MicroRNAs from Extracellular Vesicles Secreted by Bovine Embryos as Early Biomarkers of Developmental Competence
Abstract
1. Introduction
2. Results
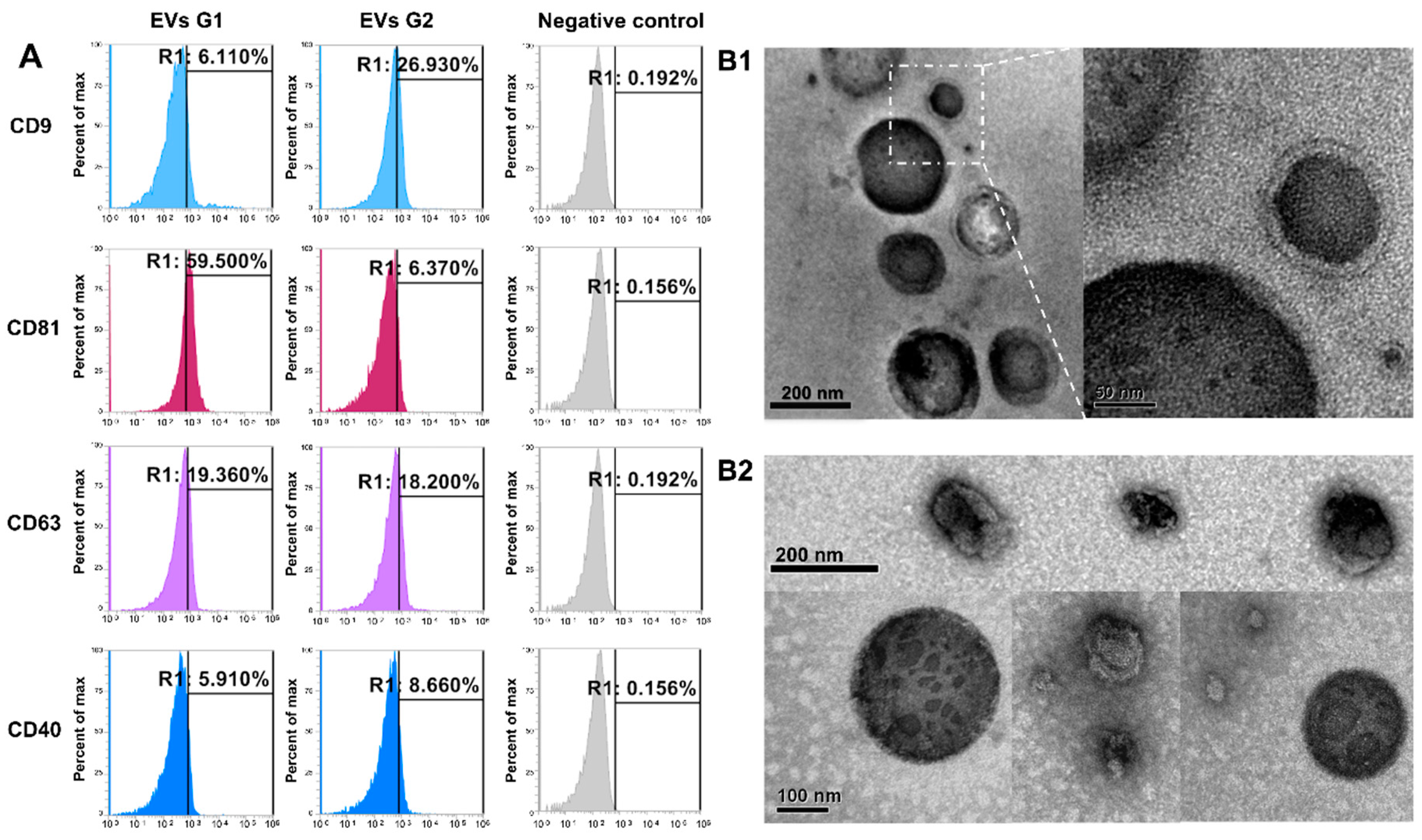
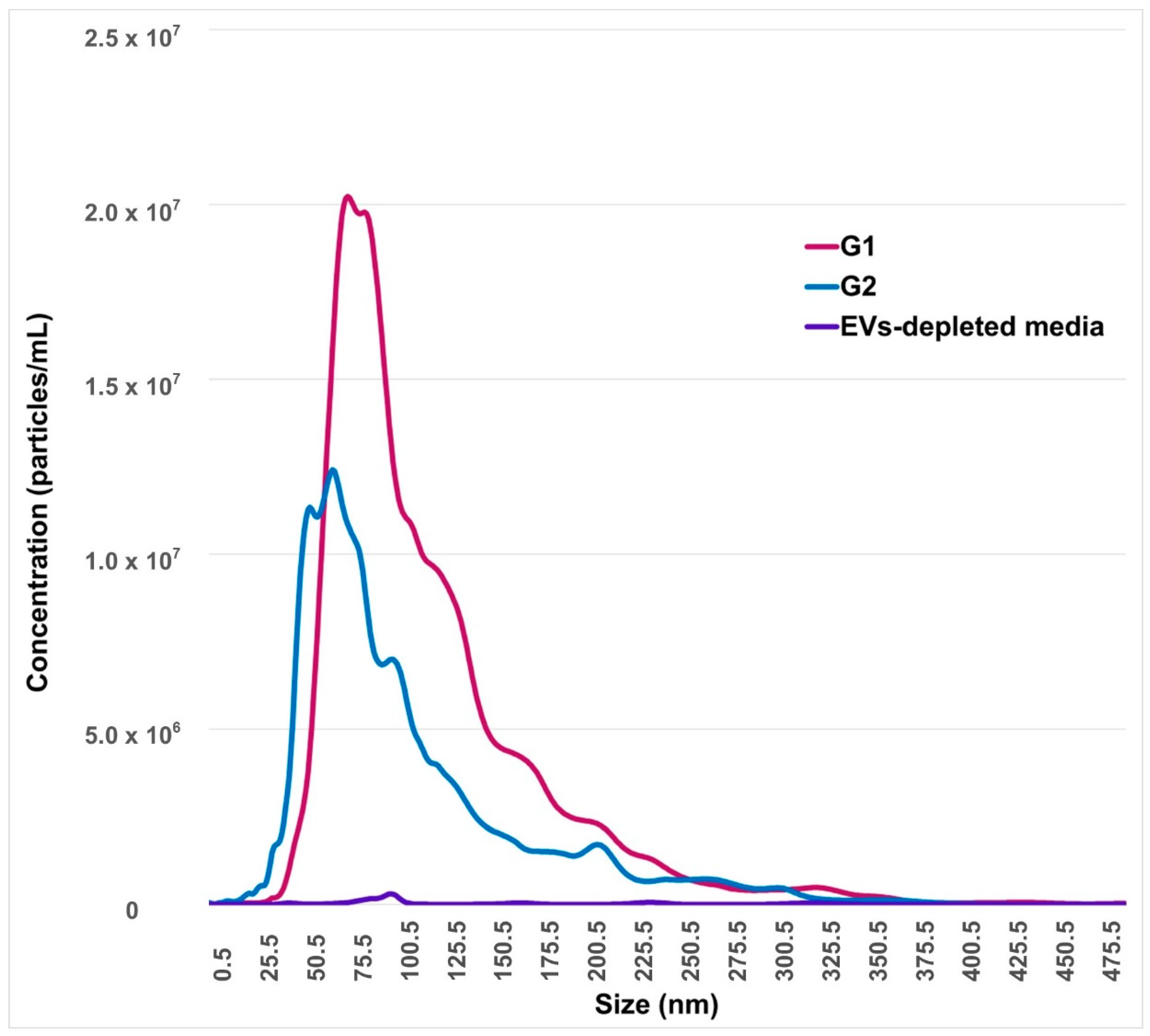
2.1. Identification and Characterization of EVs from Conditioned-Culture Media by Early Embryos
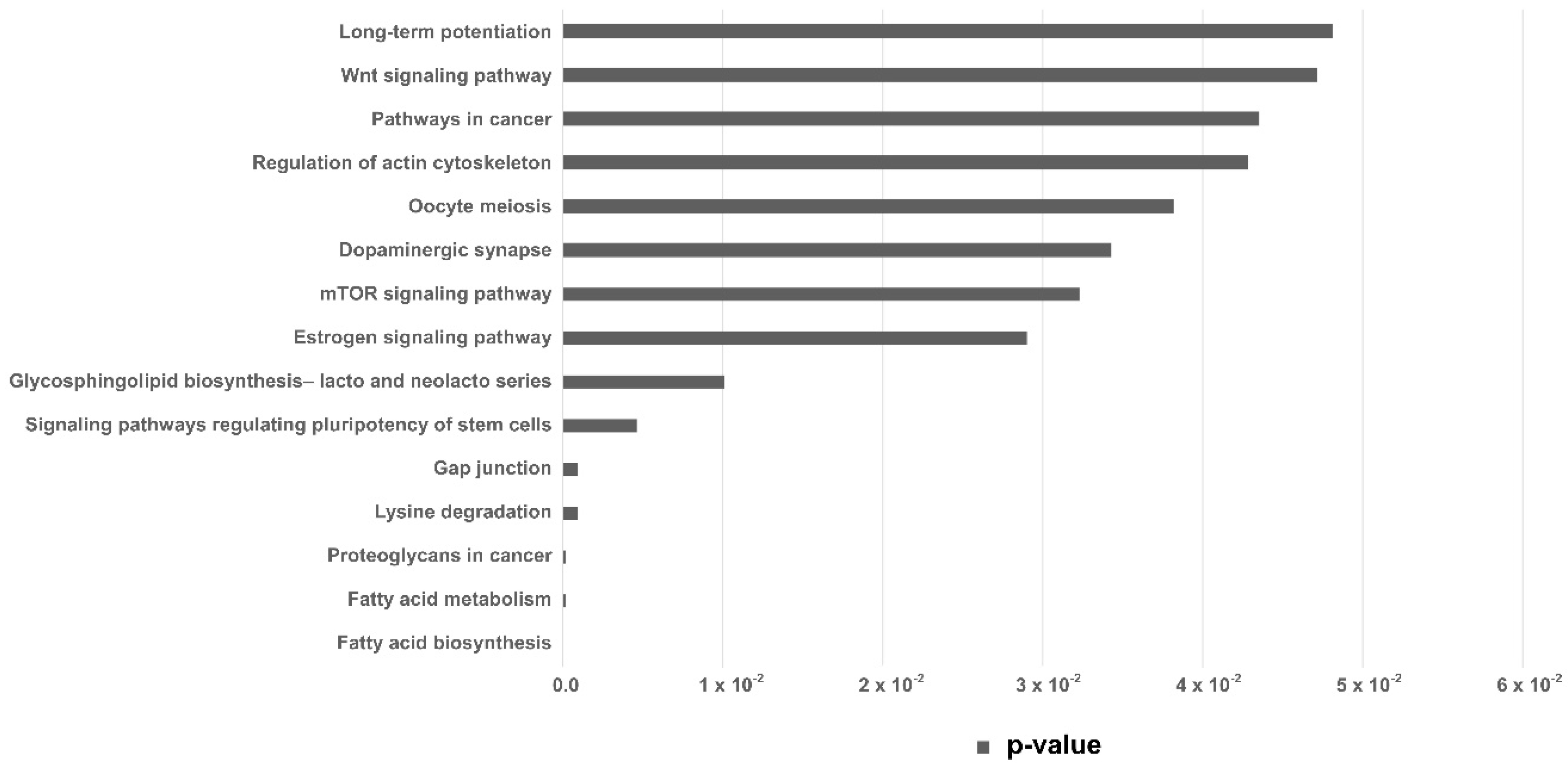
2.2. miRNAs Differential Expression Analysis
3. Discussion
4. Materials and Methods
4.1. General Design
4.2. In Vitro Embryo Production
4.3. Separation of EVs from Conditioned-Culture Media by Early Embryos
4.4. Morphological and Molecular Characterization of Nanoparticles from Conditioned-Culture Media by Early Embryos
4.4.1. Flow Cytometer for EVs Markers
4.4.2. Transmission Electron Microscopy (TEM) Analysis
4.4.3. Nanoparticles Tracking Analysis (NTA)
4.5. miRNAs Cargo Characterization of EVs from Conditioned-Culture Media by Early Embryos
4.5.1. Extracellular Vesicles RNA Isolation and Quantification
4.5.2. Small RNAs Sequencing Analysis
4.6. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| EVs | Extracellular vesicles |
| miR | microRNA |
| TEM | Transmission electron microscopy |
| NTA | Nanoparticles tracking analysis |
| NGS | Next-generation sequencing |
| G1 | Group 1: embryos arrested on 8–16-cells stage at day 3.5 of culture |
| G2 | Group 2: embryos that reached blastocyst stage at day 7 of culture |
| IVF | In vitro fertilization |
| EGA | Embryo genome activation |
| SOF | Synthetic oviduct fluid |
| SOFd | EVs-depleted SOF |
References
- Cocucci, E.; Racchetti, G.; Meldolesi, J. Shedding microvesicles: Artefacts no more. Trends Cell Biol. 2009, 19, 43–51. [Google Scholar] [CrossRef]
- György, B.; Szabó, T.G.; Pásztói, M.; Pál, Z.; Misják, P.; Aradi, B.; László, V.; Pállinger, É.; Pap, E.; Kittel, Á.; et al. Membrane vesicles, current state-of-the-art: Emerging role of extracellular vesicles. Cell. Mol. Life Sci. 2011, 68, 2667–2688. [Google Scholar] [CrossRef]
- Yáñez-Mó, M.; Siljander, P.; Andreu, Z.; Zavec, A.B.; Borràs, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological properties of extracellular vesicles and their physiological functions. J. Extracell. Vesicles 2015, 4, 27066. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.F.; Pegtel, D.M.; Lambertz, U.; Leonardi, T.; O’Driscoll, L.; Pluchino, S.; Ter-Ovanesyan, D.; Nolte-‘t Hoen, E.N. ISEV position paper: Extracellular vesicle RNA analysis and bioinformatics. J. Extracell. Vesicles 2013, 2, 22859. [Google Scholar] [CrossRef] [PubMed]
- Raposo, G.; Stoorvogel, W. Extracellular vesicles: Exosomes, microvesicles, and friends. J. Cell Biol. 2013, 200, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Machtinger, R.; Laurent, L.C.; Baccarelli, A.A. Extracellular vesicles: Roles in gamete maturation, fertilization and embryo implantation. Hum. Reprod. Update 2016, 22, 182–193. [Google Scholar] [CrossRef]
- Burns, G.; Brooks, K.; Wildung, M.; Navakanitworakul, R.; Christenson, L.K.; Spencer, T.E. Extracellular Vesicles in Luminal Fluid of the Ovine Uterus. PLoS ONE 2014, 9, e90913. [Google Scholar] [CrossRef]
- Saadeldin, I.M.; Kim, S.J.; Bin Choi, Y.; Lee, B.C. Improvement of Cloned Embryos Development by Co-Culturing with Parthenotes: A Possible Role of Exosomes/Microvesicles for Embryos Paracrine Communication. Cell. Reprogram. 2014, 16, 223–234. [Google Scholar] [CrossRef]
- Giacomini, E.; Vago, R.; Sanchez, A.M.; Podini, P.; Zarovni, N.; Murdica, V.; Rizzo, R.; Bortolotti, D.; Candiani, M.; Viganò, P. Secretome of in vitro cultured human embryos contains extracellular vesicles that are uptaken by the maternal side. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef]
- Pavani, K.C.; Hendrix, A.; Broeck, W.V.D.; Couck, L.; Szymanska, K.; Lin, X.; De Koster, J.; Van Soom, A.; Leemans, B. Isolation and Characterization of Functionally Active Extracellular Vesicles from Culture Medium Conditioned by Bovine Embryos In Vitro. Int. J. Mol. Sci. 2018, 20, 38. [Google Scholar] [CrossRef]
- Mellisho, E.; Velasquez, A.E.; Núñez, M.J.; Cabezas, J.G.; Cueto, J.A.; Fader, C.; Castro, F.O.; Rodriguez-Alvarez, L. Identification and characteristics of extracellular vesicles from bovine blastocysts produced in vitro. PLoS ONE 2017, 12, e0178306. [Google Scholar] [CrossRef] [PubMed]
- Mellisho, E.A.; Briones, M.A.; Velasquez, A.E.; Cabezas, J.; Castro, F.O.; Rodriguez-Alvarez, L. Extracellular vesicles secreted during blastulation show viability of bovine embryos. Reproduction 2019, 158, 477–492. [Google Scholar] [CrossRef] [PubMed]
- Dissanayake, K.; Nõmm, M.; Lättekivi, F.; Ressaissi, Y.; Godakumara, K.; Lavrits, A.; Midekessa, G.; Viil, J.; Bæk, R.; Jørgensen, M.M.; et al. Individually cultured bovine embryos produce extracellular vesicles that have the potential to be used as non-invasive embryo quality markers. Theriogenology 2020, 149, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.; Sun, X.; Dey, S.K. Mechanisms of implantation: Strategies for successful pregnancy. Nat. Med. 2012, 18, 1754–1767. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; An, X.; Zhang, L.; Fu, M.; Peng, J.; Han, P.; Hou, J.; Zhou, Z.; Cao, B. Identification and Profiling of microRNAs in Goat Endometrium during Embryo Implantation. PLoS ONE 2015, 10, e0122202. [Google Scholar] [CrossRef] [PubMed]
- Ozawa, M.; Sakatani, M.; Yao, J.; Shanker, S.; Yu, F.; Yamashita, R.; Wakabayashi, S.; Nakai, K.; Dobbs, K.B.; Sudano, M.J.; et al. Global gene expression of the inner cell mass and trophectoderm of the bovine blastocyst. BMC Dev. Biol. 2012, 12, 33. [Google Scholar] [CrossRef] [PubMed]
- Rizos, D.; Clemente, M.; Bermejo-Alvarez, P.; De La Fuente, J.; Lonergan, P.; Gutiérrez-Adán, A. Consequences ofIn VitroCulture Conditions on Embryo Development and Quality. Reprod. Domest. Anim. 2008, 43, 44–50. [Google Scholar] [CrossRef]
- Sirard, M.-A. Activation of the embryonic genome. Reprod. Domest. Rumin. 2010, 7, 145–158. [Google Scholar] [CrossRef]
- Mondou, E.; Dufort, I.; Gohin, M.; Fournier, E.; Sirard, M.-A. Analysis of microRNAs and their precursors in bovine early embryonic development. Mol. Hum. Reprod. 2012, 18, 425–434. [Google Scholar] [CrossRef]
- Tripurani, S.K.; Wee, G.; Lee, K.-B.; Smith, G.W.; Wang, L.; Yao, J. MicroRNA-212 Post-Transcriptionally Regulates Oocyte-Specific Basic-Helix-Loop-Helix Transcription Factor, Factor in the Germline Alpha (FIGLA), during Bovine Early Embryogenesis. PLoS ONE 2013, 8, e76114. [Google Scholar] [CrossRef]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, Y.J.; Gardiner, C.; Poli, M.; Turner, K.; Child, T.; Sargent, I.L. O-253 Human embryos release extracellular vesicles which may act as indicators of embryo quality. Session 66: Embryo quality: Does it predict pregnancy? Hum. Reprod. 2013, 28, 104. [Google Scholar] [CrossRef][Green Version]
- Abu-Halima, M.; Häusler, S.; Backes, C.; Fehlmann, T.; Staib, C.; Nestel, S.; Nazarenko, I.; Meese, E.; Keller, A. Micro-ribonucleic acids and extracellular vesicles repertoire in the spent culture media is altered in women undergoing In Vitro Fertilization. Sci. Rep. 2017, 7, 13525. [Google Scholar] [CrossRef] [PubMed]
- Pallinger, E.; Bognar, Z.; Bodis, J.; Csabai, T.; Farkas, N.; Godony, K.; Varnagy, A.; Buzas, E.; Szekeres-Bartho, J. A simple and rapid flow cytometry-based assay to identify a competent embryo prior to embryo transfer. Sci. Rep. 2017, 7, 39927. [Google Scholar] [CrossRef]
- Almiñana, C.; Tsikis, G.; Labas, V.; Uzbekov, R.; Da Silveira, J.C.; Bauersachs, S.; Mermillod, P. Deciphering the oviductal extracellular vesicles content across the estrous cycle: Implications for the gametes-oviduct interactions and the environment of the potential embryo. BMC Genom. 2018, 19, 1–27. [Google Scholar] [CrossRef]
- Ng, Y.H.; Rome, S.; Jalabert, A.; Forterre, A.; Singh, H.; Hincks, C.L.; Salamonsen, L.A. Endometrial Exosomes/Microvesicles in the Uterine Microenvironment: A New Paradigm for Embryo-Endometrial Cross Talk at Implantation. PLoS ONE 2013, 8, e58502. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, J.; Li, M.-Q.; Xu, J.; Zhang, J.-P.; Jin, L.-P. MicroRNA-184 promotes apoptosis of trophoblast cells via targeting WIG1 and induces early spontaneous abortion. Cell Death Dis. 2019, 10, 1–9. [Google Scholar] [CrossRef]
- Lin, X.; Pavani, K.C.; Smits, K.; Deforce, D.; Heindryckx, B.; Van Soom, A.; Peelman, L. Bta-miR-10b Secreted by Bovine Embryos Negatively Impacts Preimplantation Embryo Quality. Front. Genet. 2019, 10, 757. [Google Scholar] [CrossRef]
- Xiang, J.; Xing, Y.; Long, C.; Hou, D.; Liu, F.; Zhang, Y.; Lu, Z.; Wang, J.; Zuo, Y.; Li, X. Fatty acid metabolism as an indicator for the maternal–to–zygotic transition in porcine IVF embryos revealed by RNA sequencing. Theriogenology 2020, 151, 128–136. [Google Scholar] [CrossRef]
- Sugimoto, M.; Sasaki, S.; Gotoh, Y.; Nakamura, Y.; Aoyagi, Y.; Kawahara, T.; Sugimoto, Y. Genetic variants related to gap junctions and hormone secretion influence conception rates in cows. Proc. Natl. Acad. Sci. USA 2013, 110, 19495–19500. [Google Scholar] [CrossRef]
- Sun, J.; Deng, G.; Ruan, X.; Chen, S.; Liao, H.; Liu, X.; Li, J.; Zhao, G.; Gaopi, D. Exosomal MicroRNAs in Serum as Potential Biomarkers for Ectopic Pregnancy. BioMed. Res. Int. 2020, 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hromadnikova, I.; Kotlabova, K.; Hympanova, L.; Krofta, L. Gestational hypertension, preeclampsia and intrauterine growth restriction induce dysregulation of cardiovascular and cerebrovascular disease associated microRNAs in maternal whole peripheral blood. Thromb. Res. 2016, 137, 126–140. [Google Scholar] [CrossRef]
- Yang, Q.; Gu, W.-W.; Gu, Y.; Yan, N.-N.; Mao, Y.-Y.; Zhen, X.-X.; Wang, J.-M.; Yang, J.; Shi, H.-J.; Zhang, X. Association of the peripheral blood levels of circulating microRNAs with both recurrent miscarriage and the outcomes of embryo transfer in an in vitro fertilization process. J. Transl. Med. 2018, 16, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.G.; Roberts, C.W.M. SWI/SNF nucleosome remodellers and cancer. Nat. Rev. Cancer 2011, 11, 481–492. [Google Scholar] [CrossRef] [PubMed]
- Stopka, T.; Skoultchi, A.I. The ISWI ATPase Snf2h is required for early mouse development. Proc. Natl. Acad. Sci. USA 2003, 100, 14097–14102. [Google Scholar] [CrossRef] [PubMed]
- Torres-Padilla, M.E.; Zernicka-Goetz, M. Role of TIF1α as a modulator of embryonic transcription in the mouse zygote. J. Cell Biol. 2006, 174, 329–338. [Google Scholar] [CrossRef]
- Nishibori, Y.; Katayama, K.; Parikka, M.; Oddsson, A.; Nukui, M.; Hultenby, K.; Wernerson, A.; He, B.; Ebarasi, L.; Raschperger, E.; et al. Glcci1 Deficiency Leads to Proteinuria. J. Am. Soc. Nephrol. 2011, 22, 2037–2046. [Google Scholar] [CrossRef]
- Lee, Y.; Fryer, J.D.; Kang, H.; Crespo-Barreto, J.; Bowman, A.B.; Gao, Y.; Kahle, J.J.; Hong, J.S.; Kheradmand, F.; Orr, H.T.; et al. ATXN1 Protein Family and CIC Regulate Extracellular Matrix Remodeling and Lung Alveolarization. Dev. Cell 2011, 21, 746–757. [Google Scholar] [CrossRef]
- Ouyang, X.; Ahmad, I.; Johnson, M.S.; Redmann, M.; Craver, J.; Wani, W.Y.; Benavides, G.A.; Chacko, B.; Li, P.; Young, M.; et al. Nuclear receptor binding factor 2 (NRBF2) is required for learning and memory. Lab. Investig. 2020, 100, 1238–1251. [Google Scholar] [CrossRef]
- Wang, Y.; Medvid, R.; Melton, C.; Jaenisch, R.; Blelloch, R. DGCR8 is essential for microRNA biogenesis and silencing of embryonic stem cell self-renewal. Nat. Genet. 2007, 39, 380–385. [Google Scholar] [CrossRef]
- Sirohi, V.K.; Gupta, K.; Kumar, R.; Shukla, V.; Dwivedi, A. Selective estrogen receptor modulator ormeloxifene suppresses embryo implantation via inducing miR-140 and targeting insulin-like growth factor 1 receptor in rat uterus. J. Steroid Biochem. Mol. Biol. 2018, 178, 272–282. [Google Scholar] [CrossRef]
- McCarthy, S.D.; Roche, J.F.; Forde, N. Temporal changes in endometrial gene expression and protein localization of members of the IGF family in cattle: Effects of progesterone and pregnancy. Physiol. Genom. 2012, 44, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Green, D.; Dalmay, T.; Fraser, W.D. Role of miR-140 in embryonic bone development and cancer. Clin. Sci. 2015, 129, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Goissis, M.D.; Cibelli, J.B. Functional characterization of CDX2 during bovine preimplantation development in vitro. Mol. Reprod. Dev. 2014, 81, 962–970. [Google Scholar] [CrossRef] [PubMed]
- Yamada, N.; Heishima, K.; Akao, Y.; Senda, T. Extracellular Vesicles Containing MicroRNA-92a-3p Facilitate Partial Endothelial-Mesenchymal Transition and Angiogenesis in Endothelial Cells. Int. J. Mol. Sci. 2019, 20, 4406. [Google Scholar] [CrossRef] [PubMed]
- Shirayoshi, Y.; Okada, T.S.; Takeichi, M. The calcium-dependent cell-cell adhesion system regulates inner cell mass formation and cell surface polalrization in early mouse development. Cell 1983, 35, 631–638. [Google Scholar] [CrossRef]
- Stephenson, R.O.; Yamanaka, Y.; Rossant, J. Disorganized epithelial polarity and excess trophectoderm cell fate in preimplantation embryos lacking E-cadherin. Development 2010, 137, 3383–3391. [Google Scholar] [CrossRef]
- Sathanawongs, A.; Nganvongpanit, K.; Mekchay, S. Expression patterns of cell adhesion molecules in bovine preimplantation embryos cultured in vitro. Thai J. Vet. Med. 2012, 42, 455–461. [Google Scholar]
- Karagkouni, D.; Paraskevopoulou, M.D.; Chatzopoulos, S.; Vlachos, I.S.; Tastsoglou, S.; Kanellos, I.; Papadimitriou, D.; Kavakiotis, I.; Maniou, S.; Skoufos, G.; et al. DIANA-TarBase v8: A decade-long collection of experimentally supported miRNA–gene interactions. Nucleic Acids Res. 2017, 46, D239–D245. [Google Scholar] [CrossRef]
- Bogutz, A.B.; Oh-McGinnis, R.; Jacob, K.J.; Ho-Lau, R.; Gu, T.; Gertsenstein, M.; Nagy, A.; Lefebvre, L. Transcription factor ASCL2 is required for development of the glycogen trophoblast cell lineage. PLoS Genet. 2018, 14, e1007587. [Google Scholar] [CrossRef]
- Qu, H.-M.; Qu, L.-P.; Pan, X.-Z.; Mu, L.-S. Upregulated miR-222 targets BCL2L11 and promotes apoptosis of mesenchymal stem cells in preeclampsia patients in response to severe hypoxia. Int. J. Clin. Exp. Pathol. 2018, 11, 110–119. [Google Scholar]
- Fear, J.M.; Hansen, P.J. Developmental Changes in Expression of Genes Involved in Regulation of Apoptosis in the Bovine Preimplantation Embryo. Biol. Reprod. 2011, 84, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Maida, Y.; Takakura, M.; Nishiuchi, T.; Yoshimoto, T.; Kyo, S. Exosomal transfer of functional small RNAs mediates cancer-stroma communication in human endometrium. Cancer Med. 2016, 5, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Velasquez, A.E.; Castro, F.O.; Veraguas, D.; Cox, J.F.; Lara, E.; Briones, M.; Rodriguez-Alvarez, L. Splitting of IVP bovine blastocyst affects morphology and gene expression of resulting demi-embryos during in vitro culture and in vivo elongation. Zygote 2014, 24, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Bó, G.A.; Mapletoft, R.J. Evaluation and classification of bovine embryos. Anim. Reprod. 2013, 10, 344–348. [Google Scholar]
- Théry, C.; Amigorena, S.; Raposo, G.; Clayton, A. Isolation and Characterization of Exosomes from Cell Culture Supernatants and Biological Fluids. Curr. Protoc. Cell Biol. 2006, 30, 3.22.1–3.22.29. [Google Scholar] [CrossRef]
- Barturen, G.; Rueda, A.; Hamberg, M.; Alganza, A.; Lebron, R.; Kotsyfakis, M.; Shi, B.-J.; Koppers-Lalic, D.; Hackenberg, M. sRNAbench: Profiling of small RNAs and its sequence variants in single or multi-species high-throughput experiments. Methods Next Gener. Seq. 2014, 1, 21–31. [Google Scholar] [CrossRef]
- Vlachos, I.S.; Zagganas, K.; Paraskevopoulou, M.D.; Georgakilas, G.; Karagkouni, D.; Vergoulis, T.; Dalamagas, T.; Hatzigeorgiou, A.G. DIANA-miRPath v3.0: Deciphering microRNA function with experimental support. Nucleic Acids Res. 2015, 43, W460–W466. [Google Scholar] [CrossRef]
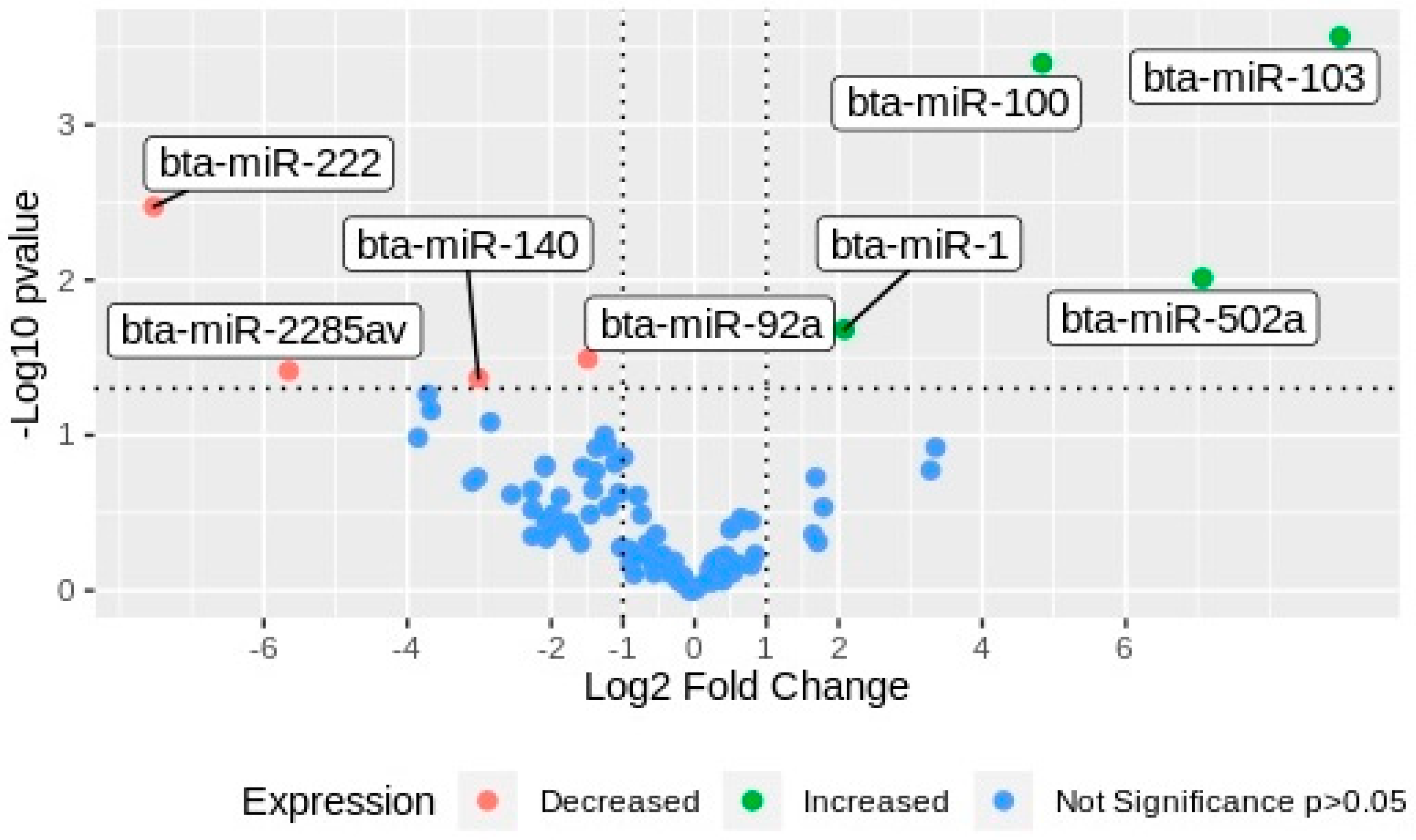
| miR | Significance Comparing G1 vs. G2 | Gene Expression Respect to G2 |
|---|---|---|
| bta-miR-103 | 0.0003 | Increased |
| bta-miR-100 | 0.0004 | Increased |
| bta-miR-502a | 0.0097 | Increased |
| bta-miR-1 | 0.0208 | Increased |
| bta-miR-101 | 0.1199 | Increased |
| bta-let-7e | 0.1682 | Increased |
| bta-miR-215 | 0.1876 | Increased |
| bta-miR-205 | 0.2915 | Increased |
| bta-miR-193a-3p | 0.4369 | Increased |
| bta-miR-224 | 0.4895 | Increased |
| bta-miR-222 | 0.0034 | Decreased |
| bta-miR-92a | 0.0323 | Decreased |
| bta-miR-2285av | 0.0385 | Decreased |
| bta-miR-140 | 0.0429 | Decreased |
| bta-miR-25 | 0.0545 | Decreased |
| bta-miR-9-5p | 0.0692 | Decreased |
| bta-miR-16b | 0.0824 | Decreased |
| bta-miR-191 | 0.1003 | Decreased |
| bta-miR-744 | 0.1038 | Decreased |
| bta-miR-451 | 0.1163 | Decreased |
| bta-miR-10b | 0.1211 | Decreased |
| bta-let-7f | 0.1519 | Decreased |
| bta-miR-2904 | 0.1561 | Decreased |
| bta-miR-11980 | 0.1609 | Decreased |
| bta-let-7g | 0.1609 | Decreased |
| bta-miR-30d | 0.1714 | Decreased |
| bta-miR-93 | 0.1873 | Decreased |
| bta-miR-378 | 0.1994 | Decreased |
| bta-let-7i | 0.2239 | Decreased |
| bta-miR-148b | 0.2253 | Decreased |
| bta-miR-143 | 0.2362 | Decreased |
| bta-miR-144 | 0.2412 | Decreased |
| bta-miR-151-3p | 0.2510 | Decreased |
| bta-miR-21-5p | 0.2891 | Decreased |
| bta-miR-221 | 0.3007 | Decreased |
| bta-miR-152 | 0.3234 | Decreased |
| bta-miR-455-5p | 0.3246 | Decreased |
| bta-miR-199a-3p | 0.3564 | Decreased |
| bta-miR-11995 | 0.3646 | Decreased |
| bta-miR-200a | 0.3960 | Decreased |
| bta-miR-96 | 0.4180 | Decreased |
| bta-miR-124b | 0.4465 | Decreased |
| bta-miR-124a | 0.4622 | Decreased |
| bta-miR-15b | 0.4951 | Decreased |
| bta-miR-92b | 0.5279 | Decreased |
| bta-miR-486 | 0.1387 | Equal |
| bta-miR-10a | 0.2450 | Equal |
| bta-miR-423-5p | 0.3259 | Equal |
| bta-let-7c | 0.3432 | Equal |
| bta-miR-27a-3p | 0.3529 | Equal |
| bta-miR-1246 | 0.3565 | Equal |
| bta-miR-27b | 0.3948 | Equal |
| bta-let-7b | 0.4056 | Equal |
| bta-miR-183 | 0.4368 | Equal |
| bta-miR-151-5p | 0.5007 | Equal |
| bta-miR-142-5p | 0.5462 | Equal |
| bta-miR-302b | 0.5876 | Equal |
| bta-miR-24-3p | 0.5886 | Equal |
| bta-miR-181a | 0.5970 | Equal |
| bta-miR-29b | 0.5970 | Equal |
| bta-miR-99a-5p | 0.6146 | Equal |
| bta-miR-29d-3p | 0.6151 | Equal |
| bta-miR-29c | 0.6178 | Equal |
| bta-miR-320a | 0.6476 | Equal |
| bta-miR-125b | 0.6481 | Equal |
| bta-miR-192 | 0.6536 | Equal |
| bta-miR-29a | 0.6548 | Equal |
| bta-miR-125a | 0.6586 | Equal |
| bta-miR-484 | 0.6634 | Equal |
| bta-miR-26b | 0.6787 | Equal |
| bta-miR-199a-5p | 0.6898 | Equal |
| bta-miR-30a-5p | 0.7072 | Equal |
| bta-miR-22-3p | 0.7340 | Equal |
| bta-miR-146a | 0.7652 | Equal |
| bta-miR-375 | 0.7673 | Equal |
| bta-miR-2478 | 0.7729 | Equal |
| bta-miR-122 | 0.7755 | Equal |
| bta-miR-141 | 0.7853 | Equal |
| bta-miR-30e-5p | 0.7925 | Equal |
| bta-miR-146b | 0.7971 | Equal |
| bta-miR-26a | 0.8062 | Equal |
| bta-miR-148a | 0.8207 | Equal |
| bta-miR-99b | 0.8406 | Equal |
| bta-miR-184 | 0.8484 | Equal |
| bta-miR-7 | 0.8697 | Equal |
| bta-miR-23b-3p | 0.8705 | Equal |
| bta-miR-423-3p | 0.8750 | Equal |
| bta-miR-30c | 0.8867 | Equal |
| bta-miR-10174-3p | 0.8955 | Equal |
| bta-let-7a-5p | 0.9635 | Equal |
| bta-miR-182 | 0.9746 | Equal |
| bta-miR-23a | 0.9817 | Equal |
| bta-miR-186 | 0.9912 | Equal |
| bta-miR-185 | 1 | Equal |
| bta-miR-2473 | 1 | Equal |
| KEEG Pathway | p-Value | Target Genes | miRNA |
|---|---|---|---|
| Fatty acid biosynthesis | 0.000005 | 1 | 1 |
| Fatty acid metabolism | 0.00017 | 4 | 2 |
| Lysine degradation | 0.00089 | 9 | 4 |
| Gap junction | 0.00089 | 19 | 4 |
| Signaling pathways regulating pluripotency of stem cells | 0.0046 | 27 | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melo-Baez, B.; Wong, Y.S.; Aguilera, C.J.; Cabezas, J.; Mançanares, A.C.F.; Riadi, G.; Castro, F.O.; Rodriguez-Alvarez, L. MicroRNAs from Extracellular Vesicles Secreted by Bovine Embryos as Early Biomarkers of Developmental Competence. Int. J. Mol. Sci. 2020, 21, 8888. https://doi.org/10.3390/ijms21238888
Melo-Baez B, Wong YS, Aguilera CJ, Cabezas J, Mançanares ACF, Riadi G, Castro FO, Rodriguez-Alvarez L. MicroRNAs from Extracellular Vesicles Secreted by Bovine Embryos as Early Biomarkers of Developmental Competence. International Journal of Molecular Sciences. 2020; 21(23):8888. https://doi.org/10.3390/ijms21238888
Chicago/Turabian StyleMelo-Baez, Bárbara, Yat S. Wong, Constanza J. Aguilera, Joel Cabezas, Ana C. F. Mançanares, Gonzalo Riadi, Fidel O. Castro, and Lleretny Rodriguez-Alvarez. 2020. "MicroRNAs from Extracellular Vesicles Secreted by Bovine Embryos as Early Biomarkers of Developmental Competence" International Journal of Molecular Sciences 21, no. 23: 8888. https://doi.org/10.3390/ijms21238888
APA StyleMelo-Baez, B., Wong, Y. S., Aguilera, C. J., Cabezas, J., Mançanares, A. C. F., Riadi, G., Castro, F. O., & Rodriguez-Alvarez, L. (2020). MicroRNAs from Extracellular Vesicles Secreted by Bovine Embryos as Early Biomarkers of Developmental Competence. International Journal of Molecular Sciences, 21(23), 8888. https://doi.org/10.3390/ijms21238888







