The Role of RIPK1 and RIPK3 in Cardiovascular Disease
Abstract
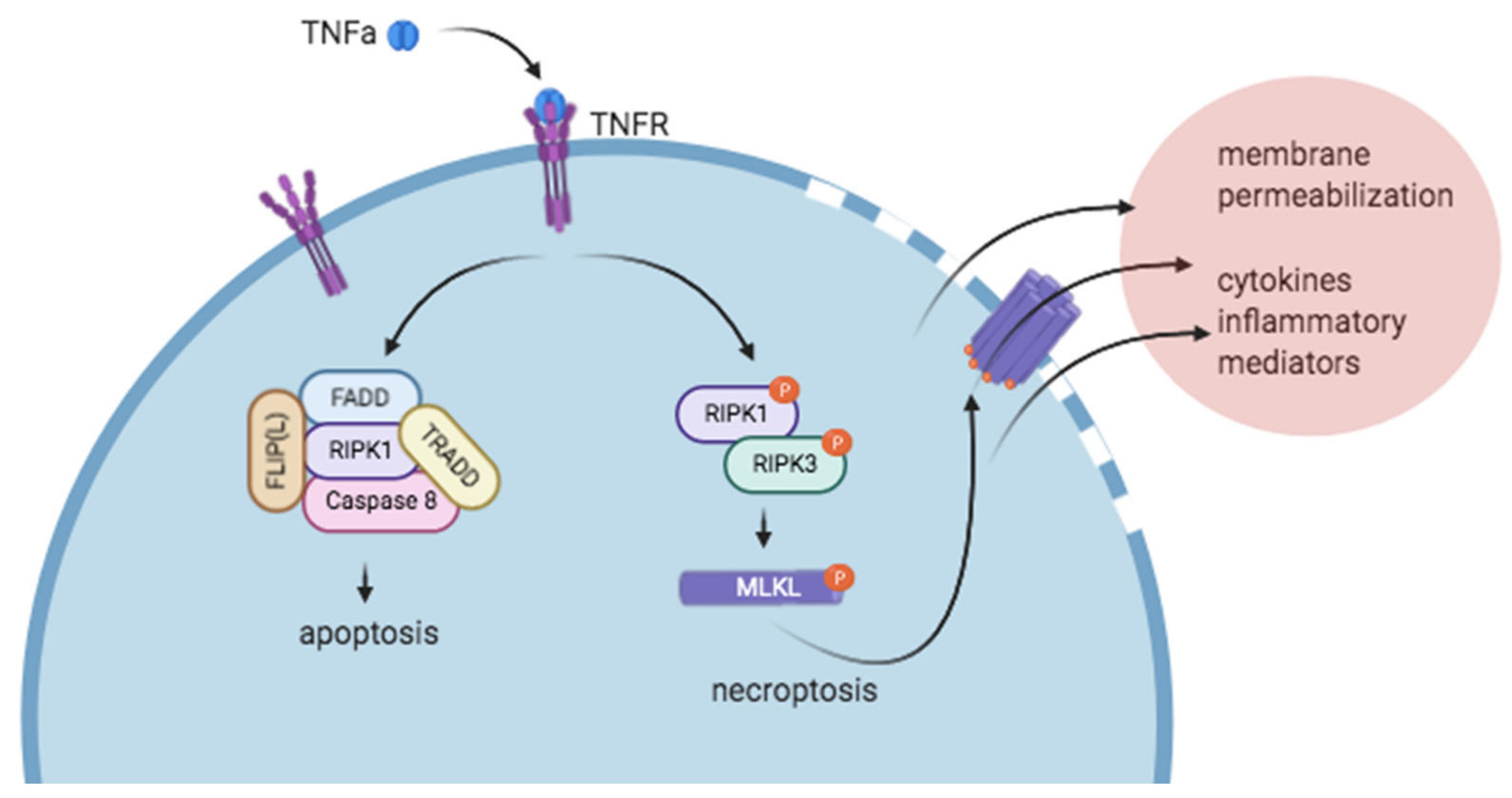
1. Introduction to RIP1 and RIPK3 and Necroptosis
2. RIPK1 and RIPK3 in Atherosclerosis
3. RIPK1 and RIPK3 in Myocardial Infarction
4. RIPK1 and RIPK3 in Stroke
5. RIPK1 and RIPK3 in Abdominal Aortic Aneurysm
6. RIPK1 and RIPK3 in Thrombosis
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AAA | abdominal aortic aneruysm |
| ALS | amyotrophic lateral sclerosis |
| AngII | angiotensin II |
| APOE | apolioprotein E |
| CAD | coronary artery disease |
| CML | chronic myeloid leukemia |
| DAMP | damage-associated molecular patterns |
| DVT | deep vein thrombosis |
| FADD | FAS-associated death domain |
| HIF1α | hypoxia inducible factor-1α |
| HUVEC | human umbilical vein endothelial cells |
| IFNγ | interferon γ |
| IL1β | Interleukin 1β |
| IVC | inferior vena cava |
| LAD | left anterior descending |
| LDL | low-density lipoprotein |
| LVEF | left ventricle ejection fraction |
| MCAO | middle cerebral artery occlusion |
| MI | myocardial infarction |
| MLKL | mixed-lineage kinase domain like protein |
| MMP | matrix metalloproteinase |
| MS | multiple sclerosis |
| Nec-1/1s | necrostain-1/1s |
| NET | neutrophil extracellular trap |
| NFKβ | Nuclear factor kappa β |
| NSA | necrosulfonamide |
| PCI | percutaneous coronary intervention |
| PE | pulmonary embolism |
| Ph+ ALL | Philadelphia chromosome positive acute lymphoblastic leukemia |
| PI | propidium iodide |
| PTLP | phospholipid transfer protein |
| RA | rheumatoid arthritis |
| RIPK1 | receptor interacting protein kinase 1 |
| RIPK3 | receptor interacting protein kinase 3 |
| SIRS | systemic inflammatory response syndrome |
| SMC | smooth muscle cell |
| STEMI | ST elevation myocardial infarction |
| STING | stimulator of interferon genes |
| TLR 3/4 | toll like receptor 3/4 |
| TNFα | tumor necrosis factor α |
| TNFR1 | tumor necrosis factor receptor 1 |
| TRAILR | tumor necrosis α related apoptosis inducing ligand receptor |
| tPA | tissue plasminogen activator |
| UC | ulcerative colitis |
| VTE | venous thromboembolism |
References
- Dhuriya, Y.K.; Sharma, D. Necroptosis: A regulated inflammatory mode of cell death. J. Neuroinflamm. 2018, 15, 1–9. [Google Scholar] [CrossRef]
- Hanson, B. Necroptosis: A new way of dying? Cancer Biol. Ther. 2016, 17, 899–910. [Google Scholar] [CrossRef]
- Leeper, N.J. The role of necroptosis in atherosclerotic disease. JACC Basic Transl. Sci. 2016, 1, 548–550. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, T.; Marchetti, P.; A Susin, S.; Dallaporta, B.; Zamzami, N.; Marzo, I.; Geuskens, M.; Kroemer, G. The apoptosis-necrosis paradox. Apoptogenic proteases activated after mitochondrial permeability transition determine the mode of cell death. Oncogene 1997, 15, 1573–1581. [Google Scholar] [CrossRef] [PubMed]
- Khoury, M.K.; Gupta, K.; Franco, S.; Liu, B. Necroptosis in the pathophysiology of disease. Am. J. Pathol. 2020, 190, 272–285. [Google Scholar] [CrossRef] [PubMed]
- Festjens, N.; Berghe, T.V.; Vandenabeele, P. Necrosis, a well-orchestrated form of cell demise: Signalling cascades, important mediators and concomitant immune response. Biochim. Biophys. Acta (BBA) Bioenerg. 2006, 1757, 1371–1387. [Google Scholar] [CrossRef] [PubMed]
- Vandenabeele, P.; Galluzzi, L.; Berghe, T.V.; Kroemer, G. Molecular mechanisms of necroptosis: An ordered cellular explosion. Nat. Rev. Mol. Cell Biol. 2010, 11, 700–714. [Google Scholar] [CrossRef] [PubMed]
- Vanlangenakker, N.; Berghe, T.V.; Vandenabeele, P. Many stimuli pull the necrotic trigger, an overview. Cell Death Differ. 2011, 19, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, W.J.; Upton, J.W.; Long, A.B.; Livingston-Rosanoff, D.; Daley-Bauer, L.P.; Hakem, R.; Caspary, T.; Mocarski, E.S. RIP3 mediates the embryonic lethality of caspase-8-deficient mice. Nat. Cell Biol. 2011, 471, 368–372. [Google Scholar] [CrossRef]
- Zhang, H.; Zhou, X.; McQuade, T.J.; Li, J.; Chan, F.K.M.; Zhang, J. Functional complementation between FADD and RIP1 in embryos and lymphocytes. Nat. Cell Biol. 2011, 471, 373–376. [Google Scholar] [CrossRef]
- Oberst, A.; Dillon, C.P.; Weinlich, R.; McCormick, L.L.; Fitzgerald, P.; Pop, C.; Hakem, R.; Salvesen, G.S.; Green, D.R. Catalytic activity of the caspase-8–FLIPL complex inhibits RIPK3-dependent necrosis. Nature 2011, 471, 363–367. [Google Scholar] [CrossRef]
- Karunakaran, D.; Geoffrion, M.; Wei, L.; Gan, W.; Richards, L.; Shangari, P.; Dekemp, E.M.; Beanlands, R.A.; Perisic, L.; Maegdefessel, L.; et al. Targeting macrophage necroptosis for therapeutic and diagnostic interventions in atherosclerosis. Sci. Adv. 2016, 2, e1600224. [Google Scholar] [CrossRef]
- Herrington, W.; Lacey, B.; Sherliker, P.; Armitage, J.; Lewington, S. Epidemiology of Atherosclerosis and the Potential to Reduce the Global Burden of Atherothrombotic Disease. Circ. Res. 2016, 118, 535–546. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Underlying Cause of Death 1999–2018; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2018.
- Virani, S.S.; Alonso, A.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Delling, F.N.; et al. Heart disease and stroke statistics—2020 update: A report from the American heart association. Circulation 2020, 141, e139–e596. [Google Scholar] [CrossRef]
- Rayner, K.J. Cell death in the Vessel wall: The good, the bad, the ugly. Arterioscler. Thromb. Vasc. Biol. 2017, 37, e75–e81. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.T.; Lin, H.Y.; Chan, Y.W.F.; Li, K.H.C.; To, O.T.L.; Yan, B.P.; Liu, T.; Li, G.; Wong, W.T.; Keung, W.; et al. Mouse models of atherosclerosis: A historical perspective and recent advances. Lipids Health Dis. 2017, 16, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bentzon, J.F.; Otsuka, F.; Virmani, R.; Falk, E. Mechanisms of Plaque Formation and Rupture. Circ. Res. 2014, 114, 1852–1866. [Google Scholar] [CrossRef]
- Tian, F.; Yao, J.; Yan, M.; Sun, X.; Wang, W.; Gao, W.; Tian, Z.; Guo, S.; Dong, Z.; Li, B.; et al. 5-Aminolevulinic Acid-Mediated Sonodynamic Therapy Inhibits RIPK1/RIPK3-Dependent Necroptosis in THP-1-Derived Foam Cells. Sci. Rep. 2016, 6, 21992. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Mehta, J.L. Oxidized LDL, a critical factor in atherogenesis. Cardiovasc. Res. 2005, 68, 353–354. [Google Scholar] [CrossRef] [PubMed]
- An, S.; Qi, Y.; Zhang, Z.; Mo, R.; Hou, L.; Yao, X.; Su, Y. Antagonism of receptor interacting protein 1 using necrostatin-1 in oxidized LDL- induced endothelial injury. Biomed. Pharmacother. 2018, 108, 1809–1815. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Li, H.; Yang, M.; Ren, J.; Huang, Z.; Han, F.; Huang, J.; Ma, J.; Zhang, D.; Zhang, Z.; et al. A Role of RIP3-mediated macrophage necrosis in atherosclerosis development. Cell Rep. 2013, 3, 200–210. [Google Scholar] [CrossRef]
- Zhang, K.; Yu, Y.; Luo, T.; Wang, L.; Ge, C.; Liu, X.; Song, J.; Jiang, X.; Zhang, Y.; Qin, S.; et al. Phospholipid Transfer Protein Destabilizes Mouse Atherosclerotic Plaque. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 2537–2544. [Google Scholar] [CrossRef]
- Kelliher, M.A.; Grimm, S.; Ishida, Y.; Kuo, F.; Stanger, B.Z.; Leder, P. The death domain kinase RIP mediates the TNF-induced NF-kB signal. Immunity 1998, 8, 297–303. [Google Scholar] [CrossRef]
- Weinlich, R.; Oberst, A.; Beere, H.M.; Green, D.R. Necroptosis in development, inflammation and disease. Nat. Rev. Mol. Cell Biol. 2017, 18, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, A.; Robichaud, S.; Nguyen, M.-A.; Geoffrion, M.; Wyatt, H.; Cottee, M.L.; Dennison, T.; Pietrangelo, A.; Lee, R.; Lagace, T.A.; et al. Loss of MLKL (Mixed Lineage Kinase Domain-Like Protein) decreases necrotic core but increases macrophage lipid accumulation in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2020, 40, 1155–1167. [Google Scholar] [CrossRef] [PubMed]
- Conos, S.A.; Chen, K.W.; De Nardo, D.; Hara, H.; Whitehead, L.; Núñez, G.; Masters, S.L.; Murphy, J.M.; Schroder, K.; Vaux, D.L.; et al. Active MLKL triggers the NLRP3 inflammasome in a cell-intrinsic manner. Proc. Natl. Acad. Sci. USA 2017, 114, E961–E969. [Google Scholar] [CrossRef]
- Yu, S.-X.; Chen, W.; Liu, Z.-Z.; Zhou, F.-H.; Yan, S.-Q.; Hu, G.-Q.; Qin, X.-X.; Zhang, J.; Ma, K.; Du, C.-T.; et al. Non-hematopoietic MLKL protects against salmonella mucosal infection by enhancing inflammasome activation. Front. Immunol. 2018, 9, 119. [Google Scholar] [CrossRef]
- Yoon, S.; Kovalenko, A.; Bogdanov, K.; Wallach, D. MLKL, the Protein that Mediates Necroptosis, Also Regulates Endosomal Trafficking and Extracellular Vesicle Generation. Immunity 2017, 47, 51–65. [Google Scholar] [CrossRef]
- Liu, Y.; Xu, H. Protective effect of necrostatin-1 on myocardial tissue in rats with acute myocardial infarction. Genet. Mol. Res. 2016, 15, 1–8. [Google Scholar] [CrossRef]
- Luedde, M.; Lutz, M.; Carter, N.; Sosna, J.; Jacoby, C.; Vucur, M.; Gautheron, J.; Roderburg, C.; Borg, N.; Reisinger, F.; et al. RIP3, a kinase promoting necroptotic cell death, mediates adverse remodelling after myocardial infarction. Cardiovasc. Res. 2014, 103, 206–216. [Google Scholar] [CrossRef]
- Degterev, A.; Huang, Z.; Boyce, M.; Li, Y.; Jagtap, P.; Mizushima, N.; Cuny, G.D.; Mitchison, T.J.; Moskowitz, M.A.; Yuan, J.; et al. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat. Chem. Biol. 2005, 1, 112–119. [Google Scholar] [CrossRef]
- Smith, C.C.T.; Davidson, S.M.; Lim, S.Y.; Simpkin, J.C.; Hothersall, J.S.; Yellon, D.M. Necrostatin: A potentially novel cardioprotective agent? Cardiovasc. Drugs Ther. 2007, 21, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Oerlemans, M.I.; Liu, J.; Arslan, F. Inhibition of RIP1-dependent necrosis prevents adverse cardiac remodeling after myocardial ischemia—Reperfusion in vivo. Basic Res. Cardiol 2012, 107, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Newton, K.; Dugger, D.L.; Maltzman, A.; Greve, J.M.; Hedehus, M.; Martin-McNulty, B.; Carano, R.A.D.; Cao, T.C.; Van Bruggen, N.; Bernstein, L.; et al. RIPK3 deficiency or catalytically inactive RIPK1 provides greater benefit than MLKL deficiency in mouse models of inflammation and tissue injury. Cell Death Differ. 2016, 23, 1565–1576. [Google Scholar] [CrossRef]
- Zhang, D.-Y.; Wang, B.-J.; Ma, M.; Yu, K.; Zhang, Q.; Zhang, X.-W. MicroRNA-325-3p protects the heart after myocardial infarction by inhibiting RIPK3 and programmed necrosis in mice. BMC Mol. Biol. 2019, 20, 1–14. [Google Scholar]
- Kaiser, W.J.; Sridharan, H.; Huang, C.; Mandal, P.; Upton, J.W.; Gough, P.J.; Sehon, C.A.; Marquis, R.W.; Bertin, J.; Mocarski, E.S. Toll-like Receptor 3-mediated Necrosis via TRIF, RIP3, and MLKL. J. Biol. Chem. 2013, 288, 31268–31279. [Google Scholar] [CrossRef]
- Dondelinger, Y.; Declercq, W.; Montessuit, S.; Roelandt, R.; Goncalves, A.; Bruggeman, I.; Hulpiau, P.; Weber, K.; Sehon, C.A.; Marquis, R.W.; et al. MLKL compromises plasma membrane integrity by binding to phosphatidylinositol phosphates. Cell Rep. 2014, 7, 971–981. [Google Scholar] [CrossRef]
- Rodriguez, D.; Weinlich, R.; Brown, S.L.; Guy, C.T.; Fitzgerald, P.J.; Dillon, C.P.; Oberst, A.; Quarato, G.; Low, J.; Cripps, J.G.; et al. Characterization of RIPK3-mediated phosphorylation of the activation loop of MLKL during necroptosis. Cell Death Differ. 2016, 23, 76–88. [Google Scholar] [CrossRef]
- Li, J.-X.; Feng, J.-M.; Wang, Y.; Li, X.-H.; Chen, X.-X.; Su, Y.; Shen, Y.-Y.; Chen, Y.; Xiong, B.; Yang, C.-H.; et al. The B-RafV600E inhibitor dabrafenib selectively inhibits RIP3 and alleviates acetaminophen-induced liver injury. Cell Death Dis. 2014, 5, e1278. [Google Scholar] [CrossRef]
- Kashlov, J.K.; Donev, I.S.; Doneva, J.G.; Valkov, V.D.; Kirkorova, A.D.; Ghenev, P.I.; Conev, N.; Radeva, T.R.; Ivanov, B.D.; Georgieva, Z.T. Serum levels of RIPK3 and troponin I as potential biomarkers for predicting impaired left ventricular function in patients with myocardial infarction with ST segment elevation and normal troponin I levels prior percutaneous coronary intervention. Biosci. Trends 2016, 10, 294–299. [Google Scholar] [CrossRef]
- Hu, X.-M.; Chen, X.; Pang, H.-Y.; Liu, H.-H.; Chen, P.-P.; Shi, J.-L.; Tang, S.; Wu, Z.-H.; Zhang, S.-Y. Plasma levels of receptor interacting protein kinase-3 correlated with coronary artery disease. Chin. Med. J. 2019, 132, 1400–1405. [Google Scholar] [CrossRef] [PubMed]
- Barthels, D.; Das, H. Current advances in ischemic stroke research and therapies. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2020, 1866, 165260. [Google Scholar] [CrossRef]
- Kim, J.S.; Nah, H.-W.; Park, S.M.; Kim, S.-K.; Cho, K.H.; Lee, J.; Lee, Y.-S.; Kim, J.; Ha, S.-W.; Kim, E.-G.; et al. Risk factors and stroke mechanisms in atherosclerotic stroke. Stroke 2012, 43, 3313–3318. [Google Scholar] [CrossRef] [PubMed]
- Hribljan, V.; Lisjak, D.; Petrović, D.J.; Mitrečić, D. Necroptosis is one of the modalities of cell death accompanying ischemic brain stroke: From pathogenesis to therapeutic possibilities. Croat. Med. J. 2019, 60, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.-S.; Yi, T.-L.; Zhang, S.; Xu, Z.-W.; Yu, Z.-Q.; Sun, H.-T.; Yang, C.; Tu, Y.; Cheng, S.-X. Hypoxia-inducible factor-1 alpha is involved in RIP-induced necroptosis caused by in vitro and in vivo ischemic brain injury. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Deng, X.; Li, S.; Sun, F. Necrostatin-1 prevents necroptosis in brains after ischemic stroke via inhibition of RIPK1-mediated RIPK3/MLKL signaling. Aging Dis. 2019, 10, 807–817. [Google Scholar] [CrossRef]
- Naito, M.G.; Xu, D.; Amin, P.; Lee, J.; Wang, H.; Li, W.; Kelliher, M.; Pasparakis, M.; Yuan, J. Sequential activation of necroptosis and apoptosis cooperates to mediate vascular and neural pathology in stroke. Proc. Natl. Acad. Sci. USA 2020, 117, 4959–4970. [Google Scholar] [CrossRef]
- Yang, J.; Zhao, Y.; Zhang, L.; Fan, H.; Qi, C.; Zhang, K.; Liu, X.; Fei, L.; Chen, S.; Wang, M.; et al. RIPK3/MLKL-mediated neuronal necroptosis modulates the M1/M2 polarization of microglia/macrophages in the ischemic cortex. Cereb. Cortex 2018, 28, 2622–2635. [Google Scholar] [CrossRef]
- Xu, X.; Chua, K.-W.; Chua, C.C.; Liu, C.-F.; Hamdy, R.C.; Chua, B.H. Synergistic protective effects of humanin and necrostatin-1 on hypoxia and ischemia/reperfusion injury. Brain Res. 2010, 1355, 189–194. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, B.; Tu, H.; Tang, Y.; Xu, C.; Chen, Y.; Zhao, Z.; Miao, Z. The degradation of mixed lineage kinase domain-like protein promotes neuroprotection after ischemic brain injury. Oncotarget 2017, 8, 68393–68401. [Google Scholar] [CrossRef]
- Chen, H.-H.; Cruz, S.A.; Qin, Z.; Stewart, A.F.R. Dabrafenib, an inhibitor of RIP3 kinase-dependent necroptosis, reduces ischemic brain injury. Neural Regen. Res. 2018, 13, 252–256. [Google Scholar] [CrossRef]
- Evans, G.H.; Stansby, G.; Hamilton, G. Suggested standards for reporting on arterial aneurysms. J. Vasc. Surg. 1992, 15, 456. [Google Scholar] [CrossRef]
- Kuivaniemi, H.; Ryer, E.J.; Elmore, J.R.; Tromp, G. Understanding the pathogenesis of abdominal aortic aneurysms. Expert Rev. Cardiovasc. Ther. 2015, 13, 975–987. [Google Scholar] [CrossRef] [PubMed]
- Chaikof, E.L.; Dalman, R.L.; Eskandari, M.K.; Jackson, B.M.; Lee, W.A.; Mansour, M.A.; Mastracci, T.M.; Mell, M.; Murad, M.H.; Nguyen, L.L.; et al. The Society for Vascular Surgery practice guidelines on the care of patients with an abdominal aortic aneurysm. J. Vasc. Surg. 2018, 67, 2–77. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Rateri, D.L.; Bruemmer, D.; Cassis, L.A.; Daugherty, A. Novel mechanisms of abdominal aortic aneurysms. Curr. Atheroscler. Rep. 2012, 14, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Mitchell, R.N.; Libby, P. Inflammation and cellular immune responses in abdominal aortic aneurysms. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 987–994. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Liu, Z.; Ren, J.; Morgan, S.; Assa, C.; Liu, B. Receptor-interacting protein kinase 3 contributes to abdominal aortic aneurysms via smooth muscle cell necrosis and inflammation. Circ. Res. 2015, 116, 600–611. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liao, M.F.; Tian, L.; Zou, S.L.; Lu, Q.S.; Bao, J.M.; Pei, Y.F.; Jing, Z.P. Overexpression of interleukin-1 b and interferon- g in type I thoracic aortic dissections and ascending thoracic aortic aneurysms: Possible correlation with matrix metalloproteinase-9 expression and apoptosis of aortic media cells. Eur. J. Cardio Thorac. Surg. 2011, 40, 17–22. [Google Scholar] [CrossRef]
- Liu, Z.; Fitzgerald, M.; Meisinger, T.; Batra, R.; Suh, M.; Greene, H.; Penrice, A.J.; Sun, L.; Baxter, B.T.; Xiong, W. CD95-ligand contributes to abdominal aortic aneurysm progression by modulating inflammation. Cardiovasc. Res. 2018, 115, 807–818. [Google Scholar] [CrossRef]
- Xiong, W.; MacTaggart, J.N.; Knispel, R.; Worth, J.M.; Persidsky, Y.; Baxter, B.T. Blocking TNF-α attenuates aneurysm formation in a murine model. J. Immunol. 2009, 183, 2741–2746. [Google Scholar] [CrossRef]
- Luo, W.; Wang, Y.; Zhang, L.; Ren, P.; Zhang, C.; Li, Y.; Azares, A.R.; Zhang, M.; Guo, J.; Ghaghada, K.B.; et al. Critical role of cytosolic DNA and its sensing adaptor STING in aortic degeneration, dissection, and rupture. Circulation 2020, 141, 42–66. [Google Scholar] [CrossRef] [PubMed]
- Juang, D.; Braverman, A.C.; Eagle, K. Aortic Dissection. Circulation 2008, 118, 507–510. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhou, T.; Liu, Z.; Ren, J.; Phan, N.; Gupta, K.; Stewart, D.M.; Morgan, S.; Assa, C.; Kent, K.C.; et al. Inhibition of receptor-interacting protein kinase 1 with necrostatin–1s ameliorates disease progression in elastase-induced mouse abdominal aortic aneurysm model. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef]
- Zhou, T.; Wang, Q.; Phan, N.; Ren, J.; Yang, H.; Feldman, C.C.; Feltenberger, J.B.; Ye, Z.; Wildman, S.A.; Tang, W.; et al. Identification of a novel class of RIP1/RIP3 dual inhibitors that impede cell death and inflammation in mouse abdominal aortic aneurysm models. Cell Death Dis. 2019, 10, 1–15. [Google Scholar] [CrossRef]
- Khoury, M.K.; Zhou, T.; Yang, H.; Prince, S.R.; Gupta, K.; Stranz, A.R.; Wang, Q.; Liu, B. GSK2593074A blocks progression of exisiting abdominal aortic dilation. JVS Vasc. Sci. 2020, 1, 123–135. [Google Scholar] [CrossRef]
- Speelman, L.; Schurink, G.W.H.; Bosboom, E.M.H.; Buth, J.; Breeuwer, M.; Van De Vosse, F.N.; Jacobs, M.H. The mechanical role of thrombus on the growth rate of an abdominal aortic aneurysm. J. Vasc. Surg. 2010, 51, 19–26. [Google Scholar] [CrossRef]
- Adolph, R.; Vorp, D.A.; Steed, D.L.; Webster, M.W.; Kameneva, M.V.; Watkins, S.C. Cellular content and permeability of intraluminal thrombus in abdominal aortic aneurysm. J. Vasc. Surg. 1997, 25, 916–926. [Google Scholar] [CrossRef]
- Piechota-Polańczyk, A.; Ejózkowicz, A.; Nowak, W.N.; Eeilenberg, W.; Eneumayer, C.; Emalinski, T.; Ehuk, I.; Brostjan, C. The abdominal aortic aneurysm and intraluminal thrombus: Current concepts of development and treatment. Front. Cardiovasc. Med. 2015, 2, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Vorp, D.A.; Lee, P.C.; Wang, D.H.; Makaroun, M.S.; Nemoto, E.M.; Ogawa, S.; Webster, M.W. Association of intraluminal thrombus in abdominal aortic aneurysm with local hypoxia and wall weakening. J. Vasc. Surg. 2001, 34, 291–299. [Google Scholar] [CrossRef]
- Moran, C.S.; Seto, S.W.; Krishna, S.M.; Sharma, S.; Jose, R.J.; Biros, E.; Wang, Y.; Morton, S.K.; Golledge, J. Parenteral administration of factor Xa/IIa inhibitors limits experimental aortic aneurysm and atherosclerosis. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Sidloff, D.A.; Stather, P.W.; Choke, E.; Bown, M.; Sayers, R.D. A systematic review and meta-analysis of the association between markers of hemostasis and abdominal aortic aneurysm presence and size. J. Vasc. Surg. 2014, 59, 528–535. [Google Scholar] [CrossRef]
- Harter, L.P.; Gross, B.H.; Callen, P.W.; Barth, R.A. Ultrasonic evaluation of abdominal aortic thrombus. J. Ultrasound Med. 1982, 1, 315–318. [Google Scholar] [CrossRef]
- Benjamin, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Chiuve, S.E.; Cushman, M.; Delling, F.N.; Deo, R.; et al. Heart disease and stroke statistics—2017 update a report from the American heart association. Circulation 2017, 135, e146–e603. [Google Scholar] [CrossRef] [PubMed]
- Gale, A.J. Currrent understanding of hemostasis. Toxicol. Pathol. 2011, 39, 273–280. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, J.; Yan, R.; Tian, J.; Zhang, Y.; Zhang, J.; Chen, M.; Cui, Q.; Zhao, L.; Hu, R.; et al. Receptor-interacting protein kinase 3 promotes platelet activation and thrombosis. Proc. Natl. Acad. Sci. USA 2017, 114, 2964–2969. [Google Scholar] [CrossRef]
- Bowie, E.J.W.; Owen, C.A. Standardization of the bleeding time. Scand. J. Haematol. 1980, 24, 87–94. [Google Scholar] [CrossRef]
- Saito, M.S.; Lourenço, A.L.; Kang, H.C.; Rodrigues, C.R.; Cabral, L.M.; Castro, H.C.; Satlher, P.C. New approaches in tail-bleeding assay in mice: Improving an important method for designing new anti-thrombotic agents. Int. J. Exp. Pathol. 2016, 97, 285–292. [Google Scholar] [CrossRef]
- Von Brühl, M.-L.; Stark, K.; Steinhart, A.; Chandraratne, S.; Konrad, I.; Lorenz, M.; Khandoga, A.; Tirniceriu, A.; Coletti, R.; Köllnberger, M.; et al. Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J. Exp. Med. 2012, 209, 819–835. [Google Scholar] [CrossRef] [PubMed]
- Martinod, K.; Demers, M.; Fuchs, T.A.; Wong, S.L.; Brill, A.; Gallant, M.; Hu, J.; Wang, Y.; Wagner, D.D. Neutrophil histone modi fi cation by peptidylarginine deiminase 4 is critical for deep vein thrombosis in mice. Proc. Natl. Acad. Sci. USA 2013, 110, 8674–8679. [Google Scholar] [CrossRef]
- Brill, A.; Fuchs, T.A.; Savchenko, A.S.; Thomas, G.M.; Martinod, K.; De Meyer, S.F.; Bhandari, A.A.; Wagner, D.D. Neutrophil extracellular traps promote deep vein thrombosis in mice. J. Thromb. Haemost. 2012, 10, 136–144. [Google Scholar] [CrossRef]
- Martinod, K.; Wagner, D.D. Thrombosis: Tangled up in NETs. Blood 2014, 123, 2768–2776. [Google Scholar] [CrossRef] [PubMed]
- Diaz, J.A.; Fuchs, T.A.; Jackson, T.O.; Hovinga, J.A.K.; Lämmle, B.; Henke, P.K.; Myers, D.D.J.; Wagner, D.D.; Wakefield, T.W.; Group, M.R.V.; et al. Plasma DNA is elevated in patients with deep vein thrombosis. J. Vasc. Surg. Venous Lym. Dis. 2013, 1, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Yipp, B.G.; Kubes, P. NETosis: How vital is it? Blood 2013, 122, 2784–2794. [Google Scholar] [CrossRef]
- Desai, J.; Kumar, S.V.; Mulay, S.R.; A Konrad, L.; Romoli, S.; Schauer, C.; Herrmann, M.; Bilyy, R.; Müller, S.; Popper, B.; et al. PMA and crystal-induced neutrophil extracellular trap formation involves RIPK1-RIPK3-MLKL signaling. Eur. J. Immunol. 2015, 46, 223–229. [Google Scholar]
- Nakazawa, D.; Desai, J.; Steiger, S.; Müller, S.; Devarapu, S.K.; Mulay, S.R.; Iwakura, T.; Anders, H.-J. Activated platelets induce MLKL-driven neutrophil necroptosis and release of neutrophil extracellular traps in venous thrombosis. Cell Death Discov. 2018, 4, 1–11. [Google Scholar] [CrossRef]
- Najjar, M.; Suebsuwong, C.; Ray, S.S.; Thapa, R.J.; Maki, J.L.; Nogusa, S.; Shah, S.; Saleh, D.; Gough, P.J.; Bertin, J.; et al. Structure guided design of potent and selective ponatinib-based hybrid inhibitors for RIPK1. Cell Rep. 2015, 10, 1850–1860. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.A.; Bandyopadhyay, D.; Berger, S.B.; Campobasso, N.; Capriotti, C.A.; Cox, J.A.; Dare, L.; Finger, J.N.; Hoffman, S.J.; Kahler, K.M.; et al. Discovery of small molecule RIP1 kinase inhibitors for the treatment of pathologies associated with necroptosis. ACS Med. Chem. Lett. 2013, 4, 1238–1243. [Google Scholar] [CrossRef]
- Berger, S.; Harris, P.; Nagilla, R.; Kasparcova, V.; Hoffman, S.; Swift, B.; Dare, L.; Schaeffer, M.; Capriotti, C.; Ouellette, M.; et al. Characterization of GSK’963: A structurally distinct, potent and selective inhibitor of RIP1 kinase. Cell Death Discov. 2015, 1, 1–7. [Google Scholar] [CrossRef]
- Ren, Y.; Su, Y.; Sun, L.; He, S.; Meng, L.; Liao, D.; Liu, X.; Ma, Y.; Liu, C.; Li, S.; et al. Discovery of a highly potent, selective, and metabolically stable inhibitor of receptor-interacting protein 1 (RIP1) for the treatment of systemic in fl ammatory response syndrome. J. Med. Chem. 2017, 60, 972–986. [Google Scholar] [CrossRef]
- Rojas-Rivera, D.; Delvaeye, T.; Roelandt, R.; Nerinckx, W.; Augustyns, K.; Vandenabeele, P.; Bertrand, M.J.M. When PERK inhibitors turn out to be new potent RIPK1 inhibitors: Critical issues on the specificity and use of GSK2606414 and GSK2656157. Cell Death Differ. 2017, 24, 1100–1110. [Google Scholar] [CrossRef]
- Le Cann, F.; Delehouzé, C.; Leverrier-Penna, S.; Filliol, A.; Comte, A.; Delalande, O.; Desban, N.; Baratte, B.; Gallais, I.; Piquet-Pellorce, C.; et al. Sibiriline, a new small chemical inhibitor of receptor-interacting protein kinase 1, prevents immune-dependent hepatitis. FEBS J. 2017, 284, 3050–3068. [Google Scholar] [CrossRef] [PubMed]
- Martens, S.; Hofmans, S.; Declercq, W.; Augustyns, K.; Vandenabeele, P. Inhibitors targeting RIPK1/RIPK3: Old and new drugs. Trends Pharmacol. Sci. 2020, 41, 209–224. [Google Scholar] [CrossRef] [PubMed]
- Fauster, A.; Rebsamen, M.; Huber, K.; Bigenzahn, J.W.; Stukalov, A.; Lardeau, C.-H.; Scorzoni, S.; Bruckner, M.K.; Gridling, M.; Parapatics, K.; et al. A cellular screen identifies ponatinib and pazopanib as inhibitors of necroptosis. Cell Death Dis. 2015, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Martens, S.; Jeong, M.; Tonnus, W.; Feldmann, F.; Hofmans, S.; Goossens, V.; Takahashi, N.; Braesen, J.H.; Lee, E.-W.; Van Der Veken, P.; et al. Sorafenib tosylate inhibits directly necrosome complex formation and protects in mouse models of inflammation and tissue injury. Cell Death Dis. 2017, 8, 1–12. [Google Scholar] [CrossRef]
| Disease | Model/Subjects | Inhibitor | Pertinent Findings | Ref. |
|---|---|---|---|---|
| Atherosclerosis | Human plaques, Apoe−/− mice, oxLDL BMDM treatment | Nec-1 | RIPK3 and MLKL expression & activation increased in human plaques, Nec-1 reduces plaque size/necrotic core in mice, reduced ox-LDL induced necroptosis in BMDM | [12] |
| Atherosclerosis | Human plaques, in vitro serum starvation foam cell model | None | RIPK1/3 expression increased in plaques, serum starvation increases RIPK1/RIPK3 expression, MLKL oligomerization | [19] |
| Atherosclerosis | ox-LDL HUVEC treatment | Nec-1 | Ox-LDL increases RIPK1 expression & inflammation, nec-1 ameliorates this effect | [21] |
| Atherosclerosis | Ripk3−/−; Ldlr−/− mice, Apoe−/− mice | None | Ripk3−/Ldlr−/− mice−mice have significantly smaller advanced plaques | [22] |
| Atherosclerosis | Apoe−/− mice | Anti-sense MLKL oligonucleotides | MLKL knockdown decreased necrotic core size but not plaque size, decreased lipid levels | [26] |
| MI | Rat LAD ligation | Nec-1 | RIPK1/RIPK3 increased in cardiac tissue after MI, Nec-1 decreased infarct size | [30] |
| MI | Mouse LAD ligation, Ripk3−/− mice | None | RIPK3 increased in cardiac tissue after MI, EF preserved in Ripk3−/− mice after LAD ligation | [31] |
| MI | Mouse and rat-derived cardiomyocytes, mouse LAD ligation | Nec-1 | Nec-1 reduced peroxide induced cell death, murine infarct size | [33] |
| MI | Mouse LAD ligation | Nec-1 | Nec-1 reduced infarct size, necrotic cell death, prevented adverse remodeling at 28 days | [34] |
| MI | Ripk3−/− mice, Mouse LAD ligation | None | Reduced infarct size in Ripk3−/− mice | [35] |
| MI | Human STEMI patients | None | In patients with normal troponin on presentation, serum RIPK3 predicts impaired LV function | [41] |
| MI | Humans with CAD, angina, unstable angina | None | Plasma RIPK3 correlates with CAD severity | [42] |
| Stroke | Mouse MCAO model | Nec-1 | Intracerebroventricular Nec-1 reduced infarct volume | [32] |
| Stroke | Oxygen-deprived glucose (ODG) in vitro model, MCAO mouse model | GSK’872 | ODG and MCAO upregulate RIPK1, RIPK3, MLKL, GSK’872 reduces infarct volume | [46] |
| Stroke | Rat MCAO model | Nec-1 | Ischemia activates RIPK1/3/MLKL signaling. Nec-1 reduces infarct volume | [47] |
| Stroke | Mouse MCAO model, Ripk3−/− mice, Ripk1D138N/D138N mice | None | Inactivation of RIPK1 and absence of RIPK3 can ultimately decrease stroke volume, improve behavioral scores | [48] |
| Stroke | Mouse MCAO model, ODG in vitro model, Ripk3−/− and Mlkl−/− mice | None | RIPK3 or MLKL deficiency decreases stroke size, neurologic deficits, polarizes macrophages to M2 phenotype | [49] |
| Stroke | Mouse MCAO model, ODG in vitro model | Nec-1 | Nec-1 protects cells from ODG related death, Nec-1 reduced infarct volume | [50] |
| Stroke | Mouse MCAO model | NSA | Decreased infarct size, neurologic deficits, MLKL levels; increased MLKL degradation after NSA treatment | [51] |
| Stroke | Photothrombosis induced ischemic injury in mouse | Dabrafenib | Dabrafenib reduced infarct size, inflammation | [52] |
| AAA | Murine elastase perfusion model, Ripk3−/− mice | None | RIPK1/RIPK3 are locally upregulated in AAA, Ripk3−/− mice are protected from AAA | [58] |
| AAA | Murine elastase perfusion model | Nec-1s | Nec-1s slows aneurysm growth, decreases inflammation, preserves vessel architecture | [64] |
| AAA | Murine CaCl2 model, murine AngII Apoe−/− model | GSK’074 | GSK’074 can prevent aneurysm growth, preserve vessel architecture in both aneurysm models | [65] |
| AAA | Murine CaCl2 model | GSK’074 | GSK’074 slows aneurysm growth, preserves vessel architecture | [66] |
| AAA | Murine AngII and CaCl2 model, cell culture | None | STING deficiency decreases necroptosis and protects mice from AAA | [62] |
| Arterial thrombosis | Murine FeCl3 injury model, tail bleeding, platelet activity assays, Ripk3−/− mice | None | Ripk3−/− mice have prolonged tail bleeding, FeCl3 arteriole time to occlusion, abnormal dense granule secretion | [76] |
| Venous Thrombosis | IVC ligation model, Mlkl−/− mice | Nec-1s, NSA | Nec-1s treatment and MLKL deficiency decrease thrombus size, decrease NETosis. Nec-1s and necrosulfonamide decrease platelet-neutrophil aggregation | [86] |
| Inhibitor Name | Molecular Target | Tested Applications | Use in Clinical Trials: Yes/No | Ref. |
|---|---|---|---|---|
| Necrostatin-1 | RIPK1 | Atherosclerosis §, stroke §, MI § | No | [12,21,30,32,33,34,47,50] |
| Necrostatin-1s | RIPK1 | AAA §, venous thrombosis § | No | [64,86] |
| PN10 | RIPK1 | TNFα induced SIRS § | No | [87] |
| cdp27 | RIPK1 | TNFα induced SIRS § | No | [88] |
| GSK′963 | RIPK1 | TNFα induced SIRS § | No | [89] |
| RIPA-56 | RIPK1 | TNFα induced SIRS § | No | [90] |
| GSK2656157 | RIPK1 | TNFα induced SIRS § | No | [91] |
| Sibiriline | RIPK1 | concanavalin A-induced hepatitis § | No | [92] |
| GSK’872 | RIPK3 | Stroke §, | No | [46] |
| GSK’074 | RIPK1 & RIPK3 | AAA §, | No | [65,66] |
| DNL747 | RIPK1 | Alzheimer’s disease, ALS, MS | Yes- Phase I | [87] |
| GSK2982772 | RIPK1 | Psoriasis, UC, RA | Yes- Phase II | [93] |
| Dabrafenib | RIPK3 | Stroke §, Metastatic melanoma | Yes- Metastatic melanoma, FDA approved | [52,87] |
| Ponatinib | RIPK1&RIPK3 | TNFα induced SIRS § | Yes- FDA approved for CML and Ph+ALL | [87,94] |
| Sorafenib | RIPK1&RIPK3 | TNFα induced SIRS § and renal ischemia–reperfusion injury § | Yes- FDA approved for advanced liver cancer; renal cancer; thyroid cancer | [95] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
DeRoo, E.; Zhou, T.; Liu, B. The Role of RIPK1 and RIPK3 in Cardiovascular Disease. Int. J. Mol. Sci. 2020, 21, 8174. https://doi.org/10.3390/ijms21218174
DeRoo E, Zhou T, Liu B. The Role of RIPK1 and RIPK3 in Cardiovascular Disease. International Journal of Molecular Sciences. 2020; 21(21):8174. https://doi.org/10.3390/ijms21218174
Chicago/Turabian StyleDeRoo, Elise, Ting Zhou, and Bo Liu. 2020. "The Role of RIPK1 and RIPK3 in Cardiovascular Disease" International Journal of Molecular Sciences 21, no. 21: 8174. https://doi.org/10.3390/ijms21218174
APA StyleDeRoo, E., Zhou, T., & Liu, B. (2020). The Role of RIPK1 and RIPK3 in Cardiovascular Disease. International Journal of Molecular Sciences, 21(21), 8174. https://doi.org/10.3390/ijms21218174





