Foliar Application of Chitosan Increases Tomato Growth and Influences Mycorrhization and Expression of Endochitinase-Encoding Genes
Abstract
1. Introduction
2. Results
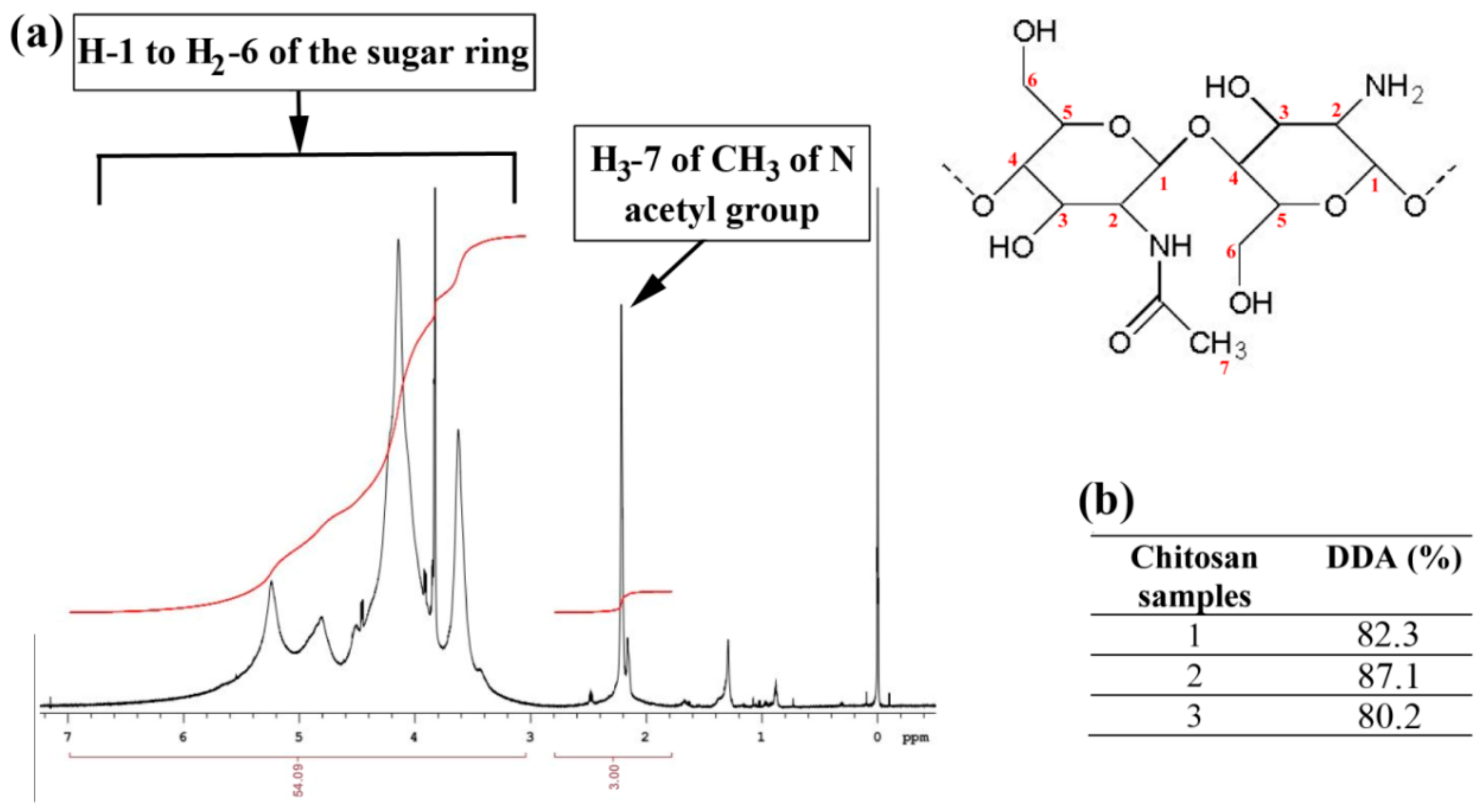
2.1. Degree of Deacetylation (DDA) of Ch and Morphology of NPs
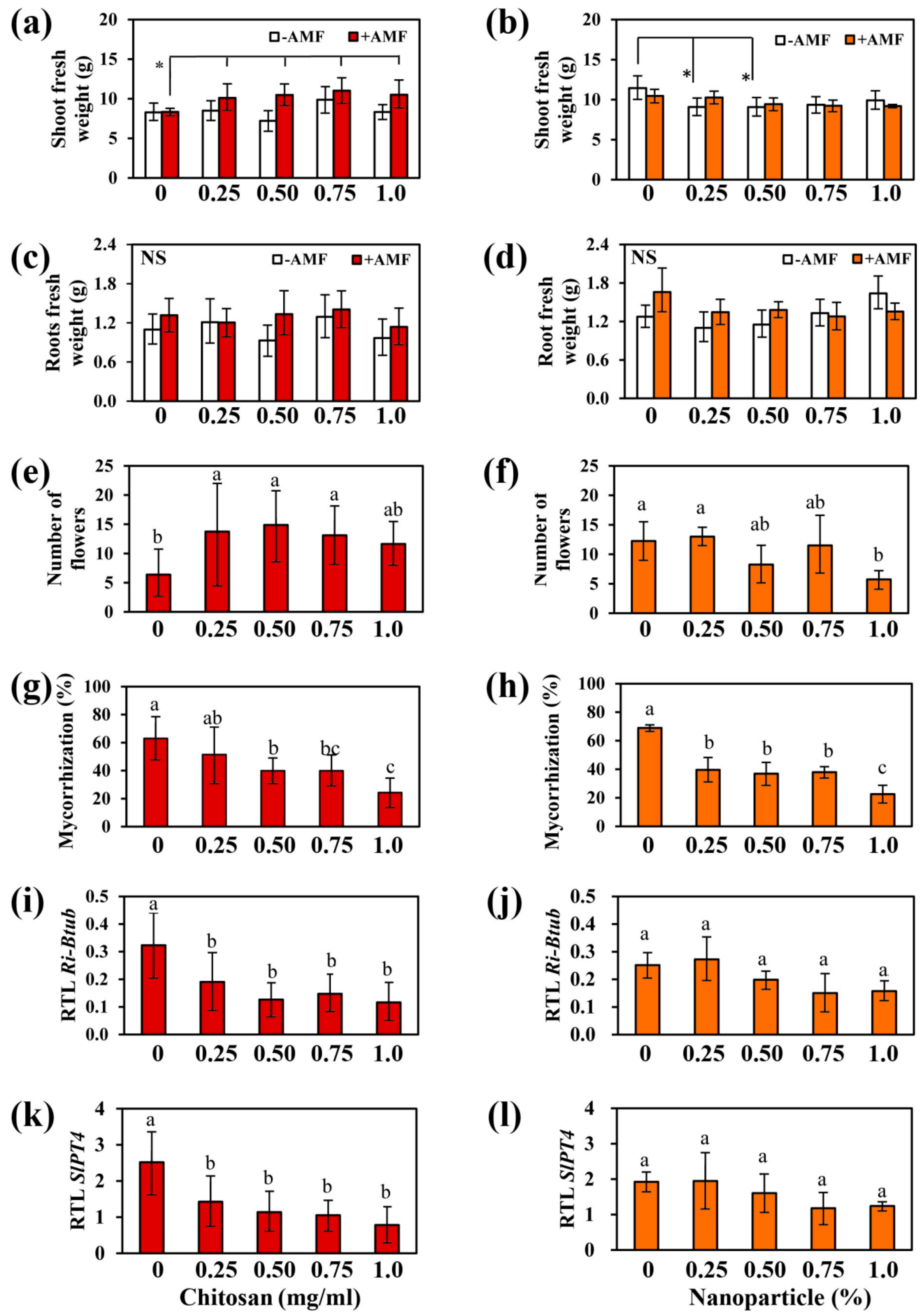
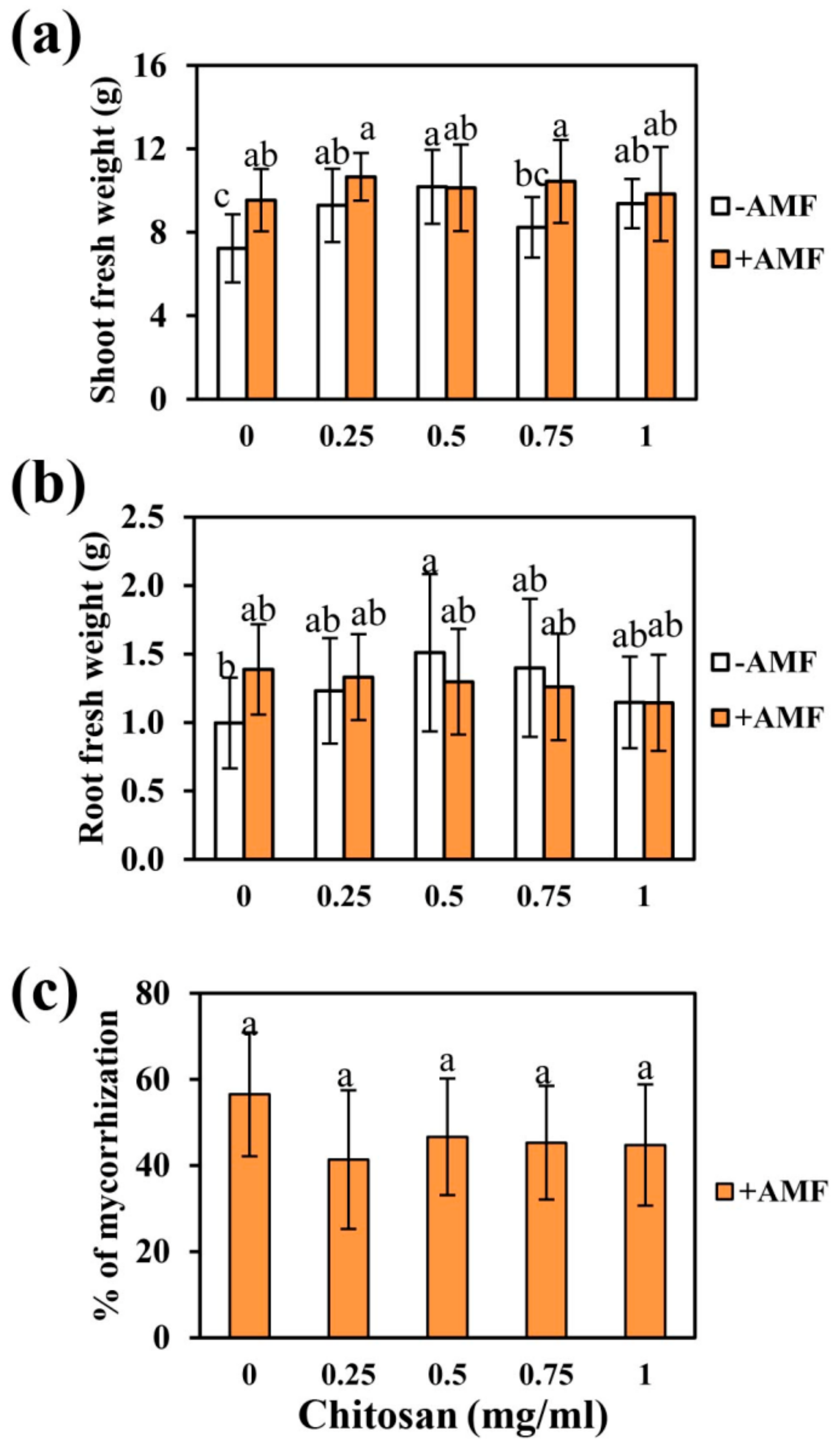
2.2. Chitosan and Nanoparticles Effects on Tomato Growth and Mycorrhization
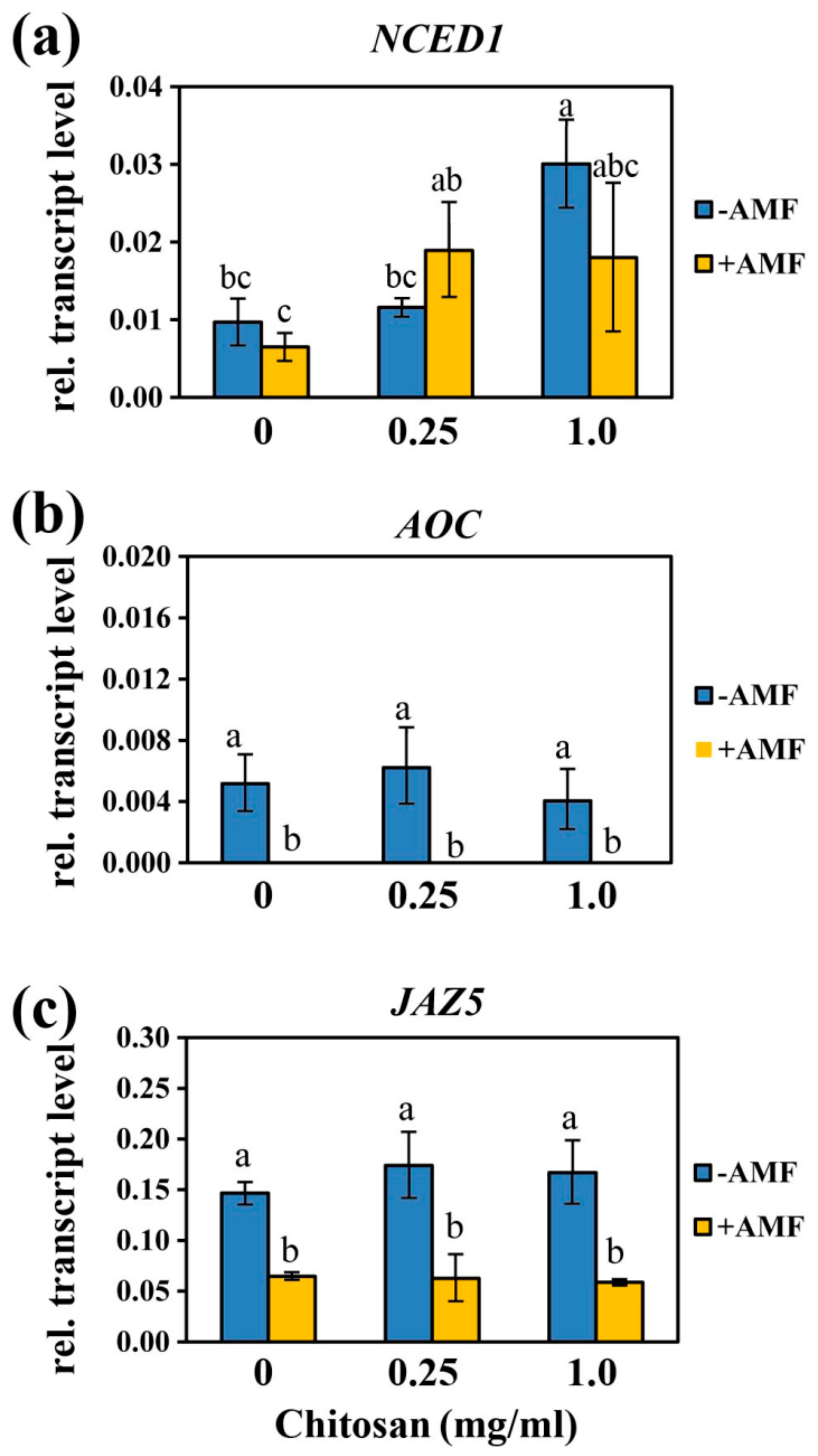
2.3. Levels of ABA and Jasmonates, and Transcript Accumulation of ABA and JA-Related Genes During Mycorrhizal Symbiosis and Foliar Application of Ch
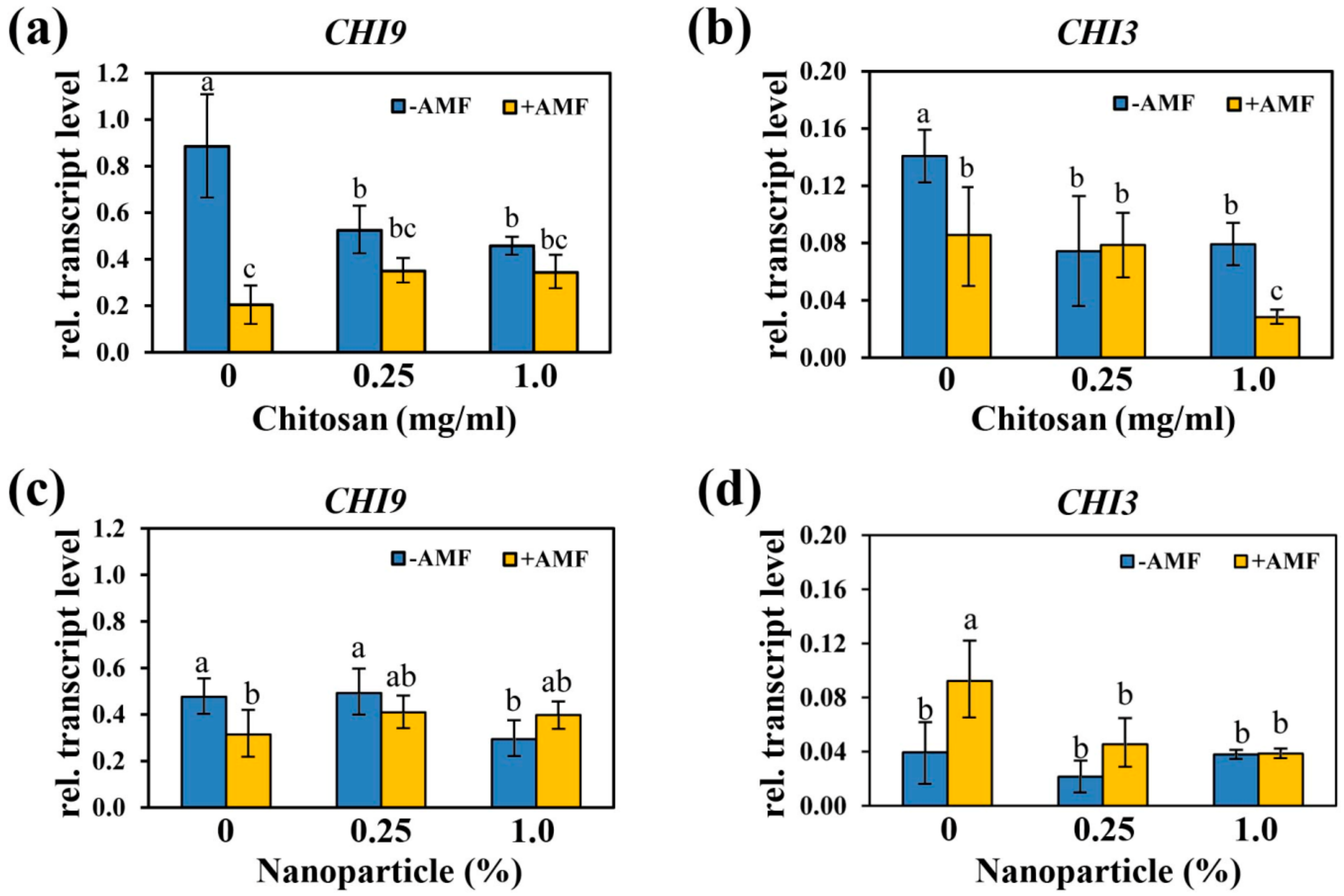
2.4. Transcript Levels of Chitinase-Encoding Genes Changed Concomitantly to the Alteration of Mycorrhization
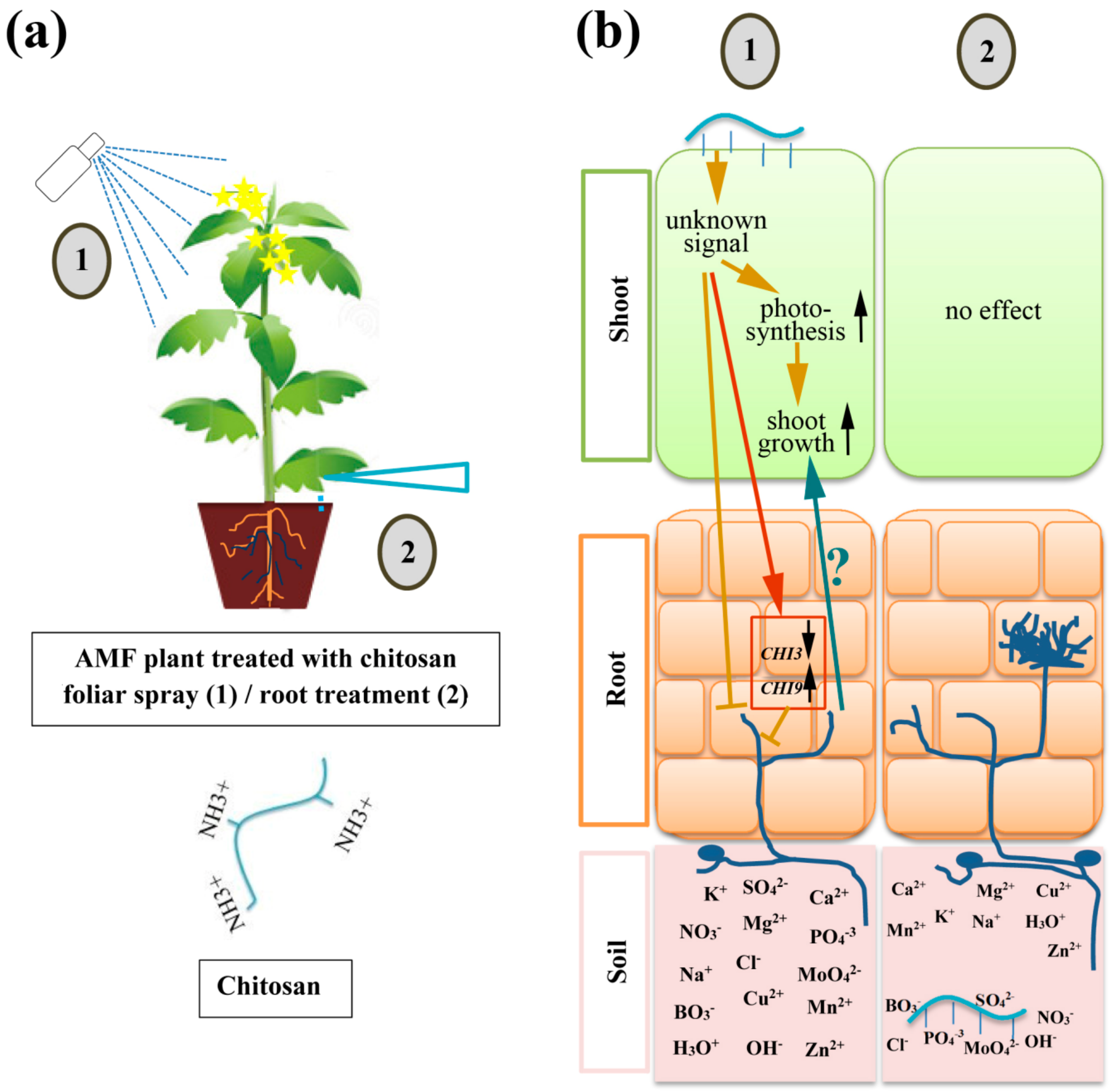
3. Discussion
4. Materials and Methods
4.1. Chitosan and Nanoparticles Preparation
4.2. Determination of Degree of Deacetylation of Chitosan by 1HNMR Spectroscopy
4.3. Analysis of Surface Morphology of NPs by Scanning Electron Microscopy (SEM)
4.4. Plant Materials and Treatments
4.5. Sugar Measurement
4.6. Phytohormone Measurement
4.7. Determination of Mycorrhization Rate by Staining
4.8. RNA Extraction, cDNA Preparation and Real Time qPCR (RT-qPCR) Reaction
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ABA | Abscisic Acid |
| AMF | Arbuscular mycorrhizal fungi |
| AOC | Allene oxide cyclase |
| Ch | Chitosan |
| Chi | Chitinase |
| DDA | Degree of deacetylation |
| EF | Elongation factor |
| JA | Jasmonic acid |
| JA-Ile | Jasmonoyl-isoleucine |
| JAZ | Jasmonate-zim-domain protein |
| GlcNAc | N-acetylglucosamine |
| NCED1 | 9-cis-epoxycarotenoid dioxygenase |
| NPs | Nanoparticles of chitosan |
| OPDA | 12-oxo-phytodienoic acid |
| SEM | Scanning electron microscopy |
| TPP | Sodium tripolyphosphate |
References
- Aktar, M.W.; Sengupta, D.; Chowdhury, A. Impact of pesticides use in agriculture: their benefits and hazards. Interdisc. Toxicol. 2009, 2, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Chun, S.C.; Chandrasekaran, M. Chitosan and chitosan nanoparticles induced expression of pathogenesis related proteins genes enhances biotic stress tolerance in tomato. Int. J. Biol. Macromol. 2019, 125, 948–954. [Google Scholar] [CrossRef] [PubMed]
- Dutta, K.P.; Dutta, J.; Tripathi, V.S. Chitin and chitosan: Chemistry, properties and applications. J. Sci. Ind. Res. 2004, 63, 20–31. [Google Scholar]
- Rinaudo, M. Chitin and chitosan: properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Aider, M. Chitosan application for active bio-based films production and potential in the food industry. LWT-Food Sci. Technol. 2010, 43, 837–842. [Google Scholar] [CrossRef]
- Dahmane, E.M.; Rhazi, M.; Taourirte, M. Chitosan Nanoparticles as a New Delivery System for the Anti-HIV Drug Zidovudine. Bull. Korean Chem. Soc. 2013, 34, 1333–1338. [Google Scholar] [CrossRef]
- Ma, Z.; Garrido-Maestu, A.; Jeong, K.C. Application, mode of action, and in vivo activity of chitosan and its micro- and nanoparticles as antimicrobial agents: A review. Carbohydr. Polym. 2017, 176, 257–265. [Google Scholar] [CrossRef]
- Mohammed, M.A.; Syeda, J.T.M.; Wasan, K.M.; Wasan, E.K. An overview of chitosan nanoparticles and its application in non-parenteral drug delivery. Pharmaceutics. 2017, 9, 53. [Google Scholar] [CrossRef]
- Benhamou, N.; Thériault, G. Treatment with chitosan enhances resistance of tomato plants to the crown and root rot pathogen Fusarium oxysporum f. sp. radicis-lycopersici. Physiol. Mol. Plant Pathol. 1992, 41, 33–52. [Google Scholar] [CrossRef]
- Sathiyabama, M.; Akila, G.; Einstein Charles, R. Chitosan-induced defence responses in tomato plants against early blight disease caused by Alternaria solani (Ellis and Martin) Sorauer. Arch. Phytopathol. Plant Protect. 2014, 47, 1777–1787. [Google Scholar] [CrossRef]
- Gryndler, M.; Jansa, J.; Hršelová, H.; Chvátalová, I.; Vosátka, M. Chitin stimulates development and sporulation of arbuscular mycorrhizal fungi. Appl. Soil Ecol. 2003, 22, 283–287. [Google Scholar] [CrossRef]
- Agbodjato, N.A.; Noumavo, P.A.; Adjanohoun, A.; Agbessi, L.; Baba-Moussa, L. Synergistic effects of plant growth promoting rhizobacteria and chitosan on In vitro seeds germination, greenhouse growth, and nutrient uptake of maize (Zea mays L.). Biotechnol. Res. Int. 2016, 11. [Google Scholar]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.Academic Press: London, UK, 2008. [Google Scholar]
- Bowles, T.M.; Barrios-Masias, F.H.; Carlisle, E.A.; Cavagnaro, T.R.; Jackson, L.E. Effects of arbuscular mycorrhizae on tomato yield, nutrient uptake, water relations, and soil carbon dynamics under deficit irrigation in field conditions. J. Sci. Total Environ. 2016, 178, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Baum, C.; El-Tohamy, W.; Gruda, N. Increasing the productivity and product quality of vegetable crops using arbuscular mycorrhizal fungi: a review. Sci. Hortic. 2015, 187, 131–141. [Google Scholar] [CrossRef]
- Bucher, M.; Hause, B.; Krajinski, F.; Küster, H. Through the doors of perception to function in arbuscular mycorrhizal symbioses. New Phytol. 2014, 204, 833–840. [Google Scholar] [CrossRef]
- Liao, D.; Wang, S.; Cui, M.; Liu, J.; Chen, A.; Xu, G. Phytohormones regulate the development of arbuscular mycorrhizal symbiosis. Int. J. Mol. Sci. 2018, 19, 3146. [Google Scholar] [CrossRef]
- Charpentier, M.; Sun, J.; Wen, J.; Mysore, K.S.; Oldroyd, G.E. Abscisic acid promotion of arbuscular mycorrhizal colonization requires a component of the PROTEIN PHOSPHATASE 2A complex. Plant Physiol. 2014, 166, 2077–2090. [Google Scholar] [CrossRef]
- Gutjahr, C.; Paszkowski, U. Weights in the balance: jasmonic acid and salicylic acid signaling in root-biotroph interactions. Mol. Plant-Microbe Interact. 2009, 22, 763–772. [Google Scholar] [CrossRef]
- Sahai, A.S.; Manocha, M.S. Chitinases of fungi and plants: their involvement in morphogenesis and host—parasite interaction. FEMS Microbiol. Rev. 1993, 11, 317–338. [Google Scholar] [CrossRef]
- Salzer, P.; Bonanomi, A.; Beyer, K.; Vögeli-Lange, R.; Aeschbacher, R.A.; Lange, J.; Wiemken, A.; Kim, D.; Cook, D.R.; Boller, T. Differential expression of eight chitinase genes in Medicago truncatula roots during mycorrhiza formation, nodulation, and pathogen infection. Mol. Plant-Microbe Interact. 2000, 13, 763–777. [Google Scholar] [CrossRef]
- Cheng, C.M.; Palloix, A.; Lefebvre, V. Isolation, mapping and characterization of allelic polymorphism of Chi3-P1, a class III chitinase of Capsicum annuum L. Plant Sci. 2002, 163, 481–489. [Google Scholar] [CrossRef]
- Whiteside, M.D.; Digman, M.A.; Gratton, E.; Treseder, K.K. Organic nitrogen uptake by arbuscular mycorrhizal fungi in a boreal forest. Soil Biol. Biochem. 2012, 55, 7–13. [Google Scholar] [CrossRef]
- El Amerany, F.; Rhazi, M.; Wahbi, S.; Taourirte, M.; Meddich, A. The effect of chitosan, arbuscular mycorrhizal fungi, and compost applied individually or in combination on growth, nutrient uptake, and stem anatomy of tomato. Sci. Hortic. 2020, 261, 109015. [Google Scholar] [CrossRef]
- Isayenkov, S.; Fester, T.; Hause, B. Rapid determination of fungal colonization and arbuscule formation in roots of Medicago truncatula using real-time (RT) PCR. J. Plant Physiol. 2004, 161, 1379–1383. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.-H.; Chague, V.; Melamed-Bessudo, C.; Kapulnik, Y.; Jain, A.; Raghothama, K.G.; Levy, A.A.; Silber, A. Functional characterization of LePT4: a phosphate transporter in tomato with mycorrhiza-enhanced expression. J. Exp. Bot. 2007, 58, 2491–2501. [Google Scholar] [CrossRef] [PubMed]
- Horváth, E.; Csiszár, J.; Gallé, Á.; Poór, P.; Szepesi, Á.; Tari, I. Hardening with salicylic acid induces concentration-dependent changes in abscisic acid biosynthesis of tomato under salt stress. J. Plant Physiol. 2015, 183, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Chini, A.; Ben-Romdhane, W.; Hassairi, A.; Aboul-Soud, M.A. Identification of TIFY/JAZ family genes in Solanum lycopersicum and their regulation in response to abiotic stresses. PloS ONE 2017, 12, e0177381. [Google Scholar] [CrossRef]
- Hause, B.; Stenzel, I.; Miersch, O.; Maucher, H.; Kramell, R.; Ziegler, J.; Wasternack, C. Tissue-specific oxylipin signature of tomato flowers: allene oxide cyclase is highly expressed in distinct flower organs and vascular bundles. Plant J. 2000, 24, 113–126. [Google Scholar] [CrossRef]
- Hause, B.; Schaarschmidt, S. The role of jasmonates in mutualistic symbioses between plants and soil-born microorganisms. Phytochemistry 2009, 70, 1589–1599. [Google Scholar] [CrossRef]
- Fracetto, G.G.; Peres, L.E.; Mehdy, M.C.; Lambais, M.R. Tomato ethylene mutants exhibit differences in arbuscular mycorrhiza development and levels of plant defense-related transcripts. Symbiosis 2013, 60, 155–167. [Google Scholar] [CrossRef]
- Malerba, M.; Cerana, R. Recent applications of chitin-and chitosan-based polymers in plants. Polymers 2019, 11, 839. [Google Scholar] [CrossRef] [PubMed]
- Petutschnig, E.K.; Jones, A.M.; Serazetdinova, L.; Lipka, U.; Lipka, V. The LysM-RLK CERK1 is a major chitin binding protein in Arabidopsis thaliana and subject to chitin-induced phosphorylation. J. Biol. Chem. 2010, jbc-M110. [Google Scholar] [CrossRef]
- Gubaeva, E.; Gubaev, A.; Melcher, R.L.; Cord-Landwehr, S.; Singh, R.; El Gueddari, N.E.; Moerschbacher, B.M. ‘Slipped sandwich’ model for chitin and chitosan perception in Arabidopsis. Mol. Plant-Microbe Interact. 2018, 31, 1145–1153. [Google Scholar] [CrossRef] [PubMed]
- Sharp, R.G. A review of the applications of chitin and its derivatives in agriculture to modify plant-microbial interactions and improve crop yields. Agronomy 2013, 3, 757–793. [Google Scholar] [CrossRef]
- Hidangmayum, A.; Dwivedi, P.; Katiyar, D.; Hemantaranjan, A. Application of chitosan on plant responses with special reference to abiotic stress. Physiol. Mol. Biol. Plants. 2019, 25, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Rendina, N.; Nuzzaci, M.; Scopa, A.; Cuypers, A.; Sofo, A. Chitosan-elicited defense responses in Cucumber mosaic virus (CMV)-infected tomato plants. J. Plant Physiol. 2019, 234, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Pivato, B.; Offre, P.; Marchelli, S.; Barbonaglia, B.; Mougel, C.; Lemanceau, P.; Berta, G. Bacterial effects on arbuscular mycorrhizal fungi and mycorrhiza development as influenced by the bacteria, fungi, and host plant. Mycorrhiza 2009, 19, 81–90. [Google Scholar] [CrossRef]
- Beltrano, J.; Ruscitti, M.; Arango, M.C.; Ronco, M. Effects of arbuscular mycorrhiza inoculation on plant growth, biological and physiological parameters and mineral nutrition in pepper grown under different salinity and p levels. J. Soil Sci. Plant Nutr. 2013, 13, 123–141. [Google Scholar] [CrossRef]
- Faessel, L.; Nassr, N.; Lebeau, T.; Walter, B. Chemically-induced resistance on soybean inhibits nodulation and mycorrhization. Plant Soil. 2010, 329, 259–268. [Google Scholar] [CrossRef]
- Herrera-Medina, M.J.; Steinkellner, S.; Vierheilig, H.; Ocampo Bote, J.A.; García Garrido, J.M. Abscisic acid determines arbuscule development and functionality in the tomato arbuscular mycorrhiza. New Phytol. 2007, 175, 554–564. [Google Scholar] [CrossRef]
- Wasternack, C.; Hause, B. Jasmonates: biosynthesis, perception, signal transduction and action in plant stress response, growth and development. An update to the 2007 review in Annals of Botany. Ann. Bot. 2013, 111, 1021–1058. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Romera, B.; Ruiz-Lozano, J.M.; Zamarreño, Á.M.; García-Mina, J.M.; Aroca, R. Arbuscular mycorrhizal symbiosis and methyl jasmonate avoid the inhibition of root hydraulic conductivity caused by drought. Mycorrhiza 2016, 26, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Song, G.; Dong, A.; Kong, X. Regulatory mechanisms of host plant defense responses to arbuscular mycorrhiza. Acta Ecol. Sin. 2011, 31, 322–327. [Google Scholar] [CrossRef]
- García-Garrido, J.; Ocampo, J. Regulation of the plant defence response in arbuscular mycorrhizal symbiosis. J. Exp. Bot. 2002, 53, 1377–1386. [Google Scholar] [CrossRef]
- Dumas-Gaudot, E.; Furlan, V.; Grenier, J.; Asselin, A. New acidic chitinase isoforms induced in tobacco roots by vesicular-arbuscular mycorrhizal fungi. Mycorrhiza 1992, 1, 133–136. [Google Scholar] [CrossRef]
- Grover, A. Plant chitinases: genetic diversity and physiological roles. Crit. Rev. Plant Sci. 2012, 31, 57–73. [Google Scholar] [CrossRef]
- Rathore, A.S.; Gupta, R.D. Chitinases from bacteria to human: properties, applications, and future perspectives. Enzyme Res. 2015, 2015. [Google Scholar] [CrossRef]
- Samac, D.A.; Shah, D.M. Developmental and pathogen-induced activation of the Arabidopsis acidic chitinase promoter. Plant Cell. 1991, 3, 1063–1072. [Google Scholar] [CrossRef][Green Version]
- Lambais, M.R.; Mehdy, M.C. Suppression of endochitinase, beta-1, 3-endoglucanase, and chalcone isomerase expression in bean vesicular arbuscular mycorrhizal roots under different soil phosphate conditions. Mol. Plant Microbe Interact. 1993, 6, 75–83. [Google Scholar] [CrossRef]
- David, R.; Itzhaki, H.; Ginzberg, I.; Gafni, Y.; Galili, G.; Kapulnik, Y. Suppression of tobacco basic chitinase gene expression in response to colonization by the arbuscular mycorrhizal fungus Glomus intraradices. Plant Microbe Interact. 1998, 11, 489–497. [Google Scholar] [CrossRef]
- Abdallah, R.A.B.; Stedel, C.; Garagounis, C.; Nefzi, A.; Jabnoun-Khiareddine, H.; Papadopoulou, K.K.; Daami-Remadi, M. Involvement of lipopeptide antibiotics and chitinase genes and induction of host defense in suppression of Fusarium wilt by endophytic Bacillus spp. in tomato. Crop Prot. 2017, 99, 45–58. [Google Scholar] [CrossRef]
- El Hadrami, A.; Adam, L.R.; El Hadrami, I.; Daayf, F. Chitosan in plant protection. Mar. Drugs. 2010, 8, 968–987. [Google Scholar] [CrossRef] [PubMed]
- Esaka, M.; Teramoto, T. cDNA cloning, gene expression and secretion of chitinase in winged bean. Plant Cell Physiol. 1998, 39, 349–356. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Takenaka, Y.; Nakano, S.; Tamoi, M.; Sakuda, S.; Fukamizo, T. Chitinase gene expression in response to environmental stresses in Arabidopsis thaliana: chitinase inhibitor allosamidin enhances stress tolerance. Biosci. Biotechnol. Biochem. 2009, 73, 1066–1071. [Google Scholar] [CrossRef]
- Al Sagheer, F.A.; Al-Sughayer, M.A.; Muslim, S.; Elsabee, M.Z. Extraction and characterization of chitin and chitosan from marin sources in Arabian Gulf. J. Carbohyd. Polym. 2009, 77, 410–419. [Google Scholar] [CrossRef]
- Landgraf, R.; Schaarschmidt, S.; Hause, B. Repeated leaf wounding alters the colonization of Medicago truncatula roots by beneficial and pathogenic microorganisms. Plant Cell Environ. 2012, 35, 1344–1357. [Google Scholar] [CrossRef]
- Schaarschmidt, S.; Qu, N.; Strack, D.; Sonnewald, U.; Hause, B. Local induction of the alc gene switch in transgenic tobacco plants by acetaldehyde. Plant Cell Physiol. 2004, 45, 1566–1577. [Google Scholar] [CrossRef]
- Balcke, G.U.; Handrick, V.; Bergau, N.; Fichtner, M.; Henning, A.; Stellmach, H.; Tissier, A.; Hause, B.; Frolov, A. An UPLC-MS/MS method for highly sensitive high-throughput analysis of phytohormones in plant tissues. Plant Methods. 2012, 8, 47. [Google Scholar] [CrossRef]
- Vierheilig, H.; Coughlan, A.P.; Wyss, U.; Piché, Y. Ink and vinegar, a simple staining technique for arbuscular-mycorrhizal fungi. Appl. Environ. Microbiol. 1998, 64, 5004–5007. [Google Scholar] [CrossRef]
- Giovanetti, M. Mosse, B. An evaluation of techniques for measuring vesicular arbuscular mycorrhizal infection in roots. New Phytol. 1980, 84, 489–500. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Eladlani, N.; Dahmane, E.M.; Rhazi, M.; Taourirte, M.; Habibi, Y. Complexation of chromium (III) ions with chitosan and its derivatives, nanoparticles and whiskers. Fresen. Environ. Bull. 2014, 23, 3278–3285. [Google Scholar]

| Treatments 1 | Glucose (mg/gFW) | Fructose (mg/gFW) | Sucrose (mg/gFW) |
|---|---|---|---|
| Ch0+AMF | 31.07 ± 4.27 a | 0.37 ± 0.11 a | 77.55 ± 9.65 a |
| Ch0.25+AMF | 27.91 ± 6.45 a | 0.42 ± 0.13 a | 68.71 ± 7.65 a |
| Ch1+AMF | 30.11 ± 6.46 a | 0.38 ± 0.10 a | 71.96 ± 9.66 a |
| Treatments 1 | ABA (pmol/gFW) | OPDA (pmol/gFW) | JA (pmol/gFW) | JA-Ile (pmol/gFW) |
|---|---|---|---|---|
| Ch0-AMF | 43.83 ± 11.01 a | 235.73 ± 43.06 a | 258.88 ± 113.59 a | 39.86 ± 11.23 ab |
| Ch0.25-AMF | 46.42 ± 14.96 a | 233.96 ± 41.14 a | 176.91 ± 115.78ab | 31.65 ± 4.44 b |
| Ch1-AMF | 51.42 ± 10.83 a | 278.97 ± 67.97 a | 300.89 ± 136.62 a | 52.05 ± 32.05 ab |
| Ch0+AMF | 53.66 ± 9.34 a | 112.49 ± 18.35 b | 58.46 ± 15.12 b | 58.48 ± 22.62 a |
| Ch0.25+AMF | 53.35 ± 13.95 a | 129.26 ± 13.82 b | 64.27 ± 25.73 b | 56.57 ± 29.78 a |
| Ch1+AMF | 56.45 ± 11.16 a | 131.69 ± 27.23 b | 71.72 ± 33.80 b | 39.56 ± 8.95 ab |
| Target Gene | Encoded Protein | Forward Primer | Reverse Primer |
|---|---|---|---|
| Ri-βTub (MT007813) | β-Tubulin | CCAACTTATGGCGATCTCAACA | AAGACGTGGAAAAGGCACCA |
| SlPT4 (Solyc06g051850.1.1) | AM-specific Phosphate Transporter | TATGGCTGGATTTTGCTGCACGT | GAACTTGTATCATTCCCATCCGTC |
| SlAOC (Solyc02g085730.2.1) | Allene Oxide Cyclase | TTCTACTTCGGCGATTACGGTC | GGTTAAGTACGCTCCCTGAACG |
| SlJAZ5 (Solyc03g118540) | Jasmonate ZIM-Domain5 | CTATAACATCCCATGGTGGC | GAAGGAGATGGAAGAACTCC |
| Chi3 (Solyc02g082920) | Chitinase (Acidic Isoform) | TGCAGGAACATTCACTGGAG | TAACGTTGTGGCATGATGGT |
| Chi9 (Solyc10g055810.1.1) | Chitinase (Basic Isoform) | GAAATTGCTGCTTTCCTTGC | CTCCAATGGCTCTTCCACAT |
| SlNCED1 (Solyc07g056570) | 9-Cis-Epoxycarotenoid Dioxygenase | ACATAATAGGCAAAGTCTCA | GTTGAAGAAGAAGAGGAGTT |
| SlEF (Solyc11g069700.1.1) | Elongation Factor | ACCACGAAGCTCTCCAGGAG | CATTGAACCCAACATTGTCACC |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El Amerany, F.; Meddich, A.; Wahbi, S.; Porzel, A.; Taourirte, M.; Rhazi, M.; Hause, B. Foliar Application of Chitosan Increases Tomato Growth and Influences Mycorrhization and Expression of Endochitinase-Encoding Genes. Int. J. Mol. Sci. 2020, 21, 535. https://doi.org/10.3390/ijms21020535
El Amerany F, Meddich A, Wahbi S, Porzel A, Taourirte M, Rhazi M, Hause B. Foliar Application of Chitosan Increases Tomato Growth and Influences Mycorrhization and Expression of Endochitinase-Encoding Genes. International Journal of Molecular Sciences. 2020; 21(2):535. https://doi.org/10.3390/ijms21020535
Chicago/Turabian StyleEl Amerany, Fatima, Abdelilah Meddich, Said Wahbi, Andrea Porzel, Moha Taourirte, Mohammed Rhazi, and Bettina Hause. 2020. "Foliar Application of Chitosan Increases Tomato Growth and Influences Mycorrhization and Expression of Endochitinase-Encoding Genes" International Journal of Molecular Sciences 21, no. 2: 535. https://doi.org/10.3390/ijms21020535
APA StyleEl Amerany, F., Meddich, A., Wahbi, S., Porzel, A., Taourirte, M., Rhazi, M., & Hause, B. (2020). Foliar Application of Chitosan Increases Tomato Growth and Influences Mycorrhization and Expression of Endochitinase-Encoding Genes. International Journal of Molecular Sciences, 21(2), 535. https://doi.org/10.3390/ijms21020535






