How Many Messenger RNAs Can Be Translated by the START Mechanism?
Abstract
:1. Introduction
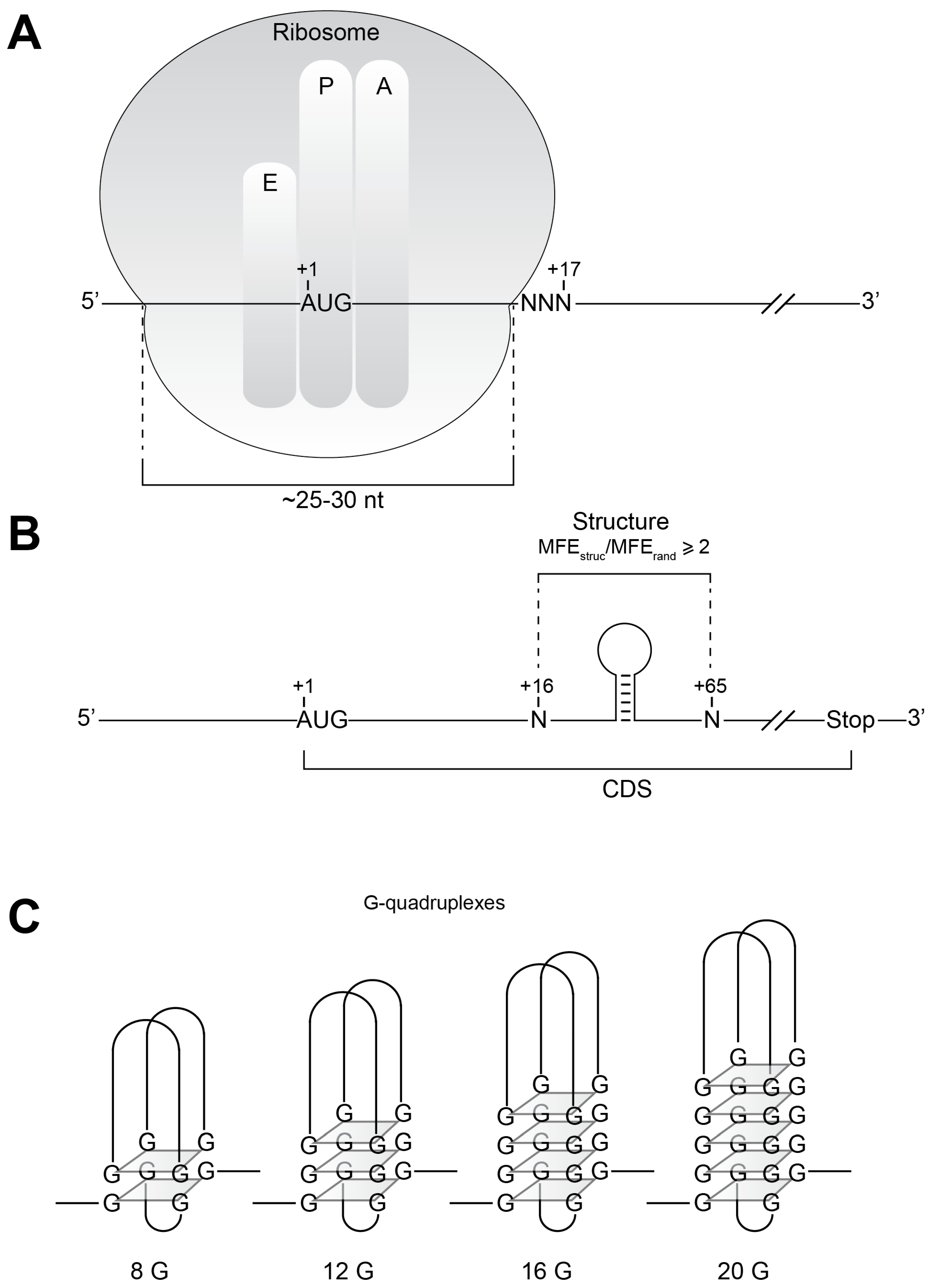
2. Results and Discussion
3. Materials and Methods
Data Processing
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rodnina, M.V. Translation in Prokaryotes. Cold Spring Harb. Perspect. Biol. 2018, 10, a032664. [Google Scholar] [CrossRef] [PubMed]
- Tollerson, R.; Ibba, M. Translational regulation of environmental adaptation in bacteria. J. Biol. Chem. 2020, 295, 10434–10445. [Google Scholar] [CrossRef] [PubMed]
- Hinnebusch, A.G.; Ivanov, I.P.; Sonenberg, N. Translational control by 5’-untranslated regions of eukaryotic mRNAs. Science 2016, 352, 1413–1416. [Google Scholar] [CrossRef] [PubMed]
- Hinnebusch, A.G. Structural Insights into the Mechanism of Scanning and Start Codon Recognition in Eukaryotic Translation Initiation. Trends Biochem. Sci. 2017, 42, 589–611. [Google Scholar] [CrossRef] [PubMed]
- Aylett, C.H.S.; Ban, N. Eukaryotic aspects of translation initiation brought into focus. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160186. [Google Scholar] [CrossRef] [Green Version]
- Akirtava, C.; McManus, J. Control of translation by eukaryotic mRNA transcript leaders—Insights from high-throughput assays and computational modeling. Wiley Interdiscip. Rev. RNA 2020, e1623. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Pelletier, J. General and Target-Specific DExD/H RNA Helicases in Eukaryotic Translation Initiation. Int. J. Mol. Sci. 2020, 21, 4402. [Google Scholar] [CrossRef]
- Pelletier, J.; Sonenberg, N. The Organizing Principles of Eukaryotic Ribosome Recruitment. Annu. Rev. Biochem. 2019, 88, 307–335. [Google Scholar] [CrossRef]
- Merrick, W.C.; Pavitt, G.D. Protein Synthesis Initiation in Eukaryotic Cells. Cold Spring Harb. Perspect. Biol. 2018, 10, a033092. [Google Scholar] [CrossRef]
- Shirokikh, N.E.; Preiss, T. Translation initiation by cap-dependent ribosome recruitment: Recent insights and open questions. Wiley Interdiscip. Rev. RNA 2018, 9, e1473. [Google Scholar] [CrossRef]
- Schmitt, E.; Coureux, P.-D.; Monestier, A.; Dubiez, E.; Mechulam, Y. Start Codon Recognition in Eukaryotic and Archaeal Translation Initiation: A Common Structural Core. Int. J. Mol. Sci. 2019, 20, 939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brion, P.; Westhof, E. Hierarchy and dynamics of RNA folding. Annu. Rev. Biophys. Biomol. Struct. 1997, 26, 113–137. [Google Scholar] [CrossRef]
- Batey, R.T.; Rambo, R.P.; Doudna, J.A. Tertiary Motifs in RNA Structure and Folding. Angew. Chem. Int. Ed. 1999, 38, 2326–2343. [Google Scholar] [CrossRef]
- Pleij, C.W.A.; Rietveld, K.; Bosch, L. A new principle of RNA folding based on pseudoknotting. Nucleic Acids Res. 1985, 13, 1717–1731. [Google Scholar] [CrossRef] [PubMed]
- Kwok, C.K.; Marsico, G.; Balasubramanian, S. Detecting RNA G-Quadruplexes (rG4s) in the Transcriptome. Cold Spring Harb. Perspect. Biol. 2018, 10, a032284. [Google Scholar] [CrossRef]
- Gualerzi, C.O.; Pon, C.L. Initiation of mRNA translation in bacteria: Structural and dynamic aspects. Cell. Mol. Life Sci. 2015, 72, 4341–4367. [Google Scholar] [CrossRef] [Green Version]
- Mailliot, J.; Martin, F. Viral internal ribosomal entry sites: Four classes for one goal. Wiley Interdiscip. Rev. RNA 2017, 9, e1458. [Google Scholar] [CrossRef]
- Mearls, E.B.; Jackter, J.; Colquhoun, J.M.; Farmer, V.J.; Matthews, A.J.; Murphy, L.S.; Fenton, C.; Camp, A. Transcription and translation of the sigG gene is tuned for proper execution of the switch from early to late gene expression in the developing Bacillus subtilis spore. PLoS Genet. 2018, 14, e1007350. [Google Scholar] [CrossRef]
- Masachis, S.; Darfeuille, F. Type I Toxin-Antitoxin Systems: Regulating Toxin Expression via Shine-Dalgarno Sequence Sequestration and Small RNA Binding. In Regulating with RNA in Bacteria and Archaea; American Society for Microbiology: Washington, DC, USA, 2018; Volume 6, pp. 171–190. [Google Scholar]
- Masachis, S.; Tourasse, N.J.; Lays, C.; Faucher, M.; Chabas, S.; Iost, I.; Darfeuille, F. A genetic selection reveals functional metastable structures embedded in a toxin-encoding mRNA. eLife 2019, 8. [Google Scholar] [CrossRef]
- Duval, M.; Korepanov, A.; Fuchsbauer, O.; Fechter, P.; Haller, A.; Fabbretti, A.; Choulier, L.; Micura, R.; Klaholz, B.P.; Romby, P.; et al. Escherichia coli ribosomal protein S1 unfolds structured mRNAs onto the ribosome for active translation initiation. PLoS Biol. 2013, 11, e1001731. [Google Scholar] [CrossRef] [Green Version]
- Byrgazov, K.; Grishkovskaya, I.; Arenz, S.; Coudevylle, N.; Temmel, H.; Wilson, D.N.; Djinović-Carugo, K.; Moll, I. Structural basis for the interaction of protein S1 with the Escherichia coli ribosome. Nucleic Acids Res. 2014, 43, 661–673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiaruttini, C.; Guillier, M. On the role of mRNA secondary structure in bacterial translation. Wiley Interdiscip. Rev. RNA 2020, 11, e1579. [Google Scholar] [CrossRef] [PubMed]
- Scharff, L.B.; Childs, L.; Walther, D.; Bock, R. Local Absence of Secondary Structure Permits Translation of mRNAs that Lack Ribosome-Binding Sites. PLoS Genet. 2011, 7, e1002155. [Google Scholar] [CrossRef] [Green Version]
- Accetto, T.; Avguštin, G. Inability of Prevotella bryantii to Form a Functional Shine-Dalgarno Interaction Reflects Unique Evolution of Ribosome Binding Sites in Bacteroidetes. PLoS ONE 2011, 6, e22914. [Google Scholar] [CrossRef]
- Leppek, K.; Das, R.; Barna, M. Functional 5′ UTR mRNA structures in eukaryotic translation regulation and how to find them. Nat. Rev. Mol. Cell Biol. 2018, 19, 158–174. [Google Scholar] [CrossRef]
- Pesole, G.; Mignone, F.; Gissi, C.; Grillo, G.; Licciulli, F.; Liuni, S. Structural and functional features of eukaryotic mRNA untranslated regions. Gene 2001, 276, 73–81. [Google Scholar] [CrossRef]
- Lynch, M.; Scofield, D.G.; Hong, X. The Evolution of Transcription-Initiation Sites. Mol. Biol. Evol. 2005, 22, 1137–1146. [Google Scholar] [CrossRef] [Green Version]
- Mignone, F.; Gissi, C.; Liuni, S.; Pesole, G. Untranslated regions of mRNAs. Genome Biol. 2002, 3, reviews0004-1. [Google Scholar] [CrossRef]
- Jan, C.H.; Friedman, R.C.; Ruby, J.G.; Bartel, D.P. Formation, regulation and evolution of Caenorhabditis elegans 3′UTRs. Nat. Cell Biol. 2011, 469, 97–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hinnebusch, A.G. The Scanning Mechanism of Eukaryotic Translation Initiation. Annu. Rev. Biochem. 2014, 83, 779–812. [Google Scholar] [CrossRef]
- Archer, S.K.; Shirokikh, N.E.; Beilharz, T.H.; Preiss, T. Dynamics of ribosome scanning and recycling revealed by translation complex profiling. Nat. Cell Biol. 2016, 535, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Shirokikh, N.E.; Dutikova, Y.S.; Staroverova, M.A.; Hannan, R.D.; Preiss, T. Migration of small ribosomal subunits on the 5’ untranslated regions of capped messenger RNA. Int. J. Mol. Sci. 2019, 20, 4464. [Google Scholar] [CrossRef] [Green Version]
- Qu, X.; Wen, J.-D.; Lancaster, L.; Noller, H.F.; Bustamante, C.D.; Tinoco, I. The ribosome uses two active mechanisms to unwind messenger RNA during translation. Nat. Cell Biol. 2011, 475, 118–121. [Google Scholar] [CrossRef] [PubMed]
- Takyar, S.; Hickerson, R.P.; Noller, H.F. mRNA Helicase Activity of the Ribosome. Cell 2005, 120, 49–58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parsyan, A.; Svitkin, Y.V.; Shahbazian, D.; Gkogkas, C.G.; Lasko, P.; Merrick, W.C.; Sonenberg, N. mRNA helicases: The tacticians of translational control. Nat. Rev. Mol. Cell Biol. 2011, 12, 235–245. [Google Scholar] [CrossRef]
- Jagodnik, J.; Chiaruttini, C.; Guillier, M. Stem-Loop Structures within mRNA Coding Sequences Activate Translation Initiation and Mediate Control by Small Regulatory RNAs. Mol. Cell 2017, 68, 158.e3–170.e3. [Google Scholar] [CrossRef] [Green Version]
- Eriani, G.; Martin, F. START: STructure-Assisted RNA Translation. RNA Biol. 2018, 15, 1250–1253. [Google Scholar] [CrossRef] [Green Version]
- Bhattacharyya, S.; Jacobs, W.M.; Adkar, B.V.; Yan, J.; Zhang, W.; Shakhnovich, E.I. Accessibility of the Shine-Dalgarno Sequence Dictates N-Terminal Codon Bias in E. coli. Mol. Cell 2018, 70, 894.e5–905.e5. [Google Scholar] [CrossRef] [Green Version]
- Boël, G.; Letso, R.; Neely, H.; Price, W.N.; Wong, K.-H.; Su, M.; Luff, J.D.; Valecha, M.; Everett, J.K.; Acton, T.B.; et al. Codon influence on protein expression in E. coli correlates with mRNA levels. Nat. Cell Biol. 2016, 529, 358–363. [Google Scholar] [CrossRef] [Green Version]
- Goodman, D.B.; Church, G.M.; Kosuri, S. Causes and Effects of N-Terminal Codon Bias in Bacterial Genes. Science 2013, 342, 475–479. [Google Scholar] [CrossRef] [Green Version]
- Kudla, G.; Murray, A.W.; Tollervey, D.; Plotkin, J.B. Coding-Sequence Determinants of Gene Expression in Escherichia coli. Science 2009, 324, 255–258. [Google Scholar] [CrossRef] [Green Version]
- Wolin, S.L.; Walter, P. Ribosome pausing and stacking during translation of a eukaryotic mRNA. EMBO J. 1988, 7, 3559–3569. [Google Scholar] [CrossRef]
- Steitz, J.A. Polypeptide Chain Initiation: Nucleotide Sequences of the Three Ribosomal Binding Sites in Bacteriophage R17 RNA. Nat. Cell Biol. 1969, 224, 957–964. [Google Scholar] [CrossRef]
- Shao, X.; Zhang, W.; Umar, M.I.; Wong, H.Y.; Seng, Z.; Xie, Y.; Zhang, Y.; Yang, L.; Kwok, C.K.; Deng, X. RNA G-Quadruplex Structures Mediate Gene Regulation in Bacteria. mBio 2020, 11. [Google Scholar] [CrossRef] [Green Version]
- Murat, P.; Marsico, G.; Herdy, B.; Ghanbarian, A.T.; Portella, G.; Balasubramanian, S. RNA G-quadruplexes at upstream open reading frames cause DHX36- and DHX9-dependent translation of human mRNAs. Genome Biol. 2018, 19, 1–24. [Google Scholar] [CrossRef]
- Yang, S.Y.; Lejault, P.; Chevrier, S.; Boidot, R.; Robertson, A.G.; Wong, J.M.Y.; Monchaud, D. Transcriptome-wide identification of transient RNA G-quadruplexes in human cells. Nat. Commun. 2018, 9, 1–11. [Google Scholar] [CrossRef]
- Crothers, D.; Cole, P.; Hilbers, C.; Shulman, R. The molecular mechanism of thermal unfolding of Escherichia coli formylmethionine transfer RNA. J. Mol. Biol. 1974, 87, 63–88. [Google Scholar] [CrossRef]
- Porschke, D. Thermodynamic and kinetic parameters of an oligonucleotide hairpin helix. Biophys. Chem. 1974, 1, 381–386. [Google Scholar] [CrossRef]
- Kennell, D.; Riezman, H. Transcription and translation initiation frequencies of the Escherichia coli lac operon. J. Mol. Biol. 1977, 114, 1–21. [Google Scholar] [CrossRef]
- Lorenz, R.; Bernhart, S.H.F.; Zu Siederdissen, C.H.; Tafer, H.; Flamm, C.; Stadler, P.F.; Hofacker, I.L. ViennaRNA Package 2.0. Algorithms Mol. Biol. 2011, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Del Campo, C.; Bartholomäus, A.; Fedyunin, I.; Ignatova, Z. Secondary Structure across the Bacterial Transcriptome Reveals Versatile Roles in mRNA Regulation and Function. PLoS Genet. 2015, 11, e1005613. [Google Scholar] [CrossRef] [Green Version]
- Burkhardt, D.H.; Rouskin, S.; Zhang, Y.; Li, G.-W.; Weissman, J.S.; A Gross, C. Operon mRNAs are organized into ORF-centric structures that predict translation efficiency. eLife 2017, 6, e22037. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Burkhardt, D.H.; Rouskin, S.; Li, G.-W.; Weissman, J.S.; Gross, C.A. A Stress Response that Monitors and Regulates mRNA Structure Is Central to Cold Shock Adaptation. Mol. Cell 2018, 70, 274.e7–286.e7. [Google Scholar] [CrossRef] [Green Version]
- Mustoe, A.M.; Busan, S.; Rice, G.M.; Hajdin, C.E.; Peterson, B.K.; Ruda, V.M.; Kubica, N.; Nutiu, R.; Baryza, J.L.; Weeks, K.M. Pervasive Regulatory Functions of mRNA Structure Revealed by High-Resolution SHAPE Probing. Cell 2018, 173, 181.e18–195.e18. [Google Scholar] [CrossRef] [Green Version]
- Kozak, M. At least six nucleotides preceding the AUG initiator codon enhance translation in mammalian cells. J. Mol. Biol. 1987, 196, 947–950. [Google Scholar] [CrossRef]
- Kozak, M. An analysis of 5’-noncoding sequences from 699 vertebrate messenger rNAS. Nucleic Acids Res. 1987, 15, 8125–8148. [Google Scholar] [CrossRef] [Green Version]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Despons, L.; Martin, F. How Many Messenger RNAs Can Be Translated by the START Mechanism? Int. J. Mol. Sci. 2020, 21, 8373. https://doi.org/10.3390/ijms21218373
Despons L, Martin F. How Many Messenger RNAs Can Be Translated by the START Mechanism? International Journal of Molecular Sciences. 2020; 21(21):8373. https://doi.org/10.3390/ijms21218373
Chicago/Turabian StyleDespons, Laurence, and Franck Martin. 2020. "How Many Messenger RNAs Can Be Translated by the START Mechanism?" International Journal of Molecular Sciences 21, no. 21: 8373. https://doi.org/10.3390/ijms21218373
APA StyleDespons, L., & Martin, F. (2020). How Many Messenger RNAs Can Be Translated by the START Mechanism? International Journal of Molecular Sciences, 21(21), 8373. https://doi.org/10.3390/ijms21218373





