Comprehensive Transcriptomic Analysis of Auxin Responses in Submerged Rice Coleoptile Growth
Abstract
1. Introduction
2. Results
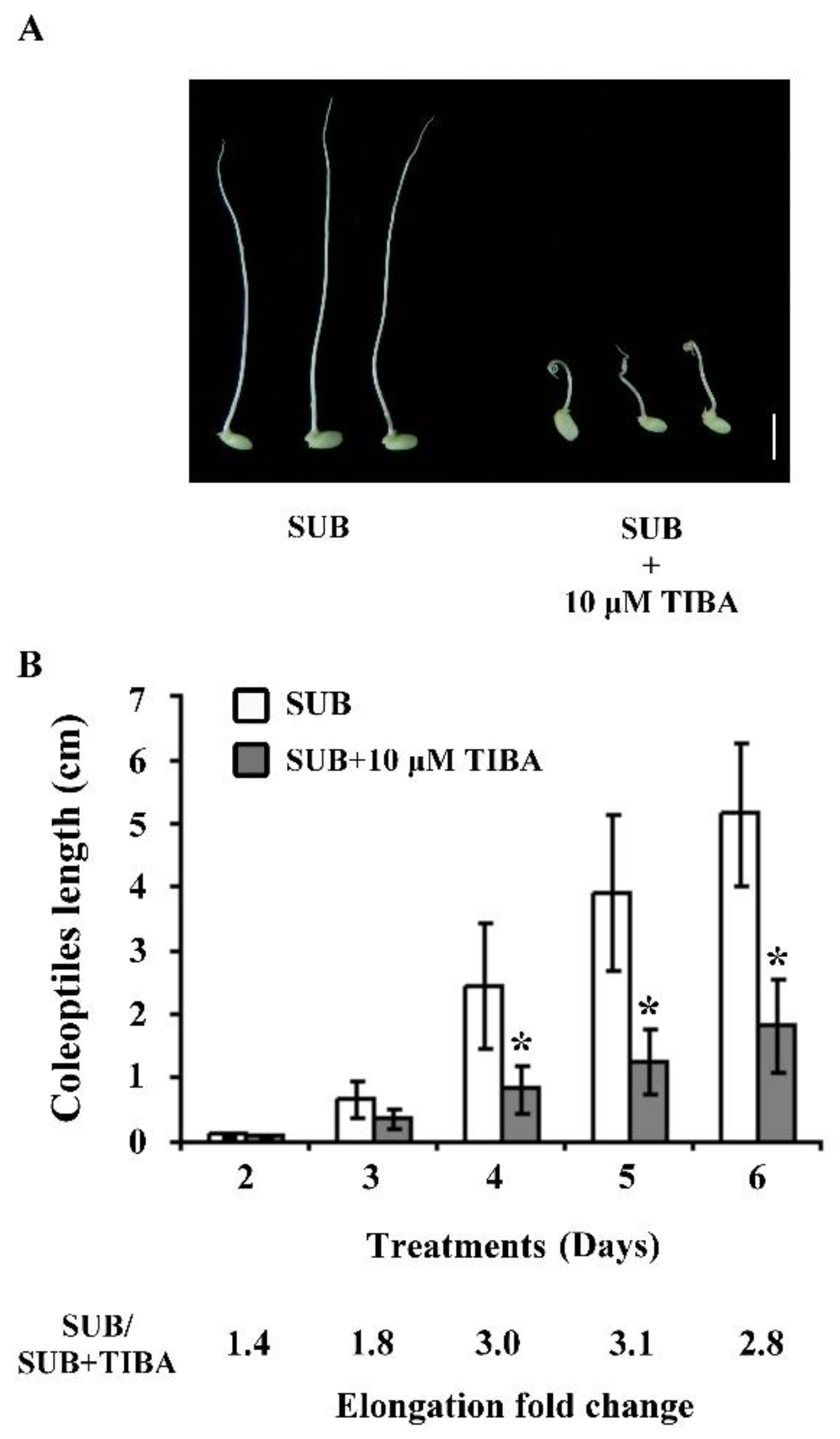
2.1. Submerged Rice Coleoptile Elongation Affected by Impeded Auxin Signaling
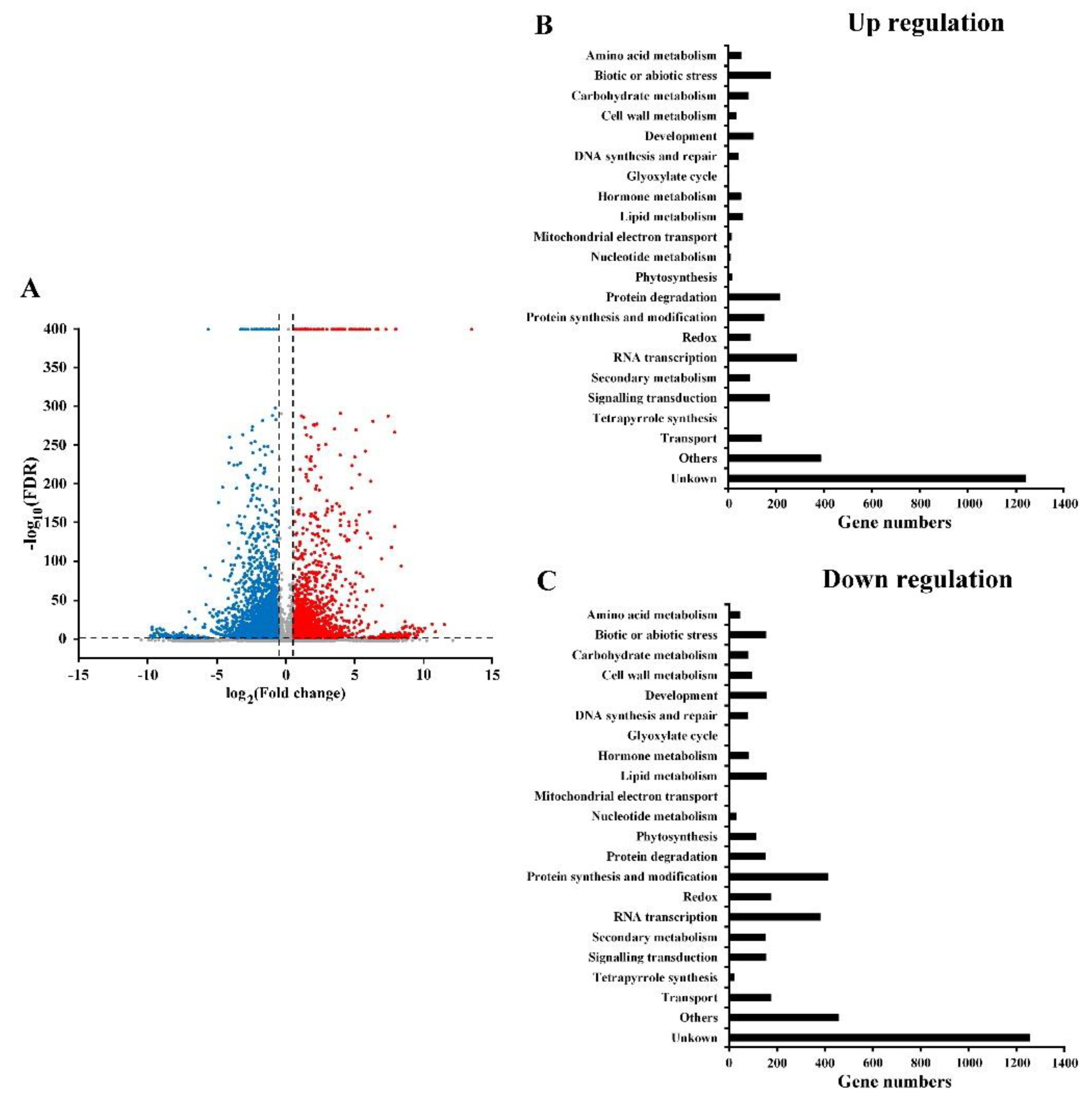
2.2. RNA Sequencing and Functional Annotation of DEGs in Rice Coleoptiles
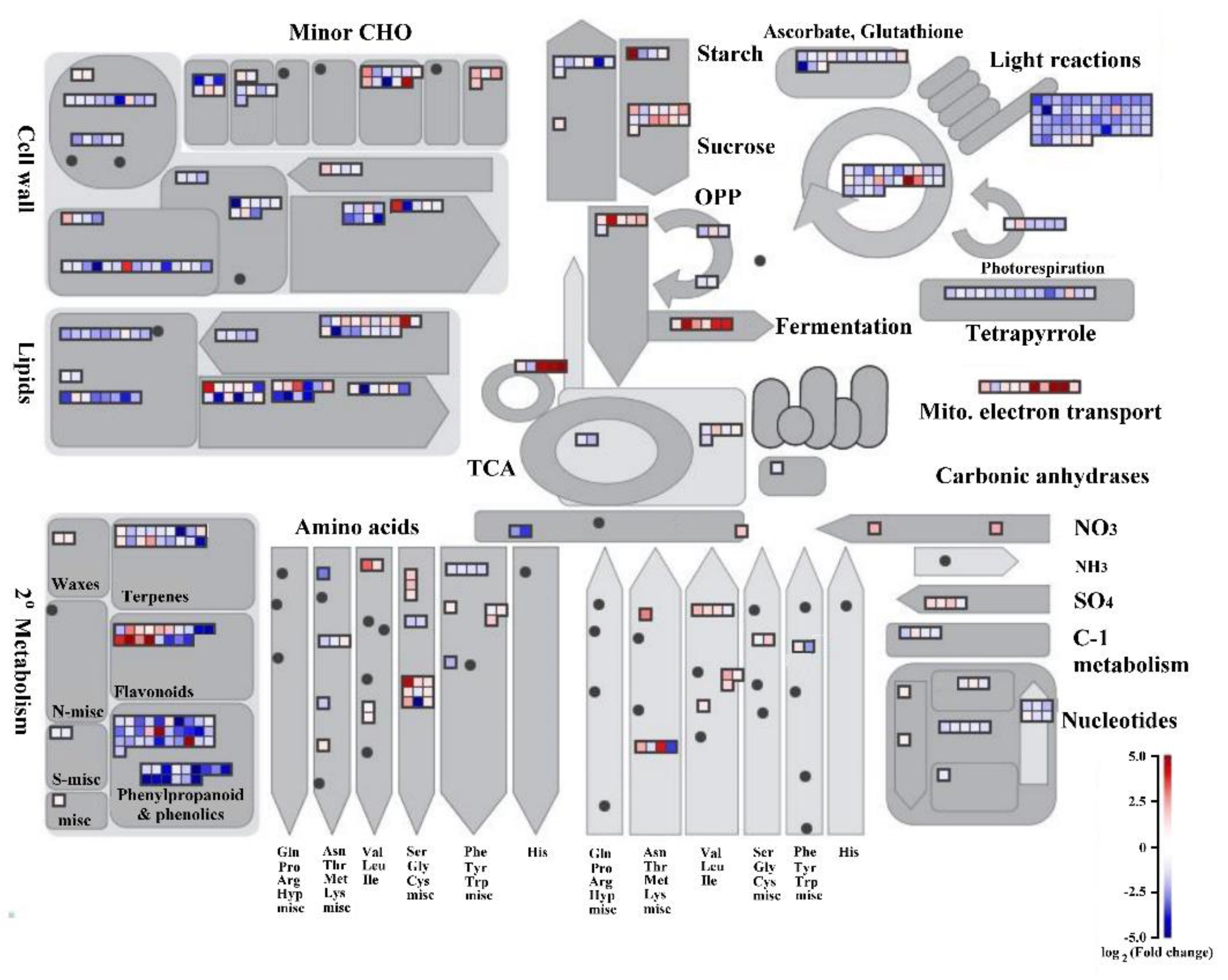
2.3. The DEGs in Metabolism Process Regulated by Auxin Signaling in Submerged Rice Coleoptiles
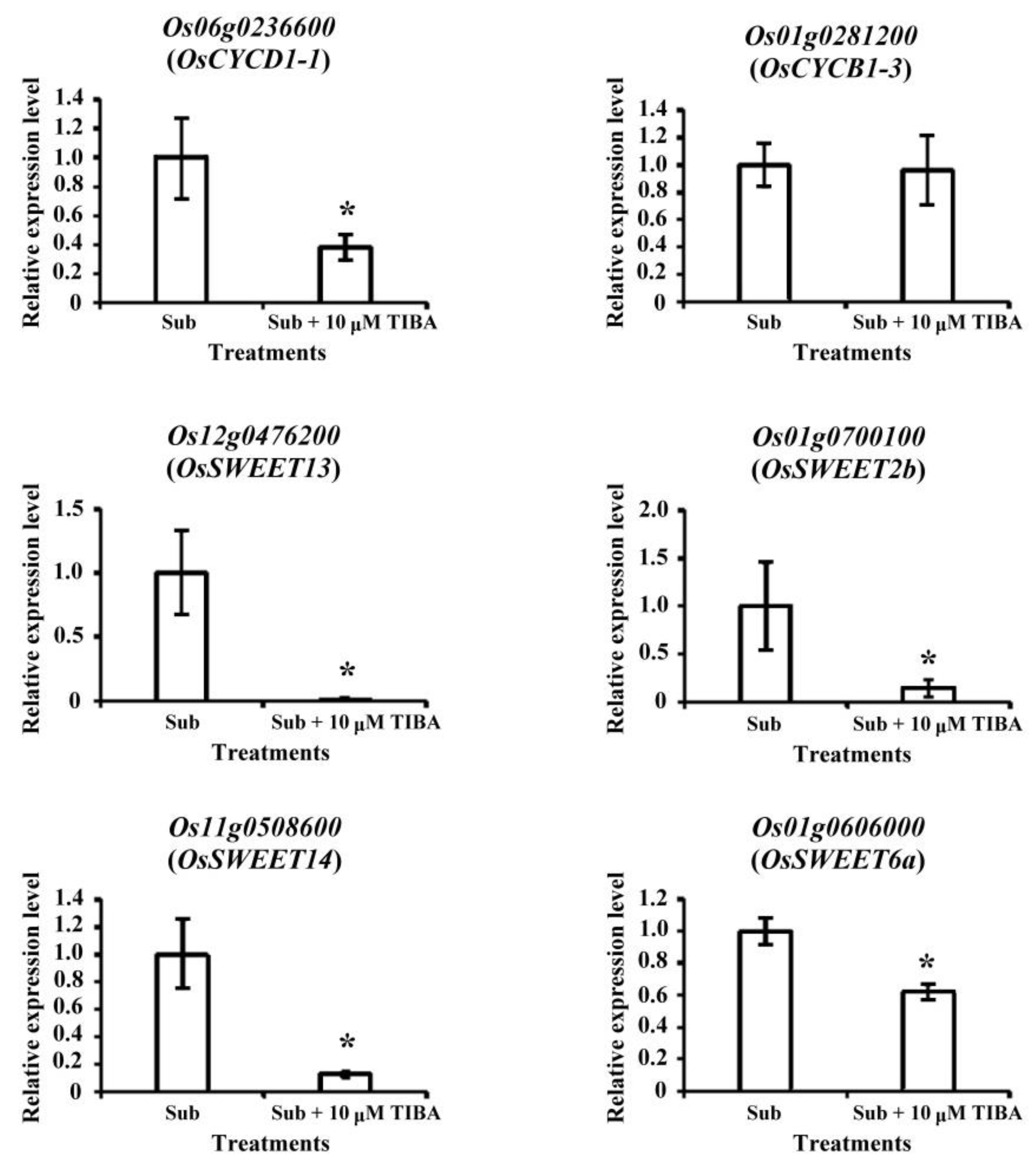
2.4. Transcript Levels of Development-Responsive Genes Regulated by Auxin Signaling in Submerged Rice Coleoptiles
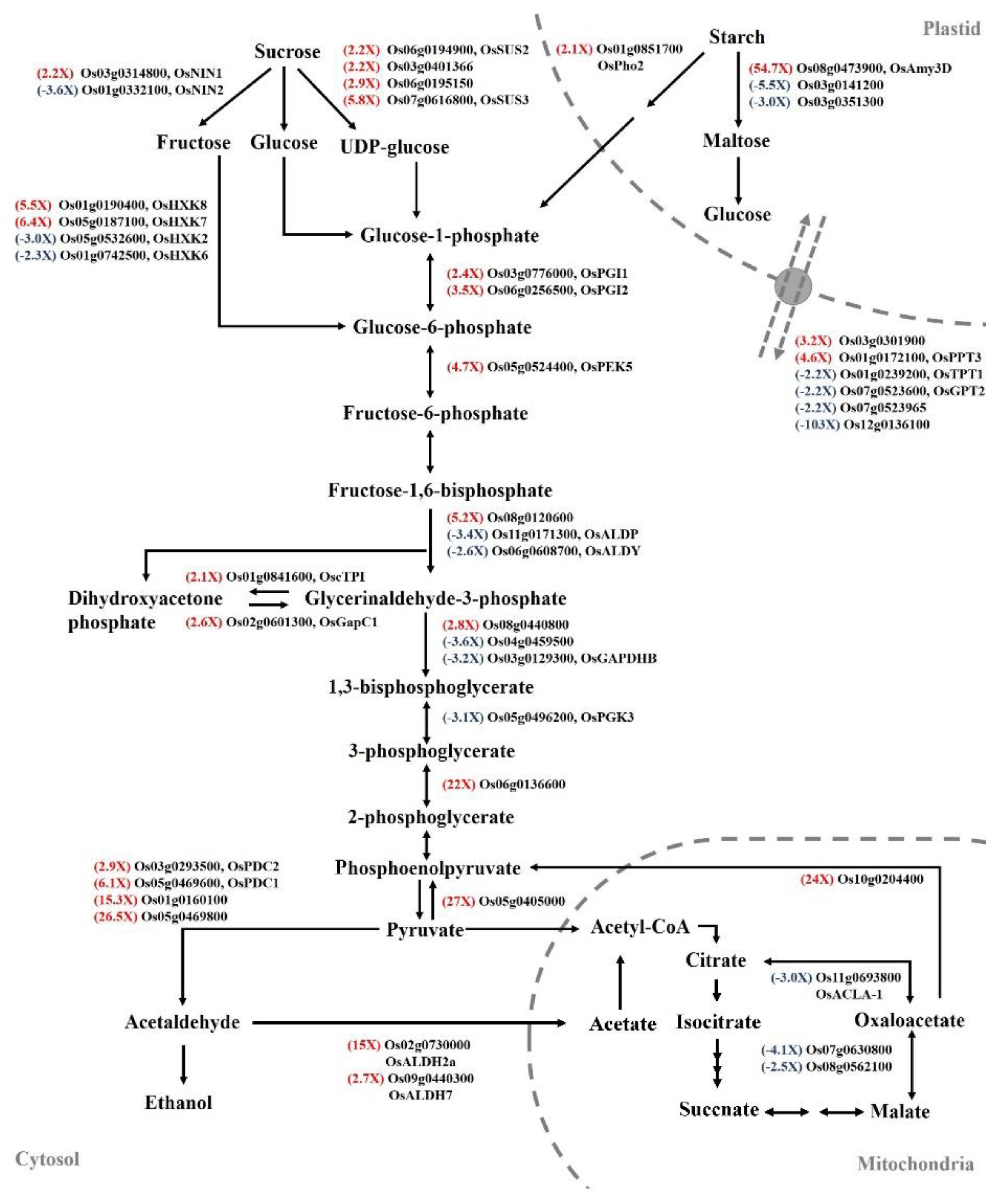
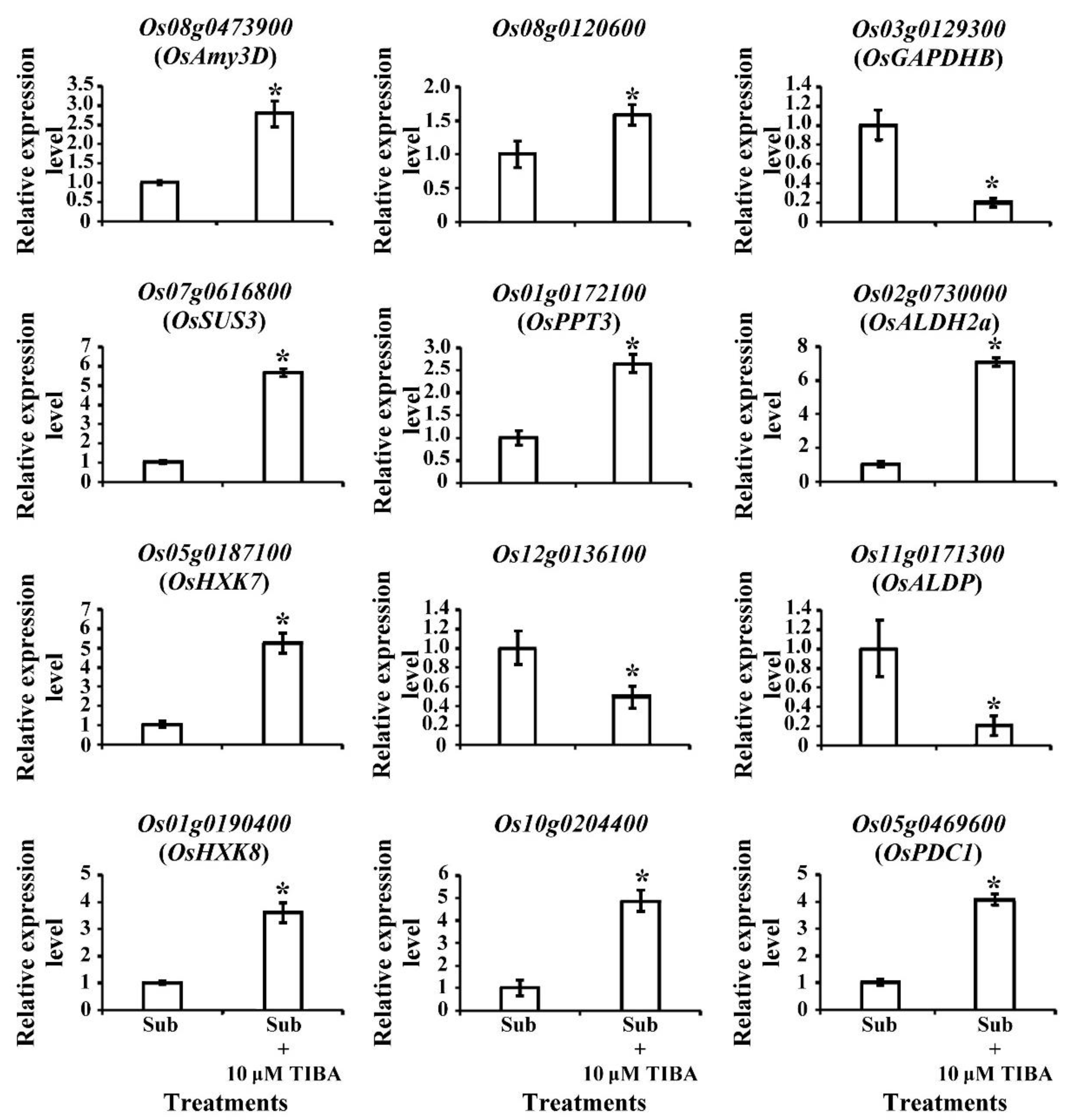
2.5. Transcript Levels of Carbohydrate-Metabolism-Associated Genes Regulated by Auxin Signaling in Submerged Rice Coleoptiles
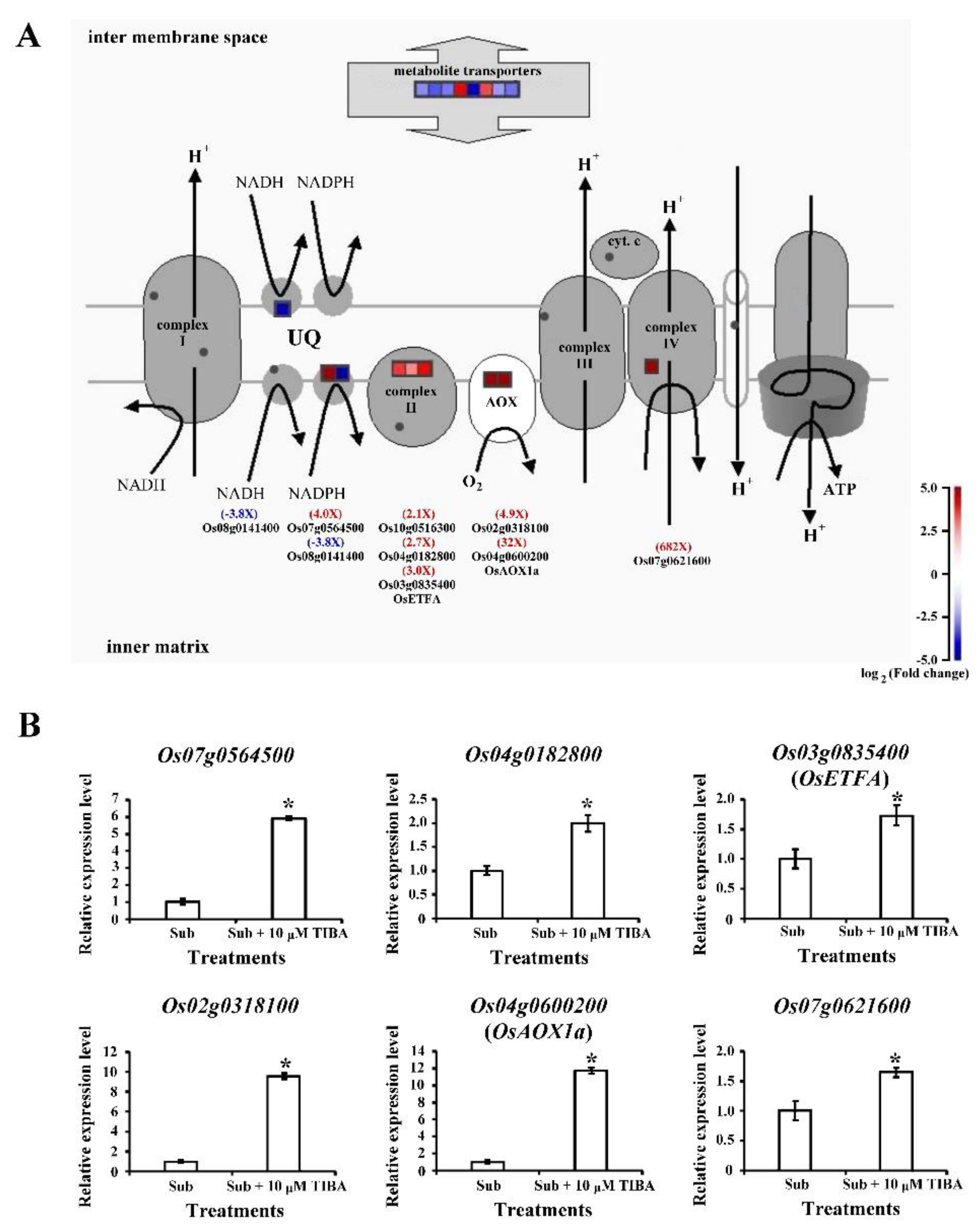
2.6. Transcript Levels of Mitochondrial-Electron-Transport–Associated Genes Regulated by Auxin Signaling in Submerged Rice Coleoptiles
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. Measurement Length of Coleoptiles
4.3. Transcriptomic Analysis by RNA Sequencing
4.4. Functional Analysis of Differentially Expressed Genes
4.5. Extraction of Total RNA and Quantitative Real-Time Polymerase Chain Reaction
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| SUB | Submergence |
| TIBA | 2,3,5-triiodobenzoic acid |
| DEGs | Differentially expressed genes |
| qRT-PCR | Quantitative real time polymerase chain reaction |
References
- Miro, B.; Ismail, A.M. Tolerance of anaerobic conditions caused by flooding during germination and early growth in rice (Oryza sativa L.). Front. Plant Sci. 2013, 4, 269. [Google Scholar] [CrossRef] [PubMed]
- de Melo, R.B.; Franco, A.C.; Silva, C.O.; Piedade, M.T.; Ferreira, C.S. Seed germination and seedling development in response to submergence in tree species of the Central Amazonian floodplains. AoB Plants 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Mahender, A.; Anandan, A.; Pradhan, S.K. Early seedling vigour, an imperative trait for direct-seeded rice: An overview on physio-morphological parameters and molecular markers. Planta 2015, 241, 1027–1050. [Google Scholar] [CrossRef] [PubMed]
- Howden, S.M.; Soussana, J.F.; Tubiello, F.N.; Chhetri, N.; Dunlop, M.; Meinke, H. Adapting agriculture to climate change. Proc. Natl. Acad. Sci. USA 2007, 104, 19691–19696. [Google Scholar] [CrossRef] [PubMed]
- Yin, C.C.; Ma, B.; Collinge, D.P.; Pogson, B.J.; He, S.J.; Xiong, Q.; Duan, K.X.; Chen, H.; Yang, C.; Lu, X.; et al. Ethylene responses in rice roots and coleoptiles are differentially regulated by a carotenoid isomerase-mediated abscisic acid pathway. Plant Cell 2015, 27, 1061–1081. [Google Scholar] [CrossRef]
- Zhao, F.Y.; Cai, F.X.; Gao, H.J.; Zhang, S.Y.; Wang, K.; Liu, T.; Wang, X. ABA plays essential roles in regulating root growth by interacting with auxin and MAPK signaling pathways and cell-cycle machinery in rice seedlings. Plant Growth Regul. 2015, 75, 535–547. [Google Scholar] [CrossRef]
- Watanabe, H.; Takahashi, K. Effects of abscisic acid, fusicoccin, and potassium on growth and morphogenesis of leaves and internodes in dark-grown rice seedlings. Plant Growth Regul. 1997, 21, 109–114. [Google Scholar] [CrossRef]
- Ishizawa, K.; Esashi, Y. Cooperation of ethylene and auxin in the growth-regulation of rice coleoptile segments. J. Exp. Bot. 1983, 34, 74–82. [Google Scholar] [CrossRef]
- Perrot-Rechenmann, C. Cellular responses to auxin: Division versus expansion. Cold Spring Harb. Perspect. Biol. 2010, 2, a001446. [Google Scholar] [CrossRef]
- Haga, K.; Takano, M.; Neumann, R.; Iino, M. The Rice COLEOPTILE PHOTOTROPISM1 gene encoding an ortholog of Arabidopsis NPH3 is required for phototropism of coleoptiles and lateral translocation of auxin. Plant Cell 2005, 17, 103–115. [Google Scholar] [CrossRef]
- Waller, F.; Furuya, M.; Nick, P. OsARF1, an auxin response factor from rice, is auxin-regulated and classifies as a primary auxin responsive gene. Plant Mol. Biol. 2002, 50, 415–425. [Google Scholar] [CrossRef]
- Burdach, Z.; Kurtyka, R.; Siemieniuk, A.; Karcz, W. Role of chloride ions in the promotion of auxin-induced growth of maize coleoptile segments. Ann. Bot. 2014, 114, 1023–1034. [Google Scholar] [CrossRef] [PubMed]
- Bailey-Serres, J.; Voesenek, L. Flooding stress: Acclimations and genetic diversity. Annu. Rev. Plant Biol. 2008, 59, 313–339. [Google Scholar] [CrossRef] [PubMed]
- Magneschi, L.; Kudahettige, R.; Alpi, A.; Perata, P. Comparative analysis of anoxic coleoptile elongation in rice varieties: Relationship between coleoptile length and carbohydrate levels, fermentative metabolism and anaerobic gene expression. Plant Biol. 2009, 11, 561–573. [Google Scholar] [CrossRef] [PubMed]
- Nghi, K.N.; Tondelli, A.; Valè, G.; Tagliani, A.; Marè, C.; Perata, P.; Pucciariello, C. Dissection of coleoptile elongation in japonica rice under submergence through integrated genome-wide association mapping and transcriptional analyses. Plant Cell Environ. 2019, 42, 1832–1846. [Google Scholar] [CrossRef]
- Narsai, R.; Secco, D.; Schultz, M.D.; Ecker, J.R.; Lister, R.; Whelan, J. Dynamic and rapid changes in the transcriptome and epigenome during germination and in developing rice (Oryza sativa) coleoptiles under anoxia and re-oxygenation. Plant J. 2017, 89, 805–824. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, T.; Wang, R.; Zhao, Y. Recent advances in auxin research in rice and their implications for crop improvement. J. Exp. Bot. 2017, 69, 255–263. [Google Scholar] [CrossRef]
- Narsai, R.; Edwards, J.M.; Roberts, T.H.; Whelan, J.; Joss, G.H.; Atwell, B.J. Mechanisms of growth and patterns of gene expression in oxygen-deprived rice coleoptiles. Plant J. 2015, 82, 25–40. [Google Scholar] [CrossRef]
- Burns, E.; Keith, B.; Refai, M.; Bothner, B.; Dyer, W. Proteomic and biochemical assays of glutathione-related proteins in susceptible and multiple herbicide resistant Avena fatua L. Pestic. Biochem. Physiol. 2017, 140, 69. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, M.M.; Wang, Y.J.; Gao, Y.T.; Li, R.; Wang, G.F.; Li, W.Q.; Liu, W.T.; Chen, K.M. The plasma membrane NADPH oxidase OsRbohA plays a crucial role in developmental regulation and drought-stress response in rice. Physiol. Plant. 2016, 156, 421–443. [Google Scholar] [CrossRef]
- Wang, Q.; Guan, Y.; Wu, Y.; Chen, H.; Chen, F.; Chu, C. Overexpression of a rice OsDREB1F gene increases salt, drought, and low temperature tolerance in both Arabidopsis and rice. Plant Mol. Biol. 2008, 67, 589–602. [Google Scholar] [CrossRef] [PubMed]
- Buti, M.; Pasquariello, M.; Ronga, D.; Milc, J.A.; Pecchioni, N.; Ho, V.T.; Pucciariello, C.; Perata, P.; Francia, E. Transcriptome profiling of short-term response to chilling stress in tolerant and sensitive Oryza sativa ssp. Japonica seedlings. Funct. Integr. Genom. 2018, 18, 627–644. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.; Dansana, P.K.; Giri, J.; Deveshwar, P.; Arora, R.; Agarwal, P.; Khurana, J.P.; Kapoor, S.; Tyagi, A.K. Modulation of transcription factor and metabolic pathway genes in response to water-deficit stress in rice. Funct. Integr. Genom. 2011, 11, 157–178. [Google Scholar] [CrossRef] [PubMed]
- Swetha, C.; Basu, D.; Pachamuthu, K.; Tirumalai, V.; Nair, A.; Prasad, M.; Shivaprasad, P.V. Major domestication-related phenotypes in Indica rice are due to loss of mirna-mediated laccase silencing. Plant Cell 2018, 30, 2649–2662. [Google Scholar] [CrossRef] [PubMed]
- Strader, L.C.; Zhao, Y. Auxin perception and downstream events. Curr. Opin. Plant Biol. 2016, 33, 8–14. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, H.; Zhao, Y.; Feng, Z.; Li, Q.; Yang, H.Q.; Luan, S.; Li, J.; He, Z.H. Auxin controls seed dormancy through stimulation of abscisic acid signaling by inducing ARF-mediated ABI3 activation in Arabidopsis. Proc. Natl. Acad. Sci. USA 2013, 110, 15485–15490. [Google Scholar] [CrossRef]
- Ramaih, S.; Guedira, M.; Paulsen, G.M. Relationship of indoleacetic acid and tryptophan to dormancy and preharvest sprouting of wheat. Funct. Plant Biol. 2003, 30, 939–945. [Google Scholar] [CrossRef]
- Umeda, M.; Umeda-Hara, C.; Yamaguchi, M.; Hashimoto, J.; Uchimiya, H. Differential expression of genes for cyclin-dependent protein kinases in rice plants. Plant Physiol. 1999, 119, 31–40. [Google Scholar] [CrossRef]
- Yu, S.M.; Lo, S.F.; Ho, T.H.D. Source–sink communication: Regulated by hormone, nutrient, and stress cross-signaling. Trends Plant Sci. 2015, 20, 844–857. [Google Scholar] [CrossRef]
- Chu, Z.; Yuan, M.; Yao, J.; Ge, X.; Yuan, B.; Xu, C.; Li, X.; Fu, B.; Li, Z.; Bennetzen, J.L. Promoter mutations of an essential gene for pollen development result in disease resistance in rice. Genes Dev. 2006, 20, 1250–1255. [Google Scholar] [CrossRef]
- Antony, G.; Zhou, J.; Huang, S.; Li, T.; Liu, B.; White, F.; Yang, B. Rice xa13 recessive resistance to bacterial blight is defeated by induction of the disease susceptibility gene Os-11N3. Plant Cell 2010, 22, 3864–3876. [Google Scholar] [CrossRef] [PubMed]
- Römer, P.; Recht, S.; Strauß, T.; Elsaesser, J.; Schornack, S.; Boch, J.; Wang, S.; Lahaye, T. Promoter elements of rice susceptibility genes are bound and activated by specific TAL effectors from the bacterial blight pathogen, Xanthomonas oryzae pv. oryzae. New Phytol. 2010, 187, 1048–1057. [Google Scholar] [CrossRef] [PubMed]
- Depta, H.; Elisele, K.H.; Hertel, R. Specific inhibitors of auxin transport: Action on tissue segments and in vitro binding to membranes from maize coleoptiles. Plant Sci. Lett. 1993, 31, 181–192. [Google Scholar] [CrossRef]
- Conesa, A.; Götz, S.; García-Gómez, J.M.; Terol, J.; Talón, M.; Robles, M. Blast2GO: A universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 2005, 21, 3674–3676. [Google Scholar] [CrossRef]
- Thimm, O.; Bläsing, O.; Gibon, Y.; Nagel, A.; Meyer, S.; Krüger, P.; Selbig, J.; Müller, L.A.; Rhee, S.Y.; Stitt, M. MAPMAN: A user-driven tool to display genomics data sets onto diagrams of metabolic pathways and other biological processes. Plant J. 2004, 37, 914–939. [Google Scholar] [CrossRef]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3-new capabilities and interfaces. Nucleic Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
| GO Function | Gene ID | Name | Description | FPKM | Fold Change | FDR | |
|---|---|---|---|---|---|---|---|
| SUB | TIBA | ||||||
| Biotic or abiotic stress | Os06g0195800 | OsDJC53 | DnaJ domain protein | 0.01 | 8.85 | 885.0 | 4.9 × 10−14 |
| Os11g0592200 | OsPR4 | Pathogenesis-related protein | 0.01 | 6.27 | 627.0 | 8.1 × 10−8 | |
| Hormone metabolism | Os11g0453900 | OsRAB16D | Responsive to ABA 16D | 0.01 | 3.15 | 315.0 | 3.3 × 10−5 |
| Os04g0339400 | OsAKR | Aaldo-keto reductase | 1.58 | 387.55 | 245.3 | 0.0 | |
| Protein degradation | Os08g0126000 | E3 ubiquitin-protein ligase | 0.01 | 3.51 | 351.0 | 1.6 × 10−15 | |
| Os09g0243200 | E3 ubiquitin-protein ligase | 0.01 | 2.7 | 270.0 | 8.8 × 10−4 | ||
| Redox | Os07g0638300 | 1-Cys peroxiredoxin | 0.01 | 7.91 | 791.0 | 3.1 × 10−5 | |
| Os01g0628000 | Cytochrome P450 | 0.01 | 3.85 | 385.0 | 8.8 × 10−6 | ||
| Os03g0785900 | Glutathione S-transferase | 4.97 | 1269.51 | 255.4 | 0.0 | ||
| Os09g0367700 | Glutathione S-transferase | 0.63 | 148.56 | 235.8 | 4.5 × 10−268 | ||
| Os10g0527400 | Glutathione S-transferase | 0.27 | 54.7 | 202.6 | 2.2 × 10−119 | ||
| RNA transcription | Os06g0719900 | Endo/excinuclease amino terminal domain-containing protein | 0.01 | 6.36 | 636.0 | 1.2 × 10−14 | |
| Os05g0223200 | Glycine-rich RNA-binding protein | 0.01 | 4.58 | 458.0 | 6.1 × 10−7 | ||
| Os05g0404700 | Methyl-CpG-binding domain-containing protein | 0.01 | 2.07 | 207.0 | 1.7 × 10−3 | ||
| Secondary metabolism | Os08g0112300 | Acetyltransferase | 0.01 | 5.07 | 507.0 | 7.9 × 10−16 | |
| Os03g0184550 | Dihydroflavonol-4-reductase | 0.01 | 3.98 | 398.0 | 2.8 × 10−9 | ||
| Signalling transduction | Os06g0225000 | OsSAR1d | GTP-binding protein | 0.01 | 5.18 | 518.0 | 6.1 × 10−7 |
| Os04g0655400 | G-type lectin S-receptor-like serine/threonine-protein kinase | 0.01 | 2.65 | 265.0 | 1.7 × 10−5 | ||
| Other | Os09g0324000 | Oleosin | 0.01 | 7.99 | 799.0 | 3.6 × 10−10 | |
| Os01g0695800 | MDR-like ABC transporter | 0.01 | 4.90 | 490.0 | 2.1 × 10−8 | ||
| Os07g0529000 | OsL85 | Isocitrate lyase | 0.01 | 4.21 | 421.0 | 2.5 × 10−17 | |
| Os05g0366600 | Beta-glucosidase | 0.01 | 3.59 | 359.0 | 6.2 × 10−15 | ||
| Os02g0586900 | Glycine-rich cell wall structural protein | 0.47 | 112.36 | 239.1 | 2.3 × 10−146 | ||
| GO Function | Gene ID | Name | Description | FPKM | Fold Change | FDR | |
|---|---|---|---|---|---|---|---|
| SUB | TIBA | ||||||
| Biotic or abiotic stress | Os07g0429600 | Tthionin like peptide | 2.71 | 0.01 | 0.0037 | 7.4 × 10−3 | |
| Os07g0431160 | Tthionin like peptide | 2.68 | 0.01 | 0.0037 | 2.2 × 10−3 | ||
| Carbohydrate metabolism | Os10g0113900 | NAD(P)H-dependent oxidoreductase | 5.09 | 0.01 | 0.0020 | 1.1 × 10−12 | |
| Cell wall metabolism | Os10g0545500 | Xyloglucan endotransglucosylase/hydrolase protein | 5.04 | 0.01 | 0.0020 | 2.9 × 10−7 | |
| Os01g0946500 | Glucan endo-1,3-beta-glucosidase | 2.48 | 0.01 | 0.0040 | 7.3 × 10−6 | ||
| Os08g0253800 | Xyloglucan glycosyltransferase | 2.07 | 0.01 | 0.0048 | 2.9 × 10−7 | ||
| Development | Os01g0606000 | OsSWEET6a | Bidirectional sugar transporter | 2.49 | 0.01 | 0.0040 | 2.6 × 10−5 |
| Hormone metabolism | Os09g0437100 | OsSAUR37 | Small auxin-up RNA | 2.86 | 0.01 | 0.0035 | 1.8 × 10−4 |
| Lipid metabolism | Os01g0882100 | Esterase | 8.07 | 0.01 | 0.0012 | 7.4 × 10−3 | |
| Os07g0174400 | Lipid transfer protein | 7.27 | 0.01 | 0.0014 | 2.6 × 10−5 | ||
| Os04g0554500 | Lipid transfer protein | 2.98 | 0.01 | 0.0034 | 3.4 × 10−4 | ||
| Os11g0299300 | Phospholipase | 2.87 | 0.01 | 0.0035 | 3.0 × 10−9 | ||
| Photosynthesis | Os04g0545600 | Stellacyanin-like protein | 8.27 | 0.01 | 0.0012 | 5.7 × 10−13 | |
| Os01g0869800 | Photosystem II 22 kDa protein | 4.26 | 0.01 | 0.0023 | 1.1 × 10−8 | ||
| Os04g0545400 | Stellacyanin | 2.72 | 0.01 | 0.0037 | 4.0 × 10−3 | ||
| Os02g0758800 | Blue copper protein | 2.00 | 0.01 | 0.0050 | 3.4 × 10−4 | ||
| Protein degradation | Os06g0278000 | Glucoamylase | 8.43 | 0.01 | 0.0012 | 3.9 × 10−16 | |
| Os06g0257600 | Esterase | 4.70 | 0.01 | 0.0021 | 5.7 × 10−13 | ||
| Os01g0727840 | Subtilisin-like protease | 2.20 | 0.01 | 0.0045 | 7.4 × 10−3 | ||
| Protein synthesis and modification | Os09g0535400 | Proline-rich receptor-like protein kinase | 2.18 | 0.01 | 0.0046 | 7.3 × 10−6 | |
| Redox | Os12g0263000 | Glutathione synthase | 5.69 | 0.01 | 0.0018 | 5.5 × 10−15 | |
| Os03g0339300 | Peroxidase | 2.36 | 0.01 | 0.0042 | 7.3 × 10−6 | ||
| Os07g0677300 | Peroxidase | 2.17 | 0.01 | 0.0046 | 2.6 × 10−5 | ||
| Os06g0203200 | Flavin-containing monooxygenase | 2.08 | 0.01 | 0.0048 | 4.1 × 10−8 | ||
| RNA transcription | Os01g0968800 | OsDREB1f | Dehydration-responsive element-binding protein | 6.54 | 0.01 | 0.0015 | 2.2 × 10−10 |
| Os05g0442400 | OsMID1 | Myb-like DNA-binding domain containing protein | 3.74 | 0.01 | 0.0027 | 1.1 × 10−6 | |
| Os01g0821300 | OsWRKY86 | WRKY transcription factor | 3.23 | 0.01 | 0.0031 | 5.0 × 10−5 | |
| Os12g0106200 | OsLBD12-1 | Lateral organ boundaries domain (LBD) family transcription factor | 2.90 | 0.01 | 0.0034 | 6.3 × 10−4 | |
| Os09g0414500 | Mini zinc finger protein | 2.59 | 0.01 | 0.0039 | 2.2 × 10−3 | ||
| Os02g0781300 | OsERF11 | Ethylene-responsive transcription factor | 2.53 | 0.01 | 0.0040 | 6.3 × 10−4 | |
| Os04g0590800 | OsbHLH94 | Basic helix-loop-helix protein | 2.40 | 0.01 | 0.0042 | 2.2 × 10−3 | |
| Os09g0522200 | Dehydration-responsive element-binding protein | 2.30 | 0.01 | 0.0043 | 3.4 × 10−4 | ||
| Os09g0538000 | Ribonuclease | 2.04 | 0.01 | 0.0049 | 9.5 × 10−5 | ||
| Secondary metabolism | Os04g0609300 | Shikimate O-hydroxycinnamoyltransferase | 2.01 | 0.01 | 0.0050 | 2.0 × 10−6 | |
| Signaling transduction | Os06g0339500 | LRR receptor-like serine/threonine-protein kinase | 2.55 | 0.01 | 0.0039 | 7.4 × 10−3 | |
| Os11g0105000 | OsCML25 | Calcium-binding protein | 2.24 | 0.01 | 0.0045 | 7.4 × 10−3 | |
| Os12g0104900 | OsCML26 | Calcium-binding protein | 2.24 | 0.01 | 0.0045 | 7.4 × 10−3 | |
| Transport | Os05g0338933 | OsNRT1 | Proton-dependent oligopeptide transporter | 4.07 | 0.01 | 0.0025 | 4.0 × 10−3 |
| Os02g0658100 | OsTIP2 | Aquaporin | 3.60 | 0.01 | 0.0028 | 7.3 × 10−6 | |
| Log2 (FC) | Gene ID | Name | Description | FPKM | Fold Change | FDR | ||
|---|---|---|---|---|---|---|---|---|
| SUB | TIBA | |||||||
| Os06g0273200 | Trichome birefringence-like protein | 0.01 | 1.54 | 154.0 | 4.6 × 10−4 |  | ||
| Os01g0306400 | OsDOG1L-3 | Delay of germination 1-like protein | 0.01 | 1.37 | 137.0 | 4.6 × 10−4 | ||
| Os02g0616600 | Meiosis arrested at leptotene like protein | 0.85 | 87.01 | 102.4 | 0.0 | |||
| Os01g0762500 | OsGLUA-1 | Glutelin type-A-1 | 0.12 | 2.25 | 18.7 | 3.7 × 10−6 | ||
| Os01g0606000 | OsSWEET6a | Bidirectional sugar transporter | 2.49 | 0.01 | 0.004 | 2.6 × 10−5 | ||
| Os09g0307300 | Microtubule-associated protein | 1.83 | 0.01 | 0.005 | 1.8 × 10−4 | |||
| Os03g0578500 | Myosin | 1.95 | 0.01 | 0.005 | 1.2 × 10−3 | |||
| Os08g0477500 | OsPLP2 | Patatin-like protein | 1.54 | 0.01 | 0.006 | 7.3 × 10−6 | ||
| Os06g0234600 | OsTBL52 | Trichome birefringence-like protein | 1.53 | 0.01 | 0.007 | 9.5 × 10−5 | ||
| Os01g0803300 | Walls are thin1 (WAT1)-related protein | 1.26 | 0.01 | 0.008 | 2.2 × 10−3 | |||
| Os09g0375300 | Trichome birefringence-like protein | 1.12 | 0.01 | 0.009 | 4.0 × 10−3 | |||
| Os03g0291200 | Trichome birefringence-like protein | 1.04 | 0.01 | 0.01 | 2.2 × 10−3 | |||
| Os01g0281200 | OsCYCB1-3 | G2/mitotic-specific cyclin-B1-3 | 0.77 | 0.01 | 0.013 | 7.4 × 10−3 | ||
| Os08g0443800 | OsTET12 | Tetraspanin | 4.96 | 0.15 | 0.03 | 7.1 × 10−9 | ||
| Os07g0671000 | Flowering-promoting factor 1-like protein | 24.62 | 0.83 | 0.034 | 6.1 × 10−15 | |||
| Os01g0563000 | OsFKBP73 | Peptidyl-prolyl cis-trans isomerase | 77.11 | 2.59 | 0.034 | 3.0 × 10−17 | ||
| Os01g0968400 | Root cap family protein | 5.33 | 0.26 | 0.049 | 8.8 × 10−10 | |||
| Os05g0557400 | OsNSL1 | Membrane attack complex/perforin (MACPF) domain containing protein | 2.08 | 0.13 | 0.063 | 2.1 × 10−7 | ||
| Os07g0691700 | OsWAVE5 | Suppressor of cAMP receptors (SCAR)-like domain containing protein | 0.76 | 0.05 | 0.066 | 7.5 × 10−4 | ||
| Os11g0508600 | OsSWEET14 | Bidirectional sugar transporter | 7.58 | 0.51 | 0.067 | 4.0 × 10−16 | ||
| Os05g0563000 | NAC transcription factor | 1.45 | 0.1 | 0.069 | 1.3 × 10−3 | |||
| Os12g0476200 | OsSWEET13 | Bidirectional sugar transporter | 9.44 | 0.65 | 0.069 | 4.9 × 10−22 | ||
| Os01g0172800 | Embryo-specific 3 family protein | 35.49 | 2.47 | 0.07 | 7.6 × 10−40 | |||
| Os04g0422300 | Walls are thin1 (WAT1)-related protein | 1.39 | 0.1 | 0.072 | 2.3 × 10−3 | |||
| Os01g0700100 | OsSWEET2b | Bidirectional sugar transporter | 1.55 | 0.13 | 0.084 | 7.1 × 10−3 | ||
| Os03g0229600 | MFP1 attachment factor 1 (MAF1)-like protein | 1.47 | 0.13 | 0.088 | 4.0 × 10−5 | |||
| Os06g0210000 | Walls are thin1 (WAT1)-related protein | 2.73 | 0.24 | 0.088 | 7.1 × 10−5 | |||
| Os06g0236600 | OsCYCD1-1 | G1/S-specific cyclin-D1-1 | 3.16 | 0.28 | 0.089 | 7.1 × 10−5 | ||
| Os07g0601600 | 3-oxo-delta(4,5)-steroid 5-beta-reductase | 2.71 | 0.24 | 0.089 | 7.1 × 10−5 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Y.-S.; Yang, C.-Y. Comprehensive Transcriptomic Analysis of Auxin Responses in Submerged Rice Coleoptile Growth. Int. J. Mol. Sci. 2020, 21, 1292. https://doi.org/10.3390/ijms21041292
Wu Y-S, Yang C-Y. Comprehensive Transcriptomic Analysis of Auxin Responses in Submerged Rice Coleoptile Growth. International Journal of Molecular Sciences. 2020; 21(4):1292. https://doi.org/10.3390/ijms21041292
Chicago/Turabian StyleWu, Yu-Sian, and Chin-Ying Yang. 2020. "Comprehensive Transcriptomic Analysis of Auxin Responses in Submerged Rice Coleoptile Growth" International Journal of Molecular Sciences 21, no. 4: 1292. https://doi.org/10.3390/ijms21041292
APA StyleWu, Y.-S., & Yang, C.-Y. (2020). Comprehensive Transcriptomic Analysis of Auxin Responses in Submerged Rice Coleoptile Growth. International Journal of Molecular Sciences, 21(4), 1292. https://doi.org/10.3390/ijms21041292





