Thymus-Pineal Gland Axis: Revisiting Its Role in Human Life and Ageing
Abstract
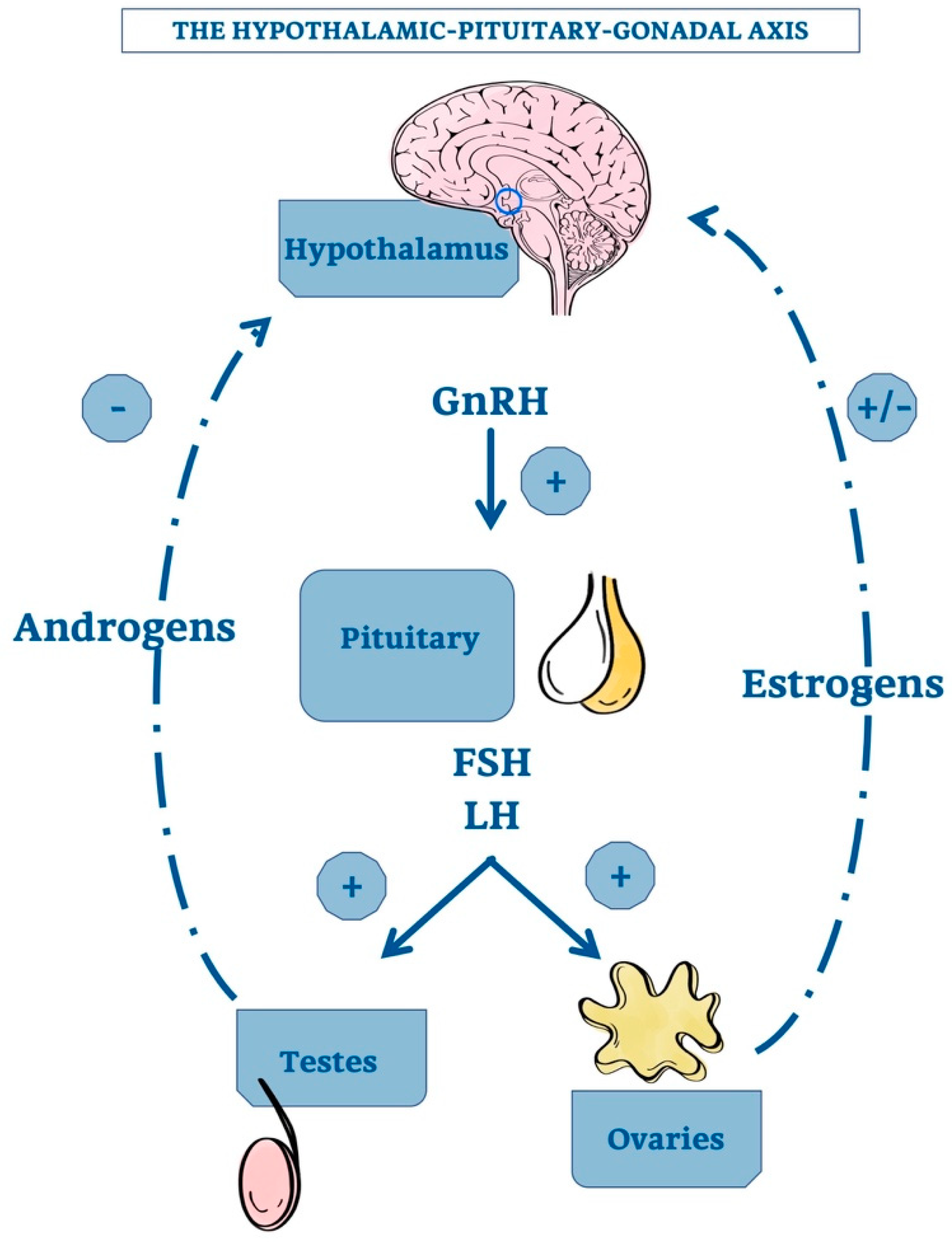
1. Introduction
2. Revisiting the Thymus Gland
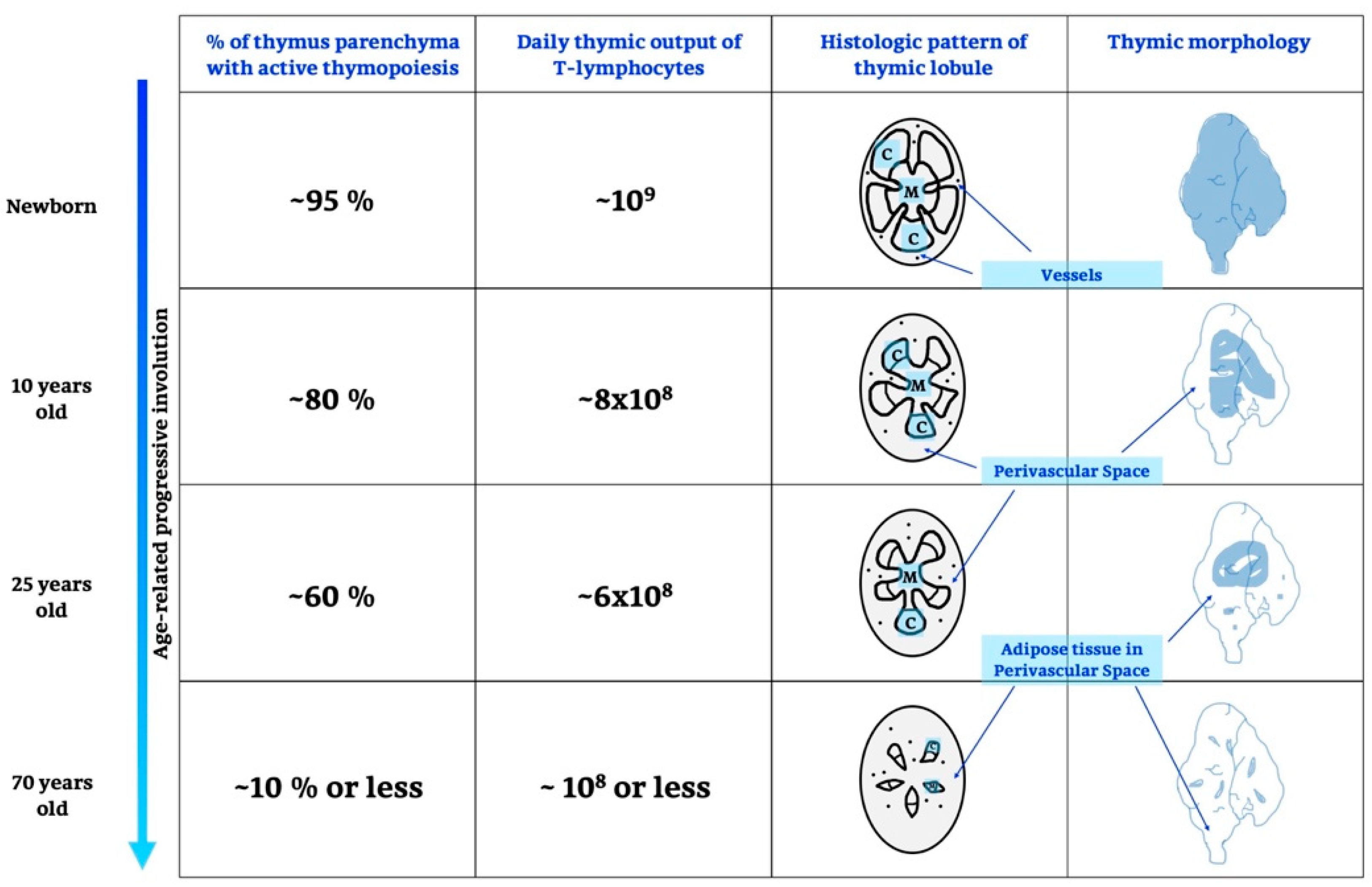
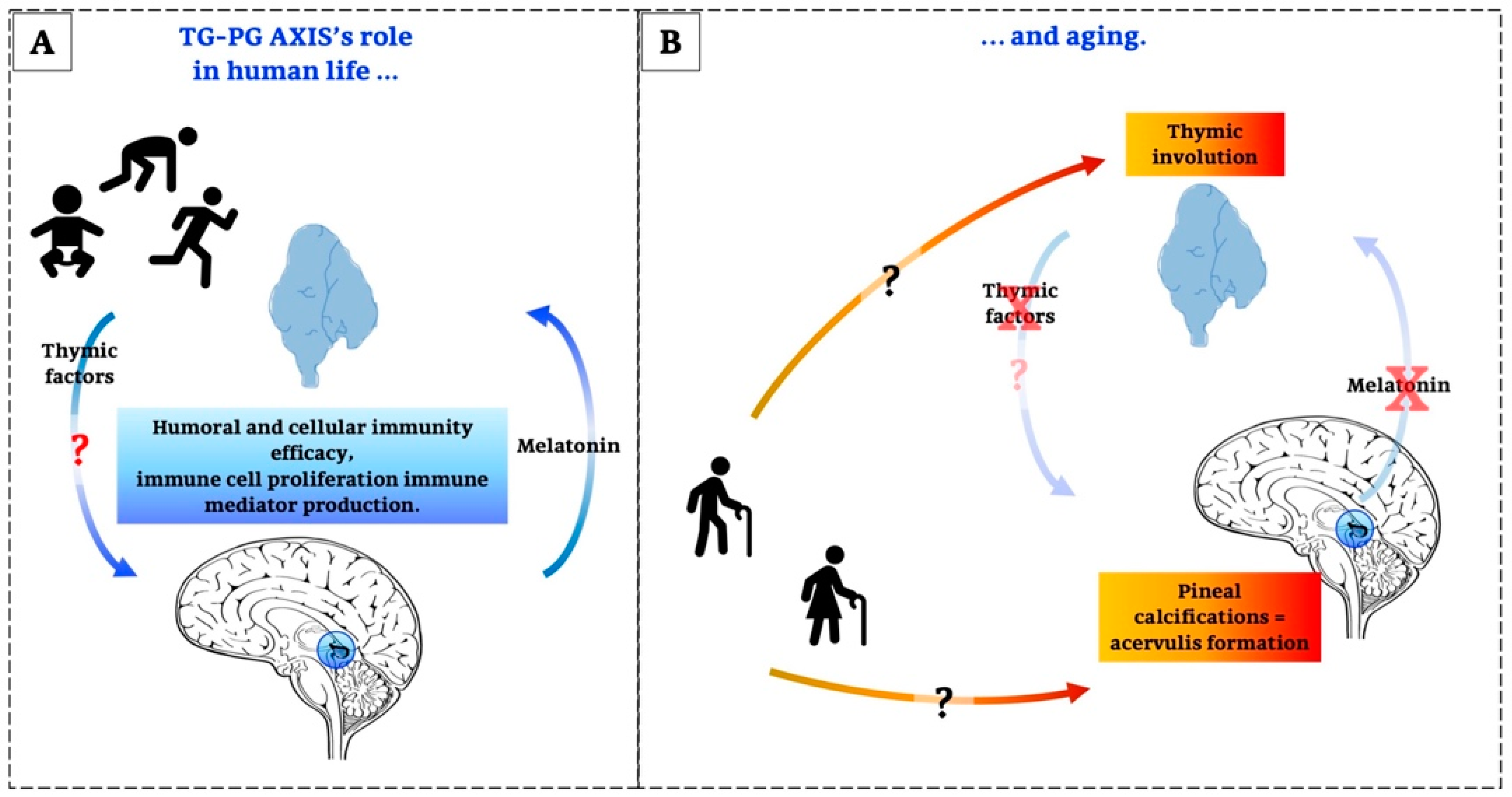
2.1. Life and Involution of the Thymus Gland
2.2. Thymic Factors and Their Effects
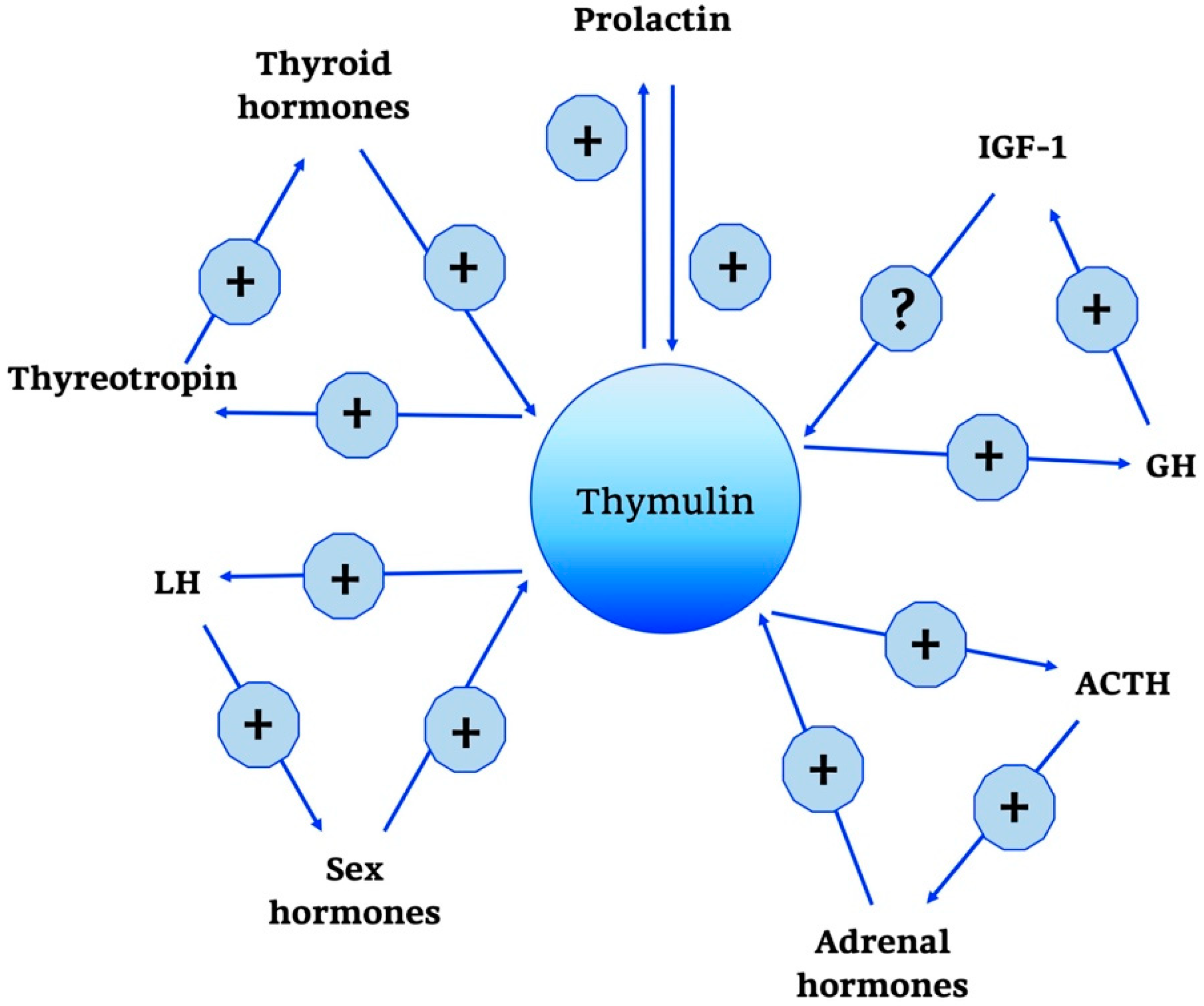
2.2.1. Thymulin
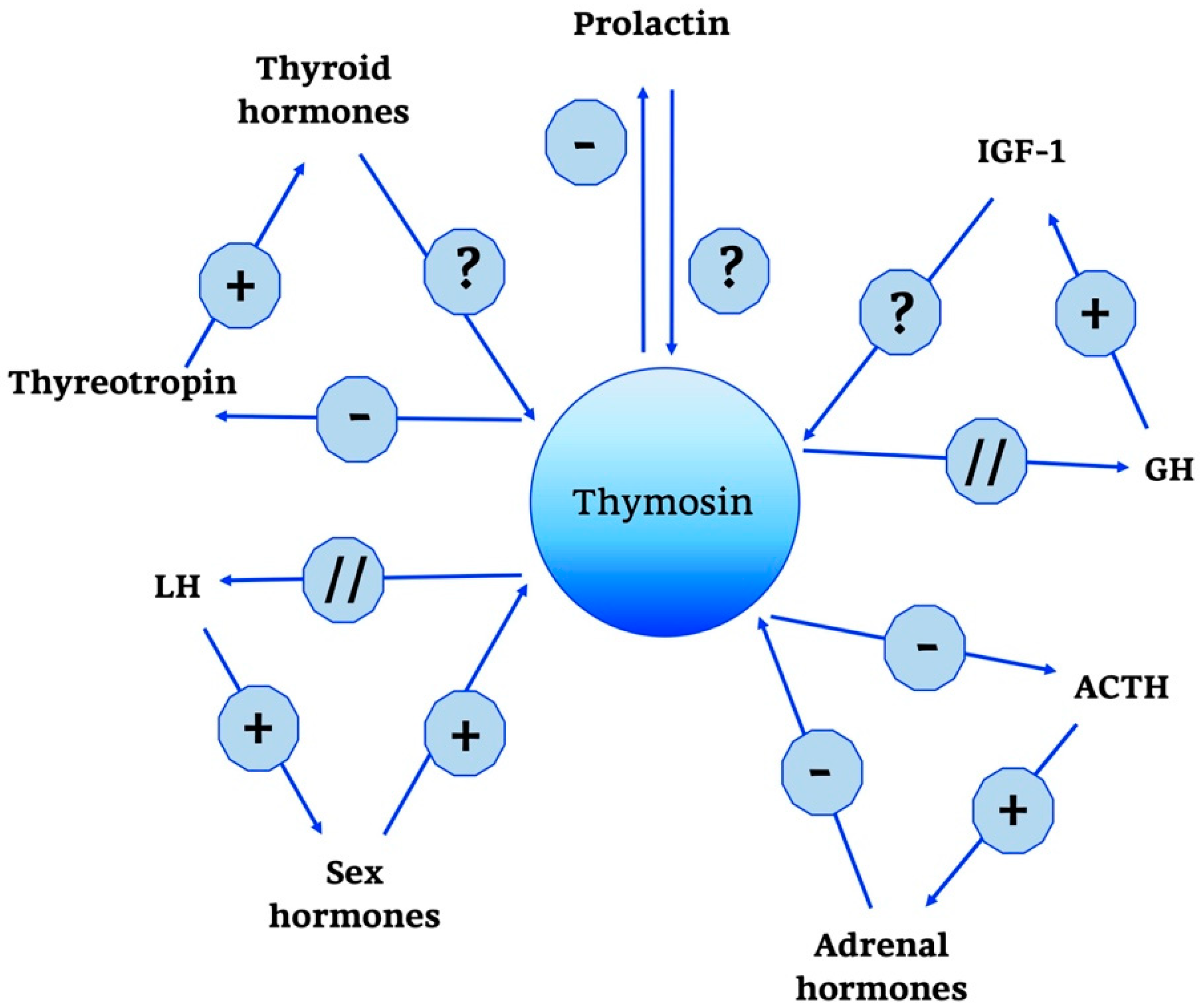
2.2.2. Thymosin
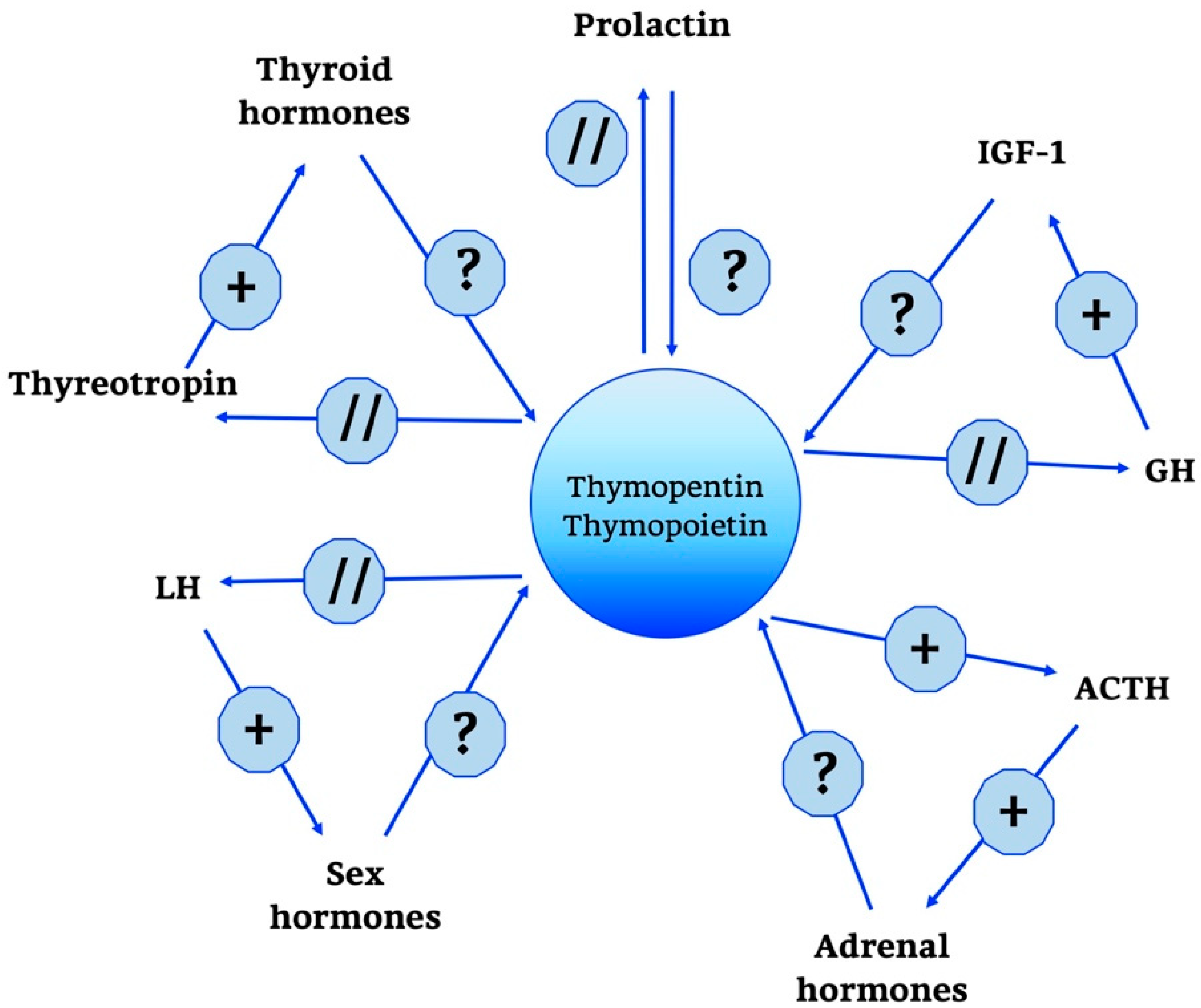
2.2.3. Thymopoietin
2.2.4. Thymic Humoral Factor
3. Revisiting the Pineal Gland
3.1. Principal Characteristics of Mammalian Pineal Gland
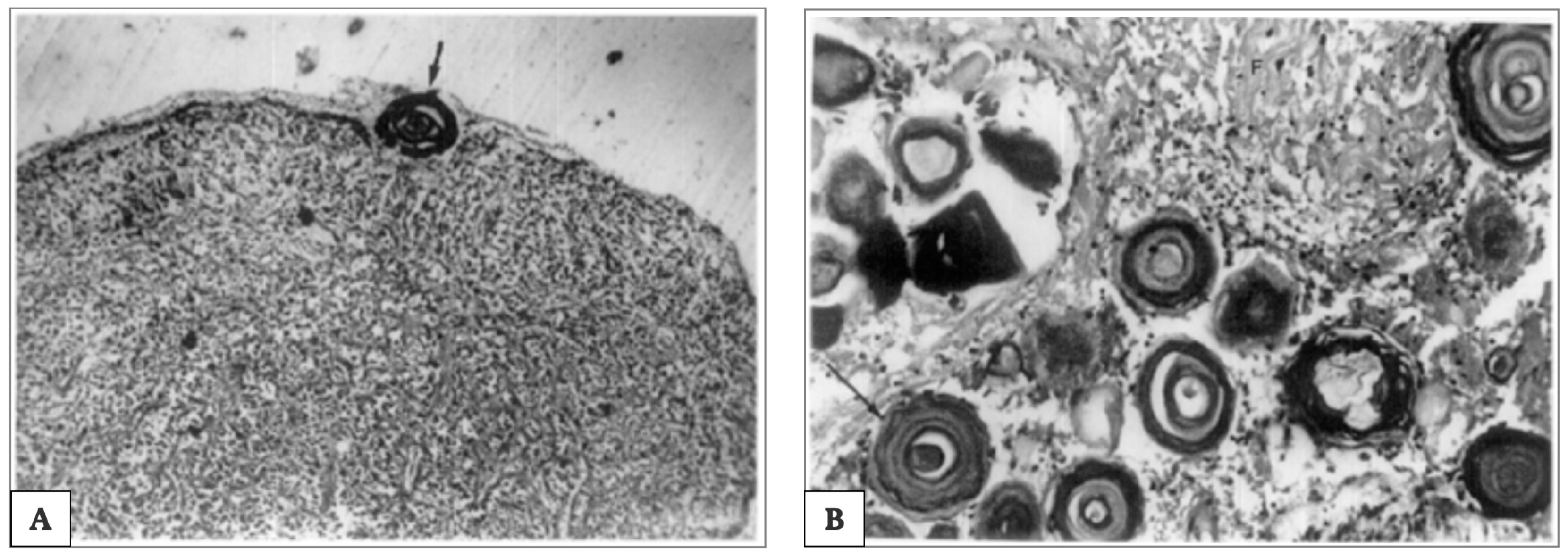
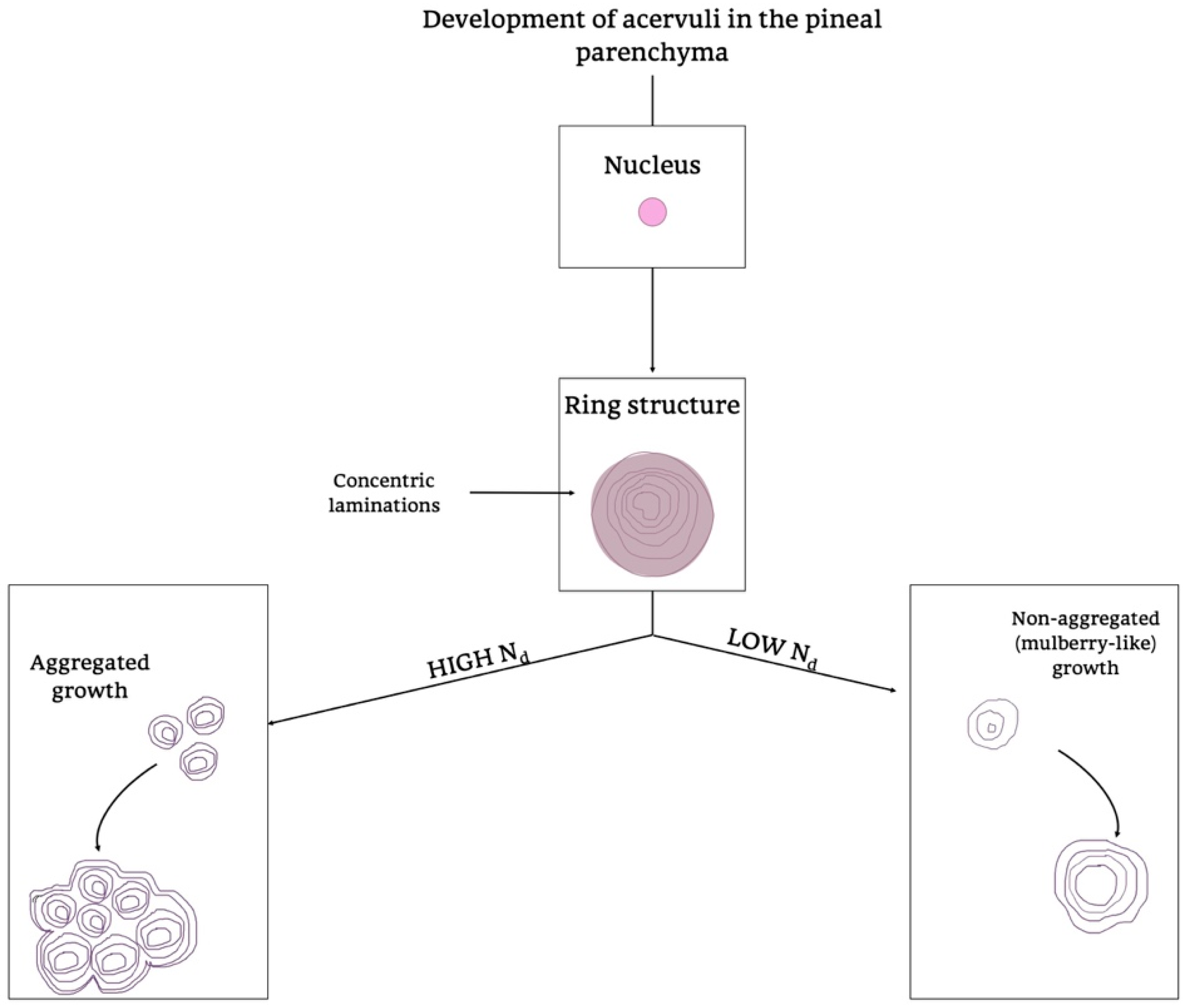
3.2. Pineal Ageing
3.3. Pineal Products and Their Effects
TRH
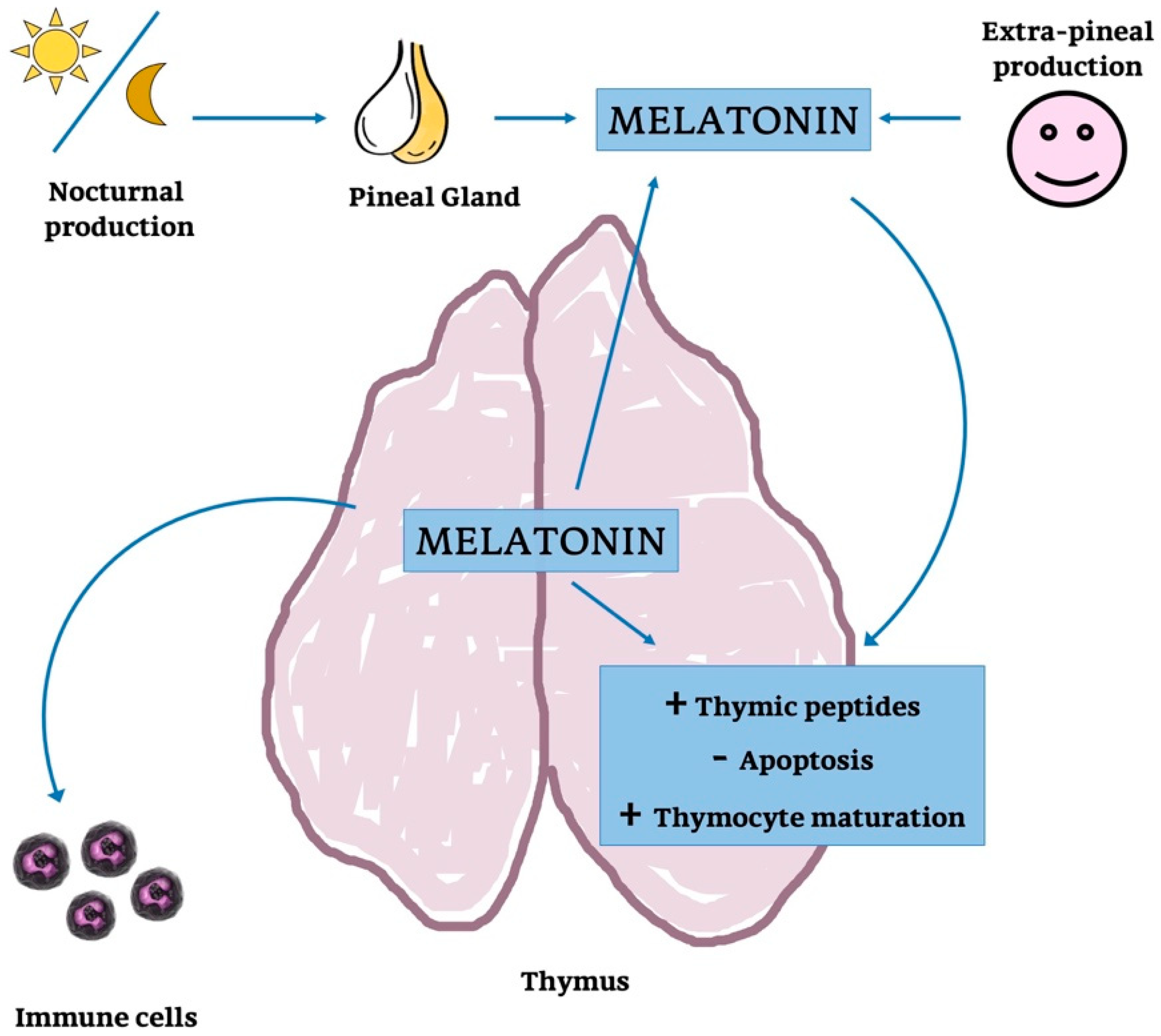
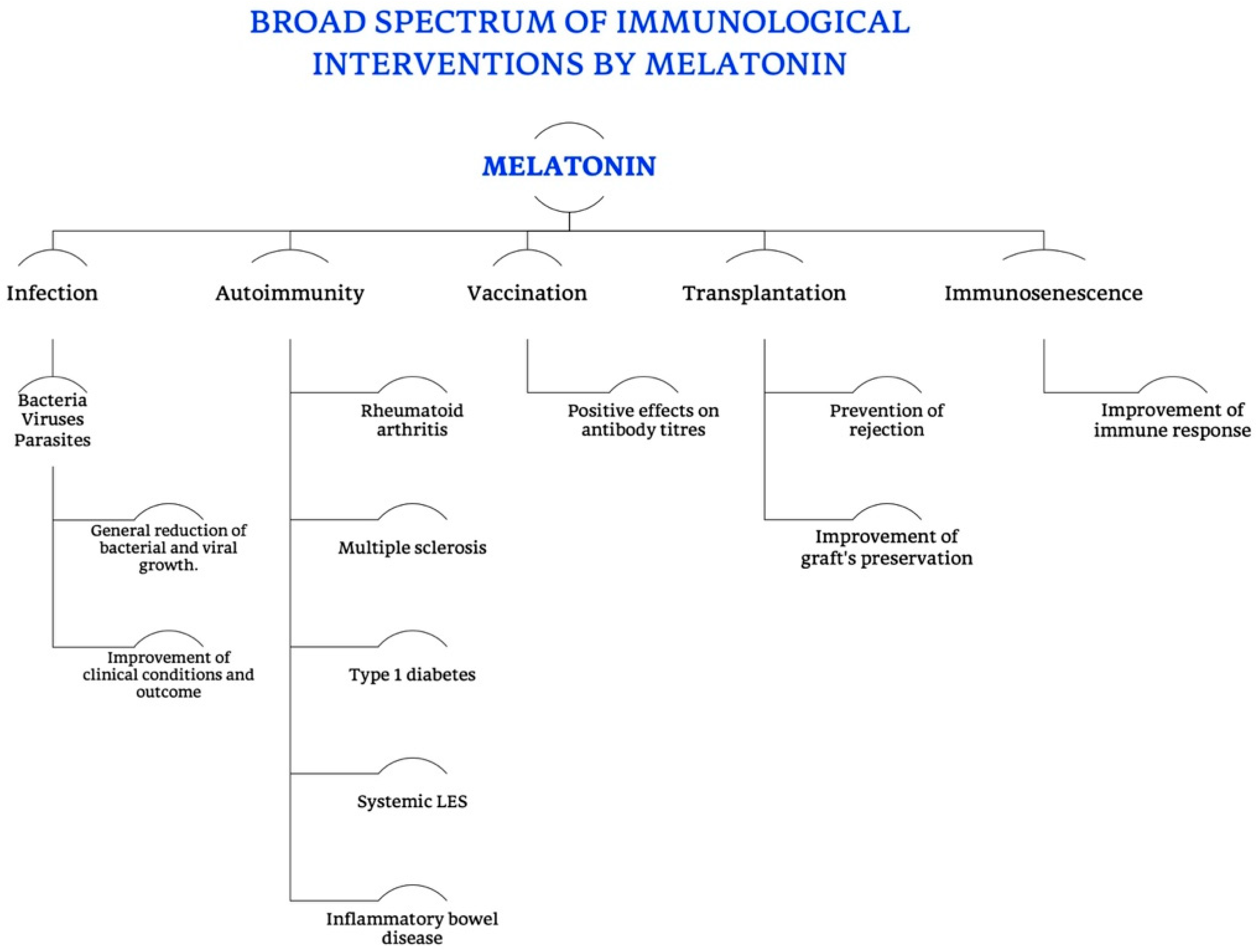
4. Melatonin
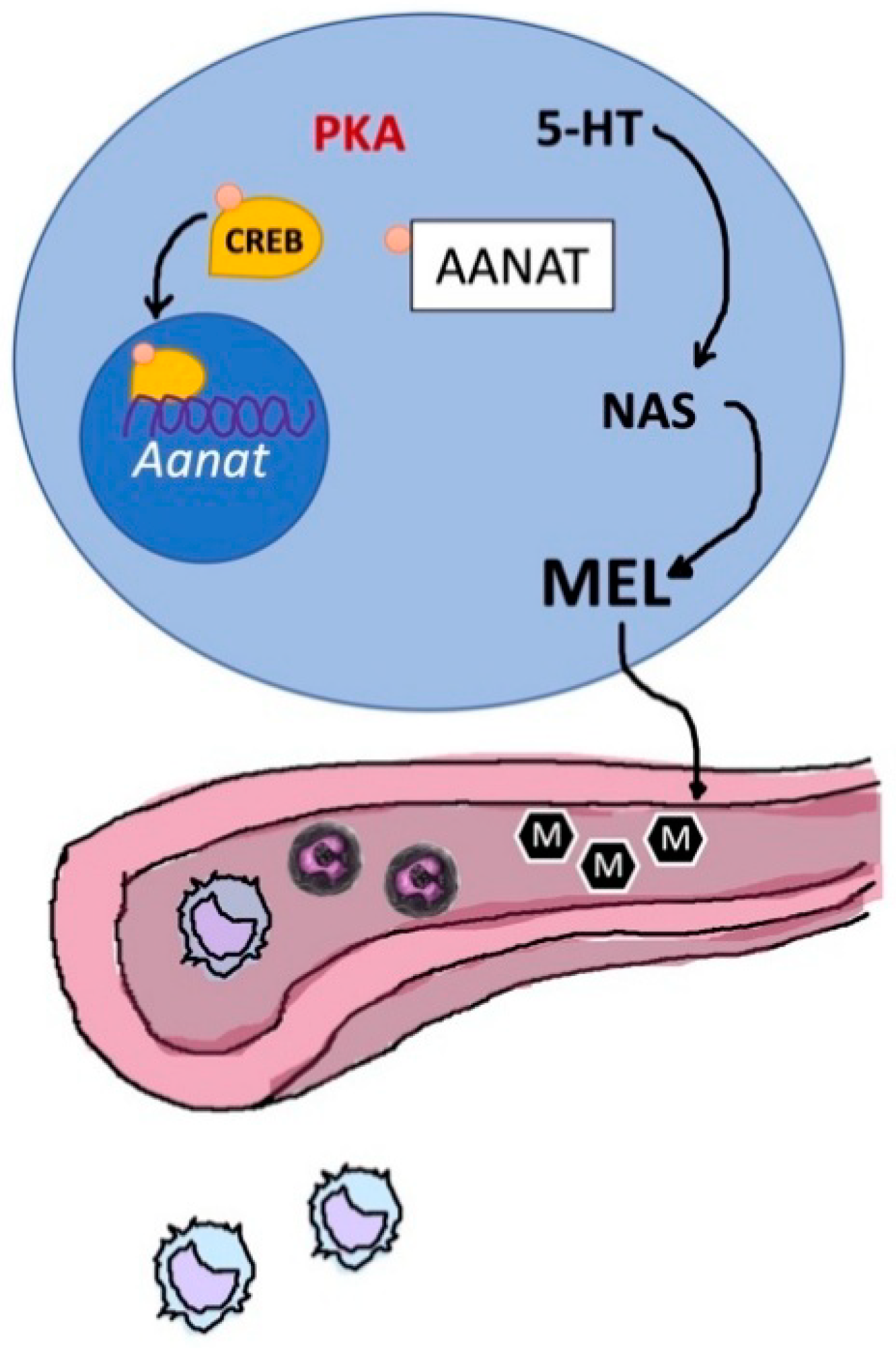
4.1. Thymic Melatonin
4.2. Pineal Melatonin
5. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| PG | Pineal Gland |
| TG | Thymus Gland |
| TG-PG axis | Thymus-Pineal Axis |
| ROS | Reactive Oxygen Species |
| NAD+ | Nicotinamide Adenine Dinucleotide |
| SIRT1 | Directory Of Open Access Journals |
| MT | Melatonin |
| miRNAs | MicroRNAs |
| circRNAs | miRNA-Sponging Circular RNAs |
| DBP | D-Box-Binding Protein |
| ETP cells | Early T Lineage Progenitor Cells |
| LIF | Leukemia Inhibitory Factor |
| OSM | Oncostatin M |
| GnRH | Gonadotropin Releasing Hormone |
| LH | Luteinizing Hormone |
| FSH | Follicle Stimulating Hormone |
| GH | Growth Hormone |
| IGF-1 | Insulin-Like Growth Factor-1 |
| SASP | Senescence-Associated Phenotype |
| FTS | Facteur Thymique Sérique - Thymulin |
| RE | Reticulo-Epithelial Tissue |
| ACTH | Adrenocorticotropic Hormone |
| TSH | Thyroid-Stimulating Hormone |
| sEAE | Severe Experimental Autoimmune Encephalomyelitis |
| TF3 | Thymosin Fraction-3 |
| TF5 | Thymosin Fraction-5 |
| Tβ4 | Thymosin Β4 |
| Tβ10 | Thymosin Β10 |
| Tα1 | Thymalfasin - Thymosin Α1 |
| ProTα | Prothymosin Α |
| INF-α | Interferon-Α |
| TRH | Thyrotropin Releasing Hormone |
| CRH | Corticotropin-Releasing Hormone |
| TP (TMPO) | Thymopoietin |
| LAP2 | Lamin-Associated Polypeptide 2 |
| HDAC1 | Histone Deacetylase 1 |
| TPα | Thymopoietin α |
| TPβ | Thymopoietin β |
| TPγ | Thymopoietin γ |
| HSP70 | Heat Shock Protein 70 |
| HSP90 | Heat Shock Protein 90 |
| THF | Thymic Humoral Factor |
| THF-γ2 | Thymic Humoral Factor-Γ2 |
| GFAP | Glial Cell Fibrillary Acid Protein |
| NPY | Neuropeptide Y |
| PACAP | Pituitary Adenylylcyclase Activating Peptide |
| VIP | Vasoactive Intestinal Polypeptide |
| Nd | Nuclear Density |
| MAO A | Monoamine Oxidase A |
| MAO B | Monoamine Oxidase B |
| AVT | Arginine Vasotocin |
| TRH-IF | TRH Inhibitory Factor |
| TPH | Tryptophan Hydroxylase |
| AADC | Aromatic Amino Acid Decarboxylase |
| AA-NAT | Aralkylamine-N-Acetyltransferase |
| THF | Thymic Humoral Factor |
| ASMT (HIOMT) | N-Acetylserotonin-O-Methyltransferase (Hydroxyindole-O-Methyltransferase) |
| SCN | Suprachiasmatic Nucleus |
| GIT | Gastro-Intestinal Tract |
| NQO2 | NRH Quinone Oxidoreductase 2 |
| CaM | Calmodulin |
| RORs | Retinoid Orphan Receptors |
| 5-HT | 5-hydroxytryptamine |
| NAS | N-acetylserotonin |
| PKA | Protein Kinases A |
| CREB | cAMP Response Element-Binding Protein |
References
- Johnsson, A. Light, circadian and circannual rhythms. In Solar Radiation and Human Health; Bjertness, E., Ed.; The Norwegian Academy of Science and Letters: Oslo, Norway, 2008; pp. 57–75. [Google Scholar]
- Labunets, I.F. Age-Related Changes in the Melatonin and Thymulin Biorhythms as Risk Factors for Human Neurodegenerative Diseases. Gerontol. Geriatr. Stud. 2017, 1, 18–22. [Google Scholar] [CrossRef]
- Reiter, R.J.; Rosales-Corral, S.; Sharma, R. Circadian disruption, melatonin rhythm perturbations and their contributions to chaotic physiology. Adv. Med. Sci. 2020, 65, 394–402. [Google Scholar] [CrossRef]
- Zhu, Q.; Belden, W.J. Molecular Regulation of Circadian Chromatin. J. Mol. Biol. 2020, 432, 3466–3482. [Google Scholar] [CrossRef]
- Pittendrigh, C.S. Temporal organization: Reflections of a Darwinian clock-watcher. Annu. Rev. Physiol. 1993, 55, 17–54. [Google Scholar] [CrossRef] [PubMed]
- Nikaido, S.S.; Johnson, C.H. Daily and circadian variation in survival from ultraviolet radiation in Chlamydomonas Reinhardtii. Photochem. Photobiol. 2000, 71, 758–765. [Google Scholar] [CrossRef]
- Johnson, C.H. Circadian clocks and cell division. What’s the pacemaker? Cell Cycle 2010, 9, 3864–3873. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Meltz, M.L. Melatonin protects human blood lymphocytes from radiation-induced chromosome damage. Mutat. Res. Lett. 1995, 346, 23–31. [Google Scholar]
- Reiter, R.J.; Tan, D.-X.; Herman, T.S.; Thomas, C.R., Jr. Melatonin as a radioprotective agent: A review. Int. J. Radiat. Oncol. Biol. Phys. 2004, 59, 639–653. [Google Scholar]
- Tahara, Y.; Aoyama, S.; Shibata, S. The mammalian circadian clock and its entrainment by stress and exercise. J. Physiol. Sci. 2017, 67, 1–10. [Google Scholar] [CrossRef]
- Rao, R.; Androulakis, I.P. The physiological significance of the circadian dynamics of the HPA axis: Interplay between circadian rhythms, allostasis and stress resilience. Horm. Behav. 2019, 110, 77–89. [Google Scholar] [CrossRef]
- Lananna, B.V.; Musiek, E.S. The wrinkling of time: Aging, inflammation, oxidative stress, and the circadian clock in neurodegeneration. Neurobiol. Dis. 2020, 139, 104832. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R. Melatonin and chromatin. Melatonin Res. 2019, 2, 67–93. [Google Scholar] [CrossRef]
- Hardeland, R. Noncoding RNAs: Bridging regulation of circadian rhythms and inflammation. Adv. Neuroimmune Biol. 2019, 7, 155–177. [Google Scholar] [CrossRef]
- Marcheva, B.; Ramsey, K.M.; Buhr, E.D.; Kobayashi, Y.; Su, H.; Ko, C.H.; Ivanova, G.; Omura, C.; Mo, S.; Vitaterna, M.H.; et al. Disruption of the clock components CLOCK and BMAL1 leads to hypoinsulinaemia and diabetes. Nature 2010, 466, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Madrid, J.A.; Tan, D.-X.; Reiter, R.J. Melatonin, the circadian multioscillator system and health: The need for detailed analyses of peripheral melatonin signaling. J. Pineal Res. 2012, 52, 139–166. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.-X.; Korkmaz, A.; Erren, T.C.; Piekarski, C.; Tamura, H.; Manchester, L.C. Light at night, chronodisruption, melatonin suppression, and cancer risk: A review. Crit. Rev. Oncog. 2007, 13, 303–328. [Google Scholar] [CrossRef] [PubMed]
- Erren, T.C.; Pape, H.G.; Reiter, R.J.; Piekarski, C. Chronodisruption and cancer. Naturwissenschaften 2008, 95, 367–382. [Google Scholar] [CrossRef] [PubMed]
- Mirza-Aghazadeh-Attari, M.; Reiter, R.J.; Rikhtegar, R.; Jalili, J.; Hajalioghli, P.; Mihanfar, A.; Majidinia, M.; Yousefi, B. Melatonin: An atypical hormone with major functions in the regulation of angiogenesis. IUBMB Life 2020, 72, 1560–1584. [Google Scholar] [CrossRef] [PubMed]
- Mate, I.; Madrid, J.A.; De la Fuente, M. Chronobiology of the neuroimmunoendocrine system and aging. Curr. Pharm. Des. 2014, 20, 4642–4655. [Google Scholar] [CrossRef]
- Goya, R.G.; Sosa, Y.E.; Cónsole, G.M.; Dardenne, M. Altered thyrotropic and somatotropic responses to environmental challenges in congenitally athymic mice. Brain Behav. Immun. 1995, 9, 79–86. [Google Scholar] [CrossRef][Green Version]
- Reiter, R.J.; Tan, D.X.; Madrid, J.A.; Erren, T.C. When the circadian clock becomes a ticking time bomb. Chronobiol. Int. 2012, 29, 1286–1287. [Google Scholar] [CrossRef]
- Paltsev, M.A.; Polyakova, V.O.; Kvetnoy, I.M.; Anderson, G.; Kvetnaia, T.V.; Linkova, N.S.; Paltseva, E.M.; Rubino, R.; De Cosmo, S.; De Cata, A.; et al. Morphofunctional and signaling molecules overlap of the pineal gland and thymus: Role and significance in aging. Oncotarget 2016, 7, 11972–11983. [Google Scholar] [CrossRef] [PubMed]
- Polyakova, V.O.; Linkova, N.S.; Kvetnoy, I.M.; Khavinson, V.K. Functional unity of the thymus and pineal gland and study of the mechanisms of aging. Bull. Exp. Biol. Med. 2011, 151, 627–630. [Google Scholar] [CrossRef] [PubMed]
- Csaba, G. The Immunoendocrine Thymus as a Pacemaker of Lifespan. Acta Microbiol. Immunol. Hung. 2016, 63, 139–158. [Google Scholar] [CrossRef] [PubMed]
- Strindhall, J.; Nilsson, B.O.; Löfgren, S.; Ernerudh, J.; Pawelec, G.; Johansson, B.; Wikby, A. No Immune Risk Profile among individuals who reach 100 years of age: Findings from the Swedish NONA immune longitudinal study. Exp. Gerontol. 2007, 42, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Ponnappan, S.; Ponnappan, U. Aging and immune function: Molecular mechanisms to interventions. Antioxid. Redox Signal. 2011, 14, 1551–1585. [Google Scholar] [CrossRef]
- Guerrero, J.M.; Reiter, R.J. Melatonin-immune system relationships. Curr. Top. Med. Chem. 2002, 2, 167–179. [Google Scholar] [CrossRef]
- Pierpaoli, W.; Bulian, D. The pineal aging and death program: Life prolongation in pre-aging pinealectomized mice. Ann. N. Y. Acad. Sci. 2005, 1057, 133–144. [Google Scholar] [CrossRef]
- Oner, H.; Ozan, E. Effects of gonadal hormones on thymus gland after bilateral ovariectomy and orchidectomy in rats. Arch. Androl. 2002, 48, 115–126. [Google Scholar] [CrossRef]
- Linkova, N.S.; Poliakova, V.O.; Trofimov, A.V.; Sevostianova, N.N.; Kvetnoĭ, I.M. Influence of peptides from pineal gland on thymus function at aging. Adv. Gerontol. 2010, 23, 543–546. [Google Scholar]
- Cardarelli, N.F. The role of a thymus-pineal axis in an immune mechanism of aging. J. Theor. Biol. 1990, 145, 397–405. [Google Scholar] [CrossRef]
- Hale, L.P. Histologic and molecular assessment of human thymus. Ann. Diagn. Pathol. 2004, 8, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Rodewald, H.R. Thymus organogenesis. Annu. Rev. Immunol. 2008, 26, 355–388. [Google Scholar] [CrossRef] [PubMed]
- Rezzani, R.; Nardo, L.; Favero, G.; Peroni, M.; Rodella, L.F. Thymus and aging: Morphological, radiological, and functional overview. Age 2014, 36, 313–351. [Google Scholar] [CrossRef]
- Ventevogel, M.S.; Sempowski, G.D. Thymic rejuvenation and aging. Curr. Opin. Immunol. 2013, 25, 516–522. [Google Scholar] [CrossRef]
- Masters, A.R.; Haynes, L.; Su, D.M.; Palmer, D.B. Immune senescence: Significance of the stromal microenvironment. Clin. Exp. Immunol. 2017, 187, 6–15. [Google Scholar] [CrossRef]
- Patel, S.; Rahmani, B.; Gandhi, J.; Seyam, O.; Joshi, G.; Reid, I.; Smith, N.L.; Waltzer, W.C.; Khan, S.A. Revisiting the pineal gland: A review of calcification, masses, precocious puberty, and melatonin functions. Int. J. Neurosci. 2020, 130, 464–475. [Google Scholar] [CrossRef]
- Boehm, T.; Swann, J.B. Thymus involution and regeneration: Two sides of the same coin? Nat. Rev. Immunol. 2013, 13, 831–838. [Google Scholar] [CrossRef]
- Hess, I.; Boehm, T. Intravital imaging of thymopoiesis reveals dynamic lympho-epithelial interactions. Immunity 2012, 36, 298–309. [Google Scholar] [CrossRef]
- Haynes, B.F.; Heinly, C.S. Early human T cell development: Analysis of the human thymus at the time of initial entry of hematopoietic stem cells into the fetal thymic microenvironment. J. Exp. Med. 1995, 181, 1445–1458. [Google Scholar] [CrossRef]
- Gray, D.H.; Seach, N.; Ueno, T.; Milton, M.K.; Liston, A.; Lew, A.M.; Goodnow, C.C.; Boyd, R.L. Developmental kinetics, turnover, and stimulatory capacity of thymic epithelial cells. Blood 2006, 108, 3777–3785. [Google Scholar] [CrossRef]
- Steinmann, G.G.; Klaus, B.; Müller-Hermelink, H.K. The involution of the ageing human thymic epithelium is independent of puberty. A morphometric study. Scand. J. Immunol. 1985, 22, 563–575. [Google Scholar] [CrossRef] [PubMed]
- Hale, J.S.; Boursalian, T.E.; Turk, G.L.; Fink, P.J. Thymic output in aged mice. Proc. Natl. Acad. Sci. USA 2006, 103, 8447–8452. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Miller, C.M.; Shadrach, J.L.; Wagers, A.J.; Serwold, T. Young, proliferative thymic epithelial cells engraft and function in aging thymuses. J. Immunol. 2015, 194, 4784–4795. [Google Scholar] [CrossRef] [PubMed]
- Olsen, N.J.; Olson, G.; Viselli, S.M.; Gu, X.; Kovacs, W.J. Androgen receptors in thymic epithelium modulate thymus size and thymocyte development. Endocrinology 2001, 142, 1278–1283. [Google Scholar] [CrossRef]
- Rijhsinghani, A.G.; Thompson, K.; Bhatia, S.K.; Waldschmidt, T.J. Estrogen blocks early T cell development in the thymus. Am. J. Reprod. Immunol. 1996, 36, 269–277. [Google Scholar] [CrossRef]
- Kumar, N.; Shan, L.X.; Hardy, M.P.; Bardin, C.W.; Sundaram, K. Mechanism of androgen-induced thymolysis in rats. Endocrinology 1995, 136, 4887–4893. [Google Scholar] [CrossRef]
- Tibbetts, T.A.; DeMayo, F.; Rich, S.; Conneely, O.M.; O’Malley, B.W. Progesterone receptors in the thymus are required for thymic involution during pregnancy and for normal fertility. Proc. Natl. Acad. Sci. USA 1999, 96, 12021–12026. [Google Scholar] [CrossRef]
- Henderson, J. On the relationship of the thymus to the sexual organs: I. The influence of castration on the thymus. J. Physiol. 1904, 31, 222–229. [Google Scholar] [CrossRef]
- Montecino-Rodriquez, E.; Min, H.; Dorshkind, K. Reevaluating current models of thymic involution. Semin. Immunol. 2005, 17, 356–361. [Google Scholar] [CrossRef]
- Min, H.; Montecino-Rodriguez, E.; Dorshkind, K. Reduction in the developmental potential of intrathymic T cell progenitors with age. J. Immunol. 2004, 173, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Sempowski, G.D.; Hale, L.P.; Sundy, J.S.; Massey, J.M.; Koup, R.A.; Douek, D.C.; Patel, D.D.; Haynes, B.F. Leukemia inhibitory factor, oncostatin M, IL-6, and stem cell factor mRNA expression in human thymus increases with age and is associated with thymic atrophy. J. Immunol. 2000, 164, 2180–2187. [Google Scholar] [CrossRef] [PubMed]
- Esashi, E.; Ito, H.; Minehata, K.; Saito, S.; Morikawa, Y.; Miyajima, A. Oncostatin M deficiency leads to thymic hypoplasia, accumulation of apoptotic thymocytes and glomerulonephritis. Eur. J. Immunol. 2009, 39, 1664–1670. [Google Scholar] [CrossRef]
- Jacobson, J.D. Gonadotropin-releasing hormone: Potential role in autoimmunity. Int. Immunopharmacol. 2001, 1, 1077–1083. [Google Scholar] [CrossRef]
- Jacobson, J.D.; Crofford, L.J.; Sun, L.; Wilder, R.L. Cyclical expression of GnRH and GnRH receptor mRNA in lymphoid organs. Neuroendocrinology 1998, 67, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Leposavić, G.; Karapetrović, B.; Obradović, S.; Vidiíc Dandović, B.; Kosec, D. Differential effects of gonadectomy on the thymocyte phenotypic profile in male and female rats. Pharmacol. Biochem. Behav. 1996, 54, 269–276. [Google Scholar] [CrossRef]
- Utsuyama, M.; Hirokawa, K. Hypertrophy of the thymus and restoration of immune functions in mice and rats by gonadectomy. Mech. Ageing. Dev. 1989, 47, 175–185. [Google Scholar] [CrossRef]
- Clark, R. The somatogenic hormones and insulin-like growth factor-1: Stimulators of lymphopoiesis and immune function. Endocr. Rev. 1997, 18, 157–179. [Google Scholar] [CrossRef] [PubMed]
- Burgess, W.; Liu, Q.; Zhou, J.; Tang, Q.; Ozawa, A.; VanHoy, R.; Arkins, S.; Dantzer, R.; Kelley, K.W. The immune-endocrine loop during aging: Role of growth hormone and insulin-like growth factor-I. Neuroimmunomodulation 1999, 6, 56–68. [Google Scholar] [CrossRef]
- Hardeland, R.; Cardinali, D.P.; Brown, G.M.; Pandi-Perumal, S.R. Melatonin and brain inflammaging. Prog. Neurobiol. 2015, 127–128, 46–63. [Google Scholar] [CrossRef]
- Franceschi, C.; Garagnani, P.; Vitale, G.; Capri, M.; Salvioli, S. Inflammaging and ‘garb-aging’. Trends Endocrinol. Metab. 2017, 28, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Savino, W.; Santa-Rosa, G.L. Histophysiology of thymic epithelial reticular cells. Arch. Histol. Jpn. 1982, 45, 139–144. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Maestroni, G.J. The immunoneuroendocrine role of melatonin. J. Pineal. Res. 1993, 14, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Csaba, G. The pineal regulation of the immune system: 40 years since the discovery. Acta Microbiol. Immunol. Hung. 2013, 60, 77–91. [Google Scholar] [CrossRef]
- Tan, D.-X.; Manchester, L.C.; Hardeland, R.; Lopez-Burillo, S.; Mayo, J.C.; Sainz, R.M.; Reiter, R.J. Melatonin—A hormone, a tissue factor, an autocoid, a paracoid, and an antioxidant vitamin. J. Pineal Res. 2003, 34, 75–78. [Google Scholar] [CrossRef]
- Naranjo, M.C.; Guerrero, J.M.; Rubio, A.; Lardonea, P.J.; Carrillo-Vicoa, A.; Carrascosa-Salmorala, M.P.; JimØnez-Jorgea, S.; Arellanob, M.V.; Leal-Novalb, S.R.; Lealc, M.; et al. Melatonin biosynthesis in the thymus of humans and rats. Cell. Mol. Life Sci. 2007, 64, 781–790. [Google Scholar] [CrossRef]
- Acuña-Castroviejo, D.; Escames, G.; Venegas, C.; Díaz-Casado, M.E.; Lima-Cabello, E.; López, L.C.; Rosales-Corral, S.; Tan, D.-X.; Reiter, R.J. Extrapineal melatonin: Sources, regulation, and potential functions. Cell. Mol. Life Sci. 2014, 71, 2997–3025. [Google Scholar] [CrossRef]
- Bubenik, G.A. Gastrointestinal melatonin: Localization, function, and clinical relevance. Dig. Dis. Sci. 2002, 47, 2336–2348. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.-X.; Rosales-Corral, S.; Galano, A.; Zhou, X.J.; Xu, B. Mitochondria: Central organelles for melatonin’s antioxidant and anti-aging actions. Molecules 2018, 23, 509. [Google Scholar] [CrossRef]
- Hardeland, R.; Pandi-Perumal, S.R. Melatonin, a potent agent in antioxidative defense: Actions as a natural food constituent, gastrointestinal factor, drug and prodrug. Nutr. Metab. 2005, 2, 22. [Google Scholar] [CrossRef]
- Dardenne, M.; Pleau, J.M. Interactions between zinc and thymulin. Met. Based Drugs 1994, 1, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Goya, R.G.; Bolognani, F. Homeostasis, thymic hormones and aging. Gerontology 1999, 45, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Bach, J.-F.; Dardenne, M. Thymulin, a zinc-dependent hormone. Med. Oncol. Tumor Pharm. 1989, 6, 25–29. [Google Scholar]
- Cohen, S.; Berrih, S.; Dardenne, M.; Bach, J.F. Feedback regulation of the secretion of a thymic hormone (thymulin) by human thymic epithelial cells in culture. Thymus 1986, 8, 109–119. [Google Scholar] [PubMed]
- Reggiani, P.C.; Schwerdt, J.I.; Console, G.M.; Roggero, E.A.; Dardenne, M.; Goya, R.G. Physiology and therapeutic potential of the thymic peptide thymulin. Curr. Pharm. Des. 2014, 20, 4690–4696. [Google Scholar] [CrossRef]
- Bodey, B.; Bodey, B., Jr.; Siegel, S.E.; Kaiser, H.E. Involution of the mammalian thymus, one of the leading regulators of aging. In Vivo 1997, 11, 421–440. [Google Scholar]
- Savino, W.; Dardenne, M. Immune-neuroendocrine interactions. Immunol. Today 1995, 16, 318–322. [Google Scholar] [CrossRef]
- Goya, R.G.; Reggiani, P.C.; Vesenbeckh, S.M.; Pléau, J.M.; Sosa, Y.E.; Cónsole, G.M.; Schade, R.; Henklein, P.; Dardenne, M. Thymulin gene therapy prevents the reduction in circulating gonadotropins induced by thymulin deficiency in mice. Am. J. Physiol. Endocrinol. Metab. 2007, 293, 182–187. [Google Scholar] [CrossRef]
- Reggiani, P.C.; Barbeito, C.G.; Zuccolilli, G.O.; Cónsole, G.M.; Flamini, A.M.; Dardenne, M.; Goya, R.G. Neonatal thymulin gene therapy prevents ovarian dysgenesis and attenuates reproductive derangements in nude female mice. Endocrinology 2012, 153, 3922–3928. [Google Scholar] [CrossRef]
- Buckingham, J.C.; Safieh, B.; Singh, S.; Arduino, L.A.; Cover, P.O.; Kendall, M.D. Interactions between the hypothalamo-pituitary adrenal axis and the thymus in the rat: A role for corticotrophin in the control of thymulin release. J. Neuroendocrinol. 1992, 4, 295–301. [Google Scholar] [CrossRef]
- Dardenne, M.; Saade, N.; Safieh-Garabedian, B. Role of thymulin or its analogue as a new analgesic molecule. Ann. N. Y. Acad. Sci. 2006, 1088, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Savino, W.; Dardenne, M. Neuroendocrine control of thymus physiology. Endocr. Rev. 2000, 21, 412–443. [Google Scholar]
- Prasad, A.S. Clinical, immunological, anti-inflammatory and antioxidant roles of zinc. Exp. Gerontol. 2008, 43, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Lunin, S.M.; Khrenov, M.O.; Novoselova, T.V.; Parfenyuk, S.B.; Glushkova, O.V.; Fesenko, E.E.; Novoselova, E.G. Modulation of inflammatory response in mice with severe autoimmune disease by thymic peptide thymulin and an inhibitor of NF-kappaB signalling. Int. Immunopharmacol. 2015, 25, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Saitoh, N.; Awaya, A.; Sakudo, A.; SungWook, S.; Saeki, K.; Matsumoto, Y.; Onodera, T. Serum thymic factor prevents LPS-induced pancreatic cell damage in mice via up-regulation of Bcl-2 expression in pancreas. Microbiol. Immunol. 2004, 48, 629–638. [Google Scholar] [CrossRef]
- da Silva, A.L.; Martini, S.V.; Abreu, S.C.; Samary Cdos, S.; Diaz, B.L.; Fernezlian, S.; de Sá, V.K.; Capelozzi, V.L.; Boylan, N.J.; Goya, R.G.; et al. DNA nanoparticle-mediated thymulin gene therapy prevents airway remodeling in experimental allergic asthma. J. Control. Release 2014, 180, 125–133. [Google Scholar] [CrossRef]
- Novoselova, E.G.; Lunin, S.M.; Khrenov, M.O.; Novoselova, T.V.; Fesenko, E.E. Involvement of NF-kappaB transcription factor in the antiinflammatory activity of thymic peptides. Dokl. Biol. Sci. 2009, 428, 484–486. [Google Scholar] [CrossRef]
- Haddad, J.J. Thymulin and zinc (Zn2+)-mediated inhibition of endotoxin-induced production of proinflammatory cytokines and NF-kappaB nuclear translocation and activation in the alveolar epithelium: Unraveling the molecular immunomodulatory, anti-inflammatory effect of thymulin/Zn2+ in vitro. Mol. Immunol. 2009, 47, 205–214. [Google Scholar]
- Novoselova, E.G.; Khrenov, M.O.; Glushkova, O.V.; Lunin, S.M.; Parfenyuk, S.B.; Novoselova, T.V.; Fesenko, E.E. Anti-inflammatory effects of IKK inhibitor XII, thymulin, and fat-soluble antioxidants in LPS-treated mice. Mediat. Inflamm. 2014, 2014, 724838. [Google Scholar] [CrossRef]
- Goldstein, A.L.; Slater, F.D.; White, A. Preparation, assay, and partial purification of a thymic lymphocytopoietic factor (thymosin). Proc. Natl. Acad. Sci. USA 1966, 56, 1010–1017. [Google Scholar] [CrossRef]
- Goldstein, A.L. History of the discovery of the thymosins. Ann. N. Y. Acad. Sci. 2007, 1112, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Severa, M.; Zhang, J.; Giacomini, E.; Rizzo, F.; Etnaa, M.P.; Cruciania, M.; Garacic, E.; Choppb, M.; Coccia, E.M. Thymosins in multiple sclerosis and its experimental models: Moving from basic to clinical application. Mult. Scler. Relat. Disord. 2019, 27, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Hannappel, E.; Iavarone, F.; Castagnola, M. Thymosin fraction 5 re-evaluated after 35 years by high-resolution mass spectrometry. Expert Opin. Biol. Ther. 2018, 18, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Zhou, B.R.; Bai, Y. Binding affinity and function of the extremely disordered protein complex containing human linker histone H1.0 and its chaperone ProTα. Biochemistry 2018, 57, 6645–6648. [Google Scholar] [CrossRef]
- Moon, H.S.; Even-Ram, S.; Kleinman, H.K.; Cha, H.J. Zyxin is upregulated in the nucleus by thymosin β4 in SiHa cells. Exp. Cell Res. 2006, 312, 3425–3431. [Google Scholar] [CrossRef]
- Zoubek, R.E.; Hannappel, E. Subcellular distribution of thymosin β4. Ann. N. Y. Acad. Sci. 2007, 1112, 442–450. [Google Scholar] [CrossRef]
- Hinkel, R.; Klett, K.; Bähr, A.; Kupatt, C. Thymosin β4-mediated protective effects in the heart. Expert Opin. Biol. Ther. 2018, 18, 121–129. [Google Scholar] [CrossRef]
- Mandaliti, W.; Nepravishta, R.; Pica, F.; Vallebona, P.S.; Garaci, E.; Paci, M. Potential mechanism of thymosin-α1-membrane interactions leading to pleiotropy: Experimental evidence and hypotheses. Expert Opin. Biol. Ther. 2018, 18, 33–42. [Google Scholar] [CrossRef]
- Hannappel, E. β-Thymosins. Ann. N. Y. Acad. Sci. 2007, 1112, 21–37. [Google Scholar] [CrossRef]
- Chen, C.; Li, M.; Yang, H.; Chai, H.; Fisher, W.; Yao, Q. Roles of thymosins in cancers and other organ systems. World J. Surg. 2005, 29, 264–270. [Google Scholar] [CrossRef]
- Papamarcaki, T.; Tsolas, O. Prothymosin alpha binds to histone H1 in vitro. FEBS Lett. 1994, 345, 71–75. [Google Scholar] [CrossRef]
- Moretti, S.; Oikonomou, V.; Garaci, E.; Romani, L. Thymosin α1: Burying secrets in the thymus. Expert Opin. Biol. Ther. 2015, 15, 51–58. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Conteas, C.N.; Mutchnick, M.G.; Palmer, K.C.; Weller, F.E.; Luk, G.D.; Naylor, P.H.; Erdos, M.R.; Goldstein, A.L.; Panneerselvam, C.; Horecker, B.L. Cellular levels of thymosin immunoreactive peptides are linked to proliferative events: Evidence for a nuclear site of action. Proc. Natl. Acad. Sci. USA 1990, 87, 3269–3273. [Google Scholar] [CrossRef]
- Pierluigi, B.; D’Angelo, C.; Fallarino, F.; Moretti, S.; Zelante, T.; Bozza, S.; De Luca, A.; Bistoni, F.; Garaci, E.; Romani, L. Thymosin α1: The regulator of regulators? Ann. N. Y. Acad. Sci. 2010, 1194, 1–5. [Google Scholar] [CrossRef]
- Romani, L.; Moretti, S.; Fallarino, F.; Bozza, S.; Ruggeri, L.; Casagrande, A.; Aversa, F.; Bistoni, F.; Velardi, A.; Garaci, E. Jack of all trades: Thymosin α1 and its pleiotropy. Ann. N. Y. Acad. Sci. 2012, 1269, 1–6. [Google Scholar] [CrossRef]
- Turrini, P.; Tirassa, P.; Vigneti, E.; Aloe, L. A role of the thymus and thymosin-alpha1 in brain NGF levels and NGF receptor expression. J. Neuroimmunol. 1998, 82, 64–72. [Google Scholar] [CrossRef]
- Huang, J.; Jiang, H.; Pan, M.; Jiang, Y.; Xie, L. Immunopotentiator thymosin alpha-1 attenuates inflammatory pain by modulating the Wnt3a/β-catenin pathway in spinal cord. Neuroreport 2020, 31, 69–75. [Google Scholar] [CrossRef]
- Billich, A. Thymosin alpha1. SciClone Pharmaceuticals. Curr. Opin. Investig. Drugs 2002, 3, 698–707. [Google Scholar]
- Tuthill, C.W.; King, R.S. Thymosin alpha 1–A peptide immune modulator with a broad range of clinical applications. Clin. Exp. Pharmacol. 2013, 3, 133. [Google Scholar]
- King, R.; Tuthill, C. Immune Modulation with Thymosin Alpha 1 Treatment. Vitam. Horm. 2016, 102, 151–178. [Google Scholar]
- Costantini, C.; Bellet, M.M.; Pariano, M.; Renga, G.; Stincardini, C.; Goldstein, A.L.; Garaci, E.; Romani, L. A Reappraisal of Thymosin Alpha1 in Cancer Therapy. Front. Oncol. 2019, 9, 873. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Wang, H.; Wang, T.; Zhang, Y.; Zhu, X. The efficacy of thymosin alpha1 as immunomodulatory treatment for sepsis: A systematic review of randomized controlled trials. BMC Infect. Dis. 2016, 16, 488. [Google Scholar] [CrossRef] [PubMed]
- Romani, L.; Bistoni, F.; Perruccio, K.; Montagnoli, C.; Gaziano, R.; Bozza, S.; Bonifazi, P.; Bistoni, G.; Rasi, G.; Velardi, A.; et al. Thymosin alpha1 activates dendritic cell tryptophan catabolism and establishes a regulatory environment for balance of inflammation and tolerance. Blood 2006, 108, 2265–2274. [Google Scholar] [CrossRef]
- Rath, N.C.; Kannan, L.; Liyanage, R.; Lay, J.O., Jr. Thymosin beta in macrophage. J. Endocrinol. Reprod. 2007, 11, 55–61. [Google Scholar]
- Huff, T.; Müller, C.S.; Otto, A.M.; Netzker, R.; Hannappel, E. beta-Thymosins, small acidic peptides with multiple functions. Int. J. Biochem. Cell Biol. 2001, 33, 205–220. [Google Scholar] [CrossRef]
- Lever, M.; Theiss, C.; Morosan-Puopolo, G.; Brand-Saberi, B. Thymosin beta4 overexpression regulates neuron production and spatial distribution in the developing avian optic tectum. Histochem. Cell Biol. 2017, 147, 555–564. [Google Scholar] [CrossRef]
- Badamchian, M.; Damavandy, A.A.; Damavandy, H.; Wadhwa, S.A.; Katz, B.; Goldstein, A.L. Identification and quantification of thymosin beta4 in human saliva and tears. Ann. N. Y. Acad. Sci. 2007, 1112, 458–465. [Google Scholar]
- Goldstein, A.L.; Hannappel, E.; Sosne, G.; Kleinman, H.K. Thymosin beta4: A multi-functional regenerative peptide. Basic properties and clinical applications. Expert Opin. Biol. Ther. 2012, 12, 37–51. [Google Scholar] [CrossRef]
- Dubé, K.N.; Smart, N. Thymosin β4 and the vasculature: Multiple roles in development, repair and protection against disease. Expert Opin. Biol. Ther. 2018, 18, 131–139. [Google Scholar]
- Lunin, S.M.; Novoselova, E.G. Thymus hormones as prospective anti-inflammatory agents. Expert Opin. Ther. Targets 2010, 14, 775–786. [Google Scholar] [CrossRef]
- Lunin, S.; Khrenov, M.; Glushkova, O.; Parfenyuk, S.; Novoselova, T.; Novoselova, E. Precursors of thymic peptides as stress sensors [published online ahead of print]. Expert Opin. Biol. Ther. 2020, 2020, 1–15. [Google Scholar] [CrossRef]
- Goldstein, G. Isolation of bovine thymin: A polypeptide hormone of the thymus. Nature 1974, 247, 11–14. [Google Scholar] [CrossRef] [PubMed]
- Harris, C.A.; Andryuk, P.J.; Cline, S.; Chan, H.K.; Natarajan, A.; Siekierka, J.J.; Goldstein, G. Three distinct human thymopoietins are derived from alternatively spliced mRNAs. Proc. Natl. Acad. Sci. USA 1994, 91, 6283–6287. [Google Scholar] [CrossRef] [PubMed]
- Theodor, L.; Shoham, J.; Berger, R.; Trachtenbrot, L.; Simon, A.J.; Brok-Simon, F.; Nir, U.; Ilan, E.; Zevin-Sonkin, D.; Friedman, E.; et al. Ubiquitous expression of a cloned murine thymopoietin cDNA. Acta Haematol. 1997, 97, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Harris, C.A.; Andryuk, P.J.; Cline, S.W.; Mathew, S.; Siekierka, J.J.; Goldstein, G. Structure and mapping of the human thymopoietin (TMPO) gene and relationship of human TMPO beta to rat lamin-associated polypeptide 2. Genomics 1995, 28, 198–205. [Google Scholar] [CrossRef]
- Furukawa, K. LAP2 binding protein 1 (L2BP1/BAF) is a candidate mediator of LAP2-chromatin interaction. J. Cell Sci. 1999, 112, 2485–2492. [Google Scholar]
- Mirza, A.N.; McKellar, S.A.; Urman, N.M.; Brown, A.S.; Hollmig, T.; Aasi, S.Z.; Oro, A.E. LAP2 proteins chaperone GLI1 movement between the lamina and chromatin to regulate transcription. Cell 2019, 176, 198–212. [Google Scholar] [CrossRef]
- Berger, R.; Theodor, L.; Shoham, J.; Gokkel, E.; Brok-Simoni, F.; Avraham, K.B.; Copeland, N.G.; Jenkins, N.A.; Rechavi, G.; Simon, A.J. The characterization and localization of the mouse thymopoietin/lamina-associated polypeptide 2 gene and its alternatively spliced products. Genome Res. 1996, 6, 361–370. [Google Scholar] [CrossRef]
- Basch, R.S.; Goldstein, G. Antigenic and functional evidence for the in vitro inductive activity of thymopoietin (thymin) on thymocyte precursors. Ann. N. Y. Acad. Sci. 1975, 249, 290–299. [Google Scholar] [CrossRef]
- Goldstein, G.; Scheid, M.P.; Boyse, E.A.; Schlesinger, D.H.; Van Wauwe, J. A synthetic pentapeptide with biological activity characteristic of the thymic hormone thymopoietin. Science 1979, 204, 1309–1310. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, J.; Wang, E. Direct binding of thymopentin to surface class II major histocompatibility complex in living cells. J. Phys. Chem. B 2010, 114, 638–642. [Google Scholar] [CrossRef] [PubMed]
- Malaise, M.G.; Hazee-Hagelstein, M.T.; Reuter, A.M.; Vrinds-Gevaert, Y.; Goldstein, G.; Franchimont, P. Thymopoietin and thymopentin enhance the levels of ACTH, beta-endorphin and beta-lipotropin from rat pituitary cells in vitro. Eur. J. Endocrinol. 1987, 115, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Angioni, S.; Iori, G.; Cellini, M.; Sardelli, S.; Massolo, F.; Petraglia, F.; Genazzan, A.R. Acute beta-interferon or thymopentin administration increases plasma growth hormone and cortisol levels in children. Eur. J. Endocrinol. 1992, 127, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Osoba, D.; Miller, J.F.A.P. Evidenc for a humoral thymus factor responsible for the maturation of immunological faculty. Nature 1963, 199, 653–654. [Google Scholar] [CrossRef]
- Ben-Efraim, S.; Keisari, Y.; Ophir, R.; Pecht, M.; Trainin, N.; Burstein, Y. Immunopotentiating and immunotherapeutic effects of thymic hormones and factors with special emphasis on thymic humoral factor THF-gamma2. Crit. Rev. Immunol. 1999, 19, 261–284. [Google Scholar]
- Goso, C.; Frasca, D.; Doria, G. Effect of synthetic thymic humoral factor (THF-gamma 2) on T cell activities in immunodeficient ageing mice. Clin. Exp. Immunol. 1992, 87, 346–351. [Google Scholar] [CrossRef]
- Ophir, R.; Pecht, M.; Rashid, G.; Halperin, D.; Lourie, S.; Burstein, Y.; Ben-Efraim, S.; Trainin, N. A synthetic thymic hormone, THF-γ2, repairs immunodeficiency of mice cured of plasmacytoma by melphalan. Int. J. Cancer 1990, 45, 1190–1194. [Google Scholar] [CrossRef]
- Handzel, Z.T.; Burstein, Y.; Buchner, V.; Pecht, M.; Trainin, N. Immunomodulation of T cell deficiency in humans by thymic humoral factor: From crude extract to synthetic thymic humoral factor-γ2. J. Biol. Response Mod. 1990, 9, 269–278. [Google Scholar]
- Calenda, V.; Silvy, F.; Chermann, J.C. Effects of thymus humoral factor gamma-2 (THF gamma 2) on lymphohaematopoietic progenitor cells: An in vitro study. Res. Immunol. 1993, 144, 395–406. [Google Scholar] [CrossRef]
- Bodey, B.; Bodey, B.J.; Siegel, S.E.; Kaiser, H.E. Review of thymic hormones in cancer diagnosis and treatment. Int. J. Immunopharmacol. 2000, 22, 261–273. [Google Scholar] [CrossRef]
- Ekström, P.; Meissl, H. Evolution of photosensory pineal organs in new light: The fate of neuroendocrine photoreceptors. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2003, 358, 1679–1700. [Google Scholar] [CrossRef] [PubMed]
- Song, J. Pineal gland dysfunction in Alzheimer’s disease: Relationship with the immune-pineal axis, sleep disturbance, and neurogenesis. Mol. Neurodegener. 2019, 14, 28. [Google Scholar] [CrossRef] [PubMed]
- Golan, J.; Torres, K.; Staśkiewicz, G.J.; Opielak, G.; Maciejewski, R. Morphometric parameters of the human pineal gland in relation to age, body weight and height. Folia Morphol 2002, 61, 111–113. [Google Scholar]
- Ooka-Souda, S.; Kadota, T.; Kabasawa, H. The preoptic nucleus: The probable location of the circadian pacemaker of the hagfish. Eptatretus Burgeri. Neurosci. Lett. 1993, 164, 33–36. [Google Scholar] [CrossRef]
- Boutet, A. The evolution of asymmetric photosensitive structures in metazoans and the Nodal connection. Mech. Dev. 2017, 147, 49–60. [Google Scholar] [CrossRef]
- Ralph, C.L. The pineal gland and geographical distribution of animals. Int. J. Biometeorol. 1975, 19, 289–303. [Google Scholar] [CrossRef]
- Tan, D.X.; Xu, B.; Zhou, X.; Reiter, R.J. Pineal Calcification, Melatonin Production, Aging, Associated Health Consequences and Rejuvenation of the Pineal Gland. Molecules 2018, 23, 301. [Google Scholar] [CrossRef]
- Itoh, M.T.; Hattori, A.; Sumi, Y.; Suzuki, T. Day-night changes in melatonin levels in different organs of the cricket (Gryllus bimaculatus). J. Pineal Res. 1995, 18, 165–169. [Google Scholar] [CrossRef]
- Tan, D.X.; Manchester, L.C.; Fuentes-Broto, L.; Paredes, S.D.; Reiter, R.J. Significance and application of melatonin in the regulation of brown adipose tissue metabolism: Relation to human obesity. Obes. Rev. 2011, 12, 167–188. [Google Scholar] [CrossRef]
- Bumb, J.M.; Schilling, C.; Enning, F.; Haddad, L.; Paul, F.; Lederbogen, F.; Deuschle, M.; Schredl, M.; Nolte, I. Pineal gland volume in primary insomnia and healthy controls: A magnetic resonance imaging study. J. Sleep Res. 2014, 23, 274–280. [Google Scholar] [CrossRef]
- Bryden, M.M.; Griffiths, D.J.; Kennaway, D.J.; Ledingham, J. The pineal gland is very large and active in newborn antarctic seals. Experientia 1986, 42, 564–566. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, M.T.; Oh, C.; Raju, A.; Choudhri, A.F. Physiologic pineal region, choroid plexus, and dural calcifications in the first decade of life. AJNR Am. J. Neuroradiol. 2015, 36, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, S.V. Age-dependent morphology of human pineal gland: Supravital study. Adv. Gerontol. 2007, 20, 60–65. [Google Scholar] [PubMed]
- Beker-Acay, M.; Turamanlar, O.; Horata, E.; Unlu, E.; Fidan, N.; Oruc, S. Assessment of Pineal Gland Volume and Calcification in Healthy Subjects: Is it Related to Aging? J. Belg. Soc. Radiol. 2016, 100, 13. [Google Scholar] [PubMed]
- Reiter, R.J. Comparative physiology: Pineal gland. Annu. Rev. Physiol. 1973, 35, 305–328. [Google Scholar] [CrossRef]
- Møller, M.; Baeres, F.M. The anatomy and innervation of the mammalian pineal gland. Cell Tissue Res. 2002, 309, 139–150. [Google Scholar] [CrossRef]
- Sapède, D.; Cau, E. The pineal gland from development to function. Curr. Top. Dev. Biol. 2013, 106, 171–215. [Google Scholar]
- Kiecker, C. The origins of the circumventricular organs. J. Anat. 2018, 232, 540–553. [Google Scholar] [CrossRef]
- Ibañez Rodriguez, M.P.; Noctor, S.C.; Muñoz, E.M. Cellular basis of pineal gland development: Emerging role of microglia as phenotype regulator. PLoS ONE 2016, 11, e0167063. [Google Scholar] [CrossRef]
- Coon, S.L.; Roseboom, P.H.; Baler, R.; Weller, J.L.; Namboodiri, M.A.A.; Koonin, E.V.; Klein, D.C. Pineal serotonin N-acetyltransferase: Expression cloning and molecular analysis. Science 1995, 270, 1681–1683. [Google Scholar] [CrossRef]
- Ishida, I.; Obinata, M.; Deguchi, T. Molecular cloning and nucleotide sequence of cDNA encoding hydroxyindole O-methyltransferase of bovine pineal glands. J. Biol. Chem. 1987, 262, 2895–2899. [Google Scholar] [PubMed]
- Møller, M.; Ingild, A.; Bock, E. Immunohistochemical demonstration of S-100 protein and GFA protein in interstitial cells of rat pineal gland. Brain Res. 1978, 140, 1–13. [Google Scholar] [CrossRef]
- Middeldorp, J.; Hol, E.M. GFAP in health and disease. Prog. Neurobiol. 2011, 93, 421–443. [Google Scholar] [CrossRef] [PubMed]
- Matsushima, S.; Sakai, Y.; Hira, Y.; Oomori, Y.; Daikoku, S. Immunohistochemical studies on sympathetic and non-sympathetic nerve fibers and neuronal cell bodies in the pineal gland of cotton rats, Sigmodon hispidus. Arch. Histol. Cytol. 1994, 57, 47–58. [Google Scholar]
- Mikkelsen, J.D.; Møller, M. Neuropeptide Y in the mammalian pineal gland. Microsc. Res. Tech. 1999, 46, 239–256. [Google Scholar] [CrossRef]
- Badiu, C.; Badiu, L.; Coculescu, M.; Vilhardt, H.; Møller, M. Presence of oxytocinergic neuronal-like cells in the bovine pineal gland: An immunocytochemical and in situ hybridization study. J. Pineal Res. 2001, 31, 273–280. [Google Scholar] [CrossRef]
- Berson, D.M.; Dunn, F.A.; Takao, M. Phototransduction by retinal ganglion cells that set the circadian clock. Science 2002, 295, 1070–1073. [Google Scholar] [CrossRef]
- Reiter, R.J. The melatonin rhythm: Both a clock and a calendar. Experientia 1993, 49, 654–664. [Google Scholar] [CrossRef]
- Hardeland, R. Melatonin, hormone of darkness and more: Occurrence, control mechanisms, actions and bioactive metabolites. Cell. Mol. Life Sci. 2008, 65, 2001–2018. [Google Scholar] [CrossRef]
- Baconnier, S.; Lang, S.B.; Polomska, M.; Hilczer, B.; Berkovic, G.; Meshulam, G. Calcite microcrystals in the pineal gland of the human brain: First physical and chemical studies. Bioelectromagnetics 2002, 23, 488–495. [Google Scholar] [CrossRef]
- Hardeland, R. Aging, Melatonin, and the Pro- and Anti-Inflammatory Networks. Int. J. Mol. Sci. 2019, 20, 1223. [Google Scholar] [CrossRef]
- Hardeland, R.; Cardinali, D.P.; Srinivasan, V.; Spence, D.W.; Brown, G.M.; Pandi-Perumal, S.R. Melatonin—A pleiotropic, orchestrating regulator molecule. Prog. Neurobiol. 2011, 93, 350–384. [Google Scholar] [CrossRef] [PubMed]
- Angervall, L.; Berger, S.; Röckert, H. A microradiographic and X-ray crystallographic study of calcium in the pineal body and in intracranial tumours. Acta Pathol. Microbiol. Scand. 2009, 44, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Gheban, B.A.; Rosca, I.A.; Crisan, M. The morphological and functional characteristics of the pineal gland. Med. Pharm. Rep. 2019, 92, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Koshy, S.; Vettivel, S.K. Varying Appearances of Calcification in Human Pineal Gland: A Light Microscopic Study. J. Anat. Soc. India 2001, 50, 17–18. [Google Scholar]
- Mahlberg, R.; Walther, S.; Kalus, P.; Bohner, G.; Haedel, S.; Reischies, F.M.; Kühl, K.-P.; Hellweg, R.; Kunz, D. Pineal calcification in Alzheimer’s disease: An in vivo study using computed tomography. Neurobiol. Aging 2008, 29, 203–209. [Google Scholar] [CrossRef]
- Sandyk, R.; Awerbuch, G.I. Pineal calcification and its relationship to the fatigue of multiple sclerosis. Int. J. Neurosci. 1994, 74, 95–103. [Google Scholar] [CrossRef]
- Sandyk, R. The relationship of thought disorder to third ventricle width and calcification of the pineal gland in chronic schizophrenia. Int. J. Neurosci. 1993, 68, 53–59. [Google Scholar] [CrossRef]
- Kim, J.; Kim, H.W.; Chang, S.; Kim, J.W.; Je, J.H.; Rhyu, I.J. Growth patterns for acervuli in human pineal gland. Sci. Rep. 2012, 2, 984–989. [Google Scholar] [CrossRef]
- Allen, D.J.; DiDio, L.J.A.; Gentry, E.R. The aged rat pineal gland as revealed in sem and tem. Age 1982, 5, 119–126. [Google Scholar] [CrossRef]
- Reiter, R.J. The role of the neurohormone melatonin as a buffer against macromolecular oxidative damage. Neurochem. Int. 1995, 27, 453–460. [Google Scholar] [CrossRef]
- Anisimov, V.N. The role of pineal gland in breast cancer development. Crit. Rev. Oncol. Hematol. 2003, 46, 221–234. [Google Scholar] [CrossRef]
- Reiter, R.J.; Vaughan, M.K. Pineal antigonadotrophic substances: Polypeptides and indoles. Life Sci. 1977, 21, 159–171. [Google Scholar] [CrossRef]
- Hardeland, R.; Poeggeler, B. Melatonin beyond its classical functions. Open Physiol. J. 2008, 1, 1–23. [Google Scholar] [CrossRef]
- Hardeland, R. Melatonin metabolism in the central nervous system. Curr. Neuropharmacol. 2010, 8, 168–181. [Google Scholar] [CrossRef]
- Pévet, P. Is 5-methoxytryptamine a pineal hormone? Psychoneuroendocrinology 1983, 8, 61–73. [Google Scholar] [CrossRef]
- Roscetti, G.; Del Carmine, R.; Trabucchi, M.; Massotti, M.; Purdy, R.H.; Barbaccia, M.L. Modulation of neurosteroid synthesis/accumulation by L-ascorbic acid in rat brain tissue: Inhibition by selected serotonin antagonists. J. Neurochem. 1998, 71, 1108–1117. [Google Scholar] [CrossRef]
- Hardeland, R.; Poeggeler, B. Actions of melatonin, its structural and functional analogs in the central nervous system and the significance of metabolism. Cent. Nerv. Syst. Agents Med. Chem. 2007, 7, 289–303. [Google Scholar] [CrossRef]
- Haldar-Misra, C.; Pévet, P. The influence of different 5-methoxyindoles on the process of protein/peptide secretion characterized by the formation of granular vesicles in the mouse pineal gland. An in vitro study. Cell Tissue Res. 1983, 230, 113–126. [Google Scholar] [CrossRef]
- Morton, D.J. Both hydroxy- and methoxyindoles modify basal temperature in the rat. J. Pineal Res. 1987, 4, 1–5. [Google Scholar] [CrossRef]
- Ouzir, M.; Bouhaddou, N.; Khalki, H.; Lakhdar-Ghazal, N. Physiological and pharmacological properties of 5-methoxytryptophol. Expert Rev. Endocrinol. Metab. 2013, 8, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Satué, M.; Ramis, J.M.; del Mar Arriero, M.; Monjo, M. A new role for 5-methoxytryptophol on bone cells function in vitro. J. Cell. Biochem. 2015, 116, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Fernando, I.N.; Francis, P.L.; Smith, M.I. Acyltryptophols reversibly inhibit muscle contractions caused by the actions of acetylcholine and raised potassium ion concentrations. J. Neural Transm. 1983, 56, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Yáñez, J.; Meissl, H. Secretion of the methoxyindoles melatonin, 5-methoxytryptophol, 5-methoxyindoleacetic acid, and 5-methoxytryptamine from trout pineal organs in superfusion culture: Effects of light intensity. Gen. Comp. Endocrinol. 1996, 101, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Barker, S.A.; Harrison, R.E.; Monti, J.A.; Brown, G.B.; Christian, S.T. Identification and quantification of 1,2,3,4-tetrahydro-beta-carboline, 2-methyl-1,2,3,4-tetrahydro-beta-carboline, and 6-methoxy-1,2,3,4-tetrahydro-beta-carboline as in vivo constituents of rat brain and adrenal gland. Biochem. Pharmacol. 1981, 30, 9–17. [Google Scholar] [CrossRef]
- Leino, M.; Kari, I.; Airaksinen, M.M.; Gynther, J. 6-Methoxy-tetrahydro-beta-carboline in the retinae of rabbits and pigs. Exp. Eye Res. 1983, 36, 135–138. [Google Scholar] [CrossRef]
- Smythe, G.A.; Duncan, M.W.; Bradshaw, J.E.; Nicholson, M.V. Effects of 6-methoxy-1,2,3,4-tetrahydro-beta-carboline and yohimbine on hypothalamic monoamine status and pituitary hormone release in the rat. Aust. J. Biol. Sci. 1983, 36, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Pähkla, R.; Kask, A.; Rägo, L. Differential effects of beta-carbolines and antidepressants on rat exploratory activity in the elevated zero-maze. Pharmacol. Biochem. Behav. 2000, 65, 737–742. [Google Scholar] [CrossRef]
- Pless, G.; Frederiksen, T.J.; Garcia, J.J.; Reiter, R.J. Pharmacological aspects of N-acetyl-5-methoxytryptamine (melatonin) and 6-methoxy-1,2,3,4-tetrahydro-β-carboline (pinoline) as antioxidants: Reduction of oxidative damage in brain region homogenates. J. Pineal Res. 1999, 26, 236–246. [Google Scholar] [CrossRef]
- Tam, B.B.; Siu, A.W.; Lam, A.F.; Lee, E.Y. Effects of vitamin E and pinoline on retinal lipid peroxidation. Clin. Exp. Optom. 2004, 87, 171–174. [Google Scholar] [CrossRef]
- Blask, D.E.; Vaughan, M.K.; Reiter, R.E. Pineal peptides and reproduction. In The Pineal Gland; Relkin, R., Ed.; Elsevier: Amsterdam, The Netherlands, 1983; pp. 201–224. [Google Scholar]
- Catrina, S.B.; Catrina, A.I.; Sirzen, F.; Griffths, W.; Bergman, T.; Biberfeld, P.; Coculescu, M.; Mutt, V. A cytotoxic, apoptotic, low-molecular weight factor from pineal gland. Life Sci. 1999, 65, 1047–1057. [Google Scholar] [CrossRef]
- Reiter, R.J. Involvement of pineal indoles and polypeptides with the neuroendocrine axis. Prog. Brain Res. 1973, 39, 281–287. [Google Scholar] [PubMed]
- Vaughan, M.K.; Little, J.C.; Johnson, L.Y.; Vaughan, G.M.; Reiter, R.J. Stimulation of rat prolactin secretion in vivo by arginine vasotocin: Influence of age of solution, nighttime administration, and dose. Neuroendocrinology 1977, 24, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Badiu, C.; Coculescu, M.; Møller, M. Arginine vasotocin mRNA revealed by in situ hybridization in bovine pineal gland cells. Cell Tissue Res. 1999, 295, 225–229. [Google Scholar]
- König, A.; Meyer, A.; Thieme, U. Die akute antidiuretische Aktivität der Epiphysis cerebri von Wistar-Ratten. Endokrinologie 1970, 55, 353–358. [Google Scholar]
- Khavinson, V.; Diomede, F.; Mironova, E.; Linkova, N.; Trofimova, S.; Trubiani, O.; Caputi, S.; Sinjari, B. AEDG Peptide (Epitalon) Stimulates Gene Expression and Protein Synthesis during Neurogenesis: Possible Epigenetic Mechanism. Molecules 2020, 25, 609. [Google Scholar] [CrossRef]
- Benson, B.; Ebels, I. Pineal peptides. J. Neural Transm. Suppl. 1978, 13, 157–173. [Google Scholar]
- Damian, E. The pineal gland as a source of releasing hormones. Endocrinologie 1979, 17, 137–138. [Google Scholar]
- Damian, E.; Ianăş, O.; Bădescu, I. Decrease of testosterone, cholesterol and ascorbic acid in the rat testis after administration of pineal polypeptides. Endocrinologie 1979, 17, 109–111. [Google Scholar]
- Damian, E.; Ianăş, O.; Bădescu, I. Purification and characterization of the antigonadotropic pineal substance. Endocrinologie 1980, 18, 79–84. [Google Scholar]
- Noteborn, H.P.; Burbach, J.P.; Ebels, I. Modified forms of vasopressin and oxytocin in a bovine pineal preparation. FEBS Lett. 1987, 216, 200–206. [Google Scholar] [CrossRef]
- Mikkelsen, J.D.; Møller, M.; Larsen, P.J.; Fahrenkrug, J. The presence of nerve fibers immunoreactive for vasoactive intestinal peptide (VIP), peptide histidine isoleucine (PHI), and preproVIP (111–122) in the mouse pineal gland. J. Pineal Res. 1994, 16, 50–56. [Google Scholar] [CrossRef]
- Khavinson, V.K.; Kopylov, A.T.; Vaskovsky, B.V.; Ryzhak, G.A.; Linkova, N.S. Identification of Peptide AEDG in the Polypeptide Complex of the Pineal Gland. Bull. Exp. Biol. Med. 2017, 164, 41–43. [Google Scholar] [CrossRef] [PubMed]
- Khavinson, V.; Linkova, N.; Diatlova, A.; Trofimova, S. Peptide Regulation of Cell Differentiation. Stem Cell Rev. Rep. 2020, 16, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Majidinia, M.; Reiter, R.J.; Shakouri, S.K.; Yousefi, B. The role of melatonin, a multitasking molecule, in retarding the processes of ageing. Ageing Res. Rev. 2018, 47, 198–213. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, V.N.; Loktionov, A.S.; Khavinson, V.K.; Morozov, V.G. Effect of low molecular-weight factors of thymus and pineal gland on life span and spontaneous tumour development in female of different age. Mech. Ageing Dev. 1989, 49, 245–257. [Google Scholar] [CrossRef]
- Goncharova, N.D.; Vengerin, A.A.; Khavinson, V.K.; Lapin, B.A. Pineal peptides restore the age-related disturbances in hormonal functions of the pineal gland and the pancreas. Exp. Gerontol. 2005, 40, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, V.N.; Khavinson, V.K. Peptide bioregulation of aging: Results and prospects. Biogerontology 2010, 11, 139–149. [Google Scholar] [CrossRef]
- Kim, S.U.; Lee, H.J.; Kim, Y.B. Neural stem cell-based treatment for neurodegenerative diseases. Neuropathology 2013, 33, 491–504. [Google Scholar] [CrossRef]
- Kang, J.M.; Yeon, B.K.; Cho, S.J.; Suh, Y.H. Stem Cell Therapy for Alzheimer’s Disease: A Review of Recent Clinical Trials. J. Alzheimers Dis. 2016, 54, 879–889. [Google Scholar] [CrossRef]
- Caputi, S.; Trubiani, O.; Sinjari, B.; Trofimova, A.; Diomede, F.; Linkova, N.; Diatlova, A.; Khavinson, V. Effect of short peptides on neuronal differentiation of stem cells. Int. J. Immunopathol. Pharmacol. 2019, 33, 1–12. [Google Scholar] [CrossRef]
- Pierpaoli, W.; Yi, C. The involvement of pineal gland and melatonin in immunity and aging. I. Thymus-mediated, immunoreconstituting and antiviral activity of thyrotropin-releasing hormone. J. Neuroimmunol. 1990, 27, 99–109. [Google Scholar] [CrossRef]
- Lesnikov, V.A.; Korneva, E.A.; Dall’ara, A.; Pierpaoli, W. The involvement of pineal gland and melatonin in immunity and aging: II. Thyrotropin-releasing hormone and melatonin forestall involution and promote reconstitution of the thymus in anterior hypothalamic area (AHA)-lesioned mice. Int. J. Neurosci. 1992, 62, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Pierpaoli, W.; Regelson, W. Pineal control of aging: Effect of melatonin and pineal grafting on aging mice. Proc. Natl. Acad. Sci. USA 1994, 91, 789–791. [Google Scholar] [CrossRef] [PubMed]
- Lew, G.M. An immunocytochemical study of thyrotropin releasing hormone in the porcine, ovine and rodent pineal gland. Histochemistry 1989, 91, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Murakami, M.; Greer, M.A. Type-II thyroxine 5′-deiodinase is present in the rat pineal gland. Biochem. Biophys. Res. Commun. 1986, 137, 863–868. [Google Scholar] [CrossRef]
- Vriend, J.; Hinkle, P.M.; Knigge, K.M. Evidence for a thyrotropin-releasing hormone inhibitor in the pineal gland. Endocrinology 1980, 107, 1791–1797. [Google Scholar] [CrossRef] [PubMed]
- Markus, R.P.; Fernandes, P.A.; Kinker, G.S.; da Silveira Cruz-Machado, S.; Marçola, M. Immune-pineal axis-acute inflammatory responses coordinate melatonin synthesis by pinealocytes and phagocytes. Br. J. Pharmacol. 2018, 175, 3239–3250. [Google Scholar] [CrossRef]
- Farhood, B.; Goradel, N.H.; Mortezaee, K.; Khanlarkhani, N.; Salehi, E.; Nashtaei, M.S.; Mirtavoos-mahyari, H.; Motevaseli, E.; Shabeeb, D.; Musa, A.E.; et al. Melatonin as an adjuvant in radiotherapy for radioprotection and radiosensitization. Clin. Transl. Oncol. 2019, 21, 268–279. [Google Scholar] [CrossRef]
- Cipolla-Neto, J.; Amaral, F.G.D. Melatonin as a Hormone: New Physiological and Clinical Insights. Endocr. Rev. 2018, 39, 990–1028. [Google Scholar] [CrossRef]
- Dawson, D.; Armstrong, S.M. Chronobiotics—Drugs that shift rhythms. Pharmacol. Ther. 1996, 69, 15–36. [Google Scholar] [CrossRef]
- Jimenez-Jorge, S.; Jimenez-Caliani, A.J.; Guerrero, J.M.; Naranjo, M.C.; Lardone, P.J.; Carrillo-Vico, A.; Osuna, C.; Molinero, P. Melatonin synthesis and melatonin-membrane receptor (MT1) expression during rat thymus development: Role of the pineal gland. J. Pineal Res. 2005, 39, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Hidalgo, M.; de la Lastra, C.A.; Carrascosa-Salmoral, M.P.; Naranjo, M.C.; Gomez-Corvera, A.; Caballero, B.; Guerrero, J.M. Age-related changes in melatonin synthesis in rat extrapineal tissues. Exp. Gerontol. 2009, 44, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Corvera, A.; Cerrillo, I.; Molinero, P.; Naranjo, M.C.; Lardone, P.J.; Sanchez-Hidalgo, M.; Carrascosa-Salmoral, M.P.; Medrano-Campillo, P.; Guerrero, J.M.; Rubio, A. Evidence of immune system melatonin production by two pineal melatonin deficient mice, C57BL/6 and Swiss strains. J. Pineal Res. 2009, 47, 15–22. [Google Scholar] [CrossRef]
- Sugden, D. Melatonin biosynthesis in the mammalian pineal gland. Experientia 1989, 45, 922–932. [Google Scholar] [CrossRef]
- Reiter, R.J. Pineal melatonin: Cell biology of its synthesis and of its physiological interactions. Endocr. Rev. 1991, 12, 151–180. [Google Scholar] [CrossRef]
- Tricoire, H.; Locatelli, A.; Chemineau, P.; Malpaux, B. Melatonin enters the cerebrospinal fluid through the pineal recess. Endocrinology 2002, 143, 84–90. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.-X.; Kim, S.J.; Cruz, M.H. Delivery of pineal melatonin to the brain and SCN: Role of canaliculi, cerebrospinal fluid, tanycytes and Virchow-Robin perivascular spaces. Brain Struct. Funct. 2014, 219, 1873–1887. [Google Scholar] [CrossRef]
- Tan, D.-X.; Manchester, L.C.; Reiter, R.J. CSF generation by pineal gland results in a robust melatonin circadian rhythm in the third ventricle as an unique light/dark signal. Med. Hypotheses 2016, 86, 3–9. [Google Scholar] [CrossRef]
- Hajak, G.; Huether, G.; Blanke, J.; Blömer, M.; Freyer, C.; Poeggeler, B.; Reimer, A.; Rodenbeck, A.; Schulz-Varszegi, M.; Rüther, E. The influence of intravenous L-tryptophan on plasma melatonin and sleep in men. Pharmacopsychiatry 1991, 24, 17–20. [Google Scholar] [CrossRef]
- Hardeland, R. A short overview of duodenal signaling by melatonin. In Mini-Reviews in Recent Melatonin Research; Hardeland, R., Ed.; Cuvillier: Göttingen, Germany, 2017; pp. 22–25. [Google Scholar]
- Kumar Vishwas, D.; Haldar, C. MT1 receptor expression and AA-NAT activity in lymphatic tissue following melatonin administration in male golden hamster. Int. Immunopharmacol. 2014, 22, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Vico, A.; García-Pergañeda, A.; Naji, L.; Calvo, J.R.; Romero, M.P.; Guerrero, J.M. Expression of membrane and nuclear melatonin receptor mRNA and protein in the mouse immune system. Cell Mol. Life Sci. 2003, 60, 2272–2278. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Hidalgo, M.; Guerrero Montávez, J.M.; Carrascosa-Salmoral, M.d.P.; Naranjo Gutierrez, M.d.C.; Lardone, P.J.; de la Lastra Romero, C.A. Decreased MT1 and MT2 melatonin receptor expression in extrapineal tissues of the rat during physiological aging. J. Pineal Res. 2009, 46, 29–35. [Google Scholar]
- Cruz-Chamorro, I.; Álvarez-Sánchez, N.; Escalante-Andicoechea, C.; Carrillo-Vico, A.; Rubio, A.; Guerrero, J.M.; Molinero, P.; Lardone, P.J. Temporal expression patterns of the melatoninergic system in the human thymus of children. Mol. Metab. 2019, 28, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Zhang, L.; Zhao, Z.; Li, N.; Wang, B.; Yang, L. Expression of melatonin receptors and CD4 in the ovine thymus, lymph node, spleen and liver during early pregnancy. Immunology 2020, 160, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.-X.; Hardeland, R.; Back, K.; Manchester, L.C.; Alatorre-Jimenez, M.A.; Reiter, R.J. On the significance of an alternate pathway of melatonin synthesis via 5-methoxytryptamine: Comparisons across species. J. Pineal Res. 2016, 61, 27–40. [Google Scholar] [CrossRef]
- Pozo, D.; Delgado, M.; Fernandez-Santos, J.M.; Calvo, J.R.; Gomariz, R.P.; Martin-Lacave, I.; Ortiz, G.G.; Guerrero, J.M. Expression of the Mel1a-melatonin receptor mRNA in T and B subsets of lymphocytes from rat thymus and spleen. FASEB J. 1997, 11, 466–473. [Google Scholar] [CrossRef]
- Carrillo-Vico, A.; Guerrero, J.M.; Lardone, P.J.; Reiter, R.J. A review of the multiple actions of melatonin on the immune system. Endocrine 2005, 27, 189–200. [Google Scholar] [CrossRef]
- Yu, H.S.; Pang, S.F.; Tang, P.L. Increase in the level of retinal melatonin and persistence of its diurnal rhythm in rats after pinealectomy. J. Endocrinol. 1981, 91, 477–481. [Google Scholar] [CrossRef]
- Vakkuri, O.; Rintamäki, H.; Leppäluoto, J. Plasma and tissue concentrations of melatonin after midnight light exposure and pinealectomy in the pigeon. J. Endocrinol. 1985, 105, 263–268. [Google Scholar] [CrossRef]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Qi, W.; Zhang, M.; Weintraub, S.T.; Cabrera, J.; Sainz, R.M.; Mayo, J.C. Identification of highly elevated levels of melatonin in bone marrow: Its origin and significance. Biochim. Biophys. Acta 1999, 1472, 206–214. [Google Scholar] [CrossRef]
- Pandi-Perumal, S.R.; Srinivasan, V.; Maestroni, G.J.; Cardinali, D.P.; Poeggeler, B.; Hardeland, R. Melatonin: Nature’s most versatile biological signal? FEBS J. 2006, 273, 2813–2838. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Pilar Terron, M.; Flores, L.J.; Koppisepi, S. Medical implications of melatonin: Receptor-mediated and receptor-independent actions. Adv. Med. Sci. 2007, 52, 11–28. [Google Scholar] [PubMed]
- Hardeland, R. Melatonin: Signaling mechanisms of a pleiotropic agent. BioFactors 2009, 35, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R. Recent findings in melatonin research and their relevance to the CNS. Cent. Nerv. Syst. Agents Med. Chem. 2018, 18, 102–114. [Google Scholar] [CrossRef] [PubMed]
- Suofu, Y.; Li, W.; Jean-Alphonse, F.G.; Jia, J.; Khattar, N.K.; Li, J.; Baranov, S.V.; Leronni, D.; Mihalik, A.C.; He, Y.; et al. Dual role of mitochondria in producing melatonin and driving GPCR signaling to block cytochrome c release. Proc. Natl. Acad. Sci. USA 2017, 114, 7997–8006. [Google Scholar] [CrossRef]
- Chu, Z.M.; Li, H.B.; Sun, S.X.; Jiang, Y.C.; Wang, B.; Dong, Y.F. Melatonin promotes osteoblast differentiation of bone marrow mesenchymal stem cells in aged rats. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 4446–4456. [Google Scholar]
- Mendivil-Perez, M.; Soto-Mercado, V.; Guerra-Librero, A.; Fernandez-Gil, B.I.; Florido, J.; Shen, Y.Q.; Tejada, M.A.; Capilla-Gonzalez, V.; Rusanova, I.; Garcia-Verdugo, J.M.; et al. Melatonin enhances neural stem cell differentiation and engraftment by increasing mitochondrial function. J. Pineal Res. 2017, 63, 1–18. [Google Scholar] [CrossRef]
- Fang, J.; Yan, Y.; Teng, X.; Wen, X.; Li, N.; Peng, S.; Liu, W.; Donadeu, F.X.; Zhao, S.; Hua, J. Melatonin prevents senescence of canine adipose-derived mesenchymal stem cells through activating NRF2 and inhibiting ER stress. Aging 2018, 10, 2954–2972. [Google Scholar] [CrossRef]
- Acuña-Castroviejo, D.; Escames, G.; León, J.; Carazo, A.; Khaldy, H. Mitochondrial regulation by melatonin and its metabolites. Adv. Exp. Med. Biol. 2003, 527, 549–557. [Google Scholar]
- Acuña Castroviejo, D.; López, L.C.; Escames, G.; López, A.; García, J.A.; Reiter, R.J. Melatonin-mitochondria interplay in health and disease. Curr. Top. Med. Chem. 2011, 11, 221–240. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R. Melatonin and the electron transport chain. Cell. Mol. Life Sci. 2017, 74, 3883–3896. [Google Scholar] [CrossRef] [PubMed]
- García, J.A.; Ortiz, F.; Miana, J.; Doerrier, C.; Fernández-Ortiz, M.; Rusanova, I.; Escames, G.; García, J.J.; Acuña-Castroviejo, D. Contribution of inducible and neuronal nitric oxide synthases to mitochondrial damage and melatonin rescue in LPS-treated mice. J. Physiol. Biochem. 2017, 73, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Coto-Montes, A.; Boga, J.A.; Rosales-Corral, S.; Fuentes-Broto, L.; Tan, D.-X.; Reiter, R.J. Role of melatonin in the regulation of autophagy and mitophagy: A review. Mol. Cell. Endocrinol. 2012, 361, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Fernández, A.; Ordóñez, R.; Reiter, R.J.; González-Gallego, J.; Mauriz, J.L. Melatonin and endoplasmic reticulum stress: Relation to autophagy and apoptosis. J. Pineal Res. 2015, 59, 292–307. [Google Scholar] [CrossRef]
- Tan, D.-X.; Manchester, L.C.; Qin, L.; Reiter, R.J. Melatonin: A mitochondrial targeting molecule involving mitochondrial protection and dynamics. Int. J. Mol. Sci. 2016, 17, 2124. [Google Scholar] [CrossRef] [PubMed]
- Favero, G.; Moretti, E.; Bonomini, F.; Reiter, R.J.; Rodella, L.F.; Rezzani, R. Promising Antineoplastic Actions of Melatonin. Front. Pharmacol. 2018, 9, 1086. [Google Scholar] [CrossRef]
- Boga, J.A.; Caballero, B.; Potes, Y.; Perez-Martinez, Z.; Reiter, R.J.; Vega-Naredo, I.; Coto-Montes, A. Therapeutic potential of melatonin related to its role as an autophagy regulator: A review. J. Pineal Res. 2019, 66, e12534. [Google Scholar] [CrossRef]
- Tan, D.-X.; Chen, L.-D.; Poeggeler, B.; Manchester, L.C.; Reiter, R.J. Melatonin: A potent, endogenous hydroxyl radical scavenger. Endocr. J. 1993, 1, 57–60. [Google Scholar]
- Hardeland, R.; Reiter, R.J.; Poeggeler, B.; Tan, D.-X. The significance of the metabolism of the neurohormone melatonin: Antioxidative protection and formation of bioactive substances. Neurosci. Biobehav. Rev. 1993, 17, 347–357. [Google Scholar] [CrossRef]
- Tan, D.-X.; Reiter, R.J.; Manchester, L.C.; Yan, M.T.; El-Sawi, M.; Sainz, R.M.; Mayo, J.C.; Kohen, R.; Allegra, M.; Hardeland, R. Chemical and physical properties and potential mechanisms: Melatonin as a broad spectrum antioxidant and free radical scavenger. Curr. Top. Med. Chem. 2002, 2, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Mayo, J.C.; Tan, D.-X.; Sainz, R.M.; Alatorre-Jimenez, M.; Qin, L. Melatonin as an antioxidant: Under promises but over delivers. J. Pineal Res. 2016, 61, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Rosales-Corral, S.; Tan, D.-X.; Jou, M.J.; Galano, A.; Xu, B. Melatonin as a mitochondria-targeted antioxidant: One of evolution’s best ideas. Cell. Mol. Life Sci. 2017, 74, 3863–3881. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, D. Pleiotropic Effects of Melatonin. Drug Res. 2019, 69, 65–74. [Google Scholar] [CrossRef]
- Hardeland, R. Antioxidative protection by melatonin: Multiplicity of mechanisms from radical detoxification to radical avoidance. Endocrine 2005, 27, 119–130. [Google Scholar] [CrossRef]
- Mailliet, F.; Ferry, G.; Vella, F.; Berger, S.; Cogé, F.; Chomarat, P.; Mallet, C.; Guénin, S.P.; Guillaumet, G.; Viaud-Massuard, M.C.; et al. Characterization of the melatoninergic MT3 binding site on the NRH:quinone oxidoreductase 2 enzyme. Biochem. Pharmacol. 2005, 71, 74–88. [Google Scholar] [CrossRef]
- Benítez-King, G.; Huerto-Delgadillo, L.; Antón-Tay, F. Binding of 3H-melatonin to calmodulin. Life Sci. 1993, 53, 201–207. [Google Scholar] [CrossRef]
- Hardeland, R.; Poeggeler, B.; Pappolla, M.A. Mitochondrial actions of melatonin—An endeavor to identify their adaptive and cytoprotective mechanisms. Abh. Sachs. Akad. Wiss. Math. Nat. Kl. 2009, 65, 14–31. [Google Scholar]
- Becker-André, M.; Wiesenberg, I.; Schaeren-Wiemers, N.; André, E.; Missbach, M.; Saurat, J.H.; Carlberg, C. Pineal gland hormone melatonin binds and activates an orphan of the nuclear receptor superfamily. J. Biol. Chem. 1994, 269, 28531–28534. [Google Scholar] [CrossRef]
- Slominski, A.T.; Zmijewski, M.A.; Jetten, A.M. RORα is not a receptor for melatonin. Bioessays 2016, 38, 1193–1194. [Google Scholar] [CrossRef]
- Hardeland, R. Melatonin and retinoid orphan receptors: Demand for new interpretations after their exclusion as nuclear melatonin receptors. Melatonin Res. 2018, 1, 77–92. [Google Scholar] [CrossRef]
- Klein, D.C.; Ganguly, S.; Coon, S.; Weller, J.L.; Obsil, T.; Hickman, A.; Dyda, F. 14–3-3 Proteins and photoneuroendocrine transduction: Role in controlling the daily rhythm in melatonin. Biochem. Soc. Trans. 2002, 30, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Cervantes, M.; Moralí, G.; Letechipía-Vallejo, G. Melatonin and ischemia-reperfusion injury of the brain. J. Pineal Res. 2008, 45, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Ni, Y.F.; Wang, W.C.; Du, H.Y.; Zhang, H.; Zhang, L.; Zhang, W.D.; Jiang, T. Melatonin attenuates intestinal ischemia‒reperfusion-induced lung injury in rats by upregulating N-myc downstream-regulated gene 2. J. Surg. Res. 2015, 194, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.R.; Liu, H.B.; Chen, Y.D.; Sha, Y.; Ma, Q.; Zhu, P.J.; Mu, Y. Melatonin attenuates myocardial ischemia/reperfusion injury by inhibiting autophagy via an AMPK/mTOR signaling pathway. Cell. Physiol. Biochem. 2018, 47, 2067–2076. [Google Scholar] [CrossRef]
- Liu, L.; Chen, H.; Jin, J.; Tang, Z.; Yin, P.; Zhong, D.; Li, G. Melatonin ameliorates cerebral ischemia/reperfusion injury through SIRT3 activation. Life Sci. 2019, 239, 117036. [Google Scholar] [CrossRef]
- Mozaffari, S.; Abdollahi, M. Melatonin, a promising supplement in inflammatory bowel disease: A comprehensive review of evidences. Curr. Pharm. Des. 2011, 17, 4372–4378. [Google Scholar] [CrossRef]
- Esteban-Zubero, E.; López-Pingarrón, L.; Alatorre-Jiménez, M.A.; Ochoa-Moneo, P.; Buisac-Ramón, C.; Rivas-Jiménez, M.; Castán-Ruiz, S.; Antoñanzas-Lombarte, Á.; Tan, D.-X.; García, J.J.; et al. Melatonin’s role as a co-adjuvant treatment in colonic diseases: A review. Life Sci. 2017, 170, 72–81. [Google Scholar] [CrossRef]
- Reiter, R.J.; Rosales-Corral, S.A.; Tan, D.X.; Acuna-Castroviejo, D.; Qin, L.; Yang, S.F.; Xu, K. Melatonin, a full service anti-cancer agent: Inhibition of initiation, progression and metastasis. Int. J. Mol. Sci. 2017, 18, 843. [Google Scholar] [CrossRef]
- Najafi, M.; Shirazi, A.; Motevaseli, E.; Geraily, G.; Norouzi, F.; Heidari, M.; Rezapoor, S. The melatonin immunomodulatory actions in radiotherapy. Biophys. Rev. 2017, 9, 139–148. [Google Scholar] [CrossRef]
- Majidinia, M.; Sadeghpour, A.; Mehrzadi, S.; Reiter, R.J.; Khatami, N.; Yousefi, B. Melatonin: A pleiotropic molecule that modulates DNA damage response and repair pathways. J. Pineal Res. 2017, 63, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Pérez-González, A.; Castañeda-Arriaga, R.; Álvarez-Idaboy, J.R.; Reiter, R.J.; Galano, A. Melatonin and its metabolites as chemical agents capable of directly repairing oxidized DNA. J. Pineal Res. 2019, 66, e12539. [Google Scholar] [CrossRef] [PubMed]
- Shirazi, A.; Ghobadi, G.; Ghazi-Khansari, M. A radiobiological review on melatonin: A novel radioprotector. J. Radiat Res. 2007, 48, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Mazzoccoli, G.; Vendemiale, G.; La Viola, M.; De Cata, A.; Carughi, S.; Greco, A.; Balzanelli, M.; Tarquini, R. Circadian variations of cortisol, melatonin and lymphocyte subpopulations in geriatric age. Int. J. Immunopathol. Pharmacol. 2010, 23, 289–296. [Google Scholar] [CrossRef]
- Mazzoccoli, G.; Sothern, R.B.; De Cata, A.; Giuliani, F.; Fontana, A.; Copetti, M.; Pellegrini, F.; Tarquini, R. A timetable of 24-hour patterns for human lymphocyte subpopulations. J. Biol. Regul. Homeost. Agents 2011, 25, 387–395. [Google Scholar]
- Hajak, G.; Rodenbeck, A.; Ehrenthal, H.D.; Leonard, S.; Wedekind, D.; Sengos, G.; Zhou, D.; Huether, G. No evidence for a physiological coupling between melatonin and glucocorticoids. Psychopharmacology 1997, 133, 313–322. [Google Scholar] [CrossRef]
- Demisch, L.; Demisch, K.; Nickelsen, T. Influence of dexamethasone on nocturnal melatonin production in healthy adult subjects. J. Pineal Res. 1988, 5, 317–322. [Google Scholar] [CrossRef]
- Maestroni, G.J.M.; Conti, A.; Pierpaoli, W. Role of the pineal gland in immunity. Circadian synthesis and release of melatonin modulates the antibody response and antagonizes the immunosuppressive effect of corticosterone. J. Neuroimmunol. 1986, 13, 19–30. [Google Scholar] [CrossRef]
- Khavinson, V.K.; Anisimov, V.N. 35-Year Experience of Investigation of Senescence Peptide Regulation. Uspekhi Gerontol. 2009, 7, 11–23. [Google Scholar]
- Pirozhkov, S.I.; Safarove, G.Z. Senescence Tendencies of Inhabitants of Russia and Ukraine. Uspekhi Gerontol. 2000, 7, 14–21. [Google Scholar]
- Giroir, B.P.; Brown, T.; Beutler, B. Constitutive synthesis of tumor necrosis factor in the thymus. Proc. Natl. Acad. Sci. USA 1992, 89, 4864–4868. [Google Scholar] [CrossRef] [PubMed]
- Markus, R.P.; Ferreira, Z.S.; Fernandes, P.A.; Cecon, E. The immune-pineal axis: A shuttle between endocrine and paracrine melatonin sources. Neuroimmunomodulation 2007, 14, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Pontes, G.N.; Cardoso, E.C.; Carneiro-Sampaio, M.M.S.; Markus, R.P. Pineal melatonin and the innate immune response: The TNF- increase after caesarean section suppresses nocturnal melatonin production. J. Pineal Res. 2007, 43, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Vico, A.; Lardone, P.J.; Naji, L.; Fernandez-Santos, J.M.; Martin-Lacave, I.; Guerrero, J.M.; Calvo, J.R. Beneficial pleiotropic actions of melatonin in an experimental model of septic shock in mice: Regulation of pro-/antiinflammatory cytokine network, protection against oxidative damage and anti-apoptotic effects. J. Pineal Res. 2005, 39, 400–408. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rezzani, R.; Franco, C.; Hardeland, R.; Rodella, L.F. Thymus-Pineal Gland Axis: Revisiting Its Role in Human Life and Ageing. Int. J. Mol. Sci. 2020, 21, 8806. https://doi.org/10.3390/ijms21228806
Rezzani R, Franco C, Hardeland R, Rodella LF. Thymus-Pineal Gland Axis: Revisiting Its Role in Human Life and Ageing. International Journal of Molecular Sciences. 2020; 21(22):8806. https://doi.org/10.3390/ijms21228806
Chicago/Turabian StyleRezzani, Rita, Caterina Franco, Rüdiger Hardeland, and Luigi Fabrizio Rodella. 2020. "Thymus-Pineal Gland Axis: Revisiting Its Role in Human Life and Ageing" International Journal of Molecular Sciences 21, no. 22: 8806. https://doi.org/10.3390/ijms21228806
APA StyleRezzani, R., Franco, C., Hardeland, R., & Rodella, L. F. (2020). Thymus-Pineal Gland Axis: Revisiting Its Role in Human Life and Ageing. International Journal of Molecular Sciences, 21(22), 8806. https://doi.org/10.3390/ijms21228806






