The Beta Subunit of Nascent Polypeptide Associated Complex Plays A Role in Flowers and Siliques Development of Arabidopsis thaliana
Abstract
1. Introduction
2. Results
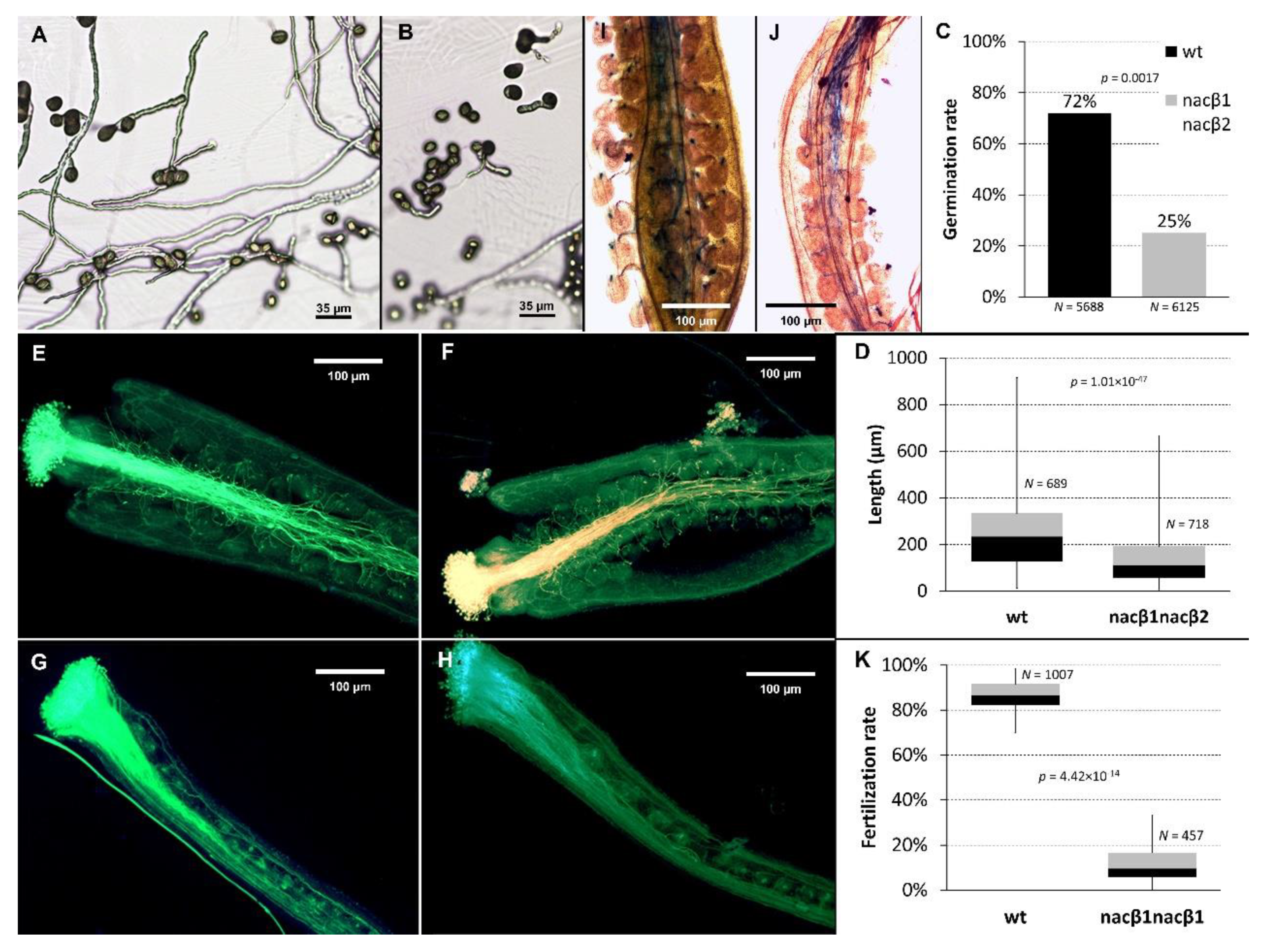
2.1. nacβ1nacβ2 Double Homozygous Plants Have Abnormal Flowers and Reduced Seed Set
2.2. nacβ1nacβ2 Double Mutation Compromises Progamic Phase and Ovule Targeting
2.3. nacβ1 and nacβ2 Alleles Show Partly Reduced Transmission Efficiency
2.4. The nacβ1nacβ2 Mutant Phenotype Was Complemented by any Functional NACβ Allele
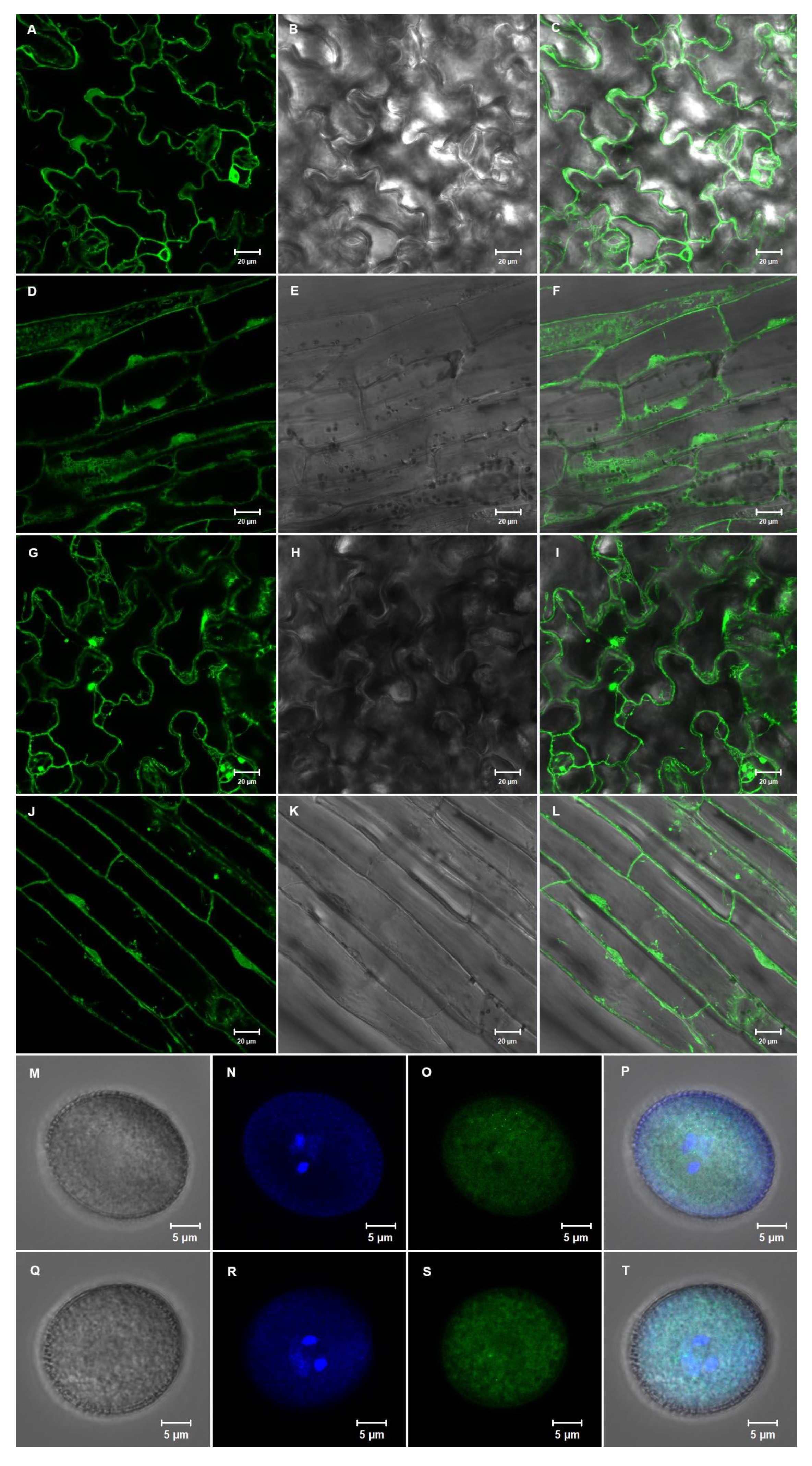
2.5. The NACβ Proteins Localized into Cytoplasm and Nuclei and Their Promoters were Active in Multiple Organs
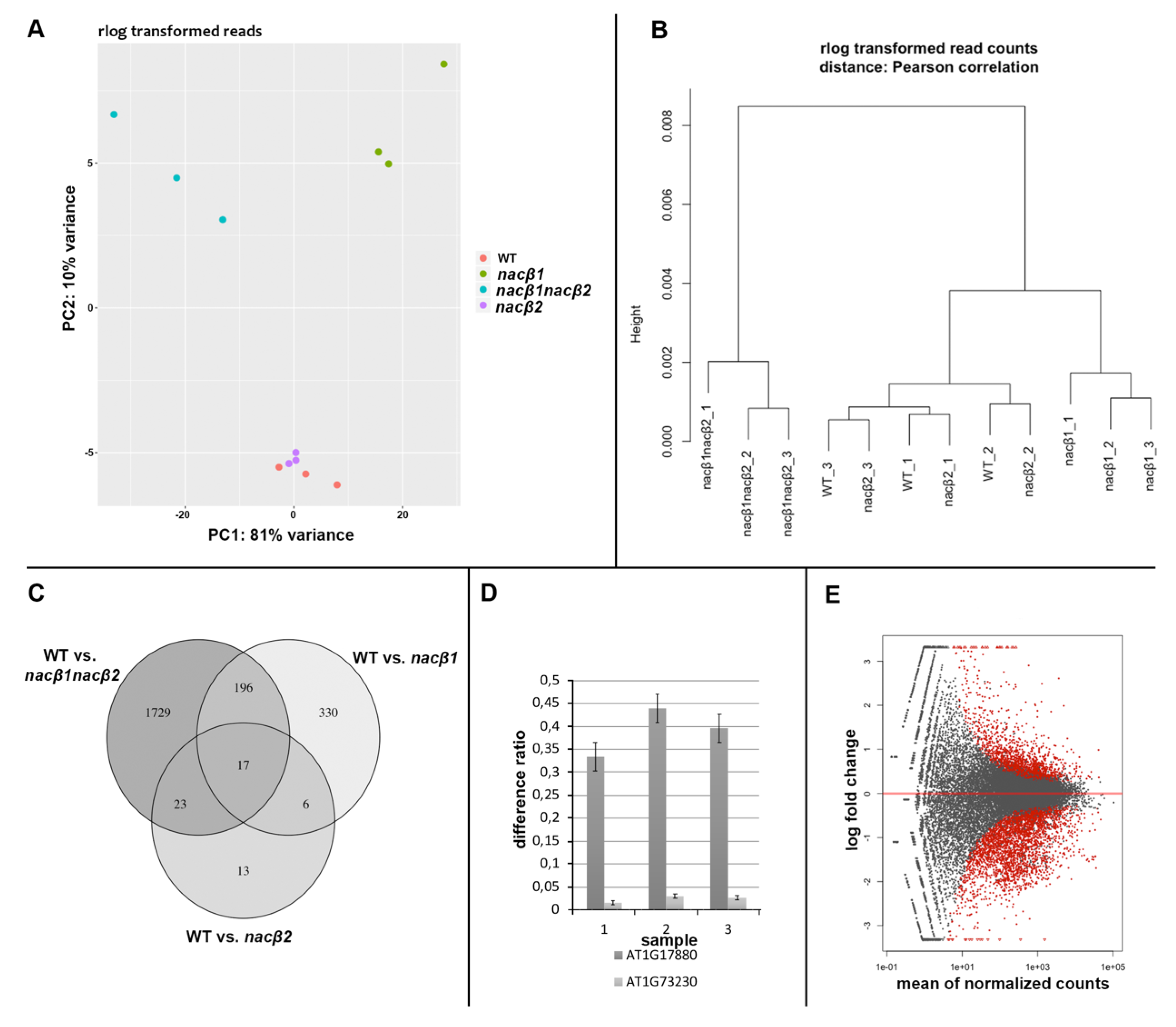
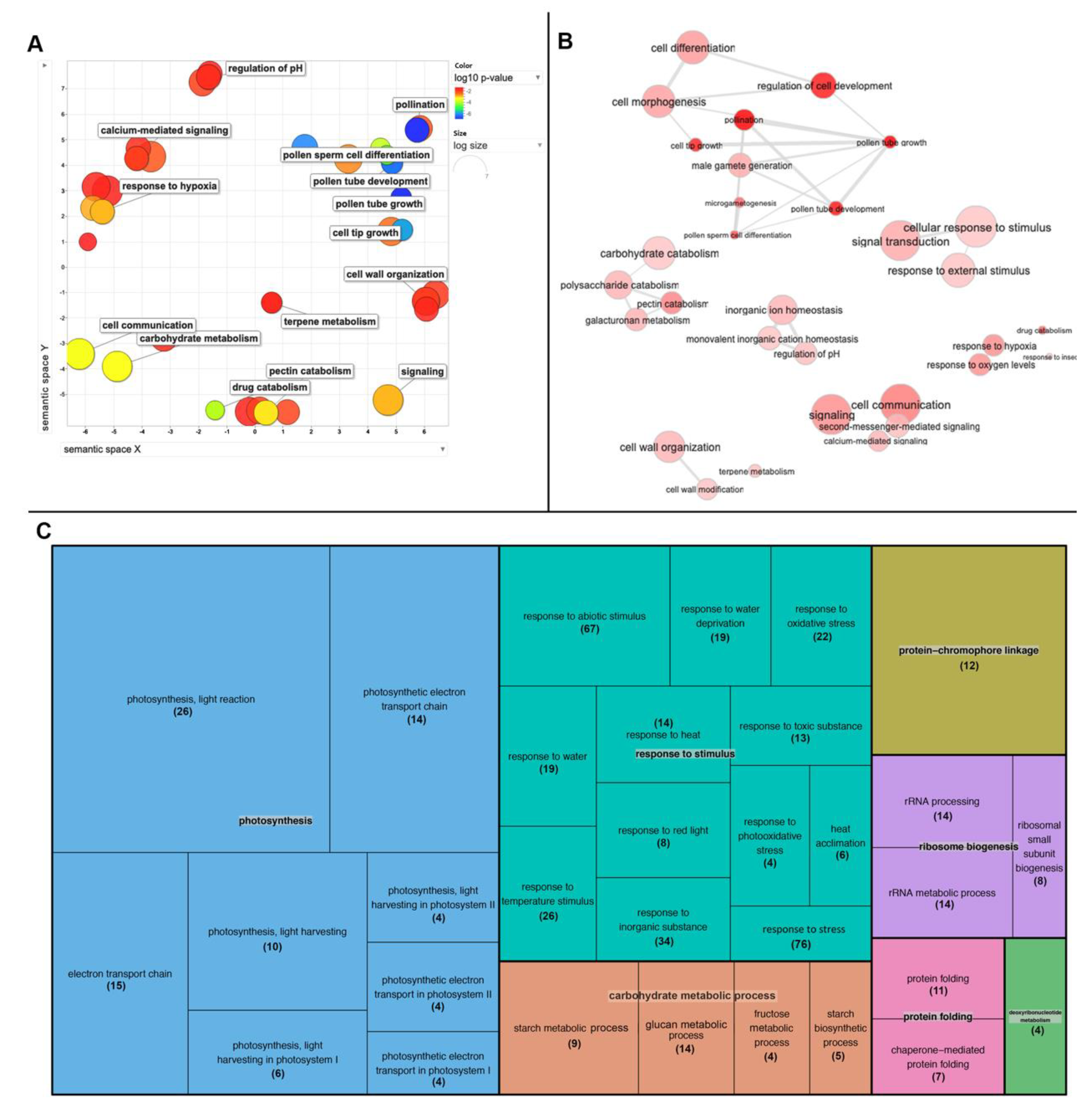
2.6. RNA Sequencing of the nacβ1nacβ2 Flower Bud Transcriptome Revealed Genes Important for Stress Responses and Male Gametophyte Development
2.7. Proteomic Analysis of the nacβ1nacβ2 Flower Buds Revealed Proteins with Role in Photosynthesis and Stress Responses
2.8. The Germination Efficiency of nacβ1nacβ2 Seeds Was Lower Under the Salt and Osmotic Stresses
3. Discussion
3.1. Phenotype Analysis of nacβ1nacβ2
3.2. NACβ Paralogues
3.3. Subcellular Localization of NACβ Proteins
3.4. Flower Bud Transcriptome and Proteome
4. Materials and Methods
4.1. Plant Cultivation
4.2. Insertion Lines and Genotyping
4.3. Phenotype Characterization
4.4. Transmission Analysis
4.5. Pollen Tube Cultivation in vitro
4.6. Pollen Tube Cultivation in vivo and Aniline Blue Staining
4.7. Blue Dot Assay
4.8. Cloning
4.9. Arabidopsis Thaliana Transformation and Microscopy
4.10. Promoter Analysis
4.11. RNA Extraction, cDNA Library Preparation and Sequencing
4.12. Mapping and Assembly Of Reads
4.13. Annotation and Enrichment Analyses
4.14. Confirmation of DEGs Expression by qRT-PCR
4.15. Protein Extraction and Proteome Analysis
4.16. Plant Cultivation Under The Salt and Osmotic Stress
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ag | agamous |
| ap3 | apetala 3 |
| BASTA | (RS)-2-Amino-4-(hydroxy(methyl)phosphonoyl)butanoic acid |
| bp | base pair |
| Col-0 | Columbia-0 |
| CaMV | cauliflower mosaic virus |
| clv1 | clavata 1 |
| clv3 | clavata 3 |
| CTAB | cetyltrimethylammonium bromide |
| DE | differential expression |
| DEG | differentially expressed gene |
| DEP | differentially expressed protein |
| dNTP | deoxyribonucleotide triphosphate |
| EDTA | ethylenediaminetetraacetic acid |
| GFP | green fluorescent protein |
| FPKM | fragments per kilobase million |
| GO | gene ontology |
| GUS | glucuronidase |
| HSP | heat shock protein |
| MS | Murashige and Skoog growth medium |
| NAC | nascent polypeptide-associated complex |
| nacβ1nacβ2 | NACβ double homozygous mutant |
| PCR | polymerase chain reaction |
| pi | pistillata |
| qRT-PCR | quantitative reverse transcriptase polymerase chain reaction |
| RNA-seq | RNA sequencing |
| sep | sepallata |
| TAE | electrophoresis buffer composed of Tris base, acetic acid and EDTA |
| Tris | 2-Amino-2-(hydroxymethyl)propane-1,3-diol |
| wig | wiggum |
| wt | wild type |
References
- Kogan, G.L.; Gvozdev, V.A. Multifunctional nascent polypeptide-associated complex (NAC). Mol. Biol. 2014, 48, 189–196. [Google Scholar] [CrossRef]
- Spreter, T.; Pech, M.; Beatrix, B. The crystal structure of archaeal nascent polypeptide-associated complex (NAC) reveals a unique fold and the presence of a ubiquitin-associated domain. J. Boil. Chem. 2005, 280, 15849–15854. [Google Scholar] [CrossRef] [PubMed]
- Rospert, S.; Dubaquié, Y.; Gautschi, M. Nascent-polypeptide-associated complex. Cell. Mol. Life Sci. 2002, 59, 1632–1639. [Google Scholar] [CrossRef] [PubMed]
- Reimann, B.; Bradsher, J.; Franke, J.; Hartmann, E.; Wiedmann, M.; Prehn, S.; Wiedmann, B. Initial characterization of the nascent polypeptide-associated complex in yeast. Yeast 1999, 15, 397–407. [Google Scholar] [CrossRef]
- Ott, A.-K.; Locher, L.; Koch, M.; Deuerling, E. Functional dissection of the nascent polypeptide-associated complex in Saccharomyces cerevisiae. PLoS ONE 2015, 10, e0143457. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Sakai, H.; Wiedmann, M. NAC covers ribosome-associated nascent chains thereby forming a protective environment for regions of nascent chains just emerging from the peptidyl transferase center. J. Cell Biol. 1995, 130, 519–528. [Google Scholar] [CrossRef]
- Wiedmann, B.; Sakai, H.; Davis, T.A.; Wiedmann, M. A protein complex required for signal-sequence-specific sorting and translocation. Nature 1994, 370, 434–440. [Google Scholar] [CrossRef]
- Martin, E.M.; Jackson, M.P.; Gamerdinger, M.; Gense, K.; Karamonos, T.K.; Humes, J.; Deuerling, E.; Ashcroft, A.E.; Radford, S.E. Conformational flexibility within the nascent polypeptide-associated complex enables its interactions with structurally diverse client proteins. J. Biol. Chem. 2018, 293, 8554–8568. [Google Scholar] [CrossRef]
- Wegrzyn, R.D.; Hofmann, D.; Merz, F.; Nikolay, R.; Rauch, T.; Graf, C.; Deuerling, E. A conserved motif is prerequisite for the interaction of NAC with ribosomal protein L23 and nascent chains. J. Biol. Chem. 2005, 281, 2847–2857. [Google Scholar] [CrossRef]
- Möller, I.; Beatrix, B.; Kreibich, G.; Sakai, H.; Lauring, B.; Wiedmann, M. Unregulated exposure of the ribosomal M-site caused by NAC depletion results in delivery of non-secretory polypeptides to the Sec61 complex. FEBS Lett. 1998, 441, 1–5. [Google Scholar] [CrossRef]
- Funfschilling, U.; Rospert, S. Nascent polypeptide–associated complex stimulates protein import into yeast mitochondria. Mol. Biol. Cell 1999, 10, 3289–3299. [Google Scholar] [CrossRef] [PubMed]
- Gamerdinger, M.; Hanebuth, M.A.; Frickey, T.; Deuerling, E. The principle of antagonism ensures protein targeting specificity at the endoplasmic reticulum. Science 2015, 348, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Koplin, A.; Preissler, S.; Ilina, Y.; Koch, M.; Scior, A.; Erhardt, M.; Deuerling, E. A dual function for chaperones SSB–RAC and the NAC nascent polypeptide–associated complex on ribosomes. J. Cell Biol. 2010, 189, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Bloss, T.A.; Witze, E.S.; Rothman, J.H. Suppression of CED-3-independent apoptosis by mitochondrial βNAC in Caenorhabditis elegans. Nature 2003, 424, 1066–1071. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.M.; Behringer, R.R. An insertional mutation in theBTF3 transcription factor gene leads to an early postimplantation lethality in mice. Transgenic Res. 1995, 4, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Markesich, D.C.; Gajewski, K.M.; Nazimiec, M.E.; Beckingham, K. bicaudal encodes the Drosophila beta NAC homolog, a component of the ribosomal translational machinery. Development 2000, 127, 559–572. [Google Scholar] [PubMed]
- Brockstedt, E.; Otto, A.; Rickers, A.; Bommert, K.; Wittmann-Liebold, B. Preparative high-resolution two-dimensional electrophoresis enables the identification of RNA polymerase B transcription factor 3 as an apoptosis-associated protein in the human BL60-2 Burkitt lymphoma cell line. Protein J. 1999, 18, 225–231. [Google Scholar] [CrossRef]
- Moreau, A.; Yotov, W.V.; Glorieux, F.H.; St-Arnaud, R. Bone-Specific Expression of the alpha chain of the nascent polypeptide-associated complex, a coactivator potentiating c-Jun-mediated transcription. Mol. Cell. Biol. 1998, 18, 1312–1321. [Google Scholar] [CrossRef]
- Yotov, W.V.; Moreau, A.; St-Arnaud, R. The alpha chain of the nascent polypeptide-associated complex functions as a transcriptional coactivator. Mol. Cell. Boil. 1998, 18, 1303–1311. [Google Scholar] [CrossRef]
- Berardini, T.; Reiser, L.; Li, N.; Mezheritsky, Y.; Muller, R.; Strait, E.; Huala, E. The Arabidopsis information resource: Making and mining the "gold standard" annotated reference plant genome. Genesis 2015, 53, 474–485. [Google Scholar] [CrossRef]
- Ding, Y.; Jia, Y.; Shi, Y.; Zhang, X.; Song, C.; Gong, Z.; Yang, S. OST 1-mediated BTF 3L phosphorylation positively regulates CBF s during plant cold responses. EMBO J. 2018, 37, e98228. [Google Scholar] [CrossRef] [PubMed]
- Park, H.L.; Cho, M.-H.; Shin, D.H.; Bhoo, S.H.; Hahn, T.-R. Molecular characterization of phytochrome-interacting nascent polypeptide associated complex protein in Arabidopsis thaliana. J. Korean Soc. Appl. Boil. Chem. 2012, 55, 323–326. [Google Scholar] [CrossRef]
- Freire, M.A. Translation initiation factor (iso) 4E interacts with BTF3, the β subunit of the nascent polypeptide-associated complex. Gene 2005, 345, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.-S.; Kim, H.-S.; Jin, U.-H.; Lee, S.S.; Park, J.-A.; Lim, Y.P.; Pai, H.-S. Silencing of NbBTF3 results in developmental defects and disturbed gene expression in chloroplasts and mitochondria of higher plants. Planta 2007, 225, 1459–1469. [Google Scholar] [CrossRef]
- Ma, H.-Z.; Liu, G.-Q.; Li, C.-W.; Kang, G.; Guo, T.-C. Identification of the TaBTF3 gene in wheat (Triticum aestivum L.) and the effect of its silencing on wheat chloroplast, mitochondria and mesophyll cell development. Biochem. Biophys. Res. Commun. 2012, 426, 608–614. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Xu, M.; Wang, Y.; Jamil, M. Basal transcription factor 3 plays an important role in seed germination and seedling growth of rice. BioMed Res. Int. 2014, 2014, 465739. [Google Scholar] [CrossRef]
- Maršálová, L.; Vítámvás, P.; Hynek, R.; Prášil, I.T.; Kosová, K. Proteomic response of Hordeum vulgare cv. Tadmor and Hordeum marinum to salinity stress: Similarities and differences between a glycophyte and a halophyte. Front. Plant Sci. 2016, 7, 2567. [Google Scholar] [CrossRef]
- Karan, R.; Subudhi, P.K. Overexpression of a nascent polypeptide associated complex gene (SaβNAC) of Spartina alterniflora improves tolerance to salinity and drought in transgenic Arabidopsis. Biochem. Biophys. Res. Commun. 2012, 424, 747–752. [Google Scholar] [CrossRef]
- Kang, G.; Ma, H.; Liu, G.; Han, Q.; Li, C.; Guo, T. Silencing of TaBTF3 gene impairs tolerance to freezing and drought stresses in wheat. Mol. Genet. Genom. 2013, 288, 591–599. [Google Scholar] [CrossRef]
- Yin, X.; Nishimura, M.; Hajika, M.; Komatsu, S. Quantitative proteomics reveals the flooding-tolerance mechanism in mutant and abscisic acid-treated soybean. J. Proteome Res. 2016, 15, 2008–2025. [Google Scholar] [CrossRef]
- Huh, S.U.; Kim, K.-J.; Paek, K.-H. Capsicum annuum basic transcription factor 3 (CaBtf3) regulates transcription of pathogenesis-related genes during hypersensitive response upon Tobacco mosaic virus infection. Biochem. Biophys. Res. Commun. 2012, 417, 910–917. [Google Scholar] [CrossRef] [PubMed]
- Kaido, M.; Inoue, Y.; Takeda, Y.; Sugiyama, K.; Takeda, A.; Mori, M.; Tamai, A.; Meshi, T.; Okuno, T.; Mise, K. Downregulation of the NbNACa1 gene encoding a movement-protein-interacting protein reduces cell-to-cell movement of Brome mosaic virus in Nicotiana benthamiana. Mol. Plant-Microbe Interactions 2007, 20, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Reňák, D.; Dupľáková, N.; Honys, D. Wide-scale screening of T-DNA lines for transcription factor genes affecting male gametophyte development in Arabidopsis. Sex. Plant Reprod. 2011, 25, 39–60. [Google Scholar] [CrossRef] [PubMed]
- Boavida, L.C.; McCormick, S. Temperature as a determinant factor for increased and reproducible in vitro pollen germination in Arabidopsis thaliana. Plant J. 2007, 52, 570–582. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Leydon, A.R.; Manziello, A.; Pandey, R.; Mount, D.; Denic, S.; Vasic, B.; Johnson, M.; Palanivelu, R. Penetration of the stigma and style elicits a novel transcriptome in pollen tubes, pointing to genes critical for growth in a pistil. PLoS Genet. 2009, 5, e1000621. [Google Scholar] [CrossRef] [PubMed]
- Regan, S.M.; Moffatt, B.A. Cytochemical analysis of pollen development in wild-type Arabidopsis and a male sterile mutant. Plant. Cell 1990, 2, 877–889. [Google Scholar] [CrossRef] [PubMed]
- Hafidh, S.; Potěšil, D.; Fila, J.; Čapková, V.; Zdráhal, Z.; Honys, D. Quantitative proteomics of the tobacco pollen tube secretome identifies novel pollen tube guidance proteins important for fertilization. Genome Biol. 2016, 17, 81. [Google Scholar] [CrossRef] [PubMed]
- Apel, K.; Bohlmann, H. Comparison of different constitutive and inducible promoters for the overexpression of transgenes in Arabidopsis thaliana. Plant Mol. Biol. 1995, 29, 637–646. [Google Scholar]
- Twell, D.; Yamaguchi, J.; McCormick, S. Pollen-specific gene expression in transgenic plants: Coordinate regulation of two different tomato gene promoters during microsporogenesis. Development 1990, 109, 705–713. [Google Scholar]
- Steffen, J.G.; Kang, I.-H.; Macfarlane, J.; Drews, G.N. Identification of genes expressed in the Arabidopsis female gametophyte. Plant J. 2007, 51, 281–292. [Google Scholar] [CrossRef]
- Nakabayashi, K.; Okamoto, M.; Koshiba, T.; Kamiya, Y.; Nambara, E. Genome-wide profiling of stored mRNA in Arabidopsis thaliana seed germination: Epigenetic and genetic regulation of transcription in seed. Plant J. 2005, 41, 697–709. [Google Scholar] [CrossRef] [PubMed]
- Schmid, M.; Davison, T.S.; Henz, S.R.; Pape, U.J.; Demar, M.; Vingron, M.; Schölkopf, B.; Weigel, D.; Lohmann, J.U. A gene expression map of Arabidopsis thaliana development. Nat. Genet. 2005, 37, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Cardarelli, M.; Cecchetti, V. Auxin polar transport in stamen formation and development: How many actors? Front. Plant Sci. 2014, 5, 333. [Google Scholar] [CrossRef] [PubMed]
- Supek, F.; Bosnjak, M.; Skunca, N.; Šmuc, T. REVIGO summarizes and visualizes long lists of gene ontology terms. PLoS ONE 2011, 6, e21800. [Google Scholar] [CrossRef] [PubMed]
- Winter, D.; Vinegar, B.; Nahal, H.; Ammar, R.; Wilson, G.; Provart, N.J. An “Electronic Fluorescent Pictograph” browser for exploring and analyzing large-scale biological data sets. PLoS ONE 2007, 2, e718. [Google Scholar] [CrossRef]
- Tsukaya, H.; Byrne, M.E.; Horiguchi, G.; Sugiyama, M.; Van Lijsebettens, M.; Lenhard, M. How do ‘housekeeping’ genes control organogenesis?—unexpected new findings on the role of housekeeping genes in cell and organ differentiation. J. Plant Res. 2012, 126, 3–15. [Google Scholar] [CrossRef]
- Riechmann, J.L. DNA-binding properties of Arabidopsis MADS domain homeotic proteins APETALA1, APETALA3, PISTILLATA and AGAMOUS. Nucleic Acids Res. 1996, 24, 3134–3141. [Google Scholar] [CrossRef]
- Running, M.P.; Fletcher, J.C.; Meyerowitz, E.M. The WIGGUM gene is required for proper regulation of floral meristem size in Arabidopsis. Development 1998, 125, 2545–2553. [Google Scholar]
- McKim, S.M.; Routier-Kierzkowska, A.-L.; Monniaux, M.; Kierzkowski, D.; Pieper, B.; Smith, R.S.; Tsiantis, M.; Hay, A.S. Seasonal regulation of petal number. Plant Physiol. 2017, 175, 886–903. [Google Scholar] [CrossRef]
- Pelaz, S.; Ditta, G.S.; Baumann, E.; Wisman, E.; Yanofsky, M.F. B and C floral organ identity functions require SEPALLATA MADS-box genes. Nature 2000, 405, 200–203. [Google Scholar] [CrossRef]
- Yotov, W.V.; St-Arnaud, R. Differential splicing-in of a proline-rich exon converts alphaNAC into a muscle-specific transcription factor. Genes Dev. 1996, 10, 1763–1772. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.M.; Black, D.; Chambon, P.; Egly, J.M. Sequencing and expression of complementary DNA for the general transcription factor BTF3. Nature 1990, 344, 556–559. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Hu, Y.; Li, X.; Niu, L.; Teng, M. The crystal structure of the human nascent polypeptide-associated complex domain reveals a nucleic acid-binding region on the NACA subunit. Biochemistry 2010, 49, 2890–2896. [Google Scholar] [CrossRef]
- Miller, D.D.; Scordilis, S.P.; Hepler, P.K. Identification and localization of three classes of myosins in pollen tubes of Lilium longiflorum and Nicotiana alata. J. Cell Sci. 1995, 108, 2549–2563. [Google Scholar] [PubMed]
- Honys, D.; Reňák, D.; Feciková, J.; Jedelský, P.; Nebesářová, J.; Dobrev, P.; Čapková, V. Cytoskeleton-associated large RNP complexes in tobacco male gametophyte (EPPs) are associated with ribosomes and are involved in protein synthesis, processing, and localization. J. Proteome Res. 2009, 8, 2015–2031. [Google Scholar] [CrossRef]
- Stinson, J.R.; Eisenberg, A.J.; Willing, R.P.; Pe, M.E.; Hanson, D.D.; Mascarenhas, J.P.; Anderson, J.A.; Ashworth, E.N. Genes expressed in the male gametophyte of flowering plants and their isolation. Plant Physiol. 1987, 83, 442–447. [Google Scholar] [CrossRef]
- Hafidh, S.; Potěšil, D.; Müller, K.; Fíla, J.; Michailidis, C.; Herrmannová, A.; Feciková, J.; Ischebeck, T.; Valášek, L.S.; Zdráhal, Z.; et al. Dynamics of the pollen sequestrome defined by subcellular coupled omics. Plant Physiol. 2018, 178, 258–282. [Google Scholar] [CrossRef]
- Adamczyk, B.J.; Fernandez, D.E. MIKC*MADS domain heterodimers are required for pollen maturation and tube growth in Arabidopsis. Plant Physiol. 2009, 149, 1713–1723. [Google Scholar] [CrossRef]
- Ge, Z.; Bergonci, T.; Zhao, Y.; Zou, Y.; Du, S.; Liu, M.-C.; Luo, X.; Ruan, H.; García-Valencia, L.E.; Zhong, S.; et al. Arabidopsis pollen tube integrity and sperm release are regulated by RALF-mediated signaling. Science 2017, 358, 1596–1600. [Google Scholar] [CrossRef]
- Rã¶ckel, N.; Wolf, S.; Kost, B.; Rausch, T.; Greiner, S.; Röckel, N. Elaborate spatial patterning of cell-wall PME and PMEI at the pollen tube tip involves PMEI endocytosis, and reflects the distribution of esterified and de-esterified pectins. Plant J. 2008, 53, 133–143. [Google Scholar] [CrossRef]
- Brownfield, L.; Hafidh, S.; Borg, M.; Sidorova, A.; Mori, T.; Twell, D. A plant germline-specific integrator of sperm specification and cell cycle progression. PLoS Genet. 2009, 5, e1000430. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Mori, T.; Ueda, K.; Yamada, L.; Nagahara, S.; Higashiyama, T.; Sawada, H.; Igawa, T. The male gamete membrane protein DMP9/DAU2 is required for double fertilization in flowering plants. Development 2018, 145, dev170076. [Google Scholar] [CrossRef]
- Maruyama, D.; Sugiyama, T.; Endo, T.; Nishikawa, S.-I. Multiple BiP Genes of Arabidopsis thaliana are required for male gametogenesis and pollen competitiveness. Plant Cell Physiol. 2014, 55, 801–810. [Google Scholar] [CrossRef]
- Sarkar, N.K.; Kim, Y.-K.; Grover, A. Rice sHsp genes: Genomic organization and expression profiling under stress and development. BMC Genom. 2009, 10, 393. [Google Scholar] [CrossRef] [PubMed]
- Wehmeyer, N.; Klann, E.M.; Hall, B.; Bennett, A.B. Synthesis of small heat-shock proteins is part of the developmental program of late seed maturation. Plant Physiol. 1996, 112, 747–757. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.-H.; Liu, R.; Yan, L.; Liu, Z.-Q.; Jiang, S.-C.; Shen, Y.-Y.; Wang, X.-F.; Zhang, D.-P. Light-harvesting chlorophyll a/b-binding proteins are required for stomatal response to abscisic acid in Arabidopsis. J. Exp. Bot. 2011, 63, 1095–1106. [Google Scholar] [CrossRef] [PubMed]
- Varotto, C.; Pesaresi, P.; Meurer, J.; Oelmüller, R.; Steiner-Lange, S.; Salamini, F.; Leister, D. Disruption of the Arabidopsis photosystem I gene psaE1 affects photosynthesis and impairs growth. Plant J. 2000, 22, 115–124. [Google Scholar] [CrossRef]
- Jaiswal, H.; Conz, C.; Otto, H.; Wölfle, T.; Fitzke, E.; Mayer, M.P.; Rospert, S. The chaperone network connected to human ribosome-associated complex. Mol. Cell. Boil. 2011, 31, 1160–1173. [Google Scholar] [CrossRef]
- Verma, A.K.; Diwan, D.; Raut, S.; Dobriyal, N.; Brown, R.E.; Gowda, V.; Hines, J.K.; Sahi, C. Evolutionary conservation and emerging functional diversity of the cytosolic Hsp70:J protein chaperone network of Arabidopsis thaliana. G3: Genes|Genomes|Genetics 2017, 7, 1941–1954. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Weigel, D.; Glazebrook, J. Arabidopsis: A Laboratory Manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2002. [Google Scholar]
- Ogden, R.C.; Adams, D.A. Electrophoresis in agarose and acrylamide gels. Methods Enzymol. 1987, 152, 61–87. [Google Scholar] [PubMed]
- Witham, F.H.; Blaydes, D.F.; Devlin, R.M. Experiment 15: Chlorophyll absorption spectrum and quantitative determinations. In Experiments in Plant Physiology; Van Nostrand Reinhold Co.: New York, NY, USA, 1971; p. 245. [Google Scholar]
- Mori, T.; Kuroiwa, H.; Higashiyama, T.; Kuroiwa, T. GENERATIVE CELL SPECIFIC 1 is essential for angiosperm fertilization. Nature 2005, 8, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Shimada, T.L.; Shimada, T.; Hara-Nishimura, I. A rapid and non-destructive screenable marker, FAST, for identifying transformed seeds of Arabidopsis thaliana. Plant J. 2010, 61, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Karimi, M.; Inzé, D.; Depicker, A. GATEWAY™ vectors for Agrobacterium-mediated plant transformation. Trends Plant Sci. 2002, 7, 193–195. [Google Scholar] [CrossRef]
- Clough, S.J.; Bent, A. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 1998, 16, 735–743. [Google Scholar] [CrossRef]
- Hu, C.-Y.; Chee, P.; Chesney, R.; Zhou, J.; Miller, P.; O’Brien, W. Intrinsic GUS-like activities in seed plants. Plant Cell Rep. 1990, 9, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Andrews, S. FastQC. A quality control tool for high throughput sequence data. 2010. Available online: https://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 6 December 2019).
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J. 2011, 17, 10. [Google Scholar] [CrossRef]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2012, 29, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Smyth, G.K.; Shi, W. The Subread aligner: Fast, accurate and scalable read mapping by seed-and-vote. Nucleic Acids Res. 2013, 41, e108. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 002832. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef]
- Fíla, J.; Čapková, V.; Feciková, J.; Honys, D. Impact of homogenization and protein extraction conditions on the obtained tobacco pollen proteomic patterns. Biol. Plant. 2011, 55, 499–506. [Google Scholar] [CrossRef]
- Wiśniewski, J.R.; Ostasiewicz, P.; Mann, M. High recovery FASP applied to the proteomic analysis of microdissected formalin fixed paraffin embedded cancer tissues retrieves known colon cancer markers. J. Proteome Res. 2011, 10, 3040–3049. [Google Scholar] [CrossRef] [PubMed]
- Stejskal, K.; Potěšil, D.; Zdráhal, Z. Suppression of peptide sample losses in autosampler vials. J. Proteome Res. 2013, 12, 3057–3062. [Google Scholar] [CrossRef] [PubMed]


| Parent | Selfing Offspring | Expected Mendelian Ratio | Observed Ratio | Change in Transmission Efficiency | χ2 |
|---|---|---|---|---|---|
| nacβ1/+;nacβ2 NACβ1 transmission (N = 119) | NACβ1nacβ2 | 29.75 | 33 | +11% | 7.5 × 10−2 |
| nacβ1/+;nacβ2 | 59.50 | 67 | +13% | ||
| nacβ1nacβ2 | 29.75 | 19 | −46% | ||
| nacβ1;nacβ2/+ NACβ2 transmission (N = 115) | nacβ1NACβ2 | 28.75 | 37 | +29% | 8.8 × 10−3 |
| nacβ1;nacβ2/+ | 57.50 | 63 | +10% | ||
| nacβ1nacβ2 | 28.75 | 15 | −48% |
 means female, whereas
means female, whereas  means male.
means male.
 means female, whereas
means female, whereas  means male.
means male.| Cross | Mendelian Rate of Mutant Allele | Observed Rate of Mutant Allele | Mutant Allele Transmission Efficiency—Wild Type Background | χ2 | |
|---|---|---|---|---|---|
nacβ1/+;nacβ2 × Col-0 (wt) × Col-0 (wt)  (N = 88) (N = 88) | 44 | 53 | nacβ1 | +20% | 5.5 × 10−2 |
nacβ1;nacβ2/+ × Col-0 (wt) × Col-0 (wt)  (N = 118) (N = 118) | 59 | 50 | nacβ2 | −15% | 9.8 × 10−2 |
Col-0 (wt)  × nacβ1/+;nacβ2 × nacβ1/+;nacβ2  (N = 128) (N = 128) | 64 | 97 | nacβ1 | +52% | 5.4 × 10−9 |
Col-0 (wt)  × nacβ1;nacβ2/+ × nacβ1;nacβ2/+  (N = 133) (N = 133) | 66.5 | 52 | nacβ2 | −22% | 1.2 × 10−2 |
 means female, whereas
means female, whereas  means male.
means male.
 means female, whereas
means female, whereas  means male.
means male.| Cross | Mendelian Rate of Mutant Allele | Observed Rate of Mutant Allele | Mutant allele Transmission Efficiency—Mutant Background | χ2 | |
|---|---|---|---|---|---|
nacβ1/+;nacβ2 × NACβ1nacβ2 × NACβ1nacβ2  (N = 114) (N = 114) | 57 | 54 | nacβ1 | −5% | 5.7 × 10−1 |
nacβ1;nacβ2/+ × nacβ1NACβ2 × nacβ1NACβ2  (N = 143) (N = 143) | 71.5 | 59 | nacβ2 | −17% | 3.7 × 10−2 |
NACβ1nacβ2 × nacβ1/+;nacβ2 × nacβ1/+;nacβ2  (N = 142) (N = 142) | 71 | 70 | nacβ1 | −1% | 8.7 × 10−1 |
nacβ1NACβ2 × nacβ1;nacβ2/+ × nacβ1;nacβ2/+  (N = 140) (N = 140) | 70 | 58 | nacβ2 | −17% | 4.3 × 10−2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fíla, J.; Klodová, B.; Potěšil, D.; Juříček, M.; Šesták, P.; Zdráhal, Z.; Honys, D. The Beta Subunit of Nascent Polypeptide Associated Complex Plays A Role in Flowers and Siliques Development of Arabidopsis thaliana. Int. J. Mol. Sci. 2020, 21, 2065. https://doi.org/10.3390/ijms21062065
Fíla J, Klodová B, Potěšil D, Juříček M, Šesták P, Zdráhal Z, Honys D. The Beta Subunit of Nascent Polypeptide Associated Complex Plays A Role in Flowers and Siliques Development of Arabidopsis thaliana. International Journal of Molecular Sciences. 2020; 21(6):2065. https://doi.org/10.3390/ijms21062065
Chicago/Turabian StyleFíla, Jan, Božena Klodová, David Potěšil, Miloslav Juříček, Petr Šesták, Zbyněk Zdráhal, and David Honys. 2020. "The Beta Subunit of Nascent Polypeptide Associated Complex Plays A Role in Flowers and Siliques Development of Arabidopsis thaliana" International Journal of Molecular Sciences 21, no. 6: 2065. https://doi.org/10.3390/ijms21062065
APA StyleFíla, J., Klodová, B., Potěšil, D., Juříček, M., Šesták, P., Zdráhal, Z., & Honys, D. (2020). The Beta Subunit of Nascent Polypeptide Associated Complex Plays A Role in Flowers and Siliques Development of Arabidopsis thaliana. International Journal of Molecular Sciences, 21(6), 2065. https://doi.org/10.3390/ijms21062065






