Advanced Medical Therapies in the Management of Non-Scarring Alopecia: Areata and Androgenic Alopecia
Abstract
:1. Introduction
- (a)
- Contains or consists of cells or tissues that have been subject to substantial manipulation so that biological characteristics, physiological functions, or structural properties relevant for the intended clinical use have been altered, or of cells or tissues that are not intended to be used for the same essential functions in the recipient and the donor;
- (b)
- Is presented as having properties for, or is used in or administered to, human beings with a view of treating, preventing, or diagnosing a disease through the pharmacological, immunological, or metabolic action of its cells or tissues.
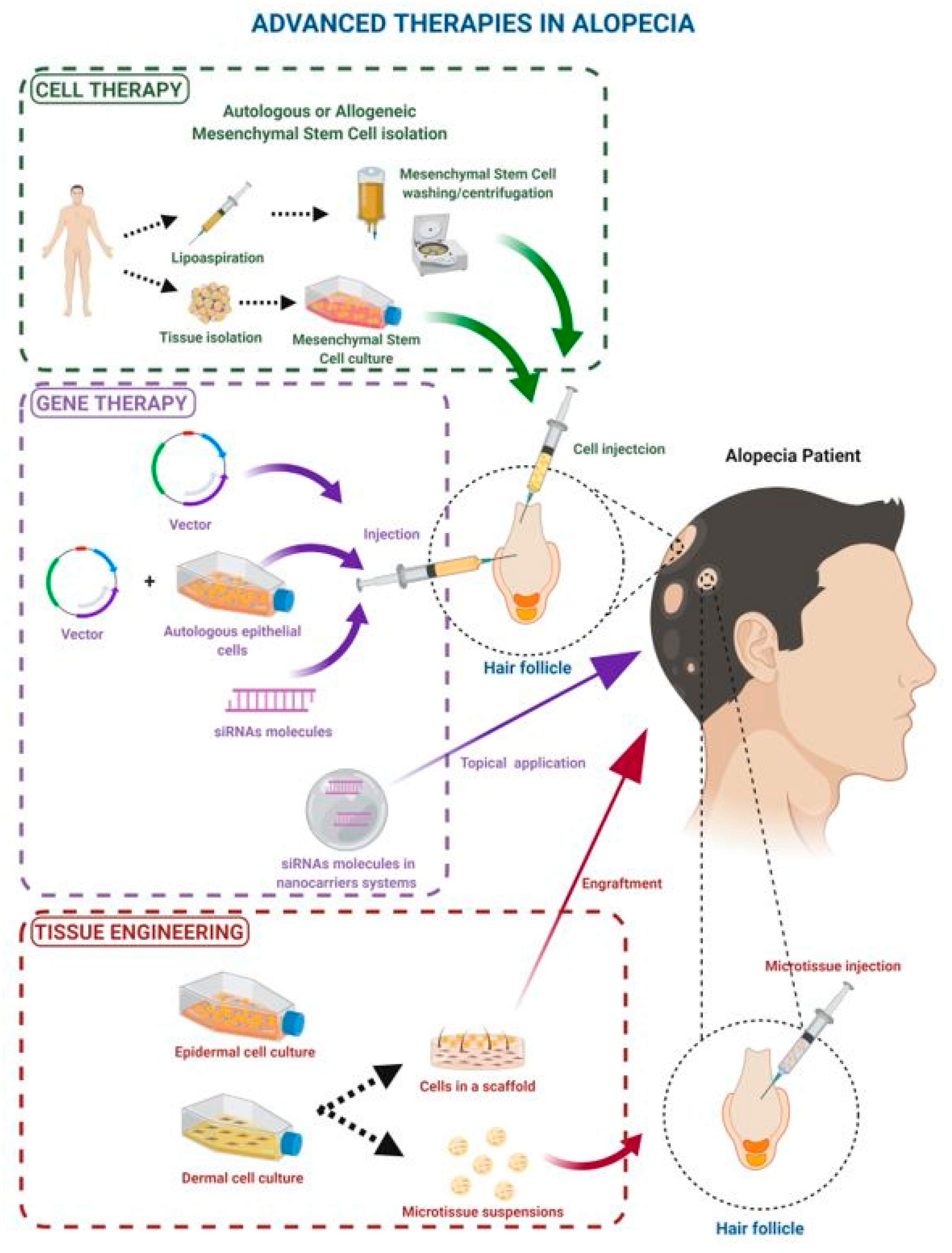
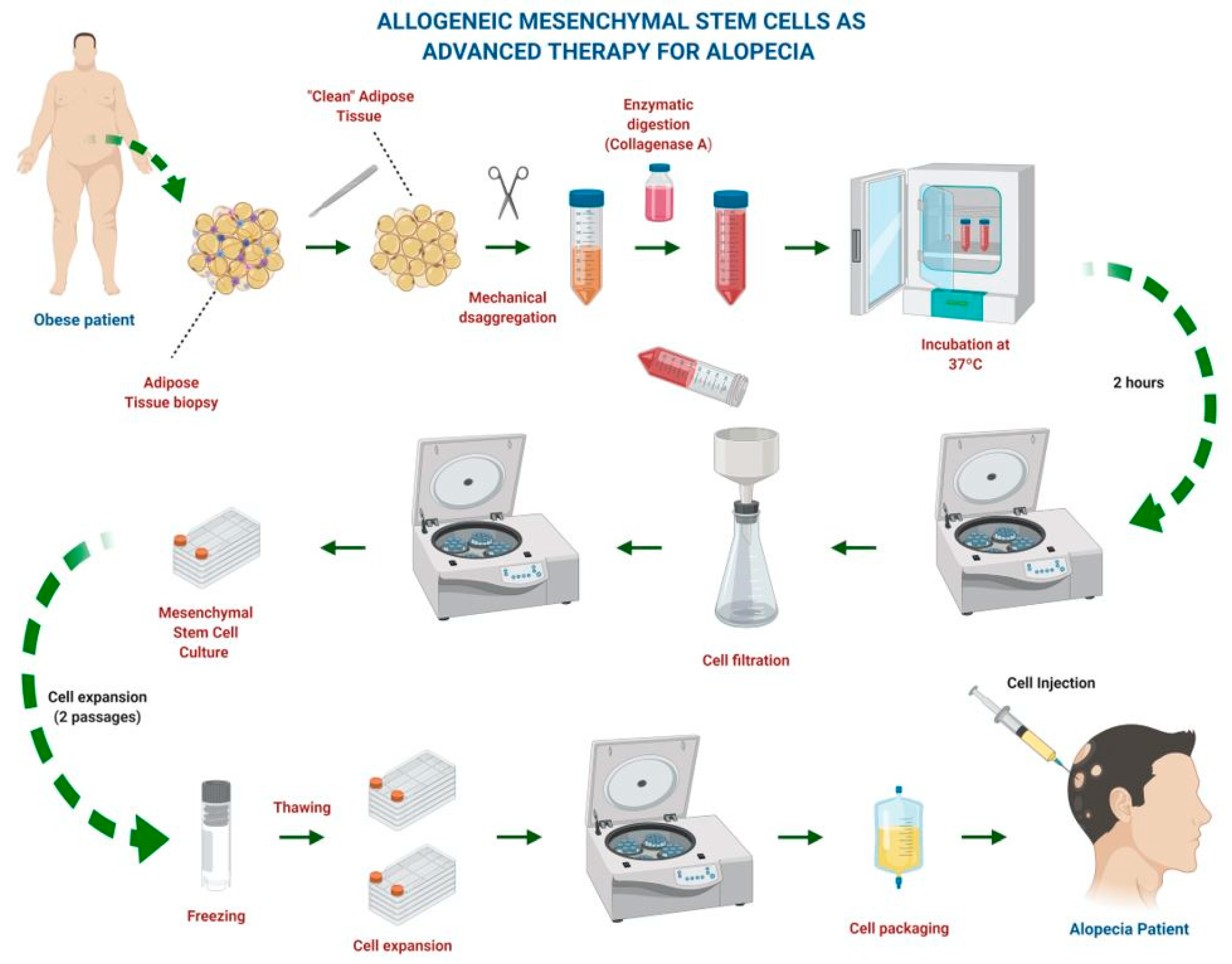
2. Mesenchymal Stem Cell Therapy
3. Gene Therapy in Alopecia
4. Tissue Engineering in Alopecia
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gonzalez-Cantero, A.; Arias-Santiago, S.; Buendía-Eisman, A.; Molina-Leyva, A.; Gilaberte, Y.; Fernández-Crehuet, P.; Husein-ElAhmed, H.; Viera-Ramírez, A.; Fernández-Peñas, P.; Taberner, R.; et al. Do Dermatologic Diagnosis Change in Hot vs Cold Periods of The Year? A Sub-Analysis of the DIADERM National Sample (Spain 2016). Actas Dermo-Sifiliográficas 2019, 110, 734–743. [Google Scholar] [CrossRef]
- Okhovat, J.-P.; Marks, D.H.; Manatis-Lornell, A.; Hagigeorges, D.; Locascio, J.J.; Senna, M.M. Association Between Alopecia Areata, Anxiety, and Depression: A Systematic Review and Meta-analysis. J. Am. Acad. Dermatol. 2019. [Google Scholar] [CrossRef]
- Almohanna, H.M.; Perper, M.; Tosti, A. Safety concerns when using novel medications to treat alopecia. Expert Opin. Drug Saf. 2018, 17, 1115–1128. [Google Scholar] [CrossRef]
- Rongioletti, F.; Christana, K. Cicatricial (Scarring) Alopecias: An overview of pathogenesis, classification, diagnosis, and treatment. Am. J. Clin. Dermatol. 2012, 13, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Strong, A.L.; Neumeister, M.W.; Levi, B. Stem Cells and Tissue Engineering: Regeneration of the Skin and Its Contents. Clin. Plast. Surg. 2017, 44, 635–650. [Google Scholar] [CrossRef]
- Campos-Muñoz, A. Advanced therapies in current Medicine. Actual. Med. 2017, 102, 133. [Google Scholar] [CrossRef]
- Erben, R.G.; Lima, B.S.; Reischl, I.; Steinhoff, G.; Tiedemann, G.; Dalemans, W.; Vos, A.; Janssen, R.T.; Le Blanc, K.; Van Osch, G.J.; et al. White paper on how to go forward with cell-based advanced therapies in Europe. Tissue Eng. Part A 2014, 20, 2549–2554. [Google Scholar] [CrossRef] [Green Version]
- Regulation (EC) No 1394/2007 of the European Parliament and of the Council of 13 November 2007 on Advanced Therapy Medicinal Products and Amending Directive 2001/83/EC and Regulation (EC) No 726/2004. Available online: https://ec.europa.eu/health//sites/health/files/files/eudralex/vol-1/reg_2007_1394/reg_2007_1394_en.pdf (accessed on 22 December 2019).
- Cuende, N.; Álvarez-Márquez, A.J.; Díaz-Aunión, C.; Castro, P.; Huet, J.; Pérez-Villares, J.M. The regenerative medicine and stem cell business: Confusion with legal implications. Gac. Sanit. 2020. [Google Scholar] [CrossRef]
- EudraLex. The Rules Governing Medicinal Products in the European Union Volume 4. Guidelines on Good Manufacturing Practice Specific to Advanced Therapy Medicinal Products. Available online: https://ec.europa.eu/health/sites/health/files/files/eudralex/vol-4/2017_11_22_guidelines_gmp_for_atmps.pdf (accessed on 22 December 2019).
- Thirabanjasak, D.; Tantiwongse, K.; Thorner, P.S. Angiomyeloproliferative lesions following autologous stem cell therapy. J. Am. Soc. Nephrol. 2010, 21, 1218–1222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moll, G.; Drzeniek, N.; Kamhieh-Milz, J.; Geissler, S.; Volk, H.-D.; Reinke, P. MSC Therapies for COVID-19: Importance of Patient Coagulopathy, Thromboprophylaxis, Cell Product Quality and Mode of Delivery for Treatment Safety and Efficacy. Front. Immunol. 2020, 11, 1091. [Google Scholar] [CrossRef]
- Wang, L.; Ji, H.; Zhou, J.; Xie, J.; Zhong, Z.; Li, M.; Bai, W.; Li, N.; Zhang, Z.; Wang, X.; et al. Therapeutic Potential of Umbilical Cord Mesenchymal Stromal Cells Transplantation for Cerebral Palsy: A Case Report. Case Rep. Transplant. 2013, 2013, 146347. [Google Scholar] [CrossRef]
- Maxson, S.; Lopez, E.A.; Yoo, D.; Danilkovitch-Miagkova, A.; Leroux, M.A. Concise Review: Role of Mesenchymal Stem Cells in Wound Repair. STEM CELLS Transl. Med. 2012, 1, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lee, S.K.; Kim, H.; Kim, T.M. Exosomes Secreted from Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Accelerate Skin Cell Proliferation. Int. J. Mol. Sci. 2018, 19, 3119. [Google Scholar] [CrossRef] [Green Version]
- Yates, C.C.; Rodrigues, M.; Nuschke, A.; Johnson, Z.I.; Whaley, D.; Stolz, D.; Newsome, J.; Wells, A. Multipotent stromal cells/mesenchymal stem cells and fibroblasts combine to minimize skin hypertrophic scarring. Stem Cell Res. Ther. 2017, 8, 193. [Google Scholar] [CrossRef] [Green Version]
- Almalki, S.G.; Agrawal, D.K. Key transcription factors in the differentiation of mesenchymal stem cells. Differentiation 2016, 92, 41–51. [Google Scholar] [CrossRef] [Green Version]
- Park, J.S.; Yang, H.N.; Woo, D.G.; Jeon, S.Y.; Park, K.-H. The promotion of chondrogenesis, osteogenesis, and adipogenesis of human mesenchymal stem cells by multiple growth factors incorporated into nanosphere-coated microspheres. Biomaterials 2011, 32, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Miyagawa, I.; Nakayamada, S.; Kondo, M.; Tanaka, Y. Regulatory Mechanism of The Induction of Regulatory T Cells through Growth Factors Released by Human Mesenchymal Stem Cells. Crit. Rev. Immunol. 2018, 38, 471–478. [Google Scholar] [CrossRef]
- Debnath, T.; Chelluri, L.K. Standardization and quality assessment for clinical grade mesenchymal stem cells from human adipose tissue. Hematol. Transfus. Cell Ther. 2019, 41, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.D.; Lopes-Pacheco, M.; Paz, A.H.R.; Cruz, F.F.; Melo, E.B.; De Oliveira, M.V.; Xisto, D.G.; Capelozzi, V.L.; Morales, M.M.; Pelosi, P.; et al. Mesenchymal Stem Cells from Bone Marrow, Adipose Tissue, and Lung Tissue Differentially Mitigate Lung and Distal Organ Damage in Experimental Acute Respiratory Distress Syndrome. Crit. Care Med. 2018, 46, e132–e140. [Google Scholar] [CrossRef]
- Zare, H.; Jamshidi, S.; Dehghan, M.M.; Saheli, M.; Piryaei, A. Bone marrow or adipose tissue mesenchymal stem cells: Comparison of the therapeutic potentials in mice model of acute liver failure. J. Cell. Biochem. 2018, 119, 5834–5842. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, K.L.; Mafi, P.; Mafi, R.; Khan, W.S. The Use of Growth Factors and Mesenchymal Stem Cells in Orthopaedics: In particular, their use in Fractures and Non-Unions: A Systematic Review. Curr. Stem Cell Res. Ther. 2017, 12, 312–325. [Google Scholar] [CrossRef]
- Liu, X.; Li, Q.; Niu, X.; Hu, B.; Chen, S.; Song, W.; Ding, J.; Zhang, C.; Wang, Y. Exosomes Secreted from Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Prevent Osteonecrosis of the Femoral Head by Promoting Angiogenesis. Int. J. Biol. Sci. 2017, 13, 232–244. [Google Scholar] [CrossRef]
- Damia, E.; Chicharro, D.; Lopez, S.; Cuervo, B.; Rubio, M.; Sopena, J.; Vilar, J.M.; Carrillo, J.M. Adipose-Derived Mesenchymal Stem Cells: Are They a Good Therapeutic Strategy for Osteoarthritis? Int. J. Mol. Sci. 2018, 19, 1926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Westhauser, F.; Senger, A.-S.; Reible, B.; Moghaddam, A. In Vivo Models for the Evaluation of the Osteogenic Potency of Bone Substitutes Seeded with Mesenchymal Stem Cells of Human Origin: A Concise Review. Tissue Eng. Part C Methods 2017, 23, 881–888. [Google Scholar] [CrossRef]
- Kim, K.H.; Blasco-Morente, G.; Cuende, N.; Arias-Santiago, S. Mesenchymal stromal cells: Properties and role in management of cutaneous diseases. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Sellheyer, K.; Krahl, D. Skin mesenchymal stem cells: Prospects for clinical dermatology. J. Am. Acad. Dermatol. 2010, 63, 859–865. [Google Scholar] [CrossRef]
- Maranda, E.L.; Rodriguez-Menocal, L.; Badiavas, E.V. Role of Mesenchymal Stem Cells in Dermal Repair in Burns and Diabetic Wounds. Curr. Stem Cell Res. Ther. 2016, 12, 61–70. [Google Scholar] [CrossRef]
- Marfia, G.; Navone, S.E.; Di Vito, C.; Ughi, N.; Tabano, S.; Miozzo, M.; Tremolada, C.; Bolla, G.; Crotti, C.; Ingegnoli, F.; et al. Mesenchymal stem cells: Potential for therapy and treatment of chronic non-healing skin wounds. Organogenesis 2015, 11, 183–206. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yu, D.; Xu, J.; Li, X.; Wang, X.; He, Z.; Zhao, T. Human umbilical cord mesenchymal stem cells improve irradiation-induced skin ulcers healing of rat models. Biomed. Pharmacother. 2018, 101, 729–736. [Google Scholar] [CrossRef]
- Zhang, P.; Kling, R.E.; Ravuri, S.K.; Kokai, L.E.; Rubin, J.P.; Chai, J.-K.; Marra, K.G. A review of adipocyte lineage cells and dermal papilla cells in hair follicle regeneration. J. Tissue Eng. 2014, 5. [Google Scholar] [CrossRef]
- Owczarczyk-Saczonek, A.; Krajewska-Włodarczyk, M.; Kruszewska, A.; Banasiak, Ł.; Placek, W.; Maksymowicz, W.; Wojtkiewicz, J. Therapeutic Potential of Stem Cells in Follicle Regeneration. Stem Cells Int. 2018, 2018, 1049641. [Google Scholar] [CrossRef] [Green Version]
- Gentile, P.; Scioli, M.G.; Bielli, A.; Orlandi, A.; Cervelli, V. Stem cells from human hair follicles: First mechanical isolation for immediate autologous clinical use in androgenetic alopecia and hair loss. Stem Cell Investig. 2017, 4, 58. [Google Scholar] [CrossRef] [Green Version]
- Byun, J.W.; Kim, H.J.; Na, K.; Ko, H.S.; Song, H.J.; Song, S.U.; Jeon, M.-S.; Choi, G.S. Bone marrow-derived mesenchymal stem cells prevent alopecia areata development through the inhibition of NKG2D expression: A pilot study. Exp. Dermatol. 2017, 26, 532–535. [Google Scholar] [CrossRef]
- Gilhar, A.; Etzioni, A.; Paus, R. Alopecia Areata. N. Engl. J. Med. 2012, 366, 1515–1525. [Google Scholar] [CrossRef] [Green Version]
- McPhee, C.G.; Duncan, F.J.; Silva, K.A.; King, L.E.; HogenEsch, H.; Roopenian, D.C.; Everts, H.B.; Sundberg, J.P. Increased Expression of Cxcr3 and Its Ligands, Cxcl9 and Cxcl10, during the Development of Alopecia Areata in the Mouse. J. Investig. Dermatol. 2012, 132, 1736–1738. [Google Scholar] [CrossRef] [Green Version]
- Garza, L.A.; Yang, C.-C.; Zhao, T.; Blatt, H.B.; Lee, M.; He, H.; Stanton, D.C.; Carrasco, L.; Spiegel, J.H.; Tobias, J.W.; et al. Bald scalp in men with androgenetic alopecia retains hair follicle stem cells but lacks CD200-rich and CD34-positive hair follicle progenitor cells. J. Clin. Investig. 2011, 121, 613–622. [Google Scholar] [CrossRef] [Green Version]
- Kang, J.-S.; Zheng, Z.; Choi, M.J.; Lee, S.-H.; Kim, D.-Y.; Cho, S.B. The effect of CD34+ cell-containing autologous platelet-rich plasma injection on pattern hair loss: A preliminary study. J. Eur. Acad. Dermatol. Venereol. 2012, 28, 72–79. [Google Scholar] [CrossRef]
- Ma, X.; Chen, S.; Jin, W.; Gao, Y. Th1/Th2 PB balance and CD200 expression of patients with active severe alopecia areata. Exp. Ther. Med. 2017, 13, 2883–2887. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, R.; Tanaka, K.; Amagai, M.; Ohyama, M. Involvement of the bulge region with decreased expression of hair follicle stem cell markers in senile female cases of alopecia areata. J. Eur. Acad. Dermatol. Venereol. 2010, 25, 1346–1350. [Google Scholar] [CrossRef]
- Leirós, G.J.; Attorresi, A.I.; Balañá, M.E. Hair follicle stem cell differentiation is inhibited through cross-talk between Wnt/β-catenin and androgen signalling in dermal papilla cells from patients with androgenetic alopecia. Br. J. Dermatol. 2012, 166, 1035–1042. [Google Scholar] [CrossRef]
- Kim, J.E.; Oh, J.H.; Woo, Y.J.; Jung, J.H.; Jeong, K.H.; Kang, H. Effects of mesenchymal stem cell therapy on alopecia areata in cellular and hair follicle organ culture models. Exp. Dermatol. 2018, 29, 265–272. [Google Scholar] [CrossRef]
- Bak, D.H.; Choi, M.J.; Kim, S.R.; Lee, B.C.; Kim, J.M.; Jeon, E.S.; Oh, W.; Lim, E.S.; Park, B.C.; Kim, M.J.; et al. Human umbilical cord blood mesenchymal stem cells engineered to overexpress growth factors accelerate outcomes in hair growth. Korean J. Physiol. Pharmacol. 2018, 22, 555–566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fawzy, M.M.; Gabrb, H.M.; Maadawy, Z.M.E. Autologous progenitor cell implantation as a novel therapeutic intervention for alopecia areata. J. Egypt Women Dermatol. Soc. 2011, 8, 11–16. [Google Scholar] [CrossRef]
- Elmaadawi, I.H.; Mohamed, B.M.; Ibrahim, Z.A.S.; Abdou, S.H.; El Attar, Y.A.; Youssef, A.; Shamloula, M.M.; Taha, A.; Metwally, H.G.; El Afandy, M.M.; et al. Stem cell therapy as a novel therapeutic intervention for resistant cases of alopecia areata and androgenetic alopecia. J. Dermatol. Treat. 2018, 29, 431–440. [Google Scholar] [CrossRef]
- Li, Y.; Yan, B.; Wang, H.; Li, H.; Li, Q.; Zhao, D.; Chen, Y.; Zhang, Y.; Li, W.; Zhang, J.; et al. Hair regrowth in alopecia areata patients following Stem Cell Educator therapy. BMC Med. 2015, 13, 87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perez-Meza, D.; Ziering, C.; Sforza, M.; Krishnan, G.; Ball, E.; Daniels, E. Hair follicle growth by stromal vascular fraction-enhanced adipose transplantation in baldness. Stem Cells Cloning Adv. Appl. 2017, 10, 1. [Google Scholar] [CrossRef] [Green Version]
- Anderi, R.; Makdissy, N.; Azar, A.; Rizk, F.; Hamade, A. Cellular therapy with human autologous adipose-derived adult cells of stromal vascular fraction for alopecia areata. Stem Cell Res. Ther. 2018, 9, 141. [Google Scholar] [CrossRef]
- Nilforoushzadeh, M.A.; Lotfi, E.; Heidari-Kharaji, M. Autologous adipose transplantation an effective method to treat alopecia after trauma: A case report. Clin. Cosmet. Investig. Dermatol. 2019, 12, 647–651. [Google Scholar] [CrossRef] [Green Version]
- Ruiz, R.G.; Rosell, J.M.C.; Ceccarelli, G.; De Sio, C.; De Angelis, G.C.; Pinto, H.; Astarita, C.; Graziano, A. Progenitor-cell-enriched micrografts as a novel option for the management of androgenetic alopecia. J. Cell. Physiol. 2019, 235, 4587–4593. [Google Scholar] [CrossRef]
- A Study to Evaluate and Compare Injections of Autologous Dermal and Epidermal Cells into the Balding Scalp of Subjects with Hair Loss (CA-0002899). ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT01451112 (accessed on 22 December 2019).
- Autologous Adipose-Derived Adult Stromal Vascular Cell Transplantation for Alopecia. ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03427905 (accessed on 22 December 2019).
- AGA Biocellular Stem/Stromal Hair Regenerative Study. ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT02849470 (accessed on 22 December 2019).
- Biocellular-Cellular Regenerative Treatment Scaring Alopecia and Alopecia Areata. ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03078686 (accessed on 22 December 2019).
- Nanofat Grafting for Treatment of Androgenetic Alopecia. ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03506503 (accessed on 22 December 2019).
- Kay, M.A. State-of-the-art gene-based therapies: The road ahead. Nat. Rev. Genet. 2011, 12, 316–328. [Google Scholar] [CrossRef]
- Williams, R.S.; Johnston, S.A.; Riedy, M.; DeVit, M.J.; McElligott, S.G.; Sanford, J.C. Introduction of foreign genes into tissues of living mice by DNA-coated microprojectiles. Proc. Natl. Acad. Sci. USA 1991, 88, 2726–2730. [Google Scholar] [CrossRef] [Green Version]
- Gorell, E.; Nguyen, N.; Lane, A.; Siprashvili, Z. Gene Therapy for Skin Diseases. Cold Spring Harb. Perspect. Med. 2014, 4, a015149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosa, J.; Suzuki, I.; Kravicz, M.; Caron, A.; Pupo, A.V.; Praça, F.G.; Bentley, M.V.L.B. Current Non-viral siRNA Delivery Systems as a Promising Treatment of Skin Diseases. Curr. Pharm. Des. 2018, 24, 2644–2663. [Google Scholar] [CrossRef]
- Hannon, G.J.; Rossi, J.J. Unlocking the potential of the human genome with RNA interference. Nat. Cell Biol. 2004, 431, 371–378. [Google Scholar] [CrossRef]
- Ruan, R.; Chen, M.; Sun, S.; Wei, P.; Zou, L.; Liu, J.; Gao, D.; Wen, L.; Ding, W. Topical and Targeted Delivery of siRNAs to Melanoma Cells Using a Fusion Peptide Carrier. Sci. Rep. 2016, 6, 29159. [Google Scholar] [CrossRef] [Green Version]
- Baroli, B. Penetration of nanoparticles and nanomaterials in the skin: Fiction or reality? J. Pharm. Sci. 2010, 99, 21–50. [Google Scholar] [CrossRef]
- Lenn, J.D.; Neil, J.; Donahue, C.; Demock, K.; Tibbetts, C.V.; Cote-Sierra, J.; Smith, S.H.; Rubenstein, D.; Therrien, J.-P.; Pendergrast, P.S.; et al. RNA Aptamer Delivery through Intact Human Skin. J. Investig. Dermatol. 2018, 138, 282–290. [Google Scholar] [CrossRef] [Green Version]
- Singhal, M.; Lapteva, M.; Kalia, Y.N. Formulation challenges for 21st century topical and transdermal delivery systems. Expert Opin. Drug Deliv. 2017, 14, 705–708. [Google Scholar] [CrossRef] [Green Version]
- Kushibiki, T.; Matsumoto, K.; Nakamura, T.; Tabata, Y. Suppression of tumor metastasis by NK4 plasmid DNA released from cationized gelatin. Gene Ther. 2004, 11, 1205–1214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biswas, S.; Torchilin, V.P. Dendrimers for siRNA Delivery. Pharmaceuticals 2013, 6, 161–183. [Google Scholar] [CrossRef] [Green Version]
- Haigh, O.; Depelsenaire, A.C.; Meliga, S.C.; Yukiko, S.R.; McMillan, N.A.; Frazer, I.H.; Kendall, M.A. CXCL1 gene silencing in skin using liposome-encapsulated siRNA delivered by microprojection array. J. Control. Release 2014, 194, 148–156. [Google Scholar] [CrossRef] [Green Version]
- Gilleron, J.; Querbes, W.; Zeigerer, A.; Borodovsky, A.; Marsico, G.; Schubert, U.; Manygoats, K.; Seifert, S.; Andree, C.; Stöter, M.; et al. Image-based analysis of lipid nanoparticle–mediated siRNA delivery, intracellular trafficking and endosomal escape. Nat. Biotechnol. 2013, 31, 638–646. [Google Scholar] [CrossRef]
- Chakraborty, C.; Sharma, A.R.; Sharma, G.; Doss, C.G.P.; Lee, S.-S. Therapeutic miRNA and siRNA: Moving from Bench to Clinic as Next Generation Medicine. Mol. Ther. Nucleic Acids 2017, 8, 132–143. [Google Scholar] [CrossRef] [Green Version]
- Nakamura, M.; Jo, J.-I.; Tabata, Y.; Ishikawa, O. Controlled Delivery of T-box21 Small Interfering RNA Ameliorates Autoimmune Alopecia (Alopecia Areata) in a C3H/HeJ Mouse Model. Am. J. Pathol. 2008, 172, 650–658. [Google Scholar] [CrossRef] [Green Version]
- Goodarzi, H.; Abbasi, A.; Saffari, M.; Haghighi, M.F.; Tabei, S.M.B.; Daloii, M.N. Differential expression analysis of balding and nonbalding dermal papilla microRNAs in male pattern baldness with a microRNA amplification profiling method. Br. J. Dermatol. 2012, 166, 1010–1016. [Google Scholar] [CrossRef]
- Sierra-Sánchez, Á.; Fernández-González, A.; Lizana-Moreno, A.; Espinosa-Ibáñez, O.; Martinez-Lopez, A.; Guerrero-Calvo, J.; Fernández-Porcel, N.; Ruiz-García, A.; Ordóñez-Luque, A.; Carriel, V.; et al. Hyaluronic acid biomaterial for human tissue-engineered skin substitutes: Preclinical comparative in vivo study of wound healing. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 2414–2427. [Google Scholar] [CrossRef]
- Papini, R. Management of burn injuries of various depths. BMJ 2004, 329, 158–160. [Google Scholar] [CrossRef] [Green Version]
- Shevchenko, R.V.; James, S.E. A review of tissue-engineered skin bioconstructs available for skin reconstruction. J. R. Soc. Interface 2009, 7, 229–258. [Google Scholar] [CrossRef] [Green Version]
- Lee, L.F.; Jiang, T.X.; Garner, W.; Chuong, C.-M. A Simplified Procedure to Reconstitute Hair-Producing Skin. Tissue Eng. Part C Methods 2011, 17, 391–400. [Google Scholar] [CrossRef] [Green Version]
- Nakao, K.; Morita, R.; Saji, Y.; Ishida, K.; Tomita, Y.; Ogawa, M.; Saitoh, M.; Tomooka, Y.; Tsuji, T. The development of a bioengineered organ germ method. Nat. Methods 2007, 4, 227–230. [Google Scholar] [CrossRef]
- Asakawa, K.; Toyoshima, K.-E.; Ishibashi, N.; Tobe, H.; Iwadate, A.; Kanayama, T.; Hasegawa, T.; Nakao, K.; Toki, H.; Noguchi, S.; et al. Hair organ regeneration via the bioengineered hair follicular unit transplantation. Sci. Rep. 2012, 2, 424. [Google Scholar] [CrossRef] [Green Version]
- Toyoshima, K.E.; Asakawa, K.; Ishibashi, N.; Toki, H.; Ogawa, M.; Hasegawa, T.; Irié, T.; Tachikawa, T.; Sato, A.J.; Takeda, A.; et al. Fully functional hair follicle regeneration through the rearrangement of stem cells and their niches. Nat. Commun. 2012, 3, 784. [Google Scholar] [CrossRef] [Green Version]
- Qiao, J.; Turetsky, A.; Kemp, P.; Teumer, J. Hair morphogenesis in vitro: Formation of hair structures suitable for implantation. Regen. Med. 2008, 3, 683–692. [Google Scholar] [CrossRef]
- Chermnykh, E.S.; Vorotelyak, E.A.; Gnedeva, K.Y.; Moldaver, M.V.; Yegorov, Y.E.; Vasiliev, A.V.; Terskikh, V.V. Dermal papilla cells induce keratinocyte tubulogenesis in culture. Histochem. Cell Biol. 2010, 133, 567–576. [Google Scholar] [CrossRef]
- Miao, Y.; Bin Sun, Y.; Liu, B.C.; Jiang, J.D.; Hu, Z.Q. Controllable Production of Transplantable Adult Human High-Passage Dermal Papilla Spheroids Using 3D Matrigel Culture. Tissue Eng. Part A 2014, 20, 2329–2338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sriwiriyanont, P.; Lynch, K.A.; Maier, E.A.; Hahn, J.M.; Supp, D.M.; Boyce, S.T. Morphogenesis of chimeric hair follicles in engineered skin substitutes with human keratinocytes and murine dermal papilla cells. Exp. Dermatol. 2012, 21, 783–785. [Google Scholar] [CrossRef]
- Ehama, R.; Ishimatsu-Tsuji, Y.; Iriyama, S.; Ideta, R.; Soma, T.; Yano, K.; Kawasaki, C.; Suzuki, S.; Shirakata, Y.; Hashimoto, K.; et al. Hair Follicle Regeneration Using Grafted Rodent and Human Cells. J. Investig. Dermatol. 2007, 127, 2106–2115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, B.M.; Kwack, M.H.; Kim, M.K.; Kim, J.C.; Sung, Y.K. Sphere Formation Increases the Ability of Cultured Human Dermal Papilla Cells to Induce Hair Follicles from Mouse Epidermal Cells in a Reconstitution Assay. J. Investig. Dermatol. 2012, 132, 237–239. [Google Scholar] [CrossRef] [Green Version]
- Thangapazham, R.L.; Klover, P.; Wang, J.-A.; Zheng, Y.; Devine, A.; Li, S.; Sperling, L.; Cotsarelis, G.; Darling, T.N. Dissociated human dermal papilla cells induce hair follicle neogenesis in grafted dermal-epidermal composites. J. Investig. Dermatol. 2013, 134, 538–540. [Google Scholar] [CrossRef] [Green Version]
- Osada, A.; Iwabuchi, T.; Kishimoto, J.; Hamazaki, T.S.; Okochi, H. Long-Term Culture of Mouse Vibrissal Dermal Papilla Cells and De Novo Hair Follicle Induction. Tissue Eng. 2007, 13, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Young, T.-H.; Lee, C.-Y.; Chiu, H.-C.; Hsu, C.-J.; Lin, S.-J. Self-assembly of dermal papilla cells into inductive spheroidal microtissues on poly (ethylene-co-vinyl alcohol) membranes for hair follicle regeneration. Biomaterials 2008, 29, 3521–3530. [Google Scholar] [CrossRef]
- Soma, T.; Fujiwara, S.; Shirakata, Y.; Hashimoto, K.; Kishimoto, J. Hair?inducing ability of human dermal papilla cells cultured under Wnt/??-catenin signalling activation. Exp. Dermatol. 2012, 21, 307–309. [Google Scholar] [CrossRef]
- Higgins, C.A.; Chen, J.C.; Cerise, J.E.; Jahoda, C.A.B.; Christiano, A.M. Microenvironmental reprogramming by three-dimensional culture enables dermal papilla cells to induce de novo human hair-follicle growth. Proc. Natl. Acad. Sci. USA 2013, 110, 19679–19688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Higgins, C.A.; Roger, M.F.; Hill, R.P.; Ali-Khan, A.S.; Garlick, J.A.; Christiano, A.M.; Jahoda, C.A. Multifaceted role of hair follicle dermal cells in bioengineered skins. Br. J. Dermatol. 2017, 176, 1259–1269. [Google Scholar] [CrossRef]
- Wu, J.J.; Zhu, T.-Y.; Lu, Y.-G.; Liu, R.-Q.; Mai, Y.; Cheng, B.; Lu, Z.-F.; Zhong, B.-Y.; Tang, S.-Q. Hair follicle reformation induced by dermal papilla cells from human scalp skin. Arch. Dermatol. Res. 2006, 298, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Leirós, G.J.; Kusinsky, A.G.; Drago, H.; Bossi, S.; Sturla, F.; Castellanos, M.L.; Stella, I.Y.; Balañá, M.E. Dermal Papilla Cells Improve the Wound Healing Process and Generate Hair Bud-Like Structures in Grafted Skin Substitutes Using Hair Follicle Stem Cells. STEM CELLS Transl. Med. 2014, 3, 1209–1219. [Google Scholar] [CrossRef]
- Abaci, H.E.; Coffman, A.; Doucet, Y.; Chen, J.; Jacków, J.; Wang, E.; Guo, Z.; Shin, J.U.; Jahoda, C.A.; Christiano, A.M. Tissue engineering of human hair follicles using a biomimetic developmental approach. Nat. Commun. 2018, 9, 1–11. [Google Scholar] [CrossRef]
- Tissue Engineering for Hair Follicle Regeneration—Full Text View—ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/show/NCT00506636 (accessed on 29 October 2020).
| MSC Source | Type of Study | Experimental Design | Results | Conclusions | Reference |
|---|---|---|---|---|---|
| Human bone marrow (hBM-MSCs) | In vivo study Alopecia Areata induced in 24 C3H/HeJ mice. | Control group (n = 11) and MSC group (n = 13). Intravenous injection of two doses of MSCs at days 1 and 7. Mice were analysed for 15 weeks. | 1-MSC-treated mice showed decreased IFNG concentration as early as 5 weeks post-transplantation. 2-Mice treated with MSCs showed less inflammatory cell infiltration in the dermis and the number of hair follicles was not decreased compared with the control group. | 1-After 15 weeks post-transplantation; 91% of control mice experimented hair loss against 23% of MSC-treated group. 2-MSCs mediate inhibition of IFNG and CD3 and CD8 + NKG2D+ T-cell infiltration which protects against the collapse of the hair follicle immune privilege. | [35] |
| Human bone marrow (hBM-MSCs) | In vitro study in Alopecia Areata induced environment. | Co-culture of human dermal papilla cells (hDPCs) (1 × 105 cells per well) pre-treated with interferon gamma (IFN-γ) to reproduce Alopecia Areata environment with hBM-MSCs (5 × 104 cells). | 1-hDPCs proliferation were increased up to ~120% compared to control cultures. 2-Levels of β-catenin protein which were suppressed by IFN-γ treatment were shown to be reversed by hBM-MSCs. 3-hMSCs induced higher phosphorylation of STAT1 and STAT3. 4-hMSCs increased mRNA expression of TNF-α and IL-1β 5-Growth factor analysis revealed that co-culture of hBM-MSCs and hDPCs enhanced expression of IGF-2, TGF-α, TGF-β3 and NT-4 which could be involved in driving anagen reentry. | 1-hHMSC co-culture could reverse suppressed hDPC proliferation and may prolong the anagen phase through activation of the β-catenin/Wnt and JAK/STAT signalling pathways. 2-hBM-MSCs in refractory AA patients might restore hair follicles (HFs) immune privilege through immunomodulatory effects and induce reentry signalling from the telogen to anagen phase | [43] |
| Human umbilical cord blood (hUCB-MSCs) | In vivo study in C3H/HeJ mice (Telogen-anagen transition model was induced by depilating the dorsal skin of mice in the telogen phase of the hair cycle). | Four groups of study (6 weeks): Control group Minoxidil-treated group Saline intra-dermal injection-treated group hUCB-MSC intra-dermal injection-treated group (8 sites—1 × 105 cells/mice) | 1-After 6 weeks post-treatment, hair regrowth was complete in the hUCB-MSCs group. 2-Minoxidil-treated group presented incomplete pigmentation and contained hairs in the early stage of the hair cycle, 3-Control and Saline-injected groups retained large areas without anagen induction. | 1-hUCB-MSCs can accelerate the initiation of the hair follicle telogen-anagen transition, increase the number of hairs in vivo, and enhance expression of proteins related to hair induction in vitro. 2-IGFBP-1 (assumed as the main secretory factor of hUCB-MSCs) restores and promotes the hair-induction ability of hDPCs via an IGF-1/IGFBP-1 co-localization. | [44] |
| In vitro analysis of the effect hUCB-MSCs co-cultured with human dermal papilla cells (hDPCs). | Co-culture of 1 × 105 hDPCs with 2 × 105 hUCB-MSCs. | 1-hUCB-MSCs co-culture enhanced hDPC proliferation and restored ALP activity after 5 days, both of which are anagen markers of the hair cycle. 2-β-catenin, AKT, and GSK3β, which are proteins contained in the pathway related to cell growth and proliferation, were up-regulated in hDPCs by co-culture with hUCB-MSCs. 3-IGFBP-1 and VEGF were upregulated in the medium of the hDPC plus hUCB-MSCs group compared to the hDPC group. |
| MSC Source/Clinical Trial Title | Experimental Design | Pre-Treatment Data | Clinical Results | Conclusions | Reference |
|---|---|---|---|---|---|
| Autologous hair follicle stem cells from the lower bulge areas | N = 8 patients with Alopecia Areata (AA). One millilitre (in a density of 105 cells/mL) was injected intradermally once per centimetre square using a 23-gauge needle. Clinical improvement was assessed by calculating the percentage of the difference in the AA at the end of 3 months and 6 months in relation to the baseline extent. | The extent of affection ranged from 25 to 70% (mean: 48 ± 15%). Patients age ranged from 6 to 17 years. (mean: 10.12 ± 3.68 years). The duration of disease ranged from 1 to 4 years (mean: 2.31 ± 0.96 years) | 1-Patients showed variable degrees of response, 20–80% (mean: 45 ± 22%) from baseline at the end of the third month and 30–100% (mean: 69 ± 27%) from baseline at the end of the sixth month. 2-After 6 months, excellent response was achieved in five patients (62.5%), good response was achieved in two patients (25%), whereas one patient (12.5%) showed poor response. 3-There was a negative correlation between the age of the patients and the grade of response they achieved, that is, the younger the patient, the better the response. | 1-Approximately 60% of the patients (the excellent responders) reported improved quality of life. | [45] |
| Autologous bone marrow derived mononuclear cells (including stem cells) and follicular stems cells | N = 40 patients: 20 patients with resistant Alopecia Areata (AA) and 20 patients with Androgenetic Alopecia (AGA). Four groups of study: *Group 1 (10 resistant AA patients) *Group 3 (10 AGA patients) *These received a single session of intradermal injections of autologous bone marrow derived mononuclear cells (BMMCs). **Group 2 (10 resistant AA patients) **Group 4 (10 AGA patients) **These received a single session of intradermal injections of autologous follicular stem cells (FSC). One millilitre (in a density of 105 cells/mL) was injected intradermally once per centimetre square using a 26-gauge needle. Clinical improvement was assessed at 3 and 6 months | The extent of affection ranged from; Mean Group 1: 44 ± 20.65% Mean Group 2: 46± 18.97% Mean Group 3: 58± 19.32% Mean Group 4: 50 ± 0% The ages of the patients ranged from; 10-50 years (mean 26 ± 8 years). All patients were resistant to conventional treatments. The duration of disease ranged from; Mean Group 1: 2 ± 1 years Mean Group 2: 1.5 ± 0.7 years Mean Group 3: 2 ± 0.9 years Mean Group 4: 1.9 ± 0.8 years | 1-The mean percentage of improvement in AA patients was 45 ± 22% in AA subjects receiving autologous BMMCs and 58 ± 34% in AA subjects receiving autologous FSC with non-statistically significant difference. 2-The mean percentage of improvement in AGA patients was 52 ± 28% in AGA subjects receiving autologous BMMCs and 42 ± 27% in AGA subjects receiving autologous FSCs with non-statistically significant difference. 3-45% of AA patients suffered recurrence of disease activity after one year of follow up mostly due to their stressful life events. | 1-BMMCs and FSCs gave significant improvement in AGA and AA with no statistically significant difference between both methods. 2-Autologous BMMCs and autologous FSC seem to be a safe, tolerable and effective treatment | [46] |
| Autologous blood mononuclear cells “educated” with allogeneic umbilical cord blood stem cells Stem Cell Educator Therapy | N = 9 patients:9 alopecia areata patients (AA). All of them received a Stem Cell Educator Therapy consisting in autologous blood mononuclear cells isolated using a Blood Cell Separator MCS+ (6–7 h) and then, transferred into the Educator device for exposure to allogeneic cord blood stem cells (CB-SCs) ex vivo cultured and prepared (2–3 h). After this time, CB-SC-treated mononuclear cells were returned to the patient’s circulation via a dorsal vein in the hand (16-gauge IV needle) with physiological saline (2 to 3 mL/min). Follow-up visits were scheduled 4, 12, 24, 40, 56, 84, and 112 weeks after treatment for clinical assessments and laboratory tests. Skin biopsies of scalps were performed before the treatment and at 12 weeks post-treatment. | The degree of affection ranged from patchy alopecia areata (3), alopecia totalis (2) to alopecia universalis (4). Mean alopecic duration: 5.1 ± 6.1 years. The ages of the patients ranged from; 12–26 years (mean 20.1 ± 4.6 years). | 1-At 4 weeks post-treatment with Stem Cell Educator therapy, there was hair regrowth in subjects with patchy AA and alopecia totalis. 2-Patients (3/4) with alopecia universalis exhibited regrowth of eyebrows and eyelashes at the 12-week follow-up. 3-All these improvements were maintained throughout the final follow-up at 2 years. 5-Only one participant with alopecia universalis failed to show a response to the Stem Cell Educator therapy, possibly due to a previous long-term therapy with oral prednisone | 1-This phase 1/phase 2 study demonstrates the safety and feasibility of Stem Cell Educator therapy in the treatment of AA subjects. 2-Stem Cell Educator therapy can control the autoimmunity and lead to hair regrowth. | [47] |
| Autologous adipose cell enriched with stromal vascular fraction (SVF) | N = 9 patientswith androgenic alopecia (AHA). The adipose cells were injected into the scalp of the patient in a single dose. 1 mL/cm2 scalp was injected. The composition was a mixture of adipose-enriched SVF cells and Lactated Ringer. Follow-up for hair count, anagen percentage, telogen percentage and cumulative thickness at weeks 6, 12 and 24. | Degree of affection: Hamilton II-VI/Ludwig I-III Average age: 29 (range 19–54) Average volume of adipose cells injected: 30 mL | 1-Hair count was significantly augmented after 24 weeks (28.4 ± 17.3; p = 0.010). 2-Percentage of anagen hair was increased but did not reach a statistical significance (p = 0.094). 3-Percentage of telogen hair was decreased but did not reach a statistical significance (p = 0.094). 4-Cumulative thickness was augmented but did not reach a statistical significance (p = 0.133). | 1-Enriched adipose cells injections may be a promising approach for treating AHA in both men and women. | [48] |
| Autologous adipose-derived stromal vascular cells (ADSVCs) | N = 20 patients with confirmed diagnostic of hair loss. The ADSVCs were injected into the scalp of the patient with a 30-gauge needle. A total of 5 mL was injected in 25 spots. 4 to 4.7 × 106 cells were transplanted: in fact, 0.2 mL containing 0.160–0.188 × 106 cells were injected per spot (total = 25 spots, 5 mL) Follow-up for hair evaluation was based on the hair cycles and was performed 1 week, 3 months, and 6 months after the procedure. | All the selected subjects showed partial alopecia grade 1 or 2 at Ludwig Scale. The ages of the patients ranged from; 20-63 years(mean 38.3 ± 2.3 years). 55% of the patients showed medium diameter hair and 45% showed fine hair. Study subjects showed abnormal hair density (density < 175 hair/ cm2 in 100% of the subjects) | 1-Hair diameter increased significantly, especially 6 months after the treatment (80.8 ± 2.4 μ and 62.8 ± 1.7 μ vs. 60.5 ± 1.8 μ for 6 and 3 months postoperatively vs. preoperatively). 2-Hair density was significantly augmented after treatment (121.1 ± 12.5 and 120.8 ± 12.6 vs. 85.1 ± 8.7 for 6 and 3 months postoperatively vs. preoperatively). 3-Results of the pull test showed a significant decrease in the number of extracted hair (0.80 ± 0.17 and 0.90 ± 0.20 vs. 4.35 ± 0.33 for 6 and 3 months postoperatively vs. preoperatively). 4-2 of the 20 patients showed no significant improvements. | 1-ADSVC injection promotes good stability of the hair by increasing the hair density, the hair diameter, and decreasing the pull test to almost zero. | [49,50,51,52] |
| Type of Gene Therapy | Type of Study | Experimental Design | Results | Conclusions | Reference |
|---|---|---|---|---|---|
| Intralesional injections of oligonucleotides and siRNAs | In vivo study Older female C3H/HeJ mice having an alopecic region on the back in the waxing phase. | N = 78 C3H/HeJ mice with alopecic lesions (develop hair loss spontaneously): Group 1: Treatment with Il4 injections (n = 10) Group 2: Treatment with 0.9% sodium chloride injections (n = 10) Group 3: Treatment with Il4 and Ifng injections (n = 10) Group 4: Treatment with anti-Ifng antibody injections (n = 6) Group 5: Treatment with rat IgG injections (n = 6) Group 6: Treatment with antisense Tbx21 oligonucleotide injections (n = 6) Group 7: Treatment with non-sense oligonucleotide injections (n = 6) Group 8: Treatment with cationized gelatin-conjugated Tbx21 siRNA injections (n = 8) Group 9: Treatment with naked Tbx21 siRNA injections (n = 8) Group 10: Treatment with cationized gelatin-conjugated non-sense siRNA injections (n = 8) | 1-Intralesional injections of recombinant Il4 (0.1 μg) (Group 1) every day for 3 weeks significantly restored hair shaft elongations when compared with the sodium chloride injections (Group 2). There was no recurrence of alopecia from these mice during a 2-month. 2-Il4 effect was suppressed by the simultaneous injection of 0.01 μg of recombinant Ifng (Group 3). 3-Anti-Ifng antibody injections (Group 4) improved the hair growth index more efficiently than the control rat IgG (Group 5). There was no disappearance of hair shafts from these mice during a 2-month observation period after the cessation of antibody application. 4-Antisense Tbx21 oligonucleotide (Group 6) was significantly more effective for alopecia than non-sense oligonucleotide (Group 7). 5-Cationized gelatin-conjugated Tbx21 siRNA injections (Group 8) were more effective than naked Tbx21 siRNA injections (Group 9) or non-sense siRNA conjugated with cationized gelatin (Group 10). There was no recurrence of alopecia in the mice during a 2-month observation period after the cessation of Tbx21 siRNA application. | 1-Intralesional injections of Il4 suppressed an enhanced expression of Ifng in alopecic skin. 2-Intralesional injections of Tbx21 antisense oligonucleotide restored the hair shaft elongation. 3-Efficient and safety delivery of Tbx21 siRNA to alopecic skin using a biodegradable cationized gelatin demonstrated specific inhibition of target gene expression (Ifng) resulting in a restoration of hair shaft elongation. | [71] |
| Cell Types and Scaffold | Type of Study | Experimental Design | Results | Conclusions | Reference |
|---|---|---|---|---|---|
| Human Dermal Papilla (DP) cells and Keratinocytes (KT) in type I collagen matrix | In vitro study | DP cells were isolated from hair follicle of human scalp. Rat vibrissa DP cells were isolated with microdissection. Keratinocytes were isolated from normal human skin. Dermal equivalents were prepared with type I collagen extracted from rat tail tendons. Human dermal fibroblasts or DP cells were embedded in a final concentration of 1 × 105 cells/mL were added. Keratinocytes were seeded on the top of the gel or embedded inside the collagen matrix. | 1-Human and rat vibrissa DP cells were able to reorganize the collagen lattices within the first 48 h. Contraction was significantly stronger with rat vibrissa DP cells than with human DP cells (60 and 40%, respectively). 2-Addition of epidermal keratinocytes enhanced contraction in both cases (75 and 56%, respectively). 3-After 1 week, rat vibrissa DP cells cultured together with keratinocytes totally disaggregated and lysed collagen lattices. 4-Human DP cells reorganized collagen matrix but were unable to disintegrate it. 5-After 10 days, DP cells embedded into collagen gel, and keratinocytes seeded on the top of the gel formed tubular structures. | 1-DP cells induced formation of multicellular tube-like outgrowths in the culture of epidermal keratinocytes | [81] |
| Human dermal papilla (DP) cells cultured in Matrigel | In vitro study | Dermal cells were isolated from dermal papillae microdissected from the bulbs of dissected hair follicles. Cells were seeded onto wells precoated with Matrigel or hyaluronan or not. For the formation of DP spheroids in 3D Matrigel culture, different conditions were evaluated. All experiments, characterization of different culture conditions was conducted after 5 days of harvesting. | 1-When 1 × 104 DP cells were cultured on the 96-well plates precoated with Matrigel for 5 days, both passage 2 and passage 8 DP cells formed spheroidal microtissues with a diameter of 150–250 μm. 2-Cells within DP spheres could disaggregate and migrate out, which was similar to primary DP. 3-Expression of several genes and proteins associated with hair follicle inductivity of DP cells, such as NCAM, Versican, and α-smooth muscle actin was elevated in the spheres compared with the dissociated DP cells. 4-DP spheroids mixed with HGMCs (hair germinal matrix cells—commercial cell line) and incubated on the Matrigel surface, developed colourless hair shafts. | 1-3D Matrigel culture technique is an ideal culture model for forming DP spheroids and that sphere formation partially models the intact DP, resulting in hair induction, even by high-passage DP cells. | [82] |
| Cell Types and Scaffold | Type of Study | Experimental Design | Results | Conclusions | Reference |
|---|---|---|---|---|---|
| Mouse epidermal and dermal cells in a gel-like endogenous matrix or in Integra™ Bilayer Wound Matrix | In vivo study Athymic nude, hairy SCID, or normal mice of the same inbred strain | Dissociated new-born mouse epidermal and dermal stem cell were mixed in different ratios. For gel-like endogenous matrix: 150–200 μL of cell suspension, containing 2–20 million cells, was pipetted onto a tissue cell culture insert. For each 1.5 cm2 piece of Integra, 12 million epidermal cells and 60 million dermal cells in 200 μL of serum-free medium. The intended area of skin to be grafted for hair bearing was excised in full thickness. | 1-At day 8, hair germs started to appear, which progress to the hair peg stage at about day 9. 2-Hairs can be seen on the surface of the wound as early as 11–15 days postgraft. 3-Histological sections of the skin at day 11 postgraft showed that normal layers of the skin were regenerated. 4-after hairs were clipped or plucked, they grew and reached normal length in about 2 months. | 1-Ratio of 1:5–10 for epidermal:dermal cells is optimal. 2-There are no differences of skin quality or hair growth when tissue culture inserts or IntegraTM are used. 3-After 18 months, hair growth and cycling are active. | [76] |
| Mouse embryonic epithelial and dermal cells using the organ germ method. Collagen gel | In vivo study -C57BL/6 mice -C57BL/6-TgN (act-EGFP) mice -C57BL/6-TgN (act-EGFP) OsbC14-Y01-FM131 mice -Balb/c nu/nu mice | Bioengineered hair follicle germs: 7.5 × 104 epithelial cells and 7.5 × 104 mesenchymal cells which were derived from skin from C57BL/6-TgN (act-EGFP) mouse embryos. After 2 days in culture, to develop and mature bioengineered hair follicles, they were transplanted into the sub-renal capsules of 8-week-old C57BL/6 mice At 14 days after engraftment, mature bioengineered hair follicles were harvested and dissected into a single or a couple of follicular units via stereomicroscopic observation. Mature bioengineered hair follicles were intracutaneously grafted into Balb/c nu/nu mice. | 1-After 7 to 10 days after the orthotopic transplantation, the wound was completely healed. 2-The eruption of the bioengineered pelage shaft was observed at 14 ± 1.8 (n = 30) days at a frequency of 90% (n = 33) 3-The bioengineered hairs repeatedly exhibited growth and regression. 4-The periods of growth and regression of bioengineered hair follicles lasted 11.0 (±2.6) days and 9.4 (±2.4) days, respectively. | 1-Bioengineered hair follicles generated by ectopic transplantation can functionally replace orthotopic FUT therapy. 2-Ectopic bioengineered pelage follicle connected to the epidermal layer of the skin, reproduced the stem cell niche and the hair cycle equivalent to the natural pelage, and repeatedly produced the same hair types during the hair cycles. | [78] |
| Mouse embryonic skin epithelial and mesenchymal cells. Epithelial cells from adult vibrissa-derived bulge region and primary cultured dermal papilla (DP) cells. Collagen gel | In vivo study -C57BL/6 -C57BL/6-TgN (act-EGFP) OsbC14-Y01-FM131 mice -Balb/c nu/nu mice | The bioengineered pelage follicle germs: mouse embryonic skin epithelial and mesenchymal cells (7.5 × 103 of each cell type) The bioengineered vibrissae follicle germ: epithelial cells (1 × 104 cells) isolated from the adult vibrissae-derived bulge region and primary cultured DP cells (3 × 103 cells) Bioengineered hair germs were ectopically engrafted into the sub-renal capsules of C57BL/6 mice or intracutaneously transplanted onto the back of Balb/c mice. | 1-Eruption and growth hair shafts were observed at a frequency of 94% (n = 78) and 74% (n = 62) for bioengineered pelage and vibrissae follicles, respectively. 2-Bioengineered pelage follicle and the vibrissae follicle formed correct structures comprising an infundibulum and sebaceous gland in the proximal region. 3-Bioengineered pelage follicle germs were found to produce all types of pelage hairs. 4-The bioengineered pelage and vibrissae follicles repeated the hair cycle at least 3 times during the 80-day period. | 1-Both bioengineered hair follicles produce follicles that can repeat the hair cycle, connect properly with surrounding skin tissues and achieve piloerection. | [79] |
| Human Dermal Papilla (DP) cells and Keratinocytes (KT) in collagen-glycosaminoglycan scaffolds | In vivo study | Human or murine DP cells, combined with foreskin-derived keratinocytes (KT) or transduced KT with pBABE-puro encoding N-terminally truncated β-catenin (KT’) (expression induced by hydroxytamoxifen (4OHT) administration) A positive control fabricated with murine hair from newborn cells was also evaluated. After 10 days incubation at air–liquid interface, Engineering Skin Substitutes (ESS) were grafted to athymic mice and were evaluated for 6 weeks. | 1-EF1 and WNT10B were significantly higher in 4OHT-treated ESS compared with vehicle-treated ESS but no hairs were observed in ESS with KTs’ and hDP cells. 2-Only ESS with mDP cells formed follicular structures, as confirmed by trichohyalin and keratin 10 immunostaining. | 1-Chimeric hair follicles were successfully generated in ESS containing combinations of mDP cells and KTs or KTs’, although they were deficient anatomically. 2-DP cells play an important role in the induction of hair morphogenesis in ESS. | [83] |
| Mouse Dermal Papilla (DP) cells and human keratinocytes in grafting chambers | In vivo study -Versican-GFP transgenic mice -Nude mice (bulb/c, nu/nu) | DP cells were isolated from skin of Versican-GPF transgenic mice. Keratinocytes were isolated from human scalps tissues and neonatal foreskins. Human epidermal cells and DP cell fractions (containing 1 × 106–107 cells of each) were transferred to grafting chambers implanted on the dorsal skins of nude mice (bulb/c, nu/nu). Chambers were removed 1 week after grafting, and hair follicle formation was assessed at 3–4 weeks. | 1-DP cells were able to induce hair follicles together with the epidermal component but only when both epithelial and mesenchymal components were present. Hair pegs were formed a week after grafting. 2-When the number of epidermal cells was reduced to 1 × 106 cells (10% of DP cells), the efficiency of hair follicle reconstitution was mostly unchanged. 3-When human keratinocytes were included, hair follicle-like structures were formed at the graft sites 4 weeks later and innermost regions of the structures were clearly keratinized. 4-Human adult cells also have the same ability to differentiate into follicular keratinocytes as neonatal foreskin-derived epidermal cells. | 1-Results show that hair follicle-like structures consisting of human keratinocytes and murine mesenchymal cells are generated. 2-Epithelial-mesenchymal interactions exists between human and mouse cells. | [84] |
| Mouse dermal and human epidermal cells. Patch assay | In vivo study | Mouse dermal and epidermal cells were freshly isolated from C57BL/6 used for control experiments. Human dermal papilla (DP) spheres (104 cells) were prepared from two-dimensional (2D) cultured DP cells using either low cell-binding plate or hydrocell plate and combined with freshly isolated mouse epidermal cells for implantation A total of 200 DP spheres (2 × 106 cells) prepared from human DP cells were mixed with fresh mouse epidermal cells (2 × 106 cells) and implanted. 50 DP spheres (5 × 105 cells) prepared from human DP cells were mixed with fresh mouse epidermal cells (5 × 105 cells) and implanted. Mice were killed 2 weeks after cell implantation in order to verify hair follicle induction. | 1-Hair follicle was observed in positive control experiments with mouse dermal and epidermal cells. 2-Hair follicle formation was observed when human DP spheres from various passages of culture were mixed with new born mouse epidermal cells. 3-Hair follicles were never observed when 2D cultures from the same population were use 4-The morphology and size of hair follicles induced by human DP spheres resembled the ones induced by mouse dermal cells. | 1-Using a reconstitution assay, sphere formation increases the ability of cultured human DP cells to induce hair follicles from mouse epidermal cells | [85] |
| Human Dermal Papilla (DP) cells embedded into rat tail collagen type 1 and neonatal foreskin keratinocytes (NFK) Dermal-epidermal composites (DEC) | In vivo study -Nude mice | Human DP cells were isolated from temporal scalp dermis. DECs were constructed by combining DP cells with rat tail collagen type 1, adding NFKs on top and bringing the constructs to the air-liquid interface for 2 days before grafting onto female nude mice. | 1-Alkaline phosphatase activity was variable between samples, with cells from 3 of the donors showing alkaline phosphatase activity in more than 50% of the cells. 2-8 weeks after grafting, hair follicles (HFs) were observed in mice grafted with the 3 human DP cells with higher alkaline phosphatase activity. 3-HFs had a bulb, dermal sheath, hair matrix and cortex 4-Cells in the region of the DP and displayed alkaline phosphatase activity, normal reactivity with specific antibodies to human nestin and versican. 5-Basal layer of the outer root sheath was immunoreactive for keratin 15. | 1-Cultured specialized human cells such as DP cells can induce complete pilosebaceous units in vivo in the grafted DEC model. | [86] |
| Cell Types and Scaffold | Type of Study | Experimental Design | Results | Conclusions | Reference |
|---|---|---|---|---|---|
| Mouse embryonic skin epidermal and dermal cells. Cell aggregates | In vitro and in vivo study -C57BL/6 mice -Male CD-1 nude albino mice | Mixed dermal and epidermal cells (keratinocytes and melanocytes) were removed from embryonic day 18 C57BL/6 mice. Aggregates were formed using the hanging droplet method. 2 × 106 cells/mL were hanging droplet in 20 µL. Aggregate formation was completed within 18–20 h. To form proto-hairs, aggregates were transferred individually to wells of a 96-well round-bottom plate. Wells were precoated with 0.24% methylcellulose medium to prevent adherence of proto-hairs. A single cultured aggregate (proto-hair) was grafted into the ear of male CD-1 nude mice. Regrowth of neo-generated hairs was regularly monitored. | 1-Few days after aggregate formation (4-7 days), hair-like structures started forming. 2-The frequency of hair morphogenesis ranged from 66 to 100% among numerous experiments. 3-Proto-hairs could undergo further maturation in vivo. 4-Within 2 weeks, black-pigmented hair fibres appeared. 5-Within 4 weeks after implantation, approximately 50–60% of implants developed follicles with pigmented hair shafts. 6-Hairs developed from implanted proto-hairs were well anchored in the skin and persisted for at least 6 months 7-Implanted proto-hairs were able to maintain their growth for many months. | 1-In vitro incubation of mixed follicular cell aggregates leads to the formation of partially developed follicle-like structures called proto-hairs. 2-Upon implantation, proto-hairs fully develop into normal hairs that persist and grow indefinitely. | [80] |
| Mouse, rat and human Dermal Papilla (DP) cells. Spheres | In vitro and in vivo studies -Versican-GFP-transgenic (versican-GFP-Tg) mice -C57BL/6J -Wistar rats -Athymic nude mice | DP cells were dissected from mouse vibrissae follicles (versican-GFP-Tg) and 104 DP cells were aggregated to form one spherical structure and maintained for 2 to 12 days. Epidermal and DP cells were dissected from C57BL/6J mouse. Fifty of the DP spheres or 5 × 105 of the dissociated DP cells were combined with 5 × 105 epidermal cells. Cells were injected subcutaneously into athymic nude mice. Mice were killed 2 weeks after cellimplantation to verify hair follicle induction | 1-Hair follicle induction could occur even from DPspheres of cells after 26 passages 2-DP cell suspensions from more than 8 passages, however, could not induce hair follicles. | 1-DP have highly aggregative properties compared withskin fibroblasts 2-DP spheres may have recovered their aggregative property by increasing their versican content, which helped them to interact with epidermal cells and induce hair follicles. | [87] |
| DP cells were isolated by scissors and forceps from cheeks of Wistar rats- 104, 2 × 104, 4 × 104, 8 × 104 and 1.6 × 105 cells were added to each well, containing poly-(ethylene-co-vinyl alcohol) (EVAL). DP microtissues were characterized in vitro and In vivo. | 1-Formation of DP microtissues on EVAL is affected by seeding cell numbers. 2-After 5 days of culture, dense microtissues were observed on EVAL at the seeding numbers of 8 × 104 cells/well or higher. 3-About 47 microtissues (diameter > 125 mm) were obtained on an EVAL surface of 1.9 cm2 with a single seeding of 1.6 × 105 DP cells. 4-Microtissues had a spheroidal structure. 5-Cell viability in DP microtissues on EVAL is much higher than that in DP spheroids generated by hanging drop method. | 1-Self-assembly of DP cells into spheroidal inductive microtissues can be facilitated when cells are seeded at appropriate densities on EVAL surface. 2- DP microtissues mixed with newborn mouse epidermal cells and injected into the hypodermis of nude mice are able to induce new hair follicles (HFs). | [88] | ||
| Human DP cells were isolated from scalp tissues obtained from plastic surgery. Human DPCs were expanded under conditions of activation of the Wnt/β-catenin signalling pathway (GSK-3 inhibitor, 6-bromoindirubin-3′-oxime (BIO)). hDP cells were analysed in the presence or absence of BIO. A cellular grafting assay to evaluate the hair-inducing ability of cultured human DP cells (5 × 106 cells/grafting) was engrafted on the dorsal skins of nude mice (bulb/c, nu/nu) | 1-Protein level of LEF1 in BIO-treated hDP cells showed a 2.7-fold increase compared with that without BIO. 2-Nuclear β-catenin was evidently observed in BIO-treated human DP cells 3-Human DP cells cultured in the presence of BIO and transplanted with murine epidermal cell fraction, formed hair follicle–like structures. | 1-Human DP cells cultured under Wnt/β-catenin signalling activation by GSK-3β inhibition maintained the expression level of DP marker genes. 2-Human DP cells showed constant hair induction when transplanted with murine epidermal cell fraction. | [89] | ||
| Primary human interfollicular dermal fibroblasts, hair follicle dermal papilla, or hair follicle dermal sheath cells into rat tail collagen and human keratinocytes | In vitro and in vivo studies -SCID mouse | Cells were isolated from occipital scalp In vitro Constructs of collagen were established in parallel from fibroblasts, dermal papilla cells and dermal sheath cells. After 7 days, keratinocytes added and cultured for 17 days. In vivo Dermal fibroblasts, dermal papilla cells, dermal sheath cells and keratinocytes were trypsinized. 107 million keratinocytes were then combined with either 107 fibroblasts, or 107 dermal papilla cells and pipetted into a hole located on a silicone chamber previously implanted under de dorsal skin of a SCID mouse. After one week the silicone chamber was removed, and cells were left for a further two weeks. | In vitro 1-There were no obvious differences in the equivalents containing hair follicle dermal cells, when compared to a fibroblast support layer. 2-Hair follicle dermal cells were capable of supporting growth and differentiation of overlying epidermal cells. 3-Type IV collagen (COL4) labeling was most intense in the basement membrane of constructs supported by dermal sheath cells from the hair follicle. In vivo 1-After grafting, all mixed cells were organized to form a skin structure. After 3 weeks, cells had reorganized with dermal cells on the inner surface, and keratinocytes exposed to the external air interface | 1-Human hair follicle dermal cells can be readily interchanged with interfollicular fibroblasts and used as an alternative cell source for establishing the dermal component of engineered skin both in vitro and in vivo. | [90,91] |
| Human dermal papilla (DP) and dermal sheath (DS) cells and epithelial cells into collagen gel. Organotypic culture | In vitro and in vivo study -Nude mice | DP cells and DS cells of the human hair follicles (HFs) were isolated from human scalp. Dermal fibroblast (FB) from the scalp skin or foreskin was cultured separately as routine method. Keratinocytes from interfollicular and follicle outer root sheath (ORS superior or inferior) were also isolated from human scalp. DP cells, DS cells or FBs were embedded onto collagen gel and 5 × 105 epithelial cells were plated on the prepared mesenchymal cell-populated collagen gels (Organotypic culture). Gels were immersed for 2 weeks and then were transplanted on the dorsal skin of the nude mice for 1 month. | 1-Compared with the free cell-populated gels, on the four types of mesenchymal cell-populated gels, the growth, differentiation and shape of interfollicular keratinocytes and ORS epithelial cells were remarkably improved. 2-Epidermal structure reformed by the interfollicular keratinocytes on four types of mesenchymal cell-populated was differentiated very well and was similar to the epidermis of nature live skin. 3-Epidermis reformed by the superior ORS cells and the interfollicular keratinocytes was the thickest while the epidermis reformed only 3–4 layers by the bulb matrical cells was the thinnest. 4-In the organotypic culture of superior + DS and inferior + DS cells, the ORS cells reformed a long-shape hair follicle structure that was seen under microscope. | 1-Dermal papilla cells induced superior and inferior epithelial cells to form hair follicle on nude mice. 2-Low passage dermal papilla cells mixed with hair follicle epithelial cells reformed many typical hair follicle structures and produced hair fibres after transplantation on nude mice. 3-Dermal part of hair follicle, such as dermal papilla cells and dermal sheath cells, has the ability to induce hair follicle formation by interaction with the epithelial cells of hair follicle. | [92] |
| Human dermal papilla (DP) cells, hair follicle enriched primary cultures (HFSCs) and immortalized human bulge stem cell line Tel-E6E7 and dermal fibroblasts (DF) on acellular dermal matrices Tissue engineered skin | In vitro and in vivo study -BALB/C nude mice | -DP cells and DF cells were isolated from occipital human scalp and HFSC cells were isolated from human skin biopsies. -Immortalized human bulge stem cell line Tel-E6E7 was acquired. -Porcine acellular dermal matrices (ADMs), were seeded with 5 × 105 cm2 DF or DP cells as the dermal cellular component. They were cultured for 7 days. After that, matrices were inverted and seeded on the opposite side with 5 × 105 cm2 HFSCs or Tel-E6E7 as the epidermal cellular and cultured for 21 days (combining liquid and air-liquid phases). -Tissue engineered skins were grafted int the back of BALB/C nude mice aged 8 weeks (n = 6 per group): one type of construct was seeded with HFSCs and DPCs (HFSC-DPC), a second type was seeded with HFSCs and DFs (HFSC-DF), and a third type was seeded exclusively with HFSCs without the dermal component. -Grafts were analysed at 14, 30 and 70 days. | 1-Air-liquid interphase improved in vitro skin constructs: skin constructs with HFSCs alone or with DFs (HFSC-DF) showed an epidermis with a proliferative basal layer, incipient and irregular stratum spinosum, frequent dyskeratosis, and a cornified layer. In the HFSC-DPC skin constructs, most regular epidermis was observed. 2-In vitro skin constructs with DPCs showed a higher number of p63-Positive epidermal basal cells and epidermal invaginations. 3-Presence of DPCs favoured the graft-take of composite skin and improved the wound healing process. 4-After 14 days of grafting, the HFSC-DPC constructs showed the highest number of blood neo-vessels. Amount of VEGF was six fold higher than the amount in the DF cultures (927 ± 87 pg/mL vs. 147 ± 54 pg/mL). 5-Only the HFSC-DPC constructs grafted in the nude mice showed notable epithelial cyst-like inclusions in the remodelling dermis and expression of k6hf. | 1-Presence of DP cells in composite skin constructs generated in air-liquid interphase led to the formation of an epidermal-like structure with the most regular stratification, more invaginations that could indicate hair follicle neogenesis attempts, and maintenance of an epidermal stem cell pool. | [93] |
| Human keratinocytes, fibroblasts, dermal papilla cells and GFP-tagged HUVECs. Human skin constructs (HSCs) in type I collagen gel. | In vitro and in vivo study -Male immunodeficient nude mice (athymic nude, Crl:NU(NCr)-Foxn1nu | -Neonatal dermal keratinocytes and fibroblasts were isolated from human foreskin. -GFP-tagged HUVECs were also cultured. -Dermal Papilla (DP) cells discarded scalp tissues from hair restoration surgery. -The molds with varying hair densities (19, 81, 255 hair follicles (HFs) per cm2) were 3D-printed. -3D skin constructs were generated with a type I collagen matrix as dermal compartment. 1.25 × 105 fibroblasts/mL were added and polymerized around the 3D-printed HF molds. -After complete polymerization, the molds were removed and 3000 DP cells per microwell were added on top of the gel. The constructs were cultured overnight and then, 106 keratinocytes (KCs) were added on top of the gel. -Constructs were cultured for 1–3 weeks. -GFP-tagged HUVECs were also studied in collagen type I gel at a concentration of 2 × 106 cells/mL. -Human skin constructs were grafted onto male immunodeficient nude mice (athymic nude, Crl:NU(NCr)-Foxn1nu. They were maintained 5 days into a silicone chamber and then maintained for 4–6 weeks for in vivo analysis. | 1-3D-printed technology allowed for precise control of DP cells aggregate size by adjusting the diameter of the microwells. The spontaneous aggregate formation restored the expression of versican (VCAN) and alkaline phosphatase (ALP) activity and suppressed the expression of smooth muscle actin (SMA). 2-Keratinocytes (KCs) over the dermal constructs and allowed the cells to settle down resembled a hair follicle-like unit (HFU) 3-Culture period of 3 weeks led to the elongation of the HFs down into the dermis and a better organization of the inner and outer root sheath layers. 4-Transfection of Lef-1 in cultured DP cells, and later formation of spheroids restored the intact DP cells gene signature. Skin constructs generated with them resulted in significant increase (up to 13-fold) in the expression of the specific hair lineage genes, including the outer and inner root sheath (K17, K71, K25) and hair companion and medulla markers (K75). 5-To generate a vascular bed, GFP-tagged human umbilical vein endothelial cells (HUVECs) in the dermis of HSCs were included together with the dermal fibroblasts. 6-Immunofluorescent wholemount imaging of the constructs revealed that these capillary-like structures were near to the HFs. 7- Grafting the vascularized HSCs onto mice promoted host vascularization into the grafts 8-Four to five weeks after grafting, vascularized HSCs presented substantial hair growth in the grafts, whereas the HSCs prepared with FB aggregates did not induce hair formation. | 1-This tissue engineering exploits the epidermal-mesenchymal interactions during hair development while synthetically guiding the physiological conformation and reconstituting the gene signature of cultured cells to induce human hair growth in vitro and in mice. | [94] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinez-Lopez, A.; Montero-Vilchez, T.; Sierra-Sánchez, Á.; Molina-Leyva, A.; Arias-Santiago, S. Advanced Medical Therapies in the Management of Non-Scarring Alopecia: Areata and Androgenic Alopecia. Int. J. Mol. Sci. 2020, 21, 8390. https://doi.org/10.3390/ijms21218390
Martinez-Lopez A, Montero-Vilchez T, Sierra-Sánchez Á, Molina-Leyva A, Arias-Santiago S. Advanced Medical Therapies in the Management of Non-Scarring Alopecia: Areata and Androgenic Alopecia. International Journal of Molecular Sciences. 2020; 21(21):8390. https://doi.org/10.3390/ijms21218390
Chicago/Turabian StyleMartinez-Lopez, Antonio, Trinidad Montero-Vilchez, Álvaro Sierra-Sánchez, Alejandro Molina-Leyva, and Salvador Arias-Santiago. 2020. "Advanced Medical Therapies in the Management of Non-Scarring Alopecia: Areata and Androgenic Alopecia" International Journal of Molecular Sciences 21, no. 21: 8390. https://doi.org/10.3390/ijms21218390
APA StyleMartinez-Lopez, A., Montero-Vilchez, T., Sierra-Sánchez, Á., Molina-Leyva, A., & Arias-Santiago, S. (2020). Advanced Medical Therapies in the Management of Non-Scarring Alopecia: Areata and Androgenic Alopecia. International Journal of Molecular Sciences, 21(21), 8390. https://doi.org/10.3390/ijms21218390









