Seminal Plasma Transcriptome and Proteome: Towards a Molecular Approach in the Diagnosis of Idiopathic Male Infertility
Abstract
1. Introduction
2. Methods
2.1. Sources
2.2. Study Selection
3. Results
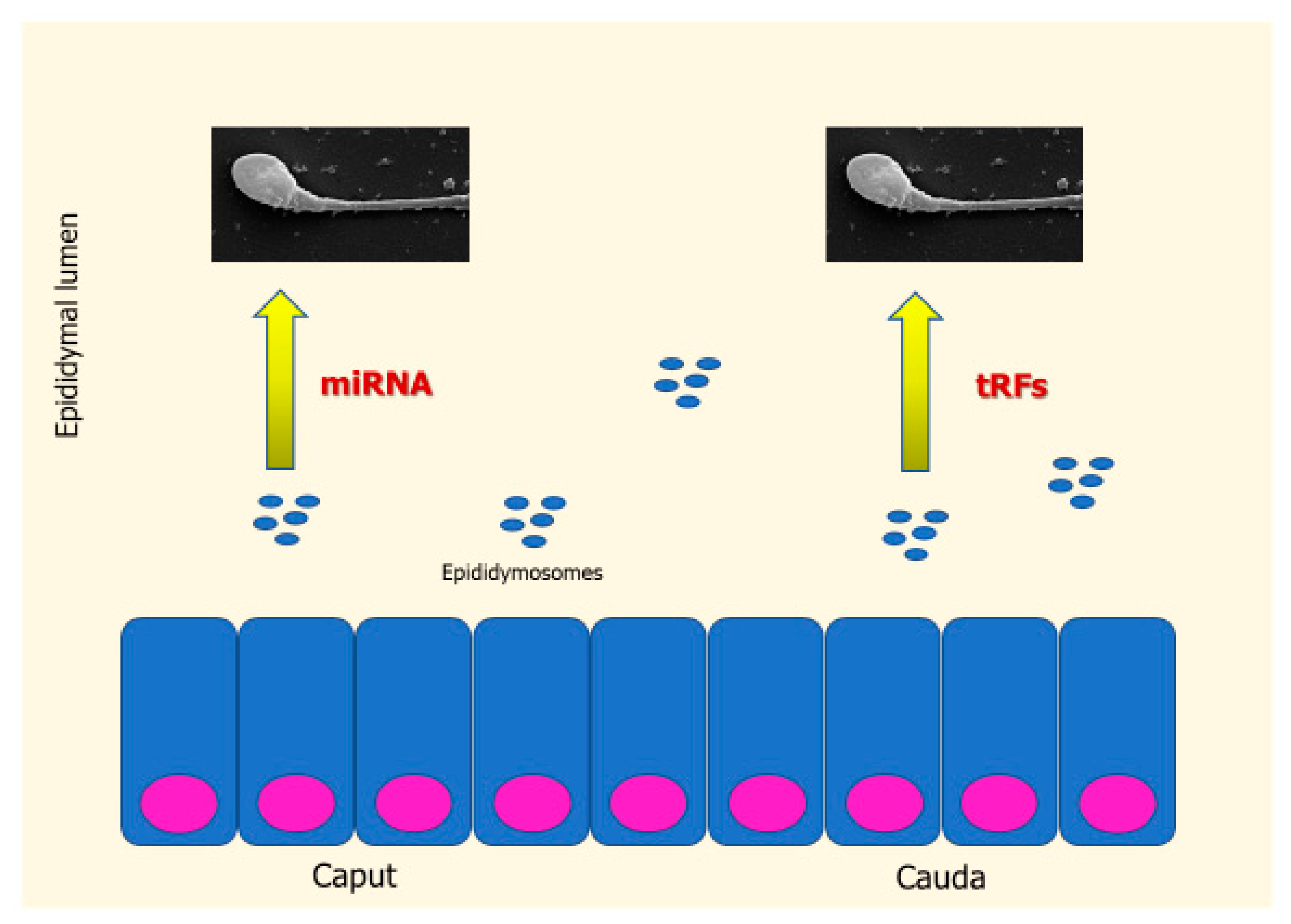
3.1. Seminal Plasma Transcriptome
3.1.1. Implication in Male Infertility
3.1.2. Conclusion
3.2. Seminal Plasma Proteome
3.2.1. Implication in Male Infertility
3.2.2. Conclusion
4. The Future of Seminal Plasma Biomarkers in Male Infertility
Funding
Conflicts of Interest
References
- Smits, R.M.; Mackenzie-Proctor, R.; Yazdani, A.; Stankiewicz, M.T.; Jordan, V.; Showell, M.G. Antioxidants for male subfertility. Cochrane Database Syst. Rev. 2019, 3, CD007411. [Google Scholar] [CrossRef]
- World Health Organization. WHO Laboratory Manual for the Examination and Processing of Human Semen, 5th ed.; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Garrido, N.; Meseguer, M.; Alvarez, J.; Simón, C.; Pellicer, A.; Remohí, J. Relationship among standard semen parameters, glutathione peroxidase/glutathione reductase activity, and mRNA expression and reduced glutathione content in ejaculated spermatozoa from fertile and infertile men. Fertil. Steril. 2004, 82, 1059–1066. [Google Scholar] [CrossRef]
- Condorelli, R.A.; Calogero, A.E.; Russo, G.I.; La Vignera, S. From Spermiogram to Bio-Functional Sperm Parameters: When and Why Request Them? J. Clin. Med. 2020, 9, 406. [Google Scholar] [CrossRef]
- Chen, Q.; Zhao, J.-Y.; Xue, X.; Zhu, G.-X. The association between sperm DNA fragmentation and reproductive outcomes following intrauterine insemination, a meta analysis. Reprod. Toxicol. 2019, 86, 50–55. [Google Scholar] [CrossRef]
- McQueen, D.B.; Zhang, J.; Robins, J.C. Sperm DNA fragmentation and recurrent pregnancy loss: A systematic review and meta-analysis. Fertil. Steril. 2019, 112, 54–60. [Google Scholar] [CrossRef]
- Tan, J.; Taskin, O.; Albert, A.; Bedaiwy, M.A. Association between sperm DNA fragmentation and idiopathic recurrent pregnancy loss: A systematic review and meta-analysis. Reprod. Biomed. Online 2019, 38, 951–960. [Google Scholar] [CrossRef]
- Tüttelmann, F.; Ruckert, C.; Röpke, A. Disorders of spermatogenesis: Perspectives for novel genetic diagnostics after 20 years of unchanged routine. Med Genet. 2018, 30, 12–20. [Google Scholar] [CrossRef]
- Barranco, I.; Padilla, L.; Martinez, C.A.; Álvarez-Rodríguez, M.; Parrilla, I.; Lucas, X.; Ferreira-Dias, G.; Yeste, M.; Rodriguez-Martínez, H.; Roca, J. Seminal Plasma Modulates miRNA Expression by Sow Genital Tract Lining Explants. Biomolecules 2020, 10, 933. [Google Scholar] [CrossRef]
- Sullivan, R.; Mieusset, R. The human epididymis: Its function in sperm maturation. Hum. Reprod. Update 2016, 22, 574–587. [Google Scholar] [CrossRef]
- Trigg, N.A.; Eamens, A.L.; Nixon, B. The contribution of epididymosomes to the sperm small RNA profile. Reproduction 2019, 157, R209–R223. [Google Scholar] [CrossRef]
- Giacone, F.; Cannarella, R.; Mongioì, L.M.; Alamo, A.; Condorelli, R.A.; Calogero, A.E.; La Vignera, S. Epigenetics of Male Fertility: Effects on Assisted Reproductive Techniques. World J. Men Health 2019, 37, 148–156. [Google Scholar] [CrossRef]
- Jodar, M.; Sendler, E.; Krawetz, S.A. The protein and transcript profiles of human semen. Cell Tissue Res. 2015, 363, 85–96. [Google Scholar] [CrossRef]
- Cannarella, R.; Condorelli, R.A.; Mongioì, L.M.; La Vignera, S.; Calogero, A.E. Molecular Biology of Spermatogenesis: Novel Targets of Apparently Idiopathic Male Infertility. Int. J. Mol. Sci. 2020, 21, 1728. [Google Scholar] [CrossRef]
- Guo, L.; Chao, S.-B.; Xiao, L.; Wang, Z.-B.; Meng, T.-G.; Li, Y.-Y.; Han, Z.-M.; Ouyang, Y.-C.; Hou, Y.; Sun, Q.-Y.; et al. Sperm-carried RNAs play critical roles in mouse embryonic development. Oncotarget 2017, 8, 67394–67405. [Google Scholar] [CrossRef]
- Sharma, U.; Conine, C.C.; Shea, J.M.; Boskovic, A.; Derr, A.G.; Bing, X.Y.; Belleannee, C.; Kucukural, A.; Serra, R.W.; Sun, F.; et al. Biogenesis and function of tRNA fragments during sperm maturation and fertilization in mammals. Science 2016, 351, 391–396. [Google Scholar] [CrossRef]
- Hutcheon, K.; McLaughlin, E.A.; Stanger, S.J.; Bernstein, I.R.; Dun, M.D.; Eamens, A.L.; Nixon, B. Analysis of the small non-protein-coding RNA profile of mouse spermatozoa reveals specific enrichment of piRNAs within mature spermatozoa. RNA Biol. 2017, 14, 1776–1790. [Google Scholar] [CrossRef]
- Lu, J.; Gu, H.; Tang, Q.; Wu, W.; Yuan, B.; Guo, D.; Wei, Y.; Sun, H.; Xia, Y.; Ding, H.; et al. Common SNP in hsa-miR-196a-2 increases hsa-miR-196a-5p expression and predisposes to idiopathic male infertility in Chinese Han population. Sci. Rep. 2016, 6, 19825. [Google Scholar] [CrossRef]
- Che, Q.; Wang, W.; Duan, P.; Fang, F.; Liu, C.; Zhou, T.; Li, H.; Xiong, C.; Zhao, K. Downregulation of miR-322 promotes apoptosis of GC-2 cell by targeting Ddx3x. Reprod. Biol. Endocrinol. 2019, 17, 1–9. [Google Scholar] [CrossRef]
- Radtke, A.; Dieckmann, K.-P.; Grobelny, F.; Salzbrunn, A.; Oing, C.; Schulze, W.; Belge, G. Expression of miRNA-371a-3p in seminal plasma and ejaculate is associated with sperm concentration. Andrology 2019, 7, 469–474. [Google Scholar] [CrossRef]
- Almstrup, K.; Lobo, J.; Mørup, N.; Belge, G.; Meyts, E.R.-D.; Looijenga, L.H.J.; Dieckmann, K.P. Application of miRNAs in the diagnosis and monitoring of testicular germ cell tumours. Nat. Rev. Urol. 2020, 17, 201–213. [Google Scholar] [CrossRef]
- Hong, Y.; Wang, C.; Fu, Z.; Liang, H.; Zhang, S.; Lu, M.; Sun, W.; Ye, C.; Zhang, C.-Y.; Zen, K.; et al. Systematic characterization of seminal plasma piRNAs as molecular biomarkers for male infertility. Sci. Rep. 2016, 6, 24229. [Google Scholar] [CrossRef]
- Wu, W.; Hu, Z.; Qin, Y.; Dong, J.; Dai, J.; Lu, C.; Zhang, W.; Shen, H.; Xia, Y.; Wang, X. Seminal plasma microRNAs: Potential biomarkers for spermatogenesis status. Mol. Hum. Reprod. 2012, 18, 489–497. [Google Scholar] [CrossRef]
- Zhu, Y.; Wu, Y.; Jin, K.; Lu, H.; Liu, F.; Guo, Y.; Yan, F.; Shi, W.; Liu, Y.; Cao, X.; et al. Differential proteomic profiling in human spermatozoa that did or did not result in pregnancy via IVF and AID. Proteom. Clin. Appl. 2013, 7, 850–858. [Google Scholar] [CrossRef]
- Gnessi, L.; Scarselli, F.; Minasi, M.G.; Mariani, S.; Lubrano, C.; Basciani, S.; Greco, P.; Watanabe, M.; Franco, G.; Farcomeni, A.; et al. Testicular histopathology, semen analysis and FSH, predictive value of sperm retrieval: Supportive counseling in case of reoperation after testicular sperm extraction (TESE). BMC Urol. 2018, 18, 63. [Google Scholar] [CrossRef]
- Barceló, M.; Mata, A.; Bassas, L.; Larriba, S. Exosomal microRNAs in seminal plasma are markers of the origin of azoospermia and can predict the presence of sperm in testicular tissue. Hum. Reprod. 2018, 33, 1087–1098. [Google Scholar] [CrossRef]
- Wu, W.; Qin, Y.; Li, Z.; Dong, J.; Dai, J.; Lu, C.; Guo, X.; Zhao, Y.; Zhu, Y.; Zhang, W.; et al. Genome-wide microRNA expression profiling in idiopathic non-obstructive azoospermia: Significant up-regulation of miR-141, miR-429 and miR-7-1-3p. Hum. Reprod. 2013, 28, 1827–1836. [Google Scholar] [CrossRef]
- Zhi, E.L.; Liang, G.Q.; Li, P.; Chen, H.X.; Tian, R.H.; Xu, P.; Li, Z. Seminal plasma miR-192a: a biomarker predicting successful resolution of nonobstructive azoospermia following varicocele repair. Asian J. Androl. 2018, 20, 396–399. [Google Scholar] [CrossRef]
- Lv, M.; Zhou, L.; Ge, P.; Li, Y.; Zhang, J.; Zhou, D.-X. Over-expression of hsa_circ_0000116 in patients with non-obstructive azoospermia and its predictive value in testicular sperm retrieval. Andrology 2020. [Google Scholar] [CrossRef]
- Schjenken, J.E.; Zhang, B.; Chan, H.Y.; Sharkey, D.J.; Fullston, T.; Robertson, S.A. miRNA Regulation of Immune Tolerance in Early Pregnancy. Am. J. Reprod. Immunol. 2016, 75, 272–280. [Google Scholar] [CrossRef]
- Wang, C.; Yang, C.; Chen, X.; Yao, B.; Yang, C.; Zhu, C.; Li, L.; Wang, J.; Li, X.; Shao, Y.; et al. Altered profile of seminal plasma microRNAs in the molecular diagnosis of male infertility. Clin. Chem. 2011, 57, 1722–1731. [Google Scholar] [CrossRef]
- Finocchi, F.; Pelloni, M.; Balercia, G.; Pallotti, F.; Radicioni, A.F.; Lenzi, A.; Lombardo, F.; Paoli, D. Seminal plasma miRNAs in Klinefelter syndrome and in obstructive and non-obstructive azoospermia. Mol. Biol. Rep. 2020, 47, 4373–4382. [Google Scholar] [CrossRef] [PubMed]
- Gholami, D.; Yazdi, R.S.; Jami, M.-S.; Ghasemi, S.; Gilani, M.A.S.; Sadeghinia, S.; Jami, M.-S. The expression of Cysteine-Rich Secretory Protein 2 (CRISP2) and miR-582-5p in seminal plasma fluid and spermatozoa of infertile men. Gene 2020, 730, 144261. [Google Scholar] [CrossRef] [PubMed]
- Wasinger, V.C.; Cordwell, S.J.; Poljak, A.; Yan, J.X.; Gooley, A.A.; Wilkins, M.R.; Duncan, M.W.; Harris, R.; Williams, K.L.; Humphery-Smith, I. Progress with gene-product mapping of the Mollicutes:Mycoplasma genitalium. Electrophoresis 1995, 16, 1090–1094. [Google Scholar] [CrossRef] [PubMed]
- Milardi, D.; Grande, G.; Vincenzoni, F.; Castagnola, M.; Marana, R. Proteomics of human seminal plasma: Identification of biomarker candidates for fertility and infertility and the evolution of technology. Mol. Reprod. Dev. 2013, 80, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Camargo, M.; Intasqui, P.; Bertolla, R.P. Understanding the seminal plasma proteome and its role in male fertility. Basic Clin. Androl. 2018, 28, 6. [Google Scholar] [CrossRef]
- Jodar, M.; Soler-Ventura, A.; Oliva, R. Molecular Biology of Reproduction and Development Research Group. Semen proteomics and male infertility. J. Proteom. 2017, 162, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Batruch, I.; Lecker, I.; Kagedan, D.; Smith, C.R.; Mullen, B.J.; Grober, E.; Lo, K.C.; Drabovich, A.P.; Jarvi, K. Proteomic Analysis of Seminal Plasma from Normal Volunteers and Post-Vasectomy Patients Identifies over 2000 Proteins and Candidate Biomarkers of the Urogenital System. J. Proteome Res. 2011, 10, 941–953. [Google Scholar] [CrossRef]
- Pilch, B.; Mann, M. Large-scale and high-confidence proteomic analysis of human seminal plasma. Genome Biol. 2006, 7, R40. [Google Scholar] [CrossRef]
- Rodriguez-Martínez, H.; Kvist, U.; Ernerudh, J.; Sanz, L.; Calvete, J.J. Seminal Plasma Proteins: What Role Do They Play? Am. J. Reprod. Immunol. 2011, 66, 11–22. [Google Scholar] [CrossRef]
- Milardi, D.; Grande, G.; Vincenzoni, F.; Messana, I.; Pontecorvi, A.; De Marinis, L.; Castagnola, M.; Marana, R. Proteomic approach in the identification of fertility pattern in seminal plasma of fertile men. Fertil. Steril. 2012, 97, 67–73. [Google Scholar] [CrossRef]
- Calvete, J.J.; Ensslin, M.; Mburu, J.; Iborra, A.; Martínez, P.; Adermann, K.; Waberski, D.; Sanz, L.; Töpfer-Petersen, E.; Weitze, K.F.; et al. Monoclonal antibodies against boar sperm zona pellucida-binding protein AWN-1. Characterization of a continuous antigenic determinant and immunolocalization of AWN epitopes in inseminated sows. Biol. Reprod. 1997, 57, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, J.; Zhang, H.-R.; Shi, H.-J.; Ma, D.; Zhao, H.; Lin, B.; Li, R. Proteomic analysis of seminal plasma from asthenozoospermia patients reveals proteins that affect oxidative stress responses and semen quality. Asian J. Androl. 2009, 11, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Diamandis, E.P.; Arnett, W.P.; Foussias, G.; Pappas, H.; Ghandi, S.; Melegos, D.N.; Mullen, B.; Yu, H.; Srigley, J.; Jarvi, K. Seminal plasma biochemical markers and their association with semen analysis findings. Urology 1999, 53, 596–603. [Google Scholar] [CrossRef]
- Chen, D.-Y.; Zhu, M.-Y.; Cui, Y.-D.; Huang, T.-H. Relationship between contents of lipocalin-type prostaglandin D synthase on the surface of infertility sperm and in seminal plasma. Biochemistry 2007, 72, 215–218. [Google Scholar] [CrossRef]
- Davalieva, K.; Kiprijanovska, S.; Noveski, P.; Plaseski, T.; Kocevska, B.; Broussard, C.; Plaseska-Karanfilska, D. Proteomic analysis of seminal plasma in men with different spermatogenic impairment. Andrologia 2012, 44, 256–264. [Google Scholar] [CrossRef]
- Drabovich, A.P.; Dimitromanolakis, A.; Saraon, P.; Soosaipillai, A.; Batruch, I.; Mullen, B.; Jarvi, K.; Drabovich, A.P. Differential Diagnosis of Azoospermia with Proteomic Biomarkers ECM1 and TEX101 Quantified in Seminal Plasma. Sci. Transl. Med. 2013, 5, 212ra160. [Google Scholar] [CrossRef]
- Freour, T.; Com, E.; Barrière, P.; Bouchot, O.; Jean, M.; Masson, D.; Pineau, C. Comparative proteomic analysis coupled with conventional protein assay as a strategy to identify predictors of successful testicular sperm extraction in patients with non-obstructive azoospermia. Andrology 2013, 1, 414–420. [Google Scholar] [CrossRef]
- Mostafa, T.; Amer, M.K.; Abdel-Malak, G.; Nsser, T.A.; Zohdy, W.; Ashour, S.; El-Gayar, D.; Awad, H.H. Seminal plasma anti-Müllerian hormone level correlates with semen parameters but does not predict success of testicular sperm extraction (TESE). Asian J. Androl. 2007, 9, 265–270. [Google Scholar] [CrossRef]
- Azpiazu, R.; Amaral, A.; Castillo, J.; Estanyol, J.M.; Guimerà, M.; Ballescà, J.L.; Balasch, J.; Oliva, R. High-throughput sperm differential proteomics suggests that epigenetic alterations contribute to failed assisted reproduction. Hum. Reprod. 2014, 29, 1225–1237. [Google Scholar] [CrossRef]
- Cannarella, R.; Condorelli, R.A.; Duca, Y.; La Vignera, S.; Calogero, A.E. New insights into the genetics of spermatogenic failure: A review of the literature. Qual. Life Res. 2019, 138, 125–140. [Google Scholar] [CrossRef]
- Cannarella, R.; A Condorelli, R.; Paolacci, S.; Barbagallo, F.; Guerri, G.; Bertelli, M.; La Vignera, S.; E Calogero, A. Next-generation sequencing: Toward an increase in the diagnostic yield in patients with apparently idiopathic spermatogenic failure. Asian J. Androl. 2020. [Google Scholar] [CrossRef]
| Authors | Population | Results |
|---|---|---|
| [31] | 118 patients with NOA and 168 fertile controls | Down-regulated miRNAs: hsa-miR-34c; hsa-miR-122; hsa-miR-509-5p |
| [23] | 96 idiopathic infertile patients (48 with oligozoospermia and 48 with NOA) and 48 fertile controls | Up-regulated miRNAs: miR-19b and let-7a |
| [27] | 100 patients with NOA and 100 fertile controls | Up-regulated miRNAs: miR-141, miR-429 and miR-7-1-3p |
| [22] | 211 infertile patients (AT or azoospermia) and 91 fertile controls | Down-regulated piRNAs: piR-31068, piR-31925, piR-43771, piR-43773 and piR-31098 |
| [18] | 1378 patients with idiopathic infertility and 486 fertile controls | Down-regulated miRNA: miR-196a-5p |
| [26] | 14 patients with NOA, 13 with OA and 9 normozoospermic controls | Down-regulated miRNAs (NOA): hsa-miR-202-3p; hsa-miR-514a-3p; hsa-miR-202-5p; hsa-miR-510-3-5p; hsa-miR-509-3-5p; hsa-miR-510-5p; hsa-miR-513c-5p; hsa-miR-518e-3p; hsa-miR-508-5p; hsa-miR-520h; hsa-miR-9-3p; hsa-miR-506-3p; hsa-miR-383-5p; hsa-miR-34c-5p; hsa-miR-517c-3p; hsa-miR-873-5p; hsa-miR-34b-5p; hsa-miR-513a-3p; hsa-miR-52; hsa-miR-452-5p; hsa-miR-122-5p; hsa-miR-449a; hsa-miR-449a-5p; hsa-miR-455-5p; has-miR-9-5p; 132-5p; hsa hsa-miR-203a Up-regulated miRNAs (NOA): hsa-miR-363-5p; hsa-miR-365a-3p; hsa-miR-550a-5p; 423-5p; hsa-miR-153-3p; hsa-miR-196b-3p; hsa-miR-96-5p; hsa-let-7-1-3p Down-regulated miRNAs (OA): hsa-miR-202-3p; hsa-miR-514a-3p; hsa-miR-202-5p; hsa-miR-510-3-5p; hsa-miR-509-3-5p; hsa-miR-510-5p; hsa-miR-513c-5p; hsa-miR-518e-3p; hsa-miR-508-5p; hsa-miR-520h; hsa-miR-9-3p; hsa-miR-506-3p; hsa-miR-383-5p; hsa-miR-34c-5p; hsa-miR-517-3p; hsa-miR-873-5p; hsa -miR-34b-5p; hsa -miR-513-3p; hsa -miR-52; hsa -miR-452-5p; hsa -miR-122-5p; hsa -miR-449a; hsa -miR-449a-5p; hsa -miR-455-5p; hsa -miR-819b; hsa -miR-890; hsa -miR-34c-3p; hsa -miR-891a-5p; hsa -miR-888-5p; hsa -miR-124-3p; hsa -miR-892a; hsa -miR-551b-3p; hsa -miR-424-5p; hsa -miR-181b-5p; hsa -miR-31-3p; hsa -miR-181a-5p; hsa -miR-31-5p; hsa -miR-10b-3p; hsa -miR-222-3p; hsa -miR-455-3p; hsa -miR-205-5p; hsa -miR-182-3p; hsa-miR-95-3p Up-regulated miRNAs (OA): hsa -miR-363-3p; hsa -miR-365a-3p; hsa -miR-29a-3p; hsa-miR-296-5p; hsa-miR-23b-5p; hsa-miR-21-3p; hsa-miR-193a-3p; hsa-miR-29c-3p; hsa-miR-361-3p |
| [19] | 30 infertile patients with high sperm DFI and 30 fertile controls | Up-regulated miRNA: miR-424 |
| [32] | 40 patients with KS, 60 with NOA, 60 OA and 40 normozoospermic controls | Down-regulated miRNAs: has-miR-509-5p; has-miR-122-5p; has-miR-34b-3p; has-miR-34c-5p |
| [33] | 17 patients with AT, 15 patients with teratozoospermia, 17 patients with AT, 18 normozoospermic infertile patients | Up-regulated miRNA: miRNA-582-5p (teratozoospermia and AT) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cannarella, R.; Barbagallo, F.; Crafa, A.; La Vignera, S.; Condorelli, R.A.; Calogero, A.E. Seminal Plasma Transcriptome and Proteome: Towards a Molecular Approach in the Diagnosis of Idiopathic Male Infertility. Int. J. Mol. Sci. 2020, 21, 7308. https://doi.org/10.3390/ijms21197308
Cannarella R, Barbagallo F, Crafa A, La Vignera S, Condorelli RA, Calogero AE. Seminal Plasma Transcriptome and Proteome: Towards a Molecular Approach in the Diagnosis of Idiopathic Male Infertility. International Journal of Molecular Sciences. 2020; 21(19):7308. https://doi.org/10.3390/ijms21197308
Chicago/Turabian StyleCannarella, Rossella, Federica Barbagallo, Andrea Crafa, Sandro La Vignera, Rosita A. Condorelli, and Aldo E. Calogero. 2020. "Seminal Plasma Transcriptome and Proteome: Towards a Molecular Approach in the Diagnosis of Idiopathic Male Infertility" International Journal of Molecular Sciences 21, no. 19: 7308. https://doi.org/10.3390/ijms21197308
APA StyleCannarella, R., Barbagallo, F., Crafa, A., La Vignera, S., Condorelli, R. A., & Calogero, A. E. (2020). Seminal Plasma Transcriptome and Proteome: Towards a Molecular Approach in the Diagnosis of Idiopathic Male Infertility. International Journal of Molecular Sciences, 21(19), 7308. https://doi.org/10.3390/ijms21197308








