Overexpression of the High-Affinity Nitrate Transporter OsNRT2.3b Driven by Different Promoters in Barley Improves Yield and Nutrient Uptake Balance
Abstract
:1. Introduction
2. Results
2.1. Confirmation of Transgenic Barley Lines with Real-Time Quantitative PCR and Western Blotting
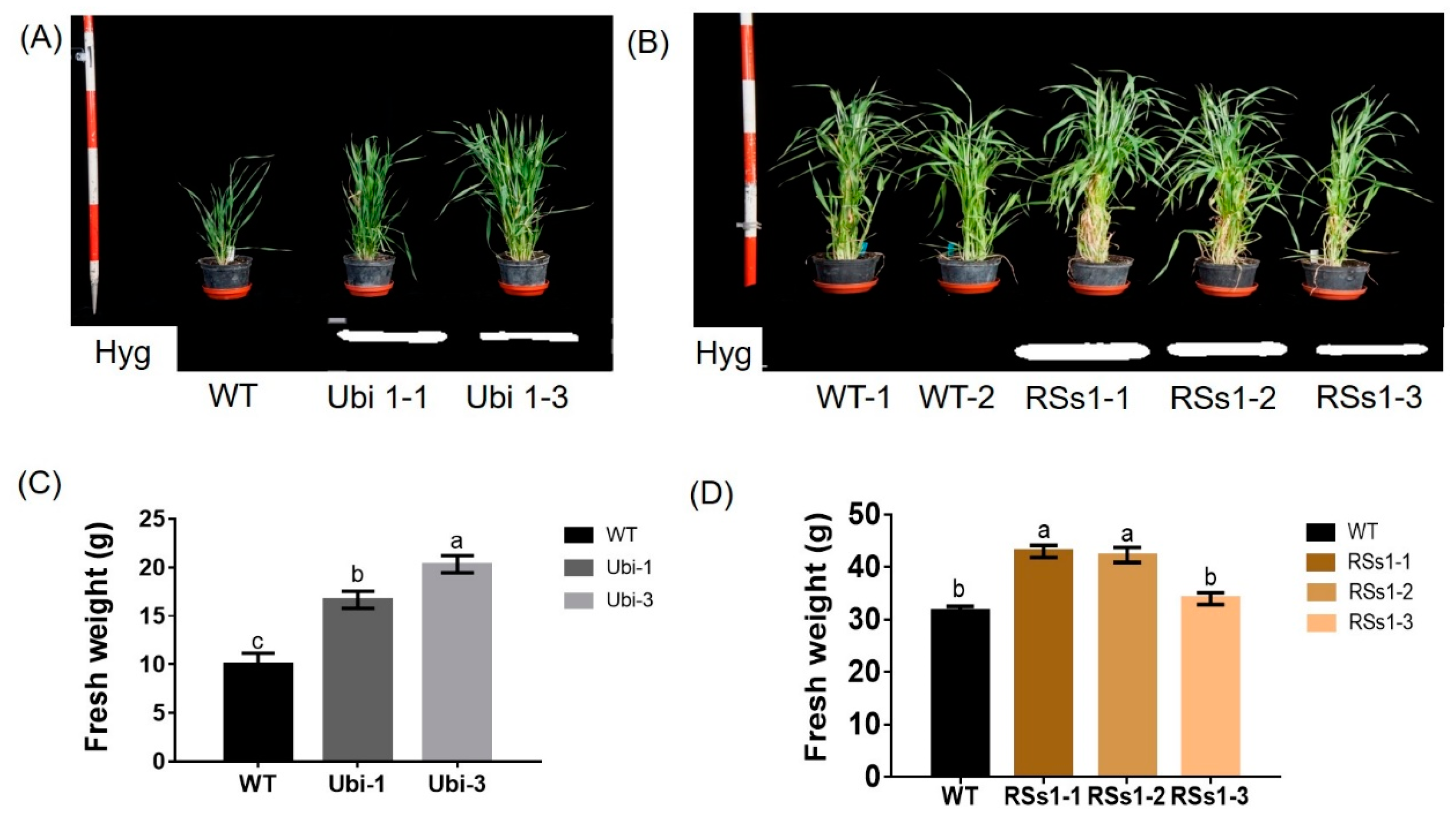
2.2. Phenotype of Transgenic Barley Lines during the Vegetative Growth Stage
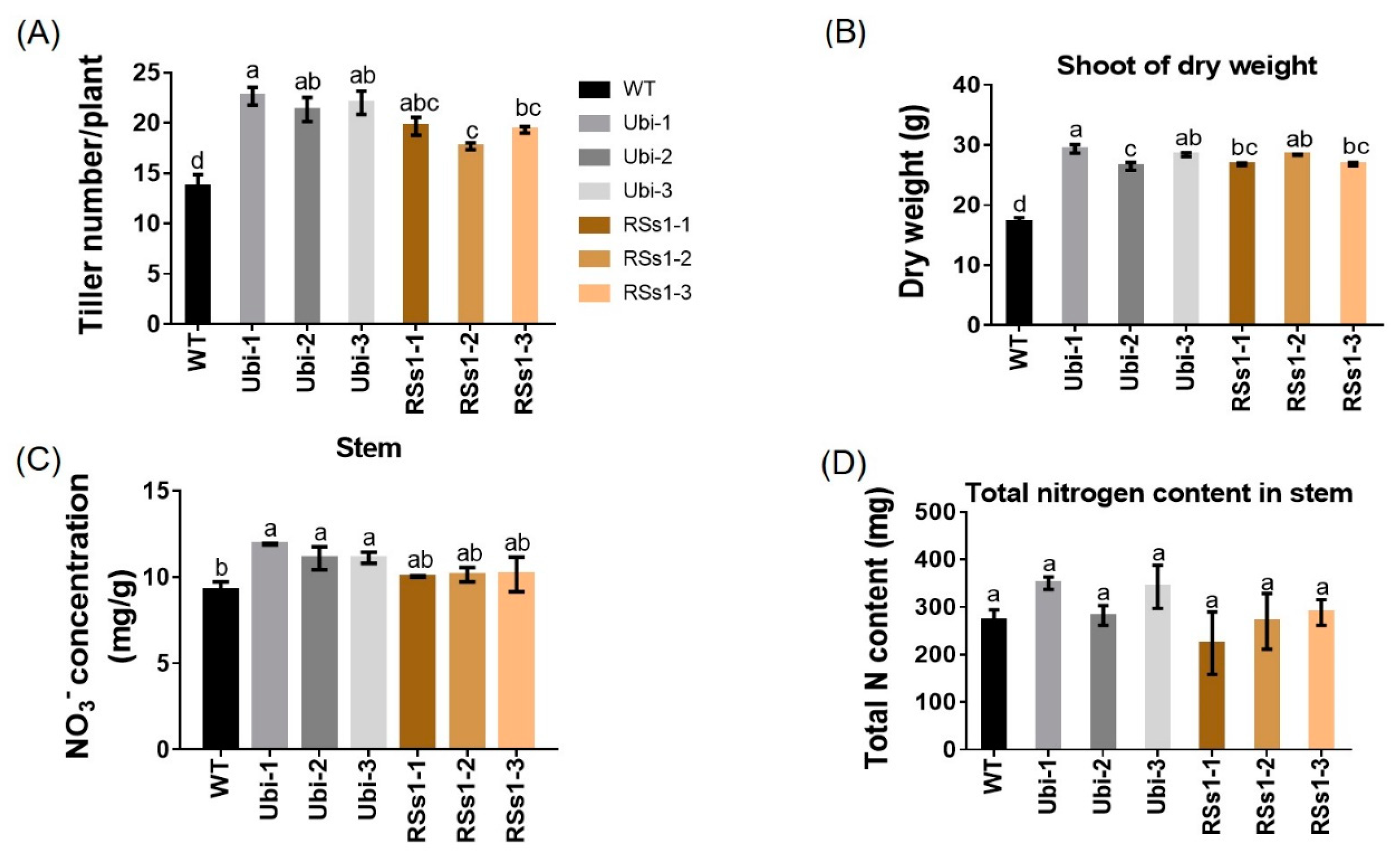
2.3. Characterization of Transgenic Barley during the Reproductive Growth Stage
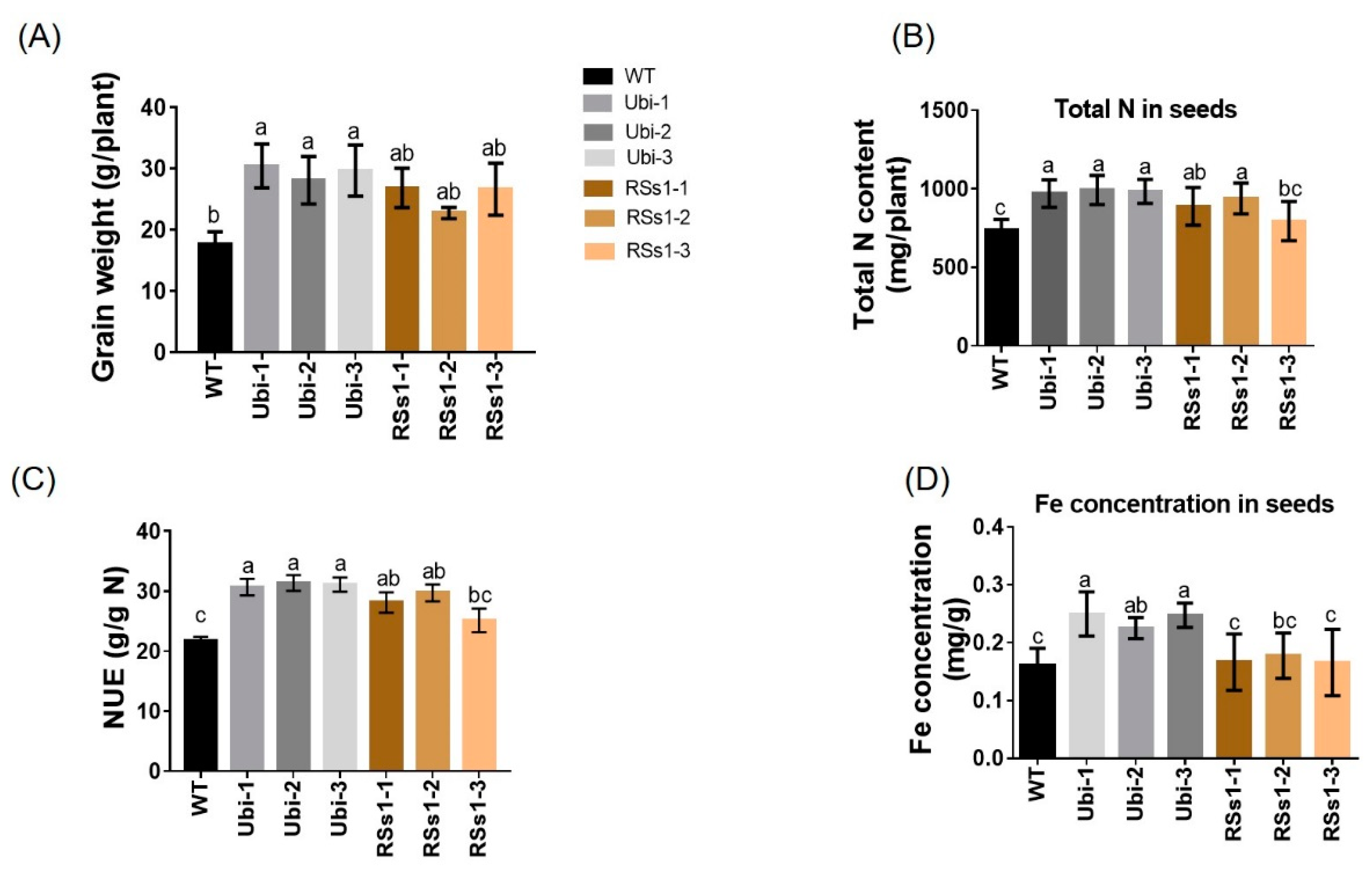
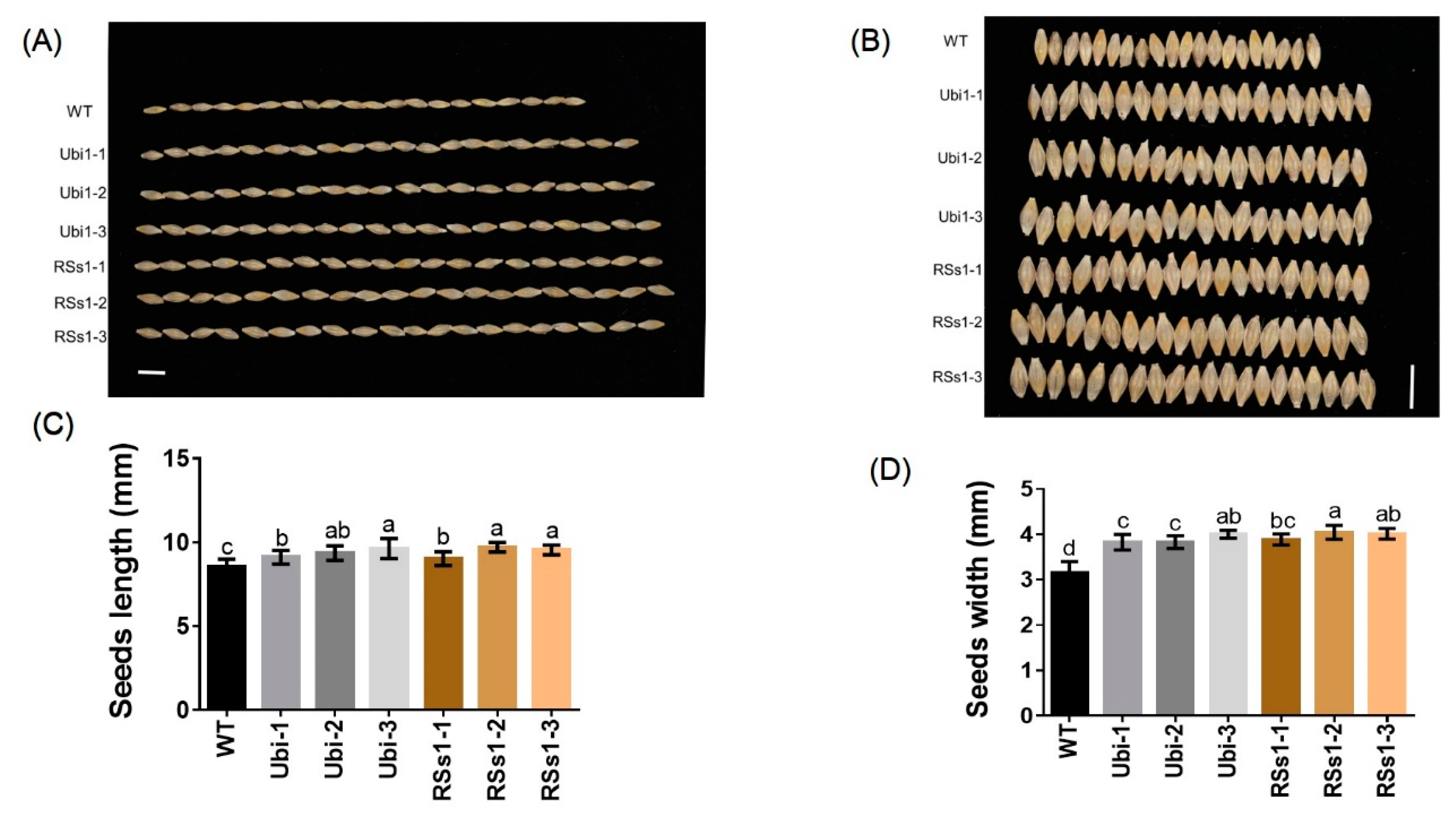
2.4. Plant Development and Metabolism at Maturity
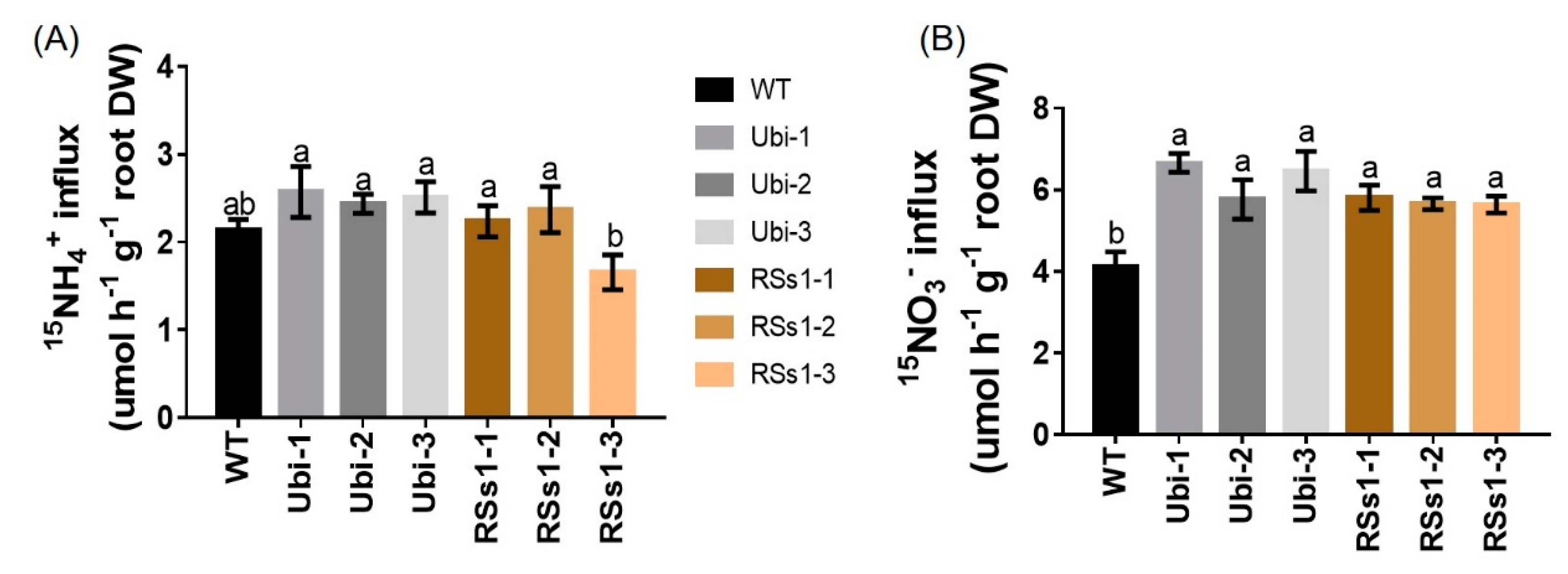
2.5. NH4+ and NO3− Influx Rates in WT and OsNRT2.3b Transgenic Barley
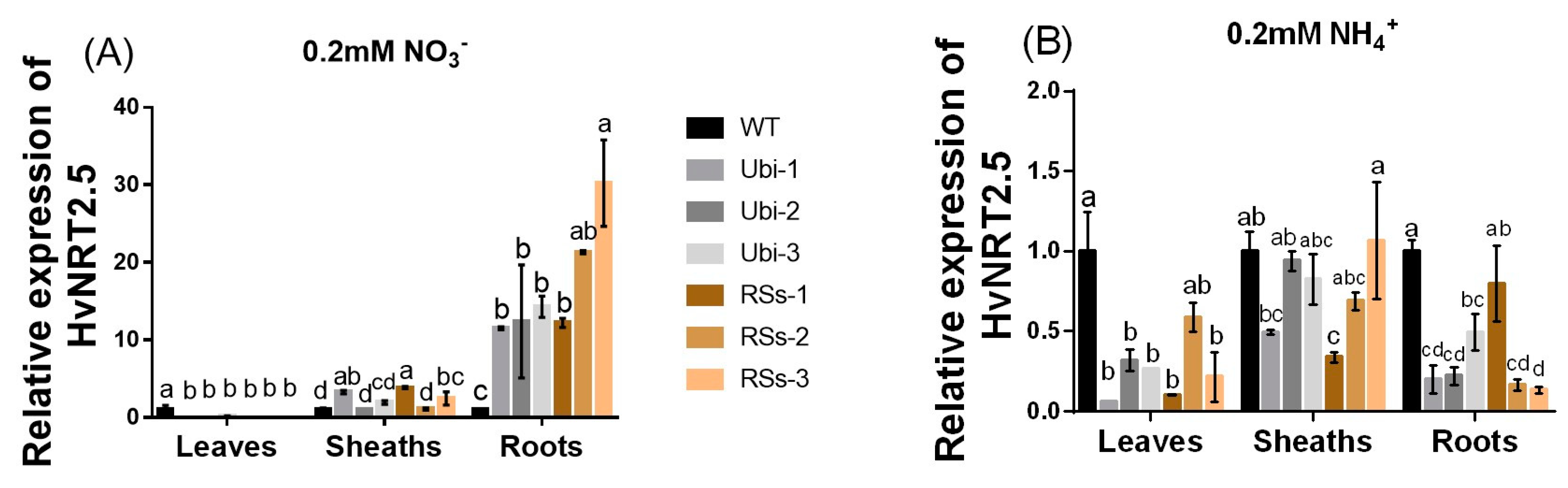
2.6. The Effect of Different N Treatments on Gene Expression, and Total N, P, and Other Fe Concentration in Different Plant Parts
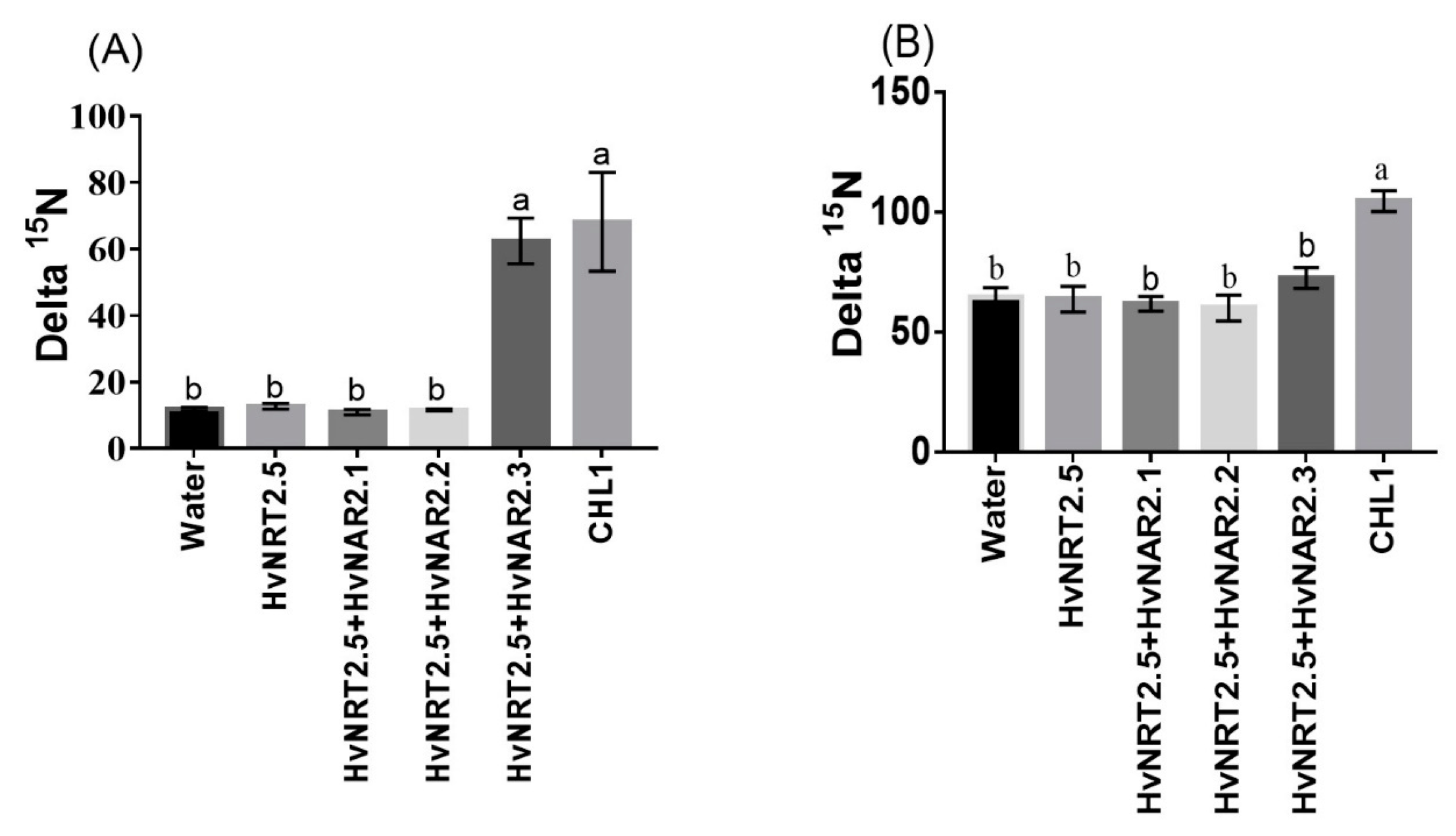
2.7. The Characteristics of HvNRT2.5, a Gene Homologous to OsNRT2.3b
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. Growth Conditions
4.3. Western Blotting
4.4. DNA/RNA Extraction and qPCR Analysis
4.5. Southern Blot Analysis
4.6. Determination of 15N-NH4+/NO3− Influx Rate in Different Barley Lines
4.7. Analysis of Agronomic Traits
4.8. Measurements of Dry Weight, Nitrate, Total N, and Metal Ion Accumulation
4.9. Cloning and cRNA Synthesis of HvNRT2.5, HvNAR2.1, HvNAR2.2, HvNAR2.3, and AtNPF6.3 (CHL1) and Nitrate Uptake Assay in Xenopus Laevis Oocytes
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Haynes, R.J.; Goh, K.M. Ammonium and nitrate nutrition of plants. Biol. Rev. 1978, 53, 465–510. [Google Scholar] [CrossRef]
- Kronzucker, H.J.; Glass, A.D.M.; Siddiqi, M.Y.; Kirk, G.J.D. Comparative kinetic analysis of ammonium and nitrate acquisition by tropical lowland rice: Implications for rice cultivation and yield potential. New Phytol. 2000, 145, 471–476. [Google Scholar] [CrossRef] [Green Version]
- Miller, A.J.; Cramer, M.D. Root nitrogen acquisition and assimilation. Plant Soil 2005, 274, 1–36. [Google Scholar] [CrossRef]
- Coruzzi, G.; Bush, D.R. Nitrogen and carbon nutrient and metabolite signaling in plants. Plant Physiol. 2001, 125, 61–64. [Google Scholar] [CrossRef] [Green Version]
- Crawford, N.M.; Glass, A.D.M. Molecular and physiological aspects of nitrate uptake in plants. Trends Plant Sci. 1998, 3, 389–395. [Google Scholar] [CrossRef]
- Siddiqi, M.Y.; Glass, A.D.; Ruth, T.J.; Rufty, T.W. Studies of the uptake of nitrate in Barley: I. kinetics of 13NO3− influx. Plant Physiol. 1990, 93, 1426–1432. [Google Scholar] [CrossRef] [Green Version]
- Forde, B.G. Nitrate transporters in plants: Structure, function and regulation. Biochim. Biophys. Acta 2000, 1465, 219–235. [Google Scholar] [CrossRef]
- Fan, X.R.; Shen, Q.R.; Ma, Z.; Zhu, H.; Yin, X.; Miller, A.J. A comparison of nitrate transport in four different rice (Oryza sativa L.) cultivars. Sci. China Ser. C 2005, 48, 897–911. [Google Scholar]
- Miller, A.J.; Fan, X.R.; Orsel, M.; Smith, S.J.; Wells, D.M. Nitrate transport and signaling. J. Exp. Bot. 2007, 58, 2297–2306. [Google Scholar] [CrossRef]
- Fan, X.R.; Naz, M.; Fan, X.R.; Wei, X.; Miller, A.J.; Xu, G.H. Plant nitrate transporter from gene function to application. J. Exp. Bot. 2016, 168, 2463–2475. [Google Scholar] [CrossRef] [PubMed]
- Rayle, D.L.; Cleland, R.E. The Acid Growth Theory of auxin induced cell elongation is alive and well. Plant Physiol. 1992, 99, 1271–1274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forde, B.G. The role of long-distance signalling in plant responses to nitrate and other nutrients. J. Exp. Bot. 2002, 53, 39–43. [Google Scholar] [PubMed]
- Tsay, Y.F.; Chiu, C.C.; Tsai, C.B.; Ho, C.H.; Hsu, P.K. Nitrate transporters and peptide transporters. FEBS Lett. 2007, 581, 2290–2300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, K.H.; Tsay, Y.F. Switching between the two action modes of the dual-affinity nitrate transporter CHL1 by phosphorylation. EMBO J. 2003, 22, 1005–1013. [Google Scholar] [CrossRef] [Green Version]
- Guo, F.Q.; Wang, R.; Chen, M.; Crawford, N.M. The Arabidopsis dual-affinity nitrate transporter gene AtNRT1.1 (CHL1) is activated and functions in nascent organ development during vegetative and reproductive growth. Plant Cell 2001, 13, 1761–1777. [Google Scholar] [CrossRef] [Green Version]
- Gojon, A.; Krouk, G.; Perrine-Walker, F.; Laugier, E. Nitrate transceptor(s) in plants. J. Exp. Bot. 2011, 62, 2299–2308. [Google Scholar] [CrossRef] [Green Version]
- Tsay, Y.F.; Schroeder, J.I.; Feldmann, K.A.; Crawford, N.M. The herbicide sensitivity gene CHL1 of Arabidopsis encodes a nitrate-inducible nitrate transporter. Cell 1993, 72, 705–713. [Google Scholar] [CrossRef]
- Ho, C.H.; Lin, S.H.; Hu, H.C.; Tsay, Y.F. CHL1 functions as a nitrate sensor in Plant. Cell 2009, 138, 1184–1194. [Google Scholar] [CrossRef] [Green Version]
- Orsel, M.; Chopin, F.; Leleu, O.; Smith, S.J.; Krapp, A.; Daniel-Vedele, F.; Miller, A.J. Characterization of a two-component high-affinity nitrate uptake system in Arabidopsis. Physiology and protein-protein interaction. Plant Physiol. 2006, 142, 1304–1317. [Google Scholar] [CrossRef] [Green Version]
- Okamoto, M.; Kumar, A.; Li, W.B.; Wang, Y.; Siddiqi, M.Y.; Crawford, N.M.; Glass, A.D.M. High-affinity nitrate transport in roots of Arabidopsis depends on expression of the NAR2-like gene AtNRT3.1. Plant Physiol. 2006, 140, 1036–1046. [Google Scholar] [CrossRef] [Green Version]
- Yong, Z.; Kotur, Z.; Glass, A.D. Characterization of an intact two component high-affinity nitrate transporter from Arabidopsis roots. Plant J. 2010, 63, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Laugier, E.; Bouguyon, E.; Mauriès, A.; Tillard, P.; Gojon, A.; Lejay, L. Regulation of high-affinity nitrate uptake in roots of Arabidopsis depends predominantly on posttranscriptional control of the NRT2.1/NAR2.1 transport system. Plant Physiol. 2012, 158, 1067–1078. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kiba, T.; Feria-Bourrellier, A.-B.; Lafouge, F.; Lezhneva, L.; Boutet-Mercey, S.; Orsel, M.; Bréhaut, V.; Miller, A.; Daniel-Vedele, F.; Sakakibara, H.; et al. The Arabidopsis nitrate transporter NRT2.4 plays a double role in roots and shoots of nitrogen-starved plants. Plant Cell Online 2012, 24, 245–258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsu, P.K.; Tsay, Y.F. Two phloem nitrate transporters, NRT1.11 and NRT1.12, are important for redistributing xylem-borne nitrate to enhance plant growth. Plant Physiol. 2013, 163, 844–856. [Google Scholar] [CrossRef] [Green Version]
- Fan, X.R.; Tang, Z.; Tan, Y.W.; Zhang, Y.; Luo, B.B.; Yang, M.; Lian, X.M.; Shen, Q.; Miller, A.J.; Xu, G. Over expression of a pH sensitive nitrate transporter in rice increases crop yields. Proc. Nat. Acad. Sci. USA 2016, 113, 7118–7123. [Google Scholar] [CrossRef] [Green Version]
- Tang, Z.; Fan, X.R.; Li, Q.; Feng, H.M.; Miller, A.J.; Shen, Q.R.; Shen, Q.; Xu, G. Knockdown of a rice stellar nitrate transporter alters long-distance translocation but not root influx. Plant Physiol. 2012, 160, 2052–2063. [Google Scholar] [CrossRef] [Green Version]
- Araki, R.; Hasegawa, H. Expression of rice (Oryza sativa L.) genes involved in high-affinity nitrate transport during the period of nitrate induction. Breed. Sci. 2006, 56, 295–302. [Google Scholar] [CrossRef] [Green Version]
- Cai, C.; Wang, J.Y.; Zhu, Y.G.; Shen, Q.R.; Li, B.; Tong, Y.P.; Li, Z.S. Gene structure and expression of the high-affinity nitrate transport system in rice roots. J. Integr. Plant Biol. 2008, 50, 443–451. [Google Scholar] [CrossRef]
- Wei, J.; Zheng, Y.; Feng, H.M.; Qu, H.Y.; Fan, X.R.; Yamaji, N.; Ma, J.F.; Xu, G.H. OsNRT2.4 encodes a dual-affinity nitrate transporter and functions in nitrate-regulated root growth and nitrate distribution in rice. J. Exp. Bot. 2018, 69, 1095–1107. [Google Scholar] [CrossRef]
- Yan, M.; Fan, X.R.; Feng, H.M.; Miller, A.J.; Shen, Q.R.; Xu, G.H. Rice OsNAR2.1 interacts with OsNRT2.1, OsNRT2.2 and OsNRT2.3a nitrate transporters to provide uptake over high and low concentration ranges. Plantcell Environ. 2011, 34, 1360–1372. [Google Scholar] [CrossRef]
- Morrell, P.L.; Clegg, M.T. Genetic evidence for a second domestication of barley (Hordeum vulgare) east of the Fertile Crescent. Proc. Nat. Acad. Sci. USA 2007, 104, 3289–3294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anbessa, Y.; Juskiw, P.; Good, A.; Nyachiro, J.; Helm, J. Genetic variability in nitrogen use efficiency of spring barley. Crop Sci. 2009, 49, 1259–1269. [Google Scholar] [CrossRef]
- Mayer, K.F.X.; Waugh, R.; Brown, J.W.; Schulman, A.; Langridge, P.; Platzer, M.; Fincher, G.B.; Muehlbauer, G.J.; Sato, K.; Close, T.J.; et al. A physical, genetic and functional sequence assembly of the barley genome. Nature 2012, 491, 711–716. [Google Scholar] [PubMed]
- Shcherbak, I.; Millar, N.; Robertson, G.P. Global metanalysis of the nonlinear response of soil nitrous oxide (N2O) emissions to fertilizer nitrogen. Proc. Nat. Acad. Sci. USA 2014, 111, 9199–9204. [Google Scholar] [CrossRef] [Green Version]
- Hinchliffe, A.; Harwood, W.A. Agrobacterium-mediated transformation of barley immature embryos. Methods Mol. Biol. 2018, 1900, 115–126. [Google Scholar]
- Luo, B.B.; Chen, J.G.; Zhu, L.L.; Liu, S.H.; Li, B.; Lu, H.; Ye, G.Y.; Xu, G.H.; Fan, X.R. Overexpression of a high-affinity transporter OsNRT2.1 increase yield and manganese accumulation in rice under alternating wet and dry condition. Front. Plant Sci. 2018, 9, 1192. [Google Scholar] [CrossRef] [Green Version]
- Tong, Y.; Zhou, J.J.; Li, Z.; Miller, A.J. A two-component high-affinity nitrate uptake system in barley. Plant J. 2005, 41, 442–450. [Google Scholar] [CrossRef]
- Vidmar, J.J.; Zhuo, D.; Siddiqi, M.Y.; Glass, A.D.M. Isolation and characterization of HvNRT2.3 and HvNRT2.4, cDNAs encoding high-Affinity nitrate transporters from roots of barley. Plant Physiol. 2000, 122, 783–792. [Google Scholar] [CrossRef] [Green Version]
- Xu, G.H.; Fan, X.R.; Miller, A.J. Plant nitrogen assimilation and use efficiency. Annu. Rev. Plant Biol. 2012, 63, 153–182. [Google Scholar] [CrossRef] [Green Version]
- Fu, Y.; Yi, H.; Bao, J.; Gong, J. LeNRT2. 3 functions in nitrate acquisition and long-distance transport in tomato. FEBS Lett. 2015, 589, 1072–1079. [Google Scholar] [CrossRef] [Green Version]
- Li, B.; Xin, W.; Sun, S.; Shen, Q.; Xu, G. Physiological and molecular responses of nitrogen-starved rice plants to re-supply of different nitrogen sources. Plant Soil 2006, 287, 145–159. [Google Scholar] [CrossRef]
- Feng, H.; Yan, M.; Fan, X.; Li, B.; Shen, Q.; Miller, A.J.; Xu, G. Spatial expression and regulation of rice high-affinity nitrate transporters by nitrogen and carbon status. J. Exp. Bot. 2011, 62, 2319–2332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Léran, S.; Varala, K.; Boyer, J.C.; Chiurazzi, M.; Crawford, N.; Daniel-Vedele, F.; David, L.; Dickstein, R.; Fernandez, E.; Forde, B.; et al. A unified nomenclature of NITRATE TRANSPORTER 1/PEPTIDE TRANSPORTER family members in plants. Trends Plant Sci. 2015, 19, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Wong, J.; Su, T.; Beatty, H.P.; Good, G.A. Identification of nitrogen use efficiency genes in barley: Searching for QTLs controlling complex physiological traits. Front. Plant Sci. 2016, 7, 1587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fischer, J.J.; Beatty, P.H.; Good, A.G.; Muench, D.G. Manipulation of microRNA expression to improve nitrogen use efficiency. Plant Sci. 2013, 210, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.G.; Zhang, Y.; Tan, Y.W.; Zhang, M.; Zhu, L.L.; Xu, G.H.; Fan, X. Agronomic nitrogen-use efficiency of rice can be increased by driving OsNRT2.1 expression with the OsNAR2.1 promoter. Plant Biotechnol. J. 2016, 14, 1705–1715. [Google Scholar] [CrossRef] [Green Version]
- Saha, P.; Chakraborti, D.; Sarkar, A.; Dutta, I.; Basu, D.; Das, S. Characterization of vascular-specific RSs1 and rolC promoters for their utilization in engineering plants to develop resistance against hemipteran insect pests. Planta 2007, 226, 429–442. [Google Scholar] [CrossRef]
- Bartlett, J.G.; Alves, S.C.; Smedley, M.; Snape, J.W.; Harwood, W.A. High-throughput Agrobacterium-mediated barley transformation. Plant Methods 2008, 4, 22. [Google Scholar] [CrossRef] [Green Version]
- Harwood, W.A.; Bartlett, J.G.; Alves, S.C.; Perry, M.; Smedley, M.A.; Leyl, N.; Snape, J.W. Barley transformation using Agrobacterium-mediated techniques. In Transgenic Wheat; Barley and Oats; Humana Press: Totowa, NJ, USA, 2009; pp. 137–147. [Google Scholar]
- Zalewski, W.; Gasparis, S.; Boczkowska, M.; Rajchel, I.K.; Kała, M.; Orczyk, W.; Nadolska-Orczyk, A. Expression patterns of HvCKX genes indicate their role in growth and reproductive development of barley. PLoS ONE 2014, 9, e115729. [Google Scholar] [CrossRef] [Green Version]
- Ausubel, F.M.; Brent, R.E.; Kingston, D. Current Protocols in Molecular Biology; John Wiley and Sons: New York, NY, USA, 1994. [Google Scholar]
- Duan, Y.H.; Zhang, Y.L.; Ye, L.T.; Fan, X.R.; Xu, G.H.; Shen, Q.R. Response of rice cultivars with different nitrogen use efficiency to partial nitrate nutrition. Ann. Bot. 2007, 99, 1153–1160. [Google Scholar] [CrossRef] [Green Version]
- Hu, B.; Wang, W.; Ou, S.; Tang, J.; Li, H.; Che, R.; Zhang, Z.; Chai, X.; Wang, H.; Wang, Y.; et al. Variation in NRT1.1B contributes to nitrate-use divergence between rice subspecies. Nature. Genetics 2015, 47, 834–838. [Google Scholar] [PubMed]
- Feng, H.; Xia, X.; Fan, X.; Xu, G.; Miller, A.J. Optimizing plant transporter expression in Xenopus oocytes. Plant Methods 2013, 9, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Distribution ration of shoot (%) | WT | Ubi-1 | Ubi-2 | Ubi-3 | RSs-1 | RSs-2 | RSs-3 |
| Total N | 72.60b | 86.76a | 85.24ab | 84.40ab | 84.10ab | 81.49ab | 81.38ab |
| Total P | 66.20b | 79.60a | 78.29a | 75.07a | 74.34a | 83.15a | 82.38a |
| Total Fe | 11.49b | 24.22b | 23.87b | 32.24b | 31.48b | 91.16a | 80.79a |
| Distribution ration of root (%) | WT | Ubi-1 | Ubi-2 | Ubi-3 | RSs-1 | RSs-2 | RSs-3 |
| Total N | 27.40a | 13.24b | 14.76ab | 15.60ab | 15.90ab | 15.81ab | 18.62ab |
| Total P | 32.97a | 20.40b | 21.71b | 24.93ab | 25.66ab | 16.85b | 17.62b |
| Total Fe | 88.51a | 75.78a | 76.13a | 67.76a | 68.52a | 8.84b | 19.21b |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, B.; Xu, M.; Zhao, L.; Xie, P.; Chen, Y.; Harwood, W.; Xu, G.; Fan, X.; Miller, A.J. Overexpression of the High-Affinity Nitrate Transporter OsNRT2.3b Driven by Different Promoters in Barley Improves Yield and Nutrient Uptake Balance. Int. J. Mol. Sci. 2020, 21, 1320. https://doi.org/10.3390/ijms21041320
Luo B, Xu M, Zhao L, Xie P, Chen Y, Harwood W, Xu G, Fan X, Miller AJ. Overexpression of the High-Affinity Nitrate Transporter OsNRT2.3b Driven by Different Promoters in Barley Improves Yield and Nutrient Uptake Balance. International Journal of Molecular Sciences. 2020; 21(4):1320. https://doi.org/10.3390/ijms21041320
Chicago/Turabian StyleLuo, Bingbing, Man Xu, Limei Zhao, Peng Xie, Yi Chen, Wendy Harwood, Guohua Xu, Xiaorong Fan, and Anthony J. Miller. 2020. "Overexpression of the High-Affinity Nitrate Transporter OsNRT2.3b Driven by Different Promoters in Barley Improves Yield and Nutrient Uptake Balance" International Journal of Molecular Sciences 21, no. 4: 1320. https://doi.org/10.3390/ijms21041320
APA StyleLuo, B., Xu, M., Zhao, L., Xie, P., Chen, Y., Harwood, W., Xu, G., Fan, X., & Miller, A. J. (2020). Overexpression of the High-Affinity Nitrate Transporter OsNRT2.3b Driven by Different Promoters in Barley Improves Yield and Nutrient Uptake Balance. International Journal of Molecular Sciences, 21(4), 1320. https://doi.org/10.3390/ijms21041320





