Enhancing the Therapeutic Potential of CCL2-Overexpressing Mesenchymal Stem Cells in Acute Stroke
Abstract
1. Introduction
2. Results
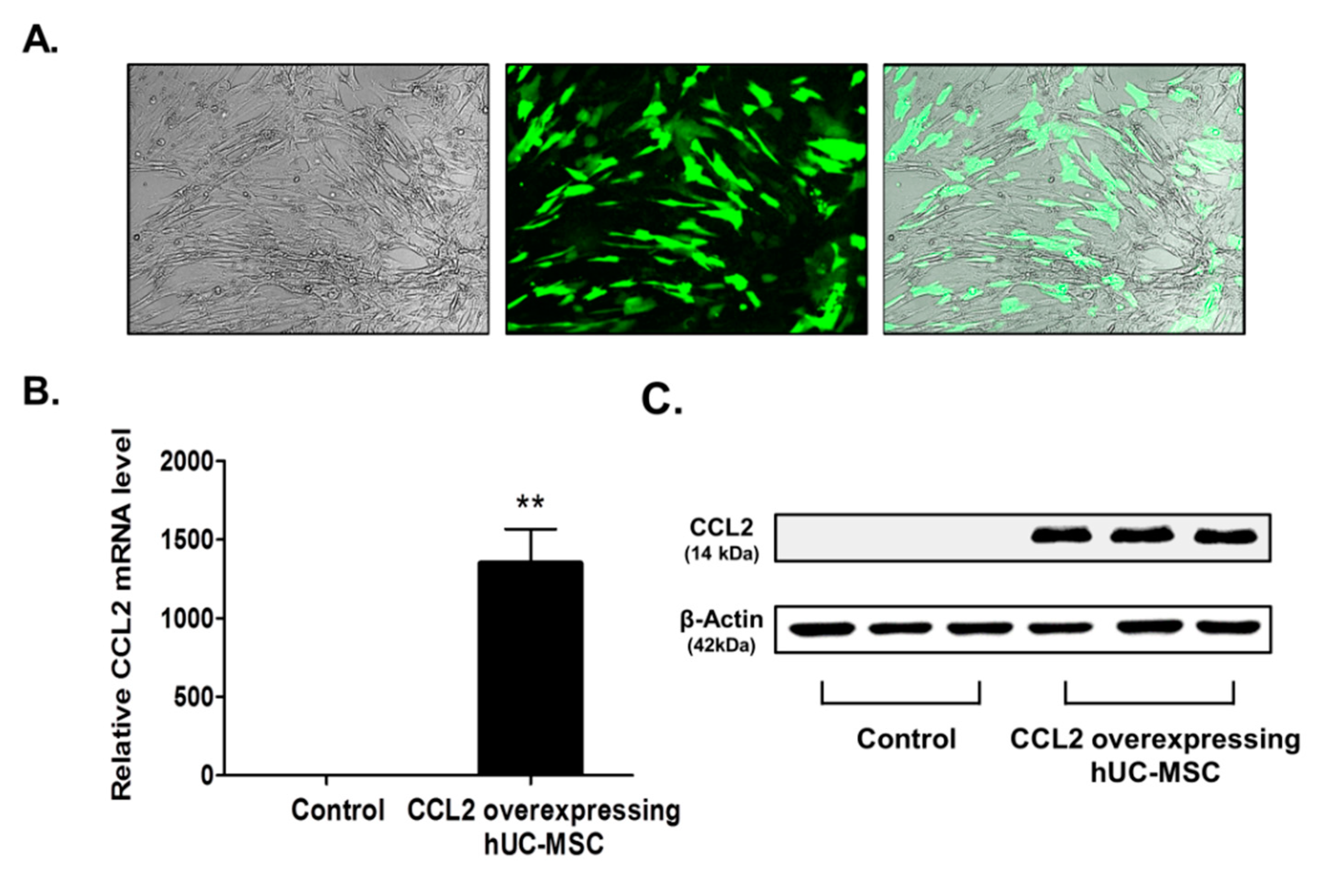
2.1. Confirmation of CCL2 Overexpression in Human Umbilical Cord-Derived Mesenchymal Stem Cell (hUC-MSCs)
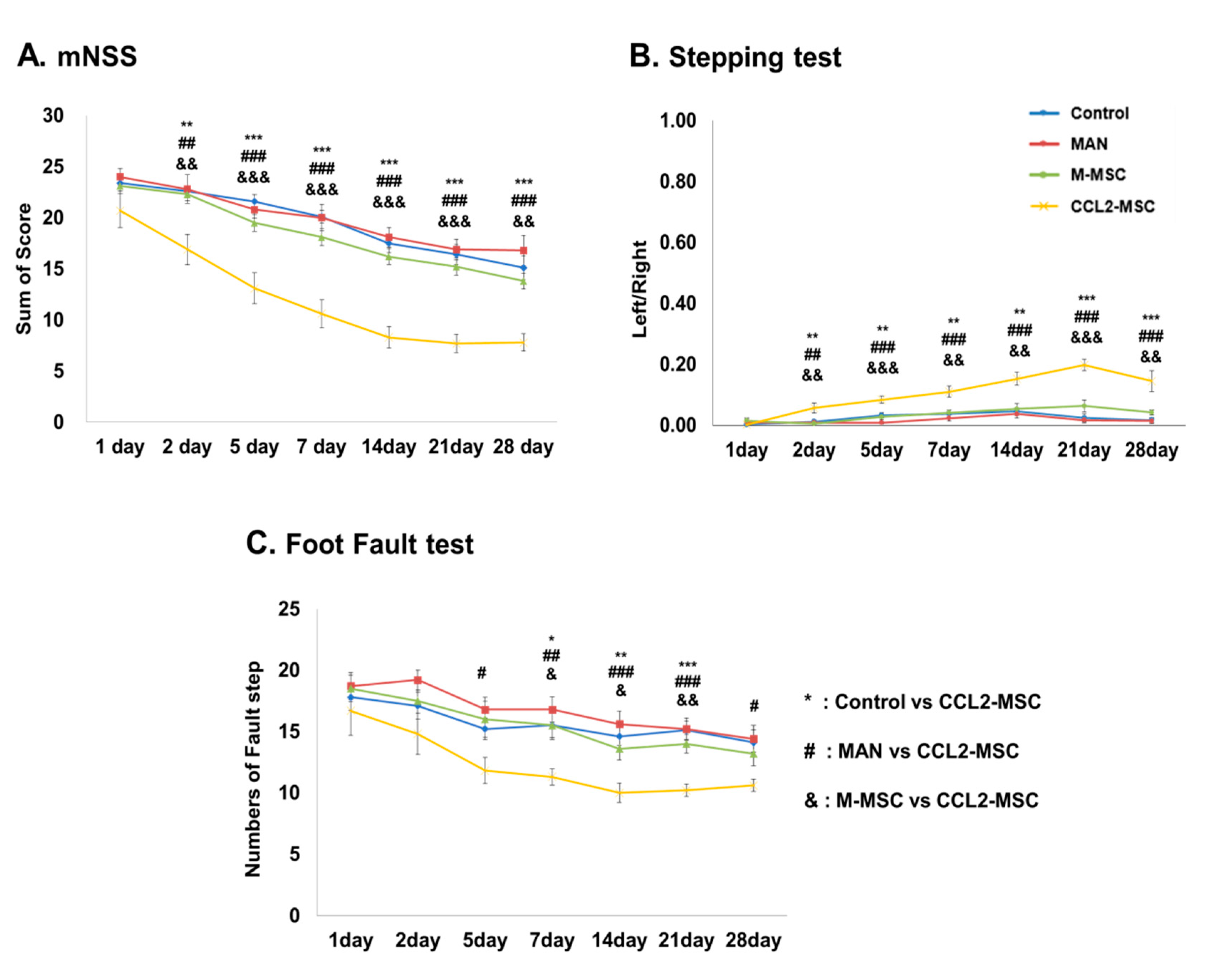
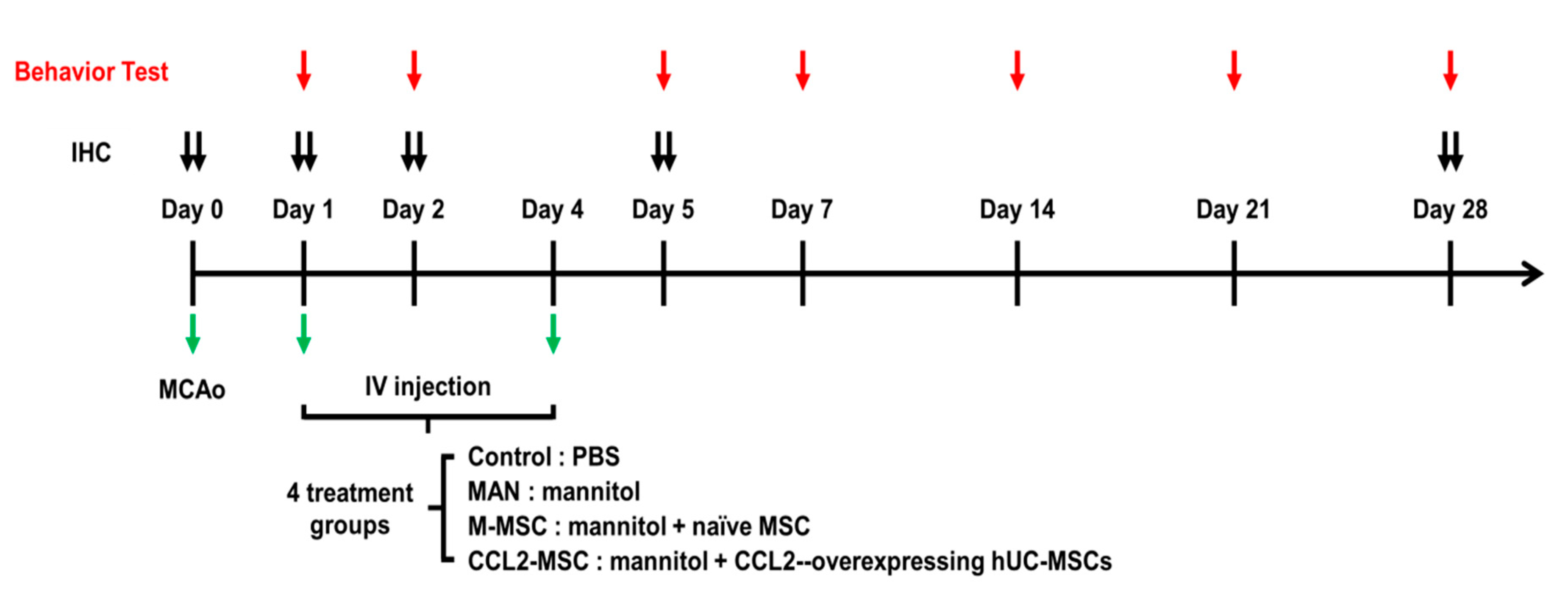
2.2. Improvement of Functional Deficits in Middle Cerebral Artery Occlusion (MCAo) Model Rats after Transplantation of CCL2-Overexpressing hUC-MSCs
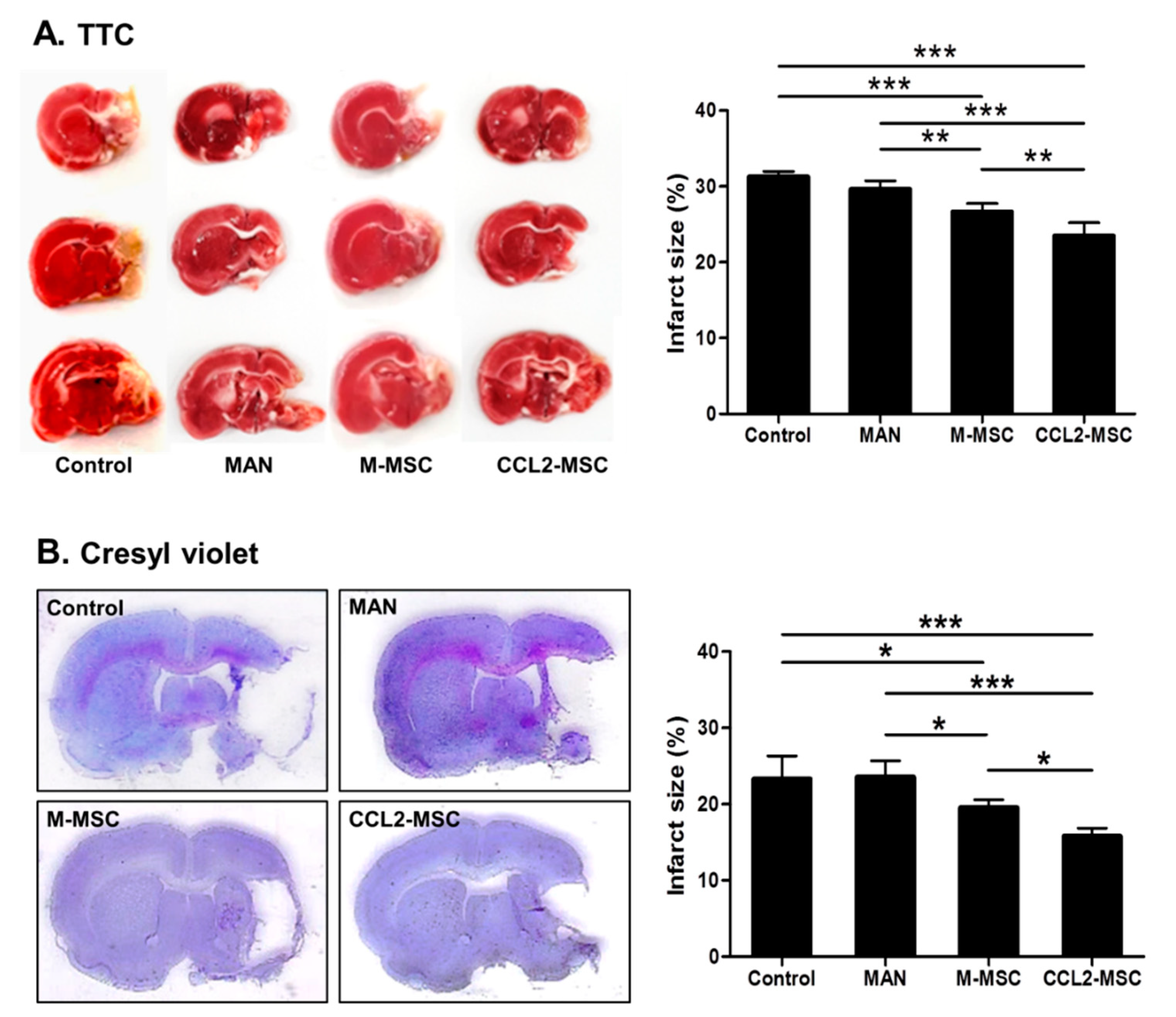
2.3. Reduced Infarct Size Following Transplantation of CCL2-Overexpressing hUC-MSCs
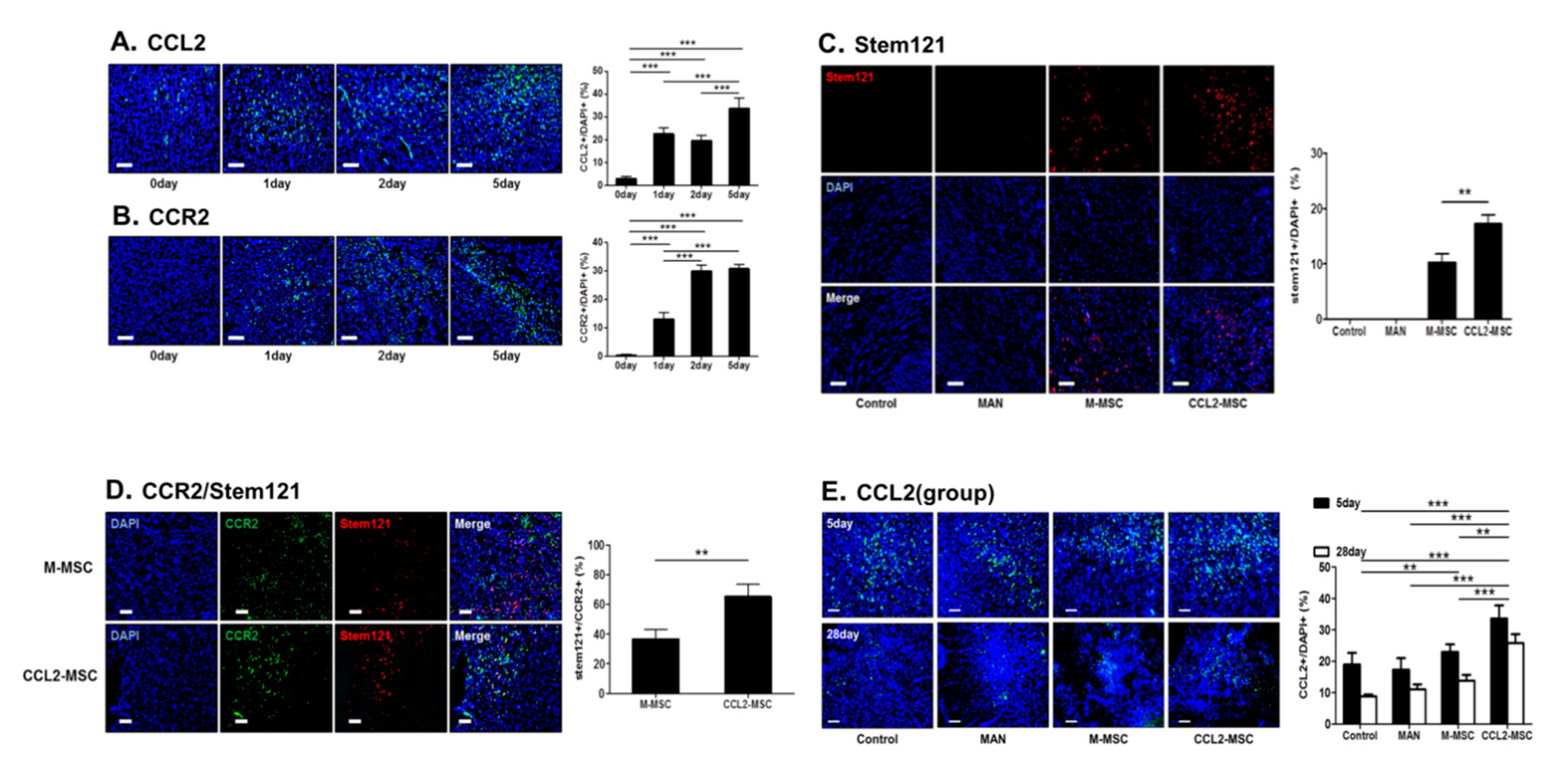
2.4. CCL2 and CCR2 Levels are Elevated in the Brain During the Acute Ischemic Phase
2.5. Increased Engraftment of CCL2-Overexpressing hUC-MSCs in Areas of Upregulated CCR2 Levels in the Ischemic Brain
2.6. CCL2 Expression is Sustained in the Brain Following CCL2-Overexpressing hUC-MSC Transplantation
2.7. Transplantation of CCL2-Overexpressing hUC-MSCs Increases Angiogenesis in the Peri-Infarct Area
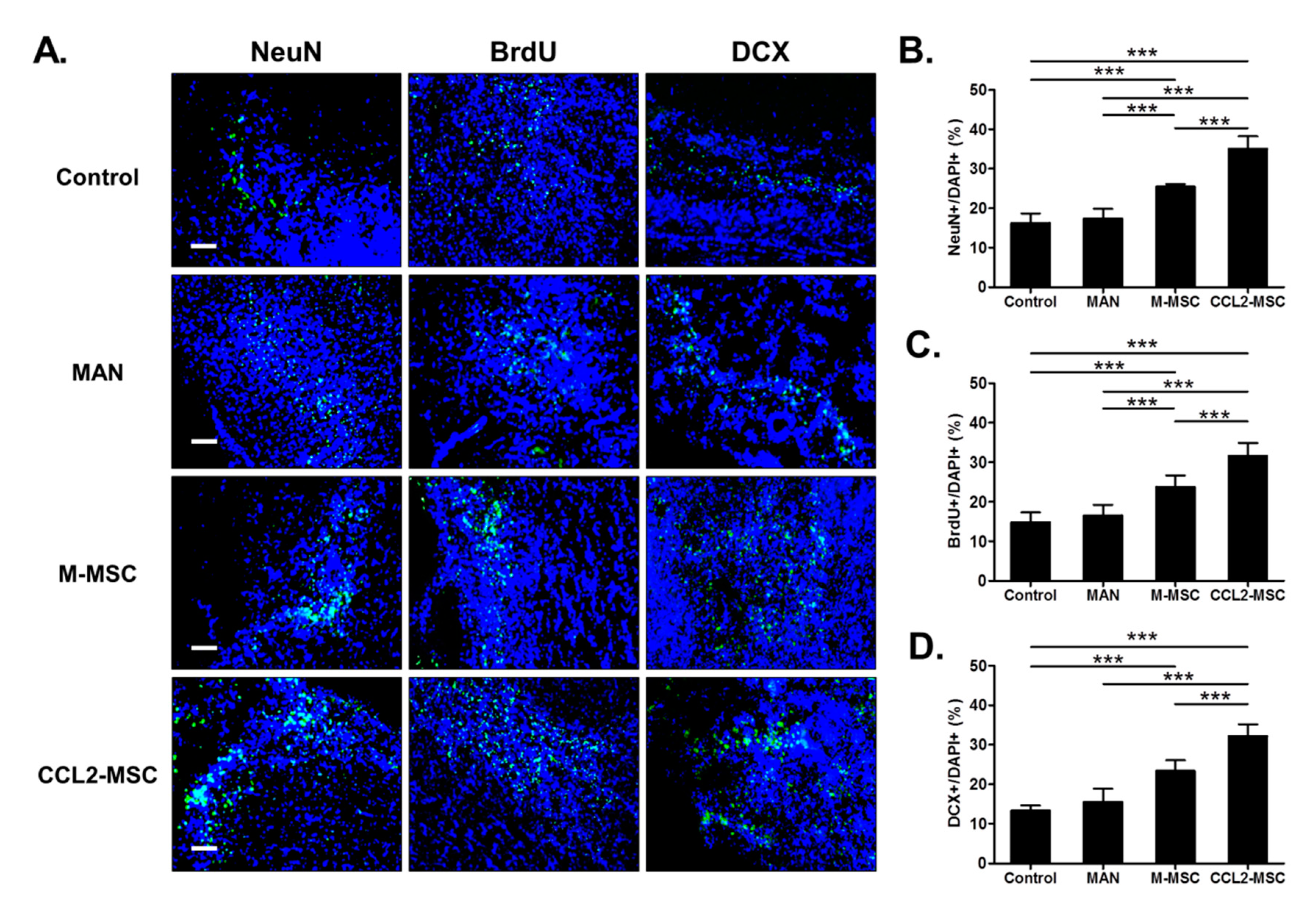
2.8. Transplantation of CCL2-Overexpressing hUC-MSCs Increases Endogenous Neurogenesis in the Peri-Infarct Area
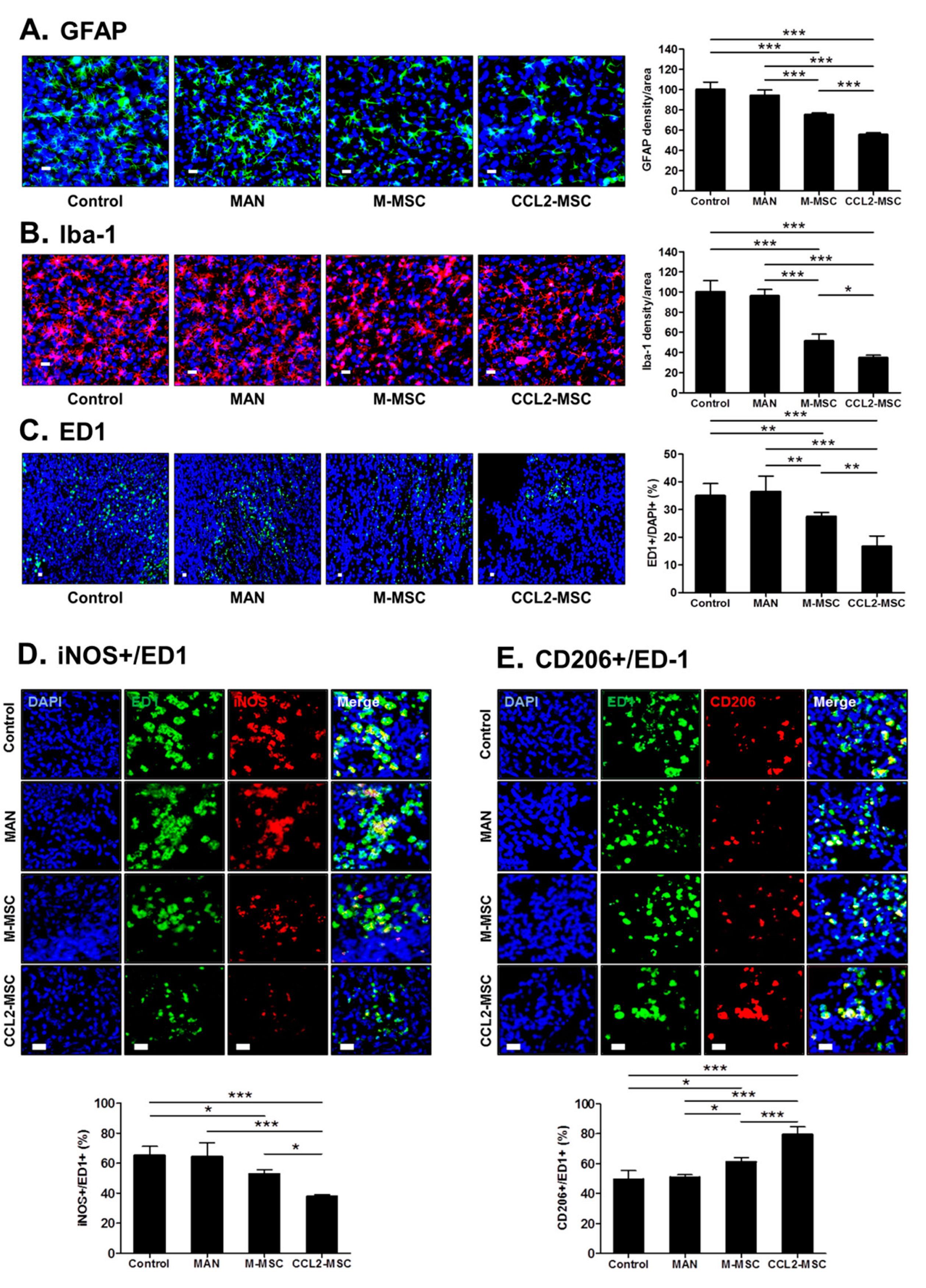
2.9. Transplantation of CCL2-Overexpressing hUC-MSCs Attenuates Inflammatory Reactions in the Peri-Infarct Area
3. Discussion
4. Materials and Methods
4.1. Ethics
4.2. Culture of hUC-MSCs
4.3. Manufacturing of CCL2-Overexpressing hUC-MSCs
4.4. Reverse Transcription Polymerase Chain Reaction (RT-PCR)
4.5. Western Blot
4.6. MCAo Stroke Model
4.7. Stem Cell Transplantation
4.8. Behavioral Tests
4.9. Measurement of Infarct Size
4.10. Immunohistochemistry
4.11. Quantitation of Vessel Density
4.12. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of variance |
| BBB | Blood–brain barrier |
| BrdU | Bromodeoxyuridine |
| CCA | Common carotid artery |
| CCL2 | C–C motif chemokine ligand 2 |
| CCL2-MSC | Mannitol+CCL2-overexpressing hUC-MSC-treatment group |
| CCR2 | C–C motif chemokine receptor 2 |
| DCX | Doublecortin |
| ECA | External carotid artery |
| GFAP | Glial fibrillary acidic protein |
| GFP | Green fluorescent protein |
| hUC-MSC | Human umbilical cord-derived mesenchymal stem cell |
| Iba-1 | Ionized calcium-binding adapter molecule 1 |
| IV | Intravenous |
| MAN | Mmannitol-treatment group |
| M-MSC | Mannitol + hUC-MSC-treatment group |
| MSC | Mesenchymal stem cell |
| MCAo | Middle cerebral artery occlusion |
| MEM | Minimum essential medium |
| mNSS | Modified neurological severity score |
| NeuN | Neuronal nuclei |
| PBS | Phosphate-buffered saline |
| PFA | Paraformaldehyde |
| r-tPA | Recombinant tissue plasminogen activator |
| RECA-1 | Rat endothelial cell antigen 1 |
| RT-PCR | Reverse transcription polymerase chain reaction |
| SEM | Standard error of the mean |
| TTC | 2:3:5-triphenyltetrazolium chloride |
| VEGF | Vascular endothelial growth factor |
References
- Simons, L.A.; McCallum, J.; Friedlander, Y.; Simons, J. Healthy ageing is associated with reduced and delayed disability. Age Ageing 2000, 29, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Kang, K.; Kang, J.; Koo, J.; Kim, D.H.; Kim, B.J.; Kim, W.J.; Kim, E.G.; Kim, J.G.; Kim, J.M.; et al. Executive summary of stroke statistics in Korea 2018: A report from the epidemiology research council of the Korean stroke society. J. Stroke 2019, 21, 42–59. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, A.; Choy, F.C.; Howell, S.; Hillier, S.; Chan, F.; Hamilton-Bruce, M.A.; Koblar, S.A. Safety and effectiveness of stem cell therapies in early-phase clinical trials in stroke: A systematic review and meta-analysis. Stem. Cell Res. Ther. 2017, 8, 191. [Google Scholar] [CrossRef] [PubMed]
- Rikhtegar, R.; Yousefi, M.; Dolati, S.; Kasmaei, H.D.; Charsouei, S.; Nouri, M.; Shakouri, S.K. Stem cell-based cell therapy for neuroprotection in stroke: A review. J Cell Biochem. 2019, 120, 8849–8862. [Google Scholar] [CrossRef]
- Tang, Y.H.; Ma, Y.Y.; Zhang, Z.J.; Wang, Y.T.; Yang, G.Y. Opportunities and challenges: Stem cell-based therapy for the treatment of ischemic stroke. CNS Neurosci. Ther. 2015, 21, 337–347. [Google Scholar] [CrossRef]
- Mohammadian, M.; Abasi, E.; Akbarzadeh, A. Mesenchymal stem cell-based gene therapy: A promising therapeutic strategy. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1206–1211. [Google Scholar] [CrossRef]
- Porada, C.D.; Almeida-Porada, G. Mesenchymal stem cells as therapeutics and vehicles for gene and drug delivery. Adv. Drug Deliv. Rev. 2010, 62, 1156–1166. [Google Scholar] [CrossRef]
- Vu, Q.; Xie, K.; Eckert, M.; Zhao, W.; Cramer, S.C. Meta-analysis of preclinical studies of mesenchymal stromal cells for ischemic stroke. Neurology 2014, 82, 1277–1286. [Google Scholar] [CrossRef]
- Wang, Q.; Duan, F.; Wang, M.X.; Wang, X.D.; Liu, P.; Ma, L.Z. Effect of stem cell-based therapy for ischemic stroke treatment: A meta-analysis. Clin. Neurol. Neurosurg. 2016, 146, 1–11. [Google Scholar] [CrossRef]
- Oggu, G.S.; Sasikumar, S.; Reddy, N.; Ella, K.K.R.; Rao, C.M.; Bokara, K.K. Gene delivery approaches for mesenchymal stem cell therapy: Strategies to increase efficiency and specificity. Stem. Cell Rev. Rep. 2017, 13, 725–740. [Google Scholar] [CrossRef]
- Andres, R.H.; Choi, R.; Pendharkar, A.V.; Gaeta, X.; Wang, N.; Nathan, J.K.; Chua, J.Y.; Lee, S.W.; Palmer, T.D.; Steinberg, G.K.; et al. The CCR2/CCL2 interaction mediates the transendothelial recruitment of intravascularly delivered neural stem cells to the ischemic brain. Stroke 2011, 42, 2923–2931. [Google Scholar] [CrossRef] [PubMed]
- Reaux-Le Goazigo, A.; Van Steenwinckel, J.; Rostene, W.; Melik Parsadaniantz, S. Current status of chemokines in the adult CNS. Prog. Neurobiol. 2013, 104, 67–92. [Google Scholar] [CrossRef] [PubMed]
- Nowakowski, A.; Andrzejewska, A.; Boltze, J.; Nitzsche, F.; Cui, L.L.; Jolkkonen, J.; Walczak, P.; Lukomska, B.; Janowski, M. Translation, but not transfection limits clinically relevant, exogenous mRNA based induction of alpha-4 integrin expression on human mesenchymal stem cells. Sci. Rep. 2017, 7, 1103. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.Y.; Liu, L.; Yang, Q.W. Functions and mechanisms of microglia/macrophages in neuroinflammation and neurogenesis after stroke. Prog. Neurobiol. 2016, 142, 23–44. [Google Scholar] [CrossRef]
- Ghazavi, H.; Hoseini, S.J.; Ebrahimzadeh-Bideskan, A.; Mashkani, B.; Mehri, S.; Ghorbani, A.; Sadri, K.; Mahdipour, E.; Ghasemi, F.; Forouzanfar, F.; et al. Fibroblast growth factor type 1 (FGF1)-overexpressed adipose-derived mesenchaymal stem cells (AD-MSC(FGF1)) induce neuroprotection and functional recovery in a rat stroke model. Stem. Cell Rev. Rep. 2017, 13, 670–685. [Google Scholar] [CrossRef]
- Kim, G.H.; Subash, M.; Yoon, J.S.; Jo, D.; Han, J.; Hong, J.M.; Kim, S.S.; Suh-Kim, H. Neurogenin-1 overexpression increases the therapeutic effects of mesenchymal stem cells through enhanced engraftment in an ischemic rat brain. Int. J. Stem. Cells 2020, 13, 127–141. [Google Scholar] [CrossRef]
- Conductier, G.; Blondeau, N.; Guyon, A.; Nahon, J.L.; Rovere, C. The role of monocyte chemoattractant protein MCP1/CCL2 in neuroinflammatory diseases. J. Neuroimmunol. 2010, 224, 93–100. [Google Scholar] [CrossRef]
- Che, X.; Ye, W.; Panga, L.; Wu, D.C.; Yang, G.Y. Monocyte chemoattractant protein-1 expressed in neurons and astrocytes during focal ischemia in mice. Brain Res. 2001, 902, 171–177. [Google Scholar] [CrossRef]
- Venkat, P.; Yan, T.; Chopp, M.; Zacharek, A.; Ning, R.; van Slyke, P.; Dumont, D.; Landschoot-Ward, J.; Liang, L.; Chen, J. Angiopoietin-1 mimetic peptide promotes neuroprotection after stroke in type 1 diabetic rats. Cell Transpl. 2018, 27, 1744–1752. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, J.; Cai, J.; Qiu, Y.; Zheng, H.; Lai, X.; Sui, X.; Wang, Y.; Lu, Q.; Zhang, Y.; et al. Targeted homing of CCR2-overexpressing mesenchymal stromal cells to ischemic brain enhances post-stroke recovery partially through PRDX4-mediated blood-brain barrier preservation. Theranostics 2018, 8, 5929–5944. [Google Scholar] [CrossRef]
- Landreneau, M.J.; Mullen, M.T.; Messe, S.R.; Cucchiara, B.; Sheth, K.N.; McCullough, L.D.; Kasner, S.E.; Sansing, L.H. Serum markers after spontaneous cerebral hemorrhage, i., ccl2 and cxcl10 are associated with poor outcome after intracerebral hemorrhage. Ann. Clin. Transl. Neurol. 2018, 5, 962–970. [Google Scholar]
- Schilling, M.; Strecker, J.K.; Schabitz, W.R.; Ringelstein, E.B.; Kiefer, R. Effects of monocyte chemoattractant protein 1 on blood-borne cell recruitment after transient focal cerebral ischemia in mice. Neuroscience 2009, 161, 806–812. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Hallenbeck, J.M.; Ruetzler, C.; Bol, D.; Thomas, K.; Berman, N.E.; Vogel, S.N. Overexpression of monocyte chemoattractant protein 1 in the brain exacerbates ischemic brain injury and is associated with recruitment of inflammatory cells. J. Cereb. Blood Flow. Metab. 2003, 23, 748–755. [Google Scholar] [CrossRef]
- Fang, W.; Zhai, X.; Han, D.; Xiong, X.; Wang, T.; Zeng, X.; He, S.; Liu, R.; Miyata, M.; Xu, B.; et al. CCR2-dependent monocytes/macrophages exacerbate acute brain injury but promote functional recovery after ischemic stroke in mice. Theranostics 2018, 8, 3530–3543. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, Y.; Chen, J.; Gautam, S.C.; Zhang, Z.; Lu, M.; Chopp, M. Ischemic cerebral tissue and MCP-1 enhance rat bone marrow stromal cell migration in interface culture. Exp. Hematol. 2002, 30, 831–836. [Google Scholar] [CrossRef]
- Yang, H.C.; Zhang, M.; Wu, R.; Zheng, H.Q.; Zhang, L.Y.; Luo, J.; Li, L.L.; Hu, X.Q. C-C chemokine receptor type 2-overexpressing exosomes alleviated experimental post-stroke cognitive impairment by enhancing microglia/macrophage M2 polarization. World J. Stem. Cells 2020, 12, 152–167. [Google Scholar] [CrossRef]
- Jiang, L.; Newman, M.; Saporta, S.; Chen, N.; Sanberg, C.; Sanberg, P.R.; Willing, A.E. MIP-1alpha and mcp-1 induce migration of human umbilical cord blood cells in models of stroke. Curr. Neurovasc. Res. 2008, 5, 118–124. [Google Scholar] [CrossRef][Green Version]
- Lee, S.H.; Jin, K.S.; Bang, O.Y.; Kim, B.J.; Park, S.J.; Lee, N.H.; Yoo, K.H.; Koo, H.H.; Sung, K.W. Differential migration of mesenchymal stem cells to ischemic regions after middle cerebral artery occlusion in rats. PLoS ONE 2015, 10, e0134920. [Google Scholar] [CrossRef]
- Wang, L.; Li, Y.; Chen, X.; Chen, J.; Gautam, S.C.; Xu, Y.; Chopp, M. MCP-1, MIP-1, IL-8 and ischemic cerebral tissue enhance human bone marrow stromal cell migration in interface culture. Hematology 2002, 7, 113–117. [Google Scholar] [CrossRef]
- Lawrence, D.M.; Seth, P.; Durham, L.; Diaz, F.; Boursiquot, R.; Ransohoff, R.M.; Major, E.O. Astrocyte differentiation selectively upregulates CCL2/monocyte chemoattractant protein-1 in cultured human brain-derived progenitor cells. Glia 2006, 53, 81–91. [Google Scholar] [CrossRef]
- Inose, Y.; Kato, Y.; Kitagawa, K.; Uchiyama, S.; Shibata, N. Activated microglia in ischemic stroke penumbra upregulate MCP-1 and CCR2 expression in response to lysophosphatidylcholine derived from adjacent neurons and astrocytes. Neuropathology 2015, 35, 209–223. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.F.; Liu, T.T.; Hu, C.H.; Chen, C.C.; Wang, J.Y. Expressions of chemokines and their receptors in the brain after heat stroke-induced cortical damage. J. Neuroimmunol. 2018, 318, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Leong, W.K.; Lewis, M.D.; Koblar, S.A. Concise review: Preclinical studies on human cell-based therapy in rodent ischemic stroke models: Where are we now after a decade? Stem Cells 2013, 31, 1040–1043. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, Y.; Murakami, M.; Kawamura, R.; Ishizaka, R.; Fukuta, O.; Nakashima, M. CXCL14 and MCP1 are potent trophic factors associated with cell migration and angiogenesis leading to higher regenerative potential of dental pulp side population cells. Stem Cell Res. Ther. 2015, 6, 111. [Google Scholar] [CrossRef]
- Hong, K.H.; Ryu, J.; Han, K.H. Monocyte chemoattractant protein-1-induced angiogenesis is mediated by vascular endothelial growth factor-A. Blood 2005, 105, 1405–1407. [Google Scholar] [CrossRef]
- Arderiu, G.; Espinosa, S.; Pena, E.; Aledo, R.; Badimon, L. Monocyte-secreted Wnt5a interacts with FZD5 in microvascular endothelial cells and induces angiogenesis through tissue factor signaling. J. Mol. Cell Biol. 2014, 6, 380–393. [Google Scholar] [CrossRef]
- Stowe, A.M.; Wacker, B.K.; Cravens, P.D.; Perfater, J.L.; Li, M.K.; Hu, R.; Freie, A.B.; Stuve, O.; Gidday, J.M. CCL2 upregulation triggers hypoxic preconditioning-induced protection from stroke. J. Neuroinflamm. 2012, 9, 33. [Google Scholar] [CrossRef]
- Eugenin, E.A.; D’Aversa, T.G.; Lopez, L.; Calderon, T.M.; Berman, J.W. MCP-1 (CCL2) protects human neurons and astrocytes from NMDA or HIV-tat-induced apoptosis. J. Neurochem. 2003, 85, 1299–1311. [Google Scholar] [CrossRef]
- Madrigal, J.L.; Leza, J.C.; Polak, P.; Kalinin, S.; Feinstein, D.L. Astrocyte-derived MCP-1 mediates neuroprotective effects of noradrenaline. J. Neurosci. 2009, 29, 263–267. [Google Scholar] [CrossRef]
- Wacker, B.K.; Perfater, J.L.; Gidday, J.M. Hypoxic preconditioning induces stroke tolerance in mice via a cascading HIF, sphingosine kinase, and CCL2 signaling pathway. J. Neurochem. 2012, 123, 954–962. [Google Scholar] [CrossRef]
- Liu, X.S.; Zhang, Z.G.; Zhang, R.L.; Gregg, S.R.; Wang, L.; Yier, T.; Chopp, M. Chemokine ligand 2 (CCL2) induces migration and differentiation of subventricular zone cells after stroke. J. Neurosci. Res. 2007, 85, 2120–2125. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.P.; Sailor, K.A.; Lang, B.T.; Park, S.W.; Vemuganti, R.; Dempsey, R.J. Monocyte chemoattractant protein-1 plays a critical role in neuroblast migration after focal cerebral ischemia. J. Cereb. Blood Flow Metab. 2007, 27, 1213–1224. [Google Scholar] [CrossRef]
- He, S.; Liu, R.; Li, B.; Huang, L.; Fan, W.; Tembachako, C.R.; Zheng, X.; Xiong, X.; Miyata, M.; Xu, B.; et al. Propagermanium, a CCR2 inhibitor, attenuates cerebral ischemia/reperfusion injury through inhibiting inflammatory response induced by microglia. Neurochem. Int. 2019, 125, 99–110. [Google Scholar] [CrossRef]
- Cho, D.I.; Kim, M.R.; Jeong, H.Y.; Jeong, H.C.; Jeong, M.H.; Yoon, S.H.; Kim, Y.S.; Ahn, Y. Mesenchymal stem cells reciprocally regulate the M1/M2 balance in mouse bone marrow-derived macrophages. Exp. Mol. Med. 2014, 46, e70. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Deng, Z.; Zhang, J.; Yang, C.; Liu, J.; Han, W.; Ye, P.; Si, Y.; Chen, G. Mesenchymal stem cells promote type 2 macrophage polarization to ameliorate the myocardial injury caused by diabetic cardiomyopathy. J. Transl. Med. 2019, 17, 251. [Google Scholar] [CrossRef] [PubMed]
- Wattananit, S.; Tornero, D.; Graubardt, N.; Memanishvili, T.; Monni, E.; Tatarishvili, J.; Miskinyte, G.; Ge, R.; Ahlenius, H.; Lindvall, O.; et al. Monocyte-derived macrophages contribute to spontaneous long-term functional recovery after stroke in mice. J. Neurosci. 2016, 36, 4182–4195. [Google Scholar] [CrossRef]
- Choi, C.; Kim, H.M.; Shon, J.; Park, J.; Kim, H.T.; Kang, S.H.; Oh, S.H.; Kim, N.K.; Kim, O.J. The combination of mannitol and temozolomide increases the effectiveness of stem cell treatment in a chronic stroke model. Cytotherapy 2018, 20, 820–829. [Google Scholar] [CrossRef]
- Choi, C.; Oh, S.H.; Noh, J.E.; Jeong, Y.W.; Kim, S.; Ko, J.J.; Kim, O.J.; Song, J. Attenuation of postischemic genomic alteration by mesenchymal stem cells: A microarray study. Mol. Cells 2016, 39, 337–344. [Google Scholar]
- Kim, S.M.; Moon, S.H.; Lee, Y.; Kim, G.J.; Chung, H.M.; Choi, Y.S. Alternative xeno-free biomaterials derived from human umbilical cord for the self-renewal ex-vivo expansion of mesenchymal stem cells. Stem. Cells Dev. 2013, 22, 3025–3038. [Google Scholar] [CrossRef]
- Tajiri, N.; Lee, J.Y.; Acosta, S.; Sanberg, P.R.; Borlongan, C.V. Breaking the blood-brain barrier with mannitol to aid stem cell therapeutics in the chronic stroke brain. Cell Transpl. 2016, 25, 1453–1460. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.; Kim, O.J.; Lee, K.O.; Jung, H.; Oh, S.-H.; Kim, N.K. Enhancing the Therapeutic Potential of CCL2-Overexpressing Mesenchymal Stem Cells in Acute Stroke. Int. J. Mol. Sci. 2020, 21, 7795. https://doi.org/10.3390/ijms21207795
Lee S, Kim OJ, Lee KO, Jung H, Oh S-H, Kim NK. Enhancing the Therapeutic Potential of CCL2-Overexpressing Mesenchymal Stem Cells in Acute Stroke. International Journal of Molecular Sciences. 2020; 21(20):7795. https://doi.org/10.3390/ijms21207795
Chicago/Turabian StyleLee, Sanghun, Ok Joon Kim, Kee Ook Lee, Hyeju Jung, Seung-Hun Oh, and Nam Keun Kim. 2020. "Enhancing the Therapeutic Potential of CCL2-Overexpressing Mesenchymal Stem Cells in Acute Stroke" International Journal of Molecular Sciences 21, no. 20: 7795. https://doi.org/10.3390/ijms21207795
APA StyleLee, S., Kim, O. J., Lee, K. O., Jung, H., Oh, S.-H., & Kim, N. K. (2020). Enhancing the Therapeutic Potential of CCL2-Overexpressing Mesenchymal Stem Cells in Acute Stroke. International Journal of Molecular Sciences, 21(20), 7795. https://doi.org/10.3390/ijms21207795




