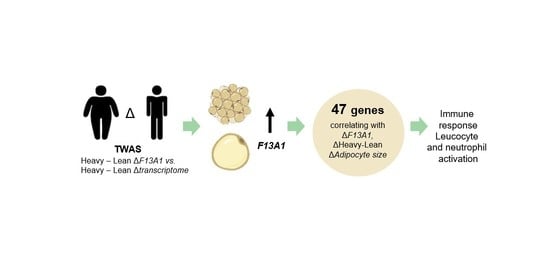
Transglutaminases and Obesity in Humans: Association of F13A1 to Adipocyte Hypertrophy and Adipose Tissue Immune Response
Abstract
:1. Introduction
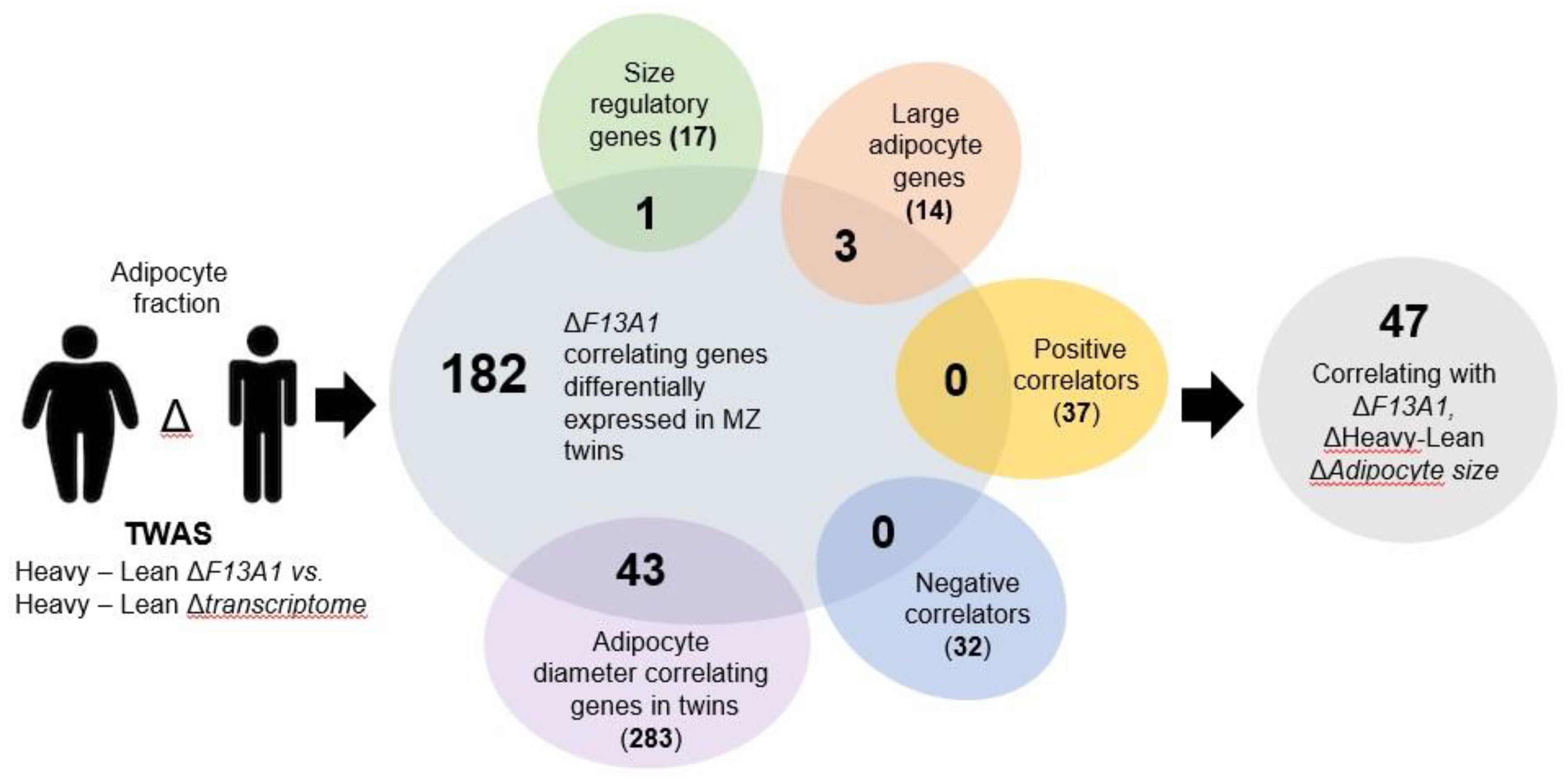
2. Results
3. Discussion
4. Materials and Methods
4.1. Weight-Discordant MZ Twins
4.2. Adipose Tissue Biopsies, Adipocyte and Tissue Preparations, and RNA Extraction
4.3. Adipocyte Morphology
4.4. Affymetrix Transcriptomics
4.5. Transcriptome-Wide Association Study, Statistics, and Bioinformatics
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Eckert, R.L.; Kaartinen, M.T.; Nurminskaya, M.; Belkin, A.M.; Colak, G.; Johnson, G.V.W.; Mehta, K. Transglutaminase Regulation of Cell Function. Physiol. Rev. 2014, 94, 383–417. [Google Scholar] [CrossRef] [Green Version]
- Iismaa, S.E.; Mearns, B.M.; Lorand, L.; Graham, R.M. Transglutaminases and Disease: Lessons from Genetically Engineered Mouse Models and Inherited Disorders. Physiol. Rev. 2009, 89, 991–1023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muszbek, L.; Bereczky, Z.; Bagoly, Z.; Komáromi, I.; Katona, É. Factor XIII: A Coagulation Factor with Multiple Plasmatic and Cellular Functions. Physiol. Rev. 2011, 91, 931–972. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lorand, L.; Graham, R.M. Transglutaminases: Crosslinking enzymes with pleiotropic functions. Nat. Rev. Mol. Cell Biol. 2003, 4, 140–156. [Google Scholar] [CrossRef] [PubMed]
- Forsprecher, J.; Wang, Z.; Goldberg, H.A.; Kaartinen, M.T. Transglutaminase-mediated oligomerization promotes osteoblast adhesive properties of osteopontin and bone sialoprotein. Cell Adhes. Migr. 2011, 5, 65–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaartinen, M.T.; Pirhonen, A.; Linnala-Kankkunen, A.; Mäenpää, P.H. Cross-linking of Osteopontin by Tissue Transglutaminase Increases Its Collagen Binding Properties. J. Biol. Chem. 1999, 274, 1729–1735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nelea, V.; Nakano, Y.; Kaartinen, M.T. Size Distribution and Molecular Associations of Plasma Fibronectin and Fibronectin Crosslinked by Transglutaminase. Protein J. 2008, 27, 223–233. [Google Scholar] [CrossRef]
- Kanchan, K.; Fuxreiter, M.; Fésüs, L. Physiological, pathological, and structural implications of non-enzymatic protein–protein interactions of the multifunctional human transglutaminase. Cell. Mol. Life Sci. 2015, 72, 3009–3035. [Google Scholar] [CrossRef] [PubMed]
- Kaartinen, M.T.; El-Maadawy, S.; Räsänen, N.H.; McKee, M.D. Tissue Transglutaminase and Its Substrates in Bone. J. Bone Miner. Res. 2002, 17, 2161–2173. [Google Scholar] [CrossRef] [PubMed]
- Mousa, A.; Cui, C.; Song, A.; Myneni, V.D.; Sun, H.; Li, J.J.; Murshed, M.; Melino, G.; Kaartinen, M.T. Transglutaminases factor XIII-A and TG2 regulate resorption, adipogenesis and plasma fibronectin homeostasis in bone and bone marrow. Cell Death Differ. 2017, 24, 844–854. [Google Scholar] [CrossRef] [Green Version]
- Nurminskaya, M. Transglutaminases in mineralized tissues. Front. Biosci. 2006, 11, 1591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Jallad, H.F.; Myneni, V.D.; Piercy-Kotb, S.A.; Chabot, N.; Mulani, A.; Keillor, J.W.; Kaartinen, M.T. Plasma Membrane Factor XIIIA Transglutaminase Activity Regulates Osteoblast Matrix Secretion and Deposition by Affecting Microtubule Dynamics. PLoS ONE 2011, 6, e15893. [Google Scholar] [CrossRef] [Green Version]
- Cui, C.; Wang, S.; Myneni, V.D.; Hitomi, K.; Kaartinen, M.T. Transglutaminase activity arising from Factor XIIIA is required for stabilization and conversion of plasma fibronectin into matrix in osteoblast cultures. Bone 2014, 59, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Heath, D.J.; Downes, S.; Verderio, E.; Griffin, M. Characterization of Tissue Transglutaminase in Human Osteoblast-like Cells. J. Bone Miner. Res. 2001, 16, 1477–1485. [Google Scholar] [CrossRef]
- Piercy-Kotb, S.A.; Mousa, A.; Al-Jallad, H.F.; Myneni, V.D.; Chicatun, F.; Nazhat, S.N.; Kaartinen, M.T. Factor XIIIA transglutaminase expression and secretion by osteoblasts is regulated by extracellular matrix collagen and the MAP kinase signaling pathway. J. Cell. Physiol. 2012, 227, 2936–2946. [Google Scholar] [CrossRef]
- Cristancho, A.G.; Lazar, M.A. Forming functional fat: A growing understanding of adipocyte differentiation. Nat. Rev. Mol. Cell Biol. 2011, 12, 722–734. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, S.; Saarinen, L.; Naukkarinen, J.; Rodríguez, A.; Frühbeck, G.; Hakkarainen, A.; Lundbom, J.; Lundbom, N.; Vuolteenaho, K.; Moilanen, E.; et al. Adipocyte morphology and implications for metabolic derangements in acquired obesity. Int. J. Obes. 2014, 38, 1423–1431. [Google Scholar] [CrossRef]
- Tandon, P.; Wafer, R.; Minchin, J.E.N. Adipose morphology and metabolic disease. J. Exp. Biol. 2018, 221, jeb164970. [Google Scholar] [CrossRef] [Green Version]
- Yin, X.; Lanza, I.R.; Swain, J.M.; Sarr, M.G.; Nair, K.S.; Jensen, M.D. Adipocyte Mitochondrial Function Is Reduced in Human Obesity Independent of Fat Cell Size. J. Clin. Endocrinol. Metab. 2014, 99, E209–E216. [Google Scholar] [CrossRef] [Green Version]
- Xiao, L.; Yang, X.; Lin, Y.; Li, S.; Jiang, J.; Qian, S.; Tang, Q.; He, R.; Li, X. Large adipocytes function as antigen-presenting cells to activate CD4+ T cells via upregulating MHCII in obesity. Int. J. Obes. 2015, 40, 112–120. [Google Scholar] [CrossRef] [Green Version]
- Engin, A.B. What Is Lipotoxicity? Adv. Exp. Med. Biol. 2017, 960, 197–220. [Google Scholar]
- Engin, A.B. Adipocyte-Macrophage Cross-Talk in Obesity. Adv. Exp. Med. Biol. 2017, 960, 327–343. [Google Scholar] [CrossRef] [PubMed]
- Kahn, S.E.; Hull, R.L.; Utzschneider, K.M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nat. Cell Biol. 2006, 444, 840–846. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Kusminski, C.M.; Scherer, P.E. Adipose tissue remodeling and obesity. J. Clin. Investig. 2011, 121, 2094–2101. [Google Scholar] [CrossRef] [Green Version]
- Myneni, V.D.; Melino, G.; Kaartinen, M.T. Transglutaminase 2—A novel inhibitor of adipogenesis. Cell Death Dis. 2015, 6, e1868. [Google Scholar] [CrossRef] [Green Version]
- Sághy, T.; Köröskényi, K.; Hegedűs, K.; Antal, M.; Bankó, C.; Bacsó, Z.; Papp, A.; Stienstra, R.; Szondy, Z. Loss of transglutaminase 2 sensitizes for diet-induced obesity-related inflammation and insulin resistance due to enhanced macrophage c-Src signaling. Cell Death Dis. 2019, 10, 439. [Google Scholar] [CrossRef] [Green Version]
- Naukkarinen, J.; Surakka, I.; Pietiläinen, K.H.; Rissanen, A.; Salomaa, V.; Ripatti, S.; Yki-Jarvinen, H.; Van Duijn, C.M.; Wichmann, H.-E.; Kaprio, J.; et al. Use of Genome-Wide Expression Data to Mine the “Gray Zone” of GWA Studies Leads to Novel Candidate Obesity Genes. PLoS Genet. 2010, 6, e1000976. [Google Scholar] [CrossRef] [PubMed]
- Myneni, V.D.; Hitomi, K.; Kaartinen, M.T. Factor XIII-A transglutaminase acts as a switch between preadipocyte proliferation and differentiation. Blood 2014, 124, 1344–1353. [Google Scholar] [CrossRef]
- Myneni, V.D.; Mousa, A.; Kaartinen, M.T. Factor XIII-A transglutaminase deficient mice show signs of metabolically healthy obesity on high fat diet. Sci. Rep. 2016, 6, 35574. [Google Scholar] [CrossRef] [Green Version]
- Kaartinen, A.; Heinonen, L.; Hakkarainen, L.; Rissanen, K.; Pietiläinen, K.H. F13A1 transglutaminase expression in human adipose tissue increases in acquired excess weight and associates with inflammatory status of adipocytes. Int. J. Obes. in press.
- Jernås, M.; Palming, J.; Sjöholm, K.; Jennische, E.; Svensson, P.-A.; Gabrielsson, B.G.; Levin, M.; Sjögren, A.; Rudemo, M.; Lystig, T.C.; et al. Separation of human adipocytes by size: Hypertrophic fat cells display distinct gene expression. FASEB J. 2006, 20, 1540–1542. [Google Scholar] [CrossRef]
- Lee, B.-C.; Lee, J. Cellular and molecular players in adipose tissue inflammation in the development of obesity-induced insulin resistance. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2014, 1842, 446–462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burhans, M.S.; Hagman, D.K.; Kuzma, J.N.; Schmidt, K.A.; Kratz, M. Contribution of Adipose Tissue Inflammation to the Development of Type 2 Diabetes Mellitus. Compr. Physiol. 2018, 9, 1–58. [Google Scholar] [CrossRef]
- Reilly, S.M.; Saltiel, A.R. Adapting to obesity with adipose tissue inflammation. Nat. Rev. Endocrinol. 2017, 13, 633–643. [Google Scholar] [CrossRef]
- Stolarczyk, E. Adipose tissue inflammation in obesity: A metabolic or immune response? Curr. Opin. Pharmacol. 2017, 37, 35–40. [Google Scholar] [CrossRef]
- Trim, W.; Turner, J.E.; Thompson, D. Parallels in Immunometabolic Adipose Tissue Dysfunction with Ageing and Obesity. Front. Immunol. 2018, 9, 169. [Google Scholar] [CrossRef] [Green Version]
- Caccamo, D.; Currò, M.; Ferlazzo, N.; Condello, S.; Ientile, R. Monitoring of transglutaminase2 under different oxidative stress conditions. Amino Acids 2011, 42, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Currò, M.; Ferlazzo, N.; Risitano, R.; Condello, S.; Vecchio, M.; Caccamo, D.; Ientile, R. Transglutaminase 2 and phospholipase A2 interactions in the inflammatory response in human Thp-1 monocytes. Amino Acids 2013, 46, 759–766. [Google Scholar] [CrossRef]
- Ientile, R.; Caccamo, D.; Griffin, M. Tissue transglutaminase and the stress response. Amino Acids 2007, 33, 385–394. [Google Scholar] [CrossRef]
- Heinonen, S.; Buzkova, J.; Muniandy, M.; Kaksonen, R.; Ollikainen, M.; Ismail, K.; Hakkarainen, A.; Lundbom, J.; Lundbom, N.; Vuolteenaho, K.; et al. Impaired Mitochondrial Biogenesis in Adipose Tissue in Acquired Obesity. Diabetes 2015, 64, 3135–3145. [Google Scholar] [CrossRef] [Green Version]
- Heinonen, S.; Jokinen, R.; Rissanen, A.; Pietiläinen, K.H. White adipose tissue mitochondrial metabolism in health and in obesity. Obes. Rev. 2019, 21, e12958. [Google Scholar] [CrossRef]
- Heinonen, S.; Muniandy, M.; Buzkova, J.; Mardinoglu, A.; Rodríguez, A.; Frühbeck, G.; Hakkarainen, A.; Lundbom, J.; Lundbom, N.; Kaprio, J.; et al. Mitochondria-related transcriptional signature is downregulated in adipocytes in obesity: A study of young healthy MZ twins. Diabetology 2016, 60, 169–181. [Google Scholar] [CrossRef] [Green Version]
- Jukarainen, S.; Heinonen, S.; Rämö, J.T.; Rinnankoski-Tuikka, R.; Rappou, E.; Tummers, M.; Muniandy, M.; Hakkarainen, A.; Lundbom, J.; Lundbom, N.; et al. Obesity Is Associated With Low NAD(+)/SIRT Pathway Expression in Adipose Tissue of BMI-Discordant Monozygotic Twins. J. Clin. Endocrinol. Metab. 2016, 101, 275–283. [Google Scholar] [CrossRef] [Green Version]
- Kaye, S.M.; Pietiläinen, K.H.; Kotronen, A.; Joutsi-Korhonen, L.; Kaprio, J.; Yki-Järvinen, H.; Silveira, A.; Hamsten, A.; Lassila, R.; Rissanen, A. Obesity-Related Derangements of Coagulation and Fibrinolysis: A Study of Obesity-Discordant Monozygotic Twin Pairs. Obesity 2012, 20, 88–94. [Google Scholar] [CrossRef]
- Naukkarinen, J.; Heinonen, S.; Hakkarainen, A.; Lundbom, J.; Vuolteenaho, K.; Saarinen, L.; Hautaniemi, S.; Rodríguez, A.; Frühbeck, G.; Pajunen, P.; et al. Characterising metabolically healthy obesity in weight-discordant monozygotic twins. Diabetology 2013, 57, 167–176. [Google Scholar] [CrossRef] [Green Version]
- Pietiläinen, K.H.; Ismail, K.; Järvinen, E.; Heinonen, S.; Tummers, M.; Bollepalli, S.; Lyle, R.; Muniandy, M.; Moilanen, E.; Hakkarainen, A.; et al. DNA methylation and gene expression patterns in adipose tissue differ significantly within young adult monozygotic BMI-discordant twin pairs. Int. J. Obes. 2015, 40, 654–661. [Google Scholar] [CrossRef]
- Sahebekhtiari, N.; Saraswat, M.; Joenväärä, S.; Jokinen, R.; Lovric, A.; Kaye, S.; Mardinoglu, A.; Rissanen, A.; Kaprio, J.; Renkonen, R.; et al. Plasma Proteomics Analysis Reveals Dysregulation of Complement Proteins and Inflammation in Acquired Obesity—A Study on Rare BMI-Discordant Monozygotic Twin Pairs. Proteom. Clin. Appl. 2019, 13, e1800173. [Google Scholar] [CrossRef]
- Church, C.D.; Berry, R.; Rodeheffer, M.S. Isolation and Study of Adipocyte Precursors. Methods Enzymol. 2014, 537, 31–46. [Google Scholar] [CrossRef] [Green Version]
- Hagberg, C.E.; Li, Q.; Kutschke, M.; Bhowmick, D.; Kiss, E.; Shabalina, I.G.; Harms, M.J.; Shilkova, O.; Kozina, V.; Nedergaard, J.; et al. Flow Cytometry of Mouse and Human Adipocytes for the Analysis of Browning and Cellular Heterogeneity. Cell Rep. 2018, 24, 2746–2756.e5. [Google Scholar] [CrossRef] [Green Version]
- Martin, J.; Collot-Teixeira, S.; McGregor, L.; McGregor, J.L. The dialogue between endothelial cells and monocytes/macrophages in vascular syndromes. Curr. Pharm. Des. 2007, 13, 1751–1759. [Google Scholar] [CrossRef]
- Bagoly, Z.; Katona, É.; Muszbek, L. Factor XIII and inflammatory cells. Thromb. Res. 2012, 129, S77–S81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, H.; Kaartinen, M.T. Transglutaminases in Monocytes and Macrophages. Med. Sci. 2018, 6, 115. [Google Scholar] [CrossRef] [Green Version]
- Mitchell, J.L.; Mutch, N.J. Let’s cross-link: Diverse functions of the promiscuous cellular transglutaminase factor XIII-A. J. Thromb. Haemost. 2019, 17, 19–30. [Google Scholar] [CrossRef] [Green Version]
- Dardik, R.; Inbal, A. Complex formation between tissue transglutaminase II (tTG) and vascular endothelial growth factor receptor 2 (VEGFR-2): Proposed mechanism for modulation of endothelial cell response to VEGF. Exp. Cell Res. 2006, 312, 2973–2982. [Google Scholar] [CrossRef]
- Dardik, R.; Leor, J.; Skutelsky, E.; Castel, D.; Holbova, R.; Schiby, G.; Shaish, A.; Dickneite, G.; Loscalzo, J.; Inbal, A. Evaluation of the pro-angiogenic effect of factor XIII in heterotopic mouse heart allografts and FXIII-deficient mice. Thromb. Haemost. 2006, 95, 546–550. [Google Scholar] [CrossRef] [PubMed]
- Dardik, R.; Loscalzo, J.; Inbal, A. Factor XIII (FXIII) and angiogenesis. J. Thromb. Haemost. 2005, 4, 19–25. [Google Scholar] [CrossRef]
- Dardik, R.; Shenkman, B.; Tamarin, I.; Eskaraev, R.; Harsfalvi, J.; Varon, D.; Inbal, A. Factor XIII mediates adhesion of platelets to endothelial cells through alpha(v)beta(3) and glycoprotein IIb/IIIa integrins. Thromb. Res. 2002, 105, 317–323. [Google Scholar] [CrossRef]
- Dardik, R.; Solomon, A.; Loscalzo, J.; Eskaraev, R.; Bialik, A.; Goldberg, I.; Schiby, G.; Inbal, A. Novel Proangiogenic Effect of Factor XIII Associated With Suppression of Thrombospondin 1 Expression. Arter. Thromb. Vasc. Biol. 2003, 23, 1472–1477. [Google Scholar] [CrossRef] [Green Version]
- Inbal, A.; Dardik, R. Role of Coagulation Factor XIII (FXIII) in Angiogenesis and Tissue Repair. Pathophysiol. Haemost. Thromb. 2006, 35, 162–165. [Google Scholar] [CrossRef]
- Akira, S.; Misawa, T.; Satoh, T. Macrophages control innate inflammation. Diabetes Obes. Metab. 2013, 15, 10–18. [Google Scholar] [CrossRef]
- Wen, H.; Gris, D.; Lei, Y.; Jha, S.; Zhang, L.; Huang, M.T.-H.; Brickey, W.J.; Ting, J.P.-Y. Fatty acid–induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat. Immunol. 2011, 12, 408–415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rheinheimer, J.; De Souza, B.M.; Cardoso, N.S.; Bauer, A.C.; Crispim, D. Current role of the NLRP3 inflammasome on obesity and insulin resistance: A systematic review. Metabolism 2017, 74, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Camell, C.D.; Günther, P.; Lee, A.; Goldberg, E.L.; Spadaro, O.; Youm, Y.-H.; Bartke, A.; Hubbard, G.B.; Ikeno, Y.; Ruddle, N.H.; et al. Aging Induces an Nlrp3 Inflammasome-Dependent Expansion of Adipose B Cells That Impairs Metabolic Homeostasis. Cell Metab. 2019, 30, 1024–1039.e6. [Google Scholar] [CrossRef]
- Stienstra, R.; Van Diepen, J.A.; Tack, C.J.; Zaki, M.H.; Van De Veerdonk, F.L.; Perera, D.; Neale, G.A.; Hooiveld, G.J.; Hijmans, A.; Vroegrijk, I.; et al. Inflammasome is a central player in the induction of obesity and insulin resistance. Proc. Natl. Acad. Sci. USA 2011, 108, 15324–15329. [Google Scholar] [CrossRef] [Green Version]
- Ballak, D.B.; Stienstra, R.; Tack, C.J.; Dinarello, C.A.; Van Diepen, J.A. IL-1 family members in the pathogenesis and treatment of metabolic disease: Focus on adipose tissue inflammation and insulin resistance. Cytokine 2015, 75, 280–290. [Google Scholar] [CrossRef] [Green Version]
- Castanheira, F.V.S.; Kubes, P. Neutrophils and NETs in modulating acute and chronic inflammation. Blood 2019, 133, 2178–2185. [Google Scholar] [CrossRef] [PubMed]
- Kolaczkowska, E.; Kubes, P. Neutrophil recruitment and function in health and inflammation. Nat. Rev. Immunol. 2013, 13, 159–175. [Google Scholar] [CrossRef]
- Liew, P.X.; Kubes, P. The Neutrophil’s Role During Health and Disease. Physiol. Rev. 2019, 99, 1223–1248. [Google Scholar] [CrossRef]
- Mantovani, A.; Cassatella, M.A.; Costantini, C.; Jaillon, S. Neutrophils in the activation and regulation of innate and adaptive immunity. Nat. Rev. Immunol. 2011, 11, 519–531. [Google Scholar] [CrossRef]
- Maurizi, G.; Della Guardia, L.; Maurizi, A.; Poloni, A. Adipocytes properties and crosstalk with immune system in obesity-related inflammation. J. Cell. Physiol. 2017, 233, 88–97. [Google Scholar] [CrossRef]
- Watanabe, Y.; Nagai, Y.; Honda, H.; Okamoto, N.; Yanagibashi, T.; Ogasawara, M.; Yamamoto, S.; Imamura, R.; Takasaki, I.; Hara, H.; et al. Bidirectional crosstalk between neutrophils and adipocytes promotes adipose tissue inflammation. FASEB J. 2019, 33, 11821–11835. [Google Scholar] [CrossRef] [Green Version]
- Hook, J.S.; Cao, M.; Potera, R.M.; Alsmadi, N.Z.; Schmidtke, D.W.; Moreland, J.G. Nox2 Regulates Platelet Activation and NET Formation in the Lung. Front. Immunol. 2019, 10, 1472. [Google Scholar] [CrossRef]
- Han, C.Y. Roles of Reactive Oxygen Species on Insulin Resistance in Adipose Tissue. Diabetes Metab. J. 2016, 40, 272–279. [Google Scholar] [CrossRef]
- Srikakulapu, P.; McNamara, C.A. B Lymphocytes and Adipose Tissue Inflammation. Arter. Thromb. Vasc. Biol. 2020, 40, 1110–1122. [Google Scholar] [CrossRef]
- Winer, D.A.; Winer, S.; Chng, M.H.Y.; Shen, L.; Engleman, E.G. B Lymphocytes in obesity-related adipose tissue inflammation and insulin resistance. Cell. Mol. Life Sci. 2013, 71, 1033–1043. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.-H.; Choi, B.-H.; Cheon, H.-G.; Do, M.-S. B cell activation factor (BAFF) is a novel adipokine that links obesity and inflammation. Exp. Mol. Med. 2009, 41, 208–216. [Google Scholar] [CrossRef] [Green Version]
- Esnault, S.; Kelly, E.A.; Sorkness, R.L.; Evans, M.D.; Busse, W.W.; Jarjour, N.N. Airway factor XIII associates with type 2 inflammation and airway obstruction in asthmatic patients. J. Allergy Clin. Immunol. 2016, 137, 767–773.e6. [Google Scholar] [CrossRef] [Green Version]
- Gualtierotti, R.; Ingegnoli, F.; Boscolo, M.; Griffini, S.; Grovetti, E.; Cugno, M. Tocilizumab Effects on Coagulation Factor XIII in Patients with Rheumatoid Arthritis. Adv. Ther. 2019, 36, 3494–3502. [Google Scholar] [CrossRef] [Green Version]
- Ichinose, A. Factor XIII is a key molecule at the intersection of coagulation and fibrinolysis as well as inflammation and infection control. Int. J. Hematol. 2012, 95, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Kovar, F.M.; Marsik, C.L.; Jilma, B.; Mannhalter, C.; Joukhadar, C.; Wagner, O.F.; Endler, G. The inflammatory response is influenced by FXIII VAL 34 LEU polymorphism in a human LPS model. Wien. Klin. Wochenschr. 2009, 121, 515–519. [Google Scholar] [CrossRef]
- Raghu, H.; Cruz, C.; Rewerts, C.L.; Frederick, M.D.; Thornton, S.; Mullins, E.S.; Schoenecker, J.G.; Degen, J.L.; Flick, M.J. Transglutaminase factor XIII promotes arthritis through mechanisms linked to inflammation and bone erosion. Blood 2015, 125, 427–437. [Google Scholar] [CrossRef] [Green Version]
- Soendergaard, C.; Kvist, P.H.; Seidelin, J.B.; Pelzer, H.; Nielsen, O.H. Systemic and intestinal levels of factor XIII-A: The impact of inflammation on expression in macrophage subtypes. J. Gastroenterol. 2015, 51, 796–807. [Google Scholar] [CrossRef]
- Kopec, A.K.; Abrahams, S.R.; Thornton, S.; Palumbo, J.S.; Mullins, E.S.; Divanovic, S.; Weiler, H.; Owens, A.P.; Mackman, N.; Goss, A.M.; et al. Thrombin promotes diet-induced obesity through fibrin-driven inflammation. J. Clin. Investig. 2017, 127, 3152–3166. [Google Scholar] [CrossRef] [PubMed]
- Porrello, A.; Leslie, P.L.; Harrison, E.B.; Gorentla, B.K.; Kattula, S.; Ghosh, S.K.; Azam, S.H.; Holtzhausen, A.; Chao, Y.L.; Hayward, M.C.; et al. Factor XIIIA-expressing inflammatory monocytes promote lung squamous cancer through fibrin cross-linking. Nat. Commun. 2018, 9, 1988. [Google Scholar] [CrossRef] [Green Version]
- Lanir, N.; Ciano, P.S.; Van De Water, L.; McDonagh, J.; Dvorak, A.M.; Dvorak, H.F. Macrophage migration in fibrin gel matrices. II. Effects of clotting factor XIII, fibronectin, and glycosaminoglycan content on cell migration. J. Immunol. 1988, 140, 2340–2349. [Google Scholar] [PubMed]
- Goldstein, B.J. Protein-Tyrosine phosphatases and the regulation of insulin action. J. Cell. Biochem. 1992, 48, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, B.J.; Ahmad, F.; Ding, W.; Li, P.-M.; Zhang, W.-R. Regulation of the insulin signalling pathway by cellular protein-tyrosine phosphatases. Mol. Cell. Biochem. 1998, 182, 91–99. [Google Scholar] [CrossRef]
- Kim, W.-K.; Jung, H.; Kim, D.-H.; Kim, E.-Y.; Chung, J.-W.; Cho, Y.-S.; Park, S.G.; Park, B.-C.; Ko, Y.; Bae, K.-H.; et al. Regulation of adipogenic differentiation by LAR tyrosine phosphatase in human mesenchymal stem cells and 3T3-L1 preadipocytes. J. Cell Sci. 2009, 122, 4160–4167. [Google Scholar] [CrossRef] [Green Version]
- Ren, J.M.; Li, P.M.; Zhang, W.R.; Sweet, L.J.; Cline, G.; Shulman, G.I.; Livingston, J.N.; Goldstein, B.J. Transgenic mice deficient in the LAR protein-tyrosine phosphatase exhibit profound defects in glucose homeostasis. Diabetes 1998, 47, 493–497. [Google Scholar] [CrossRef]
- Deng, T.; Lyon, C.J.; Minze, L.J.; Lin, J.; Zou, J.; Liu, J.Z.; Ren, Y.; Yin, Z.; Hamilton, D.J.; Reardon, P.R.; et al. Class II Major Histocompatibility Complex Plays an Essential Role in Obesity-Induced Adipose Inflammation. Cell Metab. 2013, 17, 411–422. [Google Scholar] [CrossRef] [Green Version]
- Poloni, A.; Maurizi, G.; Ciarlantini, M.; Medici, M.; Mattiucci, D.; Mancini, S.; Maurizi, A.; Falconi, M.; Olivieri, A.; Leoni, P. Interaction between human mature adipocytes and lymphocytes induces T-cell proliferation. Cytotherapy 2015, 17, 1292–1301. [Google Scholar] [CrossRef]
- Datta, R.; Podolsky, M.J.; Atabai, K. Fat fibrosis: Friend or foe? JCI Insight 2018, 3, e122289. [Google Scholar] [CrossRef]
- Khan, T.; Muise, E.S.; Iyengar, P.; Wang, Z.V.; Chandalia, M.; Abate, N.; Zhang, B.B.; Bonaldo, P.; Chua, S.; Scherer, P.E. Metabolic Dysregulation and Adipose Tissue Fibrosis: Role of Collagen VI. Mol. Cell. Biol. 2008, 29, 1575–1591. [Google Scholar] [CrossRef] [Green Version]
- Lin, D.; Chun, T.-H.; Kang, L. Adipose extracellular matrix remodelling in obesity and insulin resistance. Biochem. Pharmacol. 2016, 119, 8–16. [Google Scholar] [CrossRef] [Green Version]
- Crewe, C.; An, Y.A.; Scherer, P.E. The ominous triad of adipose tissue dysfunction: Inflammation, fibrosis, and impaired angiogenesis. J. Clin. Investig. 2017, 127, 74–82. [Google Scholar] [CrossRef] [Green Version]
- Ruiz-Ojeda, F.J.; Méndez-Gutiérrez, A.; Aguilera, C.M.; Plaza-Díaz, J. Extracellular Matrix Remodeling of Adipose Tissue in Obesity and Metabolic Diseases. Int. J. Mol. Sci. 2019, 20, 4888. [Google Scholar] [CrossRef] [Green Version]
- Cinti, S.; Mitchell, G.; Barbatelli, G.; Murano, I.; Ceresi, E.; Faloia, E.; Wang, S.; Fortier, M.; Greenberg, A.S.; Obin, M.S. Adipocyte death defines macrophage localization and function in adipose tissue of obese mice and humans. J. Lipid Res. 2005, 46, 2347–2355. [Google Scholar] [CrossRef] [Green Version]
- Trayhurn, P. Hypoxia and Adipose Tissue Function and Dysfunction in Obesity. Physiol. Rev. 2013, 93, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Sato, H.; Taketomi, Y.; Murakami, M. Metabolic regulation by secreted phospholipase A. Inflamm. Regen. 2016, 36, 7. [Google Scholar] [CrossRef] [Green Version]
- Iyer, A.; Lim, J.; Poudyal, H.; Reid, R.C.; Suen, J.Y.; Webster, J.; Prins, J.B.; Whitehead, J.P.; Fairlie, D.; Brown, L. An Inhibitor of Phospholipase A2 Group IIA Modulates Adipocyte Signaling and Protects Against Diet-Induced Metabolic Syndrome in Rats. Diabetes 2012, 61, 2320–2329. [Google Scholar] [CrossRef] [Green Version]
- Zhang, T.; He, J.; Xu, C.; Zu, L.; Jiang, H.; Pu, S.; Guo, X.; Xu, G. Mechanisms of metformin inhibiting lipolytic response to isoproterenol in primary rat adipocytes. J. Mol. Endocrinol. 2008, 42, 57–66. [Google Scholar] [CrossRef]
- Greenberg, A.S.; Shen, W.-J.; Muliro, K.; Patel, S.; Souza, S.C.; Roth, R.A.; Kraemer, F.B. Stimulation of Lipolysis and Hormone-sensitive Lipase via the Extracellular Signal-regulated Kinase Pathway. J. Biol. Chem. 2001, 276, 45456–45461. [Google Scholar] [CrossRef] [Green Version]
- Hesselbarth, N.; Kunath, A.; Kern, M.; Gericke, M.; Mejhert, N.; Rydén, M.; Stumvoll, M.; Blüher, M.; Klöting, N. Repin1 deficiency in adipose tissue improves whole-body insulin sensitivity, and lipid metabolism. Int. J. Obes. 2017, 41, 1815–1823. [Google Scholar] [CrossRef]
- Kaprio, J. Twin studies in Finland Twin research and human genetics. Off. J. Int. Soc. Twin Stud. 2006, 9, 772–777. [Google Scholar]
- Granér, M.; Seppälä-Lindroos, A.; Rissanen, A.; Hakkarainen, A.; Lundbom, N.; Kaprio, J.; Nieminen, M.S.; Pietiläinen, K.H. Epicardial Fat, Cardiac Dimensions, and Low-Grade Inflammation in Young Adult Monozygotic Twins Discordant for Obesity. Am. J. Cardiol. 2012, 109, 1295–1302. [Google Scholar] [CrossRef]
- Gentleman, R.C.; Carey, V.; Bates, D.; Bolstad, B.; Dettling, M.; Dudoit, S.; Ellis, B.; Gautier, L.; Ge, Y.; Gentry, J.; et al. Bioconductor: Open software development for computational biology and bioinformatics. Genome Biol. 2004, 5, R80. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2011. [Google Scholar]
- Zhang, Y.; Xie, L.; Gunasekar, S.K.; Tong, D.; Mishra, A.; Gibson, W.J.; Wang, C.; Fidler, T.; Marthaler, B.; Klingelhutz, A.; et al. SWELL1 is a regulator of adipocyte size, insulin signalling and glucose homeostasis. Nat. Cell Biol. 2017, 19, 504–517. [Google Scholar] [CrossRef] [Green Version]
- Abreu-Vieira, G.; Fischer, A.W.; Mattsson, C.; De Jong, J.M.; Shabalina, I.G.; Rydén, M.; Laurencikiene, J.; Arner, P.; Cannon, B.; Nedergaard, J.; et al. Cidea improves the metabolic profile through expansion of adipose tissue. Nat. Commun. 2015, 6, 7433. [Google Scholar] [CrossRef] [Green Version]
- Weingarten, A.; Turchetti, L.; Krohn, K.; Klöting, I.; Kern, M.; Kovacs, P.; Stumvoll, M.; Blüher, M.; Klöting, N. Novel genes on rat chromosome 10 are linked to body fat mass, preadipocyte number and adipocyte size. Int. J. Obes. (2005) 2016, 40, 1832–1840. [Google Scholar] [CrossRef]
- Lundbäck, V.; Kulyté, A.; Strawbridge, R.J.; Ryden, M.; Arner, P.; Marcus, C.; Dahlman, I. FAM13A and POM121C are candidate genes for fasting insulin: Functional follow-up analysis of a genome-wide association study. Diabetol. 2018, 61, 1112–1123. [Google Scholar] [CrossRef] [Green Version]
- Krautbauer, S.; Neumeier, M.; Rein-Fischboeck, L.; Haberl, E.M.; Tilg, H.; Eisinger, K.; Buechler, C. Adipocyte Hypertrophy and Improved Postprandial Lipid Response in Beta 2 Syntrophin Deficient Mice. Cell. Physiol. Biochem. 2019, 52, 1151–1165. [Google Scholar] [CrossRef]
- Geoghegan, G.; Simcox, J.; Seldin, M.M.; Parnell, T.J.; Stubben, C.; Just, S.; Begaye, L.; Lusis, A.J.; Villanueva, C.J. Targeted deletion of Tcf7l2 in adipocytes promotes adipocyte hypertrophy and impaired glucose metabolism. Mol. Metab. 2019, 24, 44–63. [Google Scholar] [CrossRef] [PubMed]
- Kilian, T.M.; Klöting, N.; Blüher, M.; Beck-Sickinger, A.G. Prenatal notch1 receptor blockade by protein delta homolog 1 (DLK1) modulates adipocyte size in vivo. Int. J. Obes. 2015, 40, 698–705. [Google Scholar] [CrossRef]
- Longo, M.; Raciti, G.A.; Zatterale, F.; Parrillo, L.; Desiderio, A.; Spinelli, R.; Hammarstedt, A.; Hedjazifar, S.; Hoffmann, J.M.; Nigro, C.; et al. Epigenetic modifications of the Zfp/ZNF423 gene control murine adipogenic commitment and are dysregulated in human hypertrophic obesity. Diabetologia 2018, 61, 369–380. [Google Scholar] [CrossRef] [Green Version]
- Huang, Z.H.; Gu, D.; Mazzone, T. Role of adipocyte-derived apoE in modulating adipocyte size, lipid metabolism, and gene expression in vivo. Am. J. Physiol. Metab. 2009, 296, E1110–E1119. [Google Scholar] [CrossRef] [Green Version]
- Acosta, J.R.; Douagi, I.; Andersson, D.P.; Bäckdahl, J.; Rydén, M.; Arner, P.; Laurencikiene, J. Increased fat cell size: A major phenotype of subcutaneous white adipose tissue in non-obese individuals with type 2 diabetes. Diabetologia 2015, 59, 560–570. [Google Scholar] [CrossRef] [Green Version]
- Ruschke, K.; Illes, M.; Kern, M.; Klöting, I.; Fasshauer, M.; Schön, M.; Kosacka, J.; Fitzl, G.; Kovacs, P.; Stumvoll, M.; et al. Repin1 maybe involved in the regulation of cell size and glucose transport in adipocytes. Biochem. Biophys. Res. Commun. 2010, 400, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Kamei, Y.; Ezaki, O. Mest/Peg1 imprinted gene enlarges adipocytes and is a marker of adipocyte size. Am. J. Physiol. Metab. 2005, 288, E117–E124. [Google Scholar] [CrossRef] [Green Version]
- Yvan-Charvet, L.; Even, P.C.; Bloch-Faure, M.; Guerre-Millo, M.; Moustaid-Moussa, N.; Ferré, P.; Quignard-Boulange, A. Deletion of the Angiotensin Type 2 Receptor (AT2R) Reduces Adipose Cell Size and Protects from Diet-Induced Obesity and Insulin Resistance. Diabetes 2005, 54, 991–999. [Google Scholar] [CrossRef]






| Clinical and Adipocity Parameters | Heavier Co-twin | Leaner Co-twin | p-Value |
|---|---|---|---|
| Height (cm) | 169.3 ± 2.89 | 169.2 ± 3.0 | 0.7530 |
| Weight (kg) | 83.8 ± 4.6 | 67.0 ± 4.1 | 0.0022 |
| BMI (kg/m2) | 29.0 ± 0.85 | 23.2 ± 0.7 | 0.0022 |
| Body fat (%) | 41.9 ± 2.0 | 32.8 ±1.8 | 0.0029 |
| Body fat (kg) | 34.7 ± 2.3 | 22.0 ± 1.9 | 0.0022 |
| Fat-free mass (kg) | 46.6 ± 3.7 | 42.8 ± 2.9 | 0.0029 |
| Subcutaneous fat (dm3) | 5762.4 ± 450.6 | 3303.6 ± 275.4 | 0.0022 |
| Intra-abdominal fat (dm3) | 1109.8 ± 186.4 | 502.2 ± 91.2 | 0.0037 |
| Liver fat (%) | 2.4 ± 0.74 | 0.60 ± 0.06 | 0.0076 |
| Triglycerides (mmol/L) | 1.14 ± 0.07 | 0.96 ± 0.11 | 0.1823 |
| fP-Leptin (pg/mL) | 34,229.0 ± 5931.3 | 18,331.8 ± 4412.6 | 0.0037 |
| fP-Adiponectin (ng/mL) | 2844.0 ± 382.1 | 3520.1 ± 380.1 | 0.0186 |
| Adipocyte diameter (mm) | 87.9 ± 2.82 | 76.6 ± 2.77 | 0.0022 |
| Adipocyte number | 8.8e + 13 ± 5.7e + 12 | 8.5e + 13 ± 8.2e + 12 | 0.4328 |
| Positive Correlation | ||||
| Gene Symbol | Gene ID | Gene Name | Pearson (r) | p-Value |
| CPVL | ENSG00000106066 | Carboxypeptidase, vitellogenic-like | 0.946 | 0.00000097 |
| PYCARD | ENSG00000103490 | PYD and CARD domain containing | 0.941 | 0.00000167 |
| CTSS | ENSG00000163131 | Cathepsin S | 0.902 | 0.00002544 |
| ITGAM | ENSG00000169896 | Integrin subunit alpha M | 0.885 | 0.00005774 |
| DCLK1 | ENSG00000133083 | Doublecortin-like kinase 1 | 0.867 | 0.00012405 |
| CYBB | ENSG00000165168 | Cytochrome b-245 beta chain | 0.856 | 0.00018955 |
| SRGN | ENSG00000122862 | Serglycin | 0.842 | 0.0003081 |
| TM4SF1 | ENSG00000169908 | Transmembrane 4 L six family member 18 | 0.834 | 0.00039092 |
| PDE4B | ENSG00000184588 | Phosphodiesterase 4B | 0.834 | 0.00039317 |
| TFPI | ENSG00000003436 | Tissue factor pathway inhibitor | 0.83 | 0.00044582 |
| ANXA4 | ENSG00000196975 | Annexin A4 | 0.827 | 0.00049387 |
| IL8 | ENSG00000169429 | Interleukin 8 | 0.806 | 0.00087858 |
| VCAN | ENSG00000038427 | Versican | 0.806 | 0.00088439 |
| BCL2A1 | ENSG00000140379 | BCL2-related protein A1 | 0.802 | 0.00097386 |
| LCP1 | ENSG00000136167 | Lymphocyte cytosolic protein 1/Plastin-2 | 0.802 | 0.00097956 |
| RECK | ENSG00000122707 | Reversion inducing cysteine-rich protein with kazal motifs | 0.799 | 0.00105162 |
| ABI3BP | ENSG00000154175 | ABI family member 3 binding protein | 0.795 | 0.00115971 |
| IL1B | ENSG00000125538 | Interleukin 1 beta | 0.786 | 0.00145102 |
| SAMSN1 | ENSG00000155307 | SAM domain, SH3 domain, and nuclear localization signals 1 | 0.78 | 0.00165797 |
| RGS1 | ENSG00000090104 | Regulator of G protein signaling 1 | 0.775 | 0.00186205 |
| CCDC109B | ENSG00000005059 | Mitochondrial calcium uniporter dominant negative beta subunit | 0.774 | 0.0018858 |
| NCEH1 | ENSG00000144959 | Neutral cholesterol ester hydrolase 1 | 0.766 | 0.0022702 |
| ARPC5 | ENSG00000162704 | Actin-related protein 2/3 complex subunit 5 | 0.764 | 0.00235294 |
| LYZ | ENSG00000090382 | Lysozyme | 0.762 | 0.00247521 |
| MAPK1 | ENSG00000100030 | Mitogen-activated protein kinase 1 | 0.743 | 0.00359262 |
| HLA-DRA | ENSG00000204287 | Major histocompatibility complex, class II, DR alpha | 0.743 | 0.00361775 |
| ARHGAP18 | ENSG00000146376 | Rho GTPase activating protein 18 | 0.743 | 0.00364017 |
| TNFSF13B | ENSG00000102524 | TNF superfamily member 13b/B cell activation factor | 0.739 | 0.00393293 |
| WIPF1 | ENSG00000115935 | WAS/WASL interacting protein family, member 1 | 0.728 | 0.00481724 |
| CNKSR3 | ENSG00000153721 | CNKSR family member 3 | 0.725 | 0.00503039 |
| MGP | ENSG00000111341 | Matrix Gla protein | 0.72 | 0.00555039 |
| EVI2A | ENSG00000126860 | Ecotropic viral integration site 2A | 0.717 | 0.00579394 |
| NPTN | ENSG00000156642 | Neuroplastin | 0.715 | 0.00596749 |
| SELE | ENSG00000007908 | Selectin-E | 0.778 | 0.00174649 |
| LPCAT1 | ENSG00000153395 | Lysophosphatidylcholine acyltransferase 1 | 0.732 | 0.00445358 |
| Negative Correlation | ||||
| Gene Symbol | Gene ID | Gene Name | Pearson (r) | p-Value |
| SCRN2 | ENSG00000141295 | Secernin 2 | –0.876 | 0.000086 |
| PTPRF | ENSG00000142949 | Protein tyrosine phosphatase receptor type F | –0.84 | 0.00033 |
| EMC3 | ENSG00000125037 | ER membrane protein complex subunit 3 | –0.816 | 0.000673 |
| ABCD1 | ENSG00000101986 | ATP binding cassette subfamily D member 1 | –0.793 | 0.001229 |
| PLA2G16 | ENSG00000176485 | Phospholipase A and acyltransferase 3 | –0.784 | 0.001527 |
| NFRKB | ENSG00000170322 | Nuclear factor related to kappaB binding protein | –0.74 | 0.00386 |
| HEPACAM | ENSG00000165478 | Hepatic and glial cell adhesion molecule | –0.734 | 0.00426 |
| RBP1 | ENSG00000114115 | Retinol binding protein 1, cellular | –0.725 | 0.00501 |
| STAT5A | ENSG00000126561 | Signal transducer and activator of transcription 5A | –0.72 | 0.00556 |
| TUSC5 | ENSG00000184811 | Tumor suppressor candidate 5/Trafficking regulator of GLUT4 | –0.717 | 0.005843 |
| COX5A | ENSG00000178741 | Cytochrome c oxidase subunit Va | –0.712 | 0.006346 |
| REPIN1 | ENSG00000214022 | Replication initiator 1 | –0.772 | 0.001987 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaartinen, M.T.; Arora, M.; Heinonen, S.; Rissanen, A.; Kaprio, J.; Pietiläinen, K.H. Transglutaminases and Obesity in Humans: Association of F13A1 to Adipocyte Hypertrophy and Adipose Tissue Immune Response. Int. J. Mol. Sci. 2020, 21, 8289. https://doi.org/10.3390/ijms21218289
Kaartinen MT, Arora M, Heinonen S, Rissanen A, Kaprio J, Pietiläinen KH. Transglutaminases and Obesity in Humans: Association of F13A1 to Adipocyte Hypertrophy and Adipose Tissue Immune Response. International Journal of Molecular Sciences. 2020; 21(21):8289. https://doi.org/10.3390/ijms21218289
Chicago/Turabian StyleKaartinen, Mari T., Mansi Arora, Sini Heinonen, Aila Rissanen, Jaakko Kaprio, and Kirsi H. Pietiläinen. 2020. "Transglutaminases and Obesity in Humans: Association of F13A1 to Adipocyte Hypertrophy and Adipose Tissue Immune Response" International Journal of Molecular Sciences 21, no. 21: 8289. https://doi.org/10.3390/ijms21218289
APA StyleKaartinen, M. T., Arora, M., Heinonen, S., Rissanen, A., Kaprio, J., & Pietiläinen, K. H. (2020). Transglutaminases and Obesity in Humans: Association of F13A1 to Adipocyte Hypertrophy and Adipose Tissue Immune Response. International Journal of Molecular Sciences, 21(21), 8289. https://doi.org/10.3390/ijms21218289