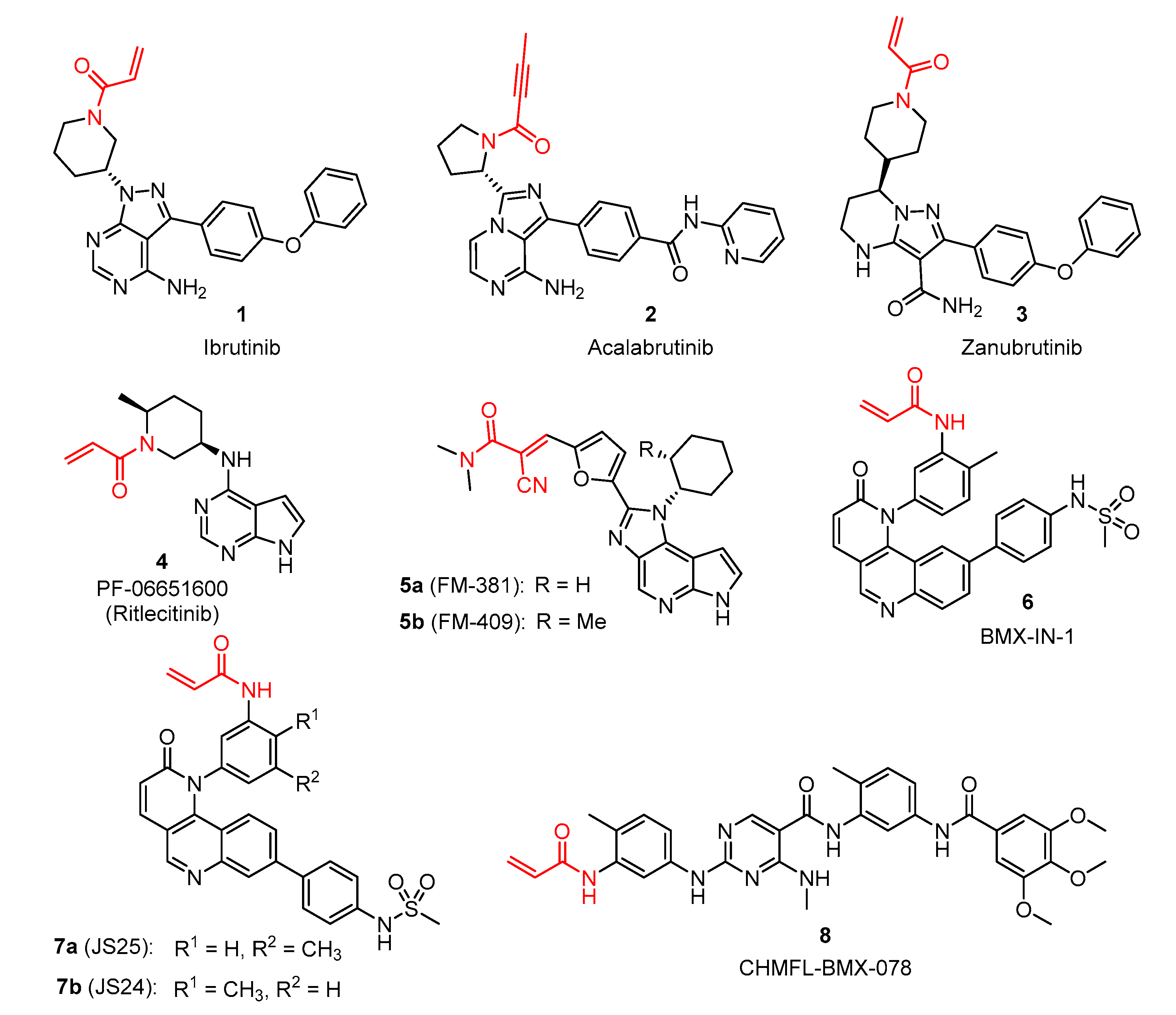
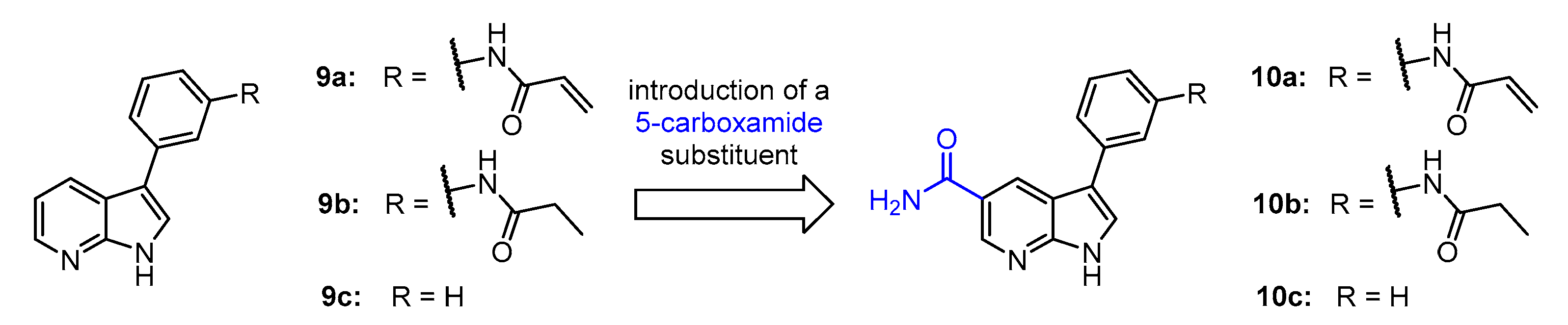
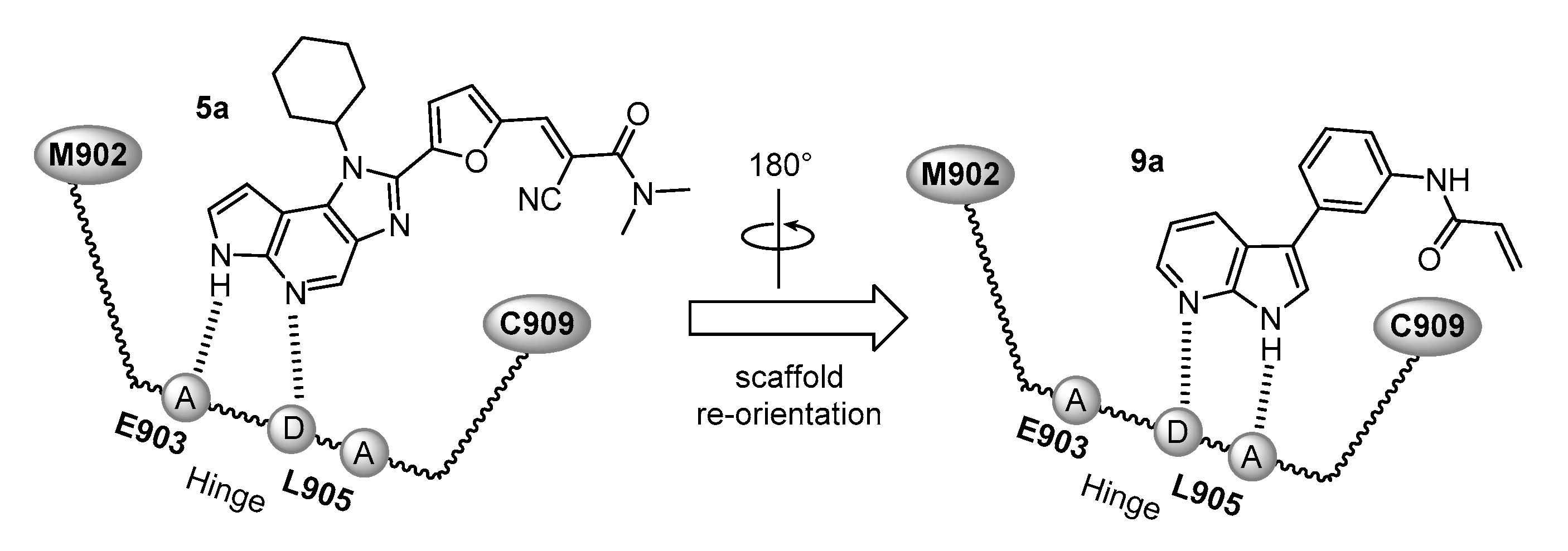
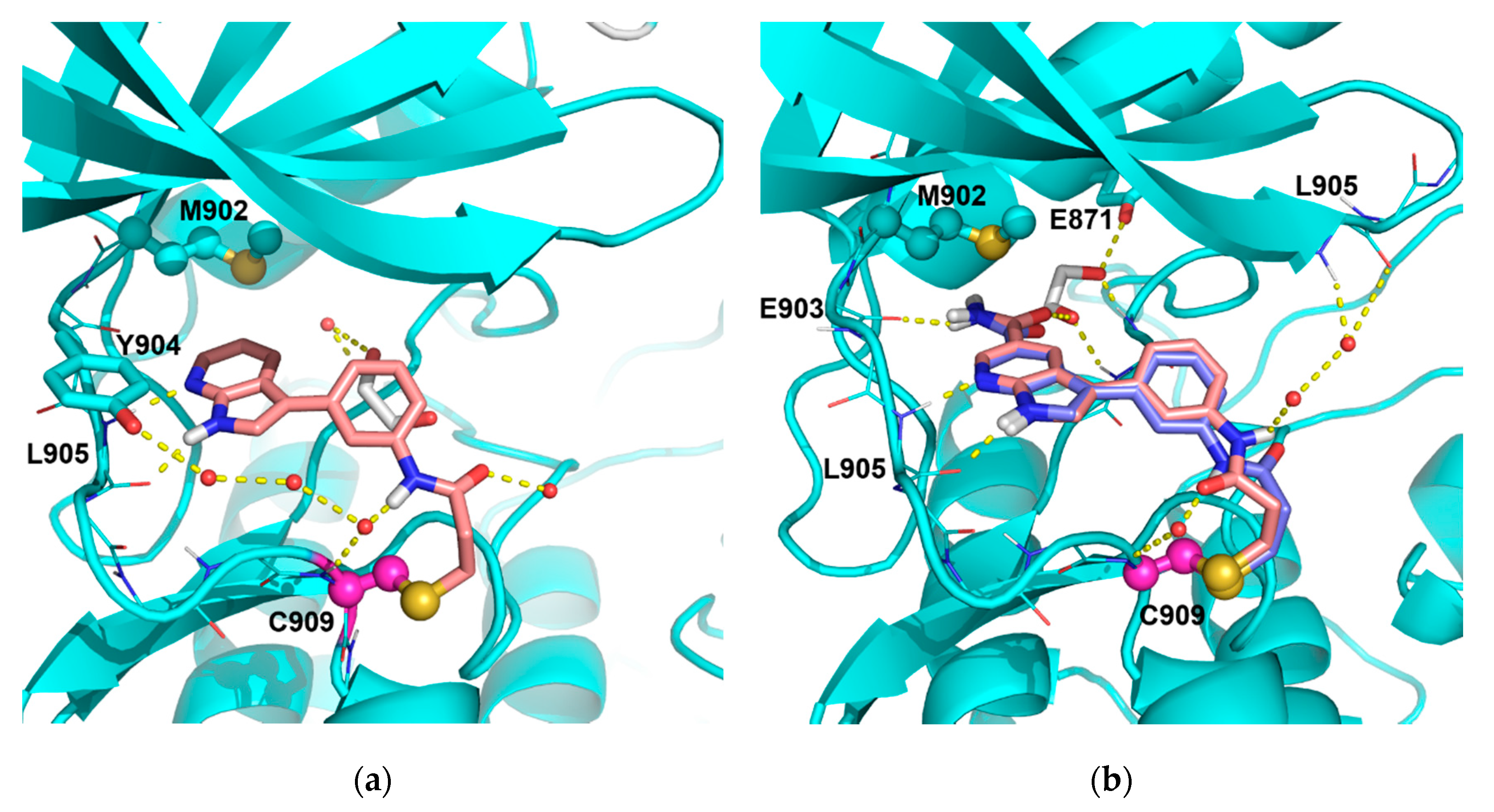
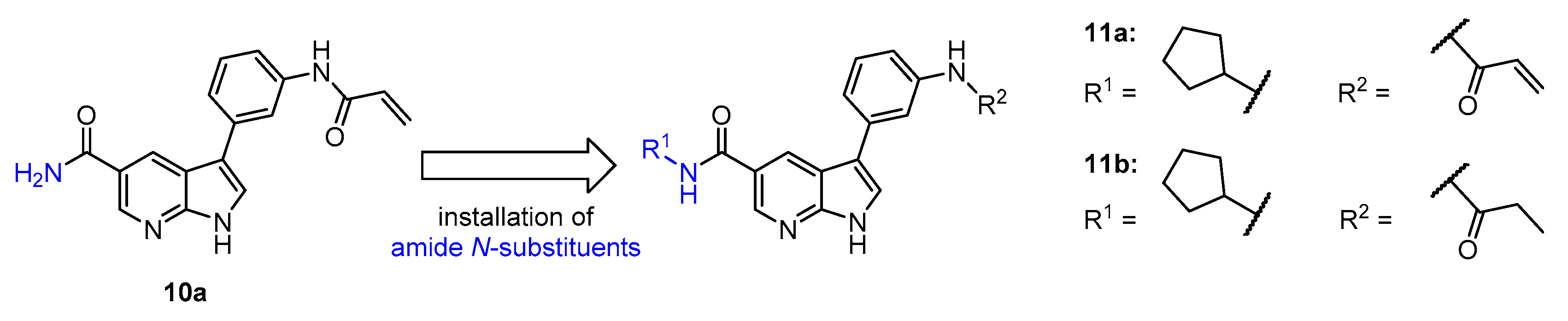
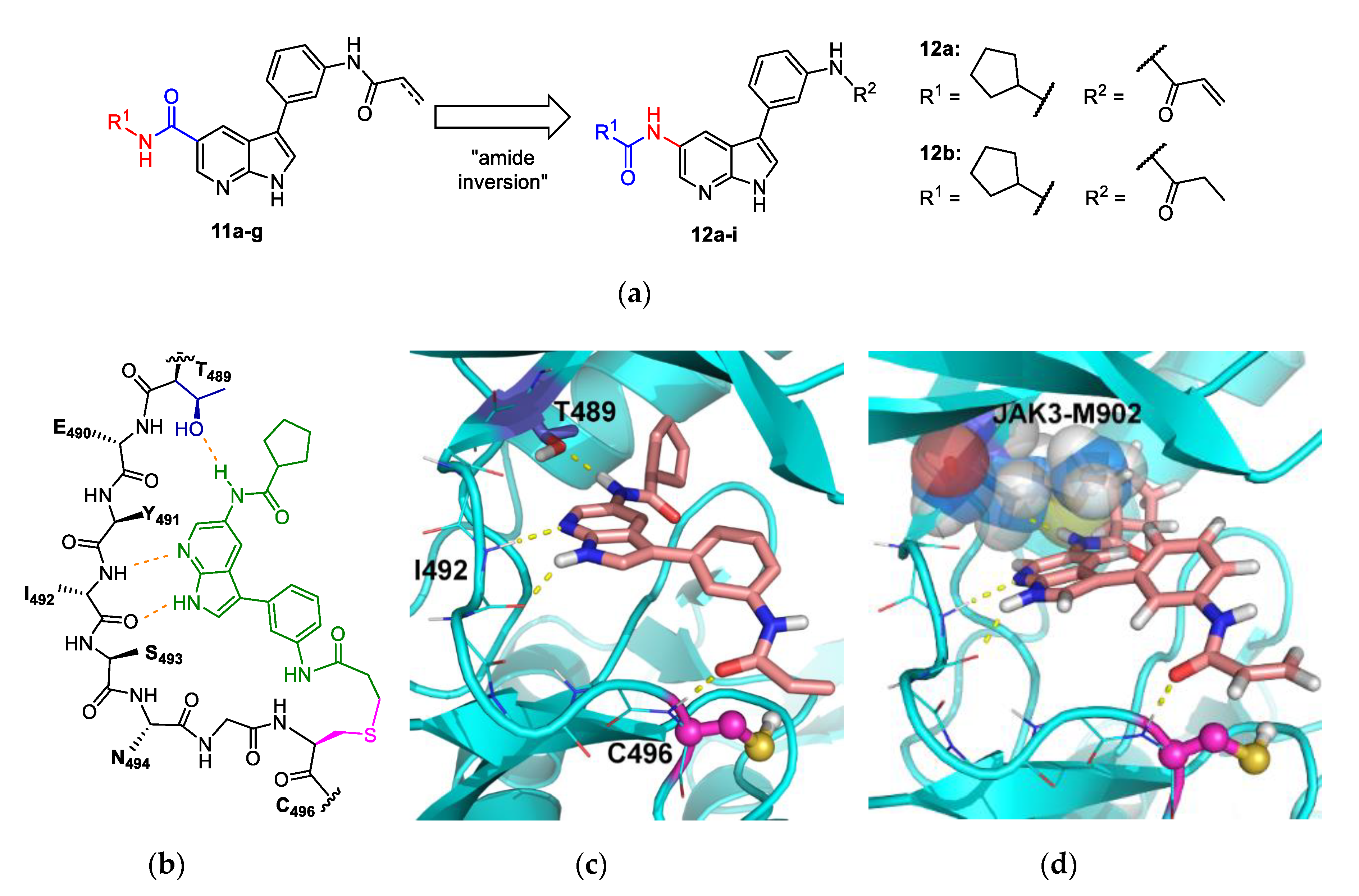
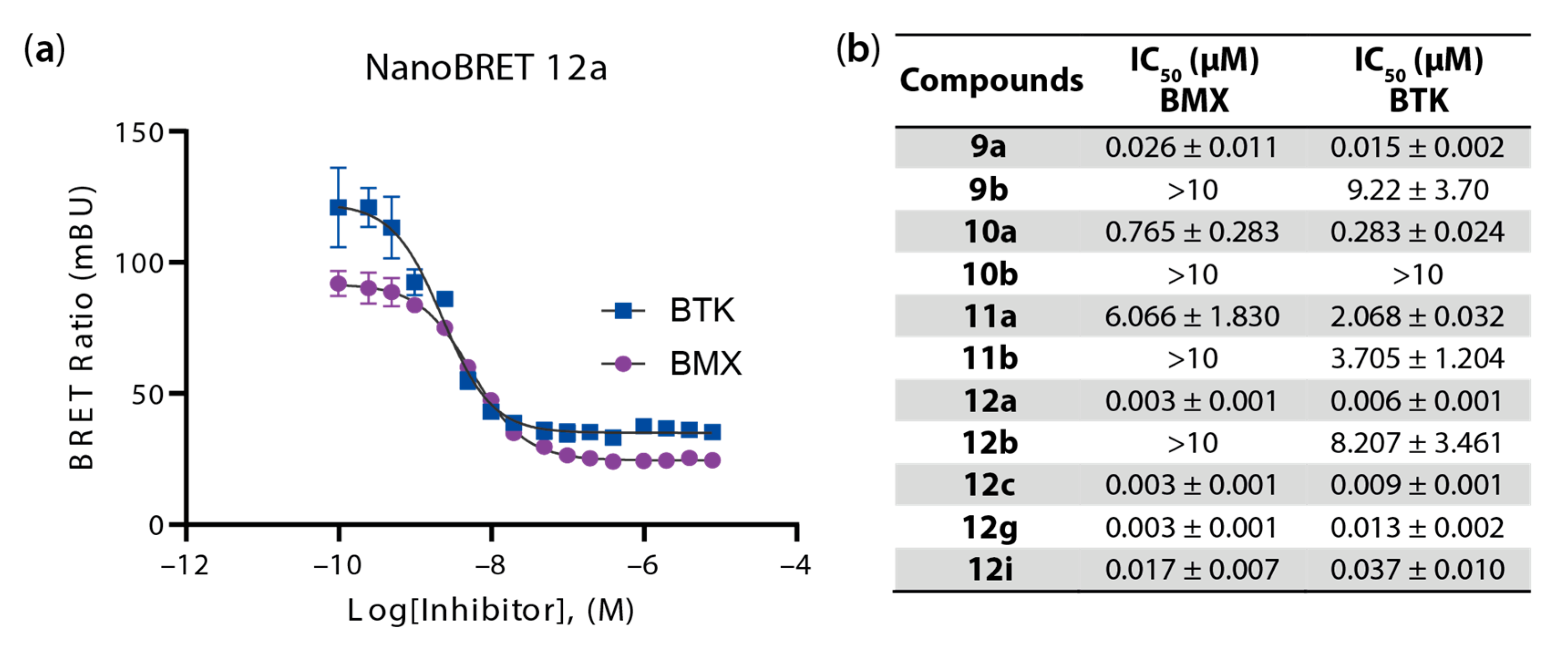
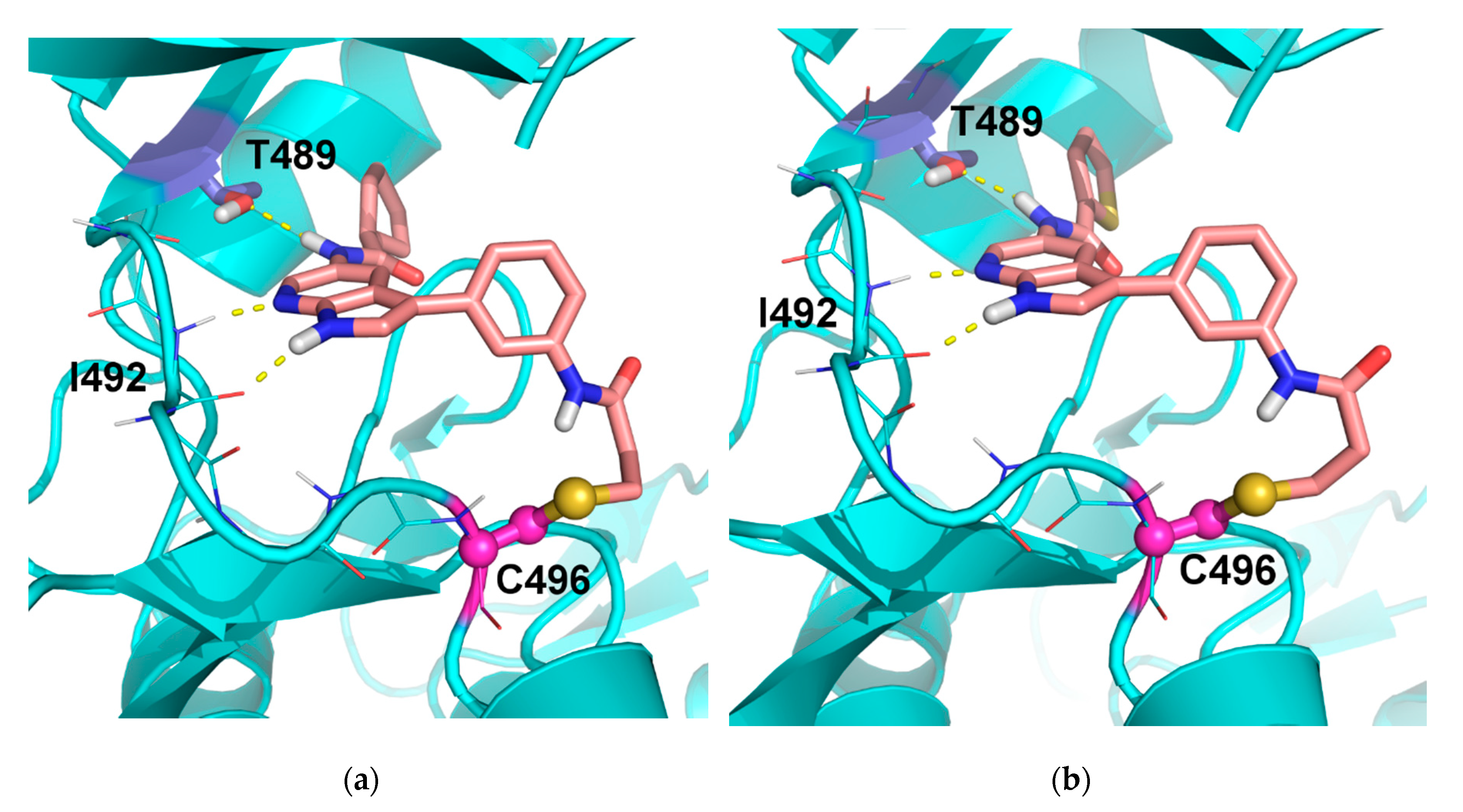
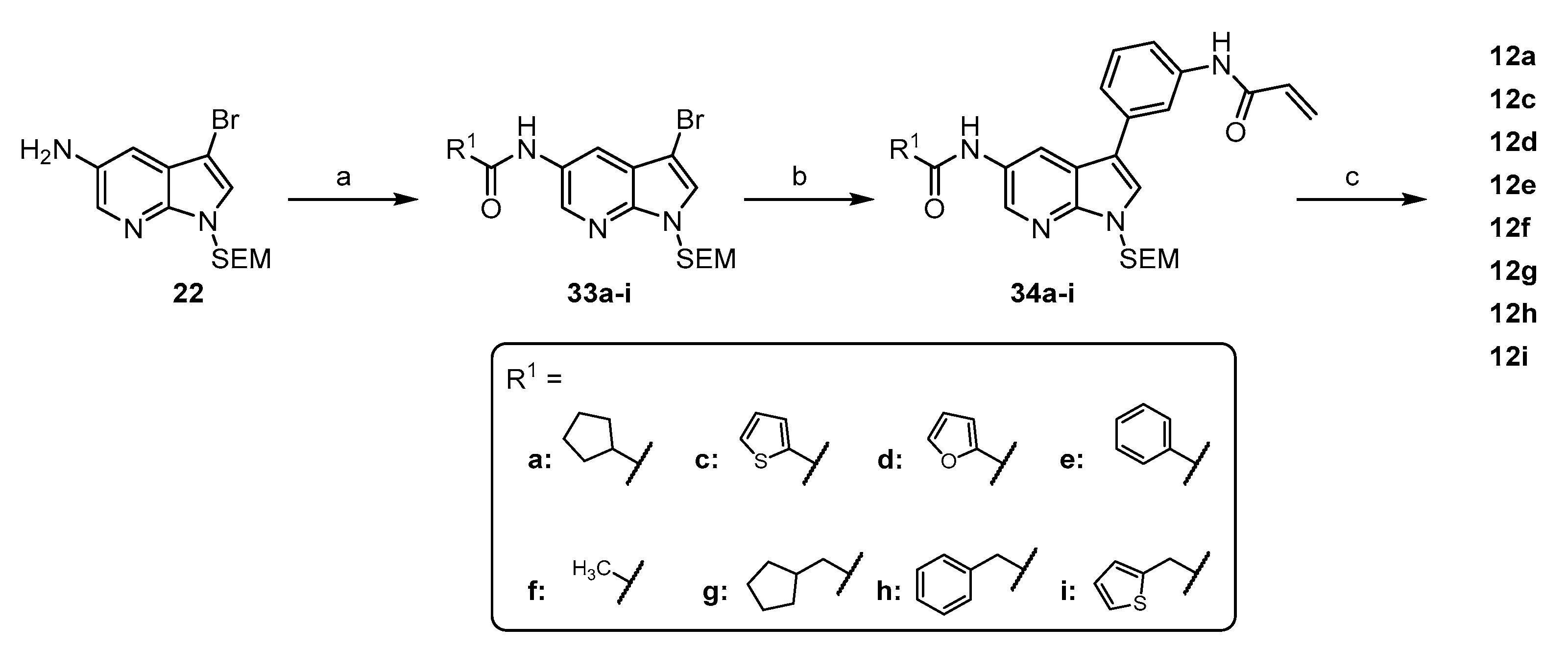
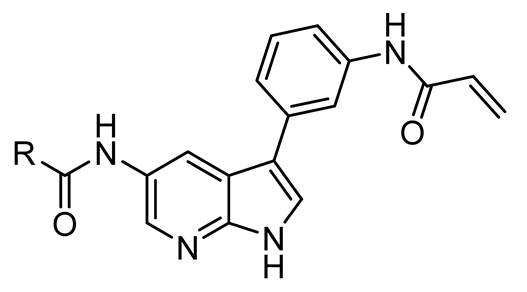
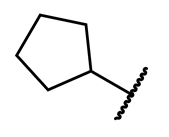
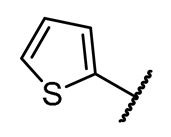
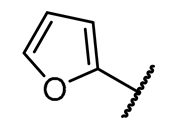
4.1.3. Synthesis and Characterization of Intermediates and Final Compounds
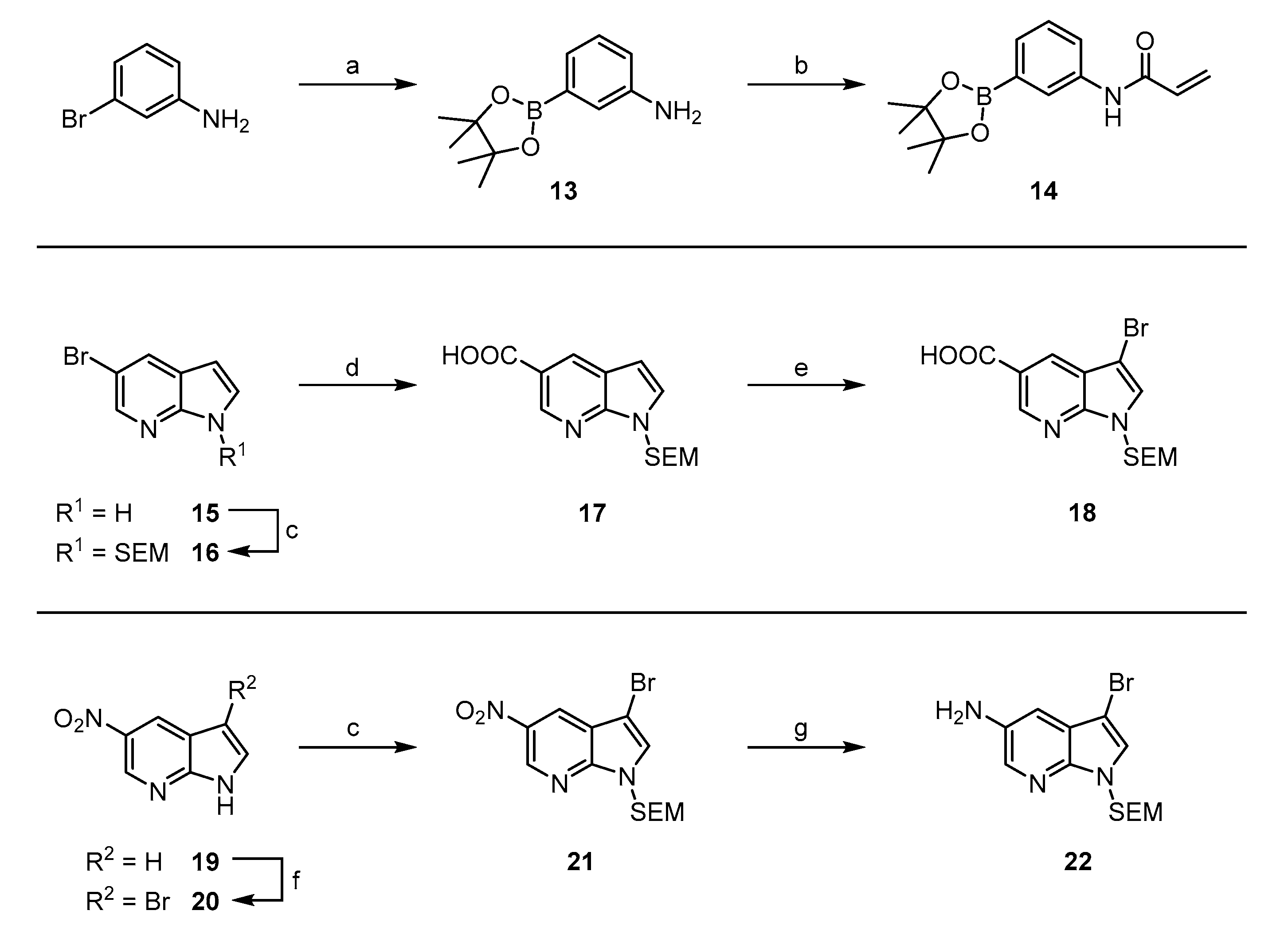
3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline (13): In a flame-dried Schlenk flask were suspended 1.47 g dry KOAc (15.0 mmol), 860 mg 3-bromoaniline (5.00 mmol) and 1.27 g bis(pinacolato)diboron (5.00 mmol) in 20 mL dry dioxane. The mixture was carefully degassed via three vacuum/argon cycles and 9 mg XPhos Pd G4 (10 µmol) was added subsequently. The degassing procedure was repeated and then the reaction was heated to 90 °C oil-bath temperature overnight. After cooling to ambient temperature, the mixture was diluted with EtOAc and filtered over a bed of celite. The filtrate was washed with water (three times) and brine, prior to drying over Na2SO4 and evaporation. The residue was triturated with heptane and the precipitate isolated by filtration to yield 914 mg (84%) of the product as a slightly brownish solid. 1H NMR (200 MHz, DMSO) δ 7.06–6.97 (m, 1H), 6.95 (d, J = 1.9 Hz, 1H), 6.82 (dt, J = 7.2, 1.1 Hz, 1H), 6.66 (ddd, J = 7.9, 2.4, 1.1 Hz, 1H), 5.02 (br s, 2H), 1.26 (s, 12H) 13C NMR (50 MHz, DMSO) δ 148.0, 128.3, 128.3, 121.9, 120.1, 116.8, 83.2, 24.7 TLC-MS (ESI) m/z: 219.9 [M + H]+ HPLC tret = 6.97 min.
N-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)acrylamide (14): To a solution of 891 mg 13 (4.07 mmol) and 715 µL Et3N (5.09 mmol) in 16 mL dry DCM were dropwise added 368 µL acryloyl chloride (4.48 mmol) at −10 °C. After 1 h, the reaction was quenched by addition of sat. NH4Cl and transferred to a separatory funnel. The phases were separated and the organic phase was washed with sat NaHCO3 and brine prior to drying over Na2SO4 and evaporation. The crude product was purified via flash chromatography (petrol ether/EtOAc + 5% MeOH (10–40%)) to obtain 780 mg (85%) of the title compound as a white solid. 1H NMR (200 MHz, DMSO) δ 10.14 (br s, 1H), 7.98 (d, J = 1.4 Hz, 1H), 7.85 (dt, J = 6.3, 2.4 Hz, 1H), 7.41–7.26 (m, 2H), 6.43 (dd, J = 17.0, 9.7 Hz, 1H), 6.25 (dd, J = 17.0, 2.4 Hz, 1H), 5.75 (dd, J = 9.7, 2.4 Hz, 1H), 1.29 (s, 12H) 13C NMR (50 MHz, CDCl3) δ 163.1, 138.6, 131.8, 129.3, 128.4, 126.8, 125.3, 122.2, 83.7, 24.7 TLC-MS (ESI) m/z: 296.4 [M + H]+ HPLC tret = 7.45 min.
5-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine (16): To an ice-cooled solution of 1.96 g 5-bromo-1H-pyrrolo[2,3-b]pyridine (10 mmol) in 10 mL dry DMF were added 518 mg sodium hydride (60%wt dispersion in min. oil, 12.9 mmol) and stirring was continued for about 30 min. Subsequently were added 1.95 mL SEM-Cl (10.9 mmol) and stirring was continued until TLC indicated complete conversion. The reaction was diluted with 120 mL EtOAc and transferred to a separatory funnel. The organic phase was successively washed with water (four times) and brine, prior to drying over Na2SO4 and evaporation. The residue was purified via flash chromatography (petrol ether/EtOAc (0–15%)) to obtain 2.55 g (78%) as a colorless oil. 1H NMR (200 MHz, CDCl3) δ 8.34 (d, J = 1.9 Hz, 1H), 8.02 (d, J = 1.9 Hz, 1H), 7.35 (d, J = 3.6 Hz, 1H), 6.46 (d, J = 3.6 Hz, 1H), 5.64 (s, 2H), 3.52 (t, J = 8.2 Hz, 2H), 0.89 (t, J = 8.2 Hz, 2H), −0.07 (s, 9H) 13C NMR (50 MHz, CDCl3) δ 146.6, 143.8, 131.1, 129.5, 122.3, 112.3, 100.7, 73.2, 66.5, 17.9, −1.3 TLC-MS (ESI) m/z: 349.3 [M + Na]+ HPLC tret = 10.84 min.
1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxylic acid (17): Under argon atmosphere were dissolved 2.49 g 16 (7.6 mmol) in 80 mL dry THF in a flame-dried Schlenk tube. At –78 °C, 3.3 mL of a 2.5 M n-BuLi solution in hexane (8.36 mmol) was added dropwise. After complete addition, stirring was continued for 30 min before dry CO2 gas was bubbled through the solution. After another 30 min, the reaction was quenched with water and slowly warmed to ambient temperature. After dilution with EtOAc, the organic phase was extracted with 0.5 M aqueous NaOH (three times). The combined extracts were acidified with 2M HCl and back-extracted with EtOAc (three times). The combined organic phases were washed with brine, dried over Na2SO4 and evaporated to dryness. The crude product was purified via flash chromatography (petrol ether/EtOAc + 5% MeOH + 2% AcOH (25–75%)) to yield 1.14 g (51%) of pure 17 as an off-white semisolid. 1H NMR (200 MHz, CDCl3) δ 10.39 (br s, 1H), 9.10 (s, 1H), 8.65 (s, 1H), 7.43 (d, J = 3.5 Hz, 1H), 6.64 (d, J = 3.5 Hz, 1H), 5.73 (s, 2H), 3.55 (t, J = 8.2 Hz, 2H), 0.90 (t, J = 8.2 Hz, 2H), -0.09 (s, 9H) 13C NMR (50 MHz, CDCl3) δ 171.4, 150.0, 145.8, 132.0, 129.8, 120.3, 118.7, 102.8, 73.3, 66.6, 17.8, −1.4 TLC-MS (ESI) m/z: 291.4 [M − H]− HPLC tret = 8.66 min.
3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxylic acid (18): In a 100 mL round-bottomed flask were dissolved 1.14 g 17 (3.89 mmol) in 50 mL dry DCM and the solution was cooled in an ice/water bath. To this were added 684 mg bromine (4.28 mmol) as 0.5 M solution in DCM in small portions until the persistence of yellow coloration was observed. The reaction was quenched by addition of aqueous Na2S2O3 solution and extracted with DCM (three times). The combined extracts were washed with sat. NH4Cl, water and brine, prior to drying over Na2SO4 and evaporation. The residue was subjected to flash purification (petrol ether/EtOAc + 5% MeOH + 2% AcOH (25–75%)) to obtain 1.29 g (89%) of 18 as a brownish oil. 1H NMR (200 MHz, CDCl3) δ 11.16 (br s, 1H), 9.10 (s, 1H), 8.59 (s, 1H), 7.48 (s, 1H), 5.70 (s, 2H), 3.56 (t, J = 8.2 Hz, 2H), 0.92 (t, J = 8.2 Hz, 2H), −0.07 (s, 9H) 13C NMR (50 MHz, CDCl3) δ δ 171.1, 149.1, 146.8, 130.9, 128.7, 119.8, 119.3, 91.8, 73.2, 66.9, 17.8, −1.4 TLC-MS (ESI) m/z: 369.2 [M − H]− HPLC tret = 9.82 min.
3-bromo-5-nitro-1H-pyrrolo[2,3-b]pyridine (20): To an ice-cooled suspension of 1.8 g 5-nitro-1H-pyrrolo[2,3-b]pyridine (11.0 mmol) in 40 mL dry DMF were added 2.36 g N-bromosuccinimide (13.2 mmol) as solid in small portions. After complete addition, the cooling bath was removed and the reaction was stirred for 3 h at ambient temperature. The mixture was diluted with water and aqueous Na2S2O3 and the precipitate was isolate by filtration. The filter cake was washed with water and dried at 60 °C in a convection oven to yield 1.86 g (70%) of the title compound as a yellow solid. 1H NMR (200 MHz, DMSO-d6) δ 12.95 (s, 1H), 9.18 (d, J = 2.5 Hz, 1H), 8.64 (d, J = 2.5 Hz, 1H), 8.09 (d, J = 2.6 Hz, 1H). TLC-MS (ESI) m/z: 239.1 [M − H]− HPLC tret = 7.73 min.
3-bromo-5-nitro-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine (21): To an ice-cooled solution of 2.14 g 20 (8.9 mmol) in 20 mL dry DMF were added 462 mg sodium hydride (60%wt dispersion in min. oil, 11.5 mmol) and stirring was continued for about one h. Subsequently were added 1.7 mL SEM-Cl (9.7 mmol) and stirring was continued until TLC indicated complete conversion. The reaction was diluted with 120 mL EtOAc and transferred to a separatory funnel. The organic phase was successively washed with water (four times) and brine, prior to drying over Na2SO4 and evaporation. The residue was purified via flash chromatography (petrol ether/EtOAc (0–20%)) to obtain 1.85 g (56%) 21 as a yellow oil. 1H NMR (200 MHz, CDCl3) δ 9.24 (d, J = 2.4 Hz, 1H), 8.73 (d, J = 2.4 Hz, 1H), 7.58 (s, 1H), 5.70 (s, 2H), 3.54 (m, 2H), 0.92 (m, 2H), −0.05 (s, 9H). HPLC tret = 11.56 min.
3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-amine (22): To a solution of 1.85 g 21 (5.0 mmol) in 40 mL abs. EtOH were added 1.63 g finely powdered zinc (24.9 mmol) and 1.57 g ammonium formate (24.9 mmol). The mixture was stirred for 22 h at 50 °C and was then filtered over a celite pad to remove unreacted zinc. The filtrate was concentrated under reduced pressure and taken up in EtOAc. The organic phase was washed with sat NH4Cl and brine, prior to drying over Na2SO4 and evaporation. The residue was purified via flash chromatography (petrol ether/EtOAc 20–60%) to obtain 0.86 g (51%) of the title compound as a brown oil. 1H NMR (200 MHz, CDCl3) δ 7.93 (d, J = 2.6 Hz, 1H), 7.27 (s, 1H), 7.14 (d, J = 2.6 Hz, 1H), 5.55 (s, 2H), 4.60 (s, 2H), 3.51 (t, 2H), 0.88 (t, 2H), −0.08 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 142.1, 137.5, 134.6, 127.2, 120.2, 112.5, 88.5, 72.9, 66.2, 17.7, −1.6. HPLC tret = 8.65 min.
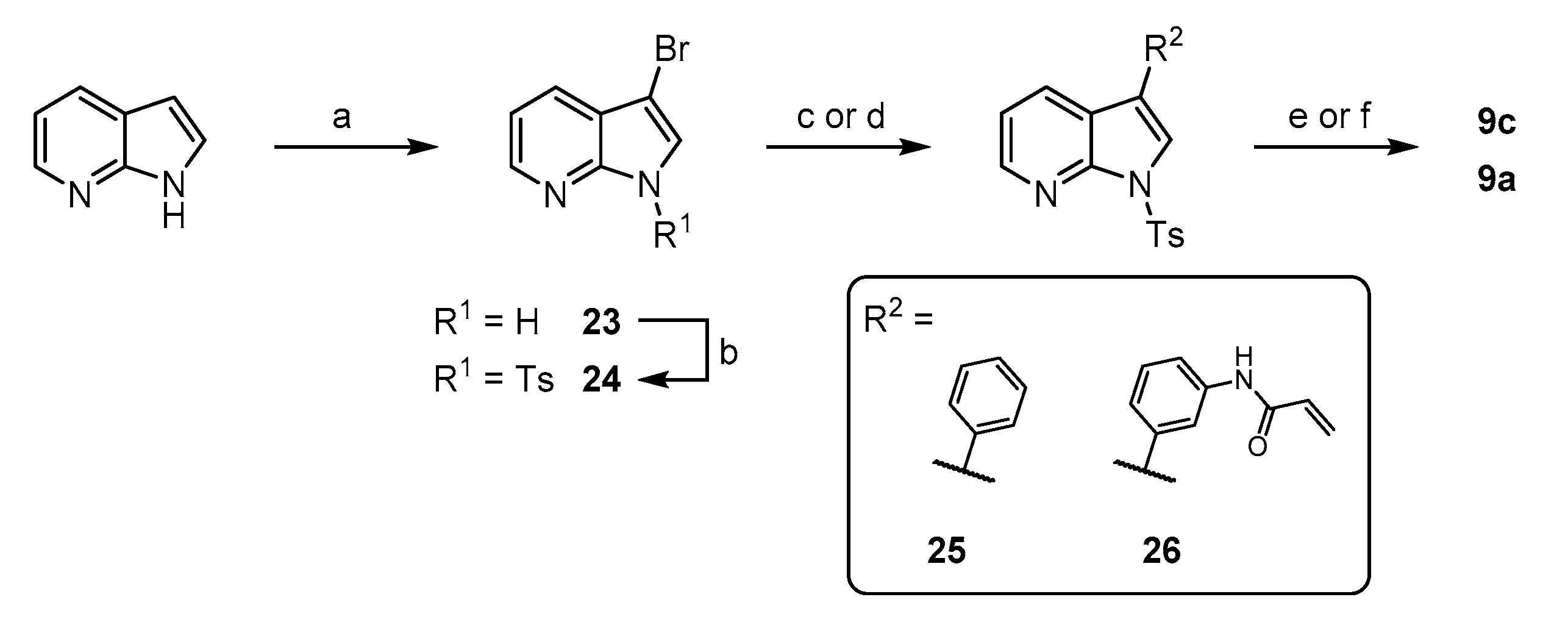
3-bromo-1H-pyrrolo[2,3-b]pyridine (23): To an ice-cooled solution of 473 mg 1H-pyrrolo[2,3-b]pyridine (4.0 mmol) in 8 mL DMF were added 726 mg N-bromosuccinimide (4.1 mmol) in several portions. After complete addition, the cooling bath was removed and stirring was continued for 4 h. The reaction mixture was poured on sat. NaHCO3/ice and the resulting suspension was stirred for ca. 10 min until a homogenous precipitate was formed. The solids were collected by filtration, washed with water and dried in vacuo to yield 753 mg (96%) of the title compound as a white solid. 1H NMR (200 MHz, CDCl3) δ 12.07 (br s, 1H), 8.29 (dd, J = 4.7, 1.5 Hz, 1H), 7.83 (dd, J = 7.9, 1.5 Hz, 1H), 7.70 (s, 1H), 7.16 (dd, J = 7.9, 4.7 Hz, 1H) 13C NMR (50 MHz, DMSO) δ 147.2, 143.9, 126.4, 125.6, 118.7, 116.3, 87.1 TLC-MS (ESI) m/z: 197.1 [M + H]+ HPLC tret = 6.56 min.
3-bromo-1-tosyl-1H-pyrrolo[2,3-b]pyridine (24): To an ice-cooled solution of 197 mg 23 (1.0 mmol) in 5 mL dry THF were added 50 mg sodium hydride (60%wt dispersion in min. oil, 1.25 mmol) and stirring was continued for about 30 min. Subsequently were added 210 mg TsCl (1.1 mmol) and stirring was continued until TLC indicated complete conversion. The reaction was diluted with 25 mL EtOAc and transferred to a separatory funnel. The organic phase was successively washed with water and brine, prior to drying over Na2SO4 and evaporation. The residue was triturated with chilled MeOH and filtered to obtain 298 mg (85%) 24 as a white solid. 1H NMR (200 MHz, DMSO) δ 8.49–8.40 (m, 1H), 8.20 (s, 1H), 8.01 (d, J = 8.4 Hz, 2H), 7.96–7.86 (m, 1H), 7.46–7.33 (m, 3H), 2.31 (s, 3H) 13C NMR (50 MHz, DMSO) δ 146.1, 146.0, 145.4, 134.2, 130.2, 128.7, 127.8, 125.7, 121.7, 120.2, 95.1, 21.1 HPLC tret = 8.86 min.
3-phenyl-1-tosyl-1H-pyrrolo[2,3-b]pyridine (25): The preparation was performed following General Procedure A from 100 mg 24 (0.25 mmol) and 38 mg phenylboronic acid (0.31 mmol), catalyzed by 1 mg Pd(OAc)2 (5 µmol) and 5 mg XPhos (10 µmol) in 3 mL dioxane and 0.75 mL of an 1 M Na2CO3 solution. The reaction was conducted at 70 °C and the crude product was purified via flash chromatography (petrol ether/EtOAc + 5% MeOH (5–30%)) to obtain 72 mg (82%) of the title compound as a colorless semisolid. 1H NMR (200 MHz, CDCl3) δ 8.50 (dd, J = 4.7, 1.2 Hz, 1H), 8.25–8.04 (m, 3H), 7.92 (s, 1H), 7.68–7.56 (m, 2H), 7.55–7.17 (m, 6H), 2.36 (s, 3H) 13C NMR (50 MHz, CDCl3) δ 147.6, 145.3, 145.1, 135.4, 132.6, 129.7, 129.1, 128.9, 128.1, 127.7, 127.4, 122.7, 121.6, 120.3, 119.1, 21.6 TLC-MS (ESI) m/z: 403.0 [M + Na + MeOH]+ HPLC tret = 9.02 min.
N-(3-(1-tosyl-1H-pyrrolo[2,3-b]pyridin-3-yl)phenyl)acrylamide (26): The preparation was performed following General Procedure A from 53 mg 24 (0.15 mmol) and 51 mg 14 (0.19 mmol), catalyzed by 3 mg XPhos Pd G3 (5 µmol) in 3.6 mL dioxane and 0.9 mL of an 0.5 M K2CO3 solution. The reaction was conducted at 50 °C and the crude product was purified via flash chromatography (petrol ether/EtOAc + 5% MeOH (40–70%)) to obtain 63 mg (quant.) of the title compound as a colorless waxy solid. 1H NMR (200 MHz, CDCl3) δ 8.47–8.35 (m, 5H), 8.23–7.95 (m, 5H), 7.85 (s, 1H), 7.57–7.45 (m, 1H), 7.40–7.11 (m, 5H), 6.52–6.24 (m, 2H), 5.74 (dd, J = 8.7, 2.5 Hz, 1H), 2.34 (s, 3H) 13C NMR (50 MHz, CDCl3) δ 164.3, 147.5, 145.5, 145.1, 138.8, 135.2, 133.2, 131.3, 129.8, 129.6, 129.2, 128.0, 127.9, 123.3, 122.8, 121.5, 120.1, 119.3, 119.2, 21.6 TLC-MS (ESI) m/z: 416.5 [M − H]− HPLC tret = 8.00 min.
3-phenyl-1H-pyrrolo[2,3-b]pyridine (9c): In a 25 mL round-bottomed flask were dissolved 72 mg 25 (0.3 mmol) in 6 mL of a 1 M solution of KOH in MeOH at 60 °C oil-bath temperature. The reaction was stirred for 3 h and was then quenched by the addition of sat. NH4Cl solution. The aqueous phase was extracted with EtOAc (4 × 15 mL) and the combined extracts were washed with brine. After drying over Na2SO4 and evaporation, the residue was purified via flash chromatography (DCM/EtOAc (10–80%)) to yield 34 mg (85%) of the final compound as a white solid. 1H NMR (400 MHz, DMSO) δ 11.92 (br s, 1H), 8.35–8.20 (m, 2H), 7.87 (s, 1H), 7.77–7.64 (m, 2H), 7.49–7.36 (m, 2H), 7.25 (t, J = 7.9 Hz, 1H), 7.17–7.08 (m, 1H) 13C NMR (100 MHz, DMSO) δ 149.1, 142.9, 135.0, 128.8, 127.4, 126.2, 125.6, 123.6, 117.3, 116.0, 114.3 TLC-MS (ESI) m/z: 193.0 [M − H]− HPLC tret = 7.60 min.
N-(3-(1H-pyrrolo[2,3-b]pyridin-3-yl)phenyl)acrylamide (9a): To a suspension of 63 mg 26 (0.15 mmol) in 10 mL tBuOH were added 561 mg finely powdered KOH (10 mmol) and the mixture was heated to 50 °C oil-bath temperature until TLC indicated complete conversion. The reaction was quenched by the addition of sat. NH4Cl solution, followed by extraction with EtOAc (3 × 20 mL). The combined extracts were washed with brine and dried over Na2SO4. After evaporation of the volatiles, the residue was purified via flash chromatography (DCM/MeOH (2–8%)) to yield 32 mg (81%) of the final compound as a white solid. 1H NMR (200 MHz, DMSO) δ 11.94 (s, 1H), 10.22 (s, 1H), 8.39–8.23 (m, 2H), 8.14 (br s, 1H), 7.86 (d, J = 2.5 Hz, 1H), 7.63–7.50 (m, 1H), 7.47–7.30 (m, 2H), 7.19 (dd, J = 7.9, 4.9 Hz, 1H), 6.48 (dd, J = 16.8, 9.7 Hz, 1H), 6.29 (dd, J = 16.8, 2.1 Hz, 1H), 5.78 (dd, J = 9.7, 2.1 Hz, 1H) 13C NMR (50 MHz, DMSO) δ 163.3, 149.1, 143.0, 139.5, 135.5, 132.0, 129.3, 127.4, 126.9, 123.7, 121.4, 117.2, 117.1, 116.6, 116.1, 114.1 TLC-MS (ESI) m/z: 318.5 [M + Na + MeOH]+ HPLC tret = 5.42 min.
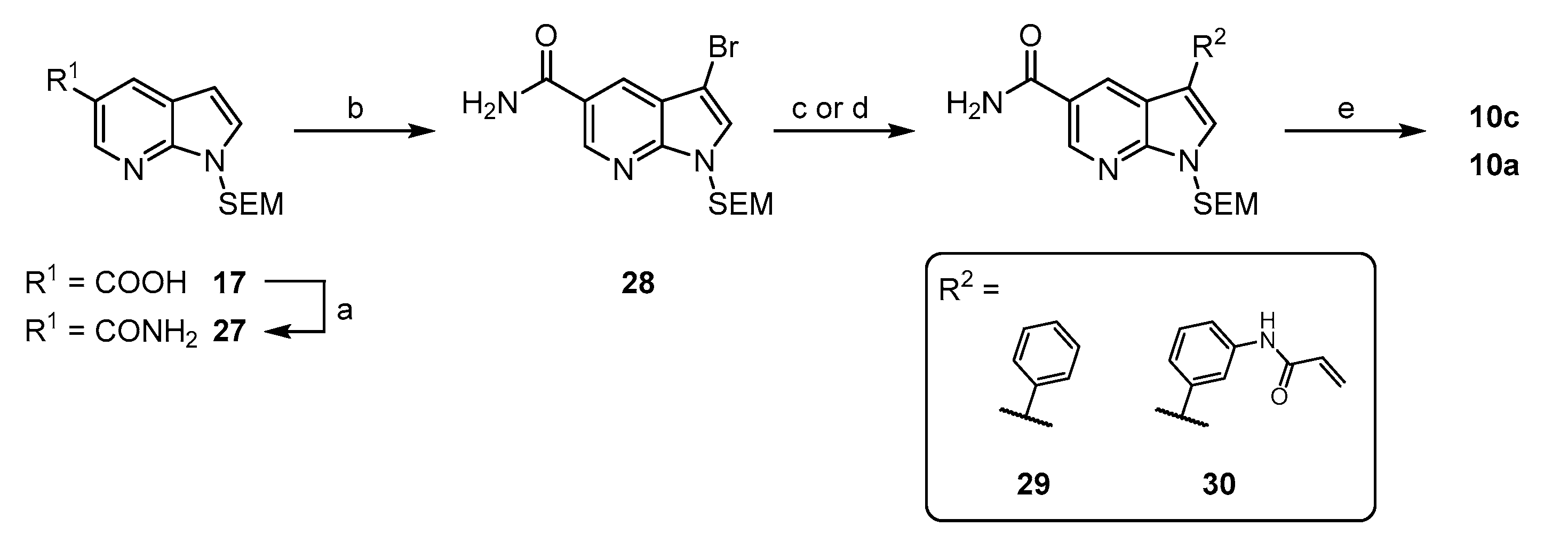
1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (27): To a solution of 235 mg 17 (0.8 mmol) were added 156 mg CDI (0.96 mmol) and stirring was continued at ambient temperature for 30 min. Then 5 mL conc. NH3 solution were added and after another hour of stirring the reaction was further diluted with water and transferred to a separatory funnel. The aqueous phase was extracted with EtOAc (3 × 20 mL) and the combined organic phases were backwashed two times with brine. After drying over Na2SO4 and evaporation, the residue was purified via flash chromatography (petrol ether/EtOAc + 5% MeOH (40–100%)) to obtain 169 mg (72%) of the title compound as a white solid. 1H NMR (200 MHz, CDCl3) δ 8.82 (s, 1H), 8.40 (s, 1H), 7.38 (d, J = 3.3 Hz, 1H), 6.60 (br s, 2H), 6.55 (d, J = 3.3 Hz, 1H), 5.66 (s, 2H), 3.51 (t, J = 8.2 Hz, 2H), 0.87 (t, J = 8.2 Hz, 2 H), −0.11 (s, 9H) 13C NMR (50 MHz, CDCl3) δ 169.4, 149.5, 142.9, 129.6, 129.1, 122.4, 120.1, 102.3, 73.2, 66.5, 17.8, -1.4 TLC-MS (ESI) m/z: 314.4 [M + Na]+ HPLC tret = 7.58 min.
3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (28): An ice-cooled solution of 150 mg 27 (0.52 mmol) in 10 mL dry DCM was treated dropwise with a 1 M bromine solution in DCM until a yellow coloration persisted. TLC indicated complete conversion at this point and the reaction was quenched by addition of aqueous Na2SO3 solution. After dilution with 40 mL EtOAc, the organic phase was washed with sat. NaHCO3 and brine, prior to drying over Na2SO4 and evaporation. The residue was purified via flash chromatography (petrol ether/EtOAc + 5% MeOH (20–100%)) to obtain 130 mg (68%) of the title compound as a white solid. 1H NMR (200 MHz, CDCl3) δ 8.85 (s, 1H), 8.31 (s, 1H), 7.42 (s, 1H), 6.81–6.45 (m, 2H), 5.64 (s, 2H), 3.52 (t, J = 8.2 Hz, 2H), 0.88 (t, J = 8.2 Hz, 2H), −0.09 (s, 9H) 13C NMR (50 MHz, CDCl3) δ 168.9, 148.4, 144.2, 128.5, 127.8, 123.1, 119.5, 91.3, 73.2, 66.8, 17.8, −1.4 TLC-MS (ESI) m/z: 424.3 [M + Na + MeOH]+ HPLC tret = 8.77 min.
3-phenyl-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (29): The preparation was performed following General Procedure A from 62 mg 28 (0.17 mmol) and 24 mg phenylboronic acid (0.20 mmol), catalyzed by 2 mg tBu3P Pd G3 (3 µmol) in 4 mL dioxane and 1 mL of an 0.5 M K3PO4 solution. The reaction was conducted at ambient temperature and the crude product was of sufficient purity to be used directly in the next step. Yield: 62 mg (quant.) as a colorless semisolid. 1H NMR (200 MHz, CDCl3/MeOD) δ 8.85 (d, J = 1.6 Hz, 1H), 8.73 (d, J = 1.9 Hz, 1H), 7.67–7.56 (m, 3H), 7.50–7.27 (m, 3 H), 5.71 (s, 2 H), 3.59 (t, J = 8.3 Hz, 2H), 0.93 (t, J = 8.3 Hz, 2H), −0.06 (s, 9H). 13C NMR (50 MHz, CDCl3/MeOD) δ 169.3, 149.6, 143.0, 133.5, 128.9, 128.6, 127.1, 126.8, 126.2, 122.5, 118.3, 117.7, 73.1, 66.5, 17.6, −1.6. TLC-MS (ESI) m/z: 422.2 [M + Na + MeOH]+ HPLC tret = 9.44 min.
3-(3-acrylamidophenyl)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (30): The preparation was performed following General Procedure A from 61 mg 28 (0.17 mmol) and 56 mg 14 (0.21 mmol), catalyzed by 3 mg tBu3P Pd G3 (5 µmol) in 4 mL dioxane and 1 mL of an 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (petrol ether/EtOAc + 5% MeOH (40–100%)). Yield: 67 mg (93%) as colorless a semisolid. 1H NMR (200 MHz, DMSO) δ 10.28 (br s, 1H), 8.87 (s, 1H), 8.78 (s, 1H), 8.18 (br s, 1H), 8.07 (s, 1H), 8.03 (s, 1H), 7.77–7.60 (m, 1 H), 7.45 (d, J = 4.7 Hz, 3H), 6.49 (dd, J = 17.0, 9.8 Hz, 1 H), 6.29 (dd, J = 17.0, 1.7 Hz, 1H), 5.85–5.53 (m, 3H), 3.58 (t, J = 7.8 Hz, 2H), 0.85 (t, J = 7.8 Hz, 2H), −0.10 (s, 9H) 13C NMR (50 MHz, DMSO) δ 167.4, 163.3, 149.2, 143.5, 143.5, 139.7, 134.2, 131.9, 129.5, 127.9, 127.8, 127.0, 123.5, 122.0, 117.6, 117.0, 115.6, 72.7, 65.7, 17.2, −1.4 TLC-MS (ESI) m/z: 459.5 [M + Na]+ HPLC tret = 8.53 min.
3-phenyl-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (10c): The preparation was carried out following General Procedure B starting from 62 mg 29 (0.17 mmol) in 6 mL DCM and 2 mL TFA. Flash purification (DCM/MeOH (8–16%)) afforded 37 mg (92%) of the final compound as a white solid. 1H NMR (200 MHz, DMSO) δ 12.18 (br s, 1H), 8.82 (d, J = 1.4 Hz, 1H), 8.77 (d, J = 1.4 Hz, 1H), 8.18 (br s, 1H), 7.96 (d, J = 2.0 Hz, 1H), 7.79 (s, 1H), 7.76 (s, 1H), 7.54–7.21 (m, 4H). 13C NMR (50 MHz, DMSO) δ 167.7, 150.2, 143.4, 134.5, 128.9, 127.2, 126.5, 126.0, 125.0, 122.4, 116.4, 115.4. TLC-MS (ESI) m/z: 238.0 [M + H]+ HPLC tret = 5.19 min.
3-(3-acrylamidophenyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (10a): The preparation was carried out following General Procedure B starting from 90 mg 30 (0.21 mmol) in 8 mL DCM and 2 mL TFA. Flash purification (DCM/MeOH (6–16%)) afforded 45 mg (71%) of the final compound as a white solid. 1H NMR (400 MHz, DMSO) δ 12.17 (s, 1H), 10.26 (s, 1H), 8.81 (s, 1H), 8.76 (s, 1H), 8.12 (br s, 1H), 7.99 (s, 1H), 7.88 (s, 1H), 7.68 (d, J = 7.4 Hz, 1H), 7.50–7.31 (m, 3H), 6.55–6.42 (m, 1H), 6.35–6.22 (m, 1H), 5.83–5.72 (m, 1H) 13C NMR (100 MHz, DMSO) δ 167.7, 163.2, 150.1, 143.2, 139.5, 134.9, 131.9, 129.3, 127.2, 126.7, 124.8, 122.6, 121.9, 117.5, 117.2, 116.4, 115.3 TLC-MS (ESI) m/z: 329.3 [M + Na]+ HPLC tret = 3.94 min.
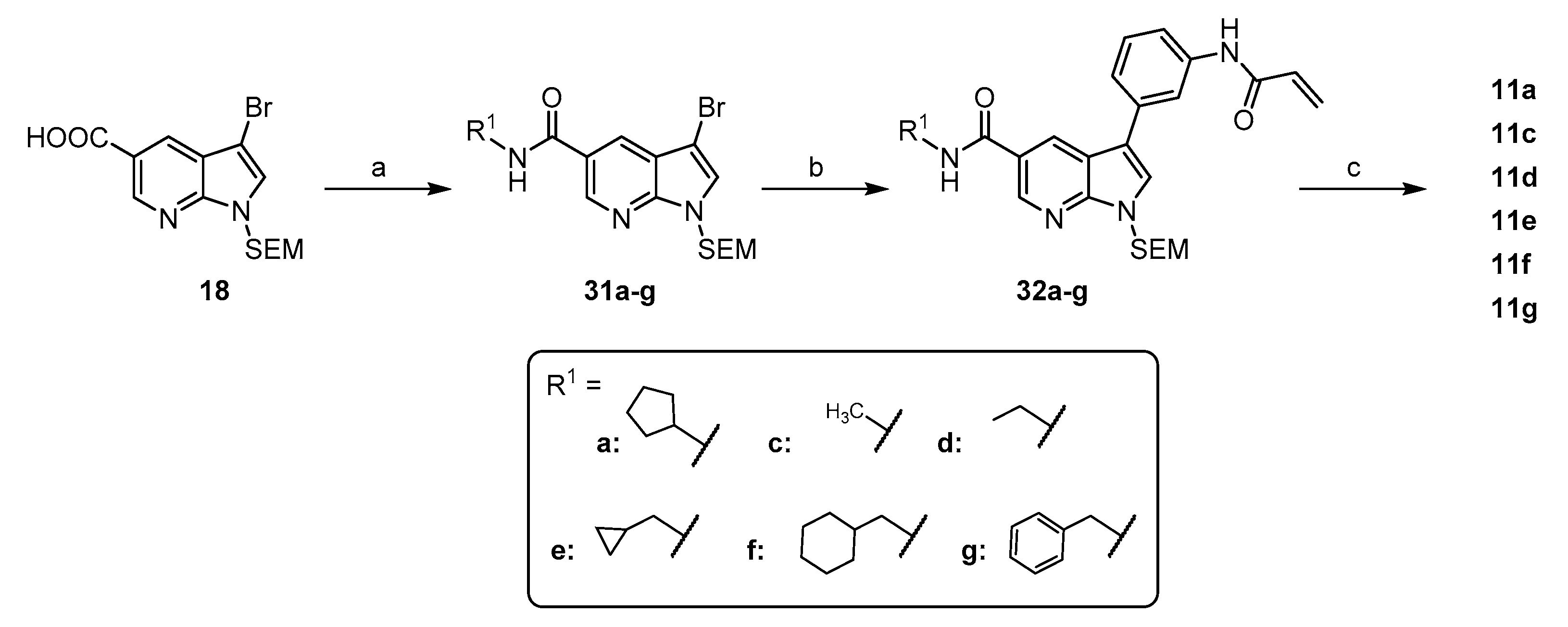
3-bromo-N-cyclopentyl-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (31a): The amide coupling was performed following General Procedure C starting from 111 mg 18 (0.30 mmol) and 58 mg CDI (0.36 mmol) followed by reaction with 59 µL cyclopentylamine (0.60 mmol). The crude product was purified via flash chromatography (hexane/EtOAc (10–40%)). Yield: 105 mg (80%) as a colorless oil. 1H NMR (200 MHz, CDCl3) δ 8.73 (d, J = 1.9 Hz, 1H), 8.15 (d, J = 1.9 Hz, 1H), 7.36 (s, 1H), 6.60 (d, J = 7.2 Hz, 1H), 5.58 (s, 2H), 4.49–4.28 (m, 1H), 3.56–3.37 (m, 2H), 2.15–1.93 (m, 2H), 1.80–1.38 (m, 6H), 0.90–0.76 (m, 2H), −0.11 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 166.2, 148.1, 144.0, 128.1, 126.7, 124.5, 119.1, 90.9, 73.0, 66.6, 51.9, 33.2, 23.9, 23.9, 17.8, −1.4. TLC-MS (ESI) m/z: 492.3 [M + Na + MeOH]+ HPLC tret = 10.49 min.
3-bromo-N-methyl-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (31c): The amide coupling was performed following General Procedure C starting from 100 mg 18 (0.27 mmol) and 53 mg CDI (0.32 mmol) followed by addition with 270 µL of a 2 M solution of methylamine in THF (0.54 mmol). The crude product was purified via flash chromatography (hexane/EtOAc (10–50%)). Yield: 60 mg (58%) as a white solid. 1H NMR (200 MHz, CDCl3) δ 8.77 (d, J = 1.9 Hz, 1H), 8.21 (d, J = 2.0 Hz, 1H), 7.38 (s, 1H), 6.90 (d, J = 5.1 Hz, 1H), 5.60 (s, 2H), 3.49 (t, J = 8.3 Hz, 2H), 3.01 (d, J = 4.7 Hz, 3H), 0.86 (t, J = 8.2 Hz, 2H), -0.11 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 167.4, 148.2, 143.9, 128.3, 126.9, 124.2, 119.2, 91.0, 73.1, 66.7, 27.1, 17.8, −1.4 TLC-MS (ESI) m/z: 405.9 [M + Na]+ HPLC tret = 9.47 min.
3-bromo-N-ethyl-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (31d): The amide coupling was performed following General Procedure C starting from 150 mg 18 (0.41 mmol) and 86 mg CDI (0.53 mmol) followed by addition with 1 mL of a 2 M solution of ethylamine in MeOH (2.0 mmol). The crude product was purified via flash chromatography (hexane/EtOAc (10–50%)). Yield: 74 mg (46%) as a yellowish solid. 1H NMR (200 MHz, CDCl3) δ 9.02 (d, J = 1.8 Hz, 1H), 8.53 (d, J = 2.0 Hz, 1H), 7.45 (s, 1H), 7.38 (s, 1H), 5.67 (s, 2H), 3.70 (dd, J = 16.2, 10.0 Hz, 2H), 3.53 (t, J = 8.3 Hz, 2H), 1.01–0.77 (m, 5H), −0.07 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 166.3, 148.0, 143.6, 128.2, 126.8, 124.2, 119.2, 90.9, 73.0, 66.6, 35.0, 17.7, 14.9, −1.5. HPLC tret = 9.85 min.
3-bromo-N-(cyclopropylmethyl)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (31e): The amide coupling was performed following General Procedure C starting from 100 mg 18 (0.27 mmol) and 53 mg CDI (0.32 mmol) followed by reaction with 94 µL cyclopropylmethanamine (1.35 mmol). The crude product was purified via flash chromatography (hexane/EtOAc (20–80%)). Yield: 83 mg (80%) as a white solid. 1H NMR (200 MHz, CDCl3 + MeOD) δ 8.77 (d, J = 2.0 Hz, 1H), 8.20 (d, J = 2.0 Hz, 1H), 7.44 (s, 1H), 6.42 (s, 1H), 5.65 (s, 2H), 3.70 (s, 2H), 3.60–3.42 (m, 2H), 3.04–2.85 (m, 1H), 0.99–0.80 (m, 4H), 0.76–0.58 (t, 2H), −0.07 (s, 9H). 13C NMR (50 MHz, CDCl3 + MeOD) δ 168.0, 148.5, 144.0, 128.4, 126.9, 124.1, 119.3, 91.2, 73.1, 67.2, 66.8, 23.4, 17.9, 7.0, −1.3. TLC-MS (ESI) m/z: 446.0 [M + Na]+ HPLC tret = 9.88 min.
3-bromo-N-(cyclohexylmethyl)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (31f): The amide coupling was performed following General Procedure C starting from 100 mg 18 (0.27 mmol) and 53 mg CDI (0.32 mmol) followed by reaction with 175 µL cyclohexylmethanamine (1.35 mmol). The crude product was purified via flash chromatography (hexane/EtOAc (0–40%)). Yield: 72 mg (53%) as a brown oil. 1H NMR (200 MHz, CDCl3) δ 8.82 (d, J = 2.1 Hz, 1H), 8.26 (d, J = 2.1 Hz, 1H), 7.46 (s, 1H), 6.27 (s, 1H), 5.67 (s, 2H), 3.53 (t, 2H), 3.37 (dd, J = 7.1, 5.4 Hz, 2H), 1.37 –1.06 (m, 4H), 0.99–0.82 (m, 4H), 0.65–0.53 (m, 2H), 0.33 (t, J = 5.2 Hz, 2H), 0.07 (s, 1H), −0.06 (s, 9H). TLC-MS (ESI) m/z: 488.0 [M + Na]+ HPLC tret = 12.44 min.
N-benzyl-3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (31g): The amide coupling was performed following General Procedure C starting from 300 mg 18 (0.81 mmol) and 171 mg CDI (1.05 mmol) followed by reaction with 441 µL benzylamine (1.35 mmol). The crude product was purified via flash chromatography (hexane/EtOAc (10–50%)). Yield: 205 mg (55%) as a brown viscous oil. 1H NMR (200 MHz, CDCl3) δ 8.83 (d, J = 2.1 Hz, 1H), 8.26 (d, J = 2.0 Hz, 1H), 7.42 (s, 1H), 7.41–7.27 (m, 5H), 6.73 (t, J = 5.7 Hz, 1H), 5.64 (s, 2H), 4.67 (d, J = 5.6 Hz, 2H), 3.58–3.41 (m, 2H), 0.96–0.81 (m, 2H), −0.07 (s, 9H). TLC-MS (ESI) m/z: 481.9 [M + Na]+ HPLC tret = 10.60 min.
3-(3-acrylamidophenyl)-N-cyclopentyl-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (32a): The preparation was performed following General Procedure A from 105 mg 31a (0.24 mmol) and 71 mg 14 (0.26 mmol), catalyzed by 3 mg tBu3P Pd G3 (5 µmol) in 5.6 mL dioxane and 1.4 mL of a 0.5 M K3PO4 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (hexane/EtOAc (30–70%)). Yield: 92 mg (78%) as a white solid. 1H NMR (200 MHz, DMSO) δ 10.28 (s, 1H), 8.82 (d, J = 1.9 Hz, 1H), 8.72 (d, J = 1.9 Hz, 1H), 8.44 (d, J = 7.3 Hz, 1H), 8.10–8.00 (m, 2H), 7.71–7.60 (m, 1H), 7.52–7.40 (m, 2H), 6.49 (dd, J = 17.0, 9.8 Hz, 1H), 6.29 (dd, J = 17.0, 2.2 Hz, 1H), 5.79 (dd, J = 9.8, 2.2 Hz, 1H), 5.71 (s, 2H), 4.38–4.18 (m, 1H), 3.58 (t, J = 7.9 Hz, 2H), 2.02–1.84 (m, 2H), 1.76–1.39 (m, 6H), 0.84 (t, J = 7.9 Hz, 2H), −0.10 (s, 9H). 13C NMR (50 MHz, DMSO) δ 165.4, 163.3, 149.0, 143.2, 139.7, 134.3, 131.9, 129.5, 127.8, 127.4, 126.9, 124.2, 121.9, 117.6, 117.5, 116.9, 115.6, 72.7, 65.7, 51.0, 32.2, 23.7, 17.2, −1.4. TLC-MS (ESI) m/z: 527.5 [M + Na]+ HPLC tret = 9.90 min.
3-(3-acrylamidophenyl)-N-methyl-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (32c): The preparation was performed following General Procedure A from 60 mg 31c (0.16 mmol) and 64 mg 14 (0.23 mmol), catalyzed by 3 mg tBu3P Pd G3 (5 µmol) in 4 mL dioxane and 0.9 mL of a 0.5 M K3PO4 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (petrol ether/EtOAc + 5% MeOH (40–100%)). Yield: 48 mg (69%) as a white solid. The material was carried on directly to the next step. TLC-MS (ESI) m/z: 473.0 [M + Na]+ HPLC tret = 8.97 min.
3-(3-acrylamidophenyl)-N-ethyl-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (32d): The preparation was performed following General Procedure A from 74 mg 31d (0.19 mmol) and 64 mg 14 (0.23 mmol), catalyzed by 3 mg tBu3P Pd G3 (5 µmol) in 4 mL dioxane and 1.1 mL of a 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (petrol ether/EtOAc (40–100%)). Yield: 48 mg (69%) as white solid. The material was carried on directly to the next step. TLC-MS (ESI) m/z: 487.2 [M + Na]+ HPLC tret = 9.11 min.
3-(3-acrylamidophenyl)-N-(cyclopropylmethyl)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (32e): The preparation was performed following General Procedure A from 83 mg 31e (0.20 mmol) and 83 mg 14 (0.30 mmol), catalyzed by 3 mg tBu3P Pd G3 (5 µmol) in 5 mL dioxane and 1.2 mL of a 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (petrol ether/EtOAc + 5% MeOH (40–100%)). Yield: 78 mg (59%) as a white solid. 1H NMR (200 MHz, CDCl3) δ 8.82 (d, J = 2.0 Hz, 1H), 8.68 (d, J = 2.0 Hz, 1H), 8.08 (s, 1H), 7.93 (s, 1H), 7.55 (s, 1H), 7.49 (s, 1H), 7.34 (q, J = 7.4 Hz, 2H), 6.73 (t, J = 5.5 Hz, 1H), 6.55–6.33 (m, 2H), 5.76 (dd, J = 8.3, 3.3 Hz, 1H), 5.69 (s, 2H), 3.56 (t, 2H), 3.37 (t, 2H), 1.13 (d, J = 9.8 Hz, 1H), 1.01–0.82 (m, 2H), 0.65–0.46 (m, 2H), 0.30 (d, J = 5.1 Hz, 2H), −0.07 (s, 9H). TLC-MS (ESI) m/z: 513.2 [M + Na]+ HPLC tret = 8.82 min.
3-(3-acrylamidophenyl)-N-(cyclohexylmethyl)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (32f): The preparation was performed following General Procedure A from 72 mg 31f (0.16 mmol) and 64 mg 14 (0.23 mmol), catalyzed by 3 mg tBu3P Pd G3 (5 µmol) in 4 mL dioxane and 0.9 mL of a 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (petrol ether/EtOAc (20–60%)). Yield: 57 mg (69%) as a yellowish solid. 1H NMR (200 MHz, CDCl3) δ 8.81 (s, 1H), 8.70 (s, 1H), 8.10 (s, 1H), 7.99 (s, 1H), 7.78–7.30 (m, 4H), 6.74 (s, 1H), 6.42 (s, 1H), 6.37 (s, 1H), 5.77 (s, 1H), 5.68 (s, 2H), 3.55 (t, 2H), 3.34 (t, 2H), 1.73 (d, J = 16.2 Hz, 9H), 1.10 (s, 1H), 0.91 (t, J = 8.1 Hz, 2H), 0.07 (s, 1H), −0.07 (s, 9H). TLC-MS (ESI) m/z: 555.3 [M + Na]+ HPLC tret = 10.98 min.
3-(3-acrylamidophenyl)-N-benzyl-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (32g): The preparation was performed following General Procedure A from 200 mg 31g (0.43 mmol) and 149 mg 14 (0.54 mmol), catalyzed by 8 mg tBu3P Pd G3 (13 µmol) in 12 mL dioxane and 2.6 mL of a 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (petrol ether/EtOAc + 5% MeOH (40–100%)). Yield: 120 mg (53%) as a brown solid. 1H NMR (200 MHz, CDCl3 + MeOD) δ 8.61 (s, 1H), 8.51 (s, 1H), 8.36 (t, J = 5.4 Hz, 1H), 7.78 (s, 1H), 7.36 (s, 1H), 7.30–7.19 (m, 1H), 7.17–6.95 (m, 8H), 6.18–6.10 (m, 2H), 5.51–5.44 (m, 1H), 5.41 (s, 2H), 4.08 (s, 2H), 3.33 (t, J = 8.2 Hz, 2H), 0.66 (t, J = 8.2 Hz, 2H), −0.31 (s, 9H). 13C NMR (50 MHz, CDCl3 + MeOD) δ 167.4, 164.7, 149.2, 143.1, 138.6, 138.3, 134.1, 130.9, 129.3, 128.3, 127.9, 127.4, 127.3, 127.0, 126.4, 123.3, 122.8, 118.7, 118.3, 118.1, 117.0, 73.0, 66.4, 43.6, 17.5, −1.9. TLC-MS (ESI) m/z: 548.9 [M+Na]+ HPLC tret = 10.08 min.
3-(3-acrylamidophenyl)-N-cyclopentyl-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (11a): The preparation was carried out following General Procedure B starting from 81 mg 32a (0.16 mmol) in 7.5 mL DCM and 2.5 mL TFA. Flash purification (DCM/MeOH (4–12%)) afforded 49 mg (82%) of the final compound as a white solid. 1H NMR (400 MHz, DMSO) δ 12.16 (br s, 1H), 10.26 (s, 1H), 8.77 (d, J = 1.6 Hz, 1H), 8.70 (d, J = 1.6 Hz, 1H), 8.39 (d, J = 7.1 Hz, 1H), 8.06–7.95 (m, 1H), 7.88 (d, J = 2.4 Hz, 1H), 7.72–7.61 (m, 1H), 7.49–7.34 (m, 2H), 6.48 (dd, J = 17.0, 10.1 Hz, 1H), 6.29 (dd, J = 17.0, 1.8 Hz, 1H), 5.78 (dd, J = 10.1, 1.8 Hz, 1H), 4.34–4.20 (m, 1H), 2.00–1.81 (m, 2H), 1.79–1.64 (m, 2H), 1.63–1.46 (m, 4H). 13C NMR (101 MHz, DMSO) δ 165.6, 163.2, 149.9, 142.9, 139.5, 135.0, 131.9, 129.3, 126.9, 126.7, 124.8, 123.3, 121.8, 117.5, 117.1, 116.3, 115.3, 51.0, 32.1, 23.6. TLC-MS (ESI) m/z: 397.0 [M + Na]+ HPLC tret = 6.94 min.
3-(3-acrylamidophenyl)-N-methyl-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (11c): The preparation was carried out following General Procedure B starting from 48 mg 32c (0.11 mmol) in 7 mL DCM and 3 mL TFA. Flash purification (DCM/MeOH (6–16%)) afforded 31 mg (91%) of the final compound as a white solid. 1H NMR (400 MHz, DMSO-d6) δ 12.18 (s, 1H), 10.27 (s, 1H), 8.76 (d, J = 2.0 Hz, 1H), 8.71 (d, J = 2.0 Hz, 1H), 8.57 (d, J = 4.6 Hz, 1H), 7.98 (s, 1H), 7.89 (d, J = 2.6 Hz, 1H), 7.69 (d, J = 7.5 Hz, 1H), 7.51–7.37 (m, 2H), 6.49 (dd, J = 16.9, 10.1 Hz, 1H), 6.29 (dd, J = 16.9, 2.0 Hz, 1H), 5.78 (dd, 1H), 2.83 (d, J = 4.4 Hz, 3H) 13C NMR (101 MHz, DMSO-d6) δ 166.9, 163.7, 150.5., 143.3, 140.0, 135.4, 132.5, 129.8, 127.3, 127.1, 125.4, 123.5, 122.4, 118.0, 117.7, 116.9, 115.8, 26.7. TLC-MS (ESI) m/z: 343.0 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C18H16N4O2: 321.1346, found: 321.1351. HPLC tret = 4.86 min.
3-(3-acrylamidophenyl)-N-ethyl-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (11d): The preparation was carried out following General Procedure B starting from 68 mg 32d (0.15 mmol) in 4.8 mL DCM and 1.2 mL TFA. Flash purification (DCM/MeOH (6–10%)) afforded 34 mg (70%) of the final compound as a white solid. 1H NMR (400 MHz, DMSO-d6) δ 12.16 (s, 1H), 10.25 (s, 1H), 8.77 (d, J = 2.0 Hz, 1H), 8.71 (d, J = 2.0 Hz, 1H), 8.58 (t, J = 5.5 Hz, 1H), 7.98 (d, J = 1.7 Hz, 1H), 7.88 (d, J = 2.6 Hz, 1H), 7.68 (dt, J = 7.1, 2.1 Hz, 1H), 7.49–7.38 (m, 2H), 6.48 (dd, J = 16.9, 10.1 Hz, 1H), 6.29 (dd, J = 17.0, 2.1 Hz, 1H), 5.78 (dd, J = 10.1, 2.1 Hz, 1H), 3.40–3.33 (m, 2H), 1.16 (t, J = 7.2 Hz, 3H). 13C NMR (101 MHz, DMSO-d6) δ 166.2, 163.7, 150.5, 143.3, 140.0, 135.5, 132.4, 129.8, 127.3, 127.2, 125.4, 123.6, 122.4, 118.0, 117.7, 116.9, 115.8, 34.5, 15.3. TLC-MS (ESI) m/z: 357.0 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C19H18N4O2: 335,1503, found: 335.1508. HPLC tret = 5.53 min.
3-(3-acrylamidophenyl)-N-(cyclopropylmethyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (11e): The preparation was carried out following General Procedure B starting from 78 mg 32e (0.16 mmol) in 4.2 mL DCM and 1.8 mL TFA. Flash purification (DCM/MeOH (6–16%)) afforded 13 mg (23%) of the final compound as a yellowish solid. 1H NMR (400 MHz, DMSO-d6) δ 12.18 (s, 1H), 10.30 (s, 1H), 8.80 (d, J = 2.0 Hz, 1H), 8.74 (d, J = 2.1 Hz, 1H), 8.71 (t, J = 5.6 Hz, 1H), 8.00 (d, J = 2.2 Hz, 1H), 7.90 (s, 1H), 7.70 (dt, J = 7.3, 1.9 Hz, 1H), 7.50–7.38 (m, 2H), 6.49 (dd, J = 17.0, 10.1 Hz, 1H), 6.29 (dd, J = 17.0, 2.1 Hz, 1H), 5.79 (dd, J = 10.0, 2.1 Hz, 1H), 3.20 (t, J = 6.2 Hz, 2H), 1.07 (ttd, J = 12.9, 6.8, 3.7 Hz, 1H), 0.51–0.37 (m, 2H), 0.36–0.22 (m, 2H). 13C NMR (101 MHz, DMSO-d6) δ 165.8, 163.2, 149.9, 142.8, 139.5, 134.9, 131.9, 129.2, 126.8, 126.7, 124.8, 123.0, 121.9, 117.5, 117.1, 116.4, 115.3, 43.5, 11.0, 3.3. TLC-MS (ESI) m/z: 383.1 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C21H20N4O2: 361.1659, found: 361.1662. HPLC tret = 6.54 min.
3-(3-acrylamidophenyl)-N-(cyclohexylmethyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (11f): The preparation was carried out following General Procedure B starting from 30 mg 32f (0.06 mmol) in 7 mL DCM and 3 mL TFA. Flash purification (DCM/MeOH (5–10%)) afforded 15 mg (64%) of the final compound as a yellowish solid. 1H NMR (400 MHz, DMSO-d6) δ 12.16 (s, 1H), 10.27 (s, 1H), 8.78 (s, 1H), 8.72 (s, 1H), 8.54 (s, 1H), 8.02 (s, 1H), 7.88 (d, J = 3.0 Hz, 1H), 7.66 (d, J = 7.2 Hz, 1H), 7.44 (s, 2H), 6.49 (ddd, J = 17.2, 10.1, 3.3 Hz, 1H), 6.29 (d, J = 16.8 Hz, 1H), 5.78 (d, J = 10.2 Hz, 1H), 3.16 (q, J = 6.5, 5.0 Hz, 2H), 1.72 (dd, J = 24.7, 11.6 Hz, 4H), 1.61 (d, J = 10.0 Hz, 2H), 1.22 (s, 1H), 1.17 (d, J = 9.2 Hz, 2H), 0.95 (t, J = 12.0 Hz, 2H). 13C NMR (101 MHz, DMSO-d6) δ 165.9, 163.2, 149.9, 142.8, 139.5, 134.9, 131.9, 129.2, 126.7, 126.6, 124.8, 123.1, 121.8, 117.5, 117.1, 116.3, 115.2, 45.4, 37.4, 30.5, 26.0, 25.4. TLC-MS (ESI) m/z: 425.3 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C24H26N4O2: 403.2129, found: 403.2133. HPLC tret = 8.50 min.
3-(3-acrylamidophenyl)-N-benzyl-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (11g): The preparation was carried out following General Procedure B starting from 80 mg 32g (0.15 mmol) in 7 mL DCM and 3 mL TFA. Flash purification (DCM/MeOH (6–8%)) afforded 58 mg (97%) of the final compound as a pale red solid. 1H NMR (400 MHz, DMSO-d6) δ 12.20 (d, J = 2.7 Hz, 1H), 10.26 (s, 1H), 9.18 (t, J = 6.0 Hz, 1H), 8.84 (d, J = 2.0 Hz, 1H), 8.79 (d, J = 2.0 Hz, 1H), 8.00 (t, J = 1.9 Hz, 1H), 7.90 (d, J = 2.6 Hz, 1H), 7.67 (dt, J = 7.7, 1.8 Hz, 1H), 7.52–7.18 (m, 7H), 6.48 (q, J = 16.9, 10.1 Hz, 1H), 6.29 (dd, J = 17.0, 2.1 Hz, 1H), 5.78 (dd, J = 10.1, 2.0 Hz, 1H), 4.54 (d, J = 5.9 Hz, 2H). 13C NMR (101 MHz, DMSO-d6) δ 166.0, 163.2, 150.1, 142.9, 139.7, 139.5, 134.9, 131.9, 129.3, 128.2, 127.2, 126.9, 126.8, 126.7, 124.9, 122.7, 121.9, 117.5, 117.2, 116.4, 115.4, 42.6. TLC-MS (ESI) m/z: 419.3 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C24H20N4O2: 397.1659, found: 397.1663. HPLC tret = 7.20 min.
N-(3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)cyclopentanecarboxamide (33a): The preparation was carried out following General Procedure D with 90 mg 22 (0.26 mmol), 111 µL Et3N (0.79 mmol) and 70 mg cyclopentanecarboxylic acid chloride (0.53 mmol). Flash purification (hexane/EtOAc (10–50%)) afforded 90 mg (78%) of the product as a colorless oily residue. 1H NMR (200 MHz, CDCl3) δ 8.30–8.21 (m, 1H), 8.20–8.15 (m, 1H), 8.11 (br s, 1H), 7.29 (s, 1H), 5.54 (s, 2H), 3.48 (t, J = 8.2 Hz, 2H), 2.82–2.60 (m, 1H), 1.95–1.80 (m, 4H), 1.78–1.43 (m, 4H), 0.86 (t, J = 8.2 Hz, 2H), −0.10 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 175.6, 144.1, 138.3, 129.5, 127.6, 120.0, 119.7, 89.9, 73.0, 66.5, 46.4, 30.7, 26.1, 17.8, −1.4. TLC-MS (ESI) m/z: 492.1 [M + Na + MeOH]+ HPLC tret = 10.84 min.
N-(3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)thiophene-2-carboxamide (33c): The preparation was carried out following General Procedure D with 100 mg 22 (0.29 mmol), 123 µL Et3N (0.88 mmol) and 41 µL thiophene-2-carbonyl chloride (0.38 mmol). Flash purification (petrol ether/EtOAc + 10% THF (0–50%)) afforded 62 mg (47%) of the product as a white solid. 1H NMR (200 MHz, CDCl3) δ 8.39 (d, J = 2.3 Hz, 1H), 8.24 (d, J = 2.3 Hz, 1H), 8.22 (s, 1H), 7.71 (dd, J = 3.8, 1.1 Hz, 1H), 7.54 (dd, J = 5.0, 1.1 Hz, 1H), 7.36 (s, 1H), 7.10 (dd, J = 5.0, 3.7 Hz, 1H), 5.59 (s, 2H), 3.52 (t, 2H), 0.89 (t, J = 11.1, 2.2, 0.9 Hz, 2H), −0.07 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 161.4, 145.1, 139.5, 139.3, 131.8, 129.6, 129.5, 128.6, 121.5, 120.6, 90.8, 73.8, 67.3, 18.5, −0.7. TLC-MS (ESI) m/z: 474.3 [M + Na]+ HPLC tret = 10.07 min.
N-(3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)furan-2-carboxamide (33d): The preparation was carried out following General Procedure D with 100 mg 22 (0.29 mmol), 123 µL Et3N (0.88 mmol) and 44 µL furane-2-carbonyl chloride (0.38 mmol). Flash purification (petrol ether/EtOAc (40–50%)) afforded 101 mg (80%) of the product as a yellow oil. 1H NMR (200 MHz, CDCl3) δ 8.44 (d, J = 2.4 Hz, 1H), 8.33 (d, J = 2.4 Hz, 1H), 8.29 (s, 1H), 7.51 (dd, J = 1.7, 0.8 Hz, 1H), 7.37 (s, 1H), 7.25 (dd, J = 3.3, 1.1 Hz, 1H), 6.56 (dd, J = 3.6, 1.8 Hz, 1H), 5.61 (s, 2H), 3.52 (t, 2H), 0.89 (t, 2H), −0.07 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 156.4, 147.5, 144.4, 144.3, 138.0, 128.5, 127.8, 119.9, 119.7, 115.4, 112.6, 89.9, 72.9, 66.4, 17.7, −1.5. TLC-MS (ESI) m/z: 458.1 [M + Na]+ HPLC tret = 9.92 min.
N-(3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)benzamide (33e): The preparation was carried out following General Procedure D with 100 mg 22 (0.29 mmol), 123 µL Et3N (0.88 mmol) and 68 µL benzoyl chloride (0.58 mmol). Flash purification (petrol ether/EtOAc (30–60%)) afforded 91 mg (70%) of the product as a yellow solid. 1H NMR (200 MHz, CDCl3) δ 8.40 (s, 1H), 8.38 (s, 1H), 8.28 (s, 1H), 7.90 (d, J = 7.5 Hz, 2H), 7.49 (q, J = 13.4, 11.4 Hz, 3H), 7.35 (s, 1H), 5.59 (s, 2H), 3.52 (t, J = 8.3 Hz, 2H), 0.89 (t, J = 8.0 Hz, 2H), −0.07 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 166.2, 144.3, 138.5, 134.3, 131.9, 129.0, 128.7, 127.8, 127.1, 120.5, 119.7, 89.9, 72.9, 66.4, 17.7, −1.5. TLC-MS (ESI) m/z: 468.2 [M + Na]+ HPLC tret = 10.20 min.
N-(3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)acetamide (33f): The preparation was carried out following General Procedure D with 130 mg 22 (0.38 mmol), 160 µL Et3N (1.14 mmol) and 54 µL acetyl chloride (0.76 mmol). Flash purification (petrol ether/EtOAc (30–80%)) afforded 134 mg (83%) of the product as a yellow solid. 1H NMR (200 MHz, CDCl3) δ 8.25 (d, J = 2.3 Hz, 1H), 8.19 (s, 1H), 8.12 (d, J = 2.3 Hz, 1H), 7.32 (s, 1H), 5.56 (s, 2H), 3.59–3.40 (t, 2H), 2.19 (s, 3H), 0.99–0.73 (t, 2H), −0.08 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 169.4, 144.3, 138.4, 129.2, 127.9, 120.4, 119.8, 90.0, 73.1, 66.6, 24.3, 17.8, −1.4. TLC-MS (ESI) m/z: 406.1 [M + Na]+ HPLC tret = 9.44 min.
N-(3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)-2-cyclopentylacetamide (33g): The preparation was carried out following General Procedure D with 100 mg 22 (0.29 mmol), 123 µL Et3N (0.88 mmol) and 79 µL 2-cyclopentylacetyl chloride (0.58 mmol). Flash purification (petrol ether/EtOAc (0–40%)) afforded 120 mg (91%) of the product as a brown oil. 1H NMR (200 MHz, CDCl3) δ 8.30 (d, J = 2.2 Hz, 1H), 8.27 (d, J = 2.1 Hz, 1H), 7.68–7.52 (m, 1H), 7.36 (s, 1H), 5.59 (s, 2H), 3.65–3.36 (m, 2H), 2.36 (d, J = 6.5 Hz, 2H), 2.23 (p, J = 7.3 Hz, 1H), 2.01–1.75 (m, 4H), 1.72–1.45 (m, 4H), 0.91 (td, J = 8.3, 7.7, 4.8 Hz, 2H), −0.06 (d, J = 2.5 Hz, 9H). TLC-MS (ESI) m/z: 474.2 [M + Na]+ HPLC tret = 11.32 min.
N-(3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)-2-phenylacetamide (33h): The preparation was carried out following General Procedure D with 100 mg 22 (0.29 mmol), 123 µL Et3N (0.88 mmol) and 77 µL 2-phenylacetyl chloride (0.58 mmol). Flash purification (petrol ether/EtOAc (40–70%)) afforded 111 mg (82%) of the product as a yellow oil. 1H NMR (200 MHz, CDCl3) δ 8.18 (d, J = 2.4 Hz, 1H), 8.11 (d, J = 2.4 Hz, 1H), 7.45 (d, J = 2.5 Hz, 1H), 7.41 (d, J = 3.4 Hz, 2H), 7.37 (d, J = 2.1 Hz, 2H), 7.34 (s, 2H), 5.57 (s, 2H), 3.77 (s, 2H), 3.49 (t, 2H), 0.88 (t, J = 8.2 Hz, 2H), −0.07 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 169.6, 144.4, 138.2, 134.3, 129.5, 129.2, 128.7, 127.7, 127.6, 120.1, 119.5, 89.8, 72.9, 66.4, 44.4, 17.7, −1.5. TLC-MS (ESI) m/z: 482.1 [M + Na]+ HPLC tret = 10.42 min.
N-(3-bromo-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)-2-(thiophen-2-yl)acetamide (33i): The preparation was carried out following General Procedure D with 100 mg 22 (0.29 mmol), 123 µL Et3N (0.88 mmol) and 72 µL 2-(thiophen-2-yl)acetyl chloride (0.58 mmol). Flash purification (petrol ether/EtOAc (20–50%)) afforded 123 mg (90%) of the product as a brown oil. 1H NMR (200 MHz, CDCl3) δ 8.22 (d, J = 2.3 Hz, 1H), 8.12 (d, J = 2.3 Hz, 1H), 7.73 (s, 1H), 7.34 (s, 1H), 7.30 (dd, 1H), 7.05 (s, 1H), 7.03 (d, J = 1.6 Hz, 1H), 5.57 (s, 2H), 3.97 (s, 2H), 3.49 (t, 2H), 0.88 (t, 2H), −0.08 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 168.6, 144.5, 138.4, 135.6, 128.7, 128.0, 127.9, 127.7, 126.1, 120.4, 119.8, 90.0, 73.1, 66.6, 38.3, 17.9, −1.3. TLC-MS (ESI) m/z: 488.1 [M + Na]+ HPLC tret = 10.15 min.
N-(3-(3-acrylamidophenyl)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)cyclopentanecarboxamide (34a): The preparation was performed following General Procedure A from 90 mg 33a (0.21 mmol) and 64 mg 14 (0.24 mmol), catalyzed by 2 mg tBu3P Pd G3 (4 µmol) in 3.3 mL dioxane and 0.4 mL of a 1.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (hexane/EtOAc (30–80%)). Yield: 76 mg (73%) as a white solid. 1H NMR (200 MHz, DMSO) δ 10.25 (s, 1H), 10.04 (s, 1H), 8.57 (d, J = 2.1 Hz, 1H), 8.49 (d, J = 2.1 Hz, 1H), 8.02–7.95 (m, 1H), 7.93 (s, 1H), 7.66–7.56 (m, 1H), 7.43 (t, J = 7.7 Hz, 1H), 7.38–7.28 (m, 1H), 6.48 (dd, J = 16.9, 9.9 Hz, 1H), 6.28 (dd, J = 16.9, 2.2 Hz, 1H), 5.78 (dd, J = 9.9, 2.2 Hz, 1H), 5.65 (s, 2H), 3.56 (t, J = 8.0 Hz, 2H), 2.91–2.70 (m, 1H), 1.97–1.46 (m, 8H), 0.84 (t, J = 8.0 Hz, 2H), −0.10 (s, 9H). 13C NMR (50 MHz, DMSO) δ 174.6, 163.3, 144.8, 139.6, 136.7, 134.8, 131.9, 130.6, 129.4, 127.3, 127.0, 121.8, 121.8, 118.6, 117.4, 117.3, 114.5, 72.6, 65.6, 45.1, 30.1, 25.7, 17.2, −1.4. TLC-MS (ESI) m/z: 527.1 [M + Na]+ HPLC tret = 10.24 min.
N-(3-(3-acrylamidophenyl)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)thiophene-2-carboxamide (34c): The preparation was performed following General Procedure A from 90 mg 33c (0.20 mmol) and 81 mg 14 (0.30 mmol), catalyzed by 3 mg tBu3P Pd G3 (5 µmol) in 5.0 mL dioxane and 1.2 mL of a 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (petrol ether/EtOAc + 10% THF (20–50%)). Yield: 46 mg (45%) as a white solid. 1H NMR (200 MHz, DMSO-d6) δ 10.44 (s, 1H), 10.24 (s, 1H), 8.61 (s, 1H), 8.60 (s, 1H), 8.14–7.97 (m, 2H), 7.99 (s, 1H), 7.87 (d, J = 5.0 Hz, 1H), 7.63 (d, J = 7.4 Hz, 1H), 7.42 (d, J = 8.1 Hz, 2H), 7.25 (t, J = 4.4 Hz, 1H), 6.48 (dd, J = 16.9, 9.9 Hz, 1H), 6.27 (dd, J = 16.8, 1.7 Hz, 1H), 5.77 (d, J = 10.0 Hz, 1H), 5.68 (s, 2H), 3.59 (t, J = 7.9 Hz, 2H), 0.86 (t, J = 7.9 Hz, 2H), −0.08 (s, 9H). 13C NMR (50 MHz, -CDCl3 + MeOD) δ 164.8, 161.5, 145.6, 139.2, 138.7, 137.7, 134.7, 131.2, 131.1, 129.5, 129.2, 128.9, 127.9, 127.4, 126.1, 122.4, 121.5, 118.5, 118.3, 118.0, 115.9, 73.2, 66.4, 17.7, −1.7 TLC-MS (ESI) m/z: 541.5 [M + Na]+ HPLC tret = 9.48 min.
N-(3-(3-acrylamidophenyl)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)furan-2-carboxamide (34d): The preparation was performed following General Procedure A from 101 mg 33d (0.23 mmol) and 95 mg 14 (0.35 mmol), catalyzed by 4 mg tBu3P Pd G3 (7 µmol) in 6.0 mL dioxane and 1.4 mL of a 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (petrol ether/EtOAc + 10% MeOH (40–60%)). Yield: 58 mg (50%) as a white solid. 1H NMR (200 MHz, CDCl3 + MeOD) δ 9.38 (s, 1H), 9.19 (s, 1H), 8.63 (d, J = 2.3 Hz, 1H), 8.46 (d, J = 2.3 Hz, 1H), 7.85 (s, 1H), 7.65 (d, J = 7.0 Hz, 1H), 7.59–7.46 (m, 2H), 7.40–7.23 (m, 2H), 7.22 (dd, J = 3.5, 0.8 Hz, 1H), 6.51 (dd, J = 3.5, 1.8 Hz, 1H), 6.38 (s, 1H), 6.35 (d, J = 3.3 Hz, 1H), 5.69 (dd, J = 7.5, 4.3 Hz, 1H), 5.60 (s, 2H), 3.62–3.43 (m, 2H), 0.96–0.78 (m, 2H), −0.12 (s, 9H). 13C NMR (50 MHz, CDCl3 + MeOD) δ 222.3, 221.4, 164.6, 147.5, 145.9, 144.9, 138.9, 137.4, 134.8, 131.3, 129.6, 128.4, 127.5, 126.3, 122.4, 121.2, 118.5, 118.2, 116.0, 115.5, 112.5, 73.2, 66.5, 17.8, −1.5. TLC-MS (ESI) m/z: 525.4 [M + Na]+ HPLC tret = 9.34 min.
N-(3-(3-acrylamidophenyl)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)benzamide (34e): The preparation was performed following General Procedure A from 91 mg 33e (0.20 mmol) and 84 mg 14 (0.31 mmol), catalyzed by 4 mg tBu3P Pd G3 (7 µmol) in 5.0 mL dioxane and 1.2 mL of a 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (petrol ether/EtOAc + 10% MeOH (20–60%)). Yield: 47 mg (45%) as a white solid. 1H NMR (200 MHz, CDCl3 + MeOD) δ 9.65 (s, 1H), 9.47 (s, 1H), 8.57 (d, J = 2.1 Hz, 1H), 8.49 (d, J = 2.1 Hz, 1H), 7.99–7.81 (m, 3H), 7.63–7.25 (m, 6H), 7.23 (s, 1H), 6.32 (s, 1H), 6.29 (d, J = 1.5 Hz, 1H), 5.63 (dd, J = 6.6, 5.1 Hz, 1H), 5.55 (s, 2H), 3.47 (t, 2H), 0.81 (t, J = 8.3 Hz, 2H), −0.17 (s, 9H). 13C NMR (50 MHz, CDCl3 + MeOD) δ 224.0, 167.3, 164.7, 145.7, 138.8, 137.7, 134.8, 134.5, 131.9, 131.2, 129.5, 129.4, 128.6, 127.5, 126.1, 122.3, 121.5, 118.5, 118.3, 118.0, 116.0, 73.2, 66.5, 17.8, −1.6. TLC-MS (ESI) m/z: 535.1 [M + Na]+ HPLC tret = 9.77 min.
N-(3-(5-acetamido-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-3-yl)phenyl)acrylamide (34f): The preparation was performed following General Procedure A from 121 mg 33f (0.32 mmol) and 129 mg 14 (0.47 mmol), catalyzed by 6 mg tBu3P Pd G3 (9 µmol) in 6.0 mL dioxane and 1.9 mL of a 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (DCM/MeOH (0–5%)). Yield: 90 mg (63%) as a white solid. 1H NMR (200 MHz, CDCl3 + MeOD) δ 9.44 (s, 1H), 8.23 (s, 1H), 8.19 (s, 1H), 7.75 (s, 1H), 7.24 (d, J = 9.3 Hz, 2H), 7.04 (dd, J = 6.9, 3.8 Hz, 2H), 6.57 (dt, J = 28.2, 8.8 Hz, 1H), 6.14 (s, 1H), 6.11 (s, 1H), 5.45 (t, J = 5.8 Hz, 1H), 5.32 (s, 2H), 3.26 (t, J = 8.1 Hz, 2H), 1.90 (s, 3H), 0.60 (t, J = 8.2 Hz, 2H), −0.37 (s, 9H). 13C NMR (50 MHz, CDCl3 + MeOD) δ 170.1, 164.7, 145.4, 138.6, 137.0, 134.7, 131.2, 129.5, 129.3, 127.2, 126.0, 122.2, 120.5, 118.4, 118.3, 117.8, 115.7, 73.1, 66.3, 23.4, 17.6, −1.7. TLC-MS (ESI) m/z: 473.1 [M + Na]+ HPLC tret = 9.18 min.
N-(3-(5-(2-cyclopentylacetamido)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-3-yl)phenyl)acrylamide (34g): The preparation was performed following General Procedure A from 120 mg 33g (0.27 mmol) and 109 mg 14 (0.40 mmol), catalyzed by 5 mg tBu3P Pd G3 (8 µmol) in 6.0 mL dioxane and 1.6 mL of a 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (DCM/MeOH (0–5%)). Yield: 85 mg (62%) as a white solid. 1H NMR (200 MHz, DMSO-d6) δ 10.26 (s, 1H), 10.04 (s, 1H), 8.54 (s, 1H), 8.47 (s, 1H), 8.01 (s, 1H), 7.94 (s, 1H), 7.58 (d, J = 6.7 Hz, 1H), 7.53–7.34 (m, 2H), 6.68–6.37 (m, 1H), 6.27 (d, J = 17.8 Hz, 1H), 5.78 (d, J = 12.2 Hz, 1H), 5.65 (s, 2H), 4.12 (t, 2H), 2.34 (s, 2H), 1.67 (d, J = 38.3 Hz, 5H), 1.23 (s, 4H), 0.84 (t, 2H), −0.10 (s, 9H). 13C NMR (50 MHz, CDCl3 + MeOD) δ 172.8, 164.7, 145.3, 138.8, 136.7, 134.8, 131.3, 129.7, 129.5, 127.4, 126.0, 122.2, 120.5, 118.6, 118.3, 117.9, 115.8, 73.2, 66.4, 43.2, 37.2, 32.5, 24.9, 17.8, −1.6. TLC-MS (ESI) m/z: 541.5 [M + Na]+ HPLC tret = 10.60 min.
N-(3-(5-(2-phenylacetamido)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-3-yl)phenyl)acrylamide (34h): The preparation was performed following General Procedure A from 111 mg 33h (0.24 mmol) and 98 mg 14 (0.36 mmol), catalyzed by 4 mg tBu3P Pd G3 (7 µmol) in 5.0 mL dioxane and 1.4 mL of a 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (petrol ether/EtOAc + 10% MeOH (20–60%)). Yield: 59 mg (47%) as a white solid. 1H NMR (200 MHz, CDCl3 + MeOD) δ 9.79 (s, 1H), 9.72 (s, 1H), 8.53 (d, J = 2.3 Hz, 1H), 8.37 (s, 1H), 7.85 (s, 1H), 7.50 (s, 1H), 7.47 (s, 1H), 7.31 (d, J = 5.3 Hz, 1H), 7.28–7.18 (m, 2H), 7.24–7.07 (m, 2H), 7.10–6.81 (m, 1H), 6.32 (s, 1H), 6.29 (s, 1H), 6.24 (d, J = 5.8 Hz, 1H), 5.62 (dt, J = 11.8, 6.3 Hz, 1H), 5.54 (s, 2H), 4.22 (s, 2H), 3.44 (t, 2H), 0.78 (t, 2H), −0.19 (s, 9H). 13C NMR (50 MHz, CDCl3 + MeOD) δ 170.8, 164.8, 144.9, 138.6, 136.2, 134.8, 134.5, 131.1, 129.6, 129.3, 129.0, 128.5, 127.1, 126.9, 126.2, 122.3, 120.8, 118.7, 118.3, 118.0, 115.9, 73.2, 66.3, 43.6, 17.6, −1.9. TLC-MS (ESI) m/z: 549.1 [M + Na]+ HPLC tret = 10.02 min.
N-(3-(5-(2-(thiophen-2-yl)acetamido)-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridin-3-yl)phenyl)acrylamide (34i): The preparation was performed following General Procedure A from 123 mg 33i (0.26 mmol) and 108 mg 14 (0.39 mmol), catalyzed by 5 mg tBu3P Pd G3 (8 µmol) in 6.0 mL dioxane and 1.6 mL of a 0.5 M K2CO3 solution. The reaction was conducted at ambient temperature and the crude product was purified via flash chromatography (DCM/MeOH (0–5%)). Yield: 87 mg (62%) as a white solid. 1H NMR (200 MHz, CDCl3) δ 8.62 (d, J = 2.3 Hz, 1H), 8.49 (d, J = 2.3 Hz, 1H), 8.35 (s, 1H), 8.23 (s, 1H), 8.05 (s, 1H), 7.73 (s, 1H), 7.68 (s, 1H), 7.52 (d, J = 3.3 Hz, 4H), 7.28 (s, 1H), 6.69 (dd, J = 16.6, 1.8 Hz, 1H), 6.55 (dd, J = 16.7, 9.4 Hz, 1H), 5.97 (dd, J = 9.5, 1.8 Hz, 1H), 5.84 (s, 2H), 4.21 (s, 2H), 3.79 (t, J = 8.1 Hz, 2H), 1.16 (t, J = 8.2 Hz, 2H), 0.19 (s, 9H). 13C NMR (50 MHz, CDCl3) δ 168.8, 146.0, 142.7, 141.4, 138.3, 137.7, 135.6, 134.9, 129.4, 128.2, 127.6, 127.4, 126.2, 125.8, 121.2, 118.2, 118.0, 115.9, 92.4, 84.8, 84.3, 73.0, 66.3, 38.0, 17.7, −1.5. TLC-MS (ESI) m/z: 555.1 [M + Na]+ HPLC tret = 9.71 min.
N-(3-(3-acrylamidophenyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)cyclopentanecarboxamide (12a): The preparation was carried out following General Procedure B starting from 70 mg 34a (0.14 mmol) in 5 mL DCM and 2 mL TFA. Flash purification (DCM/MeOH (2–8%)) afforded 40 mg (77%) of the final compound as a white solid. 1H NMR (200 MHz, DMSO) δ 11.84 (br s, 1H), 10.24 (s, 1H), 9.97 (s, 1H), 8.62–8.47 (m, 1H), 8.47–8.32 (m, 1H), 7.93 (s, 1H), 7.75 (d, J = 2.2 Hz, 1H), 7.68–7.54 (m, 1H), 7.49–7.26 (m, 2H), 6.49 (dd, J = 16.9, 9.9 Hz, 1H), 6.28 (dd, J = 16.9, 1.6 Hz, 1H), 5.78 (dd, J = 9.9, 1.6 Hz, 1H), 2.91–2.71 (m, 1H), 1.99–1.46 (m, 8H). 13C NMR (50 MHz, DMSO) δ 174.5, 163.3, 145.7, 139.5, 136.8, 135.6, 132.0, 129.7, 129.3, 126.9, 124.4, 121.7, 118.3, 117.3, 117.0, 116.7, 114.3, 45.1, 30.2, 25.8. TLC-MS (ESI) m/z: 397.0 [M + Na]+ HPLC tret = 7.38 min.
N-(3-(3-acrylamidophenyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)thiophene-2-carboxamide (12c): The preparation was carried out following General Procedure B starting from 46 mg 34c (0.09 mmol) in 4.2 mL DCM and 1.8 mL TFA. Flash purification (DCM/MeOH (4–10%)) afforded 15 mg (44%) of the final compound as a white solid. 1H NMR (200 MHz, DMSO-d6) δ 11.94 (s, 1H), 10.42 (s, 1H), 10.27 (s, 1H), 8.55 (s, 2H), 8.06 (d, J = 3.7 Hz, 1H), 7.97 (s, 1H), 7.87 (d, J = 5.1 Hz, 1H), 7.82 (s, 1H), 7.65 (dd, J = 5.8, 2.1 Hz, 1H), 7.41 (s, 1H), 7.39 (s, 1H), 7.25 (t, J = 4.4 Hz, 1H), 6.49 (dd, J = 16.9, 9.8 Hz, 1H), 6.27 (dd, J = 17.0, 1.8 Hz, 1H), 5.77 (dd, J = 9.8, 2.2 Hz, 1H). 13C NMR (50 MHz, DMSO-d6) δ 163.2, 160.1, 146.2, 139.9, 139.5, 138.0, 135.4, 131.9, 131.7, 129.3, 129.0, 128.7, 128.1, 126.9, 124.6, 121.6, 120.2, 117.2, 116.9, 116.7, 114.3. TLC-MS (ESI) m/z: 411.3 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C21H16N4O2S: 389.1067, found: 389.1072. HPLC tret = 6.44 min.
N-(3-(3-acrylamidophenyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)furan-2-carboxamide (12d): The preparation was carried out following General Procedure B starting from 58 mg 34d (0.12 mmol) in 4.2 mL DCM and 1.8 mL TFA. Flash purification (DCM/MeOH (4–10%)) afforded 23 mg (54%) of the final compound as a white solid. 1H NMR (200 MHz, DMSO-d6) δ 11.92 (s, 1H), 10.34 (s, 1H), 10.22 (s, 1H), 8.57 (s, 2H), 7.96 (s, 2H), 7.86–7.74 (m, 1H), 7.62 (d, J = 5.3 Hz, 1H), 7.50–7.33 (m, 2H), 7.33 (d, J = 3.5 Hz, 1H), 6.72 (dd, J = 3.2, 1.4 Hz, 1H), 6.48 (dd, J = 17.0, 9.8 Hz, 1H), 6.29 (dd, 1H), 5.77 (dd, J = 9.6, 2.0 Hz, 1H). 13C NMR (50 MHz, DMSO-d6) δ 163.2, 156.5, 147.6, 146.2, 145.6, 139.5, 138.0, 135.4, 131.9, 129.3, 128.4, 126.9, 124.5, 121.6, 120.2, 117.2, 116.9, 116.7, 114.6, 114.2, 112.1. TLC-MS (ESI) m/z: 395.2 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C21H16N4O3: 373.1295, found: 373.1300. HPLC tret = 5.88 min
N-(3-(3-acrylamidophenyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)benzamide (12e): The preparation was carried out following General Procedure B starting from 47 mg 34e (0.09 mmol) in 4.2 mL DCM and 1.8 mL TFA. Flash purification (DCM/MeOH (4–10%)) afforded 10 mg (29%) of the final compound as a white solid. 1H NMR (400 MHz, DMSO-d6) δ 11.92 (d, 1H), 10.39 (s, 1H), 10.23 (s, 1H), 8.62 (d, J = 2.3 Hz, 1H), 8.59 (d, J = 2.2 Hz, 1H), 8.03 (s, 1H), 8.01 (d, J = 1.6 Hz, 1H), 7.96 (s, 1H), 7.81 (d, J = 2.6 Hz, 1H), 7.66–7.61 (m, 1H), 7.60 (d, J = 6.7 Hz, 1H), 7.55 (t, J = 7.2 Hz, 2H), 7.41 (d, J = 6.5 Hz, 2H), 6.47 (dd, J = 16.9, 10.1 Hz, 1H), 6.27 (dd, J = 17.0, 2.1 Hz, 1H), 5.76 (dd, J = 10.0, 2.1 Hz, 1H). 13C NMR (101 MHz, DMSO-d6) δ 165.5, 163.2, 146.1, 139.5, 138.0, 135.4, 134.6, 131.9, 131.5, 129.2, 129.1, 128.3, 127.5, 126.7, 124.4, 121.6, 120.0, 117.2, 116.9, 116.6, 114.3. TLC-MS (ESI) m/z: 405.3 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C23H18N4O2: 383.1503, found: 383.1509. HPLC tret = 6.74 min.
N-(3-(5-acetamido-1H-pyrrolo[2,3-b]pyridin-3-yl)phenyl)acrylamide (12f): The preparation was carried out following General Procedure B starting from 90 mg 34f (0.20 mmol) in 4.2 mL DCM and 1.8 mL TFA. Flash purification (DCM/MeOH (10–16%)) afforded 26 mg (41%) of the final compound as a white solid. 1H NMR (200 MHz, DMSO-d6) δ 11.85 (s, 1H), 10.22 (s, 1H), 10.03 (s, 1H), 8.46 (s, 1H), 8.39 (s, 1H), 7.95 (s, 1H), 7.76 (s, 1H), 7.60 (d, J = 7.3 Hz, 1H), 7.38 (q, J = 7.7 Hz, 2H), 6.49 (dd, J = 16.9, 9.8 Hz, 1H), 6.28 (d, J = 16.5 Hz, 1H), 5.78 (d, J = 9.7 Hz, 1H), 2.08 (s, 3H). 13C NMR (50 MHz, DMSO-d6) δ 168.3, 163.2, 145.8, 139.5, 136.8, 135.5, 131.9, 129.4, 129.3, 126.9, 124.4, 121.6, 118.4, 117.2, 116.9, 116.7, 114.2, 23.7. TLC-MS (ESI) m/z: 343.1 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C18H16N4O2: 321.1346, found: 321.1352. HPLC tret = 4.74 min.
N-(3-(5-(2-cyclopentylacetamido)-1H-pyrrolo[2,3-b]pyridin-3-yl)phenyl)acrylamide (12g): The preparation was carried out following General Procedure B starting from 85 mg 34g (0.16 mmol) in 7 mL DCM and 3 mL TFA. Flash purification (DCM/MeOH (5–6%)) afforded 27 mg (42%) of the final compound as a white solid. 1H NMR (400 MHz, DMSO-d6) δ 11.84 (s, 1H), 10.22 (s, 1H), 9.94 (s, 1H), 8.49 (d, J = 2.2 Hz, 1H), 8.42 (d, J = 2.2 Hz, 1H), 7.96 (s, 1H), 7.76 (d, J = 2.4 Hz, 1H), 7.60 (d, J = 8.2 Hz, 1H), 7.41 (t, J = 7.8 Hz, 1H), 7.35 (d, J = 7.7 Hz, 1H), 6.48 (dd, J = 16.9, 10.1 Hz, 1H), 6.29 (dd, J = 17.0, 2.0 Hz, 1H), 5.78 (dd, J = 10.0, 2.0 Hz, 1H), 2.35 (s, 1H), 2.33 (s, 1H), 2.27 (dt, J = 15.1, 7.4 Hz, 1H), 1.78 (dq, J = 11.8, 6.2 Hz, 2H), 1.62 (pd, J = 9.0, 8.3, 5.0 Hz, 2H), 1.59–1.45 (m, 2H), 1.22 (dq, J = 11.5, 7.3 Hz, 2H). 13C NMR (101 MHz, DMSO-d6) δ 171.0, 163.3, 145.9, 139.6, 137.0, 135.7, 132.1, 129.6, 129.3, 126.9, 124.4, 121.8, 118.6, 117.5, 117.1, 116.8, 114.4, 42.5, 36.8, 32.1, 24.7. TLC-MS (ESI) m/z: 411.4 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C23H24N4O2: 389.1972, found: 389.1977. HPLC tret = 7.85 min.
N-(3-(5-(2-phenylacetamido)-1H-pyrrolo[2,3-b]pyridin-3-yl)phenyl)acrylamide (12h): The preparation was carried out following General Procedure B starting from 59 mg 34h (0.11 mmol) in 4.2 mL DCM and 1.8 mL TFA. Flash purification (DCM/MeOH (4–10%)) afforded 15 mg (34%) of the final compound as a white solid. 1H NMR (400 MHz, DMSO-d6) δ 11.86 (s, 1H), 10.28 (s, 1H), 10.22 (s, 1H), 8.51 (d, J = 2.3 Hz, 1H), 8.43 (d, J = 2.3 Hz, 1H), 7.93 (t, J = 1.9 Hz, 1H), 7.76 (d, J = 2.3 Hz, 1H), 7.60 (dt, J = 8.1, 1.6 Hz, 1H), 7.36 (ddt, J = 14.8, 12.3, 7.7 Hz, 6H), 7.28–7.22 (m, 1H), 6.47 (dd, J = 17.0, 10.1 Hz, 1H), 6.28 (dd, J = 17.0, 2.1 Hz, 1H), 5.77 (dd, J = 10.1, 2.1 Hz, 1H), 3.68 (s, 2H). 13C NMR (101 MHz, DMSO-d6) δ 169.0, 163.1, 145.8, 139.4, 136.6, 135.9, 135.4, 131.9, 129.4, 129.1, 129.1, 128.2, 126.7, 126.4, 124.3, 121.6, 118.3, 117.2, 116.9, 116.6, 114.2, 43.0. TLC-MS (ESI) m/z: 419.4 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C24H20N4O2: 397.1659, found: 397.1662. HPLC tret = 6.98 min.
N-(3-(5-(2-(thiophen-2-yl)acetamido)-1H-pyrrolo[2,3-b]pyridin-3-yl)phenyl)acrylamide (12i): The preparation was carried out following General Procedure B starting from 87 mg 34i (0.16 mmol) in 4.2 mL DCM and 1.8 mL TFA. Flash purification (DCM/MeOH (6–8%)) afforded 15 mg (34%) of the final compound as a white solid. 1H NMR (200 MHz, DMSO-d6) δ 11.88 (s, 1H), 10.33 (s, 1H), 10.23 (s, 1H), 8.49 (s, 1H), 8.43 (s, 1H), 7.94 (s, 1H), 7.77 (s, 1H), 7.61 (d, J = 6.8 Hz, 1H), 7.38 (d, J = 7.9 Hz, 3H), 7.01 (s, 2H), 6.48 (dd, 1H), 6.27 (d, J = 17.2 Hz, 1H), 5.78 (d, J = 9.6 Hz, 1H), 3.92 (s, 2H). 13C NMR (50 MHz, DMSO-d6) δ 168.1, 163.2, 145.9, 139.5, 137.1, 136.6, 135.4, 131.9, 129.2, 129.2, 126.9, 126.6, 126.3, 125.0, 124.5, 121.6, 118.4, 117.2, 116.9, 116.7, 114.2, 37.2. TLC-MS (ESI) m/z: 425.2 [M + Na]+. HRMS ESI-TOF [M + H]+ m/z calcd. for C22H18N4O2S: 403.1223, found: 403.1227. HPLC tret = 6.71 min.
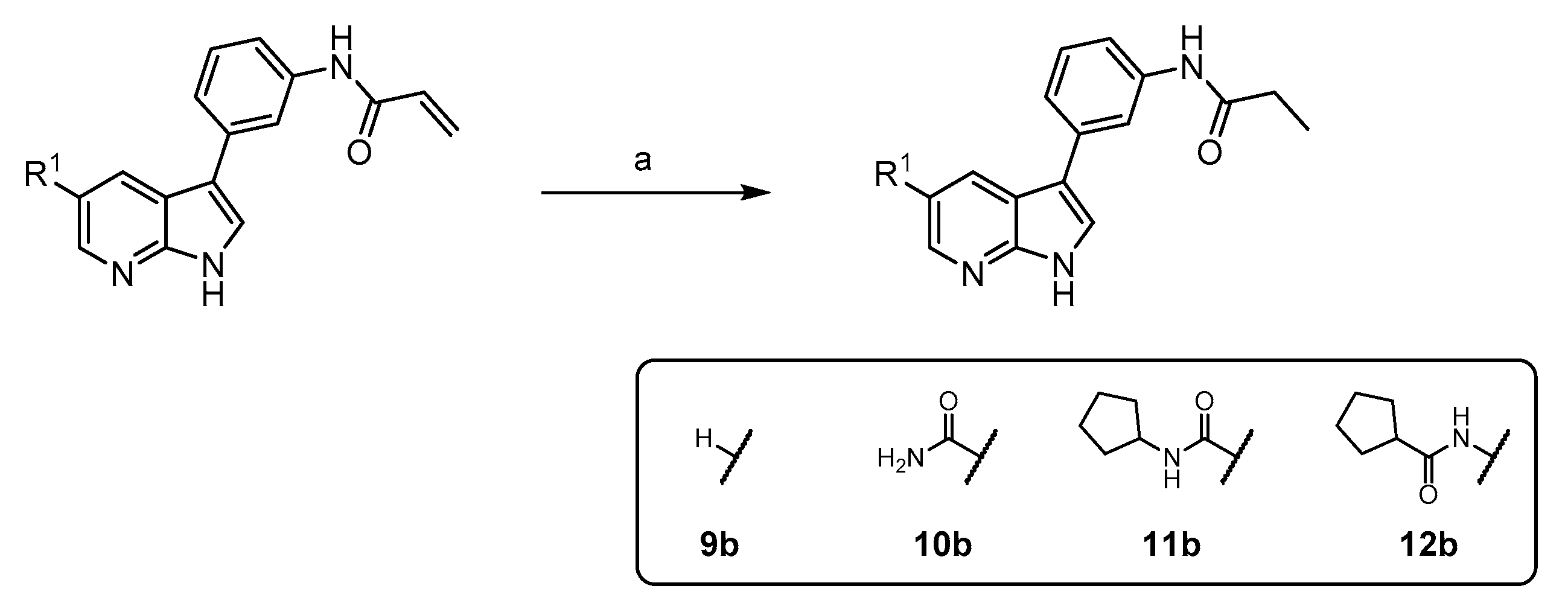
N-(3-(1H-pyrrolo[2,3-b]pyridin-3-yl)phenyl)propionamide (9b): To a solution of 17 mg 9a (0.07 mmol) in THF/MeOH (6 mL each) were added 8 mg Pd/C and the reaction was purged with hydrogen. The mixture was then stirred under hydrogen until HPLC indicated complete conversion. The catalyst was filtered off, the filtrate evaporated and the residue purified via flash chromatography (DCM/MeOH (4–8%)). Yield: 12 mg (69%) as a white solid. 1H NMR (200 MHz, DMSO) δ 11.89 (br s, 1H), 9.90 (s, 1H), 8.37–8.19 (m, 2H), 8.10–7.98 (m, 1H), 7.87–7.75 (m, 1H), 7.56–7.41 (m, 1H), 7.41–7.28 (m, 2H), 7.25–7.06 (m, 1H), 2.35 (q, J = 7.4 Hz, 2H), 1.11 (t, J = 7.4 Hz, 3H) 13C NMR (50 MHz, DMSO) δ 172.1, 149.1, 142.9, 139.8, 135.3, 129.1, 127.4, 123.6, 120.8, 117.2, 116.8, 116.3, 116.0, 114.2, 29.6, 9.7 TLC-MS (ESI) m/z: 266.4 [M + H]+ HPLC tret = 5.47 min.
3-(3-propionamidophenyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (10b): To a solution of 25 mg 10a (0.08 mmol) in THF/MeOH (5 mL each) were added 8 mg Pd/C and the reaction was purged with hydrogen. The mixture was then stirred under hydrogen until HPLC indicated complete conversion. The catalyst was filtered off, the filtrate evaporated and the residue purified via flash chromatography (DCM/MeOH (6–16%)). Yield: 15 mg (60%) as a white solid. 1H NMR (400 MHz, DMSO) δ 12.15 (s, 1H), 9.95 (s, 1H), 8.81 (s, J = 24.3 Hz, 1H), 8.75 (s, 1H), 8.10 (br s, 1H), 7.88 (d, J = 19.3 Hz, 2H), 7.61 (d, J = 7.2 Hz, 1H), 7.46–7.25 (m, 3H), 2.36 (q, J = 7.5 Hz, 2H), 1.11 (t, J = 7.5 Hz, 3H) 13C NMR (100 MHz, DMSO) δ 172.0, 167.7, 150.1, 143.2, 139.8, 134.8, 129.1, 127.2, 124.7, 122.6, 121.3, 117.1, 116.9, 116.4, 115.4, 29.5, 9.6 TLC-MS (ESI) m/z: 331.4 [M + Na]+ HPLC tret = 3.94 min.
N-cyclopentyl-3-(3-propionamidophenyl)-1H-pyrrolo[2,3-b]pyridine-5-carboxamide (11b): To a solution of 20 mg 11a (0.05 mmol) in THF/MeOH (2 mL each) were added 8 mg Pd/C and the reaction was purged with hydrogen. The mixture was then stirred under hydrogen until HPLC indicated complete conversion. The catalyst was filtered off, the filtrate evaporated and the residue purified via flash chromatography (DCM/MeOH (6–14%)). Yield: 19 mg (95%) as a white solid. 1H NMR (400 MHz, DMSO) δ 12.15 (br s, 1H), 9.98 (s, 1H), 8.76 (d, J = 1.6 Hz, 1H), 8.69 (d, J = 1.6 Hz, 1H), 8.39 (d, J = 7.2 Hz, 1H), 7.94 (s, 1H), 7.86 (d, J = 2.3 Hz, 1H), 7.62–7.55 (m, 1H), 7.43–7.32 (m, 2H), 4.33–4.21 (m, 1H), 2.36 (q, J = 7.5 Hz, 2H), 1.96–1.86 (m, 2H), 1.76–1.65 (m, 2H), 1.62–1.49 (m, 4H), 1.11 (t, J = 7.5 Hz, 3H). 13C NMR (101 MHz, DMSO) δ 172.0, 165.7, 149.9, 142.8, 139.8, 134.8, 129.1, 126.9, 124.7, 123.2, 121.2, 117.2, 116.8, 116.3, 115.4, 50.9, 32.1, 29.5, 23.6, 9.6. TLC-MS (ESI) m/z: 399.1 [M + Na]+ HPLC tret = 7.15 min.
N-(3-(3-propionamidophenyl)-1H-pyrrolo[2,3-b]pyridin-5-yl)cyclopentanecarboxamide (12b): To a solution of 18 mg 12a (0.05 mmol) in THF/MeOH (2 mL each) were added 5 mg Pd/C and the reaction was purged with hydrogen. The mixture was then stirred under hydrogen until HPLC indicated complete conversion. The catalyst was filtered off, the filtrate evaporated and the residue purified via flash chromatography (DCM/MeOH (3–10%)). Yield: 15 mg (83%) as a white solid. 1H NMR (200 MHz, DMSO) δ 11.82 (br s, 1H), 10.05–9.81 (m, 2H), 8.50 (d, J = 1.7 Hz, 1H), 8.41 (d, J = 1.7 Hz, 1H), 7.90–7.78 (m, 1H), 7.72 (d, J = 1.8 Hz, 1H), 7.63–7.46 (m, 1H), 7.44–7.20 (m, J = 7.7 Hz, 2H), 2.91–2.72 (m, 1H), 2.35 (q, J = 7.4 Hz, 2H), 1.97–1.50 (m, 8H), 1.10 (t, J = 7.4 Hz, 3H). TLC-MS (ESI) m/z: 399.2 [M + Na]+ HPLC tret = 7.40 min.