Are There Epigenetic Oxytocin-Mediated Effects on the Mother and Infant during Physiological Childbirth?
Abstract
1. Introduction
1.1. The Neuro-Endocrine Role of Oxytocin
1.2. Oxytocin in Early Life, Animal Experiments
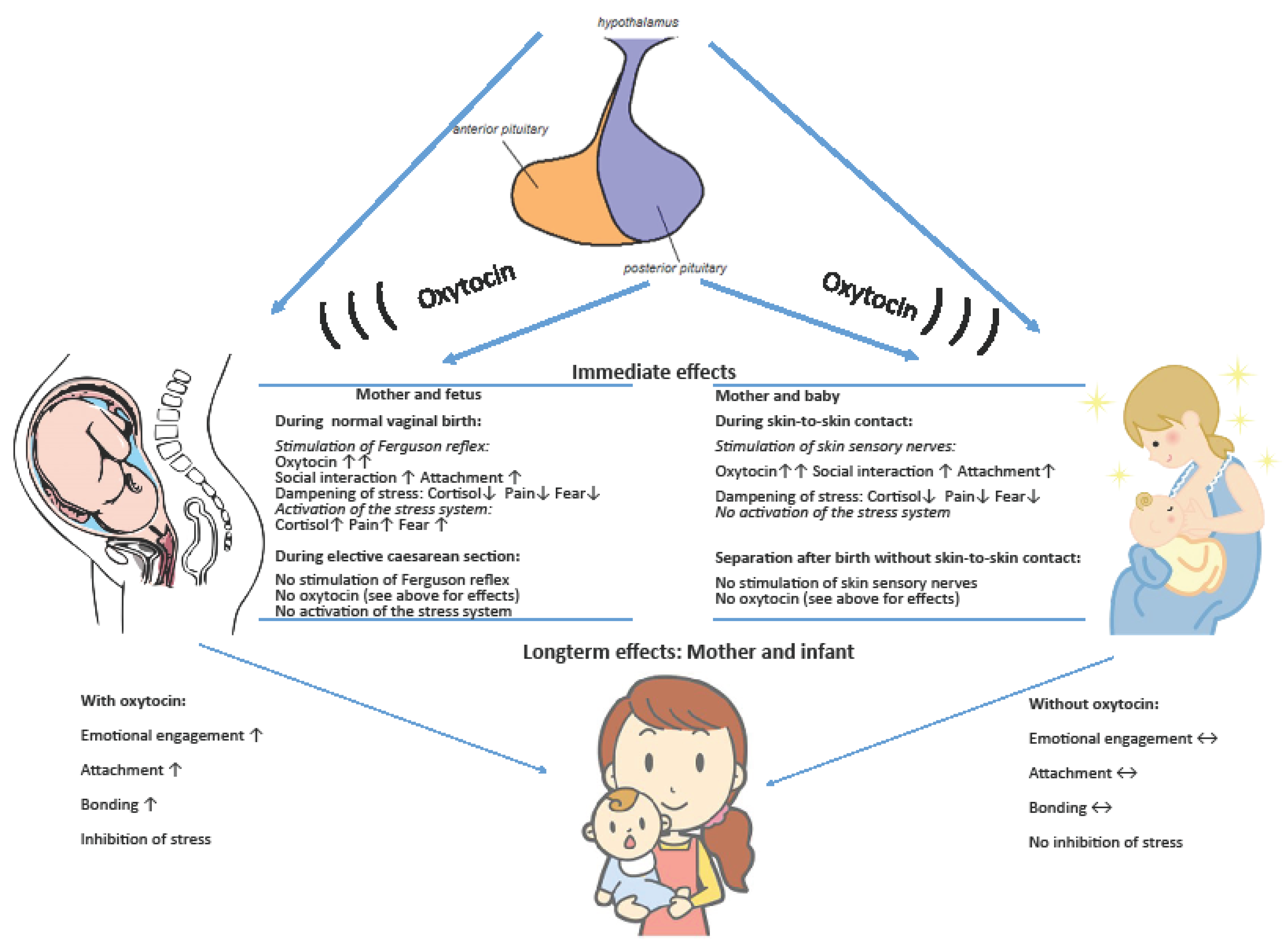
1.3. Oxytocin during Childbirth in Humans
2. Methods
3. Results
3.1. Epigenetic Changes of Oxytocin or the Oxytocin Receptor Gene in Relation to Labour and Birth
3.2. Polymorphisms of the Oxytocin Receptor
3.3. Impact of Caesarean Section on Oxytocin Levels
3.4. Longterm Impact of Mother and Baby Interaction Immediately after Birth
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hofer, M.A. Early relationships as regulators of infant physiology and behavior. Acta Paediatr. 1994, 83, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Martínez, F.; Uvnäs-Moberg, K.; Petersson, M.; Olausson, H.A.; Jiménez-Estrada, I. Neuropeptides as neuroprotective agents: Oxytocin a forefront developmental player in the mammalian brain. Prog. Neurobiol. 2014, 123, 37–78. [Google Scholar] [CrossRef]
- Anacker, C.; O’Donnell, K.J.; Meaney, M.J. Early life adversity and the epigenetic programming of hypothalamic-pituitary-adrenal function. Dialogues Clin Neurosci. 2014, 16, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Moberg, K.U.; Handlin, L.; Petersson, M. Neuroendocrine mechanisms involved in the physiological effects caused by skin-to-skin contact—With a particular focus on the oxytocinergic system. Infant Behav. Dev. 2020, 61, 101482. [Google Scholar] [CrossRef] [PubMed]
- Uvnäs-Moberg, K.; Ehandlin, L.; Epetersson, M. Self-soothing behaviors with particular reference to oxytocin release induced by non-noxious sensory stimulation. Front. Psychol. 2015, 5, 1529. [Google Scholar] [CrossRef] [PubMed]
- Monk, C.; Spicer, J.; Champagne, F.A. Linking prenatal maternal adversity to developmental outcomes in infants: The role of epigenetic pathways. Dev. Psychopathol. 2012, 24, 1361–1376. [Google Scholar] [CrossRef]
- Dahlen, H.G.; Kennedy, H.P.; Anderson, C.M.; Bell, A.F.; Clark, A.; Foureur, M.J.; Ohm, J.; Shearman, A.; Taylor, J.Y.; Wright, M.L.; et al. The EPIIC hypothesis: Intrapartum effects on the neonatal epigenome and consequent health outcomes. Med. Hypotheses 2013, 80, 656–662. [Google Scholar] [CrossRef]
- Lagercrantz, H.; Slotkin, T.A. The “Stress” of Being Born. Sci. Am. 1986, 254, 100–107. [Google Scholar] [CrossRef]
- Kalantaridou, S.; Zoumakis, E.; Makrigiannakis, A.; Lavasidis, L.; Vrekoussis, T.; Chrousos, G. Corticotropin-releasing hormone, stress and human reproduction: An update. J. Reprod. Immunol. 2010, 85, 33–39. [Google Scholar] [CrossRef]
- Olza, I.; Uvnas-Moberg, K.; Ekström, A.; Leahy-Warren, P.; Karlsdottir, S.I.; Nieuwenhuijze, M.; Villarmea, S.; Hadjigeorgiou, E.; Kazmierczak, M.; Spyridou, A.; et al. Birth as a neuro-psycho-social event: An integrative model of maternal experiences and their relation to neurohormonal events during childbirth. PLoS ONE 2020, 15, e0230992. [Google Scholar] [CrossRef]
- Uvnäs-Moberg, K.; Ekström-Bergström, A.; Berg, M.; Buckley, S.; Pajalic, Z.; Hadjigeorgiou, E.; Kotłowska, A.; Lengler, L.; Kielbratowska, B.; Leon-Larios, F.; et al. Maternal plasma levels of oxytocin during physiological childbirth—A systematic review with implications for uterine contractions and central actions of oxytocin. BMC Pregnancy Childbirth 2019. [Google Scholar] [CrossRef] [PubMed]
- Moberg, K.U.; Handlin, L.; Kendall-Tackett, K.; Petersson, M. Oxytocin is a principal hormone that exerts part of its effects by active fragments. Med. Hypotheses 2019, 133, 109394. [Google Scholar] [CrossRef] [PubMed]
- Petersson, M.; Lundeberg, T.; Uvnäs-Moberg, K. Oxytocin enhances the effects of clonidine on blood pressure and locomotor activity in rats. J. Auton. Nerv. Syst. 1999, 78, 49–56. [Google Scholar] [CrossRef]
- Pedersen, C.A. Biological Aspects of Social Bonding and the Roots of Human Violence. Ann. N. Y. Acad. Sci. 2006, 1036, 106–127. [Google Scholar] [CrossRef] [PubMed]
- Keverne, E.B.; Kendrick, K.M. Maternal behaviour in sheep and its neuroendocrine regulation. Acta Paediatr. 1994, 83, 47–56. [Google Scholar] [CrossRef]
- Uvnas Moberg, K. The Biological Guide to Motherhood; Praeclarus Press LLC: Amarillo, TX, USA, 2016. [Google Scholar]
- Inoue, T.; Kimura, T.; Azuma, C.; Inazawa, J.; Takemura, M.; Kikuchi, T.; Kubota, Y.; Ogita, K.; Saji, F. Structural organization of the human oxytocin receptor gene. J. Biol. Chem. 1994, 269. [Google Scholar]
- Kusui, C.; Kimura, T.; Ogita, K.; Nakamura, H.; Matsumura, Y.; Koyama, M.; Azuma, C.; Murata, Y. DNA Methylation of the Human Oxytocin Receptor Gene Promoter Regulates Tissue-Specific Gene Suppression. Biochem. Biophys. Res. Commun. 2001, 289, 681–686. [Google Scholar] [CrossRef]
- Kenkel, W.; Perkeybile, A.-M.; Yee, J.R.; Pournajafi-Nazarloo, H.; Lillard, T.S.; Ferguson, E.F.; Wroblewski, K.L.; Ferris, C.F.; Carter, C.S.; Connelly, J.J. Behavioral and epigenetic consequences of oxytocin treatment at birth. Sci. Adv. 2019, 5, eaav2244. [Google Scholar] [CrossRef]
- Ishak, W.W.; Kahloon, M.; Fakhry, H. Oxytocin role in enhancing well-being: A literature review. J. Affect. Disord. 2011, 130, 1–9. [Google Scholar] [CrossRef]
- Buemann, B.; Uvnäs-Moberg, K. Oxytocin may have a therapeutical potential against cardiovascular disease. Possible pharmaceutical and behavioral approaches. Med. Hypotheses 2020, 138, 109597. [Google Scholar] [CrossRef]
- Serpeloni, F.; Radtke, K.M.; Hecker, T.; Sill, J.; Vukojevic, V.; De Assis, S.G.; Schauer, M.; Elbert, T.; Nätt, D. Does Prenatal Stress Shape Postnatal Resilience?—An Epigenome-Wide Study on Violence and Mental Health in Humans. Front. Genet. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, J.C.; Mitchell, J.M. Augmenting Treatment for Posttraumatic Stress Disorder and Co-Occurring Conditions with Oxytocin. Curr. Treat. Options Psychiatry 2019, 6, 132–142. [Google Scholar] [CrossRef]
- Auyeung, B.; Lombardo, M.V.; Heinrichs, M.; Chakrabarti, B.; Sule, A.; Deakin, J.B.; Bethlehem, R.A.I.; Dickens, L.; Mooney, N.; Sipple, J.A.N.; et al. Oxytocin increases eye contact during a real-time, naturalistic social interaction in males with and without autism. Transl. Psychiatry 2015, 5, e507. [Google Scholar] [CrossRef] [PubMed]
- Champagne, F.; Diorio, J.; Sharma, S.; Meaney, M.J. Naturally occurring variations in maternal behavior in the rat are associated with differences in estrogen-inducible central oxytocin receptors. Proc. Natl. Acad. Sci. USA 2001, 98, 12736–12741. [Google Scholar] [CrossRef] [PubMed]
- Holst, S.; Uvnäs-Moberg, K.; Petersson, M. Postnatal oxytocin treatment and postnatal stroking of rats reduce blood pressure in adulthood. Auton. Neurosci. 2002, 99, 85–90. [Google Scholar] [CrossRef]
- Francis, D.D.; Champagne, F.C.; Meaney, M.J. Variations in Maternal Behaviour are Associated with Differences in Oxytocin Receptor Levels in the Rat. J. Neuroendocr. 2001, 12, 1145–1148. [Google Scholar] [CrossRef] [PubMed]
- Pruessner, J.C.; Champagne, F.; Meaney, M.J.; Dagher, A. Dopamine Release in Response to a Psychological Stress in Humans and Its Relationship to Early Life Maternal Care: A Positron Emission Tomography Study Using [11C]Raclopride. J. Neurosci. 2004, 24, 2825–2831. [Google Scholar] [CrossRef]
- Diaz-Cabiale, Z.; Narváez, M.; Petersson, M.; Uvnäs-Moberg, K.; Fuxe, K. Oxytocin/alpha(2)-Adrenoceptor interactions in feeding responses. Neuroendocrinology 2000, 71, 209–218. [Google Scholar] [CrossRef]
- Diaz-Cabiale, Z.; Olausson, H.; Sohlström, A.; Agnati, L.F.; Narvaez, J.A.; Uvnasmoberg, K.; Fuxe, K. Long-Term Modulation By Postnatal Oxytocin of the alpha2-Adrenoceptor Agonist Binding Sites in Central Autonomic Regions and the Role of Prenatal Stress. J. Neuroendocr. 2004, 16, 183–190. [Google Scholar] [CrossRef]
- Sohlström, A.; Carlsson, C.; Uvnäs-Moberg, K. Effects of oxytocin treatment in early life on body weight and corticosterone in adult offspring from ad libitum-fed and food-restricted rats. Biol. Neonate 2000, 78, 33–40. [Google Scholar] [CrossRef]
- Kim, S.H.; Bennett, P.R.; Terzidou, V. Advances in the role of oxytocin receptors in human parturition. Mol. Cell. Endocrinol. 2017, 449, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Arrowsmith, S.; Wray, S. Oxytocin: Its Mechanism of Action and Receptor Signalling in the Myometrium. J. Neuroendocr. 2014, 26, 356–369. [Google Scholar] [CrossRef] [PubMed]
- Barbara, M.A.; Abdilla, Y.; Calleja-Agius, J.; Andria, B.M.; Ylenia, A. An Introduction to Epigenetics. Neonatal Netw. 2017, 36, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Schlinzig, T.; Johansson, S.; Gunnar, A.; Ekström, T.J.; Norman, M. Epigenetic modulation at birth-altered DNA-methylation in white blood cells after Caesarean section. Acta Paediatr. 2009, 98, 1096–1099. [Google Scholar] [CrossRef]
- Virani, S.; Dolinoy, D.C.; Halubai, S.; Jones, T.R.; Domino, S.E.; Rozek, L.S.; Nahar, M.S.; Padmanabhan, V. Delivery type not associated with global methylation at birth. Clin. Epigenet. 2012, 4, 8. [Google Scholar] [CrossRef]
- Almgren, M.; Schlinzig, T.; Gomez-Cabrero, D.; Gunnar, A.; Sundin, M.; Johansson, S.; Norman, M.; Ekström, T.J. Cesarean delivery and hematopoietic stem cell epigenetics in the newborn infant: Implications for future health? Am. J. Obstet. Gynecol. 2014, 211, 502.e1–502.e8. [Google Scholar] [CrossRef]
- Franz, M.B.; Poterauer, M.; Elhenicky, M.; Stary, S.; Birner, P.; Vinatzer, U.; Husslein, P.; Streubel, B.; Husslein, H. Global and single gene DNA methylation in umbilical cord blood cells after elective caesarean: A pilot study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 179, 121–124. [Google Scholar] [CrossRef]
- Kingsbury, M.A.; Bilbo, S.D. The inflammatory event of birth: How oxytocin signaling may guide the development of the brain and gastrointestinal system. Front. Neuroendocr. 2019, 55, 100794. [Google Scholar] [CrossRef]
- Chen, F.S.; Kumsta, R.; Von Dawans, B.; Monakhov, M.; Ebstein, R.P.; Heinrichs, M. Common oxytocin receptor gene (OXTR) polymorphism and social support interact to reduce stress in humans. Proc. Natl. Acad. Sci. USA 2011, 108, 19937–19942. [Google Scholar] [CrossRef]
- Füeg, F.; Santos, S.; Haslinger, C.; Stoiber, B.; Schäffer, L.; Grünblatt, E.; Zimmermann, R.; Simões-Wüst, A.P. Influence of oxytocin receptor single nucleotide sequence variants on contractility of human myometrium: An in vitro functional study. BMC Med. Genet. 2019, 20, 1–7. [Google Scholar] [CrossRef]
- Reitman, M.E.; Conell-Price, B.J.; Evansmilth, J.; Olson, B.L.; Drosinos, M.S.; Jasper, M.N.; Randolph, M.P.; Smiley, P.R.M.; Shafer, M.S.; Flood, M.P. β2-Adrenergic Receptor Genotype and Other Variables that Contribute to Labor Pain and Progress. Anesthesiology 2011, 114, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Reinl, E.L.; Goodwin, Z.A.; Raghuraman, N.; Lee, G.Y.; Jo, E.Y.; Gezahegn, B.M.; Pillai, M.K.; Cahill, A.G.; Strong, C.D.G.; England, S.K. Novel oxytocin receptor variants in laboring women requiring high doses of oxytocin. Am. J. Obstet. Gynecol. 2017, 217, 214.e1–214.e8. [Google Scholar] [CrossRef] [PubMed]
- Marchini, G.; Lagercrantz, H.; Winberg, J.; Uvnäs-Moberg, K. Fetal and maternal plasma levels of gastrin, somatostatin and oxytocin after vaginal delivery and elective cesarean section. Early Hum. Dev. 1988, 18, 73–79. [Google Scholar] [CrossRef]
- Nissen, E.; Gustavsson, P.; Widström, A.-M.; Uvnäs-Moberg, K. Oxytocin, prolactin, milk production and their relationship with personality traits in women after vaginal delivery or Cesarean section. J. Psychosom. Obstet. Gynecol. 1998, 19, 49–58. [Google Scholar] [CrossRef]
- Nissen, E.; Uvnäs-Moberg, K.; Svensson, K.; Stock, S.; Widström, A.-M.; Winberg, J. Different patterns of oxytocin, prolactin but not cortisol release during breastfeeding in women delivered by Caesarean section or by the vaginal route. Early Hum. Dev. 1996, 45, 103–118. [Google Scholar] [CrossRef]
- Jonas, W.; Johansson, L.; Nissen, E.; Ejdebäck, M.; Ransjö-Arvidson, A.; Uvnäs-Moberg, K. Effects of Intrapartum Oxytocin Administration and Epidural Analgesia on the Concentration of Plasma Oxytocin and Prolactin, in Response to Suckling During the Second Day Postpartum. Breastfeed. Med. 2009, 4, 71–82. [Google Scholar] [CrossRef]
- Cetisli, N.E.; Arkan, G.; Top, E.D. Maternal attachment and breastfeeding behaviors according to type of delivery in the immediate postpartum period. Rev. Assoc. Médica Bras. 2018, 64, 164–169. [Google Scholar] [CrossRef]
- Chaplin, J.; Kelly, J.; Kildea, S.V.; Information, P.E.K.F.C. Maternal perceptions of breastfeeding difficulty after caesarean section with regional anaesthesia: A qualitative study. Women Birth 2016, 29, 144–152. [Google Scholar] [CrossRef]
- Prior, E.; Santhakumaran, S.; Gale, C.; Philipps, L.H.; Modi, N.; Hyde, M.J. Breastfeeding after cesarean delivery: A systematic review and meta-analysis of world literature. Am. J. Clin. Nutr. 2012, 95, 1113–1135. [Google Scholar] [CrossRef]
- Uvnäsmoberg, K.; Ekström, A.; Buckley, S.; Massarotti, C.; Pajalic, Z.; Luegmair, K.; Kotlowska, A.; Lengler, L.; Olza, I.; Grylka-Baeschlin, S.; et al. Maternal plasma levels of oxytocin during breastfeeding—A systematic review. PLoS ONE 2020, 15, e0235806. [Google Scholar] [CrossRef]
- Klaus, M.H.; Jerauld, R.; Kreger, N.C.; McAlpine, W.; Steffa, M.; Kennell, J.H. Maternal Attachment. N. Engl. J. Med. 1972, 286, 460–463. [Google Scholar] [CrossRef] [PubMed]
- Christensson, K.; Cabrera, T.; Uvnas-Moberg, K.; Winberg, J. Separation distress call in the human neonate in the absence of maternal body contact. Acta Paediatr. 1995, 84, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Velandia, M.; Uvnäs-Moberg, K.; Nissen, E. Sex differences in newborn interaction with mother or father during skin-to-skin contact after Caesarean section. Acta Paediatr. 2011, 101, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Moore, E.R.; Anderson, G.C.; Bergman, N.; Dowswell, T. Early skin-to-skin contact for mothers and their healthy newborn infants. Cochrane Database Syst. Rev. 2012, 5, CD003519. [Google Scholar] [CrossRef]
- Bystrova, K.; Ivanova, V.; Edhborg, M.; Matthiesen, A.-S.; Ransjö-Arvidson, A.-B.; Mukhamedrakhimov, R.; Uvnäs-Moberg, K.; Widström, A.-M. Early Contact versus Separation: Effects on Mother-Infant Interaction One Year Later. Birth 2009, 36, 97–109. [Google Scholar] [CrossRef]
- Bell, A.F.; Carter, C.S.; Steer, C.D.; Golding, J.; Davis, J.M.; Steffen, A.D.; Rubin, L.H.; Lillard, T.S.; Gregory, S.P.; Harris, J.C.; et al. Interaction between oxytocin receptor DNA methylation and genotype is associated with risk of postpartum depression in women without depression in pregnancy. Front. Genet. 2015, 6, 243. [Google Scholar] [CrossRef]
- Petersson, M.; Uvnäs-Moberg, K.; Erhardt, S.; Engberg, G. Oxytocin increases locus coeruleus alpha 2-adrenoreceptor responsiveness in rats. Neurosci. Lett. 1998, 255, 115–118. [Google Scholar] [CrossRef]
- Moore, S.R.; McEwen, L.M.; Quirt, J.; Morin, A.; Mah, S.M.; Barr, R.G.; Boyce, W.T.; Kobor, M.S. Epigenetic correlates of neonatal contact in humans. Dev. Psychopathol. 2017, 29, 1517–1538. [Google Scholar] [CrossRef]
- Radtke, K.M.; Ruf, M.; Gunter, H.M.; Dohrmann, K.; Schauer, M.; Meyer, A.; Elbert, T. Transgenerational impact of intimate partner violence on methylation in the promoter of the glucocorticoid receptor. Transl. Psychiatry 2011, 1, e21. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uvnäs-Moberg, K.; Gross, M.M.; Agius, A.; Downe, S.; Calleja-Agius, J. Are There Epigenetic Oxytocin-Mediated Effects on the Mother and Infant during Physiological Childbirth? Int. J. Mol. Sci. 2020, 21, 9503. https://doi.org/10.3390/ijms21249503
Uvnäs-Moberg K, Gross MM, Agius A, Downe S, Calleja-Agius J. Are There Epigenetic Oxytocin-Mediated Effects on the Mother and Infant during Physiological Childbirth? International Journal of Molecular Sciences. 2020; 21(24):9503. https://doi.org/10.3390/ijms21249503
Chicago/Turabian StyleUvnäs-Moberg, Kerstin, Mechthild M. Gross, Andee Agius, Soo Downe, and Jean Calleja-Agius. 2020. "Are There Epigenetic Oxytocin-Mediated Effects on the Mother and Infant during Physiological Childbirth?" International Journal of Molecular Sciences 21, no. 24: 9503. https://doi.org/10.3390/ijms21249503
APA StyleUvnäs-Moberg, K., Gross, M. M., Agius, A., Downe, S., & Calleja-Agius, J. (2020). Are There Epigenetic Oxytocin-Mediated Effects on the Mother and Infant during Physiological Childbirth? International Journal of Molecular Sciences, 21(24), 9503. https://doi.org/10.3390/ijms21249503




