Biomarkers for the Noninvasive Diagnosis of Endometriosis: State of the Art and Future Perspectives
Abstract
1. Introduction
2. Material and Methods
3. Endometriosis—An Inflammatory Disease
4. Immunological Aspects of Endometriosis
5. Angiogenesis in Endometriosis
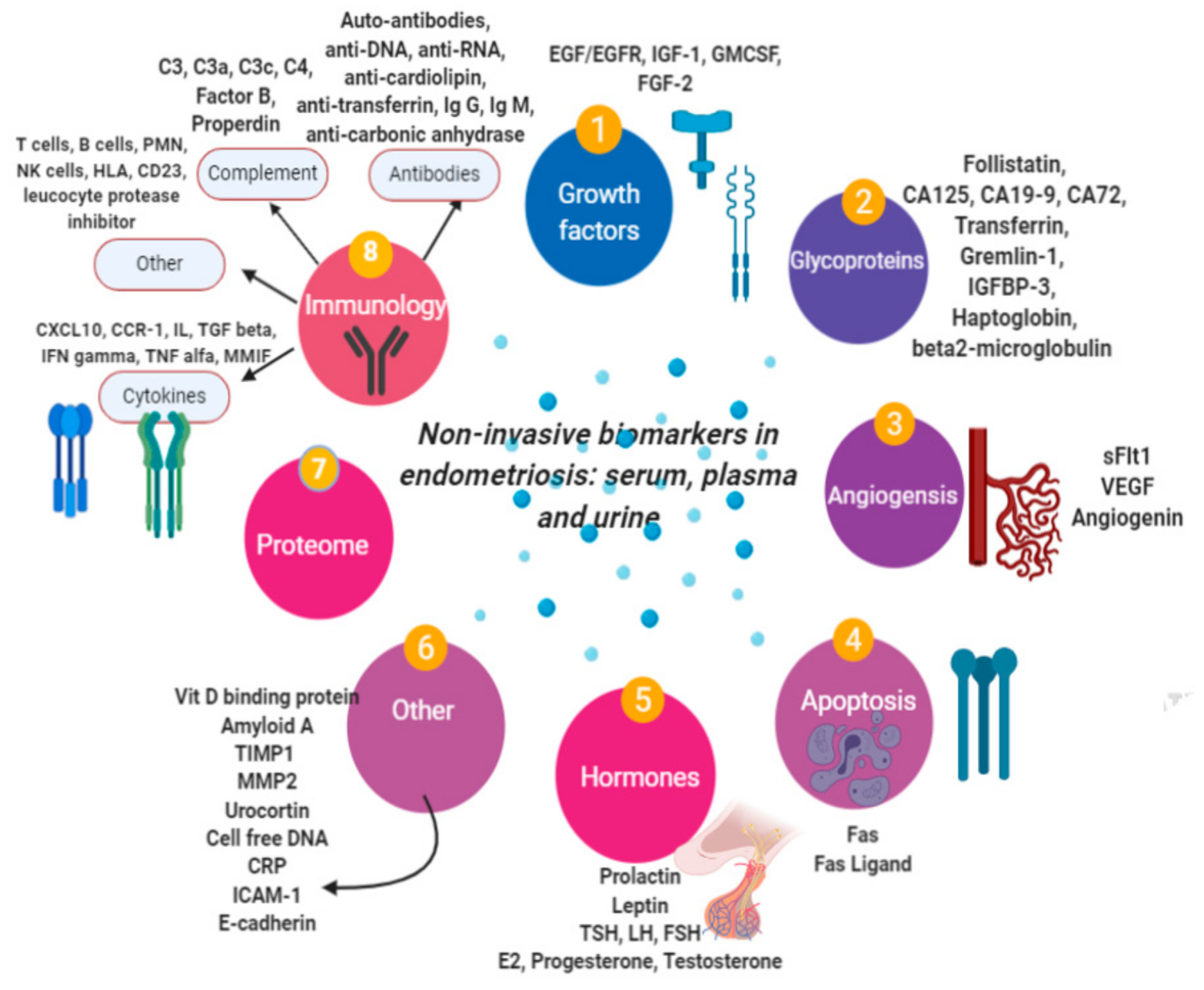
6. Studies of New Noninvasive Biomarkers in Endometriosis
6.1. Serum/Plasma Biomarkers of Endometriosis
6.1.1. Glycoproteins
6.1.2. Inflammatory Cytokines and Immunological Molecules
6.1.3. Oxidative Stress Markers
6.1.4. Growth Factors and Peptides
6.1.5. Angiogenesis Molecules
6.1.6. Autoantibodies
6.1.7. Proteomics, Metabolomics, and Genomics: The Novel Perspectives of Noninvasive Biomarkers
6.1.8. miRNAs
6.2. Urinary Biomarkers in Endometriosis
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BMI | Body mass index |
| C3 | Complement |
| CK 19 | Cytokeratin 19 |
| COX-2 | Cyclooxygenase 2 |
| CRP | C-reactive protein |
| DNA | Deoxiribonucleic acid |
| EGF | Endothelial growth factor |
| EGFR | Endothelial growth factor receptor |
| ELISA | Enzyme-linked immunosorbent assay |
| FGF | Fibroblast growth factor |
| GM-CSF | Granulocyte macrophage colony-stimulating factor |
| HOXA | Homebox A |
| ICAM | Intercellular adhesion molecule |
| IFN | Interferon |
| IGF | Insulin-like growth factor |
| Il | Interleukin |
| iNKT | Invariant natural killer T cells |
| lncRNA | Long non-coding ribonucleic acid |
| MCP-1 | Monocyte chemoattractant protein 1 |
| MIF | Macrophage inhibitory factor |
| MMP | Matrix metalloproteinase |
| miRNA | Micro ribonucleic acid |
| mtDNA | Mitochondrial DNA |
| NK | Natural killer |
| NF-KB | Nuclear factor-kappa beta |
| PDGF | Platelet-derived growth factor |
| PG | Prostaglandins |
| PGE2 | Prostaglandin E2 |
| PK-1 | Prokinetitsin 1 |
| RANTES | Regulated on Activation, Normal T Cell Expressed and Secreted |
| ROS | Reactive oxygen species |
| SOD | Superoxide dismutase |
| sFlt1 | Soluble fms-like tyrosine kinase |
| TNF | Tumor-necrosis factor |
| TSP-1 | Thrombospondin 1 |
| VDBP | Urinary vitamin D binding protein |
| VEGF | Vascular endothelial growth factor |
| VEGFR | Vascular endothelial growth factor receptor |
| Wnt/b-catenin | Wingless-type mouse mammary tumor virus integration site family |
References
- Bratila, E.; Comandasu, D.-E.; Coreleuca, C.; Cirstoiu, M.; Bohiltea, R.; Mehedintu, C.; Vladareanu, S.; Berceanu, C. Gatrointestinal Symptoms in Endometriosis Correlated With the Disease Stage. 2016. Available online: https://www.researchgate.net/publication/312552546_Gastrointestinal_symptoms_in_endometriosis_correlated_with_the_disease_stage (accessed on 27 October 2019).
- Giudice, L.C.; Kao, L.C. Endometriosis. Lancet 2004, 364, 1789–1799. [Google Scholar] [CrossRef]
- Moga, M.A.; Balan, A.; Dimienescu, O.G.; Burtea, V.; Dragomir, R.M.; Anastasiu, V.C. Circulating miRNAs as biomarkers for endometriosis and endometriosis-related ovarian cancer—An overview. J. Clin. Med. 2019, 8, 735. [Google Scholar] [CrossRef]
- Bulletti, C.; Coccia, M.E.; Battistoni, S.; Borini, A. Endometriosis and infertility. J. Assist. Reprod. Genet. 2010, 27, 441–447. [Google Scholar] [CrossRef]
- Argawal, S.K.; Chapron, C.; Giudice, L.C.; Laufer, M.R.; Lyland, N.; Missmer, S.A.; Singh, S.S.; Taylor, H.S. Clinical diagnosis of endometriosis: A call to action. Am. J. Obstet. Gynecol. 2019, 220, 354.e1–354.e12. [Google Scholar] [CrossRef]
- Nnoaham, K.E.; Hummelshoj, L.; Webster, P.; D’Hooghe, T.; de Cicco Nardone, F.; de Cicco Nardone, C.; de Cicco Nardone, C.; Jerkinson, C.; Kennedy, S.H.; Zondervan, K.T.; et al. Impact of endometriosis on quality of life and work productivity: A multicenter study across ten countries. Fertil. Steril. 2011, 96, 366–373. [Google Scholar] [CrossRef]
- Fassbender, A.; Burney, R.O.; D’Hooghe, T.; Giudice, L. Update on biomarkers for the detection of endometriosis. BioMed Res. Int. 2015, 2015, 130854. [Google Scholar] [CrossRef]
- Hadfield, R.; Mardon, H.; Barlow, D.; Kennedy, S. Delay in the diagnosis of endometriosis: A survey of women from the USA and the UK. Hum. Reprod. 1996, 11, 878–880. [Google Scholar] [CrossRef]
- Ahn, S.H.; Singh, V.; Tayade, C. Biomarkers in endometriosis: Challenges and opportunities. Fertil. Steril. 2017, 107, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Irungu, S.; Mavrelos, D.; Worthington, J.; Blyuss, O.; Saridogan, E.; Timms, J.F. Discovery of non-invasive biomarkers for the diagnosis of endometriosis. Clin. Proteom. 2019, 16, 14. [Google Scholar] [CrossRef] [PubMed]
- Biomarkers Definitions Working Group. Biomarkers and surrogate endpoints: Preferred definitions and conceptual framework. Clin. Pharmacol. Ther. 2001, 69, 89–95. [Google Scholar] [CrossRef] [PubMed]
- May, K.E.; Conduit-Hulbert, S.A.; Villar, J.; Kirtley, S.; Kennedy, S.H.; Becker, C.M. Peripheral biomarkers of endometriosis: A systematic review. Hum. Reprod. Update 2010, 16, 651–674. [Google Scholar] [CrossRef] [PubMed]
- Halme, J.; Hammond, M.G.; Hulka, J.F.; Raj, S.G.; Talbert, L.M. Retrograde menstruation in healthy women and in patients with endometriosis. Obstet. Gynecol. 1984, 64, 151–154. [Google Scholar] [PubMed]
- Lagana, A.S.; Vitale, S.G.; Salmeri, F.M.; Triolo, O.; Ban Frangez, H.; Vrtacnik-Bokal, E.; Stojanovska, L.; Apostolopoulos, V.; Granese, R.; Sofo, V. Unus pro omnibus, omnes pro uno: A novel, evidence-based, unifying theory for the pathogenesis of endometriosis. Med. Hypotheses 2017, 103, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Pluchino, N.; Taylor, H.S. Endometriosis and stem cell trafficking. Reprod. Sci. 2016, 23, 1616–1619. [Google Scholar] [CrossRef] [PubMed]
- Lagana, A.S.; Salmeri, F.M.; Vitale, S.G.; Triolo, O.; Gotte, M. Stem cell trafficking during endometriosis: May epigenetics play a pivotal role? Reprod. Sci. 2017, 25, 978–979. [Google Scholar] [CrossRef]
- Eggers, J.C.; Martino, V.; Reinbold, R.; Schafer, S.D.; Kiesel, L.; Starzinski-Powitz, A.; Schuring, A.N.; Kemper, B.; Greve, B.; Gotte, M. microRNA miR-200b affects proliferation, invasiveness and stemness of endometriotic cells by targeting ZEB1, ZEB2 and KLF4. Reprod. Biomed. Online 2016, 32, 434–445. [Google Scholar] [CrossRef]
- Marki, G.; Bokor, A.; Rigo, J.; Rigo, A. Physical pain and emotion regulation as the main predictive factors of health-related quality of life in women living with endometriosis. Hum. Reprod. 2017, 32, 1432–1438. [Google Scholar] [CrossRef]
- Lagana, A.S.; La Rosa, V.L.; Rapisarda, A.M.C. Anxiety and depression in patients with endometriosis: Impact and management challenges. Int. J. Women Health. 2017, 9, 323–330. [Google Scholar] [CrossRef]
- Zheng, W.; Cao, L.; Zheng, X.; Yuanyuan, M.; Liang, X. Anti-Angiogenic Alternative and complementary medicines for the treatment of endometriosis: A review of potential molecular mechanisms. Evid. Based Complement. Alternat. Med. 2018, 2018, 4128984. [Google Scholar] [CrossRef]
- Lousse, J.C.; Van Langendonckt, A.; Defrere, S.; Gonzalez Ramos, R.; Colette, S. Peritoneal endometriosis is an inflammatory disease. Frontiers Biosci. 2012, E4, 23–40. [Google Scholar] [CrossRef]
- Gazvani, R.; Templeton, A. Peritoneal environment, cytokines and angiogenesis in the pathophysiology of endometriosis. Reprod. 2002, 123, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Van Langendonckt, A.; Casanas-Roux, F.; Donnez, J. Oxidative stress and peritoneal endometriosis. Fertil. Steril. 2002, 77, 861–870. [Google Scholar] [CrossRef]
- Oral, E.; Olive, D.L.; Arici, A. The peritoneal environment in endometriosis. Hum. Reprod. Update 1996, 385–398. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.H.; Shoji, Y.; Chuang, P.; Tsai, S. Endometriosis: Disease pathophysiology and the role of prostaglandins. Expert Rev Mol Med. 2007, 9, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Sugino, N.; Karube-Harada, A.; Taketani, T.; Sakata, A.; Nakamura, Y. Withdrawal of ovarian steroids stimulates prostaglandin F2-alpha production through nuclear factor-kappaB activation via oxygen radicals in human endometrial stromal cells: Potential relevance to menstruation. J. Reprod. Dev. 2004, 50, 215–225. [Google Scholar] [CrossRef]
- Banu, S.K.; Lee, J.; Speights, V.O.; Starzinski-Powitz, A.; Arosh, J.A. Cyclooxygenase-2 regulates survival, migration, and invasion of human endometriotic cells through multiple mechanisms. Endocrinol 2008, 149, 1180–1189. [Google Scholar] [CrossRef]
- Wu, M.H.; Wang, C.A.; Lin, C.C.; Chen, L.-C.; Chang, W.-C.; Tsai, S.-J. Distinct regulation of cyclooxygenase-2 by interleukin-1 beta in normal and endometriotic stromal cells. J. Clin. Endocrinol. Metab. 2005, 90, 286–295. [Google Scholar] [CrossRef]
- Lagana, A.S.; Garzon, S.; Gotte, M.; Vigano, P.; Franchi, M.; Ghezzi, F.; Martin, D.C. The pathogenesis of endometriosis: Molecular and cell biology insights. Int. J. Mol. Sci. 2019, 20, 5615. [Google Scholar] [CrossRef]
- Laird, S.M.; Tuckerman, E.M.; Cork, B.A.; Li, T.C. Expression of nuclear factor kappa B in human endometrium; role in the control of interleukin 6 and leukaemia inhibitory factor production. Mol. Hum. Reprod. 2000, 6, 34–40. [Google Scholar] [CrossRef]
- Page, M.; Tuckerman, E.M.; Li, T.C.; Laird, S.M. Expression of nuclear factor kappa-B components in human endometrium. J. Reprod. Immunol. 2002, 54, 1–13. [Google Scholar] [CrossRef]
- King, A.E.; Critchley, H.; Kelly, R.W. The NF-kappaB pathway in human endometrium and first trimester decidua. Mol. Hum. Reprod. 2001, 7, 175–183. [Google Scholar] [CrossRef] [PubMed]
- González Ramos, R.; Van Langendonckt, A.; Defrère, S.; Lousse, J.C.; Colette, S.; Devoto, L.; Donnez, J. Involvement of the nuclear factor-kappaB (NF-kappa B) pathway in the pathogenesis of endometriosis. Fertil Steril 2010, 94, 1985–1994. [Google Scholar] [CrossRef] [PubMed]
- Lousse, J.C.; Van Langendonckt, A.; González Ramos, R.; Defrère, S.; Renkin, E.; Donnez, J. Increased activation of nuclear factor-kappa B (NF-kappa B) in isolated peritoneal macrophages of patients with endometriosis. Fertil. Steril. 2008, 90, 217–220. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Gupta, S.; Sikka, S. The role of oxidative stress in endometriosis. Curr. Opin. Obstet. Gynecol. 2006, 18, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Zeller, J.M.; Henig, I.; Radwanska, E.; Dmowski, W.P. Enhancement of human monocyte and peritoneal macrophage chemiluminescence activities in women with endometriosis. Am. J. Reprod. Immunol. Microbiol. 1987, 13, 78–82. [Google Scholar] [CrossRef]
- Portz, D.M.; Elkins, T.E.; White, R.; Warren, J.; Adadevoh, S.; Randolph, J. Oxygen free radicals and pelvic adhesion formation: Blocking oxygen free radical toxicity to prevent adhesion formation in an endometriosis model. Int. J. Fertil. 1991, 36, 39–42. [Google Scholar] [CrossRef]
- Wang, Y.; Sharma, R.K.; Falcone, T.; Goldberg, J.; Agarwal, A. Importance of reactive oxygen species in the peritoneal fluid of women with endometriosis or idiopathic infertility. Fertil. Steril. 1997, 68, 826–830. [Google Scholar] [CrossRef]
- Polak, G.; Koziol-Montewka, M.; Gogacz, M.; Blaszkowska, I.; Kotarski, J. Total antioxidant status of the peritoneal fluid in infertile women. Eur. J. Obstet. Gynecol. Reprod. Biol. 2001, 94, 261–263. [Google Scholar] [CrossRef]
- Wu, M.H.; Hsiao, K.Y.; Tsai, S.J. Endometriosis and possible inflammation markers. Gynecol. Minim. Invasive Ther. 2015, 4, 61–67. [Google Scholar] [CrossRef]
- Lagana, A.S.; Salmeri, F.M.; Ban Frangez, H.; Ghezzi, F.; Vrtacnik-Bokal, E.; Granese, R. Evaluation of M1 and M2 macrophages in ovarian endometriomas from women affected by endometriosis at different stages of the disease. Gynecol. Endocrinol. 2019, 30, 1–4. [Google Scholar] [CrossRef]
- Capobianco, A.; Monno, A.; Cottone, L.; Venneri, M.A.; Biziato, D.; Di Puppo, F.; Ferrari, S.; De Palma, M.; Manfredi, A.A.; Rovere-Querini, P. Proangiogenic Tie2(+) macrophages infiltrate human and murine endometriotic lesions and dictate their growth in a mouse model of the disease. Am. J. Pathol. 2011, 179, 2651–2659. [Google Scholar] [CrossRef] [PubMed]
- Lagana, A.S.; Ghezzi, F.; Vetvicka, V. Endometriosis and risk of ovarian cancer: What do we know? Arch. Gynecol. Obstet. 2019, 301, 1–10. [Google Scholar] [CrossRef]
- Wu, M.Y.; Yang, J.H.; Chao, K.H.; Hwang, J.L.; Yang, Y.S.; Ho, H.N. Increase in the expression of killer cell inhibitory receptors on peritoneal natural killer cells in women with endometriosis. Fertil. Steril. 2000, 74, 1187–1191. [Google Scholar] [CrossRef]
- Lagana, A.S.; Triolo, O.; Salmeri, F.M.; Granese, R.; Palmara, V.I.; Ban Frangez, H.; Vrtcnik Bokal, E.; Sofo, V. Natural Killer T cell subsets in eutopic and ectopic endometrium: A fresh look to a busy corner. Arch. Gynecol. Obstet. 2016, 293, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Ho, H.N.; Wu, M.Y.; Chao, K.H.; Der Chen, C.; Chen, S.U.; Yang, Y.S. Peritoneal interleukin-10 increases with decrease in activated CD4+ T lymphocytes in women with endometriosis. Hum. Reprod. 1997, 12, 2528–2533. [Google Scholar] [CrossRef] [PubMed]
- Nie, M.-F.; Xie, Q.; Wu, Y.-H.; He, H.; Zou, L.-J.; She, X.-L.; Wu, X.-Q. Serum and Ectopic Endometrium from Women with Endometriosis Modulate Macrophage M1/M2 Polarization via the Smad2/Smad3 Pathway. J. Immun. Res. 2018, 2018, 14. [Google Scholar] [CrossRef] [PubMed]
- Kralickova, M.; Vetvicka, V. Immunological aspects of endometriosis: Review. Ann. Transl. Med. 2015, 3, 153. [Google Scholar] [CrossRef]
- Osuga, Y.; Hirota, Y.; Hirata, T.; Takamura, M.; Urata, Y.; Harada, M.; Izumi, G.; Fujii, T.; Koga, K. Th2 Cells and Th17 cells in the development of endometriosis—Possible roles of interleukin-4 and interleukin-17A. J. Endometr. Pelvic Pain Dis. 2016, 8, 136–140. [Google Scholar] [CrossRef]
- Asante, A.; Taylor, R.N. Endometriosis: The role of neuroangiogenesis. Ann. Rev. Physiol. 2011, 73, 163–182. [Google Scholar] [CrossRef]
- Hanahan, D.; Folkman, J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 1996, 86, 353–364. [Google Scholar] [CrossRef]
- Ahn, S.H.; Edwards, A.K.; Singh, S.S.; Young, S.L.; Lessey, B.A.; Tayade, C. Il-17A contributes to the pathogenesis of endometriosis bu triggering proinflammatory cytokines and angiogenic growth factors. J. Immunol. 2015, 195, 2591–2600. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.K.; Qin, R.Y. Mechanism and its regulation of tumor-induced angiogenesis. World J. Gastroenterol. 2003, 9, 1144–1155. [Google Scholar] [CrossRef] [PubMed]
- Strieter, R.M.; Polverini, P.J.; Kunkel, S.L.; Arenberg, D.A.; Burdick, M.D.; Kasper, J.; Dzuiba, J.; Van Damme, J.; Walz, A.; Marriott, D.; et al. The functional role of the ELR motif in CXC chemokine-mediated angiogenesis. J. Biol. Chem. 1995, 270, 27348–27357. [Google Scholar] [CrossRef] [PubMed]
- Volpert, O.V.; Fong, T.; Koch, A.E.; Peterson, J.D.; Waltenbaugh, C.; Tepper, R.I.; Bouck, N.P. Inhibition of angiogenesis by interleukin 4. J. Exp. Med. 1998, 188, 1039–1046. [Google Scholar] [CrossRef]
- Torisu, H.; Ono, M.; Kiryu, H.; Furue, M.; Ohmoto, Y.; Nakayama, J.; Nishioka, Y.; Sone, S.; Kuwano, M. Macrophage infiltration correlates with tumor stage and angiogenesis in human malignant melanoma: Possible involvement of TNFalpha and IL-1alpha. Int. J. Cancer 2000, 85, 182–188. [Google Scholar] [CrossRef]
- Donnez, J.; Smoes, P.; Gillerot, S.; Casanas-Roux, F.; Nisolle, M. Vascular endothelial growth factor (VEGF) in endometriosis. Hum. Reprod. 1998, 3, 1686–1690. [Google Scholar] [CrossRef]
- Machado, D.E.; Rodrigues-Baptista, K.C.; Perini, J.A. Soares de Moura, R. Euterpe oleracea extract (Açaí) is a promising novel pharmacological therapeutic treatment for experimental endometriosis. PLoS ONE 2016, 11, e0166059. [Google Scholar] [CrossRef]
- Thambisetty, M.; Lovestone, S. Blood-based biomarkers of Alzheimers disease: Challenging but feasible. Biomark. Med. 2010, 4, 65–79. [Google Scholar] [CrossRef]
- Mol, B.W.J.; Bayram, N.; Lijmer, J.G.; Wiegerinck, M.A.H.M.; Bongers, M.Y.; Van der Veen, F.; Bossuyt, P. The performance of CA-125 measurement in the detection of endometriosis: A meta-analysis. Fertil. Steril. 1998, 70, 1101–1108. [Google Scholar] [CrossRef]
- Mihalyi, A.; Gevaert, O.; Kyama, C.M.; Simsa, P.; Pochet, N.; De Smet, F.; De Moor, B.; Meuleman, C.; Billen, J.; Blanckaert, N.; et al. Non-invasive diagnosis of endometriosis based on a combined analysis of six plasma biomarkers. Hum. Reprod. 2010, 25, 654–664. [Google Scholar] [CrossRef]
- Agic, A.; Djalali, S.; Wolfler, M.M.; Halis, G.; Diedrich, K.; Hornung, D. Combination of CCR1 mRNA, MCP1, and CA125 measurements in peripheral blood as a diagnostic test for endometriosis. Reprod. Sci. 2008, 15, 906–911. [Google Scholar] [CrossRef] [PubMed]
- Vodolazkaia, A.; El-Aalamat, Y.; Popovic, D. Evaluation of a panel of 28 biomarkers for the non-invasive diagnosis of endometriosis. Hum. Reprod. 2012, 27, 2698–2711. [Google Scholar] [CrossRef] [PubMed]
- Ozhan, E.; Kokcu, A.; Yanik, K.; Gunaydin, M. Investigation of diagnostic potentials of nine different biomarkers in endometriosis. Eur. J. Obstet. Gynecol. 2014, 178, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Mosbah, A.; Nabiel, Y.; Khashaba, E. Interleukin-6, intracellular adhesion molecule-1, and glycodelin A levels in serum and peritoneal fluid as biomarkers for endometriosis. Obstet. Gynecol. 2016, 134, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Kocbek, V.; Vouk, K.; Bersinger, N.A.; Mueller, M.D.; Lanišnik Rižner, T. Panels of Cytokines and Other Secretory Proteins as Potential Biomarkers of Ovarian Endometriosis. J. Mol. Diagn. 2015, 17, 325–334. [Google Scholar] [CrossRef]
- Pretice Crapper, E. Clinical Biomarkers for the Noninvasive Diagnosis of Endometriosis. 2016. Available online: http://hdl.handle.net/11375/20495 (accessed on 26 February 2020).
- Reis, F.M.; Luisi, S.; Abrão, M.S.; Rocha, A.L.L.; Viganò, P.; Rezende, C.P.; Florio, P.; Petraglia, F. Diagnostic value of serum activin A and follistatin levels in women with peritoneal, ovarian and deep infiltrating endometriosis. Hum. Reprod. 2012, 27, 1445–1450. [Google Scholar] [CrossRef]
- Vigano, P.; Infantino, M.; Lattuada, D.; Lauletta, R.; Ponti, E.; Somigliana, E.; Vignali, M.; DiBlasio, A.M. Intercellular adhesion molecule-1 (ICAM-1) gene polymorphisms in endometriosis. Mol. Hum. Reprod. 2003, 9, 47–52. [Google Scholar] [CrossRef][Green Version]
- Othman, E.E.R.; Hornung, D.; Salem, H.T.; Kalifa, E.A.; El-Metwally, T.H.; Al-Hendy, A. Serum cytokines as biomarkers for nonsurgical prediction of endometriosis. Eur. J. Obstet. Gynecol. Repd. Biol. 2008, 137, 240–246. [Google Scholar] [CrossRef]
- Borrelli, G.M.; Abrão, M.S.; Mechsner, S. Can chemokines be used as biomarkers for endometriosis? A systematic review. Hum. Reprod. 2014, 29, 253–266. [Google Scholar] [CrossRef]
- Martinez, S.; Garrido, N.; Coperias, J.L.; Pardo, F.; Desco, J.; Garcia-Velasco, J.A.; Simon, C.; Pellicer, A. Serum interleukin-6 levels are elevated in women with minimal-mild endometriosis. Hum. Reprod. 2007, 22, 836–842. [Google Scholar] [CrossRef]
- Socolov, R.; Butureanu, S.; Angioni, S.; Sindilar, A.; Boiculese, V.L.; Cozma, L.; Socolov, D. The value of serological markers in the diagnosis and prognosis of endometriosis: A prospective case-control study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2010, 154, 215–217. [Google Scholar] [CrossRef] [PubMed]
- Kalu, E.; Sumar, N.; Giannopoulos, T.; Patel, P.; Croucher, C.; Sherriff, E.; Bansal, A. Cytokine profiles in serum and peritoneal fluid from infertile women with and without endometriosis. J. Obstet. Gynaecol. Res. 2007, 33, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Pizzo, A.; Salmeri, F.M.; Ardita, F.V.; Sofo, V.; Tripepi, M.; Marsico, S. Behaviour of cytokine levels in serum and peritoneal fluid of women with endometriosis. Gynecol. Obstet. Investig. 2002, 54, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Ohata, Y.; Harada, T.; Miyakoda, H.; Taniguchi, F.; Iwabe, T.; Terakawa, N. Serum interleukin-8 levels are elevated in patients with ovarian endometrioma. Fertil. Steril. 2008, 90, 994–999. [Google Scholar] [CrossRef]
- Darai, E.; Detchev, R.; Hugol, D.; Quang, N.T. Serum and cyst fluid levels of interleukin (IL) -6, IL-8 and tumour necrosis factor-alpha in women with endometriomas and benign and malignant cystic ovarian tumours. Hum. Reprod. 2003, 18, 1681–1685. [Google Scholar] [CrossRef] [PubMed]
- Xavier, P.; Belo, L.; Beires, J.; Rebelo, I.; Martinez-de-Oliveira, J.; Lunet, N.; Barros, H. Serum levels of VEGF and TNF-a and their association with C-reactive protein in patients with endometriosis. Arch. Gynecol. Obstet. 2006, 273, 227–231. [Google Scholar] [CrossRef]
- Cho, S.H.; Oh, Y.J.; Nam, A.; Kim, H.Y.; Park, J.H.; Kim, J.H.; Cho, D.J.; Lee, B.S. Evaluation of Serum and Urinary Angiogenic Factors in Patients with Endometriosis. Am. J. Reprod. Immunol. 2007, 58, 497–504. [Google Scholar] [CrossRef]
- Seeber, B.; Sammel, M.D.; Fan, X.; Gerton, G.L.; Shaunik, A.; Chittams, J.; Barnhart, K.T. Panel of markers can accurately predict endometriosis in a subset of patients. Fertil. Steril. 2008, 89, 1073–1081. [Google Scholar] [CrossRef]
- Steff, A.M.; Gagné, D.; Pagé, M.; Rioux, A.; Hugo, P.; Gosselin, D. Insulin-like growth factor-1, soluble tumor necrosis factor receptor-1 and angiogenin in endometriosis patients. Am. J. Reprod. Immunol. 2004, 51, 166–173. [Google Scholar] [CrossRef]
- Choi, Y.S.; Kim, S.; Oh, Y.S.; Cho, S.; Hoon Kim, S. Elevated serum interleukin-32 levels in patients with endometriosis: A cross-sectional study. Am. J. Reprod. Immunol. 2019, 82, e13149. [Google Scholar] [CrossRef]
- Kikuchi, Y.; Ishikawa, N.; Hirata, J.; Imaizumi, E.; Sasa, H.; Nagata, I. Changes of peripheral blood lymphocyte subsets before and after operation of patients with endometriosis. Acta Obstet. Gynecol. Scand. 1993, 72, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Tuten, A.; Kucur, M.; Imamoglu, M.; Kaya, B.; Acikgoz, A.S.; Yilmaz, N.; Ozturk, Z.; Oncul, M. Copeptin is associated with the severity of endometriosis. Arch. Gynecol. Obstet. 2014, 290, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, L.F.; Samadder, A.N.; Agarwal, A.; Fernandes, L.F.; Abrao, M.S. Oxidative stress biomarkers in patients with endometriosis: Systematic review. Arch. Gynecol. Obstet. 2012, 286, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Scutiero, G.; Iannone, P.; Bernardi, G.; Bonaccorsi, G.; Spadaro, S.; Volta, C.A.; Greco, P.; Nappi, L. Oxidative stress and endometriosis: A systematic review of the literature. Oxidative Med. Cell Longev. 2017, 2017, 7265238. [Google Scholar] [CrossRef] [PubMed]
- Nasiri, N.; Moini, A.; Eftekhari-Yazdi, P.; Karimian, L.; Salman-Yazdi, R.; Arabipoor, A. Oxidative stress statues in serum and follicular fluid of women with endometriosis. Cell. J. 2017, 18, 582–587. [Google Scholar] [PubMed]
- Verit, F.F.; Erel, O.; Celik, N. Serum paraoxonase-1 activity in women with endometriosis and its relationship with the stage of the disease. Hum. Reprod. 2008, 100–104. [Google Scholar] [CrossRef]
- Jackson, L.W.; Schisterman, E.F.; Dey-Rao, R.; Browne, R.; Armstrong, D. Oxidative stress and endometriosis. Hum. Reprod. 2005, 20, 2014–2020. [Google Scholar] [CrossRef]
- Turkyilmaz, E.; Yildirim, M.; Cendek, B.D.; Baran, P.; Alisik, M.; Dalgaci, F.; Yavuz, A.F. Evaluation of oxidative stress markers and intra-extracellular antioxidant activities in patients with endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 199, 164–168. [Google Scholar] [CrossRef]
- Prieto, L.; Quesada, J.F.; Cambero, O.; Pacheco, A.; Pellicer, A.; Codoceo, R.; Garcia-Velasco, J.A. Analysis of follicular fluid and serum markers of oxidative stress in women with infertility related to endometriosis. Fertil. Steril. 2012, 98, 126–130. [Google Scholar] [CrossRef]
- Andrisani, A.; Dona, G.; Brunati, A.M.; Clari, G.; Armanini, D.; Ragazzi, E.; Ambrosini, G.; Bordin, L. Increased oxidation-related glutathionylation and carbonic anhydrase activity in endometriosis. Reprod. Biomed. Online 2014, 28, 773–779. [Google Scholar] [CrossRef][Green Version]
- Gurgan, T.; Bukulmez, O.; Yarali, H.; Tanir, M.; Akyildiz, S. Serum and peritoneal fluid levels of IGF I and II and insulin-like growth binding protein-3 in endometriosis. J. Reprod. Med. 1999, 44, 450–454. [Google Scholar] [CrossRef] [PubMed]
- Steff, A.M.; Gagne, D.; Page, M.; Hugo, P.; Gosselin, D. Concentration of soluble intercellular adhesion molecule-1 in serum samples from patients with endometriosis collected during the luteal phase of the menstrual cycle. Hum. Reprod. 2004, 19, 172–178. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Philippoussis, F.; Gagne, D.; Hugo, P.; Gosselin, D. Concentrations of alpha-fetoprotein, insulin-like growth factor binding protein-3, c-erbB-2, and epidermal growth factor in serum of patients with endometriosis. J. Soc. Gynecol. Investig. 2004, 11, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Galiano, D.; Navarro, V.M.; Gaytan, F.; Tena-Sempere, M. Expanding roles of NUCB2/nesfatin-1 in neuroendocrine regulation. J. Mol. Endocrinol. 2010, 45, 281–290. [Google Scholar] [CrossRef]
- Sengul, O.; Dilbaz, B.; Halici, Z.; Ferah, I.; Cadirci, E.; Yilmaz, F. Decreased serum nesfatin-1 levels in endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 177, 34–37. [Google Scholar] [CrossRef]
- Tokmak, A.; Ugur, M.; Tonguc, E.; Var, T.; Moraloglu, O.; Ozaksit, G. The value of urocortin and CA-125 in the diagnosis of endometrioma. Arch. Gynecol. Obstet. 2011, 283, 1075–1079. [Google Scholar] [CrossRef]
- Chmaj-Wierzchowska, K.; Kampioni, M.; Wilczak, M.; Sajdak, S.; Opala, T. Novel markers in the diagnostics of endometriomas: Urocortin, ghrelin and leptin or leukocytes, fibrinogen, and CA-125? Taiwan. J. Obstet. Gynecol. 2015, 54, 126–130. [Google Scholar] [CrossRef]
- Florio, P.; Reis, F.M.; Torres, P.B.; Calonaci, F.; Toti, P.; Bocchi, C.; Linton, E.A.; Petraglia, F. Plasma urocortin levels in the diagnosis of ovarian endometriosis. Obstet. Gynecol. 2007, 110, 594–600. [Google Scholar] [CrossRef]
- Mohamed, M.L.; El Behery, M.M.; Mansour, S.A.E.-A. Comparative study between VEGF-A and CA-125 in diagnosis and follow-up of advanced endometriosis after conservative laparoscopic surgery. Arch. Gynecol. Obstet. 2013, 287, 77–82. [Google Scholar] [CrossRef]
- Bourlev, V.; Iljasova, N.; Adamyan, L.; Larsson, A.; Olovsson, M. Signs of reduced angiogenic activity after surgical removal of deeply infiltrating endometriosis. Fertil. Steril. 2010, 94, 52–57. [Google Scholar] [CrossRef]
- Szubert, M.; Suzin, J.; Duechler, M.; Szuławska, A.; Czyz, M.; Kowalczyk-Amico, K. Evaluation of selected angiogenic and inflammatory markers in endometriosis before and after danazol treatment. Reprod. Fertil. Dev. 2014, 26, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Fan, R.; Huang, X.; Xu, H.; Zhang, X. Reduced levels of serum pigment epithelium-derived factor in women with endometriosis. Reprod. Sci. 2012, 19, 64–69. [Google Scholar] [CrossRef]
- Ozhan, E.; Kokcu, A.; Yanik, K.; Gunaydin, M. Investigation of diagnostic potentials of nine different biomarkers in endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 178, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Gajbhiye, R.; Sonawani, A.; Khan, S.; Suryawanshi, A.; Kadam, S.; Warty, N.; Raut, V.; Khole, V. Identification and validation of novel serum markers for early diagnosis of endometriosis. Hum. Reprod. 2012, 27, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.-C.; Wang, S.-C.; Chao, C.-C.; Su, C.-L.; Lee, Y.-L.; Chen, L.-Y. Evaluation of serum autoantibody levels in the diagnosis of ovarian endometrioma. J. Clin. Lab Anal. 2010, 24, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Nabeta, M.; Abe, Y.; Kagawa, L.; Haraguchi, R.; Kito, K.; Ueda, N.; Sugita, A.; Yokoyama, M.; Kusanagi, Y.; Ito, M. Identification of anti-α-enolase autoantibody as a novel serum marker for endometriosis. Proteom. Clin. Appl. 2009, 3, 1201–1210. [Google Scholar] [CrossRef]
- Inagaki, J.; Matsuura, E.; Kaihara, K.; Kobayashi, K.; Yasuda, T.; Nomizu, M.; Sugiura-Ogasawara, M.; Katano, K.; Aoki, K. IgG antilaminin-1 autoantibody and recurrent miscarriages. Am. J. Reprod. Immunol. 2001, 45, 232–238. [Google Scholar] [CrossRef]
- Inagaki, J.; Sugiura-Ogasawara, M.; Nomizu, M.; Nakatsuka, M.; Ikuta, K.; Suzuki, N.; Kaihara, K.; Kobayashi, K.; Yasuda, T.; Shoenfeld, Y.; et al. An association of IgG anti-laminin-1 autoantibodies with endometriosis in infertile patients. Hum. Reprod. 2003, 18, 544–549. [Google Scholar] [CrossRef]
- Mathur, S.; Garza, D.E.; Smith, L.F. Endometrial autoantigens eliciting immunoglobulin (Ig) G, IgA, and IgM responses in endometriosis. Fertil. Steril. 1990, 54, 56–63. [Google Scholar] [CrossRef]
- Odukoya, O.A.; Wheatcroft, N.; Weetman, A.P.; Cooke, I.D. The prevalence of endometrial immunoglobulin G antibodies in patients with endometriosis. Hum. Reprod. 1995, 10, 1214–1219. [Google Scholar] [CrossRef]
- Long, X.; Jinag, P.; Zhou, L.; Zhang, W. Evaluation of novel serum biomarkers and the proteomic differences of endometriosis and adenomyosis using MALDI-TOF–MS. Arch. Gynecol Obstet. 2013, 288, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Wölfler, M.M.; Schwamborn, C.; Otten, D.; Hornung, D.; Liu, H.; Rath, W. Mass spectrometry and serum pattern profiling for analyzing the individual risk for endometriosis: Promising insights? Fertil. Steril. 2009, 91, 2331–2337. [Google Scholar] [CrossRef]
- Zheng, N.; Pan, C.; Liu, W. New serum biomarkers for detection of endometriosis using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. J. Int. Med. Res. 2011, 39, 1184–1192. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zheng, W.; Mu, L.; Zhang, S.-Z. Identifying biomarkers of endometriosis using serum protein fingerprinting and artificial neural networks. Int. J. Gynaecol. Obstet. 2008, 101, 253–258. [Google Scholar] [CrossRef]
- Jing, J.; Qiao, Y.; Suginami, H.; Taniguchi, F.; Shi, H.; Wang, S. Two novel serum biomarkers for endometriosis screened by surface-enhanced laser desorption/ionization time-of-flight mass spectrometry and their change after laparoscopic removal of endometriosis. Fertil. Steril. 2008, 92, 1221–1227. [Google Scholar] [CrossRef]
- Malin, E.; Bodil, R.; Gunnar, E.; Bodil, O. AXIN1 in Plasma or Serum Is a Potential New Biomarker for Endometriosis. Int. J. Mol. Sci. 2019, 20, 189. [Google Scholar] [CrossRef]
- Signorile, P.G.; Baldi, A. Supporting evidences for potential biomarkers of endometriosis detected in peripheral blood. Data Brief. 2015, 5, 971–974. [Google Scholar] [CrossRef][Green Version]
- Signorile, P.G.; Baldi, A. Serum Biomarker of Endometriosis. J. Cell Physiol. 2014, 229, 1731–1735. [Google Scholar] [CrossRef]
- Dutta, M.; Joshi, M.; Srivastava, S.; Lodh, I.; Chakravarty, B.; Chaudhury, K. A metabonomics approach as a means for identification of potential biomarkers for early diagnosis of endometriosis. Mol. Biosyst. 2012, 8, 3281–3287. [Google Scholar] [CrossRef]
- Vouk, K.; Hevir, N.; Ribic-Pucelj, M.; Haarpaintner, G.; Scherb, H.; Osredkar, J.; Moller, G.; Prehn, C.; Lanisnik Rizner, T.; Adamski, J. Discovery of phosphatidylcholines and sphingomyelins as biomarkers for ovarian endometriosis. Hum. Reprod. 2012, 27, 2955–2965. [Google Scholar] [CrossRef]
- Zachariah, R.; Schmid, S.; Radpour, R.; Buerki, N.; Fan, A.X.-C.; Hahn, S.; Holzgreve, W.; Zhong, X.Y. Circulating cell free DNA as a potential biomarker for minimal and mild endometriosis. Reprod. Biomed. Online 2009, 18, 407–411. [Google Scholar] [CrossRef]
- Creed, J.; Maggrah, A.; Reguly, B.; Harbottle, A. Mitochondrial DNA deletions accurately detect endometriosis in symptomatic females of child-bearing age. Biomark. Med. 2019, 13, 291–306. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Weber, J.A.; Baxter, D.H.; Zhang, S.; Huang, D.Y.; Huang, K.H.; Lee, M.J.; Galas, D.J.; Wang, K. The microRNA spectrum in 12 body fluids. Clin. Chem. 2010, 56, 1733–1741. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson Teague, E.M.C.; Print, C.G.; Hull, M.L. The role of microRNAs in endometriosis and associated reproductive conditions. Hum. Reprod. Update 2009, 16, 142–165. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Tang, Q.; Wu, W.; Xia, Y.; Chen, D.; Wang, X. miR-20a contributes to endometriosis by regulating NTN4 expression. Mol. Biol. Rep. 2014, 41, 5793–5797. [Google Scholar] [CrossRef]
- Wang, W.T.; Zhao, Y.N.; Han, B.W.; Hong, S.J.; Chen, Y.Q. CirculatingMicroRNAsIdentifiedinaGenome-Wide Serum MicroRNA Expression Analysis as Noninvasive Biomarkers for Endometriosis. J. Clin. Endocrinol. Metab. 2013, 98, 281–289. [Google Scholar] [CrossRef]
- Kozomara, A.; Birgaoanu, M.; Griffiths-Jones, S. miRBase: From microRNA sequences to function. Nucleic Acids Res. 2019, 47, D155–D162. [Google Scholar] [CrossRef]
- Rekker, K.; Saare, M.; Roost, A.M.; Kaart, T.; Sõritsa, D.; Karro, H.; Sõritsa, A.; Simón, C.; Salumets, A.; Peters, M. Circulating miR-200-family micro-RNAs have altered plasma levels in patients with endometriosis and vary with blood collection time. Fertil. Steril. 2015, 104, 938–946. [Google Scholar] [CrossRef]
- Nisenblat, V.; Sharkey, D.J.; Wang, Z.; Evans, S.F.; Healey, M.; Ohlsson Teague, E.M.C.; Print, C.G.; Robertson, S.A.; Hull, M.L. Plasma miRNAs display limited potential as diagnostic tools for endometriosis. Clin. Endocrinol. Metab. 2019, 104, 1999–2022. [Google Scholar] [CrossRef]
- Vanhie, A.; Peterse, D.O.D.; Beckers, A.; Cuellar, A.; Fassbender, A.; Meuleman, C.; Mestdagh, P.; D’Hooghe, T. Plasma miRNAs as biomarkers for endometriosis. Hum. Reprod. 2019, 34, 1650–1660. [Google Scholar] [CrossRef]
- Jia, S.Z.; Yang, Y.; Lang, J.; Sun, P.; Leng, J. Plasma miR-17–5p, miR-20a and miR-22 are down-regulated in women with endometriosis. Hum. Reprod. 2013, 28, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Suryawanshi, S.; Vlad, A.M.; Lin, H.M.; Mantia-Smaldone, G.; Laskey, R.; Lee, M.; Lin, Y.; Donnellan, N.; Klein-Patel, M.; Lee, T.; et al. Plasma MicroRNAs as novel biomarkers for endometriosis and endometriosis-associated ovarian cancer. Clin. Cancer Res. 2013, 19, 1213–1224. [Google Scholar] [CrossRef] [PubMed]
- Cosar, E.; Mamillapalli, R.; Ersoy, G.S.; Cho, S.; Seifer, B.; Taylor, H.S. Serum microRNAs as diagnostic markers of endometriosis: A comprehensive array-based analysis. Fertil. Steril. 2016, 106, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-Y.; Zheng, L.-W.; Li, C.-J.; Xu, Y.; Zhou, X.; Fu, L.-I.; Li, D.-D.; Sun, L.-T.; Zhang, D.; Cui, M.-H.; et al. Dysregulated Expression of Long Noncoding RNAs in Endometriosis. Crit. Rev. Eukaryot. Expr. 2019, 29, 113–121. [Google Scholar] [CrossRef]
- Wang, W.-T.; Sun, Y.-M.; Huang, W.; He, B.; Zhao, Y.-N.; Chen, Y.-Q. Genome-wide long non-coding RNA Analysis identified circulating LncRNAs as novel non-invasive diagnostic biomarkers for gynecological disease. Sci. Rep. 2016, 6, 23343. [Google Scholar] [CrossRef]
- Qiu, J.; Zhang, X.; Ding, Y.; Hua, K. 1856 circulating exosomal long noncoding RNA-TC0101441 as a non-invasive biomarker for the prediction of endometriosis severity and recurrence. J. Minim. Invasive Gynecol. 2019, 26, S170–S171. [Google Scholar] [CrossRef]
- Gueye, N.A.; Stanhiser, J.; Valentine, L.; Kotlyar, A.; Goodman, L.; Falcone, T. Biomarkers for Endometriosis in saliva, urine, and peritoneal fluid. Biomark. endometr. 2017, 141–163. [Google Scholar] [CrossRef]
- Kuessel, C.; Jaeger-Lamsky, A.; Pateisky, P.; Rossberg, N.; Schulz, A.; Schmitz, A.A.P.; Staudigl, C.; Wenzl, R. Cytokeratin-19 as a biomarker in urine and in serum for the diagnosis of endometriosis—A prospective study. Gynecol. Endocrinol. 2014, 30, 38–41. [Google Scholar] [CrossRef]
- Hawkins, S.M.; Creighton, C.J.; Han, D.Y.; Zariff, A.; Anderson, M.L.; Gunaratne, P.H.; Matzuk, M.M. Functional microRNA involved in endometriosis. Molec Endocrinol. 2011, 25, 821–832. [Google Scholar] [CrossRef]
- Becker, C.M.; Louis, G.; Exarhopoulos, A.; Mechsner, S.; Ebert, A.D.; Zurakowski, D.; Moses, M.A. Matrix metalloproteinases are elevated in the urine of patients with endometriosis. Fertil. Steril. 2010, 94, 2343–2346. [Google Scholar] [CrossRef] [PubMed]
- Tokushige, N.; Markham, R.; Crossett, B.; Ahn, S.B.; Nelaturi, V.L.; Khan, A.; Fraser, I.S. Discovery of a novel biomarker in the urine in women with endometriosis. Fertil. Steril. 2011, 95, 46–49. [Google Scholar] [CrossRef]
- El-Kasti, M.M.; Wright, C.; Fye, H.K.S.; Roseman, F.; Kessler, B.M.; Becker, C.M. Urinary peptide profiling identifies a panel of putative biomarkers for diagnosing and staging endometriosis. Fertil. Steril. 2011, 95, 1261–1266. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Choi, Y.S.; Yim, S.Y.; Yang, H.I.; Jeon, Y.E.; Lee, K.E.; Kim, H.Y.; Seo, S.K.; Lee, B.S. Urinary vitamin D-binding protein is elevated in patients with endometriosis. Hum. Reprod. 2012, 27, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Potlog-Nahari, C.; Stratton, P.; Winkel, C.; Widra, E.; Sinaii, N.; Connors, S.; Nieman, L.K. Urine vascular endothelial growth factor-A is not a useful marker for endometriosis. Fertil. Steril. 2004, 81, 1507–1512. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Liu, H.; Sun, W.; Guo, Z.; Lang, J. Elevated urine histone 4 levels in women with ovarian endometriosis revealed by discovery and parallel reaction monitoring proteomics. J. Proteom. 2019, 204, 103398. [Google Scholar] [CrossRef]
- Gmyrek, G.B.; Sozanski, R.; Jerzak, M.; Chrobak, A.; Wickiewicz, D.; Skupnik, A.; Sierazka, U.; Fortuna, W.; Gabrys, M.; Chelmonska-Syta, A. Evaluation of monocyte chemotactic protein-1 levels in peripheral blood of infertile women with endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2005, 122, 199–205. [Google Scholar] [CrossRef]
- Gungor, T.; Kanat-Pektas, M.; Karayalcin, R.; Mollamahmutoglu, L. Peritoneal fluid and serum leptin concentrations in women with primary infertility. Arch. Gynecol. Obstet. 2009, 279, 361–364. [Google Scholar] [CrossRef]
- Huang, H.; Hong, H.; Tan, Y.; Sheng, J. Matrix metalloproteinase 2 is associated with changes in steroids hormones in the sera and peritoneal fluid of patients with endometriosis. Fertil. Steril. 2004, 81, 1235–1239. [Google Scholar] [CrossRef]
- Morin, M.; Bellehumeur, C.; Therriault, M.J.; Metz, C.; Maheux, R.; Akoum, A. Elevated levels of macrophage migration inhibitory factor in the peripheral blood of women with endometriosis. Fertil. Steril. 2005, 83, 865–872. [Google Scholar] [CrossRef]

| Author, Reference | Dysregulated miRNA | Specificity | Sensitivity |
|---|---|---|---|
| Ohlsson et al., 2009 [127] | miR-200a, miR-200b, and miR-141 | 66,7% | 84,4% |
| Jia et al., 2013 [134] | miR-22 miR-17-5p miR-20a | 90% 80% 90% | 90% 60% 60% |
| Wang et al., 2013 [129] | miR-145 miR-122 miR-199a miR-141-5p | 96% 76% 76% 96% | 70% 80% 78,3% 71,7% |
| Suryavanshi et al., 2013 [135] | miR-195, miR-16, miR-191 | 60% | 88% |
| Rekker et al., 2015 [131] | miR-200a-3p miR-200b-3p miR-141-3p | 70.8% 90.6% 70.8% | 71.9% 70.8% 71.9% |
| Cosar et al., 2016 [136] | miR-125b | 96% | 100% |
| Nisenblat et al., 2019 [132] | miR-155, miR574-3p and miR139-3p | 51% | 83% |
| Vanhie et al., 2019 [133] | hsa-miR-125b-5p, hsa-miR-28-5p and hsa-miR-29a-3p | 37% | 78% |
| Author, Reference | Source | Investigated Biomarkers |
|---|---|---|
| Agic et al, 2008 [62] | Serum | CA-125, CCR-1, miRNA, MCP-1 |
| Borrelli et al., 2014 [71] | Serum | IL-8, MCP-1, RANTES |
| Chen et al., 2012 [104] | Serum | Serum pigment epithelium-derived factor |
| Chen et al., 2019 [148] | Urine | Histone 4 |
| Chmaj-Wierzchowska et al., 2015 [99] | Serum | Urocortin, ghrelin, leptin |
| Cho et al., 2007 [79] | Serum, Urine | TNF-α, urinary sFlt-1 |
| Cho et al., 2012 [146] | Urine | Vitamin D-binding protein |
| Choi et al., 2019 [82] | Serum | IL-32, IL-6, IL-10, IL-1β, TNF-α, CA-125 |
| Cosar et al., 2016 [136] | Serum | miR-125b |
| Creed et al., 2019 [124] | Serum | mtDNA |
| Darai et al., 2003 [77] | Serum | IL-6, IL-8, TNF-α |
| Dutta et al., 2012 [91] | Serum | Lactate, 3-Hydroxybutyrate, Alanine, Glycerophosphatidylcholine, Valine, Leucine, Threonine, 2-Hydroxybutyrate, Lysine, Succinic acid |
| Gajbhiye et al., 2012 [106] | Serum | Autoantibodies against tropomodulin 3 (TMOD3), tropomyosin 3 (TPM3), stomatin-like protein 2 (SLP2) |
| Gmyrek et al., 2005 [149] | Serum | MCP-1 |
| Gungor et al., 2009 [150] | Serum | Leptin |
| Huang et al., 2004 [151] | Serum | MMP2 |
| Jia et al., 2013 [134] | Serum | miR-22 miR-17-5p miR-20a |
| Jing et al., 2008 [117] | Plasma | Proteomics-mass spectrometry |
| Kocbek et al., 2015 [66] | Serum | Glycodelin-A |
| Kuessel et al., 2014 [141] | Urine | CK19 |
| Liu et al., 2007 [139] | Plasma | Proteomics – mass spectrometry |
| Malin et al., 2019 [118] | Serum | AXIN1 |
| Martinez et al., 2007 [70] | Serum | IL-6 |
| May et al., 2010 [12] | Serum | FGF-2, angiogenin |
| Mihalyi et al., 2010 [61] | Serum | CA-125, IL-8, TNF-α |
| Morin et al., 2005 [152] | Peripheral blood | Macrophage migration inhibitory factor |
| Mosbah et al., 2016 [65] | Serum | ICAM-1, glycodelin |
| Nabeta et al., 2009 [108] | Serum | Anti-α-enolase antibodies |
| Nasiri et al., 2017 [87] | Serum | Malondialdehyde (MDA) |
| Nisenblat et al., 2019 [132] | Serum | miR-155, miR574-3p, and miR139-3p |
| Ohata et al., 2008 [76] | Seum | IL-8 |
| Ohlsson et al., 2009 [127] | Serum | miR-200a, miR-200b, and miR-141 |
| Othman et al., 2008 [70] | Serum | Il-6, MCP-1, IFN-γ IL-2, IL-8, IL-15 |
| Ozhan et al., 2014 [105] | Serum | Syntaxin-5, anti-endometrial antibody |
| Philippoussis et al., 2004 [95] | Serum | IGFBP3, EGF |
| Pizzo et al., 2002 [75] | Serum | IL-8 |
| Potlog-Nahar.i, 2004 [147] | Urine | VEGF |
| Qui et al., 2019 [139] | Serum | lncRNA |
| Reis et al., 2012 [68] | Serum | Activin A, follistatin |
| Rekker et al., 2015 [125] | Serum | miR-200a-3p miR-200b-3p miR-141-3p |
| Seeber et al., 2008 [80] | Serum | TNF-α |
| Sengul et al., 2014 [97] | Serum | Nesfatin-1 |
| Socolov et al., 2010 [73] | Serum | IL-6, IL-8, IL-1, CA-125, TNF |
| Steff et al., 2004 [94] | Serum | IGF-1, IGFBP3 |
| Suryavanshi et al., 2013 [135] | Serum | miR-195, miR-16, miR-191 |
| Tokmak et al., 2011 [98] | Serum | Urocortin |
| Tokushige et al., 2011 [144] | Urine | CK19 |
| Becker et al., 2010 [143] | Urine | MMP-2, MMP-9 |
| Tuten et al., 2014 [64] | Serum | Copeptin, CA-125, C-reactive protein, CA-15-3, CA-19-9 |
| Vanhie et al., 2019 [133] | Serum | hsa-miR-125b-5p, hsa-miR-28-5p and hsa-miR-29a-3p |
| Vigano et al., 2003 [69] | Serum | ICAM-1 |
| Vodolazkaia et al., 2012 [63] | Serum | VEGF, CA-125, Annexin V, glycodelin |
| Wang et al., 2013 [129] | Serum | miR-145 miR-122 miR-199a miR-141-5p |
| Wang et al., 2016 [138] | Serum | lncRNA |
| Xavier et al., 2006 [78] | Serum | TNF-α, VEGF |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anastasiu, C.V.; Moga, M.A.; Elena Neculau, A.; Bălan, A.; Scârneciu, I.; Dragomir, R.M.; Dull, A.-M.; Chicea, L.-M. Biomarkers for the Noninvasive Diagnosis of Endometriosis: State of the Art and Future Perspectives. Int. J. Mol. Sci. 2020, 21, 1750. https://doi.org/10.3390/ijms21051750
Anastasiu CV, Moga MA, Elena Neculau A, Bălan A, Scârneciu I, Dragomir RM, Dull A-M, Chicea L-M. Biomarkers for the Noninvasive Diagnosis of Endometriosis: State of the Art and Future Perspectives. International Journal of Molecular Sciences. 2020; 21(5):1750. https://doi.org/10.3390/ijms21051750
Chicago/Turabian StyleAnastasiu, Costin Vlad, Marius Alexandru Moga, Andrea Elena Neculau, Andreea Bălan, Ioan Scârneciu, Roxana Maria Dragomir, Ana-Maria Dull, and Liana-Maria Chicea. 2020. "Biomarkers for the Noninvasive Diagnosis of Endometriosis: State of the Art and Future Perspectives" International Journal of Molecular Sciences 21, no. 5: 1750. https://doi.org/10.3390/ijms21051750
APA StyleAnastasiu, C. V., Moga, M. A., Elena Neculau, A., Bălan, A., Scârneciu, I., Dragomir, R. M., Dull, A.-M., & Chicea, L.-M. (2020). Biomarkers for the Noninvasive Diagnosis of Endometriosis: State of the Art and Future Perspectives. International Journal of Molecular Sciences, 21(5), 1750. https://doi.org/10.3390/ijms21051750







