Comprehensive Set of Tertiary Complex Structures and Palmitic Acid Binding Provide Molecular Insights into Ligand Design for RXR Isoforms
Abstract
1. Introduction
2. Results
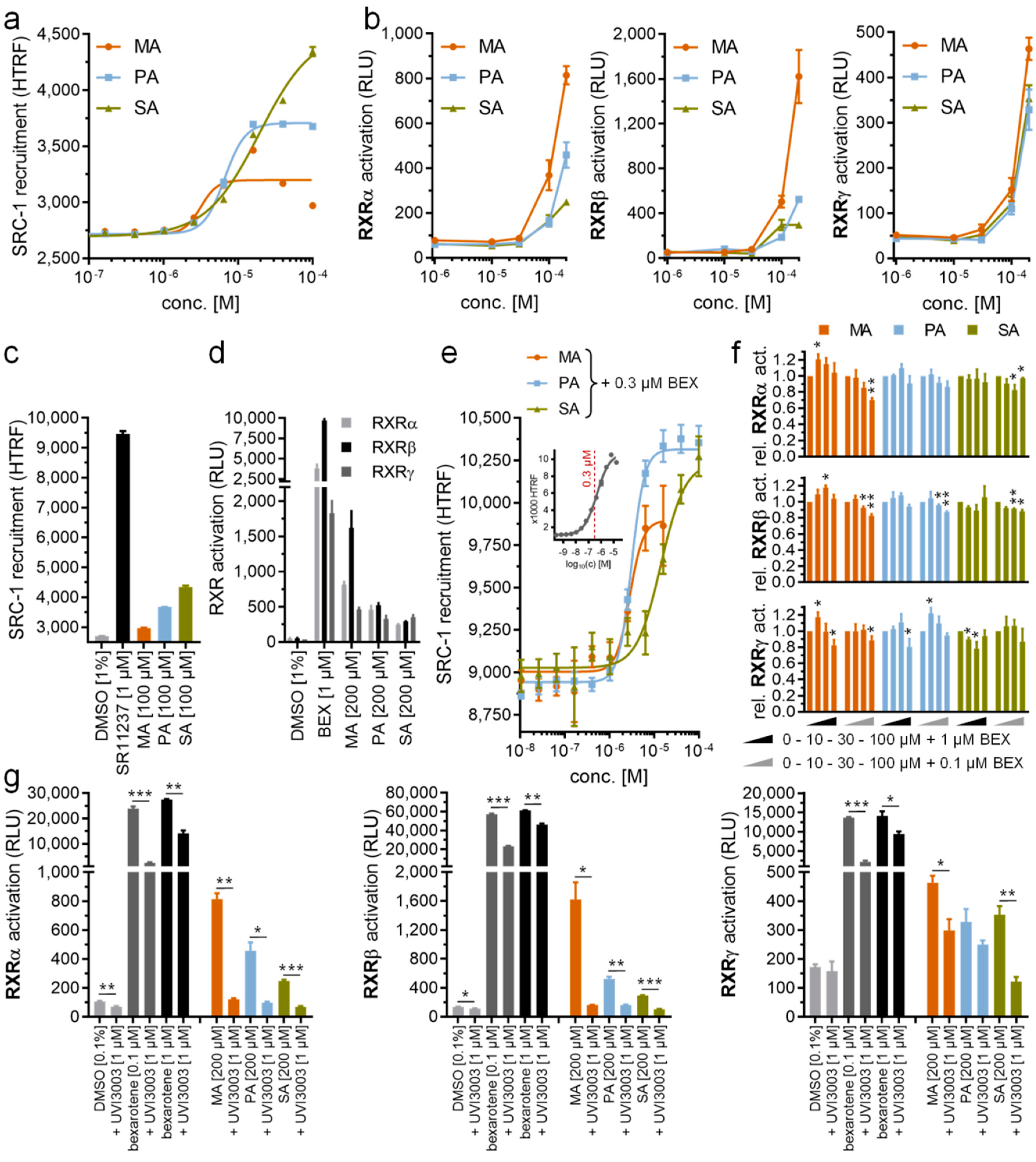
2.1. Unprecedented Identification of Palmitic Acid Binding to RXRα
2.2. Palmitic Acid Also Interacted with Other RXR Isoforms
2.3. Binding of PA in RXRs
2.4. Structural Analysis of Amino Acid Differences among RXR Isoforms
2.5. PA Acts as a Modulator of RXRs
3. Discussion
4. Materials and Methods
4.1. Expression and Purification of RXR Isoform LBDs
4.2. Crysallization and Structure Determination
4.3. Hybrid Reporter Gene Assays for RXRα, RXRβ and RXRγ
4.4. SRC-1 Recruitment Assay (HTRF)
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s disease |
| DTT | dithiotreitol |
| FI | fluorescence intensity |
| FRET | fluorescence resonance energy transfer |
| GRIP-1 | glucocorticoid receptor-interacting protein-1 |
| HTRF | homogenous time-resolved fluorescence resonance energy transfer |
| LBD | Ligand-binding domain |
| MA | myristic acid |
| Nurr1 | nuclear receptor related 1 |
| PA | palmitic acid |
| PPAR | peroxisome proliferator-activated receptor (NR1C1-3) |
| RMSD | root-mean-square deviation of atomic positions |
| RXR | retinoid X receptor (RXRα: NR2B1; RXRβ: NR2B2; RXRγ: NR2B3) |
| SA | stearic acid |
| SRC-1 | steroid receptor co-activator 1 |
| THR | thyroid hormone receptor (NR1A1-2) |
References
- Germain, P.; Chambon, P.; Eichele, G.; Evans, R.M.; Lazar, M.A.; Leid, M.; De Lera, A.R.; Lotan, R.; Mangelsdorf, D.J.; Gronemeyer, H. International Union of Pharmacology. LXIII. Retinoid X receptors. Pharmacol. Rev. 2006, 58, 760–772. [Google Scholar] [CrossRef]
- Nuclear Receptors Nomenclature Committee. A unified nomenclature system for the nuclear receptor superfamily. Cell 1999, 97, 161–163. [Google Scholar] [CrossRef]
- Alexander, S.P.; Cidlowski, J.A.; Kelly, E.; Marrion, N.V.; Peters, J.A.; Faccenda, E.; Harding, S.D.; Pawson, A.J.; Sharman, J.L.; Southan, C.; et al. THE CONCISE GUIDE TO PHARMACOLOGY 2017/18: Nuclear hormone receptors. Br. J. Pharmacol. 2017, 174, S208–S224. [Google Scholar] [CrossRef]
- Willems, S.; Kilu, W.; Ni, X.; Chaikuad, A.; Knapp, S.; Heering, J.; Merk, D. The orphan nuclear receptor Nurr1 is responsive to non-steroidal anti-inflammatory drugs. Commun. Chem. 2020, 3, 85. [Google Scholar] [CrossRef]
- Jiang, L.; Dai, S.; Li, J.; Liang, X.; Qu, L.; Chen, X.; Guo, M.; Chen, Z.; Chen, L.; Wei, H.; et al. Structural basis of binding of homodimers of the nuclear receptor NR4A2 to selective Nur-responsive DNA elements. J. Biol. Chem. 2019. [Google Scholar] [CrossRef]
- Fitzgerald, P.; Teng, M.; Chandraratna, R.A.S.; Heyman, R.A.; Allegretto, E.A. Retinoic acid receptor expression correlates with retinoid-induced growth inhibition of human breast cancer cells regardless of estrogen receptor status. Cancer Res. 1997, 57, 2642–2650. [Google Scholar] [PubMed]
- Rühl, R.; Krzyzosiak, A.; Niewiadomska-Cimicka, A.; Rochel, N.; Szeles, L.; Vaz, B.; Wietrzych-Schindler, M.; Álvarez, S.; Szklenar, M.; Nagy, L.; et al. 9- cis -13, 14-Dihydroretinoic Acid Is an Endogenous Retinoid Acting as RXR Ligand in Mice. PLoS Genet. 2015, 1–16. [Google Scholar] [CrossRef]
- de Lera, Á.; Krezel, W.; Rühl, R. An endogenous mammalian retinoid X receptor ligand, at last! ChemMedChem 2016, 11, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Lengqvist, J.; De Urquiza, A.M.; Bergman, A.; Willson, T.M.; Sjövall, J.; Perlmann, T.; Griffiths, W.J. Polyunsaturated Fatty Acids Including Docosahexaenoic and Arachidonic Acid Bind to the Retinoid X Receptor α Ligand-binding Domain. Mol. Proteomics 2004, 3, 692–703. [Google Scholar] [CrossRef] [PubMed]
- Schierle, S.; Merk, D. Therapeutic modulation of retinoid X receptors–SAR and therapeutic potential of RXR ligands and recent patents. Expert Opin. Ther. Pat. 2019, 29, 605–621. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Drew, P.D. 9-Cis-retinoic acid suppresses inflammatory responses of microglia and astrocytes. J. Neuroimmunol. 2006, 171, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Schrage, K.; Koopmans, G.; Joosten, E.A.J.; Mey, J. Macrophages and neurons are targets of retinoic acid signaling after spinal cord contusion injury. Eur. J. Neurosci. 2006, 23, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.K.; Jarjour, A.A.; Nait Oumesmar, B.; Kerninon, C.; Williams, A.; Krezel, W.; Kagechika, H.; Bauer, J.; Zhao, C.; Baron-Van Evercooren, A.; et al. Retinoid X receptor gamma signaling accelerates CNS remyelination. Nat. Neurosci. 2011, 14, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Natrajan, M.S.; De Fuente, A.G.; Crawford, A.H.; Linehan, E.; Johnson, K.R.; Wu, T.; Fitzgerald, D.C.; Ricote, M. Retinoid X receptor activation reverses age-related deficiencies in myelin debris phagocytosis and remyelination. Brain 2018, 138, 3581–3597. [Google Scholar] [CrossRef]
- Chandraratna, R.A.; Noelle, R.J.; Nowak, E.C. Treatment with retinoid X receptor agonist IRX4204 ameliorates experimental autoimmune encephalomyelitis. Am. J. Transl. Res. 2016, 8, 1016–1026. [Google Scholar]
- Kremer, D.; Akkermann, R.; Küry, P.; Dutta, R. Current advancements in promoting remyelination in multiple sclerosis. Mult. Scler. J. 2019, 25, 7–14. [Google Scholar] [CrossRef]
- Cramer, P.E.; Cirrito, J.R.; Wesson, D.W.; Lee, C.Y.D.; Karlo, J.C.; Zinn, A.E.; Casali, B.T.; Restivo, J.L.; Goebel, W.D.; James, M.J.; et al. ApoE-directed therapeutics rapidly clear β-amyloid and reverse deficits in AD mouse models. Science 2012, 335, 1503–1506. [Google Scholar] [CrossRef]
- Boehm-Cagan, A.; Michaelson, D.M. Reversal of apoE4-driven brain pathology and behavioral deficits by Bexarotene. J. Neurosci. 2014, 34, 7293–7301. [Google Scholar] [CrossRef]
- Yuan, C.; Guo, X.; Zhou, Q.; Du, F.; Jiang, W.; Zhou, X.; Liu, P.; Chi, T.; Ji, X.; Gao, J.; et al. OAB-14, a bexarotene derivative, improves Alzheimer’s disease-related pathologies and cognitive impairments by increasing β-amyloid clearance in APP/PS1 mice. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 161–180. [Google Scholar] [CrossRef]
- Koster, K.P.; Smith, C.; Valencia-Olvera, A.C.; Thatcher, G.R.J.; Tai, L.M.; LaDu, M.J. Rexinoids as therapeutics for Alzheimer disease: Role of APOE. Curr. Top. Med. Chem. 2017, 17, 708–720. [Google Scholar] [CrossRef]
- Mariani, M.M.; Malm, T.; Lamb, R.; Jay, T.R.; Neilson, L.; Casali, B.; Medarametla, L.; Landreth, G.E. Neuronally-directed effects of RXR activation in a mouse model of Alzheimer’s disease. Sci. Rep. 2017, 7, 42270. [Google Scholar] [CrossRef] [PubMed]
- Gui, Y.; Duan, S.; Xiao, L.; Tang, J.; Li, A. Bexarotent attenuated CCI-induced spinal neuroinflammation and neuropathic pain by targeting MKP-1. J. Pain 2019. [Google Scholar] [CrossRef] [PubMed]
- Tousi, B. The emerging role of bexarotene in the treatment of Alzheimer’s disease: Current evidence. Neuropsychiatr. Dis. Treat. 2015, 11, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Riancho, J.; Berciano, M.T.; Berciano, J.; Lafarga, M. Relaunching an old drug: The potential role of bexarotene in neurodegenerative diseases. J. Neurol. 2016, 263, 177–178. [Google Scholar] [CrossRef]
- Cummings, J.L.; Zhong, K.; Kinney, J.W.; Heaney, C.; Moll-Tudla, J.; Joshi, A.; Pontecorvo, M.; Devous, M.; Tang, A.; Bena, J. Double-blind, placebo-controlled, proof-of-concept trial of bexarotene in moderate Alzheimer’s disease. Alzheimers. Res. Ther. 2016, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Sherman, S.I.; Gopal, J.; Haugen, B.R.; Chiu, A.C.; Whaley, K.; Nowlakha, P.; Duvic, M. Central Hypothyroidism Associated with Retinoid X Receptor–Selective Ligands. N. Engl. J. Med. 1999, 340, 1075–1079. [Google Scholar] [CrossRef]
- Farol, L.T.; Hymes, K.B. Bexarotene: A clinical review. Expert Rev. Anticancer. Ther. 2004, 4, 180–188. [Google Scholar] [CrossRef]
- Mangelsdorf, D.J.; Borgmeyer, U.; Heyman, R.A.; Zhou, J.Y.; Ong, E.S.; Oro, A.E.; Kakizuka, A.; Evans, R.M. Characterization of three RXR genes that mediate the action of 9-cis retinoic acid. Genes Dev. 1992, 6, 329–344. [Google Scholar] [CrossRef]
- de Lera, A.R.; Bourguet, W.; Altucci, L.; Gronemeyer, H. Design of selective nuclear receptor modulators: RAR and RXR as a case study. Nat. Rev. Drug Discov. 2007, 6, 811–820. [Google Scholar] [CrossRef]
- Merk, D.; Grisoni, F.; Friedrich, L.; Gelzinyte, E.; Schneider, G. Computer-Assisted Discovery of Retinoid X Receptor Modulating Natural Products and Isofunctional Mimetics. J. Med. Chem. 2018, 61, 5442–5447. [Google Scholar] [CrossRef]
- Pollinger, J.; Schierle, S.; Gellrich, L.; Ohrndorf, J.; Kaiser, A.; Heitel, P.; Chaikuad, A.; Knapp, S.; Merk, D. A Novel Biphenyl-based Chemotype of Retinoid X Receptor Ligands Enables Subtype and Heterodimer Preferences. ACS Med. Chem. Lett. 2019, 10, 1346–1352. [Google Scholar] [CrossRef] [PubMed]
- Merk, D.; Grisoni, F.; Friedrich, L.; Gelzinyte, E.; Schneider, G. Scaffold hopping from synthetic RXR modulators by virtual screening and de novo design. Medchemcomm 2018, 9, 1289–1292. [Google Scholar] [CrossRef] [PubMed]
- Pollinger, J.; Gellrich, L.; Schierle, S.; Kilu, W.; Schmidt, J.; Kalinowsky, L.; Ohrndorf, J.; Kaiser, A.; Heering, J.; Proschak, E.; et al. Tuning Nuclear Receptor Selectivity of Wy14,643 towards Selective Retinoid X Receptor Modulation. J. Med. Chem. 2019, 62, 2112–2126. [Google Scholar] [CrossRef] [PubMed]
- Boerma, L.J.; Xia, G.; Qui, C.; Cox, B.D.; Chalmers, M.J.; Smith, C.D.; Lobo-Ruppert, S.; Griffin, P.R.; Muccio, D.D.; Renfrow, M.B. Defining the communication between agonist and coactivator binding in the retinoid X receptor α ligand binding domain. J. Biol. Chem. 2014, 289, 814–826. [Google Scholar] [CrossRef]
- Gellrich, L.; Heitel, P.; Heering, J.; Kilu, W.; Pollinger, J.; Goebel, T.; Kahnt, A.; Arifi, S.; Pogoda, W.; Paulke, A.; et al. l-Thyroxin and the Nonclassical Thyroid Hormone TETRAC Are Potent Activators of PPARγ. J. Med. Chem. 2020, 63, 6727–6740. [Google Scholar] [CrossRef]
- Proschak, E.; Heitel, P.; Kalinowsky, L.; Merk, D. Opportunities and challenges for fatty acid mimetics in drug discovery. J. Med. Chem. 2017, 60, 5235–5266. [Google Scholar] [CrossRef]
- Santín, E.P.; Germain, P.; Quillard, F.; Khanwalkar, H.; Rodríguez-Barrios, F.; Gronemeyer, H.; De Lera, Á.R.; Bourguet, W. Modulating retinoid X receptor with a series of (E)-3-[4-hydroxy-3-(3- alkoxy-5,5,8,8-tetramethyl-5,6,7,8-tetrahydronaphthalen-2-yl)phenyl]acrylic acids and their 4-alkoxy isomers. J. Med. Chem. 2009, 52, 3150–3158. [Google Scholar] [CrossRef]
- Sato, Y.; Ramalanjaona, N.; Huet, T.; Potier, N.; Osz, J.; Antony, P.; Peluso-Iltis, C.; Poussin-Courmontagne, P.; Ennifar, E.; Mély, Y.; et al. The “phantom effect” of the rexinoid LG100754: Structural and functional insights. PLoS ONE 2010, 5, e15119. [Google Scholar] [CrossRef]
- Forman, B.M. The antidiabetic agent LG100754 sensitizes cells to low concentrations of peroxisome proliferator-activated receptor γ ligands. J. Biol. Chem. 2002, 277, 12503–12506. [Google Scholar] [CrossRef]
- Kawata, K.; Morishita, K.; Nakayama, M.; Yamada, S.; Kobayashi, T.; Furusawa, Y.; Arimoto-Kobayashi, S.; Oohashi, T.; Makishima, M.; Naitou, H.; et al. RXR Partial Agonist Produced by Side Chain Repositioning of Alkoxy RXR Full Agonist Retains Antitype 2 Diabetes Activity without the Adverse Effects. J. Med. Chem. 2015, 58, 912–926. [Google Scholar] [CrossRef]
- Lagu, B.; Pio, B.; Lebedev, R.; Yang, M.; Pelton, P.D. RXR–LXR heterodimer modulators for the potential treatment of dyslipidemia. Bioorg. Med. Chem. Lett. 2007, 17, 3497–3503. [Google Scholar] [CrossRef] [PubMed]
- Scheepstra, M.; Andrei, S.A.; De Vries, R.M.J.M.; Meijer, F.A.; Ma, J.N.; Burstein, E.S.; Olsson, R.; Ottmann, C.; Milroy, L.G.; Brunsveld, L. Ligand Dependent Switch from RXR Homo- to RXR-NURR1 Heterodimerization. ACS Chem. Neurosci. 2017, 8, 2065–2077. [Google Scholar] [CrossRef] [PubMed]
- le Maire, A.; Teyssier, C.; Balaguer, P.; Bourguet, W.; Germain, P. Regulation of RXR-RAR Heterodimers by RXR- and RAR-Specific Ligands and Their Combinations. Cells 2019, 8, 1392. [Google Scholar] [CrossRef] [PubMed]
- Love, J.D.; Gooch, J.T.; Benko, S.; Li, C.; Nagy, L.; Chatterjee, V.K.K.; Evans, R.M.; Schwabe, J.W.R. The structural basis for the specificity of retinoid-X receptor-selective agonists: New insights into the role of helix H12. J. Biol. Chem. 2002, 277, 11385–11391. [Google Scholar] [CrossRef] [PubMed]
- Abdelmagid, S.A.; Clarke, S.E.; Nielsen, D.E.; Badawi, A.; El-Sohemy, A.; Mutch, D.M.; Ma, D.W.L. Comprehensive profiling of plasma fatty acid concentrations in young healthy canadian adults. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Kabsch, W. XDS. Acta Crystallogr. D. Biol. Crystallogr. 2010, 66, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Evans, P.R.; Murshudov, G.N. How good are my data and what is the resolution? Acta Crystallogr. D Biol. Crystallogr. 2013, 69, 1204–1214. [Google Scholar] [CrossRef]
- McCoy, A.J.; Grosse-Kunstleve, R.W.; Adams, P.D.; Winn, M.D.; Storoni, L.C.; Read, R.J. Phaser crystallographic software. J. Appl. Crystallogr. 2007, 40, 658–674. [Google Scholar] [CrossRef]
- Emsley, P. Tools for ligand validation in Coot. Acta Crystallogr. Sect. D Struct. Biol. 2017, 73, 203–210. [Google Scholar] [CrossRef]
- Winn, M.D.; Murshudov, G.N.; Papiz, M.Z. Macromolecular TLS Refinement in REFMAC at Moderate Resolutions. Methods Enzymol. 2003, 374, 300–321. [Google Scholar] [CrossRef]
- Williams, C.J.; Headd, J.J.; Moriarty, N.W.; Prisant, M.G.; Videau, L.L.; Deis, L.N.; Verma, V.; Keedy, D.A.; Hintze, B.J.; Chen, V.B.; et al. MolProbity: More and better reference data for improved all-atom structure validation. Protein Sci. 2018, 27, 293–315. [Google Scholar] [CrossRef] [PubMed]
- Chaikuad, A.; Knapp, S.; Von Delft, F. Defined PEG smears as an alternative approach to enhance the search for crystallization conditions and crystal-quality improvement in reduced screens. Acta Crystallogr. Sect. D Biol. Crystallogr. 2015, 71, 1627–1639. [Google Scholar] [CrossRef] [PubMed]
- Flesch, D.; Cheung, S.-Y.; Schmidt, J.; Gabler, M.; Heitel, P.; Kramer, J.S.; Kaiser, A.; Hartmann, M.; Lindner, M.; Lüddens-Dämgen, K.; et al. Non-acidic farnesoid X receptor modulators. J. Med. Chem. 2017, 60, 7199–7205. [Google Scholar] [CrossRef] [PubMed]
- Heitel, P.; Gellrich, L.; Kalinowsky, L.; Heering, J.; Kaiser, A.; Ohrndorf, J.; Proschak, E.; Merk, D. Computer-Assisted Discovery and Structural Optimization of a Novel Retinoid X Receptor Agonist Chemotype. ACS Med. Chem. Lett. 2019, 10, 203–208. [Google Scholar] [CrossRef]
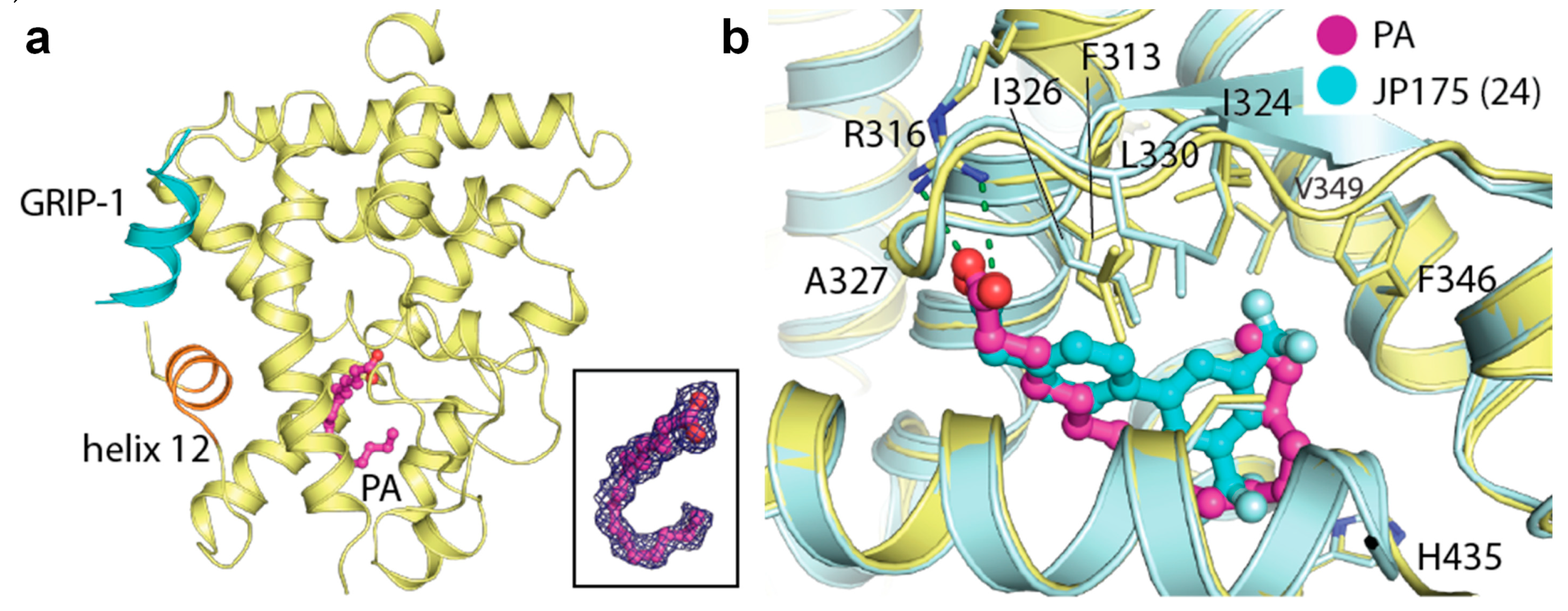
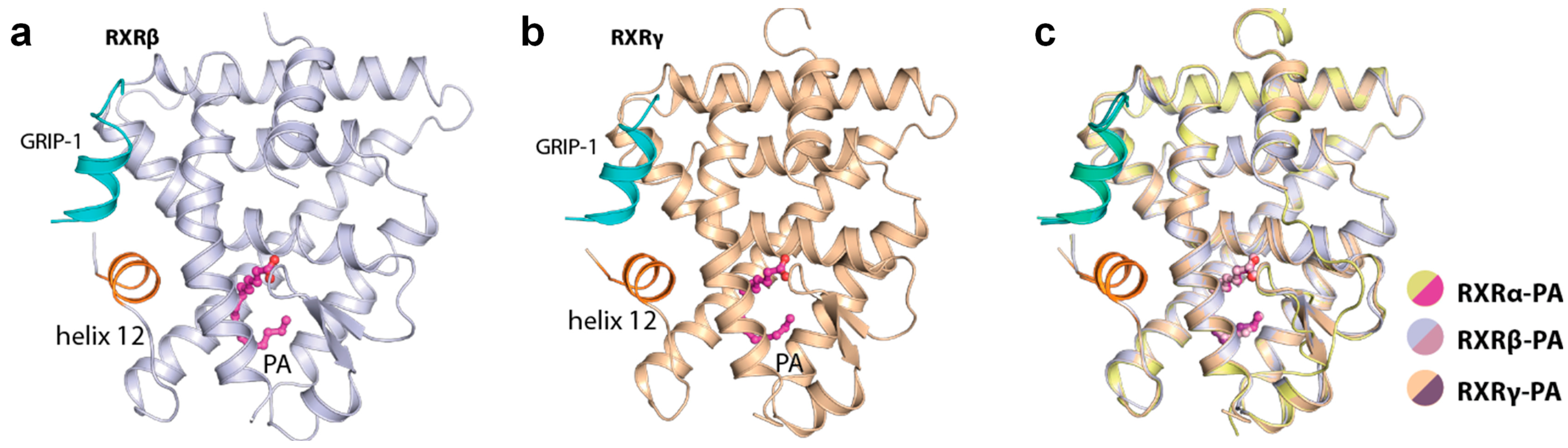
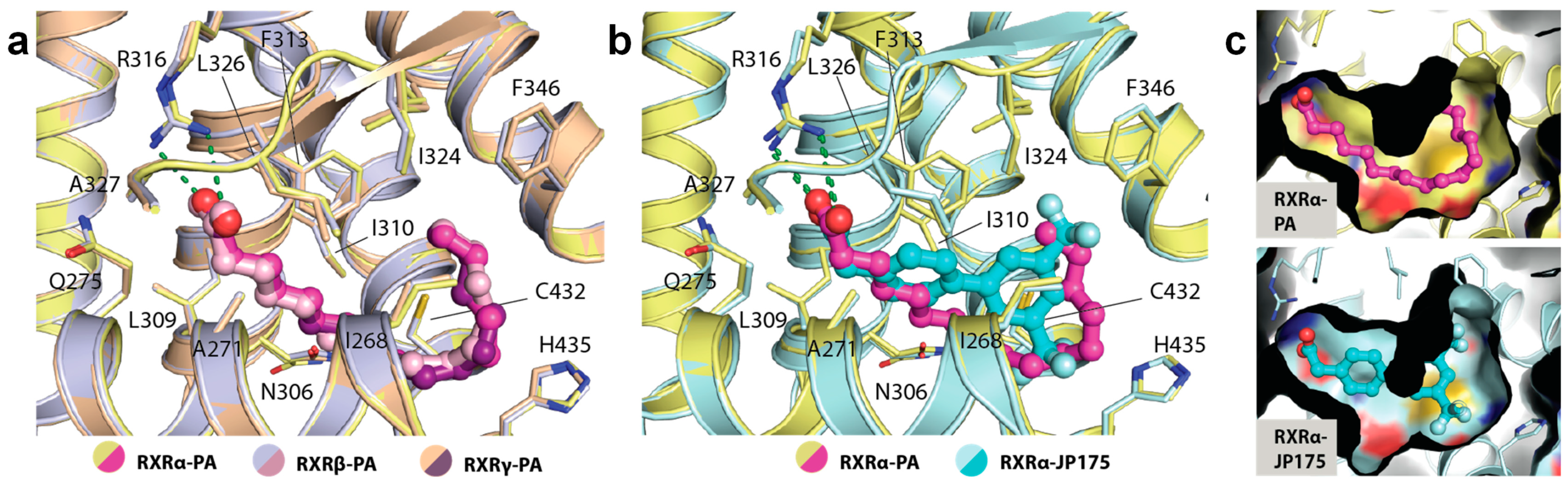
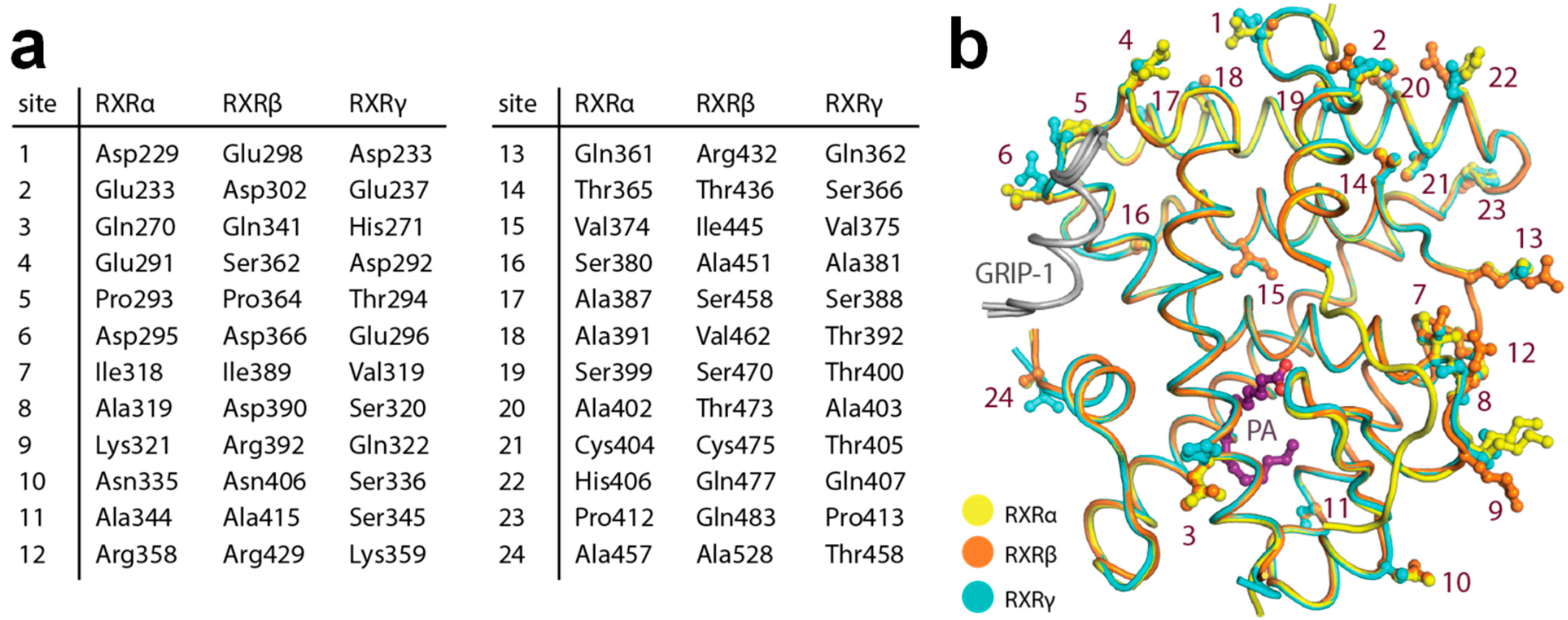
| Complex a | RXRα-PA-GRIP-1 | RXRβ-PA-GRIP-1 | RXRγ-PA-GRIP-1 |
|---|---|---|---|
| PDB codes | 7A77 | 7A78 | 7A79 |
| Beamline | DESY P13 | SLS X06SA | SLS X06SA |
| Data Collection | |||
| Resolution b (Å) | 43.10–1.50 (1.55–1.50) | 45.0–1.72 (1.78–1.72) | 46.01–2.05 (2.12–2.05) |
| Space group | P 43212 | P 43212 | P 212121 |
| Cell dimensions | a=b=66.2, c=110.6 Å | a=b=63.6, c=110.1 Å | a=63.3, b=67.1, c=110.7 Å |
| α=β=γ=90.0 | α=β=γ=90.0 | α=β=γ=90.0° | |
| Number of unique reflections | 40,177 (3855) | 24,821 (2373) | 30,235 (2910) |
| Completeness (%) | 100.0 (100.0) | 100.0 (100.0) | 99.9 (100.0) |
| I/σI | 20.0 (4.0) | 15.3 (2.3) | 8.2 (2.0) |
| Rmerge (%) | 0.073 (0.703) | 0.059 (0.875) | 0.148 (0.782) |
| CC (1/2) | 0.999 (0.876) | 0.999 (0.769) | 0.994 (0.754) |
| Redundancy | 15.6 (14.2) | 8.0 (8.3) | 8.5 (7.9) |
| Refinement | |||
| Number atoms in refinement (P/G/L/O) c | 1841/ 99/ 18/ 237 | 1688/ 111/ 18/ 160 | 3416/ 210/ 36/ 182 |
| B factor (P/G/L/O) c (Å2) | 22/ 29/ 29/ 35 | 37/ 40/ 39/ 44 | 38/ 48/ 47/ 43 |
| Rfact (%) | 15.2 | 17.7 | 19.5 |
| Rfree (%) | 18.0 | 20.4 | 24.8 |
| rms deviation bond c (Å) | 0.015 | 0.013 | 0.014 |
| rms deviation angle c (°) | 1.6 | 1.4 | 1.4 |
| Molprobity Ramachandran | |||
| Favor (%) | 97.4 | 99.0 | 98.0 |
| Outlier (%) | 0 | 0 | 0 |
| Crystallization condition d | 17% PEG 3350, 0.2 M ammonium acetate, 0.1 M tris, pH 8.0 | 16% low-molecular-weight PEG smears, 0.1 M HEPES, pH 7.5, 5% ethylene glycol, 0.1 M KCl | 17% PEG 3350, 0.1 M potassium citrate |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaikuad, A.; Pollinger, J.; Rühl, M.; Ni, X.; Kilu, W.; Heering, J.; Merk, D. Comprehensive Set of Tertiary Complex Structures and Palmitic Acid Binding Provide Molecular Insights into Ligand Design for RXR Isoforms. Int. J. Mol. Sci. 2020, 21, 8457. https://doi.org/10.3390/ijms21228457
Chaikuad A, Pollinger J, Rühl M, Ni X, Kilu W, Heering J, Merk D. Comprehensive Set of Tertiary Complex Structures and Palmitic Acid Binding Provide Molecular Insights into Ligand Design for RXR Isoforms. International Journal of Molecular Sciences. 2020; 21(22):8457. https://doi.org/10.3390/ijms21228457
Chicago/Turabian StyleChaikuad, Apirat, Julius Pollinger, Michael Rühl, Xiaomin Ni, Whitney Kilu, Jan Heering, and Daniel Merk. 2020. "Comprehensive Set of Tertiary Complex Structures and Palmitic Acid Binding Provide Molecular Insights into Ligand Design for RXR Isoforms" International Journal of Molecular Sciences 21, no. 22: 8457. https://doi.org/10.3390/ijms21228457
APA StyleChaikuad, A., Pollinger, J., Rühl, M., Ni, X., Kilu, W., Heering, J., & Merk, D. (2020). Comprehensive Set of Tertiary Complex Structures and Palmitic Acid Binding Provide Molecular Insights into Ligand Design for RXR Isoforms. International Journal of Molecular Sciences, 21(22), 8457. https://doi.org/10.3390/ijms21228457






