Comparative Proteomic Analysis by iTRAQ Reveals that Plastid Pigment Metabolism Contributes to Leaf Color Changes in Tobacco (Nicotiana tabacum) during Curing
Abstract
1. Introduction
2. Results
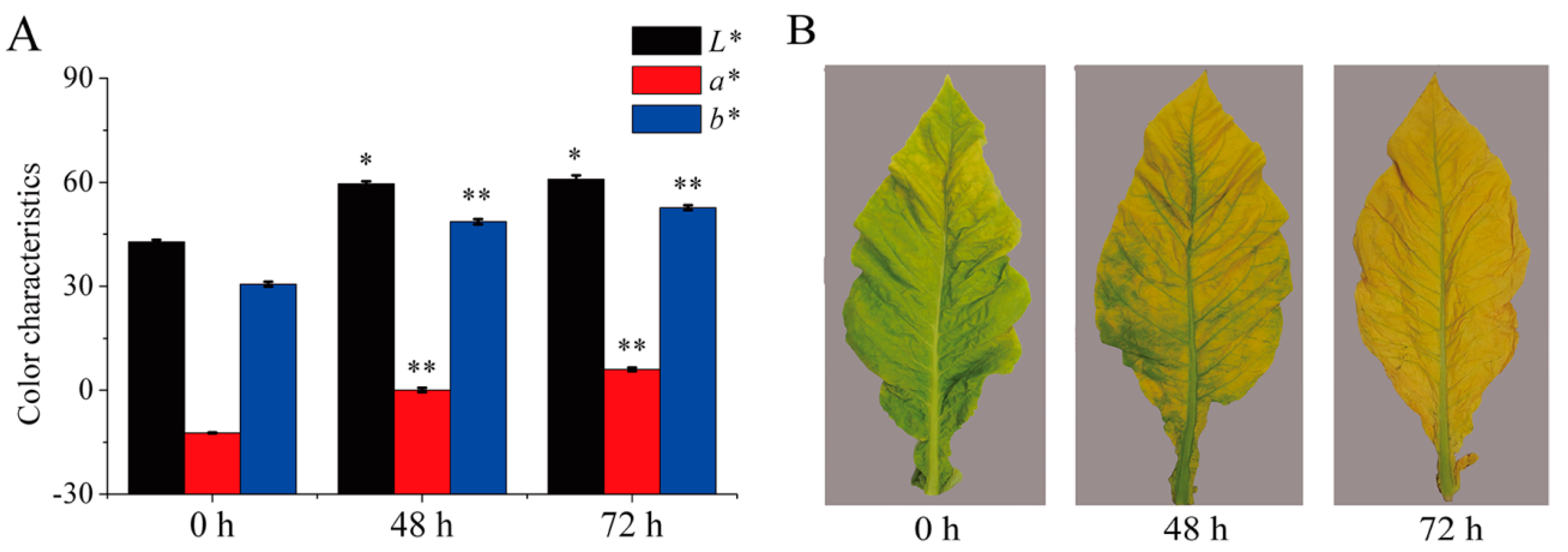
2.1. Color and Phenotypic Changes of Tobacco Leaves during Curing
2.2. Ultrastructural Observations of Tobacco Leaves during Curing
2.3. Physiological Attributes of Tobacco Leaves during Curing
2.4. Pigment Degradation Products Analysis in Tobacco Leaves during Curing
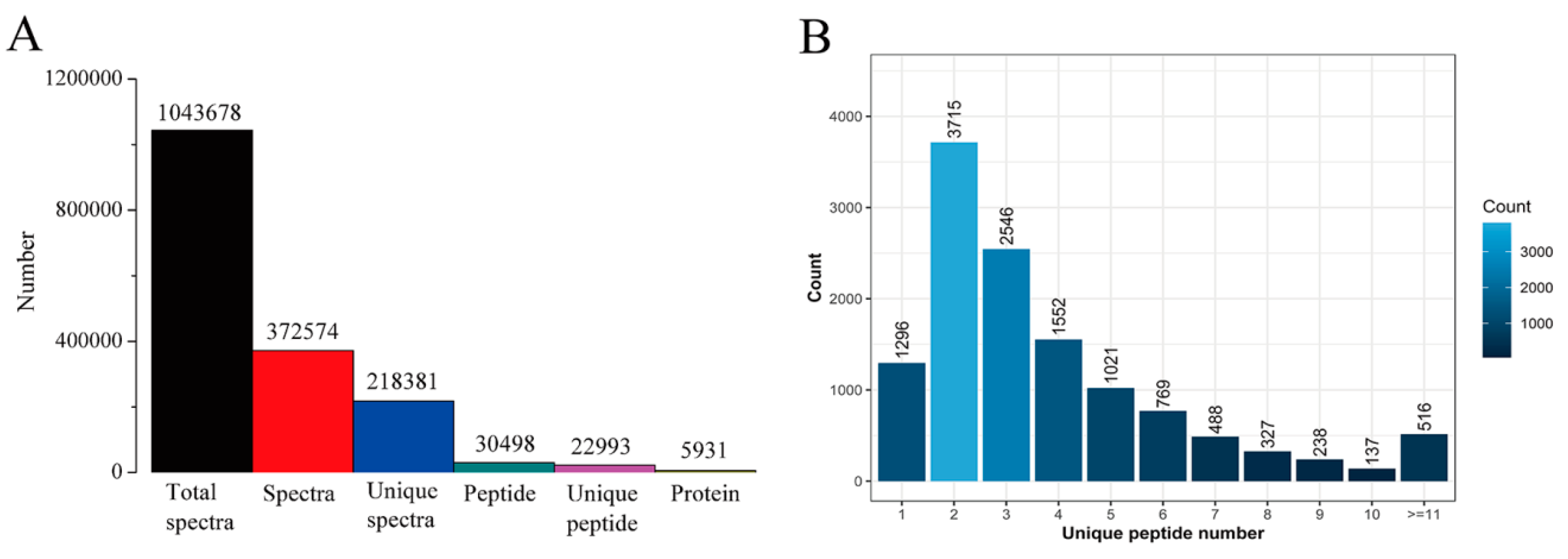
2.5. Protein Profile Analysis of Tobacco Leaves Using iTRAQ
2.6. DEPs Identified and Functional Analysis
2.7. DEPs Involved in Carotenoid and Chlorophyll Metabolism
2.8. Validation of iTRAQ Data by qRT-PCR
3. Discussion
3.1. Leaf Color Change is Determined by the Carotenoid and Chlorophyll Content
3.2. Effect of Cell Ultrastructure Damage on Pigment Metabolism and Leaf Color Change
3.3. Role of Physiological Parameters in Pigment Metabolism and Leaf Color Change
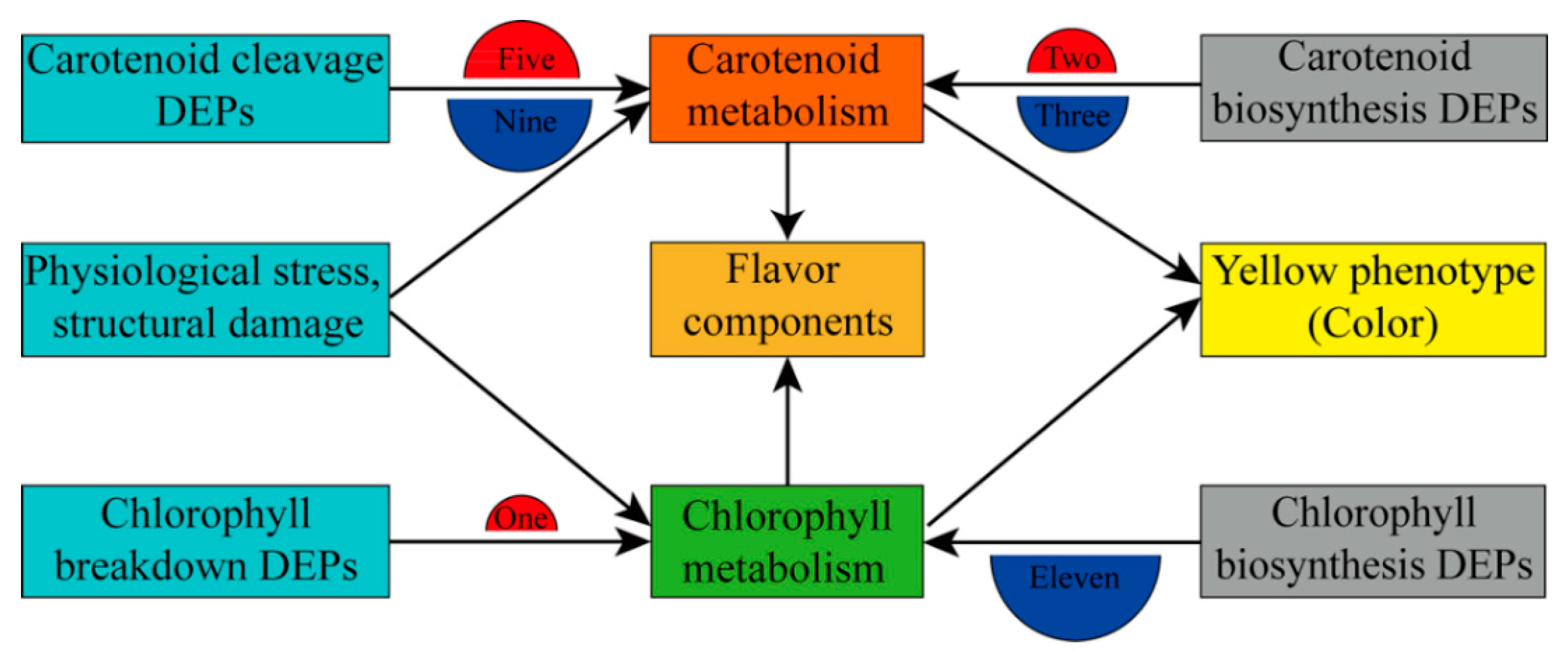
3.4. Role of DEPs in Carotenoid and Chlorophyll Metabolism and Color Change
4. Materials and Methods
4.1. Plant Material and Sampling
4.2. Color Analysis
4.3. Ultrastructural Observation
4.4. Physiological Measurements
4.5. Pigment Degradation Products Analysis
4.6. Protein Extraction
4.7. iTRAQ Labeling and Strong Cation Exchange (SCX) Fractionation
4.8. LC-ESI-MS/MS Analysis
4.9. iTRAQ protein Identification and Quantification
4.10. Bioinformatics Analysis
4.11. RNA Extraction and qRT-PCR Analysis
4.12. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ma, C.; Liang, B.; Chang, B.; Liu, L.; Yan, J.; Yang, Y.; Zhao, Z. Transcriptome Profiling Reveals Transcriptional Regulation by DNA Methyltransferase Inhibitor 5-Aza-2′-Deoxycytidine Enhancing Red Pigmentation in Bagged “Granny Smith” Apples (Malus domestica). Int. J. Mol. Sci. 2018, 19, 3133. [Google Scholar] [CrossRef]
- Chen, J.; Funnell, K.A.; Lewis, D.H.; Eason, J.R.; Woolley, D.J. Relationship between changes in colour and pigment content during spathe regreening of Zantedeschia ‘Best Gold’. Postharvest Biol. Technol. 2012, 67, 124–129. [Google Scholar] [CrossRef]
- Wei, X.; Deng, X.; Cai, D.; Ji, Z.; Wang, C.; Yu, J.; Li, J.; Chen, S. Decreased tobacco-specific nitrosamines by microbial treatment with Bacillus amyloliquefaciens DA9 during the air-curing process of burley tobacco. J. Agric. Food Chem. 2014, 62, 12701–12706. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Zhang, X.; Li, L.; Aghdam, M.S.; Wei, X.; Liu, J.; Xu, Y.; Luo, Z. Elevated CO2 delayed the chlorophyll degradation and anthocyanin accumulation in postharvest strawberry fruit. Food Chem. 2019, 285, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Deng, L.; Yin, B.; Yao, S.; Zeng, K. Effects of blue LED light irradiation on pigment metabolism of ethephon-degreened mandarin fruit. Postharvest Biol. Technol. 2017, 134, 45–54. [Google Scholar] [CrossRef]
- Yuan, H.; Zhang, J.; Nageswaran, D.; Li, L. Carotenoid metabolism and regulation in horticultural crops. Hortic. Res. 2015, 2, 15036. [Google Scholar] [CrossRef]
- Fraser, P.D.; Bramley, P.M. The biosynthesis and nutritional uses of carotenoids. Prog. Lipid Res. 2004, 43, 228–265. [Google Scholar] [CrossRef]
- Ma, C.; Cao, J.; Li, J.; Zhou, B.; Tang, J.; Miao, A. Phenotypic, histological and proteomic analyses reveal multiple differences associated with chloroplast development in yellow and variegated variants from Camellia sinensis. Sci. Rep. 2016, 6, 33369. [Google Scholar] [CrossRef]
- Davison, P.A.; Hunter, C.H.; Horton, P. Overexpression of β-carotene hydroxylase enhances stress tolerance in Arabidopsis. Nature 2002, 418, 203–206. [Google Scholar] [CrossRef]
- Karppinen, K.; Zoratti, L.; Sarala, M.; Carvalho, E.; Hirsimäki, J.; Mentula, H.; Martens, S.; Häggman, H.; Jaakola, L. Carotenoid metabolism during bilberry (Vaccinium myrtillus L.) fruit development under different light conditions is regulated by biosynthesis and degradation. BMC Plant Biol. 2016, 16, 95. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, C.; Dong, B.; Fu, J.; Hu, S.; Zhao, H. Carotenoid Accumulation and Its Contribution to Flower Coloration of Osmanthus fragrans. Front. Plant Sci. 2018, 9, 1499. [Google Scholar] [CrossRef] [PubMed]
- Leng, X.; Wang, P.; Wang, C.; Zhu, X.; Li, X.; Li, H.; Mu, Q.; Li, A.; Liu, Z.; Fang, J. Genome-wide identification and characterization of genes involved in carotenoid metabolic in three stages of grapevine fruit development. Sci. Rep. 2017, 7, 4216. [Google Scholar] [CrossRef] [PubMed]
- Gayen, D.; Ali, N.; Sarkar, S.N.; Datta, S.K.; Datta, K. Down-regulation of lipoxygenase gene reduces degradation of carotenoids of golden rice during storage. Planta 2015, 242, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Zhai, S.; Xia, X.; He, Z. Carotenoids in Staple Cereals: Metabolism, Regulation, and Genetic Manipulation. Front. Plant Sci. 2016, 7, 1197. [Google Scholar] [CrossRef] [PubMed]
- Zelena, K.; Hardebusch, B.; Hülsdau, B.; Berger, R.G.; Zorn, H. Generation of Norisoprenoid Flavors from Carotenoids by Fungal Peroxidases. J. Agric. Food Chem. 2009, 57, 9951–9955. [Google Scholar] [CrossRef]
- Wahlberg, I.; Karlsson, K.; Austin, D.J.; Junker, N.; Roeraade, J.; Enzell, C.R.; Johnson, W.H. Effects of flue-curing and ageing on the volatile, neutral and acidic constituents of Virginia tobacco. Phytochemistry 1977, 16, 1217–1231. [Google Scholar] [CrossRef]
- Hauenstein, M.; Christ, B.; Das, A.; Aubry, S.; Hörtensteiner, S. A Role for TIC55 as a Hydroxylase of Phyllobilins, the Products of Chlorophyll Breakdown during Plant Senescence. Plant Cell 2016, 28, 2510–2527. [Google Scholar] [CrossRef]
- Kräutler, B. Breakdown of Chlorophyll in Higher Plants—Phyllobilins as Abundant, Yet Hardly Visible Signs of Ripening, Senescence, and Cell Death. Angew. Chem. Int. Ed. 2016, 55, 4882–4907. [Google Scholar] [CrossRef]
- Barry, C.S.; McQuinn, R.P.; Chung, M.Y.; Besuden, A.; Giovannoni, J.J. Amino acid substitutions in homologs of the STAY-GREEN protein are responsible for the green-flesh and chlorophyll retainer mutations of tomato and pepper. Plant Physiol. 2008, 147, 179–187. [Google Scholar] [CrossRef]
- Moser, S.; Müller, T.; Holzinger, A.; Lutz, C.; Jockusch, S.; Turro, N.J.; Kräutler, B. Fluorescent chlorophyll catabolites in bananas light up blue halos of cell death. Proc. Natl. Acad. Sci. USA 2009, 106, 15538–15543. [Google Scholar] [CrossRef]
- Banala, S.; Moser, S.; Müller, T.; Kreutz, C.; Holzinger, A.; Lütz, C.; Kräutler, B. Hypermodified fluorescent chlorophyll catabolites: Source of blue luminescence in senescent leaves. Angew. Chem. Int. Ed. 2010, 49, 5174–5177. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wurst, K.; Jockusch, S.; Gruber, K.; Podewitz, M.; Liedl, K.R.; Kräutler, B. Chlorophyll-Derived Yellow Phyllobilins of Higher Plants as Medium-Responsive Chiral Photo switches. Angew. Chem. Int. Ed. 2016, 55, 15760–15765. [Google Scholar] [CrossRef] [PubMed]
- Weston, T.J. Biochemical characteristics of tobacco leaves during flue-curing. Phytochemistry 1968, 7, 921–930. [Google Scholar] [CrossRef]
- Glover, B.J.; Whitney, H.M. Structural colour and iridescence in plants: The poorly studied relations of pigment colour. Ann. Bot. 2010, 105, 505–511. [Google Scholar] [CrossRef]
- Iorizzo, M.; Ellison, S.; Senalik, D.; Zeng, P.; Satapoomin, P.; Huang, J.; Bowman, M.; Iovene, M.; Sanseverino, W.; Cavagnaro, P.; et al. A high-quality carrot genome assembly provides new insights into carotenoid accumulation and asterid genome evolution. Nat. Genet. 2016, 48, 657–666. [Google Scholar] [CrossRef]
- Galpaz, N.; Ronen, G.; Khalfa, Z.; Zamir, D.; Hirschberg, J.A. Chromoplast-specific carotenoid biosynthesis pathway is revealed by cloning of the tomato white-flower locus. Plant Cell 2006, 18, 1947–1960. [Google Scholar] [CrossRef]
- Yu, J.J.; Zhang, J.Z.; Zhao, Q.; Liu, Y.L.; Chen, S.X.; Guo, H.L.; Shi, L.; Dai, S.J. Proteomic Analysis Reveals the Leaf Color Regulation Mechanism in Chimera Hosta “Gold Standard” Leaves. Int. J. Mol. Sci. 2016, 17, 346. [Google Scholar] [CrossRef]
- Chu, P.; Yan, G.X.; Yang, Q.; Zhai, L.N.; Zhang, C.; Zhang, F.Q.; Guan, R.Z. iTRAQ-based quantitative proteomics analysis of Brassica napus leaves reveals pathways associated with chlorophyll deficiency. J. Proteom. 2015, 113, 244–259. [Google Scholar] [CrossRef]
- Lin, M.; Fang, J.; Qi, X.; Li, Y.; Chen, J.; Sun, L.; Zhong, Y. iTRAQ-based quantitative proteomic analysis reveals alterations in the metabolism of Actinidia arguta. Sci. Rep. 2017, 7, 5670. [Google Scholar] [CrossRef]
- Wu, H.; Shi, N.; An, X.; Liu, C.; Fu, H.; Cao, L.; Feng, Y.; Sun, D.; Zhang, L. Candidate Genes for Yellow Leaf Color in Common Wheat (Triticum aestivum L.) and Major Related Metabolic Pathways according to Transcriptome Profiling. Int. J. Mol. Sci. 2018, 19, 1594. [Google Scholar] [CrossRef]
- Yang, H.; Zhao, L.; Zhao, S.; Wang, J.; Shi, H. Biochemical and transcriptomic analyses of drought stress responses of LY1306 tobacco strain. Sci. Rep. 2017, 7, 17442. [Google Scholar] [CrossRef] [PubMed]
- Sierro, N.; Battey, J.N.; Ouadi, S.; Bakaher, N.; Bovet, L.; Willig, A.; Goepfert, S.; Peitsch, M.C.; Ivanov, N.V. The tobacco genome sequence and its comparison with those of tomato and potato. Nat. Commun. 2014, 5, 3833. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y. Towards systems biological understanding of leaf senescence. Plant Mol. Biol. 2013, 82, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Lee, S.J.; Min, C.W.; Kim, S.W.; Park, K.H.; Bae, D.W.; Lee, B.W.; Agrawal, G.K.; Rakwal, R.; Kim, S.T. Coupling of gel-based 2-DE and 1-DE shotgun proteomics approaches to dig deep into the leaf senescence proteome of Glycine max. J. Proteom. 2016, 148, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Wang, X.; Zhang, J.; Liu, P.; Zhao, B.; Li, G.; Dong, S. The role of nitrogen in leaf senescence of summer maize and analysis of underlying mechanisms using comparative proteomics. Plant Sci. 2015, 233, 72–81. [Google Scholar] [CrossRef]
- Li, L.; Zhao, J.; Zhao, Y.; Lu, X.; Zhou, Z.; Zhao, C.; Xu, G. Comprehensive investigation of tobacco leaves during natural early senescence via multi-platform metabolomics analyses. Sci. Rep. 2016, 6, 37976. [Google Scholar] [CrossRef]
- Burton, H.R.; Kasperbauer, M.J. Changes in chemical composition of tobacco lamina during senescence and curing. 1. Plastid pigments. J. Agric. Food Chem. 1985, 33, 879–883. [Google Scholar] [CrossRef]
- Li, S.; Su, X.; Jin, Q.; Li, G.; Sun, Y.; Abdullah, M.; Cai, Y.; Lin, Y. iTRAQ-Based Identification of Proteins Related to Lignin Synthesis in the Pear Pollinated with Pollen from Different Varieties. Molecules 2018, 23, 548. [Google Scholar] [CrossRef]
- Zhang, B.; Liu, C.; Yao, X.; Wang, F.; Wu, J.; King, G.J.; Liu, K. Disruption of a CAROTENOID CLEAVAGE DIOXYGENASE 4 gene converts flower colour from white to yellow in Brassica species. New Phytol. 2015, 206, 1513–1526. [Google Scholar] [CrossRef]
- Wang, Y.L.; Cao, C.; Zhou, H.; Zeng, Y.; Yang, J.; Wang, Y.X. Biochemical and transcriptome analyses of a novel chlorophyll-deficient chlorina tea plant cultivar. BMC Plant Biol. 2014, 14, 352. [Google Scholar] [CrossRef]
- Akram, N.A.; Shafiq, F.; Ashraf, M. Ascorbic Acid-A Potential Oxidant Scavenger and Its Role in Plant Development and Abiotic Stress Tolerance. Front. Plant Sci. 2017, 8, 613. [Google Scholar] [CrossRef]
- Yarmolinsky, D.; Brychkova, G.; Kurmanbayeva, A.; Bekturova, A.; Ventura, Y.; Khozin-Goldberg, I.; Eppel, A.; Fluhr, R.; Sagi, M. Impairment in Sulfite Reductase Leads to Early Leaf Senescence in Tomato Plants. Plant Physiol. 2014, 165, 1505–1520. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Djabou, A.S.M.; An, F.; Li, K.; Li, Z.; Yang, L.; Wang, X.; Chen, S. Proteomic analysis of injured storage roots in cassava (Manihot esculenta Crantz) under postharvest physiological deterioration. PLoS ONE 2017, 12, e0174238. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.J.; Ma, H.Y.; Zhuang, J. iTRAQ-based proteomics monitors the withering dynamics in postharvest leaves of tea plant (Camellia sinensis). Mol. Genet. Genom. 2018, 293, 45–59. [Google Scholar] [CrossRef] [PubMed]
- Lätari, K.; Wüst, F.; Hübner, M.; Schaub, P.; Beisel, K.G.; Matsubara, S.; Beyer, P.; Welsch, R. Tissue-Specific Apocarotenoid Glycosylation Contributes to Carotenoid Homeostasis in Arabidopsis Leaves. Plant Physiol. 2015, 168, 1550–1562. [Google Scholar] [CrossRef] [PubMed]
- Hörtensteiner, S. Chlorophyll degradation during senescence. Annu. Rev. Plant. Biol. 2006, 57, 55–77. [Google Scholar] [CrossRef] [PubMed]
- Bollenbach, T.J.; Lange, H.; Gutierrez, R.; Erhardt, M.; Stern, D.B.; Gagliardi, D. RNR1, a 3′-5′ exoribonuclease belonging to the RNR superfamily, catalyzes 3′ maturation of chloroplast ribosomal RNAs in Arabidopsis thaliana. Nucleic Acids Res. 2005, 33, 2751–2763. [Google Scholar] [CrossRef]
- Fargašová, A.; Molnárová, M. Assessment of Cr and Ni phytotoxicity from cutlery-washing waste-waters using biomass and chlorophyll production tests on mustard Sinapis alba L. seedlings. Environ. Sci. Pollut. Res. 2010, 17, 187–194. [Google Scholar] [CrossRef]
- Aruna, G.; Baskaran, V. Comparative study on the levels of carotenoids lutein, zeaxanthin and β-carotene in Indian spices of nutritional and medicinal importance. Food Chem. 2010, 123, 404–409. [Google Scholar] [CrossRef]
- Xiang, Z.; Cai, K.; Liang, G.; Zhou, S.; Ge, Y.; Zhang, J.; Geng, Z. Analysis of volatile flavour components in flue-cured tobacco by headspace solid-phase microextraction combined with GC×GC-TOFMS. Anal. Methods 2014, 6, 3300. [Google Scholar] [CrossRef]
- Isaacson, T.; Damasceno, C.M.; Saravanan, R.S.; He, Y.; Catalá, C.; Saladié, M.; Rose, J.K. Sample extraction techniques for enhanced proteomic analysis of plant tissues. Nat. Protoc. 2006, 1, 769–774. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal. Biochem. 1976, 722, 48–254. [Google Scholar] [CrossRef]
- Wen, B.; Zhou, R.; Feng, Q.; Wang, Q.; Wang, J.; Liu, S. IQuant: An automated pipeline for quantitative proteomics based upon isobaric tags. Proteomics 2014, 14, 2280–2285. [Google Scholar] [CrossRef] [PubMed]
- Brosch, M.; Yu, L.; Hubbard, T.; Choudhary, J. Accurate and Sensitive Peptide Identification with Mascot Percolator. J. Proteome Res. 2009, 8, 3176–3181. [Google Scholar] [CrossRef] [PubMed]
- Savitski, M.M.; Wilhelm, M.; Hahne, H.; Kuster, B.; Bantscheff, M. A Scalable Approach for Protein False Discovery Rate Estimation in Large Proteomic Data Sets. Mol. Cell. Proteom. 2015, 14, 2394–2404. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]





| Concentration | Curing Time (h) | ||
|---|---|---|---|
| 0 | 48 | 72 | |
| β-carotene (μg·gDM‒1) | 275.37 ± 7.32 | 151.19 ± 3.92** | 70.63 ± 1.20** |
| Lutein (μg·gDM‒1) | 384.29 ± 5.02 | 213.85 ±6.64** | 86.23 ± 1.93** |
| Violaxanthin (μg·gDM‒1) | 99.73 ± 2.98 | 46.49 ± 2.55** | 26.22 ± 0.62** |
| Neoxanthin (μg·gDM‒1) | 39.72 ± 1.53 | 14.85 ± 0.44** | 7.97 ± 0.28** |
| Chlorophyll a (mg·gFM‒1) | 0.71 ± 0.02 | 0.10 ± 0.01** | 0.04 ± 0.01** |
| Chlorophyll b (mg·gFM‒1) | 0.33 ± 0.01 | 0.07 ± 0.01** | 0.04 ± 0.01** |
| SPAD value | 21.87 ± 0.63 | 5.36 ± 0.39** | 1.45 ± 0.23** |
| Carotenoids/Chlorophylls | 0.29 ± 0.01 | 1.30 ± 0.06** | 2.22 ± 0.14** |
| Xanthophylls/β-carotene | 1.91 ± 0.06 | 1.84 ± 0.07 | 1.71 ± 0.03* |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, S.; Guo, Y.; Adil, M.F.; Sehar, S.; Cai, B.; Xiang, Z.; Tu, Y.; Zhao, D.; Shamsi, I.H. Comparative Proteomic Analysis by iTRAQ Reveals that Plastid Pigment Metabolism Contributes to Leaf Color Changes in Tobacco (Nicotiana tabacum) during Curing. Int. J. Mol. Sci. 2020, 21, 2394. https://doi.org/10.3390/ijms21072394
Wu S, Guo Y, Adil MF, Sehar S, Cai B, Xiang Z, Tu Y, Zhao D, Shamsi IH. Comparative Proteomic Analysis by iTRAQ Reveals that Plastid Pigment Metabolism Contributes to Leaf Color Changes in Tobacco (Nicotiana tabacum) during Curing. International Journal of Molecular Sciences. 2020; 21(7):2394. https://doi.org/10.3390/ijms21072394
Chicago/Turabian StyleWu, Shengjiang, Yushuang Guo, Muhammad Faheem Adil, Shafaque Sehar, Bin Cai, Zhangmin Xiang, Yonggao Tu, Degang Zhao, and Imran Haider Shamsi. 2020. "Comparative Proteomic Analysis by iTRAQ Reveals that Plastid Pigment Metabolism Contributes to Leaf Color Changes in Tobacco (Nicotiana tabacum) during Curing" International Journal of Molecular Sciences 21, no. 7: 2394. https://doi.org/10.3390/ijms21072394
APA StyleWu, S., Guo, Y., Adil, M. F., Sehar, S., Cai, B., Xiang, Z., Tu, Y., Zhao, D., & Shamsi, I. H. (2020). Comparative Proteomic Analysis by iTRAQ Reveals that Plastid Pigment Metabolism Contributes to Leaf Color Changes in Tobacco (Nicotiana tabacum) during Curing. International Journal of Molecular Sciences, 21(7), 2394. https://doi.org/10.3390/ijms21072394








