Restoration of Neurological Function Following Peripheral Nerve Trauma
Abstract
1. Introduction
2. Issues Influencing the Extent of Axon Regeneration and Neurological Recovery
2.1. Type of Nerve Trauma
2.2. Gender and Age
2.3. Promoting Axon Regeneration Through Crushed Nerves
2.4. Restoration of Function without Surgical Intervention
2.5. Restoration of Function with Surgical Intervention—Anastomosis
2.6. Promoting Axon Regeneration Across Nerve Gaps
2.7. Autografts
3. Sensory Nerve Grafts: Limitations
3.1. Loss of Sensory Nerve Function
3.2. Incorrect Schwann Cell Phenotype
3.3. Inflammation
3.4. Necrosis
3.5. Decreasing Recovery with Increasing Gap Length
3.6. Decreasing Recovery with Increasing Time between Nerve Injury and Repair
3.7. Schwann Cell Senescence
3.8. Neuron Loss of Ability to Regenerate
3.9. Decreasing Recovery with Patient Age
3.10. Enhancing the Efficacy of Autografts to Promote Axon Regeneration
4. Electrical Stimulation
4.1. Promoting Axotomized Neurons to Extend Axons
4.2. Activating Senescent Schwann Cells
4.3. Vascularized Nerve Grafts and Promoting Vascularization
4.4. Conclusion about Sensory Nerve Grafts
5. Allografts
Enhancing the Regeneration-Promoting Capacity of Allografts
6. Nerve Conduits
6.1. Conduit Composition
6.2. Conduit Architecture
6.3. Conduits Containing Neurotrophic and Other Factors
6.4. Conduits Containing Cells
6.5. Conduit Composition and Electrical Stimulation
6.6. Conclusion for Conduits
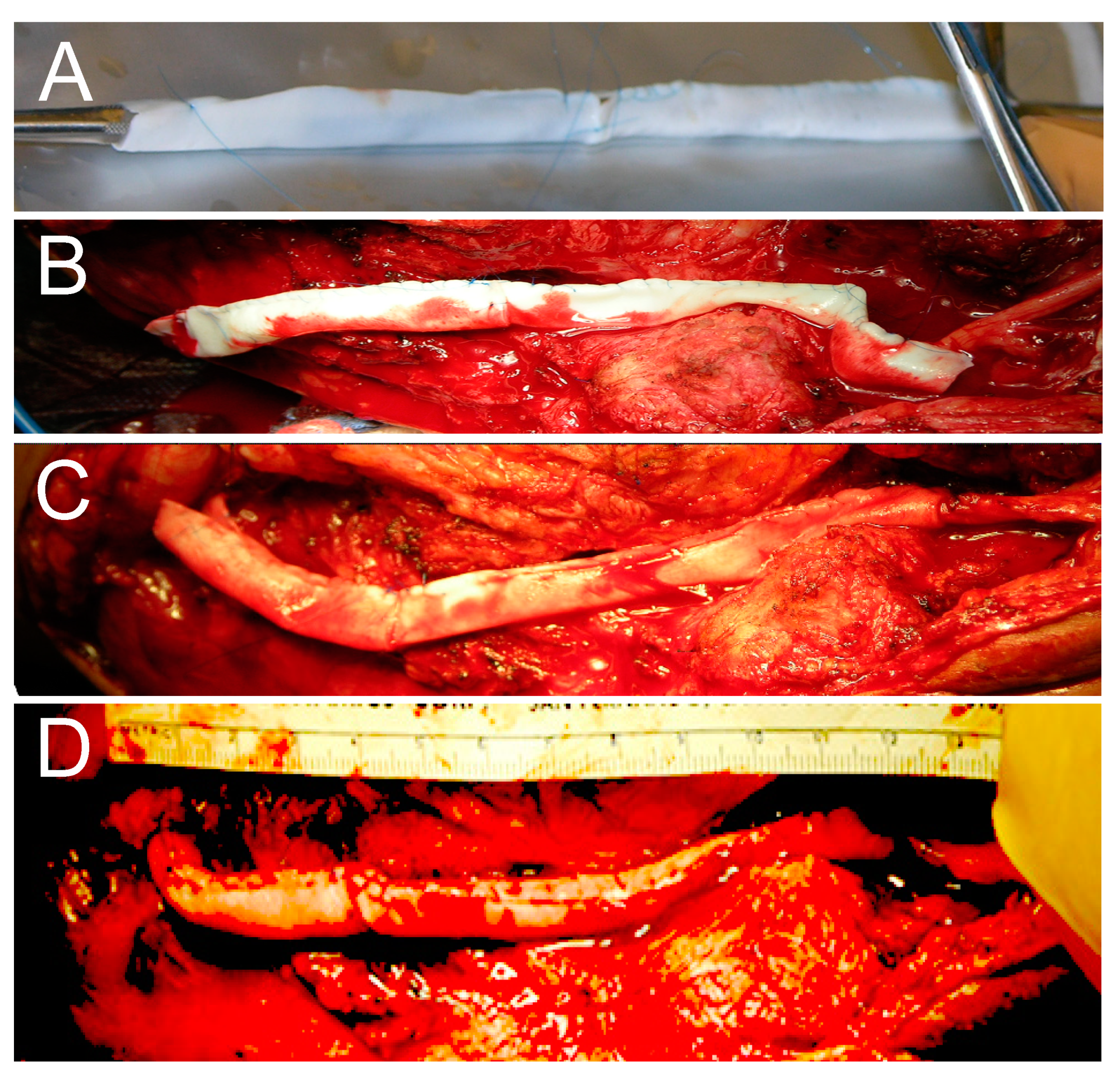
7. Novel Technique
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ADSC | adipose-derived mesenchymal stem cells |
| bFGF | basic fibroblast growth factor |
| BDNF | brain-derived neurotrophic factor |
| CNTF | ciliary neurotrophic factor |
| DMSC | bone marrow-derived mesenchymal stem cells |
| FDA | Food and Drug Administration |
| FGF | fibroblast growth factor |
| NT-3 | neurotrophin-3 |
| NGF | nerve growth factor |
| GDNF | glial-derived neurotrophic factor |
| PDGF | platelet-derived neurotrophic factor |
| PRP | platelet-rich plasma |
| TGF-β | tumor growth factor-beta |
| VEGF | vascular endothelial growth factor |
References
- Wenzinger, E.; Rivera-Barrios, A.; Gonzalez, G.; Herrera, F. Trends in Upper Extremity Injuries Presenting to US Emergency Departments. Hand 2019, 14, 408–412. [Google Scholar] [CrossRef] [PubMed]
- Huckhagel, T.; Nuchtern, J.; Regelsberger, J.; Lefering, R.; TraumaRegister, D.G.U. Nerve injury in severe trauma with upper extremity involvement: Evaluation of 49,382 patients from the TraumaRegister DGU(R) between 2002 and 2015. Scand. J. Trauma Resusc. Emerg. Med. 2018, 26, 76. [Google Scholar] [CrossRef] [PubMed]
- Domeshek, L.F.; Krauss, E.M.; Snyder-Warwick, A.K.; Laurido-Soto, O.; Hasak, J.M.; Skolnick, G.B.; Novak, C.B.; Moore, A.M.; Mackinnon, S.E. Surgical Treatment of Neuromas Improves Patient-Reported Pain, Depression, and Quality of Life. Plast. Reconstr. Surg. 2017, 139, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Holmes, S.A.; Barakat, N.; Bhasin, M.; Lopez, N.I.; Lebel, A.; Zurakowski, D.; Thomas, B.; Bhasin, S.; Silva, K.E.; Borra, R.; et al. Biological and behavioral markers of pain following nerve injury in humans. Neurobiol. Pain 2020, 7, 100038. [Google Scholar] [CrossRef]
- Bekelis, K.; Missios, S.; Spinner, R.J. Falls and peripheral nerve injuries: An age-dependent relationship. J. Neurosurg. 2015, 123, 1223–1229. [Google Scholar] [CrossRef]
- Bozkurt, A.; Lassner, F.; O’Dey, D.; Deumens, R.; Bocker, A.; Schwendt, T.; Janzen, C.; Suschek, C.V.; Tolba, R.; Kobayashi, E.; et al. The role of microstructured and interconnected pore channels in a collagen-based nerve guide on axonal regeneration in peripheral nerves. Biomaterials 2012, 33, 1363–1375. [Google Scholar] [CrossRef]
- Shergill, G.; Bonney, G.; Munshi, P.; Birch, R. The radial and posterior interosseous nerves. Results fo 260 repairs. J. Bone Joint Surg. Br. 2001, 83, 646–649. [Google Scholar] [CrossRef]
- Goransson, L.G.; Mellgren, S.I.; Lindal, S.; Omdal, R. The effect of age and gender on epidermal nerve fiber density. Neurology 2004, 62, 774–777. [Google Scholar] [CrossRef]
- Jones, K.J. Recovery from facial paralysis following crush injury of the facial nerve in hamsters: Differential effects of gender and androgen exposure. Exp. Neurol. 1993, 121, 133–138. [Google Scholar] [CrossRef]
- English, A.W.; Wilhelm, J.C.; Ward, P.J. Exercise, neurotrophins, and axon regeneration in the PNS. Physiology (Bethesda) 2014, 29, 437–445. [Google Scholar] [CrossRef]
- Sharma, N.; Marzo, S.J.; Jones, K.J.; Foecking, E.M. Electrical stimulation and testosterone differentially enhance expression of regeneration-associated genes. Exp. Neurol. 2010, 223, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Wood, K.; Wilhelm, J.C.; Sabatier, M.J.; Liu, K.; Gu, J.; English, A.W. Sex differences in the effectiveness of treadmill training in enhancing axon regeneration in injured peripheral nerves. Dev. Neurobiol. 2012, 72, 688–698. [Google Scholar] [CrossRef] [PubMed]
- Osborne, M.C.; Verhovshek, T.; Sengelaub, D.R. Androgen regulates trkB immunolabeling in spinal motoneurons. J. Neurosci. Res. 2007, 85, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Verhovshek, T.; Cai, Y.; Osborne, M.C.; Sengelaub, D.R. Androgen regulates brain-derived neurotrophic factor in spinal motoneurons and their target musculature. Endocrinology 2010, 151, 253–261. [Google Scholar] [CrossRef][Green Version]
- Alsmadi, N.Z.; Bendale, G.S.; Kanneganti, A.; Shihabeddin, T.; Nguyen, A.H.; Hor, E.; Dash, S.; Johnston, B.; Granja-Vazquez, R.; Romero-Ortega, M.I. Glial-derived growth factor and pleiotrophin synergistically promote axonal regeneration in critical nerve injuries. Acta Biomater. 2018, 78, 165–177. [Google Scholar] [CrossRef]
- Brushart, T.M.; Gerber, J.; Kessens, P.; Chen, Y.G.; Royall, R.M. Contributions of pathway and neuron to preferential motor reinnervation. J. Neurosci. 1998, 18, 8674–8681. [Google Scholar] [CrossRef]
- Chen, Y.G.; Brushart, T.M. The effect of denervated muscle and Schwann cells on axon collateral sprouting. J. Hand Surg. 1998, 23, 1025–1033. [Google Scholar] [CrossRef]
- Evans, G.R.; Brandt, K.; Ang, K.K.; Cromeens, D.; Peden, E.; Gherardini, G.; Gurlek, A.; Tinkey, P.; Williams, J. Peripheral nerve regeneration: The effects of postoperative irradiation. Plast. Reconstr. Surg. 1997, 100, 375–380. [Google Scholar] [CrossRef]
- Kuffler, D.P. Accurate reinnervation of motor end plates after disruption of sheath cells and muscle fibers. J. Comp. Neurol. 1986, 250, 228–235. [Google Scholar] [CrossRef]
- Kuffler, D.P. Isolated satellite cells of a peripheral nerve direct the growth of regenerating frog axons. J. Comp. Neurol. 1986, 249, 57–64. [Google Scholar] [CrossRef]
- Madison, R.D.; Archibald, S.J.; Lacin, R.; Krarup, C. Factors contributing to preferential motor reinnervation in the primate peripheral nervous system. J. Neurosci. 1999, 19, 11007–11016. [Google Scholar] [CrossRef] [PubMed]
- Dagum, A.B. Peripheral nerve regeneration, repair, and grafting. J. Hand Ther. 1998, 11, 111–117. [Google Scholar] [CrossRef]
- Wolberg, A.S. Thrombin generation and fibrin clot structure. Blood Rev. 2007, 21, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.D.; MacEwan, M.R.; French, A.R.; Moore, A.M.; Hunter, D.A.; Mackinnon, S.E.; Moran, D.W.; Borschel, G.H.; Sakiyama-Elbert, S.E. Fibrin matrices with affinity-based delivery systems and neurotrophic factors promote functional nerve regeneration. Biotechnol. Bioeng. 2010, 106, 970–979. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.D.; Moore, A.M.; Hunter, D.A.; Tuffaha, S.; Borschel, G.H.; Mackinnon, S.E.; Sakiyama-Elbert, S.E. Affinity-based release of glial-derived neurotrophic factor from fibrin matrices enhances sciatic nerve regeneration. Acta Biomater. 2009, 5, 959–968. [Google Scholar] [CrossRef] [PubMed]
- Schira, J.; Heinen, A.; Poschmann, G.; Ziegler, B.; Hartung, H.P.; Stuhler, K.; Kury, P. Secretome analysis of nerve repair mediating Schwann cells reveals Smad-dependent trophism. FASEB J. 2019, 33, 4703–4715. [Google Scholar] [CrossRef]
- Qin, J.; Wang, L.; Sun, Y.; Sun, X.; Wen, C.; Shahmoradi, M.; Zhou, Y. Concentrated growth factor increases Schwann cell proliferation and neurotrophic factor secretion and promotes functional nerve recovery in vivo. Int. J. Mol. Med. 2016, 37, 493–500. [Google Scholar] [CrossRef]
- Sun, X.; Zhu, Y.; Yin, H.Y.; Guo, Z.Y.; Xu, F.; Xiao, B.; Jiang, W.L.; Guo, W.M.; Meng, H.Y.; Lu, S.B.; et al. Differentiation of adipose-derived stem cells into Schwann cell-like cells through intermittent induction: Potential advantage of cellular transient memory function. Stem Cell Res. Ther. 2018, 9, 133. [Google Scholar] [CrossRef]
- Kot, M.; Musial-Wysocka, A.; Lasota, M.; Ulman, A.; Majka, M. Secretion, migration and adhesion as key processes in the therapeutic activity of mesenchymal stem cells. Acta Biochim. Pol. 2019, 66, 499–507. [Google Scholar] [CrossRef]
- Mai, J.; Fok, L.; Gao, H.; Zhang, X.; Poo, M.M. Axon initiation and growth cone turning on bound protein gradients. J. Neurosci. 2009, 29, 7450–7458. [Google Scholar] [CrossRef]
- Goodhill, G.J. Diffusion in axon guidance. Eur. J. Neurosci. 1997, 9, 1414–1421. [Google Scholar] [CrossRef] [PubMed]
- Perez, N.L.; Sosa, M.A.; Kuffler, D.P. Growth cones turn up concentration gradients of diffusible peripheral target-derived factors. Exp. Neurol. 1997, 145, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Li, Y.; Zhang, Z.; Cui, K.; Wang, S.; Yuan, X.B.; Wu, C.P.; Poo, M.M.; Duan, S. Nerve growth cone guidance mediated by G protein-coupled receptors. Nat. Neurosci. 2002, 5, 843–848. [Google Scholar] [CrossRef] [PubMed]
- De La Torre, J.R.; Hopker, V.H.; Ming, G.L.; Poo, M.M.; Tessier-Lavigne, M.; Hemmati-Brivanlou, A.; Holt, C.E. Turning of retinal growth cones in a netrin-1 gradient mediated by the netrin receptor DCC. Neuron 1997, 19, 1211–1224. [Google Scholar] [CrossRef]
- Zheng, J.Q.; Wan, J.J.; Poo, M.M. Essential role of filopodia in chemotropic turning of nerve growth cone induced by a glutamate gradient. J. Neurosci. 1996, 16, 1140–1149. [Google Scholar] [CrossRef] [PubMed]
- Houschyar, K.S.; Momeni, A.; Pyles, M.N.; Cha, J.Y.; Maan, Z.N.; Duscher, D.; Jew, O.S.; Siemers, F.; Van Schoonhoven, J. The Role of Current Techniques and Concepts in Peripheral Nerve Repair. Plast. Surg. Int. 2016, 2016, 4175293. [Google Scholar] [CrossRef]
- Horteur, C.; Forli, A.; Corcella, D.; Pailhe, R.; Lateur, G.; Saragaglia, D. Short- and long-term results of common peroneal nerve injuries treated by neurolysis, direct suture or nerve graft. Eur. J. Orthop. Surg. Traumatol. 2019, 29, 893–898. [Google Scholar] [CrossRef]
- Ronchi, G.; Cillino, M.; Gambarotta, G.; Fornasari, B.E.; Raimondo, S.; Pugliese, P.; Tos, P.; Cordova, A.; Moschella, F.; Geuna, S. Irreversible changes occurring in long-term denervated Schwann cells affect delayed nerve repair. J. Neurosurg. 2017, 127, 843–856. [Google Scholar] [CrossRef]
- Salomon, D.; Miloro, M.; Kolokythas, A. Outcomes of Immediate Allograft Reconstruction of Long-Span Defects of the Inferior Alveolar Nerve. J. Oral Maxillofac. Surg. 2016, 74, 2507–2514. [Google Scholar] [CrossRef]
- Smith, B.W.; Sakamuri, S.; Spain, D.A.; Joseph, J.R.; Yang, L.J.; Wilson, T.J. An update on the management of adult traumatic nerve injuries-replacing old paradigms: A review. J. Trauma Acute Care Surg. 2019, 86, 299–306. [Google Scholar] [CrossRef]
- Millesi, H. Techniques for nerve grafting. Hand Clin. 2000, 16, 73–91. [Google Scholar] [PubMed]
- Berger, A.; Millesi, H. Nerve grafting. Clin. Orthop. 1978, 133, 49–55. [Google Scholar] [CrossRef]
- Jenq, C.B.; Coggeshall, R.E. The effects of an autologous transplant on patterns of regeneration in rat sciatic nerve. Brain Res. 1986, 364, 45–56. [Google Scholar] [CrossRef]
- Mackinnon, S.E.; Dellon, A.L. A comparison of nerve regeneration across a sural nerve graft and a vascularized pseudosheath. J. Hand Surg. 1988, 13, 935–942. [Google Scholar] [CrossRef]
- Millesi, H. Peripheral nerve injuries. Nerve sutures and nerve grafting. Scand. J. Plast Reconstr. Surg. Suppl. 1982, 19, 25–37. [Google Scholar]
- Pereira Lopes, F.R.; Martin, P.K.; Frattini, F.; Biancalana, A.; Almeida, F.M.; Tomaz, M.A.; Melo, P.A.; Borojevic, R.; Han, S.W.; Martinez, A.M. Double gene therapy with granulocyte colony-stimulating factor and vascular endothelial growth factor acts synergistically to improve nerve regeneration and functional outcome after sciatic nerve injury in mice. Neuroscience 2013, 230, 184–197. [Google Scholar] [CrossRef]
- Reid, A.J.; De Luca, A.C.; Faroni, A.; Downes, S.; Sun, M.; Terenghi, G.; Kingham, P.J. Long term peripheral nerve regeneration using a novel PCL nerve conduit. Neurosci. Lett. 2013, 544, 125–130. [Google Scholar] [CrossRef]
- Hoke, A.; Redett, R.; Hameed, H.; Jari, R.; Zhou, C.; Li, Z.B.; Griffin, J.W.; Brushart, T.M. Schwann cells express motor and sensory phenotypes that regulate axon regeneration. J. Neurosci. 2006, 26, 9646–9655. [Google Scholar] [CrossRef]
- Forman, D.S.; Wood, D.K.; DeSilva, S. Rate of regeneration of sensory axons in transected rat sciatic nerve repaired with epineurial sutures. J. Neurol. Sci. 1979, 44, 55–59. [Google Scholar] [CrossRef]
- Bamba, R.; Riley, D.C.; Kelm, N.D.; Cardwell, N.; Pollins, A.C.; Afshari, A.; Nguyen, L.; Dortch, R.D.; Thayer, W.P. A novel conduit-based coaptation device for primary nerve repair. Int. J. Neurosci. 2018, 128, 563–569. [Google Scholar] [CrossRef]
- Pettersson, J.; Kalbermatten, D.; McGrath, A.; Novikova, L.N. Biodegradable fibrin conduit promotes long-term regeneration after peripheral nerve injury in adult rats. J. Plast. Reconstr. Aesthet. Surg. 2010, 63, 1893–1899. [Google Scholar] [CrossRef] [PubMed]
- Felix, S.P.; Pereira Lopes, F.R.; Marques, S.A.; Martinez, A.M. Comparison between suture and fibrin glue on repair by direct coaptation or tubulization of injured mouse sciatic nerve. Microsurgery 2013, 33, 468–477. [Google Scholar] [CrossRef]
- Penkert, G.; Bini, W.; Samii, M. Revascularization of nerve grafts: An experimental study. J. Reconstr. Microsurg. 1988, 4, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.I. Nerve grafting with simultaneous microvascular reconstruction. Clin. Orthop. Relat. Res. 1978, 133, 56–70. [Google Scholar] [CrossRef]
- Leckenby, J.I.; Furrer, C.; Haug, L.; Juon Personeni, B.; Vogelin, E. A retrospective case series reporting the outcomes of Avance nerve allografts in the treatment of peripheral nerve injuries. Plast. Reconstr. Surg. 2019, 45, 368e–381e. [Google Scholar] [CrossRef] [PubMed]
- Rbia, N.; Shin, A.Y. The Role of Nerve Graft Substitutes in Motor and Mixed Motor/Sensory Peripheral Nerve Injuries. J. Hand Surg. Am. 2017, 42, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Belkas, J.S.; Munro, C.A.; Shoichet, M.S.; Midha, R. Peripheral nerve regeneration through a synthetic hydrogel nerve tube. Restor. Neurol. Neurosci. 2005, 23, 19–29. [Google Scholar]
- Karabeg, R.; Jakirlic, M.; Dujso, V. Sensory recovery after forearm median and ulnar nerve grafting. Med. Arh. 2009, 63, 97–99. [Google Scholar]
- Liu, Q.; Bhat, M.; Bowen, W.D.; Cheng, J. Signaling pathways from cannabinoid receptor-1 activation to inhibition of N-methyl-D-aspartic acid mediated calcium influx and neurotoxicity in dorsal root ganglion neurons. J. Pharmacol. Exp. Ther. 2009, 331, 1062–1070. [Google Scholar] [CrossRef]
- Nichols, C.M.; Brenner, M.J.; Fox, I.K.; Tung, T.H.; Hunter, D.A.; Rickman, S.R.; Mackinnon, S.E. Effects of motor versus sensory nerve grafts on peripheral nerve regeneration. Exp. Neurol. 2004, 190, 347–355. [Google Scholar] [CrossRef]
- Strauch, R.J.; Strauch, B. Nerve conduits: An update on tubular nerve repair and reconstruction. J. Hand Surg. Am. 2013, 38, 1252–1255. [Google Scholar] [CrossRef] [PubMed]
- Matejcik, V. [Reconstructive surgery of the peripheral nerves in the upper extremities with autografts]. Acta Chir. Orthop. Traumatol. Cech. 2002, 69, 85–87. [Google Scholar] [PubMed]
- Terzis, J.K.; Kokkalis, Z.T. Outcomes of secondary reconstruction of ulnar nerve lesions: Our experience. Plast. Reconstr. Surg. 2008, 122, 1100–1110. [Google Scholar] [CrossRef]
- Aszmann, O.C.; Korak, K.J.; Luegmair, M.; Frey, M. Bridging critical nerve defects through an acellular homograft seeded with autologous schwann cells obtained from a regeneration neuroma of the proximal stump. J. Reconstr. Microsurg. 2008, 24, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Ohnishi, K.; Kiyotani, T.; Sekine, T.; Ueda, H.; Nakamura, T.; Endo, K.; Shimizu, Y. Peripheral nerve regeneration across an 80-mm gap bridged by a polyglycolic acid (PGA)-collagen tube filled with laminin-coated collagen fibers: A histological and electrophysiological evaluation of regenerated nerves. Brain Res. 2000, 868, 315–328. [Google Scholar] [CrossRef]
- Kandenwein, J.A.; Kretschmer, T.; Engelhardt, M.; Richter, H.P.; Antoniadis, G. Surgical interventions for traumatic lesions of the brachial plexus: A retrospective study of 134 cases. J. Neurosurg. 2005, 103, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Nichterwitz, S.; Hoffmann, N.; Hajosch, R.; Oberhoffner, S.; Schlosshauer, B. Bioengineered glial strands for nerve regeneration. Neurosci. Lett. 2010, 484, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Safa, B.; Buncke, G. Autograft Substitutes: Conduits and Processed Nerve Allografts. Hand Clin. 2016, 32, 127–140. [Google Scholar] [CrossRef]
- Hoben, G.M.; Ee, X.; Schellhardt, L.; Yan, Y.; Hunter, D.A.; Moore, A.M.; Snyder-Warwick, A.K.; Stewart, S.; Mackinnon, S.E.; Wood, M.D. Increasing Nerve Autograft Length Increases Senescence and Reduces Regeneration. Plast. Reconstr. Surg. 2018, 142, 952–961. [Google Scholar] [CrossRef]
- Kornfeld, T.; Vogt, P.M.; Radtke, C. Nerve grafting for peripheral nerve injuries with extended defect sizes. Wien. Med. Wochenschr. 2019, 169, 240–251. [Google Scholar] [CrossRef]
- Mani, G.V.; Shurey, C.; Green, C.J. Is early vascularization of nerve grafts necessary? J. Hand Surg. Br. 1992, 17, 536–543. [Google Scholar] [CrossRef]
- Koshima, I.; Harii, K. Experimental study of vascularized nerve grafts: Multifactorial analyses of axonal regeneration of nerves transplanted into an acute burn wound. J. Hand Surg. Am. 1985, 10, 64–72. [Google Scholar] [CrossRef]
- Roganovic, Z.; Pavlicevic, G. Difference in recovery potential of peripheral nerves after graft repairs. Neurosurgery 2006, 59, 621–633, discussion 621–633. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Kam, A.C.; Chandika, P.; Tiel, R.L.; Kline, D.G. Surgical management and outcome in patients with radial nerve lesions. J. Neurosurg. 2001, 95, 573–583. [Google Scholar] [CrossRef]
- Fu, S.Y.; Gordon, T. Contributing factors to poor functional recovery after delayed nerve repair: Prolonged denervation. J. Neurosci. 1995, 15, 3886–3895. [Google Scholar] [CrossRef]
- Matejcik, V. Peripheral nerve reconstruction by autograft. Injury 2002, 33, 627–631. [Google Scholar] [CrossRef]
- Brenner, M.J.; Hess, J.R.; Myckatyn, T.M.; Hayashi, A.; Hunter, D.A.; Mackinnon, S.E. Repair of motor nerve gaps with sensory nerve inhibits regeneration in rats. Laryngoscope 2006, 116, 1685–1692. [Google Scholar] [CrossRef]
- Jones, R.H. Repair of the trigeminal nerve: A review. Aust. Dent. J. 2010, 55, 112–119. [Google Scholar] [CrossRef]
- Tung, T.H.; Mackinnon, S.E. Nerve transfers: Indications, techniques, and outcomes. J. Hand Surg. Am. 2010, 35, 332–341. [Google Scholar] [CrossRef]
- Gordon, T.; Tyreman, N.; Raji, M.A. The basis for diminished functional recovery after delayed peripheral nerve repair. J. Neurosci. 2011, 31, 5325–5334. [Google Scholar] [CrossRef]
- Saheb-Al-Zamani, M.; Yan, Y.; Farber, S.J.; Hunter, D.A.; Newton, P.; Wood, M.D.; Stewart, S.A.; Johnson, P.J.; Mackinnon, S.E. Limited regeneration in long acellular nerve allografts is associated with increased Schwann cell senescence. Exp. Neurol. 2013, 247, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Furey, M.J.; Midha, R.; Xu, Q.G.; Belkas, J.; Gordon, T. Prolonged target deprivation reduces the capacity of injured motoneurons to regenerate. Neurosurgery 2007, 60, 723–732. [Google Scholar] [CrossRef] [PubMed]
- Fricker, F.R.; Lago, N.; Balarajah, S.; Tsantoulas, C.; Tanna, S.; Zhu, N.; Fageiry, S.K.; Jenkins, M.; Garratt, A.N.; Birchmeier, C.; et al. Axonally derived neuregulin-1 is required for remyelination and regeneration after nerve injury in adulthood. J. Neurosci. 2011, 31, 3225–3233. [Google Scholar] [CrossRef] [PubMed]
- Taveggia, C.; Zanazzi, G.; Petrylak, A.; Yano, H.; Rosenbluth, J.; Einheber, S.; Xu, X.; Esper, R.M.; Loeb, J.A.; Shrager, P.; et al. Neuregulin-1 type III determines the ensheathment fate of axons. Neuron 2005, 47, 681–694. [Google Scholar] [CrossRef]
- Lanzetta, M.; Pozzo, M.; Bottin, A.; Merletti, R.; Farina, D. Reinnervation of motor units in intrinsic muscles of a transplanted hand. Neurosci. Lett. 2005, 373, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Vikstrom, P.; Rosen, B.; Carlsson, I.K.; Bjorkman, A. The effect of early relearning on sensory recovery 4 to 9 years after nerve repair: A report of a randomized controlled study. J. Hand Surg. Eur. Vol. 2018, 43, 626–630. [Google Scholar] [CrossRef]
- Andelkovic, S.Z.; Lesic, A.R.; Bumbasirevic, M.Z.; Rasulic, L.G. The Outcomes of 150 Consecutive Patients with Digital Nerve Injuries Treated in a Single Center. Turk. Neurosurg. 2017, 27, 289–293. [Google Scholar] [CrossRef][Green Version]
- Sondell, M.; Lundborg, G.; Kanje, M. Vascular endothelial growth factor has neurotrophic activity and stimulates axonal outgrowth, enhancing cell survival and Schwann cell proliferation in the peripheral nervous system. J. Neurosci. 1999, 19, 5731–5740. [Google Scholar] [CrossRef]
- Gunin, A.G.; Petrov, V.V.; Golubtzova, N.N.; Vasilieva, O.V.; Kornilova, N.K. Age-related changes in angiogenesis in human dermis. Exp. Gerontol. 2014, 55, 143–151. [Google Scholar] [CrossRef]
- Rivard, A.; Fabre, J.E.; Silver, M.; Chen, D.; Murohara, T.; Kearney, M.; Magner, M.; Asahara, T.; Isner, J.M. Age-dependent impairment of angiogenesis. Circulation 1999, 99, 111–120. [Google Scholar] [CrossRef]
- Swift, M.E.; Kleinman, H.K.; DiPietro, L.A. Impaired wound repair and delayed angiogenesis in aged mice. Lab. Investig. 1999, 79, 1479–1487. [Google Scholar] [PubMed]
- Iijima, Y.; Ajiki, T.; Murayama, A.; Takeshita, K. Effect of Artificial Nerve Conduit Vascularization on Peripheral Nerve in a Necrotic Bed. Plast. Reconstr. Surg. Glob. Open. 2016, 4, e665. [Google Scholar] [CrossRef] [PubMed]
- Gordon, T.; Brushart, T.M.; Amirjani, N.; Chan, K.M. The potential of electrical stimulation to promote functional recovery after peripheral nerve injury--comparisons between rats and humans. Acta Neurochir. Suppl. 2007, 100, 3–11. [Google Scholar] [PubMed]
- Gordon, T.; Brushart, T.M.; Chan, K.M. Augmenting nerve regeneration with electrical stimulation. Neurol. Res. 2008, 30, 1012–1022. [Google Scholar] [CrossRef] [PubMed]
- Gordon, T.; Chan, K.M.; Sulaiman, O.A.; Udina, E.; Amirjani, N.; Brushart, T.M. Accelerating axon growth to overcome limitations in functional recovery after peripheral nerve injury. Neurosurgery 2009, 65, A132–A144. [Google Scholar] [CrossRef]
- Al-Majed, A.A.; Brushart, T.M.; Gordon, T. Electrical stimulation accelerates and increases expression of BDNF and trkB mRNA in regenerating rat femoral motoneurons. Eur. J. Neurosci. 2000, 12, 4381–4390. [Google Scholar] [PubMed]
- Al-Majed, A.A.; Tam, S.L.; Gordon, T. Electrical stimulation accelerates and enhances expression of regeneration-associated genes in regenerating rat femoral motoneurons. Cell Mol. Neurobiol. 2004, 24, 379–402. [Google Scholar] [CrossRef]
- Geremia, N.M.; Gordon, T.; Brushart, T.M.; Al-Majed, A.A.; Verge, V.M. Electrical stimulation promotes sensory neuron regeneration and growth-associated gene expression. Exp. Neurol. 2007, 205, 347–359. [Google Scholar] [CrossRef]
- Gordon, T. The role of neurotrophic factors in nerve regeneration. Neurosurg. Focus 2009, 26, E3. [Google Scholar] [CrossRef]
- Ahlborn, P.; Schachner, M.; Irintchev, A. One hour electrical stimulation accelerates functional recovery after femoral nerve repair. Exp. Neurol. 2007, 208, 137–144. [Google Scholar] [CrossRef]
- Gordon, T.; Amirjani, N.; Edwards, D.C.; Chan, K.M. Brief post-surgical electrical stimulation accelerates axon regeneration and muscle reinnervation without affecting the functional measures in carpal tunnel syndrome patients. Exp. Neurol. 2010, 223, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Gordon, T.; Udina, E.; Verge, V.M.; De Chaves, E.I. Brief electrical stimulation accelerates axon regeneration in the peripheral nervous system and promotes sensory axon regeneration in the central nervous system. Mot. Control. 2009, 13, 412–441. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.T.; Peng, C.W.; Chen, L.T.; Lin, W.S.; Chu, C.H.; Chen, J.J. Application of implantable wireless biomicrosystem for monitoring nerve impedance of rat after sciatic nerve injury. IEEE Trans. Neural Syst. Rehabil. Eng. 2013, 21, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Koppes, A.N.; Seggio, A.M.; Thompson, D.M. Neurite outgrowth is significantly increased by the simultaneous presentation of Schwann cells and moderate exogenous electric fields. J. Neural Eng. 2011, 8, 046023. [Google Scholar] [CrossRef]
- Singh, B.; Xu, Q.G.; Franz, C.K.; Zhang, R.; Dalton, C.; Gordon, T.; Verge, V.M.; Midha, R.; Zochodne, D.W. Accelerated axon outgrowth, guidance, and target reinnervation across nerve transection gaps following a brief electrical stimulation paradigm. J. Neurosurg. 2012, 116, 498–512. [Google Scholar] [CrossRef]
- Franz, C.K.; Singh, B.; Martinez, J.A.; Zochodne, D.W.; Midha, R. Brief transvertebral electrical stimulation of the spinal cord improves the specificity of femoral nerve reinnervation. Neurorehabilit. Neural Repair 2013, 27, 260–268. [Google Scholar] [CrossRef]
- Huang, J.; Zhang, Y.; Lu, L.; Hu, X.; Luo, Z. Electrical stimulation accelerates nerve regeneration and functional recovery in delayed peripheral nerve injury in rats. Eur. J. Neurosci. 2013, 38, 3691–3701. [Google Scholar] [CrossRef]
- Jo, S.; Pan, D.; Halevi, A.E.; Roh, J.; Schellhardt, L.; Hunter Ra, D.A.; Snyder-Warwick, A.K.; Moore, A.M.; Mackinnon, S.E.; Wood, M.D. Comparing electrical stimulation and tacrolimus (FK506) to enhance treating nerve injuries. Muscle Nerve 2019, 60, 629–636. [Google Scholar] [CrossRef]
- Zuo, K.J.; Shafa, G.; Antonyshyn, K.; Chan, K.; Gordon, T.; Borschel, G.H. A single session of brief electrical stimulation enhances axon regeneration through nerve autografts. Exp. Neurol. 2020, 323, 113074. [Google Scholar] [CrossRef]
- Udina, E.; Furey, M.; Busch, S.; Silver, J.; Gordon, T.; Fouad, K. Electrical stimulation of intact peripheral sensory axons in rats promotes outgrowth of their central projections. Exp. Neurol. 2008, 210, 238–247. [Google Scholar] [CrossRef]
- English, A.W.; Schwartz, G.; Meador, W.; Sabatier, M.J.; Mulligan, A. Electrical stimulation promotes peripheral axon regeneration by enhanced neuronal neurotrophin signaling. Dev. Neurobiol. 2007, 67, 158–172. [Google Scholar] [CrossRef] [PubMed]
- Behan, B.L.; DeWitt, D.G.; Bogdanowicz, D.R.; Koppes, A.N.; Bale, S.S.; Thompson, D.M. Single-walled carbon nanotubes alter Schwann cell behavior differentially within 2D and 3D environments. J. Biomed. Mater. Res. A 2011, 96, 46–57. [Google Scholar] [CrossRef]
- Pola, R.; Aprahamian, T.R.; Bosch-Marce, M.; Curry, C.; Gaetani, E.; Flex, A.; Smith, R.C.; Isner, J.M.; Losordo, D.W. Age-dependent VEGF expression and intraneural neovascularization during regeneration of peripheral nerves. Neurobiol. Aging 2004, 25, 1361–1368. [Google Scholar] [CrossRef] [PubMed]
- Kanaya, F.; Firrell, J.; Tsai, T.M.; Breidenbach, W.C. Functional results of vascularized versus nonvascularized nerve grafting. Plast. Reconstr. Surg. 1992, 89, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Doi, K.; Tamaru, K.; Sakai, K.; Kuwata, N.; Kurafuji, Y.; Kawai, S. A comparison of vascularized and conventional sural nerve grafts. J. Hand Surg. Am. 1992, 17, 670–676. [Google Scholar] [CrossRef]
- Sondell, M.; Lundborg, G.; Kanje, M. Vascular endothelial growth factor stimulates Schwann cell invasion and neovascularization of acellular nerve grafts. Brain Res. 1999, 846, 219–228. [Google Scholar] [CrossRef]
- Wongtrakul, S.; Bishop, A.T.; Friedrich, P.F. Vascular endothelial growth factor promotion of neoangiogenesis in conventional nerve grafts. J. Hand Surg. Am. 2002, 27, 277–285. [Google Scholar] [CrossRef]
- Hoyng, S.A.; De Winter, F.; Gnavi, S.; de Boer, R.; Boon, L.I.; Korvers, L.M.; Tannemaat, M.R.; Malessy, M.J.; Verhaagen, J. A comparative morphological, electrophysiological and functional analysis of axon regeneration through peripheral nerve autografts genetically modified to overexpress BDNF, CNTF, GDNF, NGF, NT3 or VEGF. Exp. Neurol. 2014, 261, 578–593. [Google Scholar] [CrossRef]
- Apel, P.J.; Ma, J.; Callahan, M.; Northam, C.N.; Alton, T.B.; Sonntag, W.E.; Li, Z. Effect of locally delivered IGF-1 on nerve regeneration during aging: An experimental study in rats. Muscle Nerve 2010, 41, 335–341. [Google Scholar] [CrossRef]
- Kawabuchi, M.; Tan, H.; Wang, S. Age affects reciprocal cellular interactions in neuromuscular synapses following peripheral nerve injury. Ageing Res. Rev. 2011, 10, 43–53. [Google Scholar] [CrossRef]
- Grinsell, D.; Keating, C.P. Peripheral nerve reconstruction after injury: A review of clinical and experimental therapies. Biomed. Res. Int. 2014, 2014, 698256. [Google Scholar] [CrossRef] [PubMed]
- Mackinnon, S.E.; Doolabh, V.B.; Novak, C.B.; Trulock, E.P. Clinical outcome following nerve allograft transplantation. Plast. Reconstr. Surg. 2001, 107, 1419–1429. [Google Scholar] [CrossRef] [PubMed]
- Tung, T.H. Tacrolimus (FK506): Safety and Applications in Reconstructive Surgery. Hand 2010, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Whitlock, E.L.; Tuffaha, S.H.; Luciano, J.P.; Yan, Y.; Hunter, D.A.; Magill, C.K.; Moore, A.M.; Tong, A.Y.; Mackinnon, S.E.; Borschel, G.H. Processed allografts and type I collagen conduits for repair of peripheral nerve gaps. Muscle Nerve 2009, 39, 787–799. [Google Scholar] [CrossRef] [PubMed]
- Azouz, S.M.; Lucas, H.D.; Mahabir, R.C.; Noland, S.S. A Survey of the Prevalence and Practice Patterns of Human Acellular Nerve Allograft Use. Plast. Reconstr. Surg. Glob. Open 2018, 6, e1803. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.J.; Wood, M.D.; Moore, A.M.; Mackinnon, S.E. Tissue engineered constructs for peripheral nerve surgery. Eur. Surg. 2013, 45. [Google Scholar] [CrossRef]
- Szynkaruk, M.; Kemp, S.W.; Wood, M.D.; Gordon, T.; Borschel, G.H. Experimental and clinical evidence for use of decellularized nerve allografts in peripheral nerve gap reconstruction. Tissue Eng. Part. B Rev. 2013, 19, 83–96. [Google Scholar] [CrossRef]
- Wood, M.D.; Kemp, S.W.; Liu, E.H.; Szynkaruk, M.; Gordon, T.; Borschel, G.H. Rat-derived processed nerve allografts support more axon regeneration in rat than human-derived processed nerve xenografts. J. Biomed. Mater. Res. A 2014, 102, 1085–1091. [Google Scholar] [CrossRef]
- Wood, M.D.; Kemp, S.W.; Weber, C.; Borschel, G.H.; Gordon, T. Outcome measures of peripheral nerve regeneration. Ann. Anat. 2011, 193, 321–333. [Google Scholar] [CrossRef]
- Rbia, N.; Bulstra, L.F.; Saffari, T.M.; Hovius, S.E.R.; Shin, A.Y. Collagen Nerve Conduits and Processed Nerve Allografts for the Reconstruction of Digital Nerve Gaps: A Single-Institution Case Series and Review of the Literature. World Neurosurg. 2019, 127, e1176–e1184. [Google Scholar] [CrossRef]
- Kuffler, D.P. Regeneration of muscle axons in the frog is directed by diffusible factors from denervated muscle and nerve tubes. J. Comp. Neurol. 1989, 281, 416–425. [Google Scholar] [CrossRef] [PubMed]
- Brooks, D.N.; Weber, R.V.; Chao, J.D.; Rinker, B.D.; Zoldos, J.; Robichaux, M.R.; Ruggeri, S.B.; Anderson, K.A.; Bonatz, E.E.; Wisotsky, S.M.; et al. Processed nerve allografts for peripheral nerve reconstruction: A multicenter study of utilization and outcomes in sensory, mixed, and motor nerve reconstructions. Microsurgery 2012, 32, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.S.; Rinker, B.D.; Weber, R.V.; Chao, J.D.; Ingari, J.V.; Brooks, D.; Buncke, G.M. Functional outcome following nerve repair in the upper extremity using processed nerve allograft. J. Hand Surg. Am. 2012, 37, 2340–2349. [Google Scholar] [CrossRef] [PubMed]
- Karabekmez, F.E.; Duymaz, A.; Moran, S.L. Early clinical outcomes with the use of decellularized nerve allograft for repair of sensory defects within the hand. Hand 2009, 4, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Taras, J.S.; Amin, N.; Patel, N.; McCabe, L.A. Allograft reconstruction for digital nerve loss. J. Hand Surg. Am. 2013, 38, 1965–1971. [Google Scholar] [CrossRef] [PubMed]
- Deiner, M.S.; Kennedy, T.E.; Fazeli, A.; Serafini, T.; Tessier-Lavigne, M.; Sretavan, D.W. Netrin-1 and DCC mediate axon guidance locally at the optic disc: Loss of function leads to optic nerve hypoplasia. Neuron 1997, 19, 575–589. [Google Scholar] [CrossRef]
- Tajdaran, K.; Gordon, T.; Wood, M.D.; Shoichet, M.S.; Borschel, G.H. A glial cell line-derived neurotrophic factor delivery system enhances nerve regeneration across acellular nerve allografts. Acta Biomater. 2016, 29, 62–70. [Google Scholar] [CrossRef]
- Zhang, Y.R.; Ka, K.; Zhang, G.C.; Zhang, H.; Shang, Y.; Zhao, G.Q.; Huang, W.H. Repair of peripheral nerve defects with chemically extracted acellular nerve allografts loaded with neurotrophic factors-transfected bone marrow mesenchymal stem cells. Neural Regen. Res. 2015, 10, 1498–1506. [Google Scholar] [CrossRef]
- Hoben, G.; Yan, Y.; Iyer, N.; Newton, P.; Hunter, D.A.; Moore, A.M.; Sakiyama-Elbert, S.E.; Wood, M.D.; Mackinnon, S.E. Comparison of acellular nerve allograft modification with Schwann cells or VEGF. Hand 2015, 10, 396–402. [Google Scholar] [CrossRef]
- Kim, B.S.; Yoo, J.J.; Atala, A. Peripheral nerve regeneration using acellular nerve grafts. J. Biomed. Mater. Res. A 2004, 68, 201–209. [Google Scholar] [CrossRef]
- Zheng, C.; Zhu, Q.; Liu, X.; Huang, X.; He, C.; Jiang, L.; Quan, D. Improved peripheral nerve regeneration using acellular nerve allografts loaded with platelet-rich plasma. Tissue Eng. Part. A 2014, 20, 3228–3240. [Google Scholar] [CrossRef] [PubMed]
- Frerichs, O.; Fansa, H.; Schicht, C.; Wolf, G.; Schneider, W.; Keilhoff, G. Reconstruction of peripheral nerves using acellular nerve grafts with implanted cultured Schwann cells. Microsurgery 2002, 22, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Hess, J.R.; Brenner, M.J.; Fox, I.K.; Nichols, C.M.; Myckatyn, T.M.; Hunter, D.A.; Rickman, S.R.; Mackinnon, S.E. Use of cold-preserved allografts seeded with autologous Schwann cells in the treatment of a long-gap peripheral nerve injury. Plast. Reconstr. Surg. 2007, 119, 246–259. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Cheng, Y.; Guo, S.; Feng, Y.; Li, Q.; Jia, H.; Wang, Y.; Tong, L.; Tong, X. Transplantation of adipose-derived stem cells for peripheral nerve repair. Int. J. Mol. Med. 2011, 28, 565–572. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Saller, M.M.; Huettl, R.E.; Mayer, J.M.; Feuchtinger, A.; Krug, C.; Holzbach, T.; Volkmer, E. Validation of a novel animal model for sciatic nerve repair with an adipose-derived stem cell loaded fibrin conduit. Neural Regen. Res. 2018, 13, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Rbia, N.; Bulstra, L.F.; Lewallen, E.A.; Hovius, S.E.R.; van Wijnen, A.J.; Shin, A.Y. Seeding decellularized nerve allografts with adipose-derived mesenchymal stromal cells: An in vitro analysis of the gene expression and growth factors produced. J. Plast. Reconstr. Aesthet. Surg. 2019, 72, 1316–1325. [Google Scholar] [CrossRef]
- Zhang, Y.; Luo, H.; Zhang, Z.; Lu, Y.; Huang, X.; Yang, L.; Xu, J.; Yang, W.; Fan, X.; Du, B.; et al. A nerve graft constructed with xenogeneic acellular nerve matrix and autologous adipose-derived mesenchymal stem cells. Biomaterials 2010, 31, 5312–5324. [Google Scholar] [CrossRef]
- Yan, Y.; Hunter, D.A.; Schellhardt, L.; Ee, X.; Snyder-Warwick, A.K.; Moore, A.M.; Mackinnon, S.E.; Wood, M.D. Nerve stepping stone has minimal impact in aiding regeneration across long acellular nerve allografts. Muscle Nerve 2018, 57, 260–267. [Google Scholar] [CrossRef]
- Abbasipour-Dalivand, S.; Mohammadi, R.; Mohammadi, V. Effects of Local Administration of Platelet Rich Plasma on Functional Recovery after Bridging Sciatic Nerve Defect Using Silicone Rubber Chamber; An Experimental Study. Bull. Emerg. Trauma 2015, 3, 1–7. [Google Scholar]
- Boecker, A.H.; van Neerven, S.G.; Scheffel, J.; Tank, J.; Altinova, H.; Seidensticker, K.; Deumens, R.; Tolba, R.; Weis, J.; Brook, G.A.; et al. Pre-differentiation of mesenchymal stromal cells in combination with a microstructured nerve guide supports peripheral nerve regeneration in the rat sciatic nerve model. Eur. J. Neurosci. 2016, 43, 404–416. [Google Scholar] [CrossRef]
- Battiston, B.; Geuna, S.; Ferrero, M.; Tos, P. Nerve repair by means of tubulization: Literature review and personal clinical experience comparing biological and synthetic conduits for sensory nerve repair. Microsurgery 2005, 25, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.E.; Leach, J.B. Neural tissue engineering: Strategies for repair and regeneration. Annu. Rev. Biomed. Eng. 2003, 5, 293–347. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Degrugillier, L.; Tremp, M.; Prautsch, K.; Sottaz, L.; Schaefer, D.J.; Madduri, S.; Kalbermatten, D. Nerve Repair With Fibrin Nerve Conduit and Modified Suture Placement. Anat. Rec. 2018, 301, 1690–1696. [Google Scholar] [CrossRef] [PubMed]
- Stocco, E.; Barbon, S.; Lora, L.; Grandi, F.; Sartore, L.; Tiengo, C.; Petrelli, L.; Dalzoppo, D.; Parnigotto, P.P.; Macchi, V.; et al. Partially oxidized polyvinyl alcohol conduitfor peripheral nerve regeneration. Sci. Rep. 2018, 8, 604. [Google Scholar] [CrossRef] [PubMed]
- Boecker, A.; Daeschler, S.C.; Kneser, U.; Harhaus, L. Relevance and Recent Developments of Chitosan in Peripheral Nerve Surgery. Front. Cell Neurosci. 2019, 13, 104. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Song, Y.; Qiao, J.; Yang, Y.; Zhang, W.; Liu, W.; Han, B. Rat sciatic nerve regeneration across a 10-mm defect bridged by a chitin/CM-chitosan artificial nerve graft. Int. J. Biol. Macromol. 2019, 129, 997–1005. [Google Scholar] [CrossRef]
- Neubrech, F.; Sauerbier, M.; Moll, W.; Seegmuller, J.; Heider, S.; Harhaus, L.; Bickert, B.; Kneser, U.; Kremer, T. Enhancing the Outcome of Traumatic Sensory Nerve Lesions of the Hand by Additional Use of a Chitosan Nerve Tube in Primary Nerve Repair: A Randomized Controlled Bicentric Trial. Plast. Reconstr. Surg. 2018, 142, 415–424. [Google Scholar] [CrossRef]
- Lopez, J.; Xin, K.; Quan, A.; Xiang, S.; Leto Barone, A.A.; Budihardjo, J.; Musavi, L.; Mulla, S.; Redett, R.; Martin, R.; et al. Poly(epsilon-Caprolactone) Nanofiber Wrap Improves Nerve Regeneration and Functional Outcomes after Delayed Nerve Repair. Plast. Reconstr. Surg. 2019, 144, 48e–57e. [Google Scholar] [CrossRef]
- Yen, C.M.; Shen, C.C.; Yang, Y.C.; Liu, B.S.; Lee, H.T.; Sheu, M.L.; Tsai, M.H.; Cheng, W.Y. Novel electrospun poly(epsilon-caprolactone)/type I collagen nanofiber conduits for repair of peripheral nerve injury. Neural Regen. Res. 2019, 14, 1617–1625. [Google Scholar] [CrossRef]
- Hou, Y.; Wang, X.; Zhang, Z.; Luo, J.; Cai, Z.; Wang, Y.; Li, Y. Repairing Transected Peripheral Nerve Using a Biomimetic Nerve Guidance Conduit Containing Intraluminal Sponge Fillers. Adv. Healthc. Mater. 2019, 8, e1900913. [Google Scholar] [CrossRef]
- Kim, S.M.; Lee, M.S.; Jeon, J.; Lee, D.H.; Yang, K.; Cho, S.W.; Han, I.; Yang, H.S. Biodegradable Nerve Guidance Conduit with Microporous and Micropatterned Poly(lactic-co-glycolic acid)-Accelerated Sciatic Nerve Regeneration. Macromol. Biosci. 2018, 18, e1800290. [Google Scholar] [CrossRef] [PubMed]
- Magaz, A.; Faroni, A.; Gough, J.E.; Reid, A.J.; Li, X.; Blaker, J.J. Bioactive Silk-Based Nerve Guidance Conduits for Augmenting Peripheral Nerve Repair. Adv. Healthc. Mater. 2018, 7, e1800308. [Google Scholar] [CrossRef] [PubMed]
- Rao, J.; Cheng, Y.; Liu, Y.; Ye, Z.; Zhan, B.; Quan, D.; Xu, Y. A multi-walled silk fibroin/silk sericin nerve conduit coated with poly(lactic-co-glycolic acid) sheath for peripheral nerve regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 73, 319–332. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Jia, Y.; Yang, W.; Zhang, C.; Zhang, K.; Chai, Y. Silk fibroin enhances peripheral nerve regeneration by improving vascularization within nerve conduits. J. Biomed. Mater. Res. A 2018, 106, 2070–2077. [Google Scholar] [CrossRef] [PubMed]
- Gontika, I.; Katsimpoulas, M.; Antoniou, E.; Kostakis, A.; Stavropoulos-Giokas, C.; Michalopoulos, E. Decellularized Human Umbilical Artery Used as Nerve Conduit. Bioengineering 2018, 5, 100. [Google Scholar] [CrossRef] [PubMed]
- Pan, D.; Mackinnon, S.E.; Wood, M.D. Advances in the repair of segmental nerve injuries and trends in reconstruction. Muscle Nerve 2019. [Google Scholar] [CrossRef]
- Patel, N.P.; Lyon, K.A.; Huang, J.H. An update-tissue engineered nerve grafts for the repair of peripheral nerve injuries. Neural Regen. Res. 2018, 13, 764–774. [Google Scholar] [CrossRef]
- Pinet, R.; Raimbeau, G.; Saint-Cast, Y.; Fouque, P.A.; Rabarin, F. Vein conduit with microsurgical suture repair of superficial branch of the radial nerve injuries at the wrist. Hand Surg. Rehabil. 2018. [Google Scholar] [CrossRef]
- Griffin, J.W.; Hogan, M.V.; Chhabra, A.B.; Deal, D.N. Peripheral nerve repair and reconstruction. J. Bone Joint Surg. Am. 2013, 95, 2144–2151. [Google Scholar] [CrossRef]
- Paprottka, F.J.; Wolf, P.; Harder, Y.; Kern, Y.; Paprottka, P.M.; Machens, H.G.; Lohmeyer, J.A. Sensory recovery outcome after digital nerve repair in relation to different reconstructive techniques: Meta-analysis and systematic review. Plast. Surg. Int. 2013, 2013, 704589. [Google Scholar] [CrossRef]
- Kim, J.Y.; Jeon, W.J.; Kim, D.H.; Rhyu, I.J.; Kim, Y.H.; Youn, I.; Park, J.W. An inside-out vein graft filled with platelet-rich plasma for repair of a short sciatic nerve defect in rats. Neural Regen. Res. 2014, 9, 1351–1357. [Google Scholar] [CrossRef] [PubMed]
- Roque, J.S.; Pomini, K.T.; Buchaim, R.L.; Buchaim, D.V.; Andreo, J.C.; Roque, D.D.; Rodrigues, A.C.; Rosa, G.M.J.; Moraes, L.H.R.; Viterbo, F. Inside-out and standard vein grafts associated with platelet-rich plasma (PRP) in sciatic nerve repair. A histomorphometric study. Acta Cir. Bras. 2017, 32, 617–625. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sabongi, R.G.; Fernandes, M.; Dos Santos, J.B. Peripheral nerve regeneration with conduits: Use of vein tubes. Neural Regen. Res. 2015, 10, 529–533. [Google Scholar] [CrossRef] [PubMed]
- Ronchi, G.; Fornasari, B.E.; Crosio, A.; Budau, C.A.; Tos, P.; Perroteau, I.; Battiston, B.; Geuna, S.; Raimondo, S.; Gambarotta, G. Chitosan Tubes Enriched with Fresh Skeletal Muscle Fibers for Primary Nerve Repair. Biomed. Res. Int. 2018, 2018, 9175248. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.X.; Lu, H.B.; Jin, X.L.; Feng, W.F.; Yang, X.N.; Qi, Z.L. Skeletal muscle-derived cells repair peripheral nerve defects in mice. Neural Regen. Res. 2020, 15, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, J.; Delaviz, H.; Mohammadi, B.; Delaviz, H.; Rad, P. Comparison of repair of peripheral nerve transection in predegenerated muscle with and without a vein graft. BMC Neurol. 2016, 16, 237. [Google Scholar] [CrossRef]
- Ramli, K.; Gasim, A.I.; Ahmad, A.A.; Htwe, O.; Mohamed Haflah, N.H.; Law, Z.K.; Hasan, S.; Naicker, A.S.; Mokhtar, S.A.; Muhamad Ariffin, M.H.; et al. Efficacy of Human Cell-Seeded Muscle-Stuffed Vein Conduit in Rat Sciatic Nerve Repair. Tissue Eng. Part. A 2019, 25, 1438–1455. [Google Scholar] [CrossRef]
- Sahin, C.; Karagoz, H.; Kulahci, Y.; Sever, C.; Akakin, D.; Kolbasi, B.; Ulkur, E.; Peker, F. Minced nerve tissue in vein grafts used as conduits in rat tibial nerves. Ann. Plast. Surg. 2014, 73, 540–546. [Google Scholar] [CrossRef]
- Siemionow, M.; Cwykiel, J.; Uygur, S.; Kwiecien, G.; Ozturk, C.; Szopinski, J.; Madajka, M. Application of epineural sheath conduit for restoration of 6-cm long nerve defects in a sheep median nerve model. Microsurgery 2019, 39, 332–339. [Google Scholar] [CrossRef]
- Ouyang, Y.; Huang, C.; Zhu, Y.; Fan, C.; Ke, Q. Fabrication of seamless electrospun collagen/PLGA conduits whose walls comprise highly longitudinal aligned nanofibers for nerve regeneration. J. Biomed. Nanotechnol. 2013, 9, 931–943. [Google Scholar] [CrossRef]
- Kriebel, A.; Hodde, D.; Kuenzel, T.; Engels, J.; Brook, G.; Mey, J. Cell-free artificial implants of electrospun fibres in a three-dimensional gelatin matrix support sciatic nerve regeneration in vivo. J. Tissue Eng. Regen. Med. 2017, 11, 3289–3304. [Google Scholar] [CrossRef]
- Ma, F.; Xu, F.; Li, R.; Zheng, Y.; Wang, F.; Wei, N.; Zhong, J.; Tang, Q.; Zhu, T.; Wang, Z.; et al. Sustained delivery of glial cell-derived neurotrophic factors in collagen conduits for facial nerve regeneration. Acta Biomater. 2018, 69, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhao, Y.; Li, X.; Xiao, Z.; Yao, Y.; Chu, Y.; Farkas, B.; Romano, I.; Brandi, F.; Dai, J. Functional Multichannel Poly(Propylene Fumarate)-Collagen Scaffold with Collagen-Binding Neurotrophic Factor 3 Promotes Neural Regeneration After Transected Spinal Cord Injury. Adv. Healthc. Mater. 2018, 7, e1800315. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Liu, C.; Hai, B.; Ma, T.; Zhang, W.; Tan, J.; Fu, X.; Wang, H.; Xu, Y.; Song, C. Chitosan conduits filled with simvastatin/Pluronic F-127 hydrogel promote peripheral nerve regeneration in rats. J. Biomed. Mater. Res. B Appl. Biomater. 2018, 106, 787–799. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yu, S.; Gu, X.; Cao, R.; Cui, S. Tissue-engineered nerve grafts using a scaffold-independent and injectable drug delivery system: A novel design with translational advantages. J. Neural Eng. 2019, 16, 036030. [Google Scholar] [CrossRef] [PubMed]
- Pfister, L.A.; Alther, E.; Papaloizos, M.; Merkle, H.P.; Gander, B. Controlled nerve growth factor release from multi-ply alginate/chitosan-based nerve conduits. Eur. J. Pharm. Biopharm. 2008, 69, 563–572. [Google Scholar] [CrossRef]
- Wu, H.; Fang, Q.; Liu, J.; Yu, X.; Xu, Y.; Wan, Y.; Xiao, B. Multi-tubule conduit-filler constructs loaded with gradient-distributed growth factors for neural tissue engineering applications. J. Mech. Behav. Biomed. Mater. 2018, 77, 671–682. [Google Scholar] [CrossRef]
- Madduri, S.; Feldman, K.; Tervoort, T.; Papaloizos, M.; Gander, B. Collagen nerve conduits releasing the neurotrophic factors GDNF and NGF. J. Control. Release 2010, 143, 168–174. [Google Scholar] [CrossRef]
- Gonzalez-Perez, F.; Hernandez, J.; Heimann, C.; Phillips, J.B.; Udina, E.; Navarro, X. Schwann cells and mesenchymal stem cells in laminin- or fibronectin-aligned matrices and regeneration across a critical size defect of 15 mm in the rat sciatic nerve. J. Neurosurg. Spine 2018, 28, 109–118. [Google Scholar] [CrossRef]
- Farzamfar, S.; Salehi, M.; Tavangar, S.M.; Verdi, J.; Mansouri, K.; Ai, A.; Malekshahi, Z.V.; Ai, J. A novel polycaprolactone/carbon nanofiber composite as a conductive neural guidance channel: An in vitro and in vivo study. Prog. Biomater. 2019, 8, 239–248. [Google Scholar] [CrossRef]
- Ikegami, Y.; Ijima, H. Development of heparin-conjugated nanofibers and a novel biological signal by immobilized growth factors for peripheral nerve regeneration. J. Biosci. Bioeng. 2019. [Google Scholar] [CrossRef]
- Beaumont, E.; Cloutier, F.C.; Atlan, M.; Rouleau, D.M.; Beaumont, P.H. Chondroitinase ABC and acute electrical stimulation are beneficial for muscle reinnervation after sciatic nerve transection in rats. Restor. Neurol. Neurosci. 2009, 27, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Chaiyasate, K.; Schaffner, A.; Jackson, I.T.; Mittal, V. Comparing FK-506 with basic fibroblast growth factor (b-FGF) on the repair of a peripheral nerve defect using an autogenous vein bridge model. J. Investig. Surg. 2009, 22, 401–405. [Google Scholar] [CrossRef] [PubMed]
- Chato-Astrain, J.; Campos, F.; Roda, O.; Miralles, E.; Durand-Herrera, D.; Saez-Moreno, J.A.; Garcia-Garcia, S.; Alaminos, M.; Campos, A.; Carriel, V. In vivo Evaluation of Nanostructured Fibrin-Agarose Hydrogels With Mesenchymal Stem Cells for Peripheral Nerve Repair. Front. Cell. Neurosci. 2018, 12, 501. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Su, X.X.; Guo, Y.C.; Li, A.; Zhang, Y.C.; Zhou, H.; Qiao, H.; Guan, L.M.; Zou, M.; Si, X.Q. Bone regeneration by nanohydroxyapatite/chitosan/poly(lactide-co-glycolide) scaffolds seeded with human umbilical cord mesenchymal stem cells in the calvarial defects of the nude mice. Biomed. Res. Int. 2015, 2015, 261938. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Crosio, A.; Fornasari, B.E.; Gambarotta, G.; Geuna, S.; Raimondo, S.; Battiston, B.; Tos, P.; Ronchi, G. Chitosan tubes enriched with fresh skeletal muscle fibers for delayed repair of peripheral nerve defects. Neural Regen. Res. 2019, 14, 1079–1084. [Google Scholar] [CrossRef]
- Lichtenfels, M.; Colome, L.; Sebben, A.D.; Braga-Silva, J. Effect of Platelet Rich Plasma and Platelet Rich Fibrin on sciatic nerve regeneration in a rat model. Microsurgery 2013, 33, 383–390. [Google Scholar] [CrossRef]
- Sariguney, Y.; Yavuzer, R.; Elmas, C.; Yenicesu, I.; Bolay, H.; Atabay, K. Effect of platelet-rich plasma on peripheral nerve regeneration. J. Reconstr. Microsurg. 2008, 24, 159–167. [Google Scholar] [CrossRef]
- Boecker, A.H.; Bozkurt, A.; Kim, B.S.; Altinova, H.; Tank, J.; Deumens, R.; Tolba, R.; Weis, J.; Brook, G.A.; Pallua, N.; et al. Cell-enrichment with olfactory ensheathing cells has limited local extra beneficial effects on nerve regeneration supported by the nerve guide Perimaix. J. Tissue Eng. Regen. Med. 2018, 12, 2125–2137. [Google Scholar] [CrossRef]
- Santiago-Figueroa, J.; Sosa, I.; Reyes, O.; Guzman, H.; Hernandez, R.; Kuffler, D. A novel technique for reducing and eliminating peripheral neuropathic pain: A clinical study. J. Pain Manag. 2011, 4, 387–394. [Google Scholar]
- Schmidt, C.E.; Shastri, V.R.; Vacanti, J.P.; Langer, R. Stimulation of neurite outgrowth using an electrically conducting polymer. Proc. Natl. Acad. Sci. USA 1997, 94, 8948–8953. [Google Scholar] [CrossRef] [PubMed]
- Prabhakaran, M.P.; Ghasemi-Mobarakeh, L.; Jin, G.; Ramakrishna, S. Electrospun conducting polymer nanofibers and electrical stimulation of nerve stem cells. J. Biosci. Bioeng. 2011, 112, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Braga Silva, J.; Marchese, G.M.; Cauduro, C.G.; Debiasi, M. Nerve conduits for treating peripheral nerve injuries: A systematic literature review. Hand Surg. Rehabil. 2017, 36, 71–85. [Google Scholar] [CrossRef] [PubMed]
- Senses, F.; Onder, M.E.; Kocyigit, I.D.; Kul, O.; Aydin, G.; Inal, E.; Atil, F.; Tekin, U. Effect of Platelet-Rich Fibrin on Peripheral Nerve Regeneration. J. Craniofac. Surg. 2016, 27, 1759–1764. [Google Scholar] [CrossRef] [PubMed]
- Kuffler, D.P.; Reyes, O.; Sosa, I.J.; Santiago-Figueroa, J. Neurological Recovery across a 12-cm Long Ulnar Nerve Gap Repaired 3.25 Years Post: A Case Report. Neurosurgery 2011. [Google Scholar] [CrossRef] [PubMed]
- Javaloy, J.; Alio, J.L.; Rodriguez, A.E.; Vega, A.; Munoz, G. Effect of platelet-rich plasma in nerve regeneration after LASIK. J. Refract. Surg. 2013, 29, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.G.; Li, S.W.; Zheng, X.M.; Hu, L.Q.; Hu, W.L.; Luo, Y. The effect of platelet-rich plasma on cavernous nerve regeneration in a rat model. Asian J. Androl. 2009, 11, 215–221. [Google Scholar] [CrossRef]
- Farrag, T.Y.; Lehar, M.; Verhaegen, P.; Carson, K.A.; Byrne, P.J. Effect of platelet rich plasma and fibrin sealant on facial nerve regeneration in a rat model. Laryngoscope 2007, 117, 157–165. [Google Scholar] [CrossRef]
- Cho, H.H.; Jang, S.; Lee, S.C.; Jeong, H.S.; Park, J.S.; Han, J.Y.; Lee, K.H.; Cho, Y.B. Effect of neural-induced mesenchymal stem cells and platelet-rich plasma on facial nerve regeneration in an acute nerve injury model. Laryngoscope 2010, 120, 907–913. [Google Scholar] [CrossRef]
- Ikumi, A.; Hara, Y.; Yoshioka, T.; Kanamori, A.; Yamazaki, M. Effect of local administration of platelet-rich plasma (PRP) on peripheral nerve regeneration: An experimental study in the rabbit model. Microsurgery 2018, 38, 300–309. [Google Scholar] [CrossRef]
- Golzadeh, A.; Mohammadi, R. Effect of local administration of platelet-derived growth factor B on functional recovery of peripheral nerve regeneration: A sciatic nerve transection model. Dent. Res. J. 2016, 13, 225–232. [Google Scholar] [CrossRef]
- Giannessi, E.; Coli, A.; Stornelli, M.R.; Miragliotta, V.; Pirone, A.; Lenzi, C.; Burchielli, S.; Vozzi, G.; De Maria, C.; Giorgetti, M. An autologously generated platelet-rich plasma suturable membrane may enhance peripheral nerve regeneration after neurorraphy in an acute injury model of sciatic nerve neurotmesis. J. Reconstr. Microsurg. 2014, 30, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.H.; Han, S.G.; Kim, S.H.; Zhu, S.J.; Huh, J.Y.; Jung, J.H.; Lee, S.H.; Kim, B.Y. Autologous fibrin glue in peripheral nerve regeneration in vivo. Microsurgery 2005, 25, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Sebben, A.D.; Lichtenfels, M.; Da Silva, J.L. Peripheral Nerve Regeneration: Cell Therapy and Neurotrophic Factors. Rev. Bras. Ortop. 2011, 46, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.; Gurney, M.E. Human platelets contain brain-derived neurotrophic factor. J. Neurosci. 1990, 10, 3469–3478. [Google Scholar] [CrossRef]
- Kuffler, D.P. An assessment of current techniques for inducing axon regeneration and neurological recovery following peripheral nerve trauma. Prog. Neurobiol. 2014, 116, 1–12. [Google Scholar] [CrossRef]
- Kuffler, D.P. Platelet-Rich Plasma Promotes Axon Regeneration, Wound Healing, and Pain Reduction: Fact or Fiction. Mol. Neurobiol. 2015, 52, 990–1014. [Google Scholar] [CrossRef]
- Wartiovaara, U.; Salven, P.; Mikkola, H.; Lassila, R.; Kaukonen, J.; Joukov, V.; Orpana, A.; Ristimaki, A.; Heikinheimo, M.; Joensuu, H.; et al. Peripheral blood platelets express VEGF-C and VEGF which are released during platelet activation. Thromb. Haemost. 1998, 80, 171–175. [Google Scholar] [CrossRef]
- Webb, N.J.; Bottomley, M.J.; Watson, C.J.; Brenchley, P.E. Vascular endothelial growth factor (VEGF) is released from platelets during blood clotting: Implications for measurement of circulating VEGF levels in clinical disease. Clin. Sci. 1998, 94, 395–404. [Google Scholar] [CrossRef]
- Geissler, J.; Stevanovic, M. Management of large peripheral nerve defects with autografting. Injury 2019, 50 (Suppl. 5), S64–S67. [Google Scholar] [CrossRef]
- Giusti, G.; Willems, W.F.; Kremer, T.; Friedrich, P.F.; Bishop, A.T.; Shin, A.Y. Return of motor function after segmental nerve loss in a rat model: Comparison of autogenous nerve graft, collagen conduit, and processed allograft (AxoGen). J. Bone Joint Surg. Am. 2012, 94, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Labroo, P.; Hilgart, D.; Davis, B.; Lambert, C.; Sant, H.; Gale, B.; Shea, J.E.; Agarwal, J. Drug-delivering nerve conduit improves regeneration in a critical-sized gap. Biotechnol. Bioeng. 2019, 116, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Saltzman, E.B.; Villa, J.C.; Doty, S.B.; Feinberg, J.H.; Lee, S.K.; Wolfe, S.W. A Comparison Between Two Collagen Nerve Conduits and Nerve Autograft: A Rat Model of Motor Nerve Regeneration. J. Hand Surg. 2019, 44, 700.e1–700.e9. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuffler, D.P.; Foy, C. Restoration of Neurological Function Following Peripheral Nerve Trauma. Int. J. Mol. Sci. 2020, 21, 1808. https://doi.org/10.3390/ijms21051808
Kuffler DP, Foy C. Restoration of Neurological Function Following Peripheral Nerve Trauma. International Journal of Molecular Sciences. 2020; 21(5):1808. https://doi.org/10.3390/ijms21051808
Chicago/Turabian StyleKuffler, Damien P., and Christian Foy. 2020. "Restoration of Neurological Function Following Peripheral Nerve Trauma" International Journal of Molecular Sciences 21, no. 5: 1808. https://doi.org/10.3390/ijms21051808
APA StyleKuffler, D. P., & Foy, C. (2020). Restoration of Neurological Function Following Peripheral Nerve Trauma. International Journal of Molecular Sciences, 21(5), 1808. https://doi.org/10.3390/ijms21051808





