Melatonin and Autophagy in Aging-Related Neurodegenerative Diseases
Abstract
1. Introduction
2. Neuronal Death in NDDs
2.1. Cell Death
2.2. Autophagy
2.2.1. Autophagy Classification
2.2.2. The Molecular Mechanism and Process of Autophagy
3. Autophagy and NDDs
3.1. Alzheimer’s Disease (AD)
3.2. Parkinson’s Disease (PD)
3.3. Huntington Disease (HD)
3.4. Organophosphate-Induced Delayed Neuropathy (OPIDN)
3.5. Amyotrophic Lateral Sclerosis (ALS)
4. Translational Applications of Melatonin against NDDs
4.1. Melatonin
4.2. Molecular Mechanisms of Protection by Melatonin against NDDs
4.2.1. The Effects of Melatonin on Autophagy
4.2.2. Inhibition of Apoptosis
4.2.3. Scavenging Free Radicals
4.2.4. Neuroprotection
4.2.5. Neurotrophic Effect
4.2.6. Anti-Aβ Neurotoxicity
4.2.7. Regulation of Neuroinflammation
4.2.8. Regulation of Cytoskeleton Proteins
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Aβ | Amyloid-β |
| AD | Alzheimer’s disease |
| Akt | serine/threonine kinase 1 |
| ALFY | autophagy-related FYVE protein |
| ALS | amyotrophic lateral sclerosis |
| AMBRA1 | autophagy and beclin1 regulator 1 |
| AMPK | AMP-activated protein kinase |
| APP | amyloid precursor protein |
| ATG | autophagy-related gene |
| ATG14L | ATG14-like |
| BAX | Bcl-2-associated X protein |
| BDNF | brain-derived neurotrophic factor |
| Beclin1 | Bcl2 interacting protein |
| BECN1 | beclin1 |
| BIF1 | Bax-interacting factor-1 |
| CDK5 | cyclin-dependent kinase 5 |
| CMA | chaperone-mediated autophagy |
| CNS | central nervous system |
| COX-2 | cyclooxygenase-2 |
| E1- | like enzyme |
| UBA5 | ubiquitin like modifier activating enzyme |
| ERK1 / 2 | extracellular signal-regulated kinases 1/2 |
| FIP200 | focal adhesion kinase family interacting protein of 200kD |
| GAPR-1 | Golgi-associated plant pathogenesis-related protein 1 |
| GDNF | glial-cell-line-derived neurotrophic factor |
| GSK3 | glycogen synthase kinase 3 |
| HD | Huntington’s disease |
| HTT | Huntington |
| JNK1/2 | c-Jun N-terminal kinase 1/2 |
| ·OH | hydroxyl radical |
| 6-OHDA | 6-hydroxydopamine |
| LC3 | microtubule-associated protein 1 light chain 3 |
| MAO-B | monoamine oxidase B |
| METH | methamphetamine |
| mHTT | mutant Huntington |
| MPPP | 1-methyl-4-phenyl-4-propionoxypiperidine |
| MPTP | methyl-4-phenyl-1,2,3,6-tetrahydropyridine |
| mTOR | mammalian rapamycin target protein |
| NDDs | neurodegenerative diseases |
| NF-κB | nuclear factor-κB |
| NRBF2 | nuclear receptor binding factor 2 |
| OPIDN | organophosphate-induced delayed neuropathy |
| OPTN | optineurin |
| Parkin | RING-domain-containing E3 ubiquitin ligase |
| PARP | poly-ADP ribose polymerase |
| PD | Parkinson’s disease |
| PI3KCIII | phosphatidylinositol 3-kinase catalytic subunit type 3 |
| PI(3)P | phosphatidylinositol 3-phosphate |
| PICALM | phosphatidylinositol-binding clathrin assembly protein |
| PINK1 | PTEN-induced kinase 1 |
| PrP | viral proteins |
| PS | presenilin |
| PtdIns3K | class III phosphatidylinositol 3-kinase complex |
| RB1CC1 | RB1-coiled helix-1 |
| ROS | reactive oxygen species |
| SNARE | soluble NSF attachment protein receptor |
| SOD | superoxide dismutase |
| SP | senile plaque |
| SQSTM1 | sequestosome 1 |
| STX17 | synaptic fusion protein-17 |
| TFEB | transcription factor EB |
| TOCP | tri-ortho-cresyl phosphate |
| ULK1 | Unc-51-like kinase 1 |
| UVRAG | ultraviolet irradiation resistance-associated gene |
| Vps | vesicular protein-sorting protein |
| Vps34 | type III PI3K |
References
- Bobkova, N.V.; Poltavtseva, R.A.; Leonov, S.V.; Sukhikh, G.T. Neuroregeneration, Regulation in Neurodegenerative Diseases and Aging. Biochem. Mosc. 2020, 85, S108–S130. [Google Scholar]
- Dumurgier, J.; Tzourio, C. Epidemiology of neurological diseases in older adults. Rev. Neurol. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Yang, Q.; BJoshi, R.; Liu, Y.; Akbar, M.; Song, B.-J.; Zhou, S.; Wang, X. Role of Alcohol Drinking in Alzheimer’s Disease, Parkinson’s Disease, and Amyotrophic Lateral Sclerosis. Int. J. Mol. Sci. 2020, 21, 2316. [Google Scholar] [CrossRef] [PubMed]
- Heemels, M.-T. Neurodegenerative diseases. Nature 2016, 539, 179. [Google Scholar] [CrossRef] [PubMed]
- Winklhofer, K.F.; Tatzelt, J.; Haass, C. The two faces of protein misfolding, gain- and loss-of-function in neurodegenerative diseases. EMBO 2008, 27, 336–349. [Google Scholar]
- Finkbeiner, E.; Haindl, M.; Raman, N.; Muller, S. SUMO routes ribosome maturation. Nucleus 2011, 2, 527–532. [Google Scholar] [CrossRef]
- Rubinsztein, D.C.; Gestwicki, J.E.; Murphy, L.O.; Klionsky, D.J. Potential therapeutic applications of autophagy. Nat. Rev. Drug Discov. 2007, 6, 304–312. [Google Scholar]
- Fuchs, Y.; Steller, H. Live to die another way, modes of programmed cell death and the signals emanating from dying cells. Nat. Rev. Mol. Cell Biol. 2015, 16, 329–344. [Google Scholar]
- Cohen, E.; Paulsson, J.F.; Blinder, P.; Burstyn-Cohen, T.; Du, D.; Estepa, G.; Adame, A.; Pham, H.M.; Holzenberger, M.; Kelly, J.W.; et al. Reduced IGF-1 Signaling Delays Age-Associated Proteotoxicity in Mice. Cell 2009, 139, 1157–1169. [Google Scholar] [CrossRef]
- Soto, C.; Pritzkow, S. Protein misfolding, aggregation, and conformational strains in neurodegenerative diseases. Nat. Neurosci. 2018, 21, 1332–1340. [Google Scholar]
- Stancu, I.-C.; Ferraiolo, M.; Terwel, D.; Dewachter, I. Tau Interacting Proteins, Gaining Insight into the Roles of Tau in Health and Disease. Adv. Exp. Med. Biol. 2019, 1184, 145–166. [Google Scholar] [PubMed]
- Zakeri, Z.; Bursch, W.; Tenniswood, M.; Lockshin, R.A. Cell death, programmed, apoptosis, necrosis, or other? Cell Death Differ. 1995, 2, 87–96. [Google Scholar] [PubMed]
- D’Arcy, M.S. Cell death, a review of the major forms of apoptosis, necrosis and autophagy. Cell Biol. Int. 2019, 43, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.J.; Henry, C.M. Distinguishing between apoptosis, necrosis, necroptosis and other cell death modalities. Methods 2013, 61, 87–89. [Google Scholar] [CrossRef]
- Galluzzi, L.; Vitale, I.; Aaronson, S.A.; Abrams, J.M.; Adam, D.; Agostinis, P.; Alnemri, E.S.; Altucci, L.; Amelio, I.; Andrews, D.W.; et al. Molecular mechanisms of cell death, recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018, 25, 486–541. [Google Scholar] [CrossRef]
- Perez-Polo, J.R.; Foreman, P.J.; Jackson, G.R.; Shan, D.; Taglialatela, G.; Thorpe, L.W.; Werrbach-Perez, K. Nerve growth factor and neuronal cell death. Mol. Neurobiol. 1990, 4, 57–91. [Google Scholar] [CrossRef]
- Fleisher, T.A. Apoptosis. Ann. Allergy Asthma Immunol. 1997, 78, 245–249. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis, a review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as anticancer mechanism, function and dysfunction of its modulators and targeted therapeutic strategies. Aging 2016, 8, 603–619. [Google Scholar] [CrossRef]
- Parzych, K.R.; Klionsky, D.J. An Overview of Autophagy, Morphology, Mechanism, and Regulation. Antioxid. Redox Signal 2014, 20, 460–473. [Google Scholar] [CrossRef]
- Ravanan, P.; Srikumar, I.F.; Talwar, P. Autophagy, The spotlight for cellular stress responses. Life Sci. 2017, 188, 53–67. [Google Scholar] [PubMed]
- Thangaraj, A.; Sil, S.; Tripathi, A.; Chivero, E.T.; Periyasamy, P.; Buch, S. Targeting endoplasmic reticulum stress and autophagy as therapeutic approaches for neurological diseases. Int. Rev. Cell Mol. Biol. 2020, 350, 285–325. [Google Scholar] [PubMed]
- Wang, D.; Peng, Z.; Ren, G.; Wang, G. The different roles of selective autophagic protein degradation in mammalian cells. Oncotarget 2015, 6, 37098–37116. [Google Scholar]
- Whitehead, N.P. Enhanced autophagy as a potential mechanism for the improved physiological function by simvastatin in muscular dystrophy. Autophagy 2016, 12, 705–706. [Google Scholar] [PubMed]
- Kobayashi, S. Choose Delicately and Reuse Adequately, The Newly Revealed Process of Autophagy. Biol. Pharm. Bull. 2015, 38, 1098–1103. [Google Scholar]
- Klionsky, D.J. Autophagy, from phenomenology to molecular understanding in less than a decade. Nat. Rev. Mol. Cell Biol. 2007, 8, 931–937. [Google Scholar]
- Yang, Y.; Klionsky, D.J. Autophagy and disease, unanswered questions. Cell Death Differ. 2020, 27, 858–871. [Google Scholar]
- Mizushima, N.; Levine, B.; Cuervo, A.M.; Klionsky, D.J. Autophagy fights disease through cellular self-digestion. Nature 2008, 451, 1069–1075. [Google Scholar]
- Levine, B.; Kroemer, G. Biological Functions of Autophagy Genes, A Disease Perspective. Cell 2019, 176, 11–42. [Google Scholar]
- Boya, P.; Reggiori, F.; Codogno, P. Emerging regulation and functions of autophagy. Nat. Cell Biol. 2013, 15, 713–720. [Google Scholar]
- Zhou, H.; Wang, J.; Jiang, J.; Stavrovskaya, I.G.; Li, M.; Li, W.; Wu, Q.; Zhang, X.; Luo, C.; Zhou, S.; et al. N-acetyl-serotonin offers neuroprotection through inhibiting mitochondrial death pathways and autophagic activation in experimental models of ischemic injury. J. Neurosci. 2014, 34, 2967–2978. [Google Scholar] [CrossRef]
- Nikoletopoulou, V.; Papandreou, M.-E.; Tavernarakis, N. Autophagy in the physiology and pathology of the central nervous system. Cell Death Differ. 2015, 22, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Pickford, F.; Masliah, E.; Britschgi, M.; Lucin, K.; Narasimhan, R.; Jaeger, P.A.; Small, S.; Spencer, B.; Rockenstein, E.; Levine, B.; et al. The autophagy-related protein beclin 1 shows reduced expression in early Alzheimer disease and regulates amyloid beta accumulation in mice. J. Clin. Investig. 2008, 118, 2190–2199. [Google Scholar] [PubMed]
- Bernard, A.; Jin, M.; Xu, Z.; Klionsky, D.J. A large-scale analysis of autophagy-related gene expression identifies new regulators of autophagy. Autophagy 2015, 11, 2114–2122. [Google Scholar] [CrossRef] [PubMed]
- Murakawa, T.; Okamoto, K.; Omiya, S.; Taneike, M.; Yamaguchi, O.; Otsu, K. A Mammalian Mitophagy Receptor, Bcl2-L-13, Recruits the ULK1 Complex to Induce Mitophagy. Cell Rep. 2019, 26, 338–345.e6. [Google Scholar] [CrossRef]
- Zachari, M.; Ganley, I.G. The mammalian ULK1 complex and autophagy initiation. Essays Biochem. 2017, 61, 585–596. [Google Scholar]
- Baskaran, R.; Poornima, P.; Priya, L.B.; Huang, C.-Y.; Padma, V.V. Neferine prevents autophagy induced by hypoxia through activation of Akt/mTOR pathway and Nrf2 in muscle cells. Biomed. Pharmacother. 2016, 83, 1407–1413. [Google Scholar] [CrossRef]
- Kalai Selvi, S.; Vinoth, A.; Varadharajan, T.; Weng, C.F.; Vijaya Padma, V. Neferine augments therapeutic efficacy of cisplatin through ROS- mediated non-canonical autophagy in human lung adenocarcinoma (A549 cells). Food Chem. Toxicol. 2017, 103, 28–40. [Google Scholar] [CrossRef]
- Decuypere, J.-P.; Parys, J.B.; Bultynck, G. Regulation of the autophagic bcl-2/beclin 1 interaction. Cells 2012, 1, 284–312. [Google Scholar] [CrossRef]
- Hu, G.; Rios, L.; Yan, Z.; Jasper, A.M.; Luera, D.; Luo, S.; Rao, H. Autophagy regulator Atg9 is degraded by the proteasome. Biochem. Biophys. Res. Commun. 2020, 522, 254–258. [Google Scholar] [CrossRef]
- Kang, S.; Shin, K.D.; Kim, J.H.; Chung, T. Autophagy-related (ATG) 11, ATG9 and the phosphatidylinositol 3-kinase control ATG2-mediated formation of autophagosomes in Arabidopsis. Plant Cell Rep. 2018, 37, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Kumari, R.; Chinchwadkar, S.; Aher, A.; Matheshwaran, S.; Manjithaya, R. Exocyst Subcomplex Functions in Autophagosome Biogenesis by Regulating Atg9 Trafficking. J. Mol. Biol. 2019, 431, 2821–2834. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhang, L. Regulation of ATG and Autophagy Initiation. Adv. Exp. Med. Biol. 2019, 1206, 41–65. [Google Scholar] [PubMed]
- Dong, G.; Zhang, Z.; Duan, K.; Shi, W.; Huang, R.; Wang, B.; Luo, L.; Zhang, Y.; Ruan, H.; Huang, H. Beclin 1 deficiency causes hepatic cell apoptosis via endoplasmic reticulum stress in zebrafish larvae. FEBS Lett. 2020, 594, 1155–1165. [Google Scholar] [CrossRef] [PubMed]
- Ağuş, H.H.; Yilmaz, S.; Şengöz, C.O. Crosstalk between autophagy and apoptosis induced by camphor in Schizosaccharomyces pombe. Turk. J. Biol. 2019, 43, 382–390. [Google Scholar] [CrossRef]
- Maejima, Y.; Isobe, M.; Sadoshima, J. Regulation of autophagy by Beclin 1 in the heart. J. Mol. Cell Cardiol. 2016, 95, 19–25. [Google Scholar] [CrossRef]
- Vega-Rubín-de-Celis, S. The Role of Beclin 1-Dependent Autophagy in Cancer. Biology 2019, 9, 4. [Google Scholar] [CrossRef]
- Towers, C.G.; Wodetzki, D.; Thorburn, A. Autophagy-dependent cancer cells circumvent loss of the upstream regulator RB1CC1/FIP200 and loss of LC3 conjugation by similar mechanisms. Autophagy 2020, 16, 1332–1340. [Google Scholar] [CrossRef]
- Zhang, M.-Q.; Li, J.-R.; Peng, Z.-G.; Zhang, J.-P. Differential Effects of Autophagy-Related 10 Protein on HCV Replication and Autophagy Flux Are Mediated by Its Cysteine44 and Cysteine135. Front. Immunol. 2018, 9, 2176. [Google Scholar] [CrossRef]
- Shams Nooraei, M.; Noori-Zadeh, A.; Darabi, S.; Rajaei, F.; Golmohammadi, Z.; Abbaszadeh, H.A. Low Level of Autophagy-Related Gene 10 (ATG10) Expression in the 6-Hydroxydopamine Rat Model of Parkinson’s Disease. Iran Biomed. J. 2018, 22, 15–21. [Google Scholar]
- Maruyama, T.; Noda, N.N. Autophagy-regulating protease Atg4, structure, function, regulation and inhibition. J. Antibiot. 2017, 71, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Agrotis, A.; Pengo, N.; Burden, J.J.; Ketteler, R. Redundancy of human ATG4 protease isoforms in autophagy and LC3/GABARAP processing revealed in cells. Autophagy 2019, 15, 976–997. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Zeng, C.; Hu, S.; Wang, L.; Liu, J. ATG3, a Target of miR-431-5p, Promotes Proliferation and Invasion of Colon Cancer via Promoting Autophagy. Cancer Manag. Res. 2019, 11, 10275–10285. [Google Scholar] [CrossRef] [PubMed]
- Frudd, K.; Burgoyne, T.; Burgoyne, J.R. Oxidation of Atg3 and Atg7 mediates inhibition of autophagy. Nat. Commun. 2018, 9, 95. [Google Scholar] [CrossRef] [PubMed]
- Manirujjaman, M.; Ozaki, I.; Murata, Y.; Guo, J.; Xia, J.; Nishioka, K.; Perveen, R.; Takahashi, H.; Anzai, K.; Matsuhashi, S. Degradation of the Tumor Suppressor PDCD4 Is Impaired by the Suppression of p62/SQSTM1 and Autophagy. Cells 2020, 9, 218. [Google Scholar] [CrossRef]
- Liao, W.; Wang, Z.; Fu, Z.; Ma, H.; Jiang, M.; Xu, A.; Zhang, W. p62/SQSTM1 protects against cisplatin-induced oxidative stress in kidneys by mediating the cross talk between autophagy and the Keap1-Nrf2 signalling pathway. Free Radic. Res. 2019, 53, 800–814. [Google Scholar] [CrossRef]
- Zhang, Y.; Mun, S.R.; Linares, J.F.; Ahn, J.; Towers, C.G.; Ji, C.H.; Fitzwalter, B.E.; Holden, M.R.; Mi, W.; Shi, X.; et al. ZZ-dependent regulation of p62/SQSTM1 in autophagy. Nat. Commun. 2018, 9, 4373. [Google Scholar] [CrossRef]
- Aparicio, R.; Rana, A.; Walker, D.W. Upregulation of the Autophagy Adaptor p62/SQSTM1 Prolongs Health and Lifespan in Middle-Aged Drosophila. Cell Rep. 2019, 28, 1029–1040.e5. [Google Scholar] [CrossRef]
- Deng, Z.; Purtell, K.; Lachance, V.; Wold, M.S.; Chen, S.; Yue, Z. Autophagy Receptors and Neurodegenerative Diseases. Trends Cell Biol. 2017, 27, 491–504. [Google Scholar] [CrossRef]
- Mancias, J.D.; Kimmelman, A.C. Mechanisms of Selective Autophagy in Normal Physiology and Cancer. J. Mol. Biol. 2016, 428, 1659–1680. [Google Scholar] [CrossRef]
- Kaiser, S.E.; Qiu, Y.; Coats, J.E.; Mao, K.; Klionsky, D.J.; Schulman, B.A. Structures of Atg7-Atg3 and Atg7-Atg10 reveal noncanonical mechanisms of E2 recruitment by the autophagy E1. Autophagy 2013, 9, 778–780. [Google Scholar] [CrossRef] [PubMed]
- Schulze-Luehrmann, J.; Eckart, R.A.; Ölke, M.; Saftig, P.; Liebler-Tenorio, E.; Lührmann, A. LAMP proteins account for the maturation delay during the establishment of the Coxiella burnetii-containing vacuole. Cell Microbiol. 2016, 18, 181–194. [Google Scholar] [CrossRef] [PubMed]
- Dikic, I.; Elazar, Z. Mechanism and medical implications of mammalian autophagy. Nat. Rev. Mol. Cell Biol. 2018, 19, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Randhawa, R.; Sehgal, M.; Singh, T.R.; Duseja, A.; Changotra, H. Unc-51 like kinase 1 (ULK1) in silico analysis for biomarker identification, a vital component of autophagy. Gene 2015, 562, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Ke, Z.; Xu, M.; Liao, M.; Wang, X.; Qi, Y.; Zhang, T.; Frank, J.A.; Bower, K.A.; Shi, X.; et al. Autophagy is a protective response to ethanol neurotoxicity. Autophagy 2012, 8, 1577–1589. [Google Scholar] [CrossRef]
- Ge, D.; Tao, H.-R.; Fang, L.; Kong, X.-Q.; Han, L.-N.; Li, N.; Xu, Y.-X.; Li, L.-Y.; Yu, M.; Zhang, H. 11-Methoxytabersonine Induces Necroptosis with Autophagy through AMPK/mTOR and JNK Pathways in Human Lung Cancer Cells. Chem. Pharm. Bull. 2020, 68, 244–250. [Google Scholar] [CrossRef]
- Kim, J.; Kundu, M.; Viollet, B.; Guan, K.-L. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat. Cell Biol. 2011, 13, 132–141. [Google Scholar] [CrossRef]
- Tan, Y.; Yin, L.; Sun, Z.; Shao, S.; Chen, W.; Man, X.; Du, Y.; Chen, Y. Astragalus polysaccharide exerts anti-Parkinson via activating the PI3K/AKT/mTOR pathway to increase cellular autophagy level in vitro. Int. J. Biol. Macromol. 2020, 153, 349–356. [Google Scholar] [CrossRef]
- Zhao, Y.; Feng, X.; Li, B.; Sha, J.; Wang, C.; Yang, T.; Cui, H.; Fan, H. Dexmedetomidine Protects Against Lipopolysaccharide-Induced Acute Kidney Injury by Enhancing Autophagy Through Inhibition of the PI3K/AKT/mTOR Pathway. Front. Pharmacol. 2020, 11, 128. [Google Scholar] [CrossRef]
- Kim, Y.C.; Guan, K.-L. mTOR, a pharmacologic target for autophagy regulation. J. Clin. Investig. 2015, 125, 25–32. [Google Scholar] [CrossRef]
- Qi, Y.; Cheng, X.; Gong, G.; Yan, T.; Du, Y.; Wu, B.; Bi, K.; Jia, Y. Synergistic neuroprotective effect of schisandrin and nootkatone on regulating inflammation, apoptosis and autophagy via the PI3K/AKT pathway. Food Funct. 2020, 11, 2427–2438. [Google Scholar] [CrossRef] [PubMed]
- Heras-Sandoval, D.; Pérez-Rojas, J.M.; Hernández-Damián, J.; Pedraza-Chaverri, J. The role of PI3K/AKT/mTOR pathway in the modulation of autophagy and the clearance of protein aggregates in neurodegeneration. Cell Signal. 2014, 26, 2694–2701. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Chen, Y.; Tooze, S.A. Autophagy pathway, Cellular and molecular mechanisms. Autophagy 2018, 14, 207–215. [Google Scholar] [CrossRef] [PubMed]
- You, Z.; Xu, Y.; Wan, W.; Zhou, L.; Li, J.; Zhou, T.; Shi, Y.; Liu, W. TP53INP2 contributes to autophagosome formation by promoting LC3-ATG7 interaction. Autophagy 2019, 15, 1309–1321. [Google Scholar] [CrossRef] [PubMed]
- Saleeb, R.S.; Kavanagh, D.M.; Dun, A.R.; Dalgarno, P.A.; Duncan, R.R. A VPS33A-binding motif on syntaxin 17 controls autophagy completion in mammalian cells. J. Biol. Chem. 2019, 294, 4188–4201. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Zheng, P.; Zhou, C.; Wang, X.; Ma, H.; Ma, W.; Zhou, X.; Teng, J.; Chen, J. DIPK2A promotes STX17- and VAMP7-mediated autophagosome-lysosome fusion by binding to VAMP7B. Autophagy 2019, 16, 797–810. [Google Scholar] [CrossRef]
- Gomes, L.R.; Menck, C.F.M.; Leandro, G.S. Autophagy Roles in the Modulation of DNA Repair Pathways. Int. J. Mol. Sci. 2017, 18, 2351. [Google Scholar] [CrossRef]
- Hartl, F.U. Protein Misfolding Diseases. Ann. Rev. Biochem. 2017, 86, 21–26. [Google Scholar] [CrossRef]
- Falcon, B.; Zhang, W.; Murzin, A.G.; Murshudov, G.; Garringer, H.J.; Vidal, R.; Crowther, R.A.; Ghetti, B.; Scheres, S.H.W.; Goedert, M. Structures of filaments from Pick’s disease reveal a novel tau protein fold. Nature 2018, 561, 137–140. [Google Scholar] [CrossRef]
- Martinez-Lopez, N.; Athonvarangkul, D.; Singh, R. Autophagy and aging. Adv. Exp. Med. Biol. 2015, 847, 73–87. [Google Scholar]
- Martinez-Lopez, N.; Garcia-Macia, M.; Sahu, S.; Athonvarangkul, D.; Liebling, E.; Merlo, P.; Cecconi, F.; Schwartz, G.J.; Singh, R. Autophagy in the CNS and Periphery Coordinate Lipophagy and Lipolysis in the Brown Adipose Tissue and Liver. Cell Metab. 2016, 23, 113–127. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Rhim, H. Therapeutic implication of autophagy in neurodegenerative diseases. BMB Rep. 2017, 50, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Vervliet, T. Ryanodine Receptors in Autophagy, Implications for Neurodegenerative Diseases? Front. Cell Neurosci. 2018, 12, 89. [Google Scholar] [CrossRef] [PubMed]
- Bekris, L.M.; Yu, C.-E.; Bird, T.D.; Tsuang, D.W. Genetics of Alzheimer Disease. J. Geriatr. Psychiatr. Neurol. 2010, 23, 213–227. [Google Scholar] [CrossRef]
- Friedrich, R.P.; Tepper, K.; Rönicke, R.; Soom, M.; Westermann, M.; Reymann, K.; Kaether, C.; Fändrich, M. Mechanism of amyloid plaque formation suggests an intracellular basis of Aβ pathogenicity. Proc. Natl. Acad. Sci. USA 2010, 107, 1942–1947. [Google Scholar] [CrossRef]
- Cho, Y.-E.; Lee, M.-H.; Song, B.-J. Neuronal Cell Death and Degeneration through Increased Nitroxidative Stress and Tau Phosphorylation in HIV-1 Transgenic Rats. PLoS ONE 2017, 12, e0169945. [Google Scholar] [CrossRef]
- Henderson, L.E.; Abdelmegeed, M.A.; Yoo, S.H.; Rhee, S.G.; Zhu, X.; Smith, M.A.; Nguyen, R.Q.; Perry, G.; Song, B.J. Enhanced Phosphorylation of Bax and Its Translocation into Mitochondria in the Brains of Individuals Affiliated with Alzheimer’s Disease. Open Neurol. J. 2017, 11, 48–58. [Google Scholar] [CrossRef]
- Baranello, R.; Bharani, K.; Padmaraju, V.; Chopra, N.; Lahiri, D.; Greig, N.; Pappolla, M.; Sambamurti, K. Amyloid-Beta Protein Clearance and Degradation (ABCD) Pathways and their Role in Alzheimer’s Disease. CAR 2015, 12, 32–46. [Google Scholar] [CrossRef]
- Pacheco-Quinto, J.; Clausen, D.; Pérez-González, R.; Peng, H.; Meszaros, A.; Eckman, C.B.; Levy, E.; Eckman, E.A. Intracellular metalloprotease activity controls intraneuronal Aβ aggregation and limits secretion of Aβ via exosomes. FASEB J. 2019, 33, 3758–3771. [Google Scholar] [CrossRef]
- Lee, J.-H.; Wolfe, D.M.; Darji, S.; McBrayer, M.K.; Colacurcio, D.J.; Kumar, A.; Stavrides, P.; Mohan, P.S.; Nixon, R.A. β2-adrenergic agonists rescue lysosome acidification and function in PSEN1 deficiency by reversing defective ER to lysosome delivery of ClC-7. J. Mol. Biol. 2020, 432, 2633–2650. [Google Scholar] [CrossRef]
- Martín-Maestro, P.; Sproul, A.; Martinez, H.; Paquet, D.; Gerges, M.; Noggle, S.; Starkov, A.A. Autophagy Induction by Bexarotene Promotes Mitophagy in Presenilin 1 Familial Alzheimer’s Disease iPSC-Derived Neural Stem Cells. Mol. Neurobiol. 2019, 56, 8220–8236. [Google Scholar] [CrossRef] [PubMed]
- Frake, R.A.; Ricketts, T.; Menzies, F.M.; Rubinsztein, D.C. Autophagy and neurodegeneration. J. Clin. Investig. 2015, 125, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Pendin, D.; Fasolato, C.; Basso, E.; Filadi, R.; Greotti, E.; Galla, L.; Gomiero, C.; Leparulo, A.; Redolfi, N.; Scremin, E.; et al. Familial Alzheimer’s disease presenilin-2 mutants affect Ca2+ homeostasis and brain network excitability. Aging Clin. Exp. Res. 2019, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Galla, L.; Redolfi, N.; Pozzan, T.; Pizzo, P.; Greotti, E. Intracellular Calcium Dysregulation by the Alzheimer’s Disease-Linked Protein Presenilin 2. Int. J. Mol. Sci. 2020, 21, 770. [Google Scholar] [CrossRef]
- Oliver, D.M.A.; Reddy, P.H. Molecular Basis of Alzheimer’s Disease, Focus on Mitochondria. J. Alzheimers Dis. 2019, 72, S95–S116. [Google Scholar] [CrossRef]
- Ando, K.; De Decker, R.; Vergara, C.; Yilmaz, Z.; Mansour, S.; Suain, V.; Sleegers, K.; de Fisenne, M.-A.; Houben, S.; Potier, M.-C.; et al. Picalm reduction exacerbates tau pathology in a murine tauopathy model. Acta Neuropathol. 2020, 139, 773–789. [Google Scholar] [CrossRef]
- Moreau, K.; Fleming, A.; Imarisio, S.; Lopez Ramirez, A.; Mercer, J.L.; Jimenez-Sanchez, M.; Bento, C.F.; Puri, C.; Zavodszky, E.; Siddiqi, F.; et al. PICALM modulates autophagy activity and tau accumulation. Nat. Commun. 2014, 5, 4998. [Google Scholar] [CrossRef]
- Van Acker, Z.P.; Bretou, M.; Annaert, W. Endo-lysosomal dysregulations and late-onset Alzheimer’s disease, impact of genetic risk factors. Mol. Neurodegener. 2019, 14, 20. [Google Scholar] [CrossRef]
- Ando, K.; Tomimura, K.; Sazdovitch, V.; Suain, V.; Yilmaz, Z.; Authelet, M.; Ndjim, M.; Vergara, C.; Belkouch, M.; Potier, M.-C.; et al. Level of PICALM, a key component of clathrin-mediated endocytosis, is correlated with levels of phosphotau and autophagy-related proteins and is associated with tau inclusions in AD, PSP and Pick disease. Neurobiol. Dis. 2016, 94, 32–43. [Google Scholar] [CrossRef]
- Roy, K.; Chakrabarti, O.; Mukhopadhyay, D. Interaction of Grb2 SH3 domain with UVRAG in an Alzheimer’s disease-like scenario. Biochem. Cell Biol. 2014, 92, 219–225. [Google Scholar] [CrossRef]
- Bieri, G.; Lucin, K.M.; O’Brien, C.E.; Zhang, H.; Villeda, S.A.; Wyss-Coray, T. Proteolytic cleavage of Beclin 1 exacerbates neurodegeneration. Mol. Neurodegener. 2018, 13, 68. [Google Scholar] [CrossRef]
- Wani, A.; Gupta, M.; Ahmad, M.; Shah, A.M.; Ahsan, A.U.; Qazi, P.H.; Malik, F.; Singh, G.; Sharma, P.R.; Kaddoumi, A.; et al. Alborixin clears amyloid-β by inducing autophagy through PTEN-mediated inhibition of the AKT pathway. Autophagy 2019, 15, 1810–1828. [Google Scholar] [CrossRef] [PubMed]
- Lucin, K.M.; O’Brien, C.E.; Bieri, G.; Czirr, E.; Mosher, K.I.; Abbey, R.J.; Mastroeni, D.F.; Rogers, J.; Spencer, B.; Masliah, E.; et al. Microglial beclin 1 regulates retromer trafficking and phagocytosis and is impaired in Alzheimer’s disease. Neuron 2013, 79, 873–886. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Yao, L.; Zhang, L.; Piao, Z.; Lu, Y. Schizandrol A protects against Aβ1-42-induced autophagy via activation of PI3K/AKT/mTOR pathway in SH-SY5Y cells and primary hippocampal neurons. Naunyn. Schmiedebergs Arch Pharmacol. 2020, 393, 1739–1752. [Google Scholar] [CrossRef]
- Mueed, Z.; Tandon, P.; Maurya, S.K.; Deval, R.; Kamal, M.A.; Poddar, N.K. Tau and mTOR, The Hotspots for Multifarious Diseases in Alzheimer’s Development. Front. Neurosci. 2018, 12, 1017. [Google Scholar] [CrossRef]
- Vartak, R.S.; Rodin, A.; Oddo, S. Differential activation of the mTOR/autophagy pathway predicts cognitive performance in APP/PS1 mice. Neurobiol. Aging 2019, 83, 105–113. [Google Scholar] [CrossRef]
- Cassano, T.; Magini, A.; Giovagnoli, S.; Polchi, A.; Calcagnini, S.; Pace, L.; Lavecchia, M.A.; Scuderi, C.; Bronzuoli, M.R.; Ruggeri, L.; et al. Early intrathecal infusion of everolimus restores cognitive function and mood in a murine model of Alzheimer’s disease. Exp. Neurol. 2019, 311, 88–105. [Google Scholar] [CrossRef] [PubMed]
- Caccamo, A.; Belfiore, R.; Oddo, S. Genetically reducing mTOR signaling rescues central insulin dysregulation in a mouse model of Alzheimer’s disease. Neurobiol. Aging 2018, 68, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Zhou, B.; Lin, M.-Y.; Sheng, Z.-H. Progressive endolysosomal deficits impair autophagic clearance beginning at early asymptomatic stages in fALS mice. Autophagy 2015, 11, 1934–1936. [Google Scholar] [CrossRef]
- Huang, L.; Lin, M.; Zhong, X.; Yang, H.; Deng, M. Galangin decreases p-tau, Aβ42 and β-secretase levels, and suppresses autophagy in okadaic acid-induced PC12 cells via an Akt/GSK3β/mTOR signaling-dependent mechanism. Mol. Med. Rep. 2019, 19, 1767–1774. [Google Scholar] [CrossRef]
- Di Domenico, F.; Tramutola, A.; Foppoli, C.; Head, E.; Perluigi, M.; Butterfield, D.A. mTOR in Down syndrome, Role in Aß and tau neuropathology and transition to Alzheimer disease-like dementia. Free Radic. Biol. Med. 2018, 114, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.-C.; Li, H.-Y.; Chen, G.-C.; Chern, Y.; Tu, P.-H. Mutations in the ubiquitin-binding domain of OPEN/optineurin interfere with autophagy-mediated degradation of misfolded proteins by a dominant-negative mechanism. Autophagy 2015, 11, 685–700. [Google Scholar] [CrossRef] [PubMed]
- Khaminets, A.; Behl, C.; Dikic, I. Ubiquitin-Dependent and Independent Signals In Selective Autophagy. Trends Cell Biol. 2016, 26, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Cai, C.-Z.; Song, J.-X.; Tan, J.-Q.; Durairajan, S.S.K.; Iyaswamy, A.; Wu, M.-Y.; Chen, L.-L.; Yue, Z.; Li, M.; et al. H. NRBF2 is involved in the autophagic degradation process of APP-CTFs in Alzheimer disease models. Autophagy 2017, 13, 2028–2040. [Google Scholar] [CrossRef]
- Kurosawa, M.; Matsumoto, G.; Sumikura, H.; Hatsuta, H.; Murayama, S.; Sakurai, T.; Shimogori, T.; Hattori, N.; Nukina, N. Serine 403-phosphorylated p62/SQSTM1 immunoreactivity in inclusions of neurodegenerative diseases. Neurosci. Res. 2016, 103, 64–70. [Google Scholar] [CrossRef]
- Anthony Jalin, A.M.A.; Jin, R.; Wang, M.; Li, G. EPPS treatment attenuates traumatic brain injury in mice by reducing Aβ burden and ameliorating neuronal autophagic flux. Exp. Neurol. 2019, 314, 20–33. [Google Scholar] [CrossRef]
- Kim, S.; Lee, D.; Song, J.C.; Cho, S.-J.; Yun, S.-M.; Koh, Y.H.; Song, J.; Johnson, G.V.W.; Jo, C. NDP52 associates with phosphorylated tau in brains of an Alzheimer disease mouse model. Biochem. Biophys. Res. Commun. 2014, 454, 196–201. [Google Scholar] [CrossRef]
- Woo, J.-A.A.; Liu, T.; Fang, C.C.; Castaño, M.A.; Kee, T.; Yrigoin, K.; Yan, Y.; Cazzaro, S.; Matlack, J.; Wang, X.; et al. β-Arrestin2 oligomers impair the clearance of pathological tau and increase tau aggregates. Proc. Natl. Acad. Sci. USA 2020, 117, 5006–5015. [Google Scholar] [CrossRef]
- Tanji, K.; Miki, Y.; Ozaki, T.; Maruyama, A.; Yoshida, H.; Mimura, J.; Matsumiya, T.; Mori, F.; Imaizumi, T.; Itoh, K.; et al. Phosphorylation of serine 349 of p62 in Alzheimer’s disease brain. Acta Neuropathol. Commun. 2014, 2, 50. [Google Scholar] [CrossRef]
- Akbar, M.; Essa, M.M.; Daradkeh, G.; Abdelmegeed, M.A.; Choi, Y.S.; Mahmood, L.; Song, B.J. Mitochondrial Dysfunction and Cell Death in Neurodegenerative Diseases through Nitroxidative Stress. Brain Res. 2016, 1637, 34–55. [Google Scholar] [CrossRef]
- Mizushima, N. Autophagy, process and function. Genes Dev. 2007, 21, 2861–2873. [Google Scholar] [PubMed]
- Barnett, R. Parkinson’s disease. Lancet 2016, 387, 217. [Google Scholar] [PubMed]
- Grayson, M. Parkinson’s disease. Nature 2016, 538, S1. [Google Scholar] [CrossRef] [PubMed]
- Hijaz, B.A.; Volpicelli-Daley, L.A. Initiation and propagation of α-synuclein aggregation in the nervous system. Mol. Neurodegener. 2020, 15, 19. [Google Scholar]
- Pozo Devoto, V.M.; Falzone, T.L. Mitochondrial dynamics in Parkinson’s disease, a role for α-synuclein? Dis. Model Mech. 2017, 10, 1075–1087. [Google Scholar]
- Visanji, N.P.; Brotchie, J.M.; Kalia, L.V.; Koprich, J.B.; Tandon, A.; Watts, J.C.; Lang, A.E. α-Synuclein-Based Animal Models of Parkinson’s Disease, Challenges and Opportunities in a New Era. Trends Neurosci. 2016, 39, 750–762. [Google Scholar]
- Wakabayashi, K.; Tanji, K.; Mori, F.; Takahashi, H. The Lewy body in Parkinson’s disease, molecules implicated in the formation and degradation of alpha-synuclein aggregates. Neuropathology 2007, 27, 494–506. [Google Scholar]
- An, W.-L.; Cowburn, R.F.; Li, L.; Braak, H.; Alafuzoff, I.; Iqbal, K.; Iqbal, I.-G.; Winblad, B.; Pei, J.-J. Up-regulation of phosphorylated/activated p70 S6 kinase and its relationship to neurofibrillary pathology in Alzheimer’s disease. Am. J. Pathol. 2003, 163, 591–607. [Google Scholar]
- Xu, C.-Y.; Kang, W.-Y.; Chen, Y.-M.; Jiang, T.-F.; Zhang, J.; Zhang, L.-N.; Ding, J.-Q.; Liu, J.; Chen, S.-D. DJ-1 Inhibits α-Synuclein Aggregation by Regulating Chaperone-Mediated Autophagy. Front. Aging Neurosci. 2017, 9, 308. [Google Scholar]
- Sun, L.; Lian, Y.; Ding, J.; Meng, Y.; Li, C.; Chen, L.; Qiu, P. The role of chaperone-mediated autophagy in neurotoxicity induced by alpha-synuclein after methamphetamine exposure. Brain Behav. 2019, 9, e01352. [Google Scholar]
- Hunn, B.H.M.; Vingill, S.; Threlfell, S.; Alegre-Abarrategui, J.; Magdelyns, M.; Deltheil, T.; Bengoa-Vergniory, N.; Oliver, P.L.; Cioroch, M.; Doig, N.M.; et al. Impairment of Macroautophagy in Dopamine Neurons Has Opposing Effects on Parkinsonian Pathology and Behavior. Cell Rep. 2019, 29, 920–931.e7. [Google Scholar] [CrossRef] [PubMed]
- di Domenico, A.; Carola, G.; Calatayud, C.; Pons-Espinal, M.; Muñoz, J.P.; Richaud-Patin, Y.; Fernandez-Carasa, I.; Gut, M.; Faella, A.; Parameswaran, J.; et al. Patient-Specific iPSC-Derived Astrocytes Contribute to Non-Cell-Autonomous Neurodegeneration in Parkinson’s Disease. Stem Cell Rep. 2019, 12, 213–229. [Google Scholar] [CrossRef] [PubMed]
- Winslow, A.R.; Chen, C.-W.; Corrochano, S.; Acevedo-Arozena, A.; Gordon, D.E.; Peden, A.A.; Lichtenberg, M.; Menzies, F.M.; Ravikumar, B.; Imarisio, S.; et al. α-Synuclein impairs macroautophagy, implications for Parkinson’s disease. J. Cell Biol. 2010, 190, 1023–1037. [Google Scholar] [CrossRef] [PubMed]
- Kam, T.-I.; Mao, X.; Park, H.; Chou, S.-C.; Karuppagounder, S.S.; Umanah, G.E.; Yun, S.P.; Brahmachari, S.; Panicker, N.; Chen, R.; et al. Poly(ADP-ribose) drives pathologic α-synuclein neurodegeneration in Parkinson’s disease. Science 2018, 362, eaat8407. [Google Scholar] [CrossRef]
- Zhang, N.; Chen, Y.; Jiang, R.; Li, E.; Chen, X.; Xi, Z.; Guo, Y.; Liu, X.; Zhou, Y.; Che, Y.; et al. PARP and RIP 1 are required for autophagy induced by 11’-deoxyverticillin A, which precees capase-dependent apoptosis. Autophagy 2011, 7, 598–612. [Google Scholar] [CrossRef]
- Gugliandolo, A.; Pollastro, F.; Bramanti, P.; Mazzon, E. Cannabidiol exerts protective effects in an in vitro model of Parkinson’s disease activating AKT/mTOR pathway. Fitoterapia 2020, 143, 104553. [Google Scholar] [CrossRef]
- Olsen, A.L.; Feany, M.B. PARP Inhibitors and Parkinson’s Disease. N. Engl. J. Med. 2019, 380, 492–494. [Google Scholar] [CrossRef]
- Pan, B.; Petersson, E.J. A PARP-1 Feed-Forward Mechanism to Accelerate α-Synuclein Toxicity in Parkinson’s Disease. Biochemistry 2019, 58, 859–860. [Google Scholar] [CrossRef]
- Vermilyea, S.C.; Babinski, A.; Tran, N.; To, S.; Guthrie, S.; Kluss, J.H.; Schmidt, J.K.; Wiepz, G.J.; Meyer, M.G.; Murphy, M.E.; et al. In Vitro CRISPR/Cas9-Directed Gene Editing to Model LRRK2 G2019S Parkinson’s Disease in Common Marmosets. Sci. Rep. 2020, 10, 3447. [Google Scholar] [CrossRef]
- Hwang, C.J.; Kim, Y.E.; Son, D.J.; Park, M.H.; Choi, D.-Y.; Park, P.-H.; Hellström, M.; Han, S.-B.; Oh, K.-W.; Park, E.K.; et al. Parkin deficiency exacerbate ethanol-induced dopaminergic neurodegeneration by P38 pathway dependent inhibition of autophagy and mitochondrial function. Redox Biol. 2017, 11, 456–468. [Google Scholar] [CrossRef]
- Meka, D.P.; Müller-Rischart, A.K.; Nidadavolu, P.; Mohammadi, B.; Motori, E.; Ponna, S.K.; Aboutalebi, H.; Bassal, M.; Annamneedi, A.; Finckh, B.; et al. Parkin cooperates with GDNF/RET signaling to prevent dopaminergic neuron degeneration. J. Clin. Investig. 2015, 125, 1873–1885. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.; Muqit, M.M.K. Therapeutic approaches to enhance PINK1/Parkin mediated mitophagy for the treatment of Parkinson’s disease. Neurosci. Lett. 2019, 705, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Sauvé, V.; Sung, G.; Soya, N.; Kozlov, G.; Blaimschein, N.; Miotto, L.S.; Trempe, J.-F.; Lukacs, G.L.; Gehring, K. Mechanism of parkin activation by phosphorylation. Nat. Struct. Mol. Biol. 2018, 25, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Du, T.-T.; Wang, L.; Duan, C.-L.; Lu, L.-L.; Zhang, J.-L.; Gao, G.; Qiu, X.-B.; Wang, X.-M.; Yang, H. GBA deficiency promotes SNCA/α-synuclein accumulation through autophagic inhibition by inactivated PPP2A. Autophagy 2015, 11, 1803–1820. [Google Scholar] [CrossRef]
- Gonzalez, A.; Valeiras, M.; Sidransky, E.; Tayebi, N. Lysosomal integral membrane protein-2, a new player in lysosome-related pathology. Mol. Genet. Metab. 2014, 111, 84–91. [Google Scholar] [CrossRef]
- Balke, D.; Tatenhorst, L.; Dambeck, V.; Ribas, V.T.; Vahsen, B.F.; Michel, U.; Bähr, M.; Lingor, P. AAV-Mediated Expression of Dominant-Negative ULK1 Increases Neuronal Survival and Enhances Motor Performance in the MPTP Mouse Model of Parkinson’s Disease. Mol. Neurobiol. 2020, 57, 685–697. [Google Scholar] [CrossRef]
- Jodeiri Farshbaf, M.; Ghaedi, K. Does any drug to treat cancer target mTOR and iron hemostasis in neurodegenerative disorders? Biometals 2017, 30, 1–16. [Google Scholar] [CrossRef]
- Ren, Z.-L.; Wang, C.-D.; Wang, T.; Ding, H.; Zhou, M.; Yang, N.; Liu, Y.-Y.; Chan, P. Ganoderma lucidum extract ameliorates MPTP-induced parkinsonism and protects dopaminergic neurons from oxidative stress via regulating mitochondrial function, autophagy, and apoptosis. Acta Pharmacol. Sin. 2019, 40, 441–450. [Google Scholar] [CrossRef]
- Zhu, F.; Fan, M.; Xu, Z.; Cai, Y.; Chen, Y.; Yu, S.; Zeng, L. Neuroprotective effect of rapamycin against Parkinson’s disease in mice. Zhejiang Da Xue Xue Bao Yi Xue Ban 2018, 47, 465–472. [Google Scholar]
- Nicklin, P.; Bergman, P.; Zhang, B.; Triantafellow, E.; Wang, H.; Nyfeler, B.; Yang, H.; Hild, M.; Kung, C.; Wilson, C.; et al. Bidirectional transport of amino acids regulates mTOR and autophagy. Cell 2009, 136, 521–534. [Google Scholar] [CrossRef]
- Orr, H.T. Beyond the Qs in the polyglutamine diseases. Genes Dev. 2001, 15, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Cepeda, C.; Tong, X.-P. Huntington’s disease, from basic science to therapeutics. CNS Neurosci. Ther. 2018, 24, 247–249. [Google Scholar] [CrossRef]
- Yhnell, E. Huntington’s disease, of mice and men. Oncotarget 2017, 8, 12552–12553. [Google Scholar] [CrossRef] [PubMed]
- Franco-Iborra, S.; Plaza-Zabala, A.; Montpeyo, M.; Sebastian, D.; Vila, M.; Martinez-Vicente, M. Mutant HTT (huntingtin) impairs mitophagy in a cellular model of Huntington disease. Autophagy 2020, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Luis-Ravelo, D.; Estévez-Silva, H.; Barroso-Chinea, P.; Afonso-Oramas, D.; Salas-Hernández, J.; Rodríguez-Núñez, J.; Acevedo-Arozena, A.; Marcellino, D.; González-Hernández, T. Pramipexole reduces soluble mutant huntingtin and protects striatal neurons through dopamine D3 receptors in a genetic model of Huntington’s disease. Exp. Neurol. 2018, 299, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Vicente, M.; Talloczy, Z.; Wong, E.; Tang, G.; Koga, H.; Kaushik, S.; de Vries, R.; Arias, E.; Harris, S.; Sulzer, D.; et al. Cargo recognition failure is responsible for inefficient autophagy in Huntington’s disease. Nat. Neurosci. 2010, 13, 567–576. [Google Scholar] [CrossRef]
- Walter, C.; Clemens, L.E.; Müller, A.J.; Fallier-Becker, P.; Proikas-Cezanne, T.; Riess, O.; Metzger, S.; Nguyen, H.P. Activation of AMPK-induced autophagy ameliorates Huntington disease pathology in vitro. Neuropharmacology 2016, 108, 24–38. [Google Scholar] [CrossRef]
- Wold, M.S.; Lim, J.; Lachance, V.; Deng, Z.; Yue, Z. ULK1-mediated phosphorylation of ATG14 promotes autophagy and is impaired in Huntington’s disease models. Mol. Neurodegener. 2016, 11, 76. [Google Scholar] [CrossRef]
- Jiang, W.; Wei, W.; Gaertig, M.A.; Li, S.; Li, X.-J. Therapeutic Effect of Berberine on Huntington’s Disease Transgenic Mouse Model. PLoS ONE 2015, 10, e0134142. [Google Scholar] [CrossRef]
- Mealer, R.G.; Murray, A.J.; Shahani, N.; Subramaniam, S.; Snyder, S.H. Rhes, a striatal-selective protein implicated in Huntington disease, binds beclin-1 and activates autophagy. J. Biol. Chem. 2014, 289, 3547–3554. [Google Scholar] [CrossRef]
- Perucho, J.; Gómez, A.; Muñoz, M.P.; de Yébenes, J.G.; Mena, M.Á.; Casarejos, M.J. Trehalose rescues glial cell dysfunction in striatal cultures from HD R6/1 mice at early postnatal development. Mol. Cell Neurosci. 2016, 74, 128–145. [Google Scholar] [CrossRef] [PubMed]
- Fox, L.; Yamamoto, A. Examining aggregates through the eyes of WDFY3/Alfy. Autophagy 2020, 16, 967–968. [Google Scholar] [CrossRef] [PubMed]
- Fox, L.M.; Kim, K.; Johnson, C.W.; Chen, S.; Croce, K.R.; Victor, M.B.; Eenjes, E.; Bosco, J.R.; Randolph, L.K.; Dragatsis, I.; et al. Huntington’s Disease Pathogenesis Is Modified In Vivo by Alfy/Wdfy3 and Selective Macroautophagy. Neuron 2020, 105, 813–821.e6. [Google Scholar] [CrossRef] [PubMed]
- Isakson, P.; Holland, P.; Simonsen, A. The role of ALFY in selective autophagy. Cell Death Differ. 2013, 20, 12–20. [Google Scholar] [CrossRef]
- Sarkar, S.; Perlstein, E.O.; Imarisio, S.; Pineau, S.; Cordenier, A.; Maglathlin, R.L.; Webster, J.A.; Lewis, T.A.; O’Kane, C.J.; Schreiber, S.L.; et al. Small molecules enhance autophagy and reduce toxicity in Huntington’s disease models. Nat. Chem. Biol. 2007, 3, 331–338. [Google Scholar] [CrossRef]
- Song, F.; Xie, K. Calcium-dependent neutral cysteine protease and organophosphate-induced delayed neuropathy. Chem. Biol. Interact. 2012, 200, 114–118. [Google Scholar] [CrossRef]
- Damodaran, T.V.; Attia, M.K.; Abou-Donia, M.B. Early differential cell death and survival mechanisms initiate and contribute to the development of OPIDN, a study of molecular, cellular, and anatomical parameters. Toxicol. Appl. Pharmacol. 2011, 256, 348–359. [Google Scholar] [CrossRef]
- Emerick, G.L.; DeOliveira, G.H.; dos Santos, A.C.; Ehrich, M. Mechanisms for consideration for intervention in the development of organophosphorus-induced delayed neuropathy. Chem. Biol. Interact. 2012, 199, 177–184. [Google Scholar] [CrossRef]
- Xu, H.-Y.; Wang, P.; Sun, Y.-J.; Xu, M.-Y.; Zhu, L.; Wu, Y.-J. Activation of Neuregulin 1/ErbB Signaling Is Involved in the Development of TOCP-Induced Delayed Neuropathy. Front. Mol. Neurosci. 2018, 11, 129. [Google Scholar] [CrossRef]
- Long, D.-X.; Hu, D.; Wang, P.; Wu, Y.-J. Induction of autophagy in human neuroblastoma SH-SY5Y cells by tri-ortho-cresyl phosphate. Mol. Cell Biochem. 2014, 396, 33–40. [Google Scholar] [CrossRef]
- Chen, J.-X.; Sun, Y.-J.; Wang, P.; Long, D.-X.; Li, W.; Li, L.; Wu, Y.-J. Induction of autophagy by TOCP in differentiated human neuroblastoma cells lead to degradation of cytoskeletal components and inhibition of neurite outgrowth. Toxicology 2013, 310, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Pandya, R.S.; Mao, L.L.J.; Zhou, E.W.; Bowser, R.; Zhu, Z.; Zhu, Y.; Wang, X. Neuroprotection for amyotrophic lateral sclerosis, role of stem cells, growth factors, and gene therapy. Cent. Nerv. Syst. Agents Med. Chem. 2012, 12, 15–27. [Google Scholar] [PubMed]
- Pandya, R.S.; Zhu, H.; Li, W.; Bowser, R.; Friedlander, R.M.; Wang, X. Therapeutic neuroprotective agents for amyotrophic lateral sclerosis. Cell Mol. Life Sci. 2013, 70, 4729–4745. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Nishiyama, A.; Kato, M.; Warita, H.; Aoki, M. Familial Amyotrophic Lateral Sclerosis. Brain Nerve 2019, 71, 1169–1181. [Google Scholar] [PubMed]
- Cristofani, R.; Crippa, V.; Rusmini, P.; Cicardi, M.E.; Meroni, M.; Nausicaa, V.; Giorgetti, E.; Grunseich, C.; Galbiati, M.; Piccolella, M.; et al. Inhibition of retrograde transport modulates misfolded protein accumulation and clearance in motoneuron diseases. Autophagy 2017, 13, 1280–1303. [Google Scholar] [CrossRef] [PubMed]
- Bowerman, M.; Murray, L.M.; Scamps, F.; Schneider, B.L.; Kothary, R.; Raoul, C. Pathogenic commonalities between spinal muscular atrophy and amyotrophic lateral sclerosis, Converging roads to therapeutic development. Eur. J. Med. Genet. 2018, 61, 685–698. [Google Scholar] [CrossRef]
- Baradaran-Heravi, Y.; Van Broeckhoven, C.; van der Zee, J. Stress granule mediated protein aggregation and underlying gene defects in the FTD-ALS spectrum. Neurobiol. Dis. 2020, 134, 104639. [Google Scholar] [CrossRef]
- Madill, M.; McDonagh, K.; Ma, J.; Vajda, A.; McLoughlin, P.; O’Brien, T.; Hardiman, O.; Shen, S. Amyotrophic lateral sclerosis patient iPSC-derived astrocytes impair autophagy via non-cell autonomous mechanisms. Mol. Brain 2017, 10, 22. [Google Scholar] [CrossRef]
- Mori, F.; Miki, Y.; Kon, T.; Tanji, K.; Wakabayashi, K. Autophagy Is a Common Degradation Pathway for Bunina Bodies and TDP-43 Inclusions in Amyotrophic Lateral Sclerosis. J. Neuropathol. Exp. Neurol. 2019, 78, 910–921. [Google Scholar] [CrossRef]
- Kochergin, I.A.; Shpilyukova, Y.A.; Lysogorskaia, E.V.; Abramycheva, N.Y.; Zakharova, M.N.; Illarioshkin, S.N. Effect of Mutations in SOD1 and C9orf72 Genes on Autophagy in Lymphomonocytes in Myotrophic Lateral Sclerosis. Bull. Exp. Biol. Med. 2019, 167, 667–670. [Google Scholar] [CrossRef]
- Dobson-Stone, C.; Hallupp, M.; Shahheydari, H.; Ragagnin, A.M.G.; Chatterton, Z.; Carew-Jones, F.; Shepherd, C.E.; Stefen, H.; Paric, E.; Fath, T.; et al. CYLD is a causative gene for frontotemporal dementia—Amyotrophic lateral sclerosis. Brain 2020, 143, 783–799. [Google Scholar] [CrossRef] [PubMed]
- Toth, R.P.; Atkin, J.D. Dysfunction of Optineurin in Amyotrophic Lateral Sclerosis and Glaucoma. Front. Immunol. 2018, 9, 1017. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, H.; Guan, Y.; Wang, Q.; Zhou, F.; Jie, L.; Ju, J.; Pu, L.; Du, H.; Wang, X. The altered autophagy mediated by TFEB in animal and cell models of amyotrophic lateral sclerosis. Am. J. Transl. Res. 2015, 7, 1574–1587. [Google Scholar] [PubMed]
- Hu, J.; Zhang, L.; Yang, Y.; Guo, Y.; Fan, Y.; Zhang, M.; Man, W.; Gao, E.; Hu, W.; Reiter, R.J.; et al. Melatonin alleviates postinfarction cardiac remodeling and dysfunction by inhibiting MstJ. Pineal Res. 2017, 62, e12368. [Google Scholar] [CrossRef]
- Jauhari, A.; Baranov, S.V.; Suofu, Y.; Kim, J.; Singh, T.; Yablonska, S.; Li, F.; Wang, X.; Oberly, P.; Minnigh, M.B.; et al. Melatonin inhibits cytosolic mitochondrial-DNA induced neuroinflammatory signaling in accelerated aging and neurodegeneration. J. Clin. Investig. 2020, 130, 3124–3136. [Google Scholar] [CrossRef]
- Tordjman, S.; Chokron, S.; Delorme, R.; Charrier, A.; Bellissant, E.; Jaafari, N.; Fougerou, C. Melatonin, Pharmacology, Functions and Therapeutic Benefits. Curr. Neuropharmacol. 2017, 15, 434–443. [Google Scholar] [CrossRef]
- Magri, F.; Sarra, S.; Cinchetti, W.; Guazzoni, V.; Fioravanti, M.; Cravello, L.; Ferrari, E. Qualitative and quantitative changes of melatonin levels in physiological and pathological aging and in centenarians. J. Pineal Res. 2004, 36, 256–261. [Google Scholar] [CrossRef]
- Schernhammer, E.; Chen, H.; Ritz, B. Circulating melatonin levels, possible link between Parkinson’s disease and cancer risk? Cancer Causes Control 2006, 17, 577–582. [Google Scholar] [CrossRef]
- Skene, D.J. Optimization of light and melatonin to phase-shift human circadian rhythms. J. Neuroendocrinol. 2003, 15, 438–441. [Google Scholar] [CrossRef]
- Wongprayoon, P.; Govitrapong, P. Melatonin as a mitochondrial protector in neurodegenerative diseases. Cell Mol. Life Sci. 2017, 74, 3999–4014. [Google Scholar] [CrossRef]
- Zhang, L.-F.; Zhou, Z.-W.; Wang, Z.-H.; Du, Y.-H.; He, Z.-X.; Cao, C.; Zhou, S.-F. Coffee and caffeine potentiate the antiamyloidogenic activity of melatonin via inhibition of Aβ oligomerization and modulation of the Tau-mediated pathway in N2a/APP cells. Drug Des. Devel. Ther. 2015, 9, 241–272. [Google Scholar] [PubMed]
- Stefanova, N.A.; Maksimova, K.Y.; Kiseleva, E.; Rudnitskaya, E.A.; Muraleva, N.A.; Kolosova, N.G. Melatonin attenuates impairments of structural hippocampal neuroplasticity in OXYS rats during active progression of Alzheimer’s disease-like pathology. J. Pineal Res. 2015, 59, 163–177. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Giordano, S.; Zhang, J. Autophagy, mitochondria and oxidative stress, cross-talk and redox signalling. Biochem. J. 2012, 441, 523–540. [Google Scholar] [CrossRef] [PubMed]
- Gelmetti, V.; De Rosa, P.; Torosantucci, L.; Marini, E.S.; Romagnoli, A.; Di Rienzo, M.; Arena, G.; Vignone, D.; Fimia, G.M.; Valente, E.M. PINK1 and BECN1 relocalize at mitochondria-associated membranes during mitophagy and promote ER-mitochondria tethering and autophagosome formation. Autophagy 2017, 13, 654–669. [Google Scholar] [CrossRef] [PubMed]
- Hamasaki, M.; Furuta, N.; Matsuda, A.; Nezu, A.; Yamamoto, A.; Fujita, N.; Oomori, H.; Noda, T.; Haraguchi, T.; Hiraoka, Y.; et al. Autophagosomes form at ER-mitochondria contact sites. Nature 2013, 495, 389–393. [Google Scholar] [CrossRef]
- Fernández, A.; Ordóñez, R.; Reiter, R.J.; González-Gallego, J.; Mauriz, J.L. Melatonin and endoplasmic reticulum stress, relation to autophagy and apoptosis. J. Pineal Res. 2015, 59, 292–307. [Google Scholar] [CrossRef]
- Scheper, W.; Nijholt, D.A.; Hoozemans, J.J. The unfolded protein response and proteostasis in Alzheimer disease, preferential activation of autophagy by endoplasmic reticulum stress. Autophagy 2011, 7, 910–911. [Google Scholar] [CrossRef]
- Polito, V.A.; Li, H.; Martini-Stoica, H.; Wang, B.; Yang, L.; Xu, Y.; Swartzlander, D.B.; Palmieri, M.; di Ronza, A.; Lee, V.M.; et al. Selective clearance of aberrant tau proteins and rescue of neurotoxicity by transcription factor EB. EMBO Mol. Med. 2014, 6, 1142–1160. [Google Scholar] [CrossRef]
- Dragicevic, N.; Copes, N.; O’Neal-Moffitt, G.; Jin, J.; Buzzeo, R.; Mamcarz, M.; Tan, J.; Cao, C.; Olcese, J.M.; Arendash, G.W.; et al. Melatonin treatment restores mitochondrial function in Alzheimer’s mice, a mitochondrial protective role of melatonin membrane receptor signaling. J. Pineal Res. 2011, 51, 75–86. [Google Scholar] [CrossRef]
- García, S.; Martín Giménez, V.M.; Mocayar Marón, F.J.; Reiter, R.J.; Manucha, W. Melatonin and cannabinoids, mitochondrial-targeted molecules that may reduce inflammaging in neurodegenerative diseases. Histol. Histopathol. 2020, 35, 789–800. [Google Scholar]
- Srivastava, A.K.; Roy Choudhury, S.; Karmakar, S. Melatonin/polydopamine nanostructures for collective neuroprotection-based Parkinson’s disease therapy. Biomat. Sci. 2020, 8, 1345–1363. [Google Scholar] [CrossRef] [PubMed]
- Fasciani, I.; Petragnano, F.; Aloisi, G.; Marampon, F.; Rossi, M.; Coppolino, M.F.; Rossi, R.; Longoni, B.; Scarselli, M.; Maggio, R. A New Threat to Dopamine Neurons, The Downside of Artificial Light. Neuroscience 2020, 432, 216–228. [Google Scholar] [CrossRef] [PubMed]
- Tamura, H.; Jozaki, M.; Tanabe, M.; Shirafuta, Y.; Mihara, Y.; Shinagawa, M.; Tamura, I.; Maekawa, R.; Sato, S.; Taketani, T.; et al. Importance of Melatonin in Assisted Reproductive Technology and Ovarian Aging. Int. J. Mol. Sci. 2020, 21, 1135. [Google Scholar] [CrossRef] [PubMed]
- Su, L.-Y.; Li, H.; Lv, L.; Feng, Y.-M.; Li, G.-D.; Luo, R.; Zhou, H.-J.; Lei, X.-G.; Ma, L.; Li, J.-L.; et al. Melatonin attenuates MPTP-induced neurotoxicity via preventing CDK5-mediated autophagy and SNCA/α-synuclein aggregation. Autophagy 2015, 11, 1745–1759. [Google Scholar] [CrossRef] [PubMed]
- Cadet, J.L.; Krasnova, I.N. Molecular bases of methamphetamine-induced neurodegeneration. Int. Rev. Neurobiol. 2009, 88, 101–119. [Google Scholar]
- Park, J.; Seo, J.; Won, J.; Yeo, H.-G.; Ahn, Y.-J.; Kim, K.; Jin, Y.B.; Koo, B.-S.; Lim, K.S.; Jeong, K.-J.; et al. Abnormal Mitochondria in a Non-human Primate Model of MPTP-induced Parkinson’s Disease, Drp1 and CDK5/p25 Signaling. Exp. Neurobiol. 2019, 28, 414–424. [Google Scholar] [CrossRef] [PubMed]
- Togashi, K.; Hasegawa, M.; Nagai, J.; Tonouchi, A.; Masukawa, D.; Hensley, K.; Goshima, Y.; Ohshima, T. Genetic suppression of collapsin response mediator protein 2 phosphorylation improves outcome in methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced Parkinson’s model mice. Genes Cells 2019, 24, 31–40. [Google Scholar] [CrossRef]
- He, F.; Qi, G.; Zhang, Q.; Cai, H.; Li, T.; Li, M.; Zhang, Q.; Chen, J.; Ming, J.; Tian, B.; et al. Quantitative Phosphoproteomic Analysis in Alpha-Synuclein Transgenic Mice Reveals the Involvement of Aberrant p25/Cdk5 Signaling in Early-stage Parkinson’s Disease. Cell Mol. Neurobiol. 2020, 40, 897–909. [Google Scholar] [CrossRef]
- Sun, Y.; Sukumaran, P.; Selvaraj, S.; Cilz, N.I.; Schaar, A.; Lei, S.; Singh, B.B. TRPM2 Promotes Neurotoxin MPP+/MPTP-Induced Cell Death. Mol. Neurobiol. 2018, 55, 409–420. [Google Scholar] [CrossRef]
- Mack, J.M.; Schamne, M.G.; Sampaio, T.B.; Pértile, R.A.N.; Fernandes, P.A.C.M.; Markus, R.P.; Prediger, R.D. Melatoninergic System in Parkinson’s Disease, From Neuroprotection to the Management of Motor and Nonmotor Symptoms. Oxid. Med. Cell Longev. 2016, 2016, 3472032. [Google Scholar] [CrossRef]
- Tapias, V.; Cannon, J.R.; Greenamyre, J.T. Melatonin treatment potentiates neurodegeneration in a rat rotenone Parkinson’s disease model. J. Neurosci. Res. 2010, 88, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Singhal, N.K.; Srivastava, G.; Agrawal, S.; Jain, S.K.; Singh, M.P. Melatonin as a neuroprotective agent in the rodent models of Parkinson’s disease, is it all set to irrefutable clinical translation? Mol. Neurobiol. 2012, 45, 186–199. [Google Scholar] [CrossRef]
- Liu, J.; Liu, W.; Lu, Y.; Tian, H.; Duan, C.; Lu, L.; Gao, G.; Wu, X.; Wang, X.; Yang, H. Piperlongumine restores the balance of autophagy and apoptosis by increasing BCL2 phosphorylation in rotenone-induced Parkinson disease models. Autophagy 2018, 14, 845–861. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Sun, S.; Sun, Y.; Song, Q.; Zhu, J.; Song, N.; Chen, M.; Sun, T.; Xia, M.; Ding, J.; et al. Small molecule-driven NLRP3 inflammation inhibition via interplay between ubiquitination and autophagy, implications for Parkinson disease. Autophagy 2019, 15, 1860–1881. [Google Scholar] [CrossRef] [PubMed]
- Nopparat, C.; Porter, J.E.; Ebadi, M.; Govitrapong, P. The mechanism for the neuroprotective effect of melatonin against methamphetamine-induced autophagy. J. Pineal Res. 2010, 49, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Schwarcz, R.; Coyle, J.T. Striatal lesions with kainic acid, neurochemical characteristics. Brain Res. 1977, 127, 235–249. [Google Scholar] [CrossRef]
- Chang, C.-F.; Huang, H.-J.; Lee, H.-C.; Hung, K.-C.; Wu, R.-T.; Lin, A.M.-Y. Melatonin attenuates kainic acid-induced neurotoxicity in mouse hippocampus via inhibition of autophagy and α-synuclein aggregation. J. Pineal Res. 2012, 52, 312–321. [Google Scholar] [CrossRef]
- Zhou, H.; Chen, J.; Lu, X.; Shen, C.; Zeng, J.; Chen, L.; Pei, Z. Melatonin protects against rotenone-induced cell injury via inhibition of Omi and Bax-mediated autophagy in Hela cells. J. Pineal Res. 2012, 52, 120–127. [Google Scholar] [CrossRef]
- Kim, B.J.; Ryu, S.W.; Song, B.J. JNK- and p38 kinase-mediated phosphorylation of Bax leads to its activation and mitochondrial translocation and to apoptosis of human hepatoma HepG2 cells. J. Biol. Chem. 2006, 281, 21256–21265. [Google Scholar] [CrossRef]
- Jenwitheesuk, A.; Nopparat, C.; Mukda, S.; Wongchitrat, P.; Govitrapong, P. Melatonin regulates aging and neurodegeneration through energy metabolism, epigenetics, autophagy and circadian rhythm pathways. Int. J. Mol. Sci. 2014, 15, 16848–16884. [Google Scholar] [CrossRef]
- Mammucari, C.; Rizzuto, R. Signaling pathways in mitochondrial dysfunction and aging. Mech. Ageing Dev. 2010, 131, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Reynolds-Peterson, C.; Xu, J.; Zhao, N.; Cruse, C.; Yonel, B.; Trasorras, C.; Toyoda, H.; Kinoshita-Toyoda, A.; Dobson, J.; Schultheis, N.; et al. Heparan Sulfate Structure Affects Autophagy, Lifespan, Responses to Oxidative Stress, and Cell Degeneration in Drosophila parkin Mutants. G3 Genes Genomes Genet. 2020, 10, 129–141. [Google Scholar] [CrossRef] [PubMed]
- Eldeeb, M.A.; Ragheb, M.A. N-degron-mediated degradation and regulation of mitochondrial PINK1 kinase. Curr. Genet. 2020, 66, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K. The PINK1-Parkin Axis, an Overview. Neurosci. Res. 2020, in press. [Google Scholar] [CrossRef]
- Ikeda, F.; Dikic, I. Atypical ubiquitin chains, new molecular signals. Protein Modifications: Beyond the Usual Suspects’ Review Series. EMBO Rep. 2008, 9, 536–542. [Google Scholar] [CrossRef]
- Scherz-Shouval, R.; Elazar, Z. Regulation of autophagy by ROS, physiology and pathology. Trends Biochem. Sci. 2011, 36, 30–38. [Google Scholar] [CrossRef]
- Kongsuphol, P.; Mukda, S.; Nopparat, C.; Villarroel, A.; Govitrapong, P. Melatonin attenuates methamphetamine-induced deactivation of the mammalian target of rapamycin signaling to induce autophagy in SK-N-SH cells. J. Pineal Res. 2009, 46, 199–206. [Google Scholar] [CrossRef]
- Frost, B.; Diamond, M.I. Prion-like mechanisms in neurodegenerative diseases. Nat. Rev. Neurosci. 2010, 11, 155–159. [Google Scholar] [CrossRef]
- Jeong, J.-K.; Moon, M.-H.; Lee, Y.-J.; Seol, J.-W.; Park, S.-Y. Melatonin-induced autophagy protects against human prion protein-mediated neurotoxicity. J. Pineal Res. 2012, 53, 138–146. [Google Scholar] [CrossRef]
- Jeong, J.-K.; Park, S.-Y. Melatonin regulates the autophagic flux via activation of alpha-7 nicotinic acetylcholine receptors. J. Pineal Res. 2015, 59, 24–37. [Google Scholar] [CrossRef]
- Zhang, Y.; Cook, A.; Kim, J.; Baranov, S.V.; Jiang, J.; Smith, K.; Cormier, K.; Bennett, E.; Browser, R.P.; Day, A.L.; et al. Melatonin inhibits the caspase-1/cytochrome c/caspase-3 cell death pathway, inhibits MT1 receptor loss and delays disease progression in a mouse model of amyotrophic lateral sclerosis. Neurobiol. Dis. 2013, 55, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Weishaupt, J.H.; Bartels, C.; Pölking, E.; Dietrich, J.; Rohde, G.; Poeggeler, B.; Mertens, N.; Sperling, S.; Bohn, M.; Hüther, G.; et al. Reduced oxidative damage in ALS by high-dose enteral melatonin treatment. J. Pineal Res. 2006, 41, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Wang, X. The Antiapoptotic Activity of Melatonin in Neurodegenerative Diseases. CNS Neurosci. Ther. 2009, 15, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sirianni, A.; Pei, Z.; Cormier, K.; Smith, K.; Jiang, J.; Zhou, S.; Wang, H.; Zhao, R.; Yano, H.; et al. The Melatonin MT1 Receptor Axis Modulates Mutant Huntingtin-Mediated Toxicity. J. Neurosci. 2011, 31, 14496–14507. [Google Scholar] [CrossRef]
- Rademakers, R.; Sleegers, K.; Theuns, J.; Van den Broeck, M.; Bel Kacem, S.; Nilsson, L.-G.; Adolfsson, R.; van Duijn, C.M.; Van Broeckhoven, C.; Cruts, M. Association of cyclin-dependent kinase 5 and neuronal activators p35 and p39 complex in early-onset Alzheimer’s disease. Neurobiol. Aging 2005, 26, 1145–1151. [Google Scholar] [CrossRef]
- Gutierrez-Cuesta, J.; Sureda, F.X.; Romeu, M.; Canudas, A.M.; Caballero, B.; Coto-Montes, A.; Camins, A.; Pallàs, M. Chronic administration of melatonin reduces cerebral injury biomarkers in SAMPJ. Pineal Res. 2007, 42, 394–402. [Google Scholar] [CrossRef]
- Alvira, D.; Tajes, M.; Verdaguer, E.; Acuña-Castroviejo, D.; Folch, J.; Camins, A.; Pallas, M. Inhibition of the cdk5/p25 fragment formation may explain the antiapoptotic effects of melatonin in an experimental model of Parkinson’s disease. J. Pineal Res. 2006, 40, 251–258. [Google Scholar] [CrossRef]
- Luchetti, F.; Canonico, B.; Curci, R.; Battistelli, M.; Mannello, F.; Papa, S.; Tarzia, G.; Falcieri, E. Melatonin prevents apoptosis induced by UV-B treatment in U937 cell line. J. Pineal Res. 2006, 40, 158–167. [Google Scholar] [CrossRef]
- Fischer, T.W.; Zbytek, B.; Sayre, R.M.; Apostolov, E.O.; Basnakian, A.G.; Sweatman, T.W.; Wortsman, J.; Elsner, P.; Slominski, A. Melatonin increases survival of HaCaT keratinocytes by suppressing UV-induced apoptosis. J. Pineal Res. 2006, 40, 18–26. [Google Scholar] [CrossRef]
- Mehrzadi, S.; Hemati, K.; Reiter, R.J.; Hosseinzadeh, A. Mitochondrial dysfunction in age-related macular degeneration, melatonin as a potential treatment. Exp. Opin. Ther. Targets 2020, 24, 359–378. [Google Scholar] [CrossRef]
- Reiter, R.; Tan, D.; Rosales-Corral, S.; Galano, A.; Zhou, X.; Xu, B. Mitochondria, Central Organelles for Melatonin′s Antioxidant and Anti-Aging Actions. Molecules 2018, 23, 509. [Google Scholar]
- Leon, J.; Acuña-Castroviejo, D.; Sainz, R.M.; Mayo, J.C.; Tan, D.-X.; Reiter, R.J. Melatonin and mitochondrial function. Life Sci. 2004, 75, 765–790. [Google Scholar] [CrossRef] [PubMed]
- Thomas, B.; Mohanakumar, K.P. Melatonin protects against oxidative stress caused by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine in the mouse nigrostriatum. J. Pineal Res. 2004, 36, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Maharaj, D.S.; Maharaj, H.; Daya, S.; Glass, B.D. Melatonin and 6-hydroxymelatonin protect against iron-induced neurotoxicity. J. Neurochem. 2006, 96, 78–81. [Google Scholar] [CrossRef]
- Wang, X.; Figueroa, B.E.; Stavrovskaya, I.G.; Zhang, Y.; Sirianni, A.C.; Zhu, S.; Day, A.L.; Kristal, B.S.; Friedlander, R.M. Methazolamide and melatonin inhibit mitochondrial cytochrome C release and are neuroprotective in experimental models of ischemic injury. Stroke 2009, 40, 1877–1885. [Google Scholar]
- Vishnoi, S.; Raisuddin, S.; Parvez, S. Glutamate Excitotoxicity and Oxidative Stress in Epilepsy, Modulatory Role of Melatonin. J. Environ. Pathol. Toxicol. Oncol. 2016, 35, 365–374. [Google Scholar] [CrossRef]
- Beker, M.C.; Caglayan, A.B.; Kelestemur, T.; Caglayan, B.; Yalcin, E.; Yulug, B.; Kilic, U.; Hermann, D.M.; Kilic, E. Effects of normobaric oxygen and melatonin on reperfusion injury, role of cerebral microcirculation. Oncotarget 2015, 6, 30604–30614. [Google Scholar] [CrossRef]
- Lee, S.-H.; Chun, W.; Kong, P.-J.; Han, J.A.; Cho, B.P.; Kwon, O.-Y.; Lee, H.J.; Kim, S.-S. Sustained activation of Akt by melatonin contributes to the protection against kainic acid-induced neuronal death in hippocampus. J. Pineal Res. 2006, 40, 79–85. [Google Scholar] [CrossRef]
- Beker, M.C.; Caglayan, B.; Caglayan, A.B.; Kelestemur, T.; Yalcin, E.; Caglayan, A.; Kilic, U.; Baykal, A.T.; Reiter, R.J.; Kilic, E. Interaction of melatonin and Bmal1 in the regulation of PI3K/AKT pathway components and cellular survival. Sci. Rep. 2019, 9, 19082. [Google Scholar]
- Armstrong, K.J.; Niles, L.P. Induction of GDNF mRNA expression by melatonin in rat C6 glioma cells. Neuroreport 2002, 13, 473–475. [Google Scholar] [CrossRef]
- Phillipson, O.T. Alpha-synuclein, epigenetics, mitochondria, metabolism, calcium traffic & circadian dysfunction in Parkinson’s disease. An integrated strategy for management. Ageing Res. Rev. 2017, 40, 149–167. [Google Scholar] [PubMed]
- Sharma, R.; McMillan, C.R.; Tenn, C.C.; Niles, L.P. Physiological neuroprotection by melatonin in a 6-hydroxydopamine model of Parkinson’s disease. Brain Res. 2006, 1068, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Olcese, J.M.; Cao, C.; Mori, T.; Mamcarz, M.B.; Maxwell, A.; Runfeldt, M.J.; Wang, L.; Zhang, C.; Lin, X.; Zhang, G.; et al. Protection against cognitive deficits and markers of neurodegeneration by long-term oral administration of melatonin in a transgenic model of Alzheimer disease. J. Pineal Res. 2009, 47, 82–96. [Google Scholar] [CrossRef] [PubMed]
- Khatoon, R.; Rasheed, M.Z.; Rawat, M.; Alam, M.M.; Tabassum, H.; Parvez, S. Effect of melatonin on Aβ42 induced changes in the mitochondrial function related to Alzheimer’s disease in Drosophila melanogaster. Neurosci. Lett. 2019, 711, 134376. [Google Scholar] [CrossRef]
- Hossain, M.F.; Uddin, M.S.; Uddin, G.M.S.; Sumsuzzman, D.M.; Islam, M.S.; Barreto, G.E.; Mathew, B.; Ashraf, G.M. Melatonin in Alzheimer’s Disease, A Latent Endogenous Regulator of Neurogenesis to Mitigate Alzheimer’s Neuropathology. Mol. Neurobiol. 2019, 56, 8255–8276. [Google Scholar] [CrossRef]
- Xu, J.; Gao, H.; Zhang, L.; Rong, S.; Yang, W.; Ma, C.; Chen, M.; Huang, Q.; Deng, Q.; Huang, F. Melatonin alleviates cognition impairment by antagonizing brain insulin resistance in aged rats fed a high-fat diet. J. Pineal Res. 2019, 67, e12584. [Google Scholar] [CrossRef]
- Yang, Z.; Bao, Y.; Chen, W.; He, Y. Melatonin exerts neuroprotective effects by attenuating astro- and microgliosis and suppressing inflammatory response following spinal cord injury. Neuropeptides 2020, 79, 102002. [Google Scholar] [CrossRef]
- Permpoonputtana, K.; Tangweerasing, P.; Mukda, S.; Boontem, P.; Nopparat, C.; Govitrapong, P. Long-term administration of melatonin attenuates neuroinflammation in the aged mouse brain. EXCLI J. 2018, 17, 634–646. [Google Scholar]
- Hardeland, R. Melatonin and inflammation-Story of a double-edged blade. J. Pineal Res. 2018, 65, e12525. [Google Scholar] [CrossRef]
- Luo, C.; Yang, Q.; Liu, Y.; Zhou, S.; Jiang, J.; Reiter, R.J.; Bhattacharya, P.; Cui, Y.; Yang, H.; Ma, H.; et al. The multiple protective roles and molecular mechanisms of melatonin and its precursor N-acetylserotonin in targeting brain injury and liver damage and in maintaining bone health. Free Radic. Biol. Med. 2019, 130, 215–233. [Google Scholar] [CrossRef]
- Ramírez-Rodríguez, G.B.; Olvera-Hernández, S.; Vega-Rivera, N.M.; Ortiz-López, L. Melatonin Influences Structural Plasticity in the Axons of Granule Cells in the Dentate Gyrus of Balb/C Mice. Int. J. Mol. Sci. 2018, 20, 73. [Google Scholar] [CrossRef] [PubMed]
- O’Neal-Moffitt, G.; Pilli, J.; Kumar, S.S.; Olcese, J. Genetic deletion of MT1/MT2 melatonin receptors enhances murine cognitive and motor performance. Neuroscience 2014, 277, 506–521. [Google Scholar] [CrossRef] [PubMed]
 ), GSK3 (
), GSK3 ( ), and Aβ plaques (
), and Aβ plaques ( ) in the absence of melatonin. Some of the dendrites and axon terminals are already degrading (
) in the absence of melatonin. Some of the dendrites and axon terminals are already degrading ( ). (B) Melatonin administration (
). (B) Melatonin administration ( ) decreases CDK-5 and GSK3 accumulations, promotes growth of new dendrites and axon terminals (
) decreases CDK-5 and GSK3 accumulations, promotes growth of new dendrites and axon terminals ( ), and flushes out some Aβ plaques.
), and flushes out some Aβ plaques.
 ), GSK3 (
), GSK3 ( ), and Aβ plaques (
), and Aβ plaques ( ) in the absence of melatonin. Some of the dendrites and axon terminals are already degrading (
) in the absence of melatonin. Some of the dendrites and axon terminals are already degrading ( ). (B) Melatonin administration (
). (B) Melatonin administration ( ) decreases CDK-5 and GSK3 accumulations, promotes growth of new dendrites and axon terminals (
) decreases CDK-5 and GSK3 accumulations, promotes growth of new dendrites and axon terminals ( ), and flushes out some Aβ plaques.
), and flushes out some Aβ plaques.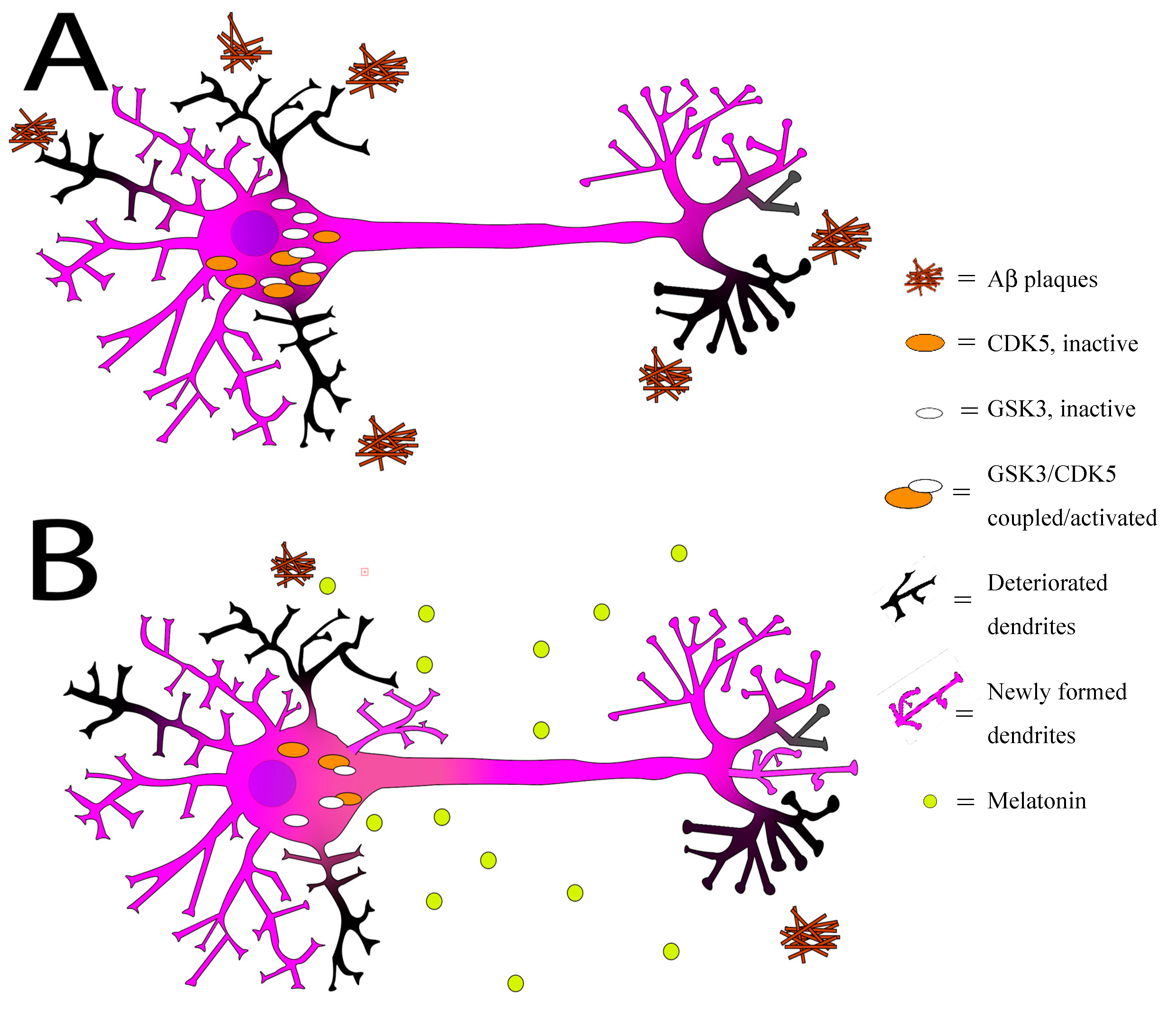
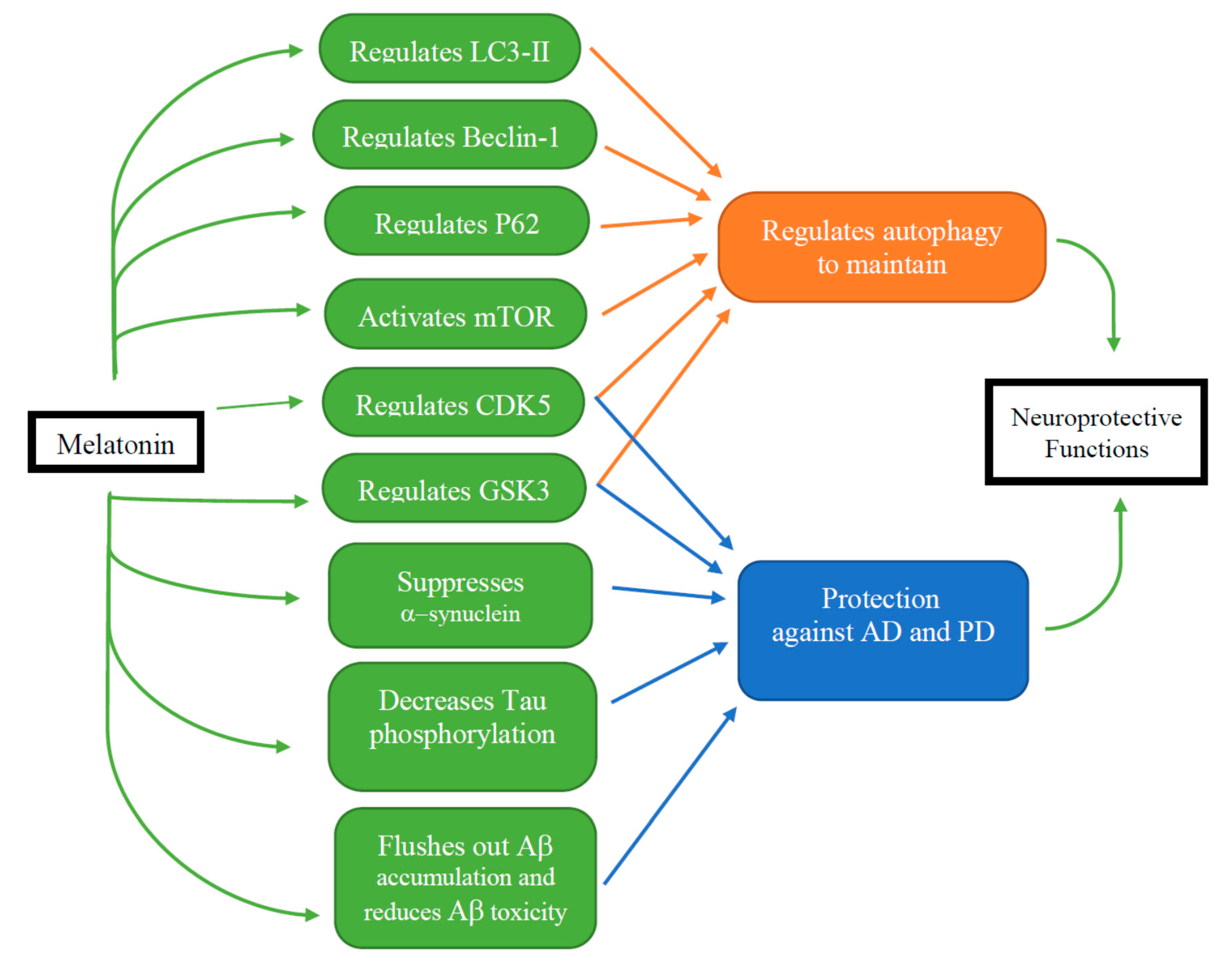
| Autophagy-Related Protein | Functions in Autophagy | References |
|---|---|---|
| ULK1 complex | Reduces phosphorylation, activates ULK1, ULK2, ATG13, ATG101, and FIP200 complexes Involved in induction | [34,35,36] |
| PI3K-III | VPS34, VPS5, AMBRA1, UVRAG, BIF1, ATG14, and Beclin1 complex Bcl-2 displacement activates VPS34 complex Participates in nucleation | [34,37,38,39] |
| ATG9 | Transmembrane proteins, only found in phagocytosis Participates in nucleation | [34,40,41,42,43] |
| Beclin1/ATG6 | Interacts with Bcl-2 Interacts with negative autophagy regulator Golgi-associated plant pathogenesis-related protein 1 (GAPR-1) | [39,44,45,46,47] |
| ATG7, ATG10 | E1 and E2-like enzyme Participates in the conjugation of ATG12-ATG5 | [34,48,49,50] |
| ATG4 | Required for autophagic elongation | [34,51,52] |
| ATG3 | Cysteine protease, cleaves LC3 | [34,53,54] |
| P62/SQSTM1 | Cellular content receptors that are preferentially degraded by autophagy | [55,56,57,58] |
| LC3/ATG8 | The only autophagy-associated protein associated with autophagosome membranes | [35,43,59,60,61] |
| NDDs | Autophagy Sites Associated with NDDs | Mechanism of Autophagy in NDDs | Changes in Autophagy | References |
|---|---|---|---|---|
| AD | PS1 and PS2 mutations | Impaired glycosylation Lysosomes damaged by acidification | Reduced autophagosomes degradation Accumulation of Aβ | [92,93,94,95] |
| PICALM decreases or disappears | Impaired phagocytic elongation, autophagosomes formation, and autophagosome-lysosomal-fusion protein | Reduced autophagosomes degradation | [96,97,98,99] | |
| Mutation of amyloid precursor protein (APP) | Increased mTOR pathway | Autophagy inhibition Aβ and tau accumulation | [86,87] | |
| Aberrant expression of P62/SQSTM1 | Hyperphosphorylated tau is associated with neurofibrillary tangles | Reduced autophagy of specific cytoplasm | [60,115,116,117,118,119] | |
| Beclin1 decreases | Defects in autophagosome synthesis | Reduced autophagosomes degradation Accumulation of Aβ | [100,101,102,103] | |
| mTOR activity is increased | Activity of autophagy reduces | Autophagy inhibition Aβ accumulation and hyperphosphorylated tau | [104,105,106,107,108,109,110,111,112,113] | |
| NRBF2 protein increases | Reduced expression of autophagy substrate SQSTM1 | Reduced APP amyloidosis | [114] | |
| PD | α-synuclein overexpression | ATG9 misalignment | Reduced macroautophagy | [133] |
| α-synuclein mutations | Inhibition of CMA binding to chaperone molecule PARP activation | Autophagy inhibition, Lewy body formation Autophagy inhibition, α- synuclein increases | [124,126,129,130,134,135,136,137] | |
| Absence of Park2 | LC3 II decreases | Downregulation of autophagy | [140] | |
| Beclin1 | Beclin1 decreases | Increased protein aggregate | [148,150] | |
| PINK1/Parkin mutation | Mitochondrial protein ubiquitin labeling reduced | Reduced autophagy in damaged mitochondria | [137,138] | |
| mTOR increases | AMPK/ mTOR-mediated autophagy/apoptosis pathway | Reduced autophagy, inhibition of MPTP induced neuron death | [146,147,148,149] | |
| HD | mHTT | Reduced antagonism to mTOR pathway | Inhibited autophagy | [157,158] |
| Beclin1 | mHTT and Beclin1 bind nonspecifically and consume Beclin1, Beclin1 level decreased | Reduced autophagosome formation and impaired autophagy | [160] | |
| Reduced ALFY mRNA expression | Reduced ALFY expression | Reduced degradation of mHTT, impaired autophagy | [162,163,164] | |
| Phosphorylation of p62/SQSTM1 | p62/SQSTM1 and ULK1 interact with each other | Increased protein aggregate clearance, impaired autophagy | [156,161] | |
| OPIDN | Beclin1 | - | Impaired autophagy, aggregation of mitochondria and vesicles | [169] |
| ALS | SOD1 mutation | Function loss of dynein | Reduced autophagy, increased protein aggregation | [175,183] |
| Gene mutation | Interact with LC3 | Increased autophagosome and autophagic matrix binding | [178,179,180] | |
| OPTN | Reduced clearance of damaged mitochondria | Reduced autophagy, increased protein aggregation | [181,182] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, F.; Sandhu, A.F.; Rungratanawanich, W.; Williams, G.E.; Akbar, M.; Zhou, S.; Song, B.-J.; Wang, X. Melatonin and Autophagy in Aging-Related Neurodegenerative Diseases. Int. J. Mol. Sci. 2020, 21, 7174. https://doi.org/10.3390/ijms21197174
Luo F, Sandhu AF, Rungratanawanich W, Williams GE, Akbar M, Zhou S, Song B-J, Wang X. Melatonin and Autophagy in Aging-Related Neurodegenerative Diseases. International Journal of Molecular Sciences. 2020; 21(19):7174. https://doi.org/10.3390/ijms21197174
Chicago/Turabian StyleLuo, Fang, Aaron F. Sandhu, Wiramon Rungratanawanich, George E. Williams, Mohammed Akbar, Shuanhu Zhou, Byoung-Joon Song, and Xin Wang. 2020. "Melatonin and Autophagy in Aging-Related Neurodegenerative Diseases" International Journal of Molecular Sciences 21, no. 19: 7174. https://doi.org/10.3390/ijms21197174
APA StyleLuo, F., Sandhu, A. F., Rungratanawanich, W., Williams, G. E., Akbar, M., Zhou, S., Song, B.-J., & Wang, X. (2020). Melatonin and Autophagy in Aging-Related Neurodegenerative Diseases. International Journal of Molecular Sciences, 21(19), 7174. https://doi.org/10.3390/ijms21197174







