Calcium Plays a Double-Edged Role in Modulating Cadmium Uptake and Translocation in Rice
Abstract
:1. Introduction
2. Results
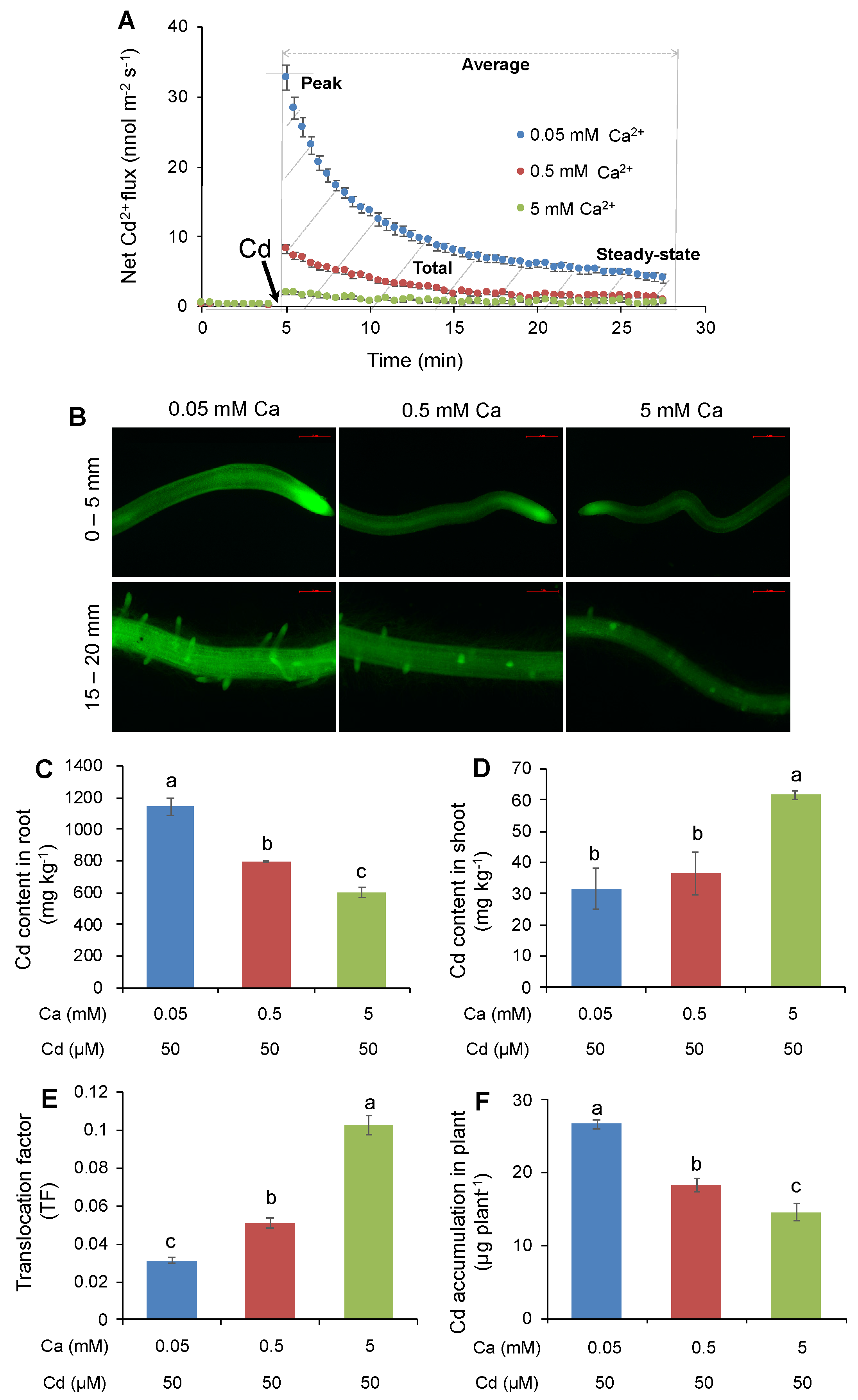
2.1. Effect of External Ca Concentrations on Net Cd2+ Flux and Distribution of Cd in Roots and Tissue Cd Contents of Rice Plants at Seedling Stage
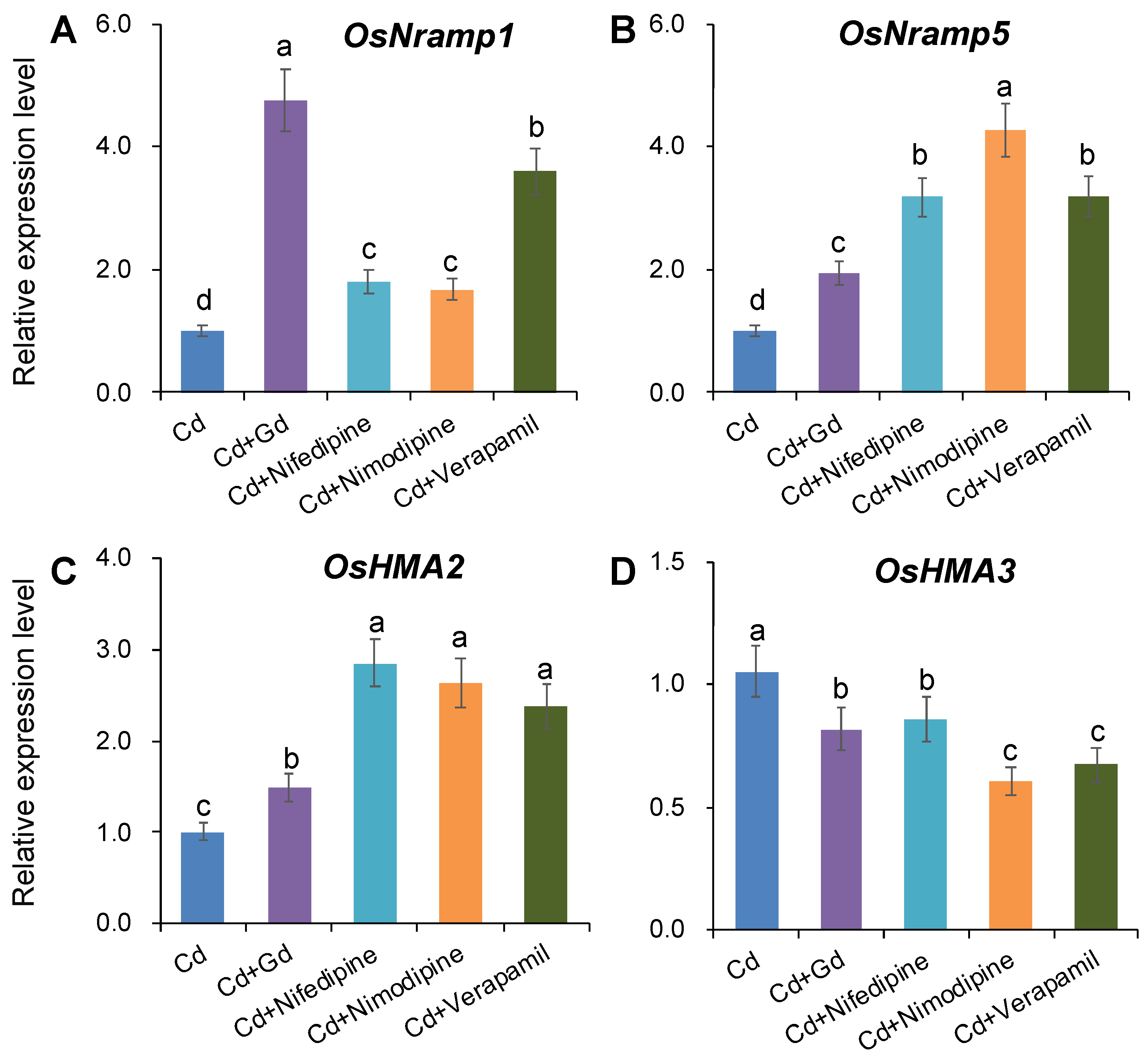
2.2. Effects of Ca Channel Blockers on Net Cd2+ Flux and Distribution of Cd in Rice Roots and Tissue Cd Contents
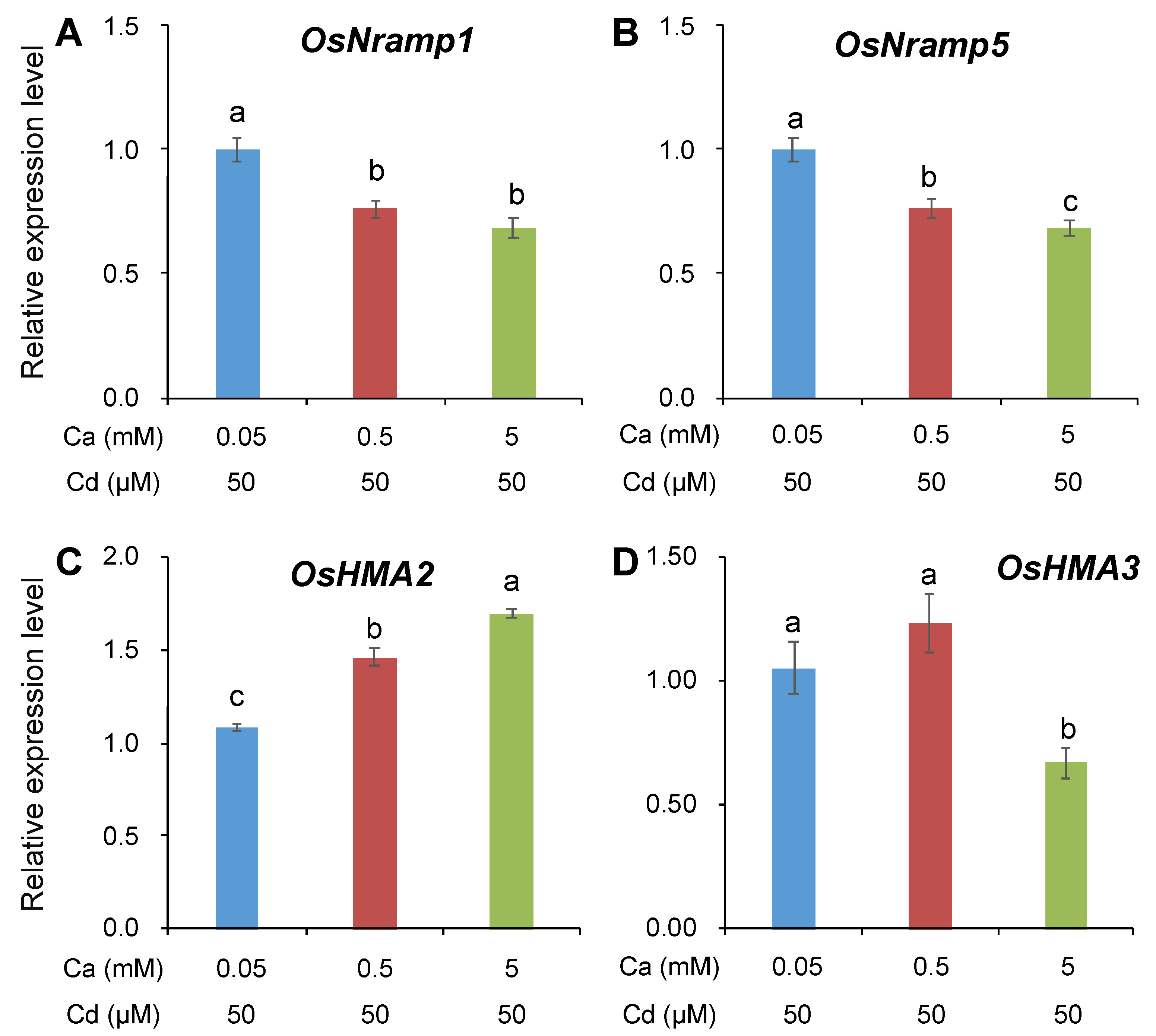
2.3. Gene Expression under Different Ca Concentrations or Pharmacological Treatments
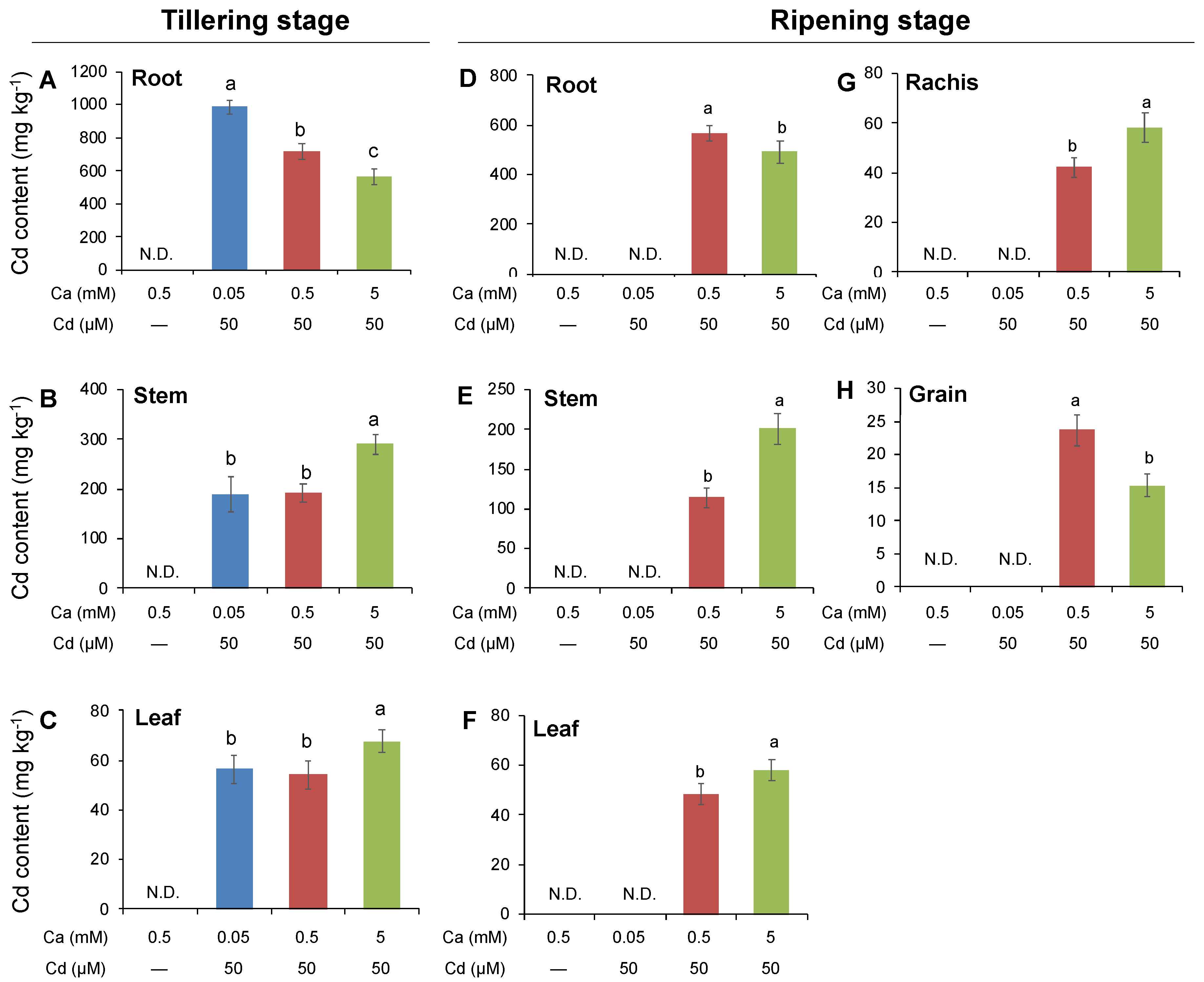
2.4. Effects of External Ca Concentration on Plant Growth and Tissue Cd Content in Rice Plants at Tillering and Ripening Stages
3. Discussion
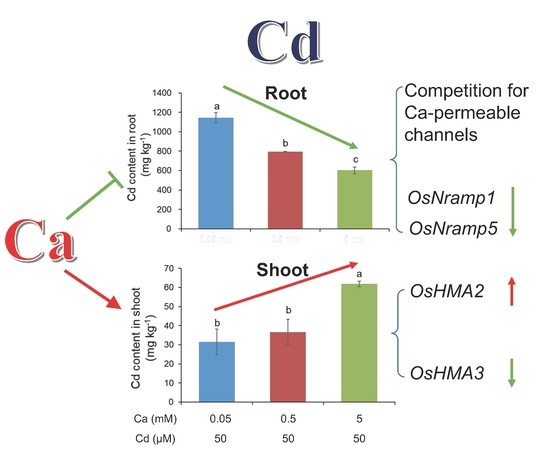
3.1. Ca Plays a Double-Edged Role in Regulating Root Cd Uptake and Root-To-Shoot Cd Translocation in Rice
3.2. Ca Decreases Root Cd Uptake in Rice Not Only by Competing for Absorption Sites but Also by Suppressing the Expression of OsNRAMPs
3.3. Ca Facilitates Root-To-Shoot Cd Translocation in Rice by Modulating the Expression of OsHMAs
3.4. The Potentiality of Ca-Based Amendments for Low-Cd Rice Production
4. Materials and Methods
4.1. Plant Materials, Growth Conditions, and Treatments
4.2. Measurement of Cd Content in Rice Tissues
4.3. Microelectrode Measurements of Net Cd2+ Flux
4.3.1. Procedure of Microelectrode Ion Flux Measurements
4.3.2. Transient Cd2+ Flux Kinetics
4.3.3. Pharmacological Experiments
4.4. Histochemical Detection of Cd in Rice Root
4.5. RNA Extraction and Gene Expression Analysis
4.6. Statistics Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Clemens, S. Safer food through plant science: Reducing toxic element accumulation in crops. J. Exp. Bot. 2019, 70, 5537–5557. [Google Scholar] [CrossRef] [PubMed]
- Gallego, S.M.; Pena, L.B.; Barcia, R.A.; Azpilicueta, C.E.; Iannone, M.F.; Rosales, E.P.; Zawoznika, M.S.; Groppaa, M.D.; Benavidesa, M.P. Unravelling cadmium toxicity and tolerance in plants: Insight into regulatory mechanisms. Environ. Exp. Bot. 2012, 83, 33–46. [Google Scholar] [CrossRef]
- Shahid, M.; Dumat, C.; Khalid, S.; Niazi, N.K.; Antunes, P.M.C. Cadmium bioavailability, uptake, toxicity and detoxification in soil-plant system. Rev. Environ. Contam.Toxicol. 2017, 241, 73–137. [Google Scholar] [PubMed]
- Ismael, M.A.; Elyamine, A.M.; Moussa, M.G.; Cai, M.M.; Zhao, X.H.; Hu, C.X. Cadmium in plants: Uptake, toxicity, and its interactions with selenium fertilizers. Metallomics 2019, 2, 255–277. [Google Scholar] [CrossRef] [PubMed]
- Bączek-Kwinta, R.; Juzoń, K.; Borek, M.; Antonkiewicz, J. Photosynthetic response of cabbage in cadmium-spiked soil. Photosynthetica 2019, 57, 731–739. [Google Scholar] [CrossRef] [Green Version]
- Abedi, T.; Mojiri, A. Cadmium uptake by wheat (Triticum aestivum L.): An overview. Plants 2020, 9, 500. [Google Scholar] [CrossRef] [Green Version]
- Abedi, T.; Mojiri, A. Arsenic uptake and accumulation mechanisms in rice species. Plants 2020, 9, 129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, H.; Luo, N.; Li, W.Y.; Cai, Q.Y.; Li, H.Y.; Mo, C.H.; Wong, M.H. Cadmium in rice: Transport mechanisms, influencing factors, and minimizing measures. Environ. Pollut. 2017, 224, 622–630. [Google Scholar] [CrossRef]
- Chang, J.D.; Huang, S.; Konoshi, N.; Wang, P.; Chen, J.; Huang, X.Y.; Ma, J.F.; Zhao, F.J. Overexpression of the manganese/cadmium transporter OsNRAMP5 reduces cadmium accumulation in rice grain. J. Exp. Bot. 2020, 71, 5705–5715. [Google Scholar] [CrossRef]
- Shi, G.L.; Li, D.J.; Wang, Y.W.; Liu, C.H.; Hu, Z.B.; Luo, L.Q.; Rengel, Z.; Cai, Q.S. Accumulation and distribution of arsenic and cadmium in winter wheat (Triticum aestivum L.) at different developmental stages. Sci. Total. Environ. 2019, 667, 532–539. [Google Scholar] [CrossRef]
- Clemens, S.; Ma, J.F. Toxic heavy metal and metalloid accumulation in crop plants and foods. Annu. Rev. Plant Biol. 2016, 67, 489–512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clemens, S. Toxic metal accumulation responses to exposure and mechanisms of tolerance in plants. Biochimie 2006, 88, 1707–1719. [Google Scholar] [CrossRef]
- Schaaf, G.; Ludewig, U.; Erenoglu, B.; Mori, S.; Kitahara, T.; von Wirén, N. ZmYS1 functions as a proton-coupled symporter for phytosiderophore- and nicotianamine-chelated metals. J. Biol.Chem. 2004, 279, 9091–9096. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.; An, G. Over-expression of OsIRT1 leads to increased iron and zinc accumulation in rice. Plant. Cell Environ. 2009, 32, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, A.; Yamaji, N.; Yokosho, K.; Ma, J.F. NRAMP5 is a major transporter responsible for manganese and cadmium uptake in rice. Plant. Cell 2012, 24, 2155–2167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, H.; Xu, W.; Xie, J. Variation of a major facilitator superfamily gene contributes to differential cadmium accumulation between rice subspecies. Nat. Commun. 2019, 10, 2562. [Google Scholar] [CrossRef] [Green Version]
- Uraguchi, S.; Fujiwara, T. Cadmium transport and tolerance in rice: Perspectives for reducing grain cadmium accumulation. Rice 2012, 5, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Lu, C.; Zhang, L.; Tang, Z.; Huang, X.Y.; Ma, J.F.; Zhao, F.J. Producing cadmium-free Indica rice by overexpressing OsHMA3. Environ. Int. 2019, 126, 619–626. [Google Scholar] [CrossRef]
- Takahashi, R.; Ishimaru, Y.; Shimo, H.; Ogo, Y.; Senoura, Y.; Nishizawa, N.K.; Nakanishi, H. The OsHMA2 transporter is involved in root-to-shoot translocation of Zn and Cd in rice. Plant Cell Environ. 2012, 35, 1948–1957. [Google Scholar] [CrossRef]
- Perfus-Barbeoch, L.; Leonhardt, N.; Vavasseur, A.; Forestier, C. Heavy metal toxicity: Cadmium permeates through calcium channels and disturbs the plant water status. Plant. J. 2002, 32, 539–548. [Google Scholar] [CrossRef]
- Chen, X.; Ouyang, Y.; Fan, Y.; Qiu, B.; Zhang, G.; Zeng, F. The pathway of transmembrane cadmium influx via calcium-permeable channels and its spatial characteristics along rice root. J. Exp. Bot. 2018, 69, 5279–5291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demidchik, V.; Shabala, S.; Isayenkov, S.; Cuin, T.A.; Pottosin, I. Calcium transport across plant membranes: Mechanisms and functions. New Phytol. 2018, 220, 49–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, C.; Zhang, Y.; Huang, C. Reduction in cadmium accumulation in japonica rice grains by CRISPR/Cas9-mediated editing of OsNRAMP5. J. Integr. Agric. 2019, 18, 688–697. [Google Scholar] [CrossRef]
- Grant, C.A.; Buckley, W.T.; Wu, R. Effect of selenium fertilizer source and rate of grain yield and selenium and cadmium concentration of durum wheat. Can. J. Plant. Sci. 2007, 87, 703–708. [Google Scholar] [CrossRef]
- Rostami, M.; Mohammadi, H.; Müller, T.; Mirzaeitalarposhti, R. Silicon application affects cadmium translocation and physiological traits of Lallemantia royleana under cadmium stress. J. Plant. Nutr. 2020, 43, 753–761. [Google Scholar] [CrossRef]
- Nakamura, S.; Suzui, N.; Yin, Y.; Ishii, S.; Fujimaki, S.; Kawachi, N.; Rai, H.; Matsumoto, T.; Sato-Izawa, K.; Ohkama-Ohtsu, N. Effects of enhancing endogenous and exogenous glutathione in roots on cadmium movement in Arabidopsis thaliana. Plant. Sci. 2020, 290, 110304. [Google Scholar] [CrossRef]
- Tian, S.; Lu, L.; Zhang, J.; Wang, K.; Brown, P.; He, Z.; Liang, J.; Yang, X. Calcium protets root of Sedum alfredii H. against cadmium-induced oxidatice stress. Chemosphere 2011, 84, 63–69. [Google Scholar] [CrossRef]
- Xu, J.; Wang, W.; Yin, H.; Liu, H.; Mi, Q. Exogenous nitric oxide improves antioxidatice capacity and reduces auxin degradation in roots of Medicago truncatula seedlings under cadmium stress. Plant. Soil 2010, 326, 321–330. [Google Scholar] [CrossRef]
- Thor, K. Calcium-nutrient and messenger. Front. Plant. Sci. 2019, 10, 440. [Google Scholar] [CrossRef]
- Huang, D.; Gong, X.; Liu, Y.; Zeng, G.; Lai, C.; Bashir, H.; Zhou, L.; Wang, D.; Xu, P.; Cheng, M.; et al. Effects of calcium at toxic concentrations of cadmium in plants. Planta 2017, 245, 863–873. [Google Scholar] [CrossRef]
- Gong, X.; Liu, Y.; Huang, D.; Zeng, G.; Liu, S.; Tang, H.; Zhou, L.; Hu, X.; Zhou, Y.; Tan, X. Effects of exogenous calcium and spermidine on cadmium stress moderation and metal accumulation in Boehmeria nivea (L.) Gaudich. Environ. Sci. Pollut. Res. 2016, 23, 8699–8708. [Google Scholar] [CrossRef] [PubMed]
- Eller, F.; Brix, H. Influence of low calcium availability on cadmium uptake and translocation in a fast-growing shrub and a metal-accumulating herb. AoB Plants 2016, 8, 143. [Google Scholar] [CrossRef] [Green Version]
- Ye, W.; Wu, F.; Zhang, G.; Fang, Q.; Lu, H.; Hu, H. Calcium decreases cadmium concentration in root but facilitates cadmium translocation from root to shoot in rice. J. Plant Growth Regul. 2020, 39, 422–429. [Google Scholar] [CrossRef]
- Demidchik, V.; Tester, M. Sodium fluxes through nonselective cation channels in the plasma membrane of protoplasts from Arabidopsis roots. Plant Physiol. 2002, 128, 379–387. [Google Scholar] [CrossRef] [PubMed]
- White, P. Calcium channels in higher plants. Biochim. Biophys. Acta Biomembr. 2000, 1465, 171–189. [Google Scholar] [CrossRef] [Green Version]
- Marchetti, C. Role of calcium channels in heavy metal toxicity. ISRN Toxicol. 2013, 2013, 184360. [Google Scholar] [CrossRef] [Green Version]
- Demidchik, V.; Davenport, R.J.; Tester, M. Nonselective cation channels in plants. Annu. Rev. Plant Biol. 2002, 53, 67–107. [Google Scholar] [CrossRef]
- Ahmad, P.; Latef, A.; Allah, E.; Hashem, A.; Sarwat, M.; Anjum, N.; Gucel, S. Calcium and potassium supplementation enhanced growth, osmolyte secondary metabolite production, and enzymatic antioxidant machinery in Cadmium-exposed chickpea (Cicer arietinum L.). Front. Plant Sci. 2016, 7, 513. [Google Scholar] [CrossRef] [Green Version]
- Wan, G.; Najeeb, U.; Jilani, G.; Naeem, M.; Zhou, W. Calcium invigorates the cadmium-stressed Brassica napus L. plants by strenthening their photosynthetic system. Environ. Sci. Pollut. Res. 2011, 18, 1478–1486. [Google Scholar] [CrossRef]
- Li, P.; Zhao, C.; Zhang, Y.; Wang, X.; Wang, X.; Wang, J.; Wang, F.; Bi, Y. Calcium alleviates cadium-induced inhibition on root growth by maintaining auxin homeostasis in Arabidopsis seedlings. Protoplasma 2015, 253, 185–200. [Google Scholar] [CrossRef]
- Tsukahara, T.; Ezaki, T.; Moriguchi, J.; Furuki, K.; Shimbo, S.; Matsuda-Inoguchi, N.; Ikeda, M. Rice as the most influential source of cadmium intake among general Japanese population. Sci. Total Environ. 2003, 305, 44–51. [Google Scholar] [CrossRef]
- Lindberg, S.; Landberg, T.; Greger, M. A new method to detect cadmium uptake in protoplasts. Planta 2004, 219, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Tian, S.; Zhang, M.; Zhang, J.; Yang, X.; Jiang, H. The role of Ca pathway in Cd uptake and translocation by the hyperaccumulator Sedum Alfredii. J. Hazard. Mater. 2010, 183, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Lux, A.; Martinka, M.; Vaculík, M.; White, P.J. Root responses to cadmium in the rhizosphere: A review. J. Exp. Bot. 2011, 62, 21–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, S.; Yu, J.; Zhu, M.; Zhao, F.; Luan, S. Cadmium impairs ion homeostasis by altering K+ and Ca2+ channel activities in rice root hair cells. Plant Cell Environ. 2012, 35, 1998–2013. [Google Scholar] [CrossRef] [PubMed]
- White, P.J.; Bowen, H.C.; Demidchik, V.; Nichols, C.; Davies, J.M. Genes for calcium-permeable channels in the plasma membrane of plant root cells. Biochim. Biophys. Acta 2002, 1564, 299–309. [Google Scholar] [CrossRef] [Green Version]
- Treesubsuntorn, C.; Thiravetyan, P. Calcium acetate-induced reduction of cadmium accumulation in Oryza sativa: Expression of auto-inhibited calcium-ATPase and cadmium transporters. Plant Biol. 2019, 21, 862–872. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; HUang, L.; Su, N.; Shabala, L.; Wang, H.; Huang, X.; Wen, R.; Yu, M.; Cui, J.; Shabala, S. Calcium-dependent hydrogen peroxide mediates hydrogen-rich water-reduced cadmium uptake in plant roots. Plant Physiol. 2020, 183, 1331–1344. [Google Scholar] [CrossRef]
- Zhou, Q.; Shao, G.S.; Zhang, Y.X.; Dong, Q.; Wang, H.; Cheng, S.H.; Cao, L.Y.; Shen, X.H. The difference of cadmium accumulation between the indica and japonica subspecies and the mechanism of it. Plant Growth Regul. 2017, 81, 523–532. [Google Scholar] [CrossRef]
- Hayakawa, N.; Tomioka, R.; Takenaka, C. Effects of calcium on cadmium uptake and transport in the tree species Gamblea innovans. Soil Sci. Plant Nutr. 2011, 57, 691–695. [Google Scholar] [CrossRef] [Green Version]
- Zeng, L.; Zhu, T.; Gao, Y.; Wang, Y.; Ning, C.; Björn, L.; Chen, D.; Li, S. Effects of Ca addition on the uptake, translocation, and distribution of Cd in Arabidopsis thaliana. Ecotox. Environ. Safe. 2017, 139, 228–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farzadfar, S.; Zarinkamar, F.; Modarres-Sanavy, S.; Hojati, M. Exogenously applied calcium alleviates cadmium toxicity in Matricaria chamomilla L. plants. Environ. Sci. Pollut. Res. Int. 2013, 20, 1413–1422. [Google Scholar] [CrossRef] [PubMed]
- Satoh-Nagasawa, N.; Mori, M.; Nakazawa, N.; Kawamoto, T.; Nagato, Y.; Sakurai, K.; Takahashi, H.; Watanabe, A.; Akagi, H. Mutations in rice (Oryza sativa) heavy metal ATPase 2 (OsHMA2) restrict the translocation of zinc and cadmium. Plant Cell Physiol. 2012, 53, 213–224. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, A.; Yamaji, N.; Ma, J.F. Overexpression of OsHMA3 enhances Cd tolerance and expression of Zn transporter genes in rice. J. Exp. Bot. 2014, 65, 6013–6021. [Google Scholar] [CrossRef] [Green Version]
- Zia-ur-Rehman, M.; Sabir, M.; Rizwan, M.; Saifullah Ahmed, H.R.; Nadeem, M. Remediating cadmium-contaminated soils by growing grain crops using inorganic amendments. In Soil Remediation and Plants: Prospects and Challenges; Academic Press: Cambridge, MA, USA, 2014; pp. 367–396. [Google Scholar]
- Yan, Y.; Zhou, Y.Q.; Liang, C.H. Evaluation of phosphate fertilizers for the immobilization of Cd in contaminated soils. PLoS ONE 2015, 10, e0124022. [Google Scholar] [CrossRef] [Green Version]
- Honma, T.; Ohba, H.; Kaneko-Kadokura, A.; Makino, T.; Nakamura, K.; Katou, H. Optimal soil Eh, pH, and water management for simultaneously minimizing arsenic and cadmium concentrations in rice grains. Environ. Sci. Technol. 2016, 50, 4178–4185. [Google Scholar] [CrossRef]
- Yu, L.L.; Zhu, J.Y.; Huang, Q.Q.; Su, D.C.; Jiang, R.F.; Li, H.F. Application of a rotation system to oilseed rape and rice fields in Cd-contaminated agricultural land to ensure food safety. Ecotox. Environ. Saf. 2014, 108, 287–293. [Google Scholar] [CrossRef]
- Song, X.; Hu, X.; Ji, P.; Li, Y.; Chi, G.; Song, Y. Phytoremediation of cadmium-contaminated farmland soil by the hyperaccumulator Beta vulgaris L. Var. Cicla. Bull. Environ. Contam. Toxicol. 2012, 88, 623–626. [Google Scholar] [CrossRef]
- Rehman, Z.U.; Rizwan, M.; Ghafoor, A.; Naeem, A.; Ali, S.; Sabir, M.; Qayyum, M.F. Effect of inorganic amendments for in situ stabilization of cadmium in contaminated soils and its phyto-availability to wheat and rice under rotation. Environ. Sci. Pollut. Res. 2015, 22, 16897–16906. [Google Scholar] [CrossRef]
- Mahar, A.; Wang, P.; Ali, A.; Lahori, A.H.; Awasthi, M.K.; Wang, Z. (Im) mobilization of soil heavy metals using CaO, FA, sulfur, and Na2S: A 1-year incubation study. Int. J. Environ. Sci. T. 2018, 15, 607–620. [Google Scholar] [CrossRef]
- Du, Y.; Wang, X.; Ji, X.; Zhang, Z.; Saha, U.K.; Xie, W.; Xie, Y.; Wu, J.; Peng, B.; Tan, C. Effectiveness and potential risk of CaO application in Cd-contaminated paddy soil. Chemosphere 2018, 204, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.; Chen, S.; Miao, Y.; Wu, F.; Zhang, G. Changes of organic acid exudation and rhizosphere pH in rice plants under chromium stress. Environ. Pollut. 2008, 155, 284–289. [Google Scholar] [CrossRef]
- Newman, I.A. Ion transport in roots: Measurement of fluxes using ionselective microelectrodes to characterize transporter function. Plant Cell Environ. 2001, 24, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Piñeros, M.A.; Shaff, J.E.; Kochian, L.V. Development, characterization, and application of a cadmium-selective microelectrode for the measurement of cadmium fluxes in roots of Thlaspi species and wheat. Plant Physiol. 1998, 116, 1393–1401. [Google Scholar] [CrossRef] [Green Version]
- Shabala, S. Non-invasive microelectrode ion flux measurements in plant stress physiology. In Plant Electrophysiology—Theory and Methods; Volkov, A., Ed.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 35–72. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using realtime quantitative PCR and the 2(-delta delta c(t)) method. Methods 2001, 25, 4. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, S.; Li, Q.; Nazir, M.M.; Ali, S.; Ouyang, Y.; Ye, S.; Zeng, F. Calcium Plays a Double-Edged Role in Modulating Cadmium Uptake and Translocation in Rice. Int. J. Mol. Sci. 2020, 21, 8058. https://doi.org/10.3390/ijms21218058
Zhang S, Li Q, Nazir MM, Ali S, Ouyang Y, Ye S, Zeng F. Calcium Plays a Double-Edged Role in Modulating Cadmium Uptake and Translocation in Rice. International Journal of Molecular Sciences. 2020; 21(21):8058. https://doi.org/10.3390/ijms21218058
Chicago/Turabian StyleZhang, Shuo, Qi Li, Muhammad Mudassir Nazir, Shafaqat Ali, Younan Ouyang, Shuzhen Ye, and Fanrong Zeng. 2020. "Calcium Plays a Double-Edged Role in Modulating Cadmium Uptake and Translocation in Rice" International Journal of Molecular Sciences 21, no. 21: 8058. https://doi.org/10.3390/ijms21218058