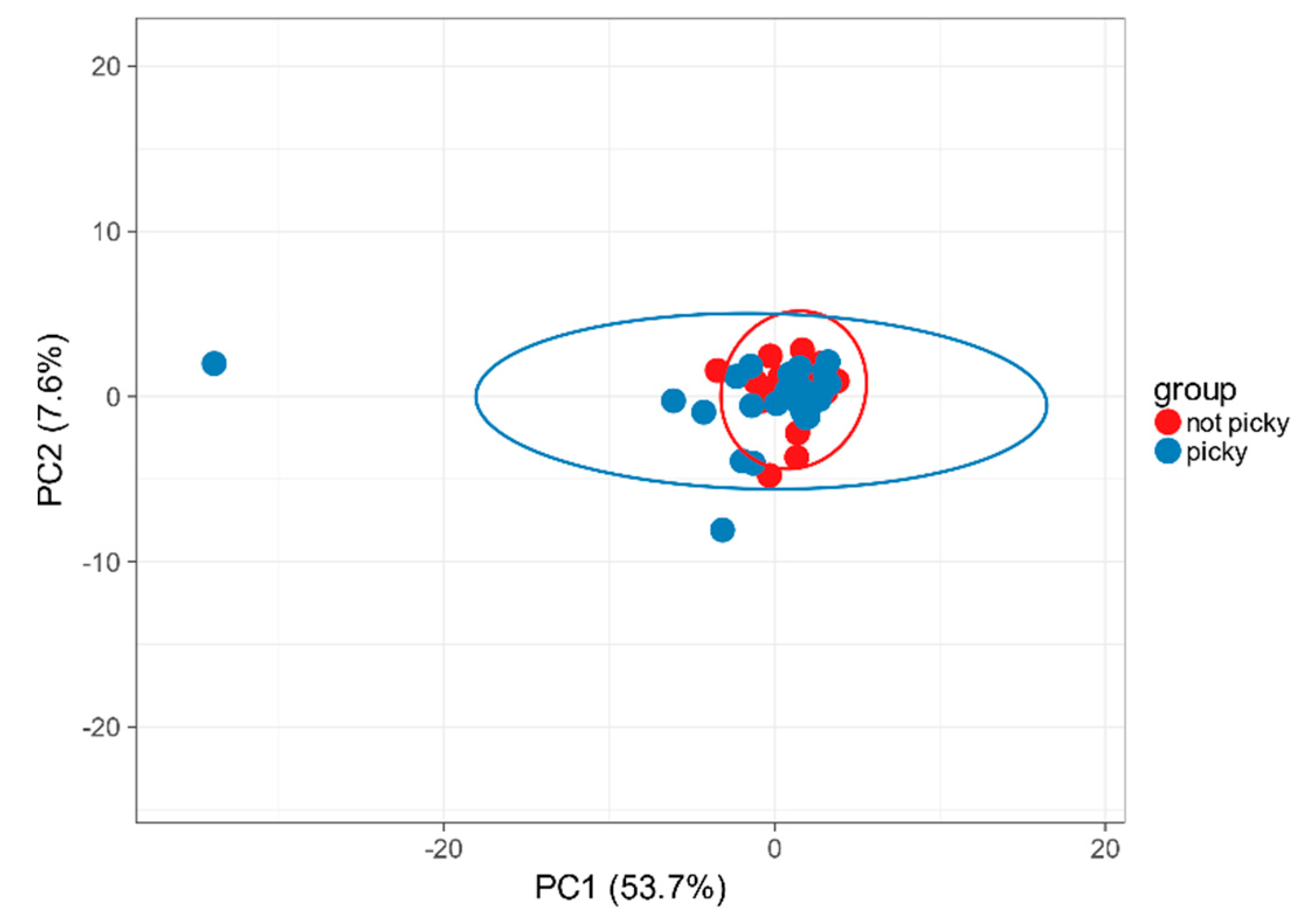
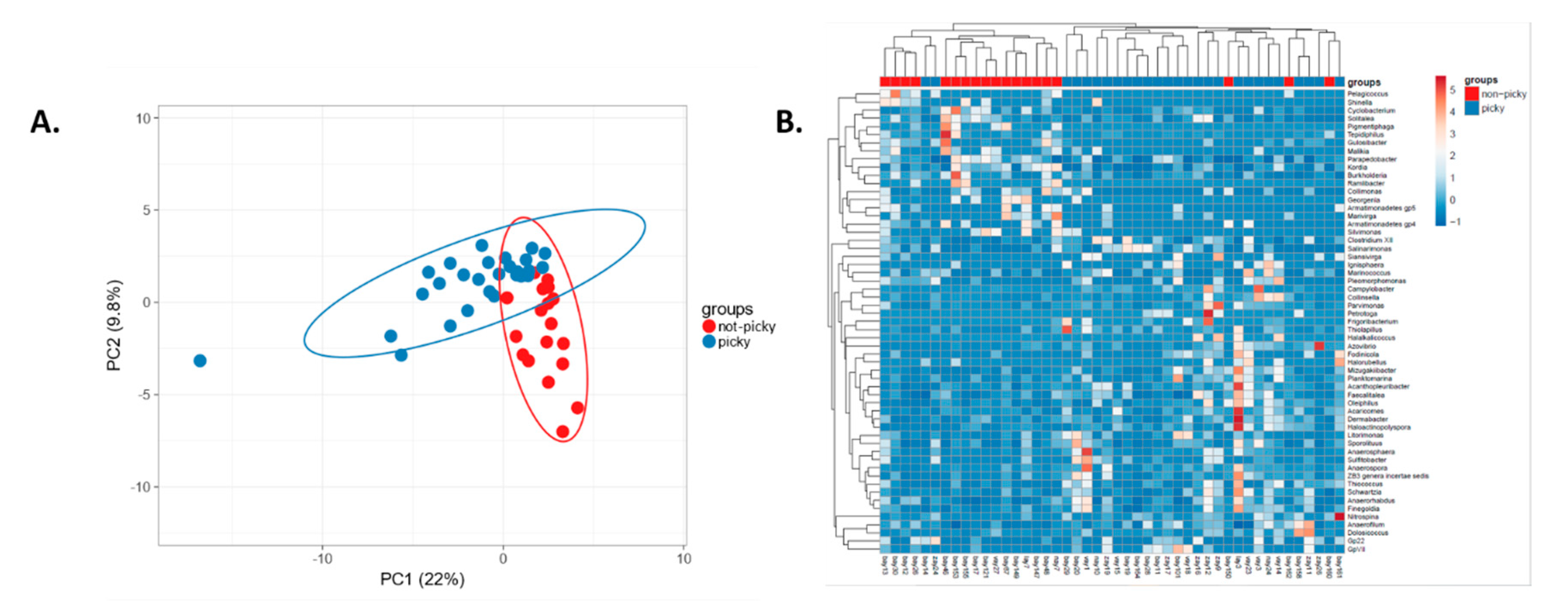
2.2. Food Selectivity Reflects the Faecal Microbiota Composition
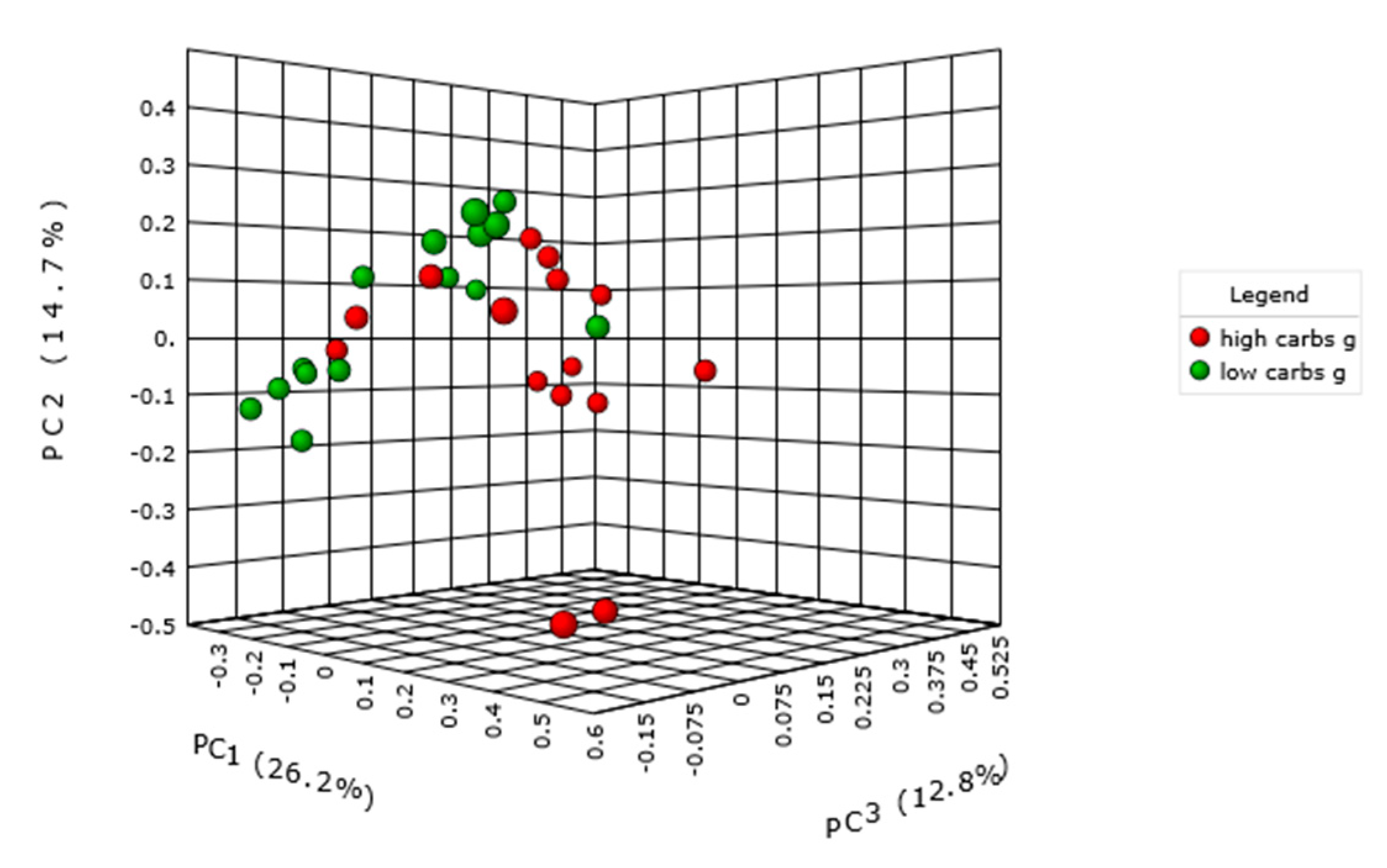
2.3. Carbohydrate Intake and Intestinal Microbiota
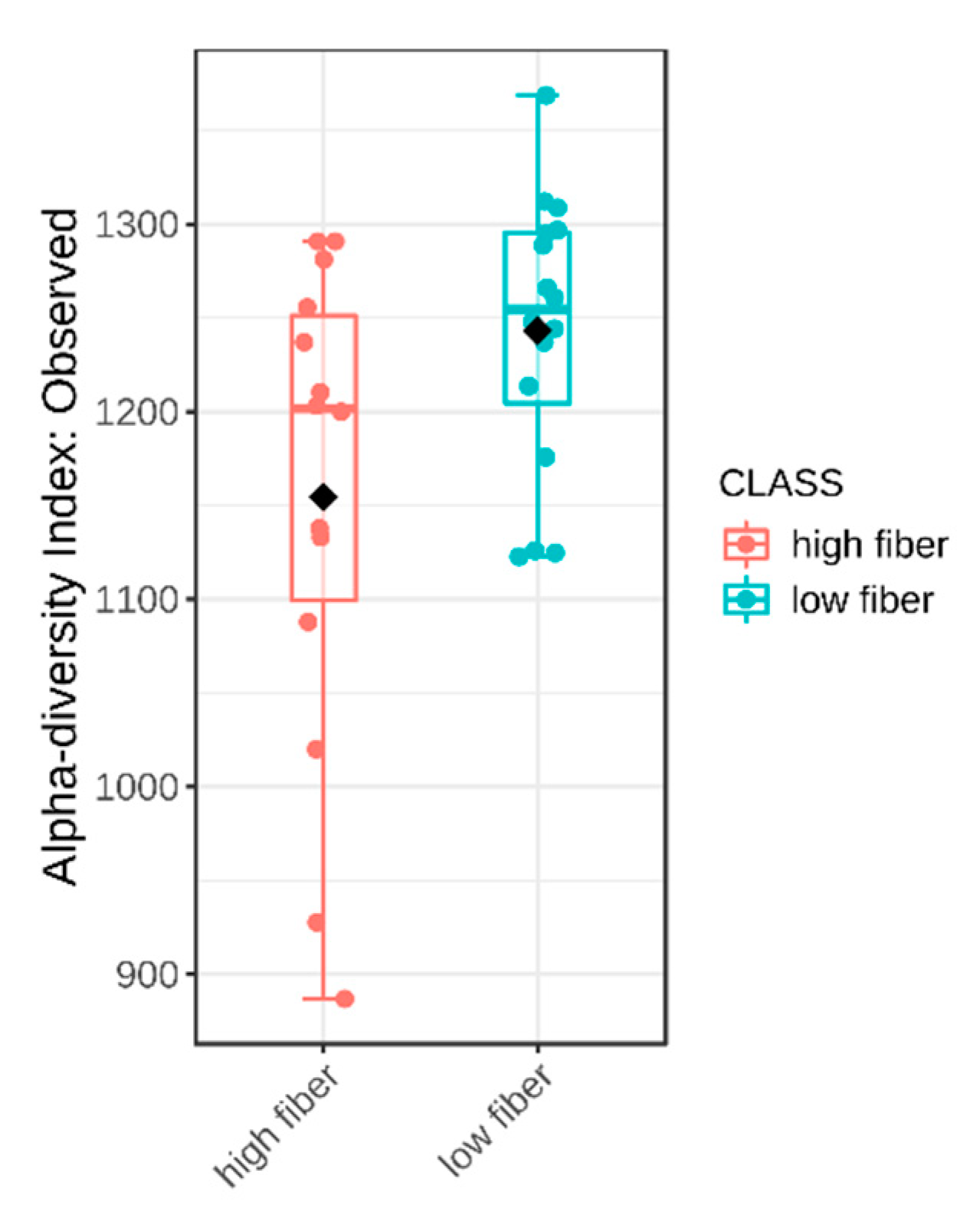
2.4. Dietary Fibre Intake and Intestinal Microbiota
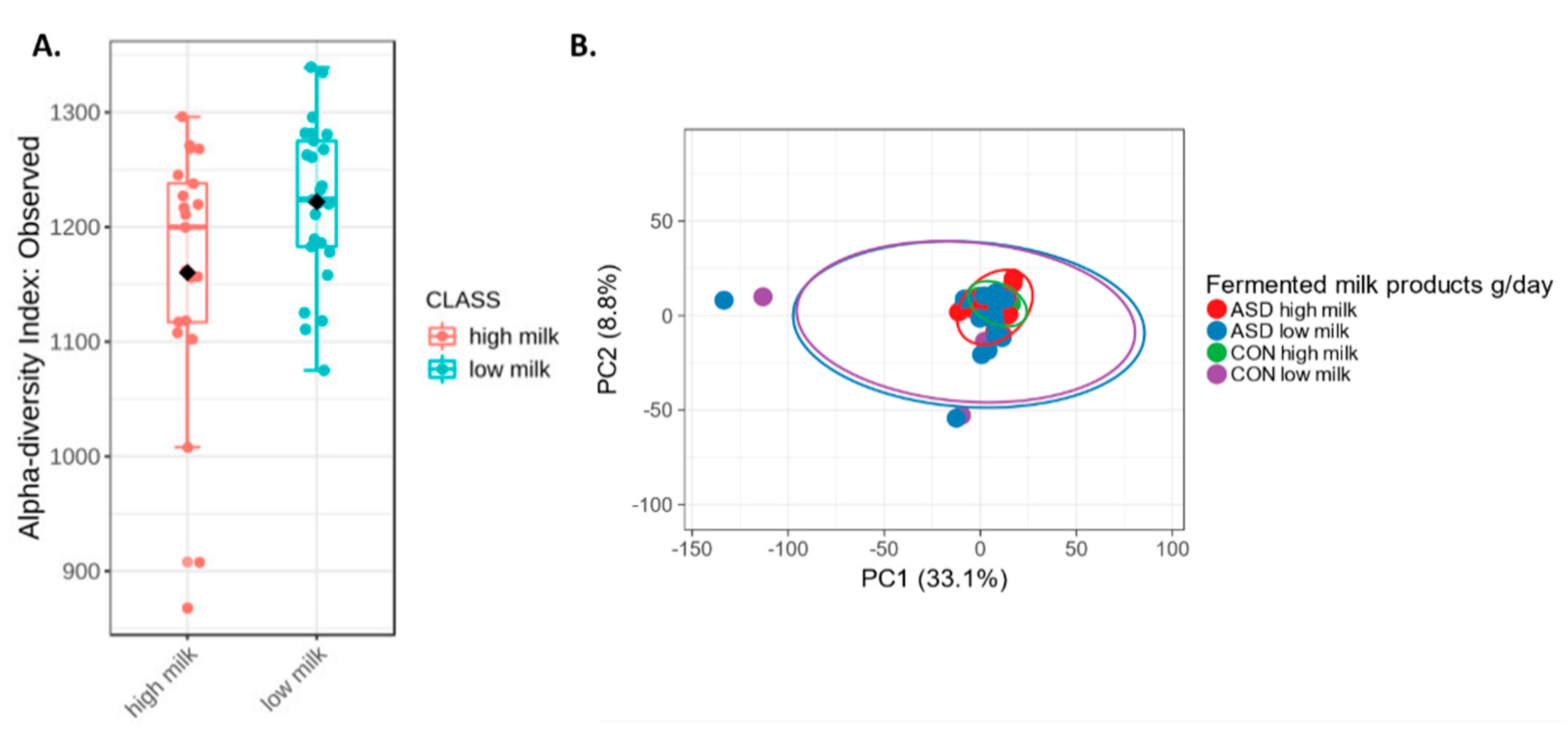
2.5. Fermented Milk Intake and Intestinal Microbiota
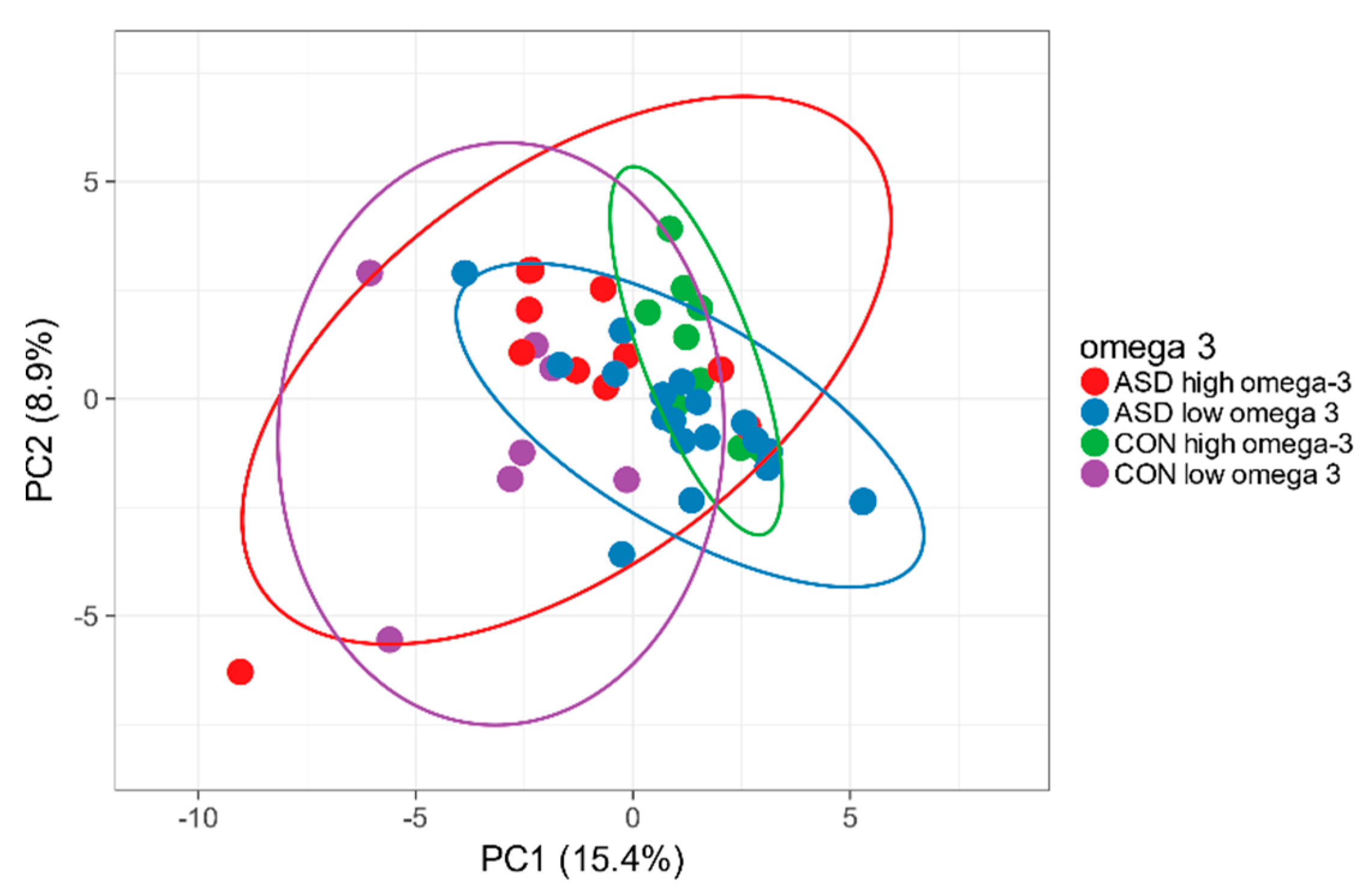
2.6. Omega-3 Intake and Intestinal Microbiota
2.7. Animal vs. Plant Protein Intake
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cermak, S.A.; Curtin, C.; Bandini, L.G. Food selectivity and sensory sensitivity in children with autism spectrum disorders. J. Am. Diet. Assoc. 2010, 110, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Marí-Bauset, S.; Zazpe, I.; Mari-Sanchis, A.; Llopis-González, A.; Morales-Suárez-Varela, M. Food selectivity in autism spectrum disorders: A systematic review. J. Child. Neurol. 2014, 29, 1554–1561. [Google Scholar] [CrossRef] [PubMed]
- Karhu, E.; Zukerman, R.; Eshraghi, R.S.; Mittal, J.; Deth, R.C.; Castejon, A.M.; Trivedi, M.; Mittal, R.; Eshraghi, A.A. Nutritional interventions for autism spectrum disorder. Nutr. Rev. 2019, nuz092. [Google Scholar] [CrossRef] [PubMed]
- Hyman, S.L.; Stewart, P.A.; Schmidt, B.; Cain, U.; Lemcke, N.; Foley, J.T.; Peck, R.; Clemons, T.; Reynolds, A.; Johnson, C.; et al. Nutrient intake from food in children with autism. Pediatrics 2012, 130 (Suppl. 2), S145–S153. [Google Scholar] [CrossRef]
- Esteban-Figuerola, P.; Canals, J.; Fernández-Cao, J.C.; Arija Val, V. Differences in food consumption and nutritional intake between children with autism spectrum disorders and typically developing children: A meta-analysis. Autism 2019, 23, 1079–1095. [Google Scholar] [CrossRef]
- Sharp, W.G.; Postorino, V.; McCracken, C.E.; Berry, R.C.; Criado, K.K.; Burrell, T.L.; Scahill, L. Dietary intake, nutrient status, and growth parameters in children with autism spectrum disorder and severe food selectivity: An Electronic Medical Record Review. J. Acad. Nutr. Diet. 2018, 118, 1943–1950. [Google Scholar] [CrossRef]
- Tomova, A.; Bukovsky, I.; Rembert, E.; Yonas, W.; Alwarith, J.; Barnard, N.D.; Kahleova, H. The Effects of Vegetarian and Vegan Diets on Gut Microbiota. Front. Nutr. 2019, 6, 47. [Google Scholar] [CrossRef]
- Wan, Y.; Wang, F.; Yuan, J.; Li, J.; Jiang, D.; Zhang, J.; Li, H.; Wang, R.; Tang, J.; Huang, T.; et al. Effects of dietary fat on gut microbiota and faecal metabolites, and their relationship with cardiometabolic risk factors: A 6-month randomised controlled-feeding trial. Gut 2019, 68, 1417–1429. [Google Scholar] [CrossRef]
- Watson, H.; Mitra, S.; Croden, F.C.; Taylor, M.; Wood, H.M.; Perry, S.L.; Spencer, J.A.; Quirke, P.; Toogood, G.J.; Lawton, C.L.; et al. A randomised trial of the effect of omega-3 polyunsaturated fatty acid supplements on the human intestinal microbiota. Gut 2018, 67, 1974–1983. [Google Scholar] [CrossRef]
- Berer, K.; Martinez, I.; Walker, A.; Kunkel, B.; Schmitt-Kopplin, P.; Walter, J.; Krishnamoorthy, G. Dietary non-fermentable fibre prevents autoimmune neurological disease by changing gut metabolic and immune status. Sci. Rep. 2018, 8, 10431. [Google Scholar] [CrossRef]
- So, D.; Whelan, K.; Rossi, M.; Morrison, M.; Holtmann, G.; Kelly, J.T.; Shanahan, E.R.; Staudacher, H.M.; Campbell, K.L. Dietary fibre intervention on gut microbiota composition in healthy adults: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2018, 107, 965–983. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhang, X.; Liu, H.; Brown, M.A.; Qiao, S. Dietary protein and gut microbiota composition and function. Curr. Protein Pept. Sci. 2019, 20, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Alcock, J.; Maley, C.C.; Aktipis, C.A. Is eating behavior manipulated by the gastrointestinal microbiota? Evolutionary pressures and potential mechanisms. Bioessays 2014, 36, 940–949. [Google Scholar] [CrossRef] [PubMed]
- Finegold, S.M.; Dowd, S.E.; Gontcharova, V.; Liu, C.; Henley, K.E.; Wolcott, R.D.; Youn, E.; Summanen, P.H.; Granpeesheh, D.; Dixon, D.; et al. Pyrosequencing study of fecal microflora of autistic and control children. Anaerobe 2010, 16, 444–453. [Google Scholar] [CrossRef]
- Tomova, A.; Soltys, K.; Repiska, G.; Palkova, L.; Filcikova, D.; Minarik, G.; Turna, J.; Prochotska, K.; Babinska, K.; Ostatnikova, D. Specificity of gut microbiota in children with autism spectrum disorder in Slovakia and its correlation with astrocytes activity marker and specific behavioural patterns. Physiol. Behav. 2020, 214, 112745. [Google Scholar] [CrossRef]
- Bezawada, N.; Phang, T.H.; Hold, G.L.; Hansen, R. Autism spectrum disorder and the gut microbiota in children: A systematic review. Ann. Nutr. Metab. 2020, 5363. [Google Scholar] [CrossRef]
- Tomova, A.; Pivovarciová, A.; Babinská, K.; Mravec, B. Os črevo-mozog: Význam interakcií medzi črevnou mikrobiotou a mozgom za fyziologických a patologických situácií [Intestinal microbiota and the brain: Multilevel interactions in health and disease]. Ceskoslovenska Fysiol. 2015, 64, 23–34. [Google Scholar]
- Srikantha, P.; Mohajeri, M.H. The possible role of the microbiota-gut-brain-axis in autism spectrum disorder. Int. J. Mol. Sci. 2019, 20, 2115. [Google Scholar] [CrossRef]
- Ristori, M.V.; Quagliariello, A.; Reddel, S.; Ianiro, G.; Vicari, S.; Gasbarrini, A.; Putignani, L. Autism, gastrointestinal symptoms and modulation of gut microbiota by nutritional interventions. Nutrients 2019, 11, 2812. [Google Scholar] [CrossRef]
- Kolodziejczyk, A.A.; Zheng, D.; Elinav, E. Diet-microbiota interactions and personalized nutrition. Nat. Rev. Microbiol. 2019, 17, 742–753. [Google Scholar] [CrossRef]
- Babinska, K.; Pivovarčiová, A.; Filčíková, D.; Tomova, A.; Ostatníková, A. Association of conduct problems and gastrointestinal symptoms in individuals with autism spectrum disorders. Act. Nerv. Super. Rediviva 2016, 58, 69–72. [Google Scholar]
- Bandini, L.G.; Anderson, S.E.; Curtin, C.; Cermak, S.; Evans, E.W.; Scampini, R.; Maslin, M.; Must, A. Food selectivity in children with autism spectrum disorders and typically developing children. J. Pediatr. 2010, 157, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.M. The immune response to Prevotella bacteria in chronic inflammatory disease. Immunology 2017, 151, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Lian, Y.; Zhao, C.; Du, H.; Han, Y.; Gao, W.; Xiao, H.; Zheng, J. Dietary fibers from fruits and vegetables and their health benefits via modulation of gut microbiota. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1514–1532. [Google Scholar] [CrossRef]
- Makki, K.; Deehan, E.C.; Walter, J.; Bäckhed, F. The impact of dietary fiber on gut microbiota in host health and disease. Cell Host Microbe 2018, 23, 705–715. [Google Scholar] [CrossRef]
- Sonnenburg, E.D.; Smits, S.A.; Tikhonov, M.; Higginbottom, S.K.; Wingreen, N.S.; Sonnenburg, J.L. Diet-induced extinctions in the gut microbiota compound over generations. Nature 2016, 529, 212–215. [Google Scholar] [CrossRef]
- Dahl, W.J.; Stewart, M.L. Position of the academy of nutrition and dietetics: Health implications of dietary fiber. J. Acad. Nutr. Diet. 2015, 115, 1861–1870. [Google Scholar] [CrossRef]
- Saghazadeh, A.; Ataeinia, B.; Keynejad, K.; Abdolalizadeh, A.; Hirbod-Mobarakeh, A.; Rezaei, N. A meta-analysis of pro-inflammatory cytokines in autism spectrum disorders: Effects of age, gender, and latitude. J. Psychiatr. Res. 2019, 115, 90–102. [Google Scholar] [CrossRef]
- Sprague, R. Fusicatenibacter is associated with kefir drinking. bioRxiv 2017, 218313. [Google Scholar] [CrossRef]
- Aoyagi, Y.; Amamoto, R.; Park, S.; Honda, Y.; Shimamoto, K.; Kushiro, A.; Tsuji, H.; Matsumoto, H.; Shimizu, K.; Miyazaki, K.; et al. Independent and interactive effects of habitually ingesting fermented milk products containing. Front. Microbiol. 2019, 10, 1477. [Google Scholar] [CrossRef]
- Nierychlo, M.; Andersen, K.S.; Xu, Y.; Green, N.; Albertsen, M.; Dueholm, M.S.; Nielsen, P.H. Species-level microbiome composition of activated sludge–introducing MiDAS 3 ecosystem-specific reference database and taxonomy. BioRxiv 2019, 842393. [Google Scholar] [CrossRef]
- Unno, T.; Choi, J.H.; Hur, H.G.; Sadowsky, M.J.; Ahn, Y.T.; Huh, C.S.; Kim, G.B.; Cha, C.J. Changes in human gut microbiota influenced by probiotic fermented milk ingestion. J. Dairy Sci. 2015, 98, 3568–3576. [Google Scholar] [CrossRef] [PubMed]
- González, S.; Fernández-Navarro, T.; Arboleya, S.; de Los Reyes-Gavilán, C.G.; Salazar, N.; Gueimonde, M. Fermented dairy foods: Impact on intestinal microbiota and health-linked biomarkers. Front. Microbiol. 2019, 10, 1046. [Google Scholar] [CrossRef] [PubMed]
- Costantini, L.; Molinari, R.; Farinon, B.; Merendino, N. Impact of omega-3 fatty acids on the gut microbiota. Int. J. Mol. Sci. 2017, 18, 2645. [Google Scholar] [CrossRef]
- Berger, F.K.; Schwab, N.; Glanemann, M.; Bohle, R.M.; Gärtner, B.; Groesdonk, H.V. Flavonifractor (Eubacterium) plautii bloodstream infection following acute cholecystitis. IDCases 2018, 14, e00461. [Google Scholar] [CrossRef]
- Gupta, A.; Dhakan, D.B.; Maji, A.; Saxena, R.; Vishnu Prasoodanan, P.K.; Mahajan, S.; Pulikkan, J.; Kurian, J.; Gomez, A.M.; Scaria, J.; et al. Association of Flavonifractor plautii, a flavonoid-degrading bacterium, with the gut microbiome of colorectal cancer patients in India. mSystems 2019, 4. [Google Scholar] [CrossRef]
- Berding, K.; Holscher, H.D.; Arthur, A.E.; Donovan, S.M. Fecal microbiome composition and stability in 4- to 8-year old children is associated with dietary patterns and nutrient intake. J. Nutr. Biochem. 2018, 56, 165–174. [Google Scholar] [CrossRef]
- Lord, C.; Rutter, M.; Dilavore, P.C.; Risi, S.; Gotham, K.; Bishop, S. Autism Diagnostic Observation Schedule. West. Psychol. Serv. Torrance CA 2012. [Google Scholar] [CrossRef]
- Lord, C.; Pickles, A.; McLennan, J.; Rutter, M.; Bregman, J.; Folstein, S.; Fombonne, E.; Leboyer, M.; Minshew, N. Diagnosing autism: Analyses of data from the Autism Diagnostic Interview. J. Autism Dev. Disord. 1997, 27, 501–517. [Google Scholar] [CrossRef]
- Sevcikova, L.; Rovny, I.; Novakova, J.; Hamada, J.; Tatara, M.; Janechova, H. Body development of infants and children in Slovakia. Results of the 6th National Survey in 2001. Bratisl. Natl. Inst. Public Health Slovak Repub. 2004, 124–143. [Google Scholar]
- Metsalu, T.; Vilo, J. ClustVis: A web tool for visualizing clustering of multivariate data using principal component analysis and heatmap. Nucleic Acids Res. 2015, 43, W566–W570. [Google Scholar] [CrossRef] [PubMed]
- Chong, J.; Liu, P.; Zhou, G.; Xia, J. Using MicrobiomeAnalyst for comprehensive statistical, functional, and meta-analysis of microbiome data. Nat. Protoc. 2020, 15, 799–821. [Google Scholar] [CrossRef] [PubMed]
- Dhariwal, A.; Chong, J.; Habib, S.; King, I.L.; Agellon, L.B.; Xia, J. MicrobiomeAnalyst: A web-based tool for comprehensive statistical, visual and meta-analysis of microbiome data. Nucleic Acids Res. 2017, 45, W180–W188. [Google Scholar] [CrossRef] [PubMed]
| ASD | Controls | p | |
|---|---|---|---|
| N (all boys) | 46 | 16 | |
| Age range (years) | 4.0–8.5 | 2.8–9.15 | |
| Age (mean ± SD) | 6.3 ± 1,5 | 5.1 ± 1.7 | 0.017 |
| BMI (kg.m-2) | 17.1 ± 3.7 | 16.2 ± 2.0 | 0.746 |
| SDS BMI | 0.12 ± 0.98 | 0.50 ± 1.56 | 0.781 |
| Mealtime problems (% of subjects) of that | 76.1% | 46.8% | 0.017 |
| Food selectivity | 58.7% | 25% | 0.020 |
| Aggressive behaviour | 34.8% | 0.0% | 0.006 |
| Stereotyped behaviour | 32.6% | 18.8% | 0.168 |
| Presence of GI symptoms (% of subjects) of that | 89.4% | 87.5% | 0.838 |
| Constipation | 28.9% | 0.0% | 0.014 |
| Diarrhoea | 2.2% | 6.3% | 0.437 |
| Bloating | 35.6% | 56.3% | 0.148 |
| Abdominal pain | 35.6% | 25.0% | 0.439 |
| Pain upon defecation | 17.8% | 6.3% | 0.284 |
| Dietary restrictions initiated by parents | 17.4% | 0.0% | 0.099 |
| Food supplement intake | 76.1% | 93.8% | 0.123 |
| ASD | Controls | p | |
|---|---|---|---|
| N (all boys) | 30 | 16 | |
| Energy and nutrients (mean ± SD) | |||
| Energy, kJ/day | 5506 ± 254 | 5666 ± 344 | 0.711 |
| Proteins, g/day | 45.5 ± 0.4 | 50.2 ± 0.9 | 0.273 |
| Animal/plant protein | 1.63 ± 0.03 | 1.76 ± 0.04 | 0.557 |
| Fats, g/day | 53.1 ± 0.6 | 55.5 ± 1.0 | 0.656 |
| Carbohydrates, g/day | 173.1 ± 1.3 | 173.9 ± 2.7 | 0.952 |
| % energy from protein | 14.2 ± 0.3 | 15.0 ± 0.47 | 0.148 |
| % energy from fat | 35.7 ± 1.1 | 36.8 ± 1.1 | 0.536 |
| % energy from carbohydrates | 50.1 ± 1.2 | 48.1 ± 1.3 | 0.306 |
| Dietary fibre, g/ day | 9.95 ± 0.56 | 11.7 ± 1.00 | 0.114 |
| Omega-3 fatty acids (g/day) of that | 0.75 ± 0.01 | 0.81 ± 0.01 | 0.473 |
| Linolenic acid (g/day) | 0.73 ± 0.01 | 0.78 ± 0.01 | 0.548 |
| Eicosapentaenoic acid (mg/day) | 10.0 ± 0.60 | 20.0 ± 1.00 | 0.325 |
| Docosahexaenoic acid (mg/day) | 7.0 ± 0.20 | 13.0 ± 0.40 | 0.022 |
| Foods (median, 95% CI) | |||
| Bakery products white (g/day) | 62.5 (38.7–90.0) | 60.5 (21.4-65.4) | 0.037 |
| Bakery products wholegrain (g/day) | 4.8 (0.0–21.4) | 21.4 (7.1-53.6) | 0.102 |
| Fermented milk products (g/day) | 125.9 (19.3–244.2) | 67.5 (42.9–109.3) | 0.393 |
| Fresh fruit (g/day) | 84.1 (47.8–133.9) | 199.1 (126.0–237.3) | 0.001 |
| Fresh vegetables (g/day) | 16.6 (2.9–26.8) | 61.4 (36.2–95.1) | 0.000 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomova, A.; Soltys, K.; Kemenyova, P.; Karhanek, M.; Babinska, K. The Influence of Food Intake Specificity in Children with Autism on Gut Microbiota. Int. J. Mol. Sci. 2020, 21, 2797. https://doi.org/10.3390/ijms21082797
Tomova A, Soltys K, Kemenyova P, Karhanek M, Babinska K. The Influence of Food Intake Specificity in Children with Autism on Gut Microbiota. International Journal of Molecular Sciences. 2020; 21(8):2797. https://doi.org/10.3390/ijms21082797
Chicago/Turabian StyleTomova, Aleksandra, Katarina Soltys, Petra Kemenyova, Miloslav Karhanek, and Katarina Babinska. 2020. "The Influence of Food Intake Specificity in Children with Autism on Gut Microbiota" International Journal of Molecular Sciences 21, no. 8: 2797. https://doi.org/10.3390/ijms21082797
APA StyleTomova, A., Soltys, K., Kemenyova, P., Karhanek, M., & Babinska, K. (2020). The Influence of Food Intake Specificity in Children with Autism on Gut Microbiota. International Journal of Molecular Sciences, 21(8), 2797. https://doi.org/10.3390/ijms21082797






