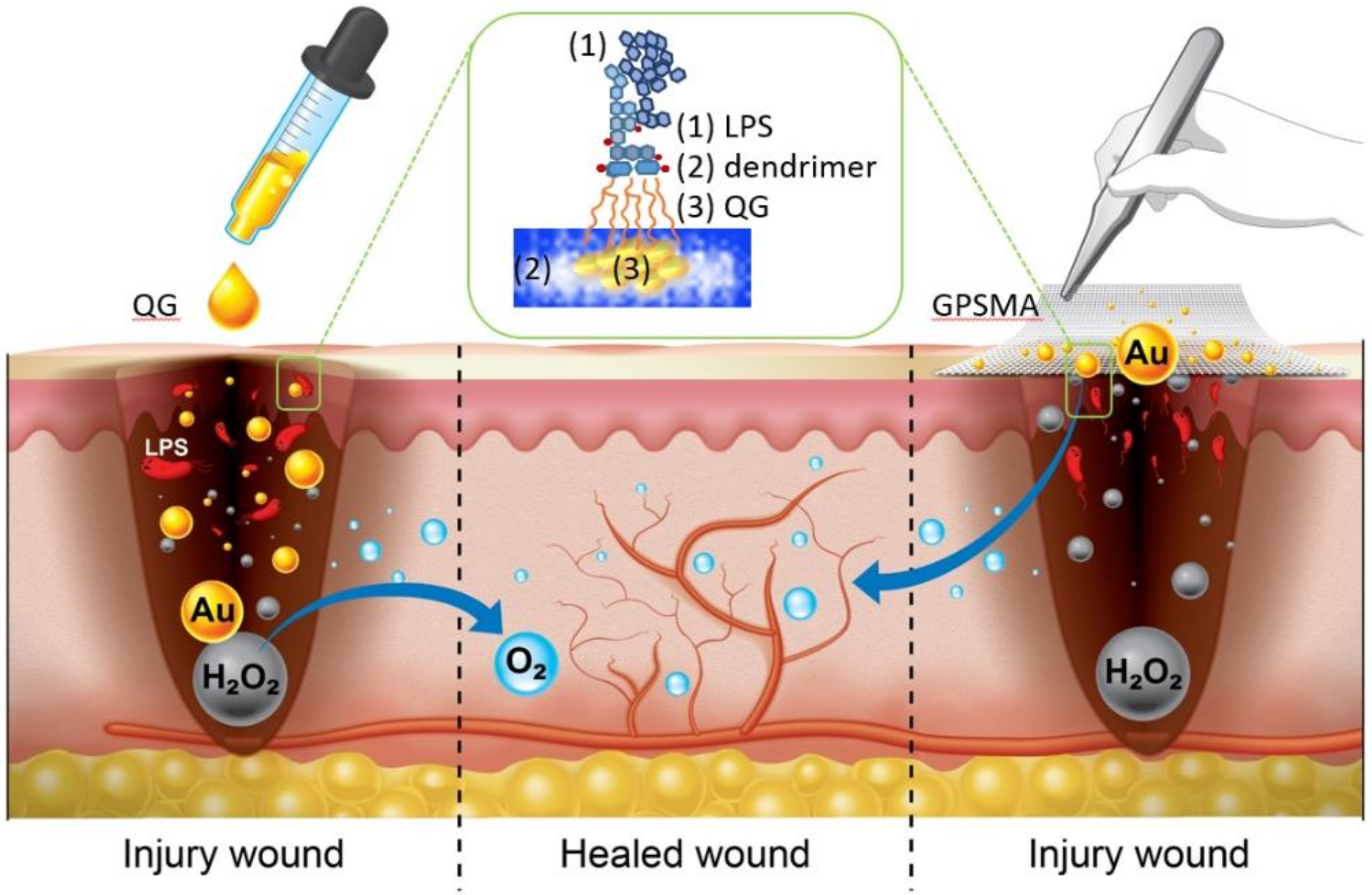
The Bioactive Core and Corona Synergism of Quantized Gold Enables Slowed Inflammation and Increased Tissue Regeneration in Wound Hypoxia
Abstract
1. Introduction
2. Results and Discussion
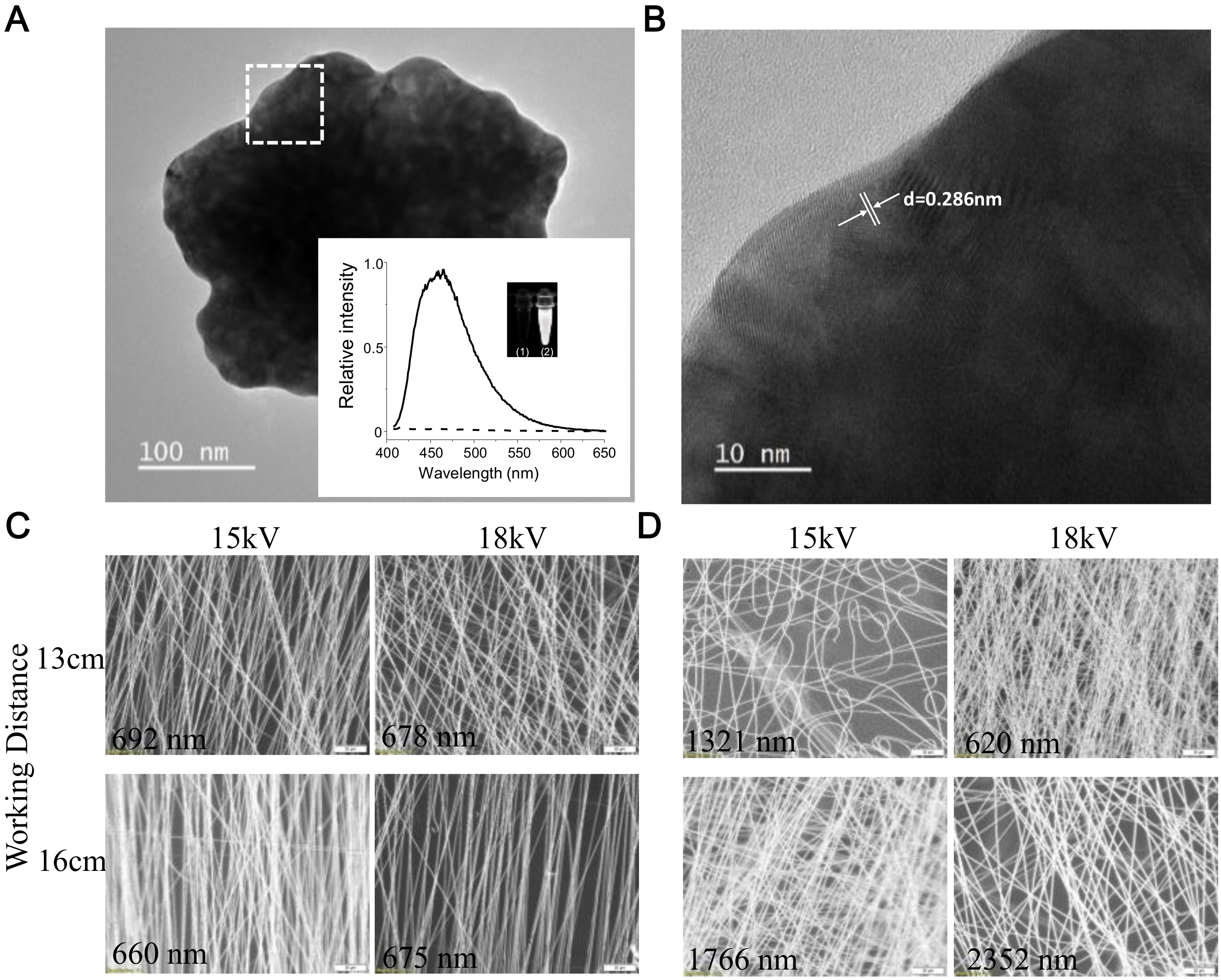
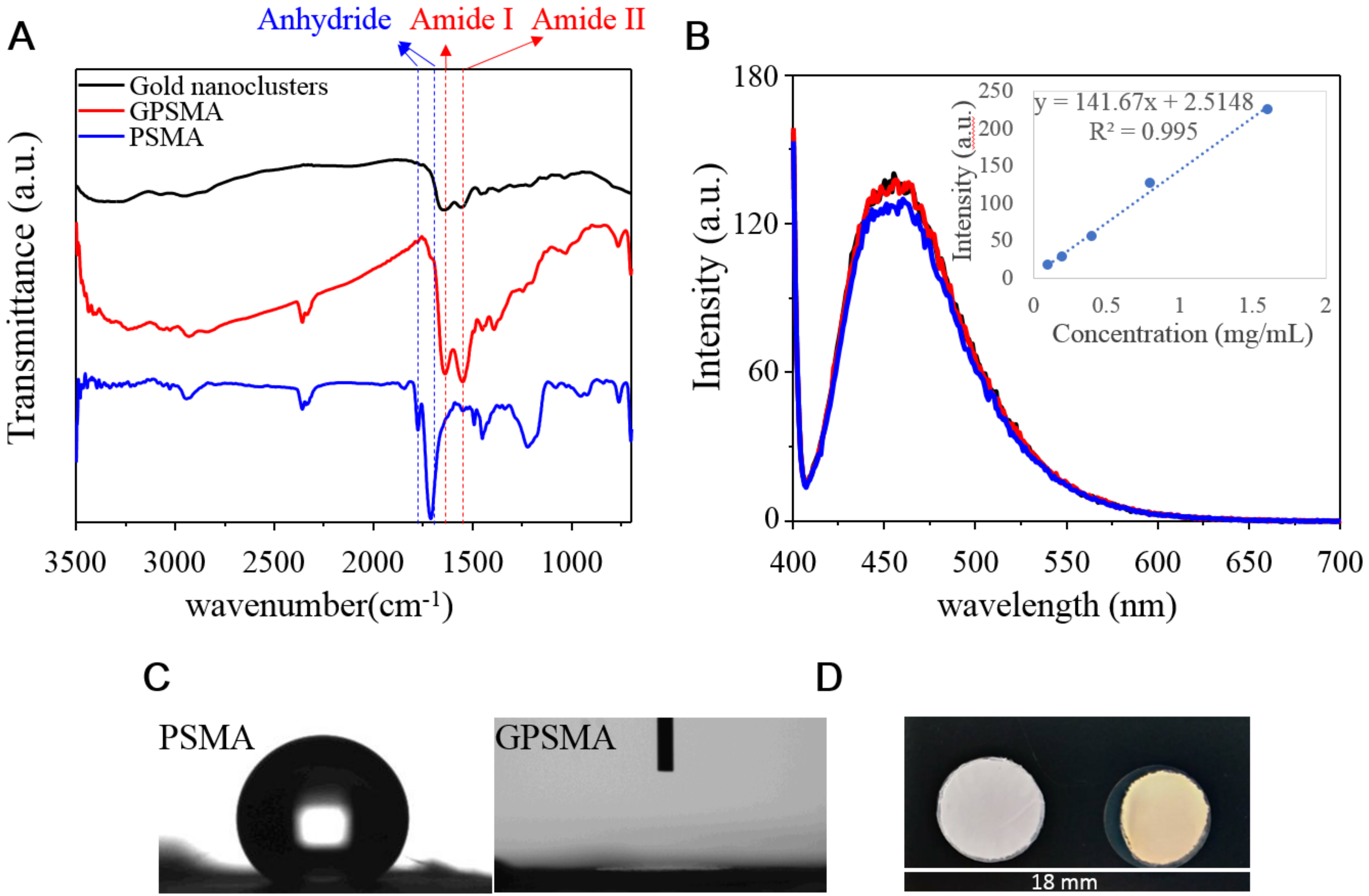
2.1. Morphology and Hydrophobic Properties of QG and GPSMA Nanofibers
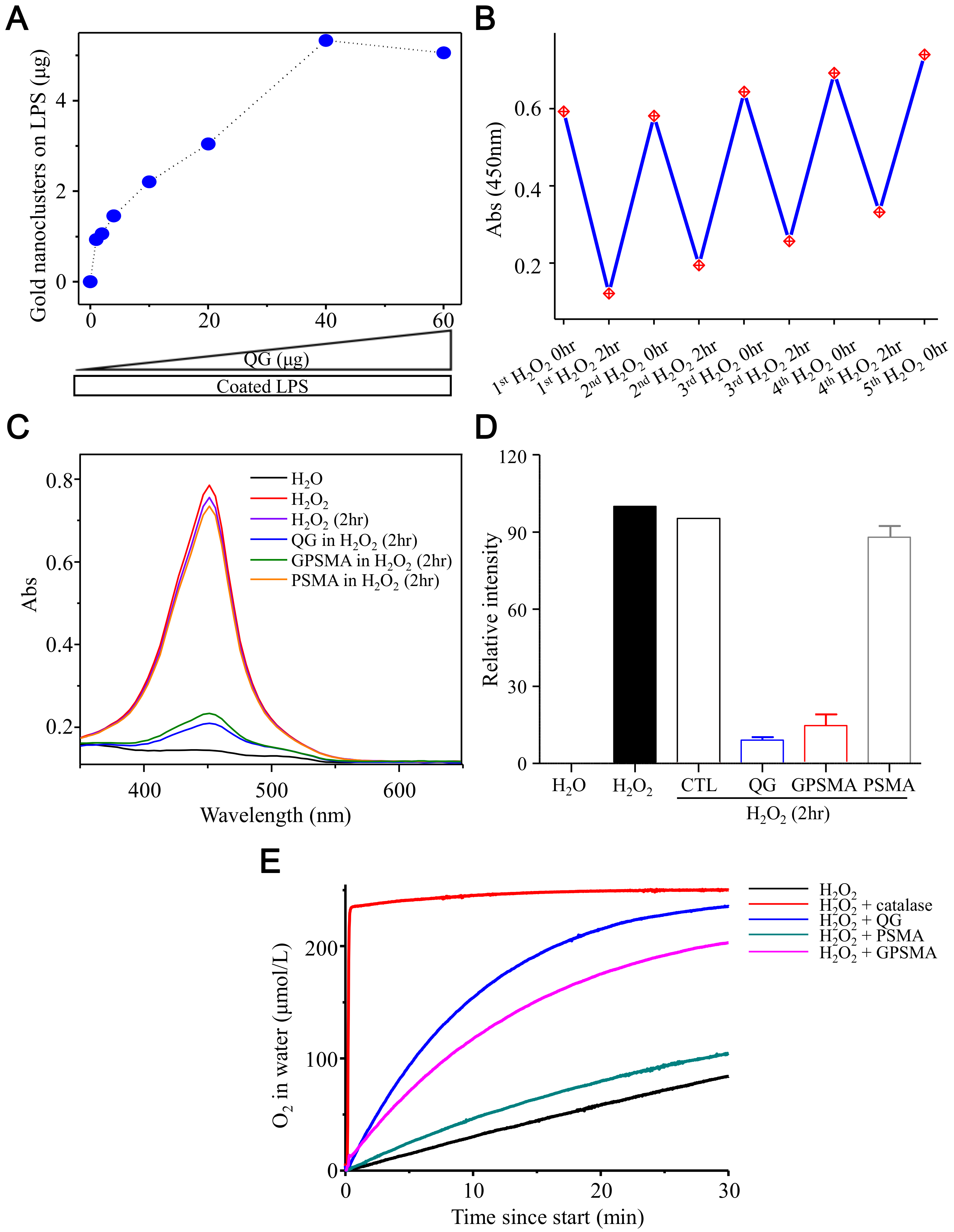
2.2. Endotoxin-Binding Capability and Catalase-Like Activity of QG and GPSMA Nanofibers
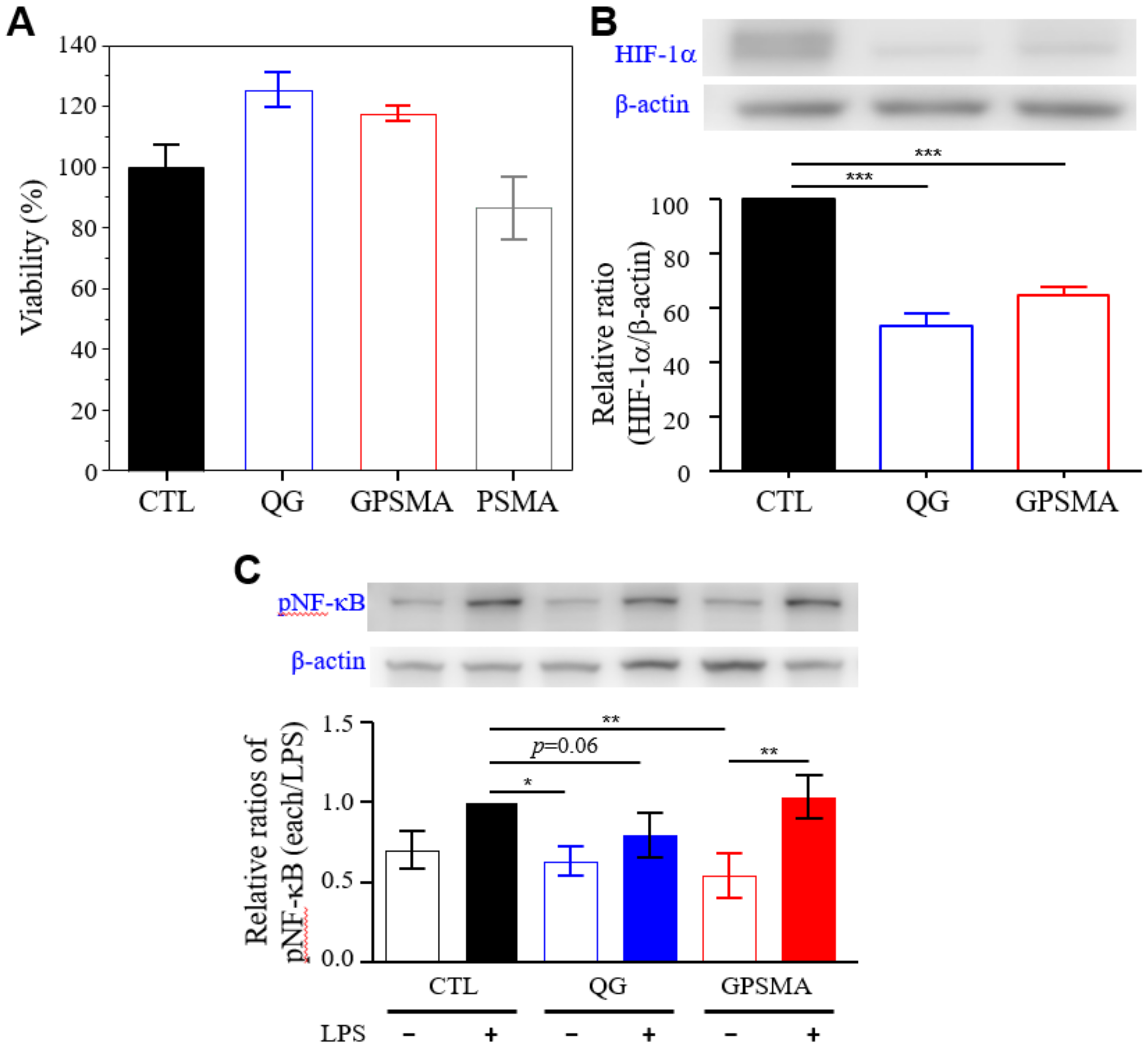
2.3. Biocompatibility Study of GPSMA
2.4. GPSMA Diminishes NF-kB Activity In Vitro and Downregulates HIF-1α in Cells in Hypoxic Microenvironments
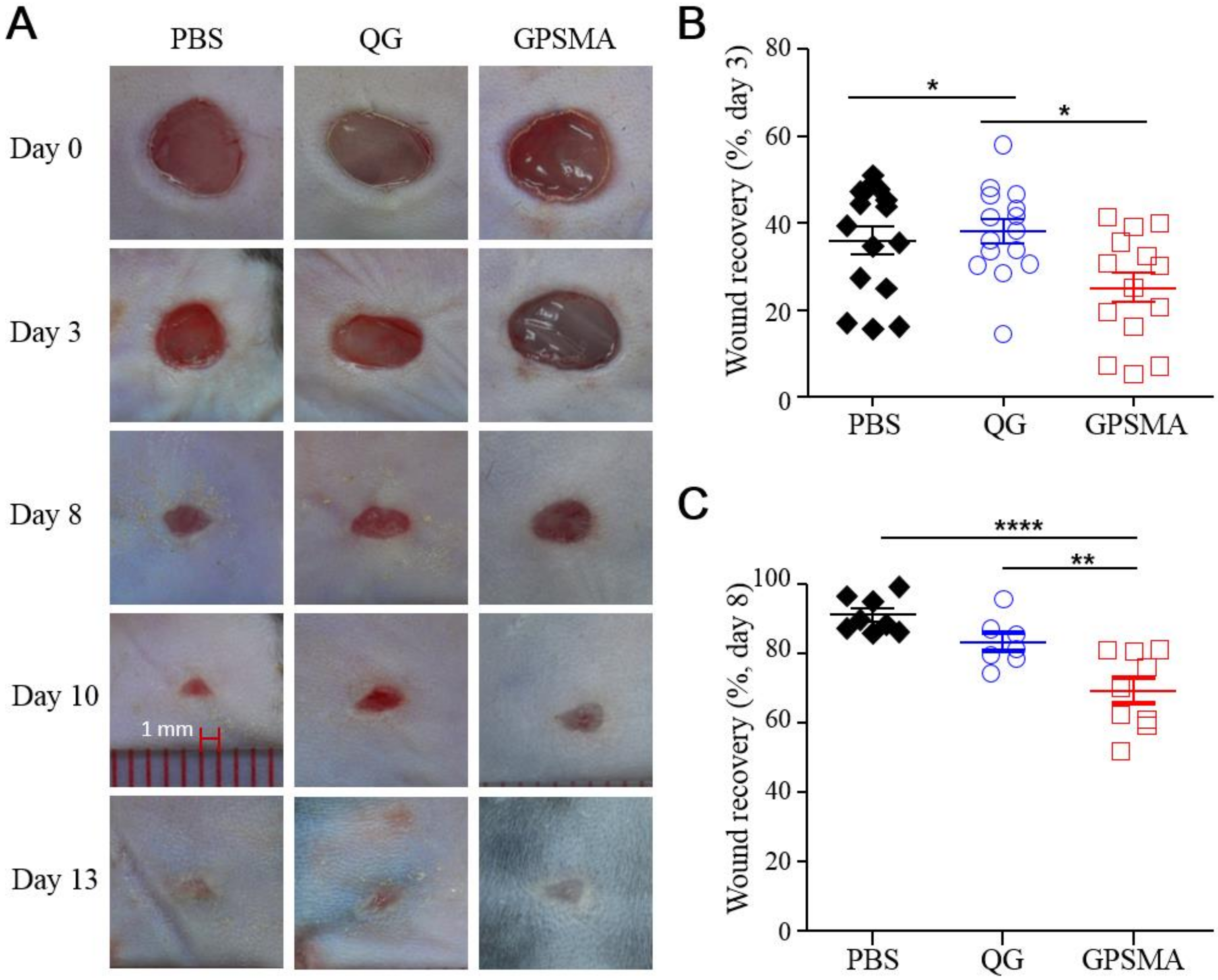
2.5. Wound Closure after Treatment with Either QG Only or GPSMA
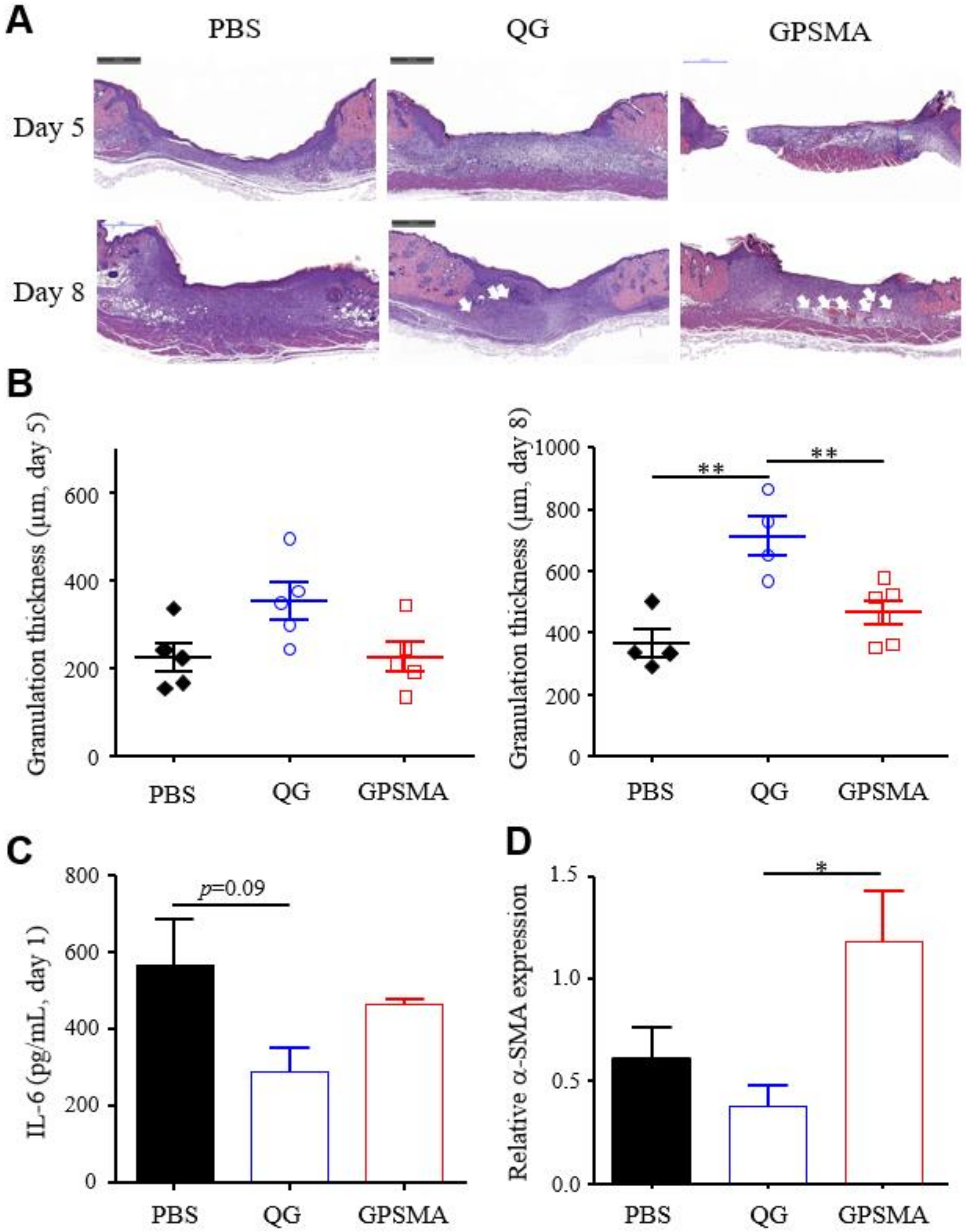
2.6. The Granulation Development and Re-Epithelialization Impact on Wound Healing after Treatment with Either QG Only or GPSMA
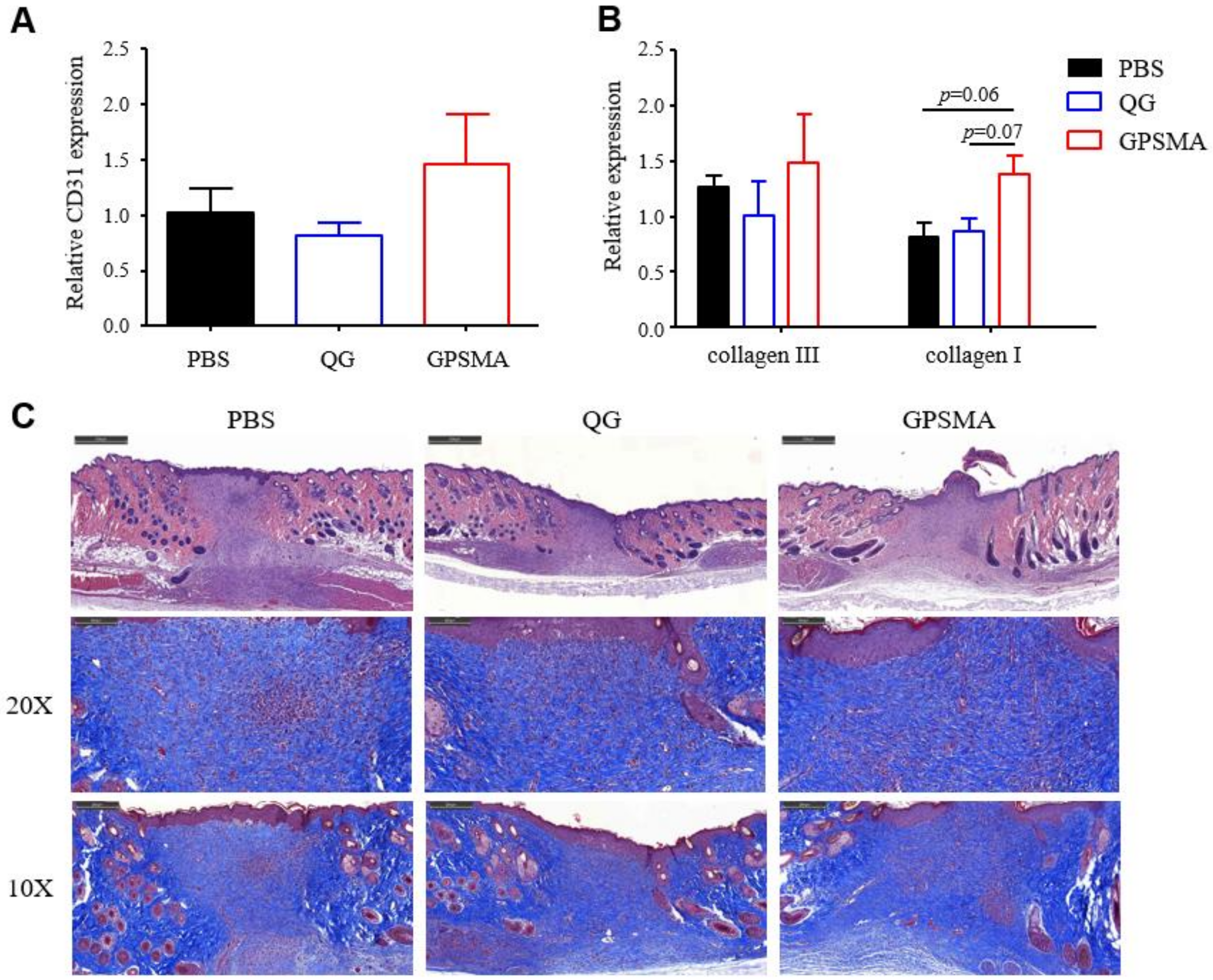
2.7. The Impacts of GPSMA on Skin Tissue Regeneration
3. Discussion
4. Materials and Methods
4.1. Fabrication and Characterization of PSMA Nanofibers and GPSMA
4.2. Endotoxin-Binding Assay
4.3. Catalase-Like Activity and Oxygen Generation of QG and GPSMA
4.4. Cell Culture and Biocompatibility
4.5. Western Blot
4.6. Wound Healing Observation
4.7. Analysis of Development and Inflammation of Granulation Tissue during Wound Healing
4.8. Statistical Analyses
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Whitfield, C.; Trent, M.S. Biosynthesis and export of bacterial lipopolysaccharides. Annu. Rev. Biochem. 2014, 83, 99–128. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Akira, S. Toll-like receptor downstream signaling. Arthritis. Res. Ther. 2005, 7, 12–19. [Google Scholar] [CrossRef] [PubMed]
- McGettrick, A.F.; O’Neill, L.A.J. Regulators of TLR4 Signaling by Endotoxins. In Endotoxins: Structure, Function and Recognition, 1st ed.; Wang, X., Quinn, P.J., Eds.; Springer: Dordrecht, The Netherlands, 2010; Volume 53, pp. 153–172. [Google Scholar]
- Park, B.S.; Lee, J.-O. Recognition of lipopolysaccharide pattern by tlr4 complexes. Exp. Mol. Med. 2013, 45, e66. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. Life with oxygen. Science 2007, 318, 62–64. [Google Scholar] [CrossRef]
- Schreml, S.; Szeimies, R.M.; Prantl, L.; Karrer, S.; Landthaler, M.; Babilas, P. Oxygen in acute and chronic wound healing. Brit. J. Dermatol. 2010, 163, 257–268. [Google Scholar] [CrossRef]
- Park, S.; Park, K.M. Hyperbaric oxygen-generating hydrogels. Biomaterials 2018, 182, 234–244. [Google Scholar] [CrossRef]
- Heyboer, M., III; Sharma, D.; Santiago, W.; McCulloch, N. Hyperbaric oxygen therapy: Side effects defined and quantified. Adv. Wound Care 2017, 6, 210–224. [Google Scholar] [CrossRef]
- Gholipourmalekabadi, M.; Zhao, S.; Harrison, B.S.; Mozafari, M.; Seifalian, A.M. Oxygen-generating biomaterials: A new, viable paradigm for tissue engineering? Trends Biotechnol. 2016, 34, 1010–1021. [Google Scholar] [CrossRef]
- Zhu, G.; Wang, Q.; Lu, S.; Niu, Y. Hydrogen peroxide: A potential wound therapeutic target. Med. Prin. Pract. 2017, 26, 301–308. [Google Scholar] [CrossRef]
- Gordillo, G.M.; Sen, C.K. Revisiting the essential role of oxygen in wound healing. Am. J. Surg. 2003, 186, 259–263. [Google Scholar] [CrossRef]
- Wei, H.; Wang, E. Nanomaterials with enzyme-like characteristics (nanozymes): Next-generation artificial enzymes. Chem. Soc. Rev. 2013, 42, 6060–6093. [Google Scholar] [CrossRef] [PubMed]
- Saha, K.; Agasti, S.S.; Kim, C.; Li, X.; Rotello, V.M. Gold nanoparticles in chemical and biological sensing. Chem. Rev. 2012, 112, 2739–2779. [Google Scholar] [CrossRef] [PubMed]
- Fratoddi, I.; Venditti, I.; Cametti, C.; Russo, M.V. Gold nanoparticles and gold nanoparticle-conjugates for delivery of therapeutic molecules. Progress and challenges. J. Mater. Chem. B 2014, 2, 4204–4220. [Google Scholar] [CrossRef]
- Myers, V.S.; Weir, M.G.; Carino, E.V.; Yancey, D.F.; Pande, S.; Crooks, R.M. Dendrimer-encapsulated nanoparticles: New synthetic and characterization methods and catalytic applications. Chem. Sci. 2011, 2, 1632–1646. [Google Scholar] [CrossRef]
- Liu, C.-P.; Wu, T.-H.; Lin, Y.-L.; Liu, C.-Y.; Wang, S.; Lin, S.-Y. Tailoring enzyme-like activities of gold nanoclusters by polymeric tertiary amines for protecting neurons against oxidative stress. Small 2016, 12, 4127–4135. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Chen, T.; He, S.; Robbins, J.P.; Podkolzin, S.G.; Tian, F. Observation and identification of an atomic oxygen structure on catalytic gold nanoparticles. Angew. Chem. 2017, 56, 12952–12957. [Google Scholar] [CrossRef]
- Lin, Y.; Zhao, A.; Tao, Y.; Ren, J.; Qu, X. Ionic liquid as an efficient modulator on artificial enzyme system: Toward the realization of high-temperature catalytic reactions. J. Am. Chem. Soc. 2013, 135, 4207–4210. [Google Scholar] [CrossRef]
- Laibinis, P.E.; Whitesides, G.M.; Allara, D.L.; Tao, Y.T.; Parikh, A.N.; Nuzzo, R.G. Comparison of the structures and wetting properties of self-assembled monolayers of n-alkanethiols on the coinage metal surfaces, copper, silver, and gold. J. Am. Chem. Soc. 1991, 113, 7152–7167. [Google Scholar] [CrossRef]
- Bhattacharya, P.; Conroy, N.; Rao, A.M.; Powell, B.A.; Ladner, D.A.; Ke, P.C. Pamam dendrimer for mitigating humic foulant. RSC Adv. 2012, 2, 7997–8001. [Google Scholar] [CrossRef]
- Duan, X.; Xiao, J.; Yin, Q.; Zhang, Z.; Mao, S.; Li, Y. Amphiphilic graft copolymer based on poly(styrene-co-maleic anhydride) with low molecular weight polyethylenimine for efficient gene delivery. Int. J. Nanomedicine 2012, 7, 4961–4972. [Google Scholar]
- Henry, S.M.; El-Sayed, M.E.H.; Pirie, C.M.; Hoffman, A.S.; Stayton, P.S. Ph-responsive poly(styrene-alt-maleic anhydride) alkylamide copolymers for intracellular drug delivery. Biomacromolecules 2006, 7, 2407–2414. [Google Scholar] [CrossRef] [PubMed]
- Cromer, J.R.; Wood, S.J.; Miller, K.A.; Nguyen, T.; David, S.A. Functionalized dendrimers as endotoxin sponges. Bioorg. Med. Chem. Lett. 2005, 15, 1295–1298. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.P.; Wu, T.H.; Liu, C.Y.; Cheng, H.J.; Lin, S.Y. Interactions of nitroxide radicals with dendrimer-entrapped au8-clusters: A fluorescent nanosensor for intracellular imaging of ascorbic acid. J. Mater. Chem. B 2015, 3, 191–197. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. Nf-κb signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Brandenburg, K.; Funari, S.S.; Koch, M.H.J.; Seydel, U. Investigation into the acyl chain packing of endotoxins and phospholipids under near physiological conditions by waxs and ftir spectroscopy. J. Struct. Biol. 1999, 128, 175–186. [Google Scholar] [CrossRef]
- Crompton, R.; Williams, H.; Ansell, D.; Campbell, L.; Holden, K.; Cruickshank, S.; Hardman, M.J. Oestrogen promotes healing in a bacterial lps model of delayed cutaneous wound repair. Lab. Invest. 2016, 96, 439. [Google Scholar] [CrossRef]
- Cheng, F.; Shen, Y.; Mohanasundaram, P.; Lindström, M.; Ivaska, J.; Ny, T.; Eriksson, J.E. Vimentin coordinates fibroblast proliferation and keratinocyte differentiation in wound healing via tgf-β–slug signaling. Proc. Natl. Acad. Sci. USA 2016, 113, E4320–E4327. [Google Scholar] [CrossRef]
- Pastar, I.; Stojadinovic, O.; Yin, N.C.; Ramirez, H.; Nusbaum, A.G.; Sawaya, A.; Patel, S.B.; Khalid, L.; Isseroff, R.R.; Tomic-Canic, M.; et al. Epithelialization in wound healing: A comprehensive review. Adv. Wound Care 2014, 3, 445–464. [Google Scholar]
- Castilla, D.M.; Liu, Z.-J.; Velazquez, O.C. Oxygen: Implications for wound healing. Adv. Wound Care 2012, 1, 225–230. [Google Scholar] [CrossRef]
- Rezvani, H.R.; Ali, N.; Serrano-Sanchez, M.; Dubus, P.; Varon, C.; Ged, C.; Pain, C.; Cario-André, M.; Seneschal, J.; Taïeb, A.; et al. Loss of epidermal hypoxia-inducible factor-1α accelerates epidermal aging and affects re-epithelialization in human and mouse. J. Cell Sci. 2011, 124, 4172–4183. [Google Scholar] [CrossRef]
- Serrano-Gomez, S.J.; Maziveyi, M.; Alahari, S.K. Regulation of epithelial-mesenchymal transition through epigenetic and post-translational modifications. Mol. Cancer 2016, 15, 18. [Google Scholar] [CrossRef] [PubMed]
- Chitturi, R.T.; Balasubramaniam, A.M.; Parameswar, R.A.; Kesavan, G.; Haris, K.T.M.; Mohideen, K. The role of myofibroblasts in wound healing, contraction and its clinical implications in cleft palate repair. J. Int. Oral Health 2015, 7, 75–80. [Google Scholar] [PubMed]
- Desmouliere, A.; Redard, M.; Darby, I.; Gabbiani, G. Apoptosis mediates the decrease in cellularity during the transition between granulation-tissue and scar. Am. J. Pathol. 1995, 146, 56–66. [Google Scholar] [PubMed]
- Hinz, B. Formation and function of the myofibroblast during tissue repair. J. Invest. Dermatol. 2007, 127, 526–537. [Google Scholar] [CrossRef] [PubMed]
- Duranteau, J.; Chandel, N.S.; Kulisz, A.; Shao, Z.; Schumacker, P.T. Intracellular signaling by reactive oxygen species during hypoxia in cardiomyocytes. J. Biol. Chem. 1998, 273, 11619–11624. [Google Scholar] [CrossRef]
- Centeno-Cerdas, C.; Jarquín-Cordero, M.; Chávez, M.N.; Hopfner, U.; Holmes, C.; Schmauss, D.; Machens, H.-G.; Nickelsen, J.; Egaña, J.T. Development of photosynthetic sutures for the local delivery of oxygen and recombinant growth factors in wounds. Acta Biomater. 2018, 81, 184–194. [Google Scholar] [CrossRef]
- DiPietro, L.A. Angiogenesis and scar formation in healing wounds. Curr. Opin. Rheumatol. 2013, 25, 87–91. [Google Scholar] [CrossRef]
- Myllyharju, J. Prolyl 4-hydroxylases, the key enzymes of collagen biosynthesis. Matrix Biol. 2003, 22, 15–24. [Google Scholar] [CrossRef]
- Peterkofsky, B.; Udenfriend, S. Enzymatic hydroxylation of proline in microsomal polypeptide leading to formation of collagen. Proc. Natl. Acad. Sci. USA 1965, 53, 335–342. [Google Scholar] [CrossRef]
- Myllyharju, J. Prolyl 4-hydroxylases, key enzymes in the synthesis of collagens and regulation of the response to hypoxia, and their roles as treatment targets. Ann. Med. 2008, 40, 402–417. [Google Scholar] [CrossRef]
- Xiong, G.; Stewart, R.L.; Chen, J.; Gao, T.; Scott, T.L.; Samayoa, L.M.; O’Connor, K.; Lane, A.N.; Xu, R. Collagen prolyl 4-hydroxylase 1 is essential for hif-1α stabilization and tnbc chemoresistance. Nat. Commun. 2018, 9, 4456. [Google Scholar] [CrossRef] [PubMed]
- Liao, F.-H.; Wu, T.-H.; Huang, Y.-T.; Lin, W.-J.; Su, C.-J.; Jeng, U.S.; Kuo, S.-C.; Lin, S.-Y. Subnanometer gold clusters adhere to lipid a for protection against endotoxin-induced sepsis. Nano Lett. 2018, 18, 2864–2869. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-P.; Wu, T.-H.; Liu, C.-Y.; Chen, K.-C.; Chen, Y.-X.; Chen, G.-S.; Lin, S.-Y. Self-supplying o2 through the catalase-like activity of gold nanoclusters for photodynamic therapy against hypoxic cancer cells. Small 2017, 13, 1700278. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yeh, L.-C.; Chen, S.-P.; Liao, F.-H.; Wu, T.-H.; Huang, Y.-T.; Lin, S.-Y. The Bioactive Core and Corona Synergism of Quantized Gold Enables Slowed Inflammation and Increased Tissue Regeneration in Wound Hypoxia. Int. J. Mol. Sci. 2020, 21, 1699. https://doi.org/10.3390/ijms21051699
Yeh L-C, Chen S-P, Liao F-H, Wu T-H, Huang Y-T, Lin S-Y. The Bioactive Core and Corona Synergism of Quantized Gold Enables Slowed Inflammation and Increased Tissue Regeneration in Wound Hypoxia. International Journal of Molecular Sciences. 2020; 21(5):1699. https://doi.org/10.3390/ijms21051699
Chicago/Turabian StyleYeh, Lu-Chen, Shu-Ping Chen, Fang-Hsuean Liao, Te-Haw Wu, Yu-Ting Huang, and Shu-Yi Lin. 2020. "The Bioactive Core and Corona Synergism of Quantized Gold Enables Slowed Inflammation and Increased Tissue Regeneration in Wound Hypoxia" International Journal of Molecular Sciences 21, no. 5: 1699. https://doi.org/10.3390/ijms21051699
APA StyleYeh, L.-C., Chen, S.-P., Liao, F.-H., Wu, T.-H., Huang, Y.-T., & Lin, S.-Y. (2020). The Bioactive Core and Corona Synergism of Quantized Gold Enables Slowed Inflammation and Increased Tissue Regeneration in Wound Hypoxia. International Journal of Molecular Sciences, 21(5), 1699. https://doi.org/10.3390/ijms21051699




