Reducing Flower Competition for Assimilates by Half Results in Higher Yield of Fagopyrum esculentum
Abstract
1. Introduction
2. Results
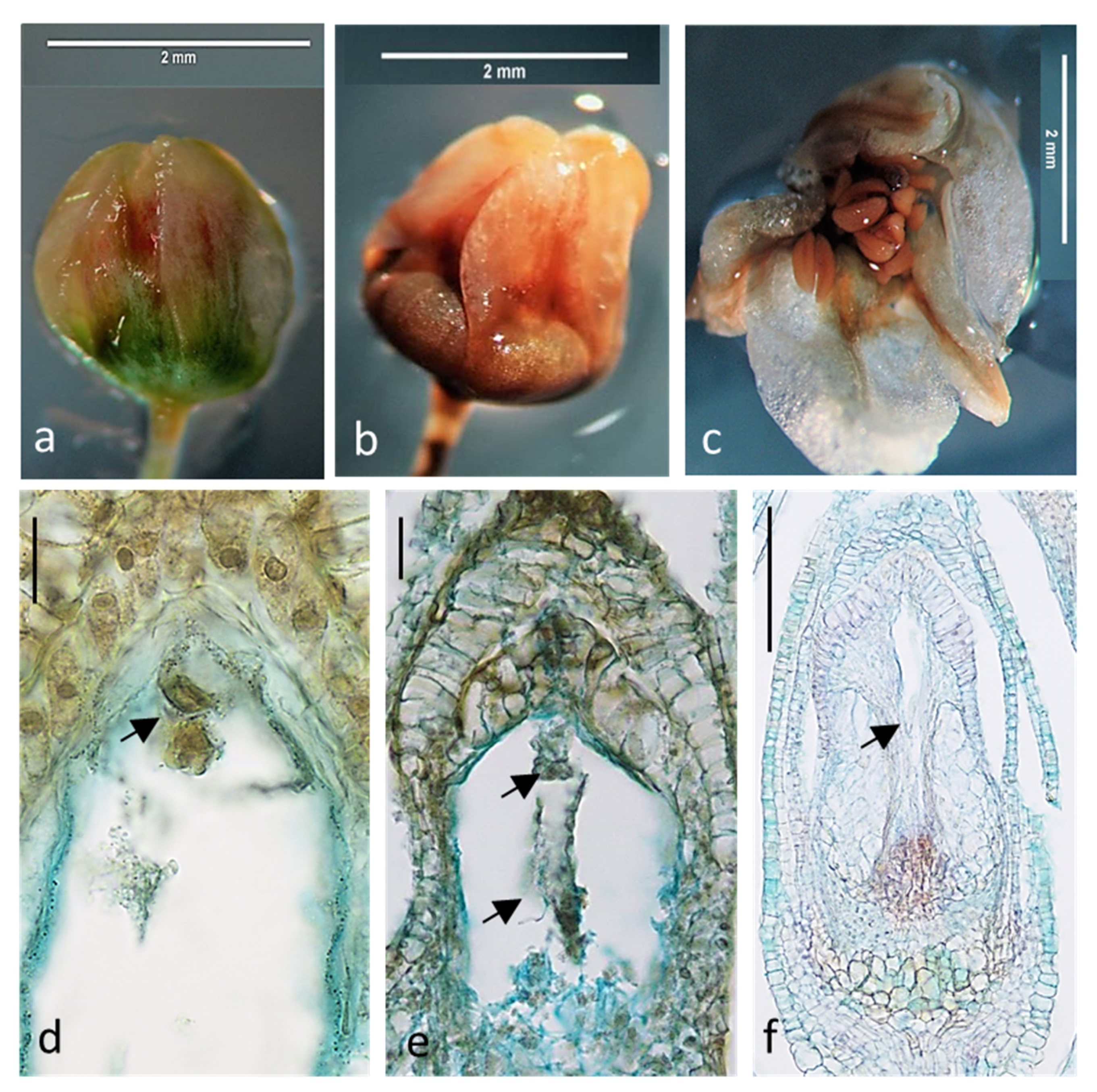
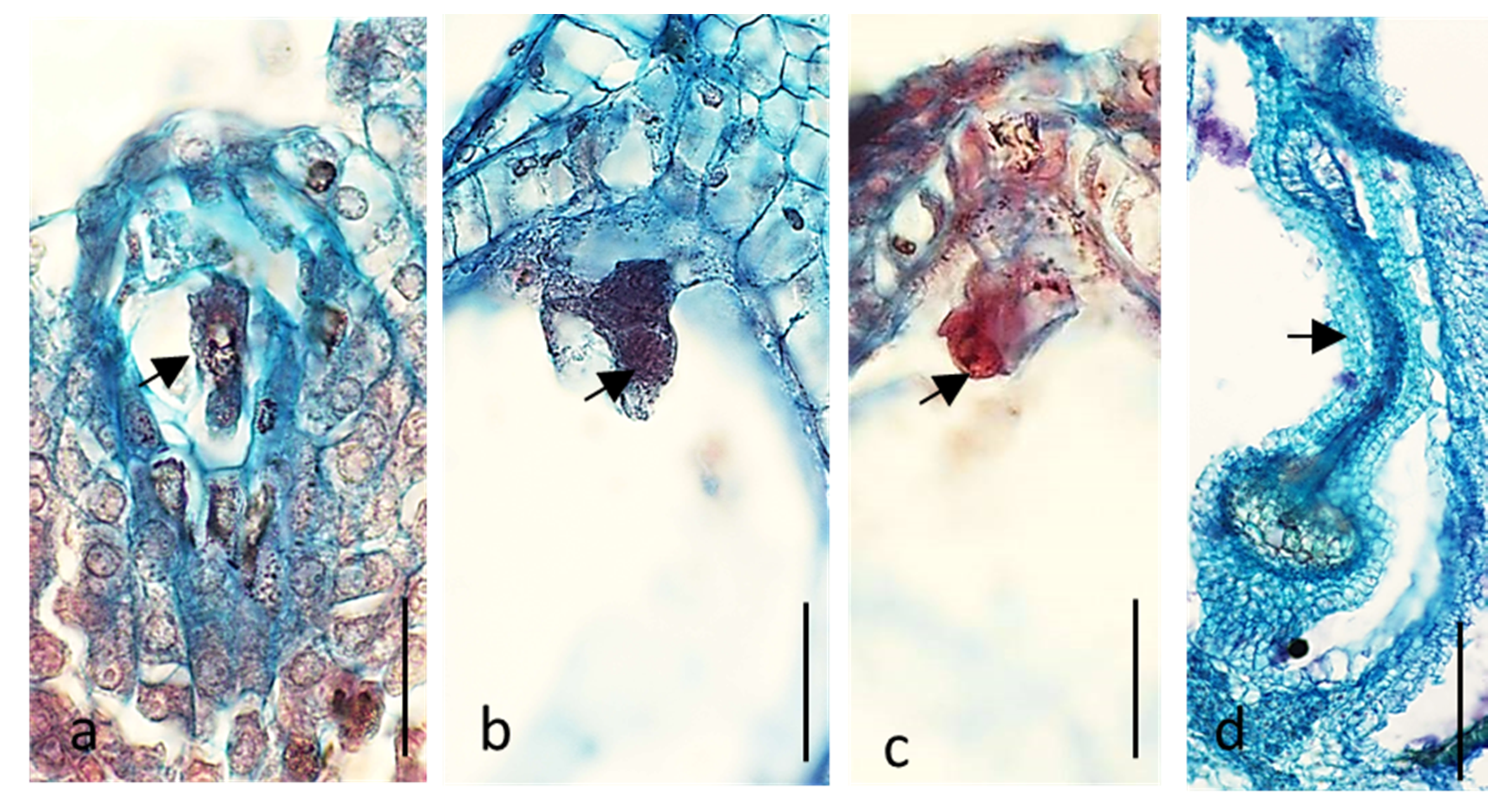
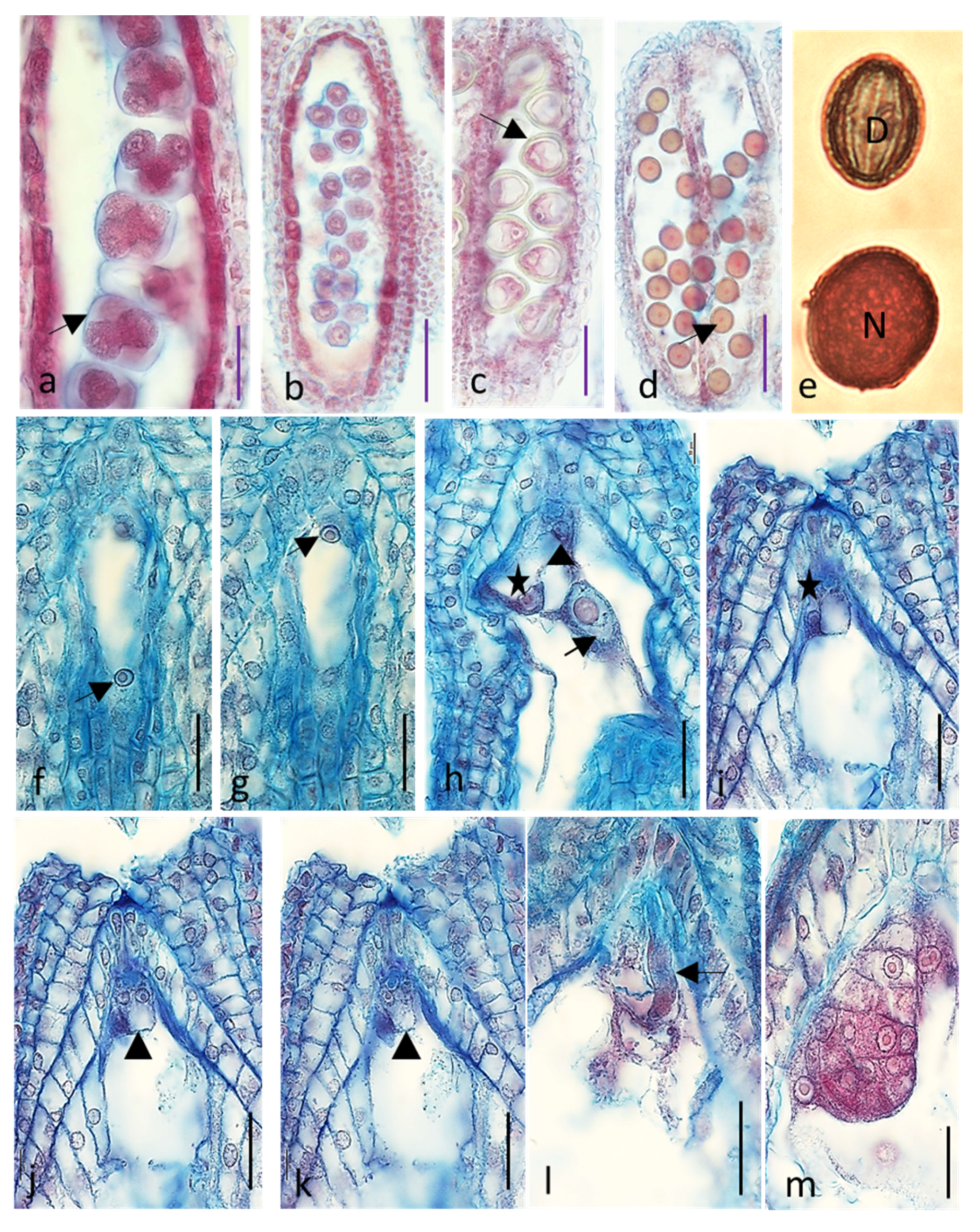
2.1. Embryological Analysis
2.2. Phytohormones
2.3. Yield
3. Discussion
4. Materials and Methods
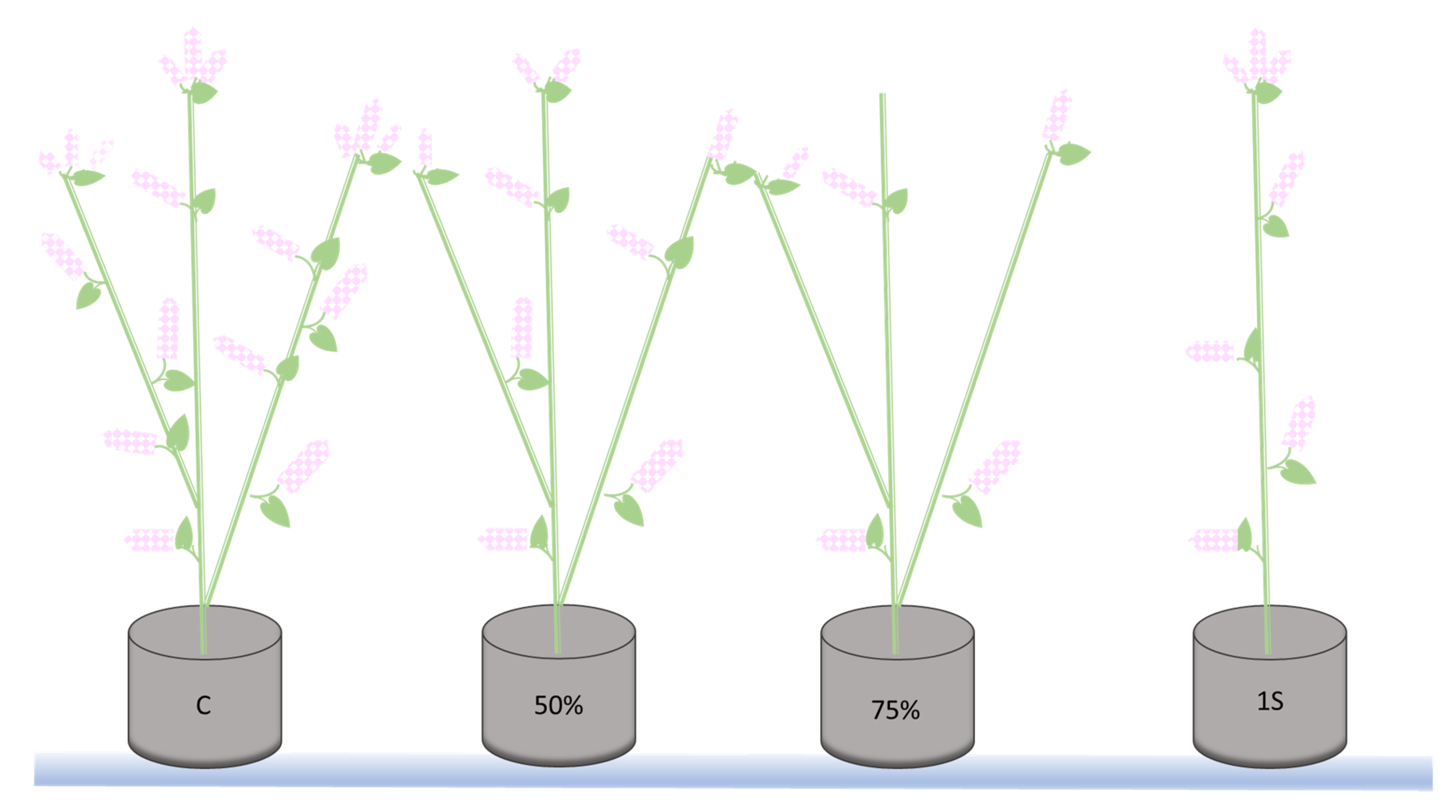
4.1. In Vitro and In Planta Experimental Design
4.1.1. In Vitro Experiment
4.1.2. In Planta Experiment
4.2. Embryological Processes in Flowers in In Vitro and In Planta Experiments
4.3. Phytohormonal Profile Analyses in Flowers in In Planta Experiments
4.4. Yield Related Measurements in In Planta Experiments
4.5. Statistical Analysis
5. Conclusions
- Following the experimental loss of some flowers, a plant initiates compensation processes including: increase in the efficiency of pollen viability by reducing the percentage of degenerated pollen grains; production of additional flowers, but, when the number of flowers turns out to be too high, the plant aborts most of them; increase the percentage of empty seeds if the above-mentioned measures are insufficient; and reduction in the amount of reserve materials accumulated in the seeds, and thereby reduction of seed mass if the other mechanisms prove insufficient.
- The critical point for seed yield is the moment of flowering and fertilization. Flower overproduction and embryo formation are the processes that exhaust the plant reserves. Failure to fill all the seeds will no longer compensate for these losses. Forming empty seeds is therefore, next to flower abortion, another form of crop regulation when the plant has produced too many flowers. Our study confirmed the common observation that the greater is the number of seeds, the smaller is the mass of a single seed.
- Removing 50% of flowers significantly reduces the percentage of defective embryo sacs, which has a direct impact on increasing the yield of mature seeds. However, this relationship was only observed in cv. ‘Korona’. In this cultivar, this effect can be attributed to higher concentration of jasmonic acid, salicylic acid (and its precursor - benzoic acid), which play an important role in attraction of pollinators.
- Plants in single-shoot cultivation finish their vegetation earlier and achieve higher mass of one seed as compared with that of the control. This result confirms our hypothesis that self-finishing plants of common buckwheat, with shorter blooming phase, could deliver higher seed yield.
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ABA | Abscisic acid |
| BA | Benzoic acid |
| CYT | Cytokinins |
| GAs | Active gibberellins (GA1, GA3, GA4, GA5, GA6, GA7) |
| IAA | Indole-3-acetic acid |
| JA | Jasmonic acid |
| SA | Salicylic acid |
| MTS | Mass of thousand seeds |
| 1S | Plant with single main shoot |
| 50% | Plant with 50% of flowers removed |
| 75% | Plant with 75% of flowers removed |
References
- Slawinska, J.; Obendorf, R.L. Buckwheat seed set in planta and during in vitro inflorescence culture: Evaluation of temperature and water deficit stress. Seed Sci. Res. 2001, 11, 223–233. [Google Scholar]
- Cawoy, V.; Lutts, S.; Ledent, J.-F.; Kinet, J.-M. Resource availability regulates reproductive meristem activity, developmental of reproductive structures and seed set in buckwheat (Fagopyrum esculentum). Physiol. Plant. 2007, 131, 341–453. [Google Scholar] [PubMed]
- Morishita, T.; Hara, T.; Hara, T. Important agronomic characteristics of yielding ability in common buckwheat; ecotype and ecological differentiation, preharvest sprouting resistance, shattering resistance, and lodging resistance. Breed Sci. 2020, 70, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Słomka, A.; Michno, K.; Dubert, F.; Dziurka, M.; Kopeć, P.; Płażek, A. Embryological background of low seed set in distylous common buckwheat (Fagopyrum esculentum Moench) with biased morph ratios, and biostimulant-induced improvement of it. Crop Past. Sci. 2017, 68, 680–690. [Google Scholar] [CrossRef]
- Płażek, A.; Hura, K.; Hura, T.; Słomka, A.; Hornyák, M.; Sychta, K. Synthesis of heat-shock proteins HSP-70 and HSP-90 in flowers of common buckwheat (Fagopyrum esculentum) under thermal stress. Crop Pasture Sci. 2020, 71, 760–767. [Google Scholar] [CrossRef]
- Płażek, A.; Słomka, A.; Kopeć, P.; Dziurka, M.; Hornyák, M.; Sychta, K.; Pastuszak, J.; Dubert, F. Effects of high temperature on embryological development and hormone profile in flowers and leaves of common buckwheat (Fagopyrum esculentum Moench). Int. J. Mol. Sci. 2019, 20, 1705. [Google Scholar] [CrossRef]
- Guan, L.M.; Adachi, T. Reproductive deterioration in buckwheat (Fagopyrum esculentum) under summer conditions. Plant Breed. 1992, 109, 304–312. [Google Scholar] [CrossRef]
- Guan, L.M.; Adachi, T. Ultrastructural changes of the mature embryo sac in buckwheat (Fagopyrum esculentum) as a result of high temperature exposure. Cytologia 1994, 59, 237–248. [Google Scholar] [CrossRef]
- Guan, L.; Chen, L.; Adachi, T. Ultrastructural studies of embryo abortion in buckwheat (Fagopyrum esculentum) as a heat-stress. Cytologia 2008, 73, 371–379. [Google Scholar] [CrossRef]
- Guglielmini, A.C.; Forcat, J.I.; Miralles, D.J. The critical period for yield determination in common buckwheat (Fagopyrum esculentum Moench). Eur. J. Agron. 2019, 110, 125933. [Google Scholar] [CrossRef]
- Yabe, S.; Hara, T.; Ueno, M.; Enoki, H.; Kimura, T.; Nishimura, S.; Yasui, Y.; Ohsawa, R.; Iwata, H. Potential of genomic selection in mass selection breeding of an allogamous crop: An empirical study to increase yield of common buckwheat. Front. Plant Sci. 2018, 9, 276. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.P.; Obendorf, R.L. Quantitative assessment of some factors limiting seed set in buckwheat. Crop Sci. 2001, 41, 1792–1799. [Google Scholar] [CrossRef]
- Halbrecq, B.; Romedenne, P.; Ledent, J.F. Evolution of flowering, ripening and seed set in buckwheat (Fagopyrum esculentum Moench): Quantitative analysis. Eur. J. Agron. 2005, 23, 209–224. [Google Scholar] [CrossRef]
- Inoue, N.; Hagiwara, M. Analysis of the yielding process based on the module concept in common buckwheat. Fagopyrum 1999, 16, 73–77. [Google Scholar]
- Inoue, N.; Hagiwara, M.; Kim, H.Y.; Matano, T. A preliminary study of modeling seed production in common buckwheat. Fagopyrum 1998, 15, 35–41. [Google Scholar]
- Matsui, K.; Yasui, Y. Genetic and genomic research for the development of an efficient breeding system in heterostylous self-incompatible common buckwheat (Fagopyrum esculentum). Theor. Appl. Gen. 2020, 133, 1641–1653. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Lv, Q.; Deng, J.; Huang, J.; Cai, F.; Liang, C.; Chen, Q.; Wang, Y.; Zhu, L.; Zhang, X.; et al. Transcriptome analysis reveals key seed-development genes in common buckwheat (Fagopyrum esculentum). Int. J. Mol. Sci. 2019, 20, 4303. [Google Scholar] [CrossRef]
- Fang, X.; Zhang, Y.; Zhang, Y.; Huang, K.; Yang, W.; Li, X.; Zhang, Z.; Wu, K.; Xu, X.; Ruan, R.; et al. De novo transcriptome assembly and identification of genes related to seed size in common buckwheat (Fagopyrum esculentum M.). Breed. Sci. 2019, 69, 487–497. [Google Scholar] [CrossRef]
- Hu, Y.; Zhou, L.; Huang, M.; He, X.; Yang, Y.; Liu, X.; Li, Y.; Hou, X. Gibberellins play an essential role in late embryogenesis of Arabidopsis. Nat. Plants 2018, 4, 289–298. [Google Scholar] [CrossRef]
- Thaler, J.S.; Karban, R.; Ullman, D.E.; Boege, K.; Bostock, R.M. Cross-talk between jasmonate and salicylate plant defense pathways: Effects on several plant parasites. Oecologia 2002, 131, 227–235. [Google Scholar] [CrossRef]
- Wang, Y.H.; Irving, H.R. Developing a model of plant hormone interactions. Plant Signal. Behav. 2011, 6, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Domagalska, M.A.; Sarnowska, E.; Nagy, F.; Davis, S.J. Genetic analyses of interactions among gibberellin, abscisic acid, and brassinosteroids in the control of flowering time in Arabidopsis thaliana. PLoS ONE 2010, 5, e14012. [Google Scholar] [CrossRef] [PubMed]
- Raskin, I. Salicylate, a new plant hormone. Plant Physiol. 1992, 99, 799–803. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, K. Study on buckwheat. Ann. Rep. Fac. Edu. Iwate Univ. 1973, 33, 1–96. [Google Scholar]
- Asako, Y.; Ujihara, A.; Matano, T. Relation between the position of flowers and their flowering of fruiting in common buckwheat. Hokuriku Crop Sci. 1980, 15, 27–30. [Google Scholar]
- Singh, R.J. Plant Cytogenetics, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Dziurka, M.; Janeczko, A.; Juhász, C.; Gullner, G.; Oklestková, J.; Novák, O.; Saja, D.; Skoczowski, A.; Tóbiász, J.; Barna, B. Local and systemic hormonal responses in pepper leaves during compatible and incompatible pepper-tobamo virus interactions. Plant Physiol. Biochem. 2016, 109, 355–364. [Google Scholar] [CrossRef]
- Hura, T.; Dziurka, M.; Hura, K.; Ostrowska, A.; Dziurka, K.; Gadzinowska, J. Wheat and rye genome confer specific phytohormone profile features and interplay under water stress in two phenotypes of triticale. Plant Physiol. Bioch. 2017, 118, 494–509. [Google Scholar] [CrossRef]
- Płażek, A.; Dubert, F.; Kopeć, P.; Dziurka, M.; Kalandyk, A.; Pastuszak, J.; Wolko, B. Seed hydropriming and smoke water significantly improve low-temperature germination of Lupinus angustifolius L. Int. J. Mol. Sci. 2018, 19, 992. [Google Scholar] [CrossRef]

| In Vitro | |||||
|---|---|---|---|---|---|
| Medium | Content of Compounds (%) | Frequency (%) of Degenerating Embryo Sacs and Ovules | |||
| ‘Panda’ cv. | ‘Korona’ cv. | ||||
| 1 | 100 | 0 | 38 | ||
| 50 | 0 | 42 * | |||
| 30 | 68 * | 50 * | |||
| 2 | 100 | 15 | 36 | ||
| 50 | 14 | 50 * | |||
| 3 | 100 | 29 | 13 | ||
| 50 | 0 * | 38 * | |||
| In Planta | |||||
| Treatment | ‘Panda’ cv. | ‘Korona’ cv. | |||
| Frequency (%) of Degenerated | |||||
| Pollen Grains | Embryo Sacs and Ovules | Pollen Grains | Embryo Sacs and Ovules | ||
| Control | 3.2 | 10 | 1.3 | 23 | |
| 1S | 3.1 | 50 * | 1.6 | 40 * | |
| 50% | 2.2 * | 15 | 0.9 | 3 * | |
| 75% | 1.1* | 66 * | 1.2 | 85 * | |
| Hormones | Buds | Open Flowers | Wilted Flowers | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | 1S | 50% | 75% | Control | 1S | 50% | 75% | Control | 1S | 50% | 75% | |
| IAA | 73.5 ± 7.1 d | 124 ± 11 b | 102 ± 10 c | 149 ± 13 a | 67.9 ± 6.2 b | 81.7 ± 8.0 a | 64.3 ± 6.1 b | 47.2 ± 4.3 c | 157 ± 15 c | 189 ± 17 b | 253 ± 21 a | 66.1 ± 6.1 d |
| GAs | 8.29 ± 0.79 a | 8.14 ± 0.72 a | 3.51 ± 0.30 b | 8.50 ± 0.81 a | 7.40 ± 0.72 a,b | 6.45 ± 0.61 b | 4.59 ± 0.47 c | 8.67 ± 0.81 a | 7.40 ± 0.73 c | 11.1 ± 0.11 b | 6.87 ± 0.65 c | 15.1 ± 0.12 a |
| CYT | 2.88 ± 0.25 c | 8.93 ± 0.83 a | 2.55 ± 0.25 c | 3.91 ± 0.36 b | 3.78 ± 0.35 a | 3.92 ± 0.32 a | 2.10 ± 0.19 b | 3.36 ± 0.32 a | 4.09 ± 0.37 a | 4.43 ± 0.42 a | 4.49 ± 0.41 a | 2.40 ± 0.21 b |
| ABA | 2.16 ± 0.19 c | 1.55 ± 0.12 d | 2.92 ± 0.25 b | 3.50 ± 0.31 a | 4.04 ± 0.36 b | 2.69 ± 0.25 d | 5.63 ± 0.52 a | 3.53 ± 0.29 c | 3.45 ± 0.33 b | 1.80 ± 0.17 d | 4.09 ± 0.39 a | 2.52 ± 0.26 c |
| JA | 51.42 ± 5.07 c | 94.61 ± 9.11 b | 151 ± 14 a | 30.82 ± 3.98 d | 24.12 ± 2.95 c | 54.14 ± 5.12 a | 30.71 ± 2.76 b | 6.46 ± 0.68 d | 34.94 ± 3.32 b | 56.22 ± 5.21 a | 58.82 ± 5.76 a | 59.15 ± 5.78 a |
| SA | 22.6 ± 2.1 c | 77.8 ± 7.2 a | 86.8 ± 8.3 a | 41.9 ± 3.9 c | 18.8 ± 1.8 c | 62.4 ± 6.1 b | 105 ± 9.5 a | 65.1 ± 5.8 b | 33.2 ± 3.2 c | 96.7 ± 9.3 b | 124 ± 10.2 a | 31.7 ± 2.8 c |
| BA * | 31.3 ± 2.8 b | 68.4 ± 5.5 a | 62.7 ± 5.2 a | 28.6 ± 1.9 b | 21.2 ± 1.6 c | 33.2 ± 2.8 b | 41.8 ± 3.8 a | 27.6 ± 2.4 c | 16.2 ± 1.1 c | 25.9 ± 2.3 b | 38.4 ± 2.9 a | 36.4 ± 1.8 a |
| Hormones | Buds | Open Flowers | Wilted Flowers | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | 1S | 50% | 75% | Control | 1S | 50% | 75% | Control | 1S | 50% | 75% | |
| IAA | 173 ± 18 a | 77 ± 6 c | 110 ± 10 b | 110 ± 9 b | 91 ± 8 a | 73 ± 7 b | 69 ± 6 b | 71 ± 6 b | 287 ± 26 a | 267 ± 24 a | 133 ± 12 b | 134 ± 12 b |
| GAs | 5.84 ± 0.42 a | 4.47 ± 0.40 b | 6.22 ± 0.52 a | 4.63 ± 0.39 b | 6.22 ± 0.59 a | 6.87 ± 0.58 a | 5.65 ± 0.57 a | 5.60 ± 0.48 a | 6.15 ± 0.57 c | 10.85 ± 0.99 a | 8.67 ± 0.82 b | 10.26 ± 0.11 a |
| CYT | 3.58 ± 0.31 b | 2.53 ± 0.21 c | 5.09 ± 0.45 a | 3.29 ± 0.31 b | 3.72 ± 0.34 a,b | 3.46 ± 0.32 b | 4.16 ± 0.41 a | 3.83 ± 0.32 a | 3.22 ± 0.35 b | 4.39 ± 0.38 a | 5.01 ± 0.46 a | 3.27 ± 0.32 b |
| ABA | 2.74 ± 0.21 a | 1.88 ± 0.19 b | 1.51 ± 0.14 b | 1.30 ± 0.14 c | 5.36 ± 0.49 a | 3.99 ± 0.32 b | 3.75 ± 0.33 b | 2.78 ± 0.27 c | 3.31 ± 0.29 a | 2.54 ± 0.26 b | 1.92 ± 0.17 c | 3.43 ± 0.34 a |
| JA | 80.42 ± 7.01 a | 80.40 ± 0.76 a | 34.61 ± 3.20 c | 53.36 ± 5.01 b | 14.43 ± 1.42 b | 20.37 ± 2.04 a | 9.67 ± 0.91 c | 9.04 ± 0.90 c | 46.91 ± 4.05 a | 48.99 ± 4.12 a | 15.50 ± 1.40 c | 25.53 ± 2.61 b |
| SA | 56.93 ± 6.02 a | 25.62 ± 2.56 c | 33.07 ± 3.01 b | 59.37 ± 6.02 a | 44.59 ± 4.8 b | 40.49 ± 4.06 b | 40.08 ± 3.89 b | 71.24 ± 7.05 a | 65.29 ± 6.21 b | 48.02 ± 4.78 c | 45.0 ± 4.31 c | 139 ± 12 a |
| BA * | 29.80 ± 2.01 c | 66.00 ± 6.26 a | 43.10 ± 4.11 b | 45.31 ± 4.23 b | 21.55 ± 2.01 b | 27.43 ± 2.36 a | 30.05 ± 3.01 a | 26.90 ± 2.48 a | 24.68 ± 2.48 b | 36.72 ± 3.76 a | 34.91 ± 3.02 a | 22.43 ± 2.01 b |
| Effects | No. of Flowers | No. of Mature Seeds | Empty Seeds (%) | Abortion of Flowers and Fruits (%) | Efficiency of Seed Setting | Mature Seed Mass | MTS |
|---|---|---|---|---|---|---|---|
| Cultivar | ns | *** | * | *** | ns | *** | *** |
| Treatment | *** | ** | *** | ns | ns | *** | *** |
| Cultivar x Treatment | ns | *** | ns | ns | * | *** | *** |
| Cultivar | Treatment | No. of Flowers per Plant | No. of Mature Seeds per Plant | No. of Empty Seeds per Plant | Abortion of Flowers and Seeds (%) | Efficiency of Fertilization (%) | Mass of One Seed | Seed Mass per Plant (g) | MTS (g) |
|---|---|---|---|---|---|---|---|---|---|
| ‘Panda’ | Control | 669 ± 55 b | 128 ± 9 a,b | 29 ± 8 a | 81 | 19 | 0.0267 ± 0.003 e | 3.42 ± 0.07 c | 26.72 ± 1.41 b |
| 50% | 823 ± 74 a | 136 ± 11 a | 25 ± 5 a | 84 | 17 | 0.0292 ± 0.003 d | 3.97 ± 0.09 a | 29.19 ± 1.55 a | |
| 75% | 321 ± 29 e | 130 ± 12 a | 26 ± 4 a | 59 | 40 | 0.0252 ± 0.002 f | 3.27 ± 0.08 d | 25.15 ± 1.89 b | |
| 1 S | 357 ± 32 ed | 107 ± 9 b | 22 ± 5 a | 70 | 30 | 0.0266 ± 0.003 e | 2.85 ± 0.05 f | 26.63 ± 1.99 b | |
| ‘Korona’ | Control | 778 ± 69 a | 105 ± 10 b,c | 14 ± 2 b | 87 | 13 | 0.0311 ± 0.003 b | 3.27 ± 0.08 d | 31.14 ± 2.05 a |
| 50% | 558 ± 52 c | 125 ± 11 b | 16 ± 4 b | 78 | 22 | 0.0303 ± 0.002 c | 3.79 ± 0.07 b | 30.32 ± 2.03 a | |
| 75% | 442 ± 45 d | 100 ± 8 b,c | 22 ± 4 a | 77 | 23 | 0.0297 ± 0.003 c,d | 2.97 ± 0.06 e | 29.70 ± 2.07 a,b | |
| 1 S | 427 ± 38 d | 91 ± 7 c | 6 ± 2 c | 79 | 21 | 0.0334±0.003 a | 3.04 ± 0.08 e | 33.40 ± 2.27 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hornyák, M.; Słomka, A.; Sychta, K.; Dziurka, M.; Kopeć, P.; Pastuszak, J.; Szczerba, A.; Płażek, A. Reducing Flower Competition for Assimilates by Half Results in Higher Yield of Fagopyrum esculentum. Int. J. Mol. Sci. 2020, 21, 8953. https://doi.org/10.3390/ijms21238953
Hornyák M, Słomka A, Sychta K, Dziurka M, Kopeć P, Pastuszak J, Szczerba A, Płażek A. Reducing Flower Competition for Assimilates by Half Results in Higher Yield of Fagopyrum esculentum. International Journal of Molecular Sciences. 2020; 21(23):8953. https://doi.org/10.3390/ijms21238953
Chicago/Turabian StyleHornyák, Marta, Aneta Słomka, Klaudia Sychta, Michał Dziurka, Przemysław Kopeć, Jakub Pastuszak, Anna Szczerba, and Agnieszka Płażek. 2020. "Reducing Flower Competition for Assimilates by Half Results in Higher Yield of Fagopyrum esculentum" International Journal of Molecular Sciences 21, no. 23: 8953. https://doi.org/10.3390/ijms21238953
APA StyleHornyák, M., Słomka, A., Sychta, K., Dziurka, M., Kopeć, P., Pastuszak, J., Szczerba, A., & Płażek, A. (2020). Reducing Flower Competition for Assimilates by Half Results in Higher Yield of Fagopyrum esculentum. International Journal of Molecular Sciences, 21(23), 8953. https://doi.org/10.3390/ijms21238953








