Wild Soybean Oxalyl-CoA Synthetase Degrades Oxalate and Affects the Tolerance to Cadmium and Aluminum Stresses
Abstract
1. Introduction
2. Results
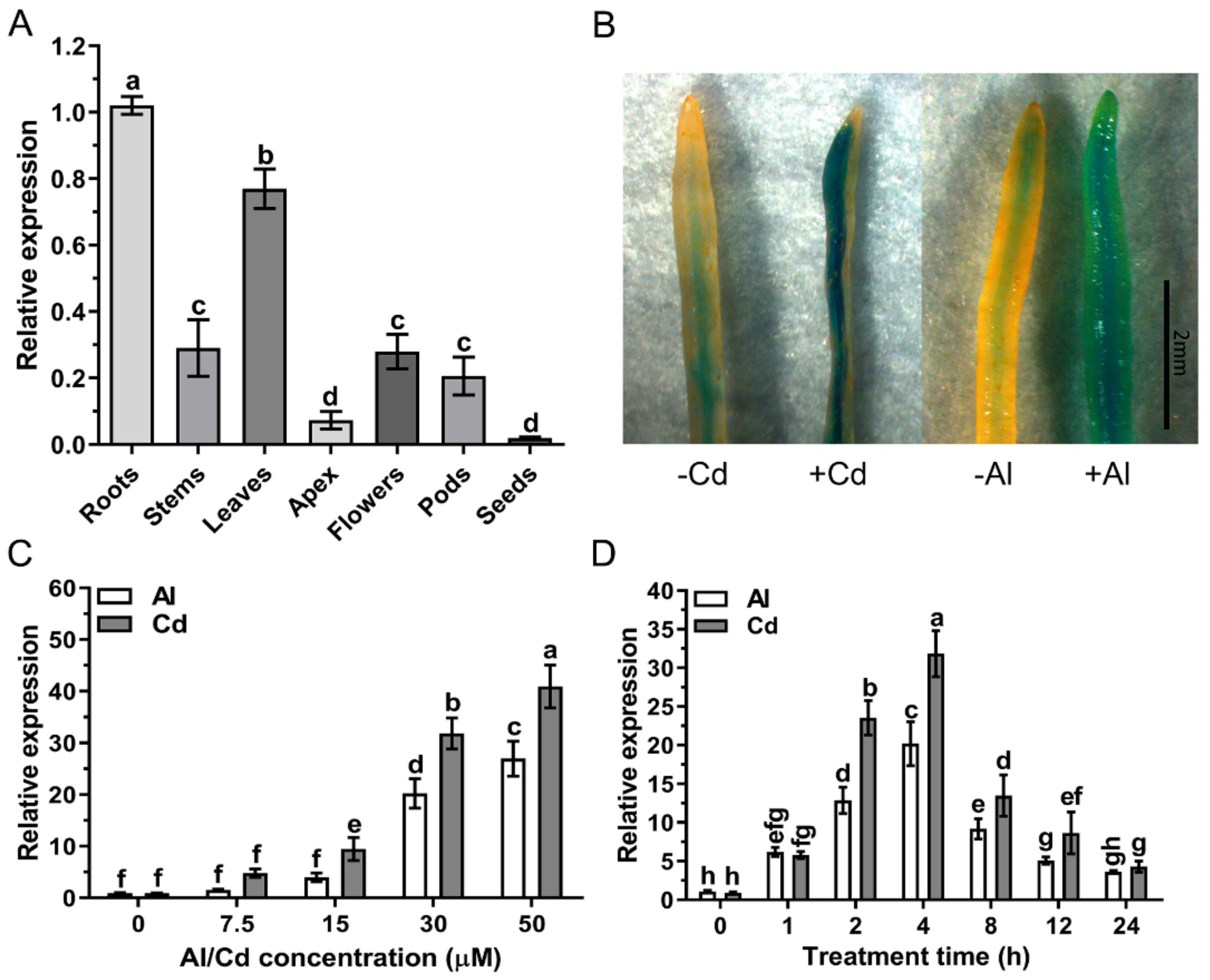
2.1. Oxalate Accumulation Induced by Cd and Al Contributes to Inhibit Root Growth in Wild Soybean
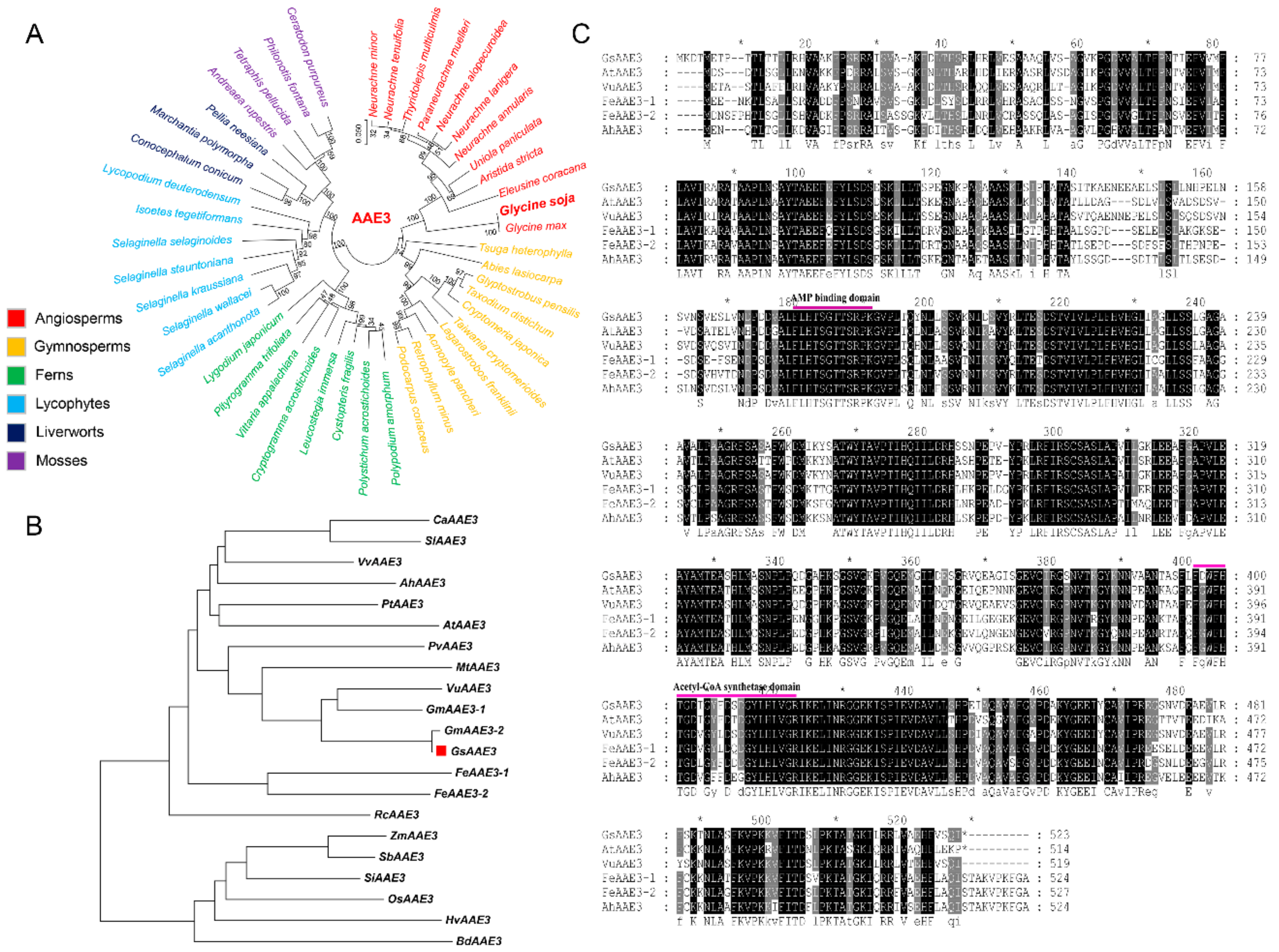
2.2. Isolation and Bioinformatics Analysis of the GsAAE3 Gene
2.3. Analysis of the GsAAE3 Expression Pattern
2.4. GsAAE3 Encodes an Oxalyl-CoA Synthetase
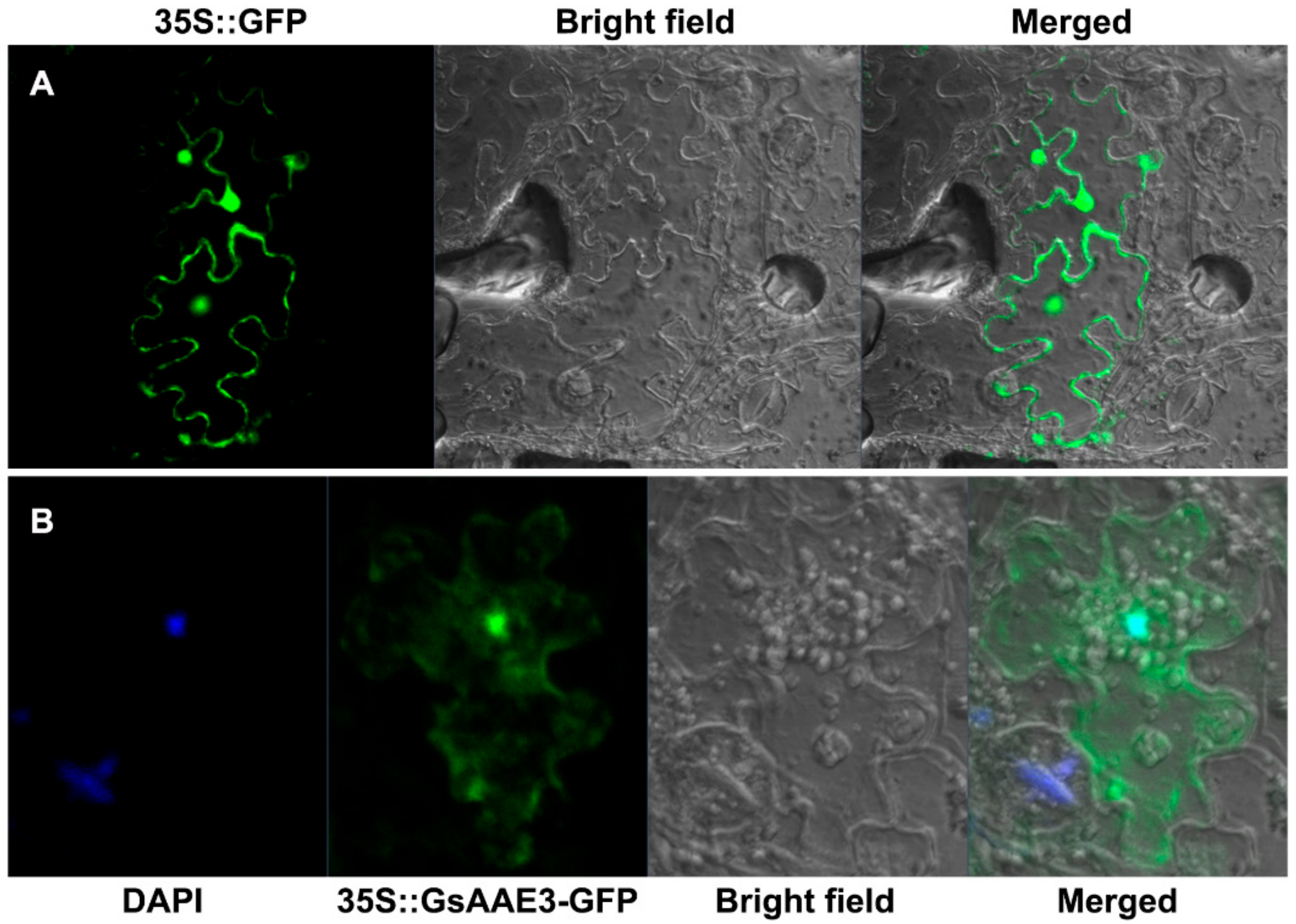
2.5. Subcellular Localization of GsAAE3
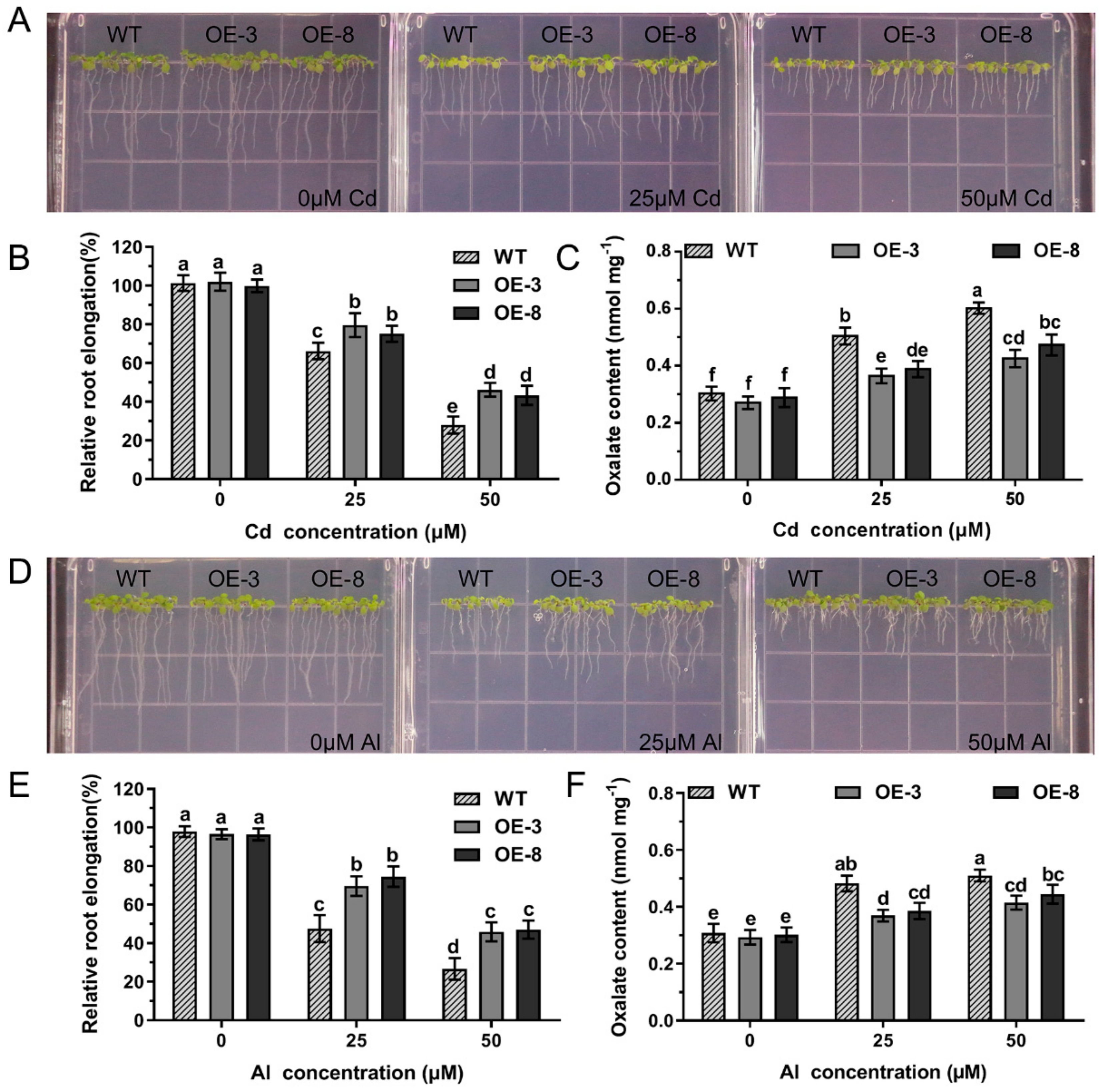
2.6. Effects of GsAAE3 Overexpression in A. thaliana on the Tolerances to Cd and Al
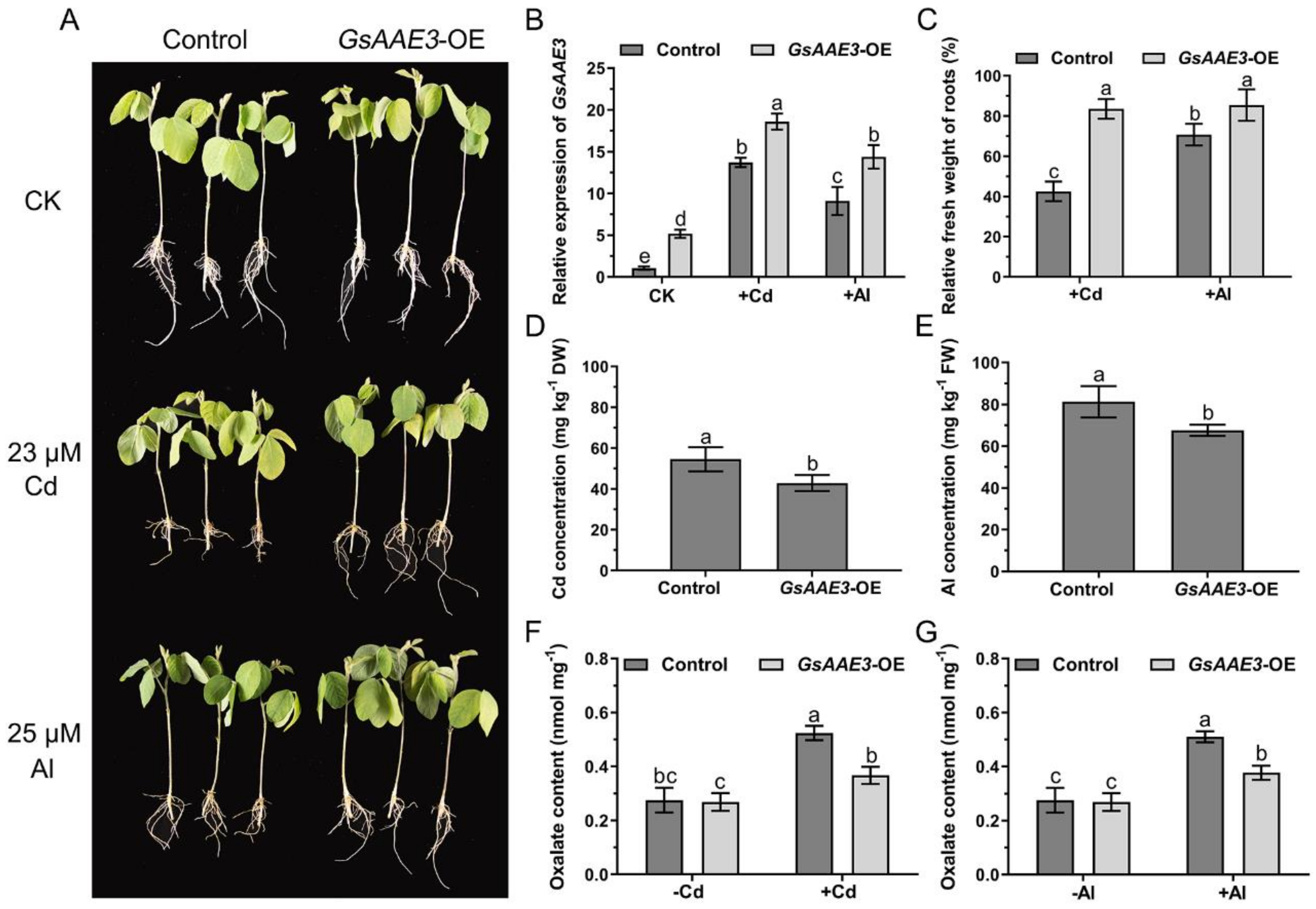
2.7. Effects of GsAAE3 Overexpression in Soybean Hairy Roots on the Tolerances to Cd and Al
3. Discussion
4. Materials and Methods
4.1. Plant Material and Treatments
4.2. Quantitative Real-Time PCR (qRT-PCR) and Gene Expression Assay
4.3. Bioinformatics Analysis and Statistical Analysis
4.4. Subcellular Localization
4.5. A. thaliana Transformation and Phenotypic Analysis
4.6. Soybean Hairy Root Transformation and Phenotypic Analysis
4.7. GUS Staining
4.8. Root Growth Analysis
4.9. Purification of the Recombinant Protein and Kinetic Analysis
4.10. Oxalate Measurement
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Cd | Cadmium |
| Al | Aluminum |
| AAE3 | ACYL-ACTIVATING ENZYME 3 |
| GFP | Green fluorescence protein |
| qRT-PCR | Real-time quantitative polymerase chain reaction |
| CDS | Coding sequence |
| WT | Wild-type |
| DTT | DL-Dithiothreitol |
| ATP | Adenosine triphosphate |
References
- Dutton, M.V.; Evans, C.S. Oxalate production by fungi: Its role in pathogenicity and ecology in the soil environment. Can. J. Microbiol. 1996, 42, 881–895. [Google Scholar] [CrossRef]
- Libert, B.; Franceschi, V.R. Oxalate in crop plants. J. Agric. Food Chem. 1987, 35, 926–938. [Google Scholar] [CrossRef]
- Franceschi, V.R.; Nakata, P.A. Calcium oxalate in plants: Formation and function. Annu. Rev. Plant Biol. 2005, 56, 41–71. [Google Scholar] [CrossRef] [PubMed]
- Nakata, P.A.; He, C.X. Oxalic acid biosynthesis is encoded by an operon in Burkholderia glumae. FEMS Microbiol. Lett. 2010, 304, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.Y.; Choi, Y.H.; Shin, T.S.; Kim, T.H.; Shin, K.S.; Park, H.W.; Kim, Y.H.; Kim, H.; Choi, G.J.; Jang, K.S.; et al. Biological Control of Meloidogyne incognita by Aspergillus niger F22 Producing Oxalic Acid. PLoS ONE 2016, 11, e0156230. [Google Scholar] [CrossRef] [PubMed]
- Molano-Flores, B. Herbivory and Calcium Concentrations Affect Calcium Oxalate Crystal Formation in Leaves of Sida (Malvaceae). Ann. Bot. 2001, 88, 387–391. [Google Scholar] [CrossRef]
- Palmieri, F.; Estoppey, A.; House, G.L.; Lohberger, A.; Bindschedler, S.; Chain, P.S.G.; Junier, P. Oxalic acid, a molecule at the crossroads of bacterial-fungal interactions. Adv. Appl. Microbiol. 2019, 106, 49–77. [Google Scholar] [PubMed]
- Zheng, S.J.; Ma, J.F.; Matsumoto, H. High aluminum resistance in buckwheat. I. Al-induced specific secretion of oxalic acid from root tips. Plant Physiol. 1998, 117, 745. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.F.; Zheng, C.; Hu, Y.T.; Jiang, T.; Liu, Y.; Dong, N.Y.; Yang, J.L.; Zheng, S.J. Cadmium-induced oxalate secretion from root apex is associated with cadmium exclusion and resistance in Lycopersicon esulentum. Plant Cell Environ. 2011, 34, 1055–1064. [Google Scholar] [CrossRef]
- Yang, Y.Y.; Jung, J.Y.; Song, W.Y.; Suh, H.S.; Lee, Y. Identification of Rice Varieties with High Tolerance or Sensitivity to Lead and Characterization of the Mechanism of Tolerance. Plant Physiol. 2000, 124, 1019–1026. [Google Scholar] [CrossRef]
- Ma, J.F.; Hiradate, S.; Matsumoto, H. High aluminium resistance in buckwheat: II. Oxalic acid detoxifies aluminium internally. Plant Physiol. (Rockville) 1998, 117, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yu, N.; Mu, G.; Shinwari, K.I.; Shen, Z.; Zheng, L. Screening for Cd-Safe Cultivars of Chinese Cabbage and a Preliminary Study on the Mechanisms of Cd Accumulation. Int. J. Environ. Res. Public Health 2017, 14, 395. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Zheng, S.J.; Matsumoto, H.; Hiradate, S. Detoxifying aluminum with buckwheat. Nature 1997, 390, 569–570. [Google Scholar] [CrossRef]
- Lou, H.Q.; Fan, W.; Xu, J.M.; Gong, Y.L.; Jin, J.F.; Chen, W.W.; Liu, L.Y.; Hai, M.R.; Yang, J.L.; Zheng, S.J. An Oxalyl-CoA Synthetase Is Involved in Oxalate Degradation and Aluminum Tolerance. Plant Physiol. 2016, 172, 1679–1690. [Google Scholar] [CrossRef]
- Wang, X.; Shi, Y.; Chen, X.; Huang, B. Screening of Cd-safe genotypes of Chinese cabbage in field condition and Cd accumulation in relation to organic acids in two typical genotypes under long-term Cd stress. Environ. Sci. Pollut. Res. 2015, 22, 16590–16599. [Google Scholar] [CrossRef]
- Lou, H.Q.; Gong, Y.L.; Fan, W.; Xu, J.M.; Liu, Y.; Cao, M.J.; Wang, M.-H.; Yang, J.L.; Zheng, S.J. A Formate Dehydrogenase Confers Tolerance to Aluminum and Low pH. Plant Physiol. 2016, 171, 294–305. [Google Scholar] [CrossRef]
- Nakata, P.A. An Assessment of Engineered Calcium Oxalate Crystal Formation on Plant Growth and Development as a Step toward Evaluating Its Use to Enhance Plant Defense. PLoS ONE 2015, 10, e0141982. [Google Scholar] [CrossRef]
- Svedruzic, D.; Jonsson, S.; Toyota, C.G.; Reinhardt, L.A.; Ricagno, S.; Linqvist, Y.; Richards, N.G.J. The enzymes of oxalate metabolism: Unexpected structures and mechanisms. Arch. Biochem. Biophys. 2005, 433, 176–192. [Google Scholar] [CrossRef]
- Yang, J.; Fu, M.; Ji, C.; Huang, Y.; Wu, Y. Maize Oxalyl-CoA Decarboxylasel Degrades Oxalate and Affects the Seed Metabolome and Nutritional Quality. Plant Cell 2018, 30, 2447–2462. [Google Scholar] [CrossRef]
- Foster, J.; Kim, H.U.; Nakata, P.A.; Browse, J. A Previously Unknown Oxalyl-CoA Synthetase Is Important for Oxalate Catabolism in Arabidopsis. Plant Cell 2012, 24, 1217–1229. [Google Scholar] [CrossRef]
- Alekseeva, A.A.; Savin, S.S.; Tishkov, V.I. NAD+-dependent formate dehydrogenase from plants. Acta Nat. 2011, 3, 38–54. [Google Scholar] [CrossRef]
- Foster, J.; Luo, B.; Nakata, P.A. An Oxalyl-CoA Dependent Pathway of Oxalate Catabolism Plays a Role in Regulating Calcium Oxalate Crystal Accumulation and Defending against Oxalate-Secreting Phytopathogens in Medicago truncatula. PLoS ONE 2016, 11, e0149850. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Suh, M.-C.; Kim, S.; Kwon, J.-K.; Kim, M.; Paek, K.-H.; Choi, D.; Kim, B.-D. Molecular cloning of a novel pathogen-inducible cDNA encoding a putative acyl-CoA synthetase from Capsicum annuum L. Plant Mol. Biol. 2001, 46, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Guo, Z.; Gu, F.; Ke, S.; Sun, D.; Dong, S.; Liu, W.; Huang, M.; Xiao, W.; Yang, G.; et al. 4-Coumarate-CoA Ligase-Like Gene OsAAE3 Negatively Mediates the Rice Blast Resistance, Floret Development and Lignin Biosynthesis. Front. Plant Sci. 2017, 7, 2041. [Google Scholar] [CrossRef]
- Carpenter, E.J.; Matasci, N.; Ayyampalayam, S.; Wu, S.; Wong, K.S. Access to RNA-sequencing data from 1173 plant species: The 1000 Plant transcriptomes initiative (1KP). Gigaence 2019, 8, giz126. [Google Scholar] [CrossRef]
- Leebens-Mack, J.H.; Barker, M.S.; Carpenter, E.J.; Deyholos, M.K.; Gitzendanner, M.A.; Graham, S.W.; Grosse, I.; Li, Z.; Melkonian, M.; Mirarab, S.; et al. One thousand plant transcriptomes and the phylogenomics of green plants. Nature 2019, 574, 679–685. [Google Scholar]
- Horton, P.; Park, K.J.; Obayashi, T.; Fujita, N.; Harada, H.; Adams-Collier, C.J.; Nakai, K. WoLF PSORT: Protein localization predictor. Nucleic Acids Res. 2007, 35, W585–W587. [Google Scholar] [CrossRef]
- Shockey, J.M.; Fulda, M.S.; Browse, J. Arabidopsis contains a large superfamily of acyl-activating enzymes. Phylogenetic and biochemical analysis reveals a new class of acyl-coenzyme A synthetases. Plant Physiol. 2003, 132, 1065–1076. [Google Scholar] [CrossRef]
- Peng, C.; Liang, X.; Liu, E.E.; Zhang, J.J.; Peng, X.X. The oxalyl-CoA synthetase-regulated oxalate and its distinct effects on resistance to bacterial blight and aluminium toxicity in rice. Plant Biol. 2017, 19, 345–353. [Google Scholar] [CrossRef]
- Dias, M.C.; Monteiro, C.; Moutinho-Pereira, J.; Correia, C.; Gonçalves, B.; Santos, C. Cadmium toxicity affects photosynthesis and plant growth at different levels. Acta Physiol. Plant. 2013, 35, 1281–1289. [Google Scholar] [CrossRef]
- Foy, C.D. Plant adaptation to acid, aluminum-toxic soils. Commun. Soil Sci. Plant Anal. 1988, 19, 959–987. [Google Scholar] [CrossRef]
- Ilarslan, H.; Palmer, R.G.; Horner, H.T. Calcium oxalate crystals in developing seeds of soybean. Ann. Bot. (Lond.) 2001, 88, 243–257. [Google Scholar] [CrossRef]
- Klug, B.; Horst, W.J. Oxalate exudation into the root-tip water free space confers protection from aluminum toxicity and allows aluminum accumulation in the symplast in buckwheat (Fagopyrum esculentum). New Phytol. 2010, 187, 380–391. [Google Scholar] [CrossRef] [PubMed]
- Korth, K.L.; Doege, S.J.; Park, S.H.; Goggin, F.L.; Wang, Q.; Gomez, S.K.; Liu, G.; Jia, L.; Nakata, P.A. Medicago truncatula mutants demonstrate the role of plant calcium oxalate crystals as an effective defense against chewing insects. Plant Physiol. 2006, 141, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Nakata, P.A. Advances in our understanding of calcium oxalate crystal formation and function in plants. Plant Sci. 2003, 164, 901–909. [Google Scholar] [CrossRef]
- Smith, L.H. The many roles of oxalate in nature. Trans. Am. Clin. Climatol. Assoc. 2002, 113, 1–20. [Google Scholar] [PubMed]
- Zhang, B.; Oakes, A.D.; Newhouse, A.E.; Baier, K.M.; Maynard, C.A.; Powell, W.A. A threshold level of oxalate oxidase transgene expression reduces Cryphonectria parasitica-induced necrosis in a transgenic American chestnut (Castanea dentata) leaf bioassay. Trans. Res. 2013, 22, 973–982. [Google Scholar] [CrossRef]
- Delhaize, E.; Ryan, P.R.; Randall, P.J. Aluminum Tolerance in Wheat (Triticum aestivum L.) (II. Aluminum-Stimulated Excretion of Malic Acid from Root Apices). Plant Physiol. 1993, 103, 695–702. [Google Scholar] [CrossRef]
- Miyasaka, S.C.; Buta, J.G.; Howell, R.K.; Foy, C.D. Mechanism of Aluminum Tolerance in Snapbeans 1: Root Exudation of Citric Acid. Plant Physiol. 1991, 96, 737–743. [Google Scholar] [CrossRef]
- Furukawa, J.; Yamaji, N.; Hua, W.; Mitani, N.; Murata, Y.; Sato, K.; Katsuhara, M.; Takeda, K.; Ma, J.F. An Aluminum-Activated Citrate Transporter in Barley. Plant Cell Physiol. 2007, 1081–1091. [Google Scholar] [CrossRef]
- Ligaba, A.; Katsuhara, M.; Ryan, P.R.; Shibasaka, M.; Matsumoto, H. The BnALMT1 and BnALMT2 genes from rape encode aluminum-activated malate transporters that enhance the aluminum resistance of plant cells. Plant Physiol. 2006, 142, 1294–1303. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, J.V.; Liu, J.; Guimaraes, C.T.; Lana, U.G.P.; Alves, V.M.C.; Wang, Y.-H.; Schaffert, R.E.; Hoekenga, O.A.; Pineros, M.A.; Shaff, J.E.; et al. A gene in the multidrug and toxic compound extrusion (MATE) family confers aluminum tolerance in sorghum. Nat. Genet. 2007, 39, 1156–1161. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, T.; Yamamoto, Y.; Ezaki, B.; Katsuhara, M.; Ahn, S.J.; Ryan, P.R.; Delhaize, E.; Matsumoto, H. A wheat gene encoding an aluminum-activated malate transporter. Plant J. 2004, 37, 645–653. [Google Scholar] [CrossRef] [PubMed]
- Yokosho, K.; Yamaji, N.; Ma, J.F. Isolation and characterisation of two MATE genes in rye. Funct. Plant Biol. 2010, 37, 296–303. [Google Scholar] [CrossRef]
- Tang, Y.; Sorrells, M.E.; Kochian, L.V.; Garvin, D.F. Identification of RFLP Markers Linked to the Barley Aluminum Tolerance Gene Alp. Crop. Sci. 2000, 40, 778–782. [Google Scholar] [CrossRef]
- Liang, C.; Pineros, M.A.; Tian, J.; Yao, Z.; Sun, L.; Liu, J.; Shaff, J.; Coluccio, A.; Kochian, L.V.; Liao, H. Low pH, aluminum, and phosphorus coordinately regulate malate exudation through GmALMT1 to improve soybean adaptation to acid soils. Plant Physiol. 2013, 161, 1347–1361. [Google Scholar] [CrossRef]
- Shen, H.; He, L.F.; Sasaki, T.; Yamamoto, Y.; Zheng, S.J.; Ligaba, A.; Yan, X.L.; Ahn, S.J.; Yamaguchi, M.; Sasakawa, H.; et al. Citrate secretion coupled with the modulation of soybean root tip under aluminum stress. Up-regulation of transcription, translation, and threonine-oriented phosphorylation of plasma membrane H+-ATPase. Plant Physiol. 2005, 139, 557. [Google Scholar] [CrossRef]
- Yang, Z.M.; Nian, H.; Sivaguru, M.; Tanakamaru, S.; Matsumoto, H. Characterization of aluminium-induced citrate secretion in aluminium-tolerant soybean (Glycine max) plants. Physiol. Plant. 2001, 113, 64–71. [Google Scholar] [CrossRef]
- Fu, S.; Lu, Y.; Zhang, X.; Yang, G.; Xia, J. The ABC transporter OsABCG36 is required for Cd tolerance in rice. J. Exp. Bot. 2019, 70, 5909–5918. [Google Scholar] [CrossRef]
- Nakanishi, H.; Ogawa, I.; Ishimaru, Y.; Mori, S.; Nishizawa, N.K. Iron deficiency enhances cadmium uptake and translocation mediated by the Fe2+ transporters OsIRT1 and OsIRT2 in rice. Soil Sci. Plant Nutr. 2006, 52, 464–469. [Google Scholar] [CrossRef]
- Sasaki, A.; Yamaji, N.; Ma, J.F. Overexpression of OsHMA3 enhances Cd tolerance and expression of Zn transporter genes in rice. J. Exp. Bot. 2014, 65, 6013–6021. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Beevers, H. Biogenesis of oxalate in plant tissues. Plant Physiol. 1968, 43, 1821–1828. [Google Scholar] [CrossRef] [PubMed]
- Fuji, N.; Watanabe, M.; Watanabe, Y.; Shimada, N. Relationship between oxalate synthesis and glycolate-cycle in spinach. J. Jpn. Soc. Hortic. Sci. 1994, 62, 789–794. [Google Scholar] [CrossRef]
- Loewus, F.A.; Loewus, M.W.; Seib, P.A. Biosynthesis and metabolism of ascorbic acid in plants. Crit. Rev. Plant Sci. 1987, 5, 101–119. [Google Scholar] [CrossRef]
- Yu, L.; Jiang, J.; Zhang, C.; Jiang, L.; Ye, N.; Lu, Y.; Yang, G.; Liu, E.; Peng, C.; He, Z.; et al. Glyoxylate rather than ascorbate is an efficient precursor for oxalate biosynthesis in rice. J. Exp. Bot. 2010, 61, 1625–1634. [Google Scholar] [CrossRef]
- Yang, X.Y.; Yang, J.L.; Zhou, Y.; Pineros, M.A.; Kochian, L.V.; Li, G.X.; Zheng, S.J. A de novo synthesis citrate transporter, Vigna umbellata multidrug and toxic compound extrusion, implicates in Al-activated citrate efflux in rice bean (Vigna umbellata) root apex. Plant Cell Environ. 2011, 34, 2138–2148. [Google Scholar] [CrossRef] [PubMed]
- Gallaher, R.N.; Perkins, H.F.; Jones, J.B., Jr. Calcium concentration and distribution in healthy and decline peach tree tissues [Physiological diseases, senescence]. Hortence 1975, 134–137. [Google Scholar]
- Kochian, L.V.; Hoekenga, O.A.; Pineros, M.A. How do crop plants tolerate acid soils?—Mechanisms of aluminum tolerance and phosphorous efficiency. Annu. Rev. Plant Biol. 2004, 55, 459–493. [Google Scholar] [CrossRef]
- Verbruggen, N.; Hermans, C.; Schat, H. Mechanisms to cope with arsenic or cadmium excess in plants. Curr. Opin. Plant Biol. 2009, 12, 364–372. [Google Scholar] [CrossRef]
- Nadal, M.; Schuhmacher, M.; Domingo, J.L. Metal pollution of soils and vegetation in an area with petrochemical industry. Sci. Total Environ. 2004, 321, 59–69. [Google Scholar] [CrossRef]
- Touceda-Gonzalez, M.; Brader, G.; Antonielli, L.; Ravindran, V.B.; Waldner, G.; Friesl-Hanl, W.; Corretto, E.; Campisano, A.; Pancher, M.; Sessitsch, A. Combined amendment of immobilizers and the plant growth-promoting strain Burkholderia phytofirmans PsJN favours plant growth and reduces heavy metal uptake. Soil Biol. Biochem. 2015, 91, 140–150. [Google Scholar] [CrossRef]
- Jiang, L.; Wang, W.; Chen, Z.; Gao, Q.; Xu, Q.; Cao, H. A role for APX1 gene in lead tolerance in Arabidopsis thaliana. Plant Sci. 2017, 256, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhao, H.; Wu, L.; Liu, A.; Zhao, F.-J.; Xu, W. Heavy metal ATPase 3 (HMA3) confers cadmium hypertolerance on the cadmium/zinc hyperaccumulator Sedum plumbizincicola. New Phytol. 2017, 215, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Kochian, L.V.; Piñeros, M.A.; Liu, J.; Magalhaes, J.V. Plant Adaptation to Acid Soils: The Molecular Basis for Crop Aluminum Resistance. Annu. Rev. Plant Biol. 2015, 66, 571. [Google Scholar] [CrossRef]
- Rao, Y.; Li, Y.; Qian, Q. Recent progress on molecular breeding of rice in China. Plant Cell Rep. 2014, 33, 551–564. [Google Scholar] [CrossRef]
- Cai, Z.D.; Cheng, Y.B.; Xian, P.Q.; Lin, R.B.; Xia, Q.J.; He, X.K.; Liang, Q.W.; Lian, T.X.; Ma, Q.B.; Nian, H. Fine-mapping QTLs and the validation of candidate genes for Aluminum tolerance using a high-density genetic map. Plant Soil 2019, 444, 119–137. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Yan, J.; Tong, T.; Li, X.; Chen, Q.; Dai, M.; Niu, F.; Yang, M.; Deyholos, M.K.; Yang, B.; Jiang, Y.-Q. A Novel NAC-Type Transcription Factor, NAC87, from Oilseed Rape Modulates Reactive Oxygen Species Accumulation and Cell Death. Plant Cell Physiol. 2018, 59, 290–303. [Google Scholar] [CrossRef]
- Zhang, X.; Henriques, R.; Lin, S.S.; Niu, Q.W.; Chua, N.H. Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nat. Protocol. 2006, 2, 1–6. [Google Scholar] [CrossRef]
- Chen, J.; Yang, L.; Gu, J.; Bai, X.; Ren, Y.; Fan, T.; Han, Y.; Jiang, L.; Xiao, F.; Liu, Y.; et al. MAN3 gene regulates cadmium tolerance through the glutathione-dependent pathway in Arabidopsis thaliana. New Phytol. 2015, 205, 570–582. [Google Scholar] [CrossRef]
- Zhu, X.F.; Sun, Y.; Zhang, B.C.; Mansoori, N.; Wan, J.X.; Liu, Y.; Wang, Z.W.; Shi, Y.Z.; Zhou, Y.H.; Zheng, S.J. TRICHOME BIREFRINGENCE-LIKE27 Affects Aluminum Sensitivity by Modulating the O-Acetylation of Xyloglucan and Aluminum-Binding Capacity in Arabidopsis. Plant Physiol. 2014, 166, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Kereszt, A.; Li, D.; Indrasumunar, A.; Nguyen, C.D.; Nontachaiyapoom, S.; Kinkema, M.; Gresshoff, P.M. Agrobacterium rhizogenes-mediated transformation of soybean to study root biology. Nat. Protoc. 2007, 2, 948–952. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Yang, B.; Wan, H.; Fang, X.; Yang, C. The differences of cell wall in roots between two contrasting soybean cultivars exposed to cadmium at young seedlings. Environ. Sci. Pollut. Res. 2018, 25, 29705–29714. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, K.; Braun, K.; Böckler, A.; Fuchs, G. Studies on the anaerobic degradation of benzoic acid and 2-aminobenzoic acid by a denitrifying Pseudomonas strain. Arch. Microbiol. 1987, 149, 62–69. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xian, P.; Cai, Z.; Cheng, Y.; Lin, R.; Lian, T.; Ma, Q.; Nian, H. Wild Soybean Oxalyl-CoA Synthetase Degrades Oxalate and Affects the Tolerance to Cadmium and Aluminum Stresses. Int. J. Mol. Sci. 2020, 21, 8869. https://doi.org/10.3390/ijms21228869
Xian P, Cai Z, Cheng Y, Lin R, Lian T, Ma Q, Nian H. Wild Soybean Oxalyl-CoA Synthetase Degrades Oxalate and Affects the Tolerance to Cadmium and Aluminum Stresses. International Journal of Molecular Sciences. 2020; 21(22):8869. https://doi.org/10.3390/ijms21228869
Chicago/Turabian StyleXian, Peiqi, Zhandong Cai, Yanbo Cheng, Rongbin Lin, Tengxiang Lian, Qibin Ma, and Hai Nian. 2020. "Wild Soybean Oxalyl-CoA Synthetase Degrades Oxalate and Affects the Tolerance to Cadmium and Aluminum Stresses" International Journal of Molecular Sciences 21, no. 22: 8869. https://doi.org/10.3390/ijms21228869
APA StyleXian, P., Cai, Z., Cheng, Y., Lin, R., Lian, T., Ma, Q., & Nian, H. (2020). Wild Soybean Oxalyl-CoA Synthetase Degrades Oxalate and Affects the Tolerance to Cadmium and Aluminum Stresses. International Journal of Molecular Sciences, 21(22), 8869. https://doi.org/10.3390/ijms21228869



