Umbilical Cord-Derived CD362+ Mesenchymal Stromal Cells Attenuate Polymicrobial Sepsis Induced by Caecal Ligation and Puncture
Abstract
:1. Introduction
2. Results
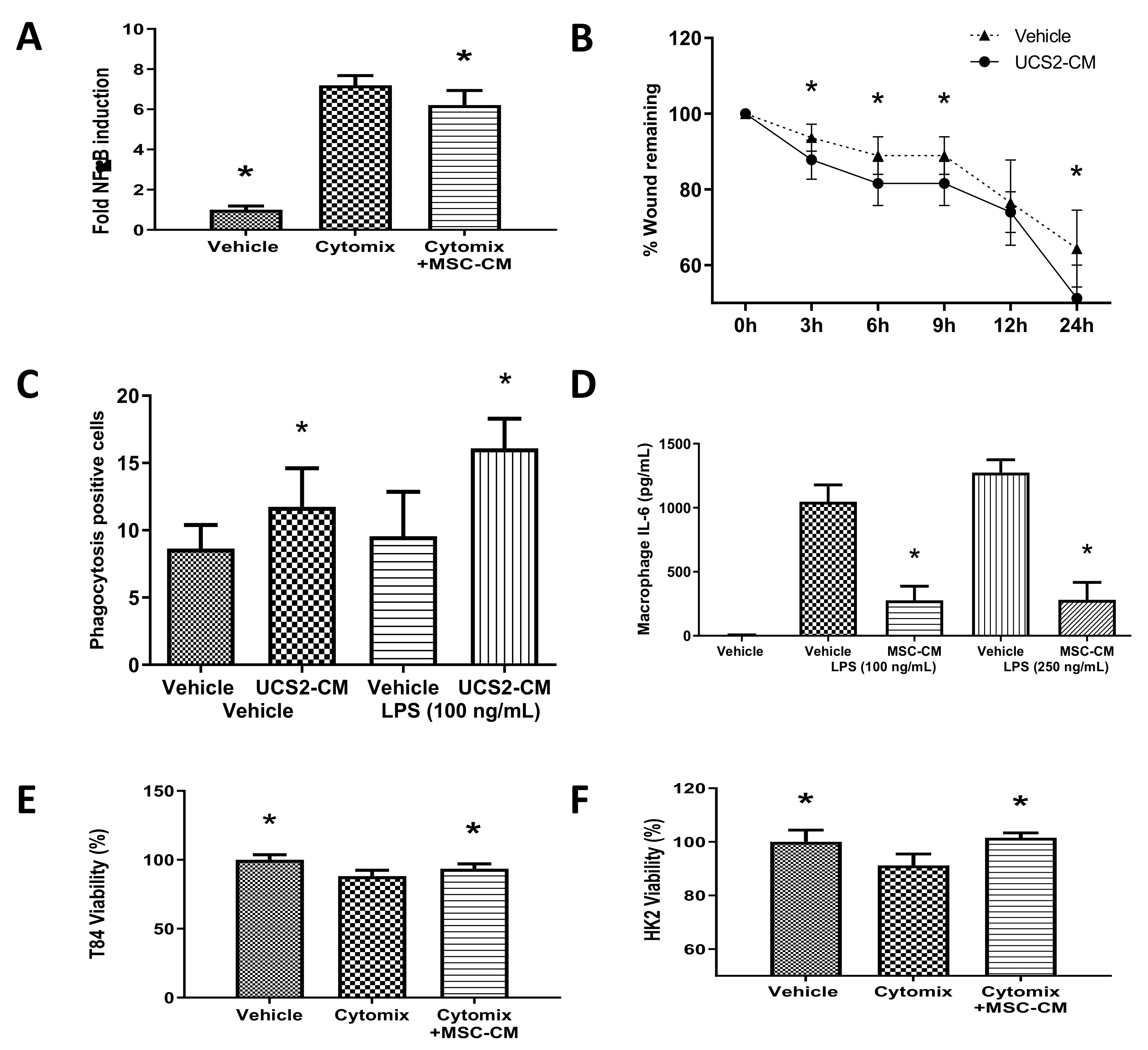
2.1. CD362+ UC-MSCs Demonstrate Immunomodulatory, Proreparative, Antibacterial and Prosurvival Effects In Vitro
2.2. CD362+ hMSCs Improve Survival in Severe Systemic Sepsis
2.3. CD362+ hMSCs Improve Hyperlactatemia and Bacterial Clearance at 4 h Administration Post Moderate CLP
2.4. CD362+ UC-MSC Administration Reduces Organ Viable Bacterial Load in Severe Systemic Sepsis
2.5. CD362+ UC-MSCs Administered 4 h Post CLP Reduce Systemic and Organ Presence of Inflammatory Cytokines
2.6. CD362+ UC-hMSCs Reduce Systemic Presence of Other Injury-Associated Molecules
3. Discussion
3.1. CD362+ UC-MSC-CM Immune Modulation In Vitro
3.2. CD362+ UC-MSCs Ameliorated CLP-Induced Sepsis
4. Materials and Methods
4.1. CD362-Targeted Isolation from Human Umbilical Cord Tissue
4.2. Generation of Conditioned Medium
4.3. In Vitro Determination of the Effects of CD362+ Human Mesenchymal Stromal Cells
- Nuclear Factor κB activation assay. A cell line derivative of type II alveolar A549 cells incorporating a stably transfected κB-luciferase reporter construct (Thermo Fisher, Waltham, MA, USA) was grown to confluence. Cell monolayers were randomised to receive cytomix (interleukin 1β (IL-1β) (10 ng/mL), TNF-α (50 ng/mL) and IFN-γ (50 ng/mL) (Immunotools Ltd., Friesoythe, Germany), or sham (vehicle) injury, then treated with either conditioned medium (CM) from CD362+ UC-MSCs, or vehicle. Cells were harvested at 24 h and assayed for luciferase content as an indicator of NF-κB activation.
- Scratch wound assay. A549 lung epithelial cells were seeded to 24-well plates at 100,000 cells per cm2 and the following day a single scratch wound was introduced per well with a p1000 pipette tip. Wells were aspirated, rinsed with PBS and re-fed with CD362+ UC-MSC-CM or vehicle. At various timepoints up to 24 h, scratch wounds were imaged by light microscopy and wound width assessed through measurement of pixel distance across the wound.
- Cell viability. 3-(4, 5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was performed using MTT reagent (thiazolyl blue tetrazolium bromide; Sigma Aldrich Ltd.) reconstituted in culture medium (5 mg/mL) to evaluate cell viability and proliferation. Human colonic adenocarcinoma cell line (T84) and human kidney proximal tubular cell line (HK-2) monolayers were randomised to receive cytomix, or sham (vehicle) activation, then treated with either CD362+ UC-MSC-CM, or vehicle. After treatment, cells were washed with PBS, followed by incubation with MTT reagent for 3 h at 37 °C in a humidified cell culture incubator. Cell supernatant was replaced with dimethyl sulfoxide and absorbance readings were measured using the Varioskan™ Flash microplate reader (Thermo Fisher Ltd.) at 595 nm wavelength. The degree of cell viability was presented as a percentage relative to uninjured control.
- Inflammatory cytokines production. Peritoneal macrophages were isolated from healthy rats by performing a peritoneal lavage with 20 mL of PBS, seeded to 96-well plates (c. 90,000 per well), and randomised to receive 100 ng/mL or 250 ng/mL of E. coli lipopolysaccharide (LPS), or sham (vehicle) activation, then treated with either CD362+ UC-MSC-CM, or vehicle. Interleukin 6—a cytokine under tight control of the NF-κB promoter [32]—production was measured by ELISA (R&D Systems, UK).
- Phagocytosis assay. THP-1 monocytes were seeded at 100,000 cells per cm2 in 96-well plates and exposed to PMA (1 µg/mL) for 72 h to force differentiation to macrophages. Lipopolysaccharide (100 or 250 ng/mL) was added for a further 24 h. Zymosan particles were then added for 4 h and phagocytosis quantified through counting of engulfed spots inside macrophages under fluorescent microscopy. Two fluorescent spots were considered positive for phagocytosis.
4.4. In Vivo Experimental Protocols
- CLP-induced sepsis. Adult male Sprague Dawley rats were anaesthetised by isoflurane inhalation. In order to induce polymicrobial sepsis, a midline laparotomy was performed, the caecum was exteriorised, and a 4–0 silk ligature was placed 5 mm from the caecal tip. The caecum was punctured twice with a biopsy punch (2 mm diameter for severe and 1 mm diameter for moderate severity) and a small amount of faecal content was extruded. The caecum was returned to the abdominal cavity and the midline incision was sutured. The rats were monitored and their status, including behavioural signs and welfare, was recorded up to 48 h after the CLP procedure.
4.5. Caecal Ligation and Puncture Systemic Sepsis Model
- Experimental series. Series 1: MSC administered within 30 min after severe CLP surgery (0 h). Groups: sham surgery (4), vehicle (9), CD362+ UC-MSC (10). Series 2: MSC administered 24 h after moderate CLP surgery (24 h). Groups: sham surgery (4), vehicle (9), CD362+ UC-MSC (11). Series 3: MSC administered 4 h after moderate CLP surgery (4 h). Groups: sham surgery (5), vehicle (12), CD362+ UC-MSC (10).
- Survival. In Series 1, animals were euthanised if they reached humane endpoints and time recorded. All animals were euthanised at 48 h.
- In Vivo assessment. At 48 h post CLP sepsis induction, animals were anaesthetised with intraperitoneal ketamine (80 mg.kg−1, Ketalar™; Pfizer, Cork, Ireland) and medetomidine (0.5 mg.kg-1, Dormidor™, Vetoquinol Ltd., Buckingham, UK). After confirmation of depth of anaesthesia by paw clamp, IV access was obtained via tail vein. Surgical tracheostomy was performed, using a 12G tracheostomy tube. Following intra-arterial access for blood sample analysis and monitoring, anaesthesia was maintained with alfaxalone (2 mg.kg-1, Alfaxan™; Vetoquinol Ltd.) and paralysis with cisatracurium besylate (0.5 mg.kg-1, Tracrium™; GlaxoSmithKline PLC., London, UK) and mechanical ventilation was commenced. Arterial blood lactate analysis was performed as previously described [10].
- Ex Vivo assessment. After exsanguination under anaesthesia, bronchoalveolar lavage (BAL) and peritoneal lavage (PL) were performed. Tissue samples from lung, liver, kidney and heart were collected for cytokine profiles and bacterial load measurements.
- Bacterial load. From the severe sepsis series, liver and spleen were immediately rinsed in 70% isopropyl alcohol and homogenised in 10 mL of PBS per gram of tissue. Homogenate was serial-diluted in PBS, plated to UTI agar plates (Fannin Ltd., Galway, Ireland) and incubated overnight at 37 °C. Total colony numbers of each indicative colour were counted. In the 4 h therapeutic moderate sepsis series, total DNA was isolated from serum, peritoneal lavage and organs by homogenisation and DNA affinity column (DNeasy Blood and Tissue Kit; Qiagen Ltd., Manchester, UK). PCR was performed using the following set of primers: forward 5′-TCCTACGGGAGGCAGCAGT-3′ (Tm 59.4 °C), reverse, 5′-GGACTACCAGGGTATCTAATCCTGTT-3′ (Tm 58.1 °C) targeting the 16S fraction of bacterial ribosomal RNA [33].
- Inflammatory cytokine profile. Cytokine-induced neutrophils chemoattractant (CINC-1), kidney injury molecule (KIM-1), interleukin 6 (IL-6) and hepcidin were quantified by ELISA (R&D Systems) and 23 other cytokines and growth factors where measured using a multiplex immunoassay system (Bio-Plex Pro Rat Cytokine, Chemokine and Growth Factor Assay; Bio-Rad Ltd., Watford, UK). The full multiplex dataset is available as Supplemental File S2.
- Statistical analyses. Data were analysed with GraphPad Prism software (GraphPad Software Ltd., San Diego, CA, USA). The distribution of all data was tested for normality with Kolmogorov–Smirnov tests. Data were analysed by two-way or one-way ANOVA or ANOVA on Ranks (Kruskall–Wallis) as appropriate, with post hoc testing by Dunnett ’s method, with the vehicle group as the single comparison group, or with Student–Newman–Keuls between-group comparisons, as appropriate. Underlying model assumptions were deemed appropriate on the basis of suitable residual plots. A two-tailed p value of less than 0.05 was considered significant.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ATMP | Advanced Therapeutic Medicinal Products |
| BM | Bone Marrow |
| CLP | Caecal Ligation and Puncture |
| ICU | Intensive Care Unit |
| IFN | Interferon |
| IL | Interleukin |
| KIM-1 | Kidney Injury Marker 1 |
| MSC | Mesenchymal Stromal Cell |
| PBS | Phosphate Buffered Saline |
| PMA | Phorbol-12-Myristate-13-Acetate |
| TNF | Tumour Necrosis Factor |
| UC | Umbilical Cord |
| UTI | Urinary Tract Infection |
References
- Gotts, J.E.; Matthay, M.A. Sepsis: Pathophysiology and clinical management. BMJ 2016, 353, i1585. [Google Scholar]
- Angus, D.C.; van der Poll, T. Severe sepsis and septic shock. N. Engl. J. Med. 2013, 369, 2063. [Google Scholar]
- Hotchkiss, R.S.; Karl, I.E. The pathophysiology and treatment of sepsis. N. Engl. J. Med. 2003, 348, 138–150. [Google Scholar]
- Ho, M.S.; Mei, S.H.; Stewart, D.J. The Immunomodulatory and Therapeutic Effects of Mesenchymal Stromal Cells for Acute Lung Injury and Sepsis. J. Cell. Physiol. 2015, 230, 2606–2617. [Google Scholar]
- Horie, S.; Gonzalez, H.E.; Laffey, J.G.; Masterson, C.H. Cell therapy in acute respiratory distress syndrome. J. Thorac. Dis. 2018, 10, 5607–5620. [Google Scholar]
- Rezoagli, E.; Murphy, E.J.; Laffey, J.; O’Toole, D. The Safety and Efficiency of Addressing ARDS Using Stem Cell Therapies in Clinical Trials. Stem Cell-Based Ther. Lung Dis. 2019, 219–238. [Google Scholar] [CrossRef]
- Weiss, A.R.R.; Dahlke, M.H. Immunomodulation by Mesenchymal Stem Cells (MSCs): Mechanisms of Action of Living, Apoptotic, and Dead MSCs. Front. Immunol. 2019, 10, 1191. [Google Scholar]
- Mei, S.H.; Haitsma, J.J.; Dos Santos, C.C.; Deng, Y.; Lai, P.F.; Slutsky, A.S.; Liles, W.C.; Stewart, D.J. Mesenchymal stem cells reduce inflammation while enhancing bacterial clearance and improving survival in sepsis. Am. J. Respir. Crit. Care Med. 2010, 182, 1047–1057. [Google Scholar]
- Nemeth, K.; Mayer, B.; Mezey, E. Modulation of bone marrow stromal cell functions in infectious diseases by toll-like receptor ligands. J. Mol. Med. 2010, 88, 5–10. [Google Scholar]
- Devaney, J.; Horie, S.; Masterson, C.; Elliman, S.; Barry, F.; O’Brien, T.; Curley, G.F.; O’Toole, D.; Laffey, J.G. Human mesenchymal stromal cells decrease the severity of acute lung injury induced by E. coli in the rat. Thorax 2015, 70, 625–635. [Google Scholar]
- Hayes, M.; Curley, G.F.; Masterson, C.; Devaney, J.; O’Toole, D.; Laffey, J.G. Mesenchymal stromal cells are more effective than the MSC secretome in diminishing injury and enhancing recovery following ventilator-induced lung injury. Intensive Care Med. Exp. 2015, 3, 29. [Google Scholar]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar]
- Phinney, D.G. Biochemical heterogeneity of mesenchymal stem cell populations: Clues to their therapeutic efficacy. Cell Cycle 2007, 6, 2884–2889. [Google Scholar]
- Masterson, C.; Devaney, J.; Horie, S.; O’Flynn, L.; Deedigan, L.; Elliman, S.; Barry, F.; O’Brien, T.; O’Toole, D.; Laffey, J.G. Syndecan-2-positive, Bone Marrow-derived Human Mesenchymal Stromal Cells Attenuate Bacterial-induced Acute Lung Injury and Enhance Resolution of Ventilator-induced Lung Injury in Rats. Anesthesiology 2018, 129, 502–516. [Google Scholar]
- Horie, S.; Masterson, C.; Brady, J.; Loftus, P.; Horan, E.; O’Flynn, L.; Elliman, S.; Barry, F.; O’Brien, T.; Laffey, J.G.; et al. Umbilical cord-derived CD362+ mesenchymal stromal cells for E. coli pneumonia: Impact of dose regimen, passage, cryopreservation, and antibiotic therapy. Stem Cell Res. Ther. 2020, 11, 116. [Google Scholar]
- Hayes, M.; Masterson, C.; Devaney, J.; Barry, F.; Elliman, S.; O’Brien, T.; O’Toole, D.; Curley, G.F.; Laffey, J.G. Therapeutic efficacy of human mesenchymal stromal cells in the repair of established ventilator-induced lung injury in the rat. Anesthesiology 2015, 122, 363–373. [Google Scholar]
- McCarthy, S.D.; Horgan, E.; Ali, A.; Masterson, C.; Laffey, J.G.; MacLoughlin, R.; O’Toole, D. Nebulized Mesenchymal Stem Cell Derived Conditioned Medium Retains Antibacterial Properties Against Clinical Pathogen Isolates. J. Aerosol Med. Pulm. Drug Deliv. 2020, 33, 140–152. [Google Scholar]
- Francois, B.; Trimoreau, F.; Vignon, P.; Fixe, P.; Praloran, V.; Gastinne, H. Thrombocytopenia in the sepsis syndrome: Role of hemophagocytosis and macrophage colony-stimulating factor. Am. J. Med. 1997, 103, 114–120. [Google Scholar]
- Huang, H.; Wang, S.; Jiang, T.; Fan, R.; Zhang, Z.; Mu, J.; Li, K.; Wang, Y.; Jin, L.; Lin, F.; et al. High levels of circulating GM-CSF+CD4+ T cells are predictive of poor outcomes in sepsis patients: A prospective cohort study. Cell. Mol. Immunol. 2019, 16, 602–610. [Google Scholar]
- Pickkers, P.; Sprong, T.; Eijk, L.; Hoeven, H.; Smits, P.; Deuren, M. Vascular endothelial growth factor is increased during the first 48 hours of human septic shock and correlates with vascular permeability. Shock 2005, 24, 508–512. [Google Scholar]
- Whitney, J.E.; Silverman, M.; Norton, J.S.; Bachur, R.G.; Melendez, E. Vascular Endothelial Growth Factor and Soluble Vascular Endothelial Growth Factor Receptor as Novel Biomarkers for Poor Outcomes in Children with Severe Sepsis and Septic Shock. Pediatr. Emerg. Care 2018. [Google Scholar] [CrossRef]
- Cecconi, M.; Evans, L.; Levy, M.; Rhodes, A. Sepsis and septic shock. Lancet 2018, 392, 75–87. [Google Scholar]
- Capcha, J.M.C.; Rodrigues, C.E.; Moreira, R.S.; Silveira, M.D.; Dourado, P.; Dos Santos, F.; Irigoyen, M.C.; Jensen, L.; Garnica, M.R.; Noronha, I.L.; et al. Wharton’s jelly-derived mesenchymal stem cells attenuate sepsis-induced organ injury partially via cholinergic anti-inflammatory pathway activation. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2020, 318, R135–R147. [Google Scholar]
- Li, S.; Wu, H.; Han, D.; Ma, S.; Fan, W.; Wang, Y.; Zhang, R.; Fan, M.; Huang, Y.; Fu, X.; et al. A Novel Mechanism of Mesenchymal Stromal Cell-Mediated Protection against Sepsis: Restricting Inflammasome Activation in Macrophages by Increasing Mitophagy and Decreasing Mitochondrial ROS. Oxid. Med. Cell. Longev. 2018, 2018, 3537609. [Google Scholar]
- Park, K.S.; Svennerholm, K.; Shelke, G.V.; Bandeira, E.; Lasser, C.; Jang, S.C.; Chandode, R.; Gribonika, I.; Lotvall, J. Mesenchymal stromal cell-derived nanovesicles ameliorate bacterial outer membrane vesicle-induced sepsis via IL-10. Stem Cell Res. Ther. 2019, 10, 231. [Google Scholar]
- Pedrazza, L.; Cubillos-Rojas, M.; de Mesquita, F.C.; Luft, C.; Cunha, A.A.; Rosa, J.L.; de Oliveira, J.R. Mesenchymal stem cells decrease lung inflammation during sepsis, acting through inhibition of the MAPK pathway. Stem Cell Res. Ther. 2017, 8, 289. [Google Scholar]
- Boomer, J.S.; Green, J.M.; Hotchkiss, R.S. The changing immune system in sepsis: Is individualized immuno-modulatory therapy the answer? Virulence 2014, 5, 45–56. [Google Scholar]
- Venet, F.; Demaret, J.; Gossez, M.; Monneret, G. Myeloid cells in sepsis-acquired immunodeficiency. Ann. N. Y. Acad. Sci. 2020. [Google Scholar] [CrossRef]
- Gauer, R.; Forbes, D.; Boyer, N. Sepsis: Diagnosis and Management. Am. Fam. Physician 2020, 101, 409–418. [Google Scholar]
- Krasnodembskaya, A.; Song, Y.; Fang, X.; Gupta, N.; Serikov, V.; Lee, J.W.; Matthay, M.A. Antibacterial effect of human mesenchymal stem cells is mediated in part from secretion of the antimicrobial peptide LL-37. Stem Cells 2010, 28, 2229–2238. [Google Scholar]
- Wood, C.R.; Al Dhahri, D.; Al Delfi, I.; Pickles, N.A.; Sammons, R.L.; Worthington, T.; Wright, K.T.; Johnson, W.E.B. Human adipose tissue-derived mesenchymal stem/stromal cells adhere to and inhibit the growth of Staphylococcus aureus and Pseudomonas aeruginosa. J. Med. Microbiol. 2018, 67, 1789–1795. [Google Scholar]
- Libermann, T.A.; Baltimore, D. Activation of interleukin-6 gene expression through the NF-kappa B transcription factor. Mol. Cell. Biol. 1990, 10, 2327–2334. [Google Scholar]
- Rampini, S.K.; Bloemberg, G.V.; Keller, P.M.; Buchler, A.C.; Dollenmaier, G.; Speck, R.F.; Bottger, E.C. Broad-range 16S rRNA gene polymerase chain reaction for diagnosis of culture-negative bacterial infections. Clin. Infect. Dis. 2011, 53, 1245–1251. [Google Scholar]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonzalez, H.; Keane, C.; Masterson, C.H.; Horie, S.; Elliman, S.J.; Higgins, B.D.; Scully, M.; Laffey, J.G.; O’Toole, D. Umbilical Cord-Derived CD362+ Mesenchymal Stromal Cells Attenuate Polymicrobial Sepsis Induced by Caecal Ligation and Puncture. Int. J. Mol. Sci. 2020, 21, 8270. https://doi.org/10.3390/ijms21218270
Gonzalez H, Keane C, Masterson CH, Horie S, Elliman SJ, Higgins BD, Scully M, Laffey JG, O’Toole D. Umbilical Cord-Derived CD362+ Mesenchymal Stromal Cells Attenuate Polymicrobial Sepsis Induced by Caecal Ligation and Puncture. International Journal of Molecular Sciences. 2020; 21(21):8270. https://doi.org/10.3390/ijms21218270
Chicago/Turabian StyleGonzalez, Hector, Colm Keane, Claire H. Masterson, Shahd Horie, Stephen J. Elliman, Brendan D. Higgins, Michael Scully, John G. Laffey, and Daniel O’Toole. 2020. "Umbilical Cord-Derived CD362+ Mesenchymal Stromal Cells Attenuate Polymicrobial Sepsis Induced by Caecal Ligation and Puncture" International Journal of Molecular Sciences 21, no. 21: 8270. https://doi.org/10.3390/ijms21218270
APA StyleGonzalez, H., Keane, C., Masterson, C. H., Horie, S., Elliman, S. J., Higgins, B. D., Scully, M., Laffey, J. G., & O’Toole, D. (2020). Umbilical Cord-Derived CD362+ Mesenchymal Stromal Cells Attenuate Polymicrobial Sepsis Induced by Caecal Ligation and Puncture. International Journal of Molecular Sciences, 21(21), 8270. https://doi.org/10.3390/ijms21218270




