Protective Effects of [6]-Gingerol Against Chemical Carcinogens: Mechanistic Insights
Abstract
1. Introduction
1.1. Chemical Carcinogens
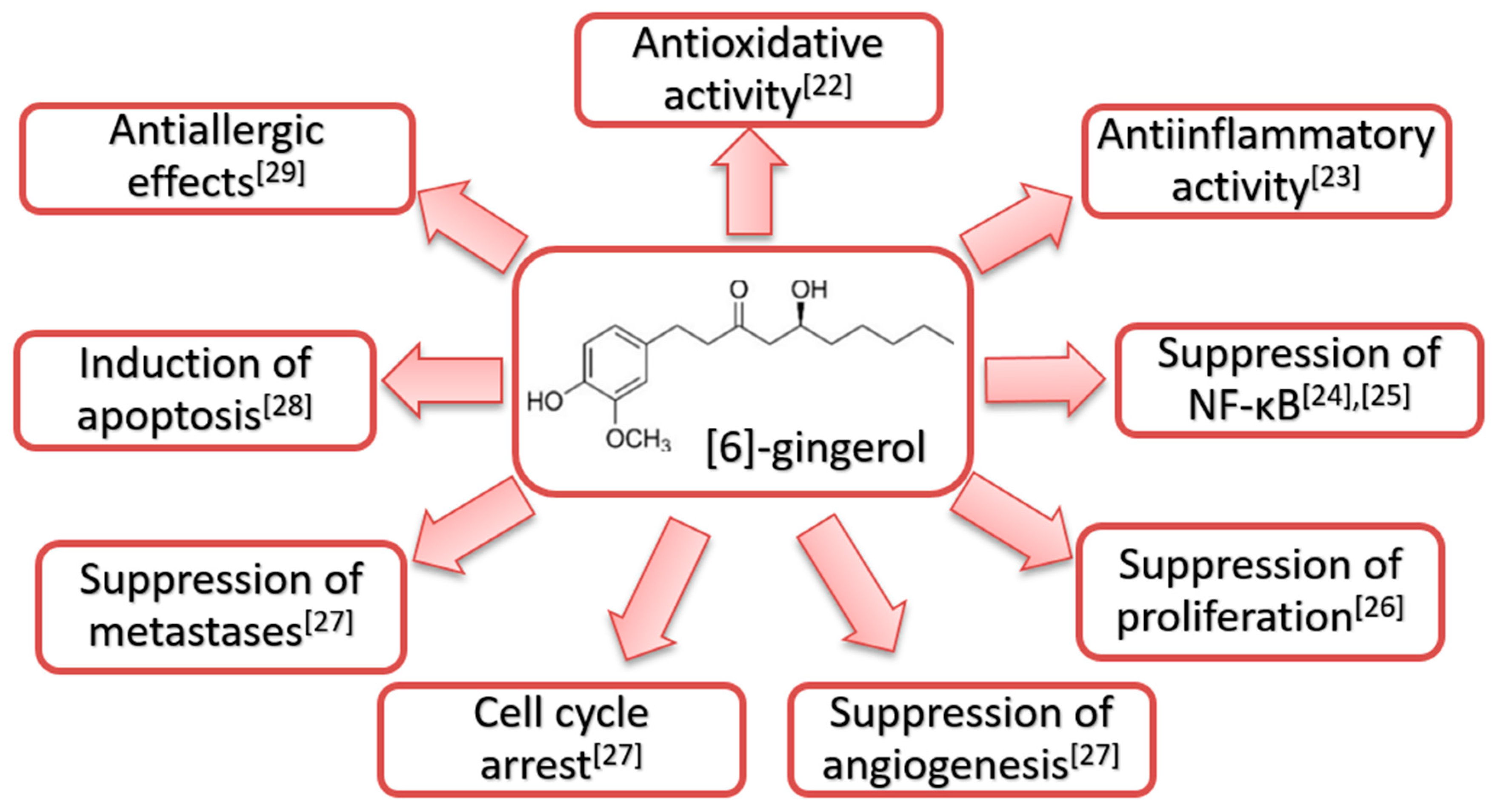
1.2. [6]-Gingerol and Glutathione
2. Results and Discussion
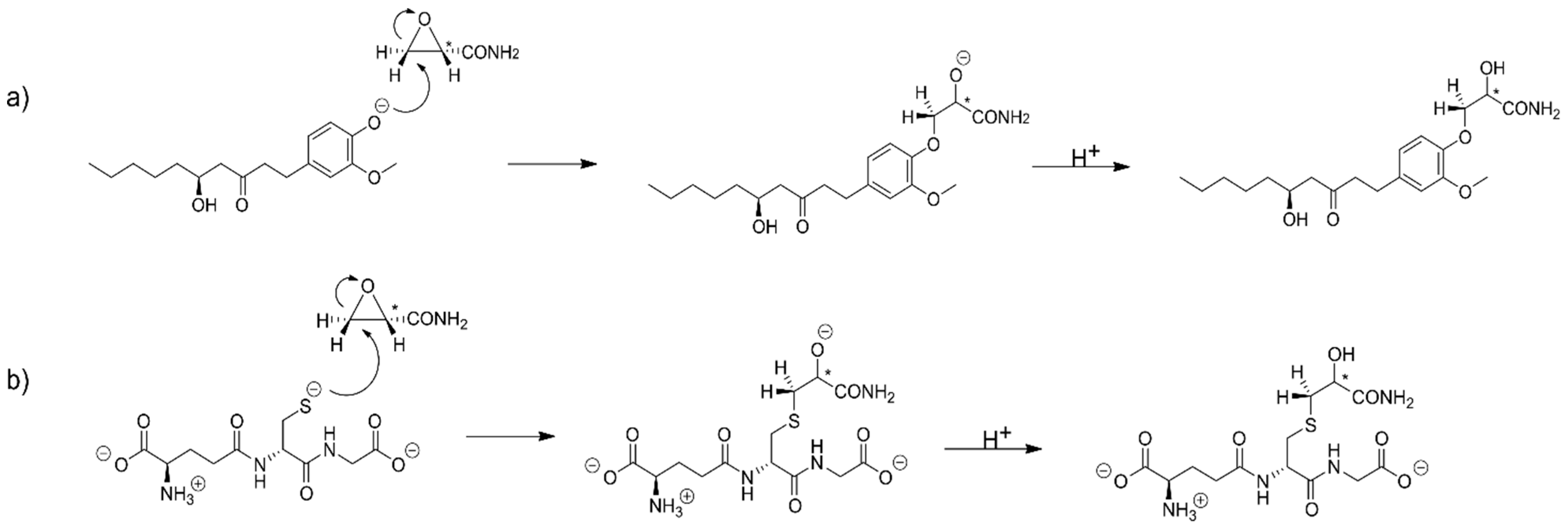
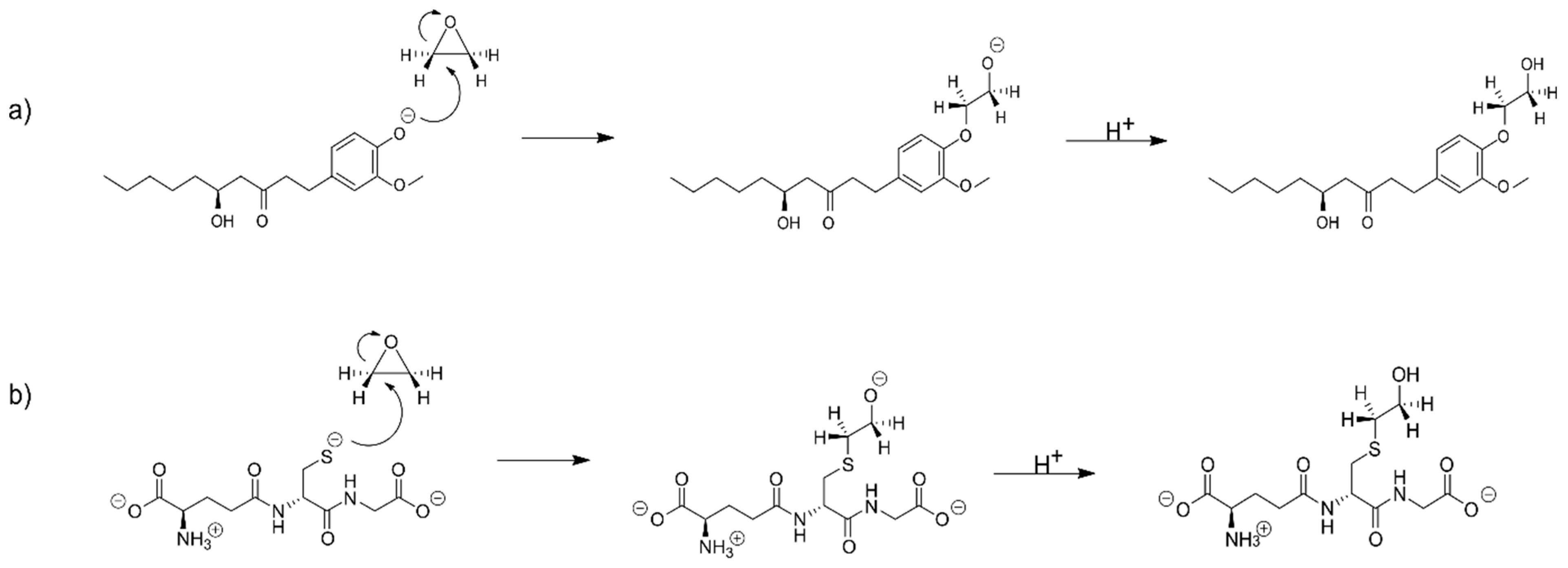
2.1. The Alkylation Reaction of Glycidamide with [6]-Gingerol and Glutathione
2.2. The Alkylation Reaction of Styrene Oxide with [6]-Gingerol and Glutathione
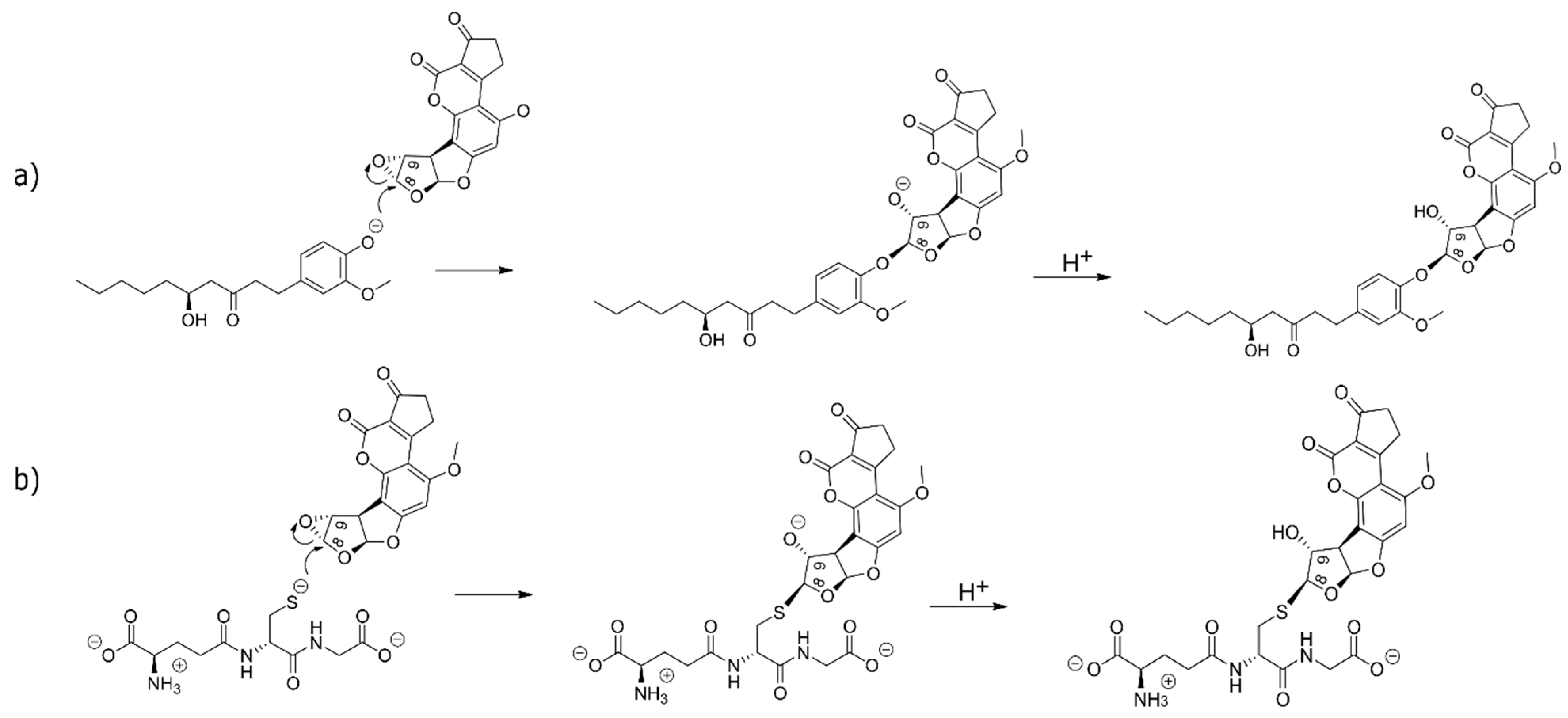
2.3. The Alkylation Reaction of AFB1 Exo-8,9-Epoxide with [6]-Gingerol and Glutathione
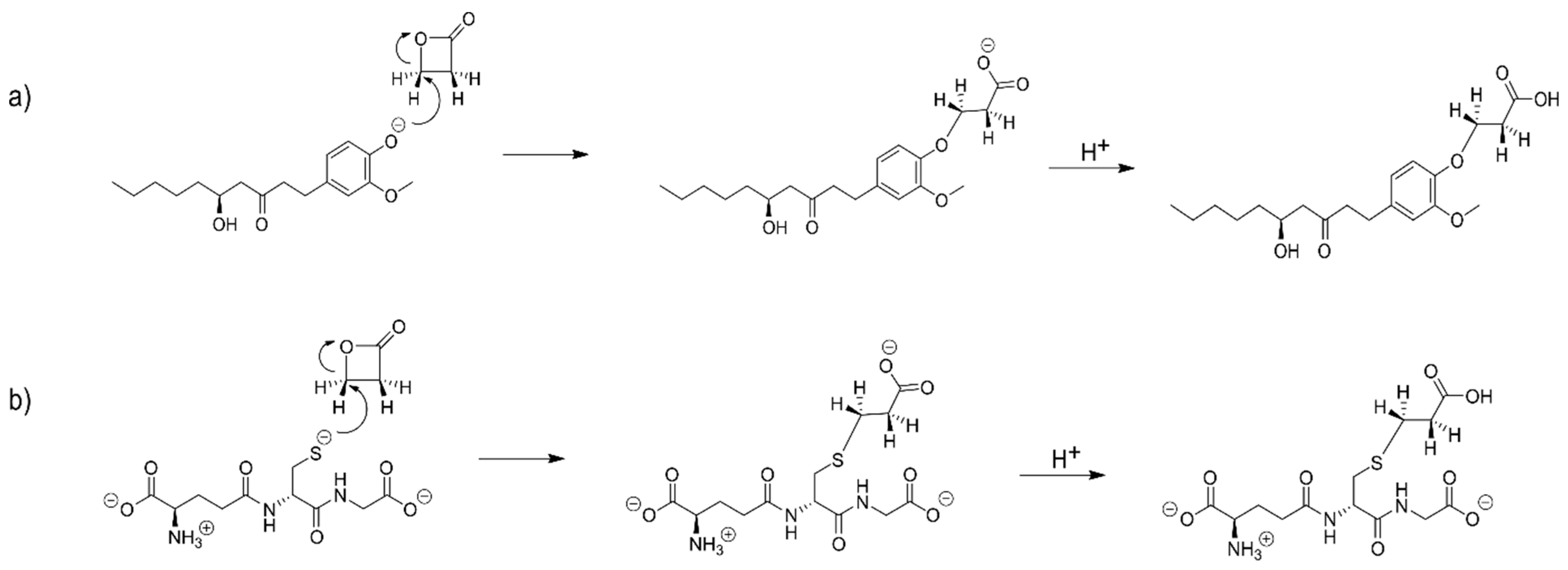
2.4. The Alkylation Reaction of β-Propiolactone with [6]-Gingerol and Glutathione
2.5. The Alkylation Reaction of Ethylene Oxide with [6]-Gingerol and Glutathione
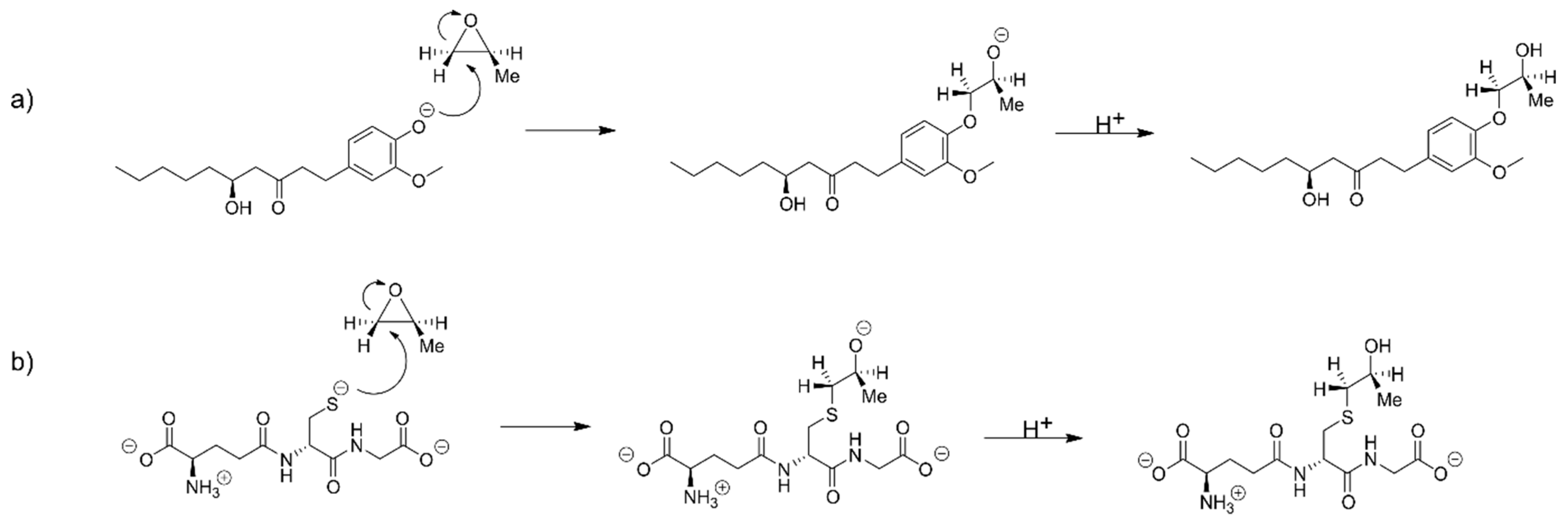
2.6. The Alkylation Reaction of Propylene Oxide with [6]-Gingerol and Glutathione
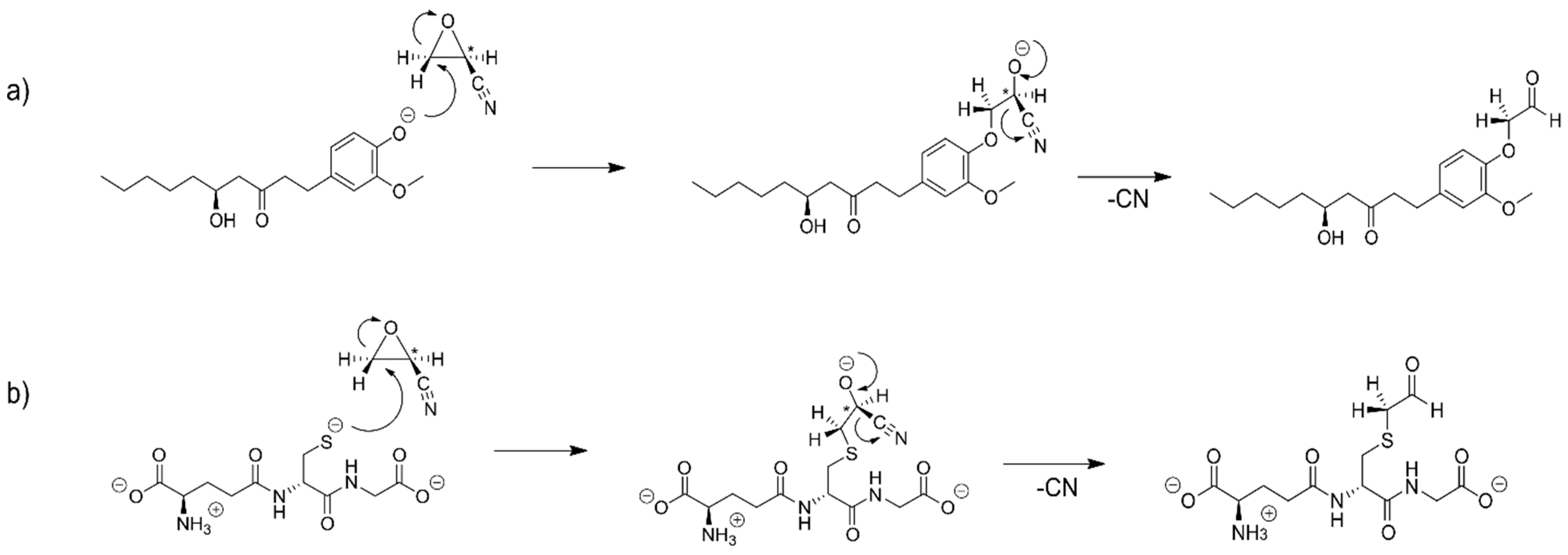
2.7. The Alkylation Reaction of 2-Cyanoethylene Oxide with [6]-Gingerol and Glutathione
2.8. The Alkylation Reaction of Chloroethylene Oxide with [6]-Gingerol and Glutathione
2.9. The Alkylation Reaction of Vinyl Carbamate Epoxide with [6]-Gingerol and Glutathione
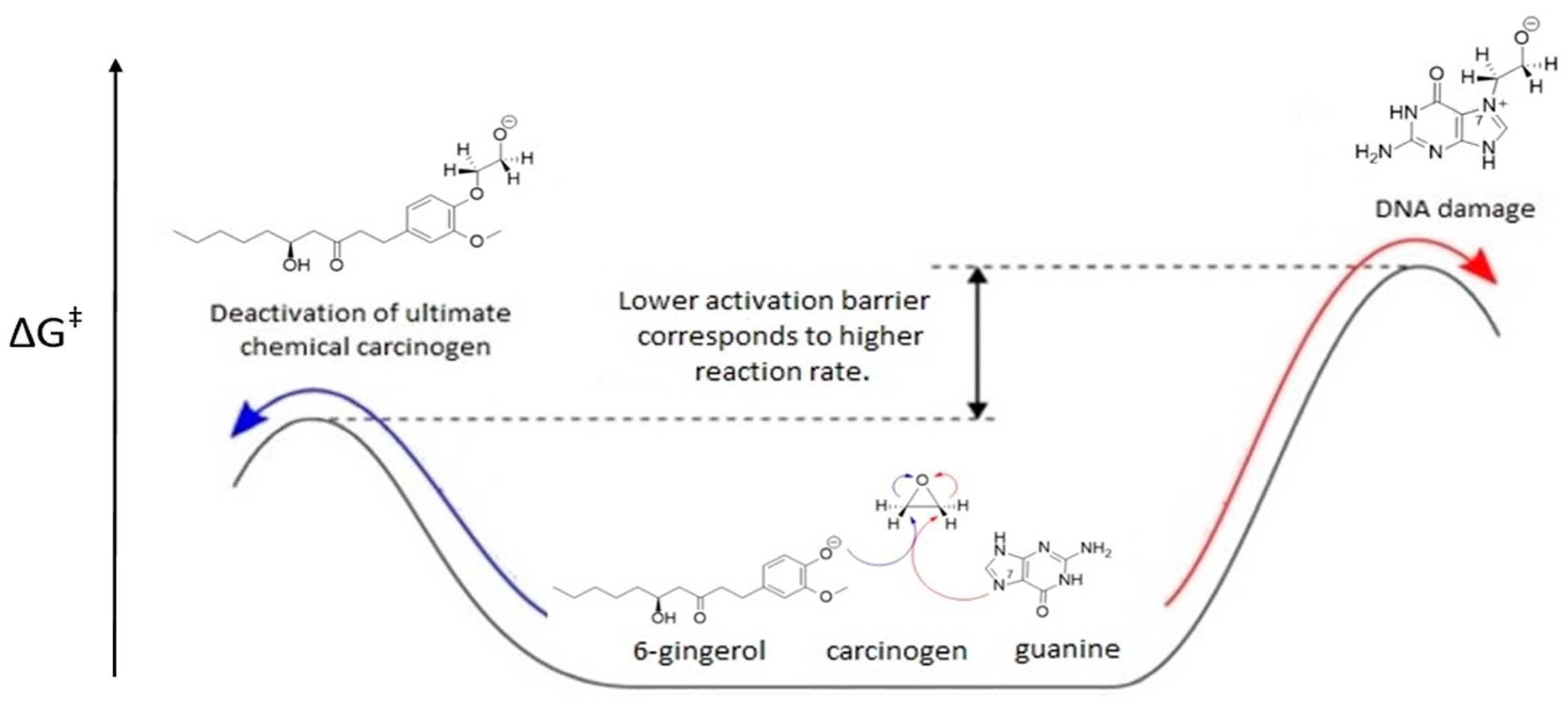
2.10. General Remarks
2.11. The Comparison of Activation Free Energies Obtained with Implicit Solvation Models for Alkylation Reactions of the Studied Chemical Carcinogens with [6]-Gingerol and Glutathione
3. Computational Methods
4. Conclusions and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AFB1 | Aflatoxin B1 |
| AP-1 | Activator protein 1 |
| BPL | Beta-propiolactone |
| CEO | Chloroethylene oxide |
| DNA | Deoxyribonucleic acid |
| ERK1/2 | Extracellular signal–regulated kinase |
| ETO | Ethylene oxide |
| GSH | Glutathione |
| HF | Hartree−Fock level of theory |
| JNK | c-Jun N-terminal kinase |
| LD | Langevin dipoles implicit solvation model |
| MAPK | Mitogen-activated protein kinase |
| MMP-9 | Matrix metalloproteinase 9 |
| NADP | Nicotinamide adenine dinucleotide phosphate |
| NF- κB | Nuclear factor kappa B |
| PAHs | Polyaromatic hydrocarbons |
| PO | Propylene oxide |
| SCRF | Self-consistent reaction field implicit solvation model |
| STO | Styrene-7,8-oxide |
| TPA | Tetradecanoylphorbol-13-acetate |
| VC | Vinyl chloride |
Appendix A



References
- Galesa, K.; Bren, U.; Kranjc, A.; Mavri, J. Carcinogenicity of acrylamide: A computational study. J. Agric. Food. Chem. 2008, 56, 8720–8727. [Google Scholar] [CrossRef]
- Mottram, D.S.; Wedzicha, B.L.; Dodson, A.T. Food chemistry: Acrylamide is formed in the Maillard reaction. Nature 2002, 419, 448. [Google Scholar] [CrossRef]
- Sumner, S.C.; Fennell, T.R.; Moore, T.A.; Chanas, B.; Gonzalez, F.; Ghanayem, B.I. Role of cytochrome P450 2E1 in the metabolism of acrylamide and acrylonitrile in mice. Chem. Res. Toxicol. 1999, 12, 1110–1116. [Google Scholar] [CrossRef]
- Kržan, M.; Mavri, J. Carcinogenicity of styrene oxide: Calculation of chemical reactivity. Croat. Chem. Acta 2009, 82, 317–322. [Google Scholar]
- Kolman, A.; Chovanec, M.; Osterman-Golkar, S. Genotoxic effects of ethylene oxide, propylene oxide and epichlorohydrin in humans: Update review (1990–2001). Mutat. Res. 2002, 512, 173–194. [Google Scholar] [CrossRef]
- Giri, I.; Jenkins, M.D.; Schnetz-Boutaud, N.C.; Stone, M.P. Structural Refinement of the 8, 9-Dihydro-8-(N7-guanyl)-9-hydroxy-aflatoxin B1 Adduct in a 5′-CpAFBG-3′Sequence. Chem. Res. Toxicol. 2002, 15, 638–647. [Google Scholar] [CrossRef] [PubMed]
- Bren, U.; Guengerich, F.P.; Mavri, J. Guanine alkylation by the potent carcinogen aflatoxin B1: Quantum chemical calculations. Chem. Res. Toxicol. 2007, 20, 1134–1140. [Google Scholar] [CrossRef] [PubMed]
- Bonnafous, P.; Nicolaï, M.-C.; Taveau, J.-C.; Chevalier, M.; Barrière, F.; Medina, J.; Le Bihan, O.; Adam, O.; Ronzon, F.; Lambert, O. Treatment of influenza virus with beta-propiolactone alters viral membrane fusion. Biochim. Biophys. Acta 2014, 1838, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Boutwell, R.; Colburn, N.H.; Muckerman, C.C. In vivo reactions of β-propiolactone. Ann. N.Y. Acad. Sci. 1969, 163, 751–763. [Google Scholar] [CrossRef]
- Kranjc, A.; Mavri, J. Guanine alkylation by ethylene oxide: Calculation of chemical reactivity. J. Phys. Chem. A 2006, 110, 5740–5744. [Google Scholar] [CrossRef]
- Mavri, J. Can the chemical reactivity of an ultimate carcinogen be related to its carcinogenicity? An application to propylene oxide. Toxicol. In Vitro 2013, 27, 479–485. [Google Scholar] [CrossRef]
- Gladović, M.; Španinger, E.; Bren, U. Nucleic bases alkylation with acrylonitrile and cyanoethylene oxide: A computational study. Chem. Res. Toxicol. 2017, 31, 97–104. [Google Scholar] [CrossRef]
- Bren, U.; Zupan, M.; Guengerich, F.P.; Mavri, J. Chemical reactivity as a tool to study carcinogenicity: Reaction between chloroethylene oxide and guanine. J. Org. Chem. 2006, 71, 4078–4084. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F.P.; Crawford Jr, W.M.; Watanabe, P.G. Activation of vinyl chloride to covalently bound metabolites: Roles of 2-chloroethylene oxide and 2-chloroacetaldehyde. Biochemistry 1979, 18, 5177–5182. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F.P.; Mason, P.S.; Stott, W.T.; Fox, T.R.; Watanabe, P.G. Roles of 2-haloethylene oxides and 2-haloacetaldehydes derived from vinyl bromide and vinyl chloride in irreversible binding to protein and DNA. Cancer Res. 1981, 41, 4391–4398. [Google Scholar] [PubMed]
- Lajovic, A.; Nagy, L.D.; Guengerich, F.P.; Bren, U. Carcinogenesis of urethane: Simulation versus experiment. Chem. Res. Toxicol. 2015, 28, 691. [Google Scholar] [CrossRef]
- Guengerich, F.P.; Kim, D.-H. Enzymatic oxidation of ethyl carbamate to vinyl carbamate and its role as an intermediate in the formation of 1, N6-ethenoadenosine. Chem. Res. Toxicol. 1991, 4, 413–421. [Google Scholar] [CrossRef]
- Landis-Piwowar, K.R.; Iyer, N.R. Cancer Chemoprevention: Current State of the Art. Cancer Growth Metastasis 2014, 2014, 19–25. [Google Scholar] [CrossRef]
- Srinivasan, K. Ginger rhizomes (Zingiber officinale): A spice with multiple health beneficial potentials. PharmaNutrition 2017, 5, 18–28. [Google Scholar] [CrossRef]
- Ali, B.H.; Blunden, G.; Tanira, M.O.; Nemmar, A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): A review of recent research. Food Chem. Toxicol. 2008, 46, 409–420. [Google Scholar] [CrossRef]
- Chrubasik, S.; Pittler, M.; Roufogalis, B. Zingiberis rhizoma: A comprehensive review on the ginger effect and efficacy profiles. Phytomedicine 2005, 12, 684–701. [Google Scholar] [CrossRef] [PubMed]
- Ippoushi, K.; Azuma, K.; Ito, H.; Horie, H.; Higashio, H. [6]-Gingerol inhibits nitric oxide synthesis in activated J774. 1 mouse macrophages and prevents peroxynitrite-induced oxidation and nitration reactions. Life Sci. 2003, 73, 3427–3437. [Google Scholar] [CrossRef] [PubMed]
- Tzeng, T.-F.; Liou, S.-S.; Chang, C.J.; Liu, I.-M. [6]-gingerol dampens hepatic steatosis and inflammation in experimental nonalcoholic steatohepatitis. Phytomedicine 2015, 22, 452–461. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.O.; Chun, K.-S.; Kundu, J.K.; Surh, Y.-J. Inhibitory effects of [6]-gingerol on PMA-induced COX-2 expression and activation of NF-κB and p38 MAPK in mouse skin. Biofactors 2004, 21, 27–31. [Google Scholar] [CrossRef]
- Prasad, S.; Tyagi, A.K. Ginger and its constituents: Role in prevention and treatment of gastrointestinal cancer. Gastroenterol. Res. Pract. 2015. [Google Scholar] [CrossRef]
- Davies, M.; Robinson, M.; Smith, E.; Huntley, S.; Prime, S.; Paterson, I. Induction of an epithelial to mesenchymal transition in human immortal and malignant keratinocytes by TGF-β1 involves MAPK, Smad and AP-1 signalling pathways. J. Cell. Biochem. 2005, 95, 918–931. [Google Scholar] [CrossRef]
- Kim, E.-C.; Min, J.-K.; Kim, T.-Y.; Lee, S.-J.; Yang, H.-O.; Han, S.; Kim, Y.-M.; Kwon, Y.-G. [6]-Gingerol, a pungent ingredient of ginger, inhibits angiogenesis in vitro and in vivo. Biochem. Biophys. Res. Commun. 2005, 335, 300–308. [Google Scholar] [CrossRef]
- Radhakrishnan, E.; Bava, S.V.; Narayanan, S.S.; Nath, L.R.; Thulasidasan, A.K.T.; Soniya, E.V.; Anto, R.J. [6]-Gingerol induces caspase-dependent apoptosis and prevents PMA-induced proliferation in colon cancer cells by inhibiting MAPK/AP-1 signaling. PLoS ONE 2014, 9, e104401. [Google Scholar] [CrossRef]
- Kawamoto, Y.; Ueno, Y.; Nakahashi, E.; Obayashi, M.; Sugihara, K.; Qiao, S.; Iida, M.; Kumasaka, M.Y.; Yajima, I.; Goto, Y. Prevention of allergic rhinitis by ginger and the molecular basis of immunosuppression by 6-gingerol through T cell inactivation. J. Nutr. Biochem. 2016, 27, 112–122. [Google Scholar] [CrossRef]
- Kim, S.O.; Kim, M.R. [6]-gingerol prevents disassembly of cell junctions and activities of MMPs in invasive human pancreas cancer cells through ERK/NF-κB/snail signal transduction pathway. Evid. Based Complement. Alternat. Med. 2013. [Google Scholar] [CrossRef]
- Hayes, J.D.; McLellan, L.I. Glutathione and glutathione-dependent enzymes represent a co-ordinately regulated defence against oxidative stress. Free Radical Res. 1999, 31, 273–300. [Google Scholar] [CrossRef] [PubMed]
- Pompella, A.; Visvikis, A.; Paolicchi, A.; De Tata, V.; Casini, A.F. The changing faces of glutathione, a cellular protagonist. Biochem. Pharmacol. 2003, 66, 1499–1503. [Google Scholar] [CrossRef]
- Dringen, R.; Brandmann, M.; Hohnholt, M.C.; Blumrich, E.-M. Glutathione-dependent detoxification processes in astrocytes. Neurochem. Res. 2015, 40, 2570–2582. [Google Scholar] [CrossRef]
- MarvinSketch. version 18.5.0, developed by ChemAxon. 2019. Available online: http://www.chemaxon.com/products/marvin/marvinsketch/ (accessed on 20 January 2020).
- Schaftenaar, G.; Noordik, J.H. Molden: A pre-and post-processing program for molecular and electronic structures. J. Comput. Aided Mol. Des. 2000, 14, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Hanwell, M.D.; Curtis, D.E.; Lonie, D.C.; Vandermeersch, T.; Zurek, E.; Hutchison, G.R. Avogadro: An advanced semantic chemical editor, visualization, and analysis platform. J. Cheminform. 2012, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Manso, J.A.; Pérez-Prior, M.T.; García-Santos, M.d.P.; Calle, E.; Casado, J. A kinetic approach to the alkylating potential of carcinogenic lactones. Chem. Res. Toxicol. 2005, 18, 1161–1166. [Google Scholar] [CrossRef] [PubMed]
- Miertuš, S.; Scrocco, E.; Tomasi, J. Electrostatic interaction of a solute with a continuum. A direct utilizaion of AB initio molecular potentials for the prevision of solvent effects. Chem. Phys. 1981, 55, 117–129. [Google Scholar] [CrossRef]
- Florián, J.; Warshel, A. Langevin dipoles model for ab initio calculations of chemical processes in solution: Parametrization and application to hydration free energies of neutral and ionic solutes and conformational analysis in aqueous solution. J. Phys. Chem. B 1997, 101, 5583–5595. [Google Scholar] [CrossRef]
- Florián, J.; Warshel, A. Calculations of hydration entropies of hydrophobic, polar, and ionic solutes in the framework of the Langevin dipoles solvation model. J. Phys. Chem. B 1999, 103, 10282–10288. [Google Scholar] [CrossRef]
- Borštnik, U.; Hodošcek, M.; Janežič, D. Improving the performance of molecular dynamics simulations on parallel clusters. J. Chem. Inf. Comput. Sci. 2004, 44, 359–364. [Google Scholar] [CrossRef]
- Frisch, M.; Trucks, G.; Schlegel, H.; Scuseria, G.; Robb, M.; Cheeseman, J.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. Gaussian 09, revision D. 01; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Oakley, A. Glutathione transferases: A structural perspective. Drug Metab. Rev. 2011, 43, 138–151. [Google Scholar] [CrossRef] [PubMed]

| Method/Basis Set HF/6-311++G(d,p) | ωTS [i cm−1] d | ωR [cm−1] e | dTS [Å] f | dR [Å] g | |||
|---|---|---|---|---|---|---|---|
| Glycidamide | |||||||
| [6]-Gingerol | 30.81 | −5.58 | 25.23 | 646.37 | 6.99 | 1.91 | 3.18 |
| Glutathione | 35.50 | −3.28 | 32.22 | 585.04 | 14.51 | 2.40 | 4.12 |
| Styrene Oxide | |||||||
| [6]-Gingerol | 26.42 | −3.76 | 21.65 | 647.91 | 2.34 | 1.96 | 3.19 |
| Glutathione | 30.23 | −1.33 | 31.56 | 581.77 | 12.00 | 2.44 | 4.14 |
| AFB1 Exo-8.9-Epoxide | |||||||
| [6]-Gingerol | 20.87 | −5.49 | 15.38 | 317.15 | 7.02 | 2.21 | 3.62 |
| Glutathione | 17.56 | −1.88 | 15.68 | 213.25 | 10.74 | 2.60 | 3.97 |
| Beta Propiolactone | |||||||
| [6]-Gingerol | 16.50 | −1.57 | 14.93 | 644.29 | 6.27 | 2.05 | 2.81 |
| Glutathione | 22.89 | −0.38 | 22.51 | 614.21 | 14.72 | 2.55 | 3.85 |
| Ethylene Oxide | |||||||
| [6]-Gingerol | 24.48 | −0.45 | 24.03 | 629.98 | 8.29 | 1.95 | 3.26 |
| Glutathione | 30.63 | −0.61 | 30.02 | 569.22 | 11.68 | 2.44 | 4.12 |
| Propylene Oxide | |||||||
| [6]-Gingerol | 24.41 | −0.04 | 24.37 | 615.67 | 7.58 | 1.94 | 3.28 |
| Glutathione | 31.01 | −0.99 | 30.02 | 560.64 | 4.92 | 2.42 | 4.04 |
| 2-Cyanoethylene Oxide | |||||||
| [6]-Gingerol | 20.72 | −0.45 | 20.27 | 677.50 | 7.67 | 1.98 | 3.07 |
| Glutathione | 26.36 | −2.35 | 24.01 | 623.35 | 6.57 | 2.48 | 3.77 |
| Chloroethylene Oxide | |||||||
| [6]-Gingerol | 21.10 | −2.37 | 18.73 | 651.63 | 6.55 | 2.05 | 3.18 |
| Glutathione | 21.60 | −0.02 | 21.59 | 627.92 | 14.19 | 2.57 | 3.91 |
| Vinyl Carbamate Epoxide | |||||||
| [6]-Gingerol | 19.65 | −0.18 | 18.97 | 620.03 | 5.70 | 2.06 | 3.17 |
| Glutathione | 25.28 | −0.70 | 24.59 | 584.39 | 15.41 | 2.47 | 4.92 |
| Method/Basis Set HF/6-311++G(d,p) | [6]-Gingerol [kcal/mol] | Glutathione [kcal/mol] | Guanine [kcal/mol] | a Experimental Value for Guanine [kcal/mol] | |||
|---|---|---|---|---|---|---|---|
| b | |||||||
| Styrene Oxide | 21.65 | 26.03 | 31.56 | 30.16 | -[4] | 27.65 [4] | 26.5 [4] |
| Propylene Oxide | 24.37 | 23.57 | 30.02 | 26.06 | 21.26 [11] | 25.15 [11] | 25.4 [11] |
| Ethylene Oxide | 24.03 | 21.99 | 30.02 | 24.17 | 21 [10] | 24.62 [10] | 24.7 [10] |
| Glycidamide | 25.23 | 19.65 | 32.22 | 28.19 | 25.78 [1] | 23.55 [1] | 22.8 [1] |
| Vinyl Carbamate Epoxide | 18.97 | 19.16 | 24.59 | 22.09 | 22.15 [16] | 19.13 [16] | 22.4 [16] |
| β-Propiolactone | 14.93 | 12.10 | 22.51 | 17.79 | 22.45 | 12.06 | 20.8 [37] |
| Chloroethylene oxide | 18.73 | 17.57 | 21.59 | 18.78 | 17.26 [13] | 22.87 [13] | 19.5 [13] |
| 2-Cyanoethylene Oxide | 20.27 | 18.19 | 26.36 | 26.78 | 28.48 [12] | 19.02 [12] | 19.2 [12] |
| AFB1 Exo-8.9-Epoxide | 15.38 | 5.44 | 15.68 | 5.21 | 18.9 [7] | 14.25 [7] | 15.1 [7] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Furlan, V.; Bren, U. Protective Effects of [6]-Gingerol Against Chemical Carcinogens: Mechanistic Insights. Int. J. Mol. Sci. 2020, 21, 695. https://doi.org/10.3390/ijms21030695
Furlan V, Bren U. Protective Effects of [6]-Gingerol Against Chemical Carcinogens: Mechanistic Insights. International Journal of Molecular Sciences. 2020; 21(3):695. https://doi.org/10.3390/ijms21030695
Chicago/Turabian StyleFurlan, Veronika, and Urban Bren. 2020. "Protective Effects of [6]-Gingerol Against Chemical Carcinogens: Mechanistic Insights" International Journal of Molecular Sciences 21, no. 3: 695. https://doi.org/10.3390/ijms21030695
APA StyleFurlan, V., & Bren, U. (2020). Protective Effects of [6]-Gingerol Against Chemical Carcinogens: Mechanistic Insights. International Journal of Molecular Sciences, 21(3), 695. https://doi.org/10.3390/ijms21030695






