Ligand-Specific Factors Influencing GLP-1 Receptor Post-Endocytic Trafficking and Degradation in Pancreatic Beta Cells
Abstract
1. Introduction
2. Results
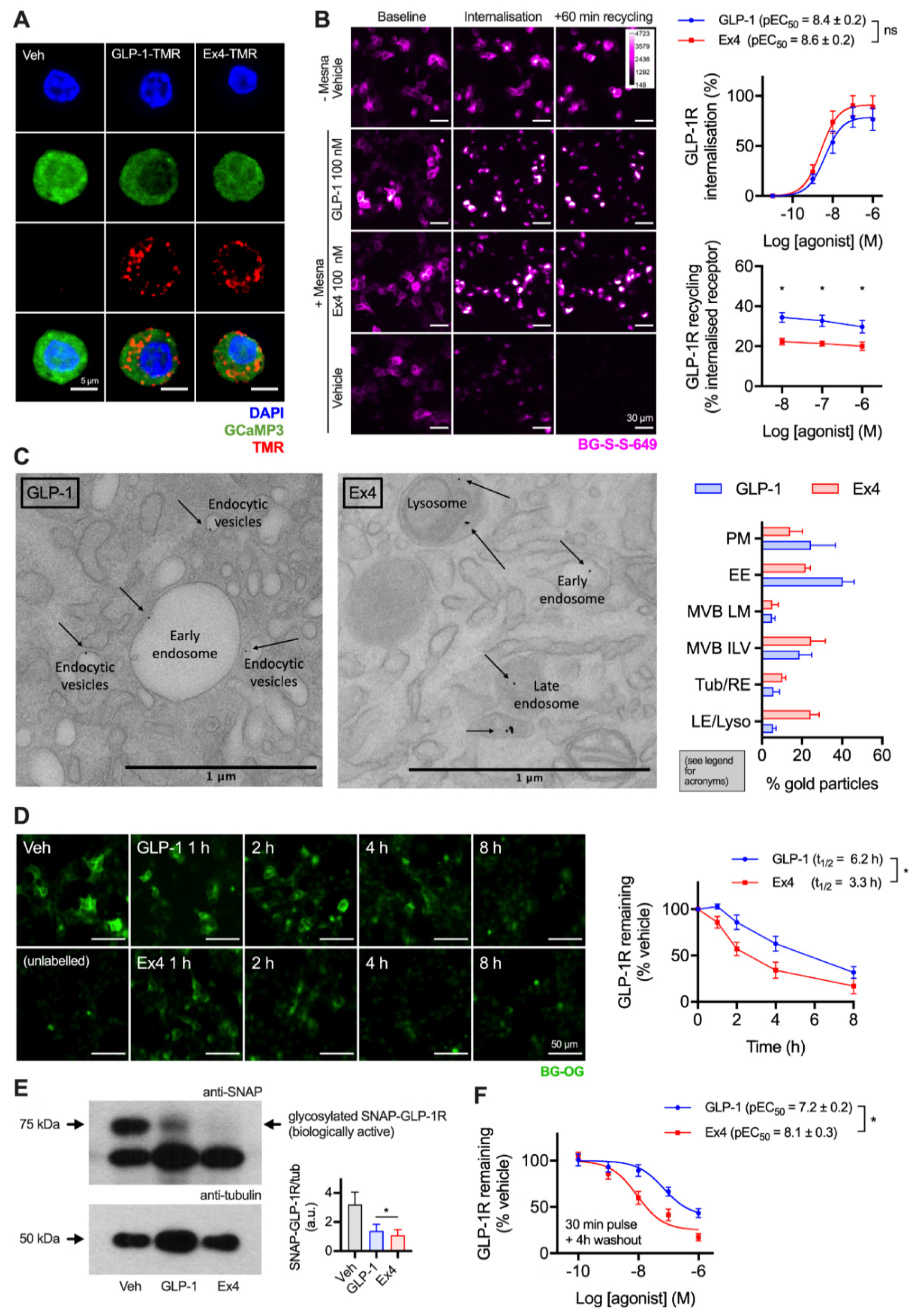
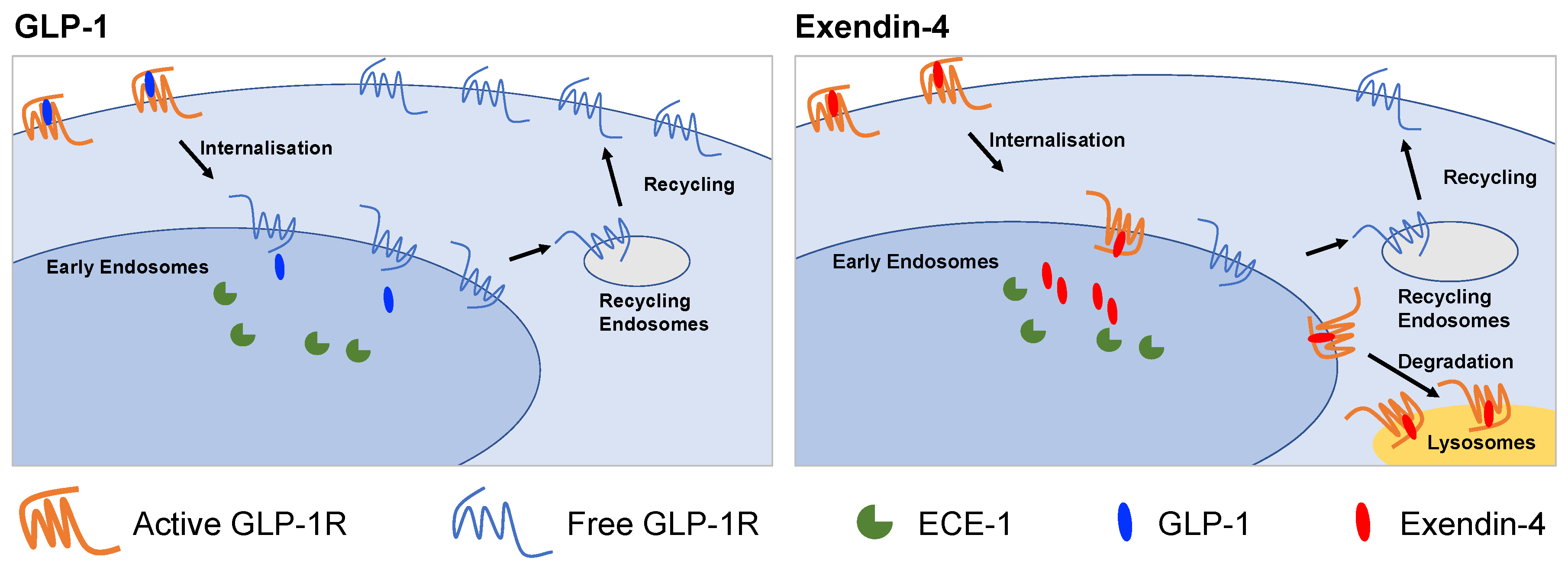
2.1. GLP-1R Degradation in Beta Cells is More Prominent after Treatment with Exendin-4 versus GLP-1
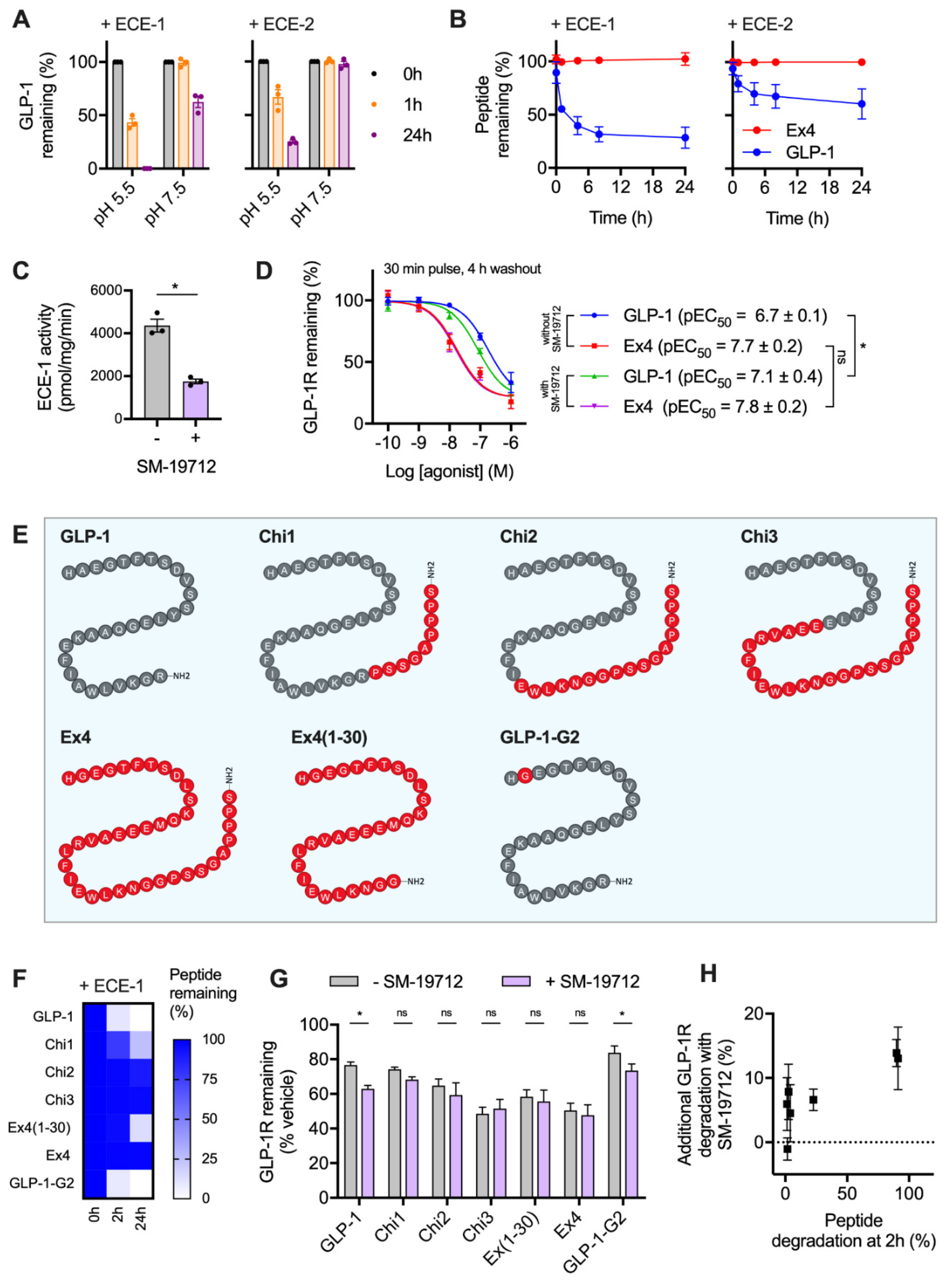
2.2. GLP-1R Degradation Rates are Associated with Ligand Sensitivity to ECE-1
2.3. Peptide Agonist N-Terminal Substitutions Influence GLP-1R Degradation
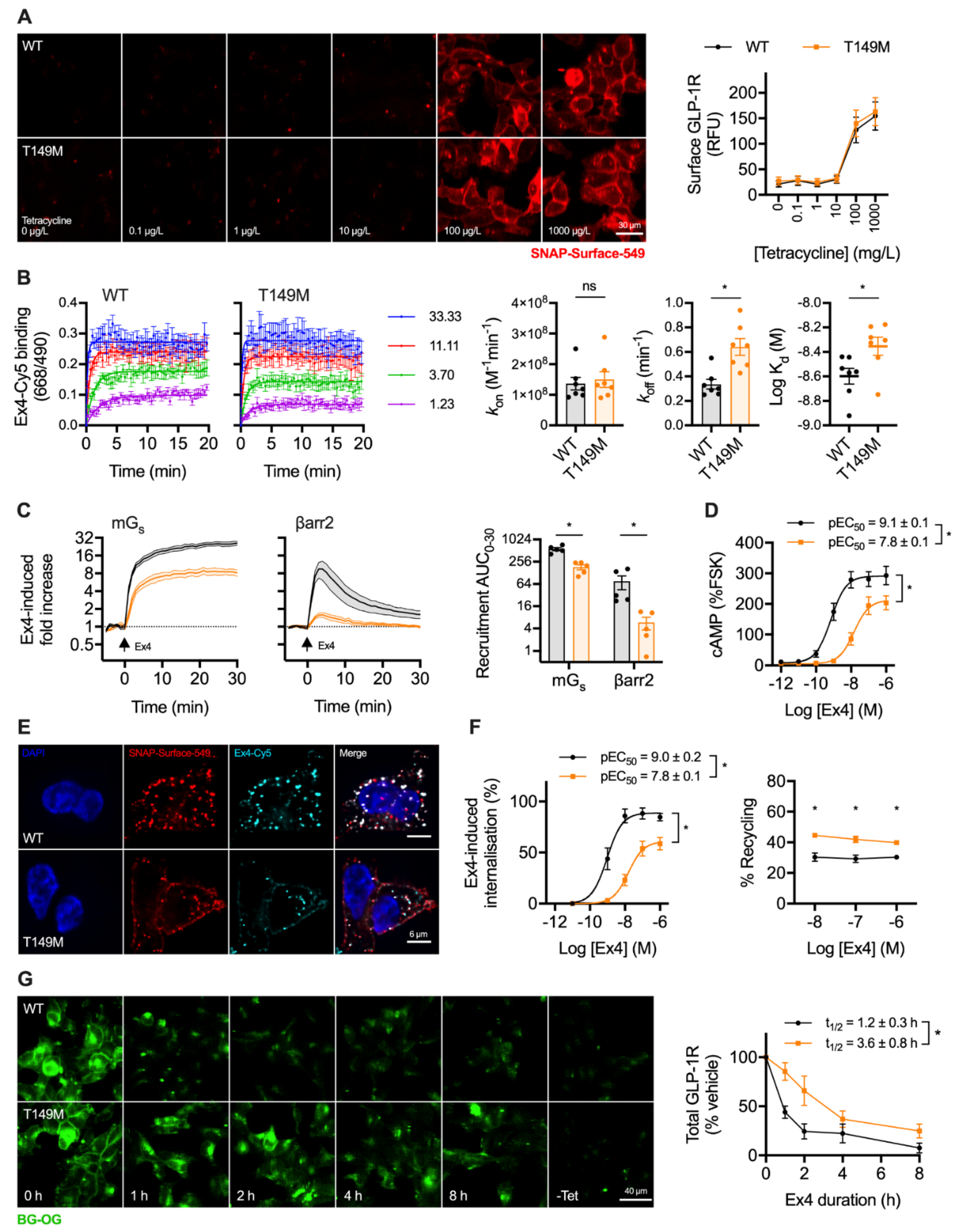
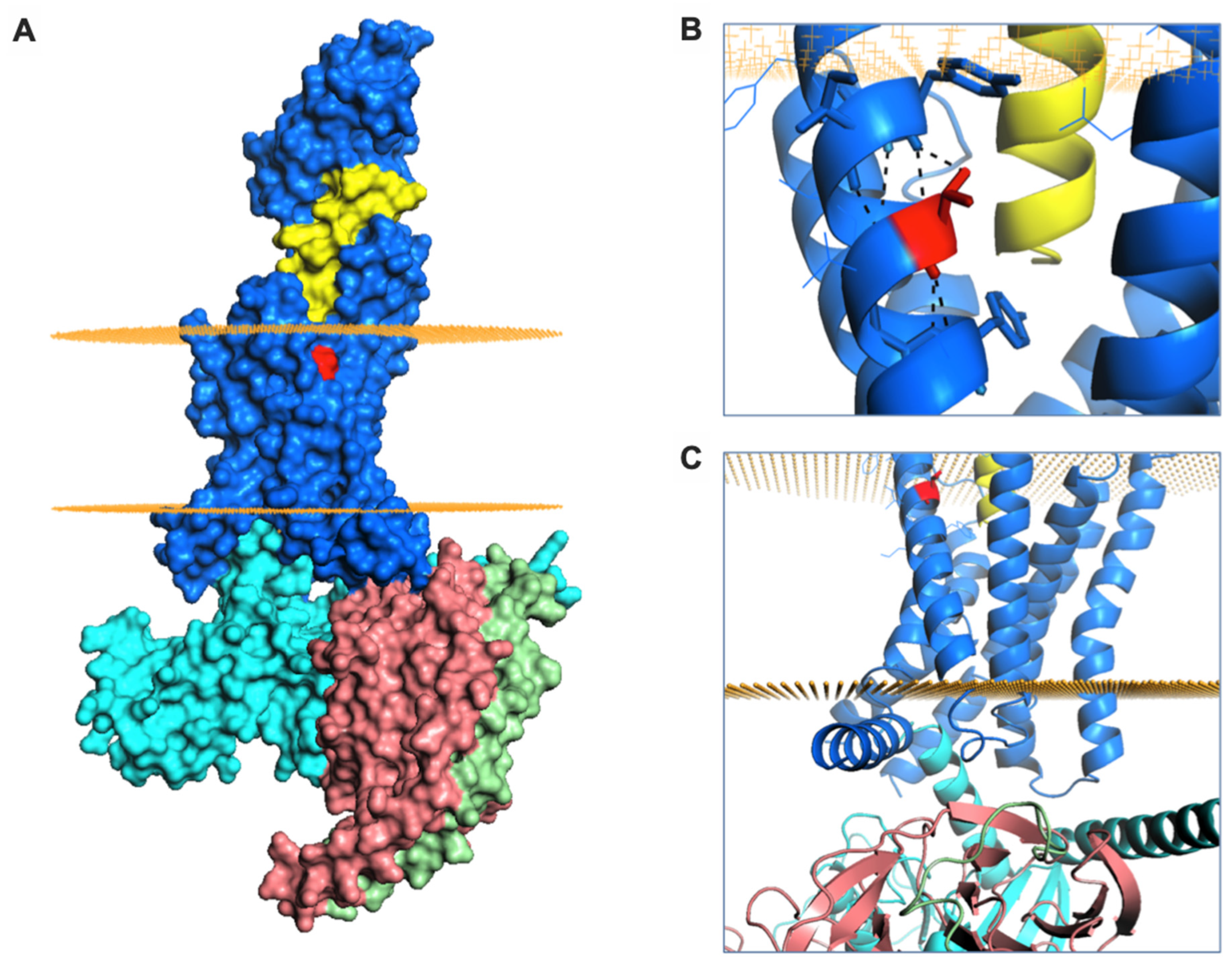
2.4. A Natural GLP-1R Variant Shows Altered Trafficking Response
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Plasmids
4.3. Cell Culture
4.4. Fluorescence Imaging of Fixed Samples
4.5. Electron Microscopy
4.6. Fluorescent GLP-1R Ligand Uptake Assay in Primary Dispersed Pancreatic Islets
4.7. High Content Imaging Internalisation and Recycling Assay
4.8. SNAP-GLP-1R Degradation Assay by Microscopy
4.9. SNAP-GLP-1R Degradation Assay by Immunoblotting
4.10. Measurement of Binding Affinities
4.11. NanoBiT Assays
4.12. Cyclic AMP Assay
4.13. Measurement of ECE-1 Activity
4.14. Measurement of Peptide Stability by HPLC
4.15. Structural Analysis
4.16. Data Analysis and Statistics
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- De Graaf, C.; Donnelly, D.; Wootten, D.; Lau, J.; Sexton, P.M.; Miller, L.J.; Ahn, J.-M.; Liao, J.; Fletcher, M.M.; Yang, D.; et al. Glucagon-Like Peptide-1 and Its Class B G Protein–Coupled Receptors: A Long March to Therapeutic Successes. Pharmacol. Rev. 2016, 68, 954–1013. [Google Scholar] [CrossRef] [PubMed]
- Andersen, A.; Lund, A.; Knop, F.K.; Vilsbøll, T. Glucagon-like peptide 1 in health and disease. Nat. Rev. Endocrinol. 2018, 14, 390–403. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, M.J.; Gaunt, P.; Aithal, G.P.; Barton, D.; Hull, D.; Parker, R.; Hazlehurst, J.M.; Guo, K.; Aldersley, M.; Abouda, G.; et al. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): A multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet 2016, 13, 679–690. [Google Scholar] [CrossRef]
- Grieco, M.; Giorgi, A.; Gentile, M.C.; D’Erme, M.; Morano, S.; Maras, B.; Filardi, T. Glucagon-Like Peptide-1: A Focus on Neurodegenerative Diseases. Front. Neurosci. 2019, 13, 1112. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Sturchler, E.; Zhu, J.; Nieto, A.; Cistrone, P.A.; Xie, J.; Rachel, T.; Yea, K.; Jones, T.; Turn, R.; et al. Autocrine selection of a GLP-1R G-protein biased agonist with potent antidiabetic effects. Nat. Commun. 2015, 6, 8918. [Google Scholar] [CrossRef] [PubMed]
- Jones, B.; Buenaventura, T.; Kanda, N.; Chabosseau, P.; Owen, B.M.; Scott, R.; Goldin, R.; Angkathunyakul, N.; Ivan, R.C.J.; Bosco, D.; et al. Targeting GLP-1 receptor trafficking to improve agonist efficacy. Nat. Commun. 2018, 9, 1–17. [Google Scholar] [CrossRef]
- Lucey, M.; Pickford, P.; Bitsi, S.; Minnion, J.; Ungewiss, J.; Schoeneberg, K.; Rutter, G.A.; Bloom, S.R.; Tomas, A.; Jones, B.J. Disconnect between signalling potency and in vivo efficacy of pharmacokinetically optimised biased glucagon-like peptide-1 receptor agonists. Mol. Metab. 2020, 37, 100991. [Google Scholar] [CrossRef]
- Wang, M.; Yao, P.; Gao, M.; Jin, J.; Yu, Y. Novel fatty chain-modified GLP-1R G-protein biased agonist exerts prolonged anti-diabetic effects through targeting receptor binding sites. RSC Adv. 2020, 10, 8044–8053. [Google Scholar] [CrossRef]
- Girada, S.B.; Kuna, R.S.; Bele, S.; Zhu, Z.; Chakravarthi, N.; DiMarchi, R.D.; Mitra, P. Gαs regulates Glucagon-Like Peptide 1 Receptor-mediated cyclic AMP generation at Rab5 endosomal compartment. Mol. Metab. 2017, 6, 1173–1185. [Google Scholar] [CrossRef]
- Buenaventura, T.; Kanda, N.; Douzenis, P.C.; Jones, B.; Bloom, S.R.; Chabosseau, P.; Corrêa, I.R.; Bosco, D.; Piemonti, L.; Marchetti, P.; et al. A Targeted RNAi Screen Identifies Endocytic Trafficking Factors That Control GLP-1 Receptor Signaling in Pancreatic β-Cells. Diabetes 2017, 67, 385–399. [Google Scholar] [CrossRef] [PubMed]
- Fang, Z.; Chen, S.; Pickford, P.; Broichhagen, J.; Hodson, D.J.; Corrêa, I.R.; Kumar, S.; Görlitz, F.; Dunsby, C.; French, P.M.W.; et al. The Influence of Peptide Context on Signaling and Trafficking of Glucagon-like Peptide-1 Receptor Biased Agonists. ACS Pharmacol. Transl. Sci. 2020, 3, 345–360. [Google Scholar] [CrossRef]
- Pickford, P.; Lucey, M.; Fang, Z.; Bitsi, S.; De La Serna, J.B.; Broichhagen, J.; Hodson, D.J.; Minnion, J.; Rutter, G.A.; Bloom, S.R.; et al. Signalling, trafficking and glucoregulatory properties of glucagon-like peptide-1 receptor agonists exendin-4 and lixisenatide. Br. J. Pharmacol. 2020, 177, 3905–3923. [Google Scholar] [CrossRef]
- Eng, J.; Kleinman, W.A.; Singh, L.; Singh, G.; Raufman, J.P. Isolation and characterization of exendin-4, an exendin-3 analogue, from Heloderma suspectum venom. Further evidence for an exendin receptor on dispersed acini from guinea pig pancreas. J. Biol. Chem. 1992, 267, 7402–7405. [Google Scholar]
- Jorgensen, R.; Kubale, V.; Vrecl, M.; Schwartz, T.W.; Elling, C.E. Oxyntomodulin differentially affects glucagon-like peptide-1 receptor beta-arrestin recruitment and signaling through Galpha(s). J. Pharmacol. Exp. Ther. 2007, 322, 148–154. [Google Scholar] [CrossRef]
- Roed, S.N.; Wismann, P.; Underwood, C.R.; Kulahin, N.; Iversen, H.; Cappelen, K.A.; Schäffer, L.; Lehtonen, J.; Hecksher-Soerensen, J.; Secher, A.; et al. Real-time trafficking and signaling of the glucagon-like peptide-1 receptor. Mol. Cell. Endocrinol. 2014, 382, 938–949. [Google Scholar] [CrossRef]
- Fletcher, M.M.; Halls, M.L.; Zhao, P.; Clydesdale, L.; Christopoulos, A.; Sexton, P.M.; Wootten, D. Glucagon-like peptide-1 receptor internalisation controls spatiotemporal signalling mediated by biased agonists. Biochem. Pharmacol. 2018, 156, 406–419. [Google Scholar] [CrossRef]
- Baggio, L.L.; Kim, J.-G.; Drucker, D.J. Chronic exposure to GLP-1R agonists promotes homologous GLP-1 receptor desensitization in vitro but does not attenuate GLP-1R-dependent glucose homeostasis in vivo. Diabetes 2004, 53, S205–S214. [Google Scholar] [CrossRef]
- Lu, J.; Willars, G.B. Endothelin-converting enzyme-1 regulates glucagon-like peptide-1 receptor signalling and resensitisation. Biochem. J. 2019, 476, 513–533. [Google Scholar] [CrossRef]
- Tobin, A.B.; Butcher, A.J.; Kong, K.C. Location, location, location…site-specific GPCR phosphorylation offers a mechanism for cell-type-specific signalling. Trends Pharmacol. Sci. 2008, 29, 413–420. [Google Scholar] [CrossRef]
- Tokuyama, Y.; Matsui, K.; Egashira, T.; Nozaki, O.; Ishizuka, T.; Kanatsuka, A. Five missense mutations in glucagon-like peptide 1 receptor gene in Japanese population. Diabetes Res. Clin. Pr. 2004, 66, 63–69. [Google Scholar] [CrossRef]
- Koole, C.; Wootten, D.; Simms, J.; Valant, C.; Miller, L.J.; Christopoulos, A.; Sexton, P.M. Polymorphism and ligand dependent changes in human glucagon-like peptide-1 receptor (GLP-1R) function: Allosteric rescue of loss of function mutation. Mol. Pharmacol. 2011, 80, 486–497. [Google Scholar] [CrossRef] [PubMed]
- Shaaban, G.; Oriowo, M.; Al-Sabah, S. Rate of Homologous Desensitization and Internalization of the GLP-1 Receptor. Molecules 2016, 22, 22. [Google Scholar] [CrossRef] [PubMed]
- Richards, P.; Parker, H.E.; Adriaenssens, A.E.; Hodgson, J.M.; Cork, S.C.; Trapp, S.; Gribble, F.M.; Reimann, F. Identification and Characterization of GLP-1 Receptor-Expressing Cells Using a New Transgenic Mouse Model. Diabetes 2013, 63, 1224–1233. [Google Scholar] [CrossRef]
- Buenaventura, T.; Bitsi, S.; Laughlin, W.E.; Burgoyne, T.; Lyu, Z.; Oqua, A.I.; Norman, H.; McGlone, E.R.; Klymchenko, A.S.; Ivan, R.C.J.; et al. Agonist-induced membrane nanodomain clustering drives GLP-1 receptor responses in pancreatic beta cells. PLoS Biol. 2019, 17, e3000097. [Google Scholar] [CrossRef] [PubMed]
- Poc, P.; Gutzeit, V.A.; Ast, J.; Lee, J.; Jones, B.J.; D’Este, E.; Mathes, B.; Lehmann, M.; Hodson, D.J.; Levitz, J.; et al. Interrogating surface versus intracellular transmembrane receptor populations using cell-impermeable SNAP-tag substrates. Chem. Sci. 2020, 11, 7871–7883. [Google Scholar] [CrossRef]
- Roosterman, D.; Kempkes, C.; Cottrell, G.S.; Padilla, B.E.; Bunnett, N.W.; Turck, C.W.; Steinhoff, M. Endothelin-Converting Enzyme-1 Degrades Internalized Somatostatin-14. Endocrinology 2008, 149, 2200–2207. [Google Scholar] [CrossRef][Green Version]
- Zhao, P.; Canals, M.; Murphy, J.E.; Klingler, D.; Eriksson, E.M.; Pelayo, J.-C.; Hardt, M.; Bunnett, N.W.; Poole, D.P. Agonist-biased Trafficking of Somatostatin Receptor 2A in Enteric Neurons. J. Biol. Chem. 2013, 288, 25689–25700. [Google Scholar] [CrossRef]
- Hupe-Sodmann, K.; McGregor, G.P.; Bridenbaugh, R.; Göke, R.; Göke, B.; Thole, H.; Zimmermann, B.; Voigt, K. Characterisation of the processing by human neutral endopeptidase 24.11 of GLP-1(7–36) amide and comparison of the substrate specificity of the enzyme for other glucagon-like peptides. Regul. Pept. 1995, 58, 149–156. [Google Scholar] [CrossRef]
- Umekawa, K.; Hasegawa, H.; Tsutsumi, Y.; Sato, K.; Matsumura, Y.; Ohashi, N. Pharmacological Characterization of a Novel Sulfonylureid-Pyrazole Derivative, SM-19712, a Potent Nonpeptidic Inhibitor of Endothelin Converting Enzyme. Jpn. J. Pharmacol. 2000, 84, 7–15. [Google Scholar] [CrossRef]
- Lee, J.G.; Ryu, J.H.; Kim, S.-M.; Park, M.-Y.; Kim, S.-H.; Shin, Y.G.; Sohn, J.-W.; Kim, H.H.; Park, Z.-Y.; Seong, J.Y.; et al. Replacement of the C-terminal Trp-cage of exendin-4 with a fatty acid improves therapeutic utility. Biochem. Pharmacol. 2018, 151, 59–68. [Google Scholar] [CrossRef]
- Jones, B.J.; Scopelliti, R.; Tomas, A.; Bloom, S.R.; Hodson, D.J.; Broichhagen, J. Potent Prearranged Positive Allosteric Modulators of the Glucagon-like Peptide-1 Receptor. ChemistryOpen 2017, 6, 501–505. [Google Scholar] [CrossRef]
- Dixon, A.S.; Schwinn, M.K.; Hall, M.P.; Zimmerman, K.; Otto, P.; Lubben, T.H.; Butler, B.L.; Binkowski, B.F.; Machleidt, T.; Kirkland, T.A.; et al. NanoLuc Complementation Reporter Optimized for Accurate Measurement of Protein Interactions in Cells. ACS Chem. Biol. 2016, 11, 400–408. [Google Scholar] [CrossRef]
- Beinborn, M.; Worrall, C.I.; McBride, E.W.; Kopin, A.S. A human glucagon-like peptide-1 receptor polymorphism results in reduced agonist responsiveness. Regul. Pept. 2005, 130, 1–6. [Google Scholar] [CrossRef]
- Thompson, A.; Kanamarlapudi, V. Agonist-induced internalisation of the glucagon-like peptide-1 receptor is mediated by the Gαq pathway. Biochem. Pharmacol. 2015, 93, 72–84. [Google Scholar] [CrossRef]
- Ast, J.; Arvaniti, A.; Fine, N.H.F.; Nasteska, D.; Ashford, F.B.; Stamataki, Z.; Koszegi, Z.; Bacon, A.; Jones, B.J.; Lucey, M.A.; et al. Super-resolution microscopy compatible fluorescent probes reveal endogenous glucagon-like peptide-1 receptor distribution and dynamics. Nat. Commun. 2020, 24, 467. [Google Scholar] [CrossRef]
- Wan, Q.; Okashah, N.; Inoue, A.; Nehmé, R.; Carpenter, B.; Tate, C.G.; Lambert, N.A. Mini G protein probes for active G protein–coupled receptors (GPCRs) in live cells. J. Biol. Chem. 2018, 293, 7466–7473. [Google Scholar] [CrossRef]
- Liang, Y.-L.; Khoshouei, M.; Glukhova, A.; Furness, S.G.B.; Zhao, P.; Clydesdale, L.; Koole, C.; Truong, T.T.; Thal, D.M.; Lei, S.; et al. Phase-plate cryo-EM structure of a biased agonist-bound human GLP-1 receptor–Gs complex. Nat. Cell Biol. 2018, 555, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sun, B.; Feng, D.; Hu, H.; Chu, M.; Qu, Q.; Tarrasch, J.T.; Li, S.; Kobilka, T.S.; Kobilka, B.K.; et al. Cryo-EM structure of the activated GLP-1 receptor in complex with a G protein. Nat. Cell Biol. 2017, 546, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, J.A.; Deupi, X.; Olivella, M.; Haaksma, E.E.; Pardo, L. Serine and threonine residues bend alpha-helices in the chi(1) = g(-) conformation. Biophys. J. 2000, 79, 2754–2760. [Google Scholar] [CrossRef]
- Lee, Y.; Warne, T.; Nehmé, R.; Pandey, S.; Dwivedi-Agnihotri, H.; Chaturvedi, M.; Edwards, P.C.; García-Nafría, J.; Leslie, A.G.W.; Shukla, A.K.; et al. Molecular basis of β-arrestin coupling to formoterol-bound β1-adrenoceptor. Nat. Cell Biol. 2020, 583, 862–866. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.; Im, W. Revisiting Hydrophobic Mismatch with Free Energy Simulation Studies of Transmembrane Helix Tilt and Rotation. Biophys. J. 2010, 99, 175–183. [Google Scholar] [CrossRef]
- Heydenreich, F.M.; Vuckovic, Z.; Matkovic, M.; Veprintsev, D.B. Stabilization of G protein-coupled receptors by point mutations. Front. Pharmacol. 2015, 6, 82. [Google Scholar] [CrossRef]
- Vilardaga, J.-P.; Jean-Alphonse, F.G.; Gardella, T.J. Endosomal generation of cAMP in GPCR signaling. Nat. Chem. Biol. 2014, 10, 700–706. [Google Scholar] [CrossRef]
- Kuna, R.S.; Girada, S.B.; Asalla, S.; Vallentyne, J.; Maddika, S.; Patterson, J.T.; Smiley, D.L.; DiMarchi, R.D.; Mitra, P. Glucagon-like peptide-1 receptor-mediated endosomal cAMP generation promotes glucose-stimulated insulin secretion in pancreatic β-cells. Am. J. Physiol. Metab. 2013, 305, E161–E170. [Google Scholar] [CrossRef]
- Fremaux, J.; Venin, C.; Mauran, L.; Zimmer, R.; Koensgen, F.; Rognan, D.; Bitsi, S.; Lucey, M.A.; Jones, B.; Tomas, A.; et al. Ureidopeptide GLP-1 analogues with prolonged activity in vivo via signal bias and altered receptor trafficking. Chem. Sci. 2019, 10, 9872–9879. [Google Scholar] [CrossRef]
- Gupta, A.; Fujita, W.; Gomes, I.; Bobeck, E.N.; Devi, L.A. Endothelin-converting enzyme 2 differentially regulates opioid receptor activity. Br. J. Pharmacol. 2014, 172, 704–719. [Google Scholar] [CrossRef]
- Jeng, A.Y.; de Lombaert, S.; Beil, M.E.; Bruseo, C.W.; Savage, P.; Chou, M.; Trapani, A.J. Design and synthesis of a potent and selective endothelin-converting enzyme inhibitor, CGS 35066. J. Cardiovasc. Pharmacol. 2000, 36 (Suppl. 1), S36–S39. [Google Scholar] [CrossRef]
- Schmid, C.L.; Kennedy, N.M.; Ross, N.C.; Lovell, K.M.; Yue, Z.; Morgenweck, J.; Cameron, M.D.; Bannister, T.D.; Bohn, L.M. Bias Factor and Therapeutic Window Correlate to Predict Safer Opioid Analgesics. Cell 2017, 171, 1165–1175. [Google Scholar] [CrossRef] [PubMed]
- Kenakin, T. Is the Quest for Signaling Bias Worth the Effort? Mol. Pharmacol. 2018, 93, 266–269. [Google Scholar] [CrossRef]
- Borden, L.A.; Einstein, R.; Gabel, C.A.; Maxfield, F.R. Acidification-dependent dissociation of endocytosed insulin precedes that of endocytosed proteins bearing the mannose 6-phosphate recognition marker. J. Biol. Chem. 1990, 265, 8497–8504. [Google Scholar]
- Tomas, A.; Vaughan, S.O.; Burgoyne, T.; Sorkin, A.; Hartley, J.A.; Hochhauser, D.; Futter, C.E. WASH and Tsg101/ALIX-dependent diversion of stress-internalized EGFR from the canonical endocytic pathway. Nat. Commun. 2015, 12, 7324. [Google Scholar] [CrossRef]
- Godbole, A.; Lyga, S.; Lohse, M.J.; Calebiro, D. Internalized TSH receptors en route to the TGN induce local Gs-protein signaling and gene transcription. Nat. Commun. 2017, 5, 443. [Google Scholar] [CrossRef]
- Jong, Y.-J.I.; Harmon, S.K.; O’Malley, K.L. GPCR signalling from within the cell. Br. J. Pharmacol. 2017, 175, 4026–4035. [Google Scholar] [CrossRef]
- Hauser, A.S.; Chavali, S.; Masuho, I.; Jahn, L.J.; Martemyanov, K.A.; Gloriam, D.E.; Babu, M.M. Pharmacogenomics of GPCR Drug Targets. Cell 2018, 172, 41–54. [Google Scholar] [CrossRef]
- Koole, C.; Wootten, D.; Simms, J.; Miller, L.J.; Christopoulos, A.; Sexton, P.M. Differential Impact of Amino Acid Substitutions on Critical Residues of the Human Glucagon-Like Peptide-1 Receptor Involved in Peptide Activity and Small-Molecule Allostery. J. Pharmacol. Exp. Ther. 2015, 353, 52–63. [Google Scholar] [CrossRef]
- Hennen, S.; Kodra, J.T.; Soroka, V.; Krogh, B.O.; Wu, X.; Kaastrup, P.; Ørskov, C.; Rønn, S.G.; Schluckebier, G.; Barbateskovic, S.; et al. Structural insight into antibody-mediated antagonism of the Glucagon-like peptide-1 Receptor. Sci. Rep. 2016, 6, 26236. [Google Scholar] [CrossRef]
- Hodson, D.J.; Mitchell, R.K.; Bellomo, E.A.; Sun, G.; Vinet, L.; Meda, P.; Li, D.; Li, W.-H.; Bugliani, M.; Marchetti, P.; et al. Lipotoxicity disrupts incretin-regulated human β cell connectivity. J. Clin. Investig. 2013, 123, 4182–4194. [Google Scholar] [CrossRef]
- Salem, V.; Silva, L.D.; Suba, K.; Georgiadou, E.; Gharavy, S.N.M.; Akhtar, N.; Martin-Alonso, A.; Gaboriau, D.C.A.; Rothery, S.M.; Stylianides, T.; et al. Leader β-cells coordinate Ca2+ dynamics across pancreatic islets in vivo. Nat. Metab. 2019, 1, 615–629. [Google Scholar] [CrossRef]
- gnomAD. Available online: https://gnomad.broadinstitute.org (accessed on 12 January 2020).
- Naylor, J.; Suckow, A.T.; Seth, A.; Baker, D.J.; Sermadiras, I.; Ravn, P.; Howes, R.; Li, J.; Snaith, M.R.; Coghlan, M.P.; et al. Use of CRISPR/Cas9-engineered INS-1 pancreatic β cells to define the pharmacology of dual GIPR/GLP-1R agonists. Biochem. J. 2016, 473, 2881–2891. [Google Scholar] [CrossRef]
- Lilla, V.; Webb, G.; Rickenbach, K.; Maturana, A.; Steiner, N.F.; Halban, P.A.; Irminger, J.-C. Differential Gene Expression in Well-Regulated and Dysregulated Pancreatic β-Cell (MIN6) Sublines. Endocrinology 2003, 144, 1368–1379. [Google Scholar] [CrossRef]
- High Content Analysis. Available online: http://www.imperial.ac.uk/a-z-research/photonics/research/biophotonics/instruments--software/high-content-analysis/ (accessed on 12 February 2020).
- Edelstein, A.D.; Amodaj, N.; Hoover, K.H.; Vale, R.D.; Stuurman, N. Computer Control of Microscopes Using µManager. Curr. Protoc. Mol. Biol. 2010, 92, 14.20.1–14.20.17. [Google Scholar] [CrossRef]
- Sage, D.; Donati, L.; Soulez, F.; Fortun, D.; Schmit, G.; Seitz, A.; Guiet, R.; Vonesch, C.; Unser, M. DeconvolutionLab2: An open-source software for deconvolution microscopy. Methods 2017, 115, 28–41. [Google Scholar] [CrossRef]
- Peng, T.; Thorn, K.; Schroeder, T.; Wang, L.; Theis, F.J.; Marr, C.; Navab, N. A BaSiC tool for background and shading correction of optical microscopy images. Nat. Commun. 2017, 8, 14836. [Google Scholar] [CrossRef]
- Jaccard, N.; Griffin, L.D.; Keser, A.; Macown, R.J.; Super, A.; Veraitch, F.S.; Szita, N. Automated method for the rapid and precise estimation of adherent cell culture characteristics from phase contrast microscopy images. Biotechnol. Bioeng. 2013, 111, 504–517. [Google Scholar] [CrossRef]
- Widmann, C.; Dolci, W.; Thorens, B. Agonist-induced internalization and recycling of the glucagon-like peptide-1 receptor in transfected fibroblasts and in insulinomas. Biochem. J. 1995, 310, 203–214. [Google Scholar] [CrossRef]
- Liang, Y.-L.; Khoshouei, M.; Deganutti, G.; Glukhova, A.; Koole, C.; Peat, T.S.; Radjainia, M.; Plitzko, J.M.; Baumeister, W.; Miller, L.J.; et al. Cryo-EM structure of the active, Gs-protein complexed, human CGRP receptor. Nat. Cell Biol. 2018, 561, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Ittisoponpisan, S.; Islam, S.A.; Khanna, T.; Alhuzimi, E.; David, A.; Sternberg, M.J. Can Predicted Protein 3D Structures Provide Reliable Insights into whether Missense Variants Are Disease Associated? J. Mol. Biol. 2019, 431, 2197–2212. [Google Scholar] [CrossRef]
- Schymkowitz, J.; Borg, J.; Stricher, F.; Nys, R.; Rousseau, F.; Serrano, L. The FoldX web server: An online force field. Nucleic Acids Res. 2005, 33, W382–W388. [Google Scholar] [CrossRef]
- David, A.; Razali, R.B.M.; Wass, M.N.; Sternberg, M.J.E. Protein-protein interaction sites are hot spots for disease-associated nonsynonymous SNPs. Hum. Mutat. 2011, 33, 359–363. [Google Scholar] [CrossRef]
- Metsalu, T.; Vilo, J. ClustVis: A web tool for visualizing clustering of multivariate data using Principal Component Analysis and heatmap. Nucleic Acids Res. 2015, 43, W566–W570. [Google Scholar] [CrossRef] [PubMed]
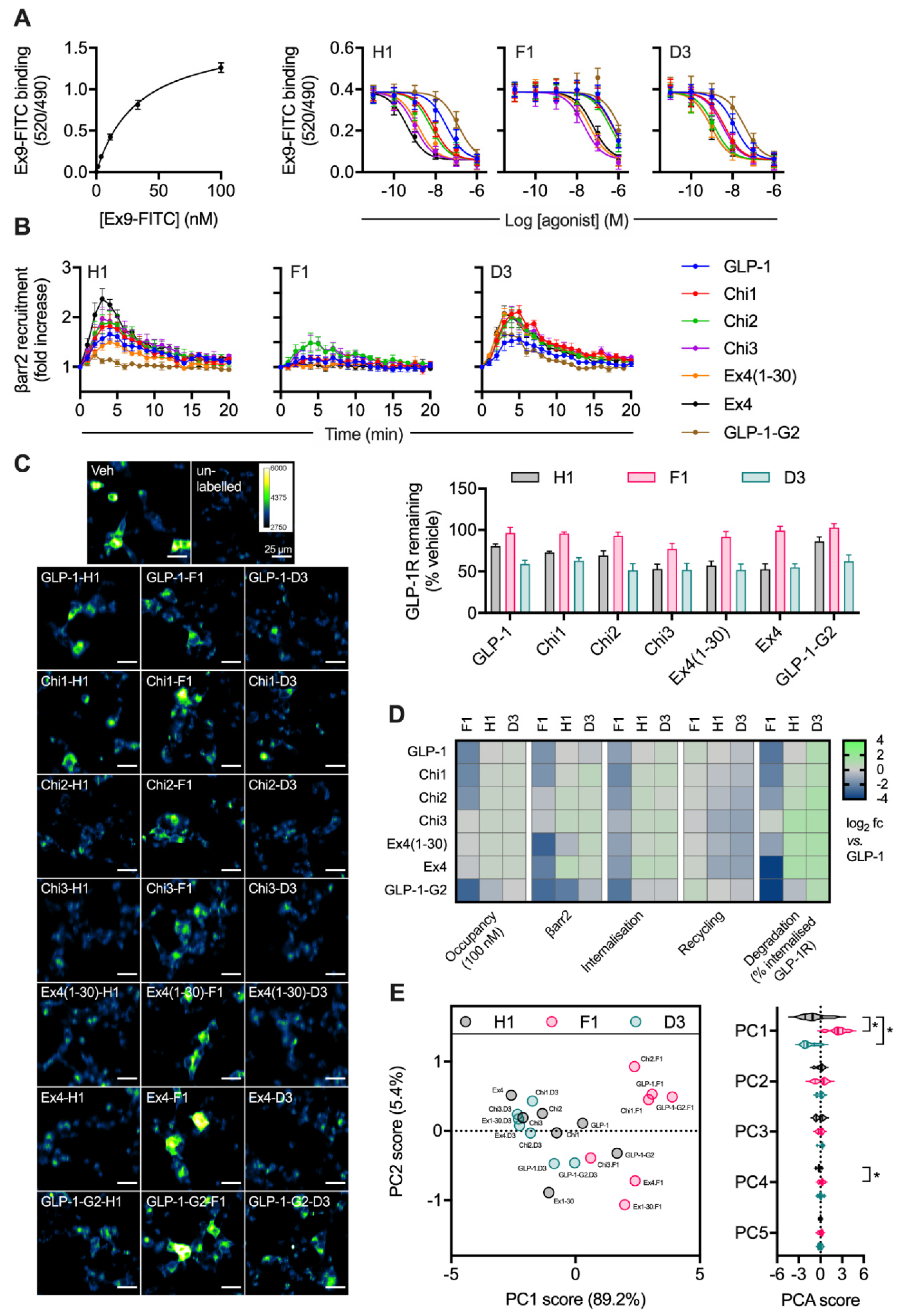
| Assay | N-Terminus | GLP-1 | Chi1 | Chi2 | Chi3 | Ex4(1-30) | Ex4 | GLP-1-G2 |
|---|---|---|---|---|---|---|---|---|
| Log Ki (M) | H1 | −7.6 ± 0.1 | −8.2 ± 0.1 | −8.4 ± 0.1 | −9.1 ± 0.0 | −9.0 ± 0.0 | −9.5 ± 0.1 | −7.0 ± 0.1 |
| F1 | −6.4 ± 0.1 | −6.4 ± 0.1 | −6.5 ± 0.0 | −7.8 ± 0.1 | −7.5 ± 0.0 | −7.3 ± 0.1 | −6.1 ± 0.0 | |
| D3 | −8.0 ± 0.1 | −8.5 ± 0.1 | −9.0 ± 0.1 | 8.6 ± 0.1 | −9.2 ± 0.1 | −9.2 ± 0.1 | −7.6 ± 0.1 | |
| β-arrestin-2 recruitment (AUC) | H1 | 6.2 ± 0.9 | 7.0 ± 1.6 | 8.4 ± 2.3 | 8.9 ± 2.1 | 4.0 ± 0.5 | 10.2 ± 1.6 | 1.3 ± 0.5 |
| F1 | 2.0 ± 1.3 | 2.0 ± 0.8 | 4.6 ± 1.6 | 4.8 ± 1.4 | 0.8 ± 0.2 | 1.3 ± 1 | 0.9 ± 0.5 | |
| D3 | 4.9 ± 0.9 | 9.4 ± 0.5 | 8.1 ± 1.2 | 8.9 ± 1.4 | 8.8 ± 0.3 | 8.3 ± 0.3 | 4.0 ± 0.7 | |
| Internalisation (%) | H1 | 69.6 ± 14.1 | 88.9 ± 12.7 | 97.3 ± 14.3 | 93 ± 13.7 | 84.5 ± 17.4 | 110.7 ± 9.9 | 58.9 ± 11.4 |
| F1 | 28.1 ± 11.1 | 18.5 ± 8.7 | 24.4 ± 9.3 | 40.6 ± 19.4 | 29.1 ± 11.3 | 25.5 ± 11.8 | 12.7 ± 7.8 | |
| D3 | 88.5 ± 16.9 | 97.7 ± 14.1 | 78.6 ± 15.7 | 101.5 ± 12.2 | 98 ± 12.3 | 105.4 ± 11.7 | 60.3 ± 17 | |
| Recycling (% internalised GLP-1R) | H1 | 44.1 ± 3.1 | 35.3 ± 2.8 | 30.9 ± 3.2 | 21.8 ± 3.8 | 26.4 ± 2.8 | 20.8 ± 3.6 | 44.4 ± 3.8 |
| F1 | 55.2 ± 3.4 | 48.7 ± 4.8 | 48.5 ± 5.2 | 40.7 ± 2.7 | 45.9 ± 3.6 | 49.9 ± 3.2 | 62.7 ± 17.9 | |
| D3 | 33.2 ± 4.1 | 28.6 ± 4.0 | 22.9 ± 5.1 | 19.0 ± 4.3 | 19.2 ± 3.2 | 19.0 ± 3.5 | 35.4 ± 5.6 | |
| Degradation (% remaining GLP-1R) | H1 | 80.4 ± 2.7 | 72.9 ± 1.6 | 69.1 ± 5.7 | 52.8 ± 5.9 | 57.0 ± 5.3 | 52.6 ± 6.6 | 86.2 ± 5.5 |
| F1 | 96.4 ± 6.7 | 95.6 ± 2 | 92.8 ± 4.5 | 77.1 ± 6.6 | 91.8 ± 6.4 | 99.3 ± 5.0 | 102.8 ± 4.7 | |
| D3 | 58.9 ± 4.4 | 62.9 ± 3.8 | 51.3 ± 8.1 | 51.8 ± 7.7 | 51.9 ± 7.1 | 55.0 ± 4.2 | 62.2 ± 7.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fang, Z.; Chen, S.; Manchanda, Y.; Bitsi, S.; Pickford, P.; David, A.; Shchepinova, M.M.; Corrêa Jr, I.R.; Hodson, D.J.; Broichhagen, J.; et al. Ligand-Specific Factors Influencing GLP-1 Receptor Post-Endocytic Trafficking and Degradation in Pancreatic Beta Cells. Int. J. Mol. Sci. 2020, 21, 8404. https://doi.org/10.3390/ijms21218404
Fang Z, Chen S, Manchanda Y, Bitsi S, Pickford P, David A, Shchepinova MM, Corrêa Jr IR, Hodson DJ, Broichhagen J, et al. Ligand-Specific Factors Influencing GLP-1 Receptor Post-Endocytic Trafficking and Degradation in Pancreatic Beta Cells. International Journal of Molecular Sciences. 2020; 21(21):8404. https://doi.org/10.3390/ijms21218404
Chicago/Turabian StyleFang, Zijian, Shiqian Chen, Yusman Manchanda, Stavroula Bitsi, Philip Pickford, Alessia David, Maria M. Shchepinova, Ivan R. Corrêa Jr, David J. Hodson, Johannes Broichhagen, and et al. 2020. "Ligand-Specific Factors Influencing GLP-1 Receptor Post-Endocytic Trafficking and Degradation in Pancreatic Beta Cells" International Journal of Molecular Sciences 21, no. 21: 8404. https://doi.org/10.3390/ijms21218404
APA StyleFang, Z., Chen, S., Manchanda, Y., Bitsi, S., Pickford, P., David, A., Shchepinova, M. M., Corrêa Jr, I. R., Hodson, D. J., Broichhagen, J., Tate, E. W., Reimann, F., Salem, V., Rutter, G. A., Tan, T., Bloom, S. R., Tomas, A., & Jones, B. (2020). Ligand-Specific Factors Influencing GLP-1 Receptor Post-Endocytic Trafficking and Degradation in Pancreatic Beta Cells. International Journal of Molecular Sciences, 21(21), 8404. https://doi.org/10.3390/ijms21218404






