Transient Receptor Potential Canonical (TRPC) Channels as Modulators of Migration and Invasion
Abstract
1. Introduction
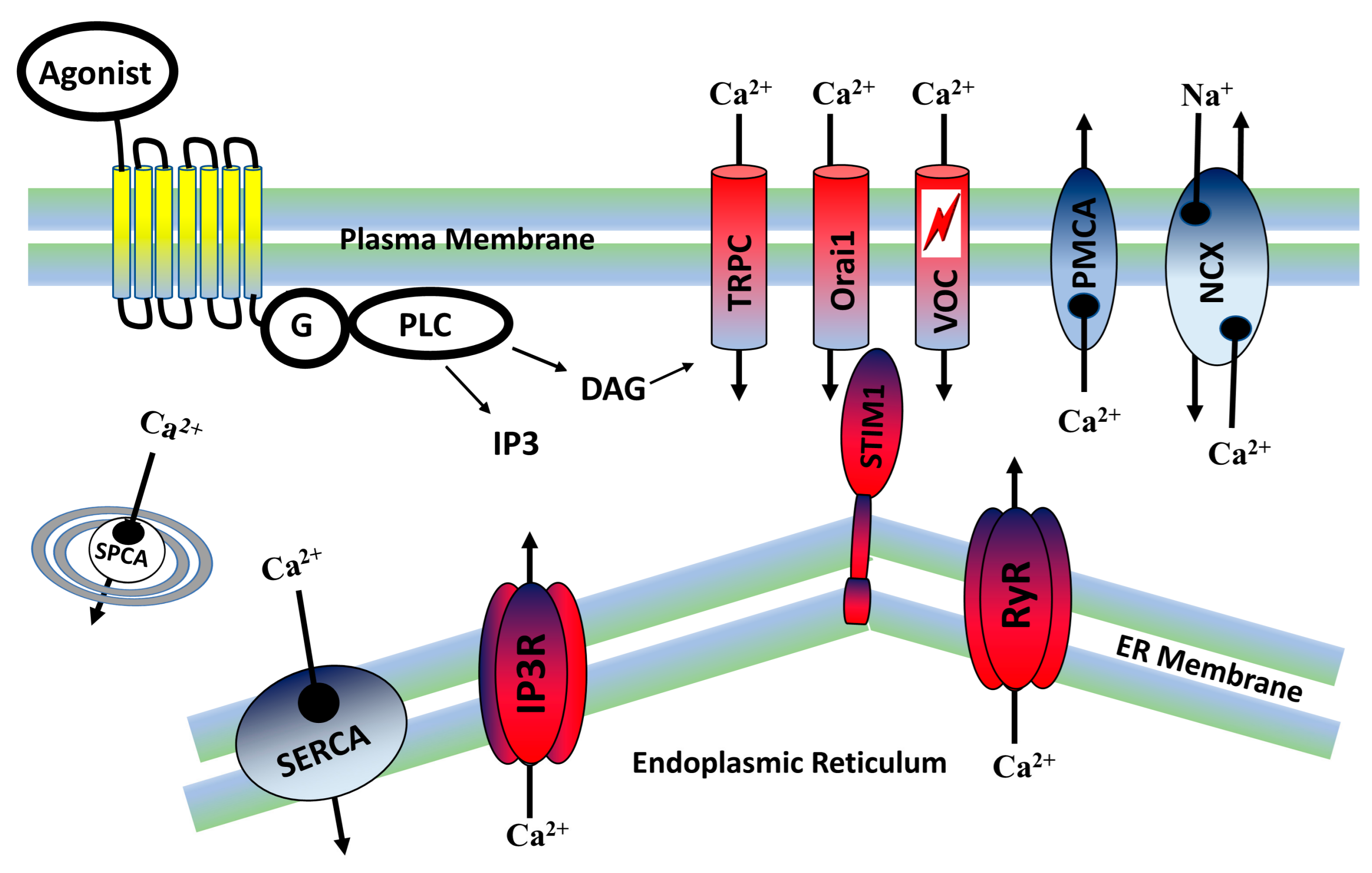
2. Calcium Signaling
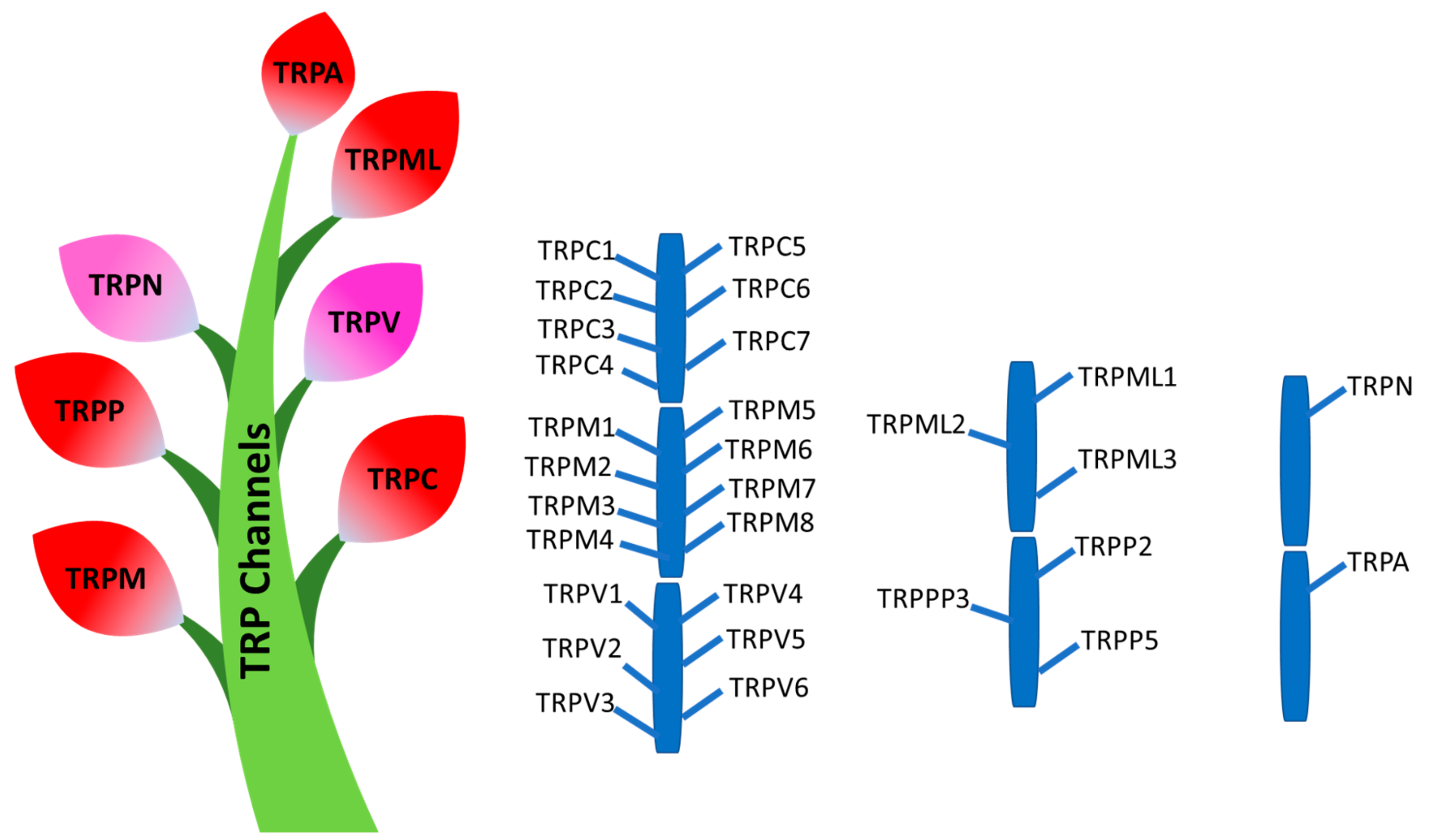
3. Transient Receptor Potential (TRP) Channels
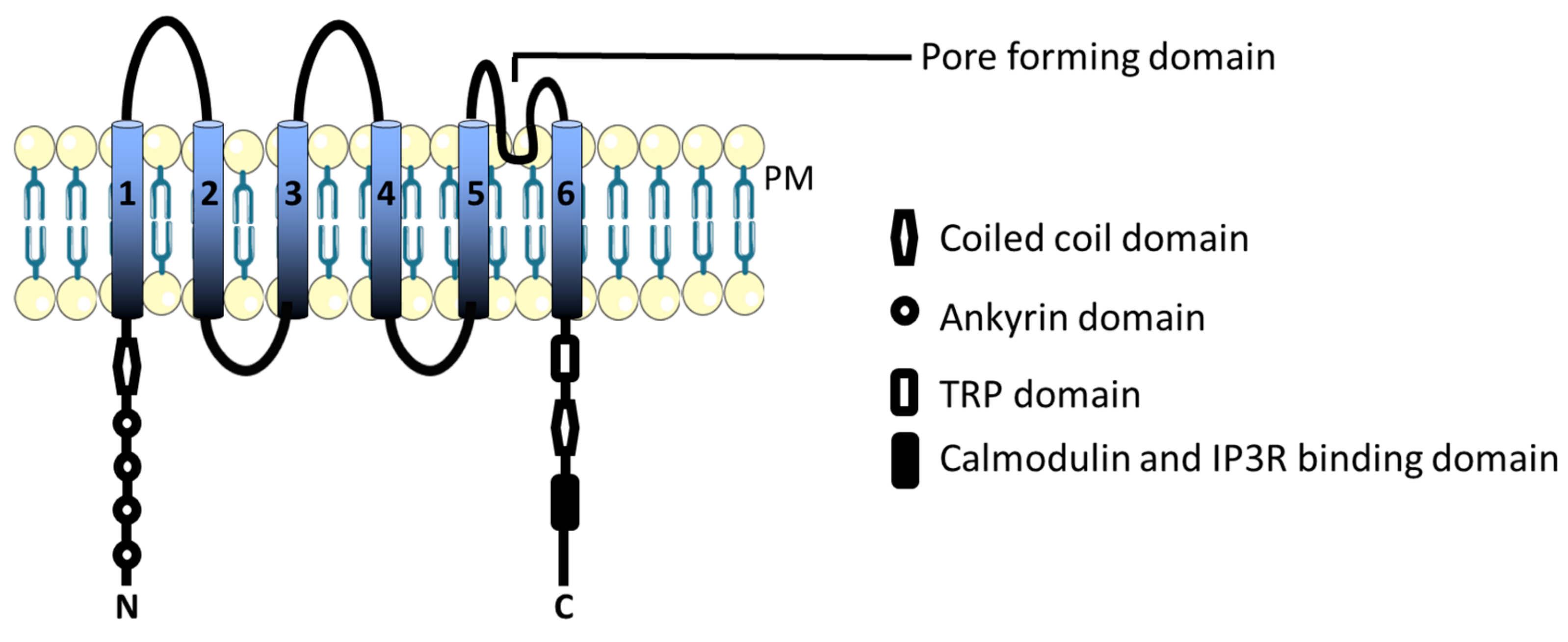
3.1. TRPC Channels
3.1.1. TRPC1
3.1.2. TRPC2
3.1.3. TRPC3
3.1.4. TRPC4
3.1.5. TRPC5
3.1.6. TRPC6
3.1.7. TRPC7
4. TRPC Channels as Regulators of Migration and Invasion
4.1. TRPC Channels, Migration and Invasion
4.2. TRPC Channels and Angiogenesis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Prevarskaya, N.; Skryma, R.; Shuba, Y. Ion channels and the hallmarks of cancer. Trends Mol. Med. 2010, 16, 107–121. [Google Scholar] [CrossRef] [PubMed]
- Monteith, G.R.; Prevarskaya, N.; Roberts-Thomson, S.J. The calcium-cancer signalling nexus. Nat. Rev. Cancer 2017, 17, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Prevarskaya, N.; Skryma, R.; Shuba, Y. Ion Channels in Cancer: Are Cancer Hallmarks Oncochannelopathies? Physiol. Rev. 2018, 98, 559–621. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J.; Lipp, P.; Bootman, M.D. The versatility and universality of calcium signalling. Nat. Rev. Mol. Cell Biol. 2000, 1, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J.; Bootman, M.D.; Roderick, H.L. Calcium signalling: Dynamics, homeostasis and remodelling. Nat. Rev. Mol. Cell Biol. 2003, 4, 517–529. [Google Scholar] [CrossRef]
- Brini, M.; Carafoli, E. Calcium pumps in health and disease. Physiol. Rev. 2009, 89, 1341–1378. [Google Scholar] [CrossRef]
- Goodman, M.B.; Schwarz, E.M. Transducing Touch in Caenorhabditis elegans. Annu. Rev. Physiol. 2003, 65, 429–452. [Google Scholar] [CrossRef]
- Liu, X.; Cheng, K.T.; Bandyopadhyay, B.C.; Pani, B.; Dietrich, A.; Paria, B.C.; Swaim, W.D.; Beech, D.; Yildrim, E.; Singh, B.B.; et al. Attenuation of store-operated Ca2+ current impairs salivary gland fluid secretion in TRPC1(-/-) mice. Proc. Natl. Acad. Sci. USA 2007, 104, 17542–17547. [Google Scholar] [CrossRef]
- Montell, C. The TRP superfamily of cation channels: Science’s STKE: Signal transduction knowledge environment. Science 2005, 2005, re3. [Google Scholar] [CrossRef]
- Pedersen, S.F.; Owsianik, G.; Nilius, B. TRP channels: An overview. Cell Calcium 2005, 38, 233–252. [Google Scholar] [CrossRef]
- Louis, M.; Zanou, N.; Van Schoor, M.; Gailly, P. TRPC1 regulates skeletal myoblast migration and differentiation. J. Cell Sci. 2008, 121, 3951–3959. [Google Scholar] [CrossRef] [PubMed]
- Nilius, B.; Owsianik, G. The transient receptor potential family of ion channels. Genome Biol. 2011, 12, 218. [Google Scholar] [CrossRef] [PubMed]
- Riccio, A.; Medhurst, A.D.; Mattei, C.; Kelsell, R.E.; Calver, A.R.; Randall, A.D.; Benham, C.D.; Pangalos, M.N. mRNA distribution analysis of human TRPC family in CNS and peripheral tissues. Mol. Brain Res. 2002, 109, 95–104. [Google Scholar] [CrossRef]
- Abramowitz, J.; Birnbaumer, L. Physiology and pathophysiology of canonical transient receptor potential channels. FASEB J. 2009, 23, 297–328. [Google Scholar] [CrossRef]
- Clapham, D.E. Calcium Signaling. Cell 2007, 131, 1047–1058. [Google Scholar] [CrossRef]
- Pinto, M.C.X.; Kihara, A.H.; Goulart, V.A.M.; Tonelli, F.M.P.; Gomes, K.N.; Ulrich, H.; Resende, R.R. Calcium signaling and cell proliferation. Cell. Signal. 2015, 27, 2139–2149. [Google Scholar] [CrossRef]
- Venkatachalam, K.; Zheng, F.; Gill, D.L. Regulation of canonical transient receptor potential (TRPC) channel function by diacylglycerol and protein kinase C. J. Biol. Chem. 2003, 278, 29031–29040. [Google Scholar] [CrossRef]
- Montell, C.; Birnbaumer, L.; Flockerzi, V. The TRP channels, a remarkably functional family. Cell 2002, 108, 595–598. [Google Scholar] [CrossRef]
- Venkatachalam, K.; Montell, C. TRP channels. Annu. Rev. Biochem. 2007, 76, 387–417. [Google Scholar] [CrossRef]
- Berridge, M.J. The Inositol Trisphosphate/Calcium Signaling Pathway in Health and Disease. Physiol. Rev. 2016, 96, 1261–1296. [Google Scholar] [CrossRef]
- Stutzmann, G.E.; Mattson, M.P. Endoplasmic reticulum Ca(2+) handling in excitable cells in health and disease. Pharmacol. Rev. 2011, 63, 700–727. [Google Scholar] [CrossRef] [PubMed]
- Cosens, D.J.; Manning, A. Abnormal electroretinogram from a Drosophila mutant. Nature 1969, 224, 285–287. [Google Scholar] [CrossRef] [PubMed]
- Minke, B.; Wu, C.-F.; Pak, W.L. Induction of photoreceptor voltage noise in the dark in Drosophila mutant. Nature 1975, 258, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Montell, C.; Rubin, G.M. Molecular characterization of the drosophila trp locus: A putative integral membrane protein required for phototransduction. Neuron 1989, 2, 1313–1323. [Google Scholar] [CrossRef]
- Clapham, D.E. TRP channels as cellular sensors. Nature 2003, 426, 517–524. [Google Scholar] [CrossRef]
- Julius, D. TRP channels and pain. Annu. Rev. Cell Dev. Biol. 2013, 29, 355–384. [Google Scholar] [CrossRef]
- Montell, C. Physiology, Phylogeny, and Functions of the TRP Superfamily of Cation Channels. Sci. Signal. 2001, 2001, re1. [Google Scholar] [CrossRef]
- Minke, B.; Cook, B. TRP channel proteins and signal transduction. Physiol. Rev. 2002, 82, 429–472. [Google Scholar] [CrossRef]
- Minke, B. The history of the Drosophila TRP channel: the birth of a new channel superfamily. J. Neurogenet. 2010, 24, 216–233. [Google Scholar] [CrossRef]
- Nilius, B.; Mahieu, F. A Road Map for TR(I)Ps. Mol. Cell 2006, 22, 297–307. [Google Scholar] [CrossRef]
- Benemei, S.; Patacchini, R.; Trevisani, M.; Geppetti, P. TRP channels. Curr. Opin. Pharmacol. 2015, 22, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Nilius, B.; Flockerzi, V. Mammalian Transient Receptor Potential (TRP) Cation Channels. Handb. Exp. Pharmacol. 2014, 223, 795–826. [Google Scholar] [CrossRef]
- Kaneko, Y.; Szallasi, A. Transient receptor potential (TRP) channels: A clinical perspective. Br. J. Pharmacol. 2014, 171, 2474–2507. [Google Scholar] [CrossRef]
- Gees, M.; Owsianik, G.; Voets, T. TRP Channels. Compr. Physiol. 2012, 2, 563–608. [Google Scholar] [CrossRef]
- Nilius, B. TRP channels in disease. Biochim. Biophys. Acta Mol. Basis Dis. 2007, 1772, 805–812. [Google Scholar] [CrossRef]
- Prevarskaya, N.; Zhang, L.; Barritt, G. TRP channels in cancer. Biochim. Biophys. Acta Mol. Basis Dis. 2007, 1772, 937–946. [Google Scholar] [CrossRef]
- Van Haute, C.; De Ridder, D.; Nilius, B. TRP channels in human prostate. Sci. World J. 2010, 10, 1597–1611. [Google Scholar] [CrossRef]
- Lehen’Kyi, V.; Prevarskaya, N. Oncogenic TRP channels. Adv. Exp. Med. Biol. 2010, 704, 929–945. [Google Scholar] [CrossRef]
- Ouadid-Ahidouch, H.; Dhennin-Duthille, I.; Gautier, M.; Sevestre, H.; Ahidouch, A. TRP channels: Diagnostic markers and therapeutic targets for breast cancer? Trends Mol. Med. 2013, 19, 117–124. [Google Scholar] [CrossRef]
- Lepage, P.K.; Lussier, M.P.; Barajas-Martinez, H.; Bousquet, S.M.; Blanchard, A.P.; Francoeur, N.; Dumaine, R.; Boulay, G. Identification of two domains involved in the assembly of transient receptor potential canonical channels. J. Biol. Chem. 2006, 281, 30356–30364. [Google Scholar] [CrossRef]
- Birnbaumer, L. The TRPC class of ion channels: a critical review of their roles in slow, sustained increases in intracellular Ca(2+) concentrations. Annu. Rev. Pharmacol. Toxicol. 2009, 49, 395–426. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Zagranichnaya, T.K.; Gurda, G.T.; Eves, E.M.; Villereal, M.L. A TRPC1/TRPC3-mediated increase in store-operated calcium entry is required for differentiation of H19-7 hippocampal neuronal cells. J. Biol. Chem. 2004, 279, 43392–43402. [Google Scholar] [CrossRef]
- Kuang, C.; Yu, Y.; Wang, K.; Qian, D.; Den, M.; Huang, L. Knockdown of Transient Receptor Potential Canonical-1 Reduces the Proliferation and Migration of Endothelial Progenitor Cells. Stem. Cells Dev. 2012, 21, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Wes, P.D.; Chevesich, J.; Jeromin, A.; Rosenberg, C.; Stetten, G.; Montell, C. TRPC1, a human homolog of a Drosophila store-operated channel. Proc. Natl. Acad. Sci. USA 1995, 92, 9652–9656. [Google Scholar] [CrossRef]
- Parekh, A.B.; Putney, J.W. Store-operated calcium channels. Physiol. Rev. 2005, 85, 757–810. [Google Scholar] [CrossRef] [PubMed]
- Beech, D.J. TRPC1: Store-operated channel and more. Pflug. Arch. Eur. J. Physiol. 2005, 451, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Ambudkar, I.S. TRPC1: a core component of store-operated calcium channels. Biochem. Soc. Trans. 2007, 35, 96–100. [Google Scholar] [CrossRef]
- Storch, U.; Forst, A.L.; Philipp, M.; Gudermann, T.; Mederos, Y.; Schnitzler, M. Transient receptor potential channel 1 (TRPC1) reduces calcium permeability in heteromeric channel complexes. J. Biol. Chem. 2012, 287, 3530–3540. [Google Scholar] [CrossRef]
- Nesin, V.; Tsiokas, L. TRPC1. Handb. Exp. Pharmacol. 2014, 222, 15–51. [Google Scholar] [CrossRef]
- Liu, X.; Bandyopadhyay, B.C.; Singh, B.B.; Groschner, K.; Ambudkar, I.S. Molecular analysis of a store-operated and OAG sensitive non-selective cation channel: Heteromeric assembly of TRPC1-TRPC3. J. Biol. Chem. 2005, 280, 21600–21606. [Google Scholar] [CrossRef]
- Rao, J.N.; Platoshyn, O.; Golovina, V.; Liu, L.; Zou, T.; Marasa, B.S.; Turner, D.J.; Yuan, J.X.J.; Wang, J.Y. TRPC1 functions as a store-operated Ca2+ channel in intestinal epithelial cells and regulates early mucosal restitution after wounding. Am. J. Physiol. Gastrointest Liver Physiol. 2006, 290, G782–G792. [Google Scholar] [CrossRef]
- Bomben, V.C.; Sontheimer, H.W. Inhibition of transient receptor potential canonical channels impairs cytokinesis in human malignant gliomas. Cell Prolif. 2008, 41, 98–121. [Google Scholar] [CrossRef]
- Dhennin-Duthille, I.; Gautier, M.; Faouzi, M.; Guilbert, A.; Brevet, M.; Vaudry, D.; Ahidouch, A.; Sevestre, H.; Ouadid-Ahidouch, H. High expression of transient receptor potential channels in human breast cancer epithelial cells and tissues: Correlation with pathological parameters. Cell. Physiol. Biochem. 2011, 28, 813–822. [Google Scholar] [CrossRef]
- Antigny, F.; Sabourin, J.; Saüc, S.; Bernheim, L.; Koenig, S.; Frieden, M. TRPC1 and TRPC4 channels functionally interact with STIM1L to promote myogenesis and maintain fast repetitive Ca2??+ release in human myotubes. Biochim. Biophys. Acta Mol. Cell Res. 2017, 1864, 806–813. [Google Scholar] [CrossRef]
- Wissenbach, U.; Schroth, G.; Philipp, S.; Flockerzi, V. Structure and mRNA expression of a bovine trp homologue related to mammalian trp2 transcripts. FEBS Lett. 1998, 429, 61–66. [Google Scholar] [CrossRef]
- Vannier, B.; Peyton, M.; Boulay, G.; Brown, D.; Qin, N.; Jiang, M.; Zhu, X.; Birnbaumer, L. Mouse trp2, the homologue of the human trpc2 pseudogene, encodes mTrp2, a store depletion-activated capacitative Ca2+ entry channel. Proc. Natl. Acad. Sci. USA 1999, 96, 2060–2064. [Google Scholar] [CrossRef]
- Zufall, F. TRPs in olfaction. Handb. Exp. Pharmacol. 2014, 223, 917–933. [Google Scholar] [CrossRef]
- Miller, B.A. TRPC2. Handb. Exp. Pharmacol. 2014, 222, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.P.; Kiselyov, K.; Shin, D.M.; Chen, J.; Shcheynikov, N.; Kang, S.H.; Dehoff, M.H.; Schwarz, M.K.; Seeburg, P.H.; Muallem, S.; et al. Homer binds TRPC family channels and is required for gating of TRPC1 by IP3 receptors. Cell 2003, 114, 777–789. [Google Scholar] [CrossRef]
- Lockwich, T.P.; Liu, X.; Singh, B.B.; Jadlowiec, J.; Weiland, S.; Ambudkar, I.S. Assembly of Trp1 in a signaling complex associated with caveolin-scaffolding lipid raft domains. J. Biol. Chem. 2000, 275, 11934–11942. [Google Scholar] [CrossRef] [PubMed]
- Mast, T.G.; Brann, J.H.; Fadool, D.A. The TRPC2 channel forms protein-protein interactions with Homer and RTP in the rat vomeronasal organ. BMC Neurosci. 2010, 11, 61. [Google Scholar] [CrossRef] [PubMed]
- Sukumaran, P.; Löf, C.; Pulli, I.; Kemppainen, K.; Viitanen, T.; Törnquist, K. Significance of the transient receptor potential canonical 2 (TRPC2) channel in the regulation of rat thyroid FRTL-5 cell proliferation, migration, adhesion and invasion. Mol. Cell Endocrinol. 2013, 374, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Törnquist, K.; Sukumaran, P.; Kemppainen, K.; Löf, C.; Viitanen, T. Canonical transient receptor potential channel 2 (TRPC2): old name-new games. Importance in regulating of rat thyroid cell physiology. Pflug. Arch. Eur. J. Physiol. 2014, 466, 2025–2034. [Google Scholar] [CrossRef] [PubMed]
- Putney, J.W. Physiological mechanisms of TRPC activation. Pflug. Arch. Eur. J. Physiol. 2005, 451, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Owsianik, G.; Talavera, K.; Voets, T.; Nilius, B. Permeation and selectivity of trp channels. Annu. Rev. Physiol. 2006, 68, 685–717. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Plummer, N.W.; George, M.D.; Abramowitz, J.; Zhu, M.X.; Birnbaumer, L. A role for Orai in TRPC-mediated Ca2+ entry suggests that a TRPC:Orai complex may mediate store and receptor operated Ca2+ entry. Proc. Natl. Acad. Sci. USA 2009, 106, 3202–3206. [Google Scholar] [CrossRef]
- Woodard, G.E.; López, J.J.; Jardín, I.; Salido, G.M.; Rosado, J.A. TRPC3 regulates agonist-stimulated Ca2+ mobilization by mediating the interaction between type I inositol 1,4,5-trisphosphate receptor, RACK1, and Orai1. J. Biol. Chem. 2010, 285, 8045–8053. [Google Scholar] [CrossRef]
- Trebak, M.; Bird, G.S.J.; McKay, R.R.; Birnbaumer, L.; Putney, J.W. Signaling mechanism for receptor-activated canonical transient receptor potential 3 (TRPC3) channels. J. Biol. Chem. 2003, 278, 16244–16252. [Google Scholar] [CrossRef]
- Yuan, J.P.; Zeng, W.; Huang, G.N.; Worley, P.F.; Muallem, S. STIM1 heteromultimerizes TRPC channels to determine their function as store-operated channels. Nat. Cell Biol. 2007, 9, 636–645. [Google Scholar] [CrossRef]
- Qin, W.; Xie, W.; Xia, N.; He, Q.; Sun, T. Silencing of transient receptor potential channel 4 alleviates oxLDL-induced angiogenesis in human coronary artery endothelial cells by inhibition of VEGF and NF-κB. Med. Sci. Monit. 2016, 22, 930–936. [Google Scholar] [CrossRef]
- Schaefer, M.; Plant, T.D.; Obukhov, A.G.; Hofmann, T.; Gudermann, T.; Schultz, G. Receptor-mediated regulation of the nonselective cation channels TRPC4 and TRPC5. J. Biol. Chem. 2000, 275, 17517–17526. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.; Wong, C.O.; Lau, O.C.; Woo, T.; Bai, S.; Huang, Y.; Yao, X. Plasma membrane mechanical stress activates TRPC5 channels. PLoS ONE 2015, 10, e0122227. [Google Scholar] [CrossRef]
- Earley, S.; Brayden, J.E. Transient receptor potential channels in the vasculature. Physiol. Rev. 2015, 95, 645–690. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, A.; Mederos YSchnitzler, M.; Gollasch, M.; Gross, V.; Storch, U.; Dubrovska, G.; Obst, M.; Yildirim, E.; Salanova, B.; Kalwa, H.; et al. Increased vascular smooth muscle contractility in TRPC6-/- mice. Mol. Cell Biol. 2005, 25, 6980–6989. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Erxleben, C.; Yildirim, E.; Abramowitz, J.; Armstrong, D.L.; Birnbaumer, L. Orai proteins interact with TRPC channels and confer responsiveness to store depletion. Proc. Natl. Acad. Sci. USA 2007, 104, 4682–4687. [Google Scholar] [CrossRef]
- Shirakawa, H.; Katsumoto, R.; Iida, S.; Miyake, T.; Higuchi, T.; Nagashima, T.; Nagayasu, K.; Nakagawa, T.; Kaneko, S. Sphingosine-1-phosphate induces Ca2+ signaling and CXCL1 release via TRPC6 channel in astrocytes. Glia 2017, 65, 1005–1016. [Google Scholar] [CrossRef]
- Park, S.; Lee, S.; Park, E.J.; Kang, M.J.; So, I.; Jeon, J.H.; Chun, J.N. TGFβ1 induces stress fiber formation through upregulation of TRPC6 in vascular smooth muscle cells. Biochem. Biophys. Res. Commun. 2017, 483, 129–134. [Google Scholar] [CrossRef]
- Weber, E.W.; Han, F.; Tauseef, M.; Birnbaumer, L.; Mehta, D.; Muller, W.A. TRPC6 is the endothelial calcium channel that regulates leukocyte transendothelial migration during the inflammatory response. J. Exp. Med. 2015, 212, 1883–1899. [Google Scholar] [CrossRef]
- Nagamine, K.; Kudoh, J.; Minoshima, S.; Kawasaki, K.; Asakawa, S.; Ito, F.; Shimizu, N. Molecular cloning of a novel putative Ca2+ channel protein (TRPC7) highly expressed in brain. Genomics 1998, 54, 124–131. [Google Scholar] [CrossRef]
- Okada, T.; Inoue, R.; Yamazaki, K.; Maeda, A.; Kurosaki, T.; Yamakuni, T.; Tanaka, I.; Shimizu, S.; Ikenaka, K.; Imoto, K.; et al. Molecular and functional characterization of a novel mouse transient receptor potential protein homologue TRP7. J. Biol. Chem. 1999, 274, 27359–27370. [Google Scholar] [CrossRef]
- Zagranichnaya, T.K.; Wu, X.; Villereal, M.L. Endogenous TRPC1, TRPC3, and TRPC7 proteins combine to form native store-operated channels in HEK-293 cells. J. Biol. Chem. 2005, 280, 29559–29569. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, K.; Matsuda, T.; Tsuji, A. Functional regulation of transient receptor potential canonical 7 by cGMP-dependent protein kinase Iα. Cell. Signal. 2011, 23, 1179–1187. [Google Scholar] [CrossRef] [PubMed]
- Satoh, S.; Tanaka, H.; Ueda, Y.; Oyama, J.I.; Sugano, M.; Sumimoto, H.; Mori, Y.; Makino, N. Transient receptor potential (TRP) protein 7 acts as a G protein-activated Ca2+ channel mediating angiotensin II-induced myocardial apoptosis. Mol. Cell. Biochem. 2007, 294, 205–215. [Google Scholar] [CrossRef]
- Schwab, A.; Fabian, A.; Hanley, P.J.; Stock, C. Role of Ion Channels and Transporters in Cell Migration. Physiol. Rev. 2012, 92, 1865–1913. [Google Scholar] [CrossRef]
- Déliot, N.; Constantin, B. Plasma membrane calcium channels in cancer: Alterations and consequences for cell proliferation and migration. Biochim. Biophys. Acta Biomembr. 2015, 1848, 2512–2522. [Google Scholar] [CrossRef] [PubMed]
- Vrenken, K.S.; Jalink, K.; van Leeuwen, F.N.; Middelbeek, J. Beyond ion-conduction: Channel-dependent and -independent roles of TRP channels during development and tissue homeostasis. Biochim. Biophys. Acta Mol. Cell Res. 2016, 1863, 1436–1446. [Google Scholar] [CrossRef] [PubMed]
- Fabian, A.; Fortmann, T.; Dieterich, P.; Riethmüller, C.; Schön, P.; Mally, S.; Nilius, B.; Schwab, A. TRPC1 channels regulate directionality of migrating cells. Pflug. Arch. Eur. J. Physiol. 2008, 457, 475–484. [Google Scholar] [CrossRef]
- Bomben, V.C.; Turner, K.L.; Barclay, T.T.C.; Sontheimer, H. Transient receptor potential canonical channels are essential for chemotactic migration of human malignant gliomas. J. Cell Physiol. 2011, 226, 1879–1888. [Google Scholar] [CrossRef]
- Guéguinou, M.; Harnois, T.; Crottes, D.; Uguen, A.; Deliot, N.; Gambade, A.; Chantôme, A.; Haelters, J.P.; Jaffrès, P.A.; Jourdan, M.L.; et al. SK3/TRPC1/Orai1 complex regulates SOCE-dependent colon cancer cell migration: A novel opportunity to modulate anti- EGFR mAb action by the alkyl-lipid Ohmline. Oncotarget 2016, 7, 36168–36184. [Google Scholar] [CrossRef]
- He, B.; Liu, F.; Ruan, J.; Li, A.; Chen, J.; Li, R.; Shen, J.; Zheng, D.; Luo, R. Silencing TRPC1 expression inhibits invasion of CNE2 nasopharyngeal tumor cells. Oncol. Rep. 2012, 27. [Google Scholar] [CrossRef]
- Asghar, M.Y.; Magnusson, M.; Kemppainen, K.; Sukumaran, P.; Löf, C.; Pulli, I.; Kalhori, V.; Törnquist, K. Transient receptor potential canonical 1 (TRPC1) channels as regulators of sphingolipid and VEGF receptor expression: Implications for thyroid cancer cell migration and proliferation. J. Biol. Chem. 2015, 290, 16116–16131. [Google Scholar] [CrossRef] [PubMed]
- Törnquist, K. Sphingosine 1-phosphate, sphingosine kinase and autocrine calcium signalling in thyroid cells. Acta Physiol. 2012, 204, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Wildi-Runge, S.; Stoppa-Vaucher, S.; Lambert, R.; Turpin, S.; Van Vliet, G.; Deladoëy, J. A high prevalence of dual thyroid ectopy in congenital hypothyroidism: Evidence for insufficient signaling gradients during embryonic thyroid migration or for the polyclonal nature of the thyroid gland? J. Clin. Endocrinol. Metab. 2012, 97, E978–E981. [Google Scholar] [CrossRef] [PubMed]
- Kimchi, T.; Xu, J.; Dulac, C. A functional circuit underlying male sexual behaviour in the female mouse brain. Nature 2007, 448, 1009–1014. [Google Scholar] [CrossRef]
- Yang, S.L.; Cao, Q.; Zhou, K.C.; Feng, Y.J.; Wang, Y.Z. Transient receptor potential channel C3 contributes to the progression of human ovarian cancer. Oncogene 2009, 28, 1320–1328. [Google Scholar] [CrossRef]
- Storck, H.; Hild, B.; Schimmelpfennig, S.; Sargin, S.; Nielsen, N.; Zaccagnino, A.; Budde, T.; Novak, I.; Kalthoff, H.; Schwab, A. Ion channels in control of pancreatic stellate cell migration. Oncotarget 2016, 8, 769–784. [Google Scholar] [CrossRef]
- Oda, K.; Umemura, M.; Nakakaji, R.; Tanaka, R.; Sato, I.; Nagasako, A.; Oyamada, C.; Baljinnyam, E.; Katsumata, M.; Xie, L.H.; et al. Transient receptor potential cation 3 channel regulates melanoma proliferation and migration. J. Physiol. Sci. 2016, 67, 497–505. [Google Scholar] [CrossRef]
- Turunen, P.M.; Louhivuori, L.M.; Louhivuori, V.; Kukkonen, J.P.; Åkerman, K.E. Endocannabinoid Signaling in Embryonic Neuronal Motility and Cell–Cell Contact—Role of mGluR5 and TRPC3 Channels. Neuroscience 2018, 375, 135–148. [Google Scholar] [CrossRef]
- Kim, J.M.; Heo, K.; Choi, J.; Kim, K.; An, W. The histone variant MacroH2A regulates Ca2+ influx through TRPC3 and TRPC6 channels. Oncogenesis 2013, 2, e77. [Google Scholar] [CrossRef]
- Zeng, B.; Yuan, C.; Yang, X.; LAtkin, S.; Xu, S.-Z. TRPC Channels and Their Splice Variants are Essential for Promoting Human Ovarian Cancer Cell Proliferation and Tumorigenesis. Curr. Cancer Drug Targets 2012, 13, 103–116. [Google Scholar] [CrossRef]
- Wei, W.C.; Huang, W.C.; Lin, Y.P.; Becker, E.B.E.; Ansorge, O.; Flockerzi, V.; Conti, D.; Cenacchi, G.; Glitsch, M.D. Functional expression of calcium-permeable canonical transient receptor potential 4-containing channels promotes migration of medulloblastoma cells. J. Physiol. 2017, 595, 5525–5544. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.N.; Zeng, B.; Zhang, Y.; Daskoulidou, N.; Fan, H.; Qu, J.M.; Xu, S.Z. Involvement of TRPC Channels in Lung Cancer Cell Differentiation and the Correlation Analysis in Human Non-Small Cell Lung Cancer. PLoS ONE 2013, 8, e67637. [Google Scholar] [CrossRef] [PubMed]
- Greka, A.; Navarro, B.; Oancea, E.; Duggan, A.; Clapham, D.E. TRPC5 is a regulator of hippocampal neurite length and growth cone morphology. Nat. Neurosci. 2003, 6, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Tian, D.; Jacobo, S.M.P.; Billing, D.; Rozkalne, A.; Gage, S.D.; Anagnostou, T.; Pavenstaedt, H.; Hsu, H.H.; Schlondorff, J.; Ramos, A.; et al. Antagonistic regulation of actin dynamics and cell motility by TRPC5 and TRPC6 channels. Sci. Signal. 2010, 3, ra77. [Google Scholar] [CrossRef]
- Carson, C.; Raman, P.; Tullai, J.; Xu, L.; Henault, M.; Thomas, E.; Yeola, S.; Lao, J.; McPate, M.; Martin Verkuyl, J.; et al. Englerin a agonizes the TRPC4/C5 cation channels to inhibit tumor cell line proliferation. PLoS ONE 2015, 10, e0127498. [Google Scholar] [CrossRef]
- Muraki, K.; Ohnishi, K.; Takezawa, A.; Suzuki, H.; Hatano, N.; Muraki, Y.; Hamzah, N.; Foster, R.; Waldmann, H.; Nussbaumer, P.; et al. Na+ entry through heteromeric TRPC4/C1 channels mediates (-)Englerin A-induced cytotoxicity in synovial sarcoma cells. Sci. Rep. 2017, 7, 16988. [Google Scholar] [CrossRef]
- Bernaldo de Quirós, S.; Merlo, A.; Secades, P.; Zambrano, I.; de Santa María, I.S.; Ugidos, N.; Jantus-Lewintre, E.; Sirera, R.; Suarez, C.; Chiara, M.D. Identification of TRPC6 as a possible candidate target gene within an amplicon at 11q21-q22.2 for migratory capacity in head and neck squamous cell carcinomas. BMC Cancer 2013, 13, 116. [Google Scholar] [CrossRef]
- Chigurupati, S.; Venkataraman, R.; Barrera, D. Receptor channel TRPC6 is a key mediator of Notch-driven glioblastoma growth and invasiveness. Cancer Res. 2010, 70, 418–427. [Google Scholar] [CrossRef]
- Thebault, S.; Flourakis, M.; Vanoverberghe, K.; Vandermoere, F.; Roudbaraki, M.; Lehen’kyi, V.; Slomianny, C.; Beck, B.; Mariot, P.; Bonnal, J.L.; et al. Differential role of transient receptor potential channels in Ca2+ entry and proliferation of prostate cancer epithelial cells. Cancer Res. 2006, 66, 2038–2047. [Google Scholar] [CrossRef]
- Yang, L.L.; Liu, B.C.; Lu, X.Y.; Yan, Y.; Zhai, Y.J.; Bao, Q.; Doetsch, P.W.; Deng, X.; Thai, T.L.; Alli, A.A.; et al. Inhibition of TRPC6 reduces non-small cell lung cancer cell proliferation and invasion. Oncotarget 2016, 8, 5123–5134. [Google Scholar] [CrossRef]
- Diez-Bello, R.; Jardin, I.; Lopez, J.J.; El Haouari, M.; Ortega-Vidal, J.; Altarejos, J.; Salido, G.M.; Salido, S.; Rosado, J.A. (−)-Oleocanthal inhibits proliferation and migration by modulating Ca2+ entry through TRPC6 in breast cancer cells. Biochim. Biophys. Acta Mol. Cell Res. 2019, 1866, 474–485. [Google Scholar] [CrossRef] [PubMed]
- Jardin, I.; Diez-Bello, R.; Lopez, J.J.; Redondo, P.C.; Salido, G.M.; Smani, T.; Rosado, J.A. Trpc6 channels are required for proliferation, migration and invasion of breast cancer cell lines by modulation of orai1 and orai3 surface exposure. Cancers 2018, 10, 331. [Google Scholar] [CrossRef] [PubMed]
- Andrikopoulos, P.; Eccles, S.A.; Yaqoob, M.M. Coupling between the TRPC3 ion channel and the NCX1 transporter contributed to VEGF-induced ERK1/2 activation and angiogenesis in human primary endothelial cells. Cell. Signal. 2017, 37, 12–30. [Google Scholar] [CrossRef] [PubMed]
- Song, H.B.; Jun, H.O.; Kim, J.H.; Fruttiger, M.; Kim, J.H. Suppression of transient receptor potential canonical channel 4 inhibits vascular endothelial growth factor-induced retinal neovascularization. Cell Calcium 2015, 57, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Antigny, F.; Girardin, N.; Frieden, M. Transient receptor potential canonical channels are required for in vitro endothelial tube formation. J. Biol. Chem. 2011, 287, 5917–5927. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Gao, M.; Zhou, T.; Xie, M.; Mao, A.; Feng, L.; Yao, X.; Wong, W.T.; Ma, X. The TRPC5 channel regulates angiogenesis and promotes recovery from ischemic injury in mice. J. Biol. Chem. 2018, 294, 28–37. [Google Scholar] [CrossRef]
- Hamdollah Zadeh, M.A.; Glass, C.; Magnussen, A.; Hancox, J.; Bates, D. VEGF-Mediated elevated intracellular calcium and angiogenesis in human microvascular endothelial cells In vitro are inhibited by dominant negative TRPC6. Microcirculation 2008, 15, 605–614. [Google Scholar] [CrossRef]
- Kini, V.; Chavez, A.; Mehta, D. A new role for PTEN in regulating transient receptor potential canonical channel 6-mediated Ca2+ entry, endothelial permeability, and angiogenesis. J. Biol. Chem. 2010, 285, 33082–33091. [Google Scholar] [CrossRef]
- Smani, T.; Gómez, L.J.; Regodon, S.; Woodard, G.E.; Siegfried, G.; Khatib, A.M.; Rosado, J.A. Trp channels in angiogenesis and other endothelial functions. Front. Physiol. 2018, 9. [Google Scholar] [CrossRef]
- Moccia, F.; Negri, S.; Shekha, M.; Faris, P.; Guerra, G. Endothelial Ca2+ signaling, angiogenesis and vasculogenesis: Just what it takes to make a blood vessel. Int. J. Mol. Sci. 2019, 20, 3962. [Google Scholar] [CrossRef]
- Thakore, P.; Earley, S. Transient Receptor Potential Channels and Endothelial Cell Calcium Signaling. Compr. Physiol. 2019, 9, 1249–1277. [Google Scholar] [CrossRef] [PubMed]
- Petros, R.A.; Desimone, J.M. Strategies in the design of nanoparticles for therapeutic applications. Nat. Rev. Drug Discov. 2010, 9, 615–627. [Google Scholar] [CrossRef] [PubMed]
- Rahikkala, A.; Rosenholm, J.M.; Santos, H.A. Biofunctionalized Mesoporous Silica Nanomaterials for Targeted Drug Delivery. Biomed. Appl. Funct. Nanomater. 2018, 489–520. [Google Scholar] [CrossRef]
 |  |  |  | |
|---|---|---|---|---|
| TRPC | 3 | 3 | 7 | 6 |
| TRPV | 5 | 2 | 6 | 6 |
| TRPM | 4 | 1 | 8 | 8 |
| TRPA | 2 | 4 | 1 | 1 |
| TRPML | 1 | 1 | 3 | 3 |
| TRPP | 1 | 1 | 3 | 3 |
| TRPN | 1 | 1 | 0 | 0 |
| Total | 17 | 13 | 28 | 27 |
| Channel | Tissue | Cell Line | Cell function | References |
|---|---|---|---|---|
| TRPC1 | Kidney | MDCK-F | Migration | [87] |
| Brain cancer | D54MG | Migration | [88] | |
| Colon cancer | HCT-116 | Migration | [89] | |
| Skeletal Muscle | C2C12 | Migration, differentiation | [11] | |
| Nasopharyngeal cancer | CNE2 | Migration, Invasion | [90] | |
| Thyroid cancer | ML-1 | Migration and Invasion | [91] | |
| TRPC2 | Rat Thyroid | FRTL5 | Proliferation, adhesion, migration and invasion | [62] |
| TRPC3 | Ovarian cancer | SKOV3 | Migration | [95] |
| Pancreatic stellate cells | RLT-PSC | Migration | [96] | |
| Melanoma | C8161 | Migration | [97] | |
| Neuronal progenitor cell | NPCs | Migration | [98] | |
| Bladder Cancer | LD611 | Migration and Invasion | [99] | |
| TRPC4 | Coronary artery | HCAECs | Proliferation | [70] |
| Ovarian cancer | SKOV3 | Proliferation | [100] | |
| Medulloblastoma | DAOY | Migration, Invasion | [101] | |
| Lung cancer | A549 | Proliferation | [102] | |
| TRPC5 | Rat hippocampal neuron | E18 | Inhibition of neurite extension | [103] |
| Mouse Kidney | Podocytes | Promote migration | [104] | |
| TRPC6 | Mouse Kidney | Podocytes | Inhibit migration | [109] |
| Head and neck cancer | HNSCC | Promote Invasion | [107] | |
| Brain cancer | U373MG | Migration, cell growth | [108] | |
| Prostate cancer | hPCE | Proliferation | [109] | |
| Lung cancer | A549 | Proliferation | [110] | |
| Breast Cancer | MDA-MB-231 | Migration | [111] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asghar, M.Y.; Törnquist, K. Transient Receptor Potential Canonical (TRPC) Channels as Modulators of Migration and Invasion. Int. J. Mol. Sci. 2020, 21, 1739. https://doi.org/10.3390/ijms21051739
Asghar MY, Törnquist K. Transient Receptor Potential Canonical (TRPC) Channels as Modulators of Migration and Invasion. International Journal of Molecular Sciences. 2020; 21(5):1739. https://doi.org/10.3390/ijms21051739
Chicago/Turabian StyleAsghar, Muhammad Yasir, and Kid Törnquist. 2020. "Transient Receptor Potential Canonical (TRPC) Channels as Modulators of Migration and Invasion" International Journal of Molecular Sciences 21, no. 5: 1739. https://doi.org/10.3390/ijms21051739
APA StyleAsghar, M. Y., & Törnquist, K. (2020). Transient Receptor Potential Canonical (TRPC) Channels as Modulators of Migration and Invasion. International Journal of Molecular Sciences, 21(5), 1739. https://doi.org/10.3390/ijms21051739






