Leptin Mediated Pathways Stabilize Posttraumatic Insulin and Osteocalcin Patterns after Long Bone Fracture and Concomitant Traumatic Brain Injury and Thus Influence Fracture Healing in a Combined Murine Trauma Model
Abstract
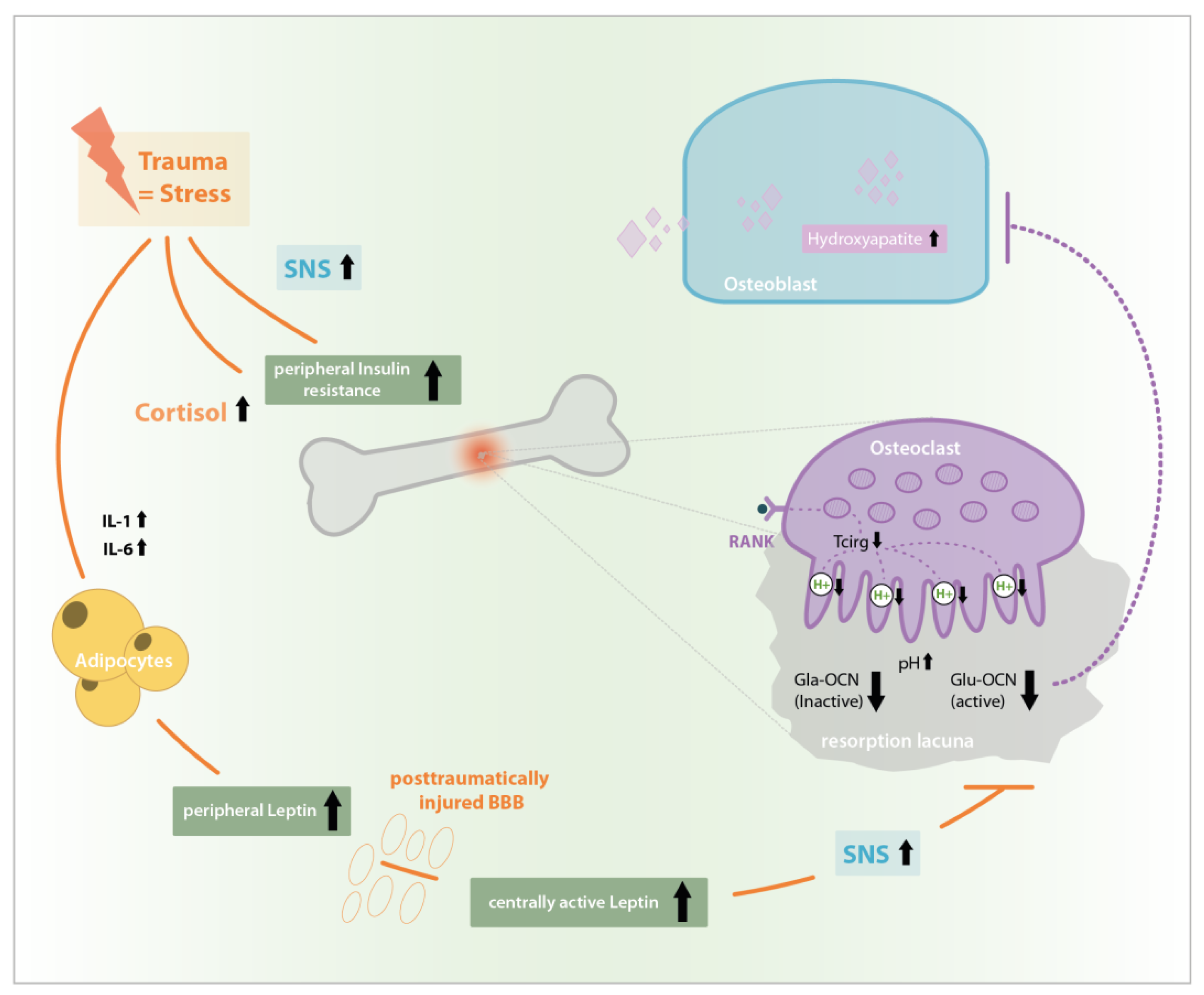
1. Introduction
2. Results
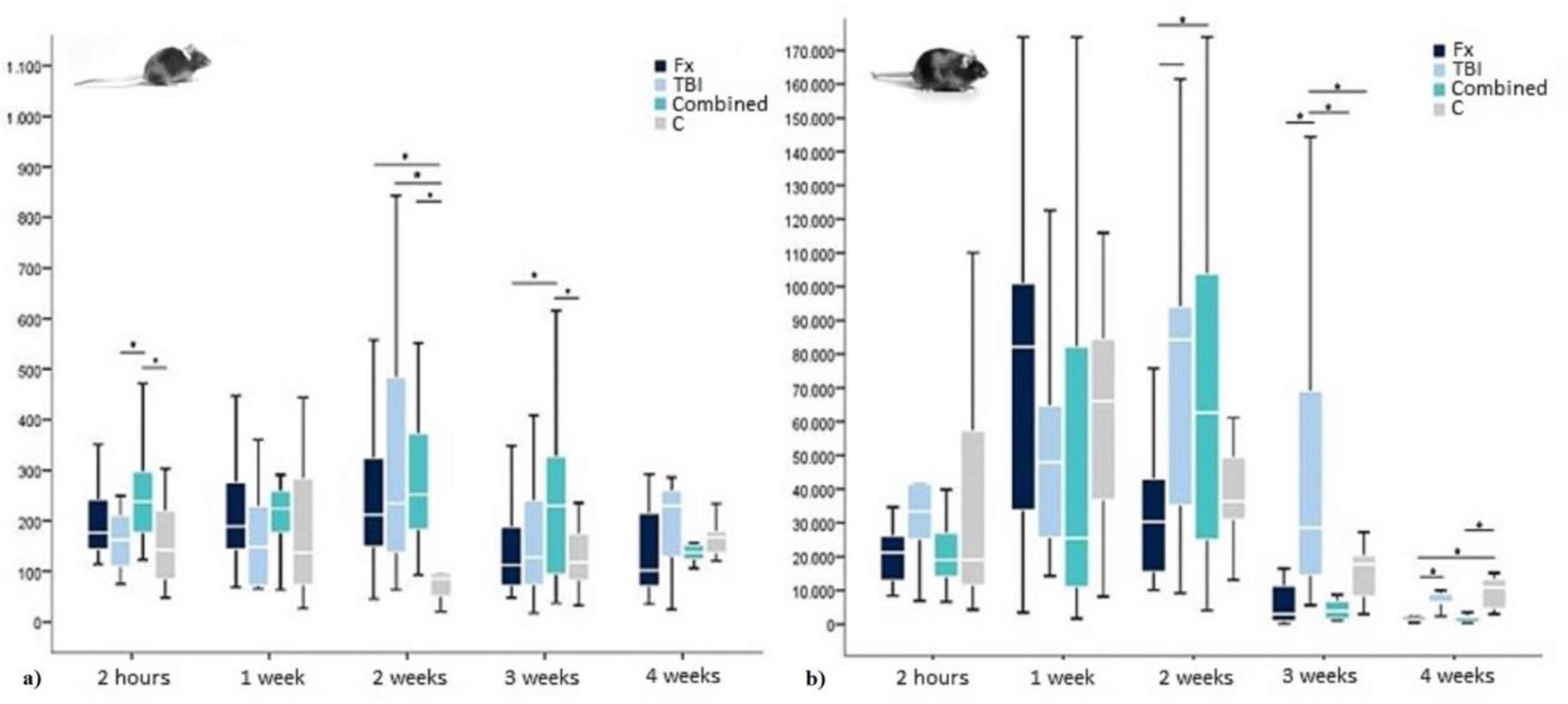
2.1. Insulin in WT-Mice
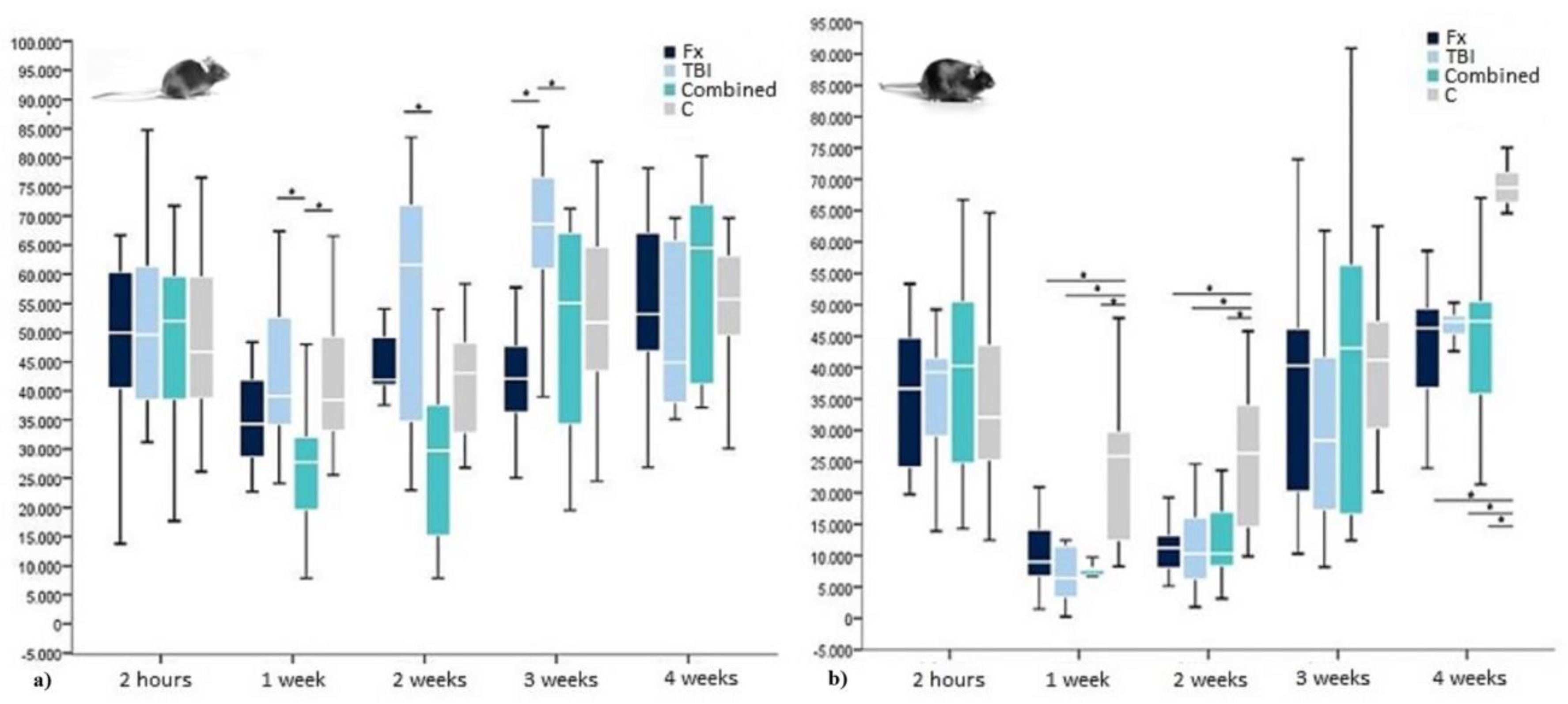
2.2. Osteocalcin in WT-Mice
2.3. Insulin in Leptin-Deficient Mice
2.4. OCN in Leptin-Deficient Mice
2.5. WT-Versus Leptin-Deficient Mice
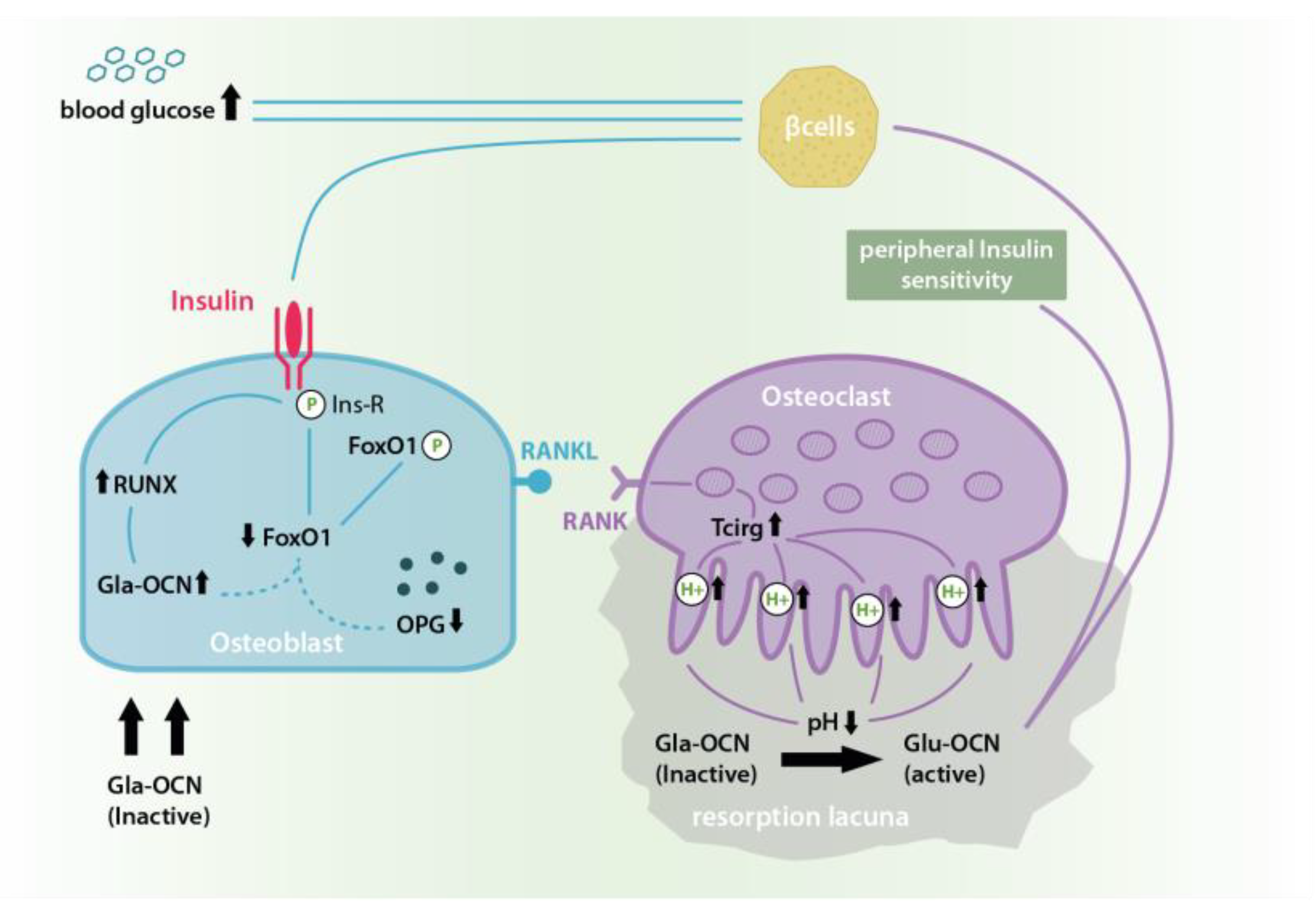
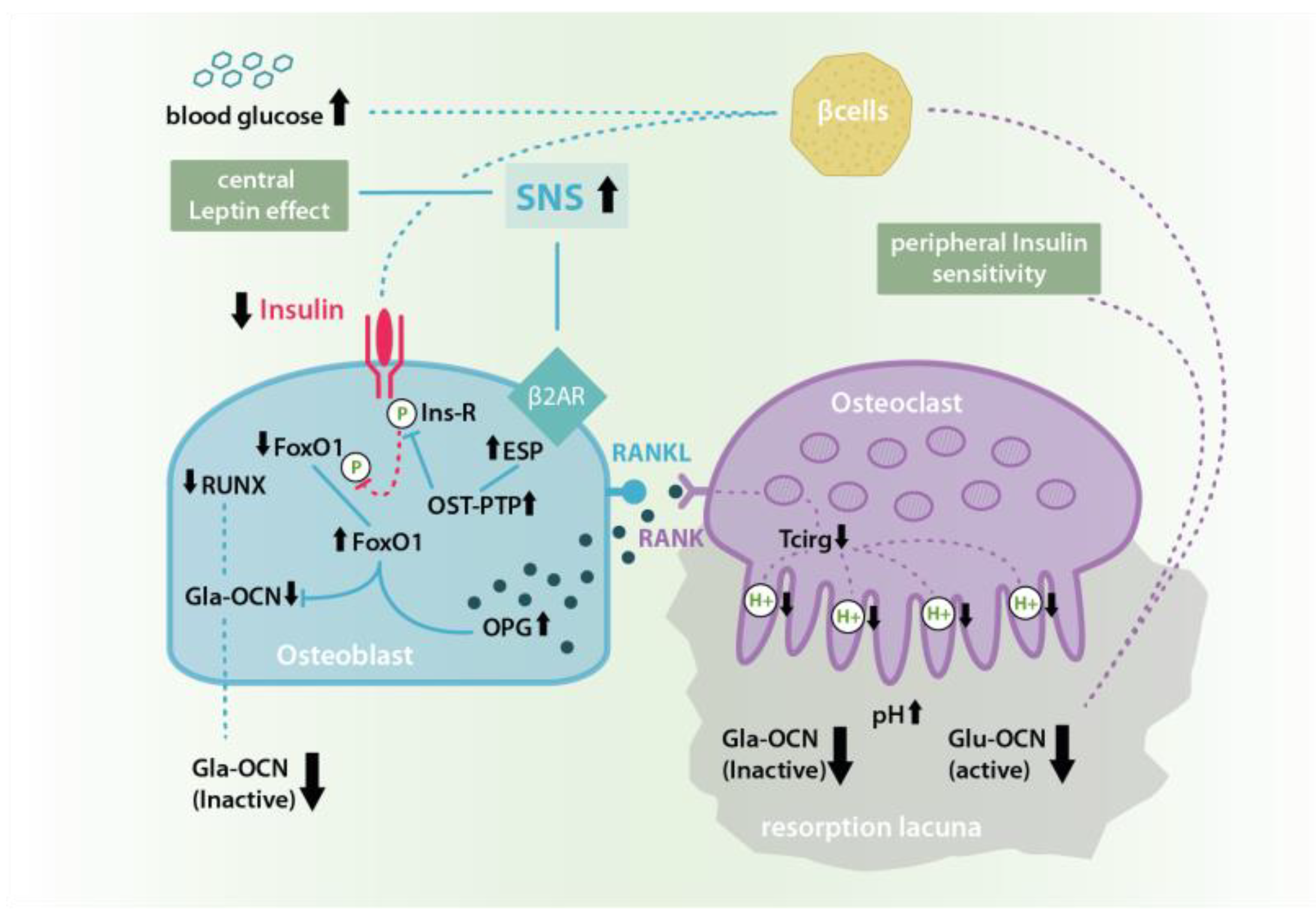
3. Discussion
4. Materials and Methods
4.1. Experimental Design
4.2. Animal Care and Perioperative Management
4.3. Surgical Procedures
4.4. Blood Sampling
4.5. Hormone Analysis
4.6. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| OCN | Osteocalcin |
| WT | Wild-Type |
| Fx | Fracture |
| TBI | Traumatic Brain Injury |
| Combined | Combined Trauma |
| C | Control |
| KO | Knock-Out |
| ECM | Extra Cellular Matrix |
| SNS | Sympathetic Nervous System |
| ESP | Embryonic-Stem-Cell-Phosphatase |
| OST-PTP | Osteoblast Testicular Tyrosinphosphatase |
| FoxO-1 | Forkhead Box Protein O-1 |
| OPG | Ostoeprotegrin |
| Runx 2 | Runt-Related Transcription Factor 2 |
| RANK | Receptor Activator of NF-κB |
| RANKL | Receptor Activator of NF-κB-Ligand |
| ANOVA | Analysis of Variance |
| pg | Picogram |
| mL | Milliliter |
| CT | Computer Tomography |
| CNS | Central Nervous System |
| BMD | Bone Mineral Density |
| BM | Bone Mass |
| BBB | Blood Brain Barrier |
| IL | Interleukin |
| CCII | Controlled Cortical Impact Injury |
| n | Number |
| ELISA | Enzyme Linked Immuno Sorbent Assay |
References
- Calandriello, B. Callus formation in severe brain injuries. Bull. Hosp. Jt. Dis. 1964, 25, 170–175. [Google Scholar]
- Gautschi, O.P.; Toffoli, A.M.; Joesbury, K.A.; Skirving, A.P.; Filgueira, L.; Zellweger, R. Osteoinductive effect of cerebrospinal fluid from brain-injured patients. J. Neurotrauma 2007, 24, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Gautschi, O.P.; Cadosch, D.; Frey, S.P.; Skirving, A.P.; Filgueira, L.; Zellweger, R. Serum-mediated osteogenic effect in traumatic brain-injured patients. ANZ J. Surg. 2009, 79, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Hofman, M.; Koopmans, G.; Kobbe, P.; Poeze, M.; Andruszkow, H.; Brink, P.R.; Pape, H.C. Improved fracture healing in patients with concomitant traumatic brain injury: Proven or not? Mediat. Inflamm. 2015, 2015, 1–14. [Google Scholar] [CrossRef]
- Amling, M.; Takeda, S.; Karsenty, G. A neuro (endo)crine regulation of bone remodeling. BioEssays News Rev. Mol. Cell. Dev. Biol. 2000, 22, 970–975. [Google Scholar] [CrossRef]
- Karsenty, G. Central control of bone formation. Adv. Nephrol. Necker Hosp. 2001, 31, 119–133. [Google Scholar]
- Ducy, P.; Amling, M.; Takeda, S.; Priemel, M.; Schilling, A.F.; Beil, F.T.; Shen, J.; Vinson, C.; Rueger, J.M.; Karsenty, G. Leptin inhibits bone formation through a hypothalamic relay: A central control of bone mass. Cell 2000, 100, 197–207. [Google Scholar] [CrossRef]
- Takeda, S.; Elefteriou, F.; Levasseur, R.; Liu, X.; Zhao, L.; Parker, K.L.; Armstrong, D.; Ducy, P.; Karsenty, G. Leptin regulates bone formation via the sympathetic nervous system. Cell 2002, 111, 305–317. [Google Scholar] [CrossRef]
- Hauschka, P.V.; Lian, J.B.; Gallop, P.M. Direct identification of the calcium-binding amino acid, gamma-carboxyglutamate, in mineralized tissue. Proc. Natl. Acad. Sci. USA 1975, 72, 3925–3929. [Google Scholar] [CrossRef]
- Confavreux, C.B. Bone: From a reservoir of minerals to a regulator of energy metabolism. Kidney Int. 2011, 79 (Suppl. 121), S14–S19. [Google Scholar] [CrossRef]
- Ducy, P. The role of osteocalcin in the endocrine cross-talk between bone remodelling and energy metabolism. Diabetologia 2011, 54, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Fulzele, K.; Riddle, R.C.; DiGirolamo, D.J.; Cao, X.; Wan, C.; Chen, D.; Faugere, M.C.; Aja, S.; Hussain, M.A.; Bruning, J.C.; et al. Insulin receptor signaling in osteoblasts regulates postnatal bone acquisition and body composition. Cell 2010, 142, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.K.; Karsenty, G. Reciprocal regulation of bone and energy metabolism. J. Musculoskelet. Neuronal Interact. 2008, 8, 351. [Google Scholar] [CrossRef] [PubMed]
- Ferron, M.; Wei, J.; Yoshizawa, T.; Del Fattore, A.; DePinho, R.A.; Teti, A.; Ducy, P.; Karsenty, G. Insulin signaling in osteoblasts integrates bone remodeling and energy metabolism. Cell 2010, 142, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, G.; Perego, S.; Luzi, L.; Banfi, G. A four-season molecule: Osteocalcin. Updates in its physiological roles. Endocrine 2015, 48, 394–404. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.K.; Sowa, H.; Hinoi, E.; Ferron, M.; Ahn, J.D.; Confavreux, C.; Dacquin, R.; Mee, P.J.; McKee, M.D.; Jung, D.Y.; et al. Endocrine regulation of energy metabolism by the skeleton. Cell 2007, 130, 456–469. [Google Scholar] [CrossRef]
- Ferron, M.; Hinoi, E.; Karsenty, G.; Ducy, P. Osteocalcin differentially regulates beta cell and adipocyte gene expression and affects the development of metabolic diseases in wild-type mice. Proc. Natl. Acad. Sci. USA 2008, 105, 5266–5270. [Google Scholar] [CrossRef]
- Hinoi, E.; Gao, N.; Jung, D.Y.; Yadav, V.; Yoshizawa, T.; Myers, M.G., Jr.; Chua, S.C., Jr.; Kim, J.K.; Kaestner, K.H.; Karsenty, G. The sympathetic tone mediates leptin’s inhibition of insulin secretion by modulating osteocalcin bioactivity. J. Cell Biol. 2008, 183, 1235–1242. [Google Scholar] [CrossRef]
- Hinoi, E.; Gao, N.; Jung, D.Y.; Yadav, V.; Yoshizawa, T.; Kajimura, D.; Myers, M.G., Jr.; Chua, S.C., Jr.; Wang, Q.; Kim, J.K.; et al. An Osteoblast-dependent mechanism contributes to the leptin regulation of insulin secretion. Ann. N. Y. Acad. Sci. 2009, 1173 (Suppl. 1), E20–E30. [Google Scholar] [CrossRef]
- Ferron, M.; Lacombe, J. Regulation of energy metabolism by the skeleton: Osteocalcin and beyond. Arch. Biochem. Biophys. 2014, 561, 137–146. [Google Scholar] [CrossRef]
- Takeda, S.; Karsenty, G. Central control of bone formation. J. Bone Miner. Metab. 2001, 19, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Yadav, V.K.; Karsenty, G. Leptin-dependent co-regulation of bone and energy metabolism. Aging 2009, 1, 954–956. [Google Scholar] [CrossRef]
- Karsenty, G.; Oury, F. Biology without walls: The novel endocrinology of bone. Annu. Rev. Physiol. 2012, 74, 87–105. [Google Scholar] [CrossRef] [PubMed]
- Karsenty, G.; Ferron, M. The contribution of bone to whole-organism physiology. Nature 2012, 481, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Ducy, P.; Desbois, C.; Boyce, B.; Pinero, G.; Story, B.; Dunstan, C.; Smith, E.; Bonadio, J.; Goldstein, S.; Gundberg, C.; et al. Increased bone formation in osteocalcin-deficient mice. Nature 1996, 382, 448–452. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Wang, L.; Clark, J.C.; Dass, C.R.; Choong, P.F. Elevated leptin expression in a rat model of fracture and traumatic brain injury. J. Pharm. Pharmacol. 2008, 60, 1667–1672. [Google Scholar] [CrossRef]
- Wang, L.; Tang, X.; Zhang, H.; Yuan, J.; Ding, H.; Wei, Y. Elevated leptin expression in rat model of traumatic spinal cord injury and femoral fracture. J. Spinal Cord Med. 2011, 34, 501–509. [Google Scholar] [CrossRef]
- Peters, A.; Schweiger, U.; Pellerin, L.; Hubold, C.; Oltmanns, K.M.; Conrad, M.; Schultes, B.; Born, J.; Fehm, H.L. The selfish brain: Competition for energy resources. Neurosci. Biobehav. Rev. 2004, 28, 143–180. [Google Scholar] [CrossRef]
- Peters, A. The selfish brain: Competition for energy resources. Am. J. Hum. Biol. Off. J. Hum. Biol. Counc. 2011, 23, 29–34. [Google Scholar] [CrossRef]
- Tsitsilonis, S.; Seemann, R.; Misch, M.; Wichlas, F.; Haas, N.P.; Schmidt-Bleek, K.; Kleber, C.; Schaser, K.D. The effect of traumatic brain injury on bone healing: An experimental study in a novel in vivo animal model. Injury 2015, 46, 661–665. [Google Scholar] [CrossRef]
- Locher, R.J.; Lunnemann, T.; Garbe, A.; Schaser, K.; Schmidt-Bleek, K.; Duda, G.; Tsitsilonis, S. Traumatic brain injury and bone healing: Radiographic and biomechanical analyses of bone formation and stability in a combined murine trauma model. J. Musculoskelet. Neuronal Interact. 2015, 15, 309–315. [Google Scholar] [PubMed]
- Graef, F.; Seemann, R.; Garbe, A.; Schmidt-Bleek, K.; Schaser, K.D.; Keller, J.; Duda, G.; Tsitsilonis, S. Impaired fracture healing with high non-union rates remains irreversible after traumatic brain injury in leptin-deficient mice. J. Musculoskelet. Neuronal Interact. 2017, 17, 78–85. [Google Scholar] [PubMed]
- Seemann, R.; Graef, F.; Garbe, A.; Keller, J.; Huang, F.; Duda, G.; Schmidt-Bleek, K.; Schaser, K.D.; Tsitsilonis, S. Leptin-deficiency eradicates the positive effect of traumatic brain injury on bone healing: Histological analyses in a combined trauma mouse model. J. Musculoskelet. Neuronal Interact. 2018, 18, 32–41. [Google Scholar] [PubMed]
- Kerimoglu, G.; Yulug, E.; Kerimoglu, S.; Citlak, A. Effects of leptin on fracture healing in rat tibia. Eklem Hastalik. Cerrahisi Jt. Dis. Relat. Surg. 2013, 24, 102–107. [Google Scholar] [CrossRef]
- Khan, S.N.; DuRaine, G.; Virk, S.S.; Fung, J.; Rowland, D.J.; Reddi, A.H.; Lee, M.A. The temporal role of leptin within fracture healing and the effect of local application of recombinant leptin on fracture healing. J. Orthop. Trauma 2013, 27, 656–662. [Google Scholar] [CrossRef]
- Hamrick, M.W.; Della-Fera, M.A.; Choi, Y.H.; Pennington, C.; Hartzell, D.; Baile, C.A. Leptin treatment induces loss of bone marrow adipocytes and increases bone formation in leptin-deficient ob/ob mice. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2005, 20, 994–1001. [Google Scholar] [CrossRef]
- Steppan, C.M.; Crawford, D.T.; Chidsey-Frink, K.L.; Ke, H.; Swick, A.G. Leptin is a potent stimulator of bone growth in ob/ob mice. Regul. Pept. 2000, 92, 73–78. [Google Scholar] [CrossRef]
- Turner, R.T.; Kalra, S.P.; Wong, C.P.; Philbrick, K.A.; Lindenmaier, L.B.; Boghossian, S.; Iwaniec, U.T. Peripheral leptin regulates bone formation. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2013, 28, 22–34. [Google Scholar] [CrossRef]
- Caro, J.F.; Kolaczynski, J.W.; Nyce, M.R.; Ohannesian, J.P.; Opentanova, I.; Goldman, W.H.; Lynn, R.B.; Zhang, P.L.; Sinha, M.K.; Considine, R.V. Decreased cerebrospinal-fluid/serum leptin ratio in obesity: A possible mechanism for leptin resistance. Lancet 1996, 348, 159–161. [Google Scholar] [CrossRef]
- Ghazali, A.; Grados, F.; Oprisiu, R.; Bunea, D.; Moriniere, P.; El Esper, N.; El Esper, I.; Brazier, M.; Souberbielle, J.C.; Fournier, A.; et al. Bone mineral density directly correlates with elevated serum leptin in haemodialysis patients. Nephrol. Dial. Transplant. 2003, 18, 1882–1890. [Google Scholar] [CrossRef]
- Histing, T.; Garcia, P.; Holstein, J.H.; Klein, M.; Matthys, R.; Nuetzi, R.; Steck, R.; Laschke, M.W.; Wehner, T.; Bindl, R.; et al. Small animal bone healing models: Standards, tips, and pitfalls results of a consensus meeting. Bone 2011, 49, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.M.; Kaluarachi, K.; Andrew, G.; Lu, W.; Chan, D.; Cheah, K.S. An externally fixed femoral fracture model for mice. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 2003, 21, 685–690. [Google Scholar] [CrossRef]
- Lighthall, J.W. Controlled cortical impact: A new experimental brain injury model. J. Neurotrauma 1988, 5, 1–15. [Google Scholar] [CrossRef] [PubMed]
| Fracture | TBI | Combined | Control | ||
|---|---|---|---|---|---|
| Insulin 2 Hours | Median in pg/mL | 177 | 162 | 237 | 142 |
| Interquartile Range in pg/mL | 106 | 115 | 137 | 146 | |
| Insulin 1 Week | Median in pg/mL | 189 | 148.5 | 225 | 138 |
| Interquartile Range in pg/mL | 138 | 159 | 84 | 240 | |
| Insulin 2 Weeks | Median in pg/mL | 211.5 | 234 | 252 | 85.5 |
| Interquartile Range in pg/mL | 176 | 423 | 210 | 71 | |
| Insulin 3 Weeks | Median in pg/mL | 113 | 127.5 | 229.5 | 117.5 |
| Interquartile Range in pg/mL | 124 | 171 | 241 | 98 | |
| Insulin 4 Weeks | Median in pg/mL | 102 | 229 | 138 | 167 |
| Interquartile Range in pg/mL | 180 | 153 | 33 | 47 |
| Fracture | TBI | Combined | Control | ||
|---|---|---|---|---|---|
| OCN 2 Hours | Median in pg/mL | 50,064 | 49,638 | 51,891 | 46,701 |
| Interquartile Range in pg/mL | 22,098 | 27,636 | 21,870 | 21,951 | |
| OCN 1 Week | Median in pg/mL | 34,263 | 39,078 | 27,711 | 38,490 |
| Interquartile Range in pg/mL | 14,703 | 24,588 | 14,965 | 16,536 | |
| OCN 2 Weeks | Median in pg/mL | 41,892 | 61,596 | 29,733 | 43,104 |
| Interquartile Rangein pg/mL | 10,686 | 44,613 | 23,728 | 16,818 | |
| OCN 3 Weeks | Median in pg/mL | 42,093 | 68,655 | 55,104 | 51,717 |
| Interquartile Range in pg/mL | 13,593 | 16,605 | 35,964 | 22,371 | |
| OCN 4 Weeks | Median in pg/mL | 53,106 | 44,862 | 64,470 | 55,719 |
| Interquartile Rangein pg/mL | 24,728 | 30,109 | 36,600 | 16,362 |
| Fracture | TBI | Combined | Control | ||
|---|---|---|---|---|---|
| Insulin 2 Hours | Median in pg/mL | 21,320 | 33,472 | 19,064 | 19,026 |
| Interquartile Range in pg/mL | 14,484 | 26,935 | 16,096 | 49,030 | |
| Insulin 1 Week | Median in pg/mL | 82,188 | 47,934 | 25,536 | 66,022 |
| Interquartile Range in pg/mL | 84,972 | 44,307 | 73,852 | 53,700 | |
| Insulin 2 Weeks | Median in pg/mL | 30,419 | 84,300 | 62,772 | 36,444 |
| Interquartile Range in pg/mL | 31,299 | 62,324 | 88,050 | 24,174 | |
| Insulin 3 Weeks | Median in pg/mL | 3058 | 28,572 | 3992 | 17,760 |
| Interquartile Range in pg/mL | 11,615.5 | 74,414 | 6104 | 13,398 | |
| Insulin 4 Weeks | Median in pg/mL | 1204 | 6396 | 2177.5 | 10,825 |
| Interquartile Range in pg/mL | 1309.5 | 3407 | 1683.5 | 10,025 |
| Fracture | TBI | Combined | Control | ||
|---|---|---|---|---|---|
| OCN 2 Hours | Median in pg/mL | 36,669 | 39,258 | 40,236 | 32,046 |
| Interquartile Range in pg/mL | 21,219 | 19,158 | 28,836 | 22,068 | |
| OCN 1 Week | Median in pg/mL | 9030 | 6416 | 7761 | 25,803 |
| Interquartile Range in pg/mL | 9051 | 8339 | 1284 | 17,713.5 | |
| OCN 2 Weeks | Median in pg/mL | 11,255 | 10,334 | 10,376 | 26,370 |
| Interquartile Range in pg/mL | 5901 | 10,270 | 8772 | 20,022 | |
| OCN 3 Weeks | Median in pg/mL | 40,365 | 28,419 | 43,098 | 41,238 |
| Interquartile Range in pg/mL | 30,585 | 24,532.5 | 42,006 | 18,667.5 | |
| OCN 4 Weeks | Median in pg/mL | 46,320 | 47,280 | 47,334 | 68,631 |
| Interquartile Range in pg/mL | 14,170 | 4149 | 24,642 | 5623.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garbe, A.; Graef, F.; Appelt, J.; Schmidt-Bleek, K.; Jahn, D.; Lünnemann, T.; Tsitsilonis, S.; Seemann, R. Leptin Mediated Pathways Stabilize Posttraumatic Insulin and Osteocalcin Patterns after Long Bone Fracture and Concomitant Traumatic Brain Injury and Thus Influence Fracture Healing in a Combined Murine Trauma Model. Int. J. Mol. Sci. 2020, 21, 9144. https://doi.org/10.3390/ijms21239144
Garbe A, Graef F, Appelt J, Schmidt-Bleek K, Jahn D, Lünnemann T, Tsitsilonis S, Seemann R. Leptin Mediated Pathways Stabilize Posttraumatic Insulin and Osteocalcin Patterns after Long Bone Fracture and Concomitant Traumatic Brain Injury and Thus Influence Fracture Healing in a Combined Murine Trauma Model. International Journal of Molecular Sciences. 2020; 21(23):9144. https://doi.org/10.3390/ijms21239144
Chicago/Turabian StyleGarbe, Anja, Frank Graef, Jessika Appelt, Katharina Schmidt-Bleek, Denise Jahn, Tim Lünnemann, Serafeim Tsitsilonis, and Ricarda Seemann. 2020. "Leptin Mediated Pathways Stabilize Posttraumatic Insulin and Osteocalcin Patterns after Long Bone Fracture and Concomitant Traumatic Brain Injury and Thus Influence Fracture Healing in a Combined Murine Trauma Model" International Journal of Molecular Sciences 21, no. 23: 9144. https://doi.org/10.3390/ijms21239144
APA StyleGarbe, A., Graef, F., Appelt, J., Schmidt-Bleek, K., Jahn, D., Lünnemann, T., Tsitsilonis, S., & Seemann, R. (2020). Leptin Mediated Pathways Stabilize Posttraumatic Insulin and Osteocalcin Patterns after Long Bone Fracture and Concomitant Traumatic Brain Injury and Thus Influence Fracture Healing in a Combined Murine Trauma Model. International Journal of Molecular Sciences, 21(23), 9144. https://doi.org/10.3390/ijms21239144






