Hsp90 Relieves Heat Stress-Induced Damage in Mouse Kidneys: Involvement of Antiapoptotic PKM2-AKT and Autophagic HIF-1α Signaling
Abstract
1. Introduction
2. Results
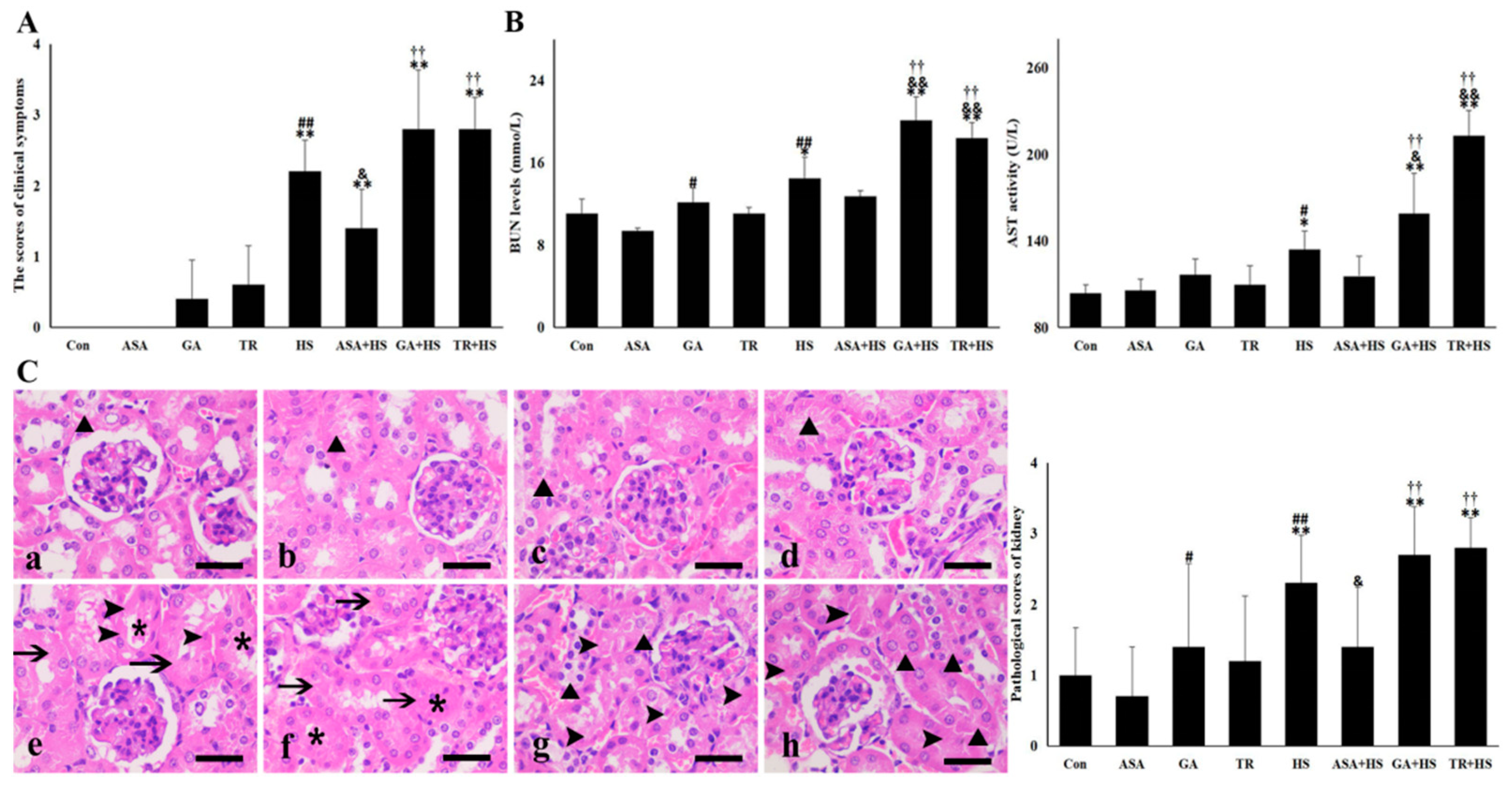
2.1. Heat-Stressed Kidney Damage under Different Treatments
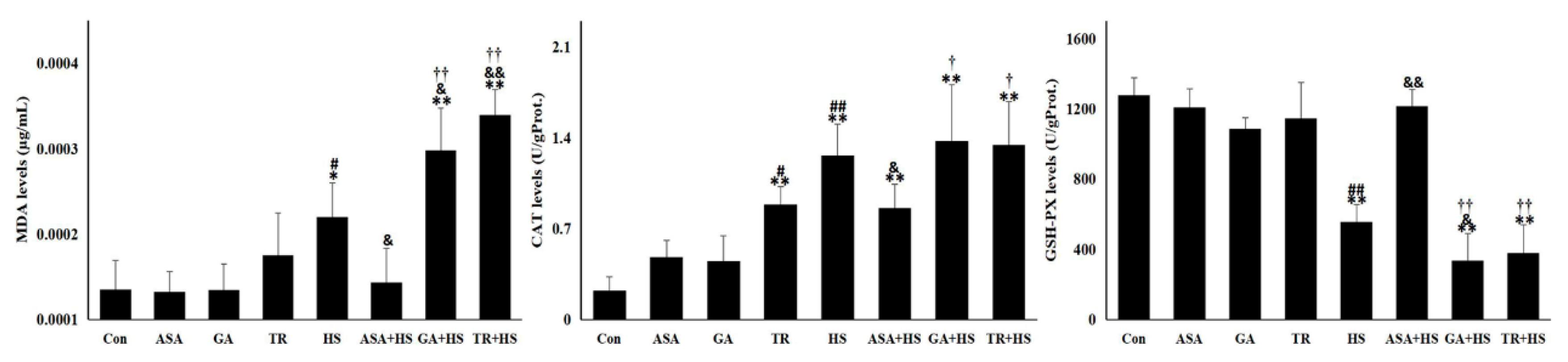
2.2. The Oxidative and Antioxidant Condition of the Kidney under Different Treatments
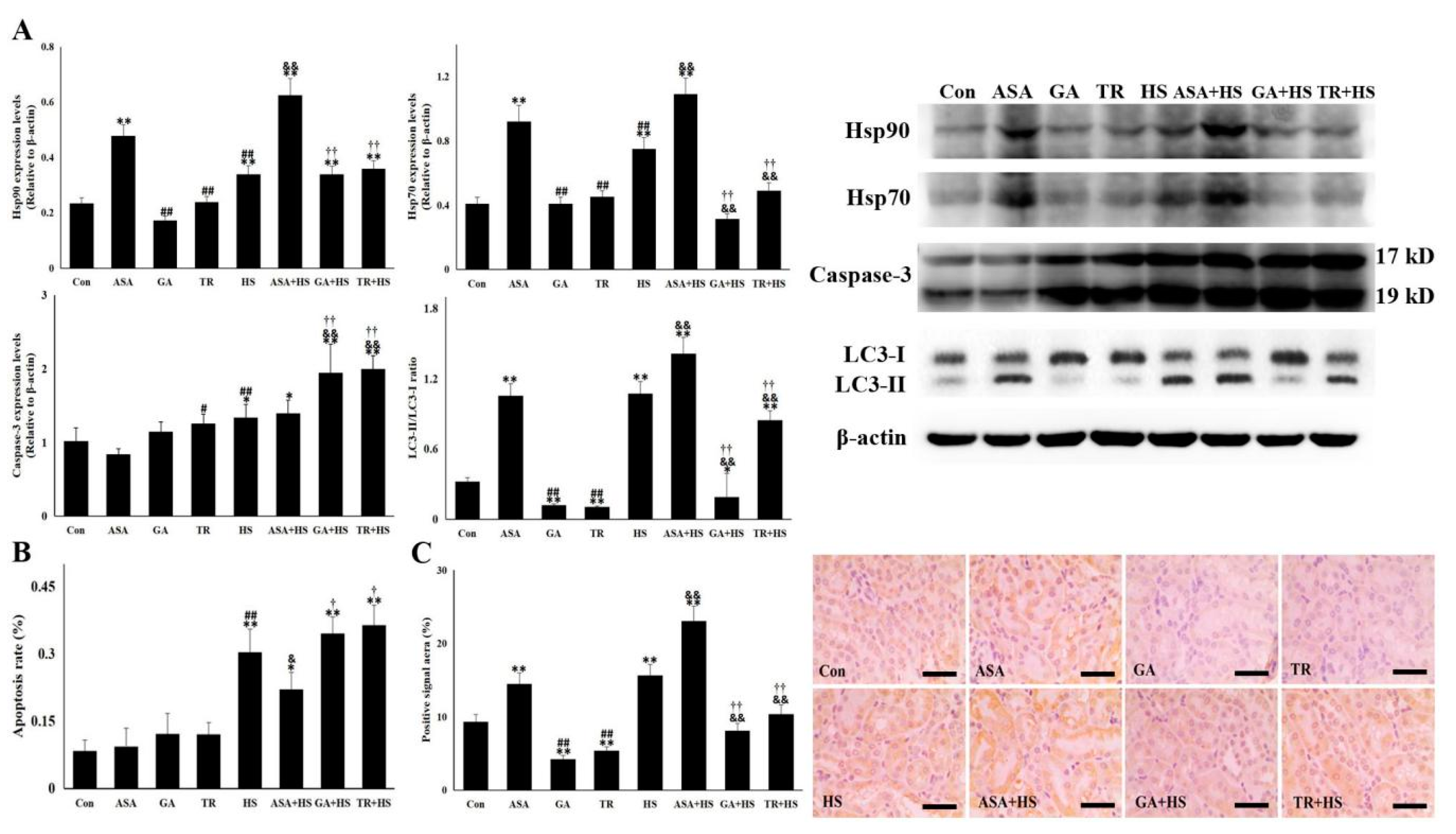
2.3. Hsp90 Correlates with Cellular Apoptosis and Autophagy-Mediated Survival in Conjunction with Hsp70
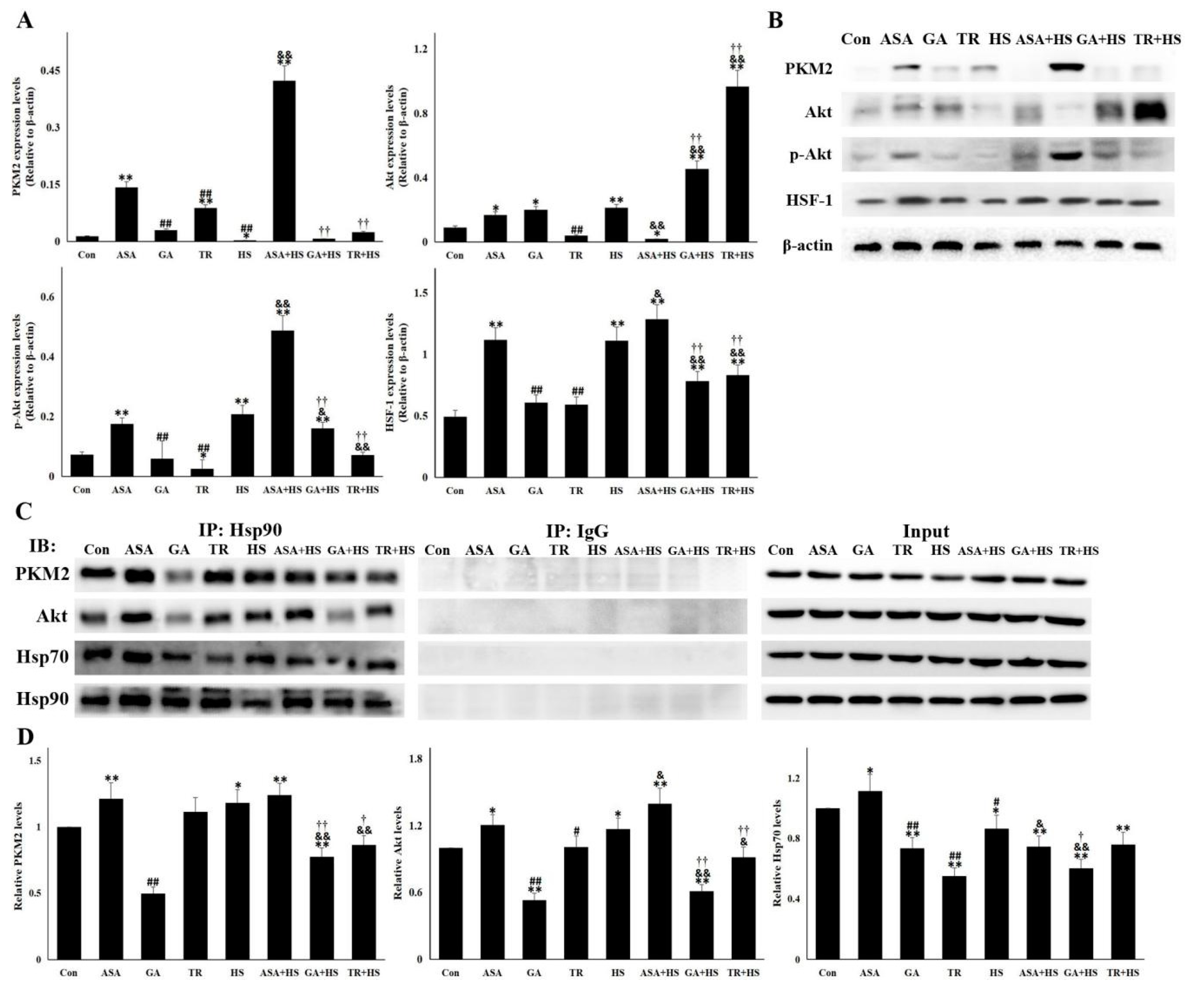
2.4. The Antiapoptotic Pathway Hsp90-PKM2-Akt Signaling Is Affected by Different Treatments
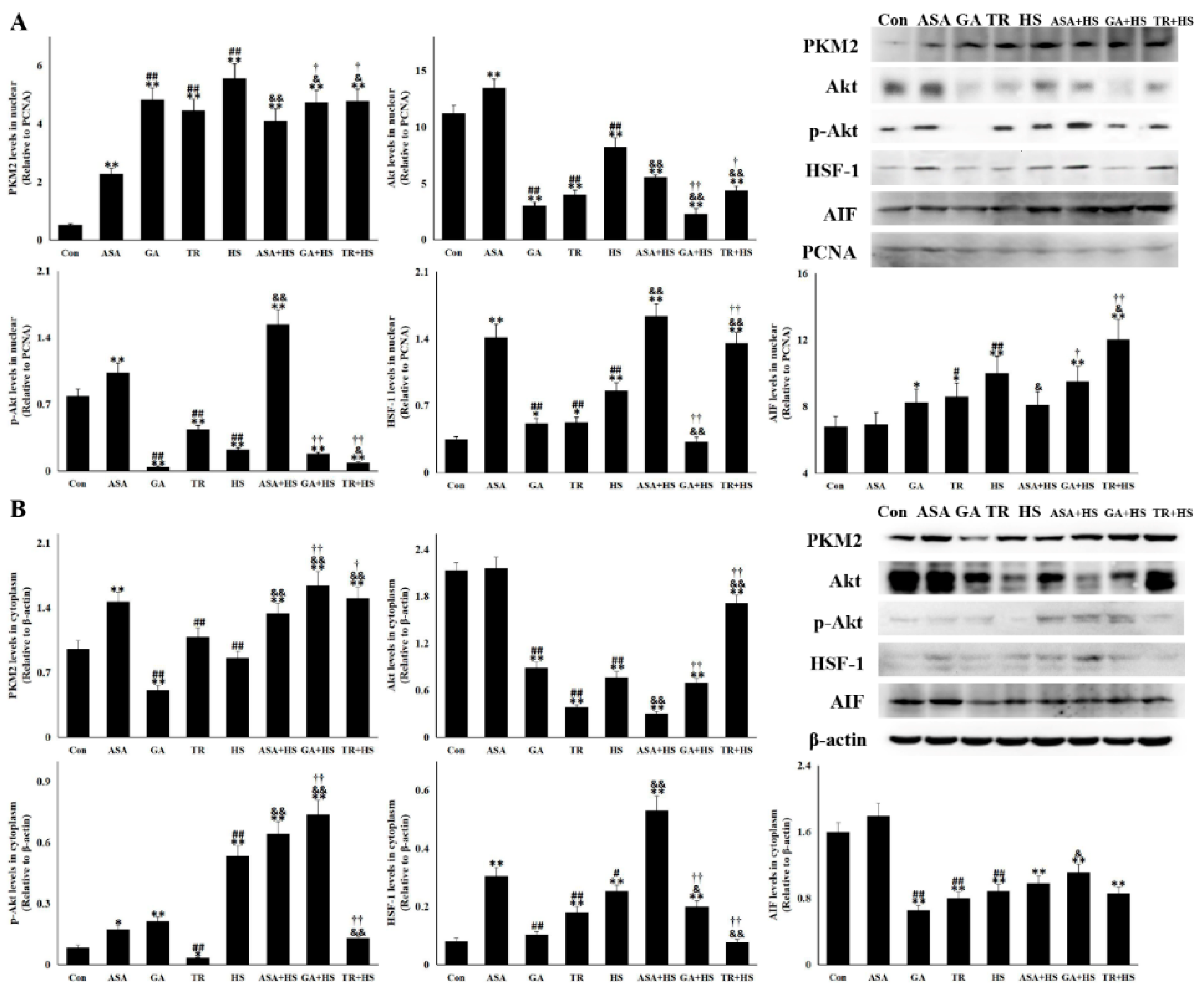
2.5. The Nuclear PKM2, Akt, p-Akt, and HSF-1 Is Also Altered for Their Renal Protection
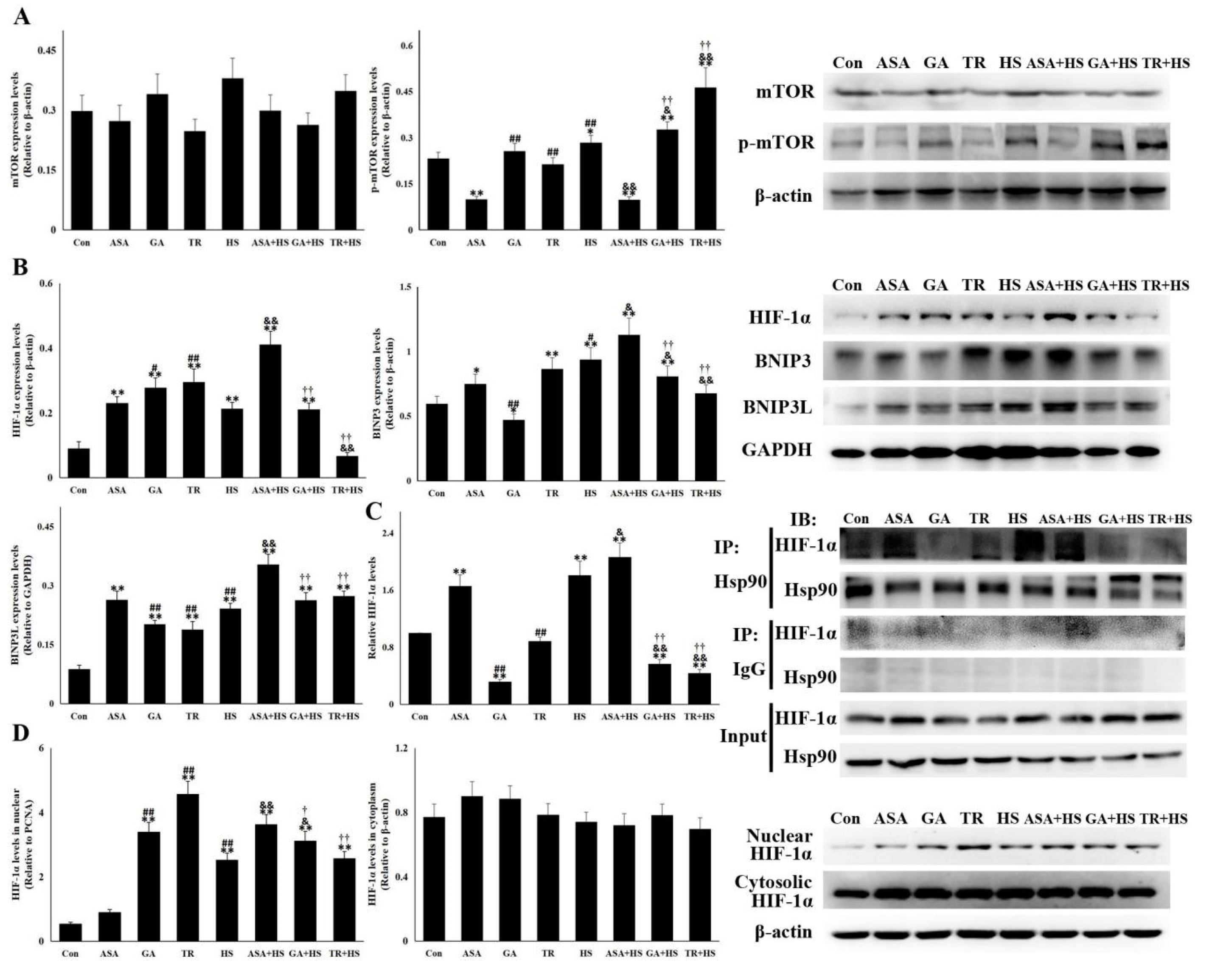
2.6. The Hsp90-HIF-1α-BNIP3/BNIP3L Pathway Was Correlated with Heat Stress-Mediated Autophagy
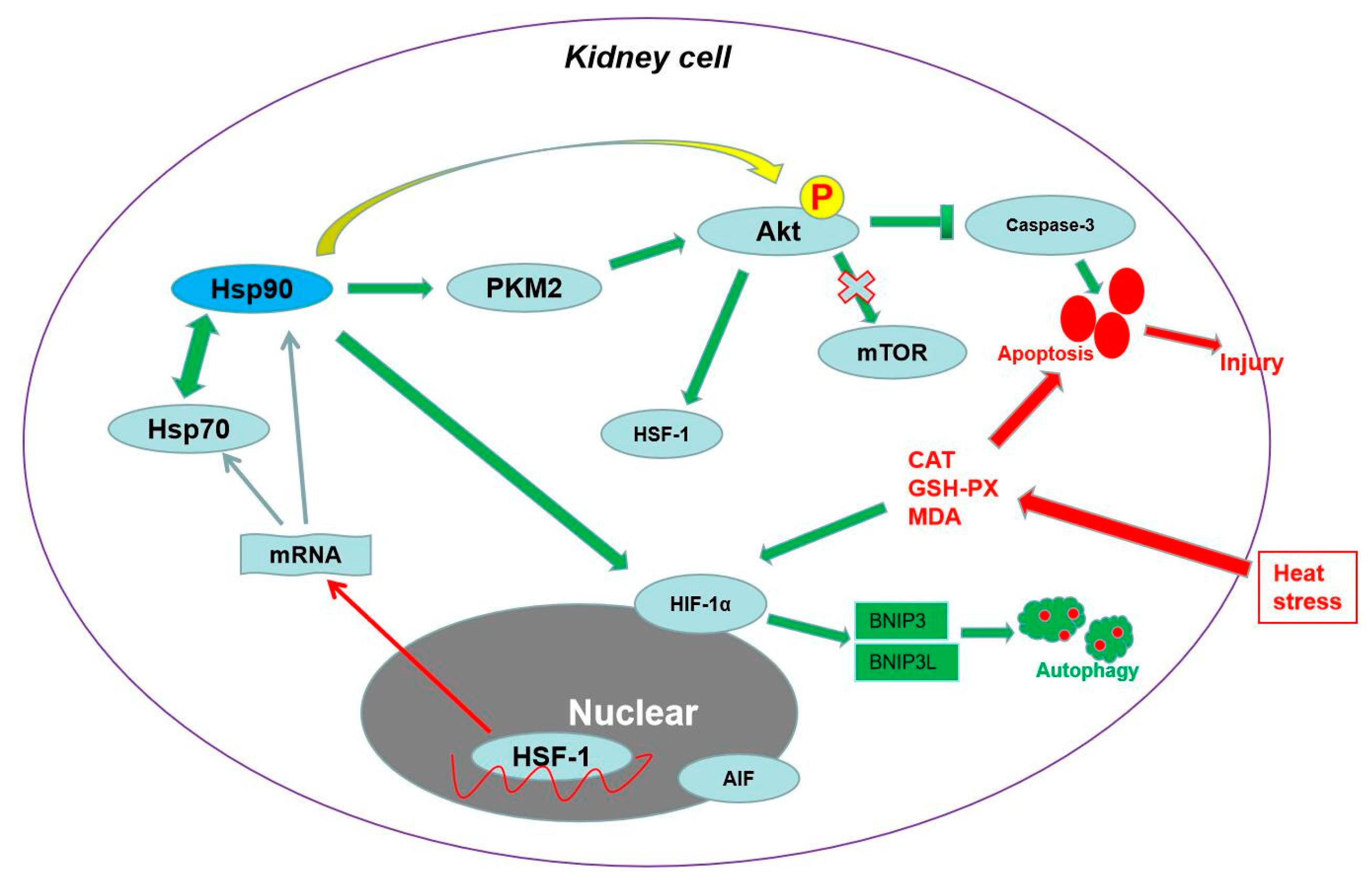
3. Discussion
4. Materials and Methods
4.1. Animals and Sampling
4.2. Clinical Observation and Scoring
4.3. Assessment of Kidney Damage
4.4. Extraction of Total Protein and Determination of Oxidation and Antioxidant Markers
4.5. Extraction of Nuclear and Cytoplasmic Proteins
4.6. Western Blotting
4.7. Immunohistochemistry
4.8. Coimmunoprecipitation
4.9. TdT-Mediated dUTP Nick End Labeling (TUNEL) Staining
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Asserraji, M.; Benameur, I.; Maoujoud, O.; El Kharras, A.; Hajbi, H.; Filali, K. Late care in marathon runs leading to exertional heat stroke with multiple organ failure. Asian J. Sports Med. 2014, 5, 136–138. [Google Scholar]
- Casa, D.J.; Armstrong, L.E.; Ganio, M.S.; Yeargin, S.W. Exertional heat stroke in competitive athletes. Curr. Sports Med. Rep. 2005, 4, 309–317. [Google Scholar] [CrossRef]
- Glaser, J.; Lemery, J.; Rajagopalan, B.; Diaz, H.F.; García-Trabanino, R.; Taduri, G.; Madero, M.; Amarasinghe, M.; Abraham, G.; Anutrakulchai, S.; et al. Climate change and the emergent epidemic of CKD from heat stress in rural communities: The case for heat stress nephropathy. Clin. J. Am. Soc. Nephrol. 2016, 11, 1472–1483. [Google Scholar] [CrossRef]
- Im, E.S.; Pal, J.S.; Eltahir, E.A.B. Deadly heat waves projected in the densely populated agricultural regions of South Asia. Sci. Adv. 2017, 3, e1603322. [Google Scholar] [CrossRef]
- Mohanaselvan, A.; Bhaskar, E. Mortality from non-exertional heat stroke still high in India. Int. J. Occup. Environ. Med. 2014, 5, 222–224. [Google Scholar]
- Rahmstorf, S.; Coumou, D. Increase of extreme events in a warming world. Proc. Natl. Acad. Sci. USA 2011, 108, 17905–17909. [Google Scholar] [CrossRef]
- Suzuki, S.; Maruyama, S.; Sato, W.; Morita, Y.; Sato, F.; Miki, Y.; Kato, S.; Katsuno, M.; Sobue, G.; Yuzawa, Y.; et al. Geranylgeranylacetone ameliorates ischemic acute renal failure via induction of Hsp70. Kidney Int. 2005, 7, 2210–2220. [Google Scholar] [CrossRef]
- Devarajan, P. Update on mechanisms of ischemic acute kidney injury. J. Am. Soc. Nephrol. 2006, 17, 1503–1520. [Google Scholar] [CrossRef]
- Bonventre, J.V. Dedifferentiation and proliferation of surviving epithelial cells in acute renal failure. J. Am. Soc. Nephrol. 2003, 14, S55–S61. [Google Scholar] [CrossRef]
- Wallin, A.; Zhang, G.; Jones, T.W.; Jaken, S.; Stevens, J. Mechanism of the nephrogenic repair response. Studies on proliferation and vimentin expression after 35S-1, 2-dichlorovinyl-L-cysteine nephrotoxicity in vivo and in cultured proximal tubule epithelial cells. Lab. Investig. 1992, 66, 474–484. [Google Scholar]
- Picone, P.; Nuzzo, D.; Caruana, L.; Messina, E.; Scafidi, V.; Di Carlo, M. Curcumin induces apoptosis in human neuroblastoma cells via inhibition of AKT and Foxo3a nuclear translocation. Free Radic. Res. 2014, 48, 1397–1408. [Google Scholar] [CrossRef]
- Inoue, K.; Kuwana, H.; Shimamura, Y.; Ogata, K.; Taniguchi, Y.; Kagawa, T.; Horino, T.; Takao, T.; Morita, T.; Sasaki, S.; et al. Cisplatin-induced macroautophagy occurs prior to apoptosis in proximal tubules in vivo. Clin. Exp. Nephrol. 2010, 14, 112–122. [Google Scholar] [CrossRef]
- Huber, T.B.; Edelstein, C.L.; Hartleben, B.; Inoki, K.; Jiang, M.; Koya, D.; Kume, S.; Lieberthal, W.; Pallet, N.; Quiroga, A.; et al. Emerging role of autophagy in kidney function, diseases and aging. Autophagy 2012, 8, 1–12. [Google Scholar] [CrossRef]
- Jiang, M.; Liu, K.; Luo, J.; Dong, Z. Autophagy is a renoprotective mechanism during in vitro hypoxia and in vivo ischemia-reperfusion injury. Am. J. Pathol. 2010, 176, 1181–1192. [Google Scholar] [CrossRef]
- Mizushima, N.; Yoshimori, T.; Levine, B. Methods in mammalian autophagy research. Cell 2010, 140, 313–326. [Google Scholar] [CrossRef]
- Sun, Q.; Chen, X.; Ma, J.; Peng, H.; Wang, F.; Zha, X.; Wang, Y.; Jing, Y.; Yang, H.; Chen, R. Mammalian target of rapamycin up-regulation of pyruvate kinase isoenzyme type M2 is critical for aerobic glycolysis and tumor growth. Proc. Natl. Acad. Sci. USA 2011, 108, 4129–4134. [Google Scholar] [CrossRef]
- Gabriele, F.; Martinelli, C.; Comincini, S. Prostate cancer cells at a therapeutic gunpoint of the autophagy process. J. Cancer Metastasis Treat 2018, 4, 1–17. [Google Scholar] [CrossRef]
- Moore, J.; Megaly, M.; MacNeil, A.J.; Klentrou, P.; Tsiani, E. Rosemary extract reduces Akt/mTOR/p70S6K activation and inhibits proliferation and survival of A549 human lung cancer cells. Biomed. Pharmacother. 2016, 83, 725–732. [Google Scholar] [CrossRef]
- He, C.; Klionsky, D.J. Regulation mechanisms and signaling pathways of autophagy. Annu. Rev. Genet. 2009, 43, 67–93. [Google Scholar] [CrossRef]
- Zhang, X.H.; Wu, H.; Tang, S.; Li, Q.N.; Xu, J.; Zhang, M.; Su, Y.N.; Yin, B.; Zhao, Q.L.; Kemper, N.; et al. Apoptosis in response to heat stress is positively associated with heat-shock protein 90 expression in chicken myocardial cells in vitro. J. Vet. Sci. 2017, 18, 129–140. [Google Scholar] [CrossRef]
- Bellot, G.; Garcia-Medina, R.; Gounon, P.; Chiche, J.; Roux, D.; Pouysségur, J.; Mazure, N.M. Hypoxia-induced autophagy is mediated through hypoxia-inducible factor induction of BNIP3 and BNIP3L via their BH3 domains. Mol. Cell Biol. 2009, 29, 2570–2581. [Google Scholar] [CrossRef] [PubMed]
- Hecker, J.G.; McGarvey, M. Heat shock proteins as biomarkers for the rapid detection of brain and spinal cord ischemia: A review and comparison to other methods of detection in thoracic aneurysm repair. Cell Stress Chaperones 2011, 16, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Hecker, J.G.; Sundram, H.; Zou, S.; Praestgaard, A.; Bavaria, J.E.; Ramchandren, S.; McGarvey, M. Heat shock proteins HSP70 and HSP27 in the cerebral spinal fluid of patients undergoing thoracic aneurysm repair correlate with the probability of postoperative paralysis. Cell Stress Chaperones 2008, 13, 435–446. [Google Scholar] [CrossRef]
- Sreedharan, R.; Van Why, S.K. Heat shock proteins in the kidney. Pediatr. Nephrol. 2016, 31, 1561–1570. [Google Scholar] [CrossRef]
- Chiosis, G. Targeting chaperones in transformed systems: A focus on Hsp90 and cancer. Expert Opin. Ther. Targets 2006, 10, 37–50. [Google Scholar] [CrossRef]
- Hipp, M.S.; Park, S.H.; Hartl, F.U. Proteostasis impairment in proteinmisfolding and aggregation diseases. Trends Cell Biol. 2014, 24, 506–514. [Google Scholar] [CrossRef]
- Taipale, M.; Jarosz, D.F.; Lindquist, S. HSP90 at the hub of protein homeostasis: Emerging mechanistic insights. Nat. Rev. Mol. Cell Biol. 2010, 11, 515–528. [Google Scholar] [CrossRef]
- Pearl, L.H. The HSP90 molecular chaperone-an enigmaticATPase. Biopolymers 2016, 105, 594–607. [Google Scholar] [CrossRef]
- Acunzo, J.; Katsogiannou, M.; Rocchi, P. Small heat shock proteins HSP27 (HspB1), αB-crystallin (HspB5) and HSP22 (HspB8) as regulators of cell death. Int. J. Biochem. Cell Biol. 2012, 44, 1622–1631. [Google Scholar] [CrossRef]
- Chatterjee, M.; Andrulis, M.; Stühmer, T.; Müller, E.; Hofmann, C.; Steinbrunn, T.; Heimberger, T.; Schraud, H.; Kressmann, S.; Einsele, H.; et al. The PI3K/Akt signaling pathway regulates the expression of Hsp70, which critically contributes to Hsp90-chaperone function and tumor cell survival in multiple myeloma. Haematologica 2013, 98, 1132–1141. [Google Scholar] [CrossRef]
- Liang, J.; Cao, R.; Wang, X.; Zhang, Y.; Wang, P.; Gao, H.; Li, C.; Yang, F.; Zeng, R.; Wei, P.; et al. Mitochondrial PKM2 regulates oxidative stress-induced apoptosis by stabilizing Bcl2. Cell Res. 2017, 27, 329–351. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, S.; Tu, J.; Cai, W.; Xu, Q. HSP90 inhibits apoptosis and promotes growth by regulating HIF-1α abundance in hepatocellular carcinoma. Int. J. Mol. Med. 2016, 37, 825–835. [Google Scholar] [CrossRef] [PubMed]
- Dey, P.; Kundu, A.; Sachan, R.; Park, J.H.; Ahn, M.Y.; Yoon, K.; Lee, J.; Kim, N.D.; Kim, I.S.; Lee, B.M.; et al. PKM2 knockdown induces autophagic cell death via AKT/mTOR pathway in human prostate cancer cells. Cell Physiol. Biochem. 2019, 52, 1535–1552. [Google Scholar] [CrossRef] [PubMed]
- Chu, B.; Wang, J.; Wang, Y.; Yang, G. Knockdown of PKM2 induces apoptosis and autophagy in human A549 alveolar adenocarcinoma cells. Mol. Med. Rep. 2015, 1, 4358–4363. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Feng, G.; Bao, G.; Xu, G.; Sun, Y.; Li, W.; Wang, L.; Chen, J.; Jin, H.; Cui, Z. Nuclear translocation of PKM2 modulates astrocyte proliferation via p27 and β-catenin pathway after spinal cord injury. Cell Cycle 2015, 14, 2609–2618. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Bosch-Marce, M.; Shimoda, L.A.; Tan, Y.S.; Baek, J.H.; Wesley, J.B.; Gonzalez, F.J.; Semenza, G.L. Mitochondrial autophagy is an HIF-1-dependent adaptive metabolic response to hypoxia. J. Biol. Chem. 2008, 283, 10892–10903. [Google Scholar] [CrossRef]
- Lu, H.H.; Kao, S.Y.; Liu, T.Y.; Liu, S.T.; Huang, W.P.; Chang, K.W.; Lin, S.C. Areca nut extract induced oxidative stress and upregulated hypoxia inducing factor leading to autophagy in oral cancer cells. Autophagy 2010, 6, 725–737. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine; Clarendon: Oxford, UK, 2007. [Google Scholar]
- Vysakh, A.; Raji, N.R.; Suma, D.; Jayesh, K.; Jyothis, M.; Latha, M.S. Role of antioxidant defence, renal toxicity markers and inflammatory cascade in disease progression of acute pyelonephritis in experimental rat model. Microb. Pathog. 2017, 109, 189–194. [Google Scholar] [CrossRef]
- Smolka, M.B.; Zoppi, C.C.; Alves, A.A.; Silveira, L.R.; Marangoni, S.; Pereira-Da-Silva, L.; Novello, J.C.; Macedo, D.V. HSP72 as a complementary protection against oxidative stress induced by exercise in the soleus muscle of rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 279, 1539–1545. [Google Scholar] [CrossRef]
- Henstridge, D.C.; Febbraio, M.A.; Hargreaves, M. Heat shock proteins and exercise adaptations. Our knowledge thus far and the road still ahead. J. Appl. Physiol. 2016, 120, 683–691. [Google Scholar] [CrossRef]
- Atalay, M.; Oksala, N.K.; Laaksonen, D.E.; Khanna, S.; Nakao, C.; Lappalainen, J.; Roy, S.; Hänninen, O.; Sen, C.K. Exercise training modulates heat shock protein response in diabetic rats. J. Appl. Physiol. 2004, 97, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, M.; Su, Y.; Wang, Z.; Zhao, Q.; Zhu, H.; Qian, Z.; Xu, J.; Tang, S.; Wu, D.; et al. Inhibition of heat stress-related apoptosis of chicken myocardial cells through inducing Hsp90 expression by aspirin administration in vivo. Br. Poult. Sci. 2018, 59, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Tang, S.; Yin, B.; Sun, J.; Song, E.; Bao, E. Co-enzyme Q10 and acetyl salicylic acid enhance Hsp70 expression in primary chicken myocardial cells to protect the cells during heat stress. Mol. Cell Biochem. 2017, 435, 73–86. [Google Scholar] [CrossRef] [PubMed]
- He, C.L.; Bian, Y.Y.; Xue, Y.; Liu, Z.X.; Zhou, K.Q.; Yao, C.F.; Lin, Y.; Zou, H.F.; Luo, F.X.; Qu, Y.Y.; et al. Pyruvate kinase M2 activates mTORC1 by phosphorylating AKT1S1. Sci. Rep. 2016, 6, 21524. [Google Scholar] [CrossRef]
- Biggs, W.H.; Meisenhelder, J.; Hunter, T.; Cavanee, W.K.; Arden, K.C. Protein kinase B/Akt-mediated phosphorylation promotes nuclear exclusion of the winged helix transcription factor FKHR1. Proc. Natl. Acad. Sci. USA 1999, 96, 7421–7426. [Google Scholar] [CrossRef]
- Brunet, A.; Bonni, A.; Zigmond, M.J.; Lin, M.Z.; Juo, P.; Hu, L.S.; Anderson, M.J.; Arden, K.C.; Blenis, J.; Greenberg, M.E. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 1999, 6, 857–868. [Google Scholar] [CrossRef]
- Pugazhenthi, S.; Nesterova, A.; Sable, C.; Heidenreich, K.A.; Boxer, L.M.; Heasley, L.E.; Reusch, J.E. Akt/protein kinase B up-regulates Bcl-2 expression through cAMP-response element-binding protein. J. Biol. Chem. 2000, 275, 10761–10766. [Google Scholar] [CrossRef]
- Mazurek, S.; Boschek, C.B.; Hugo, F.; Eigenbrodt, E. Pyruvate kinase type M2 and its role in tumor growth and spreading. Semin. Cancer Biol. 2005, 15, 300–308. [Google Scholar] [CrossRef]
- Bramanti, V.; Tomassoni, D.; Avitabile, M.; Amenta, F.; Avola, R. Biomarkers of glial cell proliferation and differentiation in culture. Front. Biosci. 2010, 2, 558–570. [Google Scholar] [CrossRef]
- Alao, J.P. The regulation of cyclin D1 degradation: Roles in cancer development and the potential for therapeutic invention. Mol. Cancer 2007, 6, 24. [Google Scholar] [CrossRef]
- Takahashi-Yanaga, F.; Sasaguri, T. GSK-3beta regulates cyclin D1 expression: A new target for chemotherapy. Cell Signal. 2008, 20, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Yang, Y.; Duan, J.; Wu, P.; Jiang, Q.; Xu, C. PTEN-regulated AKT/FoxO3a/Bim signaling contributes to reactive oxygen species-mediated apoptosis in selenite-treated colorectal cancer cells. Cell Death Dis. 2013, 4, e481. [Google Scholar] [CrossRef]
- Park, Y.S.; Kim, D.J.; Koo, H.; Jang, S.H.; You, Y.M.; Cho, J.H.; Yang, S.J.; Yu, E.S.; Jung, Y.; Lee, D.C.; et al. AKT-induced PKM2 phosphorylation signals for IGF-1-stimulated cancer cell growth. Oncotarget 2016, 7, 48155–48167. [Google Scholar] [CrossRef] [PubMed]
- Klumpen, E.; Hoffschröer, N.; Zeis, B.; Gigengack, U.; Dohmen, E.; Paul, R.J. Reactive oxygen species (ROS) and the heat stress response of Daphnia pulex: ROS-mediated activation of hypoxia-inducible factor 1 (HIF-1) and heat shock factor 1 (HSF-1) and the clustered expression of stress genes. Biol. Cell. 2017, 109, 39–64. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Liu, R.; Li, J.; Mao, J.; Lei, Y.; Wu, J.; Zeng, J.; Zhang, T.; Wu, H.; Chen, L.; et al. Quercetin induces protective autophagy in gastric cancer cells: Involvement of Akt-mTOR- and hypoxia-induced factor 1α-mediated signaling. Autophagy 2011, 7, 966–978. [Google Scholar] [CrossRef] [PubMed]
- Palsson-McDermott, E.M.; Curtis, A.M.; Goel, G.; Lauterbach, M.A.; Sheedy, F.J.; Gleeson, L.E.; van den Bosch, M.W.; Quinn, S.R.; Domingo Fernandez, R.; Johnston, D.G.; et al. Pyruvate kinase M2 regulates Hif-1α activity and IL-1β induction and is a critical determinant of the Warburg effect in LPS-activated macrophages. Cell Metab. 2015, 21, 65–80. [Google Scholar] [CrossRef]
- Wang, G.L.; Jiang, B.H.; Rue, E.A.; Semenza, G.L. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc. Natl. Acad. Sci. USA 1995, 92, 5510–5514. [Google Scholar] [CrossRef]
- Luo, W.; Hu, H.; Chang, R.; Zhong, J.; Knabel, M.; O’Meally, R.; Cole, R.N.; Pandey, A.; Semenza, G.L. Pyruvate kinase M2 is a PHD3-stimulated coactivator for hypoxia-inducible factor 1. Cell 2011, 145, 732–744. [Google Scholar] [CrossRef]
- Ahmad, N.; Wang, Y.; Haider, K.H.; Wang, B.; Pasha, Z.; Uzun, O.; Ashraf, M. Cardiac protection by mitoKATP channels is dependent on Akt transloca- tion from cytosol to mitochondria during late preconditioning. Am. J. Physiol. Heart Circ. Physiol. 2006, 290, H2402–H2408. [Google Scholar] [CrossRef]
- Mejía-García, T.A.; Portugal, C.C.; Encarnação, T.G.; Prado, M.A.; Paes-de-Carvalho, R. Nitric oxide regulates AKT phosphorylation and nuclear translocation in cultured retinal cells. Cell Signal. 2013, 25, 2424–2439. [Google Scholar] [CrossRef]
- Xuan Nguyen, T.L.; Choi, J.W.; Lee, S.B.; Ye, K.; Woo, S.D.; Lee, K.H.; Ahn, J.Y. Akt phosphorylation is essential for nuclear translocation and retention in NGF-stimulated PC12 cells. Biochem. Biophys. Res. Commun. 2006, 349, 789–798. [Google Scholar] [CrossRef] [PubMed]
- Xavier, I.J.; Mercier, P.A.; McLoughlin, C.M.; Ali, A.; Woodgett, J.R.; Ovsenek, N. Glycogen synthase kinase 3 beta negatively regulates both DNA-binding and transcriptional activities of heat shock factor 1. J. Biol. Chem. 2000, 275, 29147–29152. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, B.; Yang, B.; Zhu, J.; Wu, J.; Sha, J.; Sun, J.; Bao, E.; Zhang, X. Hsp90 Relieves Heat Stress-Induced Damage in Mouse Kidneys: Involvement of Antiapoptotic PKM2-AKT and Autophagic HIF-1α Signaling. Int. J. Mol. Sci. 2020, 21, 1646. https://doi.org/10.3390/ijms21051646
Chen B, Yang B, Zhu J, Wu J, Sha J, Sun J, Bao E, Zhang X. Hsp90 Relieves Heat Stress-Induced Damage in Mouse Kidneys: Involvement of Antiapoptotic PKM2-AKT and Autophagic HIF-1α Signaling. International Journal of Molecular Sciences. 2020; 21(5):1646. https://doi.org/10.3390/ijms21051646
Chicago/Turabian StyleChen, Bixia, Bo Yang, Jie Zhu, Jiaxin Wu, Junzhou Sha, Jiarui Sun, Endong Bao, and Xiaohui Zhang. 2020. "Hsp90 Relieves Heat Stress-Induced Damage in Mouse Kidneys: Involvement of Antiapoptotic PKM2-AKT and Autophagic HIF-1α Signaling" International Journal of Molecular Sciences 21, no. 5: 1646. https://doi.org/10.3390/ijms21051646
APA StyleChen, B., Yang, B., Zhu, J., Wu, J., Sha, J., Sun, J., Bao, E., & Zhang, X. (2020). Hsp90 Relieves Heat Stress-Induced Damage in Mouse Kidneys: Involvement of Antiapoptotic PKM2-AKT and Autophagic HIF-1α Signaling. International Journal of Molecular Sciences, 21(5), 1646. https://doi.org/10.3390/ijms21051646




