Abstract
Preclinical studies have shown that exposure of the developing brain to inhalational anesthetics can cause neurotoxicity. However, other studies have claimed that anesthetics can exert neuroprotective effects. We investigated the mechanisms associated with the neurotoxic and neuroprotective effects exerted by inhalational anesthetics. Neuroblastoma cells were exposed to sevoflurane and then cultured in 1% oxygen. We evaluated the expression of proteins related to the unfolded protein response (UPR). Next, we exposed adult mice in which binding immunoglobulin protein (BiP) had been mutated, and wild-type mice, to sevoflurane, and evaluated their cognitive function. We compared our results to those from our previous study in which mice were exposed to sevoflurane at the fetal stage. Pre-exposure to sevoflurane reduced the expression of CHOP in neuroblastoma cells exposed to hypoxia. Anesthetic pre-exposure also significantly improved the cognitive function of adult wild-type mice, but not the mutant mice. In contrast, mice exposed to anesthetics during the fetal stage showed cognitive impairment. Our data indicate that exposure to inhalational anesthetics causes endoplasmic reticulum (ER) stress, and subsequently leads to an adaptive response, the UPR. This response may enhance the capacity of cells to adapt to injuries and improve neuronal function in adult mice, but not in developing mice.
1. Introduction
General anesthesia is an essential medical treatment for surgery. Inhalational anesthetics provide adequate loss of consciousness and a certain degree of analgesia, and are widely used in current medial practice. The premise of general anesthesia is that it is reversible. It is widely accepted that an inhalational anesthetic loses its effect if it is excreted from the site of effect. The whole body then returns to its original state, including the nervous system. However, over recent years, it has become clear that the action of inhalational anesthetics is not necessarily reversible. For example, Jevtovic-Todorovic showed that pediatric anesthesia in 7-day-old infant rats, using midazolam, nitrous oxide, and isoflurane, caused widespread apoptotic neurodegeneration in the developing brain, and persistent cognitive impairments [1]. Subsequent studies confirmed that common inhalational anesthetics, such as isoflurane and sevoflurane, cause neuronal degeneration in the developing brain [2]. These anesthetics resulted in impaired neurogenesis, and induced delayed-onset neurocognitive dysfunction in infant rats [3]. Early exposure of infant rhesus monkeys to sevoflurane, within one month of birth, caused increased anxiety-related emotional behaviors by the time monkeys reached 6 months of age [4], and late-onset impairments in visual recognition memory by 12 months of age [5].
Retrospective human clinical cohort studies further revealed that multiple exposures to anesthetics prior to the age of 2 years [6], or 4 years [7], was a significant risk factor for subsequent learning disabilities. On the other hand, a sibling-matched cohort study (the Pediatric Anesthesia Neurodevelopment Assessment, PANDA study), exposed otherwise healthy children undergoing inguinal hernia surgery to a single dose of anesthetic prior to 36 months of age; interestingly, these children did not develop cognitive dysfunction in their subsequent childhood [8]. The General Anesthesia compared to Spinal anesthesia (GAS) trial also revealed that exposure to sevoflurane less than 1 h into infancy did not increase the risk of an adverse neurodevelopmental outcome at 2 years of age compared with awake-regional anesthesia [9]. Healthy infants therefore appear to withstand single exposure to anesthetics for a short duration.
Several laboratory studies have demonstrated the neuroprotective ability of inhalational anesthetics. For example, isoflurane was shown to reduce the proliferation of neuronal progenitors in rats on postnatal day 7, but increased such proliferation by postnatal day 60 [3]. In another study, sevoflurane was shown to reduce focal ischemic brain damage in an experimental rat model [10]. Evidence suggests that the neuroprotective effects of inhalational anesthetics, such as sevoflurane and isoflurane, are due to a reduction in the excitotoxicity of N-methyl-d-aspartate (NMDA) [11,12]. Exposure to sevoflurane has also been shown to improve cognitive function in both young (8–10 weeks) and aged rats (19 months) [13]. The general mechanism responsible for these conflicting effects, neurotoxicity and neuroprotection, remains unelucidated [14,15,16].
One possible factor underlying the neurotoxicity of inhalational anesthetics is a change in intracellular Ca2+ kinetics [17]. The endoplasmic reticulum (ER) is the main Ca2+ pool in cells; Ca2+ is released from the ER into the cytosol via the inositol 1,4,5-triphosphate receptor (ITPR), and the ryanodine receptor, on the ER membrane [18,19,20]. Inhalational anesthetics can exert influence over these Ca2+ channel receptors, and thus induce an aberrant rise in cytoplasmic Ca2+ [21]. An excess of cytoplasmic Ca2+ would then enter the mitochondria, thus resulting in the activation of caspases and ultimately, cell death [22,23]. Since ER molecular chaperones are Ca2+ binding proteins, it follows that aberrant Ca2+ mobilization will lead to the disruption of protein folding in the ER, thereby causing ER stress [24].
Secreted proteins, and membrane proteins, are synthesized by ribosomes on the ER membrane. These proteins are subsequently translocated to the ER membrane and undergo folding to become functional proteins with mature structures; this process involves significant interaction with molecular chaperones in the ER, including binding immunoglobulin protein (BiP). Subsequently, these proteins are transported to the secretory pathway to carry out their functional role; for example, as such as cell surface receptors or secretory proteins [24,25]. ER stresses, such as hypoxia, ischemia, malnutrition, or mutation, initiates an adaptive response referred to as the unfolded protein response (UPR) [26]. The UPR improves the capacity of cells to cope with ER stress, and do so by increasing the production of ER molecular chaperones, degrading unfolded proteins, and by repressing the synthesis of new proteins [27]. However, the capacity for the UPR to yield protective effects is limited. Extensive ER stress, that is beyond the capacity to adapt, has been shown to cause cellular dysfunction and cell death, thus leading to a range of human disorders, including cardiovascular disease [28], tumorigenesis [29], and neurodegenerative diseases [30,31,32,33].
We previously showed that exposure to inhalational anesthetics induced ER stress, leading to neuronal cell death and cognitive dysfunction in the developing brain of a knock-in mouse model expressing a mutant form of the Bip gene [34,35]. The BiP protein is a major ER-resident molecular chaperone that helps to fold proteins and regulate the UPR [36]. ER membrane proteins, such as protein kinase RNA (PKR)-like ER kinase (PERK, or eukaryotic translation initiation factor 2 alpha kinase 3; EIF2AK3), inositol requiring enzyme 1 (IRE1, or endoplasmic reticulum to nucleus signaling 1; ERN1), and activating transcription factor 6 (ATF6), are normally bound to BiP in the resting state [26]. Under ER stress, BiP binds to unfolded proteins and these membrane proteins become activated. IRE1 and PERK are multiplexed, and become activated by autophosphorylation [37]. IRE1 contains an endoribonuclease domain that splices X-Box Binding Protein 1 (XBP1) mRNA, and the active form of XBP1 mRNA is then translated [38]. The XBP1 protein promotes the transcription of various genes required for the UPR [39]. PERK is a serine/threonine kinase that inactivates eukaryotic translation initiation factor 2A, thereby suppressing the translation of most proteins [40]. ATF6 is transported to the Golgi complex, cleaved, and the cytosolic N-terminus acts as a transcription factor, thus facilitating the transcription of molecular chaperones, including BiP and transcriptional factors such as XBP1 and C/EBP homologous protein (CHOP) [41,42].
Wild-type BiP is secreted from the ER to the Golgi at a constant rate, while the carboxyl-terminal Lys-Asp-Glu-Leu (KDEL) sequence of BiP is recognized by the KDEL receptor in the Golgi [43]. BiP is returned back to the ER by coat protein I (COPI) vesicles [44]. Mutant BiP lacks the KDEL sequence and exhibits impairment in terms of ER function [25]. Homozygous mutant Bip mice die on the first day after birth because their ability to synthesize pulmonary surfactant is impaired [45]. The heterozygous Bip mutant mouse, however, is viable and develops late-onset neurodegenerative diseases [35,46].
In this study, we examined the relationship between the UPR and the neurotoxic and neuroprotective effects of inhalational anesthetics using Bip mutant mice and cultured neuronal cells.
2. Results
2.1. Exposure to Inhalational Anesthetics Induces the UPR
In order to investigate whether inhalational anesthetics influence the function of the ER at the cellular level, we exposed a mouse neuroblastoma cell line, neuro2a, to sevoflurane. Neuro2a cells were cultured at 37 °C in an atmosphere containing 21% O2 and 5% CO2. Neuro2a cells were then exposed to 3% sevoflurane for 2.5 h, 5 h, or 7 h. Subsequently, cells were collected and analyzed by western blotting for protein expression related to the UPR (Figure 1). We found that exposure to sevoflurane caused an increase in the expression of cleaved ATF6α, thus indicating that the UPR had been initiated. The expression of XBP1 and BiP was observed even under the control condition, but their expression levels increased as the length of exposure to sevoflurane increased. CHOP, a transcription factor that is expressed in response to ER stress and induces cell death [47,48], was expressed at very low levels under the control conditions, but was expressed at higher levels in cells exposed to sevoflurane. A significant increase in the expression of CHOP was observed at 7.5 h. These results suggested that sevoflurane affected cells and inhibited the protein folding of nascent proteins in the ER, thus inducing the UPR. Subsequently, the UPR enhanced the expression of cytoprotective ER chaperones, such as BiP, but also induced the expression of CHOP, thus resulting in cell death under conditions in which the ER is seriously stressed.
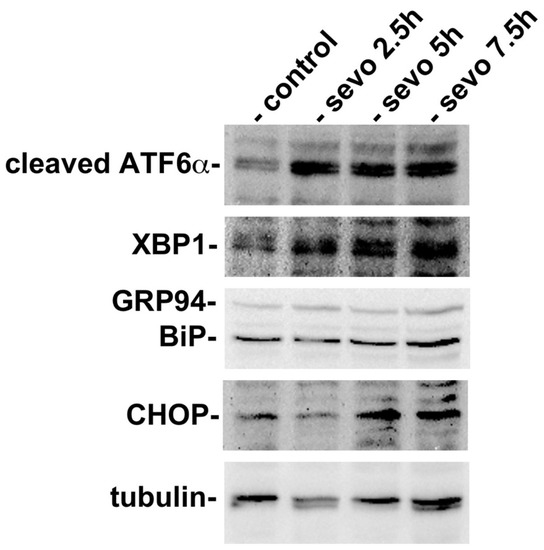
Figure 1.
Exposure to sevoflurane resulted in the initiation of the unfolded protein response (UPR). Neuro2a cells were cultured and exposed to 3% sevoflurane for 2.5, 5, or 7.5 h. The cells were then collected and analyzed by western blotting. Sevoflurane exposure activated ATF6 and induced the expression of XBP-1, a transcription factor that enhanced the expression of BiP, a major endoplasmic reticulum (ER) chaperone. GRP94, another ER chaperone is recognized by the anti-KDEL antibody. CHOP, a transcription factor responsible for initiating apoptotic cell death during ER stress, was expressed at significantly higher levels after 7.5 h of exposure.
2.2. Anesthetic Exposure Caused Cognitive Impairment in the Developing Brain
Next, we investigated the effects of inhalational anesthetics on the developing mouse brain in vivo. Since the exposure of neonatal mice to inhalational anesthetics could suppress their respiration and circulation, possibly resulting in hypoxia and low blood flow, we exposed pregnant mice to sevoflurane 2 days before delivery. Female wild-type mice were mated with male heterozygous mutant Bip mice. On day 17.5 of gestation, pregnant mice were exposed to 3% sevoflurane for 3 h under 40% O2. During exposure, we measured body temperature, blood pressure in the tail, blood flow in the tail, and percutaneous oxygen saturation. Overall, we observed a reduction in blood pressure, although blood flow and SpO2 did not decrease. The newborns showed a ratio of 1:1 (wild-type:heterozygous mutant Bip mice) and were raised by the parent mice. At 10 weeks of age, we evaluated their cognitive function [35]. Radial maze testing is widely used as a spatial cognitive function test in mice [49]. Testing was performed for 5 consecutive days, during which learning effects (shorter completion time and higher correct answer rate) were observed in individual mice. Radial maze testing at 10 weeks revealed that both wild-type mice, and the heterozygous mutant Bip mice, exposed to anesthetic, had a significantly longer completion time (p < 0.01), and a lower correct answer rate (p < 0.05), compared with unexposed mice (Figure 2, Supplementary Materials S1).
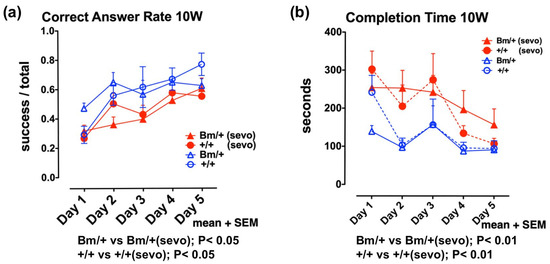
Figure 2.
Anesthetic exposure during the perinatal period had negative effects on spatial working memory in young mice. Eight-arm radial maze testing was performed in wild-type mice (+/+; circles) and Bip mutant mice (Bm; triangles) at 10 weeks after sevoflurane exposure (+/+ sevo, n = 12, closed red circles; Bm/+ sevo, n = 16, closed red triangles), and without exposure (+/+, n = 6, open blue circles; Bm/+, n = 8; open blue triangles). Tests were performed for 5 days. Statistical significance was determined by one-way repeated measures analysis of variance (ANOVA) followed by Bonferroni’s multiple comparison testing. (a) Correct answer rates are shown as the mean value of each group + standard error of the mean (SEM). The correct answer rate was defined as the ratio of successful attempts to overall attempts (0.0–1.0). (b) Mean completion times for each group + SEM (data extracted from DATASET S1 and DATASET S2, Front. Neurosci. 12:753. doi:10.3389/fnins. 2018.00753). Fetal anesthetic exposure resulted in a lower correct answer rate (p < 0.05) (a), and a longer completion time (p < 0.01) (b), compared with unexposed mice; this was the case for both Bip mutant mice and wild-type mice.
2.3. Moderate Exposure to Anesthetics Causes Cytoprotective Effects through the UPR
We considered that exposure to inhalational anesthetic caused ER stress at the cellular level, thus inducing the expression of CHOP (Figure 1). We also considered that the UPR also induces molecular chaperones, such as BiP, that can exert cytoprotective effects. Then, we exposed neuro2a cells to 3% sevoflurane for 5 h, and returned them to normal culture conditions (37 °C, 21% O2, and 5% CO2) for 12 h, thus allowing them to compensate for the stress. Subsequently, cells were cultured for 17 h in hypoxic conditions of 1% O2. The expression of CHOP was then evaluated by western blotting. When cells were exposed to hypoxia alone, the expression of CHOP was induced to some extent, but in cells that had been previously exposed to sevoflurane, the expression of CHOP was clearly suppressed, thus indicating the existence of some form of neuroprotection. When the expression of BiP was suppressed by siRNA, the expression of CHOP was notably increased (p < 0.05.) and the neuroprotective effect of sevoflurane appeared to disappear. These results suggested that pre-exposure to sevoflurane induced the UPR, which subsequently increased the capacity of cells to tolerate hypoxic conditions via the expression of cytoprotective chaperones, such as BiP (Figure 3, Supplementary Materials S2).
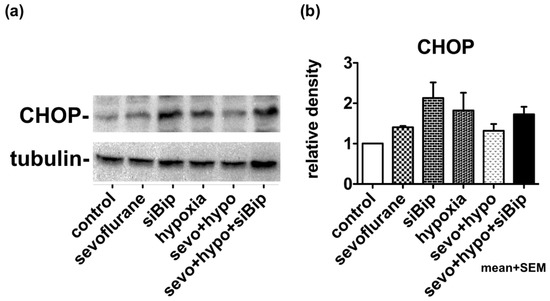
Figure 3.
Pre-exposure to sevoflurane may induce the UPR and reduce the damage caused by hypoxia. (a) Cells were exposed to 3% sevoflurane for 5 h. After 12 h of recovery time, cells were exposed to hypoxic conditions (1% O2) for 17 h before collection. Hypoxia alone induced the expression of CHOP. Pre-exposure to sevoflurane reduced the expression of CHOP while siBiP enhanced the expression of CHOP. (b) Densitometry was performed for three experiments. The density of CHOP relative to that of tubulin was measured by Image J and analyzed by one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparison test. Values indicate mean + standard error of the mean (SEM). Control vs. siBip; p < 0.05.
2.4. Pre-Exposure to Sevoflurane Improves Cognitive Function in Adult Wild-Type Mice, but not in Bip Mutant Mice
Finally, we evaluated the effect of inhalational anesthetics on the cognitive function of adult mice. One-year old wild-type, and heterozygous Bip mutant mice, were exposed to 3% sevoflurane for 3 h in an atmosphere containing 40% O2. A radial maze test was then carried out each day from the 7th day after exposure, for a total period of 5 days. There was no significant difference in terms of cognitive function when compared between the unexposed wild-type and the mutant Bip mice, whereas the wild-type mice exposed to sevoflurane were associated with a significantly higher rate of correct answers when compared with unexposed Bip mutant mice (p < 0.05). In addition, the wild-type mice exposed to sevoflurane showed a significantly shorter completion time when compared with the unexposed wild-type mice (p < 0.05). Pre-exposure to inhalational anesthetics significantly improved cognitive function in the wild-type mice, while no such effect was evident in the Bip mutant mice (Figure 4, Supplementary Materials S3).
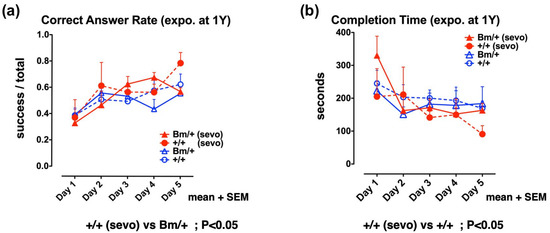
Figure 4.
Anesthetic exposure had positive effects on spatial working memory in adult wild-type mice, but not mutant BiP mice. Eight-arm radial maze testing was performed in adult wild-type mice (+/+; circles) and mutant-Bip mice (Bm; triangles) 7 days after sevoflurane (sevo) exposure (+/+ sevo, n = 4, closed red circles; Bm/+ sevo, n = 5, closed red triangles) and without exposure (+/+, n = 7, open circles; Bm/+, n = 10; open triangles). Tests were performed over 5 days. Significance was determined by one-way repeated measures ANOVA followed by Bonferroni’s multiple comparison testing. (a) Correct answer rates are shown as the mean value for each group + standard error of the mean (SEM). The correct answer rate was defined as the ratio of successful attempts to the number of overall attempts (0.0–1.0). (b) Completion times represent the mean for each group + SEM.
These results suggest that exposure to inhalational anesthetics may confer neurotoxic effects, thus inducing adaptive responses of UPR. The UPR enhances the capacity of cells, and individuals, to tolerate such insults. This scenario applies to healthy wild-type individuals, but not sensitive individuals, such as neonates with developing neuronal cells, and aged individuals with degenerative neuronal cells (Figure 2 and Figure 4).
3. Discussion
The exposure to inhalational anesthetics has neurotoxic effects, the extent of which depend on the duration and dose. However, moderate levels of exposure many have beneficial effects on appropriate subjects. The exposure to inhalational anesthetics appears to exert a pre-conditioning function. The concept of preconditioning was conceived some time ago [50]. For example, in cardiac surgery, it is now well known that a period of moderate ischemia can cause preconditioning and thereby enhancing resistance to a subsequent period of ischemia over a longer period [51]. It has also been shown that ischemic preconditioning is effective not only in the heart [52] but also in the nervous system [53], skeletal muscle [54], and liver [55]. The mechanism underlying these beneficial effects is thought to be related to changes in the expression of various genes in response to moderate preconditioning, including the production of heat-shock proteins [56], and the ER molecular chaperone, BiP [57]. Injuries that perturb protein folding in cells can induce a heat shock response in the cytoplasm, and the UPR in the ER, thus increasing the production of cytoprotective molecular chaperones, including heat shock protein 70 (HSP70) and BiP. Consequently, cells gain enhanced resistance to further cytotoxic injuries. This integrated form of stress response is involved in a variety of pathological conditions [58,59]. Other research has shown that during anesthesia for cardiac surgery, inhalational anesthetics produce similar effects as those induced by ischemia [60]. Thus, sevoflurane has been used as a clinically beneficial agent for anesthetic preconditioning, or pharmacological preconditioning [61]. Inhalational anesthetics act on cells throughout the whole body, and the phenomenon observed in the myocardium is thought to occur in all tissues, including the nervous system. In fact, several studies have reported the neuroprotective effects of anesthetic preconditioning [62,63,64,65].
In this study, we showed that exposure to sevoflurane induces the UPR; this concurred with our previous findings [34]. The UPR is initiated by the activation of ATF6, PERK, and IRE1 [26]. Distinctive roles of these three pathways are suggested by analysis of their gene knockout mice [66]. We assessed the initiation of the UPR induced by anesthetic exposure with western blot analysis of ATF6α and XBP1. Since XBP1 protein are translated after splicing of XBP1 mRNA by activated IRE1 [38], the detection of XBP1 protein represents the activation of IRE1. ATF6α and ATF6β double knockout mice are embryonic lethal [67]. XBP1 knockout mice are embryonic lethal [68]. IRE1α knockout mice are also embryonic lethal [69]. While PERK knockout mice develop diabetes mellitus, they are viable [70]. Although the PERK pathway was not evaluated in this study, the detection of cleaved ATF6α and XBP1 may have been able to assess, if not completely, the initiation of the UPR.
The UPR can enhance the resistance of cells to injuries. However, sevoflurane can also cause ER stress, and can eventually induce cell death if the exposure is excessive. The developing brain is highly sensitive to injuries. Moreover, it is thought that certain types of cells in the same individual are highly sensitive, such as actively dividing stem cells. If important neuronal cells suffer from a failure to migrate, or die, then the consequences of these effects may later manifest as cognitive dysfunction or higher brain damage, although it might not be immediately apparent. A previous study, involving the rat model, reported that inhalational anesthetics could disturb the neuronal migration in the developing brain [71]. Fetal isoflurane exposure was also shown to diminish the expression of reelin, a large glycoprotein secreted by neurosecretory Cajal-Retzius cells and responsible for mediating cortical neuronal migration in the developing brain [72]. The dysfunction of reelin has been suggested to cause higher brain disorders, such as autism [73]. In our previous paper, homozygous Bip mutant mice showed impaired neuronal migration [74]; the production of reelin was shown to be diminished in the Cajal-Retzius cells, while transcription of the reelin gene was preserved. The dysfunction of BiP in the Bip mutant neonates may affect the folding of large secretory proteins, such as reelin. The protein folding environment in the ER is highly sensitive to injuries, including ischemia, hypoxia, toxic substances, and malnutrition. Thus, the UPR may represent one of the most important cellular adaptive mechanisms to injury. We were the first to report that fetal exposure to inhalational anesthetics causes ER stress and neuronal cell death [34]; since then, several other papers have confirmed that general anesthesia causes ER stress [75,76,77,78,79,80,81].
In our previous study, the fetal exposure of homozygous Bip mutant mice to 3% sevoflurane for 3 h resulted in neuronal cell death, but not in the heterozygous mutant or the wild-type mice [34]. After delivery, both the heterozygous mutant, and wild-type mice, appeared to be normal, despite the fact that they had been exposed to sevoflurane during their time as a fetus. However, further analysis showed that these mice suffered from cognitive dysfunction when examined 10 weeks after birth. In contrast, the same level of exposure (3% sevoflurane for 3 h) resulted in a slight improvement in cognitive function when tested 7 days after exposure in adult wild-type mice, but not the heterozygous Bip mutant. The overall outcome of anesthetic exposure appears to depend on the balance between the extent of injury, and the sensitivity of the subjects. In the heterozygous Bip mutant mice, cognitive function tended to decline faster than in the wild-type mice. However, the cognitive function of wild-type mice also fades with advanced age; thus, there is no significant difference between the aged wild-type and mutants [35]. Cognitive function is superior in young mice, and aging exerts more of an influence than genotype. The exposure of fetuses to sevoflurane results in a decline of cognitive function, regardless of genotype; however, when adults were exposed, wild-type mice showed slight improvements in cognitive function. No such changes in cognitive function were observed in the Bip mutant mice (Figure 4).
There is no clear evidence, from either animal experiments or clinical studies, that sevoflurane anesthesia subsequently leads to poorer cognitive function. One study showed that one minimum alveolar concentration (MAC) of sevoflurane exposure led to an improvement in the cognitive function of 10 week-old rats and 20 month-old rats [13]. In another study, using APP23 Alzheimer’s disease transgenic mice expressing mutant amyloid-β precursor protein, and wild-type control mice, exposure to one MAC of isoflurane for 2 h led to an improvement in cognitive function in both types of mice. Regardless of genotype, young mice (4 months-old) were superior in terms of cognitive function when compared with aged mice (14–16 months-old) [82].
Several clinical studies have also reported the beneficial effect of inhalational anesthetics in terms of post-operative cognitive function; however, other studies have reported an increased risk for post-operative cognitive decline. In a randomized, controlled trial involving patients undergoing on-pump cardiac surgery, sevoflurane anesthesia showed better results in terms of a cognitive function test on days 2–6 post-surgery, when compared with propofol anesthesia, [83]. In another study, propofol anesthesia was reported to exert less cognitive impairment one week after surgery than sevoflurane anesthesia when patients over 65 years of age underwent cancer surgery for more than 2 h [84]. Another study described the postoperative cognitive function of twin surgical patients, but only reported a slight effect with regards to surgical anesthesia; cognitive function was reduced slightly following major surgery, and showed slight improvements following hip and knee surgery [85]. A retrospective cohort study of patients with mild cognitive impairment (MCI) and healthy controls aged 70–89 years, showed that MCI was not related to general anesthesia at ages over 40 years [86]. In a prospective cohort study of people over 65 years of age free of dementia at baseline, there was no relationship between anesthesia experience and the incidence of dementia or Alzheimer’s disease; people who underwent less invasive forms of surgery had a lower incidence of dementia than people with no history of anesthesia [87]. Another study compared patients over 50 years-of-age who had been newly diagnosed with dementia with a control group; those who had experienced general anesthesia had a higher incidence of dementia. In addition, people with a history of diabetes, hypertension, or cerebral infarction, were also shown to have a higher incidence of dementia [88].
Inhalational anesthetics are associated with longer exposure times; furthermore, the higher the exposure concentration, the greater the effect. It is difficult to determine the specific concentration and duration to use for preconditioning. Sensitivity is known to vary from individual to individual; it also appears that sensitivity varies according to cell type, and even within the same individual. The degree of invasion that is appropriate for preconditioning therefore shows individual variation and may also depend on the patient’s condition at the time of exposure.
4. Materials and Methods
4.1. Cells and Reagents
The murine neuroblastoma cell line, neuro2a, were cultured with a complete medium that consisted of Dulbecco’s modified Eagle’s medium (DMEM; Sigma Chemical, Saint Louis, MO, USA) with 10% fetal bovine serum, 2 mM glutamine, 50 µg/mL streptomycin, and 50 U/mL penicillin G at 37 °C in a 5% CO2 incubator. For hypoxic experiments, N2 was injected into the incubator to create a 1% oxygen concentration. The following antibodies were used during our experiments: Mouse mAb against γ-tubulin (Sigma Chemical, Saint Louis, MO, USA), mouse mAb SPA-827 against BiP (KDEL sequence; Stressgen, Victoria, BC, Canada), rabbit antiserum against CHOP/GADD153 (Santa Cruz Biotechnology, Dallas, TX, USA), mouse mAb 37-1, 73–505 against ATF6α (Bio Academia, Suita, Japan), and mouse mAbs M-186 and sc-7160 against XBP-1 (Santa Cruz Biotechnology, Dallas, TX, USA). siRNA oligonucleotides for Bip (sc-35522), and control siRNA (sc-37007) were purchased from Santa Cruz Biotechnology. For siRNA uptake, neuro2a cells were transfected with siRNA oligonucleotides in accordance with the manufacturer’s instructions. At 24 h post-transfection, the medium was changed to a complete medium and cells were exposed to sevoflurane.
4.2. Animals
This study was carried out in accordance with the recommendations of the guidelines for animal experiments of Chiba University. The protocol (30–132) was approved by the Institutional Animal Care Committee of Chiba University, Chiba, Japan. The generation of the knock-in mouse, expressing a mutant form of BiP that lacked the carboxyl-terminal KDEL sequence, was described previously [45]. The missing KDEL sequence was replaced with a hemagglutinin (HA) tag. The heterozygous Bip mutant male mice (Bm/+) were mated with wild-type (C57BL/6, +/+) female mice, and offspring (wild-type and heterozygous mutant) were bred for use in experiments.
4.3. Inhalational Anesthetic Exposure
Adult heterozygous Bip mutant mice, and wild-type mice (approximately one year-of-age), were exposed to 3% sevoflurane for 3 h, as previously described [34]. Neuro2a cells were exposed to 3% sevoflurane for 2.5–7.5 h, as previously described [34]. Previous studies suggested that 3.0% sevoflurane was almost equivalent to 0.6 MAC for mice [89].
4.4. Eight-Arm Radial Maze Test
Mice were subjected to the eight-arm radial maze test 7 days after being exposed to sevoflurane, as previously described [35]. The completion time (in seconds) was defined as the total time taken by each mouse to eat all of the food in the eight arms of the maze. Attempts to enter arms without bait were counted as errors, while attempts to enter arms with food were counted as successes. The correct answer rate was defined as the ratio of successful attempts to the overall number of attempts (0.0–1.0). Testing was carried out daily for 5 days (Supplementary Materials S3, Figure 4). Data related to fetal sevoflurane exposure were extracted from DATASET S1 and DATASET S2, as described in our previous study (Supplementary Materials S1, Figure 2) [35].
4.5. Western Blotting
Cultured cells were homogenized in a buffer containing 0.4% (w/v) Nonidet P-40, 0.2% N-lauroylsarcosine, 10 mM Tris/HCl pH 8.0, 30 mM EDTA, 10 µg/mL aprotinin, 10 µg/mL leupeptin, and 30 µg/mL N-acetyl-l-leucinal-l-lecinal-l-norleucinal (ALLN, Sigma Chemical). Western blotting was then performed and analyzed with LAS-1000 and Image Gauge Software (Fuji Photo Film Co. Ltd., Tokyo, Japan), as described previously [34]. Densitometry was performed using ImageJ software (NIH, Bethesda, MD, USA).
4.6. Statistical Analysis
The results of the radial maze test are shown as means + standard error of the mean (SEM). Repeated measures analyses of variance (ANOVA), followed by Bonferroni multiple comparison tests, were then performed to compare values between groups (GraphPad Prism 4.0, GraphPad Software, San Diego, CA, USA). Statistical significance was accepted at p < 0.05. Densitometry was carried out for the CHOP images and resultant data analyzed by one-way ANOVA, followed by Dunnett’s multiple comparison test.
5. Conclusions
Inhalational anesthetics cause neurotoxicity, but can also induce adaptive responses, such as the UPR in neuronal cells. Therefore, inhaled anesthetics may exert neuroprotective effects. It is therefore necessary to use inhalational anesthetics in an appropriate manner, depending on the patient’s individual condition.
Supplementary Materials
Supplementary Materials can be found at https://www.mdpi.com/1422-0067/21/2/450/s1.
Author Contributions
Conceptualization, T.A.; formal analysis, H.K., H.J., M.K. and T.A.; investigation, H.K., H.J., M.K. and T.A.; data curation, T.A.; preparation of the original draft manuscript, T.A.; reviewed and edited the manuscript, T.A., H.K. and H.J; project administration, T.A.; funding acquisition, M.K., H.K. and T.A. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by Grants-in-Aid for Japan Society for the Promotion of Science (KAKENHI) to H.K. (18K16473), M.K. (15K20031) and T.A. (17K11114).
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Abbreviations
| ER | Endoplasmic reticulum |
| UPR | Unfolded protein response |
| BiP | Binding immunoglobulin protein |
| KDEL | Lys-Asp-Glu-Leu |
| COPI | Coat protein I |
| CHOP | C/EBP homologous protein |
| PERK | Protein kinase RNA (PKR)-like ER kinase |
| IRE1 | Inositol requiring enzyme 1 |
| ATF6 | Activating transcription factor 6 |
| XBP1 | X-box-binding protein 1 |
| NMDA | N-methyl-D-aspartate |
| ITPR | Inositol 1,4,5-triphosphate receptor |
| MAC | Minimum alveolar concentration |
| MCI | Mild cognitive impairment |
References
- Jevtovic-Todorovic, V.; Hartman, R.E.; Izumi, Y.; Benshoff, N.D.; Dikranian, K.; Zorumski, C.F.; Olney, J.W.; Wozniak, D.F. Early exposure to common anesthetic agents causes widespread neurodegeneration in the developing rat brain and persistent learning deficits. J. Neurosci. 2003, 23, 876–882. [Google Scholar] [CrossRef] [PubMed]
- Jevtovic-Todorovic, V. General Anesthetics and Neurotoxicity: How Much Do We Know? Anesthesiol. Clin. 2016, 34, 439–451. [Google Scholar] [CrossRef]
- Stratmann, G.; Sall, J.W.; May, L.D.; Bell, J.S.; Magnusson, K.R.; Rau, V.; Visrodia, K.H.; Alvi, R.S.; Ku, B.; Lee, M.T.; et al. Isoflurane differentially affects neurogenesis and long-term neurocognitive function in 60-day-old and 7-day-old rats. Anesthesiology 2009, 110, 834–848. [Google Scholar] [CrossRef] [PubMed]
- Raper, J.; Alvarado, M.C.; Murphy, K.L.; Baxter, M.G. Multiple Anesthetic Exposure in Infant Monkeys Alters Emotional Reactivity to an Acute Stressor. Anesthesiology 2015, 123, 1084–1092. [Google Scholar] [CrossRef]
- Alvarado, M.C.; Murphy, K.L.; Baxter, M.G. Visual recognition memory is impaired in rhesus monkeys repeatedly exposed to sevoflurane in infancy. Br. J. Anaesth. 2017, 119, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Flick, R.P.; Katusic, S.K.; Colligan, R.C.; Wilder, R.T.; Voigt, R.G.; Olson, M.D.; Sprung, J.; Weaver, A.L.; Schroeder, D.R.; Warner, D.O. Cognitive and behavioral outcomes after early exposure to anesthesia and surgery. Pediatrics 2011, 128, e1053–e1061. [Google Scholar] [CrossRef]
- Wilder, R.T.; Flick, R.P.; Sprung, J.; Katusic, S.K.; Barbaresi, W.J.; Mickelson, C.; Gleich, S.J.; Schroeder, D.R.; Weaver, A.L.; Warner, D.O. Early exposure to anesthesia and learning disabilities in a population-based birth cohort. Anesthesiology 2009, 110, 796–804. [Google Scholar] [CrossRef]
- Sun, L.S.; Li, G.; Miller, T.L.; Salorio, C.; Byrne, M.W.; Bellinger, D.C.; Ing, C.; Park, R.; Radcliffe, J.; Hays, S.R.; et al. Association Between a Single General Anesthesia Exposure Before Age 36 Months and Neurocognitive Outcomes in Later Childhood. JAMA 2016, 315, 2312–2320. [Google Scholar] [CrossRef]
- Davidson, A.J.; Disma, N.; de Graaff, J.C.; Withington, D.E.; Dorris, L.; Bell, G.; Stargatt, R.; Bellinger, D.C.; Schuster, T.; Arnup, S.J.; et al. Neurodevelopmental outcome at 2 years of age after general anaesthesia and awake-regional anaesthesia in infancy (GAS): An international multicentre, randomised controlled trial. Lancet 2016, 387, 239–250. [Google Scholar] [CrossRef]
- Warner, D.S.; McFarlane, C.; Todd, M.M.; Ludwig, P.; McAllister, A.M. Sevoflurane and halothane reduce focal ischemic brain damage in the rat. Possible influence on thermoregulation. Anesthesiology 1993, 79, 985–992. [Google Scholar] [CrossRef]
- Harada, H.; Kelly, P.J.; Cole, D.J.; Drummond, J.C.; Patel, P.M. Isoflurane reduces N-methyl-D-aspartate toxicity in vivo in the rat cerebral cortex. Anesth. Analg. 1999, 89, 1442–1447. [Google Scholar] [CrossRef] [PubMed]
- Kudo, M.; Aono, M.; Lee, Y.; Massey, G.; Pearlstein, R.D.; Warner, D.S. Effects of volatile anesthetics on N-methyl-D-aspartate excitotoxicity in primary rat neuronal-glial cultures. Anesthesiology 2001, 95, 756–765. [Google Scholar] [CrossRef] [PubMed]
- Callaway, J.K.; Jones, N.C.; Royse, A.G.; Royse, C.F. Sevoflurane anesthesia does not impair acquisition learning or memory in the Morris water maze in young adult and aged rats. Anesthesiology 2012, 117, 1091–1101. [Google Scholar] [CrossRef] [PubMed]
- Eizaga Rebollar, R.; Garcia Palacios, M.V.; Morales Guerrero, J.; Torres Morera, L.M. Neurotoxicity versus Neuroprotection of Anesthetics: Young Children on the Ropes? Paediatr. Drugs 2017, 19, 271–275. [Google Scholar] [CrossRef]
- Alam, A.; Suen, K.C.; Hana, Z.; Sanders, R.D.; Maze, M.; Ma, D. Neuroprotection and neurotoxicity in the developing brain: An update on the effects of dexmedetomidine and xenon. Neurotoxicol. Teratol. 2017, 60, 102–116. [Google Scholar] [CrossRef]
- Wu, L.; Zhao, H.; Weng, H.; Ma, D. Lasting effects of general anesthetics on the brain in the young and elderly: “mixed picture” of neurotoxicity, neuroprotection and cognitive impairment. J. Anesth. 2019, 33, 321–335. [Google Scholar] [CrossRef]
- Wei, H. The role of calcium dysregulation in anesthetic-mediated neurotoxicity. Anesth. Analg. 2011, 113, 972–974. [Google Scholar] [CrossRef]
- Berridge, M.J. Inositol trisphosphate and calcium signalling mechanisms. Biochim. Biophys. Acta 2009, 1793, 933–940. [Google Scholar] [CrossRef]
- Rizzuto, R.; De Stefani, D.; Raffaello, A.; Mammucari, C. Mitochondria as sensors and regulators of calcium signalling. Nat. Rev. Mol. Cell Biol. 2012, 13, 566–578. [Google Scholar] [CrossRef]
- Trychta, K.A.; Back, S.; Henderson, M.J.; Harvey, B.K. KDEL Receptors Are Differentially Regulated to Maintain the ER Proteome under Calcium Deficiency. Cell Rep. 2018, 25, 1829–1840.e6. [Google Scholar] [CrossRef]
- Yang, H.; Liang, G.; Hawkins, B.J.; Madesh, M.; Pierwola, A.; Wei, H. Inhalational anesthetics induce cell damage by disruption of intracellular calcium homeostasis with different potencies. Anesthesiology 2008, 109, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Kang, B.; Wei, W.; Liang, G.; Meng, Q.C.; Li, Y.; Eckenhoff, R.G. Isoflurane and sevoflurane affect cell survival and BCL-2/BAX ratio differently. Brain Res. 2005, 1037, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Dong, Y.; Wu, X.; Lu, Y.; Xu, Z.; Knapp, A.; Yue, Y.; Xu, T.; Xie, Z. The mitochondrial pathway of anesthetic isoflurane-induced apoptosis. J. Biol. Chem. 2010, 285, 4025–4037. [Google Scholar] [CrossRef] [PubMed]
- Kokubun, H.; Jin, H.; Aoe, T. Pathogenic Effects of Impaired Retrieval between the Endoplasmic Reticulum and Golgi Complex. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Komita, M.; Aoe, T. The Role of BiP Retrieval by the KDEL Receptor in the Early Secretory Pathway and its Effect on Protein Quality Control and Neurodegeneration. Front. Mol. Neurosci. 2017, 10, 222. [Google Scholar] [CrossRef]
- Hetz, C.; Papa, F.R. The Unfolded Protein Response and Cell Fate Control. Mol. Cell 2018, 69, 169–181. [Google Scholar] [CrossRef]
- Almanza, A.; Carlesso, A.; Chintha, C.; Creedican, S.; Doultsinos, D.; Leuzzi, B.; Luis, A.; McCarthy, N.; Montibeller, L.; More, S.; et al. Endoplasmic reticulum stress signalling—From basic mechanisms to clinical applications. FEBS J. 2019, 286, 241–278. [Google Scholar] [CrossRef]
- He, M.; Guo, H.; Yang, X.; Zhou, L.; Zhang, X.; Cheng, L.; Zeng, H.; Hu, F.B.; Tanguay, R.M.; Wu, T. Genetic variations in HSPA8 gene associated with coronary heart disease risk in a Chinese population. PLoS ONE 2010, 5, e9684. [Google Scholar] [CrossRef]
- Minakshi, R.; Rahman, S.; Jan, A.T.; Archana, A.; Kim, J. Implications of aging and the endoplasmic reticulum unfolded protein response on the molecular modality of breast cancer. Exp. Mol. Med. 2017, 49, e389. [Google Scholar] [CrossRef]
- Ogen-Shtern, N.; Ben David, T.; Lederkremer, G.Z. Protein aggregation and ER stress. Brain Res. 2016, 1648, 658–666. [Google Scholar] [CrossRef]
- Kalinderi, K.; Bostantjopoulou, S.; Fidani, L. The genetic background of Parkinson’s disease: Current progress and future prospects. Acta Neurol. Scand. 2016, 134, 314–326. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.; Jan, A.T.; Ayyagari, A.; Kim, J.; Kim, J.; Minakshi, R. Entanglement of UPR(ER) in Aging Driven Neurodegenerative Diseases. Front. Aging Neurosci. 2017, 9, 341. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.; Archana, A.; Jan, A.T.; Minakshi, R. Dissecting Endoplasmic Reticulum Unfolded Protein Response (UPR(ER)) in Managing Clandestine Modus Operandi of Alzheimer’s Disease. Front. Aging Neurosci. 2018, 10, 30. [Google Scholar] [CrossRef] [PubMed]
- Komita, M.; Jin, H.; Aoe, T. The effect of endoplasmic reticulum stress on neurotoxicity caused by inhaled anesthetics. Anesth Analg. 2013, 117, 1197–1204. [Google Scholar] [CrossRef]
- Jin, H.; Komita, M.; Aoe, T. Decreased Protein Quality Control Promotes the Cognitive Dysfunction Associated With Aging and Environmental Insults. Front. Neurosci. 2018, 12, 753. [Google Scholar] [CrossRef]
- Pobre, K.F.R.; Poet, G.J.; Hendershot, L.M. The endoplasmic reticulum (ER) chaperone BiP is a master regulator of ER functions: Getting by with a little help from ERdj friends. J. Biol. Chem. 2019, 294, 2098–2108. [Google Scholar] [CrossRef]
- Bertolotti, A.; Zhang, Y.; Hendershot, L.M.; Harding, H.P.; Ron, D. Dynamic interaction of BiP and ER stress transducers in the unfolded- protein response. Nat. Cell Biol. 2000, 2, 326–332. [Google Scholar] [CrossRef]
- Lee, K.; Tirasophon, W.; Shen, X.; Michalak, M.; Prywes, R.; Okada, T.; Yoshida, H.; Mori, K.; Kaufman, R.J. IRE1-mediated unconventional mRNA splicing and S2P-mediated ATF6 cleavage merge to regulate XBP1 in signaling the unfolded protein response. Genes Dev. 2002, 16, 452–466. [Google Scholar] [CrossRef]
- Adams, C.J.; Kopp, M.C.; Larburu, N.; Nowak, P.R.; Ali, M.M.U. Structure and Molecular Mechanism of ER Stress Signaling by the Unfolded Protein Response Signal Activator IRE1. Front. Mol. Biosci. 2019, 6, 11. [Google Scholar] [CrossRef]
- Hughes, D.; Mallucci, G.R. The unfolded protein response in neurodegenerative disorders—Therapeutic modulation of the PERK pathway. FEBS J. 2019, 286, 342–355. [Google Scholar] [CrossRef]
- Haze, K.; Yoshida, H.; Yanagi, H.; Yura, T.; Mori, K. Mammalian transcription factor ATF6 is synthesized as a transmembrane protein and activated by proteolysis in response to endoplasmic reticulum stress. Mol. Biol. Cell 1999, 10, 3787–3799. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Chen, X.; Hendershot, L.; Prywes, R. ER Stress Regulation of ATF6 Localization by Dissociation of BiP/GRP78 Binding and Unmasking of Golgi Localization Signals. Dev. Cell 2002, 3, 99–111. [Google Scholar] [CrossRef]
- Lewis, M.J.; Pelham, H.R. A human homologue of the yeast HDEL receptor. Nature 1990, 348, 162–163. [Google Scholar] [CrossRef]
- Yamamoto, K.; Fujii, R.; Toyofuku, Y.; Saito, T.; Koseki, H.; Hsu, V.W.; Aoe, T. The KDEL receptor mediates a retrieval mechanism that contributes to quality control at the endoplasmic reticulum. Embo J. 2001, 20, 3082–3091. [Google Scholar] [CrossRef] [PubMed]
- Mimura, N.; Hamada, H.; Kashio, M.; Jin, H.; Toyama, Y.; Kimura, K.; Iida, M.; Goto, S.; Saisho, H.; Toshimori, K.; et al. Aberrant quality control in the endoplasmic reticulum impairs the biosynthesis of pulmonary surfactant in mice expressing mutant BiP. Cell Death Differ. 2007, 14, 1475–1485. [Google Scholar] [CrossRef]
- Jin, H.; Mimura, N.; Kashio, M.; Koseki, H.; Aoe, T. Late-onset of spinal neurodegeneration in knock-in mice expressing a mutant BiP. PLoS ONE 2014, 9, e112837. [Google Scholar] [CrossRef]
- Oyadomari, S.; Mori, M. Roles of CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Differ. 2004, 11, 381–389. [Google Scholar] [CrossRef]
- Lu, H.Y.; Chen, X.Q.; Tang, W.; Wang, Q.X.; Zhang, J. GRP78 silencing enhances hyperoxia-induced alveolar epithelial cell apoptosis via CHOP pathway. Mol. Med. Rep. 2017, 16, 1493–1501. [Google Scholar] [CrossRef][Green Version]
- Olton, D.S.; Papas, B.C. Spatial memory and hippocampal function. Neuropsychologia 1979, 17, 669–682. [Google Scholar] [CrossRef]
- Murry, C.E.; Jennings, R.B.; Reimer, K.A. Preconditioning with ischemia: A delay of lethal cell injury in ischemic myocardium. Circulation 1986, 74, 1124–1136. [Google Scholar] [CrossRef]
- Annachhatre, A.S.; Annachhatre, S.R. Preconditioning in cardiac anesthesia…… where are we? Ann. Card Anaesth. 2019, 22, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Yellon, D.M.; Alkhulaifi, A.M.; Pugsley, W.B. Preconditioning the human myocardium. Lancet 1993, 342, 276–277. [Google Scholar] [CrossRef]
- Gidday, J.M.; Fitzgibbons, J.C.; Shah, A.R.; Park, T.S. Neuroprotection from ischemic brain injury by hypoxic preconditioning in the neonatal rat. Neurosci. Lett. 1994, 168, 221–224. [Google Scholar] [CrossRef]
- Lee, H.T.; Lineaweaver, W.C. Protection against ischemic-reperfusion injury of skeletal muscle: Role of ischemic preconditioning and adenosine pretreatment. J. Reconstr. Microsurg. 1996, 12, 383–388. [Google Scholar] [CrossRef]
- Lloris-Carsi, J.M.; Cejalvo, D.; Toledo-Pereyra, L.H.; Calvo, M.A.; Suzuki, S. Preconditioning: Effect upon lesion modulation in warm liver ischemia. Transplant. Proc. 1993, 25, 3303–3304. [Google Scholar]
- Benjamin, I.J.; McMillan, D.R. Stress (heat shock) proteins: Molecular chaperones in cardiovascular biology and disease. Circ. Res. 1998, 83, 117–132. [Google Scholar] [CrossRef]
- Hayashi, T.; Saito, A.; Okuno, S.; Ferrand-Drake, M.; Chan, P.H. Induction of GRP78 by ischemic preconditioning reduces endoplasmic reticulum stress and prevents delayed neuronal cell death. J. Cereb. Blood Flow Metab. 2003, 23, 949–961. [Google Scholar] [CrossRef]
- Walter, P.; Ron, D. The unfolded protein response: From stress pathway to homeostatic regulation. Science 2011, 334, 1081–1086. [Google Scholar] [CrossRef]
- Labbadia, J.; Morimoto, R.I. The biology of proteostasis in aging and disease. Annu. Rev. Biochem. 2015, 84, 435–464. [Google Scholar] [CrossRef]
- Guerrero-Orriach, J.L.; Escalona Belmonte, J.J.; Ramirez Fernandez, A.; Ramirez Aliaga, M.; Rubio Navarro, M.; Cruz Manas, J. Cardioprotection with halogenated gases: How does it occur? Drug Des. Dev. Ther. 2017, 11, 837–849. [Google Scholar] [CrossRef]
- Warltier, D.C.; Kersten, J.R.; Pagel, P.S.; Gross, G.J. Anesthetic preconditioning: Serendipity and science. Anesthesiology 2002, 97, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Zuo, Z. Isoflurane preconditioning decreases glutamate receptor overactivation-induced Purkinje neuronal injury in rat cerebellar slices. Brain Res. 2005, 1054, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Liang, G.; Yang, H. Isoflurane preconditioning inhibited isoflurane-induced neurotoxicity. Neurosci. Lett. 2007, 425, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Yang, Z.; Liang, G.; Wu, Z.; Peng, Y.; Joseph, D.J.; Inan, S.; Wei, H. Dual effects of isoflurane on proliferation, differentiation, and survival in human neuroprogenitor cells. Anesthesiology 2013, 118, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Deng, B.; Zhao, X.; Gao, C.; Yang, L.; Zhao, H.; Yu, D.; Zhang, F.; Xu, L.; Chen, L.; et al. Isoflurane preconditioning provides neuroprotection against stroke by regulating the expression of the TLR4 signalling pathway to alleviate microglial activation. Sci. Rep. 2015, 5, 11445. [Google Scholar] [CrossRef] [PubMed]
- Bommiasamy, H.; Popko, B. Animal models in the study of the unfolded protein response. Methods Enzymol. 2011, 491, 91–109. [Google Scholar] [CrossRef]
- Yamamoto, K.; Sato, T.; Matsui, T.; Sato, M.; Okada, T.; Yoshida, H.; Harada, A.; Mori, K. Transcriptional induction of mammalian ER quality control proteins is mediated by single or combined action of ATF6alpha and XBP1. Dev. Cell 2007, 13, 365–376. [Google Scholar] [CrossRef]
- Reimold, A.M.; Etkin, A.; Clauss, I.; Perkins, A.; Friend, D.S.; Zhang, J.; Horton, H.F.; Scott, A.; Orkin, S.H.; Byrne, M.C.; et al. An essential role in liver development for transcription factor XBP-1. Genes Dev. 2000, 14, 152–157. [Google Scholar]
- Zhang, K.; Wong, H.N.; Song, B.; Miller, C.N.; Scheuner, D.; Kaufman, R.J. The unfolded protein response sensor IRE1alpha is required at 2 distinct steps in B cell lymphopoiesis. J. Clin. Investig. 2005, 115, 268–281. [Google Scholar] [CrossRef]
- Harding, H.P.; Zeng, H.; Zhang, Y.; Jungries, R.; Chung, P.; Plesken, H.; Sabatini, D.D.; Ron, D. Diabetes mellitus and exocrine pancreatic dysfunction in perk-/- mice reveals a role for translational control in secretory cell survival. Mol. Cell 2001, 7, 1153–1163. [Google Scholar] [CrossRef]
- Gluncic, V.; Moric, M.; Chu, Y.; Hanko, V.; Li, J.; Lukic, I.K.; Lukic, A.; Edassery, S.L.; Kroin, J.S.; Persons, A.L.; et al. In utero Exposure to Anesthetics Alters Neuronal Migration Pattern in Developing Cerebral Cortex and Causes Postnatal Behavioral Deficits in Rats. Cereb. Cortex 2019. [Google Scholar] [CrossRef] [PubMed]
- Ishii, K.; Kubo, K.I.; Nakajima, K. Reelin and Neuropsychiatric Disorders. Front. Cell Neurosci. 2016, 10, 229. [Google Scholar] [CrossRef] [PubMed]
- Fatemi, S.H.; Snow, A.V.; Stary, J.M.; Araghi-Niknam, M.; Reutiman, T.J.; Lee, S.; Brooks, A.I.; Pearce, D.A. Reelin signaling is impaired in autism. Biol. Psychiatry 2005, 57, 777–787. [Google Scholar] [CrossRef] [PubMed]
- Mimura, N.; Yuasa, S.; Soma, M.; Jin, H.; Kimura, K.; Goto, S.; Koseki, H.; Aoe, T. Altered quality control in the endoplasmic reticulum causes cortical dysplasia in knock-in mice expressing a mutant BiP. Mol. Cell Biol. 2008, 28, 293–301. [Google Scholar] [CrossRef]
- Wang, H.; Dong, Y.; Zhang, J.; Xu, Z.; Wang, G.; Swain, C.A.; Zhang, Y.; Xie, Z. Isoflurane induces endoplasmic reticulum stress and caspase activation through ryanodine receptors. Br. J. Anaesth. 2014, 113, 695–707. [Google Scholar] [CrossRef]
- Liu, B.; Xia, J.; Chen, Y.; Zhang, J. Sevoflurane-Induced Endoplasmic Reticulum Stress Contributes to Neuroapoptosis and BACE-1 Expression in the Developing Brain: The Role of eIF2alpha. Neurotox. Res. 2017, 31, 218–229. [Google Scholar] [CrossRef]
- Zhu, G.; Tao, L.; Wang, R.; Xue, Y.; Wang, X.; Yang, S.; Sun, X.; Gao, G.; Mao, Z.; Yang, Q. Endoplasmic reticulum stress mediates distinct impacts of sevoflurane on different subfields of immature hippocampus. J. Neurochem. 2017, 142, 272–285. [Google Scholar] [CrossRef]
- Coghlan, M.; Richards, E.; Shaik, S.; Rossi, P.; Vanama, R.B.; Ahmadi, S.; Petroz, C.; Crawford, M.; Maynes, J.T. Inhalational Anesthetics Induce Neuronal Protein Aggregation and Affect ER Trafficking. Sci. Rep. 2018, 8, 5275. [Google Scholar] [CrossRef]
- Shen, F.Y.; Song, Y.C.; Guo, F.; Xu, Z.D.; Li, Q.; Zhang, B.; Ma, Y.Q.; Zhang, Y.Q.; Lin, R.; Li, Y.; et al. Cognitive Impairment and Endoplasmic Reticulum Stress Induced by Repeated Short-Term Sevoflurane Exposure in Early Life of Rats. Front. Psychiatry 2018, 9, 332. [Google Scholar] [CrossRef]
- Liu, B.; Ou, G.; Chen, Y.; Zhang, J. Inhibition of protein tyrosine phosphatase 1B protects against sevoflurane-induced neurotoxicity mediated by ER stress in developing brain. Brain Res. Bull 2019, 146, 28–39. [Google Scholar] [CrossRef]
- Seo, E.H.; Piao, L.; Park, H.J.; Lee, J.Y.; Sa, M.; Oh, C.S.; Lee, S.H.; Kim, S.H. Impact of general anaesthesia on endoplasmic reticulum stress: Propofol vs. isoflurane. Int. J. Med. Sci. 2019, 16, 1287–1294. [Google Scholar] [CrossRef] [PubMed]
- Eckel, B.; Ohl, F.; Starker, L.; Rammes, G.; Bogdanski, R.; Kochs, E.; Blobner, M. Effects of isoflurane-induced anaesthesia on cognitive performance in a mouse model of Alzheimer’s disease: A randomised trial in transgenic APP23 mice. Eur. J. Anaesthesiol. 2013, 30, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Schoen, J.; Husemann, L.; Tiemeyer, C.; Lueloh, A.; Sedemund-Adib, B.; Berger, K.U.; Hueppe, M.; Heringlake, M. Cognitive function after sevoflurane- vs. propofol-based anaesthesia for on-pump cardiac surgery: A randomized controlled trial. Br. J. Anaesth. 2011, 106, 840–850. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Shan, G.J.; Zhang, Y.X.; Cao, S.J.; Zhu, S.N.; Li, H.J.; Ma, D.; Wang, D.X.; First Study of Perioperative Organ Protection (SPOP1) investigators. Propofol compared with sevoflurane general anaesthesia is associated with decreased delayed neurocognitive recovery in older adults. Br. J. Anaesth. 2018, 121, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Dokkedal, U.; Hansen, T.G.; Rasmussen, L.S.; Mengel-From, J.; Christensen, K. Cognitive Functioning after Surgery in Middle-aged and Elderly Danish Twins. Anesthesiology 2016, 124, 312–321. [Google Scholar] [CrossRef]
- Sprung, J.; Roberts, R.O.; Knopman, D.S.; Price, L.L.; Schulz, H.P.; Tatsuyama, C.L.; Weingarten, T.N.; Schroeder, D.R.; Hanson, A.C.; Petersen, R.C.; et al. Mild Cognitive Impairment and Exposure to General Anesthesia for Surgeries and Procedures: A Population-Based Case-Control Study. Anesth. Analg. 2017, 124, 1277–1290. [Google Scholar] [CrossRef]
- Aiello Bowles, E.J.; Larson, E.B.; Pong, R.P.; Walker, R.L.; Anderson, M.L.; Yu, O.; Gray, S.L.; Crane, P.K.; Dublin, S. Anesthesia Exposure and Risk of Dementia and Alzheimer’s Disease: A Prospective Study. J. Am. Geriatr Soc. 2016, 64, 602–607. [Google Scholar] [CrossRef]
- Chen, C.W.; Lin, C.C.; Chen, K.B.; Kuo, Y.C.; Li, C.Y.; Chung, C.J. Increased risk of dementia in people with previous exposure to general anesthesia: A nationwide population-based case-control study. Alzheimers Dement. 2014, 10, 196–204. [Google Scholar] [CrossRef]
- Istaphanous, G.K.; Howard, J.; Nan, X.; Hughes, E.A.; McCann, J.C.; McAuliffe, J.J.; Danzer, S.C.; Loepke, A.W. Comparison of the neuroapoptotic properties of equipotent anesthetic concentrations of desflurane, isoflurane, or sevoflurane in neonatal mice. Anesthesiology 2011, 114, 578–587. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).