The Role of Biofilms Developed under Different Anthropogenic Pressure on Recruitment of Macro-Invertebrates
Abstract
1. Introduction
2. Results
2.1. Biofilm Characterization
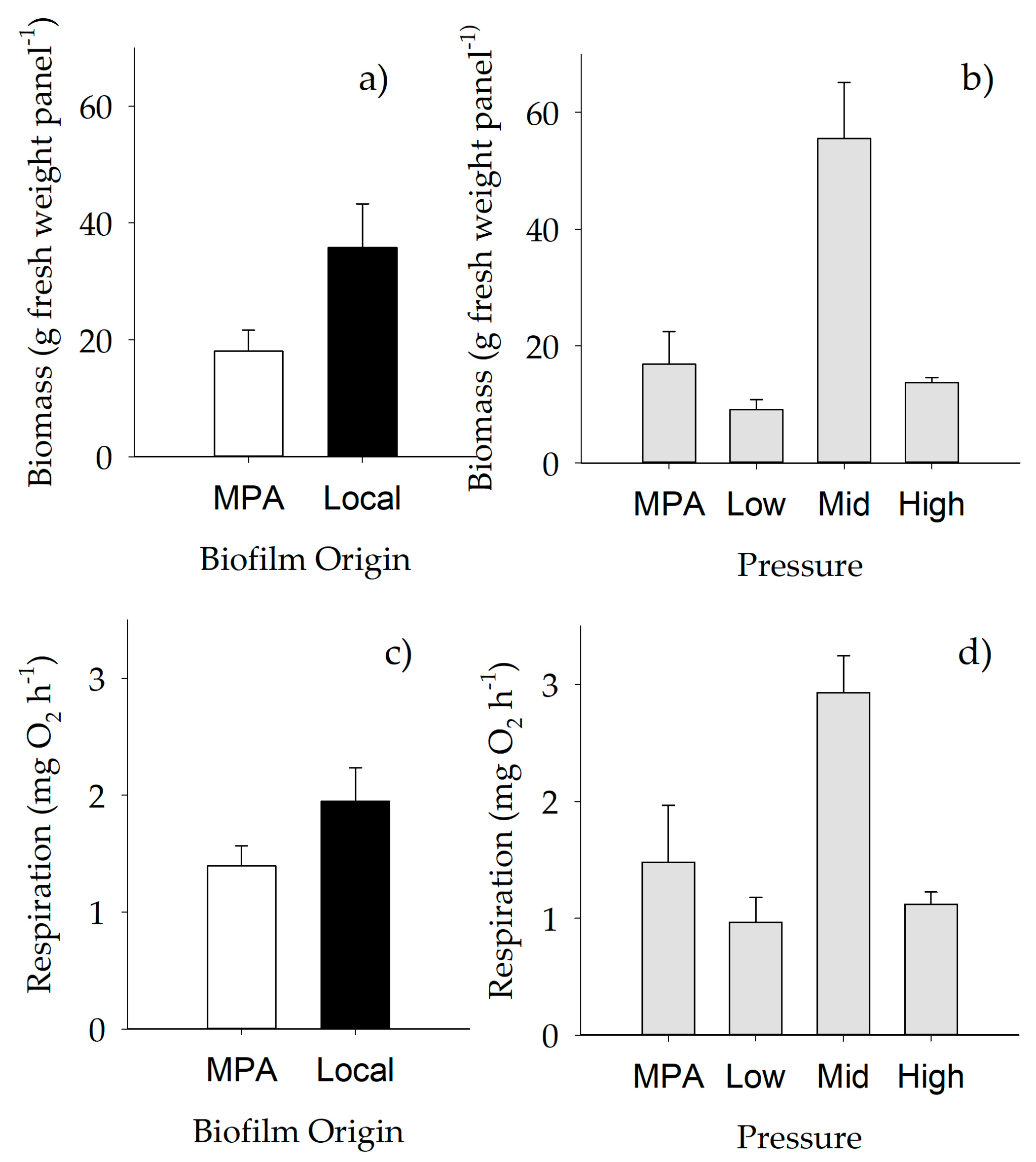
2.1.1. Weight of Biofilm
2.1.2. Bacteria
2.1.3. Diatoms
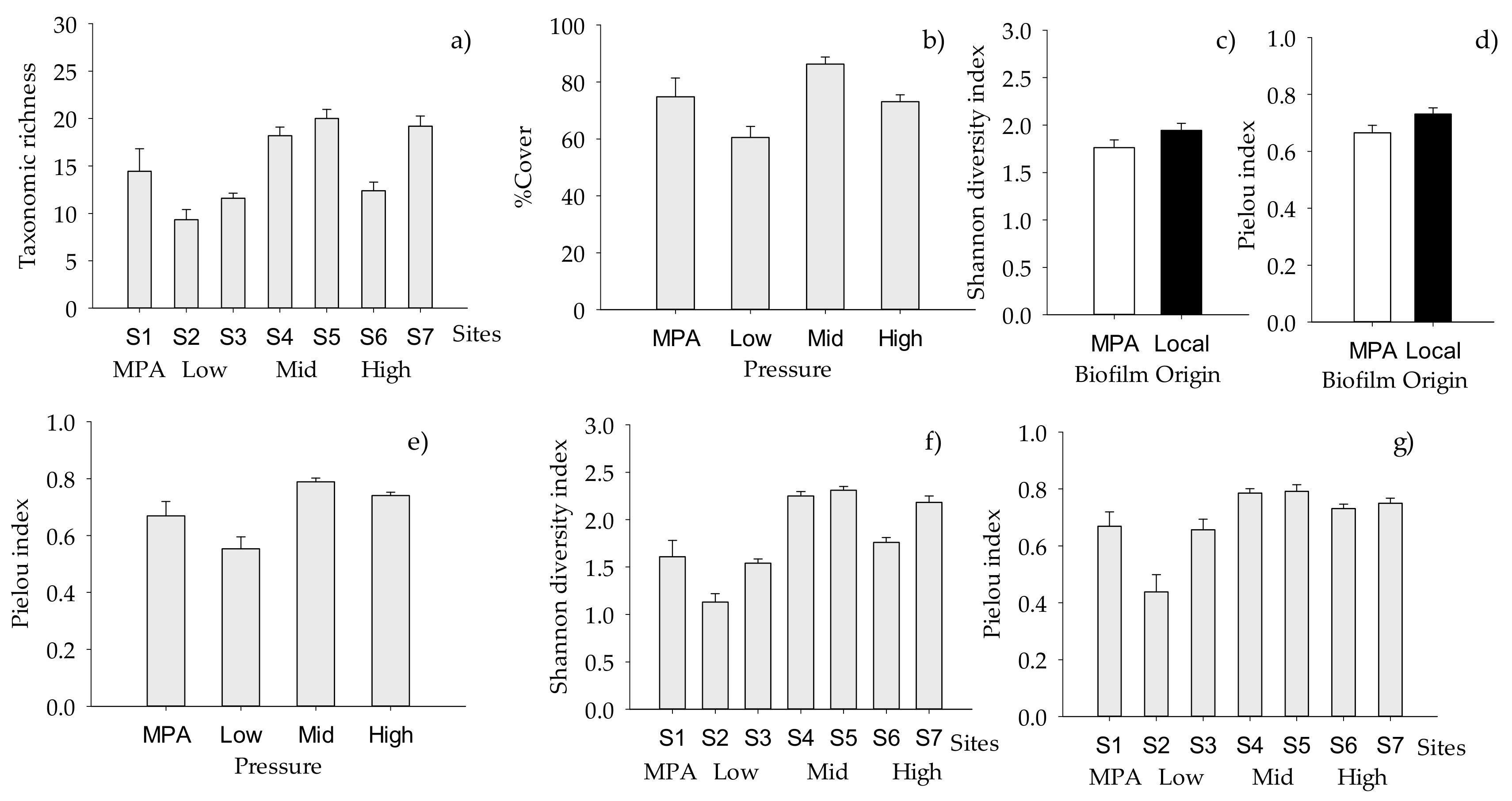
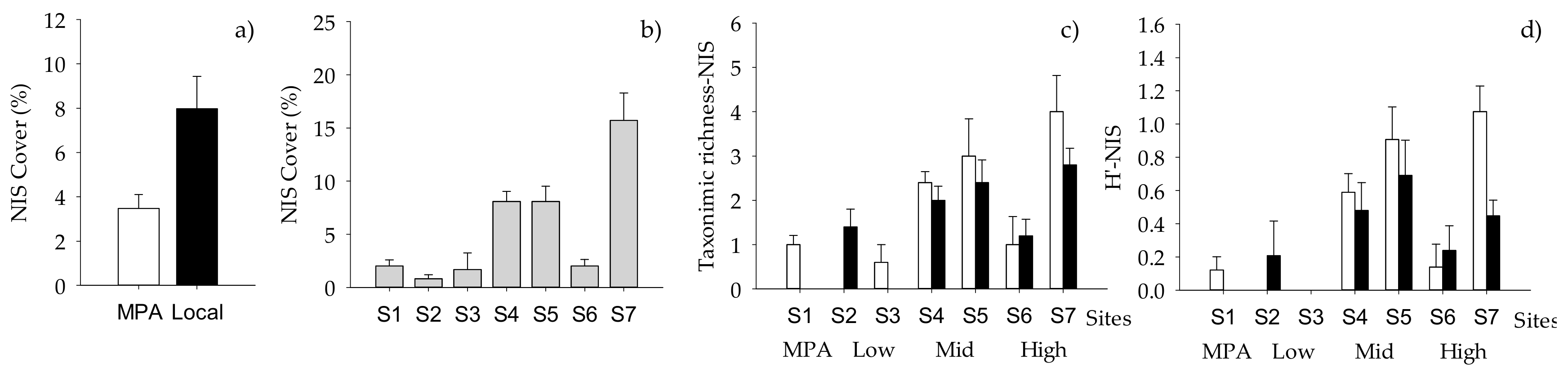
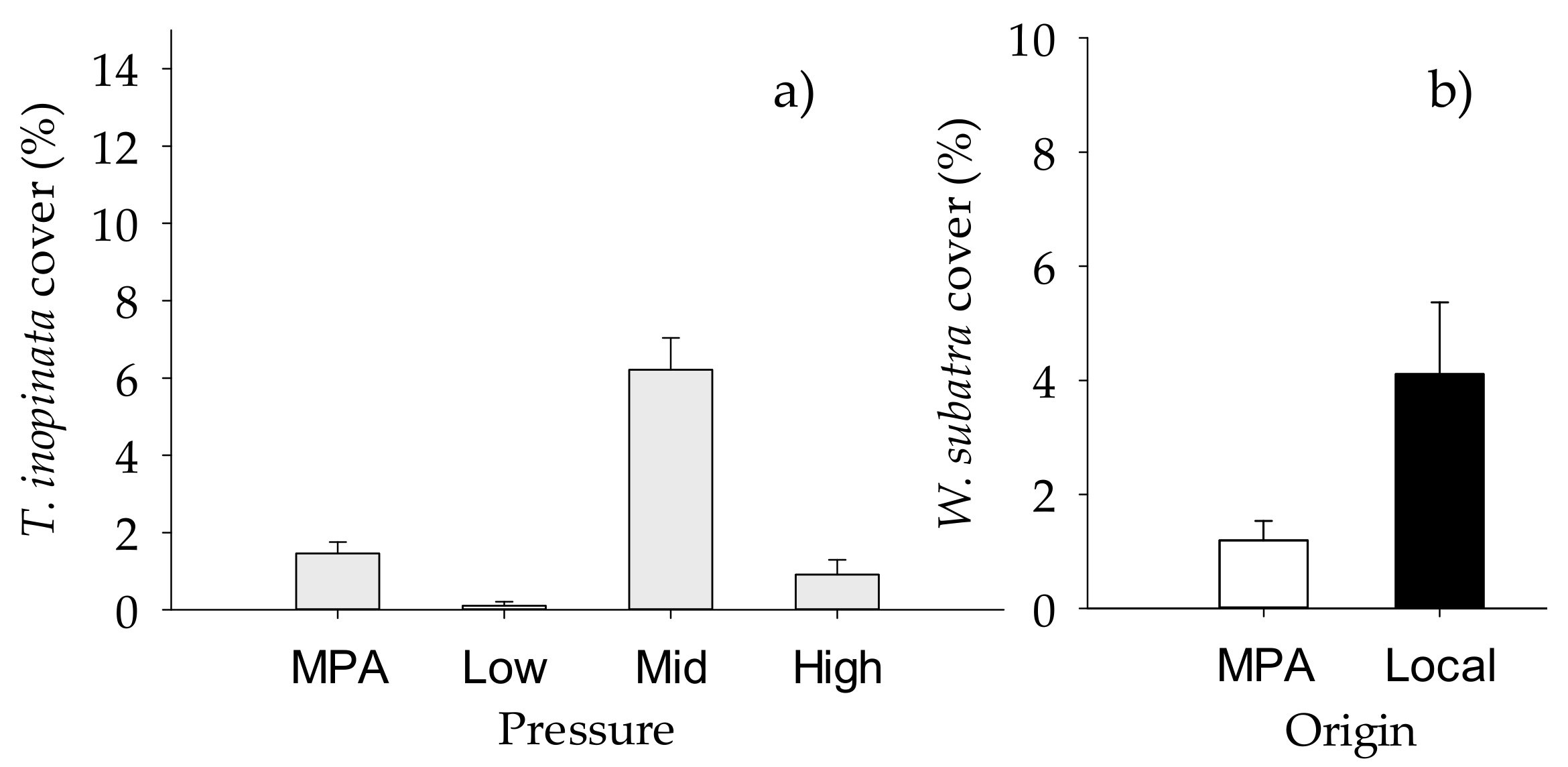
2.2. Structure of Macrofouling Assemblages
2.3. Functioning of Macrofouling Assemblage
3. Discussion
4. Materials and Methods
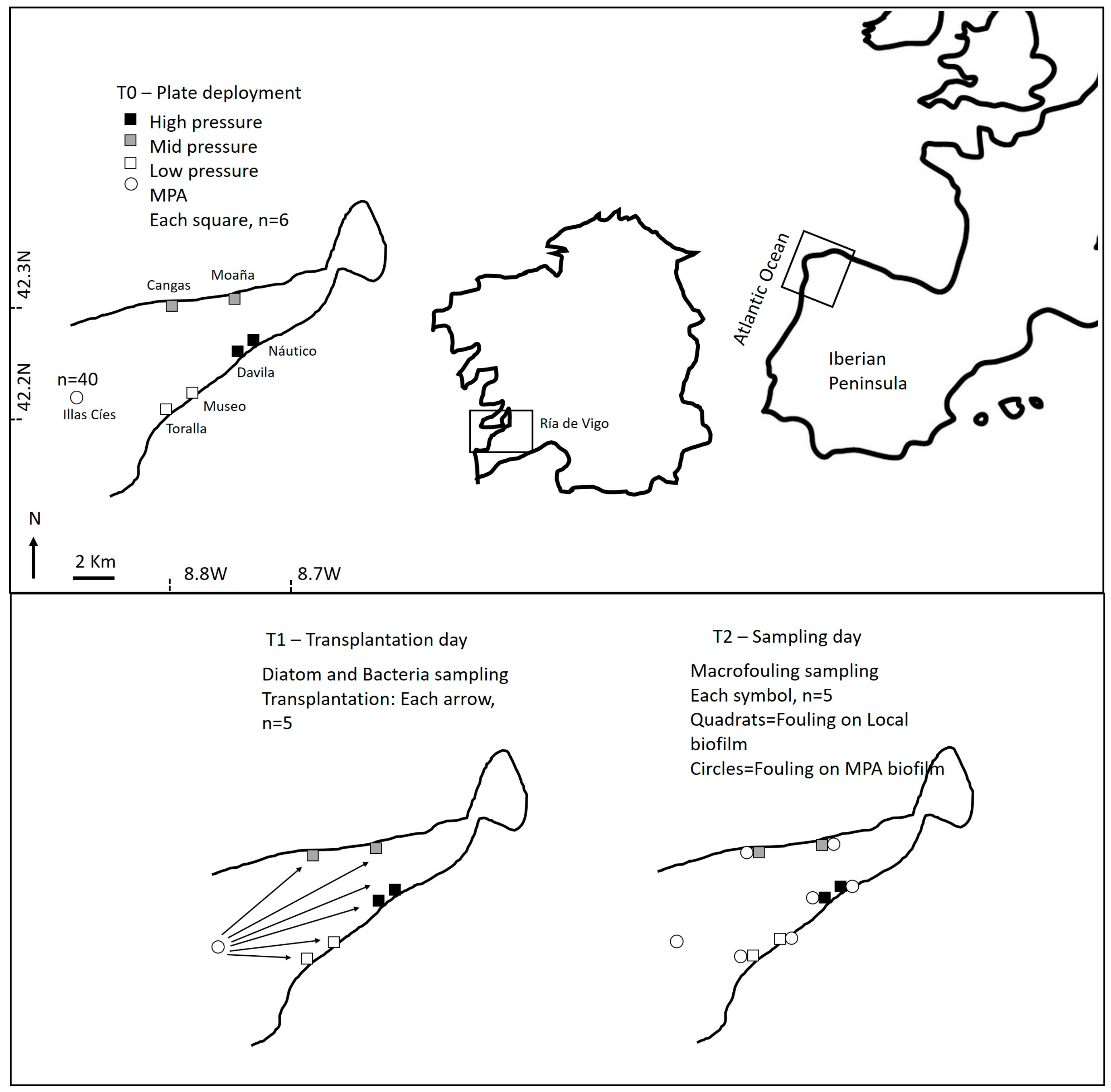
4.1. General Approach
4.2. Biofilm Characterization
4.1.1. Field Sampling
4.1.2. Laboratory Analysis
4.3. Transplantation Experiment
4.4. Macrofouling Characterization
4.5. Data Analysis
4.5.1. Biofilm
4.5.2. Macrofouling Assemblages
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| MPA | Marine protected areas |
| NIS | Non-indigenous species |
| Pr | Pressure |
| Or | Origin of biofilm |
| Si | Site |
| Pl | Panel |
| S | Taxonomic richness |
| H’ | Shannon diversity index |
| J | Pielou evenness index |
| N | Percentage cover |
| PERMDISP | Test for homogeneity of multivariate dispersion |
| SIMPER | Similarity percentages |
| nMDS | Non-metric multidimensional scaling |
| PERMANOVA | Permutational multivariate analysis of variance |
| MC | Monte Carlo |
References
- Global Invasive Species Programme. Marine Biofouling and Invasive Species: An Assessment of Risks and Management Initiatives; Compiled by Lynn Jackson on behalf of the Global Invasive Species Programme and the UNEP Regional Seas Programme; United Nations: Wellington, New Zealand, 2008; p. 68. [Google Scholar]
- Bressy, C.; Lejars, M.N. Marine Fouling: An Overview Marine antifouling. J. Ocean Technol. 2014, 9, 19–28. [Google Scholar]
- Fitridge, I.; Dempster, T.; Guenther, J.; de Nys, R. The impact and control of biofouling in marine aquaculture: A review. Biofouling 2012, 28, 649–669. [Google Scholar] [CrossRef]
- Molnar, J.L.; Gamboa, R.L.; Revenga, C.; Spalding, M.D. Assessing the global threat of invasive species to marine biodiversity. Front. Ecol. Environ. 2008, 6, 485–492. [Google Scholar] [CrossRef]
- Minchin, D.; Gollasch, S. Fouling and ships’ hulls: How changing circumstances and spawning events may result in the spread of exotic species. Biofouling 2003, 19, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Carlton, J.T.; Geller, J.B. Ecological Roulette: The Global Transport of Nonindigenous Marine Organisms. Science 1993, 261, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Dafforn, K.A.; Glasby, T.M.; Johnston, E.L. Links between estuarine condition and spatial distributions of marine invaders. Divers. Distrib. 2009, 15, 807–821. [Google Scholar] [CrossRef]
- Mineur, F.; Cook, E.; Minchin, D.; Bohn, K.; MacLeod, A.; Maggs, C. Changing coasts: Marine aliens and artificial structures. Oceanogr. Mar. Biol. An. Annu. Rev. 2012, 50, 189–234. [Google Scholar]
- Bax, N.; Williamson, A.; Aguero, M.; Gonzalez, E.; Geeves, W. Marine invasive alien species: A threat to global biodiversity. Mar. Policy 2003, 27, 313–323. [Google Scholar] [CrossRef]
- Wahl, M. Marine epibiosis. I. Fouling and antifouling: Some basic aspects. Mar. Ecol. Prog. Ser. 1989, 58, 175–189. [Google Scholar] [CrossRef]
- Railkin, A.I. Marine Biofouling: Colonization Processes and Defenses; Gant, T.A., Manylov, O., Eds.; CRC Press: Boca Raton, FL, USA, 2004; p. 316. [Google Scholar]
- Grzegorczyk, M.; Pogorzelski, S.J.; Pospiech, A.; Boniewicz-Szmyt, K. Monitoring of marine biofilm formation dynamics at submerged solid surfaces with multitechnique sensors. Front. Mar. Sci. 2018, 5, 363. [Google Scholar] [CrossRef]
- Dobretsov, S.; Thomason, J.C. The development of marine biofilms on two commercial non-biocidal coatings: A comparison between silicone and fluoropolymer technologies. Biofouling 2011, 27, 869–880. [Google Scholar] [CrossRef]
- Dang, H.; Lovell, C.R. Microbial Surface Colonization and Biofilm Development in Marine Environments. Microbiol. Mol. Biol. Rev. 2016, 80, 91–138. [Google Scholar] [CrossRef] [PubMed]
- Dang, H.; Li, T.; Chen, M.; Huang, G. Cross-ocean distribution of Rhodobacterales bacteria as primary surface colonizers in temperate coastal marine waters. Appl. Environ. Microbiol. 2008, 74, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Salta, M.; Wharton, J.A.; Blache, Y.; Stokes, K.R.; Briand, J.F. Marine biofilms on artificial surfaces: Structure and dynamics. Environ. Microbiol. 2013, 15, 2879–2893. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Lyu, Y.; Richlen, M.L.; Anderson, D.M.; Cai, Z. Quorum Sensing Is a Language of Chemical Signals and Plays an Ecological Role in Algal-Bacterial Interactions. CRC Crit. Rev. Plant. Sci. 2016, 35, 81–105. [Google Scholar] [CrossRef]
- Patil, J.; Anil, A.C. Biofilm diatom community structure: Influence of temporal and substratum variability. Biofouling 2005, 21, 189–206. [Google Scholar] [CrossRef]
- Rossi, F.; De Philippis, R. Role of cyanobacterial exopolysaccharides in phototrophic biofilms and in complex microbial mats. Life 2015, 5, 1218–1238. [Google Scholar] [CrossRef]
- Keough, M.J.; Raimondi, P.T. Responses of settling invertebrate larvae to bioorganic films: Effects of different types of films. J. Exp. Mar. Biol. Ecol. 1995, 185, 235–253. [Google Scholar] [CrossRef]
- Hadfield, M.G.; Paul, V.J. Natural Chemical Cues for Settlement and Metamorphosis of Marine-Invertebrate Larvae. In Marine Chemical Ecology; McClintock, J.B., Baker, B.J., Eds.; CRC Press: Boca Raton, FL, USA, 2001; pp. 431–461. [Google Scholar]
- Qian, P.Y.; Lau, S.C.K.; Dahms, H.U.; Dobretsov, S.; Harder, T. Marine biofilms as mediators of colonization by marine macroorganisms: Implications for antifouling and aquaculture. Mar. Biotechnol. 2007, 9, 399–410. [Google Scholar] [CrossRef]
- Dobretsov, S.; Abed, R.M.M.; Teplitski, M. Mini-review: Inhibition of biofouling by marine microorganisms. Biofouling 2013, 29, 423–441. [Google Scholar] [CrossRef]
- Krug, P.J. Defense of benthic invertebrates against surface colonization by larvae: A chemical arms race. Prog. Mol. Subcell. Biol. 2006, 42, 1–53. [Google Scholar] [PubMed]
- Bao, W.-Y. A Study on Roles of Naturally Formed Biofilms and Cultured Bacterial Biofilms on Larval Settlement and Metamorphosis of the Mussel Mytilus galloprovincialis. Ph.D. Thesis, Nagasaki University, Nagasaki, Japan, 2008; p. 78. [Google Scholar]
- Cao, S.; Wang, J.D.; Chen, H.S.; Chen, D.R. Progress of marine biofouling and antifouling technologies. Chin. Sci. Bull. 2011, 56, 598–612. [Google Scholar] [CrossRef]
- Hadfield, M.G. Biofilms and Marine Invertebrate Larvae: What Bacteria Produce That Larvae Use to Choose Settlement Sites. Ann. Rev. Mar. Sci. 2011, 3, 453–470. [Google Scholar] [CrossRef] [PubMed]
- Wieczorek, S.K.; Clare, A.S.; Todd, C.D. Inhibitory and facilitatory effects of microbial films on settlement of Balanus amphitrite amphitrite larvae. Mar. Ecol. Prog. Ser. 1995, 119, 221–228. [Google Scholar] [CrossRef]
- Hung, O.S.; Thiyagarajan, V.; Wu, R.S.S.; Qian, P.Y. Effect of ultraviolet radiation on biofilms and subsequent larval settlement of Hydroides elegans. Mar. Ecol. Prog. Ser. 2005, 304, 155–166. [Google Scholar] [CrossRef]
- Huang, Y.L.; Dobretsov, S.; Xiong, H.; Qian, P.Y. Effect of biofilm formation by Pseudoalteromonas spongiae on induction of larval settlement of the polychaete Hydroides elegans. Appl. Environ. Microbiol. 2007, 73, 6284–6288. [Google Scholar] [CrossRef]
- De Carvalho, C.C.C.R. Marine biofilms: A successful microbial strategy with economic implications. Front. Mar. Sci. 2018, 5, 1–11. [Google Scholar] [CrossRef]
- Sweat, L.H.; Swain, G.W.; Hunsucker, K.Z.; Johnson, K.B. Transported biofilms and their influence on subsequent macrofouling colonization. Biofouling 2017, 33, 433–449. [Google Scholar] [CrossRef]
- Dahms, H.-U.; Dobretsov, S.; Qian, P.-Y. The effect of bacterial and diatom biofilms on the settlement of the bryozoan Bugula neritina. J. Exp. Mar. Bio. Ecol. 2004, 313, 191–209. [Google Scholar] [CrossRef]
- Webster, N.O.; Negri, A. Site-specific variation in Antarctic marine biofilms established on artificial surfaces. Environ. Microbiol. 2006, 8, 1177–1190. [Google Scholar] [CrossRef]
- Lau, S.C.K.; Thiyagarajan, V.; Cheung, S.C.K.; Qian, P.Y. Roles of bacterial community composition in biofilms as a mediator for larval settlement of three marine invertebrates. Aquat. Microb. Ecol. 2005, 38, 41–51. [Google Scholar] [CrossRef]
- Chiu, J.M.Y.; Zhang, R.; Wang, H.; Thiyagarajan, V.; Qian, P.Y. Nutrient effects on intertidal community: From bacteria to invertebrates. Mar. Ecol. Prog. Ser. 2008, 358, 41–50. [Google Scholar] [CrossRef][Green Version]
- Wahl, M.; Goecke, F.; Labes, A.; Dobretsov, S.; Weinberger, F. The second skin: Ecological role of epibiotic biofilms on marine organisms. Front. Microbiol. 2012, 3, 1–21. [Google Scholar] [CrossRef]
- Thompson, R.C.; Norton, T.A.; Hawkins, S.J. Physical stress and biological control regulate the producer-consumer balance in intertidal biofilms. Ecology 2005, 85, 1372–1382. [Google Scholar] [CrossRef]
- Stocker, R. Marine microbes see a sea of gradients. Science 2012, 338, 628–633. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, S.J.; Moore, P.J.; Burrows, M.T.; Poloczanska, E.; Mieszkowska, N.; Herbert, R.J.H.; Jenkins, S.R.; Thompson, R.C.; Genner, M.J.; Southward, A.J. Complex interactions in a rapidly changing world: Responses of rocky shore communities to recent climate change. Clim. Res. 2008, 37, 123–133. [Google Scholar] [CrossRef]
- Gerwing, T.G.; Drolet, D.; Hamilton, D.J.; Barbeau, M.A. Relative Importance of Biotic and Abiotic Forces on the Composition and Dynamics of a Soft-Sediment Intertidal Community. PLoS ONE 2016, 11, 1–15. [Google Scholar] [CrossRef]
- Naser, H.A. Variability of marine macrofouling assemblages in a marina and a mariculture centre in Bahrain, Arabian Gulf. Reg. Stud. Mar. Sci. 2017, 16, 162–170. [Google Scholar] [CrossRef]
- Nogueira, E.; Pérez, F.F.; Ríos, A.F. Seasonal Patterns and Long-term Trends in an Estuarine Upwelling Ecosystem (Ría de Vigo, NW Spain). Estuar. Coast. Shelf Sci. 1997, 44, 285–300. [Google Scholar] [CrossRef]
- Vilas, F.; Bernabeu, A.M.; Méndez, G. Sediment distribution pattern in the Rias Baixas (NW Spain): Main facies and hydrodynamic dependence. J. Mar. Syst. 2005, 54, 261–276. [Google Scholar] [CrossRef]
- Gilconto, M.; Pardo, P.C.; Álvarez-Salgado, X.A.; Pérez, F.F. Exchange fluxes between the Ría de Vigo and the shelf: A bidirectional flow forced by remote wind. J. Geophys. Res. 2007, 112. [Google Scholar] [CrossRef]
- Babarro, J.M.F.; Carrington, E. Byssus secretion of Mytilus galloprovincialis: Effect of site at macro- and micro-geographical scales within Ŕa de Vigo (NW Spain). Mar. Ecol. Prog. Ser. 2011, 435, 125–140. [Google Scholar] [CrossRef]
- Mitarai, S.; Siegel, D.A.; Watson, J.R.; Dong, C.; McWilliams, J.C. Quantifying connectivity in the coastal ocean with application to the Southern California Bight. J. Geophys. Res. Ocean. 2009, 114, 1–21. [Google Scholar] [CrossRef]
- Watson, J.R.; Mitarai, S.; Siegel, D.A.; Caselle, J.E.; Dong, C.; McWilliams, J.C. Realized and potential larval connectivity in the southern California bight. Mar. Ecol. Prog. Ser. 2010, 401, 31–48. [Google Scholar] [CrossRef]
- Marshall, D.J.; Keough, M.J. Variation in the dispersal potential of non-feeding invertebrate larvae: The desperate larva hypothesis and larval size. Mar. Ecol. Prog. Ser. 2003, 255, 145–153. [Google Scholar] [CrossRef]
- Sams, M.A.; Warren-Myers, F.; Keough, M.J. Increased larval planktonic duration and post-recruitment competition influence survival and growth of the bryozoan Watersipora subtorquata. Mar. Ecol. Prog. Ser. 2015, 531, 179–191. [Google Scholar] [CrossRef]
- Nogata, Y.; Yoshimura, E.; Shinshima, K.; Kitano, Y.; Sakaguchi, I. Antifouling substances against larvae of the barnacle Balanus amphitrite from the marine sponge, Acanthella cavernosa. Biofouling 2003, 19, 193–196. [Google Scholar] [CrossRef]
- Dürr, S.; Thomason, J.C. Biofouling; Wiley-Blackwell: Chichester, UK, 2008; p. 456. [Google Scholar]
- Butler, A.J.; Connolly, R.M. Assemblages of sessile marine invertebrates: Still changing after all these years? Mar. Ecol. Prog. Ser. 1999, 182, 109–118. [Google Scholar] [CrossRef]
- Wahl, M.; Link, H.; Alexandridis, N.; Thomason, J.C.; Cifuentes, M.; Costello, M.J.; da Gama, B.A.P.; Hillock, K.; Hobday, A.J.; Kaufmann, M.J.; et al. Re-structuring of marine communities exposed to environmental change: A global study on the interactive effects of species and functional richness. PLoS ONE 2011, 6, 3–5. [Google Scholar] [CrossRef]
- Keough, M.J.; Raimondi, P.T. Responses of settling invertebrate larvae to bioorganic films: Effects of large-scale variation in films. J. Exp. Mar. Biol. Ecol. 1996, 207, 59–78. [Google Scholar] [CrossRef]
- Lockwood, J.L.; Cassey, P.; Blackburn, T. The role of propagule pressure in explaining species invasions. Trends Ecol. Evol. 2005, 20, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Floerl, O.; Inglis, G.J.; Dey, K.; Smith, A. The importance of transport hubs in stepping-stone invasions. J. Appl. Ecol. 2009, 46, 37–45. [Google Scholar] [CrossRef]
- Foster, V.; Giesler, R.J.; Wilson, A.M.W.; Nall, C.R.; Cook, E.J. Identifying the physical features of marina infrastructure associated with the presence of non-native species in the UK. Mar. Biol. 2016, 163, 1–14. [Google Scholar] [CrossRef]
- López-Legentil, S.; Legentil, M.L.; Erwin, P.M.; Turon, X. Harbor networks as introduction gateways: Contrasting distribution patterns of native and introduced ascidians. Biol. Invasions 2015, 17, 1623–1638. [Google Scholar] [CrossRef] [PubMed]
- Ferrario, J.; Caronni, S.; Occhipinti-Ambrogi, A.; Marchini, A. Role of commercial harbours and recreational marinas in the spread of non-indigenous fouling species. Biofouling 2017, 33, 651–660. [Google Scholar] [CrossRef]
- Page, H.M.; Simons, R.D.; Zaleski, S.F.; Miller, R.J.; Dugan, J.E.; Schroeder, D.M.; Doheny, B.; Goddard, J.H.R. Distribution and potential larval connectivity of the non-native Watersipora (Bryozoa) among harbors, offshore oil platforms, and natural reefs. Aquat. Invasions 2019, 14, 615–637. [Google Scholar] [CrossRef]
- Clark, G.F.; Johnston, E.L. Propagule pressure and disturbance interact to overcome biotic resistance of marine invertebrate communities. Oikos 2009, 118, 1679–1686. [Google Scholar] [CrossRef]
- Ramalhosa, P.; Gestoso, I.; Duarte, B.; Caçador, I.; Canning-Clode, J. Metal pollution affects both native and non-indigenous biofouling recruitment in a subtropical island system. Mar. Pollut. Bull. 2019, 141, 373–386. [Google Scholar] [CrossRef]
- Piola, R.F.; Johnston, E.L. Differential resistance to extended copper exposure in four introduced bryozoans. Mar. Ecol. Prog. Ser. 2006, 311, 103–114. [Google Scholar] [CrossRef]
- Mackie, J.A.; Darling, J.A.; Geller, J.B. Ecology of cryptic invasions: Latitudinal segregation among Watersipora (Bryozoa) species. Sci. Rep. 2012, 2, 871. [Google Scholar] [CrossRef]
- Lauer, A. Watersipora subtorquata and the Possible Role of Its Associated Microbes: An Attempt to Explain the Extraordinary Invasion Success of This Marine Bryozoan Species. In The Mechanistic Benefits of Microbial Symbionts Advances in Environmental Microbiology; Hurst, C.J., Ed.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 239–268. [Google Scholar]
- Carlton, J.T. Transoceanic and interoceanic dispersal of coastal marine organisms: The biology of ballast water. Oceanogr. Mar. Biol. An. Annu. Rev. 1985, 23, 313–371. [Google Scholar]
- Trevathan-Tackett, S. A horizon scan of priorities for coastal microbiome research. Nat. Ecol. Evol. 2019, 3, 1509–1520. [Google Scholar] [CrossRef] [PubMed]
- El Nagar, A.; Huys, R.; Bishop, J.D.D. Widespread occurrence of the Southern Hemisphere ascidian Corella eumyota Traustedt, 1882 on the Atlantic coast of Iberia. Aquat. Invasions 2010, 5, 169–173. [Google Scholar] [CrossRef]
- Canning-Clode, J.; Fofonoff, P.; McCann, L.; Carlton, J.T.; Ruiz, G. Marine invasions on a subtropical island: Fouling studies and new records in a recent marina on Madeira island (Eastern Atlantic Ocean). Aquat. Invasions 2013, 8, 261–270. [Google Scholar] [CrossRef]
- Marraffini, M.L.; Ashton, G.V.; Brown, C.W.; Chang, A.L.; Ruiz, G.M. Settlement plates as monitoring devices for non-indigenous species in marine fouling communities. Manag. Biol. Invasions 2017, 8, 559–566. [Google Scholar] [CrossRef]
- Echenique-Subiabre, I.; Dalle, C.; Duval, C.; Heath, M.W.; Couté, A.; Wood, S.A.; Humbert, J.; Quiblier, C. Application of a spectrofluorimetric tool (bbe BenthoTorch) for monitoring potentially toxic benthic cyanobacteria in rivers. Water Res. 2016, 101, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Herlemann, D.P.R.; Labrenz, M.; Jürgens, K.; Bertilsson, S.; Waniek, J.J.; Andersson, A.F. Transitions in bacterial communities along the 2000 km salinity gradient of the Baltic Sea. ISME J. 2011, 5, 1571–1579. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Round, F.E.; Crawford, R.M.; Mann, D.G. The Diatoms. Biology and Morphology of the Genera; Cambridge University Press: Cambridge, UK, 1990; p. 747. [Google Scholar]
- Hasle, G.R.; Syvertsen, E.E. Marine Diatoms. In Identifying Marine Diatoms and Dinoflagellates; Tomas, C.R., Ed.; Academic Press, Inc.: San Diego, CA, USA, 1996; pp. 1–385. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 1. Teil: Naviculaceae. In Süßwasserflora von Mitteleuropa, Band 2/1; Ettl, H., Gärtner, G., Gerloff, J., Heynig, H., Mollenhauer, D., Eds.; Gustav Fischer Verlag: Berlin/Heidelberg, Germany, 1986; p. 876. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 2. Teil: Bacillariaceae, Epithemiaceae, Surirellaceae. In Süßwasserflora von Mitteleuropa, Band 2/2; Ettl, H., Gärtner, G., Gerloff, J., Heynig, H., Mollenhauer, D., Eds.; Gustav Fischer Verlag: Berlin/Heidelberg, Germany, 1988; p. 611. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 3. Teil: Centrales, Fragilariaceae, Eunotiaceae. In Süßwasserflora von Mitteleuropa, Band 2/3; Ettl, H., Gärtner, G., Gerloff, J., Heynig, H., Mollenhauer, D., Eds.; Gustav Fischer Verlag: Berlin/Heidelberg, Germany, 1991; p. 599. [Google Scholar]
- Krammer, K.; Lange-Bertalot, H. Bacillariophyceae 4. Teil: Achnanthaceae. In Süßwasserfloravon Mitteleuropa, Band 2/4; Ettl, H., Gärtner, G., Gerloff, J., Heynig, H., Mollenhauer, D., Eds.; Gustav Fischer Verlag: Berlin/Heidelberg, Germany, 1991; p. 437. [Google Scholar]
- Lange-Bertalot, H. 85 neue Taxa und über 100 weitere neu definierte Taxa ergänzend zur Süsswasserflora von Mitteleuropa 2/1-4. Bibl. Diatomol. 1993, 27, 1–454. [Google Scholar]
- Lange-Bertalot, H. Navicula sensu stricto. 10 genera separated from Navicula sensu lato. Frustulia. In Diatoms of Europe; Lange-Bertalot, H., Ed.; A.R.G. Gantner Verlag K.G.: Ruggell, Liechtenstein, 2001; Volume 2, p. 526. [Google Scholar]
- Levkov, Z.; Caput Mihalič, K.; Ector, L. A taxonomical study of Rhoicosphenia Grunow (Bacillariophyceae) with a key for identification of selected taxa. Fottea 2010, 10, 145–200. [Google Scholar] [CrossRef]
- Trobajo, R.; Rovira, L.; Ector, L.; Wetzel, C.E.; Kelly, M.; Mann, D.G. Morphology and identity of some ecologically important small Nitzschia species. Diatom Res. 2013, 28, 37–59. [Google Scholar] [CrossRef]
- Witkowski, A.; Lange-Bertalot, H.; Metzeltin, D. Diatom Flora of Marine Coasts I; Lange-Bertalot, H., Ed.; Koeltz Scientific Books: Königstein, Germany, 2000; p. 925. [Google Scholar]
- Bokulich, N.A.; Kaehler, B.D.; Rideout, J.R.; Dillon, M.; Bolyen, E.; Knight, R.; Huttley, G.A.; Gregory Caporaso, J. Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2’s q2-feature-classifier plugin. Microbiome 2018, 6, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Vázquez, E.; Lafargue, F.; Urgorri, V. Nuevos datos sobre la distribución de trece especies de la familia Didemnidae (Tunicata, Ascidiacea) en las costas ibéricas. Bol. Real Soc. Esp. Hist. Nat. (SEc. Biol.) 1994, 91, 219–229. [Google Scholar]
- Reverter-Gil, O.; Fernández-Pulpeiro, E. Inventario y Cartografía de Los Briozoos Marinos de Galicia (N.O. de España); Monografás de Nova Acta Científica Compostelana, Serie Biología; Universidad de Santiago de Compostela: Santiago de Compostela, Spain, 2001; p. 243. [Google Scholar]
- Kerckhof, F.; Haelters, J.; Gollasch, S. Alien species in the marine and brackish ecosystem: The situation in Belgian waters. Aquat. Invasions 2007, 2, 243–257. [Google Scholar] [CrossRef]
- Minchin, D.; Cook, E.J.; Clark, P.F. Alien species in British brackish and marine waters. Aquat. Invasions 2013, 8, 3–19. [Google Scholar] [CrossRef]
- Chainho, P.; Fernandes, A.; Amorim, A.; Ávila, S.; Canning-Clode, J.; Castro, J.J.; Costa, A.C.; Costa, J.; Cruz, T.; Gollasch, S.; et al. Non-indigenous species in Portuguese coastal areas, coastal lagoons, estuaries, and islands. Estuar. Coast. Shelf Sci. 2015, 167, 199–211. [Google Scholar] [CrossRef]
- AquaNIS Information System on Aquatic Non-indigenous and Cryptogenic Species. World Wide Web Electronic Publication. Version 2.36+. 2015. Available online: http://www.corpi.ku.lt/databases/aquanis (accessed on 18 September 2019).
- Ahyong, S.; Galil, B.; Gollasch, S.; Hutchings, P.; Katsanevakis, S.; Lejeusne, C.; Marchini, A.; Occhipinti, A.; Pagad, S.; Poore, G.; et al. World Register of Introduced Marine Species (WRiMS). 2018. Available online: http://www.marinespecies.org/introduced (accessed on 14 October 2018).
- Fofonoff, P.; Ruiz, G.; Steves, B.; Simkanin, C.; Carlton, J. National Exotic Marine and Estuarine Species Information System. 2018. Available online: http://invasions.si.edu/nemesis/ (accessed on 18 September 2019).
- WoRMS Editorial Board World Register of Marine Species (WoRMS). Available online: http://www.marinespecies.org (accessed on 4 May 2018).
- Kohler, K.E.; Gill, S.M. Coral Point Count with Excel extensions (CPCe): A Visual Basic program for the determination of coral and substrate coverage using random point count methodology. Comput. Geosci. 2006, 32, 1259–1269. [Google Scholar] [CrossRef]
- Anderson, M.; Gorley, R.N.; Clarke, K.R. Permanova+ for Primer: Guide to Software and Statistical Methods; PRIMER-E Ltd: Plymouth, UK, 2008; p. 214. [Google Scholar]
- Anderson, M.J. Permutational Multivariate Analysis of Variance (PERMANOVA). Wiley StatsRef Stat. 2017, 1–15. [Google Scholar] [CrossRef]
- Clarke, K.R.; Warwick, R.M. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation; PRIMER-E Ltd: Plymouth, UK, 2001; p. 176. [Google Scholar]
- Clarke, K.R.; Gorley, R.N. PRIMER v6: User Manual/Tutorial (Plymouth Routines in Multivariate Ecological Research); PRIMER-E Ltd.: Plymouth, UK, 2006. [Google Scholar]
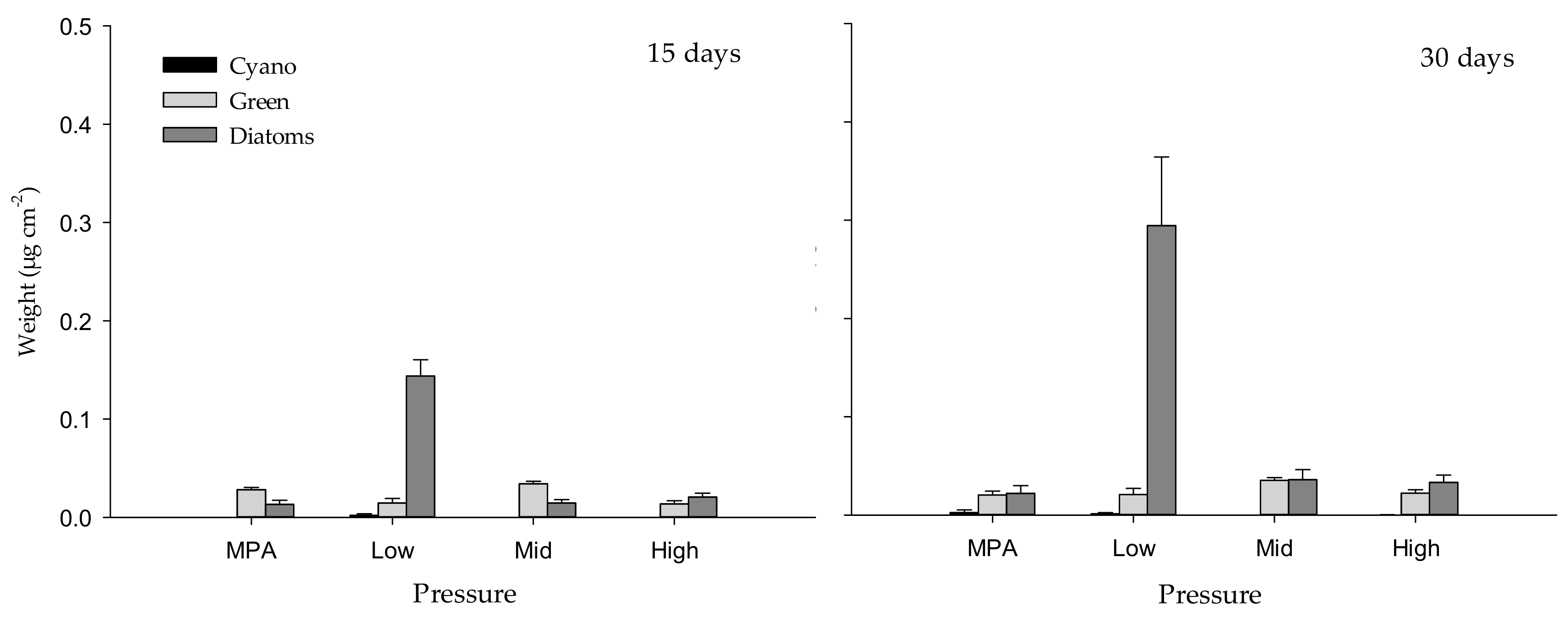

| 15 days | 30 days | |||
|---|---|---|---|---|
| Source | df | Pseudo-F | df | Pseudo-F |
| Pr | 3 | 4.551 * | 3 | 6.108 ** |
| Si(Pr) | 3 | 5.888 *** | 3 | 2.831 ** |
| Pl(Si(Pr)) | 32 | 2.968 *** | 31 | 2.677 *** |
| Res | 76 | 76 | ||
| Total | 114 | 113 | ||
| PERMDISP | P(perm): 0.004 | P(perm) ns |
| Bacteria | Diatoms | |||||||
|---|---|---|---|---|---|---|---|---|
| (a) Total Assemblage | (b) | S | H’ | J | (c) Total assemblage | |||
| Source | df | Pseudo-F | df | Pseudo-F | Pseudo-F | Pseudo-F | df | Pseudo-F |
| Pr | 3 | 2.408 ** | 3 | 12.642 (p = 0.051) | 1.712 | 0.459 | 3 | 1.682 * |
| Res | 3 | 3 | 2 | |||||
| Total | 6 | 6 | 5 | |||||
| (a) Low (Av. Similarity: 67.97%) | Av.Abund | Av.Sim | Contrib% | ||
| Bacteroidia_uncultured | 8.64 | 0.92 | 1.35 | ||
| Bacteroidia_Ulvibacter | 6.63 | 0.69 | 1.01 | ||
| Gammaproteobacteria_Halioglobus | 7.11 | 0.68 | 1.01 | ||
| Gammaproteobacteria_Halioglobus | 6.19 | 0.67 | 0.98 | ||
| Bacteroidia_Aquibacter | 6.02 | 0.67 | 0.98 | ||
| Gammaproteobacteria_Granulosicoccus | 6.25 | 0.64 | 0.94 | ||
| Gammaproteobacteria_Leucothrix | 6.51 | 0.64 | 0.94 | ||
| Bacteroidia_Lewinella | 5.99 | 0.62 | 0.92 | ||
| Alphaproteobacteria_Loktanella | 6.42 | 0.61 | 0.9 | ||
| Bacteroidia_Lutimonas | 6.12 | 0.61 | 0.9 | ||
| Oxyphotobacteria_Minutocellus sp. CCMP1701 | 5.74 | 0.61 | 0.9 | ||
| Mid (Av. Similarity: 74.44%) | Av.Abund | Av.Sim | Contrib% | ||
| Bacteroidia_Ulvibacter | 9.05 | 0.79 | 1.07 | ||
| Bacteroidia_uncultured | 7.58 | 0.66 | 0.88 | ||
| Gammaproteobacteria_Halioglobus | 7.71 | 0.64 | 0.86 | ||
| Bacteroidia_Aquibacter | 6.97 | 0.61 | 0.82 | ||
| Alphaproteobacteria_Loktanella | 6.93 | 0.6 | 0.81 | ||
| Bacteroidia_Dokdonia | 6.84 | 0.57 | 0.77 | ||
| Gammaproteobacteria_Halioglobus | 6.92 | 0.55 | 0.74 | ||
| Bacteroidia_Winogradskyella | 6.41 | 0.55 | 0.74 | ||
| Gammaproteobacteria_Marinicella | 6.24 | 0.54 | 0.72 | ||
| Bacteroidia_uncultured | 5.97 | 0.52 | 0.7 | ||
| Oxyphotobacteria_Minutocellus sp. CCMP1701 | 6.37 | 0.51 | 0.68 | ||
| Alphaproteobacteria_Sulfitobacter | 6.22 | 0.5 | 0.67 | ||
| Bacteroidia_Maribacter | 5.57 | 0.5 | 0.67 | ||
| High (Av. Similarity: 70.05%) | Av.Abund | Av.Sim | Contrib% | ||
| Gammaproteobacteria_Francisella | 8.52 | 1 | 1.43 | ||
| Bacteroidia_Pseudofulvibacter | 8.2 | 0.97 | 1.38 | ||
| Bacteroidia_Ulvibacter | 8.36 | 0.95 | 1.36 | ||
| Alphaproteobacteria_Amylibacter | 8.87 | 0.95 | 1.35 | ||
| Alphaproteobacteria_Candidatus Megaira | 7.45 | 0.88 | 1.25 | ||
| Oxyphotobacteria_bacterium WHC4–2 | 8.15 | 0.88 | 1.25 | ||
| Bacteroidia_uncultured | 7.34 | 0.84 | 1.19 | ||
| Gammaproteobacteria_Halioglobus | 6.4 | 0.79 | 1.12 | ||
| (b) High and Low (Av. Dissimilarity: 46.81%) | High Av.Abund | Low Av.Abund | Av.Diss | Diss/SD | Contrib% |
| Gammaproteobacteria_Francisella | 8.52 | 0 | 0.5 | 224.12 | 1.06 |
| Bacteroidia_Pseudofulvibacter | 8.2 | 1.76 | 0.38 | 3.16 | 0.8 |
| Gammaproteobacteria_Thiothrix | 3.63 | 8.88 | 0.31 | 1.33 | 0.66 |
| Gammaproteobacteria_uncultured | 5.15 | 0 | 0.3 | 5.95 | 0.64 |
| Campylobacteria_Arcobacter | 4.94 | 0 | 0.29 | 2.25 | 0.63 |
| Bacteroidia_Bernardetia | 4.62 | 0 | 0.27 | 2.57 | 0.58 |
| Oxyphotobacteria_Virgulinella fragilis | 0 | 4.66 | 0.27 | 4.7 | 0.58 |
| Gammaproteobacteria_Aliikangiella | 4.54 | 0 | 0.26 | 46.84 | 0.57 |
| Deinococci_Truepera | 1 | 5.42 | 0.26 | 3.25 | 0.56 |
| Alphaproteobacteria_Candidatus Megaira | 7.45 | 3.03 | 0.26 | 9.61 | 0.55 |
| Alphaproteobacteria_Allorhizobium–Neorhizobium–Pararhizobium–Rhizobium | 1.04 | 5.27 | 0.25 | 2.41 | 0.53 |
| Bacteroidia_Mesoflavibacter | 4.31 | 0 | 0.25 | 3.7 | 0.53 |
| Alphaproteobacteria_Roseobacter clade NAC11-7 lineage | 4.05 | 0 | 0.24 | 1.86 | 0.52 |
| Oxyphotobacteria_Podosira stelligera | 0 | 4.06 | 0.24 | 5.31 | 0.51 |
| Bacteroidia_[Polaribacter] huanghezhanensis | 4.03 | 0 | 0.23 | 11.67 | 0.5 |
| Alphaproteobacteria_Jannaschia | 5.07 | 1.16 | 0.23 | 2.08 | 0.49 |
| Oxyphotobacteria_bacterium WHC4-2 | 8.15 | 4.2 | 0.23 | 3.16 | 0.49 |
| High and Mid (Av. Dissimilarity: 39.40%) | High Av.Abund | Mid Av.Abund | Av.Diss | Diss/SD | Contrib% |
| Gammaproteobacteria_uncultured | 0 | 6.4 | 0.33 | 1.96 | 0.84 |
| Gammaproteobacteria_Francisella | 8.52 | 2.45 | 0.31 | 2.1 | 0.8 |
| Alphaproteobacteria_Halocynthiibacter | 4.08 | 0 | 0.21 | 2.48 | 0.54 |
| Oxyphotobacteria_bacterium WHC4-2 | 8.15 | 4.28 | 0.2 | 2.56 | 0.5 |
| Deltaproteobacteria_Desulfosarcina | 0 | 3.72 | 0.19 | 10.2 | 0.49 |
| Gammaproteobacteria_Verticia | 1.45 | 5.23 | 0.19 | 1.61 | 0.49 |
| Gammaproteobacteria_Marimicrobium | 0 | 3.61 | 0.19 | 5.12 | 0.48 |
| Oxyphotobacteria_Proteobacteria bacterium JGI 0000113-L05 | 7.11 | 3.49 | 0.19 | 2.79 | 0.47 |
| Bacteroidia_Kordia | 3.44 | 0 | 0.18 | 2.53 | 0.46 |
| Gammaproteobacteria_Candidatus Thiobios | 0 | 3.45 | 0.18 | 12.83 | 0.45 |
| Alphaproteobacteria_Thalassobius | 3.49 | 0 | 0.18 | 2.66 | 0.45 |
| Bacteroidia_Muriicola | 0 | 3.42 | 0.18 | 13.22 | 0.45 |
| Actinobacteria_Intrasporangiaceae | 0 | 3.39 | 0.18 | 20.42 | 0.45 |
| Gammaproteobacteria_uncultured | 1.89 | 5.2 | 0.18 | 1.43 | 0.44 |
| Thermoanaerobaculia_Subgroup 24 | 0 | 3.35 | 0.17 | 8.04 | 0.44 |
| Bacteroidia_Algibacter | 0 | 3.32 | 0.17 | 14.55 | 0.44 |
| Gammaproteobacteria_Perspicuibacter | 3.13 | 0 | 0.16 | 2.94 | 0.42 |
| Bacteroidia_Eudoraea | 0 | 3.16 | 0.16 | 22.31 | 0.42 |
| Bacteroidia_Bernardetia | 4.62 | 1.53 | 0.16 | 1.26 | 0.41 |
| Deltaproteobacteria_Sva0081 sediment group | 0 | 3.14 | 0.16 | 8.13 | 0.41 |
| Verrucomicrobiae_Roseibacillus | 1.37 | 4.46 | 0.16 | 1.82 | 0.41 |
| Low and Mid (Av. Dissimilarity: 36.13%) | Low Av.Abund | Mid Av.Abund | Av.Diss | Diss/SD | Contrib% |
| Gammaproteobacteria_Thiothrix | 8.88 | 3.85 | 0.25 | 1.27 | 0.69 |
| Gammaproteobacteria_uncultured | 3.18 | 6.4 | 0.23 | 1.51 | 0.63 |
| Deinococci_Truepera | 5.42 | 1.17 | 0.21 | 3.08 | 0.58 |
| Gammaproteobacteria_Alcanivorax | 0 | 4.21 | 0.21 | 7.79 | 0.58 |
| Bacteroidia_Pseudofulvibacter | 1.76 | 5.83 | 0.2 | 1.94 | 0.56 |
| Oxyphotobacteria_Podosira stelligera | 4.06 | 0 | 0.2 | 5.5 | 0.55 |
| Gammaproteobacteria_Cycloclasticus | 0 | 4.04 | 0.2 | 14.3 | 0.55 |
| Gracilibacteria_Gracilibacteria | 4.46 | 1.25 | 0.17 | 1.22 | 0.48 |
| Bacteroidia_[Polaribacter] huanghezhanensis | 0 | 3.42 | 0.17 | 3.53 | 0.47 |
| Actinobacteria_Intrasporangiaceae | 0 | 3.39 | 0.17 | 46.32 | 0.46 |
| Gammaproteobacteria_Oleiphilus | 0 | 3.38 | 0.17 | 6.85 | 0.46 |
| Gammaproteobacteria_Verticia | 2.2 | 5.23 | 0.17 | 1.33 | 0.46 |
| Oxyphotobacteria_Phormidesmis ANT.LACV5.1 | 3.33 | 0 | 0.16 | 8.29 | 0.46 |
| Campylobacteria_Arcobacter | 0 | 3.23 | 0.16 | 3.44 | 0.44 |
| Gammaproteobacteria_Hydrogenophaga | 0 | 3.2 | 0.16 | 7.9 | 0.44 |
| Bacteroidia_Polaribacter 5 | 0 | 3.17 | 0.16 | 3.07 | 0.43 |
| Gammaproteobacteria_Candidatus Tenderia | 0 | 3.14 | 0.16 | 4.15 | 0.43 |
| Alphaproteobacteria_Roseobacter | 0 | 3.09 | 0.15 | 20.29 | 0.42 |
| Gammaproteobacteria_uncultured | 0 | 3.09 | 0.15 | 11.82 | 0.42 |
| Gammaproteobacteria_Coxiella | 1.16 | 4.23 | 0.15 | 1.2 | 0.42 |
| Gammaproteobacteria_Psychromonas | 4.02 | 0.97 | 0.15 | 2.53 | 0.42 |
| High and MPA (Av. Dissimilarity: 35.54%) | High Av.Abund | MPA Av.Abund | Av.Diss | Diss/SD | Contrib% |
| Gammaproteobacteria_Francisella | 8.52 | 0 | 0.51 | 309.51 | 1.45 |
| Gracilibacteria_marine metagenome | 0 | 5.88 | 0.36 | 16.02 | 1 |
| Oxyphotobacteria_bacterium WHC4-2 | 8.15 | 2.43 | 0.34 | 5.09 | 0.96 |
| Oxyphotobacteria_Proteobacteria bacterium JGI 0000113-L05 | 7.11 | 1.97 | 0.31 | 5.64 | 0.87 |
| Bacteroidia_Bernardetia | 4.62 | 0 | 0.28 | 2.09 | 0.79 |
| Gammaproteobacteria_Aliikangiella | 4.54 | 0 | 0.27 | 41.41 | 0.77 |
| Parcubacteria_uncultured organism | 0 | 4.22 | 0.25 | 16.02 | 0.72 |
| Alphaproteobacteria_Candidatus Megaira | 7.45 | 3.25 | 0.25 | 18.95 | 0.71 |
| Alphaproteobacteria_Halocynthiibacter | 4.08 | 0 | 0.25 | 2 | 0.7 |
| Oxyphotobacteria_environmental clone OCS182 | 6.61 | 2.43 | 0.25 | 3.3 | 0.7 |
| Alphaproteobacteria_Roseobacter clade NAC11-7 lineage | 4.05 | 0 | 0.25 | 1.52 | 0.7 |
| Bacteroidia_Muriicola | 0 | 4.04 | 0.24 | 16.02 | 0.69 |
| Low and MPA (Av. Dissimilarity: 35.48%) | Low Av.Abund | MPA Av.Abund | Av.Diss | Diss/SD | Contrib% |
| Deinococci_Truepera | 5.42 | 0 | 0.31 | 460.95 | 0.87 |
| Alphaproteobacteria_Allorhizobium–Neorhizobium–Pararhizobium–Rhizobium | 5.27 | 0 | 0.3 | 4.05 | 0.85 |
| Gammaproteobacteria_uncultured | 0 | 4.72 | 0.27 | 578.22 | 0.76 |
| Gammaproteobacteria_Thiothrix | 8.88 | 4.59 | 0.25 | 0.89 | 0.69 |
| Alphaproteobacteria_Lentibacter | 4.15 | 0 | 0.24 | 1.64 | 0.67 |
| Oxyphotobacteria_Podosira stelligera | 4.06 | 0 | 0.23 | 4.5 | 0.65 |
| Gammaproteobacteria_HTCC5015 | 1.72 | 5.49 | 0.21 | 1.55 | 0.61 |
| Oxyphotobacteria_Psammodictyon panduriforme | 0 | 3.7 | 0.21 | 578.22 | 0.6 |
| Gracilibacteria_uncultured bacterium | 0 | 3.65 | 0.21 | 578.22 | 0.59 |
| Gammaproteobacteria_uncultured | 0 | 3.57 | 0.2 | 578.22 | 0.57 |
| Gammaproteobacteria_Perspicuibacter | 0 | 3.55 | 0.2 | 578.22 | 0.57 |
| Alphaproteobacteria_Aliiroseovarius | 0 | 3.52 | 0.2 | 578.22 | 0.57 |
| Oxyphotobacteria_Cylindrotheca closterium | 0 | 3.46 | 0.2 | 578.22 | 0.56 |
| Bacteroidia_Mesoflavibacter | 0 | 3.45 | 0.2 | 578.22 | 0.56 |
| Gracilibacteria_marine metagenome | 2.47 | 5.88 | 0.19 | 8.06 | 0.55 |
| Sericytochromatia_Hyaloperonospora arabidopsidis | 0.93 | 4.33 | 0.19 | 2.57 | 0.55 |
| Mid and MPA (Av. Dissimilarity: 37.24%) | Mid Av.Abund | MPA Av.Abund | Av.Diss | Diss/SD | Contrib% |
| Gammaproteobacteria_uncultured | 6.4 | 0 | 0.32 | 1.61 | 0.87 |
| Gracilibacteria_marine metagenome | 0 | 5.88 | 0.3 | 109.31 | 0.8 |
| Gammaproteobacteria_Verticia | 5.23 | 0 | 0.27 | 2.46 | 0.71 |
| Alphaproteobacteria_Marivita | 4.66 | 0 | 0.24 | 5.29 | 0.64 |
| Parcubacteria_uncultured bacterium | 1.15 | 5.57 | 0.22 | 2.66 | 0.6 |
| Parcubacteria_uncultured prokaryote | 1.17 | 5.58 | 0.22 | 2.61 | 0.6 |
| Alphaproteobacteria_Pseudorhodobacter | 4.21 | 0 | 0.21 | 7.5 | 0.57 |
| Gammaproteobacteria_Alcanivorax | 4.21 | 0 | 0.21 | 6.35 | 0.57 |
| Alphaproteobacteria_Boseongicola | 4.15 | 0 | 0.21 | 22.36 | 0.57 |
| Gammaproteobacteria_Cycloclasticus | 4.04 | 0 | 0.2 | 11.71 | 0.55 |
| Alphaproteobacteria_Albirhodobacter | 4 | 0 | 0.2 | 274.32 | 0.55 |
| Gracilibacteria_Gracilibacteria | 1.25 | 5.24 | 0.2 | 2.22 | 0.54 |
| Gammaproteobacteria_Pseudomonas | 3.85 | 0 | 0.2 | 13.15 | 0.52 |
| Gammaproteobacteria_Enterobacteriaceae | 3.8 | 0 | 0.19 | 1.9 | 0.52 |
| Alphaproteobacteria_Lentibacter | 3.78 | 0 | 0.19 | 2.13 | 0.51 |
| Bacteroidia_Aquibacter | 2.28 | 6.03 | 0.19 | 39.9 | 0.51 |
| Deltaproteobacteria_Desulfosarcina | 3.72 | 0 | 0.19 | 9.33 | 0.51 |
| (a) | Low (Av. Similarity: 33.51%) | Av.Abund | Av.Sim | Contrib% | ||
| Cocconeis costata | 3.81 | 6.41 | 19.13 | |||
| Grammatophora oceanica | 3.95 | 4.68 | 13.97 | |||
| Nitzschia aff. inconspicua | 1.6 | 2.87 | 8.56 | |||
| Amphora sp.1 | 1.37 | 2.34 | 6.99 | |||
| Grammatophora serpentina | 1.51 | 2.32 | 6.92 | |||
| Nitzschia aff. distans | 1.78 | 2.32 | 6.92 | |||
| Gomphonemopsis sp. | 1.19 | 2.03 | 6.05 | |||
| Opephora sp.1 | 2.03 | 1.66 | 4.94 | |||
| Ulnaria biceps | 0.97 | 1.66 | 4.94 | |||
| High (Av. Similarity: 59.12%) | Av.Abund | Av.Sim | Contrib% | |||
| Bacillaria socialis | 4.6 | 7.52 | 12.72 | |||
| Nitzschia aff. distans | 2.95 | 4.92 | 8.32 | |||
| Navicula aff. subagnita | 2.46 | 3.97 | 6.72 | |||
| Amphora aff. acutiuscula | 2.32 | 3.69 | 6.24 | |||
| Fragilaria aff. investiens | 2.25 | 3.55 | 6.01 | |||
| Nitzschia aff. medioconstricta | 2.15 | 3.55 | 6 | |||
| Cocconeis costata | 1.86 | 3.08 | 5.2 | |||
| Amphora sp.1 | 1.62 | 2.71 | 4.59 | |||
| Cocconeis sp. | 1.35 | 2.29 | 3.87 | |||
| Nitzschia aff. perindistincta | 1.76 | 2.29 | 3.87 | |||
| Astartiella sp.1 | 1.15 | 2.05 | 3.46 | |||
| Nitzschia aff. laevis | 2.07 | 2.05 | 3.46 | |||
| Plagiotropis sp. | 1.28 | 2.05 | 3.46 | |||
| Gomphonemopsis sp. | 1.26 | 1.78 | 3 | |||
| (b) | Low and High (Av. Dissimilarity: 68.23%) | Low Av.Abund | High Av.Abund | Av.Diss | Diss/SD | Contrib% |
| Grammatophora oceanica | 3.95 | 0 | 3.91 | 1.79 | 5.73 | |
| Bacillaria socialis | 0.7 | 4.6 | 3.82 | 2.96 | 5.6 | |
| Nitzschia aff. medioconstricta | 0 | 2.15 | 2.07 | 5.96 | 3.03 | |
| Opephora sp.1 | 2.03 | 0 | 2.04 | 1.33 | 2.99 | |
| Fragilaria aff. investiens | 2 | 2.25 | 1.94 | 3.83 | 2.84 | |
| Cocconeis costata | 3.81 | 1.86 | 1.93 | 2.01 | 2.83 | |
| Nitzschia aff. perindistincta | 0 | 1.76 | 1.71 | 2.66 | 2.51 | |
| Amphora aff. acutiuscula | 0.69 | 2.32 | 1.59 | 3.43 | 2.33 | |
| Navicula aff. subagnita | 0.99 | 2.46 | 1.47 | 1.2 | 2.16 | |
| Achnanthes aff. delicatissima | 1.62 | 0 | 1.44 | 0.86 | 2.1 | |
| Grammatophora serpentina | 1.51 | 0 | 1.43 | 4.64 | 2.09 | |
| Plagiotropis sp. | 0 | 1.28 | 1.24 | 5.08 | 1.81 | |
| Navicula sp.1 | 1.23 | 0.98 | 1.2 | 2.92 | 1.76 | |
| Nitzschia aff. distans | 1.78 | 2.95 | 1.17 | 1.41 | 1.71 | |
| Amphora aff. exilitata | 1.18 | 0.98 | 1.12 | 4.86 | 1.64 | |
| Astartiella sp.1 | 0 | 1.15 | 1.1 | 9.41 | 1.62 | |
| Nitzschia aff. laevis | 1.21 | 2.07 | 1.1 | 1.15 | 1.61 | |
| Nitzschia sp.6 | 0 | 1.11 | 1.08 | 2.66 | 1.59 | |
| Halamphora sp. | 1.02 | 0 | 1.05 | 0.86 | 1.55 | |
| Ulnaria biceps | 0.97 | 0 | 0.94 | 3.53 | 1.38 | |
| Cocconeis sp. | 0.95 | 1.35 | 0.94 | 1.74 | 1.38 | |
| Mid and High (Av. Dissimilarity: 68.85%) | Mid Av.Abund | High Av.Abund | Av.Diss | Diss/SD | Contrib% | |
| Licmophora flabellata | 3.23 | 0 | 3.14 | 13.85 | 4.56 | |
| Nitzschia aff. distans | 0 | 2.95 | 2.86 | 48.77 | 4.15 | |
| Navicula aff. subagnita | 0 | 2.46 | 2.38 | 16.15 | 3.45 | |
| Amphora aff. acutiuscula | 0 | 2.32 | 2.27 | 4.47 | 3.29 | |
| Nitzschia aff. medioconstricta | 0 | 2.15 | 2.09 | 5.74 | 3.04 | |
| Nitzschia aff. laevis | 0 | 2.07 | 2.05 | 1.46 | 2.98 | |
| Seminavis sp. | 1.9 | 0 | 1.84 | 13.85 | 2.67 | |
| Nitzschia aff. perindistincta | 0 | 1.76 | 1.73 | 2.24 | 2.51 | |
| Nitzschia sp.2 | 1.51 | 0 | 1.47 | 13.85 | 2.13 | |
| Bacillaria socialis | 6.08 | 4.6 | 1.41 | 3.37 | 2.05 | |
| Gomphonemopsis exigua | 1.4 | 0 | 1.36 | 13.85 | 1.97 | |
| Grammatophora oceanica | 1.4 | 0 | 1.36 | 13.85 | 1.97 | |
| Fragilaria sp.1 | 1.62 | 0.29 | 1.31 | 2.68 | 1.9 | |
| Nitzschia sp.1 | 1.71 | 0.41 | 1.25 | 2.68 | 1.82 | |
| Plagiotropis sp. | 0 | 1.28 | 1.25 | 4.65 | 1.81 | |
| Gomphonemopsis sp. | 0 | 1.26 | 1.21 | 4.46 | 1.76 | |
| Astartiella sp.1 | 0 | 1.15 | 1.12 | 13.63 | 1.62 | |
| Achnanthes sp.1 | 1.14 | 0 | 1.11 | 13.85 | 1.61 | |
| Lyrella abrupta | 1.14 | 0 | 1.11 | 13.85 | 1.61 | |
| Nitzschia aff. coarctata | 1.81 | 0.7 | 1.11 | 1.06 | 1.61 | |
| Nitzschia sp.6 | 0 | 1.11 | 1.09 | 2.24 | 1.59 | |
| Low and Mid (Av. Dissimilarity: 74.21%) | Low Av.Abund | Mid Av.Abund | Av.Diss | Diss/SD | Contrib% | |
| Bacillaria socialis | 0.7 | 6.08 | 5.7 | 3.36 | 7.68 | |
| Licmophora flabellata | 0.4 | 3.23 | 2.94 | 11.88 | 3.96 | |
| Grammatophora oceanica | 3.95 | 1.4 | 2.81 | 1.01 | 3.79 | |
| Fragilaria aff. investiens | 2 | 2.06 | 2.1 | 6.3 | 2.83 | |
| Nitzschia aff. coarctata | 0 | 1.81 | 1.9 | 8.58 | 2.56 | |
| Nitzschia aff. distans | 1.78 | 0 | 1.81 | 2.44 | 2.44 | |
| Nitzschia sp.1 | 0 | 1.71 | 1.8 | 8.58 | 2.42 | |
| Seminavis sp. | 0.29 | 1.9 | 1.71 | 2.76 | 2.31 | |
| Fragilaria sp.1 | 0 | 1.62 | 1.7 | 8.58 | 2.29 | |
| Nitzschia aff. inconspicua | 1.6 | 0 | 1.69 | 3.49 | 2.28 | |
| Nitzschia sp.2 | 0 | 1.51 | 1.59 | 8.58 | 2.14 | |
| Achnanthes aff. delicatissima | 1.62 | 0 | 1.56 | 0.71 | 2.1 | |
| Grammatophora serpentina | 1.51 | 0 | 1.56 | 4.08 | 2.1 | |
| Gomphonemopsis exigua | 0 | 1.4 | 1.47 | 8.58 | 1.98 | |
| Cocconeis sp. | 0.95 | 2.21 | 1.41 | 0.9 | 1.9 | |
| Navicula sp.1 | 1.23 | 0 | 1.4 | 0.71 | 1.89 | |
| Opephora sp.1 | 2.03 | 0.99 | 1.37 | 0.81 | 1.85 | |
| Gomphonemopsis sp. | 1.19 | 0 | 1.26 | 2.88 | 1.7 | |
| Odontella aurita | 0.4 | 1.62 | 1.24 | 2.79 | 1.67 | |
| Nitzschia aff. laevis | 1.21 | 0 | 1.23 | 2.69 | 1.66 | |
| MPA and High (Av. Dissimilarity: 57.14%) | MPA Av.Abund | High Av.Abund | Av.Diss | Diss/SD | Contrib% | |
| Achnanthes aff. delicatissima | 3.51 | 0 | 3.06 | 15.43 | 5.35 | |
| Parlibellus aff. coxieae | 2.81 | 0 | 2.45 | 15.43 | 4.28 | |
| Bacillaria socialis | 1.86 | 4.6 | 2.4 | 3.85 | 4.2 | |
| Nitzschia aff. laevis | 0 | 2.07 | 1.84 | 1.47 | 3.22 | |
| Seminavis sp. | 1.78 | 0 | 1.55 | 15.43 | 2.71 | |
| Achnanthes sp.1 | 1.59 | 0 | 1.38 | 15.43 | 2.42 | |
| Achnanthes aff. vistulana | 1.59 | 0 | 1.38 | 15.43 | 2.42 | |
| Plagiotropis sp. | 0 | 1.28 | 1.12 | 4.81 | 1.96 | |
| Nitzschia aff. perindistincta | 0.56 | 1.76 | 1.06 | 1.63 | 1.85 | |
| Astartiella sp.1 | 0 | 1.15 | 1 | 15.15 | 1.75 | |
| Nitzschia sp.6 | 0 | 1.11 | 0.98 | 2.28 | 1.71 | |
| Caloneis sp. | 1.12 | 0 | 0.98 | 15.43 | 1.71 | |
| Licmophora gracilis | 1.12 | 0 | 0.98 | 15.43 | 1.71 | |
| Cocconeis sp. | 2.45 | 1.35 | 0.96 | 64.73 | 1.67 | |
| Nitzschia aff. inconspicua | 2.03 | 0.98 | 0.92 | 3.45 | 1.6 | |
| Amphora aff. exilitata | 2.03 | 0.98 | 0.9 | 6.03 | 1.58 | |
| Nitzschia aff. medioconstricta | 1.12 | 2.15 | 0.9 | 3.59 | 1.57 | |
| Nitzschia sp.4 | 0 | 0.98 | 0.86 | 3.27 | 1.51 | |
| Amphora sp.3 | 0.97 | 0 | 0.85 | 15.43 | 1.48 | |
| Cocconeiopsis sp. | 0.97 | 0 | 0.85 | 15.43 | 1.48 | |
| Cyclotella aff. atomus | 0.97 | 0 | 0.85 | 15.43 | 1.48 | |
| Grammatophora oceanica | 0.97 | 0 | 0.85 | 15.43 | 1.48 | |
| Navicula aff. cincta | 0.97 | 0 | 0.85 | 15.43 | 1.48 | |
| MPA and Low (Av. Dissimilarity: 59.22%) | MPA Av.Abund | Low Av.Abund | Av.Diss | Diss/SD | Contrib% | |
| Grammatophora oceanica | 0.97 | 3.95 | 2.89 | 1.17 | 4.87 | |
| Parlibellus aff. coxieae | 2.81 | 0 | 2.62 | 9.63 | 4.42 | |
| Opephora sp.1 | 0 | 2.03 | 1.98 | 1.09 | 3.34 | |
| Fragilaria aff. investiens | 3.13 | 2 | 1.94 | 1.16 | 3.28 | |
| Achnanthes aff. delicatissima | 3.51 | 1.62 | 1.87 | 0.81 | 3.17 | |
| Cocconeis costata | 1.86 | 3.81 | 1.87 | 1.72 | 3.15 | |
| Navicula aff. subagnita | 2.69 | 0.99 | 1.66 | 1.13 | 2.8 | |
| Cocconeis sp. | 2.45 | 0.95 | 1.46 | 1.05 | 2.47 | |
| Seminavis sp. | 1.78 | 0.29 | 1.41 | 2.7 | 2.38 | |
| Grammatophora serpentina | 0 | 1.51 | 1.39 | 3.88 | 2.34 | |
| Achnanthes sp.1 | 1.59 | 0.29 | 1.24 | 2.45 | 2.09 | |
| Fragilaria pinnata | 1.26 | 0 | 1.17 | 9.63 | 1.98 | |
| Amphora aff. exilitata (pediculus) | 2.03 | 1.18 | 1.16 | 0.94 | 1.95 | |
| Navicula sp.1 | 1.49 | 1.23 | 1.13 | 5.32 | 1.91 | |
| Bacillaria socialis | 1.86 | 0.7 | 1.13 | 1.09 | 1.91 | |
| Nitzschia aff. laevis | 0 | 1.21 | 1.1 | 2.61 | 1.86 | |
| Caloneis sp. | 1.12 | 0 | 1.05 | 9.63 | 1.77 | |
| Nitzschia aff. medioconstricta | 1.12 | 0 | 1.05 | 9.63 | 1.77 | |
| Halamphora sp. | 0 | 1.02 | 1.02 | 0.71 | 1.73 | |
| Eunotogramma marinum | 0 | 1.04 | 0.95 | 4.45 | 1.61 | |
| MPA and Mid (Av. Dissimilarity: 70.47%) | MPA Av.Abund | Mid Av.Abund | Av.Diss | Diss/SD | Contrib% | |
| Bacillaria socialis | 1.86 | 6.08 | 3.97 | - | 5.64 | |
| Achnanthes aff. delicatissima | 3.51 | 0 | 3.31 | - | 4.69 | |
| Parlibellus aff. coxieae | 2.81 | 0 | 2.65 | - | 3.76 | |
| Navicula aff. subagnita | 2.69 | 0 | 2.54 | - | 3.6 | |
| Licmophora flabellata | 0.79 | 3.23 | 2.3 | - | 3.26 | |
| Nitzschia aff. distans | 2.1 | 0 | 1.98 | - | 2.81 | |
| Nitzschia aff. inconspicua | 2.03 | 0 | 1.91 | - | 2.71 | |
| Nitzschia aff. coarctata | 0 | 1.81 | 1.7 | - | 2.42 | |
| Nitzschia sp.1 | 0 | 1.71 | 1.62 | - | 2.29 | |
| Fragilaria sp.1 | 0 | 1.62 | 1.52 | - | 2.16 | |
| Odontella aurita | 0 | 1.62 | 1.52 | - | 2.16 | |
| Achnanthes aff. vistulana | 1.59 | 0 | 1.5 | - | 2.12 | |
| Nitzschia sp.2 | 0 | 1.51 | 1.43 | - | 2.02 | |
| Amphora aff. acutiuscula | 1.49 | 0 | 1.4 | - | 1.99 | |
| Navicula sp.1 | 1.49 | 0 | 1.4 | - | 1.99 | |
| Gomphonemopsis exigua | 0 | 1.4 | 1.32 | - | 1.87 | |
| Fragilaria pinnata | 1.26 | 0 | 1.18 | - | 1.68 | |
| Amphora aff. exilitata | 2.03 | 0.81 | 1.15 | - | 1.63 | |
| Lyrella abrupta | 0 | 1.14 | 1.08 | - | 1.53 |
| S | H’ | J | N | |||
|---|---|---|---|---|---|---|
| Pooled terms | Si(Pr) × Or | Si(Pr) × Or | Si(Pr) × Or | Si(Pr); Si(Pr) × Or | ||
| Transf. | None | None | None | Square root | ||
| Source | df | Pseudo-F | Pseudo-F | Pseudo-F | df | Pseudo-F |
| Pr | 3 | 2.892 | 5.982 | 6.659 * | 3 | 10.482 *** |
| Or | 1 | 0.009 | 4.270 * | 7.922 ** | 1 | 1.573 |
| Si(Pr) | 3 | 6.237 *** | 7.954 *** | 5.744 ** | 2 | 0.902 |
| Pr × Or + | 2 | 2.457 | 1.362 | 0.060 | ||
| Pooled | 58 | 61 | ||||
| Total | 67 | 67 | ||||
| PERMDISP | P(perm): 0.004 | P(perm): 0.001 | P(perm): ns. | P(perm):0.17 |
| (a) Macrofouling assemblage | (b) NIS assemblage | ||||
|---|---|---|---|---|---|
| Source | df | Pseudo-F | df | Pseudo-F | |
| Pr | 3 | 4.115 ** | Pr | 3 | 3.567 (p = 0.07) |
| Or | 1 | 3.201 *** | Or | 1 | 1.305 |
| Si(Pr) | 3 | 5.320 *** | Si(Pr) | 3 | 8.593 *** |
| Pr × Or + | 2 | 2.667 *** | PrxOr + | 2 | 0.843 |
| Pooled | 58 | Si(Pr) × Or | 3 | 2.753 * | |
| Res | 55 | ||||
| Total | 67 | Total | 67 | ||
| PERMDISP | P(perm): ns. | P(perm): 0.001 | |||
| (a) | Status | MPA (Av. Similarity: 47.47%) | Av.Abund | Av.Sim | Sim/SD | Contrib% | |
| N | Perforatus perforatus | 34.79 | 24.86 | 1.61 | 52.36 | ||
| Bare space | 25.14 | 13.72 | 1.13 | 28.9 | |||
| U | Cyanobact | 4.71 | 1.54 | 0.44 | 3.25 | ||
| N | Spirobranchus triqueter | 2.13 | 1.18 | 1.24 | 2.48 | ||
| N | Lithophyllum incrustans | 3.37 | 0.95 | 0.49 | 2.01 | ||
| NIS | Tricellaria inopinata | 1.46 | 0.95 | 1.5 | 2.01 | ||
| Low (Av. Similarity: 48.22%) | Av.Abund | Av.Sim | Sim/SD | Contrib% | |||
| Bare space | 38.92 | 29.5 | 2.06 | 60.94 | |||
| U | Cyanobacteria | 23.82 | 10.46 | 0.71 | 21.61 | ||
| N | Perforatus perforatus | 13.77 | 4.05 | 0.46 | 8.41 | ||
| N | Lithophyllum incrustans | 4.36 | 1.69 | 0.7 | 3.51 | ||
| Mid (Av. Similarity: 48.80%) | Av.Abund | Av.Sim | Sim/SD | Contrib% | |||
| C | Diplosoma cf. listerianum | 19.97 | 12.58 | 1.47 | 25.77 | ||
| Bare space | 13.74 | 7.7 | 1.32 | 15.78 | |||
| N | Ciona intestinalis | 9.3 | 5.05 | 1.08 | 10.34 | ||
| C | Botryllus schlosseri | 9.5 | 4.43 | 0.95 | 9.07 | ||
| NIS | Tricellaria inopinata | 6.22 | 4.12 | 1.76 | 8.43 | ||
| N | Perforatus perforatus | 7.33 | 3.81 | 1.15 | 7.8 | ||
| N | Spirobranchus triqueter | 3.84 | 2.38 | 1.4 | 4.87 | ||
| N | Ascidia aspersa | 7.53 | 2.36 | 0.47 | 4.83 | ||
| U | Microcosmus sp. | 2.93 | 1.4 | 1.17 | 2.88 | ||
| NIS | Watersipora subatra | 1.62 | 0.88 | 1.17 | 1.81 | ||
| High (Av. Similarity: 54.56%) | Av.Abund | Av.Sim | Sim/SD | Contrib% | |||
| Bare space | 26.72 | 20.63 | 2.23 | 37.32 | |||
| N | Spirobranchus triqueter | 18.23 | 13.23 | 2.19 | 24.25 | ||
| C | Diplosoma cf. listerianum | 12.53 | 6.15 | 0.86 | 11.28 | ||
| N | Perforatus perforatus | 10.5 | 4.78 | 0.84 | 8.77 | ||
| U | Foliculina sp. | 8.28 | 3.9 | 0.91 | 7.15 | ||
| NIS | Watersipora subatra | 6.11 | 2.19 | 0.67 | 4.02 | ||
| (b) | Status | Low: MPA and Local (Av. Dissimilarity: 53.01%) | MPA Av.Abund | Local Av.Abund | Av.Diss | Diss/SD | Contrib% |
| U | Cyanobacteria | 31.01 | 15.82 | 14.71 | 1.31 | 27.41 | |
| N | Perforatus perforatus | 7.98 | 20.2 | 10.22 | 1.02 | 19.27 | |
| Bare space | 43.03 | 34.34 | 9.97 | 1.45 | 18.81 | ||
| C | Kirchenpaueria halecioides | 4.55 | 5.95 | 4.16 | 0.64 | 7.85 | |
| N | Lithophyllum incrustans | 3.64 | 5.16 | 2.65 | 1.1 | 5 | |
| C | Diplosoma cf. listerianum | 0.1 | 2.81 | 1.45 | 0.4 | 2.73 | |
| Status | Mid: MPA and Local (Av. Dissimilarity: 53.07%) | MPA Av.Abund | Local Av.Abund | Av.Diss | Diss/SD | Contrib% | |
| C | Diplosoma cf. listerianum | 26.1 | 13.84 | 8.22 | 1.5 | 15.48 | |
| Bare space | 12.43 | 15.05 | 5.86 | 1.15 | 11.04 | ||
| N | Ascidia aspersa | 3.44 | 11.62 | 5.6 | 1.13 | 10.54 | |
| C | Botryllus schlosseri | 8.49 | 10.51 | 4.97 | 1.11 | 9.37 | |
| N | Ciona intestinalis | 6.88 | 11.72 | 4.47 | 1.5 | 8.43 | |
| N | Perforatus perforatus | 8.8 | 5.86 | 3.47 | 1.15 | 6.54 | |
| NIS | Tricellaria inopinata | 5.36 | 7.07 | 2.13 | 1.42 | 4.02 | |
| N | Spirobranchus triqueter | 5.36 | 2.32 | 1.71 | 1.5 | 3.22 | |
| U | Microcosmus sp. | 1.62 | 4.24 | 1.64 | 0.92 | 3.08 | |
| U | Foliculina sp. | 3.03 | 1.52 | 1.61 | 0.96 | 3.03 | |
| C | Kirchenpaueria halecioides | 1.52 | 2.73 | 1.28 | 0.78 | 2.42 | |
| U | Cradoscrupocellaria sp2. | 1.01 | 2.02 | 1.09 | 1.01 | 2.06 | |
| N | Celleporella hyalina | 1.92 | 0 | 0.96 | 0.87 | 1.81 | |
| Status | High: MPA and Local (Av. Dissimilarity: 47.59%) | MPA Av.Abund | Local Av.Abund | Av.Diss | Diss/SD | Contrib% | |
| Bare space | 30.81 | 22.63 | 6.65 | 1.42 | 13.98 | ||
| C | Diplosoma cf. listerianum | 8.59 | 16.46 | 6.63 | 1.38 | 13.93 | |
| N | Spirobranchus triqueter | 23.74 | 12.73 | 6.16 | 1.73 | 12.94 | |
| N | Perforatus perforatus | 11.11 | 9.9 | 5.59 | 1.22 | 11.76 | |
| U | Foliculina sp. | 4.65 | 11.92 | 4.79 | 1.27 | 10.06 | |
| NIS | Watersipora subatra | 2.63 | 9.6 | 4.26 | 1.05 | 8.95 | |
| U | Bryozoa sp.1 | 1.52 | 1.62 | 1.25 | 0.77 | 2.62 | |
| N | Cryptosula pallasiana | 1.72 | 1.11 | 1.12 | 0.67 | 2.36 | |
| U | Spirorbis sp. | 1.31 | 1.82 | 1.06 | 1.13 | 2.23 | |
| N | Celleporella hyalina | 2.12 | 0.3 | 1.06 | 0.84 | 2.23 |
| N-NIS | S-NIS | H’-NIS | ||||
|---|---|---|---|---|---|---|
| Pooled terms | Si(Pr) x Or | |||||
| Transf | Square-root | |||||
| Source | df | Source | df | Pseudo-F | Pseudo-F | |
| Pr | 3 | 1.870 | Pr | 3 | 1.645 | 2.366 |
| Or | 1 | 14.246 *** | Or | 1 | 0.031 | 0.696 |
| Si(Pr) | 3 | 27.484 *** | Si(Pr) | 3 | 13.552 *** | 7.051 *** |
| Pr × Or + | 2 | 1.718 | PrxOr + | 2 | 0.842 | 0.713 |
| Pooled | 58 | Si(Pr)xOr | 3 | 3.1524 (p = 0.05) | 2.530 (p = 0.07) | |
| Total | 67 | Res | 55 | |||
| Total | 67 | |||||
| PERMDISP | P(perm): 0.001 | P(perm): 0.006 | P(perm): 0.002 |
| Tricellaria inopinata | Watersipora subatra | ||||
|---|---|---|---|---|---|
| Pooled terms: | Si(Pr) x Or; Pr × Or | ||||
| Source | df | Pseudo-F | Source | df | Pseudo-F |
| Pr | 3 | 13.701 * | Pr | 3 | 1.345 |
| Or | 1 | 0.005 | Or | 1 | 7.561 * |
| Si(Pr) | 3 | 4.446 ** | Si(Pr) | 3 | 18.375 *** |
| Pooled | 5 | 2.235 | Pr × Or + | 2 | 3.651 |
| Res | 55 | Si(Pr) x Or | 3 | 1.607 | |
| Total | 67 | Res | 55 | ||
| Total | 67 | ||||
| PERMDISP | P(perm): 0.001 | P(perm): 0.002 | |||
| Biomass | Respiration Rate | ||
|---|---|---|---|
| Pooled terms | Si(Pr); Si(Pr) x Or | Si(Pr); Si(Pr) x Or | |
| Transf. | Fourth root | None | |
| Source | df | Pseudo-F | Pseudo-F |
| Pr | 3 | 26.520 *** | 16.233 *** |
| Or | 1 | 14.990 *** | 4.590 * |
| Pr × Or + | 2 | 2.434 | 0.853 |
| Pooled | 56 | ||
| Total | 62 | ||
| PERMDISP | P(perm): ns | P(perm): 0.026 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cacabelos, E.; Ramalhosa, P.; Canning-Clode, J.; Troncoso, J.S.; Olabarria, C.; Delgado, C.; Dobretsov, S.; Gestoso, I. The Role of Biofilms Developed under Different Anthropogenic Pressure on Recruitment of Macro-Invertebrates. Int. J. Mol. Sci. 2020, 21, 2030. https://doi.org/10.3390/ijms21062030
Cacabelos E, Ramalhosa P, Canning-Clode J, Troncoso JS, Olabarria C, Delgado C, Dobretsov S, Gestoso I. The Role of Biofilms Developed under Different Anthropogenic Pressure on Recruitment of Macro-Invertebrates. International Journal of Molecular Sciences. 2020; 21(6):2030. https://doi.org/10.3390/ijms21062030
Chicago/Turabian StyleCacabelos, Eva, Patrício Ramalhosa, João Canning-Clode, Jesús S. Troncoso, Celia Olabarria, Cristina Delgado, Sergey Dobretsov, and Ignacio Gestoso. 2020. "The Role of Biofilms Developed under Different Anthropogenic Pressure on Recruitment of Macro-Invertebrates" International Journal of Molecular Sciences 21, no. 6: 2030. https://doi.org/10.3390/ijms21062030
APA StyleCacabelos, E., Ramalhosa, P., Canning-Clode, J., Troncoso, J. S., Olabarria, C., Delgado, C., Dobretsov, S., & Gestoso, I. (2020). The Role of Biofilms Developed under Different Anthropogenic Pressure on Recruitment of Macro-Invertebrates. International Journal of Molecular Sciences, 21(6), 2030. https://doi.org/10.3390/ijms21062030







