Deciphering Adipose Tissue Extracellular Vesicles Protein Cargo and Its Role in Obesity
Abstract
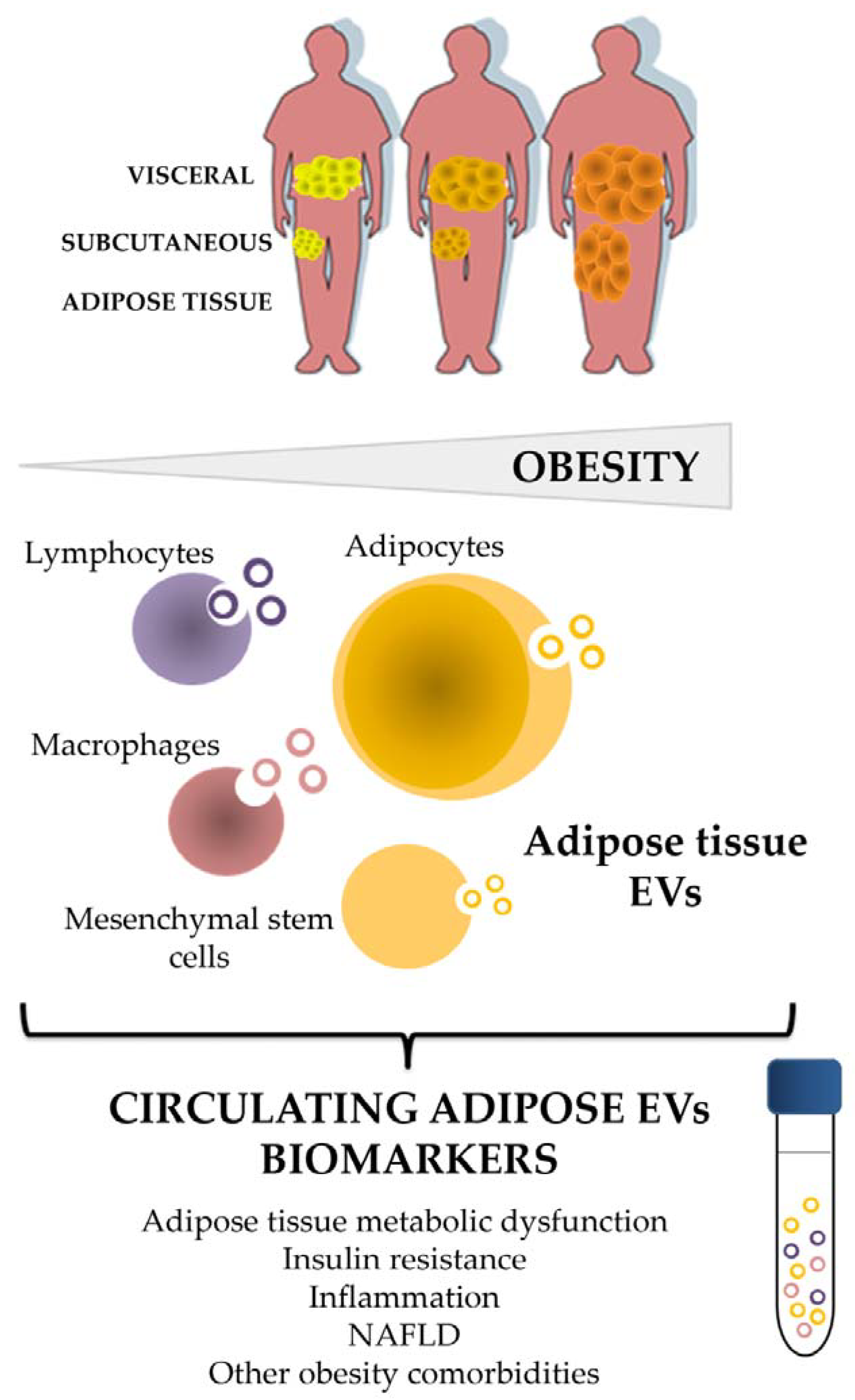
1. Introduction
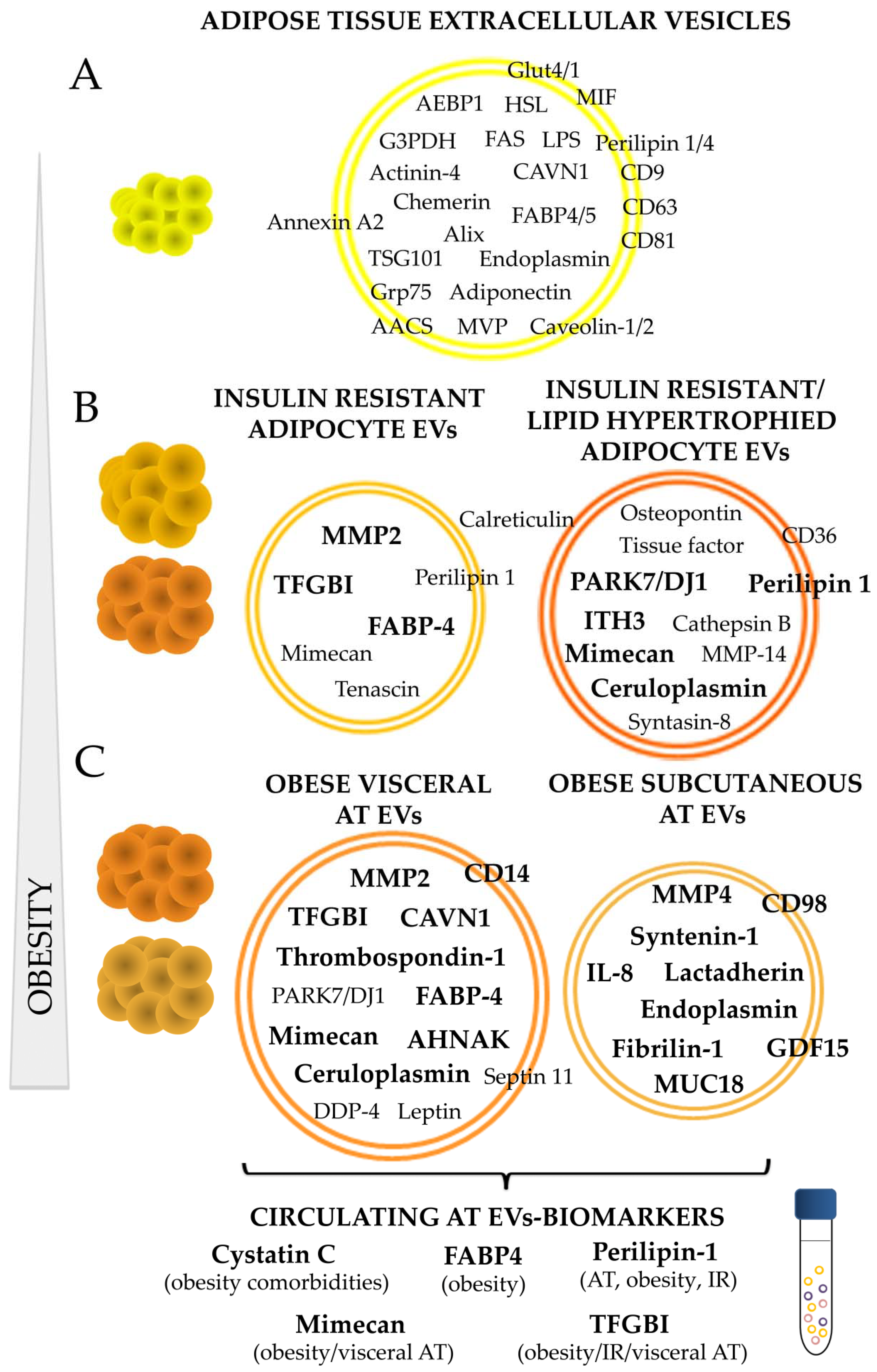
2. Protein Content Characterization of EVs Shed by Adipose Tissue
2.1. EVs Isolated from Individual Adipose Tissue Cell Components
2.1.1. Cultured Adipocytes
2.1.2. Cultured Adipose-Derived Mesenchymal Stem Cells (ADSCs)
2.2. EVs Secreted by Whole Adipose Tissue Explants
2.3. Circulating EVs in Obesity
3. Conclusions
4. Future Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| EVs | Extracellular Vesicles |
| NAFLD | Non-Alcoholic Fatty Liver Disease |
| AT | Adipose Tissue |
| IR | Insulin Resistance |
| AEBP1 | Adipocyte Enhancer-Binding Protein 1 |
| HSL | Hormone Sensitive-Lipase |
| FAS | Fatty Acid Synthase |
| MVP | Major Vault Protein |
| MIF | Macrophage Migration Inhibitory Factor |
| AACS | Acetoacetyl-CoA Synthetase |
| LPS | Lipoprotein Lipase |
| FABP | Fatty Acid Binding Protein |
| G3PDH | Glyceraldehyde 3-Phosphate Dehydrogenase |
| CAVN1 | Caveolae Associated Protein 1 |
| MMP | Matrix Metalloproteinase |
| ITH3 | Inter-α-Trypsin Inhibitor Heavy Chain H3 |
| GDF15 | Growth Differentiation Factor 15 |
| IL | Interleukin |
| MUC18 | Cell Surface Glycoprotein |
| AHNAK | Neuroblast Differentiation-Associated Protein |
| TGFBI | Transforming Growth Factor-Beta-Induced Protein ig-h3 |
| dUC | Differential Ultracentrifugation |
| SDG | Sucrose Density Grandient |
| UF | Ultrafiltration |
| TEIR | Total Exosome Isolation Reagent |
| LC-MS/MS | Liquid Chromatography Coupled to Mass Spectrometry |
| SWATH | Sequential Window Acquisition of all Theoretical Mass Spectra |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| MV | Microvesicles |
| EXO | Exosomes |
| CRP | C-Reactive Protein |
| ICAM-1 | Intercellular Adhesion Molecule 1 |
| DDP-4 | Dipeptidyl Peptidase-4 |
| GDM | Gestational Diabetes Mellitus |
| MSCs | Mesenchymal Stem Cells |
| VAT | Visceral Adipose Tissue |
| SAT | Subcutaneous Adipose Tissue |
References
- Li, M.-D. Leptin and beyond: An odyssey to the central control of body weight. Yale J. Biol. Med. 2011, 84, 1–7. [Google Scholar]
- Balistreri, C.R.; Caruso, C.; Candore, G. The Role of Adipose Tissue and Adipokines in Obesity-Related Inflammatory Diseases. Mediat. Inflamm. 2010, 2010, 1–19. [Google Scholar] [CrossRef]
- Tanaka, M.; Nozaki, M.; Fukuhara, A.; Segawa, K.; Aoki, N.; Matsuda, M.; Komuro, R.; Shimomura, I. Visfatin is released from 3T3-L1 adipocytes via a non-classical pathway. Biochem. Biophys. Res. Commun. 2007, 359, 194–201. [Google Scholar] [CrossRef]
- Connolly, K.D.; Wadey, R.M.; Mathew, D.; Johnson, E.; Rees, D.A.; James, P.E. Evidence for Adipocyte-Derived Extracellular Vesicles in the Human Circulation. Endocrinology 2018, 159, 3259–3267. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Kerr, A.; Jiao, H.; Hon, C.-C.; Rydén, M.; Dahlman, I.; Arner, P. Long Non-Coding RNAs Associated with Metabolic Traits in Human White Adipose Tissue. EBioMedicine 2018, 30, 248–260. [Google Scholar] [CrossRef] [PubMed]
- Lorente-Cebrián, S.; González-Muniesa, P.; Milagro, F.I.; Martínez, J.A. MicroRNAs and other non-coding RNAs in adipose tissue and obesity: Emerging roles as biomarkers and therapeutic targets. Clin. Sci. 2019, 133, 23–40. [Google Scholar] [CrossRef] [PubMed]
- Lo Cicero, A.; Stahl, P.D.; Raposo, G. Extracellular vesicles shuffling intercellular messages: For good or for bad. Curr. Opin. Cell Biol. 2015, 35, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Poon, I.K.H.; Lucas, C.D.; Rossi, A.G.; Ravichandran, K.S. Apoptotic cell clearance: Basic biology and therapeutic potential. Nat. Rev. Immunol. 2014, 14, 166–180. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Raposo, G.; Théry, C. Biogenesis, Secretion, and Intercellular Interactions of Exosomes and Other Extracellular Vesicles. Annu. Rev. Cell Dev. Biol. 2014, 30, 255–289. [Google Scholar] [CrossRef]
- Raposo, G.; Stoorvogel, W. Extracellular vesicles: Exosomes, microvesicles, and friends. J. Cell Biol. 2013, 200, 373–383. [Google Scholar] [CrossRef]
- Dehbashi Behbahani, G.; Khani, S.; Mahmoodzadeh Hosseini, H.; Abbaszadeh-Goudarzi, K.; Nazeri, S. The role of exosomes contents on genetic and epigenetic alterations of recipient cancer cells. Iran. J. Basic Med. Sci. 2016, 19, 1031–1039. [Google Scholar]
- O’Brien, K.; Breyne, K.; Ughetto, S.; Laurent, L.C.; Breakefield, X.O. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat. Rev. Mol. Cell Biol. 2020, 21, 585–606. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, N.; Whiteside, T.L.; Reichert, T.E. Challenges in exosome isolation and analysis in health and disease. Int. J. Mol. Sci. 2019, 20, 4684. [Google Scholar] [CrossRef] [PubMed]
- Brennan, K.; Martin, K.; FitzGerald, S.P.; O’Sullivan, J.; Wu, Y.; Blanco, A.; Richardson, C.; Mc Gee, M.M. A comparison of methods for the isolation and separation of extracellular vesicles from protein and lipid particles in human serum. Sci. Rep. 2020, 10, 1039. [Google Scholar] [CrossRef] [PubMed]
- Camino, T.; Lago-Baameiro, N.; Bravo, S.B.; Sueiro, A.; Couto, I.; Santos, F.; Baltar, J.; Casanueva, F.F.; Pardo, M. Vesicles Shed by Pathological Murine Adipocytes Spread Pathology: Characterization and Functional Role of Insulin Resistant/Hypertrophied Adiposomes. Int. J. Mol. Sci. 2020, 21, 2252. [Google Scholar] [CrossRef] [PubMed]
- Revenfeld, A.L.S.; Bæk, R.; Nielsen, M.H.; Stensballe, A.; Varming, K.; Jørgensen, M. Diagnostic and prognostic potential of extracellular vesicles in peripheral blood. Clin. Ther. 2014, 36, 830–846. [Google Scholar] [CrossRef]
- Royo, F.; Zuñiga-Garcia, P.; Sanchez-Mosquera, P.; Egia, A.; Perez, A.; Loizaga, A.; Arceo, R.; Lacasa, I.; Rabade, A.; Arrieta, E.; et al. Different EV enrichment methods suitable for clinical settings yield different subpopulations of urinary extracellular vesicles from human samples. J. Extracell. Vesicles 2016, 5, 29497. [Google Scholar] [CrossRef]
- Crewe, C.; Joffin, N.; Rutkowski, J.M.; Kim, M.; Zhang, F.; Towler, D.A.; Gordillo, R.; Scherer, P.E. An Endothelial-to-Adipocyte Extracellular Vesicle Axis Governed by Metabolic State. Cell 2018, 175, 695–708. [Google Scholar] [CrossRef]
- Samuelson, I.; Vidal-Puig, A.J. Fed-EXosome: Extracellular vesicles and cell–cell communication in metabolic regulation. Essays Biochem. 2018, 62, 165–175. [Google Scholar]
- Huang-Doran, I.; Zhang, C.-Y.; Vidal-Puig, A. Extracellular Vesicles: Novel Mediators of Cell Communication in Metabolic Disease. Trends Endocrinol. Metab. 2017, 28, 3–18. [Google Scholar] [CrossRef]
- Kranendonk, M.E.G.; Visseren, F.L.J.; van Herwaarden, J.A.; Nolte-’t Hoen, E.N.M.; de Jager, W.; Wauben, M.H.M.; Kalkhoven, E. Effect of extracellular vesicles of human adipose tissue on insulin signaling in liver and muscle cells. Obesity 2014, 22, 2216–2223. [Google Scholar] [CrossRef] [PubMed]
- Kranendonk, M.E.G.; Visseren, F.L.J.; van Balkom, B.W.M.; Nolte-’t Hoen, E.N.M.; van Herwaarden, J.A.; de Jager, W.; Schipper, H.S.; Brenkman, A.B.; Verhaar, M.C.; Wauben, M.H.M.; et al. Human adipocyte extracellular vesicles in reciprocal signaling between adipocytes and macrophages. Obesity 2014, 22, 1296–1308. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.-B.; Poliakov, A.; Hardy, R.W.; Clements, R.; Liu, C.; Liu, Y.; Wang, J.; Xiang, X.; Zhang, S.; Zhuang, X.; et al. Adipose Tissue Exosome-Like Vesicles Mediate Activation of Macrophage-Induced Insulin Resistance. Diabetes 2009, 58, 2498–2505. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Hui, X.; Chong Hoo, R.L.; Ye, D.; Cheung Chan, C.Y.; Feng, T.; Wang, Y.; Ling Lam, K.S.; Xu, A. Adipocyte-secreted exosomal microRNA-34a inhibits M2 macrophage polarization to promote obesity-induced adipose inflammation. J. Clin. Investig. 2019, 129, 834–849. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Han, L.; Chen, F.F.; Wang, D.; Wang, F.; Zhang, L.; Wang, Z.H.; Zhong, M.; Tang, M.X.; Zhang, W. Adipocyte-derived exosomes carrying sonic hedgehog mediate M1 macrophage polarization-induced insulin resistance via Ptch and PI3K pathways. Cell. Physiol. Biochem. 2018, 48, 1416–1432. [Google Scholar] [CrossRef]
- Koeck, E.S.; Iordanskaia, T.; Sevilla, S.; Ferrante, S.C.; Hubal, M.J.; Freishtat, R.J.; Nadler, E.P. Adipocyte exosomes induce transforming growth factor beta pathway dysregulation in hepatocytes: A novel paradigm for obesity-related liver disease. J. Surg. Res. 2014, 192, 268–275. [Google Scholar] [CrossRef]
- Eguchi, A.; Mulya, A.; Lazic, M.; Radhakrishnan, D.; Berk, M.P.; Povero, D.; Gornicka, A.; Feldstein, A.E. Microparticles Release by Adipocytes Act as “Find-Me” Signals to Promote Macrophage Migration. PLoS ONE 2015, 10, e0123110. [Google Scholar] [CrossRef]
- Gao, J.; Li, X.; Wang, Y.; Cao, Y.; Yao, D.; Sun, L.; Qin, L.; Qiu, H.; Zhan, X. Adipocyte-derived extracellular vesicles modulate appetite and weight through mTOR signalling in the hypothalamus. Acta Physiol. 2019, 228, e13339. [Google Scholar] [CrossRef]
- Zhang, H.; Deng, T.; Ge, S.; Liu, Y.; Bai, M.; Zhu, K.; Fan, Q.; Li, J.; Ning, T.; Tian, F.; et al. Exosome circRNA secreted from adipocytes promotes the growth of hepatocellular carcinoma by targeting deubiquitination-related USP7. Oncogene 2019, 38, 2844–2859. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Garcovich, S. Concise Review: Adipose-Derived Stem Cells (ASCs) and Adipocyte-Secreted Exosomal microRNA (A-SE-miR) Modulate Cancer Growth and proMote Wound Repair. J. Clin. Med. 2019, 8, 855. [Google Scholar] [CrossRef] [PubMed]
- Lazar, I.; Clement, E.; Dauvillier, S.; Milhas, D.; Ducoux-Petit, M.; LeGonidec, S.; Moro, C.; Soldan, V.; Dalle, S.; Balor, S.; et al. Adipocyte Exosomes Promote Melanoma Aggressiveness through Fatty Acid Oxidation: A Novel Mechanism Linking Obesity and Cancer. Cancer Res. 2016, 76, 4051–4057. [Google Scholar] [CrossRef] [PubMed]
- Quan, M.; Kuang, S. Exosomal Secretion of Adipose Tissue during Various Physiological States. Pharm. Res. 2020, 37, 221. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Buyel, J.J.; Hanssen, M.J.W.; Siegel, F.; Pan, R.; Naumann, J.; Schell, M.; Van Der Lans, A.; Schlein, C.; Froehlich, H.; et al. Exosomal microRNA miR-92a concentration in serum reflects human brown fat activity. Nat. Commun. 2016, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Booth, A.; Magnuson, A.; Foster, M. Detrimental and protective fat: Body fat distribution and its relation to metabolic disease. Horm. Mol. Biol. Clin. Investig. 2014, 17, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Ojeda, F.J.; Méndez-Gutiérrez, A.; Aguilera, C.M.; Plaza-Díaz, J. Extracellular Matrix Remodeling of Adipose Tissue in Obesity and Metabolic Diseases. Int. J. Mol. Sci. 2019, 20, 4888. [Google Scholar] [CrossRef] [PubMed]
- Thomou, T.; Mori, M.A.; Dreyfuss, J.M.; Konishi, M.; Sakaguchi, M.; Wolfrum, C.; Rao, T.N.; Winnay, J.N.; Garcia-Martin, R.; Grinspoon, S.K.; et al. Adipose-derived circulating miRNAs regulate gene expression in other tissues. Nature 2017, 542, 450–455. [Google Scholar] [CrossRef]
- Greenhill, C. Exosomal microRNAs as novel adipokines. Nat. Rev. Genet. 2017, 18, 212. [Google Scholar] [CrossRef]
- Yang, Z.; Wei, Z.; Wu, X.; Yang, H. Screening of exosomal miRNAs derived from subcutaneous and visceral adipose tissues: Determination of targets for the treatment of obesity and associated metabolic disorders. Mol. Med. Rep. 2018, 18, 3314–3324. [Google Scholar] [CrossRef]
- Meng, Y.; Eirin, A.; Zhu, X.-Y.; O’Brien, D.R.; Lerman, A.; van Wijnen, A.J.; Lerman, L.O. The metabolic syndrome modifies the mRNA expression profile of extracellular vesicles derived from porcine mesenchymal stem cells. Diabetol. Metab. Syndr. 2018, 10, 58. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, O.-K. Potential Roles of Adipocyte Extracellular Vesicle-Derived miRNAs in Obesity-Mediated Insulin Resistance. Adv. Nutr. 2020, nmaa105. [Google Scholar] [CrossRef]
- Santamaria-Martos, F.; Benitez, I.D.; Latorre, J.; Lluch, A.; Moreno-Navarrete, J.M.; Sabater, M.; Ricart, W.; Sanchez de la Torre, M.; Mora, S.; Fernández-Real, J.M.; et al. Comparative and functional analysis of plasma membrane-derived extracellular vesicles from obese vs. nonobese women. Clin. Nutr. 2020, 39, 1067–1076. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, S.; De Filippo, E.; Göddeke, S.; Knebel, B.; Kotzka, J.; Al-Hasani, H.; Roden, M.; Lehr, S.; Sell, H. Exosomal proteins constitute an essential part of the human adipose tissue secretome. Biochim. Biophys. Acta Proteins Proteom. 2018, 1867, 140172. [Google Scholar] [CrossRef] [PubMed]
- Durcin, M.; Fleury, A.; Taillebois, E.; Hilairet, G.; Krupova, Z.; Henry, C.; Truchet, S.; Trötzmüller, M.; Köfeler, H.; Mabilleau, G.; et al. Characterisation of adipocyte-derived extracellular vesicle subtypes identifies distinct protein and lipid signatures for large and small extracellular vesicles. J. Extracell. Vesicles 2017, 6, 1305677. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-E.; Moon, P.-G.; Lee, I.-K.; Baek, M.-C. Proteomic Analysis of Extracellular Vesicles Released by Adipocytes of Otsuka Long-Evans Tokushima Fatty (OLETF) Rats. Protein J. 2015, 34, 220–235. [Google Scholar] [CrossRef]
- Eirin, A.; Zhu, X.-Y.; Puranik, A.S.; Woollard, J.R.; Tang, H.; Dasari, S.; Lerman, A.; van Wijnen, A.J.; Lerman, L.O. Comparative proteomic analysis of extracellular vesicles isolated from porcine adipose tissue-derived mesenchymal stem/stromal cells. Sci. Rep. 2016, 6, 36120. [Google Scholar] [CrossRef]
- Xing, X.; Han, S.; Cheng, G.; Ni, Y.; Li, Z.; Li, Z. Proteomic Analysis of Exosomes from Adipose-Derived Mesenchymal Stem Cells: A Novel Therapeutic Strategy for Tissue Injury. Biomed. Res. Int. 2020, 2020, 6094562. [Google Scholar] [CrossRef]
- Jayabalan, N.; Lai, A.; Ormazabal, V.; Adam, S.; Guanzon, D.; Palma, C.; Scholz-Romero, K.; Lim, R.; Jansson, T.; McIntyre, H.D.; et al. Adipose Tissue Exosomal Proteomic Profile Reveals a Role on Placenta Glucose Metabolism in Gestational Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2019, 104, 1735–1752. [Google Scholar] [CrossRef]
- Camino, T.; Lago-Baameiro, N.; Bravo, S.B.; Molares-Vila, A.; Sueiro, A.; Couto, I.; Baltar, J.; Casanueva, F.F.; Pardo, M. Human obese white adipose tissue sheds depot-specific extracellular vesicles and reveals candidate obesity biomarkers. Rev. Transl. Res. 2020. under review. [Google Scholar]
- Zhang, Y.; Yu, M.; Dong, J.; Wu, Y.; Tian, W. Identification of Novel Adipokines through Proteomic Profiling of Small Extracellular Vesicles Derived from Adipose Tissue. J. Proteome Res. 2020, 19, 3130–3142. [Google Scholar] [CrossRef]
- Kranendonk, M.E.; de Kleijn, D.P.; Kalkhoven, E.; Kanhai, D.A.; Uiterwaal, C.S.; van der Graaf, Y.; Pasterkamp, G.; Visseren, F.L. Extracellular vesicle markers in relation to obesity and metabolic complications in patients with manifest cardiovascular disease. Cardiovasc. Diabetol. 2014, 13, 37. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Eguchi, A.; Tempaku, M.; Honda, T.; Togashi, K.; Iwasa, M.; Hasegawa, H.; Takei, Y.; Sumida, Y.; Taguchi, O. Circulating extracellular vesicles are associated with lipid and insulin metabolism. Am. J. Physiol. Metab. 2018, 315, E574–E582. [Google Scholar] [CrossRef] [PubMed]
- Amosse, J.; Durcin, M.; Malloci, M.; Vergori, L.; Fleury, A.; Gagnadoux, F.; Dubois, S.; Simard, G.; Boursier, J.; Hue, O.; et al. Phenotyping of circulating extracellular vesicles (EVs) in obesity identifies large EVs as functional conveyors of Macrophage Migration Inhibitory Factor. Mol. Metab. 2018, 18, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Witczak, J.K.; Min, T.; Prior, S.L.; Stephens, J.W.; James, P.E.; Rees, A. Bariatric Surgery Is Accompanied by Changes in Extracellular Vesicle-Associated and Plasma Fatty Acid Binding Protein 4. Obes. Surg. 2018, 28, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Phoonsawat, W.; Aoki-Yoshida, A.; Tsuruta, T.; Sonoyama, K. Adiponectin is partially associated with exosomes in mouse serum. Biochem. Biophys. Res. Commun. 2014, 448, 261–266. [Google Scholar] [CrossRef]
- Gao, X.; Salomon, C.; Freeman, D.J. Extracellular Vesicles from Adipose Tissue-A Potential Role in Obesity and Type 2 Diabetes? Front. Endocrinol. 2017, 8, 202. [Google Scholar] [CrossRef] [PubMed]
- Eguchi, A.; Lazic, M.; Armando, A.M.; Phillips, S.A.; Katebian, R.; Maraka, S.; Quehenberger, O.; Sears, D.D.; Feldstein, A.E. Circulating adipocyte-derived extracellular vesicles are novel markers of metabolic stress. J. Mol. Med. 2016, 94, 1241–1253. [Google Scholar] [CrossRef]
- Aryani, A.; Denecke, B. Exosomes as a Nanodelivery System: A Key to the Future of Neuromedicine? Mol. Neurobiol. 2016, 53, 818–834. [Google Scholar] [CrossRef]
- Akbar, N.; Azzimato, V.; Choudhury, R.P.; Aouadi, M. Extracellular vesicles in metabolic disease. Diabetologia 2019, 62, 2179–2187. [Google Scholar] [CrossRef]
- Li, C.-J.; Fang, Q.-H.; Liu, M.-L.; Lin, J.-N. Current understanding of the role of Adipose-derived Extracellular Vesicles in Metabolic Homeostasis and Diseases: Communication from the distance between cells/tissues. Theranostics 2020, 10, 7422–7435. [Google Scholar] [CrossRef]
- Kita, S.; Maeda, N.; Shimomura, I. Interorgan communication by exosomes, adipose tissue, and adiponectin in metabolic syndrome. J. Clin. Investig. 2019, 129, 4041–4049. [Google Scholar] [CrossRef]
- Sáez, T.; Toledo, F.; Sobrevia, L. Impaired signalling pathways mediated by extracellular vesicles in diabesity. Mol. Aspects Med. 2019, 66, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Hubal, M.J.; Nadler, E.P.; Ferrante, S.C.; Barberio, M.D.; Suh, J.-H.; Wang, J.; Dohm, G.L.; Pories, W.J.; Mietus-Snyder, M.; Freishtat, R.J. Circulating adipocyte-derived exosomal MicroRNAs associated with decreased insulin resistance after gastric bypass. Obesity 2017, 25, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, M.; Dai, M.; Chen, C.; Tang, Q.; Jing, W.; Wang, H.; Tian, W. miR-450a-5p within rat adipose tissue exosome-like vesicles promotes adipogenic differentiation by targeting WISP2. J. Cell Sci. 2017, 130, 1158–1168. [Google Scholar] [CrossRef] [PubMed]
| Ref | Isolation Method | Type of Protein Analysis | Cell/Tissue Type/Circulating | Species | EVs Markers |
|---|---|---|---|---|---|
| Cultured adipocytes | |||||
| Kranendonk et al., 2014 [22] | dUC | Immunoblot + multiplex immunoassay | Simpson Golabi Behmel Syndrome (SGBS) adipocytes cell line | Human | FABP4, adiponectin, TNF-α, MCSF, RBP4, MIF |
| Hartwing et al., 2019 [42] | UF + dUC | NanoLC-MS/MS | Human primary adipocytes from lean to moderate overweight woman | Human | Adiponectin, FABP4, metalloproteases |
| Lazar et al., 2016 [31] | SDG | NanoLC-MS/MS | 3T3-F442A adipocyte cell line | Mouse | ECHA, HCDH (FAO INVOLVED) |
| Durcin et al., 2017 [43] | dUC | NanoLC-MS/MS | 3T3-L1 adipocyte cell line | Mouse | L-Evs: FABP4, 14-3-3, Annexin A2, endoplasmin, actinin-4 sEVs: MVP, FAS, adiponectin |
| Camino et al., 2020 [15] | dUC | Nano-LC/MS-MS + SWATH | C3H10T1/2 adipocyte cell line: models of IR and lipid hypertrophy | Mouse | Ceruloplasmin, mimecan, perilipin 1, TFGBI |
| Lee et al., 2015 [44] | dUC | NanoLC-MS/MS+ label-free | Obese diabetic and obese nondiabetic adipocytes of Otsuka Long-Evans Tokusima Fatty (OLETF) primary cell culture | Rat | Caveolin, LPL, AQ7 |
| Cultured adipose-derived mesenchymal stem cells | |||||
| Eirin et al., 2016 [45] | dUC | LC-MS/MS | MSCs from abdominal fat | Porcine | C2, VEGF, vWF (vonWillebrand factor) |
| Xing et al., 2020 [46] | dUC | LC-MS/MS | Adipose-derived mesenchymal stem cells (ADSCs) | Mouse | Tissue repair |
| Adipose tissue explants | |||||
| Kranendonk et al., 2014 [22] | dUC | Adipokine profile array | Subjects undergoing surgery for aneurysmatic aortic disease (obese/overweight) | Human | MCP-1, IL-6, and MIF in omental AT vs. subc |
| Jayabalan et al., 2019 [47] | dUC | LC-MS/MS(SWATH) | Omental adipose tissue from woman with GDM after baby delivery | Human | Proteins related to glucose metabolism |
| Camino et al., 2020 [48] | dUC | NanoLC-MS/MS (SWATH) | Visceral and Subcutaneous whole AT explants from obese | Human | VAT: TGFBI, CAVN1, CD14, mimecan, thrombospondin-1, FABP-4,AHNAK/↓Syntenin 1 |
| Zhang Y et al., 2020 [49] | TEIR | LC-MS/MS | Inguinal fat pads obese Sprague-Dawley rats | Rat | NPM3, DAD1and STEAP3 |
| Circulating EVs | |||||
| Kranendonk et al., 2014 [50] | ExoQuick | Multiplex immunoassay | Circulating:plasma patients with vascular disease | Human | Cystatin C positive related to obesity metabolic complications/CD14 negative related |
| Kobayashi et al., 2018 [51] | qEV column | Immunoblot | Circulating: plasma patients with metabolic disease | Human | Perilipin 1 elevated with metab risk factors |
| Amosse et al., 2018 [52] | dUC | Protein arrays/multiplex | Circulating MVs and EXO plasma from metabolic syndrome patients | Human | Adiponectin, adipsin, cathepsin D, CRP, ICAM-1 chemerin, DDP-4 among others. |
| Witczak et al., 2018 [53] | dUC | Immunoassay | Circulating plasma obese before and after bariatric surgery | Human | FABP4 |
| Phoonsawat et al., 2014 [54] | dUC | Immunoblot/ELISA | Circulating: serum obese mice | Mouse | Adiponectin, resistin |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Camino, T.; Lago-Baameiro, N.; Martis-Sueiro, A.; Couto, I.; Santos, F.; Baltar, J.; Pardo, M. Deciphering Adipose Tissue Extracellular Vesicles Protein Cargo and Its Role in Obesity. Int. J. Mol. Sci. 2020, 21, 9366. https://doi.org/10.3390/ijms21249366
Camino T, Lago-Baameiro N, Martis-Sueiro A, Couto I, Santos F, Baltar J, Pardo M. Deciphering Adipose Tissue Extracellular Vesicles Protein Cargo and Its Role in Obesity. International Journal of Molecular Sciences. 2020; 21(24):9366. https://doi.org/10.3390/ijms21249366
Chicago/Turabian StyleCamino, Tamara, Nerea Lago-Baameiro, Aurelio Martis-Sueiro, Iván Couto, Francisco Santos, Javier Baltar, and María Pardo. 2020. "Deciphering Adipose Tissue Extracellular Vesicles Protein Cargo and Its Role in Obesity" International Journal of Molecular Sciences 21, no. 24: 9366. https://doi.org/10.3390/ijms21249366
APA StyleCamino, T., Lago-Baameiro, N., Martis-Sueiro, A., Couto, I., Santos, F., Baltar, J., & Pardo, M. (2020). Deciphering Adipose Tissue Extracellular Vesicles Protein Cargo and Its Role in Obesity. International Journal of Molecular Sciences, 21(24), 9366. https://doi.org/10.3390/ijms21249366







