Mesenchymal Stem Cells in Multiple Sclerosis: Recent Evidence from Pre-Clinical to Clinical Studies
Abstract
1. Introduction
2. Mesenchymal Stem Cells
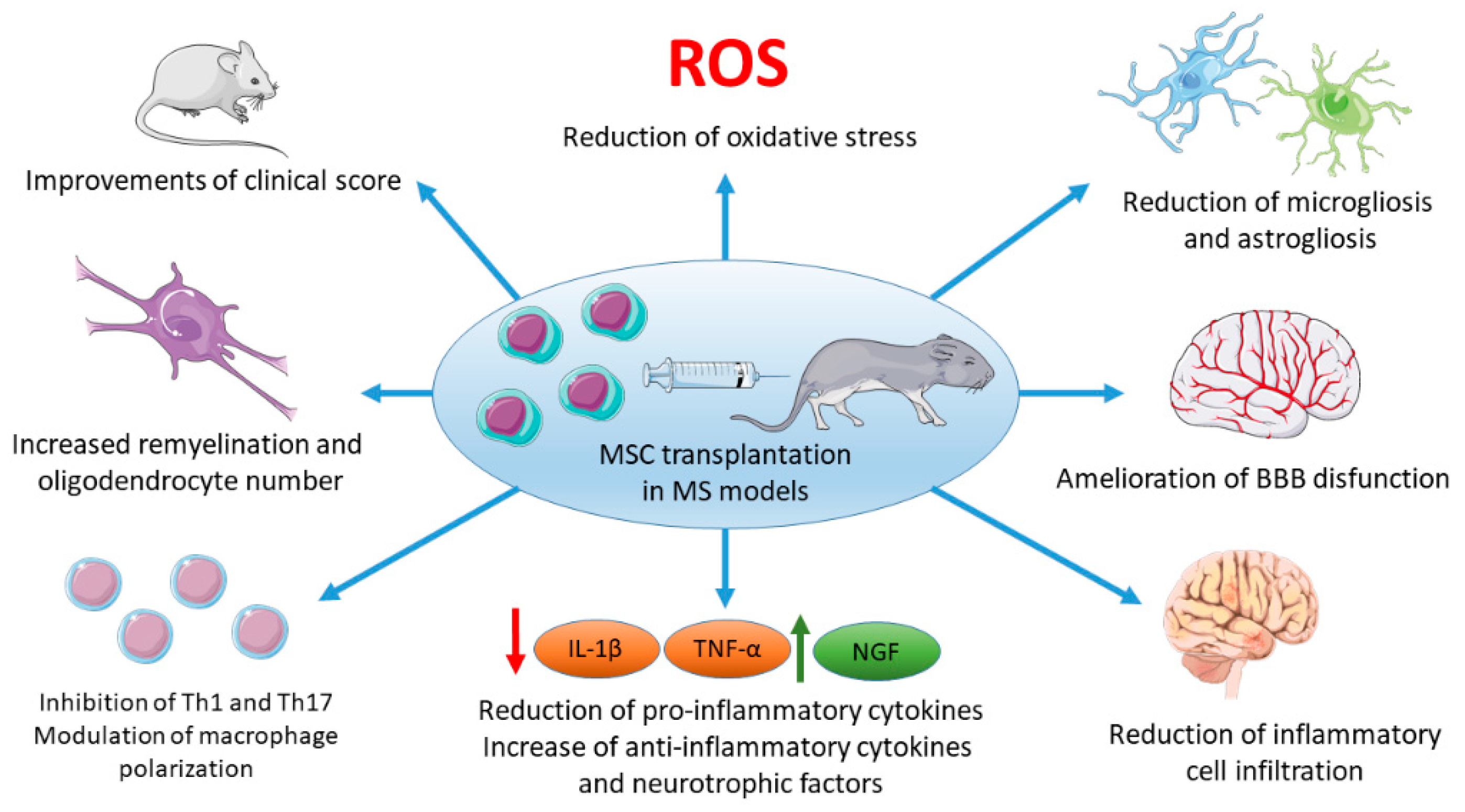
3. MSCs Administration in MS Experimental Models
3.1. Use of Differentiated MSCs in Multiple Sclerosis Experimental Models
3.2. Use of Preconditioned MSCs in Multiple Sclerosis Experimental Models
3.3. Use of Engineered MSCs in Multiple Sclerosis Experimental Models
4. MSCs Secretome in Multiple Sclerosis Experimental Models
5. Combined Therapy Using MSCs in Multiple Sclerosis Experimental Models
6. Clinical Trials Using MSCs
7. Autologous Therapy: Is It Possible in MS?
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| MS | Multiple sclerosis |
| CNS | Central nervous system |
| CIS | Clinically isolated syndrome |
| RRMS | Relapsing remitting MS |
| SPMS | Secondary progressive MS |
| PPMS | Primary progressive MS |
| Th | T helper |
| MSCs | Mesenchymal stem cells |
| CM | conditioned medium |
| EVs | Extracellular vesicles |
| Exo | Exosomes |
| OPC | Oligodendrocyte progenitor cell |
| Olig2 | Oligodendrocyte transcription factor |
| MBP | Myelin basic protein |
| GFAP | Glial fibrillary acidic protein |
| Treg | Regulatory T |
| BM-MSCs | Bone marrow MSCs |
| EAE | Experimental autoimmune encephalomyelitis |
| TNF | Tumor necrosis factor |
| MPO | Myeloperoxidase |
| NO | Nitric oxide |
| IL | Interleukin |
| GSH | Glutathione |
| MP | Methylprednisolone |
| IL17RA | IL17 Receptor A |
| BBB | Blood-brain barrier |
| AQP4 | Aquaporin 4 |
| A2BAR | A2B adenosine receptor |
| HIF-1 | Hypoxia-inducible factor 1 |
| T-bet | T-box transcription factor TBX21 |
| RORγT | RAR-related orphan receptor γ |
| Foxp3 | Forkhead box P3 |
| TGF | Transforming growth factor |
| AD-MSCs | Adipose tissue-derived MSCs |
| DC | Dendritic cell |
| SVF | Stromal vascular fraction |
| PDLSCs | Periodontal Ligament Stem Cells |
| NGF | Nerve growth factor |
| BDNF | Brain derived neurotrophic factor |
| S-MSCs | Skin-derived MSCs |
| IFN | Interferon |
| sTNFR1 | soluble TNF receptor 1 |
| WJ-MSCs | Wharton’s jelly-derived MSCs |
| MDA | Malondialdehyde |
| SOD | Superoxide dismutase |
| IDO1 | indoleamine 2,3-dioxygenase 1 |
| UCMSCs | Umbilical cord MSCs |
| NK | Natural killer |
| AMCs | Amnion mesenchymal cells |
| PMSCs | Placental derived MSCs |
| EMSCs | Embryonic MSCs |
| DMSCs | Decidua derived MSCs |
| mMAPCs | Mouse multipotent adult progenitor cells |
| NT-3 | Neurotrophin-3 |
| NMSCs | Neutralized MSCs |
| MSCs-NPs | MSCs-derived neural progenitor |
| MOG | Myelin oligodendrocyte glycoprotein |
| UCB-MSC | Umbilical cord blood mesenchymal stromal cells |
| TMP | Tetramethylpyrazine |
| SDF-1α | stromal cell-derived factor-1α |
| CXCR4 | C-X-C chemokinereceptor type 4 |
| APC | Adenomatous polyposis coli |
| 17β-ED | 17β-estradiol |
| MMP | Metalloproteinase |
| PSGL1 | P-selectin glycoprotein ligand-1 |
| SLeX | Sialyl-Lewisx |
| SPK1 | Sphingosine kinase 1 |
| rMOG | recombinant MOG |
| SHED | human exfoliated deciduous teeth |
| ED–Siglec-9 | secreted ectodomain of sialic acid–binding Ig-like lectin-9 |
| HGF | Hepatocyte growth factor |
| H-PDLSCs-CM | CM obtained from human PDLSCs under hypoxia |
| EMVs | Exo/microvesicles |
| TMEV | Theiler’s murine encephalomyelitis virus |
| GDNF | glial cell-derived neurotrophic factor |
| CNTF | ciliary neurotrophic factor |
| RA | retinoic acid |
| NR-MSCs | preinduced MSCs with NT-3 and RA |
| EA | Electroacupuncture |
| EDSS | Expanded Disability Status Scale |
| MRI | Magnetic resonance imaging |
| RD | radial diffusivity |
| AD | axial diffusivity |
References
- Filippi, M.; Bar-Or, A.; Piehl, F.; Preziosa, P.; Solari, A.; Vukusic, S.; Rocca, M.A. Multiple sclerosis. Nat. Rev. Dis. Primers 2018, 4, 43. [Google Scholar] [CrossRef] [PubMed]
- Lublin, F.D.; Reingold, S.C.; Cohen, J.A.; Cutter, G.R.; Sorensen, P.S.; Thompson, A.J.; Wolinsky, J.S.; Balcer, L.J.; Banwell, B.; Barkhof, F.; et al. Defining the clinical course of multiple sclerosis: The 2013 revisions. Neurology 2014, 83, 278–286. [Google Scholar] [CrossRef]
- Atlas of MS. PART 1: Mapping Multiple Sclerosis Around the World Key Epidemiology Findings, 3rd ed.; The Multiple Sclerosis International Federation (MSIF): New York, NY, USA, 2020. [Google Scholar]
- Baecher-Allan, C.; Kaskow, B.J.; Weiner, H.L. Multiple Sclerosis: Mechanisms and Immunotherapy. Neuron 2018, 97, 742–768. [Google Scholar] [PubMed]
- Gharibi, T.; Babaloo, Z.; Hosseini, A.; Marofi, F.; Ebrahimi-Kalan, A.; Jahandideh, S.; Baradaran, B. The role of B cells in the immunopathogenesis of multiple sclerosis. Immunology 2020, 160, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Negron, A.; Robinson, R.R.; Stuve, O.; Forsthuber, T.G. The role of B cells in multiple sclerosis: Current and future therapies. Cell. Immunol. 2019, 339, 10–23. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Patterson, K.R.; Bar-Or, A. Reassessing B cell contributions in multiple sclerosis. Nat. Immunol. 2018, 19, 696–707. [Google Scholar] [CrossRef]
- Hauser, S.L.; Cree, B.A.C. Treatment of Multiple Sclerosis: A Review. Am. J. Med. 2020. [Google Scholar] [CrossRef]
- Sormani, M.P.; Muraro, P.A.; Schiavetti, I.; Signori, A.; Laroni, A.; Saccardi, R.; Mancardi, G.L. Autologous hematopoietic stem cell transplantation in multiple sclerosis: A meta-analysis. Neurology 2017, 88, 2115–2122. [Google Scholar] [CrossRef]
- Wei, X.; Yang, X.; Han, Z.P.; Qu, F.F.; Shao, L.; Shi, Y.F. Mesenchymal stem cells: A new trend for cell therapy. Acta Pharmacol. Sin. 2013, 34, 747–754. [Google Scholar] [CrossRef]
- Berebichez-Fridman, R.; Montero-Olvera, P.R. Sources and Clinical Applications of Mesenchymal Stem Cells: State-of-the-art review. Sultan Qaboos Univ. Med. J. 2018, 18, e264–e277. [Google Scholar]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Vizoso, F.J.; Eiro, N.; Cid, S.; Schneider, J.; Perez-Fernandez, R. Mesenchymal Stem Cell Secretome: Toward Cell-Free Therapeutic Strategies in Regenerative Medicine. Int. J. Mol. Sci. 2017, 18, 1852. [Google Scholar] [CrossRef] [PubMed]
- Eleuteri, S.; Fierabracci, A. Insights into the Secretome of Mesenchymal Stem Cells and Its Potential Applications. Int. J. Mol. Sci. 2019, 20, 4597. [Google Scholar] [CrossRef] [PubMed]
- Harrell, C.R.; Fellabaum, C.; Jovicic, N.; Djonov, V.; Arsenijevic, N.; Volarevic, V. Molecular Mechanisms Responsible for Therapeutic Potential of Mesenchymal Stem Cell-Derived Secretome. Cells 2019, 8, 467. [Google Scholar] [CrossRef]
- Ghasemi, N. Transdifferentiation of human adipose-derived mesenchymal stem cells into oligodendrocyte progenitor cells. Iran. J. Neurol. 2018, 17, 24–30. [Google Scholar]
- Jadasz, J.J.; Tepe, L.; Beyer, F.; Samper Agrelo, I.; Akkermann, R.; Spitzhorn, L.S.; Silva, M.E.; Oreffo, R.O.C.; Hartung, H.P.; Prigione, A.; et al. Human mesenchymal factors induce rat hippocampal- and human neural stem cell dependent oligodendrogenesis. Glia 2018, 66, 145–160. [Google Scholar] [CrossRef]
- Ullah, M.; Liu, D.D.; Thakor, A.S. Mesenchymal Stromal Cell Homing: Mechanisms and Strategies for Improvement. iScience 2019, 15, 421–438. [Google Scholar] [CrossRef]
- Song, N.; Scholtemeijer, M.; Shah, K. Mesenchymal Stem Cell Immunomodulation: Mechanisms and Therapeutic Potential. Trends Pharmacol. Sci. 2020, 41, 653–664. [Google Scholar] [CrossRef]
- Jiang, W.; Xu, J. Immune modulation by mesenchymal stem cells. Cell Prolif. 2020, 53, e12712. [Google Scholar] [CrossRef]
- Ransohoff, R.M. A polarizing question: Do M1 and M2 microglia exist? Nat. Neurosci. 2016, 19, 987–991. [Google Scholar] [CrossRef]
- Xue, J.; Schmidt, S.V.; Sander, J.; Draffehn, A.; Krebs, W.; Quester, I.; De Nardo, D.; Gohel, T.D.; Emde, M.; Schmidleithner, L.; et al. Transcriptome-Based Network Analysis Reveals a Spectrum Model of Human Macrophage Activation. Immunity 2014, 40, 274–288. [Google Scholar] [CrossRef] [PubMed]
- Xin, Y.; Gao, J.; Hu, R.; Li, H.; Li, Q.; Han, F.; He, Z.; Lai, L.; Su, M. Changes of immune parameters of T lymphocytes and macrophages in EAE mice after BM-MSCs transplantation. Immunol. Lett. 2020, 225, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Mahfouz, M.M.; Abdelsalam, R.M.; Masoud, M.A.; Mansour, H.A.; Ahmed-Farid, O.A.; Kenawy, S.A. The neuroprotective effect of mesenchymal stem cells on an experimentally induced model for multiple sclerosis in mice. J. Biochem. Mol. Toxicol. 2017, 31. [Google Scholar] [CrossRef] [PubMed]
- Kurte, M.; Luz-Crawford, P.; Vega-Letter, A.M.; Contreras, R.A.; Tejedor, G.; Elizondo-Vega, R.; Martinez-Viola, L.; Fernandez-O’Ryan, C.; Figueroa, F.E.; Jorgensen, C.; et al. IL17/IL17RA as a Novel Signaling Axis Driving Mesenchymal Stem Cell Therapeutic Function in Experimental Autoimmune Encephalomyelitis. Front. Immunol. 2018, 9, 802. [Google Scholar] [CrossRef]
- Liu, Y.; Ma, Y.; Du, B.; Wang, Y.; Yang, G.Y.; Bi, X. Mesenchymal Stem Cells Attenuated Blood-Brain Barrier Disruption via Downregulation of Aquaporin-4 Expression in EAE Mice. Mol. Neurobiol. 2020, 57, 3891–3901. [Google Scholar] [CrossRef]
- Cedola, A.; Bravin, A.; Bukreeva, I.; Fratini, M.; Pacureanu, A.; Mittone, A.; Massimi, L.; Cloetens, P.; Coan, P.; Campi, G.; et al. X-Ray Phase Contrast Tomography Reveals Early Vascular Alterations and Neuronal Loss in a Multiple Sclerosis Model. Sci. Rep. 2017, 7, 5890. [Google Scholar] [CrossRef]
- Gramlich, O.W.; Brown, A.J.; Godwin, C.R.; Chimenti, M.S.; Boland, L.K.; Ankrum, J.A.; Kardon, R.H. Systemic Mesenchymal Stem Cell Treatment Mitigates Structural and Functional Retinal Ganglion Cell Degeneration in a Mouse Model of Multiple Sclerosis. Transl. Vis. Sci. Technol. 2020, 9, 16. [Google Scholar] [CrossRef]
- Kurte, M.; Bravo-Alegria, J.; Torres, A.; Carrasco, V.; Ibanez, C.; Vega-Letter, A.M.; Fernandez-O’Ryan, C.; Irarrazabal, C.E.; Figueroa, F.E.; Fuentealba, R.A.; et al. Intravenous administration of bone marrow-derived mesenchymal stem cells induces a switch from classical to atypical symptoms in experimental autoimmune encephalomyelitis. Stem Cells Int. 2015, 2015, 140170. [Google Scholar] [CrossRef]
- Glenn, J.D.; Smith, M.D.; Kirby, L.A.; Baxi, E.G.; Whartenby, K.A. Disparate Effects of Mesenchymal Stem Cells in Experimental Autoimmune Encephalomyelitis and Cuprizone-Induced Demyelination. PLoS ONE 2015, 10, e0139008. [Google Scholar] [CrossRef]
- Barati, S.; Ragerdi Kashani, I.; Moradi, F.; Tahmasebi, F.; Mehrabi, S.; Barati, M.; Joghataei, M.T. Mesenchymal stem cell mediated effects on microglial phenotype in cuprizone-induced demyelination model. J. Cell. Biochem. 2019, 120, 13952–13964. [Google Scholar] [CrossRef]
- Barati, S.; Kashani, I.R.; Tahmasebi, F.; Mehrabi, S.; Joghataei, M.T. Effect of mesenchymal stem cells on glial cells population in cuprizone induced demyelination model. Neuropeptides 2019, 75, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Marzban, M.; Mousavizadeh, K.; Bakhshayesh, M.; Vousooghi, N.; Vakilzadeh, G.; Torkaman-Boutorabi, A. Effect of Multiple Intraperitoneal Injections of Human Bone Marrow Mesenchymal Stem Cells on Cuprizone Model of Multiple Sclerosis. Iran. Biomed. J. 2018, 22, 312–321. [Google Scholar] [CrossRef] [PubMed][Green Version]
- El-Akabawy, G.; Rashed, L.A. Beneficial effects of bone marrow-derived mesenchymal stem cell transplantation in a non-immune model of demyelination. Ann. Anat. Anat. Anz. 2015, 198, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Rivera, F.J.; de la Fuente, A.G.; Zhao, C.; Silva, M.E.; Gonzalez, G.A.; Wodnar, R.; Feichtner, M.; Lange, S.; Errea, O.; Priglinger, E.; et al. Aging restricts the ability of mesenchymal stem cells to promote the generation of oligodendrocytes during remyelination. Glia 2019, 67, 1510–1525. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P.; Gonzalez-Rey, E.; O’Valle, F.; Martin, F.; Oliver, F.J.; Delgado, M. Allogeneic Adipose-Derived Mesenchymal Stromal Cells Ameliorate Experimental Autoimmune Encephalomyelitis by Regulating Self-Reactive T Cell Responses and Dendritic Cell Function. Stem Cells Int. 2017, 2017, 2389753. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, L.L.; de Lima, A.R.; Martins, D.M.; de Oliveira, E.H.C.; Souza, M.P.C.; de Carvalho Miranda, C.M.F.; Baleeiro Beltrao-Braga, P.C.; Russo, F.B.; Pignatari, G.C.; da Silva Filho, E.; et al. Mesenchymal stem cells in dogs with demyelinating leukoencephalitis as an experimental model of multiple sclerosis. Heliyon 2019, 5, e01857. [Google Scholar] [CrossRef]
- Strong, A.L.; Bowles, A.C.; Wise, R.M.; Morand, J.P.; Dutreil, M.F.; Gimble, J.M.; Bunnell, B.A. Human Adipose Stromal/Stem Cells from Obese Donors Show Reduced Efficacy in Halting Disease Progression in the Experimental Autoimmune Encephalomyelitis Model of Multiple Sclerosis. Stem Cells 2016, 34, 614–626. [Google Scholar] [CrossRef]
- Bowles, A.C.; Strong, A.L.; Wise, R.M.; Thomas, R.C.; Gerstein, B.Y.; Dutreil, M.F.; Hunter, R.S.; Gimble, J.M.; Bunnell, B.A. Adipose Stromal Vascular Fraction-Mediated Improvements at Late-Stage Disease in a Murine Model of Multiple Sclerosis. Stem Cells 2017, 35, 532–544. [Google Scholar] [CrossRef]
- Bowles, A.C.; Wise, R.M.; Gerstein, B.Y.; Thomas, R.C.; Ogelman, R.; Manayan, R.C.; Bunnell, B.A. Adipose stromal vascular fraction attenuates TH1 cell-mediated pathology in a model of multiple sclerosis. J. Neuroinflamm. 2018, 15, 77. [Google Scholar] [CrossRef]
- Trubiani, O.; Giacoppo, S.; Ballerini, P.; Diomede, F.; Piattelli, A.; Bramanti, P.; Mazzon, E. Alternative source of stem cells derived from human periodontal ligament: A new treatment for experimental autoimmune encephalomyelitis. Stem Cell Res. Ther. 2016, 7, 1. [Google Scholar] [CrossRef]
- Ke, F.; Zhang, L.; Liu, Z.; Yan, S.; Xu, Z.; Bai, J.; Zhu, H.; Lou, F.; Cai, W.; Sun, Y.; et al. Soluble Tumor Necrosis Factor Receptor 1 Released by Skin-Derived Mesenchymal Stem Cells Is Critical for Inhibiting Th17 Cell Differentiation. Stem Cells Transl. Med. 2016, 5, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Shiri, E.; Pasbakhsh, P.; Borhani-Haghighi, M.; Alizadeh, Z.; Nekoonam, S.; Mojaverrostami, S.; Pirhajati Mahabadi, V.; Mehdi, A.; Zibara, K.; Kashani, I.R. Mesenchymal Stem Cells Ameliorate Cuprizone-Induced Demyelination by Targeting Oxidative Stress and Mitochondrial Dysfunction. Cell. Mol. Neurobiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Donders, R.; Vanheusden, M.; Bogie, J.F.; Ravanidis, S.; Thewissen, K.; Stinissen, P.; Gyselaers, W.; Hendriks, J.J.; Hellings, N. Human Wharton’s Jelly-Derived Stem Cells Display Immunomodulatory Properties and Transiently Improve Rat Experimental Autoimmune Encephalomyelitis. Cell Transplant. 2015, 24, 2077–2098. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, J.; Han, R.; Meng, M.; Wang, W.; Zhao, Y.; Yang, F.; Yang, L.; Gao, H.; Zhao, Y.; et al. Therapeutic effect of transplanted umbilical cord mesenchymal stem cells in a cynomolgus monkey model of multiple sclerosis. Am. J. Transl. Res. 2019, 11, 2516–2531. [Google Scholar]
- Shu, J.; He, X.; Li, H.; Liu, X.; Qiu, X.; Zhou, T.; Wang, P.; Huang, X. The Beneficial Effect of Human Amnion Mesenchymal Cells in Inhibition of Inflammation and Induction of Neuronal Repair in EAE Mice. J. Immunol. Res. 2018, 2018, 5083797. [Google Scholar] [CrossRef]
- Selim, A.O.; Selim, S.A.; Shalaby, S.M.; Mosaad, H.; Saber, T. Neuroprotective effects of placenta-derived mesenchymal stromal cells in a rat model of experimental autoimmune encephalomyelitis. Cytotherapy 2016, 18, 1100–1113. [Google Scholar] [CrossRef]
- Jiang, H.; Zhang, Y.; Tian, K.; Wang, B.; Han, S. Amelioration of experimental autoimmune encephalomyelitis through transplantation of placental derived mesenchymal stem cells. Sci. Rep. 2017, 7, 41837. [Google Scholar] [CrossRef]
- Yan, L.; Jiang, B.; Niu, Y.; Wang, H.; Li, E.; Yan, Y.; Sun, H.; Duan, Y.; Chang, S.; Chen, G.; et al. Intrathecal delivery of human ESC-derived mesenchymal stem cell spheres promotes recovery of a primate multiple sclerosis model. Cell Death Discov. 2018, 4, 28. [Google Scholar] [CrossRef]
- Bravo, B.; Gallego, M.I.; Flores, A.I.; Bornstein, R.; Puente-Bedia, A.; Hernandez, J.; de la Torre, P.; Garcia-Zaragoza, E.; Perez-Tavarez, R.; Grande, J.; et al. Restrained Th17 response and myeloid cell infiltration into the central nervous system by human decidua-derived mesenchymal stem cells during experimental autoimmune encephalomyelitis. Stem Cell Res. Ther. 2016, 7, 43. [Google Scholar] [CrossRef]
- Abramowski, P.; Krasemann, S.; Ernst, T.; Lange, C.; Ittrich, H.; Schweizer, M.; Zander, A.R.; Martin, R.; Fehse, B. Mesenchymal Stromal/Stem Cells Do Not Ameliorate Experimental Autoimmune Encephalomyelitis and Are Not Detectable in the Central Nervous System of Transplanted Mice. Stem Cells Dev. 2016, 25, 1134–1148. [Google Scholar] [CrossRef]
- Salinas Tejedor, L.; Berner, G.; Jacobsen, K.; Gudi, V.; Jungwirth, N.; Hansmann, F.; Gingele, S.; Prajeeth, C.K.; Baumgartner, W.; Hoffmann, A.; et al. Mesenchymal stem cells do not exert direct beneficial effects on CNS remyelination in the absence of the peripheral immune system. Brain Behav. Immun. 2015, 50, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.P.; Jadhav, S.H.; Chaturvedi, C.P.; Nityanand, S. Therapeutic efficacy of multipotent adult progenitor cells versus mesenchymal stem cells in experimental autoimmune encephalomyelitis. Regen. Med. 2017, 12, 377–396. [Google Scholar] [CrossRef] [PubMed]
- Sargent, A.; Bai, L.; Shano, G.; Karl, M.; Garrison, E.; Ranasinghe, L.; Planchon, S.M.; Cohen, J.; Miller, R.H. CNS disease diminishes the therapeutic functionality of bone marrow mesenchymal stem cells. Exp. Neurol. 2017, 295, 222–232. [Google Scholar] [CrossRef]
- Servier Medical Art by Servier. Available online: http://smart.servier.com/ (accessed on 16 November 2020).
- Creative Commons Attribution 3.0 Unported License. Available online: https://creativecommons.org/licenses/by/3.0/ (accessed on 16 November 2020).
- Han, J.; Zhu, K.; Zhou, K.; Hakim, R.; Sankavaram, S.R.; Blomgren, K.; Lund, H.; Zhang, X.M.; Harris, R.A. Sex-Specific Effects of Microglia-Like Cell Engraftment during Experimental Autoimmune Encephalomyelitis. Int. J. Mol. Sci. 2020, 21, 6824. [Google Scholar] [CrossRef] [PubMed]
- Catuneanu, A.; Paylor, J.W.; Winship, I.; Colbourne, F.; Kerr, B.J. Sex differences in central nervous system plasticity and pain in experimental autoimmune encephalomyelitis. Pain 2019, 160, 1037–1049. [Google Scholar] [CrossRef]
- Papenfuss, T.L.; Rogers, C.J.; Gienapp, I.; Yurrita, M.; McClain, M.; Damico, N.; Valo, J.; Song, F.; Whitacre, C.C. Sex differences in experimental autoimmune encephalomyelitis in multiple murine strains. J. Neuroimmunol. 2004, 150, 59–69. [Google Scholar] [CrossRef]
- Ben-Zwi, M.; Petrou, P.; Halimi, M.; Karussis, D.; Kassis, I. Neuralized mesenchymal stem cells (NMSC) exhibit phenotypical, and biological evidence of neuronal transdifferentiation and suppress EAE more effectively than unmodified MSC. Immunol. Lett. 2019, 212, 6–13. [Google Scholar] [CrossRef]
- Nasri, F.; Mohtasebi, M.S.; Hashemi, E.; Zarrabi, M.; Gholijani, N.; Sarvestani, E.K. Therapeutic Efficacy of Mesenchymal Stem Cells and Mesenchymal Stem Cells-derived Neural Progenitors in Experimental Autoimmune Encephalomyelitis. Int. J. Stem Cells 2018, 11, 68–77. [Google Scholar] [CrossRef]
- Rafieemehr, H.; Kheyrandish, M.; Soleimani, M. Neuroprotective Effects of Transplanted Mesenchymal Stromal Cells-derived Human Umbilical Cord Blood Neural Progenitor Cells in EAE. Iran. J. Allergy AsthmaImmunol. 2015, 14, 596–604. [Google Scholar]
- Nazm Bojnordi, M.; Ghasemi, H.H.; Akbari, E. Remyelination after Lysophosphatidyl Choline-Induced Demyelination Is Stimulated by Bone Marrow Stromal Cell-Derived Oligoprogenitor Cell Transplantation. Cells Tissues Organs 2015, 200, 300–306. [Google Scholar] [CrossRef]
- Zhou, X.Y.; Liu, X.L.; Liu, L.; Han, C.; Xie, Z.H.; Liu, X.T.; Xu, Y.Y.; Li, F.; Bi, J.Z.; Zheng, C.Y. Transplantation of IFN-gamma Primed hUCMSCs Significantly Improved Outcomes of Experimental Autoimmune Encephalomyelitis in a Mouse Model. Neurochem. Res. 2020, 45, 1510–1517. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, X.; Lu, X.; Ma, Y.; Xin, X.; Xu, X.; Wang, S.; Hou, Y. Tetramethylpyrazine enhanced the therapeutic effects of human umbilical cord mesenchymal stem cells in experimental autoimmune encephalomyelitis mice through Nrf2/HO-1 signaling pathway. Stem Cell Res. Ther. 2020, 11, 186. [Google Scholar] [CrossRef] [PubMed]
- Boroujeni, F.B.; Pasbakhsh, P.; Mortezaee, K.; Pirhajati, V.; Alizadeh, R.; Aryanpour, R.; Madadi, S.; Kashani, I.R. Intranasal delivery of SDF-1 alpha-preconditioned bone marrow mesenchymal cells improves remyelination in the cuprizone-induced mouse model of multiple sclerosis. Cell Biol. Int. 2020, 44, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Heidari Barchi Nezhad, R.; Asadi, F.; Mirzaei, M.R.; Abtahi Froushani, S.M. Comparison of the effects of 17beta- estradiol treated and untreated mesenchymal stem cells on ameliorating animal model of multiple sclerosis. Iran. J. Basic Med. Sci. 2018, 21, 936–942. [Google Scholar] [CrossRef]
- Heidari Barchi Nezhad, R.; Asadi, F.; Abtahi Froushani, S.M.; Hassanshahi, G.; Kaeidi, A.; Khanamani Falahati-Pour, S.; Hashemi, Z.; Mirzaei, M.R. The effects of transplanted mesenchymal stem cells treated with 17-b estradiol on experimental autoimmune encephalomyelitis. Mol. Biol. Rep. 2019, 46, 6135–6146. [Google Scholar] [CrossRef]
- Abdallah, A.N.; Shamaa, A.A.; El-Tookhy, O.S. Evaluation of treatment of experimentally induced canine model of multiple sclerosis using laser activated non-expanded adipose derived stem cells. Res. Vet. Sci. 2019, 125, 71–81. [Google Scholar] [CrossRef]
- Mohammadzadeh, A.; Pourfathollah, A.A.; Shahrokhi, S.; Fallah, A.; Tahoori, M.T.; Amari, A.; Forouzandeh, M.; Soleimani, M. Evaluation of AD-MSC (adipose-derived mesenchymal stem cells) as a vehicle for IFN-beta delivery in experimental autoimmune encephalomyelitis. Clin. Immunol. 2016, 169, 98–106. [Google Scholar] [CrossRef]
- Marin-Banasco, C.; Benabdellah, K.; Melero-Jerez, C.; Oliver, B.; Pinto-Medel, M.J.; Hurtado-Guerrero, I.; de Castro, F.; Clemente, D.; Fernandez, O.; Martin, F.; et al. Gene therapy with mesenchymal stem cells expressing IFN-ss ameliorates neuroinflammation in experimental models of multiple sclerosis. Br. J. Pharm. 2017, 174, 238–253. [Google Scholar] [CrossRef]
- Liao, W.; Pham, V.; Liu, L.; Riazifar, M.; Pone, E.J.; Zhang, S.X.; Ma, F.; Lu, M.; Walsh, C.M.; Zhao, W. Mesenchymal stem cells engineered to express selectin ligands and IL-10 exert enhanced therapeutic efficacy in murine experimental autoimmune encephalomyelitis. Biomaterials 2016, 77, 87–97. [Google Scholar] [CrossRef]
- Wang, Y.L.; Xue, P.; Xu, C.Y.; Wang, Z.; Liu, X.S.; Hua, L.L.; Bai, H.Y.; Zeng, Z.L.; Duan, H.F.; Li, J.F. SPK1-transfected UCMSC has better therapeutic activity than UCMSC in the treatment of experimental autoimmune encephalomyelitis model of Multiple sclerosis. Sci. Rep. 2018, 8, 1756. [Google Scholar] [CrossRef]
- Wang, X.; Zhai, W.; Zhu, J.; Zhao, W.; Zou, X.; Qu, S.; Wang, S.; He, Z.; Li, Z.; Wang, L.; et al. Treatment of the bone marrow stromal stem cell supernatant by nasal administration-a new approach to EAE therapy. Stem Cell Res. Ther. 2019, 10, 325. [Google Scholar] [CrossRef]
- Shimojima, C.; Takeuchi, H.; Jin, S.; Parajuli, B.; Hattori, H.; Suzumura, A.; Hibi, H.; Ueda, M.; Yamamoto, A. Conditioned Medium from the Stem Cells of Human Exfoliated Deciduous Teeth Ameliorates Experimental Autoimmune Encephalomyelitis. J. Immunol. 2016, 196, 4164–4171. [Google Scholar] [CrossRef]
- Yousefi, F.; Ebtekar, M.; Soudi, S.; Soleimani, M.; Hashemi, S.M. In vivo immunomodulatory effects of adipose-derived mesenchymal stem cells conditioned medium in experimental autoimmune encephalomyelitis. Immunol. Lett. 2016, 172, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Giacoppo, S.; Thangavelu, S.R.; Diomede, F.; Bramanti, P.; Conti, P.; Trubiani, O.; Mazzon, E. Anti-inflammatory effects of hypoxia-preconditioned human periodontal ligament cell secretome in an experimental model of multiple sclerosis: A key role of IL-37. FASEB J. 2017, 31, 5592–5608. [Google Scholar] [CrossRef] [PubMed]
- Soundara Rajan, T.; Giacoppo, S.; Diomede, F.; Bramanti, P.; Trubiani, O.; Mazzon, E. Human periodontal ligament stem cells secretome from multiple sclerosis patients suppresses NALP3 inflammasome activation in experimental autoimmune encephalomyelitis. Int. J. Immunopathol. Pharmacol. 2017, 30, 238–252. [Google Scholar] [CrossRef] [PubMed]
- Borhani-Haghighi, M.; Mohamadi, Y. Intranasal administration of conditioned medium derived from mesenchymal stem cells-differentiated oligodendrocytes ameliorates experimental autoimmune encephalomyelitis. J. Chem. Neuroanat. 2020, 106, 101792. [Google Scholar] [CrossRef]
- Laso-Garcia, F.; Ramos-Cejudo, J.; Carrillo-Salinas, F.J.; Otero-Ortega, L.; Feliu, A.; Gomez-de Frutos, M.; Mecha, M.; Diez-Tejedor, E.; Guaza, C.; Gutierrez-Fernandez, M. Therapeutic potential of extracellular vesicles derived from human mesenchymal stem cells in a model of progressive multiple sclerosis. PLoS ONE 2018, 13, e0202590. [Google Scholar] [CrossRef]
- Jafarinia, M.; Alsahebfosoul, F.; Salehi, H.; Eskandari, N.; Azimzadeh, M.; Mahmoodi, M.; Asgary, S.; Hakemi, M.G. Therapeutic effects of extracellular vesicles from human adipose-derived mesenchymal stem cells on chronic experimental autoimmune encephalomyelitis. J. Cell. Physiol. 2020. [Google Scholar] [CrossRef]
- Clark, K.; Zhang, S.; Barthe, S.; Kumar, P.; Pivetti, C.; Kreutzberg, N.; Reed, C.; Wang, Y.; Paxton, Z.; Farmer, D.; et al. Placental Mesenchymal Stem Cell-Derived Extracellular Vesicles Promote Myelin Regeneration in an Animal Model of Multiple Sclerosis. Cells 2019, 8, 1497. [Google Scholar] [CrossRef]
- Riazifar, M.; Mohammadi, M.R.; Pone, E.J.; Yeri, A.; Lasser, C.; Segaliny, A.I.; McIntyre, L.L.; Shelke, G.V.; Hutchins, E.; Hamamoto, A.; et al. Stem Cell-Derived Exosomes as Nanotherapeutics for Autoimmune and Neurodegenerative Disorders. Acs Nano 2019, 13, 6670–6688. [Google Scholar] [CrossRef]
- Li, Z.; Liu, F.; He, X.; Yang, X.; Shan, F.; Feng, J. Exosomes derived from mesenchymal stem cells attenuate inflammation and demyelination of the central nervous system in EAE rats by regulating the polarization of microglia. Int. Immunopharmacol. 2019, 67, 268–280. [Google Scholar] [CrossRef] [PubMed]
- Hosseini Shamili, F.; Alibolandi, M.; Rafatpanah, H.; Abnous, K.; Mahmoudi, M.; Kalantari, M.; Taghdisi, S.M.; Ramezani, M. Immunomodulatory properties of MSC-derived exosomes armed with high affinity aptamer toward mylein as a platform for reducing multiple sclerosis clinical score. J. Control. Release 2019, 299, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Ryu, C.H.; Kim, S.M.; Lim, J.Y.; Kim, W.S.; Jeun, S.S. Combined Treatment with Methylprednisolone and Human Bone Marrow-Derived Mesenchymal Stem Cells Ameliorate Experimental Autoimmune Encephalomyelitis. Tissue Eng. Regen. Med. 2018, 15, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Lim, J.Y.; Park, S.A.; Park, S.I.; Kim, W.S.; Ryu, C.H.; Jeun, S.S. Effective combination of methylprednisolone and interferon beta-secreting mesenchymal stem cells in a model of multiple sclerosis. J. Neuroimmunol. 2018, 314, 81–88. [Google Scholar] [CrossRef]
- Wang, D.; Li, S.P.; Fu, J.S.; Bai, L.; Guo, L. Resveratrol augments therapeutic efficiency of mouse bone marrow mesenchymal stem cell-based therapy in experimental autoimmune encephalomyelitis. Int. J. Dev. Neurosci. 2016, 49, 60–66. [Google Scholar] [CrossRef]
- Khezri, S.; Abtahi Froushani, S.M.; Shahmoradi, M. Nicotine Augments the Beneficial Effects of Mesenchymal Stem Cell-based Therapy in Rat Model of Multiple Sclerosis. Immunol. Investig. 2018, 47, 113–124. [Google Scholar] [CrossRef]
- Togha, M.; Jahanshahi, M.; Alizadeh, L.; Jahromi, S.R.; Vakilzadeh, G.; Alipour, B.; Gorji, A.; Ghaemi, A. Rapamycin Augments Immunomodulatory Properties of Bone Marrow-Derived Mesenchymal Stem Cells in Experimental Autoimmune Encephalomyelitis. Mol. Neurobiol. 2017, 54, 2445–2457. [Google Scholar] [CrossRef]
- Yu, J.W.; Li, Y.H.; Song, G.B.; Yu, J.Z.; Liu, C.Y.; Liu, J.C.; Zhang, H.F.; Yang, W.F.; Wang, Q.; Yan, Y.P.; et al. Synergistic and Superimposed Effect of Bone Marrow-Derived Mesenchymal Stem Cells Combined with Fasudil in Experimental Autoimmune Encephalomyelitis. J. Mol. Neurosci. 2016, 60, 486–497. [Google Scholar] [CrossRef]
- Liu, Z.; He, B.; Zhang, R.Y.; Zhang, K.; Ding, Y.; Ruan, J.W.; Ling, E.A.; Wu, J.L.; Zeng, Y.S. Electroacupuncture Promotes the Differentiation of Transplanted Bone Marrow Mesenchymal Stem Cells Preinduced With Neurotrophin-3 and Retinoic Acid Into Oligodendrocyte-Like Cells in Demyelinated Spinal Cord of Rats. Cell Transplant. 2015, 24, 1265–1281. [Google Scholar] [CrossRef]
- Tahmasebi, F.; Pasbakhsh, P.; Barati, S.; Madadi, S.; Kashani, I.R. The effect of microglial ablation and mesenchymal stem cell transplantation on a cuprizone-induced demyelination model. J. Cell Physiol. 2020. [Google Scholar] [CrossRef]
- Clinicaltrials.gov. Available online: https://www.clinicaltrials.gov/ (accessed on 17 October 2020).
- Cohen, J.A.; Imrey, P.B.; Planchon, S.M.; Bermel, R.A.; Fisher, E.; Fox, R.J.; Bar-Or, A.; Sharp, S.L.; Skaramagas, T.T.; Jagodnik, P.; et al. Pilot trial of intravenous autologous culture-expanded mesenchymal stem cell transplantation in multiple sclerosis. Mult. Scler. 2018, 24, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Planchon, S.M.; Lingas, K.T.; Reese Koc, J.; Hooper, B.M.; Maitra, B.; Fox, R.M.; Imrey, P.B.; Drake, K.M.; Aldred, M.A.; Lazarus, H.M.; et al. Feasibility of mesenchymal stem cell culture expansion for a phase I clinical trial in multiple sclerosis. Mult. Scler. J. Exp. Transl. Clin. 2018, 4, 2055217318765288. [Google Scholar] [CrossRef] [PubMed]
- Dahbour, S.; Jamali, F.; Alhattab, D.; Al-Radaideh, A.; Ababneh, O.; Al-Ryalat, N.; Al-Bdour, M.; Hourani, B.; Msallam, M.; Rasheed, M.; et al. Mesenchymal stem cells and conditioned media in the treatment of multiple sclerosis patients: Clinical, ophthalmological and radiological assessments of safety and efficacy. CNS Neurosci. Ther. 2017, 23, 866–874. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, O.; Izquierdo, G.; Fernandez, V.; Leyva, L.; Reyes, V.; Guerrero, M.; Leon, A.; Arnaiz, C.; Navarro, G.; Paramo, M.D.; et al. Adipose-derived mesenchymal stem cells (AdMSC) for the treatment of secondary-progressive multiple sclerosis: A triple blinded, placebo controlled, randomized phase I/II safety and feasibility study. PLoS ONE 2018, 13, e0195891. [Google Scholar] [CrossRef] [PubMed]
- Riordan, N.H.; Morales, I.; Fernandez, G.; Allen, N.; Fearnot, N.E.; Leckrone, M.E.; Markovich, D.J.; Mansfield, D.; Avila, D.; Patel, A.N.; et al. Clinical feasibility of umbilical cord tissue-derived mesenchymal stem cells in the treatment of multiple sclerosis. J. Transl. Med. 2018, 16, 57. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Zhu, L.; Liu, Z.; Wu, J.; Xu, Y.; Zhang, C.J. IV/IT hUC-MSCs Infusion in RRMS and NMO: A 10-Year Follow-Up Study. Front. Neurol. 2020, 11, 967. [Google Scholar] [CrossRef] [PubMed]
- Harris, V.K.; Vyshkina, T.; Sadiq, S.A. Clinical safety of intrathecal administration of mesenchymal stromal cell-derived neural progenitors in multiple sclerosis. Cytotherapy 2016, 18, 1476–1482. [Google Scholar] [CrossRef] [PubMed]
- Harris, V.K.; Stark, J.; Vyshkina, T.; Blackshear, L.; Joo, G.; Stefanova, V.; Sara, G.; Sadiq, S.A. Phase I Trial of Intrathecal Mesenchymal Stem Cell-derived Neural Progenitors in Progressive Multiple Sclerosis. EBioMedicine 2018, 29, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Baldassari, L.E.; Planchon, S.M.; Bermel, R.A.; Nakamura, K.; Fisher, E.; Feng, J.; Sakaie, K.E.; Ontaneda, D.; Cohen, J.A. Serum neurofilament light chain concentration in a phase 1/2 trial of autologous mesenchymal stem cell transplantation. Mult. Scler. J. Exp. Transl. Clin. 2019, 5. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Offerman, E.; Lin, J.; Fisher, E.; Planchon, S.M.; Sakaie, K.; Lowe, M.; Nakamura, K.; Cohen, J.A.; Ontaneda, D. Exploratory MRI measures after intravenous autologous culture-expanded mesenchymal stem cell transplantation in multiple sclerosis. Mult. Scler. J. Exp. Transl. Clin. 2019, 5. [Google Scholar] [CrossRef] [PubMed]
- Sahraian, M.A.; Mohyeddin Bonab, M.; Baghbanian, S.M.; Owji, M.; Naser Moghadasi, A. Therapeutic Use of Intrathecal Mesenchymal Stem Cells in patients with Multiple Sclerosis: A Pilot Study with Booster Injection. Immunol. Investig. 2019, 48, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Meng, M.; Liu, Y.; Wang, W.; Wei, C.; Liu, F.; Du, Z.; Xie, Y.; Tang, W.; Hou, Z.; Li, Q. Umbilical cord mesenchymal stem cell transplantation in the treatment of multiple sclerosis. Am. J. Transl. Res. 2018, 10, 212–223. [Google Scholar] [PubMed]
- Duma, C.; Kopyov, O.; Kopyov, A.; Berman, M.; Lander, E.; Elam, M.; Arata, M.; Weiland, D.; Cannell, R.; Caraway, C.; et al. Human intracerebroventricular (ICV) injection of autologous, non-engineered, adipose-derived stromal vascular fraction (ADSVF) for neurodegenerative disorders: Results of a 3-year phase 1 study of 113 injections in 31 patients. Mol. Biol. Rep. 2019, 46, 5257–5272. [Google Scholar] [CrossRef] [PubMed]
- Siennicka, K.; Zolocinska, A.; Stepien, K.; Lubina-Dabrowska, N.; Maciagowska, M.; Zolocinska, E.; Slysz, A.; Piusinska-Macoch, R.; Mazur, S.; Zdanowicz, U.; et al. Adipose-Derived Cells (Stromal Vascular Fraction) Transplanted for Orthopedical or Neurological Purposes: Are They Safe Enough? Stem Cells Int. 2016, 2016, 5762916. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, X.; Xue, H.; Liu, L.; Zhu, J.; Jin, T. Autologous Mesenchymal Stem Cell Transplantation in Multiple Sclerosis: A Meta-Analysis. Stem Cells Int. 2019, 2019, 8536785. [Google Scholar] [CrossRef] [PubMed]
- Diomede, F.; Rajan, T.S.; D’Aurora, M.; Bramanti, P.; Merciaro, I.; Marchisio, M.; Gatta, V.; Mazzon, E.; Trubiani, O. Stemness Characteristics of Periodontal Ligament Stem Cells from Donors and Multiple Sclerosis Patients: A Comparative Study. Stem Cells Int. 2017, 2017, 1606125. [Google Scholar] [CrossRef]
- Redondo, J.; Sarkar, P.; Kemp, K.; Virgo, P.F.; Pawade, J.; Norton, A.; Emery, D.C.; Guttridge, M.G.; Marks, D.I.; Wilkins, A.; et al. Reduced cellularity of bone marrow in multiple sclerosis with decreased MSC expansion potential and premature ageing in vitro. Mult. Scler. 2018, 24, 919–931. [Google Scholar] [CrossRef]
- de Oliveira, G.L.; de Lima, K.W.; Colombini, A.M.; Pinheiro, D.G.; Panepucci, R.A.; Palma, P.V.; Brum, D.G.; Covas, D.T.; Simoes, B.P.; de Oliveira, M.C.; et al. Bone marrow mesenchymal stromal cells isolated from multiple sclerosis patients have distinct gene expression profile and decreased suppressive function compared with healthy counterparts. Cell Transplant. 2015, 24, 151–165. [Google Scholar] [CrossRef]
- Redondo, J.; Sarkar, P.; Kemp, K.; Heesom, K.J.; Wilkins, A.; Scolding, N.J.; Rice, C.M. Dysregulation of Mesenchymal Stromal Cell Antioxidant Responses in Progressive Multiple Sclerosis. Stem Cells Transl. Med. 2018, 7, 748–758. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, P.; Redondo, J.; Kemp, K.; Ginty, M.; Wilkins, A.; Scolding, N.J.; Rice, C.M. Reduced neuroprotective potential of the mesenchymal stromal cell secretome with ex vivo expansion, age and progressive multiple sclerosis. Cytotherapy 2018, 20, 21–28. [Google Scholar] [CrossRef] [PubMed]
| Age and Strain | MS Model | MSCs | MSCs Administration | Results | Ref. |
|---|---|---|---|---|---|
| 4-week-old female C57BL/6 mice | MOG-EAE | Mouse BM-MSCs | 1 × 107 cells; i.p. at the disease onset (day 14) and day 20 | ↑ clinical score; ↓ inflammatory infiltration and demyelination of spinal cord | [23] |
| Adult male Swiss mice | Spinal cord homogenate EAE | Rat BM-MSCs | 1 × 106; i.p. on the same day of immunization | ↑ clinical score; ↓ inflammation | [24] |
| 10–14-week-old female C57BL/6 mice | MOG-EAE | Mouse BM-MSCs | 1 × 106 cells; i.p. 5 days after EAE induction | IL17/IL17RA signaling mediated the therapeutic function of MSCs in EAE | [25] |
| 6–8-week-old female C57BL/6 mice | MOG-EAE | BM-MSCs | 5 × 105 cells; i.v. at the disease onset (day 11) | ↑ neurobehavioral outcomes; ↓ BBB disruption, inflammatory infiltration and demyelination in spinal cord | [26] |
| 6–8-week-old female C57BL/6J mice | MOG-EAE | Mouse BM-MSCs | 1 × 106; i.v. on the day of clinical onset | ↓ vascular alteration of vessels, myelin, and neuronal damage | [27] |
| 2-month-old female C57BL6/J mice | MOG-EAE | Human BM-MSCs | 1 × 106; i.v. 7 days after EAE induction | ↑ retinal ganglion cell function and motor-sensory impairment | [28] |
| 10–14-week-old females C57BL/6 mice | MOG-EAE | Mouse BM-MSCs | 1 × 106; i.v. administrated at the onset of the disease (day 10), at the peak of the disease (day 18), or at the time of EAE stabilization (day 30) | ↑ clinical score; ↓ Th1, Th17, and Treg | [29] |
| Female C57BL/6 mice | MOG-EAE and cuprizone model | Mouse BM-MSCs | 5 × 106 cells; EAE: i.p. on day 3 and 8. Cuprizone: i.p. weekly | ↓ Th17 activation and function; positive effects only in cuprizone model | [30] |
| 6-week-old male C57BL/6 mice | Cuprizone model | Mouse BM-MSCs | 3 × 105 cells; lateral ventricle after cuprizone treatment | ↑ M2 phenotype; ↓ M1 phenotype | [31] |
| 8-week-old male C57BL/6 mice | Cuprizone model | Mouse BM-MSCs | 3 × 105 cells; lateral ventricle after 12 weeks of diet with cuprizone | ↑ remyelination; ↓ microgliosis and astrocytosis | [32] |
| 7–8-week-old male C57BL/6 mice | Cuprizone model | Human BM-MSCs | 2 × 106 cells for 2 consecutive weeks i.p. after 4 weeks of cuprizone treatment | ↓ demyelination in corpus callosum | [33] |
| 7–8-week-old male C57BL/6 mice | Cuprizone model | Mouse BM-MSCs | 1 × 106; i.v. 2 weeks after cuprizone diet | ↑ remyelination; ↓ demyelination and apoptosis | [34] |
| 12-month-old male Fischer 344 rats | Ethidium bromide | Rat BM-MSCs (2-month-old and 17–20-month-old rat donors) | 1.5–2.0 × 106 MSCs; i.v. at 1, 2, and 3 days post-lesion induction | ↑ differentiation of OPC with young MSCs | [35] |
| 6–8-week-old female C57BL/6 mice | MOG-EAE | Mouse AD-MSCs expanded in hypoxia or human AD-MSCs | 1 × 106 cells; i.p. at the disease onset or at the acute phase | ↓ disease severity, inflammatory cell infiltration, and demyelination | [36] |
| Dogs | Demyelinating leukoencephalitis caused by the distemper virus | Canine AD-MSCs | 1 × 107; 3 doses into the femoral artery at 30-day intervals | ↓ myoclonus | [37] |
| 6–8-week-old female C57Bl/6 mice | MOG-EAE | Human AD-MSCs | 1 × 106; i.p. before disease onset or at the peak of disease severity | Obese derived AD-MSCs failed to improve EAE | [38] |
| 6–8-week-old female C57Bl/6 mice | MOG-EAE | Mouse AD-MSCs | 1 × 106; i.p. 20 days after immunization | ↑ clinical score, behavior, motor function, and histopathologic analyses | [39] |
| 6–8-week-old female C57Bl/6 mice | MOG-EAE | Mouse AD-MSCs | 1 × 106; i.p. 8 days after immunization | ↑ SVF cells and AD-MSCs administration transiently increased disease severity. SVF cells were able to overcome the advancing pathogenesis and showed improvements | [40] |
| 12-week-old male C57BL/6 mice | MOG-EAE | Human PDLSCs | 1 × 106 cells; i.v. at the disease onset (day 14) | ↑ clinical score, lymphocytic infiltration and demyelination; ↓ apoptosis | [41] |
| 6–8-week-old C57BL/6J mice | MOG-EAE | Mouse S-MSCs | 1 × 106; i.p. 3 days before immunization or on day 8 post immunization | ↑ expression of Th2- and Treg-associated genes; ↓ disease onset, disease severity, demyelination, inflammatory cell infiltrate, Th17 cell induction | [42] |
| 8-week-old male C57BL/6 mice | Cuprizone model | Human WJ-MSCs | 3 × 105 cells; lateral ventricle after 12 weeks of diet with cuprizone | ↑ remyelination and oligodendrocyte; ↓ astrogliosis, microgliosis, oxidative stress, and mitochondrial dysfunction | [43] |
| 8-week-old female dark agouti rats | MOG-EAE | Human WJ-MSCs | 2 × 106; i.v. at the onset of clinical symptoms. In order to assess the effect on chronic disease course, rats received a dose at 28 days post immunization | ↑ improved clinical score in EAE rats; ↓ proliferation of activated T | [44] |
| 3–5-year-old cynomolgus monkeys | MOG-EAE | Human UCMSCs | 1 × 106 cells/kg/mL; i.v. at days 74 and 84 | ↓ demyelination and inflammation | [45] |
| 8–10-week-old female C57BL/6 mice | MOG-EAE | Human AMCs | 1 × 106 cells; i.p. at the disease onset (day 14) | ↑ remyelination and neurotrophic factors; ↓ clinical score, inflammation | [46] |
| 6–8-week-old female Wistar rats | MOG-EAE | Human PMSCs | 1 × 106 cells; i.v. at 9 or 16 days post immunization | ↑ oligodendrocyte precursors; ↓ inflammation, axonal injury and degenerating neurons | [47] |
| 9–10-week-old male Lewis rats | Spinal cord homogenate EAE | Rat PMSCs; EMSCs | 1 × 106 cells; ICV 10 days after EAE induction | ↑ neurological functions; ↓ infiltrating inflammatory cells, gliosis, apoptosis, and demyelination | [48] |
| 8–9-year-old female cynomolgus monkeys | MOG-EAE | EMSCs | 2 × 107 cells/monkey; i.t. 3 doses | ↓ clinical symptoms, brain lesion, and demyelination | [49] |
| 10–14-week-old female C57BL/6 mice | MOG-EAE | Human DMSCs | 1 × 106 cells; i.p. at days 1, 3 and 6, or days 6-10 after MOG inoculation. | ↓ demyelination and inflammatory cell infiltration | [50] |
| Female C57BL/6J mice | MOG-EAE | Mouse BM-MSCs | 1 × 106 cells; i.v. at the disease onset (day 11) or at day 3 and 8 | No improvements in EAE | [51] |
| 10-week-old male C57BL/6J mice | Cuprizone model | Human and murine BM-MSCs; canine AD-MSCs | 1 × 106 cells; intraventricularly or intralesion at week 3 or week 4 | No regenerative effects | [52] |
| 12-week-old male C57BL/6 mice | MOG-EAE | Mouse BM-MSCs | 1 × 106; i.v. 8 days after immunization | Superior effects of mMAPCs compared to MSCs | [53] |
| 10–12-week-old female C57BL/6 mice | MOG-EAE | EAE or naïve mouse BM-MSCs; human BM-MSCs obtained from RRMS or control subjects | 0.8 × 106; i.v. 15 days post immunization | BM-MSCs obtained from EAE mice did not improve EAE | [54] |
| Age and Strain | MS Model | MSCs | Differentiation Method | MSCs Administration | Results | Ref. |
|---|---|---|---|---|---|---|
| 6–7-week-old female C57BL/6 mice | MOG-EAE | Human and mouse BM-MSCs | DMEM-F12 supplemented with B-27, bFGF, EGF for 14 days | Intraventricularly 8 days after EAE induction | ↑ disease score; ↓ inflammation, axonal loss, and demyelination | [60] |
| 12-week-old female C57BL/6 mice | MOG-EAE | Mouse BM-MSCs | Neurobasal media supplemented with B27, insulin-transferring-selenite, L-glutamine, penicillin and streptomycin, bFGF and dehydroepiandrosterone | 1 × 106; i.v. on days 22, 29, and 36 after immunization | ↑ IL-10; ↓ proliferation of pathogenic MOG35-55-specific T cells, IFN-γ production | [61] |
| 8–10-week-old female C57BL/6 mice | MOG-EAE | Human UCB-MSCs | Basal medium supplemented with B-27, L-glutamin, retinoic acid, bFGF, epidermal growth factor, nerve growth factor, 3-isobutylmethyl-xanthin, and ascorbic acid | 5 × 105 UCB-MSCs or MDNPC; i.v. on days 14 and 21 after immunization | ↑ clinical score; ↓ leukocyte infiltration | [62] |
| Female Sprague-Dawley rats | LPC | Rat BM-MSCs | DMEM/F12 with N-2 supplement, B27 supplement, bFGF and epidermal growth factor for 8–12 days | 2 × 105; injected into the corpus callosum | ↓ demyelination | [63] |
| Age and Strain | MS Model | MSCs | Pre-Conditioning | MSCs Administration | Results | Ref. |
|---|---|---|---|---|---|---|
| 6–8-week-old female C57BL/6J mice | MOG-EAE | Human UCMSCs | 20 ng/mL IFN-γ for 48 h | 1 × 106; i.v. 14 days after immunization | ↑ clinical symptoms; ↓ serum IL-17A and TNF-α levels | [64] |
| 10-week-old female C57BL/6 mice | MOG-EAE | Human UCMSCs | TMP 100 µM | 1 × 106; i.v. 13 days after immunization | ↑ clinical score; ↓ inflammatory cell infiltration and NLRP3 levels, demyelination, and BBB disruption | [65] |
| 6–8-week-old male C57BL/6 mice | Cuprizone | Mouse BM-MSCs | 100 ng/mL SDF-1α for 24 h | 1 × 106; intranasal | ↑ remyelination | [66] |
| 6-8-week-old male Wistar rats | Spinal cord homogenate EAE | Rat BM-MSCs | 100 nM 17β-estradiol for 24 h | 2 × 106 cells/rat; i.p. | ↑ clinical score and neuropathological changes | [67] |
| 8–12-week-old male Wistar rats | Spinal cord homogenate EAE | Rat BM-MSCs | 100 nM 17β-estradiol for 24 h | 2 × 106 cells/rat; i.p. when all of the rats showed disease symptoms | ↓ lymphocyte infiltration | [68] |
| 2–5-year old and of both sexes mongrel dogs | Ethidium bromide | Dog SVF | low level laser for 20 min | 10 × 106 nucleated cells; injected directly in the Cerebrospinal fluid 14 days after induction | ↑ remyelination | [69] |
| Age and Strain | MS Model | MSCs | Gene Expression | MSCs Administration | Results | Ref. |
|---|---|---|---|---|---|---|
| 6–8-week-old female C57Bl/6 mice | MOG-EAE | Mouse AD-MSCs | Mouse IFN-β gene | 5 × 105/each time; i.v. on day 16 and 18 after immunization | ↑ Tregs and IL-10 production; ↓ inflammatory cell infiltration | [70] |
| 6–8-week-old female SJL/JCrl (RR-EAE) and C57Bl/6 mice (CP-EAE) | Proteolipid protein 139–151 peptide for RR-EAE, MOG for CP-EAE | Mouse AD-MSCs | Mouse IFN-β gene | 1 × 106; i.v. | ↓ disease score, activated microglia and inflammatory cell infiltration and demyelination | [71] |
| C57BL/6 mice | MOG-EAE | Human BM-MSCs | PSGL-1, FUT-7 and IL-10 | 1 × 106; i.v. 14 days after immunization | ↑ clinical score and myelination; ↓ inflammatory infiltration | [72] |
| 6–8-week-old female C57BL/6 J mice | MOG-EAE | Human UCMSCs | SPK1 | 5.5 × 106; i.v. on the day 7, 14, 21, 28 | ↓ neurological deficits, inflammatory cell infiltration, demyelination, and astrogliosis | [73] |
| Age and Strain | MS Model | MSCs Source | Secretome | Administration | Results | Ref. |
|---|---|---|---|---|---|---|
| Female C57BL/6 mice | MOG-EAE | Mouse BM-MSCs | CM | 60 μL/mouse (30 μL on each nostril); i.n. from the day 3 after immunization until the onset of symptoms | ↓ disease onset and disease severity demyelination B cells infiltration and microglia activation | [74] |
| 8-week-old female C57BL/6J mice | MOG-EAE | SHED | CM | 500 µl; i.v. 14 days after immunization corresponding to the peak of EAE | ↑ clinical scores; ↓ demyelination, axonal injury, inflammatory infiltrates, and the expression of proinflammatory cytokines | [75] |
| 6–7-week-old female C57BL/6 mice | MOG-EAE | Mouse AD-MSCs | CM | 1 mL; i.p. on day 10 after immunization and the same amount once a week for four weeks. 1 × 106 AD-MSCs i.p. | ↑ clinical scores; ↓ inflammatory infiltration | [76] |
| 6–7-week-old female C57BL/6 mice | MOG-EAE | Human PDLSCs | CM obtained in hypoxic condition | 1.0 mg/mouse; i.v. after 14 days from EAE induction | ↑ IL-37 and BDNF levels; ↓ inflammatory cell infiltration, demyelination, oxidative stress, and apoptosis | [77] |
| 12-week-old male C57BL/6 mice | MOG-EAE | Human PDLSCs derived from RRMS patients and healthy controls | CM and EMVs | 1600 μG for CM or 24 μG for EMVs; i.v. after 14 days of EAE induction | ↓ inflammasome, NF-κB and inflammatory cell infiltration | [78] |
| 10-week-old male C57BL/6 mice | MOG-EAE | Human WJ-MSCs differentiated toward oligodendrocytes | CM | i.n. 10 days after immunization | ↑ remyelination and neurological scores; ↓ inflammatory cell infiltration and the expression of proinflammatory markers | [79] |
| 4–6-week-old female SJL/J mice | TMEV-induced demyelinating disease | Human AD-MSCs | EVs | 25 μg; i.v. on day 60 post infection | ↑ motor function; ↓ brain atrophy, inflammatory infiltration and plasma cytokine levels | [80] |
| 6–8-week-old female C57Bl/6 mice | MOG-EAE | Human AD-MSCs | EVs | 60 μg EVs or 1 × 106 AD-MSC; i.v. 10 days after immunization | ↑ clinical score; ↓ inflammatory infiltrates and demyelination | [81] |
| 3-month-old male and female C57BL/6J mice | MOG-EAE | Human PMSCs | EVs | 1 × 107 (low dose), 1 × 1010 (high dose) EVs, 1 × 106 PMSCs; i.v. on day 19 (peak of the disease) | ↑ motor function, myelination | [82] |
| 6–8-week-old female C57BL/6J mice | MOG-EAE | Human BM-MSCs | Exo | 150 μg or 1 × 106 native or IFN-γ stimulated MSCs; i.v. at the peak of the disease (day 18) | ↑ Tregs; ↓ demyelination and neuroinflammation | [83] |
| Female Sprague Dawley rats | Spinal cord homogenate EAE | Rat BM-MSCs | Exo | 100 μg or 400 µg; i.v. the day after EAE induction. 1 × 106 BM-MSCs | ↑ M2-related markers and behavioral scores; ↓ demyelination and inflammatory cell infiltration | [84] |
| 10–13-week-old female C57BL/6 mice | MOG-EAE | Mouse BM-MSCs | Exo | 200 μg of Exo or Exo-APT; i.v. on day 1, 3, 6 after immunization in the prophylactic model or on day 12, 15, and 18 in the therapeutic model | ↓ disease severity, inflammation, and demyelination | [85] |
| Age and Strain | MS Model | MSCs | Combination | MSCs Administration | Results | Ref. |
|---|---|---|---|---|---|---|
| 9-week-old female C57BL/6 mice | MOG-EAE | Human BM-MSCs | MP (20 mg/kg) and BM-MSCs | 1 × 106; i.v. (MSCs) or i.p. (MP)14 days after immunization | ↑ remyelination; ↓ inflammatory cell infiltration | [86] |
| 9-week-old female C57BL/6 mice | MOG-EAE | Human BM-MSCs | MP (10 mg/kg) and MSCs-IFNβ | 1 × 106; i.v. (MSCs) or i.p. (MP) 14 days after immunization | ↑ remyelination; ↓ inflammatory infiltration, BBB disruption | [87] |
| 8–12-week-old female C57BL/6 mice | MOG-EAE | Mouse BM-MSCs | Resveratrol (i.p. 30 mg/kg) and BM-MSCs | 1.5×106 cells; i.v. 7 days after immunization | ↓ symptom onset, clinical scores, and inflammatory cell infiltration | [88] |
| 6–8-week-old male Wistar rats | Spinal cord homogenate EAE | Rat BM-MSCs | Nicotine (i.p. 2.5 mg/kg body weight every day) and BM-MSCs | 2 × 106; i.p. 12 days after immunization | ↓ disease disability | [89] |
| 8–10-week-old female C57BL/6 mice | MOG-EAE | Mouse BM-MSCs | Rapamycin (0.3 mg/kg) and BM-MSCs | 2 × 106; i.p. 10 and 17 days after EAE induction | ↑ clinical score; ↓ inflammatory cell infiltration and demyelination | [90] |
| 8–12-week-old female C57BL/6 mice | MOG-EAE | Mouse BM-MSCs | Fasudil (i.p. 400 μg/mice on day 14 p.i. till day 27) and BM-MSCs | 1.5 × 105; intranasally on day 12 post immunization | ↑ clinical score, demyelination, and inflammatory cell infiltration | [91] |
| Female SD rats | Ethidium bromide | Rat BM-MSCs | NT-3 (20 ng/mL) and/or RA (1 μM/L) preinduced MSCs and electroacupuncture | 1 × 105; intralesion | ↑ conduction of cortical motor-evoked potentials; ↓ demyelination | [92] |
| 8-week-old male C57BL/6 mice | Cuprizone | Mouse BM-MSCs | PLX3397 (290 mg/kg) for 21 days and BM-MSCs | 3 × 105 cells; intraventricular at the 13th week | ↑ oligodendrocytes and remyelination; ↓ microglia, astrocytes, and neurobehavioral deficits | [93] |
| NCT Number | MSCs | MSCs Administration | Safety | Severe Adverse Events | Improvements | Ref. |
|---|---|---|---|---|---|---|
| NCT00813969 | Autologous BM-MSCs | 1–2 × 106/Kg body weight; i.v. | Yes | No | - | [95] |
| NCT01895439 | Autologous BM-MSCs and CM | Average number of 110 × 106 MSCs injected per patient; intrathecal. A month after, an average volume of 18 mL of CM was given intrathecally | Yes | No | Yes | [97] |
| NCT01056471 | Autologous AD-MSCs | 1 × 106 cells/kg (low-dose) or 4 × 106 cells/kg (high-dose); i.v. | Yes | No | - | [98] |
| NCT02034188 | UCMSC | 20 × 106/day; i.v. over the course of 7 visit separated by 1–4 days | Yes | No | Yes | [99] |
| NCT01364246 | UCMSCs | Four doses: 4 × 107, 2 × 107, 2 × 107, 2 × 107 i.v. on day 0, 7, 14, and 21. Three doses: 2 × 107, 2 × 107, 2 × 107 intrathecally on day 7, 14, and 21 | Yes | No | - | [100] |
| NCT01933802 | Neural progenitors derived from autologous BM-MSCs | Up to 1 × 107; intrathecal, 3 doses spaced three months apart | Yes | No | Yes | [102] |
| MSCs | MSCs Administration | Severe Adverse Events | Improvements | Ref. |
|---|---|---|---|---|
| Autologous BM-MSCs | A mean of 57 × 106 cells; intrathecally | No | Yes | [105] |
| UCMSC | 1 to 2 × 106 cells/kg; i.v. at 3-month intervals for 7 times | No | Yes | [106] |
| Autologous SVF | Patients received intraventricular SVF injection volumes of 3.5–20 cc (median: 4 cc) containing 4.05 × 105 to 6.2 × 107 cells/cc and contained on average 8% hematopoietic and 7.5% ADSCs | - | Yes | [107] |
| SVF | 4.2 × 106 cells (females) and 12.8 × 106 cells (males); intrathecally | No | - | [108] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gugliandolo, A.; Bramanti, P.; Mazzon, E. Mesenchymal Stem Cells in Multiple Sclerosis: Recent Evidence from Pre-Clinical to Clinical Studies. Int. J. Mol. Sci. 2020, 21, 8662. https://doi.org/10.3390/ijms21228662
Gugliandolo A, Bramanti P, Mazzon E. Mesenchymal Stem Cells in Multiple Sclerosis: Recent Evidence from Pre-Clinical to Clinical Studies. International Journal of Molecular Sciences. 2020; 21(22):8662. https://doi.org/10.3390/ijms21228662
Chicago/Turabian StyleGugliandolo, Agnese, Placido Bramanti, and Emanuela Mazzon. 2020. "Mesenchymal Stem Cells in Multiple Sclerosis: Recent Evidence from Pre-Clinical to Clinical Studies" International Journal of Molecular Sciences 21, no. 22: 8662. https://doi.org/10.3390/ijms21228662
APA StyleGugliandolo, A., Bramanti, P., & Mazzon, E. (2020). Mesenchymal Stem Cells in Multiple Sclerosis: Recent Evidence from Pre-Clinical to Clinical Studies. International Journal of Molecular Sciences, 21(22), 8662. https://doi.org/10.3390/ijms21228662




