Angiopoietin-Like Protein 4 Overexpression in Visceral Adipose Tissue from Obese Subjects with Impaired Glucose Metabolism and Relationship with Lipoprotein Lipase
Abstract
1. Introduction
2. Results
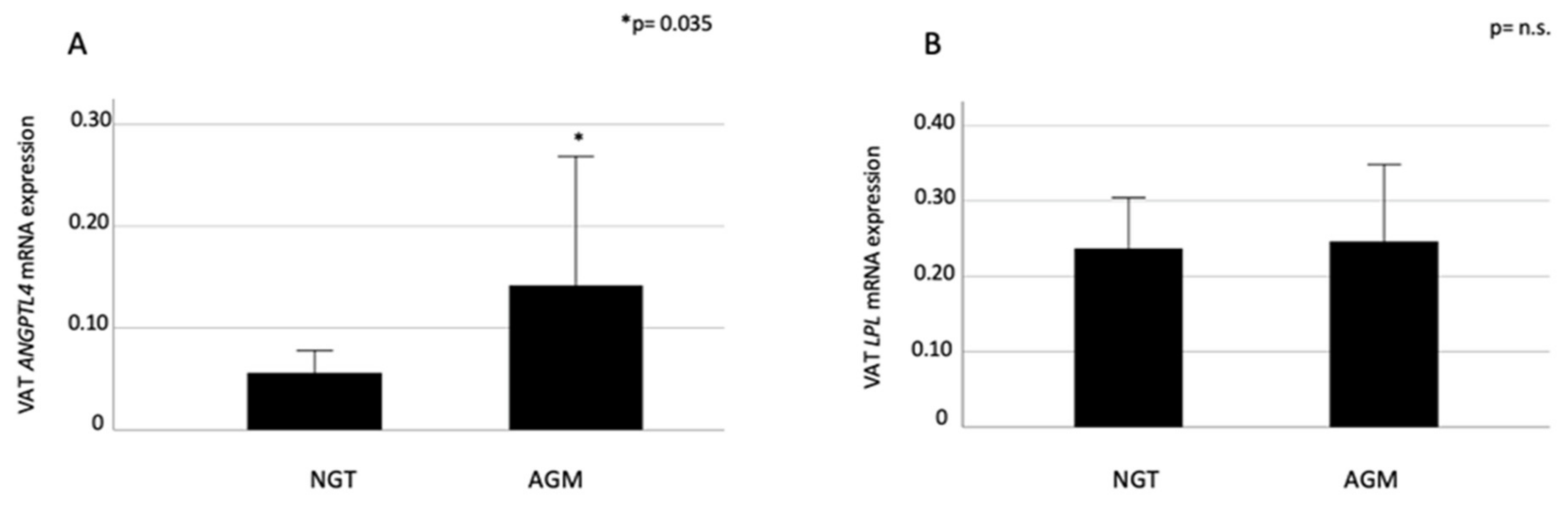
2.1. VAT ANGPTL4 and LPL expression in Relation to Glucose Metabolism
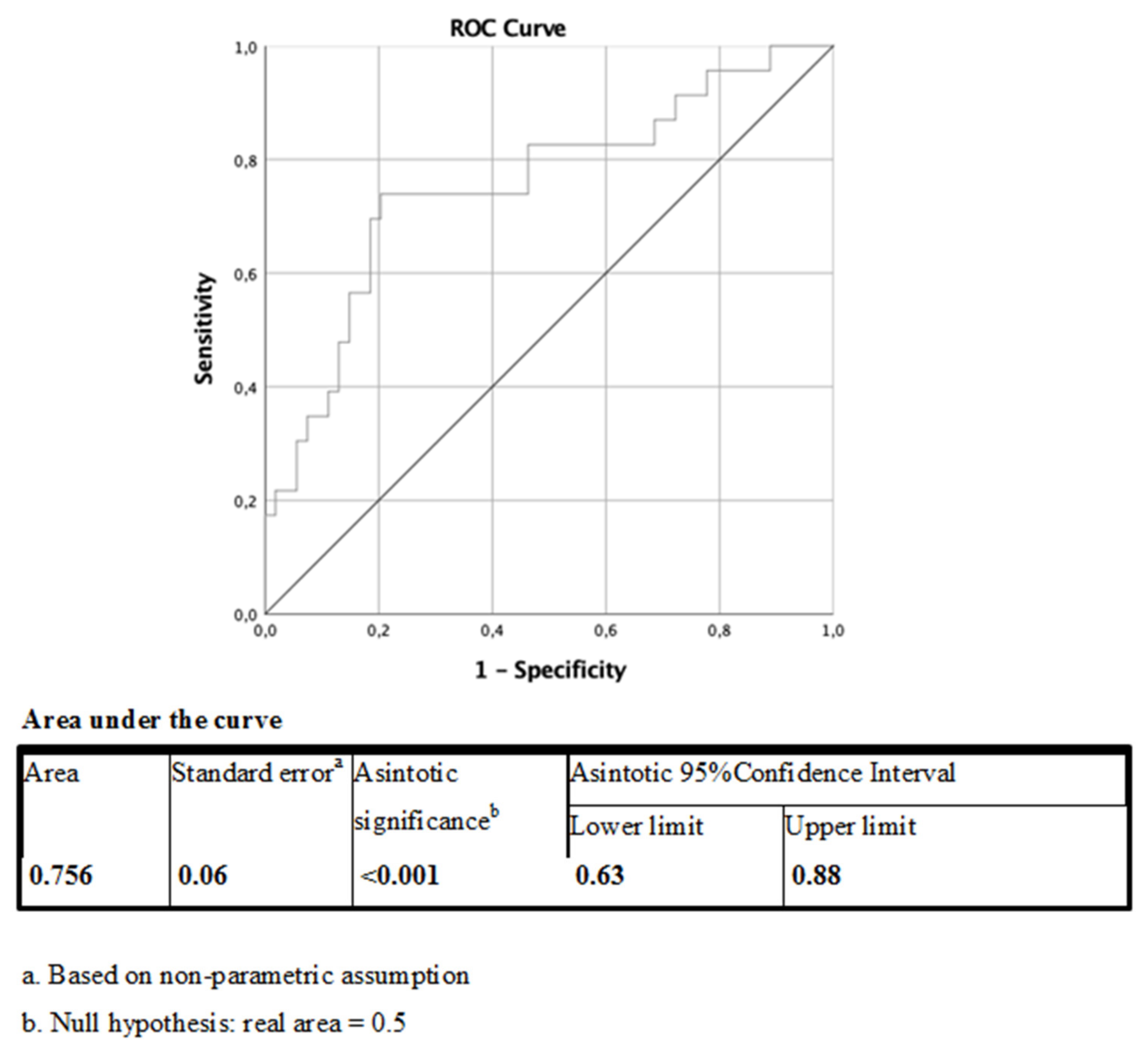
2.2. Plasma ANGPTL4 Concentrations and Relation to ANGPTL4 and LPL Expression, and Glucose Metabolism
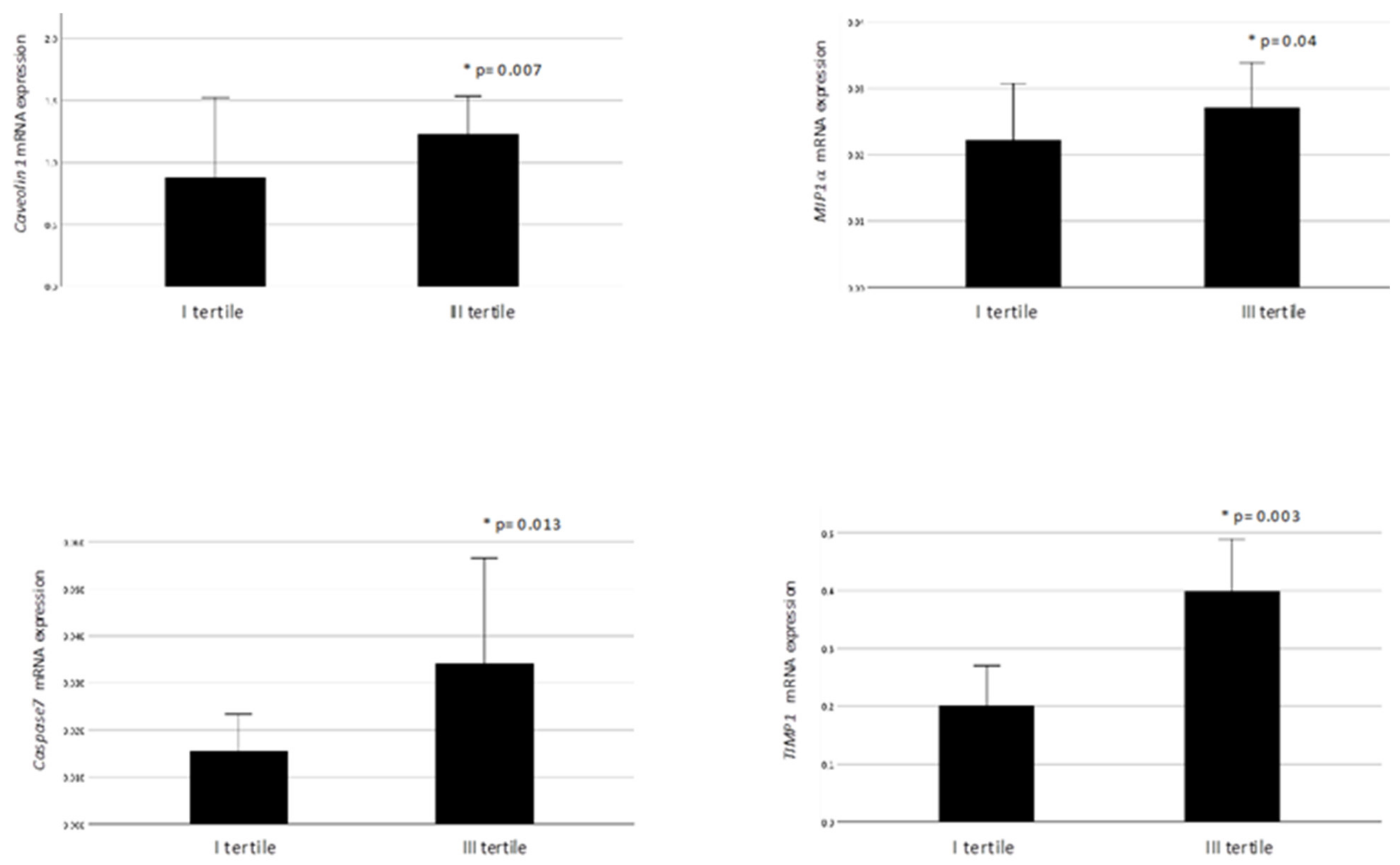
2.3. VAT ANGPTL4 and LPL Expression in Relation to VAT Homeostasis
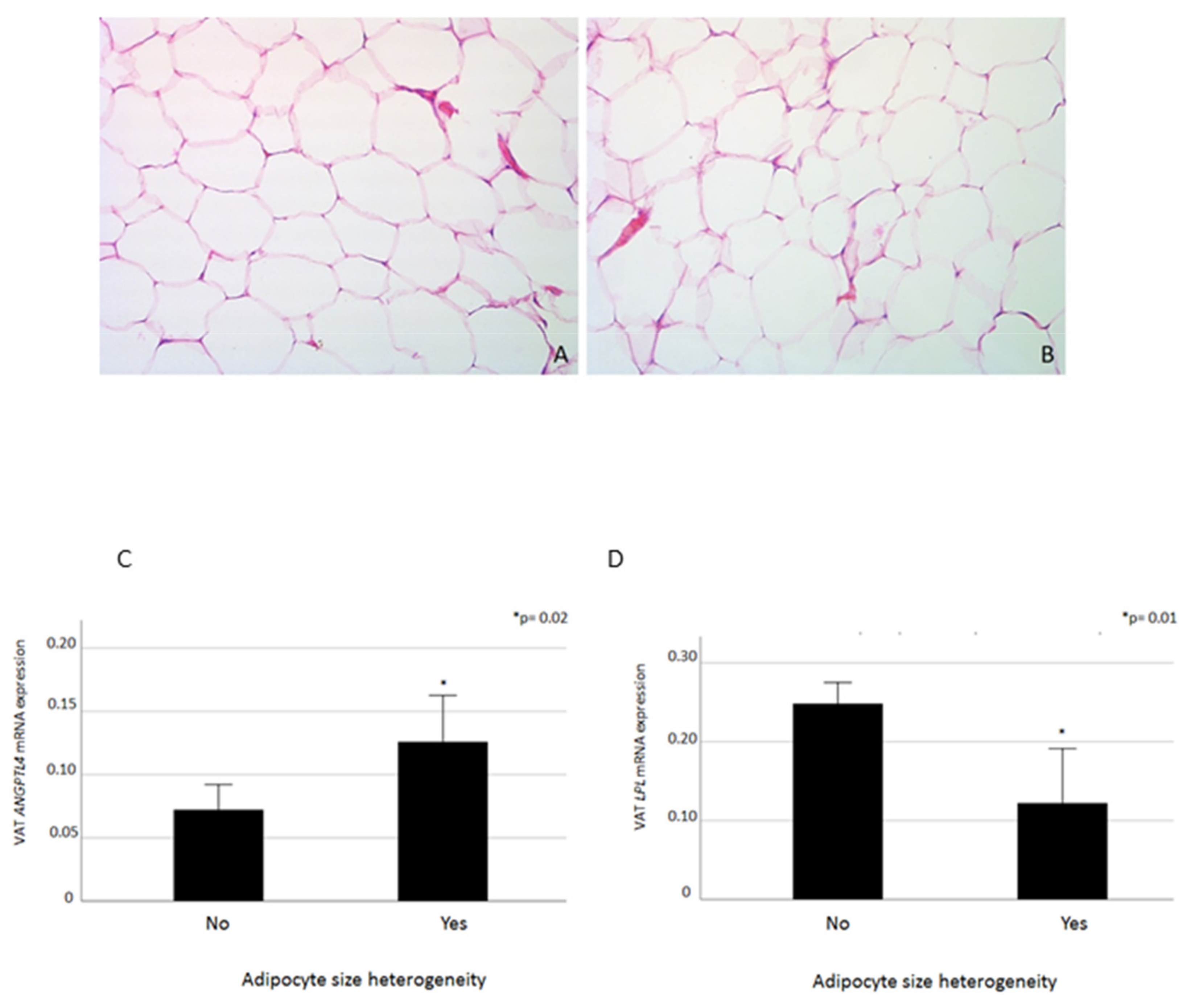
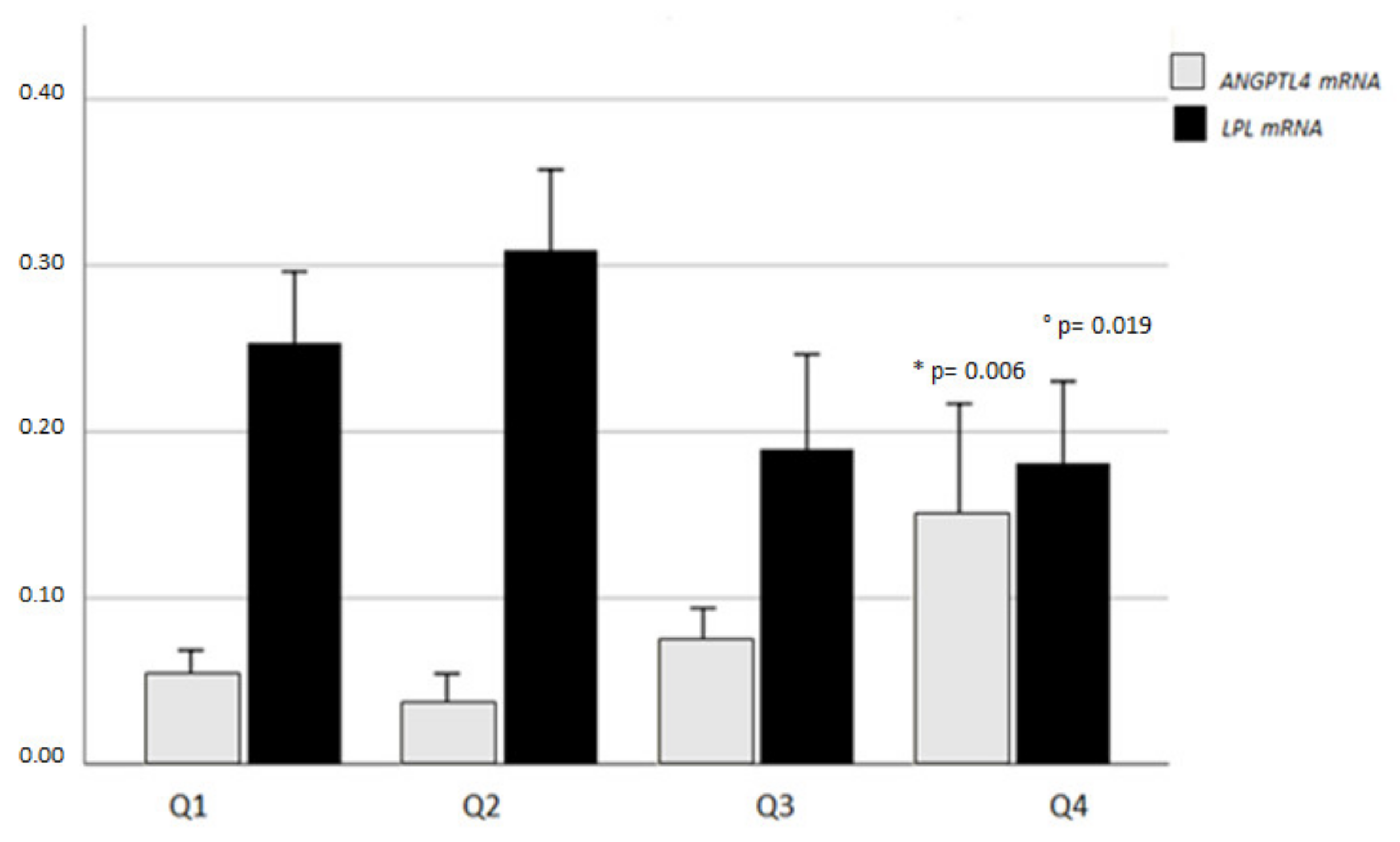
2.4. VAT ANGPTL4 and LPL mRNA Levels in Relation to Adipocyte Heterogeneity
2.5. VAT ANGPTL4, LPL, and VDR mRNA Expression
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Laboratory Tests
4.3. Vat and Liver Biopsy Collection, Immunohistochemistry and Gene Expression Analyses
4.4. Statistics
4.5. Ethics Standards
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AGM | abnormal glucose metabolism |
| ALT | alanine aminotransferase |
| ANGPTL | angiopoietin-like protein |
| AST | aspartate aminotransferase |
| AT | adipose tissue |
| BMI | body mass index |
| DBP | diastolic blood pressure |
| FBG | fasting blood glucose |
| FBI | fasting blood insulin |
| FFA | free fatty acid |
| GGT | gamma-glutamyl transpeptidase |
| HbA1c | glycosylated hemoglobin |
| HDL | high-density lipoprotein |
| IFG | impaired fasting glucose |
| IGT | impaired glucose tolerance |
| LPL | lipoprotein lipase |
| MS | metabolic syndrome |
| NAFLD | nonalcoholic fatty liver disease |
| NAS | NAFLD Activity Score |
| NASH | nonalcoholic steatohepatitis |
| NGT | normal glucose tolerance |
| SBP | systolic blood pressure |
| T2D | type 2 diabetes |
| VDR | vitamin D receptor |
References
- Dijk, W.; Kersten, S. Regulation of lipid metabolism by angiopoietin-like proteins. Curr. Opin. Lipidol. 2016, 27, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Kersten, S. New insights into angiopoietin-like proteins in lipid metabolism and cardiovascular disease risk. Curr. Opin. Lipidol. 2019, 30, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Sonnenburg, W.K.; Yu, D.; Lee, E.-C.; Xiong, W.; Gololobov, G.; Key, B.; Gay, J.; Wilganowski, N.; Hu, Y.; Zhao, S.; et al. GPIHBP1 stabilizes lipoprotein lipase and prevents its inhibition by angiopoietin-like 3 and angiopoietin-like 4. J. Lipid Res. 2009, 50, 2421–2429. [Google Scholar] [CrossRef] [PubMed]
- Romeo, S.; Pennacchio, L.A.; Fu, Y.; Boerwinkle, E.; Tybjaerg-Hansen, A.; Hobbs, H.H.; Cohen, J.C. Population-based resequencing of ANGPTL4 uncovers variations that reduce triglycerides and increase HDL. Nat. Genet. 2007, 39, 513–516. [Google Scholar] [CrossRef] [PubMed]
- Romeo, S.; Yin, W.; Kozlitina, J.; Pennacchio, L.A.; Boerwinkle, E.; Hobbs, H.H.; Cohen, J.C. Rare loss-of-function mutations in ANGPTL family members contribute to plasma triglyceride levels in humans. J. Clin. Investig. 2009, 119, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Bailetti, D.; Bertoccini, L.; Mancina, R.M.; Barchetta, I.; Capoccia, D.; Cossu, E.; Pujia, A.; Lenzi, A.; Leonetti, F.; Cavallo, M.G.; et al. ANGPTL4 gene E40K variation protects against obesity-associated dyslipidemia in participants with obesity. Obes. Sci. Pract. 2018, 5, 83–90. [Google Scholar] [CrossRef]
- Gusarova, V.; O’Dushlaine, C.; Teslovich, T.M.; Benotti, P.N.; Mirshahi, T.; Gottesman, O.; Van Hout, C.V.; Murray, M.F.; Mahajan, A.; Nielsen, J.B.; et al. Genetic inactivation of ANGPTL4 improves glucose homeostasis and is associated with reduced risk of diabetes. Nat. Commun. 2018, 9, 2252. [Google Scholar] [CrossRef]
- Ruscica, M.; Zimetti, F.; Adorni, M.P.; Sirtori, C.R.; Lupo, M.G.; Ferri, N. Pharmacological aspects of ANGPTL3 and ANGPTL4 inhibitors: New therapeutic approaches for the treatment of atherogenic dyslipidemia. Pharmacol. Res. 2020, 153, 104653. [Google Scholar] [CrossRef]
- Mandard, S.; Zandbergen, F.; Escher, P.; Patsouris, D.; Koenig, W.; Bakker, A.; Veenman, F.; Wahli, W.; Kersten, S.; Tan, N.S.; et al. The Direct Peroxisome Proliferator-activated Receptor Target Fasting-induced Adipose Factor (FIAF/PGAR/ANGPTL4) Is Present in Blood Plasma as a Truncated Protein That Is Increased by Fenofibrate Treatment. J. Biol. Chem. 2004, 279, 34411–34420. [Google Scholar] [CrossRef]
- Xu, A.; Lam, M.C.; Chan, K.W.; Wang, Y.; Zhang, J.; Hoo, R.L.C.; Xu, J.Y.; Chen, B.; Chow, W.-S.; Tso, A.W.K.; et al. Angiopoietin-like protein 4 decreases blood glucose and improves glucose tolerance but induces hyperlipidemia and hepatic steatosis in mice. Proc. Natl. Acad. Sci. USA 2005, 102, 6086–6091. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, L.-M.; Wei, L.; Ye, W.-W.; Meng, X.-Y.; Chen, F.; Xiao, Q.; Chen, J.-Y.; Zhou, Y. Angiopoietin-like protein 4 improves glucose tolerance and insulin resistance but induces liver steatosis in high-fat-diet mice. Mol. Med. Rep. 2016, 14, 3293–3300. [Google Scholar] [CrossRef] [PubMed]
- Janssen, A.W.F.; Katiraei, S.; Bartosinska, B.; Eberhard, D.; Van Dijk, K.W.; Kersten, S. Loss of angiopoietin-like 4 (ANGPTL4) in mice with diet-induced obesity uncouples visceral obesity from glucose intolerance partly via the gut microbiota. Diabetologia 2018, 61, 1447–1458. [Google Scholar] [CrossRef]
- Arca, M.; Minicocci, I.; Maranghi, M. The angiopoietin-like protein 3: A hepatokine with expanding role in metabolism. Curr. Opin. Lipidol. 2013, 24, 313–320. [Google Scholar] [CrossRef]
- Barchetta, I.; Cimini, F.A.; Chiappetta, C.; Bertoccini, L.; Ceccarelli, V.; Capoccia, D.; Gaggini, M.; Di Cristofano, C.; Rocca, C.D.; Silecchia, G.; et al. Relationship between hepatic and systemic angiopoietin-like 3, hepatic Vitamin D receptor expression and NAFLD in obesity [published online ahead of print, 8 June 2020]. Liver Int. 2020, 40, 2139–2147. [Google Scholar] [CrossRef]
- Dijk, W.; Ruppert, P.M.; Oost, L.J.; Kersten, S. Angiopoietin-like 4 promotes the intracellular cleavage of lipoprotein lipase by PCSK3/furin in adipocytes. J. Biol. Chem. 2018, 7, 14134–14145. [Google Scholar] [CrossRef]
- Dijk, W.; Schutte, S.; Aarts, E.O.; Janssen, I.M.; Afman, L.; Kersten, S. Regulation of angiopoietin-like 4 and lipoprotein lipase in human adipose tissue. J. Clin. Lipidol. 2018, 12, 773–783. [Google Scholar] [CrossRef]
- Catoire, M.; Alex, S.; Paraskevopulos, N.; Mattijssen, F.; Gogh, I.E.-V.; Schaart, G.; Jeppesen, J.; Kneppers, A.; Mensink, M.; Voshol, P.J.; et al. Fatty acid-inducible ANGPTL4 governs lipid metabolic response to exercise. Proc. Natl. Acad. Sci. USA 2014, 111, E1043–E1052. [Google Scholar] [CrossRef]
- Cushing, E.M.; Chi, X.; Sylvers, K.; Shetty, S.K.; Potthoff, M.J.; Davies, B.S.J. Angiopoietin-like 4 directs uptake of dietary fat away from adipose during fasting. Mol. Metab. 2017, 6, 809–818. [Google Scholar] [CrossRef]
- Kroupa, O.; Vorrsjö, E.; Stienstra, R.; Mattijssen, F.; Nilsson, S.K.; Sukonina, V.; Kersten, S.; Olivecrona, G.; Olivecrona, T. Linking nutritional regulation of Angptl4, Gpihbp1, and Lmf1 to lipoprotein lipase activity in rodent adipose tissue. BMC Physiol. 2012, 12, 13. [Google Scholar] [CrossRef]
- Xu, Y.; Lou, Y.; Kong, J. VDR regulates energy metabolism by modulating remodeling in adipose tissue. Eur. J. Pharmacol. 2019, 865, 172761. [Google Scholar] [CrossRef]
- Bozic, M.; Guzmán, C.; Benet, M.; Sánchez-Campos, S.; García-Monzón, C.; Garí, E.; Gatius, S.; Valdivielso, J.M.; Jover, R. Hepatocyte vitamin D receptor regulates lipid metabolism and mediates experimental diet-induced steatosis. J. Hepatol. 2016, 65, 748–757. [Google Scholar] [CrossRef]
- Wong, K.E.; Kong, J.; Zhang, W.; Szeto, F.L.; Ye, H.; Deb, D.K.; Brady, M.J.; Li, Y.C. Targeted Expression of Human Vitamin D Receptor in Adipocytes Decreases Energy Expenditure and Induces Obesity in Mice. J. Boil. Chem. 2011, 286, 33804–33810. [Google Scholar] [CrossRef]
- Jahn, D.; Dorbath, D.; Schilling, A.-K.; Gildein, L.; Meier, C.; Vuille-Dit-Bille, R.N.; Schmitt, J.; Kraus, D.; Fleet, J.C.; Hermanns, H.M.; et al. Intestinal vitamin D receptor modulates lipid metabolism, adipose tissue inflammation and liver steatosis in obese mice. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2019, 1865, 1567–1578. [Google Scholar] [CrossRef]
- Szántó, M.; Bai, P. The role of ADP-ribose metabolism in metabolic regulation, adipose tissue differentiation, and metabolism. Genes Dev. 2020, 34, 321–340. [Google Scholar] [CrossRef]
- Cimini, F.A.; Barchetta, I.; Ciccarelli, G.; Leonetti, F.; Silecchia, G.; Chiappetta, C.; Di Cristofano, C.; Capoccia, D.; Bertoccini, L.; Ceccarelli, V.; et al. Adipose tissue remodelling in obese subjects is a determinant of presence and severity of fatty liver disease [published online ahead of print, 29 May 2020]. Diabetes/Metab. Res. Rev. 2020, e3358. [Google Scholar] [CrossRef]
- Barchetta, I.; Angelico, F.; Del Ben, M.; Di Martino, M.; Cimini, F.A.; Bertoccini, L.; Polimeni, L.; Catalano, C.; Fraioli, A.; Del Vescovo, R.; et al. Phenotypical heterogeneity linked to adipose tissue dysfunction in patients with Type 2 diabetes. Clin. Sci. 2016, 130, 1753–1762. [Google Scholar] [CrossRef]
- Zatterale, F.; Longo, M.; Naderi, J.; Raciti, G.A.; Desiderio, A.; Miele, C.; Beguinot, F. Chronic Adipose Tissue Inflammation Linking Obesity to Insulin Resistance and Type 2 Diabetes. Front. Physiol. 2020, 10, 1607. [Google Scholar] [CrossRef]
- Greenwood, M.R. The relationship of enzyme activity to feeding behaviour in rats: Lipoprotein lipase as the metabolic gatekeeper. Int. J. Obes. 1985, 9 (Suppl. 1), 67–70. [Google Scholar]
- Gonzales, A.M.; Orlando, R.A. Role of adipocyte-derived lipoprotein lipase in adipocyte hypertrophy. Nutr. Metab. 2007, 4, 22. [Google Scholar] [CrossRef]
- Koike, T.; Liang, J.; Wang, X.; Ichikawa, T.; Shiomi, M.; Liu, G.; Sun, H.; Kitajima, S.; Morimoto, M.; Watanabe, T.; et al. Overexpression of Lipoprotein Lipase in Transgenic Watanabe Heritable Hyperlipidemic Rabbits Improves Hyperlipidemia and Obesity. J. Biol. Chem. 2003, 279, 7521–7529. [Google Scholar] [CrossRef]
- Kern, P.A.; Ong, J.M.; Saffari, B.; Carty, J. The Effects of Weight Loss on the Activity and Expression of Adipose-Tissue Lipoprotein Lipase in Very Obese Humans. New Engl. J. Med. 1990, 322, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- McCulloch, L.J.; Bramwell, L.R.; Knight, B.; Kos, K. Circulating and tissue specific transcription of angiopoietin-like protein 4 in human Type 2 diabetes. Metabolism 2020, 106, 154192. [Google Scholar] [CrossRef] [PubMed]
- Geldenhuys, W.J.; Lin, L.; Darvesh, A.S.; Sadana, P. Emerging strategies of targeting lipoprotein lipase for metabolic and cardiovascular diseases. Drug Discov. Today 2016, 22, 352–365. [Google Scholar] [CrossRef] [PubMed]
- Trayhurn, P. Hypoxia and adipocyte physiology: Implications for adipose tissue dysfunction in obesity. Ann. Rev. Nutr. 2014, 34, 207–236. [Google Scholar] [CrossRef]
- Hosogai, N.; Fukuhara, A.; Oshima, K.; Miyata, Y.; Tanaka, S.; Segawa, K.; Furukawa, S.; Tochino, Y.; Komuro, R.; Matsuda, M.; et al. Adipose Tissue Hypoxia in Obesity and Its Impact on Adipocytokine Dysregulation. Diabetes 2007, 56, 901–911. [Google Scholar] [CrossRef]
- Strissel, K.J.; Stancheva, Z.; Miyoshi, H.; Perfield, J.W.; DeFuria, J.; Jick, Z.; Greenberg, A.S.; Obin, M.S. Adipocyte Death, Adipose Tissue Remodeling, and Obesity Complications. Diabetes 2007, 56, 2910–2918. [Google Scholar] [CrossRef]
- González-Muniesa, P.; De Oliveira, C.; De Heredia, F.P.; Thompson, M.P.; Trayhurn, P. Fatty Acids and Hypoxia Stimulate the Expression and Secretion of the Adipokine ANGPTL4 (Angiopoietin-Like Protein 4/ Fasting-Induced Adipose Factor) by Human Adipocytes. J. Nutr. Nutr. 2011, 4, 146–153. [Google Scholar] [CrossRef]
- Tjeerdema, N.; Georgiadi, A.; Jonker, J.T.; Van Glabbeek, M.; Dehnavi, R.A.; Tamsma, J.T.; Smit, J.W.A.; Kersten, S.; Rensen, P.C.N. Inflammation increases plasma angiopoietin-like protein 4 in patients with the metabolic syndrome and type 2 diabetes. BMJ Open Diabetes Res. Care 2014, 2, e000034. [Google Scholar] [CrossRef]
- Lu, B.; Moser, A.; Shigenaga, J.K.; Grunfeld, C.; Feingold, K.R. The acute phase response stimulates the expression of angiopoietin like protein 4. Biochem. Biophys. Res. Commun. 2010, 391, 1737–1741. [Google Scholar] [CrossRef]
- Aryal, B.; Singh, A.K.; Zhang, X.; Varela, L.; Rotllan, N.; Goedeke, L.; Chaube, B.; Camporez, J.-P.; Vatner, D.F.; Horvath, T.L.; et al. Absence of ANGPTL4 in adipose tissue improves glucose tolerance and attenuates atherogenesis. JCI Insight 2018, 3, 97918. [Google Scholar] [CrossRef]
- Neeland, I.J.; Ross, R.; Després, J.-P.; Matsuzawa, Y.; Yamashita, S.; Shai, I.; Seidell, J.; Magni, P.; Santos, R.D.; Arsenault, B.; et al. Visceral and ectopic fat, atherosclerosis, and cardiometabolic disease: A position statement. Lancet Diabetes Endocrinol. 2019, 7, 715–725. [Google Scholar] [CrossRef]
- Sironi, A.M.; Petz, R.; De Marchi, D.; Buzzigoli, E.; Ciociaro, D.; Positano, V.; Lombardi, M.; Ferrannini, E.; Gastaldelli, A. Impact of increased visceral and cardiac fat on cardiometabolic risk and disease. Diabet. Med. 2012, 29, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, N.; Antoniou, S.A.; Batterham, R.L.; Busetto, L.; Godoroja, D.; Iossa, A.; Carrano, F.M.; Agresta, F.; Alarçon, I.; Azran, C.; et al. Clinical practice guidelines of the European Association for Endoscopic Surgery (EAES) on bariatric surgery: Update 2020 endorsed by IFSO-EC, EASO and ESPCOP. Surg. Endosc. 2020, 34, 2332–2358. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association 1. Improving Care and Promoting Health in Populations: Standards of Medical Care in Diabetes—2020. Diabetes Care 2019, 43 (Suppl. 1), S7–S13. [Google Scholar] [CrossRef]
- Grundy, S.M.; Cleeman, J.I.; Daniels, S.R.; Donato, K.A.; Eckel, R.; Franklin, B.A.; Gordon, D.J.; Krauss, R.M.; Savage, P.J.; Smith, S.C.; et al. Diagnosis and Management of the Metabolic Syndrome. An American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Executive Summary. Circulation 2005, 112, 2735–2752. [Google Scholar] [CrossRef]
- Barchetta, I.; Cimini, F.A.; Capoccia, D.; Bertoccini, L.; Ceccarelli, V.; Chiappetta, C.; Leonetti, F.; Di Cristofano, C.; Silecchia, G.; Orho-Melander, M.; et al. Neurotensin Is a Lipid-Induced Gastrointestinal Peptide Associated with Visceral Adipose Tissue Inflammation in Obesity. Nutrients 2018, 10, 526. [Google Scholar] [CrossRef]
- Brunt, E.M.; Janney, C.G.; Bisceglie, A.M.; Neuschwander-Tetri, B.A.; Bacon, B.R. Nonalcoholic steatohepatitis: A proposal for grading and staging the histological lesions. Am. J. Gastroenterol. 1999, 94, 2467–2474. [Google Scholar] [CrossRef] [PubMed]
- Kleiner, D.E.; Brunt, E.M.; Van Natta, M.; Behling, C.; Contos, M.J.; Cummings, O.W.; Ferrell, L.D.; Liu, Y.-C.; Torbenson, M.S.; Unalp-Arida, A.; et al. Nonalcoholic Steatohepatitis Clinical Research Network. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005, 41, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
| All Participants; n = 88 | NGT; n= 65 | AGM; n = 23 | p-value | |
|---|---|---|---|---|
| Age (years) | 44.1 ± 9.9 | 42.4 ± 9.8 | 48.4 ± 8.9 | 0.017 |
| Sex (male/female) | 26/62 | 17/48 | 9/14 | 0.131 |
| BMI (kg/m2) | 42.4 ± 4.8 | 42 ± 4.9 | 43.5 ± 4.7 | 0.24 |
| Waist circumference (cm) | 126.2 ± 13 | 125.4 ± 14.3 | 128.9 ± 9.3 | 0.36 |
| SBP (mmHg) | 129.4 ± 14 | 129.2 ± 13.4 | 130.7 ± 15.7 | 0.67 |
| DBP (mmHg) | 84.2 ± 14 | 84.3 ± 7.7 | 84 ± 23 | 0.92 |
| Total cholesterol (mg/dl) | 194.5 ± 32.2 | 196.3 ± 31 | 188.3 ± 32 | 0.34 |
| HDL-C (mg/dl) | 47.5 ± 10.9 | 49.3 ± 11 | 42.9 ± 9.8 | 0.02 |
| LDL-C (mg/dl) | 118.5 ± 31.2 | 120.6 ± 31 | 111.7 ± 30 | 0.28 |
| Triglycerides (mg/dl) | 138.8 ± 62 | 130.5 ± 58.7 | 158 ± 70.3 | 0.039 |
| FBG (mg/dl) | 100 ± 23 | 89.9 ± 5.3 | 126.8 ± 29.2 | <0.001 |
| HbA1c - mmol/mol; % | 38.3 ± 8.8; 5.6 ± 0.7 | 35.2 ± 3; 5.3 ± 0.3 | 49 ± 13.8; 6.1 ± 1 | 0.003 |
| AST (IU/l) | 27.6 ± 14.4 | 25.5 ± 11.5 | 33 ± 18.9 | 0.06 |
| ALT (IU/l) | 36.2 ± 25 | 31.8 ± 23 | 46.6 ± 28 | 0.005 |
| GGT (IU/l) | 30 ± 25.6 | 25.5 ± 13 | 42.8 ± 42.6 | 0.06 |
| Uric acid (mg/dl) | 5.6 ± 1.4 | 5.4 ± 1 | 6.0 ± 1.95 | 0.56 |
| Serum creatinine (mg/dl) | 0.76 ± 0.1 | 0.74 ± 0.1 | 0.79 ± 0.2 | 0.28 |
| Plasma ANGPTL4 (pg/mL) | 174 ± 64.8 | 175.6 ± 67 | 160.2 ± 56.8 | 0.33 |
| ANGPTL4 mRNA | LPL mRNA | |||
|---|---|---|---|---|
| r Coefficient | p-Value | r Coefficient | p-Value | |
| Age | −0.14 | 0.22 | −0.15 | 0.18 |
| BMI | −0.16 | 0.18 | 0.07 | 0.56 |
| Waist circumference | 0.10 | 0.44 | −0.12 | 0.36 |
| FBG | 0.10 | 0.39 | −0.04 | 0.97 |
| FSI | 0.51 | 0.008 | 0.46 | 0.019 |
| HbA1c | 0.13 | 0.29 | 0.21 | 0.20 |
| Total Cholesterol | 0.06 | 0.63 | −0.15 | 0.22 |
| HDL | −0.24 | 0.048 | 0.04 | 0.73 |
| LDL | 0.12 | 0.34 | −0.18 | 0.13 |
| Triglycerides | 0.08 | 0.48 | −0.06 | 0.61 |
| AST | −0.07 | 0.56 | −0.23 | 0.045 |
| ALT | 0.06 | 0.59 | −0.36 | 0.001 |
| GGT | −0.11 | 0.12 | −0.22 | 0.045 |
| HOMA-IR | 0.43 | 0.029 | 0.44 | 0.024 |
| LPL | 0.13 | 0.22 | - | - |
| Caveolin1 | 0.25 | 0.018 | 0.18 | 0.10 |
| MIP-1α | 0.22 | 0.043 | 0.07 | 0.49 |
| HIF-1α | 0.10 | 0.35 | 0.38 | <0.001 |
| TIMP1 | 0.30 | 0.005 | 0.16 | 0.14 |
| Caspase7 | 0.29 | 0.006 | −0.16 | 0.13 |
| PARP1 | 0.06 | 0.57 | 0.23 | 0.03 |
| Plasma ANGPTL4 | 0.03 | 0.81 | −0.001 | 0.99 |
| Unstandardized Coefficients | Standardized Coefficient | t | p-Value | ||
|---|---|---|---|---|---|
| B | Standard Error | Beta | |||
| (Constant) | 5.69 | 5.07 | 1.12 | 0.31 | |
| VAT VDR mRNA | 2.58 | 0.91 | 1.28 | 2.82 | 0.037 |
| Age | −0.043 | 0.03 | −0.44 | −1.52 | 0.19 |
| Sex | −0.010 | 0.63 | −0.005 | −0.016 | 0.98 |
| BMI | −0.075 | 0.057 | 0.47 | −1.33 | 0.19 |
| Waist circumference | 0.28 | 0.31 | 0.287 | 0.92 | 0.40 |
| HOMA-IR | 0.53 | 0.72 | 0.85 | 1.94 | 0.11 |
| Presence of AGM | −0.24 | 1.38 | −1.99 | −1.77 | 0.137 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barchetta, I.; Chiappetta, C.; Ceccarelli, V.; Cimini, F.A.; Bertoccini, L.; Gaggini, M.; Cristofano, C.D.; Silecchia, G.; Lenzi, A.; Leonetti, F.; et al. Angiopoietin-Like Protein 4 Overexpression in Visceral Adipose Tissue from Obese Subjects with Impaired Glucose Metabolism and Relationship with Lipoprotein Lipase. Int. J. Mol. Sci. 2020, 21, 7197. https://doi.org/10.3390/ijms21197197
Barchetta I, Chiappetta C, Ceccarelli V, Cimini FA, Bertoccini L, Gaggini M, Cristofano CD, Silecchia G, Lenzi A, Leonetti F, et al. Angiopoietin-Like Protein 4 Overexpression in Visceral Adipose Tissue from Obese Subjects with Impaired Glucose Metabolism and Relationship with Lipoprotein Lipase. International Journal of Molecular Sciences. 2020; 21(19):7197. https://doi.org/10.3390/ijms21197197
Chicago/Turabian StyleBarchetta, Ilaria, Caterina Chiappetta, Valentina Ceccarelli, Flavia Agata Cimini, Laura Bertoccini, Melania Gaggini, Claudio Di Cristofano, Gianfranco Silecchia, Andrea Lenzi, Frida Leonetti, and et al. 2020. "Angiopoietin-Like Protein 4 Overexpression in Visceral Adipose Tissue from Obese Subjects with Impaired Glucose Metabolism and Relationship with Lipoprotein Lipase" International Journal of Molecular Sciences 21, no. 19: 7197. https://doi.org/10.3390/ijms21197197
APA StyleBarchetta, I., Chiappetta, C., Ceccarelli, V., Cimini, F. A., Bertoccini, L., Gaggini, M., Cristofano, C. D., Silecchia, G., Lenzi, A., Leonetti, F., Baroni, M. G., Gastaldelli, A., & Cavallo, M. G. (2020). Angiopoietin-Like Protein 4 Overexpression in Visceral Adipose Tissue from Obese Subjects with Impaired Glucose Metabolism and Relationship with Lipoprotein Lipase. International Journal of Molecular Sciences, 21(19), 7197. https://doi.org/10.3390/ijms21197197









