Associations of Arginine with Gestational Diabetes Mellitus in a Follow-Up Study
Abstract
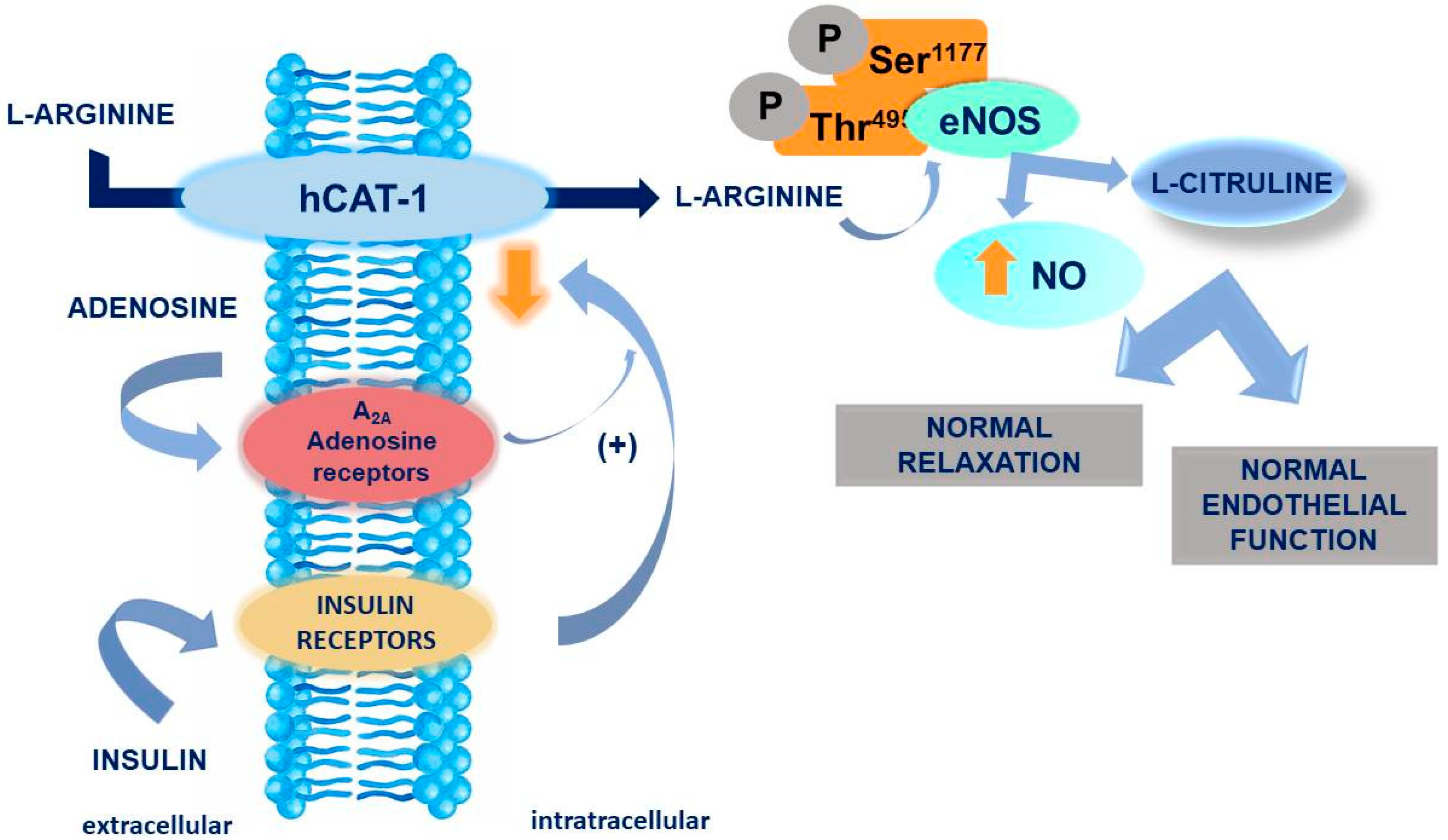
:1. Introduction
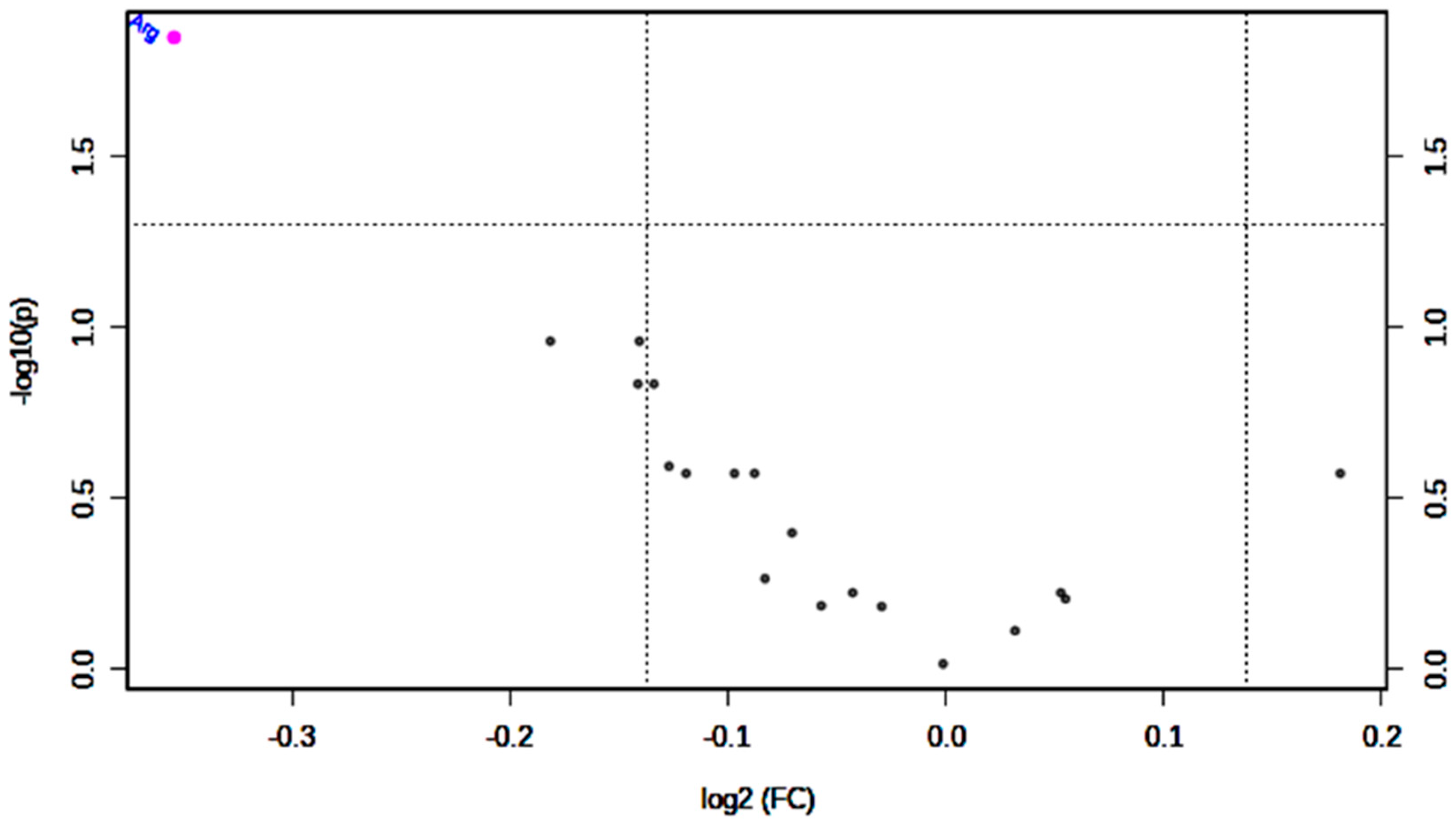
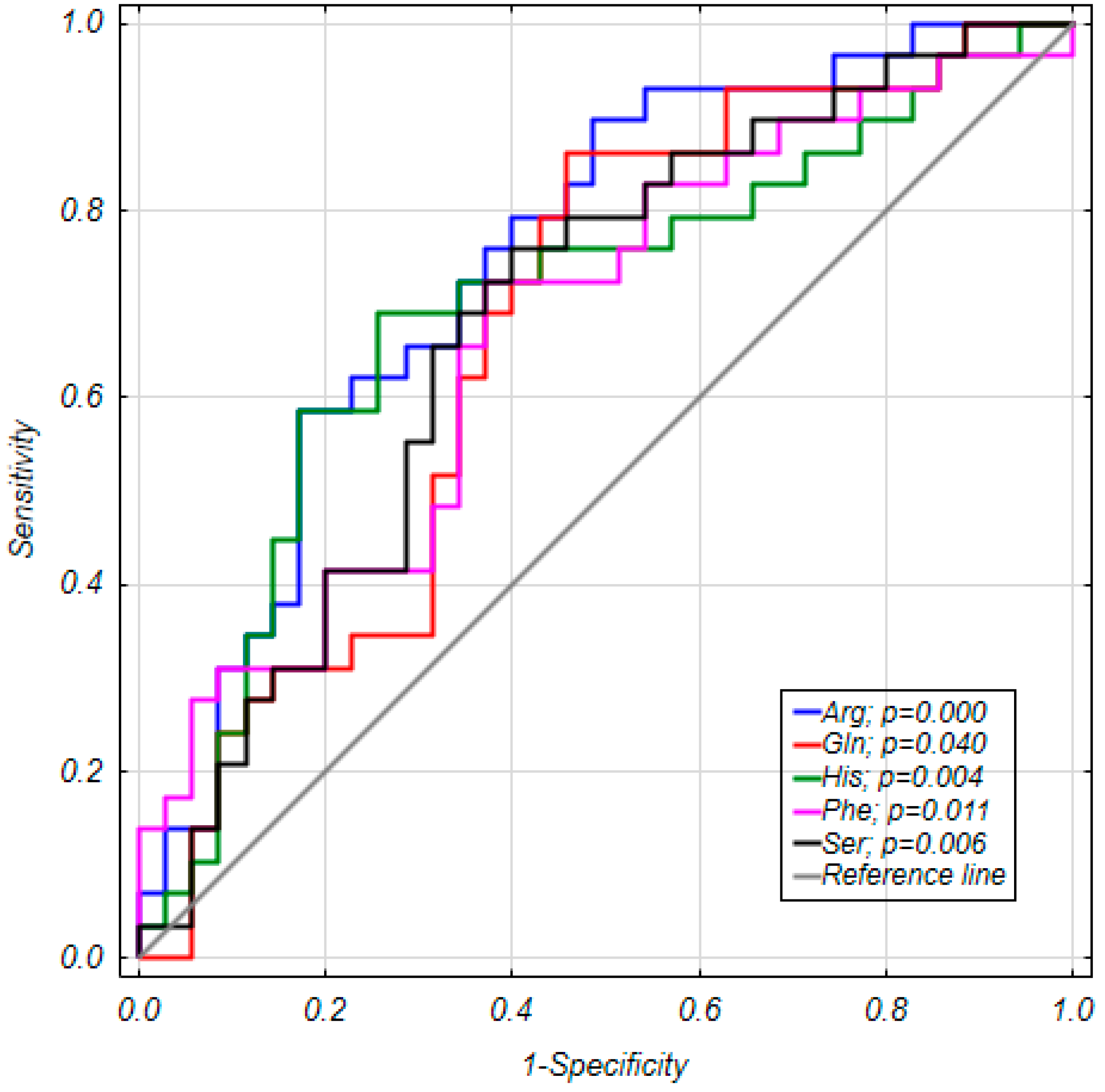
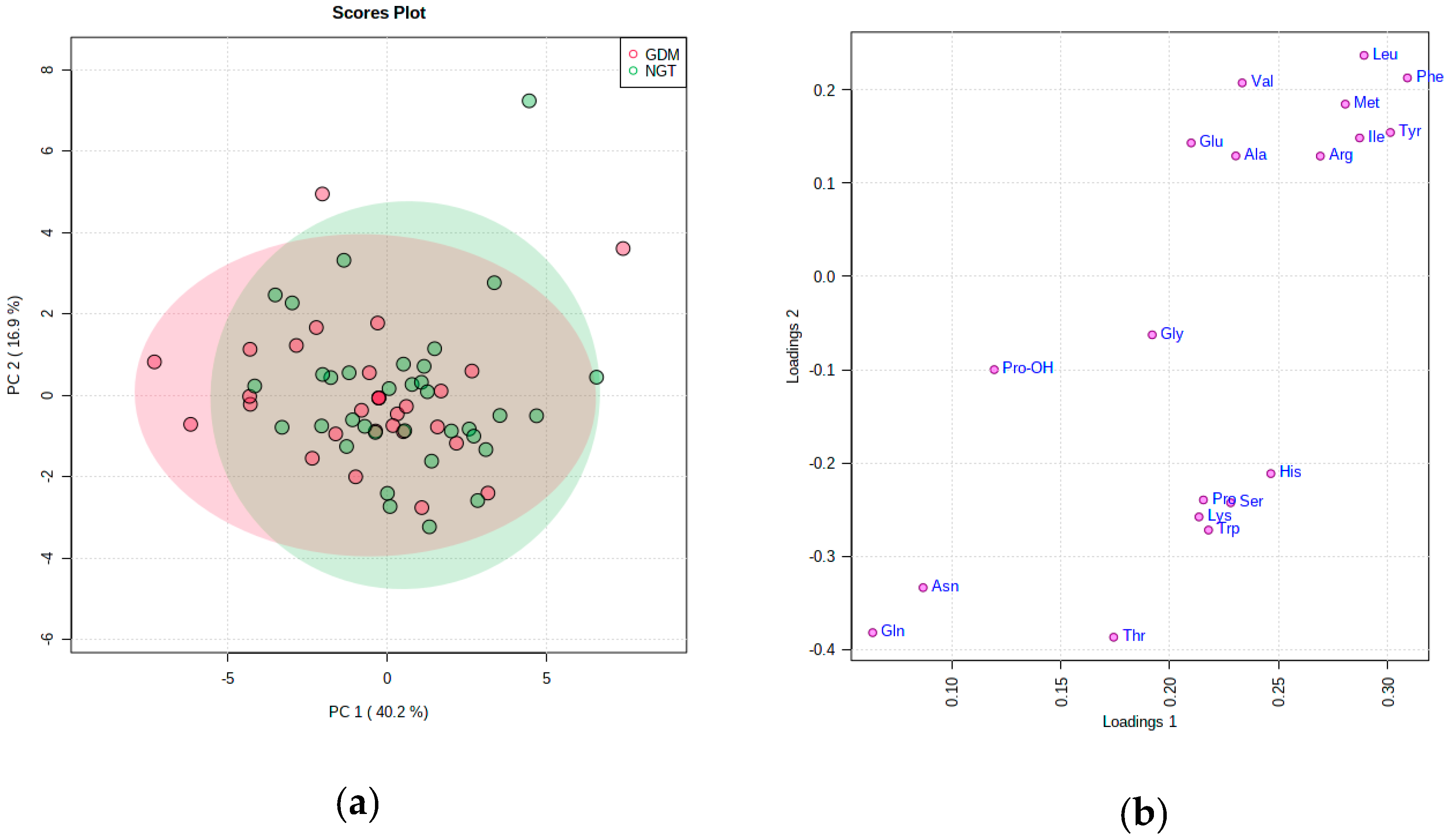
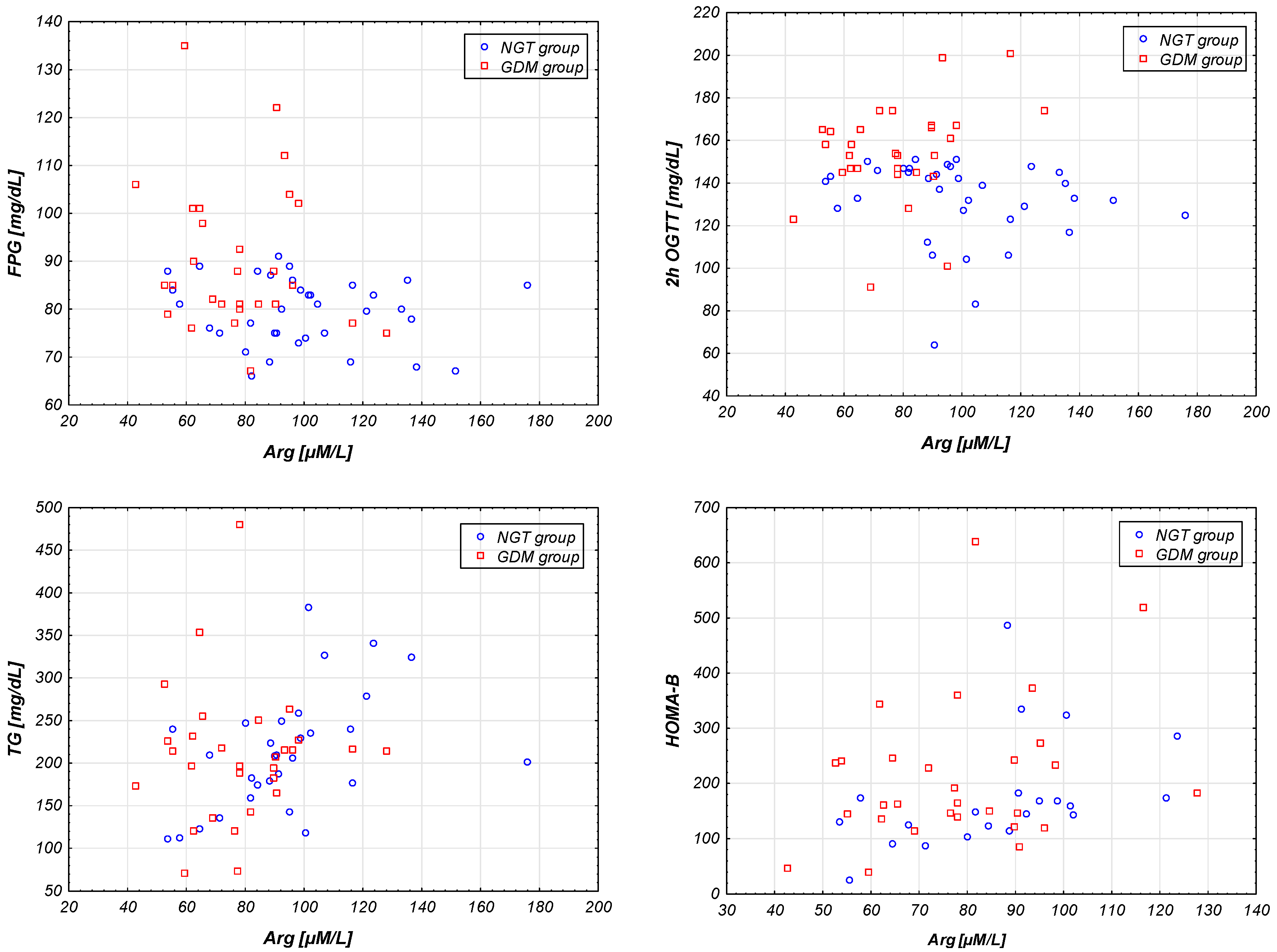
2. Results
PCA Analysis
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Anthropometric Measurements and Biochemical Data
4.3. Chemicals and Reagents
4.4. Sample Preparation
4.5. HPLC–MS Analysis
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AAs | Amino Acids |
| FPG | Fasting Plasma Glucose |
| GDM | Gestational Diabetes Mellitus |
| HCA | Hierarchical Cluster Analysis |
| IGT | Impaired Glucose Tolerance |
| IR | Insulin Resistance |
| IUGR | Intrauterine Growth Restriction |
| NGT | Normal Glucose Tolerance |
| NO | Nitric Oxide |
| OGTT | Oral Glucose Tolerance Test |
| PCA | Principal Component Analysis |
| T2DM | Type 2 Diabetes Mellitus |
References
- American Diabetes Association. (2) Classification and diagnosis of diabetes. Diabetes Care 2015, 3, S8–S16. [Google Scholar] [CrossRef] [Green Version]
- Nezami, N.; Safa, J.; Eftekhar-Sadat, A.T.; Salari, B.; Ghorashi, S.; Sakhaee, K.; Khosraviani, K. Lovastatin raises serum osteoprotegerin level in people with type 2 diabetic nephropathy. Clin. Biochem. 2010, 43, 1294–1299. [Google Scholar] [CrossRef]
- Barbour, L.A.; McCurdy, C.E.; Hernandez, T.L.; Kirwan, J.P.; Catalano, P.M.; Friedman, J.E. Cellular mechanisms for insulin resistance in normal pregnancy and gestational diabetes. Diabetes Care 2007, 30, S112–S119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carr, D.B.; Utzschneider, K.M.; Hull, R.L.; Tong, J.; Wallace, T.M.; Kodama, K.; Shofer, J.B.; Heckbert, S.R.; Boyko, E.J.; Fujimoto, W.Y.; et al. Gestational diabetes mellitus increases the risk of cardiovascular disease in women with a family history of type 2 diabetes. Diabetes Care 2006, 29, 2078–2083. [Google Scholar] [CrossRef] [Green Version]
- Group, H.S.C.R.; Metzger, B.E.; Lowe, L.P.; Dyer, A.R.; Trimble, E.R.; Chaovarindr, U.; Coustan, D.R.; Hadden, D.R.; McCance, D.R.; Hod, M.; et al. Hyperglycemia and adverse pregnancy outcomes. N. Engl. J. Med. 2008, 358, 1991–2002. [Google Scholar] [CrossRef] [Green Version]
- Rodrigo, N.; Glastras, S.J. The Emerging Role of Biomarkers in the Diagnosis of Gestational Diabetes Mellitus. J. Clin. Med. 2018, 7, 120. [Google Scholar] [CrossRef] [Green Version]
- A World Health Organization Guideline. Diagnostic criteria and classification of hyperglycaemia first detected in pregnancy. Diabetes Res. Clin. Pract. 2014, 103, 341–363. [CrossRef]
- Ruth, C.E.; Hughes, J.R.; Chris, M. Florkowski Is There a Role for HbA1c in Pregnancy? Curr. Diabates Rep. 2016, 16. [Google Scholar] [CrossRef]
- Hughes, R.C.; Moore, M.P.; Gullam, J.E.; Mohamed, K.; Rowan, J. An early pregnancy HbA1c >/=5.9% (41 mmol/mol) is optimal for detecting diabetes and identifies women at increased risk of adverse pregnancy outcomes. Diabetes Care 2014, 37, 2953–2959. [Google Scholar] [CrossRef] [Green Version]
- Corrado, F.; Anna, R.D.; Cannata, M.L.; Interdonato, M.L.; Pintaudi, B.; Di Benedetto, A. Correspondence between first-trimester fasting glycaemia, and oral glucose tolerance test in gestational diabetes diagnosis. Diabetes Metab. 2012, 38, 458–461. [Google Scholar] [CrossRef]
- Zhu, W.W.; Yang, H.X.; Wei, Y.M.; Yan, J.; Wang, Z.L.; Li, X.L.; Wu, H.R.; Li, N.; Zhang, M.H.; Liu, X.H.; et al. Evaluation of the value of fasting plasma glucose in the first prenatal visit to diagnose gestational diabetes mellitus in china. Diabetes Care 2013, 36, 586–590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cosson, E.; Carbillon, L.; Valensi, P. High Fasting Plasma Glucose during Early Pregnancy: A Review about Early Gestational Diabetes Mellitus. J. Diabetes Res. 2017, 2017. [Google Scholar] [CrossRef]
- Wojcik, M.; Zieleniak, A.; Mac-Marcjanek, K.; Wozniak, L.; Cypryk, K. The elevated gene expression level of the A(2B) adenosine receptor is associated with hyperglycemia in women with gestational diabetes mellitus. Diabetes Metab. Res. Rev. 2014, 30, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Mac-Marcjanek, K.; Zieleniak, A.; Zurawska-Klis, M.; Cypryk, K.; Wozniak, L.; Wojcik, M. Expression Profile of Diabetes-Related Genes Associated with Leukocyte Sirtuin 1 Overexpression in Gestational Diabetes. Int. J. Mol. Sci. 2018, 19, 3826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wojcik, M.; Zieleniak, A.; Zurawska-Klis, M.; Cypryk, K.; Wozniak, L.A. Increased expression of immunerelated genes in leukocytes of patients with diagnosed gestational diabetes mellitus (GDM). Exp. Biol. Med. 2016, 241, 457–465. [Google Scholar] [CrossRef] [Green Version]
- Bukowiecka-Matusiak, M.; Burzynska-Pedziwiatr, I.; Sansone, A.; Malachowska, B.; Zurawska-Klis, M.; Ferreri, C.; Chatgilialoglu, C.; Ochedalski, T.; Cypryk, K.; Wozniak, L.A. Lipid profile changes in erythrocyte membranes of women with diagnosed GDM. PLoS ONE 2018, 13, e0203799. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.J.; Larson, M.G.; Vasan, R.S.; Cheng, S.; Rhee, E.P.; McCabe, E.; Lewis, G.D.; Fox, C.S.; Jacques, P.F.; Fernandez, C.; et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 2011, 17, 448–453. [Google Scholar] [CrossRef]
- Leitner, M.; Fragner, L.; Danner, S.; Holeschofsky, N.; Leitner, K.; Tischler, S.; Doerfler, H.; Bachmann, G.; Sun, X.; Jaeger, W.; et al. Combined Metabolomic Analysis of Plasma and Urine Reveals AHBA, Tryptophan and Serotonin Metabolism as Potential Risk Factors in Gestational Diabetes Mellitus (GDM). Front. Mol. Biosci. 2017, 4. [Google Scholar] [CrossRef] [Green Version]
- Lopez-Hernandez, Y.; Herrera-Van Oostdam, A.S.; Toro-Ortiz, J.C.; Lopez, J.A.; Salgado-Bustamante, M.; Murgu, M.; Torres-Torres, L.M. Urinary Metabolites Altered during the Third Trimester in Pregnancies Complicated by Gestational Diabetes Mellitus: Relationship with Potential Upcoming Metabolic Disorders. Int. J. Mol. Sci. 2019, 20, 1186. [Google Scholar] [CrossRef] [Green Version]
- Bentley-Lewis, R.; Huynh, J.; Xiong, G.; Lee, H.; Wenger, J.; Clish, C.; Nathan, D.; Thadhani, R.; Gerszten, R. Metabolomic profiling in the prediction of gestational diabetes mellitus. Diabetologia 2015, 58, 1329–1332. [Google Scholar] [CrossRef] [Green Version]
- Sandgren, J.A.; Deng, G.; Linggonegoro, D.W.; Scroggins, S.M.; Perschbacher, K.J.; Nair, A.R.; Nishimura, T.E.; Zhang, S.Y.; Agbor, L.N.; Wu, J.; et al. Arginine vasopressin infusion is sufficient to model clinical features of preeclampsia in mice. JCI Insight 2018, 3. [Google Scholar] [CrossRef] [PubMed]
- Bourdon, A.; Hannigsberg, J.; Misbert, E.; Tran, T.N.; Amarger, V.; Ferchaud-Roucher, V.; Winer, N.; Darmaun, D. Maternal supplementation with citrulline or arginine during gestation impacts fetal amino acid availability in a model of intrauterine growth restriction (IUGR). Clin. Nutr. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Bazer, F.W.; Satterfield, M.C.; Li, X.; Wang, X.; Johnson, G.A.; Burghardt, R.C.; Dai, Z.; Wang, J.; Wu, Z. Impacts of arginine nutrition on embryonic and fetal development in mammals. Amino Acids 2013, 45, 241–256. [Google Scholar] [CrossRef] [PubMed]
- Vega, M.; Johnson, M.C.; Diaz, H.A.; Urrutia, L.R.; Troncoso, J.L.; Devoto, L. Regulation of human luteal steroidogenesis in vitro by nitric oxide. Endocrine 1998, 8, 185–191. [Google Scholar] [CrossRef]
- Dong, M.; Shi, Y.; Cheng, Q.; Hao, M. Increased nitric oxide in peritoneal fluid from women with idiopathic infertility and endometriosis. J. Reprod. Med. 2001, 46, 887–891. [Google Scholar] [PubMed]
- Rosselli, M.; Keller, P.J.; Dubey, R.K. Role of nitric oxide in the biology, physiology and pathophysiology of reproduction. Hum. Reprod. Update 1998, 4, 3–24. [Google Scholar] [CrossRef] [Green Version]
- Conrad, K.P.; Joffe, G.M.; Kruszyna, H.; Kruszyna, R.; Rochelle, L.G.; Smith, R.P.; Chavez, J.E.; Mosher, M.D. Identification of increased nitric oxide biosynthesis during pregnancy in rats. FASEB J. 1993, 7, 566–571. [Google Scholar] [CrossRef]
- Morris, N.H.; Eaton, B.M.; Dekker, G. Nitric oxide, the endothelium, pregnancy and pre-eclampsia. Br. J. Obstet. Gynaecol. 1996, 103, 4–15. [Google Scholar] [CrossRef]
- Bogdanski, P.; Suliburska, J.; Grabanska, K.; Musialik, K.; Cieslewicz, A.; Skoluda, A.; Jablecka, A. Effect of 3-month L-arginine supplementation on insulin resistance and tumor necrosis factor activity in patients with visceral obesity. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 816–823. [Google Scholar]
- Suliburska, J.; Bogdanski, P.; Szulinska, M.; Pupek-Musialik, D.; Jablecka, A. Changes in mineral status are associated with improvements in insulin sensitivity in obese patients following L-arginine supplementation. Eur. J. Nutr. 2014, 53, 387–393. [Google Scholar] [CrossRef] [Green Version]
- Lorin, J.; Zeller, M.; Guilland, J.C.; Cottin, Y.; Vergely, C.; Rochette, L. Arginine and nitric oxide synthase: Regulatory mechanisms and cardiovascular aspects. Mol. Nutr. Food Res. 2014, 58, 101–116. [Google Scholar] [CrossRef]
- Boger, R.H. The pharmacodynamics of L-arginine. Altern. Ther. Health Med. 2014, 20, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Chong, J.; Wishart, D.S.; Xia, J. Using MetaboAnalyst 4.0 for Comprehensive and Integrative Metabolomics Data Analysis. Curr. Protoc. Bioinf. 2019, 68, e86. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, H.D.; Colagiuri, S.; Roglic, G.; Hod, M. Diagnosis of GDM: A suggested consensus. Best Pract. Res. Clin. Obstet. Gynaecol. 2015, 29, 194–205. [Google Scholar] [CrossRef] [Green Version]
- Catalano, P.M.; Huston, L.; Amini, S.B.; Kalhan, S.C. Longitudinal changes in glucose metabolism during pregnancy in obese women with normal glucose tolerance and gestational diabetes mellitus. Am. J. Obstet. Gynecol. 1999, 180, 903–916. [Google Scholar] [CrossRef]
- Bao, W.; Dar, S.; Zhu, Y.; Wu, J.; Rawal, S.; Li, S.; Weir, N.L.; Tsai, M.Y.; Zhang, C. Plasma concentrations of lipids during pregnancy and the risk of gestational diabetes mellitus: A longitudinal study. J. Diabetes 2018, 10, 487–495. [Google Scholar] [CrossRef]
- Nurjhan, N.; Bucci, A.; Perriello, G.; Stumvoll, M.; Dailey, G.; Bier, D.M.; Toft, I.; Jenssen, T.G.; Gerich, J.E. Glutamine: A major gluconeogenic precursor and vehicle for interorgan carbon transport in man. J. Clin. Investig. 1995, 95, 272–277. [Google Scholar] [CrossRef]
- Mirzaei, H.; Suarez, J.A.; Longo, V.D. Protein and amino acid restriction, aging and disease: From yeast to humans. Trends Endocrinol. Metab. 2014, 25, 558–566. [Google Scholar] [CrossRef] [Green Version]
- Cetin, I.; de Santis, M.S.; Taricco, E.; Radaelli, T.; Teng, C.; Ronzoni, S.; Spada, E.; Milani, S.; Pardi, G. Maternal and fetal amino acid concentrations in normal pregnancies and in pregnancies with gestational diabetes mellitus. Am. J. Obstet. Gynecol. 2005, 192, 610–617. [Google Scholar] [CrossRef] [PubMed]
- de Seymour, J.V.; Conlon, C.A.; Sulek, K.; Villas Bôas, S.G.; McCowan, L.M.; Kenny, L.C.; Baker, P.N. Early pregnancy metabolite profiling discovers a potential biomarker for the subsequent development of gestational diabetes mellitus. Acta Diabetol. 2014, 51, 887–890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sachse, D.; Sletner, L.; Mørkrid, K.; Jenum, A.K.; Birkeland, K.I.; Rise, F.; Piehler, A.P.; Berg, J.P. Metabolic changes in urine during and after pregnancy in a large, multiethnic population-based cohort study of gestational diabetes. PLoS ONE 2012, 7, e52399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pappa, K.I.; Vlachos, G.; Theodora, M.; Roubelaki, M.; Angelidou, K.; Antsaklis, A. Intermediate metabolism in association with the amino acid profile during the third trimester of normal pregnancy and diet-controlled gestational diabetes. Am. J. Obstet. Gynecol. 2007, 196, 65.e1–65.e5. [Google Scholar] [CrossRef] [PubMed]
- Hou, W.; Meng, X.; Zhao, A.; Zhao, W.; Pan, J.; Tang, J.; Huang, Y.; Li, H.; Jia, W.; Liu, F. Development of Multimarker Diagnostic Models from Metabolomics Analysis for Gestational Diabetes Mellitus (GDM). Mol. Cell. Proteomics 2018, 17, 431–441. [Google Scholar] [CrossRef] [Green Version]
- Lowe, W.L.; Karban, J. Genetics, genomics and metabolomics: New insights into maternal metabolism during pregnancy. Diabet. Med. 2014, 31, 254–262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, J.; Ren, W.; Chen, S.; Li, Y.; Han, H.; Gao, J.; Liu, G.; Wu, X.; Li, T.; Woo Kim, S.; et al. Metabolic Regulation of Methionine Restriction in Diabetes. Mol. Nutr. Food Res. 2018, 62, e1700951. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, N.; Razi, F.; Nasli-Esfahani, E.; Qorbani, M.; Shirzad, N.; Larijani, B. Amino acid profiling in the gestational diabetes mellitus. J. Diabetes Metab. Disord. 2017, 16, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, S.C.; Mato, J.M. S-Adenosylmethionine in cell growth, apoptosis and liver cancer. J. Gastroenterol. Hepatol. 2008, 23, S73–S77. [Google Scholar] [CrossRef] [Green Version]
- Kamal, M.A.; Greig, N.H. Editorial: Frontier Views in Designing Therapeutic Candidates for Management of Diverse Diseases. Curr. Pharm. Des. 2017, 23, 1571–1574. [Google Scholar] [CrossRef] [Green Version]
- Dejong, C.H.; van de Poll, M.C.; Soeters, P.B.; Jalan, R.; Olde Damink, S.W. Aromatic amino acid metabolism during liver failure. J. Nutr. 2007, 137, 1579S–1585S. [Google Scholar] [CrossRef]
- Butte, N.F.; Hsu, H.W.; Thotathuchery, M.; Wong, W.W.; Khoury, J.; Reeds, P. Protein metabolism in insulin-treated gestational diabetes. Diabetes Care 1999, 22, 806–811. [Google Scholar] [CrossRef]
- Metzger, B.E.; Phelps, R.L.; Freinkel, N.; Navickas, I.A. Effects of gestational diabetes on diurnal profiles of plasma glucose, lipids, and individual amino acids. Diabetes Care 1980, 3, 402–409. [Google Scholar] [CrossRef]
- Roy, C.; Tremblay, P.Y.; Anassour-Laouan-Sidi, E.; Lucas, M.; Forest, J.C.; Giguere, Y.; Ayotte, P. Risk of gestational diabetes mellitus in relation to plasma concentrations of amino acids and acylcarnitines: A nested case-control study. Diabetes Res. Clin. Pract. 2018, 140, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Chorell, E.; Hall, U.A.; Gustavsson, C.; Berntorp, K.; Puhkala, J.; Luoto, R.; Olsson, T.; Holmang, A. Pregnancy to postpartum transition of serum metabolites in women with gestational diabetes. Metabolism 2017, 72, 27–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piatti, P.M.; Monti, L.D.; Valsecchi, G.; Magni, F.; Setola, E.; Marchesi, F.; Galli-Kienle, M.; Pozza, G.; Alberti, K.G. Long-term oral L-arginine administration improves peripheral and hepatic insulin sensitivity in type 2 diabetic patients. Diabetes Care 2001, 24, 875–880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vasilijevic, A.; Buzadzic, B.; Korac, A.; Petrovic, V.; Jankovic, A.; Korac, B. Beneficial effects of L-arginine nitric oxide-producing pathway in rats treated with alloxan. J. Physiol. 2007, 584, 921–933. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Han, M.; Rezaei, A.; Li, D.; Wu, G.; Ma, X. L-Arginine Modulates Glucose and Lipid Metabolism in Obesity and Diabetes. Curr. Protein Pept. Sci. 2017, 18, 599–608. [Google Scholar] [CrossRef]
- San Martin, R.; Sobrevia, L. Gestational diabetes and the adenosine/L-arginine/nitric oxide (ALANO) pathway in human umbilical vein endothelium. Placenta 2006, 27, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Guzmán-Gutiérrez, E.; Abarzúa, F.; Belmar, C.; Nien, J.K.; Ramírez, M.A.; Arroyo, P.; Salomón, C.; Westermeier, F.; Puebla, C.; Leiva, A.; et al. Functional link between adenosine and insulin: A hypothesis for fetoplacental vascular endothelial dysfunction in gestational diabetes. Curr. Vasc. Pharmacol. 2011, 9, 750–762. [Google Scholar] [CrossRef]
- Sydow, K.; Mondon, C.E.; Cooke, J.P. Insulin resistance: Potential role of the endogenous nitric oxide synthase inhibitor ADMA. Vasc. Med. 2005, 10, S35–S43. [Google Scholar] [CrossRef] [Green Version]
- Matsumoto, T.; Kobayashi, T.; Ishida, K.; Taguchi, K.; Kamata, K. Enhancement of mesenteric artery contraction to 5-HT depends on Rho kinase and Src kinase pathways in the ob/ob mouse model of type 2 diabetes. Br. J. Pharmacol. 2010, 160, 1092–1104. [Google Scholar] [CrossRef] [Green Version]
- Zou, M.H.; Hou, X.Y.; Shi, C.M.; Kirkpatrick, S.; Liu, F.; Goldman, M.H.; Cohen, R.A. Withdrawal: Activation of 5′-AMP-activated kinase is mediated through c-Src and phosphoinositide 3-kinase activity during hypoxia-reoxygenation of bovine aortic endothelial cells: Role of peroxynitrite. J. Biol. Chem. 2019, 294, 18016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaneto, H.; Fujii, J.; Seo, H.G.; Suzuki, K.; Matsuoka, T.; Nakamura, M.; Tatsumi, H.; Yamasaki, Y.; Kamada, T.; Taniguchi, N. Apoptotic cell death triggered by nitric oxide in pancreatic beta-cells. Diabetes 1995, 44, 733–738. [Google Scholar] [CrossRef] [PubMed]
- Kaneto, H.; Nakatani, Y.; Kawamori, D.; Miyatsuka, T.; Matsuoka, T.A.; Matsuhisa, M.; Yamasaki, Y. Role of oxidative stress, endoplasmic reticulum stress, and c-Jun N-terminal kinase in pancreatic beta-cell dysfunction and insulin resistance. Int. J. Biochem. Cell Biol. 2005, 37, 1595–1608. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Xia, W.; Li, H.; Liu, F.; Li, Y.; Sun, X.; Lu, S.; Xu, S. Normal pregnancy induced glucose metabolic stress in a longitudinal cohort of healthy women: Novel insights generated from a urine metabolomics study. Medicine 2018, 97, e12417. [Google Scholar] [CrossRef] [PubMed]
- Kirwan, J.P.; Varastehpour, A.; Jing, M.; Presley, L.; Shao, J.; Friedman, J.E.; Catalano, P.M. Reversal of insulin resistance postpartum is linked to enhanced skeletal muscle insulin signaling. J. Clin. Endocrinol. Metab. 2004, 89, 4678–4684. [Google Scholar] [CrossRef] [Green Version]
- Allain, C.C.; Poon, L.S.; Chan, C.S.; Richmond, W.; Fu, P.C. Enzymatic determination of total serum cholesterol. Clin. Chem. 1974, 20, 470–475. [Google Scholar] [CrossRef]
- Willms, B.; Soling, H.D. Methodology of enzymatic triglyceride determination in the serum. Z. Klin. Chem. Klin. Biochem. 1967, 5, 276–277. [Google Scholar]
- Metus, P.; Ruzzante, N.; Bonvicini, P.; Meneghetti, M.; Zaninotto, M.; Plebani, M. Immunoturbidimetric assay of glycated hemoglobin. J. Clin. Lab. Anal. 1999, 13, 5–8. [Google Scholar] [CrossRef]
- Eda, S.; Kaufmann, J.; Roos, W.; Pohl, S. Development of a new microparticle-enhanced turbidimetric assay for C-reactive protein with superior features in analytical sensitivity and dynamic range. J. Clin. Lab. Anal. 1998, 12, 137–144. [Google Scholar] [CrossRef]
- Sapin, R.; Le Galudec, V.; Gasser, F.; Pinget, M.; Grucker, D. Elecsys insulin assay: Free insulin determination and the absence of cross-reactivity with insulin lispro. Clin. Chem. 2001, 47, 602–605. [Google Scholar] [CrossRef] [Green Version]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Variable | NGT (n = 35) | GDM (n = 29) | p-Value |
|---|---|---|---|
| Age [years] | 28.0 (26.0–31.0) | 30.0 (28.0–34.0) | 0.124 |
| Pre-pregnancy BMI [kg/m2] | 23.9 (21.8–26.7) | 23.8 (20.2–26.3) | 0.728 |
| Gestational weight gain [kg] | 8.0 (5.8–11.0) | 9.2 (6.0–11.6) | 0.401 |
| FPG [mg/dL] | 80.0 (75.0–85.0) | 85.0 (81.0–101.0) | 0.002 |
| 2h OGTT [mg/dL] | 139.0 (139.0–125.0) | 154.0 (145.0–166.0) | 0.000 |
| HbA1C [%] | 5.1 (5.0–5.5) | 5.1 (5.0–5.3) | 0.799 |
| insulin [µlU/mL] | 8.0 (6.1–9.0) | 12.1 (7.3–15.8) | 0.020 |
| HOMA-B | 149.1 (122.4–174.5) | 165.4 (139.8–241.9) | 0.242 |
| HOMA-IR | 1.6 (1.1–1.8) | 2.6 (1.5–3.7) | 0.005 |
| TG [mg/dL] | 208.5 (174.5–246.8) | 214.3 (172.7–227.0) | 0.779 |
| TC [mg/dL] | 268.2 (230.5–288.3) | 245.2 (224.2–275.6) | 0.146 |
| HDL-C [mg/dL] | 83.3 (71.8–98.0) | 75.2 (65.8–87.3) | 0.120 |
| LDL-C [mg/dL] | 141.0 (121.0–168.0) | 126.0 (116.0–150.0) | 0.203 |
| CRP [mg/L] | 3.7 (2.1–7.3) | 3.8 (2.1–5.3) | 0.569 |
| AA $ | NGT (n = 35) | GDM (n = 29) | FC | p-Value (U Mann–Whitney Test) | OR (95%CI) | p-Value (LR) | p-Value (Wald’s Test) | ROC AUC |
|---|---|---|---|---|---|---|---|---|
| Ala | 209.5 (195.0–243.0) | 213.2 (192.3–232.5) | 1.02 | 0.686 | 0.998 (0.987–1.009) | 0.699 | 0.701 | 0.530 |
| Arg | 96.1 (82.3–116.6) | 78.0 (62.6–90.4) | 0.81 | 0.001 # | 0.96 (0.934–0.986) | 0.000 # | 0.003 | 0.749 |
| Asn | 23.8 (21.8–26.8) | 25.2 (21.7–27.5) | 1.06 | 0.671 | 1.035 (0.94–1.139) | 0.486 | 0.489 | 0.532 |
| Gln | 1054.1 (883.2–1127.3) | 924.4 (807.8–1014.0) | 0.88 | 0.018 | 0.997 (0.993–0.999) | 0.024 | 0.032 | 0.673 |
| Glu | 74.4 (65.6–86.6) | 76.2 (65.9–82.4) | 1.02 | 0.656 | 0.988 (0.957–1.019) | 0.443 | 0.448 | 0.533 |
| Gly | 113.5 (102.1–128.5) | 110.0 (97.2–127.9) | 0.97 | 0.458 | 0.995 (0.975–1.015) | 0.587 | 0.590 | 0.555 |
| His | 132.2 (123.1–144.3) | 119.9 (107.6–130.0) | 0.91 | 0.007 | 0.968 (0.941–0.995) | 0.013 | 0.020 | 0.698 |
| Ile | 43.2 (39.3–49.4) | 41.5 (36.0–44.8) | 0.96 | 0.142 | 0.955 (0.892–1.023) | 0.172 | 0.187 | 0.608 |
| Leu | 107.1 (98.7–122.7) | 107.0 (96.8–115.7) | 0.99 | 0.345 | 0.987 (0.962–1.012) | 0.295 | 0.309 | 0.569 |
| Lys | 136.3 (115.9–149.2) | 126.0 (103.5–138.5) | 0.92 | 0.084 | 0.986 (0.966–1.007) | 0.169 | 0.183 | 0.627 |
| Met | 14.9 (13.3–17.8) | 14.5 (12.3–15.7) | 0.97 | 0.010 | 0.882 (0.747–1.042) | 0.113 | 0.139 | 0.621 |
| Phe | 54.8 (49.5–61.9) | 52.1 (46.4–55.2) | 0.95 | 0.019 | 0.949 (0.895–1.006) | 0.051 | 0.081 | 0.672 |
| Pro | 88.0 (77.6–101.7) | 81.1 (75.6–92.2) | 0.92 | 0.318 | 0.987 (0.963–1.012) | 0.311 | 0.320 | 0.573 |
| Pro-OH | 6.7 (4.1–9.6) | 8.0 (6.8–8.4) | 1.19 | 0.366 | 1.081 (0.933–1.253) | 0.281 | 0.297 | 0.566 |
| Ser | 99.6 (83.8–112.1) | 85.9 (73.5–96.9) | 0.86 | 0.012 | 0.963 (0.934–0.993) | 0.010 | 0.016 | 0.684 |
| Thr | 108.1 (91.0–122.4) | 109.8 (94.2–123.3) | 1.02 | 0.696 | 1.005 (0.982–1.029) | 0.651 | 0.652 | 0.529 |
| Trp | 60.3 (54.8–65.7) | 59.7 (54.0–64.3) | 0.99 | 0.866 | 0.999 (0.949–1.053) | 0.983 | 0.983 | 0.513 |
| Tyr | 26.8 (24.3–30.9) | 26.1 (22.5–27.6) | 0.97 | 0.134 | 0.938 (0.855–1.029) | 0.144 | 0.177 | 0.610 |
| Val | 121.7 (110.8–134.5) | 129.1 (115.8–139.3) | 1.06 | 0.418 | 1.008 (0.988–1.028) | 0.449 | 0.455 | 0.560 |
| Variable | Rho | p-Value | FDR |
|---|---|---|---|
| Age [years] | −0.08 | 0.549 | 0.676 |
| BMI [kg/m2] | 0.14 | 0.275 | 0.400 |
| Weight gein [kg] | −0.01 | 0.909 | 0.909 |
| FPG [mg/dL] | −0.27 | 0.032 | 0.060 |
| 2h OGTT [mg/dL] | −0.27 | 0.034 | 0.060 |
| HbA1C [mmol/mol] | 0.03 | 0.813 | 0.839 |
| Insulin [µlU/mL] | 0.16 | 0.261 | 0.398 |
| HOMA-B | 0.31 | 0.030 | 0.060 |
| HOMA-IR | 0.09 | 0.515 | 0.659 |
| TG [mg/dL] | 0.33 | 0.011 | 0.027 |
| TC [mg/dL] | 0.05 | 0.692 | 0.764 |
| HDL-C [mg/dL] | −0.14 | 0.308 | 0.429 |
| LDL-C [mg/dL] | 0.06 | 0.671 | 0.764 |
| CRP [mg/L] | 0.19 | 0.194 | 0.310 |
| Ala [µM/L] | 0.38 | 0.002 | 0.007 |
| Asn [µM/L] | 0.07 | 0.587 | 0.695 |
| Gln [µM/L] | 0.04 | 0.749 | 0.799 |
| Glu [µM/L] | 0.44 | 0.000 | 0.001 |
| Gly [µM/L] | 0.32 | 0.011 | 0.027 |
| His [µM/L] | 0.54 | 0.000 | 0.000 |
| Ile [µM/L] | 0.46 | 0.000 | 0.001 |
| Leu [µM/L] | 0.54 | 0.000 | 0.000 |
| Lys [µM/L] | 0.34 | 0.006 | 0.018 |
| Met [µM/L] | 0.61 | 0.000 | 0.000 |
| Phe [µM/L] | 0.81 | 0.000 | 0.000 |
| Pro [µM/L] | 0.38 | 0.002 | 0.007 |
| Pro-OH [µM/L] | 0.26 | 0.041 | 0.069 |
| Ser [µM/L] | 0.51 | 0.000 | 0.000 |
| Thr [µM/L] | 0.09 | 0.462 | 0.617 |
| Trp [µM/L] | 0.30 | 0.017 | 0.040 |
| Tyr [µM/L] | 0.66 | 0.000 | 0.000 |
| Val [µM/L] | 0.27 | 0.033 | 0.060 |
| Variable | 3 Mo | FC * | 1 Y | FC * | 3Mo/GDM-1Y/3Mo Tendency | p-Value (Friedman’s Test) |
|---|---|---|---|---|---|---|
| FPG [mg/dL] | 87.0 (82.0–91.0) | 1.02 | 91.0 (82.0–101.0) | 1.07 | →-↗ | 0.001 # |
| 2h OGTT [mg/dL] | 96.0 (82.0–108.0) | 0.62 | 101.5 (84.0–107.0) | 0.66 | ↘-→ | 0.000 # |
| HbA1C [%] | 5.2 (5.0–5.5) | 1.02 | 5.1 (4.9–5.3) | 1.00 | →-→ | 0.094 |
| insulin [µlU/mL | 7.9 (5.9–10.0) | 0.65 | 9.3 (5.5–11.1) | 0.77 | ↘-→ | 0.000 # |
| HOMA-B | 127.3 (94.2–178.1) | 0.77 | 121.5 (89.1–168.0) | 0.73 | →-→ | 0.003 # |
| HOMA-IR | 1.6 (1.1–2.3) | 0.62 | 2.2 (1.1–2.7) | 0.85 | →-→ | 0.004 # |
| TG-C [mg/dL] | 81.4 (56.9–102.3) | 0.38 | 89.6 (53.9–103.1) | 0.42 | ↘-→ | 0.000 # |
| TC [mg/dL] | 187.9 (172.5–214.3) | 0.77 | 185.0 (165.0–202.0) | 0.75 | ↘-→ | 0.000 # |
| HDL-C [mg/dL] | 61.1 (51.2–75.3) | 0.81 | 63.0 (54.0–78.0) | 0.84 | ↘-→ | 0.001 # |
| LDL-C [mg/dL] | 115.0 (100.0–127.0) | 0.91 | 95.0 (87.0–132.0) | 0.75 | ↘-→ | 0.018 # |
| CRP [mg/L] | 1.4 (0.7–2.6) | 0.37 | 1.1 (0.8–1.9) | 0.29 | ↘-→ | 0.000 # |
| Ala [µM/L] | 266.4 (205.8–300.0) | 1.25 | 237.3 (205.8–274.5) | 1.11 | ↗-→ | 0.006 # |
| Arg [µM/L] | 85.2 (76.5–99.1) | 1.09 | 71.4 (63.1–81.1) | 0.92 | →-→ | 0.065 |
| Asn [µM/L] | 24.7 (23.7–26.6) | 0.98 | 29.8 (26.3–32.5) | 1.18 | →-↗ | 0.009 # |
| Gln [µM/L] | 1258.6 (1161.0–1450.5) | 1.36 | 1323.8 (1179.1–1472.7) | 1.43 | ↗-→ | 0.000 # |
| Glu [µM/L] | 91.5 (78.9–99.0) | 1.20 | 86.5 (72.2–96.0) | 1.14 | ↗-→ | 0.001 # |
| Gly [µM/L] | 229.0 (178.1–267.9) | 2.08 | 196.3 (165.9–266.2) | 1.78 | ↗-→ | 0.000 # |
| His [µM/L] | 109.2 (96.5–123.7) | 0.91 | 124.8 (113.1–136.7) | 1.04 | ↘-↗ | 0.032 # |
| Ile [µM/L] | 49.5 (40.2–58.9) | 1.19 | 49.8 (46.0–54.6) | 1.20 | ↗-→ | 0.001 # |
| Leu [µM/L] | 138.2 (119.1–156.3) | 1.29 | 137.2 (122.4–150.0) | 1.28 | ↗-→ | 0.000 # |
| Lys [µM/L] | 141.8 (128.9–151.3) | 1.13 | 159.6 (132.1–185.1) | 1.27 | ↗-→ | 0.002 # |
| Met [µM/L] | 16.8 (14.7–17.8) | 1.16 | 16.2 (14.5–18.7) | 1.12 | ↗-→ | 0.003 # |
| Phe [µM/L] | 58.1 (52.7–62.9) | 1.12 | 58.3 (54.4–61.0) | 1.12 | ↗-→ | 0.000 # |
| Pro [µM/L] | 128.4 (116.3–145.4) | 1.58 | 121.0 (101.9–145.8) | 1.49 | ↗-→ | 0.000 # |
| Pro-OH [µM/L] | 10.8 (7.0–15.5) | 1.35 | 8.6 (6.7–10.6) | 1.08 | ↗-→ | 0.028 # |
| Ser [µM/L] | 92.2 (85.8–114.6) | 1.07 | 116.6 (110.8–127.1) | 1.36 | ↗-↗ | 0.002 # |
| Thr [µM/L] | 64.6 (53.4–86.5) | 0.59 | 84.3 (68.1–90.3) | 0.77 | ↘-↗ | 0.000 # |
| Trp [µM/L] | 66.5 (57.3–73.7) | 1.11 | 75.8 (69.8–79.8) | 1.27 | ↗-↗ | 0.000 # |
| Tyr [µM/L] | 41.2 (36.3–47.8) | 1.58 | 41.4 (37.4–44.3) | 1.59 | ↗-→ | 0.000 # |
| Val [µM/L] | 177.5 (152.8–196.8) | 1.37 | 178.8 (159.9–196.6) | 1.38 | ↗-→ | 0.000 # |
| AA | FC 3 Mo/NGT | p-Value | FC 1Y/NGT | p-Value |
|---|---|---|---|---|
| Ala | 1.27 | 0.004 # | 1.13 | 0.075 |
| Arg | 0.89 | 0.067 | 0.74 | 0.000 # |
| Asn | 1.04 | 0.517 | 1.25 | 0.001 # |
| Gln | 1.19 | 0.000 # | 1.26 | 0.000 # |
| Glu | 1.23 | 0.003 # | 1.16 | 0.075 |
| Gly | 2.02 | 0.000 # | 1.73 | 0.000 # |
| His | 0.83 | 0.000 # | 0.94 | 0.080 |
| Ile | 1.15 | 0.071 | 1.15 | 0.002 # |
| Leu | 1.29 | 0.000 # | 1.28 | 0.000 # |
| Lys | 1.04 | 0.269 | 1.17 | 0.008 # |
| Met | 1.13 | 0.169 | 1.09 | 0.094 |
| Phe | 1.06 | 0.526 | 1.06 | 0.215 |
| Pro | 1.46 | 0.000 # | 1.38 | 0.000 # |
| Pro-OH | 1.61 | 0.001 # | 1.28 | 0.133 |
| Ser | 0.93 | 0.647 | 1.17 | 0.000 # |
| Thr | 0.60 | 0.000 # | 0.78 | 0.000 # |
| Trp | 1.10 | 0.018 # | 1.26 | 0.000 # |
| Tyr | 1.54 | 0.000 # | 1.54 | 0.000 # |
| Val | 1.46 | 0.000 # | 1.47 | 0.000 # |
| AA | Preg. NGT | Preg. HMDB | FC | 1 Y Post-GDM | Norm. HMDB | FC |
|---|---|---|---|---|---|---|
| ALA | 217.5 (42.0) | 258.6 (85.4) | 0.84 * | 244.4 (58.4) | 319.4 (71.7) | 0.77 * |
| ARG | 99.7 (27.7) | 122.8 (39.7) | 0.81 * | 73.9 (18.3) | 75.0 (24.0) | 0.98 |
| ASN | 24.3 (5.4) | 31.9 (13.6) | 0.76 * | 29.8 (7.5) | 47.0 (9.0) | 0.63 * |
| GLN | 1019.5 (180.4) | 250.2 (77.3) | 4.07 * | 1323.8 (290.5) | 578.0 (85.0) | 2.29 * |
| GLU | 79.3 (18.2) | NA | 91.3 (26.7) | 46.0 (13.0) | 1.98 * | |
| GLY | 116.2 (22.3) | 209.8 (74.9) | 0.55 * | 215.1 (65.9) | 258.0 (64.0) | 0.83 * |
| HIS | 132.2 (19.2) | NA | 122.4 (21.4) | 83.0 (14.0) | 1.47 * | |
| ILE | 44.4 (7.0) | 40.9 (11.7) | 1.09 *. | 50.4 (7.0) | 56.0 (12.0) | 0.90 |
| LEU | 112.4 (19.9) | 71.6 (40.6) | 1.57 * | 138.0 (22.1) | 160.0 (27.0) | 0.86 * |
| LYS | 134.8 (24.4) | 97.4 (29.5) | 1.38 * | 154.5 (37.7) | 183.0 (34.0) | 0.84 * |
| MET | 15.8 (3.4) | 26.1 (11.3) | 0.61 * | 16.8 (3.1) | 27.0 (5.0) | 0.62 * |
| PHE | 57.1 (10.5) | 58.2 (28.7) | 0.98 | 58.5 (7.3) | 52.0 (5.0) | 1.12 * |
| PRO | 92.2 (21.8) | 125.4 (48.5) | 0.73 * | 127.0 (38.5) | 168.0 (49.0) | 0.76 * |
| H-Pro | 7.2 (3.4) | NA | 9.6 (6.8) | 16.0 (9.0) | 0.60 * | |
| SER | 97.4 (18.8) | 132.0 (50.6) | 0.74 * | 115.4 (19.6) | 127.0 (29.0) | 0.91 |
| THR | 107.4 (20.1) | 108.8 (42.5) | 0.99 | 84.3 (22.2) | 154.0 (40.0) | 0.55 * |
| TRP | 59.4 (9.8) | 74.4 (11.2) | 48.7 (11.6) | 1.53 * | ||
| TYR | 28.3 (6.7) | 55.8 (19.6) | 0.51 * | 41.4 (7.6) | 61.0 (13.0) | 0.68 * |
| VAL | 124.7 (22.8) | 119.6 (38.1) | 1.04 | 178.8 (35.4) | 209.0 (31.0) | 0.86 * |
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burzynska-Pedziwiatr, I.; Jankowski, A.; Kowalski, K.; Sendys, P.; Zieleniak, A.; Cypryk, K.; Zurawska-Klis, M.; Wozniak, L.A.; Bukowiecka-Matusiak, M. Associations of Arginine with Gestational Diabetes Mellitus in a Follow-Up Study. Int. J. Mol. Sci. 2020, 21, 7811. https://doi.org/10.3390/ijms21217811
Burzynska-Pedziwiatr I, Jankowski A, Kowalski K, Sendys P, Zieleniak A, Cypryk K, Zurawska-Klis M, Wozniak LA, Bukowiecka-Matusiak M. Associations of Arginine with Gestational Diabetes Mellitus in a Follow-Up Study. International Journal of Molecular Sciences. 2020; 21(21):7811. https://doi.org/10.3390/ijms21217811
Chicago/Turabian StyleBurzynska-Pedziwiatr, Izabela, Adrian Jankowski, Konrad Kowalski, Przemyslaw Sendys, Andrzej Zieleniak, Katarzyna Cypryk, Monika Zurawska-Klis, Lucyna A. Wozniak, and Malgorzata Bukowiecka-Matusiak. 2020. "Associations of Arginine with Gestational Diabetes Mellitus in a Follow-Up Study" International Journal of Molecular Sciences 21, no. 21: 7811. https://doi.org/10.3390/ijms21217811






