Matrix Metalloproteinases and Tissue Inhibitors of Metalloproteinases in Extracellular Matrix Remodeling during Left Ventricular Diastolic Dysfunction and Heart Failure with Preserved Ejection Fraction: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Results
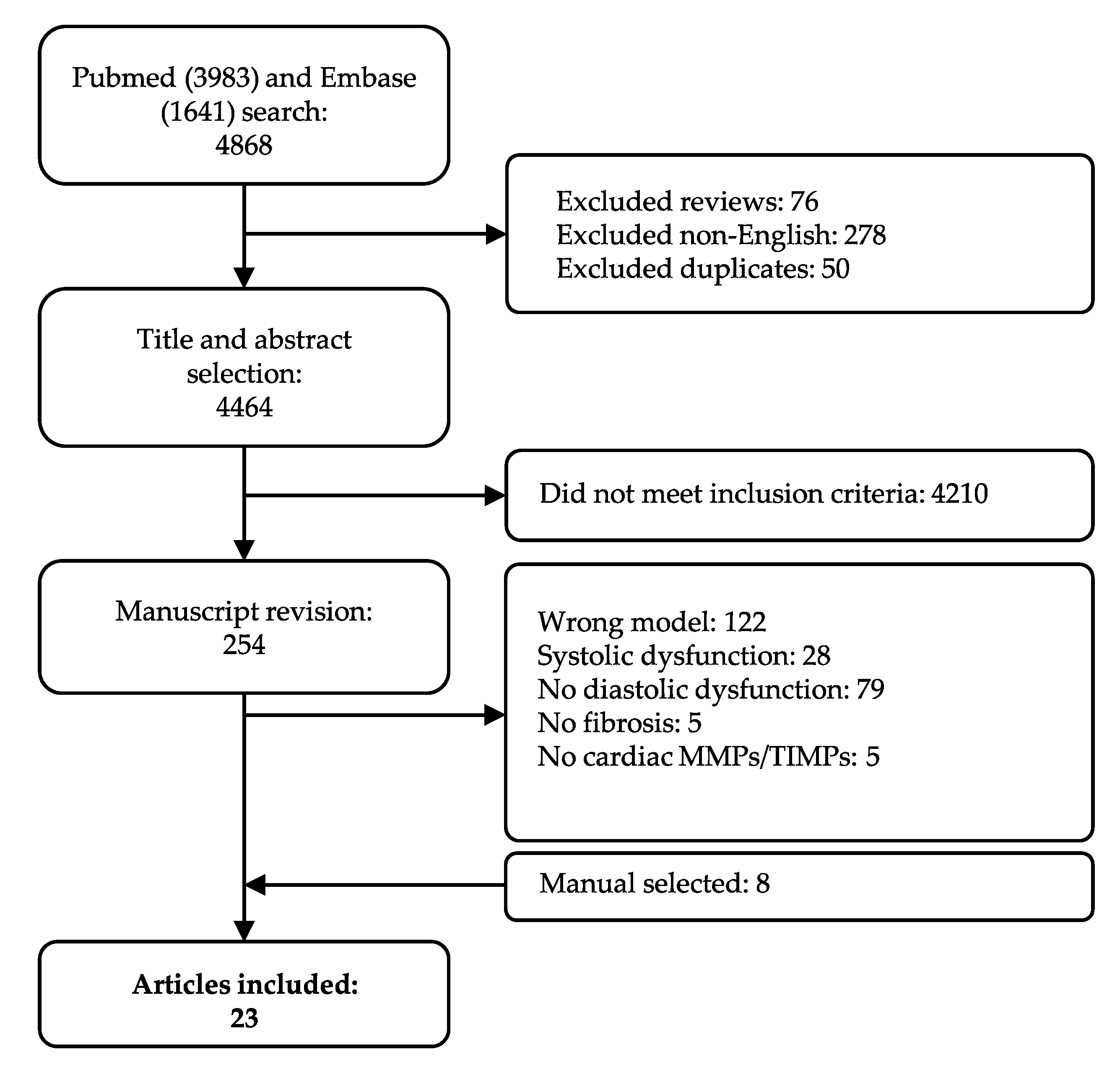
2.1. Study Population Selection and Overall Characteristics
2.2. Quality Assessment of the Studies
2.3. Meta-Analysis on Diastolic Function and Fibrosis in Models of LVDD/HFpEF
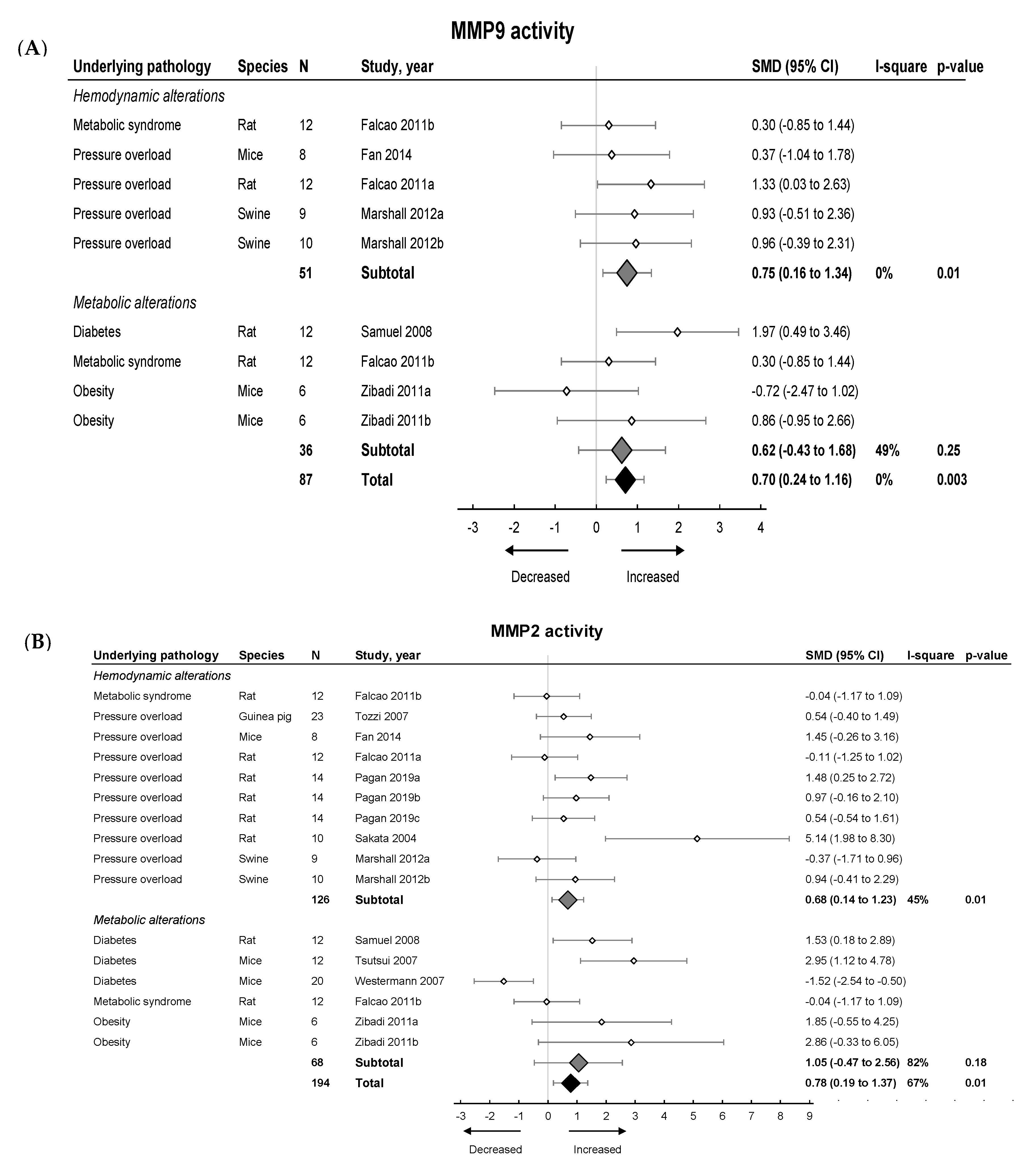
2.3.1. Meta-Analysis on MMPs and TIMPs in Pooled Models of LVDD/HFpEF
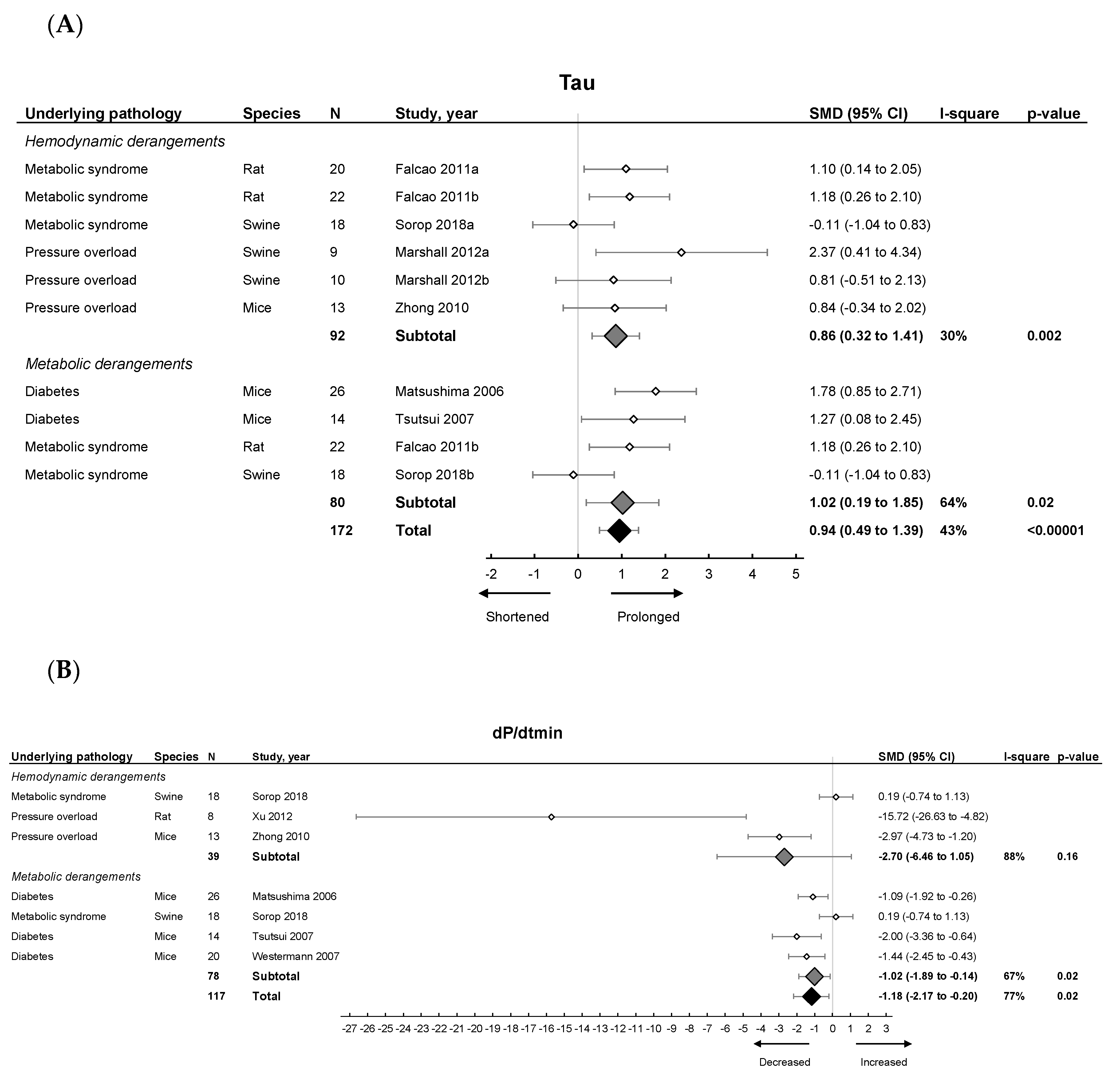
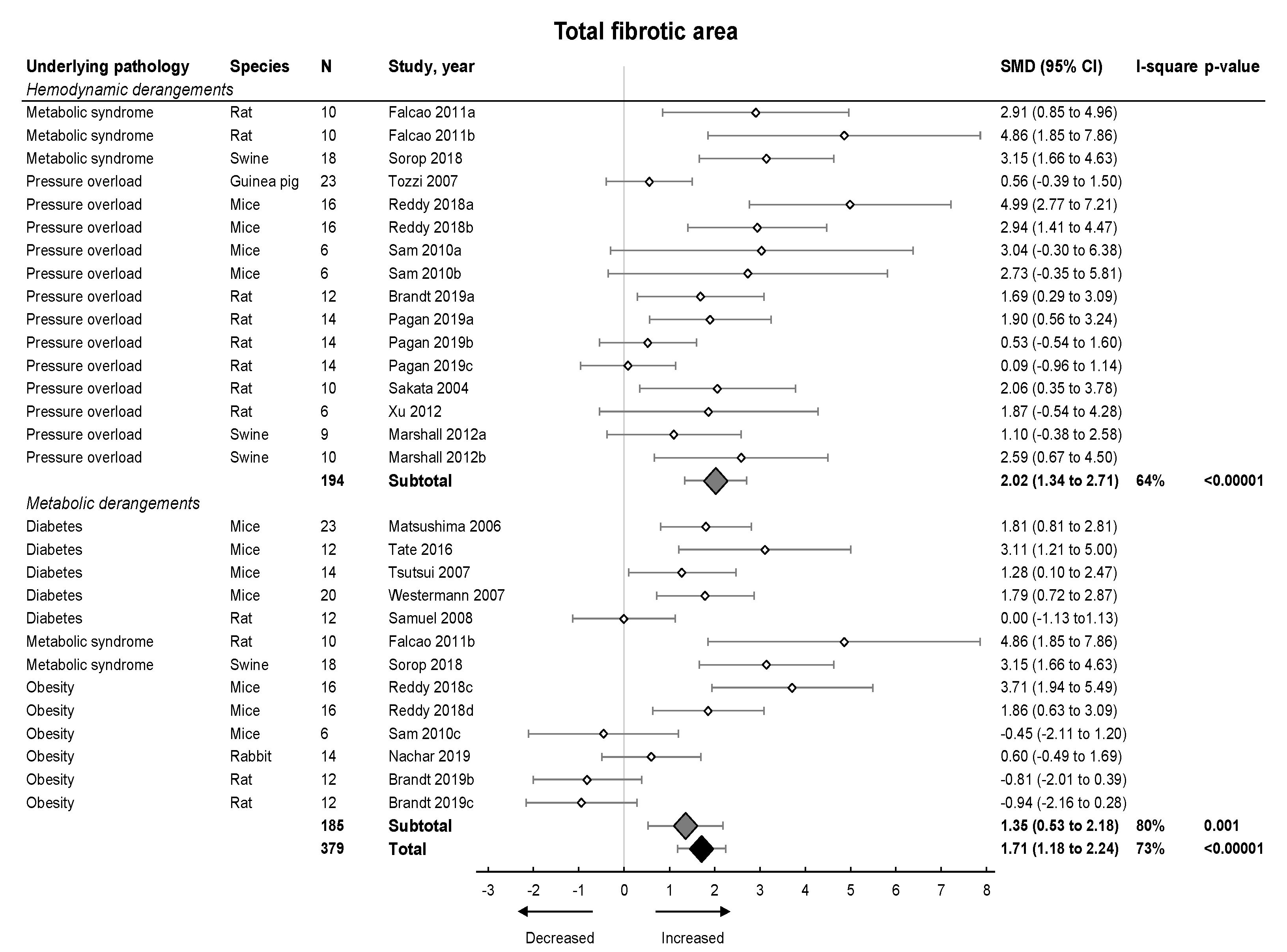
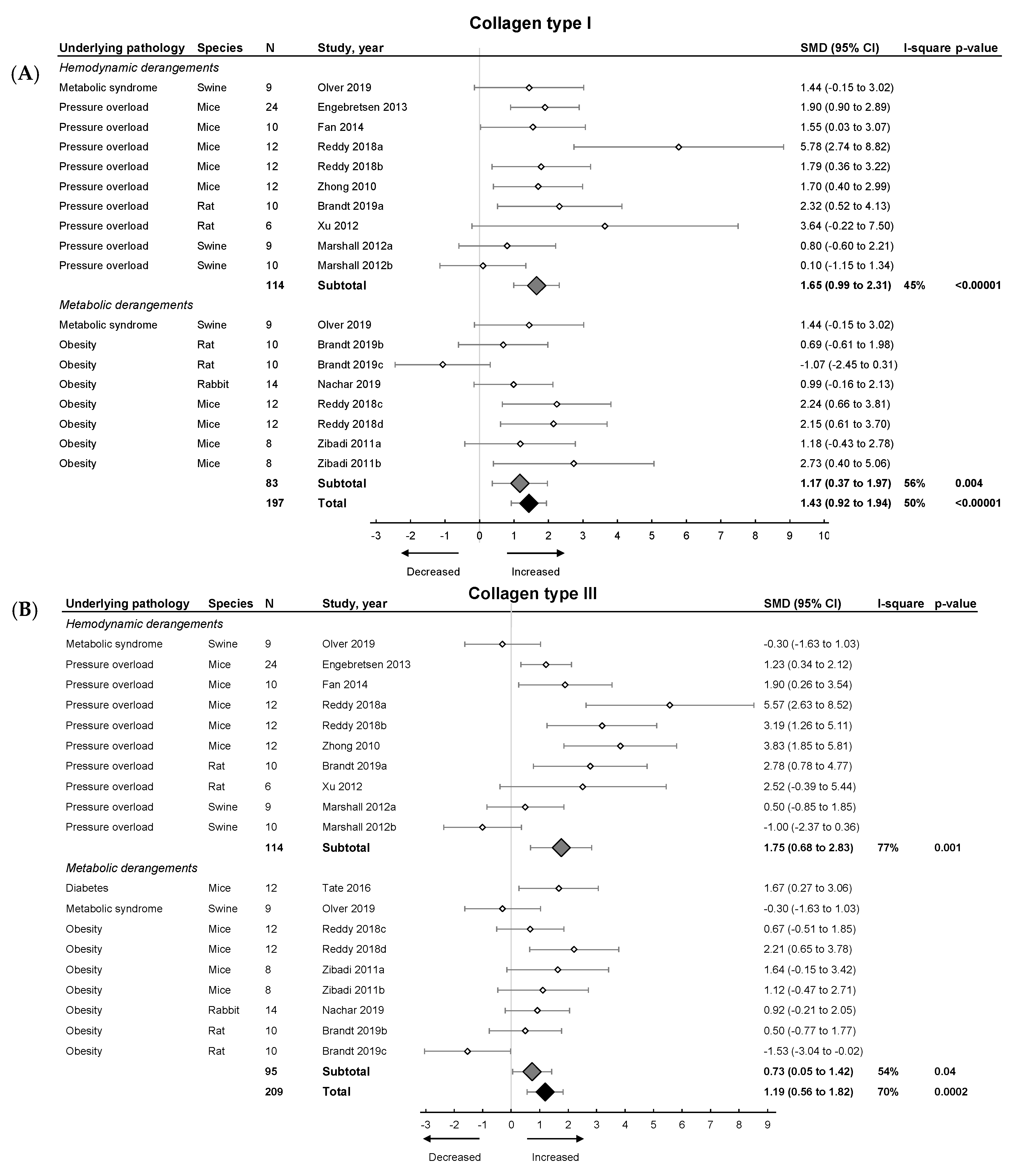
2.3.2. Meta-Analysis on MMPs and TIMPs in Models Involving Hemodynamic and Metabolic Alterations
2.3.3. Descriptive Effect on Models Involving Ageing (All in Mice)
3. Discussion
3.1. Echocardiography and Tissue Doppler Parameters of LVDD and HFpEF
3.2. Influence of Fibrosis on Development and Progression of LVDD and HFpEF
3.3. MMP and TIMP Activity in LVDD and HFpEF
3.4. Study Limitations
4. Materials and Methods
4.1. Literature Search
4.2. Study Selection
4.3. Quality Assesment
4.4. Data Extraction
4.5. Data Analysis
5. Conclusions
6. Recommendations for Future Studies on LVDD/HFpEF
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AngII | Angiotensin 2 |
| AVF | Arterio-venous fistula |
| CAD | Coronary artery disease |
| CI | Confidence interval |
| CKD | Chronic kidney disease |
| CVD | Cardiovascular disease |
| DCM | Dilated cardiomyopathy |
| DOCA | Deoxycorticosterone acetate |
| ECM | Extracellular matrix |
| EDP | End diastolic pressure |
| EDPVR | End diastolic pressure volume relationship |
| EF | Ejection fraction |
| FS | Fractional shortening |
| HF | Heart failure |
| HFpEF | Heart failure with preserved ejection fraction |
| HFrEF | Heart failure with reduced ejection fraction |
| IHC | Immunohistochemistry |
| IVRT | Isovolumic relaxation time |
| LV | Left ventricle |
| LVDD | Left ventricular diastolic dysfunction |
| MI | Myocardial infarction |
| MMP | Matrix metalloproteinase |
| SHR | Spontaneously-hypertensive rat |
| SMD | Standardized mean difference |
| SR | Sirius red |
| STZ | Streptozotocin |
| TAC | Transaortic constriction |
| TIMP | Tissue inhibitor of metalloproteinase |
| WB | Western blot |
References
- Kloch-Badelek, M.; Kuznetsova, T.; Sakiewicz, W.; Tikhonoff, V.; Ryabikov, A.; Gonzalez, A.; Lopez, B.; Thijs, L.; Jin, Y.; Malyutina, S.; et al. Prevalence of left ventricular diastolic dysfunction in European populations based on cross-validated diagnostic thresholds. Cardiovasc. Ultrasound 2012, 10, 10. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen-Torvik, L.J.; Colangelo, L.A.; Lima, J.A.C.; Jacobs, D.R.; Rodriguez, C.J.; Gidding, S.S.; Lloyd-Jones, D.M.; Shah, S.J. Prevalence and predictors of diastolic dysfunction according to different classification criteria: The coronary artery risk development in young in adults study. Am. J. Epidemiol. 2017, 185, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Palmiero, P.; Zito, A.; Maiello, M.; Cameli, M.; Modesti, P.A.; Muiesan, M.L.; Novo, S.; Saba, P.S.; Scicchitano, P.; Pedrinelli, R.; et al. Left ventricular diastolic function in hypertension: Methodological considerations and clinical implications. J. Clin. Med. Res. 2015, 7, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Nagueh, S.F.; Smiseth, O.A.; Appleton, C.P.; Byrd, B.F.; Dokainish, H.; Edvardsen, T.; Flachskampf, F.A.; Gillebert, T.C.; Klein, A.L.; Lancellotti, P.; et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: An update from the american society of echocardiography and the european association of cardiovascular imaging. Eur. J. Echocardiogr. 2016, 17, 1321–1360. [Google Scholar] [CrossRef]
- Mitter, S.S.; Shah, S.J.; Thomas, J.D. A Test in context: E/A and E/e’ to assess diastolic dysfunction and LV filling pressure. J. Am. Coll Cardiol. 2017, 69, 1451–1464. [Google Scholar] [CrossRef]
- Valstar, G.B.; Bots, S.H.; Groepenhoff, F.; Gohar, A.; Rutten, F.H.; Leiner, T.; Cramer, M.J.M.; Teske, A.J.; Suciadi, L.P.; Menken, R.; et al. Discovery of biomarkers for the presence and progression of left ventricular diastolic dysfunction and heart failure with preserved ejection fraction in patients at risk for cardiovascular disease: Rationale and design of the helpful case-cohort study in a dutch cardiology outpatient clinic. BMJ Open 2019, 9, e028408. [Google Scholar] [CrossRef] [PubMed]
- Guan, Z.; Liu, S.; Wang, Y.; Meng, P.; Zheng, X.; Jia, D.; Yang, J.; Ma, C. Left ventricular systolic dysfunction potentially contributes to the symptoms in heart failure with preserved ejection fraction. Echocardiography 2019, 36, 1825–1833. [Google Scholar] [CrossRef]
- Senni, M.; Paulus, W.J.; Gavazzi, A.; Fraser, A.G.; Diez, J.; Solomon, S.D.; Smiseth, O.A.; Guazzi, M.; Lam, C.S.; Maggioni, A.P.; et al. New strategies for heart failure with preserved ejection fraction: The importance of targeted therapies for heart failure phenotypes. Eur. Heart J. 2014, 35, 2797–2815. [Google Scholar] [CrossRef]
- Bhatia, R.S.; Tu, J.V.; Lee, D.S.; Austin, P.C.; Fang, J.; Haouzi, A.; Gong, Y.; Liu, P.P. Outcome of heart failure with preserved ejection fraction in a population-based study. N. Engl. J. Med. 2006, 355, 260–269. [Google Scholar] [CrossRef]
- Devereux, R.B.; Roman, M.J.; Liu, J.E.; Welty, T.K.; Lee, E.L.; Rodeheffer, R.; Fabsitz, R.R.; Howard, B.V. Congestive heart failure despite normal left ventricular systolic function in a population-based sample: The strong heart study. Am. J. Cardiol. 2000, 86, 1090–1096. [Google Scholar] [CrossRef]
- Valero-Munoz, M.; Backman, W.; Sam, F. Murine models of heart failure with preserved ejection fraction: A “fishing expedition”. JACC Basic Transl. Sci. 2017, 2, 770–789. [Google Scholar] [CrossRef] [PubMed]
- Scantlebury, D.C.; Borlaug, B.A. Why are women more likely than men to develop heart failure with preserved ejection fraction? Curr. Opin. Cardiol. 2011, 26, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Dewan, P.; Rorth, R.; Raparelli, V.; Campbell, R.T.; Shen, L.; Jhund, P.S.; Petrie, M.C.; Anand, I.S.; Carson, P.E.; Desai, A.S.; et al. Sex-related differences in heart failure with preserved ejection fraction. Circ. Heart Fail. 2019, 12, e006539. [Google Scholar] [CrossRef] [PubMed]
- Borlaug, B.A.; Redfield, M.M. Diastolic and systolic heart failure are distinct phenotypes within the heart failure spectrum. Circulation 2011, 123, 2006–2014. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, A.M.; St Sauver, J.L.; Gerber, Y.; Manemann, S.M.; Boyd, C.M.; Dunlay, S.M.; Rocca, W.A.; Finney Rutten, L.J.; Jiang, R.; Weston, S.A.; et al. Multimorbidity in heart failure: A community perspective. Am. J. Med. 2015, 128, 38–45. [Google Scholar] [CrossRef]
- Swierblewska, E.; Wolf, J.; Kunicka, K.; Graff, B.; Polonis, K.; Hoffmann, M.; Chrostowska, M.; Szyndler, A.; Bandosz, P.; Graff, B.; et al. Prevalence and distribution of left ventricular diastolic dysfunction in treated patients with long-lasting hypertension. Blood Press. 2018, 27, 376–384. [Google Scholar] [CrossRef]
- Van De Wouw, J.; Broekhuizen, M.; Sorop, O.; Joles, J.A.; Verhaar, M.C.; Duncker, D.J.; Danser, A.H.J.; Merkus, D. Chronic kidney disease as a risk factor for heart failure with preserved ejection fraction: A focus on microcirculatory factors and therapeutic targets. Front. Physiol. 2019, 10, 1108. [Google Scholar] [CrossRef]
- Bouthoorn, S.; Valstar, G.B.; Gohar, A.; den Ruijter, H.M.; Reitsma, H.B.; Hoes, A.W.; Rutten, F.H. The prevalence of left ventricular diastolic dysfunction and heart failure with preserved ejection fraction in men and women with type 2 diabetes: A systematic review and meta-analysis. Diab. Vasc. Dis. Res. 2018, 15, 477–493. [Google Scholar] [CrossRef]
- Russo, C.; Jin, Z.; Homma, S.; Rundek, T.; Elkind, M.S.; Sacco, R.L.; Di Tullio, M.R. Effect of obesity and overweight on left ventricular diastolic function: A community-based study in an elderly cohort. J. Am. Coll. Cardiol. 2011, 57, 1368–1374. [Google Scholar] [CrossRef]
- Savji, N.; Meijers, W.C.; Bartz, T.M.; Bhambhani, V.; Cushman, M.; Nayor, M.; Kizer, J.R.; Sarma, A.; Blaha, M.J.; Gansevoort, R.T.; et al. The association of obesity and cardiometabolic traits with incident HFpEF and HFrEF. JACC Heart Fail. 2018, 6, 701–709. [Google Scholar] [CrossRef]
- van Riet, E.E.; Hoes, A.W.; Wagenaar, K.P.; Limburg, A.; Landman, M.A.; Rutten, F.H. Epidemiology of heart failure: The prevalence of heart failure and ventricular dysfunction in older adults over time. A systematic review. Eur. J. Heart Fail. 2016, 18, 242–252. [Google Scholar] [CrossRef]
- Mohammed, S.F.; Hussain, S.; Mirzoyev, S.A.; Edwards, W.D.; Maleszewski, J.J.; Redfield, M.M. Coronary microvascular rarefaction and myocardial fibrosis in heart failure with preserved ejection fraction. Circulation 2015, 131, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Mocan, M.; Mocan Hognogi, L.D.; Anton, F.P.; Chiorescu, R.M.; Goidescu, C.M.; Stoia, M.A.; Farcas, A.D. Biomarkers of inflammation in left ventricular diastolic dysfunction. Dis. Markers 2019, 2019, 7583690. [Google Scholar] [CrossRef] [PubMed]
- de Boer, R.A.; De Keulenaer, G.; Bauersachs, J.; Brutsaert, D.; Cleland, J.G.; Diez, J.; Du, X.J.; Ford, P.; Heinzel, F.R.; Lipson, K.E.; et al. Towards better definition, quantification and treatment of fibrosis in heart failure. A scientific roadmap by the Committee of Translational Research of the Heart Failure Association (HFA) of the European Society of Cardiology. Eur. J. Heart Fail. 2019, 21, 272–285. [Google Scholar] [CrossRef] [PubMed]
- Frangogiannis, N.G. The extracellular matrix in myocardial injury, repair, and remodeling. J. Clin. Investig. 2017, 127, 1600–1612. [Google Scholar] [CrossRef]
- Li, Y.Y.; McTiernan, C.F.; Feldman, A.M. Proinflammatory cytokines regulate tissue inhibitors of metalloproteinases and disintegrin metalloproteinase in cardiac cells. Cardiovasc. Res. 1999, 42, 162–172. [Google Scholar] [CrossRef]
- Fan, D.; Takawale, A.; Lee, J.; Kassiri, Z. Cardiac fibroblasts, fibrosis and extracellular matrix remodeling in heart disease. Fibrogenesis Tissue Repair 2012, 5, 15. [Google Scholar] [CrossRef]
- Reinhardt, D.; Sigusch, H.H.; Henβe, J.; Tyagi, S.C.; Körfer, R.; Figulla, H.R. Cardiac remodelling in end stage heart failure: Upregulation of matrix metalloproteinase (MMP) irrespective of the underlying disease, and evidence for a direct inhibitory effect of ACE inhibitors on MMP. Heart 2002, 88, 525–530. [Google Scholar] [CrossRef]
- Antonov, I.B.; Kozlov, K.L.; Pal’Tseva, E.M.; Polyakova, O.V.; Lin’Kova, N.S. Matrix metalloproteinases MMP-1 and MMP-9 and their inhibitor TIMP-1 as markers of dilated cardiomyopathy in patients of different age. Bull. Exp. Boil. Med. 2018, 164, 550–553. [Google Scholar] [CrossRef]
- Ngu, J.M.; Teng, G.; Meijndert, H.C.; Mewhort, H.E.; Turnbull, J.D.; Stetler-Stevenson, W.G.; Fedak, P.W. Human cardiac fibroblast extracellular matrix remodeling: Dual effects of tissue inhibitor of metalloproteinase-2. Cardiovasc. Pathol. 2014, 23, 335–343. [Google Scholar] [CrossRef]
- Pfeffer, M.A.; Shah, A.M.; Borlaug, B.A. Heart failure with preserved ejection fraction in perspective. Circ. Res. 2019, 124, 1598–1617. [Google Scholar] [CrossRef] [PubMed]
- Sava, R.I.; Pepine, C.J.; March, K.L. Immune dysregulation in HFpEF: A target for mesenchymal stem/stromal cell therapy. J. Clin. Med. 2020, 9, 241. [Google Scholar] [CrossRef] [PubMed]
- Zachariah, J.P.; Colan, S.D.; Lang, P.; Triedman, J.K.; Alexander, M.E.; Walsh, E.P.; Berul, C.I.; Cecchin, F. Circulating matrix metalloproteinases in adolescents with hypertrophic cardiomyopathy and ventricular arrhythmia. Circ. Heart Fail. 2012, 5, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Eiros, R.; Romero-Gonzalez, G.; Gavira, J.J.; Beloqui, O.; Colina, I.; Fortun Landecho, M.; Lopez, B.; Gonzalez, A.; Diez, J.; Ravassa, S. Does chronic kidney disease facilitate malignant myocardial fibrosis in heart failure with preserved ejection fraction of hypertensive origin? J. Clin. Med. 2020, 9, 404. [Google Scholar] [CrossRef] [PubMed]
- Agrinier, N.; Thilly, N.; Boivin, J.M.; Dousset, B.; Alla, F.; Zannad, F. Prognostic value of serum PIIINP, MMP1 and TIMP1 levels in hypertensive patients: A community-based prospective cohort study. Fundam. Clin. Pharmacol. 2013, 27, 572–580. [Google Scholar] [CrossRef]
- DeLeon-Pennell, K.Y.; Meschiari, C.A.; Jung, M.; Lindsey, M.L. Matrix metalloproteinases in myocardial infarction and heart failure. Prog. Mol. Biol. Transl. Sci. 2017, 147, 75–100. [Google Scholar] [CrossRef] [PubMed]
- Chiao, Y.A.; Ramirez, T.A.; Zamilpa, R.; Okoronkwo, S.M.; Dai, Q.; Zhang, J.; Jin, Y.F.; Lindsey, M.L. Matrix metalloproteinase-9 deletion attenuates myocardial fibrosis and diastolic dysfunction in ageing mice. Cardiovasc. Res. 2012, 96, 444–455. [Google Scholar] [CrossRef]
- Ma, Y.; Chiao, Y.A.; Zhang, J.; Manicone, A.M.; Jin, Y.F.; Lindsey, M.L. Matrix metalloproteinase-28 deletion amplifies inflammatory and extracellular matrix responses to cardiac aging. Microsc. Microanal. 2012, 18, 81–90. [Google Scholar] [CrossRef]
- Engebretsen, K.V.; Lunde, I.G.; Strand, M.E.; Waehre, A.; Sjaastad, I.; Marstein, H.S.; Skrbic, B.; Dahl, C.P.; Askevold, E.T.; Christensen, G.; et al. Lumican is increased in experimental and clinical heart failure, and its production by cardiac fibroblasts is induced by mechanical and proinflammatory stimuli. FEBS J. 2013, 280, 2382–2398. [Google Scholar] [CrossRef]
- Fan, D.; Takawale, A.; Basu, R.; Patel, V.; Lee, J.; Kandalam, V.; Wang, X.; Oudit, G.Y.; Kassiri, Z. Differential role of TIMP2 and TIMP3 in cardiac hypertrophy, fibrosis, and diastolic dysfunction. Cardiovasc. Res. 2014, 103, 268–280. [Google Scholar] [CrossRef]
- Reddy, S.S.; Agarwal, H.; Barthwal, M.K. Cilostazol ameliorates heart failure with preserved ejection fraction and diastolic dysfunction in obese and non-obese hypertensive mice. J. Mol. Cell. Cardiol. 2018, 123, 46–57. [Google Scholar] [CrossRef]
- Sam, F.; Duhaney, T.A.; Sato, K.; Wilson, R.M.; Ohashi, K.; Sono-Romanelli, S.; Higuchi, A.; De Silva, D.S.; Qin, F.; Walsh, K.; et al. Adiponectin deficiency, diastolic dysfunction, and diastolic heart failure. Endocrinology 2010, 151, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Basu, R.; Guo, D.; Chow, F.L.; Byrns, S.; Schuster, M.; Loibner, H.; Wang, X.H.; Penninger, J.M.; Kassiri, Z.; et al. Angiotensin-converting enzyme 2 suppresses pathological hypertrophy, myocardial fibrosis, and cardiac dysfunction. Circulation 2010, 122, 717–728. [Google Scholar] [CrossRef] [PubMed]
- Westermann, D.; Rutschow, S.; Jager, S.; Linderer, A.; Anker, S.; Riad, A.; Unger, T.; Schultheiss, H.P.; Pauschinger, M.; Tschope, C. Contributions of inflammation and cardiac matrix metalloproteinase activity to cardiac failure in diabetic cardiomyopathy: The role of angiotensin type 1 receptor antagonism. Diabetes 2007, 56, 641–646. [Google Scholar] [CrossRef] [PubMed]
- Tate, M.; Robinson, E.; Green, B.D.; McDermott, B.J.; Grieve, D.J. Exendin-4 attenuates adverse cardiac remodelling in streptozocin-induced diabetes via specific actions on infiltrating macrophages. Basic Res. Cardiol. 2016, 111, 1. [Google Scholar] [CrossRef] [PubMed]
- Matsushima, S.; Kinugawa, S.; Ide, T.; Matsusaka, H.; Inoue, N.; Ohta, Y.; Yokota, T.; Sunagawa, K.; Tsutsui, H. Overexpression of glutathione peroxidase attenuates myocardial remodeling and preserves diastolic function in diabetic heart. Am. J. Physiol. Heart Circ. Physiol. 2006, 291, H2237–H2245. [Google Scholar] [CrossRef]
- Tsutsui, H.; Matsushima, S.; Kinugawa, S.; Ide, T.; Inoue, N.; Ohta, Y.; Yokota, T.; Hamaguchi, S.; Sunagawa, K. Angiotensin II type 1 receptor blocker attenuates myocardial remodeling and preserves diastolic function in diabetic heart. Hypertens Res. 2007, 30, 439–449. [Google Scholar] [CrossRef]
- Zibadi, S.; Cordova, F.; Slack, E.H.; Watson, R.R.; Larson, D.F. Leptin’s regulation of obesity-induced cardiac extracellular matrix remodeling. Cardiovasc. Toxicol. 2011, 11, 325–333. [Google Scholar] [CrossRef]
- Brandt, M.M.; Nguyen, I.T.N.; Krebber, M.M.; van de Wouw, J.; Mokry, M.; Cramer, M.J.; Duncker, D.J.; Verhaar, M.C.; Joles, J.A.; Cheng, C. Limited synergy of obesity and hypertension, prevalent risk factors in onset and progression of heart failure with preserved ejection fraction. J. Cell. Mol. Med. 2019, 23, 6666–6678. [Google Scholar] [CrossRef]
- Falcao-Pires, I.; Palladini, G.; Goncalves, N.; Van Der Velden, J.; Moreira-Goncalves, D.; Miranda-Silva, D.; Salinaro, F.; Paulus, W.J.; Niessen, H.W.; Perlini, S.; et al. Distinct mechanisms for diastolic dysfunction in diabetes mellitus and chronic pressure-overload. Basic Res. Cardiol. 2011, 106, 801–814. [Google Scholar] [CrossRef]
- Pagan, L.U.; Damatto, R.L.; Gomes, M.J.; Lima, A.R.R.; Cezar, M.D.M.; Damatto, F.C.; Reyes, D.R.A.; Caldonazo, T.M.M.; Polegato, B.F.; Okoshi, M.P.; et al. Low-intensity aerobic exercise improves cardiac remodelling of adult spontaneously hypertensive rats. J. Cell. Mol. Med. 2019, 23, 6504–6507. [Google Scholar] [CrossRef] [PubMed]
- Sakata, Y.; Yamamoto, K.; Mano, T.; Nishikawa, N.; Yoshida, J.; Hori, M.; Miwa, T.; Masuyama, T. Activation of matrix metalloproteinases precedes left ventricular remodeling in hypertensive heart failure rats: Its inhibition as a primary effect of angiotensin-converting enzyme inhibitor. Circulation 2004, 109, 2143–2149. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Ding, F.; Pang, J.; Gao, X.; Xu, R.K.; Hao, W.; Cao, J.M.; Chen, C. Chronic administration of hexarelin attenuates cardiac fibrosis in the spontaneously hypertensive rat. Am. J. Physiol. Heart Circ. Physiol. 2012, 303, H703–H711. [Google Scholar] [CrossRef] [PubMed]
- Samuel, C.S.; Hewitson, T.D.; Zhang, Y.; Kelly, D.J. Relaxin ameliorates fibrosis in experimental diabetic cardiomyopathy. Endocrinology 2008, 149, 3286–3293. [Google Scholar] [CrossRef] [PubMed]
- Marshall, K.D.; Muller, B.N.; Krenz, M.; Hanft, L.M.; McDonald, K.S.; Dellsperger, K.C.; Emter, C.A. Heart failure with preserved ejection fraction: Chronic low-intensity interval exercise training preserves myocardial O2 balance and diastolic function. J. Appl. Physiol. 2013, 114, 131–147. [Google Scholar] [CrossRef] [PubMed]
- Olver, T.D.; Edwards, J.C.; Jurrissen, T.J.; Veteto, A.B.; Jones, J.L.; Gao, C.; Rau, C.; Warren, C.M.; Klutho, P.J.; Alex, L.; et al. Western diet-fed, aortic-banded ossabaw swine: A preclinical model of cardio-metabolic heart failure. JACC Basic Transl. Sci. 2019, 4, 404–421. [Google Scholar] [CrossRef]
- Sorop, O.; Heinonen, I.; van Kranenburg, M.; Van De Wouw, J.; de Beer, V.J.; Nguyen, I.T.N.; Octavia, Y.; van Duin, R.W.B.; Stam, K.; Van Geuns, R.J.; et al. Multiple common comorbidities produce left ventricular diastolic dysfunction associated with coronary microvascular dysfunction, oxidative stress, and myocardial stiffening. Cardiovasc. Res. 2018, 114, 954–964. [Google Scholar] [CrossRef]
- Nachar, W.; Merlet, N.; Maafi, F.; Shi, Y.; Mihalache-Avram, T.; Mecteau, M.; Ferron, M.; Rheaume, E.; Tardif, J.C. Cardiac inflammation and diastolic dysfunction in hypercholesterolemic rabbits. PLoS ONE 2019, 14, e0220707. [Google Scholar] [CrossRef]
- Tozzi, R.; Palladini, G.; Fallarini, S.; Nano, R.; Gatti, C.; Presotto, C.; Schiavone, A.; Micheletti, R.; Ferrari, P.; Fogari, R.; et al. Matrix metalloprotease activity is enhanced in the compensated but not in the decompensated phase of pressure overload hypertrophy. Am. J. Hypertens 2007, 20, 663–669. [Google Scholar] [CrossRef]
- Ferrari, R.; Bohm, M.; Cleland, J.G.; Paulus, W.J.; Pieske, B.; Rapezzi, C.; Tavazzi, L. Heart failure with preserved ejection fraction: Uncertainties and dilemmas. Eur. J. Heart Fail. 2015, 17, 665–671. [Google Scholar] [CrossRef]
- Thomas, L.; Marwick, T.H.; Popescu, B.A.; Donal, E.; Badano, L.P. Left atrial structure and function, and left ventricular diastolic dysfunction: JACC state-of-the-art review. J. Am. Coll. Cardiol. 2019, 73, 1961–1977. [Google Scholar] [CrossRef] [PubMed]
- Nagueh, S.F. Left ventricular diastolic function: Understanding pathophysiology, diagnosis, and prognosis with echocardiography. JACC Cardiovasc. Imaging 2020, 13, 228–244. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Khoury, D.S.; Yue, Y.; Torre-Amione, G.; Nagueh, S.F. Preserved left ventricular twist and circumferential deformation, but depressed longitudinal and radial deformation in patients with diastolic heart failure. Eur. Heart J. 2008, 29, 1283–1289. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, A.; Magoon, R.; Malik, V.; Kapoor, P.M.; Ramakrishnan, S. Studying diastology with speckle tracking echocardiography: The essentials. Ann. Card. Anaesth. 2017, 20, S57–S60. [Google Scholar] [CrossRef] [PubMed]
- Mottram, P.M.; Marwick, T.H. Assessment of diastolic function: What the general cardiologist needs to know. Heart 2005, 91, 681–695. [Google Scholar] [CrossRef] [PubMed]
- Garcia, M.J.; Thomas, J.D.; Klein, A.L. New Doppler echocardiographic applications for the study of diastolic function. J. Am. Coll. Cardiol. 1998, 32, 865–875. [Google Scholar] [CrossRef]
- Schnelle, M.; Catibog, N.; Zhang, M.; Nabeebaccus, A.A.; Anderson, G.; Richards, D.A.; Sawyer, G.; Zhang, X.; Toischer, K.; Hasenfuss, G.; et al. Echocardiographic evaluation of diastolic function in mouse models of heart disease. J. Mol. Cell. Cardiol. 2018, 114, 20–28. [Google Scholar] [CrossRef]
- Schaefer, A.; Meyer, G.P.; Brand, B.; Hilfiker-Kleiner, D.; Drexler, H.; Klein, G. Effects of anesthesia on diastolic function in mice assessed by echocardiography. Echocardiography 2005, 22, 665–670. [Google Scholar] [CrossRef]
- Heerdt, P.M.; Gandhi, C.D.; Dickstein, M.L. Disparity of isoflurane effects on left and right ventricular afterload and hydraulic power generation in swine. Anesth. Analg. 1998, 87, 511–521. [Google Scholar]
- Hettrick, D.A.; Pagal, P.S.; Kersten, J.R.; Lowe, D.; Warltier, D.C. The effects of isoflurane and halothane on left ventricular afterload in dogs with dilated cardiomyopathy. Anesth. Analg. 1997, 85, 979–986. [Google Scholar] [CrossRef]
- Shillcutt, S.K.; Chacon, M.M.; Brakke, T.R.; Roberts, E.K.; Schulte, T.E.; Markin, N. Heart failure with preserved ejection fraction: A perioperative review. J. Cardiothorac. Vasc. Anesth. 2017, 31, 1820–1830. [Google Scholar] [CrossRef] [PubMed]
- Penicka, M.; Bartunek, J.; Trakalova, H.; Hrabakova, H.; Maruskova, M.; Karasek, J.; Kocka, V. Heart failure with preserved ejection fraction in outpatients with unexplained dyspnea: A pressure-volume loop analysis. J. Am. Coll. Cardiol. 2010, 55, 1701–1710. [Google Scholar] [CrossRef] [PubMed]
- Runte, K.E.; Bell, S.P.; Selby, D.E.; Haussler, T.N.; Ashikaga, T.; LeWinter, M.M.; Palmer, B.M.; Meyer, M. Relaxation and the role of calcium in isolated contracting myocardium from patients with hypertensive heart disease and heart failure with preserved ejection fraction. Circ. Heart Fail. 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Kasner, M.; Westermann, D.; Lopez, B.; Gaub, R.; Escher, F.; Kuhl, U.; Schultheiss, H.P.; Tschope, C. Diastolic tissue Doppler indexes correlate with the degree of collagen expression and cross-linking in heart failure and normal ejection fraction. J. Am. Coll. Cardiol. 2011, 57, 977–985. [Google Scholar] [CrossRef]
- Van Dijk, C.G.; Oosterhuis, N.R.; Xu, Y.J.; Brandt, M.; Paulus, W.J.; Van Heerebeek, L.; Duncker, D.J.; Verhaar, M.C.; Fontoura, D.; Lourenco, A.P.; et al. Distinct endothelial cell responses in the heart and kidney microvasculature characterize the progression of heart failure with preserved ejection fraction in the obese ZSF1 rat with cardiorenal metabolic syndrome. Circ. Heart Fail. 2016, 9, e002760. [Google Scholar] [CrossRef] [PubMed]
- She, G.; Ren, Y.J.; Wang, Y.; Hou, M.C.; Wang, H.F.; Gou, W.; Lai, B.C.; Lei, T.; Du, X.J.; Deng, X.L. KCa3.1 channels promote cardiac fibrosis through mediating inflammation and differentiation of monocytes into myofibroblasts in angiotensin II-treated rats. J. Am. Heart Assoc. 2019, 8, e010418. [Google Scholar] [CrossRef]
- Santiago, J.J.; Dangerfield, A.L.; Rattan, S.G.; Bathe, K.L.; Cunnington, R.H.; Raizman, J.E.; Bedosky, K.M.; Freed, D.H.; Kardami, E.; Dixon, I.M. Cardiac fibroblast to myofibroblast differentiation in vivo and in vitro: Expression of focal adhesion components in neonatal and adult rat ventricular myofibroblasts. Dev. Dyn. 2010, 239, 1573–1584. [Google Scholar] [CrossRef]
- Rosenkranz, S. TGF-beta1 and angiotensin networking in cardiac remodeling. Cardiovasc. Res. 2004, 63, 423–432. [Google Scholar] [CrossRef]
- Das, S.; Frisk, C.; Eriksson, M.J.; Walentinsson, A.; Corbascio, M.; Hage, C.; Kumar, C.; Asp, M.; Lundeberg, J.; Maret, E.; et al. Transcriptomics of cardiac biopsies reveals differences in patients with or without diagnostic parameters for heart failure with preserved ejection fraction. Sci. Rep. 2019, 9, 3179. [Google Scholar] [CrossRef]
- Martos, R.; Baugh, J.; Ledwidge, M.; O’Loughlin, C.; Murphy, N.F.; Conlon, C.; Patle, A.; Donnelly, S.C.; McDonald, K. Diagnosis of heart failure with preserved ejection fraction: Improved accuracy with the use of markers of collagen turnover. Eur. J. Heart Fail. 2009, 11, 191–197. [Google Scholar] [CrossRef]
- Zile, M.R.; Desantis, S.M.; Baicu, C.F.; Stroud, R.E.; Thompson, S.B.; McClure, C.D.; Mehurg, S.M.; Spinale, F.G. Plasma biomarkers that reflect determinants of matrix composition identify the presence of left ventricular hypertrophy and diastolic heart failure. Circ. Heart Fail. 2011, 4, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Müller-Brunotte, R.; Kahan, T.; López, B.; Edner, M.; González, A.; Díez, J.; Malmqvist, K. Myocardial fibrosis and diastolic dysfunction in patients with hypertension: Results from the Swedish Irbesartan Left Ventricular Hypertrophy Investigation versus Atenolol (SILVHIA). J. Hypertens. 2007, 25, 1958–1966. [Google Scholar] [CrossRef]
- Xu, H.; Qing, T.; Shen, Y.; Huang, J.; Liu, Y.; Li, J.; Zhen, T.; Xing, K.; Zhu, S.; Luo, M. RNA-seq analyses the effect of high-salt diet in hypertension. Gene 2018, 677, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, M.M.; Maxwell, P.; Dunn, F.G. TIMP-1: A marker of left ventricular diastolic dysfunction and fibrosis in hypertension. Hypertension 2002, 40, 136–141. [Google Scholar] [CrossRef]
- Moore, L.; Fan, D.; Basu, R.; Kandalam, V.; Kassiri, Z. Tissue inhibitor of metalloproteinases (TIMPs) in heart failure. Heart Fail. Rev. 2012, 17, 693–706. [Google Scholar] [CrossRef] [PubMed]
- Gaffney, J.; Solomonov, I.; Zehorai, E.; Sagi, I. Multilevel regulation of matrix metalloproteinases in tissue homeostasis indicates their molecular specificity in vivo. Matrix Biol. 2015, 44, 191–199. [Google Scholar] [CrossRef]
- Zhang, Y.; Shao, L.; Ma, A.; Guan, G.; Wang, J.; Wang, Y.; Tian, G. Telmisartan delays myocardial fibrosis in rats with hypertensive left ventricular hypertrophy by TGF-beta1/Smad signal pathway. Hypertens. Res. 2014, 37, 43–49. [Google Scholar] [CrossRef]
- Lu, L.; Zhang, Q.; Pu, L.J.; Peng, W.H.; Yan, X.X.; Wang, L.J.; Chen, Q.J.; Zhu, Z.B.; Michel, J.B.; Shen, W.F. Dysregulation of matrix metalloproteinases and their tissue inhibitors is related to abnormality of left ventricular geometry and function in streptozotocin-induced diabetic minipigs. Int. J. Exp. Pathol. 2008, 89, 125–137. [Google Scholar] [CrossRef]
- Sakamuri, S.V.P.; Watts, R.; Takawale, A.; Wang, X.; Hernandez-Anzaldo, S.; Bahitham, W.; Fernandez-Patron, C.; Lehner, R.; Kassiri, Z. Absence of tissue inhibitor of metalloproteinase-4 (TIMP4) ameliorates high fat diet-induced obesity in mice due to defective lipid absorption. Sci. Rep. 2017, 7, 6210. [Google Scholar] [CrossRef]
- Chaturvedi, P.; Tyagi, S.C. Epigenetic silencing of TIMP4 in heart failure. J. Cell. Mol. Med. 2016, 20, 2089–2101. [Google Scholar] [CrossRef]
- Ahmed, S.H.; Clark, L.L.; Pennington, W.R.; Webb, C.S.; Bonnema, D.D.; Leonardi, A.H.; McClure, C.D.; Spinale, F.G.; Zile, M.R. Matrix metalloproteinases/tissue inhibitors of metalloproteinases: Relationship between changes in proteolytic determinants of matrix composition and structural, functional, and clinical manifestations of hypertensive heart disease. Circulation 2006, 113, 2089–2096. [Google Scholar] [CrossRef] [PubMed]
- Uijl, A.; Koudstaal, S.; Vaartjes, I.; Boer, J.M.; Verschuren, W.M.; Van Der Schouw, Y.T.; Asselbergs, F.W.; Hoes, A.W.; Sluijs, I. Risk for heart failure. JACC Heart Fail. 2019, 7, 637–647. [Google Scholar] [CrossRef] [PubMed]
- Davison, B.A.; Takagi, K.; Senger, S.; Koch, G.; Metra, M.; Kimmoun, A.; Mebazaa, A.; Voors, A.A.; Nielsen, O.W.; Chioncel, O.; et al. Mega-trials in heart failure: Effects of dilution in examination of new therapies. Eur. J. Heart Fail. 2020. [Google Scholar] [CrossRef]
- Papazova, D.A.; Oosterhuis, N.R.; Gremmels, H.; van Koppen, A.; Joles, J.A.; Verhaar, M.C. Cell-based therapies for experimental chronic kidney disease: A systematic review and meta-analysis. Dis. Model. Mech. 2015, 8, 281–293. [Google Scholar] [CrossRef] [PubMed]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krebber, M.M.; van Dijk, C.G.M.; Vernooij, R.W.M.; Brandt, M.M.; Emter, C.A.; Rau, C.D.; Fledderus, J.O.; Duncker, D.J.; Verhaar, M.C.; Cheng, C.; et al. Matrix Metalloproteinases and Tissue Inhibitors of Metalloproteinases in Extracellular Matrix Remodeling during Left Ventricular Diastolic Dysfunction and Heart Failure with Preserved Ejection Fraction: A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2020, 21, 6742. https://doi.org/10.3390/ijms21186742
Krebber MM, van Dijk CGM, Vernooij RWM, Brandt MM, Emter CA, Rau CD, Fledderus JO, Duncker DJ, Verhaar MC, Cheng C, et al. Matrix Metalloproteinases and Tissue Inhibitors of Metalloproteinases in Extracellular Matrix Remodeling during Left Ventricular Diastolic Dysfunction and Heart Failure with Preserved Ejection Fraction: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences. 2020; 21(18):6742. https://doi.org/10.3390/ijms21186742
Chicago/Turabian StyleKrebber, Merle M., Christian G. M. van Dijk, Robin W. M. Vernooij, Maarten M. Brandt, Craig A. Emter, Christoph D. Rau, Joost O. Fledderus, Dirk J. Duncker, Marianne C. Verhaar, Caroline Cheng, and et al. 2020. "Matrix Metalloproteinases and Tissue Inhibitors of Metalloproteinases in Extracellular Matrix Remodeling during Left Ventricular Diastolic Dysfunction and Heart Failure with Preserved Ejection Fraction: A Systematic Review and Meta-Analysis" International Journal of Molecular Sciences 21, no. 18: 6742. https://doi.org/10.3390/ijms21186742
APA StyleKrebber, M. M., van Dijk, C. G. M., Vernooij, R. W. M., Brandt, M. M., Emter, C. A., Rau, C. D., Fledderus, J. O., Duncker, D. J., Verhaar, M. C., Cheng, C., & Joles, J. A. (2020). Matrix Metalloproteinases and Tissue Inhibitors of Metalloproteinases in Extracellular Matrix Remodeling during Left Ventricular Diastolic Dysfunction and Heart Failure with Preserved Ejection Fraction: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences, 21(18), 6742. https://doi.org/10.3390/ijms21186742






