In Vitro Cytological Responses against Laser Photobiomodulation for Periodontal Regeneration
Abstract
1. Introduction
2. Interaction with Tissues
3. Effects of Laser Irradiation on Osteoblasts
3.1. Diode Lasers
3.2. Nd:YAG Laser
3.3. Er:YAG Laser
3.4. Er,Cr:YSGG Laser
3.5. CO2 Laser
3.6. Summary
4. Effects of Laser Irradiation on Fibroblasts
4.1. Diode Laser
4.2. Nd:YAG Laser
4.3. Er:YAG and Er,Cr:YSGG Lasers
4.4. CO2 Laser
4.5. Summary
5. Effects of Laser Irradiation on Periodontal Ligament Cells
5.1. Diode Laser
5.2. Er:YAG Laser
5.3. Nd:YAG Laser, Er,Cr:YSGG Laser, and CO2 Laser
5.4. Summary
6. Effects of Laser Irradiation on Endothelial Cells
6.1. Diode Laser
6.2. Nd:YAG Laser
6.3. Er:YAG, Er,Cr:YSGG, and CO2 Lasers
6.4. Summary
7. Effects of Laser Irradiation on Cementoblasts
8. Effects of Laser Irradiation on Epithelial Cells
8.1. Diode Laser
8.2. Summary
9. Effects of Laser Irradiation on Osteocytes
10. Effects of Laser Irradiation on Osteoclasts
11. Effects of Laser Irradiation on Stem Cells
11.1. Diode Laser
11.2. Nd:YAG Laser
11.3. CO2 Laser
11.4. Summary
12. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lang, N.P.; Lindhe, J. Clinical Periodontology and Implant Dentistry; John Wiley & Sons: Hoboken, NJ, USA, 2015; Volume 2. [Google Scholar]
- Kinane, D.F.; Stathopoulou, P.G.; Papapanou, P.N. Periodontal diseases. Nat. Rev. Dis. Primers 2017, 3, 17038. [Google Scholar] [CrossRef] [PubMed]
- Suvan, J.; Leira, Y.; Moreno Sancho, F.M.; Graziani, F.; Derks, J.; Tomasi, C. Subgingival instrumentation for treatment of periodontitis. A systematic review. J. Clin. Periodontol. 2020, 47 (Suppl. 22), 155–175. [Google Scholar] [CrossRef] [PubMed]
- Aoki, A.; Mizutani, K.; Schwarz, F.; Sculean, A.; Yukna, R.A.; Takasaki, A.A.; Romanos, G.E.; Taniguchi, Y.; Sasaki, K.M.; Zeredo, J.L.; et al. Periodontal and peri-implant wound healing following laser therapy. Periodontoloy 2000 2015, 68, 217–269. [Google Scholar] [CrossRef] [PubMed]
- Mizutani, K.; Aoki, A.; Coluzzi, D.; Yukna, R.; Wang, C.Y.; Pavlic, V.; Izumi, Y. Lasers in minimally invasive periodontal and peri-implant therapy. Periodontoloy 2000 2016, 71, 185–212. [Google Scholar] [CrossRef] [PubMed]
- Gholami, L.; Asefi, S.; Hooshyarfard, A.; Sculean, A.; Romanos, G.E.; Aoki, A.; Fekrazad, R. Photobiomodulation in Periodontology and Implant Dentistry: Part 1. Photobiomodul. Photomed. Laser Surg. 2019, 37, 739–765. [Google Scholar] [CrossRef]
- Ruh, A.C.; Frigo, L.; Cavalcanti, M.; Svidnicki, P.; Vicari, V.N.; Lopes-Martins, R.A.B.; Leal Junior, E.C.P.; De Isla, N.; Diomede, F.; Trubiani, O.; et al. Laser photobiomodulation in pressure ulcer healing of human diabetic patients: Gene expression analysis of inflammatory biochemical markers. Lasers Med. Sci. 2018, 33, 165–171. [Google Scholar] [CrossRef]
- Cavalcanti, M.F.; Silva, U.H.; Leal-Junior, E.C.; Lopes-Martins, R.A.; Marcos, R.L.; Pallotta, R.C.; Diomede, F.; Trubiani, O.; De Isla, N.; Frigo, L. Comparative Study of the Physiotherapeutic and Drug Protocol and Low-Level Laser Irradiation in the Treatment of Pain Associated with Temporomandibular Dysfunction. Photomed. Laser Surg. 2016, 34, 652–656. [Google Scholar] [CrossRef]
- Heiskanen, V.; Hamblin, M.R. Photobiomodulation: Lasers vs. light emitting diodes? Photochem. Photobiol. Sci. 2018, 17, 1003–1017. [Google Scholar] [CrossRef]
- Anders, J.J.; Lanzafame, R.J.; Arany, P.R. Low-level light/laser therapy versus photobiomodulation therapy. Photomed. Laser Surg. 2015, 33, 183–184. [Google Scholar] [CrossRef]
- Hale, G.M.; Querry, M.R. Optical constants of water in the 200-nm to 200-µm wavelength region. Appl. Opt. 1973, 12, 555–563. [Google Scholar] [CrossRef]
- Niemz, M.H. Laser-Tissue Interaction. In Fundamentals and Applications; Springer: Berlin, Germany, 1996. [Google Scholar]
- Aoki, A.; Sasaki, K.; Watanabe, H.; Ishikawa, I. Lasers in non-surgical periodontal therapy. Periodontology 2000 2004, 36, 59–97. [Google Scholar] [CrossRef] [PubMed]
- Hadjidakis, D.J.; Androulakis, I.I. Bone remodeling. Ann. N. Y. Acad. Sci. 2006, 1092, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Tamura, K.; Hiratsuka, K.; Abiko, Y. Stimulation of MCM3 gene expression in osteoblast by low level laser irradiation. Lasers Med. Sci. 2001, 16, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Hamajima, S.; Hiratsuka, K.; Kiyama-Kishikawa, M.; Tagawa, T.; Kawahara, M.; Ohta, M.; Sasahara, H.; Abiko, Y. Effect of low-level laser irradiation on osteoglycin gene expression in osteoblasts. Lasers Med. Sci. 2003, 18, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Renno, A.C.; McDonnell, P.A.; Parizotto, N.A.; Laakso, E.L. The effects of laser irradiation on osteoblast and osteosarcoma cell proliferation and differentiation in vitro. Photomed. Laser Surg. 2007, 25, 275–280. [Google Scholar] [CrossRef]
- Fujimoto, K.; Kiyosaki, T.; Mitsui, N.; Mayahara, K.; Omasa, S.; Suzuki, N.; Shimizu, N. Low-intensity laser irradiation stimulates mineralization via increased BMPs in MC3T3-E1 cells. Lasers Surg. Med. 2010, 42, 519–526. [Google Scholar] [CrossRef]
- Kanenari, M.; Zhao, J.; Abiko, Y. Enhancement of microtubule-associated protein-1 Alpha gene expression in osteoblasts by low level laser irradiation. Laser 2011, 20, 47–51. [Google Scholar] [CrossRef][Green Version]
- Migliario, M.; Pittarella, P.; Fanuli, M.; Rizzi, M.; Reno, F. Laser-induced osteoblast proliferation is mediated by ROS production. Lasers Med. Sci. 2014, 29, 1463–1467. [Google Scholar] [CrossRef]
- Pagin, M.T.; de Oliveira, F.A.; Oliveira, R.C.; Sant’Ana, A.C.; de Rezende, M.L.; Greghi, S.L.; Damante, C.A. Laser and light-emitting diode effects on pre-osteoblast growth and differentiation. Lasers Med. Sci. 2014, 29, 55–59. [Google Scholar] [CrossRef]
- Oliveira, F.A.; Matos, A.A.; Matsuda, S.S.; Buzalaf, M.A.; Bagnato, V.S.; Machado, M.A.; Damante, C.A.; Oliveira, R.C.; Peres-Buzalaf, C. Low level laser therapy modulates viability, alkaline phosphatase and matrix metalloproteinase-2 activities of osteoblasts. J. Photochem. Photobiol. B 2017, 169, 35–40. [Google Scholar] [CrossRef]
- Son, J.H.; Park, B.S.; Kim, I.R.; Sung, I.Y.; Cho, Y.C.; Kim, J.S.; Kim, Y.D. A novel combination treatment to stimulate bone healing and regeneration under hypoxic conditions: Photobiomodulation and melatonin. Lasers Med. Sci. 2017, 32, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Chen, Y.; Dong, S.; Liu, S.; Zhang, X.; Si, X.; Zhou, Y. Laser irradiation promotes the proliferation of mouse pre-osteoblast cell line MC3T3-E1 through hedgehog signaling pathway. Lasers Med. Sci. 2017, 32, 1489–1496. [Google Scholar] [CrossRef] [PubMed]
- Kunimatsu, R.; Gunji, H.; Tsuka, Y.; Yoshimi, Y.; Awada, T.; Sumi, K.; Nakajima, K.; Kimura, A.; Hiraki, T.; Abe, T.; et al. Effects of high-frequency near-infrared diode laser irradiation on the proliferation and migration of mouse calvarial osteoblasts. Lasers Med. Sci. 2018, 33, 959–966. [Google Scholar] [CrossRef] [PubMed]
- Mikami, R.; Mizutani, K.; Aoki, A.; Tamura, Y.; Aoki, K.; Izumi, Y. Low-level ultrahigh-frequency and ultrashort-pulse blue laser irradiation enhances osteoblast extracellular calcification by upregulating proliferation and differentiation via transient receptor potential vanilloid 1. Lasers Surg. Med. 2018, 50, 340–352. [Google Scholar] [CrossRef]
- Coombe, A.R.; Ho, C.T.; Darendeliler, M.A.; Hunter, N.; Philips, J.R.; Chapple, C.C.; Yum, L.W. The effects of low level laser irradiation on osteoblastic cells. Clin. Orthod. Res. 2001, 4, 3–14. [Google Scholar] [CrossRef]
- Bayram, H.; Kenar, H.; Tasar, F.; Hasirci, V. Effect of low level laser therapy and zoledronate on the viability and ALP activity of Saos-2 cells. Int. J. Oral. Maxillofac. Surg. 2013, 42, 140–146. [Google Scholar] [CrossRef]
- Bloise, N.; Ceccarelli, G.; Minzioni, P.; Vercellino, M.; Benedetti, L.; De Angelis, M.G.; Imbriani, M.; Visai, L. Investigation of low-level laser therapy potentiality on proliferation and differentiation of human osteoblast-like cells in the absence/presence of osteogenic factors. J. Biomed. Opt. 2013, 18, 128006. [Google Scholar] [CrossRef]
- Incerti Parenti, S.; Checchi, L.; Fini, M.; Tschon, M. Different doses of low-level laser irradiation modulate the in vitro response of osteoblast-like cells. J. Biomed. Opt. 2014, 19, 108002. [Google Scholar] [CrossRef]
- Tschon, M.; Incerti-Parenti, S.; Cepollaro, S.; Checchi, L.; Fini, M. Photobiomodulation with low-level diode laser promotes osteoblast migration in an in vitro micro wound model. J. Biomed. Opt. 2015, 20, 78002. [Google Scholar] [CrossRef]
- Heymann, P.G.; Ziebart, T.; Kammerer, P.W.; Mandic, R.; Saydali, A.; Braun, A.; Neff, A.; Draenert, G.F. The enhancing effect of a laser photochemotherapy with cisplatin or zolendronic acid in primary human osteoblasts and osteosarcoma cells in vitro. J. Oral. Pathol. Med. 2016, 45, 803–809. [Google Scholar] [CrossRef]
- Tani, A.; Chellini, F.; Giannelli, M.; Nosi, D.; Zecchi-Orlandini, S.; Sassoli, C. Red (635 nm), Near-Infrared (808 nm) and Violet-Blue (405 nm) Photobiomodulation Potentiality on Human Osteoblasts and Mesenchymal Stromal Cells: A Morphological and Molecular in vitro Study. Int. J. Mol. Sci. 2018, 19, 1946. [Google Scholar] [CrossRef] [PubMed]
- Stein, E.; Koehn, J.; Sutter, W.; Wendtlandt, G.; Wanschitz, F.; Thurnher, D.; Baghestanian, M.; Turhani, D. Initial effects of low-level laser therapy on growth and differentiation of human osteoblast-like cells. Wien Klin Wochenschr 2008, 120, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Saracino, S.; Mozzati, M.; Martinasso, G.; Pol, R.; Canuto, R.A.; Muzio, G. Superpulsed laser irradiation increases osteoblast activity via modulation of bone morphogenetic factors. Lasers Surg. Med. 2009, 41, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.H.; Lu, Y.C.; Kao, C.T. Low-level diode laser therapy reduces lipopolysaccharide (LPS)-induced bone cell inflammation. Lasers Med. Sci. 2012, 27, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Huertas, R.M.; Luna-Bertos, E.D.; Ramos-Torrecillas, J.; Leyva, F.M.; Ruiz, C.; Garcia-Martinez, O. Effect and clinical implications of the low-energy diode laser on bone cell proliferation. Biol. Res. Nurs. 2014, 16, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Incerti Parenti, S.; Panseri, S.; Gracco, A.; Sandri, M.; Tampieri, A.; Alessandri Bonetti, G. Effect of low-level laser irradiation on osteoblast-like cells cultured on porous hydroxyapatite scaffolds. Ann. Ist. Super Sanita 2013, 49, 255–260. [Google Scholar] [PubMed]
- Medina-Huertas, R.; Manzano-Moreno, F.J.; De Luna-Bertos, E.; Ramos-Torrecillas, J.; Garcia-Martinez, O.; Ruiz, C. The effects of low-level diode laser irradiation on differentiation, antigenic profile, and phagocytic capacity of osteoblast-like cells (MG-63). Lasers Med. Sci. 2014, 29, 1479–1484. [Google Scholar] [CrossRef] [PubMed]
- Manzano-Moreno, F.J.; Medina-Huertas, R.; Ramos-Torrecillas, J.; Garcia-Martinez, O.; Ruiz, C. The effect of low-level diode laser therapy on early differentiation of osteoblast via BMP-2/TGF-beta1 and its receptors. J. Craniomaxillofac. Surg. 2015, 43, 1926–1932. [Google Scholar] [CrossRef]
- Pyo, S.J.; Song, W.W.; Kim, I.R.; Park, B.S.; Kim, C.H.; Shin, S.H.; Chung, I.K.; Kim, Y.D. Low-level laser therapy induces the expressions of BMP-2, osteocalcin, and TGF-beta1 in hypoxic-cultured human osteoblasts. Lasers Med. Sci. 2013, 28, 543–550. [Google Scholar] [CrossRef]
- Jawad, M.M.; Husein, A.; Azlina, A.; Alam, M.K.; Hassan, R.; Shaari, R. Effect of 940 nm low-level laser therapy on osteogenesis in vitro. J. Biomed. Opt. 2013, 18, 128001. [Google Scholar] [CrossRef]
- Walter, C.; Pabst, A.M.; Ziebart, T. Effects of a low-level diode laser on oral keratinocytes, oral fibroblasts, endothelial cells and osteoblasts incubated with bisphosphonates: An in vitro study. Biomed. Rep. 2015, 3, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.H.; Kim, K.H.; Choi, N.R.; Kim, I.R.; Park, B.S.; Kim, Y.D.; Kim, U.K.; Kim, C.H. Effect of low-level laser therapy on bisphosphonate-treated osteoblasts. Maxillofac. Plast Reconstr. Surg. 2016, 38, 48. [Google Scholar] [CrossRef] [PubMed]
- Bolukbasi Ates, G.; Ak Can, A.; Gulsoy, M. Investigation of photobiomodulation potentiality by 635 and 809 nm lasers on human osteoblasts. Lasers Med. Sci. 2017, 32, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Ates, G.B.; Ak, A.; Garipcan, B.; Gulsoy, M. Indocyanine green-mediated photobiomodulation on human osteoblast cells. Lasers Med. Sci. 2018, 33, 1591–1599. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.Y.; Hong, J.U.; Song, J.M.; Kim, I.R.; Park, B.S.; Kim, C.H.; Shin, S.H. Combined effect of recombinant human bone morphogenetic protein-2 and low level laser irradiation on bisphosphonate-treated osteoblasts. J. Korean Assoc. Oral. Maxillofac. Surg. 2018, 44, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Fujihara, N.A.; Hiraki, K.R.; Marques, M.M. Irradiation at 780 nm increases proliferation rate of osteoblasts independently of dexamethasone presence. Lasers Surg. Med. 2006, 38, 332–336. [Google Scholar] [CrossRef] [PubMed]
- Fukuhara, E.; Goto, T.; Matayoshi, T.; Kobayashi, S.; Takahashi, T. Optimal low-energy laser irradiation causes temporal G2/M arrest on rat calvarial osteoblasts. Calcif. Tissue Int. 2006, 79, 443–450. [Google Scholar] [CrossRef]
- Shimizu, N.; Mayahara, K.; Kiyosaki, T.; Yamaguchi, A.; Ozawa, Y.; Abiko, Y. Low-intensity laser irradiation stimulates bone nodule formation via insulin-like growth factor-I expression in rat calvarial cells. Lasers Surg. Med. 2007, 39, 551–559. [Google Scholar] [CrossRef]
- Xu, M.; Deng, T.; Mo, F.; Deng, B.; Lam, W.; Deng, P.; Zhang, X.; Liu, S. Low-intensity pulsed laser irradiation affects RANKL and OPG mRNA expression in rat calvarial cells. Photomed. Laser Surg. 2009, 27, 309–315. [Google Scholar] [CrossRef]
- Ozawa, Y.; Shimizu, N.; Kariya, G.; Abiko, Y. Low-energy laser irradiation stimulates bone nodule formation at early stages of cell culture in rat calvarial cells. Bone 1998, 22, 347–354. [Google Scholar] [CrossRef]
- Ueda, Y.; Shimizu, N. Pulse irradiation of low-power laser stimulates bone nodule formation. J. Oral. Sci. 2001, 43, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Ueda, Y.; Shimizu, N. Effects of pulse frequency of low-level laser therapy (LLLT) on bone nodule formation in rat calvarial cells. J. Clin. Laser Med. Surg. 2003, 21, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Khadra, M.; Lyngstadaas, S.P.; Haanaes, H.R.; Mustafa, K. Effect of laser therapy on attachment, proliferation and differentiation of human osteoblast-like cells cultured on titanium implant material. Biomaterials 2005, 26, 3503–3509. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, M.V.; do Vale Placa, R.; Sant’Ana, A.C.P.; Greghi, S.L.A.; Zangrando, M.S.R.; de Rezende, M.L.R.; Oliveira, R.C.; Damante, C.A. Laser and LED photobiomodulation effects in osteogenic or regular medium on rat calvaria osteoblasts obtained by newly forming bone technique. Lasers Med. Sci. 2020. [Google Scholar] [CrossRef]
- Petri, A.D.; Teixeira, L.N.; Crippa, G.E.; Beloti, M.M.; de Oliveira, P.T.; Rosa, A.L. Effects of low-level laser therapy on human osteoblastic cells grown on titanium. Braz. Dent J. 2010, 21, 491–498. [Google Scholar] [CrossRef]
- Emes, Y.A.K.; Aybar, B. Low-level laser therapy vs. pulsed electromagnetic field on neonatal rat calvarial osteoblast-like cells. Lasers Med. Sci. 2013, 28, 901–909. [Google Scholar] [CrossRef]
- Morsoleto, M.; Sella, V.; Machado, P.; Bomfim, F.D.; Fernandes, M.H.; Morgado, F.; Lopes Filho, G.J.; Plapler, H. Effect of low power laser in biomodulation of cultured osteoblastic cells of Wistar rats1. Acta Cir. Bras 2019, 34, e201900210. [Google Scholar] [CrossRef]
- Pires Oliveira, D.A.; de Oliveira, R.F.; Zangaro, R.A.; Soares, C.P. Evaluation of low-level laser therapy of osteoblastic cells. Photomed Laser Surg. 2008, 26, 401–404. [Google Scholar] [CrossRef]
- Dortbudak, O.; Haas, R.; Mallath-Pokorny, G. Biostimulation of bone marrow cells with a diode soft laser. Clin. Oral. Implant. Res. 2000, 11, 540–545. [Google Scholar] [CrossRef]
- Grassi, F.R.; Ciccolella, F.; D’Apolito, G.; Papa, F.; Iuso, A.; Salzo, A.E.; Trentadue, R.; Nardi, G.M.; Scivetti, M.; De Matteo, M.; et al. Effect of low-level laser irradiation on osteoblast proliferation and bone formation. J. Biol. Regul. Homeost. Agents 2011, 25, 603–614. [Google Scholar]
- Mergoni, G.; Vescovi, P.; Belletti, S.; Uggeri, J.; Nammour, S.; Gatti, R. Effects of 915 nm laser irradiation on human osteoblasts: A preliminary in vitro study. Lasers Med. Sci. 2018, 33, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Hirata, S.; Kitamura, C.; Fukushima, H.; Nakamichi, I.; Abiko, Y.; Terashita, M.; Jimi, E. Low-level laser irradiation enhances BMP-induced osteoblast differentiation by stimulating the BMP/Smad signaling pathway. J. Cell Biochem. 2010, 111, 1445–1452. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, T.; Nomura, S.; Yamaguchi, A.; Suda, T.; Yoshiki, S. In situ hybridization of bone matrix proteins in undecalcified adult rat bone sections. J. Histochem. Cytochem. 1992, 40, 1079–1088. [Google Scholar] [CrossRef] [PubMed]
- Komori, T. Molecular Mechanism of Runx2-Dependent Bone Development. Mol. Cells 2020, 43, 168–175. [Google Scholar]
- Nakashima, K.; Zhou, X.; Kunkel, G.; Zhang, Z.; Deng, J.M.; Behringer, R.R.; de Crombrugghe, B. The Novel Zinc Finger-Containing Transcription Factor Osterix Is Required for Osteoblast Differentiation and Bone Formation. Cell 2002, 108, 17–29. [Google Scholar] [CrossRef]
- Miyazono, K.; Kamiya, Y.; Morikawa, M. Bone morphogenetic protein receptors and signal transduction. J. Biochem. 2010, 147, 35–51. [Google Scholar] [CrossRef]
- Heldin, C.H.; Miyazono, K.; ten Dijke, P. TGF-beta signalling from cell membrane to nucleus through SMAD proteins. Nature 1997, 390, 465–471. [Google Scholar] [CrossRef]
- Fujihara, S.; Yokozeki, M.; Oba, Y.; Higashibata, Y.; Nomura, S.; Moriyama, K. Function and regulation of osteopontin in response to mechanical stress. J. Bone Min. Res. 2006, 21, 956–964. [Google Scholar] [CrossRef]
- Miyazono, K.; Kusanagi, K.; Inoue, H. Divergence and convergence of TGF-beta/BMP signaling. J. Cell Physiol. 2001, 187, 265–276. [Google Scholar] [CrossRef]
- Ryoo, H.M.; Hoffmann, H.M.; Beumer, T.; Frenkel, B.; Towler, D.A.; Stein, G.S.; Stein, J.L.; van Wijnen, A.J.; Lian, J.B. Stage-specific expression of Dlx5 during osteoblast differentiation: Involvement in regulation of osteocalcin gene expression. Mol. Endocrinol. 1997, 11, 1681–1694. [Google Scholar] [CrossRef]
- Deckx, S.; Heymans, S.; Papageorgiou, A.P. The diverse functions of osteoglycin: A deceitful dwarf, or a master regulator of disease? Faseb J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2016, 30, 2651–2661. [Google Scholar] [CrossRef] [PubMed]
- Starup-Linde, J.; Viggers, R.; Handberg, A. Osteoglycin and Bone-a Systematic Review. Curr. Osteoporos. Rep. 2019, 17, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Arisu, H.D.; Turkoz, E.; Bala, O. Effects of Nd:Yag laser irradiation on osteoblast cell cultures. Lasers Med. Sci. 2006, 21, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Chellini, F.; Sassoli, C.; Nosi, D.; Deledda, C.; Tonelli, P.; Zecchi-Orlandini, S.; Formigli, L.; Giannelli, M. Low pulse energy Nd:YAG laser irradiation exerts a biostimulative effect on different cells of the oral microenvironment: “an in vitro study”. Lasers Surg. Med. 2010, 42, 527–539. [Google Scholar] [CrossRef]
- Kara, C.; Selamet, H.; Gokmenoglu, C.; Kara, N. Low level laser therapy induces increased viability and proliferation in isolated cancer cells. Cell Prolif. 2018, 51, e12417. [Google Scholar] [CrossRef]
- Tsuka, Y.; Kunimatsu, R.; Gunji, H.; Nakajima, K.; Kimura, A.; Hiraki, T.; Nakatani, A.; Tanimoto, K. Effects of Nd:YAG low-level laser irradiation on cultured human osteoblasts migration and ATP production: In vitro study. Lasers Med. Sci. 2019, 34, 55–60. [Google Scholar] [CrossRef]
- Tsuka, Y.; Kunimatsu, R.; Gunji, H.; Abe, T.; Medina, C.C.; Nakajima, K.; Kimura, A.; Hiraki, T.; Nakatani, A.; Tanimoto, K. Examination of the Effect of the Combined Use of Nd: YAG Laser Irradiation and Mechanical Force Loading on Bone Metabolism Using Cultured Human Osteoblasts. J. Lasers Med. Sci. 2020, 11, 138–143. [Google Scholar] [CrossRef]
- Kim, I.S.; Cho, T.H.; Kim, K.; Weber, F.E.; Hwang, S.J. High power-pulsed Nd:YAG laser as a new stimulus to induce BMP-2 expression in MC3T3-E1 osteoblasts. Lasers Surg. Med. 2010, 42, 510–518. [Google Scholar] [CrossRef]
- Middleton, J.; Arnott, N.; Walsh, S.; Beresford, J. Osteoblasts and osteoclasts in adult human osteophyte tissue express the mRNAs for insulin-like growth factors I and II and the type 1 IGF receptor. Bone 1995, 16, 287–293. [Google Scholar] [CrossRef]
- Schwarz, F.; Rothamel, D.; Herten, M.; Bieling, K.; Scherbaum, W.; Becker, J. Effects of an Er:YAG laser on mitochondrial activity of human osteosarcoma-derived osteoblasts in vitro. Lasers Med. Sci. 2004, 19, 37–40. [Google Scholar] [CrossRef]
- Aleksic, V.; Aoki, A.; Iwasaki, K.; Takasaki, A.A.; Wang, C.Y.; Abiko, Y.; Ishikawa, I.; Izumi, Y. Low-level Er:YAG laser irradiation enhances osteoblast proliferation through activation of MAPK/ERK. Lasers Med. Sci. 2010, 25, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Niimi, H.; Ohsugi, Y.; Katagiri, S.; Watanabe, K.; Hatasa, M.; Shimohira, T.; Tsuchiya, Y.; Maekawa, S.; Hirota, T.; Kadokura, H.; et al. Effects of Low-Level Er:YAG Laser Irradiation on Proliferation and Calcification of Primary Osteoblast-Like Cells Isolated From Rat Calvaria. Front. Cell Dev. Biol. 2020, 8, 459. [Google Scholar] [CrossRef] [PubMed]
- Cargnello, M.; Roux, P.P. Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol. Mol. Biol. Rev. 2011, 75, 50–83. [Google Scholar] [CrossRef] [PubMed]
- Kawaki, H.; Kubota, S.; Suzuki, A.; Suzuki, M.; Kohsaka, K.; Hoshi, K.; Fujii, T.; Lazar, N.; Ohgawara, T.; Maeda, T.; et al. Differential roles of CCN family proteins during osteoblast differentiation: Involvement of Smad and MAPK signaling pathways. Bone 2011, 49, 975–989. [Google Scholar] [CrossRef]
- Lin, G.L.; Hankenson, K.D. Integration of BMP, Wnt, and notch signaling pathways in osteoblast differentiation. J. Cell Biochem. 2011, 112, 3491–3501. [Google Scholar] [CrossRef]
- Sasaki, Y.; Wang, S.; Ogata, Y. Transcriptional regulation of bone sialoprotein gene by CO(2) laser irradiation. J. Oral. Sci. 2011, 53, 51–59. [Google Scholar] [CrossRef][Green Version]
- Dekoninck, S.; Blanpain, C. Stem cell dynamics, migration and plasticity during wound healing. Nat. Cell Biol. 2019, 21, 18–24. [Google Scholar] [CrossRef]
- Bainbridge, P. Wound healing and the role of fibroblasts. J. Wound Care 2013, 22, 407–411. [Google Scholar]
- Grinnell, F. Fibroblasts, myofibroblasts, and wound contraction. J. Cell Biol. 1994, 124, 401–404. [Google Scholar] [CrossRef]
- Hamblin, M.R. Mechanisms and applications of the anti-inflammatory effects of photobiomodulation. Aims Biophys. 2017, 4, 337–361. [Google Scholar] [CrossRef]
- Solmaz, H.; Dervisoglu, S.; Gulsoy, M.; Ulgen, Y. Laser biostimulation of wound healing: Bioimpedance measurements support histology. Lasers Med. Sci. 2016, 31, 1547–1554. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, Y.; Yamaguchi, M.; Abiko, Y. Inhibitory effect of low-level laser irradiation on LPS-stimulated prostaglandin E2 production and cyclooxygenase-2 in human gingival fibroblasts. Eur. J. Oral. Sci. 2000, 108, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Takema, T.; Yamaguchi, M.; Abiko, Y. Reduction of Plasminogen Activator Activity Stimulated by Lipopolysaccharide from Periodontal Pathogen in Human Gingival Fibroblasts by Low-energy Laser Irradiation. Lasers Med. Sci. 2000, 15, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Nomura, K.; Yamaguchi, M.; Abiko, Y. Inhibition of interleukin-1beta production and gene expression in human gingival fibroblasts by low-energy laser irradiation. Lasers Med. Sci. 2001, 16, 218–223. [Google Scholar] [CrossRef]
- Almeida-Lopes, L.; Rigau, J.; Zângaro, R.A.; Guidugli-Neto, J.; Jaeger, M.M. Comparison of the low level laser therapy effects on cultured human gingival fibroblasts proliferation using different irradiance and same fluence. Lasers Surg. Med. 2001, 29, 179–184. [Google Scholar] [CrossRef]
- Basso, F.G.; Pansani, T.N.; Turrioni, A.P.; Bagnato, V.S.; Hebling, J.; de Souza Costa, C.A. In vitro wound healing improvement by low-level laser therapy application in cultured gingival fibroblasts. Int. J. Dent 2012, 2012, 719452. [Google Scholar] [CrossRef]
- Frozanfar, A.; Ramezani, M.; Rahpeyma, A.; Khajehahmadi, S.; Arbab, H.R. The Effects of Low Level Laser Therapy on the Expression of Collagen Type I Gene and Proliferation of Human Gingival Fibroblasts (Hgf3-Pi 53): In vitro Study. Iran J. Basic Med. Sci. 2013, 16, 1071–1074. [Google Scholar]
- Saygun, I.; Karacay, S.; Serdar, M.; Ural, A.U.; Sencimen, M.; Kurtis, B. Effects of laser irradiation on the release of basic fibroblast growth factor (bFGF), insulin like growth factor-1 (IGF-1), and receptor of IGF-1 (IGFBP3) from gingival fibroblasts. Lasers Med. Sci. 2008, 23, 211–215. [Google Scholar] [CrossRef]
- Basso, F.G.; Pansani, T.N.; Soares, D.G.; Scheffel, D.L.; Bagnato, V.S.; de Souza Costa, C.A.; Hebling, J. Biomodulation of Inflammatory Cytokines Related to Oral Mucositis by Low-Level Laser Therapy. Photochem. Photobiol. 2015, 91, 952–956. [Google Scholar] [CrossRef]
- Hakki, S.S.; Bozkurt, S.B. Effects of different setting of diode laser on the mRNA expression of growth factors and type I collagen of human gingival fibroblasts. Lasers Med. Sci. 2012, 27, 325–331. [Google Scholar] [CrossRef]
- Damante, C.A.; De Micheli, G.; Miyagi, S.P.; Feist, I.S.; Marques, M.M. Effect of laser phototherapy on the release of fibroblast growth factors by human gingival fibroblasts. Lasers Med. Sci. 2009, 24, 885–891. [Google Scholar] [CrossRef] [PubMed]
- Kreisler, M.; Daubländer, M.; Willershausen-Zönnchen, B.; d’Hoedt, B. Effect of diode laser irradiation on the survival rate of gingival fibroblast cell cultures. Lasers Surg. Med. 2001, 28, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.N.; Eduardo Cde, P.; Matson, E.; Marques, M.M. Effect of low-power laser irradiation on cell growth and procollagen synthesis of cultured fibroblasts. Lasers Surg. Med. 2002, 31, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Sassoli, C.; Chellini, F.; Squecco, R.; Tani, A.; Idrizaj, E.; Nosi, D.; Giannelli, M.; Zecchi-Orlandini, S. Low intensity 635 nm diode laser irradiation inhibits fibroblast-myofibroblast transition reducing TRPC1 channel expression/activity: New perspectives for tissue fibrosis treatment. Lasers Surg. Med. 2016, 48, 318–332. [Google Scholar] [CrossRef]
- Kaibuchi, N.; Iwata, T.; Yamato, M.; Okano, T.; Ando, T. Multipotent mesenchymal stromal cell sheet therapy for bisphosphonate-related osteonecrosis of the jaw in a rat model. Acta Biomater. 2016, 42, 400–410. [Google Scholar] [CrossRef]
- Marques, M.M.; Pereira, A.N.; Fujihara, N.A.; Nogueira, F.N.; Eduardo, C.P. Effect of low-power laser irradiation on protein synthesis and ultrastructure of human gingival fibroblasts. Lasers Surg. Med. 2004, 34, 260–265. [Google Scholar] [CrossRef]
- Castro, D.J.; Abergel, R.P.; Meeker, C.; Dwyer, R.M.; Lesavoy, M.A.; Uitto, J. Effects of the Nd:YAG laser on DNA synthesis and collagen production in human skin fibroblast cultures. Ann. Plast Surg. 1983, 11, 214–222. [Google Scholar] [CrossRef]
- Abergel, R.P.; Meeker, C.A.; Dwyer, R.M.; Lesavoy, M.A.; Uitto, J. Nonthermal effects of ND:YAG laser on biological functions of human skin fibroblasts in culture. Lasers Surg. Med. 1984, 3, 279–284. [Google Scholar] [CrossRef]
- Cisneros, J.L.; Río, R.; Palou, J. The Q-switched neodymium (Nd):YAG laser with quadruple frequency. Clinical histological evaluation of facial resurfacing using different wavelengths. Derm. Surg. 1998, 24, 345–350. [Google Scholar] [CrossRef]
- Jansen, P.L.; Rosch, R.; Jansen, M.; Binnebösel, M.; Junge, K.; Alfonso-Jaume, A.; Klinge, U.; Lovett, D.H.; Mertens, P.R. Regulation of MMP-2 gene transcription in dermal wounds. J. Investig. Derm. 2007, 127, 1762–1767. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, A.; Perfetto, B.; Guerrera, L.P.; Oliviero, G.; Baroni, A. Q-switched 1064 nm Nd-Yag nanosecond laser effects on skin barrier function and on molecular rejuvenation markers in keratinocyte-fibroblasts interaction. Lasers Med. Sci. 2019, 34, 595–605. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Wang, L.; Dang, Y.; Liu, B.; Zhao, D. Investigation of the 1064 nm Q-switched Nd:YAG laser on collagen expression in an animal model. Photomed. Laser Surg. 2012, 30, 604–609. [Google Scholar] [CrossRef] [PubMed]
- Dang, Y.; Ye, X.; Weng, Y.; Tong, Z.; Ren, Q. Effects of the 532-nm and 1064-nm Q-switched Nd: YAG lasers on collagen turnover of cultured human skin fibroblasts: A comparative study. Lasers Med. Sci. 2010, 25, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Ishikawa, I.; Suzuki, M.; Hasegawa, K. Clinical assessments of the erbium:YAG laser for soft tissue surgery and scaling. J. Clin. Laser Med. Surg. 1996, 14, 67–75. [Google Scholar] [CrossRef]
- Aoki, A.; Mizutani, K.; Takasaki, A.A.; Sasaki, K.M.; Nagai, S.; Schwarz, F.; Yoshida, I.; Eguro, T.; Zeredo, J.L.; Izumi, Y. Current status of clinical laser applications in periodontal therapy. Gen Dent 2008, 56, 674–687, quiz 688-9, 767. [Google Scholar]
- Pourzarandian, A.; Watanabe, H.; Ruwanpura, S.M.; Aoki, A.; Ishikawa, I. Effect of low-level Er:YAG laser irradiation on cultured human gingival fibroblasts. J. Periodontol. 2005, 76, 187–193. [Google Scholar] [CrossRef]
- Pourzarandian, A.; Watanabe, H.; Ruwanpura, S.M.; Aoki, A.; Noguchi, K.; Ishikawa, I. Er:YAG laser irradiation increases prostaglandin E production via the induction of cyclooxygenase-2 mRNA in human gingival fibroblasts. J. Periodontal Res. 2005, 40, 182–186. [Google Scholar] [CrossRef]
- Ogita, M.; Tsuchida, S.; Aoki, A.; Satoh, M.; Kado, S.; Sawabe, M.; Nanbara, H.; Kobayashi, H.; Takeuchi, Y.; Mizutani, K.; et al. Increased cell proliferation and differential protein expression induced by low-level Er:YAG laser irradiation in human gingival fibroblasts: Proteomic analysis. Lasers Med. Sci. 2015, 30, 1855–1866. [Google Scholar] [CrossRef]
- Kong, S.; Aoki, A.; Iwasaki, K.; Mizutani, K.; Katagiri, S.; Suda, T.; Ichinose, S.; Ogita, M.; Pavlic, V.; Izumi, Y. Biological effects of Er:YAG laser irradiation on the proliferation of primary human gingival fibroblasts. J. Biophotonics 2018, 11, 201700157. [Google Scholar] [CrossRef]
- Talebi-Ardakani, M.R.; Torshabi, M.; Karami, E.; Arbabi, E.; Rezaei Esfahrood, Z. In Vitro Study of Er:YAG and Er, Cr:YSGG Laser Irradiation on Human Gingival Fibroblast Cell Line. Acta Med. Iran 2016, 54, 251–255. [Google Scholar]
- Tsuka, Y.; Kunimatsu, R.; Gunji, H.; Abe, T.; Medina, C.C.; Hiraki, T.; Nakatani, A.; Sakata, S.; Rikitake, K.; Aisyah, P.N.; et al. Examination of the effect of combined use of Er:YAG laser irradiation and mechanical force loading on bone metabolism using primary human gingival fibroblasts. Lasers Med. Sci. 2020, 35, 2059–2064. [Google Scholar] [CrossRef] [PubMed]
- Thomson, P.J.; Wylie, J. Interventional laser surgery: An effective surgical and diagnostic tool in oral precancer management. Int. J. Oral. Maxillofac. Surg. 2002, 31, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Iwasaka, K.; Hemmi, E.; Tomita, K.; Ishihara, S.; Katayama, T.; Sakagami, H. Effect of CO₂ laser irradiation on hormesis induction in human pulp and periodontal ligament fibroblasts. In Vivo 2011, 25, 787–793. [Google Scholar] [PubMed]
- Pant, V.; Dixit, J.; Agrawal, A.K.; Seth, P.K.; Pant, A.B. Behavior of human periodontal ligament cells on CO2 laser irradiated dentinal root surfaces: An in vitro study. J. Periodontal Res. 2004, 39, 373–379. [Google Scholar] [CrossRef]
- Yamasaki, A.; Tamamura, K.; Sakurai, Y.; Okuyama, N.; Yusa, J.; Ito, H. Remodeling of the rat gingiva induced by CO2 laser coagulation mode. Lasers Surg. Med. 2008, 40, 695–703. [Google Scholar] [CrossRef]
- Nowak, K.C.; McCormack, M.; Koch, R.J. The effect of superpulsed carbon dioxide laser energy on keloid and normal dermal fibroblast secretion of growth factors: A serum-free study. Plast Reconstr. Surg. 2000, 105, 2039–2048. [Google Scholar] [CrossRef]
- Shingyochi, Y.; Kanazawa, S.; Tajima, S.; Tanaka, R.; Mizuno, H.; Tobita, M. A Low-Level Carbon Dioxide Laser Promotes Fibroblast Proliferation and Migration through Activation of Akt, ERK, and JNK. PLoS ONE 2017, 12, e0168937. [Google Scholar] [CrossRef]
- Apfelberg, D.B.; Mittelman, H.; Chadi, B. Carcinogenic potential of in vitro carbon dioxide laser exposure of fibroblast. Obs. Gynecol. 1983, 61, 493–496. [Google Scholar]
- Barczyk, M.; Bolstad, A.I.; Gullberg, D. Role of integrins in the periodontal ligament: Organizers and facilitators. Periodontoloy 2000 2013, 63, 29–47. [Google Scholar] [CrossRef]
- Wu, J.Y.; Chen, C.H.; Yeh, L.Y.; Yeh, M.L.; Ting, C.C.; Wang, Y.H. Low-power laser irradiation promotes the proliferation and osteogenic differentiation of human periodontal ligament cells via cyclic adenosine monophosphate. Int. J. Oral. Sci. 2013, 5, 85–91. [Google Scholar] [CrossRef]
- Huang, T.H.; Chen, C.C.; Liu, S.L.; Lu, Y.C.; Kao, C.T. A low-level diode laser therapy reduces the lipopolysaccharide (LPS)-induced periodontal ligament cell inflammation. Laser Phys. Lett. 2014, 11, 075602. [Google Scholar] [CrossRef]
- Mayahara, K.; Yamaguchi, A.; Sakaguchi, M.; Igarashi, Y.; Shimizu, N. Effect of Ga-Al-As laser irradiation on COX-2 and cPLA2-alpha expression in compressed human periodontal ligament cells. Lasers Surg. Med. 2010, 42, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, N.; Yamaguchi, M.; Goseki, T.; Shibata, Y.; Takiguchi, H.; Iwasawa, T.; Abiko, Y. Inhibition of prostaglandin E2 and interleukin 1-beta production by low-power laser irradiation in stretched human periodontal ligament cells. J. Dent Res. 1995, 74, 1382–1388. [Google Scholar] [CrossRef]
- Ozawa, Y.; Shimizu, N.; Abiko, Y. Low-energy diode laser irradiation reduced plasminogen activator activity in human periodontal ligament cells. Lasers Surg. Med. 1997, 21, 456–463. [Google Scholar] [CrossRef]
- Huang, T.H.; Liu, S.L.; Chen, C.L.; Shie, M.Y.; Kao, C.T. Low-level laser effects on simulated orthodontic tension side periodontal ligament cells. Photomed. Laser Surg. 2013, 31, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.J.; Yim, J.Y.; Koo, K.T.; Seol, Y.J.; Lee, Y.M.; Ku, Y.; Rhyu, I.C.; Chung, C.P.; Kim, T.I. Biological effects of a semiconductor diode laser on human periodontal ligament fibroblasts. J. Periodontal. Implant Sci. 2010, 40, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Dehdashtizadeh, A.; Esnaashari, N.; Farhad, S.Z.; Ejeian, F.; Amini, S. The effect of laser irradiation and doxycycline application on the production of matrix metalloproteinase-8 and collagen I from cultured human periodontal ligament cells. Dent. Res. J. Isfahan 2020, 17, 213–218. [Google Scholar] [PubMed]
- Kreisler, M.; Christoffers, A.B.; Willershausen, B.; d’Hoedt, B. Effect of low-level GaAlAs laser irradiation on the proliferation rate of human periodontal ligament fibroblasts: An in vitro study. J. Clin. Periodontol. 2003, 30, 353–358. [Google Scholar] [CrossRef]
- Lin, T.; Yu, C.C.; Liu, C.M.; Hsieh, P.L.; Liao, Y.W.; Yu, C.H.; Chen, C.J. Er:YAG laser promotes proliferation and wound healing capacity of human periodontal ligament fibroblasts through Galectin-7 induction. J. Med. Assoc. 2020. [Google Scholar] [CrossRef]
- Peplow, P.V.; Chung, T.Y.; Baxter, G.D. Laser photobiomodulation of wound healing: A review of experimental studies in mouse and rat animal models. Photomed. Laser Surg. 2010, 28, 291–325. [Google Scholar] [CrossRef]
- Schwarz, F.; Aoki, A.; Sculean, A.; Becker, J. The impact of laser application on periodontal and peri-implant wound healing. Periodontology 2000 2009, 51, 79–108. [Google Scholar] [CrossRef] [PubMed]
- Vannella, K.M.; Wynn, T.A. Mechanisms of Organ Injury and Repair by Macrophages. Annu. Rev. Physiol. 2017, 79, 593–617. [Google Scholar] [CrossRef] [PubMed]
- Amaroli, A.; Ravera, S.; Baldini, F.; Benedicenti, S.; Panfoli, I.; Vergani, L. Photobiomodulation with 808-nm diode laser light promotes wound healing of human endothelial cells through increased reactive oxygen species production stimulating mitochondrial oxidative phosphorylation. Lasers Med. Sci. 2019, 34, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Protasiewicz, M.; Kuliczkowski, W.; Woznicka, A.K.; Szymkiewicz, P.; Derkacz, A.; Andrzej, M.; Dziegiel, P. Influence of low-power laser illumination on pro-inflammatory response in human endothelial cells stimulated with interleukin-1a. In Vivo 2013, 27, 313–319. [Google Scholar]
- Schindl, A.; Merwald, H.; Schindl, L.; Kaun, C.; Wojta, J. Direct stimulatory effect of low-intensity 670 nm laser irradiation on human endothelial cell proliferation. Br. J. Derm. 2003, 148, 334–336. [Google Scholar] [CrossRef]
- Góralczyk, K.; Szymańska, J.; Łukowicz, M.; Drela, E.; Kotzbach, R.; Dubiel, M.; Michalska, M.; Góralczyk, B.; Zając, A.; Rość, D. Effect of LLLT on endothelial cells culture. Lasers Med. Sci. 2015, 30, 273–278. [Google Scholar] [CrossRef]
- Du, S.; Zhang, Q.; Zhang, S.; Wang, L.; Lian, J. Heat shock protein 70 expression induced by diode laser irradiation on choroid-retinal endothelial cells in vitro. Mol. Vis. 2012, 18, 2380–2387. [Google Scholar]
- Masuda, Y.; Yokose, S.; Sakagami, H. Gene Expression Analysis of Cultured Rat-Endothelial Cells after Nd:YAG Laser Irradiation by Affymetrix GeneChip Array. In Vivo 2017, 31, 51–54. [Google Scholar] [CrossRef][Green Version]
- Giannelli, M.; Bani, D.; Tani, A.; Pini, A.; Margheri, M.; Zecchi-Orlandini, S.; Tonelli, P.; Formigli, L. In vitro evaluation of the effects of low-intensity Nd:YAG laser irradiation on the inflammatory reaction elicited by bacterial lipopolysaccharide adherent to titanium dental implants. J. Periodontol. 2009, 80, 977–984. [Google Scholar] [CrossRef]
- Saygin, N.E.; Giannobile, W.V.; Somerman, M.J. Molecular and cell biology of cementum. Periodontoloy 2000 2000, 24, 73–98. [Google Scholar] [CrossRef]
- Bozkurt, S.B.; Hakki, E.E.; Kayis, S.A.; Dundar, N.; Hakki, S.S. Biostimulation with diode laser positively regulates cementoblast functions, in vitro. Lasers Med. Sci. 2017, 32, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 2009, 119, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Rathinam, V.A.K.; Chan, F.K. Inflammasome, Inflammation, and Tissue Homeostasis. Trends Mol. Med. 2018, 24, 304–318. [Google Scholar] [CrossRef] [PubMed]
- de Farias Gabriel, A.; Wagner, V.P.; Correa, C.; Webber, L.P.; Pilar, E.F.S.; Curra, M.; Carrard, V.C.; Martins, M.A.T.; Martins, M.D. Photobiomodulation therapy modulates epigenetic events and NF-kappaB expression in oral epithelial wound healing. Lasers Med. Sci. 2019, 34, 1465–1472. [Google Scholar] [CrossRef]
- Luomanen, M.; Rauhamaa-Makinen, R.; Meurman, J.H.; Kosloff, T.; Tiitta, O. Healing of rat mouth mucosa after irradiation with CO2, Nd:YAG, and CO2-Nd:YAG combination lasers. Scand. J. Dent. Res. 1994, 102, 223–228. [Google Scholar] [CrossRef]
- Sawabe, M.; Aoki, A.; Komaki, M.; Iwasaki, K.; Ogita, M.; Izumi, Y. Gingival tissue healing following Er:YAG laser ablation compared to electrosurgery in rats. Lasers Med. Sci. 2015, 30, 875–883. [Google Scholar] [CrossRef]
- Posten, W.; Wrone, D.A.; Dover, J.S.; Arndt, K.A.; Silapunt, S.; Alam, M. Low-level laser therapy for wound healing: Mechanism and efficacy. Derm. Surg. 2005, 31, 334–340. [Google Scholar] [CrossRef]
- Grossman, N.; Schneid, N.; Reuveni, H.; Halevy, S.; Lubart, R. 780 nm low power diode laser irradiation stimulates proliferation of keratinocyte cultures: Involvement of reactive oxygen species. Lasers Surg. Med. 1998, 22, 212–218. [Google Scholar] [CrossRef]
- Basso, F.G.; Oliveira, C.F.; Kurachi, C.; Hebling, J.; Costa, C.A. Biostimulatory effect of low-level laser therapy on keratinocytes in vitro. Lasers Med. Sci. 2013, 28, 367–374. [Google Scholar] [CrossRef]
- Donnarumma, G.; De Gregorio, V.; Fusco, A.; Farina, E.; Baroni, A.; Esposito, V.; Contaldo, M.; Petruzzi, M.; Pannone, G.; Serpico, R. Inhibition of HSV-1 replication by laser diode-irradiation: Possible mechanism of action. Int. J. Immunopathol. Pharm. 2010, 23, 1167–1176. [Google Scholar] [CrossRef]
- Yang, H.Q.; Wang, Y.H.; Chen, J.X.; Zheng, L.Q.; Xie, S.S. [Low level laser irradiation in the visible spectra induces HeLa cells proliferation]. Spectrosc. Spectr. Anal. 2012, 32, 1024–1027. [Google Scholar]
- Mognato, M.; Squizzato, F.; Facchin, F.; Zaghetto, L.; Corti, L. Cell growth modulation of human cells irradiated in vitro with low-level laser therapy. Photomed. Laser Surg. 2004, 22, 523–526. [Google Scholar] [CrossRef] [PubMed]
- Szabó, A.; Varga, V.; Toimela, T.; Hiitelä, K.; Tähti, H.; Oja, S.S.; Süveges, I.; Salminen, L. Laser treatment of cultured retinal pigment epithelial cells-evaluation of the cellular damage in vitro. J. Ocul. Pharm. 2004, 20, 246–255. [Google Scholar] [CrossRef] [PubMed]
- Barak, A.; Goldkorn, T.; Morse, L.S. Laser induces apoptosis and ceramide production in human retinal pigment epithelial cells. Investig. Ophthalmol. Vis. Sci. 2005, 46, 2587–2591. [Google Scholar] [CrossRef] [PubMed]
- Hattenbach, L.O.; Beck, K.F.; Pfeilschifter, J.; Koch, F.; Ohrloff, C.; Schacke, W. Pigment-epithelium-derived factor is upregulated in photocoagulated human retinal pigment epithelial cells. Ophthalmic Res. 2005, 37, 341–346. [Google Scholar] [CrossRef]
- Powell, K.; Low, P.; McDonnell, P.A.; Laakso, E.L.; Ralph, S.J. The effect of laser irradiation on proliferation of human breast carcinoma, melanoma, and immortalized mammary epithelial cells. Photomed. Laser Surg. 2010, 28, 115–123. [Google Scholar] [CrossRef]
- Tang, E.; Khan, I.; Andreana, S.; Arany, P.R. Laser-activated transforming growth factor-β1 induces human β-defensin 2: Implications for laser therapies for periodontitis and peri-implantitis. J. Periodontal. Res. 2017, 52, 360–367. [Google Scholar] [CrossRef]
- Ejiri, K.; Aoki, A.; Yamaguchi, Y.; Ohshima, M.; Izumi, Y. High-frequency low-level diode laser irradiation promotes proliferation and migration of primary cultured human gingival epithelial cells. Lasers Med. Sci. 2014, 29, 1339–1347. [Google Scholar] [CrossRef]
- Nagahara, A.; Mitani, A.; Fukuda, M.; Yamamoto, H.; Tahara, K.; Morita, I.; Ting, C.C.; Watanabe, T.; Fujimura, T.; Osawa, K.; et al. Antimicrobial photodynamic therapy using a diode laser with a potential new photosensitizer, indocyanine green-loaded nanospheres, may be effective for the clearance of Porphyromonas gingivalis. J. Periodontal. Res. 2013, 48, 591–599. [Google Scholar] [CrossRef]
- Fujimura, T.; Mitani, A.; Fukuda, M.; Mogi, M.; Osawa, K.; Takahashi, S.; Aino, M.; Iwamura, Y.; Miyajima, S.; Yamamoto, H.; et al. Irradiation with a low-level diode laser induces the developmental endothelial locus-1 gene and reduces proinflammatory cytokines in epithelial cells. Lasers Med. Sci 2014, 29, 987–994. [Google Scholar] [CrossRef]
- Yokose, S.K.H. Low-power carbon dioxide laser irradiation reduces sclerostin expression, but stimulates Dmp-1 expression in osteocyte-like cells of rats. J. Bio-Integ. 2013, 3, 8. [Google Scholar] [CrossRef]
- Ohsugi, Y.; Katagiri, S.; Hirota, T.; Niimi, H.; Hatasa, M.; Watanabe, K.; Shimohira, T.; Mizutani, K.; Kitazawa, M.; Matsuzawa, A.; et al. Laser irradiation decreases sclerostin expression in bone and osteogenic cells. Faseb. J. 2020. [Google Scholar] [CrossRef]
- Suda, T.; Takahashi, N.; Udagawa, N.; Jimi, E.; Gillespie, M.T.; Martin, T.J. Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr. Rev. 1999, 20, 345–357. [Google Scholar] [CrossRef]
- Aihara, N.; Yamaguchi, M.; Kasai, K. Low-energy irradiation stimulates formation of osteoclast-like cells via RANK expression in vitro. Lasers Med. Sci. 2006, 21, 24–33. [Google Scholar] [CrossRef]
- Atala, A.L.R. Handbook of Stem Cells; Academic Press: Cambridge, MA, USA, 2012; Volume 452. [Google Scholar]
- Mvula, B.; Mathope, T.; Moore, T.; Abrahamse, H. The effect of low level laser irradiation on adult human adipose derived stem cells. Lasers Med. Sci. 2008, 23, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Seo, B.M.; Miura, M.; Gronthos, S.; Bartold, P.M.; Batouli, S.; Brahim, J.; Young, M.; Robey, P.G.; Wang, C.Y.; Shi, S. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 2004, 364, 149–155. [Google Scholar] [CrossRef]
- Gronthos, S.; Mankani, M.; Brahim, J.; Robey, P.G.; Shi, S. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2000, 97, 13625–13630. [Google Scholar] [CrossRef] [PubMed]
- Gurtner, G.C.; Callaghan, M.J.; Longaker, M.T. Progress and potential for regenerative medicine. Annu. Rev. Med. 2007, 58, 299–312. [Google Scholar] [CrossRef] [PubMed]
- Zare, F.; Moradi, A.; Fallahnezhad, S.; Ghoreishi, S.K.; Amini, A.; Chien, S.; Bayat, M. Photobiomodulation with 630 plus 810nm wavelengths induce more in vitro cell viability of human adipose stem cells than human bone marrow-derived stem cells. J. Photochem. Photobiol. BBiol. 2019, 201, 111658. [Google Scholar] [CrossRef]
- Wu, Y.H.; Wang, J.; Gong, D.X.; Gu, H.Y.; Hu, S.S.; Zhang, H. Effects of low-level laser irradiation on mesenchymal stem cell proliferation: A microarray analysis. Lasers Med. Sci. 2012, 27, 509–519. [Google Scholar] [CrossRef]
- Hou, J.F.; Zhang, H.; Yuan, X.; Li, J.; Wei, Y.J.; Hu, S.S. In vitro effects of low-level laser irradiation for bone marrow mesenchymal stem cells: Proliferation, growth factors secretion and myogenic differentiation. Lasers Surg. Med. 2008, 40, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Giannelli, M.; Chellini, F.; Sassoli, C.; Francini, F.; Pini, A.; Squecco, R.; Nosi, D.; Bani, D.; Zecchi-Orlandini, S.; Formigli, L. Photoactivation of bone marrow mesenchymal stromal cells with diode laser: Effects and mechanisms of action. J. Cell. Physiol. 2013, 228, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Li, X.; Zhu, S.; Xie, Y.; Du, J.; Ge, H.; Bai, Y.; Liu, Y.; Guo, L. Photobiomodulation with 808-nm diode laser enhances gingival wound healing by promoting migration of human gingival mesenchymal stem cells via ROS/JNK/NF-kappaB/MMP-1 pathway. Lasers Med. Sci. 2020, 35, 1831–1839. [Google Scholar] [CrossRef] [PubMed]
- Amaroli, A.; Agas, D.; Laus, F.; Cuteri, V.; Hanna, R.; Sabbieti, M.G.; Benedicenti, S. The Effects of Photobiomodulation of 808 nm Diode Laser Therapy at Higher Fluence on the in Vitro Osteogenic Differentiation of Bone Marrow Stromal Cells. Front. Physiol. 2018, 9, 123. [Google Scholar] [CrossRef]
- Horvat-Karajz, K.; Balogh, Z.; Kovacs, V.; Drrernat, A.H.; Sreter, L.; Uher, F. In vitro effect of carboplatin, cytarabine, paclitaxel, vincristine, and low-power laser irradiation on murine mesenchymal stem cells. Lasers Surg. Med. 2009, 41, 463–469. [Google Scholar] [CrossRef]
- Ferreira, L.S.; Diniz, I.M.A.; Maranduba, C.M.S.; Miyagi, S.P.H.; Rodrigues, M.; Moura-Netto, C.; Marques, M.M. Short-term evaluation of photobiomodulation therapy on the proliferation and undifferentiated status of dental pulp stem cells. Lasers Med. Sci. 2019, 34, 659–666. [Google Scholar] [CrossRef]
- Garrido, P.R.; Pedroni, A.C.F.; Cury, D.P.; Moreira, M.S.; Rosin, F.; Sarra, G.; Marques, M.M. Effects of photobiomodulation therapy on the extracellular matrix of human dental pulp cell sheets. J. Photochem. Photobiol. BBiol. 2019, 194, 149–157. [Google Scholar] [CrossRef]
- Yurtsever, M.C.; Kiremitci, A.; Gumusderelioglu, M. Dopaminergic induction of human dental pulp stem cells by photobiomodulation: Comparison of 660 nm laser light and polychromatic light in the nir. J. Photochem. Photobiol. BBiol. 2020, 204, 111742. [Google Scholar] [CrossRef]
- Wang, L.; Wu, F.; Liu, C.; Song, Y.; Guo, J.; Yang, Y.; Qiu, Y. Low-level laser irradiation modulates the proliferation and the osteogenic differentiation of bone marrow mesenchymal stem cells under healthy and inflammatory condition. Lasers Med. Sci. 2019, 34, 169–178. [Google Scholar] [CrossRef]
- Peat, F.J.; Colbath, A.C.; Bentsen, L.M.; Goodrich, L.R.; King, M.R. In Vitro Effects of High-Intensity Laser Photobiomodulation on Equine Bone Marrow-Derived Mesenchymal Stem Cell Viability and Cytokine Expression. Photomed. Laser Surg. 2018, 36, 83–91. [Google Scholar] [CrossRef]
- Anwer, A.G.; Gosnell, M.E.; Perinchery, S.M.; Inglis, D.W.; Goldys, E.M. Visible 532 nm laser irradiation of human adipose tissue-derived stem cells: Effect on proliferation rates, mitochondria membrane potential and autofluorescence. Lasers Surg. Med. 2012, 44, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Constantin, A.; Dumitrescu, M.; Mihai Corotchi, M.C.; Jianu, D.; Simionescu, M. CO2 laser increases the regenerative capacity of human adipose-derived stem cells by a mechanism involving the redox state and enhanced secretion of pro-angiogenic molecules. Lasers Med. Sci. 2017, 32, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Iwata, T.; Yamato, M.; Washio, K.; Yoshida, T.; Tsumanuma, Y.; Yamada, A.; Onizuka, S.; Izumi, Y.; Ando, T.; Okano, T.; et al. Periodontal regeneration with autologous periodontal ligament-derived cell sheets—A safety and efficacy study in ten patients. Regen 2018, 9, 38–44. [Google Scholar]
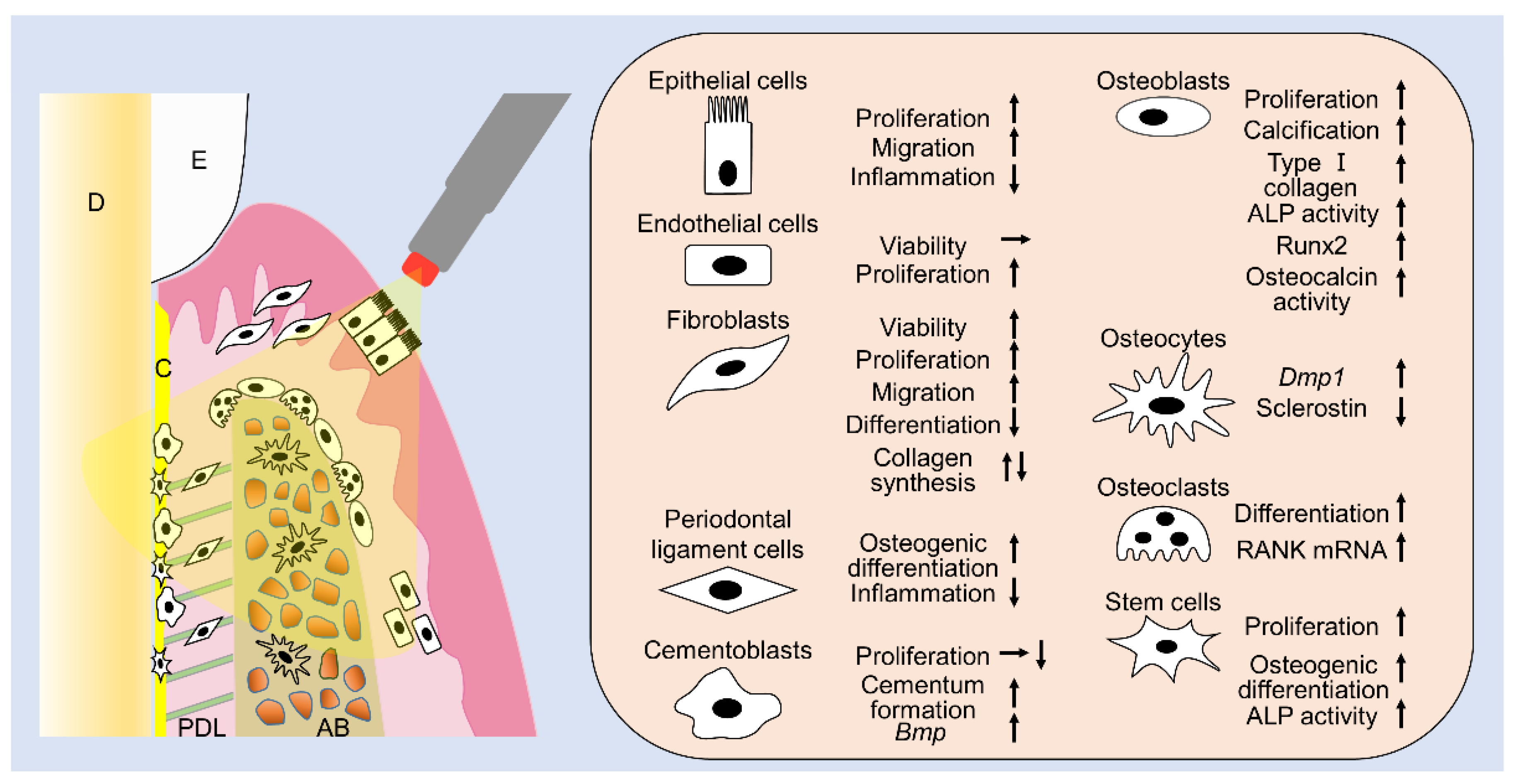
| Reference No. | Laser | Cell | Year Author | Irradiation Protocol | Major Findings |
|---|---|---|---|---|---|
| [15] | Diode | MC3T3-E1 | 2001 Yamamoto, et al. | 830 nm 7.64 J/cm2 CW 20 min | Irradiation may enhance DNA replication and play a role in stimulating proliferation of osteoblast through the enhancement of the mouse minichromosome maintenance 3 gene expression. |
| [16] | Diode | MC3T3-E1 | 2003 Hamajima et al. | 830 nm 7.64 J/cm2 CW 20 min | The osteoglycin gene was upregulated at 2 h after low level laser irradiation. |
| [17] | Diode | MC3T3-E1, MG-63 | 2007 Renno, et al. | 670, 780, or 830 nm 0.5, 1, 5, or 10 J/cm2 CW | Osteoblast proliferation increased significantly after 830 nm laser irradiation (10 J/cm2) but decreased after 780 nm laser irradiation (at 1, 5, and 10 J/cm2). MG-63 cell proliferation increased significantly after 670 nm (at 5 J/cm2) and 780 nm (at 1, 5, and 10 J/cm2) laser irradiation, but not after 830 nm laser irradiation. Alkaline phosphatase (ALP) activity in the osteoblast line was increased after 830 nm laser irradiation at 10 J/cm2 |
| [18] | Diode | MC3T3-E1 | 2010 Fujimoto et al. | 830 nm 0.97, 1.91, or 3.82 J/cm2 2 Hz 5, 10, or 20 min | Expression of bone morphogenetic protein (BMP)-2, 4, and 7 were significantly increased at 6, 9, 12 h. Runt-related protein transcription factor 2 (Runx2), Osx, distal-less homeobox 5 (Dlx5), and Msh homeobox 2 (Msx2) expression was significantly increased at 12, 24, 48 h. |
| [19] | Diode | MC3T3-E1 | 2011 Kanenari, et al. | 830 nm 7.64 J/cm2 20 min | Laser irradiation enhances Map1a gene expression and modulates microtubule assembly and the functional structure of microtubules, in turn, stimulates osteoblastic proliferation and differentiation. |
| [20] | Diode | MC3T3-E1 | 2014 Migliario, et al. | 980 nm 1.57, 7.87, 15.74, or 78.75 J/cm2 CW 1, 5, 10, 25, or 50 s | Laser irradiation enhances cell proliferation via ROS production. |
| [21] | Diode | MC3T3-E1 | 2014 Pagin, et al. | 660 and 780 nm 3, 5 J/cm2 Punctual irradiation mode 2 and 5 s | Laser irradiation significantly promoted cell growth at 24 h. |
| [22] | Diode | MC3T3-E1 | 2017 Oliveira, et al. | 660 or 780 nm 1.9, or 3.8 J/cm2 CW 4 or 8 s | Laser irradiation at both wavelengths significantly increased cell viability on 24 and 48 h. Infrared Ga-Al-As laser at 780 nm significantly increased ALP activity on 24 and 72 h. Red laser at 660 nm significantly increased matrix metalloproteinase (MMP)-2 activities on 48 and 72 h. |
| [23] | Diode | MC3T3-E1 | 2017 Son, et al. | 808 nm ± 5 nm 1.2 J/cm2 CW15 s 3 times at 0, 24, and 48 h | Laser irradiation with melatonin treatment increased significantly Osterix (Sp7) expression at 48 and 72 h and ALP activity and calcification on day 7 and 14. |
| [24] | Diode | MC3T3-E1 | 2017 Li, et al. | 808 nm 1.25, 3.75, or 6.25 J/cm2 CW 30, 90, or 150 s | Irradiation at 3.75 J/cm2 increased the cell amount at S phase and promoted cell proliferation through hedgehog signaling pathway at 24 h. The expressions of Ihh, Ptch, Smo, and Gli were significantly increased by 3.75 J/cm2 irradiation. |
| [25] | Diode | MC3T3-E1 | 2018 Kunimatsu, et al. | 910 nm 0, 1.42, 2.85, 5.7, or 17.1 J/cm2 Pulsed 30 kHz | Cell proliferation was significantly increased by laser irradiation at a dose of 2.85, 5.7, or 17.1 J/cm2. Laser irradiation at a dose of 2.85 J/cm2 induced MC3T3-E1 cells to migrate more rapidly than nonirradiated control cells. Irradiation with the high-frequency 910 nm diode laser at a dose of 2.85 J/cm2 induces phosphorylation of Mitogen‑activated protein kinase (MAPK)/extracellular signal-regulated kinase (ERK)1/2 at 15 and 30 min later irradiation |
| [26] | Diode | MC3T3-E1 | 2018 Mikami, et al. | 405 nm 1.9, 5.6, 9.4, 13.1, or 16.9 J/cm2 Pulsed 80 MHz 1 min | Laser irradiation significantly accelerated cell proliferation activity on day 3 and ALP activity on day 7 via transient receptor potential vanilloid 1(TRPV1). Expression of Alpl, Sp7, and Runx2 mRNAs was significantly increased. Calcification was significantly increased 3 weeks later after final irradiation on day 2, 4, 6, 9, and 11. |
| [27] | Diode | Saos-2 | 2000 Coombe, et al. | 830 nm 1.7–25.1 J/cm2 CW Single or multiple for 10 days | Cellular proliferation or activation of osteoblastic cells was not significantly affected by laser irradiation. |
| [28] | Diode | Saos-2 | 2013 Bayram, et al. | 808 nm 1.316, or 2.63 J/cm2 CW 10 s | Laser irradiation lessened the detrimental effects of zoledronate, improved cell function and/or proliferation, and ALP activity. |
| [29] | Diode | Saos-2 | 2013 Bloise, et al. | 659 nm 1 and 3 J/cm2 200 or 600 s Single or multiple for 3 days | Cell proliferation is significantly increased on day 2 by single dose of 1 J/cm2 and on day 2, 3, and 7 with multiple doses of 1 and 3 J/cm2. ALP activity on day 14 and calcification on day 14 were increased significantly. |
| [30] | Diode | Saos-2 | 2014 Incerti Parenti, et al. | 915 nm 1, 5, 10, 20, or 50 J/cm2 Pulsed 100 Hz 10, 48, 96, 193, or 482 s | Cell viability was significantly increased on day 3 by irradiation at 10 J/cm2, but significantly decreased by irradiation at 20 and 50 J/cm2. A rapid and transitory trend toward increased receptor activator of NF-κB ligand (RANKL)/osteoprotegerin ( OPG) ratio and a tendency toward a delayed increase in VEGF release for doses of 1 to 10 J/cm2 was found. |
| [31] | Diode | Saos-2 | 2015 Tschon, et al. | 915 nm 5, 10, or 15 J/cm2 Pulsed 100 Hz 0, 48, 96, or 144 s | Wound healing was significantly promoted at 72 and 96 h. COL1A1 expression was significantly increased at 24, 48, and 72 h. transforming growth factor (TGF) B1 expression significantly decreased TGFB1 expression at 48 and 72 h. |
| [32] | Diode | HOB and Saos-2 | 2016 Heymann, et al. | 670 nm 100 mW/cm2 CW 120 s | Laser irradiation alone increased cell bioavailability. |
| [33] | Diode | Saos-2 | 2018 Tani, et al. | 635 or 808 nm 0.378 J/cm2 CW 30 s | Laser irradiation caused no differences in viability at 24 h. Laser irradiation increased expression of RUNX2, ALPL, and osteopontin (SPP1) on 7 days and calcification on 18 days by activation of Akt signaling. |
| [34] | Diode | Saos-2 | 2008 Stein, et al. | 670 nm 1 or 2 J/cm2 CW 30 s or 1 min | Cell viability, alkaline phosphatase activity, and the expression of osteopontin and collagen type I mRNA were slightly enhanced in cells irradiated with 1 J/cm2. |
| [35] | Diode | MG-63 | 2009 Saracino, et al. | 910 nm 6.7 J/cm2 30 kHz 5 min | Laser irradiation decreased cell growth, induced expression of TGFB2, BMP-4, and BMP-7, type I collagen, ALPL, and osteocalcin, and increased the size and the number of calcium deposits. |
| [36] | Diode | MG-63 | 2012 Huang, et al. | 920 nm 5, or 10 J/cm2 50–60 Hz 2.5 or 5 s | Laser irradiation promoted cell adhesion at 12 h and cell viability at 1 and 12 h. Laser irradiation reduced the expression of the lipopolysaccharide (LPS)-induced inflammatory markers iNOS (INOS), tumour necrosis factor α (TNFA), and IL1B and increased the expression of phospho-ERK. |
| [37] | Diode | MG-63 | 2014 Huertas, et al. | 940 nm 0.5, 1, 1.5, or 2 W/cm2 70 mW 1, 2, 3, 4, or 5 J | At 24 h culture, cell proliferation was increased. |
| [38] | Diode | MG-63 | 2013 Incerti Parenti, et al. | 915 nm 2 J/cm2 CW 17, 31, or 157 s | Laser irradiation did not interfere in cell viability and proliferation. |
| [39] | Diode | MG-63 | 2014 Medina-Huertas, et al. | 940 nm 1, 1.5 W/cm2 3, 4 J 10.6, 12.96, 14.7, or 19.31 s | ALP activity was increased significantly at 24 h by irradiation at 1 W/cm2 and 3 J. CD54, CD86, and HLA-DR were decreased at 24 h. |
| [40] | Diode | MG-63 | 2015 Manzano-Moreno, et al. | 940 nm 1, or 1.5 W 3 or 4 J | Laser irradiation significantly increased gene expression of RUNX2, SP7, COL1A, ALPL, BMP2, and TGFB1 at 24 h. |
| [41] | Diode | hFOB 1.19 | 2013 Pyo, et al. | 808 ± 3 nm 1.2, 2.4, or 3.6 J/cm2 CW 15 s at 0, 24, 48 h | Laser irradiation on hypoxic-cultured osteoblast stimulates osteoblast differentiation and proliferation at 24 and 72 h. Laser irradiation significantly increased expression of BMP-2, osteocalcin, Type 1 collagen, and TGFB1. Type 1 collagen expression were significantly decreased at 48 and 72 h. |
| [42] | Diode | hFOB 1.19 | 2013 Jawad, et al. | 940 nm 22.92, 45.85, 68.78, 91.79, or 137.57 J/cm2 CW 3 or 6 min/day for 7 days | Laser irradiation significantly increased proliferation and ALP activity on day 3 and 7. Osteocalcin activity was significantly increased on day 7. |
| [43] | Diode | HHOB-c; Human osteogenic cells | 2015 Walter, et al. | 670 nm 280 mW 60 s CW | Laser irradiation increased the viability of cells, but was significant only in the experimental approach with pamidronate. |
| [44] | Diode | hFOB 1.19 | 2016 Shin, et al. | 808 ± 3 nm 1.2 J/cm2 CW 15 s 3 times at 1, 24, 48 h | Laser irradiation significantly increased cell viability at 72 h. Expression of RANKL and M-CSF (CSF1) were significantly increased at 72 h. |
| [45] | Diode | Human osteoblasts cell line (ATCC® CRL-11372) | 2017 Bolukbasi Ateş, et al. | 635 or 809 nm 0.5, 1, or 2 J/cm2 CW 10, 20, or 40 s | Viability was significantly increased at 48 and 72 h. Expression of COL1A was significantly increased by 1 and 2 J/cm2 at day 14. Expression of BGLAP was significantly decreased by 0.5, 1 and 2 J/cm2 at day 14. |
| [46] | Diode | Human osteoblasts cell line (ATCC® CRL-11372) | 2018 Ateş, et al. | 809 nm 0.5, 1, or 2 J/cm2 CW 10, 20, or 40 s | Laser irradiation at 2 J/cm2 significantly increased cell viability at 24 h. ALP activity was significantly enhanced on day 7. Mineralization was significantly increased on day 14. The expression of ALPL, COL1A, and BGLAP was significantly increased on day 7 and/or 14. |
| [47] | Diode | hFOB 1.19 | 2018 Jeong, et al. | 808 ± 3 nm 1.2 J/cm2 CW 15 s | Combined application of rhBMP-2 and laser irradiation was more effective than application of either modality alone. Expression of RANKL, OPG, and M-CSF in hFOB cells were increased following application of rhBMP-2 and laser irradiation. The expression of TGFB1, BMP2, collagen type I, and osteopontin were increased following combined application of rhBMP-2 and laser irradiation. |
| [48] | Diode | Osteo-1; Rat calvarial osteoblast-like cells | 2006 Fujihara, et al. | 780 nm 3 J/cm2 CW 12 s | Irradiation significantly increased cell proliferation with and without dexamethasone. |
| [49] | Diode | Rat primary calvarial osteoblastic cells | 2006 Fukuhara, et al. | 905 nm 1.25, 3.75, or 6.25 J/cm2 150, 450, or 750 s Every day for 1–3 weeks | Irradiation energy of 3.75 J/cm2 induced an increased number of cells at day 3 and the greatest bone formation at day 21. Low-energy laser irradiation increased Runx2 expression and ALP-positive colonies. FACS data demonstrated a higher proportion of cells in the G2/M phase of the cell cycle 12 h after irradiation compared with the control. |
| [50] | Diode | Rat primary calvarial osteoblastic cells | 2007 Shimizu, et al. | 830 nm 3.82 J/cm2 CW 10 min | Irradiation increased bone nodule formation at day 24 post-irradiation which is partly mediated by insulin-like growth factor-1 (IGF-I) expression. |
| [51] | Diode | Rat primary calvarial osteoblastic cells | 2009, Xu, et al. | 650 nm 1.14 or 2.28 J/cm2 6000 Hz 5 or 10 min | Laser irradiation may directly promote osteoblast proliferation on day 3 and differentiation. Irradiation significantly downregulated RANKL and upregulated OPG, downregulating the RANKL:OPG mRNA ratio in osteoblasts. |
| [52] | Diode | Rat primary calvarial osteoblastic cells | 1998 Ozawa, et al. | 830 nm 3.82 J/cm2 CW 10 min | Laser irradiation significantly stimulated cellular proliferation, ALP activity, and osteocalcin gene expression thereafter. Laser irradiation at earlier stages of culture significantly stimulated bone nodules formation in the culture dish on day 21. |
| [53] | Diode | Rat primary calvarial osteoblastic cells | 2001 Ueda and Shimizu | 830 nm 0.48–3.84 J/cm2 CW or pulsed (1,2, 8 Hz) 1.25–10 for 2.5–20 min | Both CW and pulsed irradiation significantly enhanced cell proliferation, bone nodule formation, ALP activity, and Alpl gene expression as compared with the nonirradiated group. |
| [54] | Diode | Rat primary calvarial osteoblastic cells | 2003 Ueda and Shimizu | 830 nm 0.48–3.84 J/cm2 CW or pulsed (1, 2, and 8 Hz) 1.25–10 for 2.5–20 min | Laser irradiation on day 1 at all conditions significantly stimulated cellular proliferation on day 6, 9, and 12 as compared with the controls. |
| [55] | Diode | Primary human osteoblast-like cells from mandibular | 2005 Khadra, et al. | 830 nm 1.5 and 3 J/cm2 CW For 3 consecutive days | Greater cell proliferation in the irradiated groups was observed first after 96 h. Osteocalcin synthesis and TGF-b1 production were significantly greater on the samples exposed to 3 J/cm2 |
| [56] | Diode | Rat primary calvarial osteoblastic cells | 2020 Cardoso, et al. | 660 or 808 nm 5, 8.3 J/cm2 CW 3 and 5 s | Laser irradiation at both wavelengths promoted cell proliferation and wound healing. ALP activity and mineralization were significantly increased. |
| [57] | Diode | Primary human osteoblast-like cells from alveolar | 2010 Petri, et al. | 780 nm 3 J/cm2 CW 9 min on day 3 and 7 | Laser irradiation did not influence culture growth, ALP activity, and mineralized matrix formation. Gene expression of ALPL, BGLAP, IBSP, and BMP7 was higher in laser-treated cultures, while RUNX2, SPP1, and OPG (TNFRSF11B) were lower on day 14. |
| [58] | Diode | Rat primary calvarial osteoblast-like cells | 2013 Emes, et al. | 808 nm 1.5 J/cm2 CW 90 s | Laser irradiation did not affect cell proliferation and ALP activity. |
| [59] | Diode | Primary human osteoblast-like cells from femur | 2019 Morsoleto, et al. | 808 nm 2 J/cm2 5 s Every day for 8 days | Laser irradiations on 1–8 days enhanced cell viability and matrix mineralization on day 18. |
| [60] | Diode | A mouse OFCOL Ⅱ cell line | 2008 Pires Oliveira, et al. | 830 nm 3 J/cm2 CW 36 s | Cellular viability was significantly increased at 24, 48, and 72 h after irradiation. Intense grouping of mitochondria in the perinuclear region was observed at 24 and 48 h. Changes from a filamentous to a granular appearance in mitochondrial morphology and mitochondria distributed throughout the cytoplasm were observed 72 h. |
| [61] | Diode | Osteoblasts from rat bone marrow stem cells | 2000 Dortbudak, et al. | 690 nm 1.6 J/cm2 CW 60 s 3 times on day 3, 5, and 7 | Irradiations 3 times on day 3, 5, and 7 significantly enhanced more fluorescent bone deposits than the nonirradiated cultures. |
| [62] | Diode | Primary human osteoblast-like cells from alveolar | 2011 Grassi, et al. | 920 nm 3 J/cm2 CW 60 s | Laser irradiation significantly enhanced Alp activity on day 7 and 14 and mineralization after 5 weeks. Laser irradiation decreased Runx2 and Sp7 mRNA at 3, 6, and 12 h. |
| [63] | Diode | Primary human osteoblast-like cells from mandibular | 2018 Mergoni, et al. | 915 nm 5, 15, and 45 J/cm2 CW 4, 12, 36, 41.7, 125, and 375 s Every day for 3 or 6 days | Irradiation for 6 days significantly increased bone deposits 3 weeks after irradiation. |
| [64] | Diode | Mouse primary calvarial osteoblasts | 2010 Hirata, et al. | 805 nm 2–12 J/cm2 CW 2 min | Irradiation stimulated BMP2-induced phosphorylation of Smad1/5/8 and Bmp2 expression, but had no effect on the expression of inhibitory Smads6, Smad7, Bmp4, or insulin-like growth factor 1. Laser irradiation enhanced Smad-induced Id1 reporter activity and BMP-induced transcription factors such as Id1, Osterix, and Runx2. Laser irradiation also stimulated BMP-induced expressions of type I collagen, osteonectin, and osteocalcin mRNA. |
| [75] | Nd:YAG | Saos-2 | 2006 Arisu, et al. | 1064 nm 20, 60, 80, and 120 mJ 0.2, 0.6, 0.8, 0.9, 1.2, 1.6, 1.8, 2.4, and 3.6 W Pulsed 10, 15, 20, and 30 Hz 10 s | Irradiation had a stimulatory effect on the cell viability and proliferation at 7, 14, and 21 days. |
| [76] | Nd:YAG | Saos-2 | 2010 Chellini, et al. | 1064 nm 1.5 J/cm2 Pulsed 50 and 70 Hz 1.4 W 20 mJ 10 s | Laser irradiation did not affect cell viability but significant increased proliferation at 48 h. Laser irradiation significantly induced the expression of ALPL, RUNX2, and SPP1 on day 7. Laser irradiation increased the intracellular Ca2+ levels through the activation of transient receptor potential 1 (TRPC1) ion channels. |
| [77] | Nd:YAG | Saos-2 | 2018 Kara, et al. | 1064 nm Pulsed 5, 10, 20, and 30 Hz 0.5, 1, 2, and 3 W 100 mJ 30 s | The proliferation rates on day 4 increased as the number of applications increased, especially in those cases in which the irradiation was performed 2 or 3 times more. |
| [78] | Nd:YAG | Saos-2 | 2019 Tsuka, et al. | 1064 nm 10.34 J/cm2 10 pps 0.3 W 30 mJ 60 s | Laser irradiation accelerated migration of cells until 24 h, significant enhancement of ATP production. |
| [79] | Nd:YAG | Saos-2 | 2020 Tsuka, et al. | 1064 nm 5.17, 17.23, 34.47, and 51.7 J/cm2 Pulsed 20–30 Hz 0.6, 2.0, 4.0, and 6.0 W 15 s | Laser irradiation significantly increased expression of ALPL, RANKL, TNFRSF11B, and RANKL/OPG ratio at 24 h. |
| [80] | Nd:YAG | MC3T3-E1 | 2010 Kim, et al. | 1064 nm 1.5, 3, and 5 J/cm2 15 pps 0.75 W 4–12 s | Laser irradiation significantly decreased cell proliferation at day 3, but significantly increased ALP activity on day 3, 7, and 14. Laser irradiation highly intensified calcium deposition at all fluences on day 12 and significantly enhanced mineralization on day 21. Laser irradiation significantly increased expression of Bmp2, Cbfa1, SP7, Dlx5, Igf1, and Vegf. |
| [82] | Er:YAG | Saos-2 | 2004 Schwarz, et al. | 2940 nm 5.08, 7.62, 10.16, and 12.7 J/cm2 Pulsed 10 Hz 40, 60, 80, and 100 mJ 10 s | Mitochondrial activity increased significantly with decreasing energy settings and increasing distances. |
| [83] | Er:YAG | MC3T3-E1 | 2010 Aleksic, et al. | 2940 nm 0.7, 1.0, 1.4, 2.1, 2.9, 3.1, 3.6, 4.3, 4.7, 6.4, 6.7, 8.6, 10.8, 12.9, 15.1, and 17.2 J/cm2 Pulsed 10, 20, 30, 40, and 50 Hz 23, 39, 50, and 68 mJ 30, 60, 90, and 120 s and 2.5, 3, 3.5, and 4 min | Significantly higher proliferation was also observed in laser-irradiated MC3T3-E1 cells at a fluence of approximately 1.0–15.1 J/cm2, whereas no increase in lactate dehydrogenase (LDH) activity was observed. Low-level Er:YAG irradiation induced phosphorylation of extracellular signal-regulated protein kinase (MAPK/ERK) 5–30 min after irradiation. |
| [84] | Er:YAG | Rat primary calvarial osteoblast-like cell, MC3T3-E1 | 2020 Niimi, et al. | 2940 nm 2.2, 3.3, and 4.3 J/cm2 Pulsed 20 Hz 17.6, 26.4, and 34.5 mJ 60 s | Calcification and Bglap expression were significantly increased after Er:YAG laser irradiation at 3.3 J/cm2. Laser irradiation at 3.3 J/cm2 caused upregulation of inflammation-related genes, downregulation of Wisp2, and enrichment of inflammation-related and Notch signaling gene sets. |
| [88] | CO2 | Rat osteoblast-like ROS17/2.8 cells | 2011 Sasaki, et al. | 0.357, 0.715, 1.07, and 1.43 J/cm2 0.5, 1, 1.5, and 2 W 20 s | Ibsp mRNA levels were increased at 12 h after irradiation at /.1.43 J/cm2. |
| Reference No. | Laser | Cell | Year Author | Irradiation Protocol | Major Finding |
|---|---|---|---|---|---|
| [43] | Diode | Primary human gingival fibroblast cells (HGF cells) | 2015 Walter et al. | 670 nm 280 mW CW 60 s | Laser irradiation nonsignificantly increased cell viability compared to nonirradiated control HGFs. In bisphosphonate-treated HGFs, laser irradiation significantly increased cell viability compared to the control. |
| [94] | Diode | Lipopolysaccharide (LPS)-challenged human gingival fibroblast cells (HGF cells) | 2000 Sakurai et al. | 830 nm 0.95–6.32 J/cm2 CW 3–20 min | Laser irradiation suppressed LPS-induced PGE2 production by reducing cyclooxygenase (COX)-2 mRNA expression. |
| [95] | Diode | LPS-challenged human gingival fibroblast cells (HGF cells) | 2000 Takema et al. | 830 nm 7.90 J/cm2 CW 10 min | Plasminogen activator activity was dramatically elevated by LPS in cultured medium of HGF cells, which was significantly inhibited by laser irradiation in a dose-dependent manner. |
| [96] | Diode | LPS-challenged human gingival fibroblast cells (HGF cells) | 2001 Nomura et al. | 830 nm 3.95–7.90 J/cm2 CW 10 min | Interleukin (IL)-1β was dramatically elevated by LPS in cultured medium of HGF cells, which was significantly inhibited by laser irradiation in a dose-dependent manner. |
| [97] | Diode | Human gingival fibroblast cell line (LMF) | 2001 Almeida-Lopez et al. | 670, 780, 692, or 782 nm 2.0 J/cm2 CW | Laser irradiation promoted cell proliferation in vitro. Shorter irradiation time resulted in higher proliferation. |
| [98] | Diode | Continuous cell line; Ethics Committee 64/99-Piracicaba Dental School | 2012 Basso et al. | 730 ± 3 nm 0.5, 1.5, 3.0, 5.0, or 7.0 J/cm2 CW 40, 120, 240, 400, or 560 s | 0.5 and 3.0 J/cm2 laser irradiation significantly increased cell metabolism, cell number, and cell migration. |
| [99] | Diode | Human gingival fibroblast cell line (HGF3-PI 53) | 2013 Frozanfar et al. | 810 nm 4.0 J/cm2 CW 32 s for 3 consecutive days | Significant increase in cell proliferation was observed on day 2 and 3. Expression of collagen type 1 gene was dramatically increased on day 3. |
| [100] | Diode | Primary human gingival fibroblast cells (HGF cells) | 2008 Saygun et al. | 685 nm 2.0 J/cm2 CW 140 s for 1 or 2 days (one irradiation per day) | Cell proliferation was promoted in both single-dosed and double-dosed group. Single dose significantly promoted basic fibroblast growth factor (bFGF) and insulin-like growth factor-1 (IGF-1) compared to control. Double dose significantly promoted bFGF, IGF-1, and receptor of IGF-1 (IGFBP3) compared to control. None of the parameters showed significant difference between single-dosed and double-dosed group. |
| [101] | Diode | LPS-treated primary human gingival fibroblast cells (HGF cells) | 2015 Basso et al. | 780 nm 0, 0.5, 1.5, or 3.0 J/cm2 CW 40–240 s | Laser irradiation at 1.3 and 3.0 J/cm2 decreased tumor necrosis factor α (TNFA), IL6, and IL8 gene expression, which were induced by LPS. |
| [102] | Diode | Primary human gingival fibroblast cells (HGF cells) | 2012 Hakki et al. | 940 nm 6.0, 15.0, or 20.0 J/cm2 CW 20 s | No significant difference was observed between laser and control group in proliferation experiment. Laser irradiation at 6, 15, and 20 J/cm2 significantly increased IGF, VEGF, and transforming growth factor (TGF)-β (TGFB) mRNA expressions. Collagen type Ⅰ mRNA expression was enhanced by 6.0 J/cm2 irradiation. |
| [103] | Diode | Primary human gingival fibroblast cells (HGF cells) | 2009 Damante et al. | 780 nm 3.0 or 5.0 J/cm2 CW 3 and 5 s | Production of bFGF was significantly higher in laser-treated group. |
| [104] | Diode | Primary human gingival fibroblast cells (HGF cells) | 2001 Kreisler et al. | 810 nm 24.64–492.8 J/cm2 CW 60–240 s | Laser irradiation caused significant reduction in cell numbers. Exposure time was more relevant to cell reduction than power output. |
| [105] | Diode | Fibroblast cell line (NIH-3T3) | 2002 Pereira et al. | 904 nm 3.0-5.0 J/cm2 CW 8–24 s for 1–6 days | Cell numbers were about 3- to 6-fold higher in laser-irradiated (3.0 and 4.0 J/cm2) culture compared to control. Irradiation at 5.0 J/cm2 had no significant effect on cell growth. 3.0 J/cm2 irradiation increased cell growth without affecting procollagen synthesis. |
| [106] | Diode | Fibroblast cell line (NIH-3T3) | 2016 Sassoli et al. | 635 ± 5 nm 0.3 J/cm2 CW | Laser irradiation inhibited TGF-β- induced fibroblast-myoblast transition. Upregulation of matrix metalloproteinase (MMP)-2 and MMP-9 and downregulation of tissue inhibitors of metalloproteinase (TIMP)-1 and TIMP-2 was shown in laser-treated group. |
| [108] | Diode | Human gingival fibroblast cell line (FMM1) | 2004 Marques et al. | 904 nm 3.0 J/cm2 CW 24 s | Laser irradiation caused ultrastructural changes. Procollagen synthesis was unaffected, but significant reduction in the amount of protein was observed in the medium conditioned by irradiated cells. |
| [76] | Nd:YAG | Fibroblast cell line (NIH-3T3) | 2010 Chellini et al. | 1064 nm 1.5 J/cm2 Pulsed 50, 70 Hz 10 s | Proliferation and cell viability was not significantly affected by laser irradiation. Type 1 collagen expression was significantly induced by 20 mJ/50 Hz laser irradiation. |
| [109] | Nd:YAG | Primary human skin fibroblast cells (HSF cells) | 1983 Castro et al. | 1060 nm 1.1 × 103–2.3 × 103 J/cm2 Pulsed | Significant reduction in DNA synthesis and collagen production was observed at 1.7 × 103 J/cm2 irradiation. At 2.3 × 103 J/cm2, suppression of DNA synthesis was accompanied by cell nonviability. Collagen production was inhibited, while DNA synthesis was unaffected with 1.1 × 103 J/cm2 irradiation. |
| [110] | Nd:YAG | Primary human skin fibroblast cells (HSF cells) | 1984 Abergel et al. | 1064 nm 1.2 × 103–4.7 × 103 J/cm2 Pulsed 3–12 s | Collagen production and DNA replication was significantly decreased by laser irradiation. |
| [113] | Nd:YAG | Human normal epidermal keratinocyte cell line (HaCaT)/keratinocyte-conditioned medium (KCM) stimulated human dermal fibroblast cells (HDF cells) | 2019 De Filippis et al. | 1064 nm 2.0, 4.0, 6.0, or 8.0 J/cm2 Pulsed | Expression of aquaporins, filaggrin, TGase, and HSP70 was upregulated in HaCaT cells by laser irradiation. In HDF cells stimulated by KCM, reduction in MMP-1 and increase in procollagen, collagen type1, and elastin was induced by laser irradiation. |
| [115] | Nd:YAG | Primary human skin fibroblast cells (HSF cells) | 2010 Dang et al. | 532 or 1064 nm 1.5 J/cm2 Pulsed Irradiated twice | Both lasers upregulated collagen synthesis and gene expression of TIMPs expression, but downregulated MMPs mRNA expression at 24 and 48 h postirradiation. TGFB mRNA expression was promoted by 1064 nm laser. Gene expression of HSP70 and IL6 were promoted by 532 nm laser. |
| [118] | Er:YAG | Primary human gingival fibroblast cells (HGF cells) | 2005 Pourzarandian et al. | 2940 nm 1.68–5.0 J/cm2 Pulsed 20 Hz | Faster cell growth was observed in laser-treated cultures. The optimal energy was found to be 3.37 J/cm2. |
| [119] | Er:YAG | Primary human gingival fibroblast cells (HGF cells) | 2005 Pourzarandian et al. | 2940 nm 3.37 J/cm2 Pulsed 20 Hz | Laser irradiation significantly increased PGE2 production by HGFs. mRNA expression of COX2 was significantly increased after laser irradiation. Inhibition of COX-2 completely suppressed PGE2 synthesis induced by laser irradiation. |
| [120] | Er:YAG | Primary human gingival fibroblast cells (HGF cells) | 2015 Ogita et al. | 2940 nm 1.65, 2.11, or 2.61 J/cm2 Pulsed 30 s | A significant cell proliferation without cell damage was shown on day 3 after irradiation. mRNA and protein expression of galectin-7 was increased after laser irradiation. |
| [121] | Er:YAG | Primary human gingival fibroblast cells (HGF cells) | 2018 Kong et al. | 2940 nm 3.6, 4.2, 4.9, 6.3, 8.1, or 9.7 J/cm2 Pulsed 20 or 30 Hz 20 or 30 s | Laser irradiation at 6.3 J/cm2 enhanced maximal cell proliferation, however, lactate dehydrogenase (LDH) release was observed on day 3 after irradiation. Laser irradiation affected cell cycle and increased proliferating cells. Transient damage was observed at 3 h. mRNA expression of HSP70 family was increased by laser irradiation. Inhibition of thermosensory transient receptor potential channels suppressed laser-induced cell proliferation. |
| [122] | Er:YAG/ Er,Cr:YSGG | Human gingival fibroblast cell line (NCBI code: C-165) | 2016 Talebi-Ardakani et al. | 2940 nm/2780 nm 1 W/0.5 W Pulsed 10 Hz 10 or 30 s | Significant increasement in cell proliferation was shown in all laser-irradiated groups at 24 and 48 h. |
| [123] | Er:YAG | Primary human gingival fibroblast | 2020 Tsuka et al. | 2940 nm 30–150 mJ Pulsed 20 Hz | Gene expression of COX2, IL1B, TNFA, BMP2, and BMP4 significantly increased in laser-irradiated group in a dose-dependent manner compared to the control group. COX2 gene expression showed significant increase in the centrifugal load group compared to control group, whereas gene expression of COX2, IL1B, TNFA, BMP2, and BMP4 was significantly higher in the laser and centrifugal loaded group than in the centrifugal load group. |
| [128] | CO2 | Keloid and normal fibroblast cell line | 2000 Nowak et al. | 10,600 nm 2.4, 4.7, or 7.3 J/cm2 Pulsed 16 Hz | Population doubling time for keloid fibroblasts was shortened by 2.4 and 4.7 J/cm2 irradiation. Secretion of bFGF was increased by 4.7 J/cm2 laser irradiation in both keloid and normal group; significant in keloid group. TGF-β1 was suppressed by laser irradiation in both keloid and normal group; maximal effect occurred at 4.7 J/cm2. |
| [129] | CO3 | Primary human dermal fibroblast cells (HDF cells) | 2017 Shingyochi et al. | 10,600 nm 0.1, 0.5, 1.0, 2.0, or 5.0 J/cm2 CW 2–40 s | Cell proliferation and migration were promoted after 1.0 J/cm2 irradiation. Akt, ERK, and JNK pathways were activated after 1.0 J/cm2 irradiation. Inhibition of Akt, ERK, or JNK pathways suppressed cell proliferation and migration induced by laser irradiation. |
| [130] | CO4 | Primary fibroblast cells | 1983 Apfelberg et al. | 10,600 nm 5 W CW 1–3 s | CO2 laser does not produce a greater incidence of malignant transformation that normal controls. |
| Reference No. | Laser | Cell | Year Author | Irradiation Protocol | Major Finding |
|---|---|---|---|---|---|
| [132] | Diode | Human periodontal ligament cells (hPDLCs) | 2013 Wu, et al. | 660 nm 1, 2, or 4 J/cm2 CW 66, 132, or 264 s | Laser irradiation significantly promotes proliferation of hPDLCs. Laser irradiation enhanced the mRNA expression of osteogenic maker genes. |
| [133] | Diode | Periodontal ligament cell line | 2014 Huang, et al. | 670 nm 5, or 10 J/cm2 CW 2.5, or 5 s | Laser irradiation significantly decreased the protein expressions of inflammatory makers. The protein expression of osteocalcin was significantly increased in laser-irradiated cells. |
| [134] | Diode | Human periodontal ligament cells (hPDLCs) | 2010 Mayahara, et al. | 830 nm 3.82 J/cm2 CW 10 min | Laser irradiation significantly inhibited cyclooxygenase (COX)-2 and cytosolic phospholipaseA2-α (PLA2G4A) mRNA expression. |
| [135] | Diode | Stretched Human periodontal ligament cells (hPDLCs) | 1995 Shimizu, et al. | 830 nm 346–1152 J/cm2 CW 0, 3, 6, or 10 min | The protein expression of prostaglandin E2 was significantly decreased in laser-irradiated cells. Laser irradiation was tended to decrease the protein expression of interleukin (IL) 1-β. |
| [136] | Diode | Human periodontal ligament cells (hPDLCs) | 1997 Ozawa, et al. | 830 nm 3.95–7.90 J/cm2 CW 10 or 20 min/day | Laser irradiation in a dose-dependent manner significantly inhibited the plasminogen activator (PA) activity in hPDLCs with stretching. |
| [137] | Diode | Periodontal ligament cell line | 2013 Huang, et al. | 920 nm 5–10 J/cm2 CW 2.5, or 5 s | The mRNA and protein expressions of inducible NO synthase (iNOS), TNF-a, IL-1 was decreased in lipopolysaccharide-exposed periodontal ligament cells after laser irradiation. The protein expression of pErk was significantly increased in the laser-irradiated cells compared with the nonirradiated cells. |
| [138] | Diode | Human periodontal ligament fibroblasts | 2010 Choi, et al. | 810 nm 1.97, 3.94, or 5.91 J/cm2 CW 10, 20, or 30 s | Proliferation, alkaline phosphatase activity, and phosphorylated ERK level were significantly increased in laser-irradiated cell at limited time point. |
| [139] | Diode | Human periodontal ligament fibroblasts | 2020 Dehdashtizadeh, et al. | 810 nm 10 J/cm2 CW 5 s/day | Laser irradiation reduced the protein expression of matrix metalloproteinase (MMP)-8. |
| [140] | Diode | Human periodontal ligament fibroblasts | 2003 Kreisler, et al. | 809 nm 1.96, 3.92, or 7.84 J/cm2 CW 75, 150, or 300 s | The proliferation rates of laser-irradiated culture were significant up to 72 h compared to control culture. |
| [141] | Er:YAG | Human periodontal ligament cells (hPDLCs) | 2020 Lin, et al. | 2940 nm 3.6, 4.2, or 6.3 J/cm2 Pulsed 20 Hz 75, 150, or 300 s | The proliferation, migration, and invasion abilities were induced through the upregulation of galectin-7 after laser irradiation. |
| Reference No. | Laser | Cell | Year Author | Irradiation Protocol | Major Finding |
|---|---|---|---|---|---|
| [43] | Diode | Human umbilical vein endothelial cells (HUVECs) | 2015 Walter, et al. | 670 nm 280 mW CW 60 s | Laser irradiation had a positive effect on cell viability. |
| [145] | Diode | Human endothelial cell line (HECV) | 2019 Amaroli, et al. | 808 nm 57 J/cm2 CW 1 min | Laser-irradiated cells demonstrated higher proliferation rate and increased migration ability. Laser irradiation stimulated mitochondrial oxygen consumption and ATP synthesis in HECV cell. |
| [146] | Diode | Human umbilical vein endothelial cells (HUVECs) | 2013 Protasiewicz, et al. | 808 nm 1.5, 4.5 J/cm2 CW 90 or 270 s | Laser irradiation diminished the pro-inflammatory and procoagulant activity of Interleukin (IL)-1β-stimulated HUVECs. |
| [147] | Diode | Human umbilical vein endothelial cells (HUVECs) | 2003 Schindl, et al. | 670 nm 2–8 J/cm2 CW every 48 h for a period of 6 days | Doses of between 2 and 8 J/cm2 induced statistically significant cell proliferation. |
| [148] | Diode | Human umbilical vein endothelial cells (HUVECs) | 2015 Góralczyk, et al. | 635 nm 2, 4, or 8 J/cm2 2 times with 1-day break | Laser irradiation was significantly increased in proliferation of endothelial cells. Laser irradiation significantly reduced the concentration of soluble VEGF receptor (sVEGFR)-1 in the supernatant. |
| [149] | Diode | Cultured rhesus macaque choroid-retinal endothelial cells (RF/6A) | 2012 Du, et al. | 810 nm 45.86–76.43 J/cm2 CW 1 min | 810 nm diode laser irradiation can induce Hsp70 hyperexpression from 12 to 18 h postirradiation in cultured choroid-retinal endothelial cells without obvious cell death. |
| [76] | Nd:YAG | H-end endothelial cells | 2010 Chellini, et al. | 1064 nm 1.5 J/cm2 Pulsed 50 or 70 Hz 10 s | Vinculin expression in endothelial cells could be observed in the irradiated cells. Laser irradiation did not affect cell viability. |
| [150] | Nd:YAG | Rat aortic endothelial cells | 2017 Masuda, et al. | 1064 nm 100 mJ Pulsed 5 Hz 30 s | Upregulated genes with laser irradiation were related to cell migration and cell structure (membrane stretch, actin regulation, and junctional complexes), neurotransmission, and inflammation. |
| [151] | Nd:YAG | Human umbilical vein endothelial cells (HUVECs) | 2009 Giannelli, et al. | 1064 nm 15 mJ/mm2 Pulsed 70 Hz 1 min | Laser irradiation attenuated intercellular adhesion molecule-1 and vascular cell adhesion molecule expression. |
| Reference No. | Laser | Cell | Year Author | Irradiation Protocol | Major Finding |
|---|---|---|---|---|---|
| [153] | Diode | Cementoblasts | 2017 Bozkurt, et al. | 940 nm 15, 11.4, or 105 J/cm2 CW 60 s/cm2 | The mRNA expression related to only cementoblast and bone morphogenetic protein were increased in laser-irradiated cells. |
| Reference No. | Laser | Cell | Year Author | Irradiation Protocol | Major Finding |
|---|---|---|---|---|---|
| [169] | Diode | Normal human oral keratinocytes (NOKSI) | 2017 Tang et al. | 810 nm 1.0 and 4.0 J/cm2 CW 5 min | Laser treatments induced Human β defensing (HBD)-2 expression in keratinocyte cell line. transforming growth factor (TGF) β-1 pathway was activated by laser irradiation. |
| [170] | Diode | Human gingival epithelial cells (HGECs) | 2014 Ejiri et al. | 904–910 nm 5.7–56.7 J/cm2 Pulsed 30 kHz 1. 3, 5, or 10 min | The laser irradiation significantly increased cell proliferation and [3H]thymidine incorporation at various irradiation time periods. Migration of the irradiated cells was significantly accelerated compared with the nonirradiated control. Laser irradiation induced phosphorylation of MAPK/ERK at 5, 15, 60, and 120 min after irradiation. Stress-activated protein kinases/c-Jun N-terminal kinase and p38 MAPK remained unphosphorylated. |
| [172] | Diode | Human oral squamous epithelial carcinoma cell lines (Ca9-22 and SCC-25) | 2014 Fujimura et al. | 805 ± 20 nm 0.475 W Pulsed (pulse width: 100 ms) 60 s | The mRNA expression of DEL1 was significantly upregulated by laser irradiation (p < 0.01). Lipopolysaccharide (LPS)-induced interleukin (IL)-6 and IL-8 expression was significantly suppressed in the LPS+laser group (p < 0.01). Intercellular adhesion molecule (ICAM)-1 expression was significantly higher in the LPS+laser group than in the LPS only or control groups. Compared with the control, the migration of epithelial cells was significantly increased by diode laser irradiation. |
| Reference No. | Laser | Cell | Year Author | Irradiation Protocol | Major Finding |
|---|---|---|---|---|---|
| [173] | CO2 | Osteocyte-like cells | 2013 Yokose, et al. | 10,600 nm 0.71, 1.42, and 2.83 J/cm2 CW 10 s | The mRNA expression of Sost was decreased and that of Dmp1 was increased in the cells after dose-dependent laser irradiation. |
| [174] | Er:YAG | Osteogenic cells (osteoblast-like cells incubated for 21 days by osteoinduction) | 2020 Ohsugi, et al. | 2940 nm 1.5 and 3.1 J/cm2 Pulsed 20 Hz 30 and 60 s | The mRNA expression of Sost was decreased and that of Mef2c was increased in laser-irradiated cells. |
| Reference No. | Laser | Cell | Year Author | Irradiation Protocol | Major Finding |
|---|---|---|---|---|---|
| [176] | Diode | Osteoclasts | 2006 Aihara, et al. | 810 nm 9.33, 27.99, 55.98, or 93.30 J/cm2 CW 1, 3, 6, or 10 min/day | Laser irradiated cells showed greater amounts of staining compared to non-irradiated cells in immunohistochemistry for receptor activator of NF-kappaB (RANK). The mRNA expression of receptor activator of RANK was upregulated in low-energy irradiated cells. |
| Reference No. | Laser | Cell | Year Author | Irradiation Protocol | Major Finding |
|---|---|---|---|---|---|
| [182] | Diode | Human bone marrow-derived mesenchymal stem cells (hBM-MSCs) human adipose-derived stem cells (hASCs) | 2019 Zare et al. | 630 and 810 nm 0.6, 1.2, or 2.4 J/cm2 10 s | Laser irradiation combined 630 and 810 nm significantly stimulated cell viability, and decreased apoptosis in hBM-MSCs and hASCs. |
| [183] | Diode | Mesenchymal stem cells (MSCs) isolated from femurs and tibias in rat | 2012 Wu et al. | 635 nm 0.5 J/cm2 75 s | Microarray analysis revealed 119 differentially expressed genes after laser irradiation. mRNA expression of Akt1, Ccnd1, and Pik3ca were upregulated and Ptpn6 and Skt17b expression were downregulated. |
| [184] | Diode | Mesenchymal stem cells (MSCs) isolated from femurs and tibias in rat | 2008 Hou et al. | 635 nm 0.5–5.0 J/cm2 CW 75–750 s | Laser irradiation at 0.5 J/cm2 stimulated MSCs proliferation. Laser irradiation at 5.0 J/cm2 increased VEGF and nerve growth factor secretion and dramatically facilitated the differentiation. |
| [33] | Diode | Human mesenchymal stromal cells (hMSCs) | 2018 Tani et al. | 635, or 808 nm 0.378 J/cm2 CW 30 s | Irradiation at 635 nm increased Runx2 and Alpl mRNA expression, and expression of osteopontin and Ki67. |
| [185] | Diode | Mesenchymal stem cells (MSCs) isolated from femurs and tibias in male C2F1 mice | 2013 Giannelli et al. | 635 nm 0.3 J/cm2 CW 10, 26 s | Cell proliferation was increased without change of cell viability. Increase in cell proliferation was associated with the upregulation and activation of Notch-1 pathway. |
| [186] | Diode | Human gingival mesenchymal stem cells (HGMSCs) | 2020 Feng et al. | 808 nm 0.5–4.0 J/cm2 CW | Laser irradiation promoted cell migration but not cell proliferation. Laser irradiation at 1.0 J/cm2 activated mitochondrial ROS after 2 h. |
| [187] | Diode | Bone marrow stromal cells (MSCs) isolated from 3-old female BALB-c mice | 2018 Amaroli et al. | 808 nm 64 J/cm2 CW 60 s | Laser irradiation increased Runx2 and Osterix and decreased Pparγ protein expression. Positive areas of alkaline phosphatase and Arizarin Red S staining were significantly increased after irradiation. |
| [188] | Diode | Bone marrow stem cells (MSCs) isolated from young adult C57Bl/6 mice | 2009 Horvat-Karajz et al. | 660 nm 1.9–11.7 J/cm2 CW 25–75 s | Laser irradiation at 1.9 J/cm2 enhanced cell proliferation, although irradiation at 11.7 J/cm2 suppressed cell proliferation. |
| [189] | Diode | Stem cells from human exfoliated deciduous teeth (SHEDs) | 2019 Ferreira et al. | 660 nm 1–20 J/cm2 CW 1–28 s | Laser irradiation at 5 J/cm2 enhanced cell proliferation. mRNA expression of OCT4, NES, and CD90 was increased, although that of CD105 was decreased after irradiation at 5 J/cm2. |
| [190] | Diode | Human dental pulp stem cells (hDPSCs) | 2019 Garrido et al. | 660 nm 3 and 5 J/cm2 CW 4 and 7 s | Laser irradiation at 3 J/cm2 increased fibronectin expression. hDPSCs irradiated at 5 J/cm2 showed sign of apoptosis and necrosis. |
| [191] | Diode | Human dental pulp stem cells (hDPSCs) | 2020 Yurtsever et al. | 660 nm 0.6, or 1.6 J/cm2 240 or 600 s | Laser irradiation at 1.6 J/cm2 increased mRNA expression of brain-derived neurotrophic factor (BDNF), glial cell line-derived neurotrophic factor (GDNF), matrix-associated protein 2 (MAP2), nuclear receptor-related 1 protein (NURR1), and dopamine transporter (DAT) in hDPSCs. |
| [192] | Nd:YAG | Human bone marrow mesenchymal stem cells (BMSCs) | 2019 Wang et al. | 1064 nm 2, 4, 8, or 16 J/cm2 20 s | Laser irradiation at 2 and 4 J/cm2 promoted proliferation and osteogenesis in BMSCs. Laser irradiation at 16 J/cm2 suppressed proliferation and osteogenesis in BMSCs. |
| [193] | Nd:YAG | Horse bone marrow mesenchymal stem cells (BMSCs) | 2018 Peat et al. | 1064 nm 9.77 J/cm2 Pulsed 10 Hz 10 s | Laser-irradiated BMSCs did not show a difference in viability. Laser-irradiated BMSCs exhibited slightly lower proliferation. Interleukin (IL)10 and VEGF mRNA expression was increased after laser irradiation. |
| [194] | Nd:YAG | Human adipose-derived stem cells (hADSCs) | 2012 Anwer et al. | 532 nm 5–45 J/cm2 CW 30–300 s | Laser irradiation at 5–9.2 J/cm2 increased cell proliferation by increasing mitochondrial activity in hADSCs. Laser irradiation at 28 and 45 J/cm2 decreased cell proliferation in hADSCs. |
| [195] | CO2 | Human adipose-derived stem cells (hADSCs) | 2017 Constantin et al. | 10,600 nm 5, 9, or 10 W Pulsed 2–7 ms/shot | Laser irradiation (output power 9 W, exposure time 4 ms/shot) increased proliferation, mitochondrial ROS, the capacity to restore Δψm after rotenone-induced depolarization and the secretion of matrix metalloproteinase (MMP)-2. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ohsugi, Y.; Niimi, H.; Shimohira, T.; Hatasa, M.; Katagiri, S.; Aoki, A.; Iwata, T. In Vitro Cytological Responses against Laser Photobiomodulation for Periodontal Regeneration. Int. J. Mol. Sci. 2020, 21, 9002. https://doi.org/10.3390/ijms21239002
Ohsugi Y, Niimi H, Shimohira T, Hatasa M, Katagiri S, Aoki A, Iwata T. In Vitro Cytological Responses against Laser Photobiomodulation for Periodontal Regeneration. International Journal of Molecular Sciences. 2020; 21(23):9002. https://doi.org/10.3390/ijms21239002
Chicago/Turabian StyleOhsugi, Yujin, Hiromi Niimi, Tsuyoshi Shimohira, Masahiro Hatasa, Sayaka Katagiri, Akira Aoki, and Takanori Iwata. 2020. "In Vitro Cytological Responses against Laser Photobiomodulation for Periodontal Regeneration" International Journal of Molecular Sciences 21, no. 23: 9002. https://doi.org/10.3390/ijms21239002
APA StyleOhsugi, Y., Niimi, H., Shimohira, T., Hatasa, M., Katagiri, S., Aoki, A., & Iwata, T. (2020). In Vitro Cytological Responses against Laser Photobiomodulation for Periodontal Regeneration. International Journal of Molecular Sciences, 21(23), 9002. https://doi.org/10.3390/ijms21239002






