Mammalian Homologue NME3 of DYNAMO1 Regulates Peroxisome Division
Abstract
:1. Introduction
2. Results
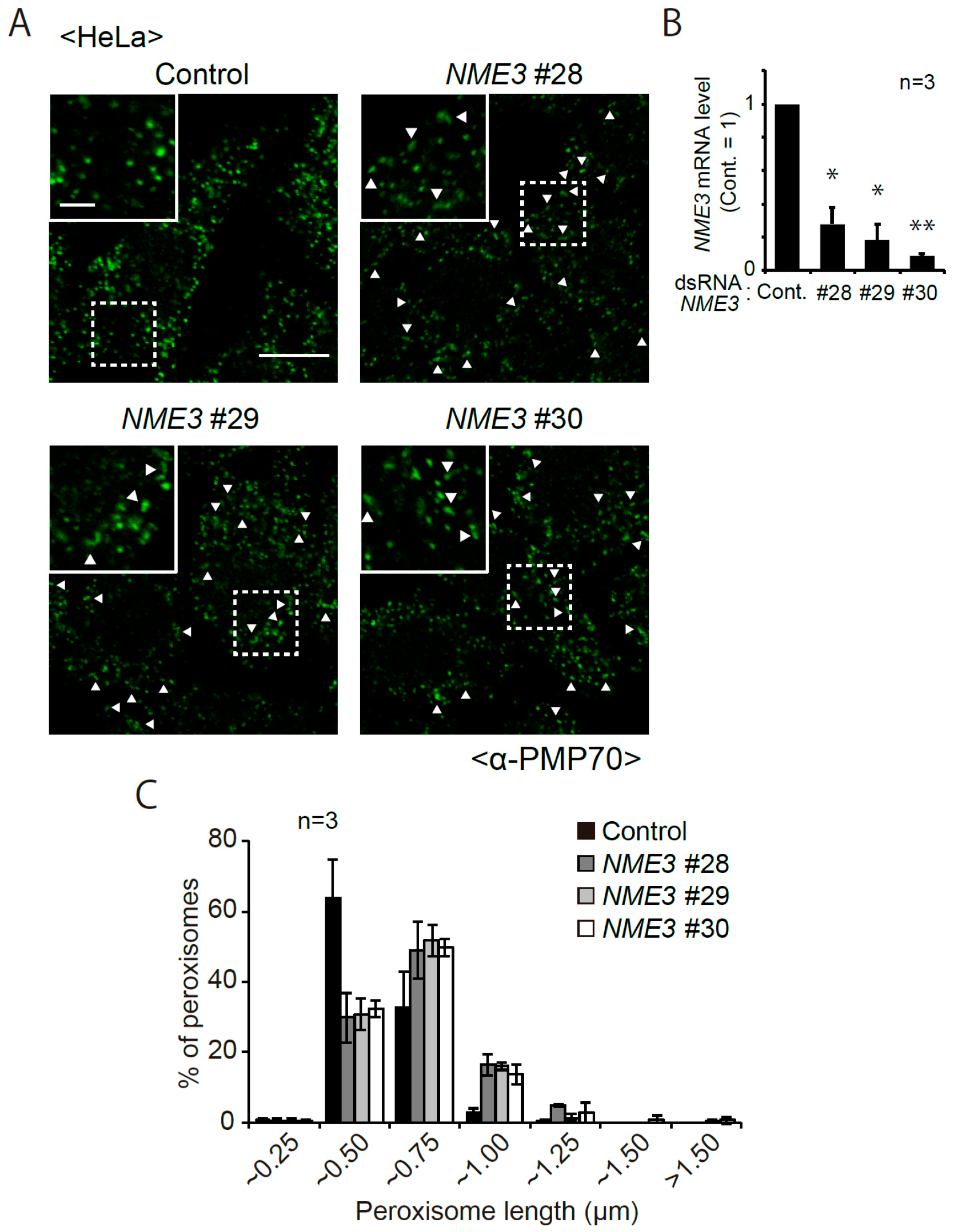
2.1. Morphological Changes of Peroxisomes in Cells Reduced Expression of NME3
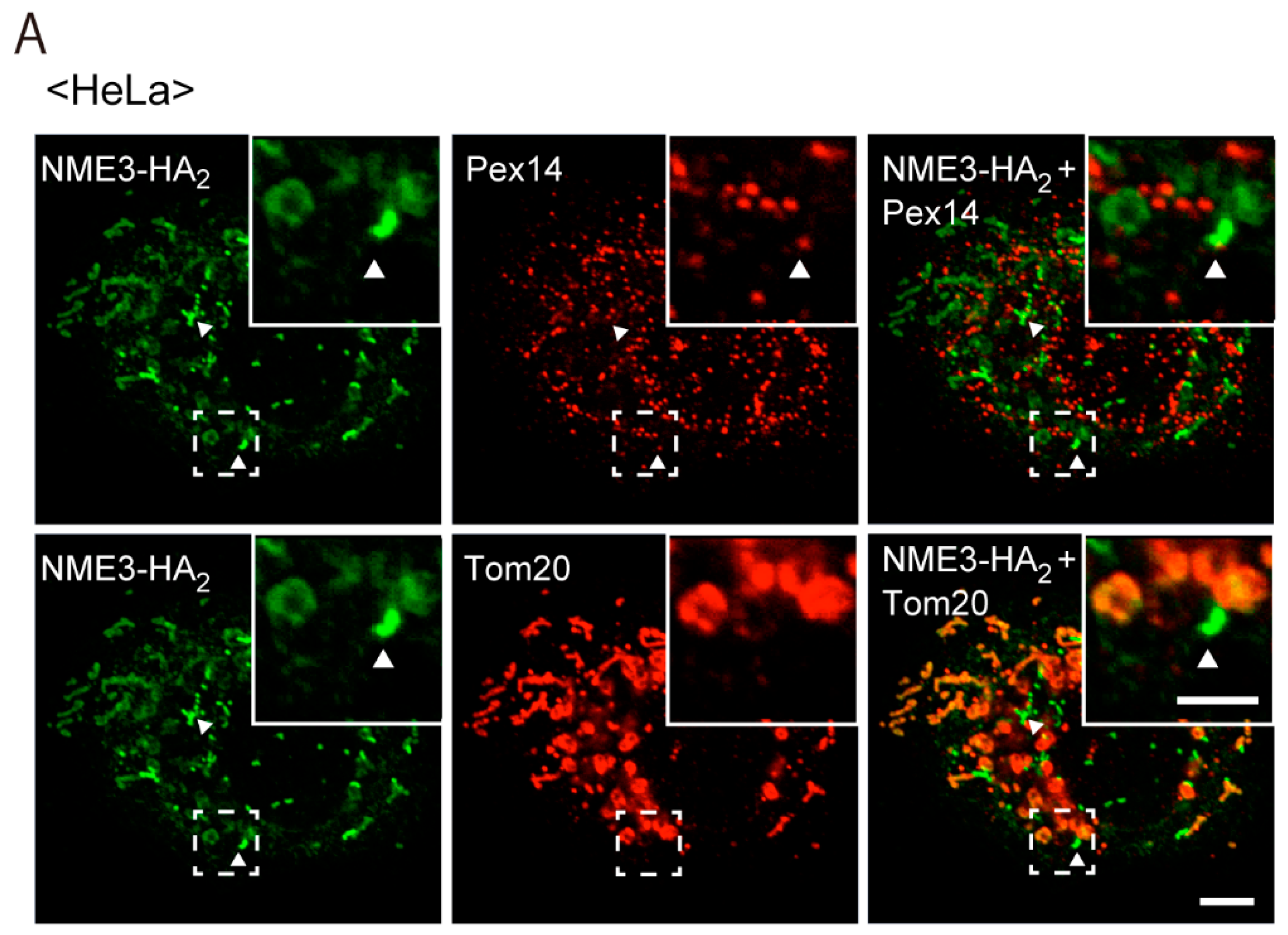
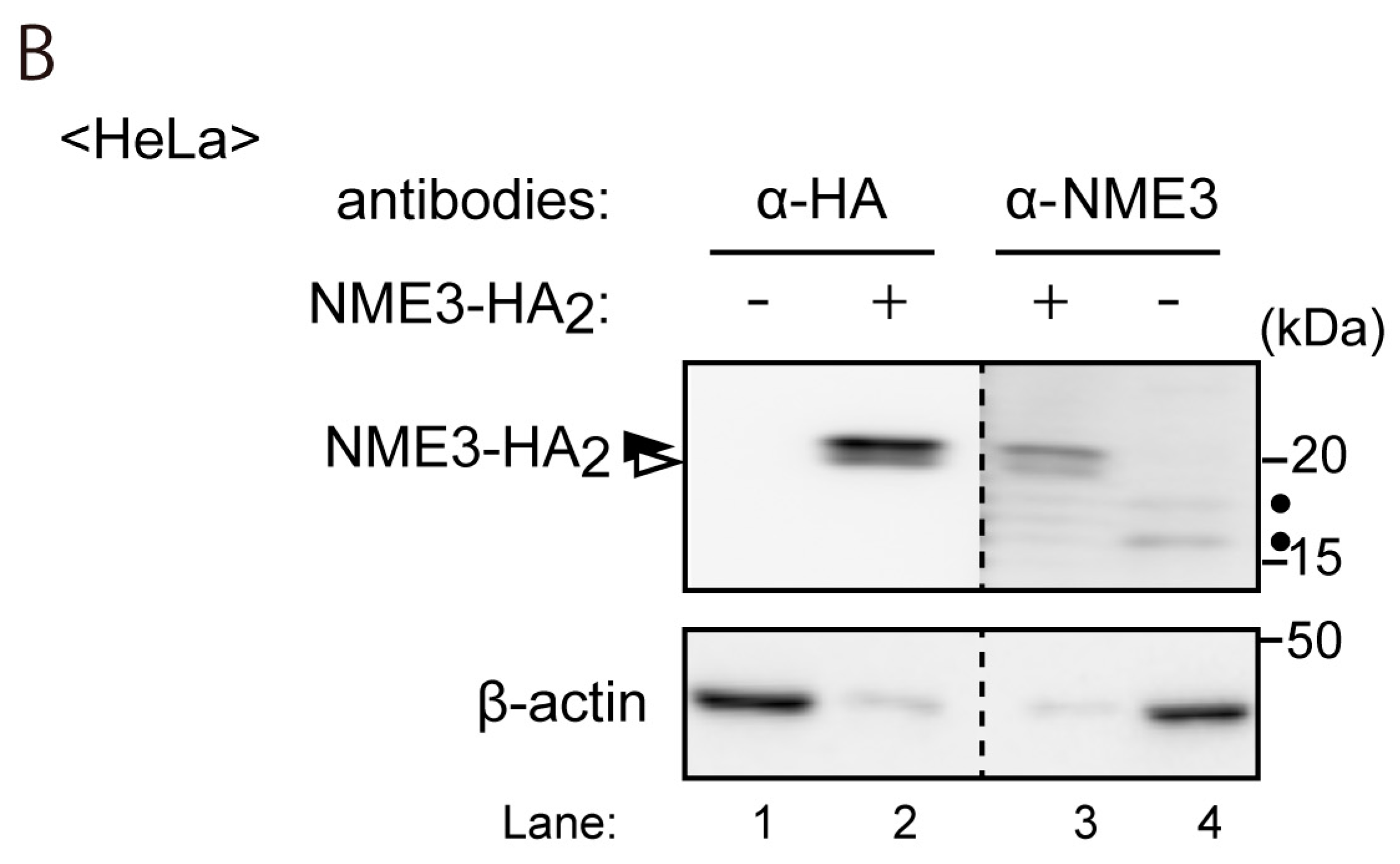
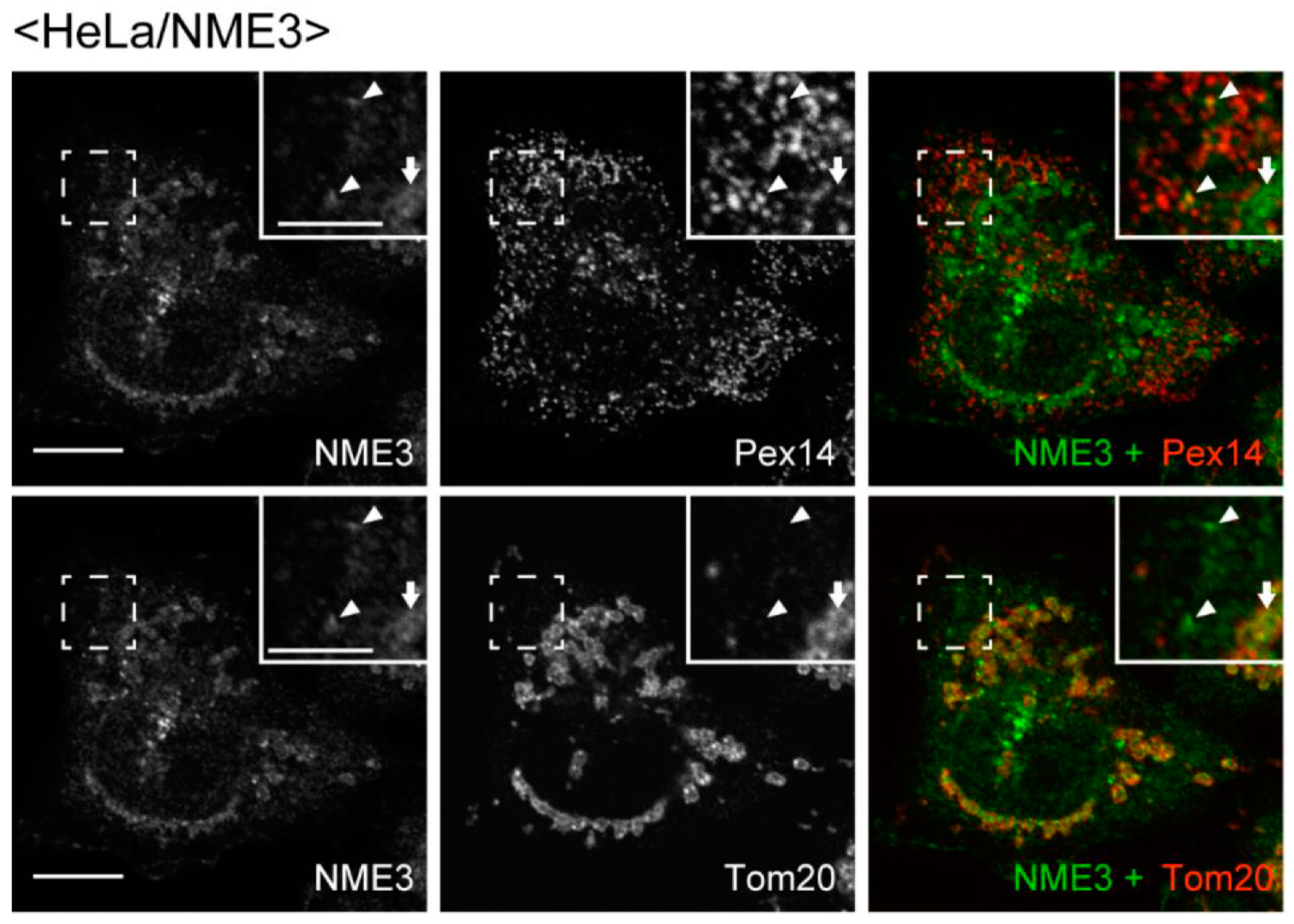
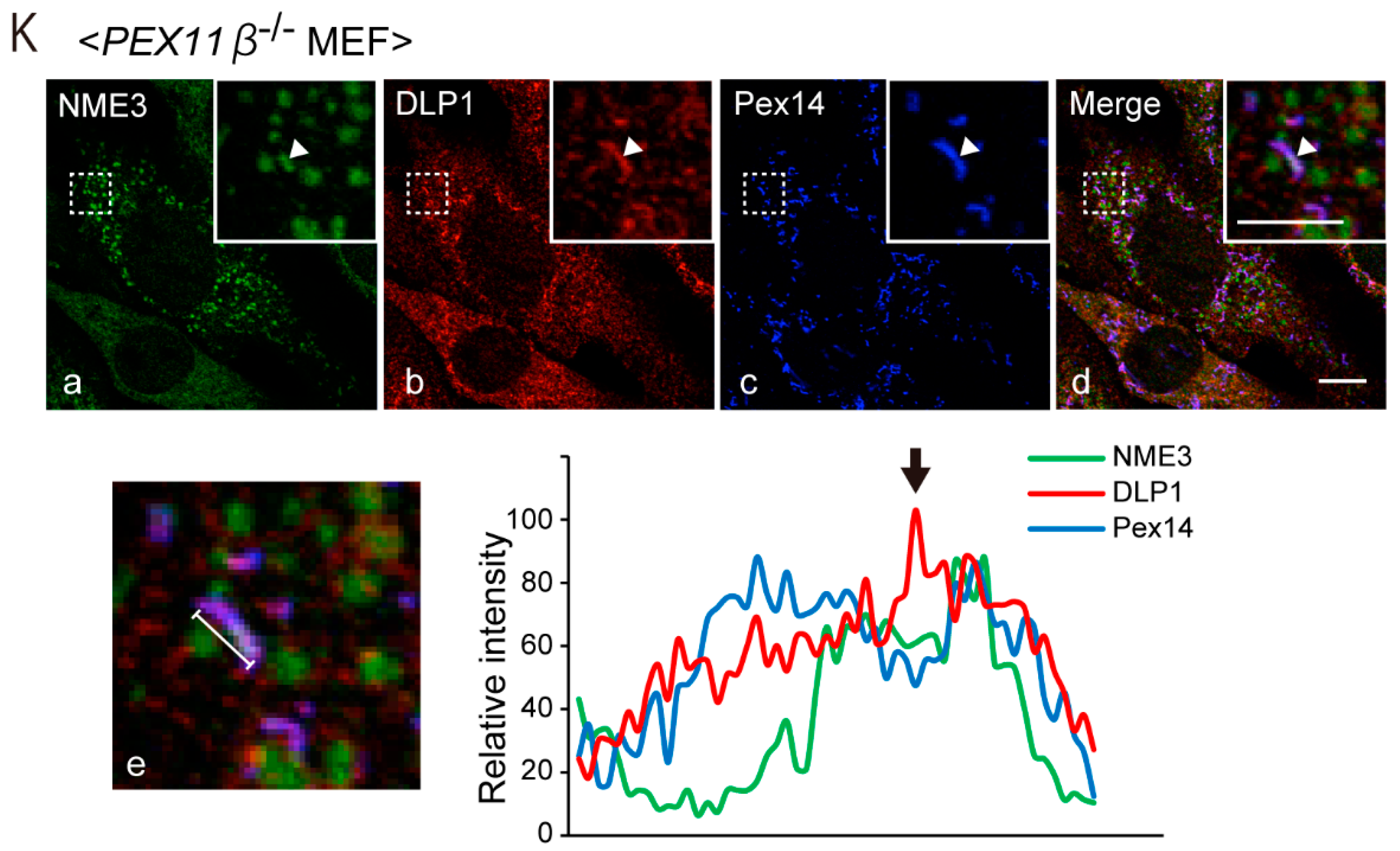
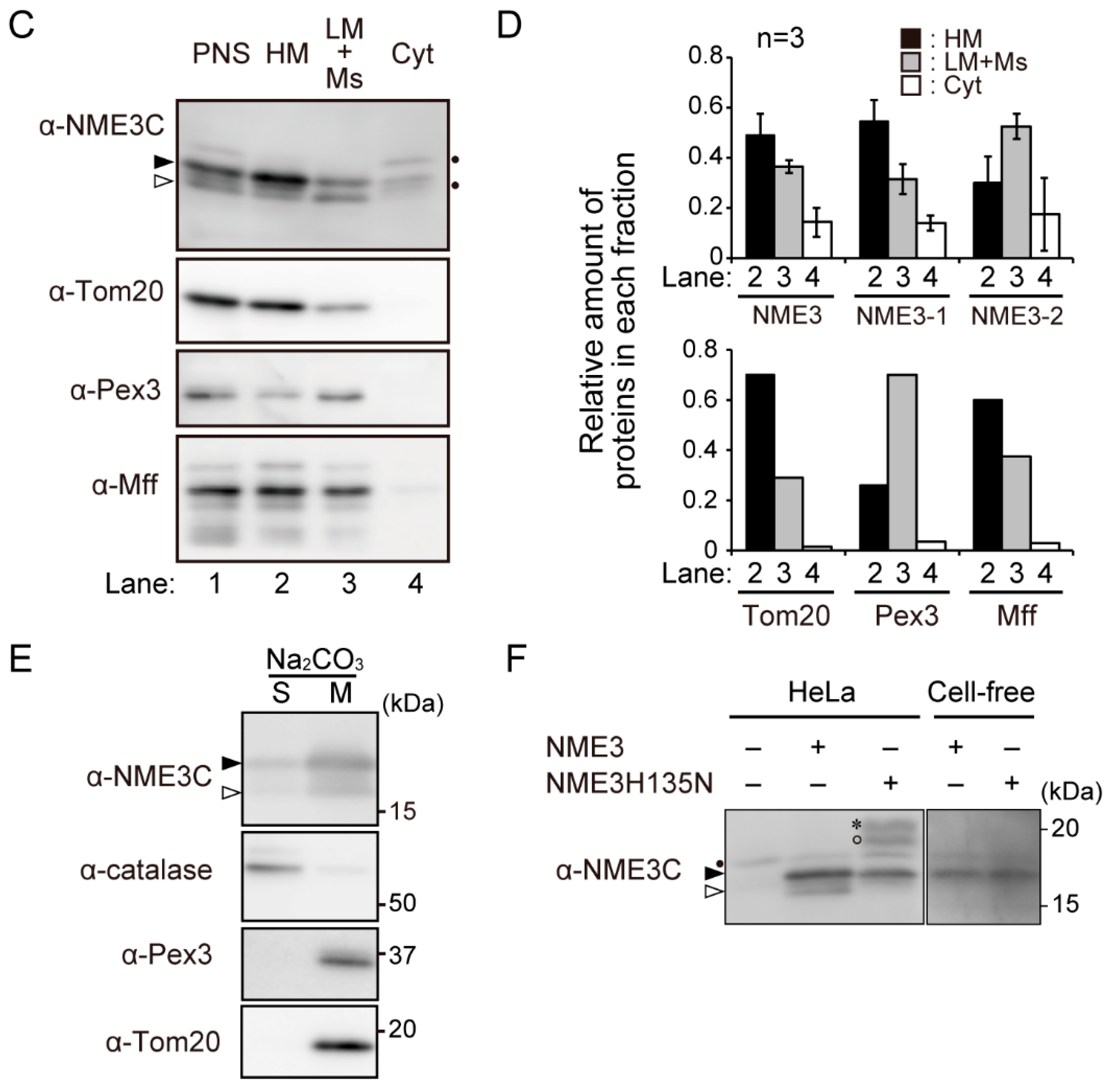
2.2. Intracellular Localization of NME3
2.3. NME3 Is Elevated by Knockdown of ATAD1
2.4. Peroxisomes Are Increased in Number upon Knocking down ATAD1
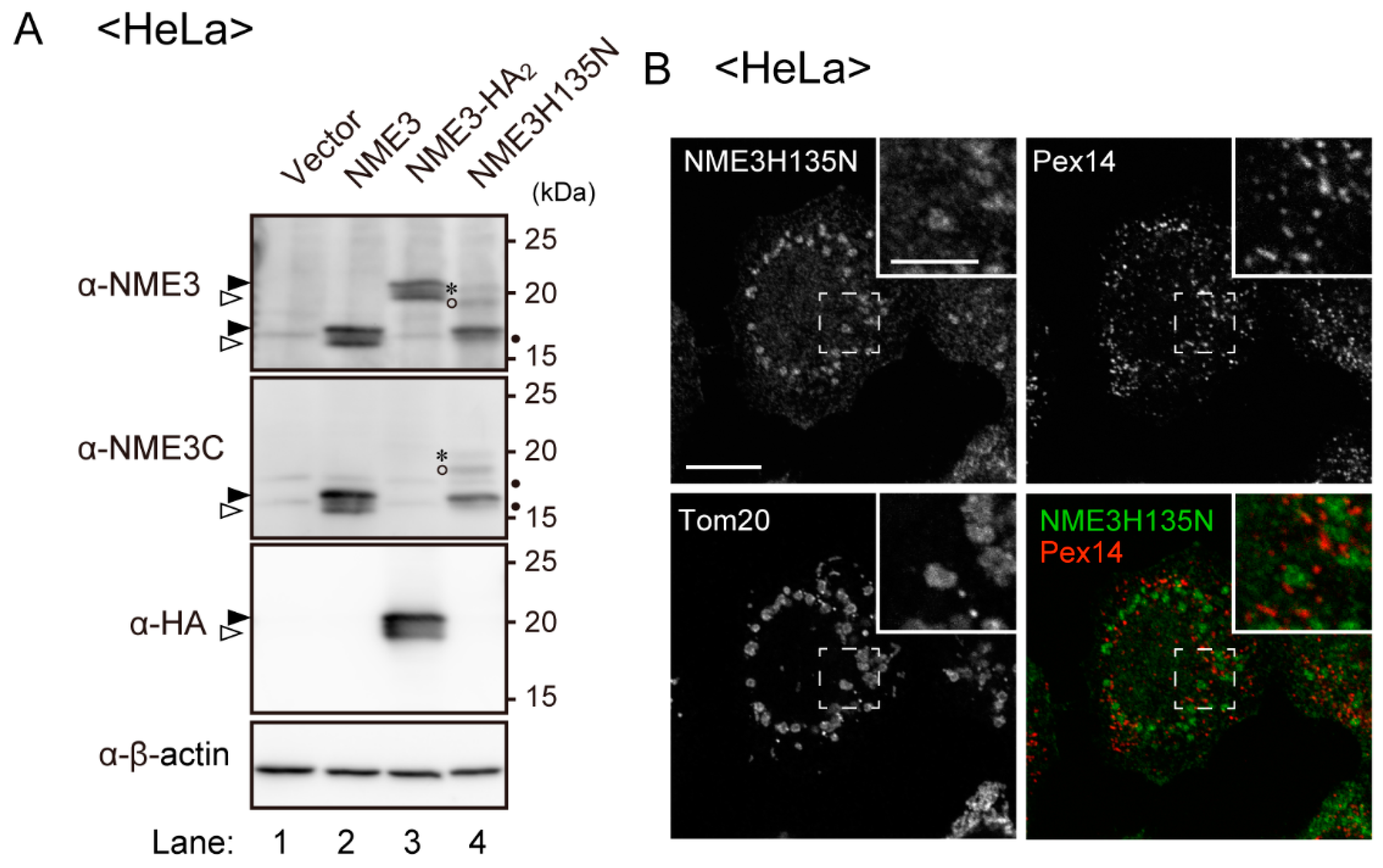
2.5. Peroxisomes Are Elongated by Expression of Catalytically Inactive NME3
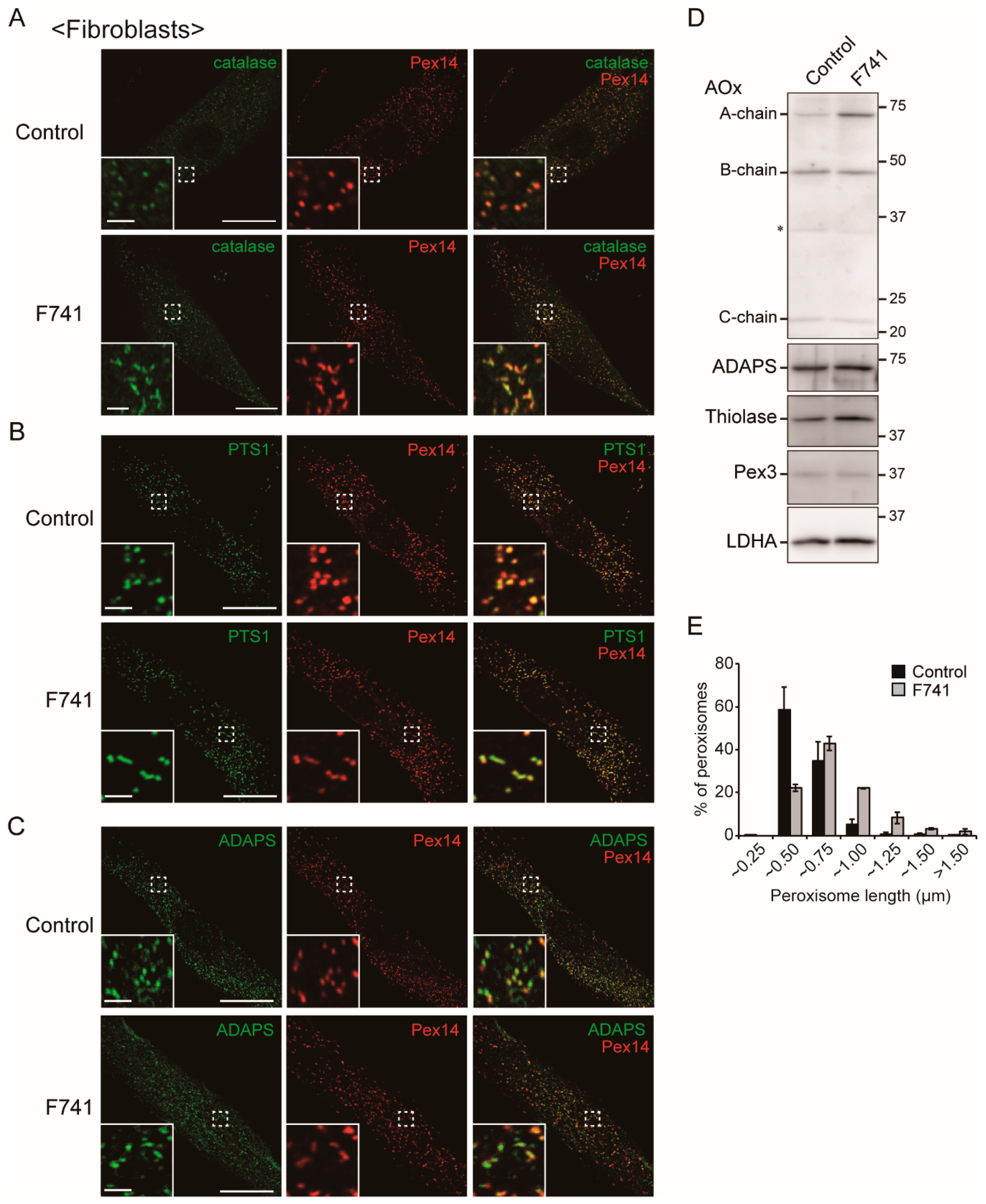
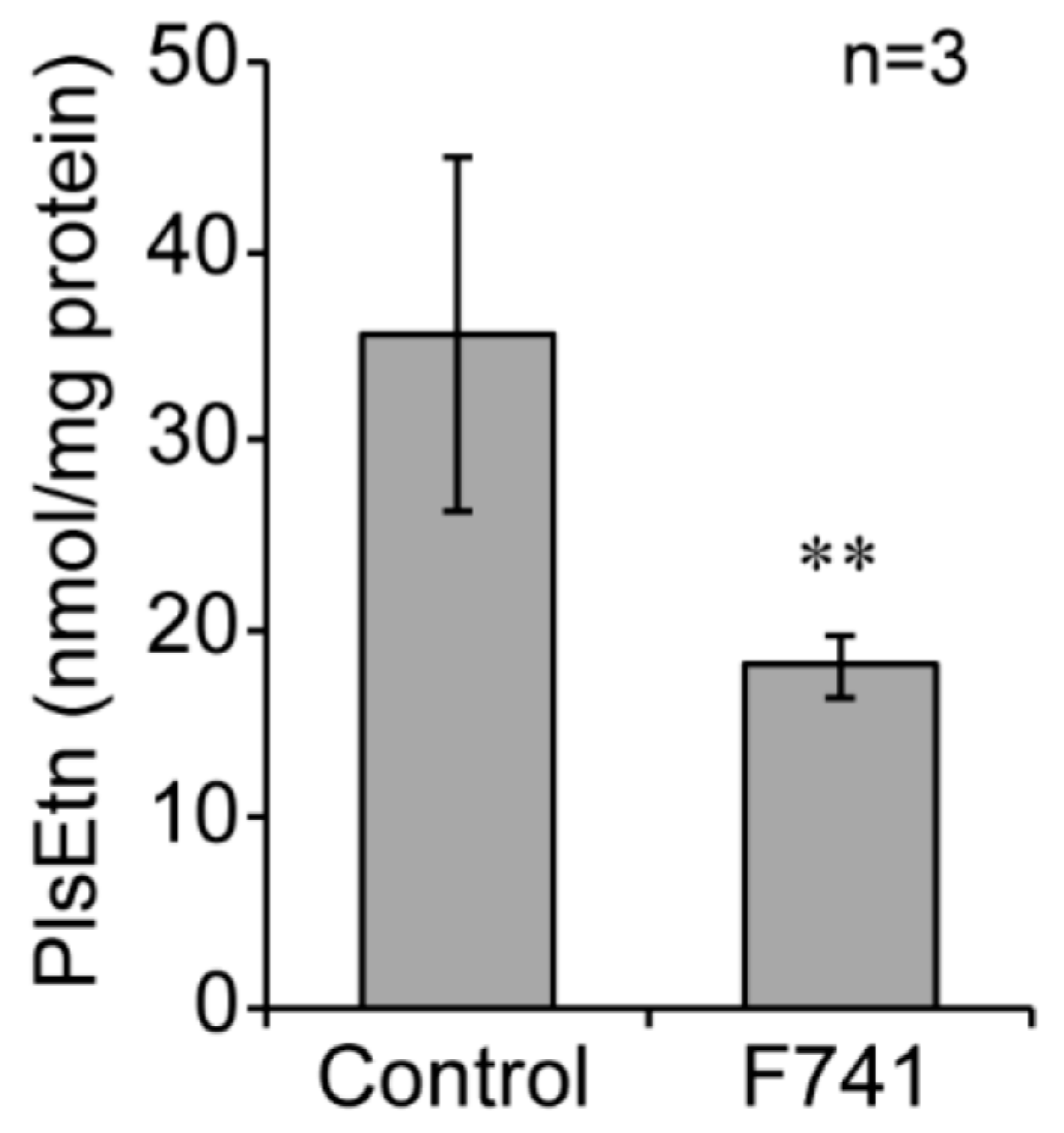
2.6. Decreased Level of Plasmalogens in F741 Patient Fibroblasts
3. Discussion
4. Materials and Methods
4.1. Cell Culture, DNA Transfection, and RNAi
4.2. Antibodies
4.3. RT-PCR
4.4. Plasmids
4.5. Immunoblotting
4.6. Immunofluorescence Microscopy
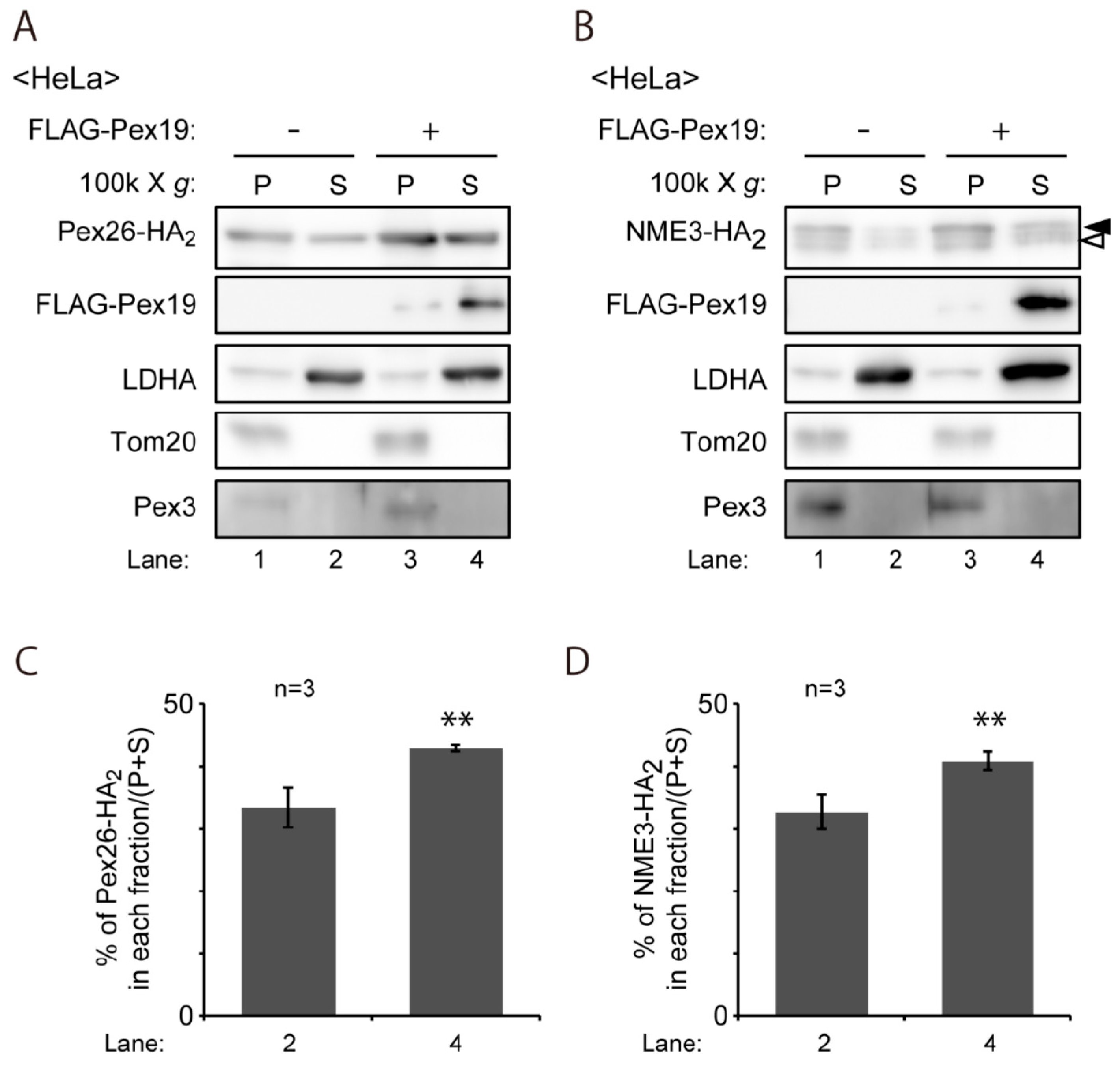
4.7. Subcellular Fractionation and Biochemical Analysis
4.8. Lipid Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Statistical Analysis and Data Presentation
References
- Lazarow, P.B.; Fujiki, Y. Biogenesis of peroxisomes. Annu. Rev. Cell Biol. 1985, 1, 489–530. [Google Scholar] [CrossRef] [PubMed]
- Schrader, M.; Costello, J.L.; Godinho, L.F.; Azadi, A.S.; Islinger, M. Proliferation and fission of peroxisomes—An update. Biochim. Biophys. Acta-Mol. Cell Res. 2016, 1863, 971–983. [Google Scholar] [CrossRef] [PubMed]
- Honsho, M.; Yamashita, S.; Fujiki, Y. Peroxisome homeostasis: Mechanisms of division and selective degradation of peroxisomes in mammals. Biochim. Biophys. Acta-Mol. Cell Res. 2016, 1863, 984–991. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Gould, S.J. The dynamin-like GTPase DLP1 is essential for peroxisome division and is recruited to peroxisomes in part by PEX11. J. Biol. Chem. 2003, 278, 17012–17020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanaka, A.; Kobayashi, S.; Fujiki, Y. Peroxisome division is impaired in a CHO cell mutant with an inactivating point-mutation in dynamin-like protein 1 gene. Exp. Cell Res. 2006, 312, 1671–1684. [Google Scholar] [CrossRef] [PubMed]
- Gandre-Babbe, S.; van der Bliek, A.M. The novel tail-anchored membrane protein Mff controls mitochondrial and peroxisomal fission in mammalian cells. Mol. Biol. Cell 2008, 19, 2402–2412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Itoyama, A.; Honsho, M.; Abe, Y.; Moser, A.; Yoshida, Y.; Fujiki, Y. Docosahexaenoic acid mediates peroxisomal elongation, a prerequisite for peroxisome division. J. Cell Sci. 2012, 125, 589–602. [Google Scholar] [CrossRef] [Green Version]
- Koch, A.; Yoon, Y.; Bonekamp, N.A.; McNiven, M.A.; Schrader, M. A role for Fis1 in both mitochondrial and peroxisomal fission in mammalian cells. Mol. Biol. Cell 2005, 16, 5077–5086. [Google Scholar] [CrossRef]
- Kobayashi, S.; Tanaka, A.; Fujiki, Y. Fis1, DLP1, and Pex11p coordinately regulate peroxisome morphogenesis. Exp. Cell Res. 2007, 313, 1675–1686. [Google Scholar] [CrossRef]
- Fujiki, Y.; Abe, Y.; Imoto, Y.; Tanaka, A.J.; Okumoto, K.; Honsho, M.; Tamura, S.; Miyata, N.; Yamashita, T.; Chung, W.K.; et al. Recent insights into peroxisome biogenesis and associated diseases. J. Cell Sci. 2020, 133, jcs236943. [Google Scholar] [CrossRef]
- Itoyama, A.; Michiyuki, S.; Honsho, M.; Yamamoto, T.; Moser, A.; Yoshida, Y.; Fujiki, Y. Mff functions with Pex11pβ and DLP1 in peroxisomal fission. Biol. Open 2013, 2, 998–1006. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Baumgart, E.; Morrell, J.C.; Jimenez-Sanchez, G.; Valle, D.; Gould, S.J. PEX11β deficiency is lethal and impairs neuronal migration but does not abrogate peroxisome function. Mol. Cell Biol. 2002, 22, 4358–4365. [Google Scholar] [CrossRef] [Green Version]
- Ebberink, M.S.; Koster, J.; Visser, G.; van Spronsen, F.; Stolte-Dijkstra, I.; Smit, G.P.A.; Fock, J.M.; Kemp, S.; Wanders, R.J.A.; Waterham, H.R. A novel defect of peroxisome division due to a homozygous non-sense mutation in the PEX11β gene. J. Med. Genet. 2012, 49, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Thoms, S.; Gaertner, J. First PEX11β patient extends spectrum of peroxisomal biogenesis disorder phenotypes. J. Med. Genet. 2012, 49, 314–316. [Google Scholar] [CrossRef]
- Opaliński, Ł.; Kiel, J.A.; Williams, C.; Veenhuis, M.; van der Klei, I.J. Membrane curvature during peroxisome fission requires Pex11. EMBO J. 2011, 30, 5–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoshida, Y.; Niwa, H.; Honsho, M.; Itoyama, A.; Fujiki, Y. Pex11p mediates peroxisomal proliferation by promoting deformation of the lipid membrane. Biol. Open 2015, 4, 710–721. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, J.; Thomas, A.S.; Grabietz, T.; Landgraf, C.; Volkmer, R.; Marrink, S.; Williams, C.; Melo, M.N. The N-terminal amphipathic helix of Pex11p self-interacts to induce membrane remodelling during peroxisome fission. Biochim. Biophys. Acta-Biomembr. 2018, 1860, 1292–1300. [Google Scholar] [CrossRef] [PubMed]
- Sesaki, H.; Jensen, R.E. Division versus fusion: Dnm1p and Fzo1p antagonistically regulate mitochondrial shape. J. Cell Biol. 1999, 147, 699–706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koch, A.; Thiemann, M.; Grabenbauer, M.; Yoon, Y.; McNiven, M.A.; Schrader, M. Dynamin-like protein 1 is involved in peroxisomal fission. J. Biol. Chem. 2003, 278, 8597–8605. [Google Scholar] [CrossRef] [Green Version]
- Bleazard, W.; McCaffery, J.M.; King, E.J.; Bale, S.; Mozdy, A.; Tieu, Q.; Nunnari, J.; Shaw, J.M. The dynamin-related GTPase Dnm1 regulates mitochondrial fission in yeast. Nat. Cell Biol. 1999, 1, 298–304. [Google Scholar] [CrossRef]
- Waterham, H.R.; Koster, J.; van Roermund, C.W.T.; Mooyer, P.A.W.; Wanders, R.J.A.; Leonard, J.V. A lethal defect of mitochondrial and peroxisomal fission. N. Eng. J. Med. 2007, 356, 1736–1741. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, N.; Nomura, M.; Jofuku, A.; Kato, H.; Suzuki, S.O.; Masuda, K.; Otera, H.; Nakanishi, Y.; Nonaka, I.; Goto, Y.; et al. Mitochondrial fission factor Drp1 is essential for embryonic development and synapse formation in mice. Nat. Cell Biol. 2009, 11, 958–966. [Google Scholar] [CrossRef] [PubMed]
- Islinger, M.; Voelkl, A.; Fahimi, H.D.; Schrader, M. The peroxisome: An update on mysteries 2.0. Histochem. Cell Biol. 2018, 150, 443–471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williams, C.; Opalinski, L.; Landgraf, C.; Costello, J.; Schrader, M.; Krikken, A.M.; Knoops, K.; Kram, A.M.; Volkmer, R.; van der Klei, I.J. The membrane remodeling protein Pex11p activates the GTPase Dnm1p during peroxisomal fission. Proc. Natl. Acad. Sci. USA 2015, 112, 6377–6382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ford, M.G.; Chappie, J.S. The structural biology of the dynamin-related proteins: New insights into a diverse, multitalented family. Traffic 2019, 20, 717–740. [Google Scholar] [CrossRef] [PubMed]
- Imoto, Y.; Abe, Y.; Okumoto, K.; Honsho, M.; Kuroiwa, H.; Kuroiwa, T.; Fujiki, Y. Defining the dynamin-based ring organizing center on the peroxisome-dividing machinery isolated from Cyanidioschyzon merolae. J. Cell Sci. 2017, 130, 853–867. [Google Scholar] [CrossRef] [Green Version]
- Imoto, Y.; Abe, Y.; Honsho, M.; Okumoto, K.; Ohnuma, M.; Kuroiwa, H.; Kuroiwa, T.; Fujiki, Y. Onsite GTP fuelling via DYNAMO1 drives division of mitochondria and peroxisomes. Nat. Commun. 2018, 9, 4634. [Google Scholar] [CrossRef]
- Boissan, M.; Montagnac, G.; Shen, Q.; Griparic, L.; Guitton, J.; Romao, M.; Sauvonnet, N.; Lagache, T.; Lascu, I.; Raposo, G.; et al. Nucleoside diphosphate kinases fuel dynamin superfamily proteins with GTP for membrane remodeling. Science 2014, 344, 1510–1515. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.-W.; Wang, H.-L.; Huang, C.-W.; Huang, C.-Y.; Lim, W.K.; Tu, I.-C.; Koorapati, A.; Hsieh, S.-T.; Kan, S.-W.; Tzeng, S.-R.; et al. Two separate functions of NME3 critical for cell survival underlie a neurodegenerative disorder. Proc. Natl. Acad. Sci. USA 2019, 116, 566–574. [Google Scholar] [CrossRef] [Green Version]
- Satoh, M.; Hamamoto, T.; Seo, N.; Kagawa, Y.; Endo, H. Differential sublocalization of the dynamin-related protein OPA1 isoforms in mitochondria. Biochem. Biophys. Res. Commun. 2003, 300, 482–493. [Google Scholar] [CrossRef]
- Griparic, L.; van der Wel, N.N.; Orozco, I.J.; Peters, P.J.; van der Bliek, A.M. Loss of the intermembrane space protein Mgm1/OPA1 induces swelling and localized constrictions along the lengths of mitochondria. J. Biol. Chem. 2004, 279, 18792–18798. [Google Scholar] [CrossRef] [Green Version]
- Ishihara, N.; Fujita, Y.; Oka, T.; Mihara, K. Regulation of mitochondrial morphology through proteolytic cleavage of OPA1. EMBO J. 2006, 25, 2966–2977. [Google Scholar] [CrossRef]
- Osumi, T.; Tsukamoto, T.; Hata, S.; Yokota, S.; Miura, S.; Fujiki, Y.; Hijikata, M.; Miyazawa, S.; Hashimoto, T. Amino-terminal presequence of the precursor of peroxisomal 3-ketoacyl-CoA thiolase is a cleavable signal peptide for peroxisomal targeting. Biochem. Biophys. Res. Commun. 1991, 181, 947–954. [Google Scholar] [CrossRef]
- Kurochkin, I.V.; Mizuno, Y.; Konagaya, A.; Sakaki, Y.; Schönbach, C.; Okazaki, Y. Novel peroxisomal protease Tysnd1 processes PTS1- and PTS2-containing enzymes involved in β-oxidation of fatty acids. EMBO J. 2007, 26, 835–845. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, Y.; Ninomiya, Y.; Nakachi, Y.; Iseki, M.; Iwasa, H.; Akita, M.; Tsukui, T.; Shimozawa, N.; Ito, C.; Toshimori, K.; et al. Tysnd1 deficiency in mice interferes with the peroxisomal localization of PTS2 enzymes, causing lipid metabolic abnormalities and male infertility. PLoS Genet. 2013, 9, e1003286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okumoto, K.; Kametani, Y.; Fujiki, Y. Two proteases, trypsin domain-containing 1 (Tysnd1) and peroxisomaal Lon protease (PsLon), cooperatively regulate fatty-acid β-oxidation in peroxisomal matrix. J. Biol. Chem. 2011, 286, 44367–44379. [Google Scholar] [CrossRef] [Green Version]
- Koch, J.; Feichtinger, R.G.; Freisinger, P.; Pies, M.; Schrödl, F.; Iuso, A.; Sperl, W.; Mayr, J.A.; Prokisch, H.; Haack, T.B. Disturbed mitochondrial and peroxisomal dynamics due to loss of MFF causes Leigh-like encephalopathy, optic atrophy and peripheral neuropathy. J. Med. Genet. 2016, 53, 270–278. [Google Scholar] [CrossRef]
- Nasca, A.; Nardecchia, F.; Commone, A.; Semeraro, M.; Legati, A.; Garavaglia, B.; Ghezzi, D.; Leuzzi, V. Clinical and biochemical features in a patient with Mitochondrial Fission Factor gene alteration. Front. Genet. 2018, 9, 625. [Google Scholar] [CrossRef] [Green Version]
- Boissan, M.; Dabernat, S.; Peuchant, E.; Schlattner, U.; Lascu, I.; Lacombe, M.-L. The mammalian Nm23/NDPK family: From metastasis control to cilia movement. Mol. Cell Biochem. 2009, 329, 51–62. [Google Scholar] [CrossRef]
- Fujiki, Y.; Matsuzono, Y.; Matsuzaki, T.; Fransen, M. Import of peroxisomal membrane proteins: The interplay of Pex3p- and Pex19p-mediated interactions. Biochim. Biophys. Acta-Mol. Cell Res. 2006, 1763, 1639–1646. [Google Scholar] [CrossRef] [Green Version]
- Yagita, Y.; Hiromasa, T.; Fujiki, Y. Tail-anchored PEX26 targets peroxisomes via a PEX19-dependent and TRC40-independent class I pathway. J. Cell Biol. 2013, 200, 651–666. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Yagita, Y.; Fujiki, Y. Assembly of peroxisomal membrane proteins via the direct Pex19p-Pex3p pathway. Traffic 2016, 17, 433–455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.-C.; Umanah, G.K.E.; Dephoure, N.; Andrabi, S.A.; Gygi, S.P.; Dawson, T.M.; Dawson, V.L.; Rutter, J. Msp1/ATAD1 maintains mitochondrial function by facilitating the degradation of mislocalized tail-anchored proteins. EMBO J. 2014, 33, 1548–1564. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Gould, S.J. PEX11 promotes peroxisome division independently of peroxisome metabolism. J. Cell Biol. 2002, 156, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Ghaedi, K.; Tamura, S.; Okumoto, K.; Matsuzono, Y.; Fujiki, Y. The peroxin Pex3p initiates membrane assembly in peroxisome biogenesis. Mol. Biol. Cell 2000, 11, 2085–2102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagan, N.; Zoeller, R.A. Plasmalogens: Biosynthesis and functions. Prog. Lipid Res. 2001, 40, 199–229. [Google Scholar] [CrossRef]
- Weller, S.; Gould, S.J.; Valle, D. Peroxisome biogenesis disorders. Annu. Rev. Genom. Hum. Genet. 2003, 4, 165–211. [Google Scholar] [CrossRef]
- Wanders, R.J.A.; Waterham, H.R. Peroxisomal disorders: The single peroxisomal enzyme deficiencies. Biochim. Biophys. Acta 2006, 1763, 1707–1720. [Google Scholar] [CrossRef] [Green Version]
- Koch, J.; Pranjic, K.; Huber, A.; Ellinger, A.; Hartig, A.; Kragler, F.; Brocard, C. PEX11 family members are membrane elongation factors that coordinate peroxisome proliferation and maintenance. J. Cell Sci. 2010, 123, 3389–3400. [Google Scholar] [CrossRef] [Green Version]
- Ferguson, S.M.; Raimondi, A.; Paradise, S.; Shen, H.; Mesaki, K.; Ferguson, A.; Destaing, O.; Ko, G.; Takasaki, J.; Cremona, O.; et al. Coordinated actions of actin and BAR proteins upstream of dynamin at endocytic clathrin-coated pits. Dev. Cell 2009, 17, 811–822. [Google Scholar] [CrossRef] [Green Version]
- Park, R.J.; Shen, H.; Liu, L.; Liu, X.; Ferguson, S.M. Dynamin triple knockout cells reveal off target effects of commonly used dynamin inhibitors. J. Cell Sci. 2013, 126, 5305–5312. [Google Scholar] [CrossRef] [Green Version]
- Wohlever, M.L.; Mateja, A.; McGilvray, P.T.; Day, K.J.; Keenan, R.J. Msp1 is a membrane protein dislocase for tail-anchored proteins. Mol. Cell 2017, 67, 194–202.e6. [Google Scholar] [PubMed] [Green Version]
- Matsumoto, S.; Nakatsukasa, K.; Kakuta, C.; Tamura, Y.; Esaki, M.; Endo, T. Msp1 clears mistargeted proteins by facilitating their transfer from mitochondria to the ER. Mol. Cell 2019, 76, 191–205.e10. [Google Scholar]
- Abe, Y.; Honsho, M.; Itoh, R.; Kawaguchi, R.; Fujitani, M.; Fujiwara, K.; Hirokane, M.; Matsuzaki, T.; Nakayama, K.; Ohgi, R.; et al. Peroxisome biogenesis deficiency attenuates the BDNF-TrkB pathway-mediated development of the cerebellum. Life Sci. Alliance 2018, 1, e201800062. [Google Scholar]
- Honsho, M.; Dorninger, F.; Abe, Y.; Setoyama, D.; Ohgi, R.; Uchiumi, T.; Kang, D.; Berger, J.; Fujiki, Y. Impaired plasmalogen synthesis dysregulates liver X receptor-dependent transcription in cerebellum. J. Biochem. 2019, 166, 353–361. [Google Scholar]
- Tsukamoto, T.; Yokota, S.; Fujiki, Y. Isolation and characterization of Chinese hamster ovary cell mutants defective in assembly of peroxisomes. J. Cell Biol. 1990, 110, 651–660. [Google Scholar] [PubMed]
- Honsho, M.; Yagita, Y.; Kinoshita, N.; Fujiki, Y. Isolation and characterization of mutant animal cell line defective in alkyl-dihydroxyacetonephosphate synthase: Localization and transport of plasmalogens to post-Golgi compartments. Biochim. Biophys. Acta 2008, 1783, 1857–1865. [Google Scholar] [PubMed] [Green Version]
- Otera, H.; Okumoto, K.; Tateishi, K.; Ikoma, Y.; Matsuda, E.; Nishimura, M.; Tsukamoto, T.; Osumi, T.; Ohashi, K.; Higuchi, O.; et al. Peroxisome targeting signal type 1 (PTS1) receptor is involved in import of both PTS1 and PTS2: Studies with PEX5-defective CHO cell mutants. Mol. Cell Biol. 1998, 18, 388–399. [Google Scholar] [PubMed] [Green Version]
- Shimizu, N.; Itoh, R.; Hirono, Y.; Otera, H.; Ghaedi, K.; Tateishi, K.; Tamura, S.; Okumoto, K.; Harano, T.; Mukai, S.; et al. The peroxin Pex14p: cDNA cloning by functional complementation on a Chinese hamster ovary cell mutant, characterization, and functional analysis. J. Biol. Chem. 1999, 274, 12593–12604. [Google Scholar] [PubMed] [Green Version]
- Mukai, S.; Ghaedi, K.; Fujiki, Y. Intracellular localization, function, and dysfunction of the peroxisome-targeting signal type 2 receptor, Pex7p, in mammalian cells. J. Biol. Chem. 2002, 277, 9548–9561. [Google Scholar] [CrossRef] [Green Version]
- Otera, H.; Harano, T.; Honsho, M.; Ghaedi, K.; Mukai, S.; Tanaka, A.; Kawai, A.; Shimizu, N.; Fujiki, Y. The mammalian peroxin Pex5pL, the longer isoform of the mobile peroxisome targeting signal (PTS) type 1 transporter, translocates Pex7p-PTS2 protein complex into peroxisomes via its initial docking site, Pex14p. J. Biol. Chem. 2000, 275, 21703–21714. [Google Scholar] [CrossRef] [Green Version]
- Honsho, M.; Abe, Y.; Fujiki, Y. Plasmalogen biosynthesis is spatiotemporally regulated by sensing plasmalogens in the inner leaflet of plasma membranes. Sci. Rep. 2017, 7, 43936. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Honsho, M.; Asaoku, S.; Fukumoto, K.; Fujiki, Y. Topogenesis and homeostasis of fatty acyl-CoA reductase 1. J. Biol. Chem. 2013, 288, 34588–34598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fujiki, Y.; Hubbard, A.L.; Fowler, S.; Lazarow, P.B. Isolation of intracellular membranes by means of sodium carbonate treatment: Application to endoplasmic reticulum. J. Cell Biol. 1982, 93, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Abe, Y.; Honsho, M.; Nakanishi, H.; Taguchi, R.; Fujiki, Y. Very-long-chain polyunsaturated fatty acids accumulate in phosphatidylcholine of fibroblasts from patients with Zellweger syndrome and acyl-CoA oxidase1 deficiency. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2014, 1841, 610–619. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Honsho, M.; Abe, Y.; Imoto, Y.; Chang, Z.-F.; Mandel, H.; Falik-Zaccai, T.C.; Fujiki, Y. Mammalian Homologue NME3 of DYNAMO1 Regulates Peroxisome Division. Int. J. Mol. Sci. 2020, 21, 8040. https://doi.org/10.3390/ijms21218040
Honsho M, Abe Y, Imoto Y, Chang Z-F, Mandel H, Falik-Zaccai TC, Fujiki Y. Mammalian Homologue NME3 of DYNAMO1 Regulates Peroxisome Division. International Journal of Molecular Sciences. 2020; 21(21):8040. https://doi.org/10.3390/ijms21218040
Chicago/Turabian StyleHonsho, Masanori, Yuichi Abe, Yuuta Imoto, Zee-Fen Chang, Hanna Mandel, Tzipora C. Falik-Zaccai, and Yukio Fujiki. 2020. "Mammalian Homologue NME3 of DYNAMO1 Regulates Peroxisome Division" International Journal of Molecular Sciences 21, no. 21: 8040. https://doi.org/10.3390/ijms21218040
APA StyleHonsho, M., Abe, Y., Imoto, Y., Chang, Z.-F., Mandel, H., Falik-Zaccai, T. C., & Fujiki, Y. (2020). Mammalian Homologue NME3 of DYNAMO1 Regulates Peroxisome Division. International Journal of Molecular Sciences, 21(21), 8040. https://doi.org/10.3390/ijms21218040




