Relevance of Porcine Stroke Models to Bridge the Gap from Pre-Clinical Findings to Clinical Implementation
Abstract
1. Introduction
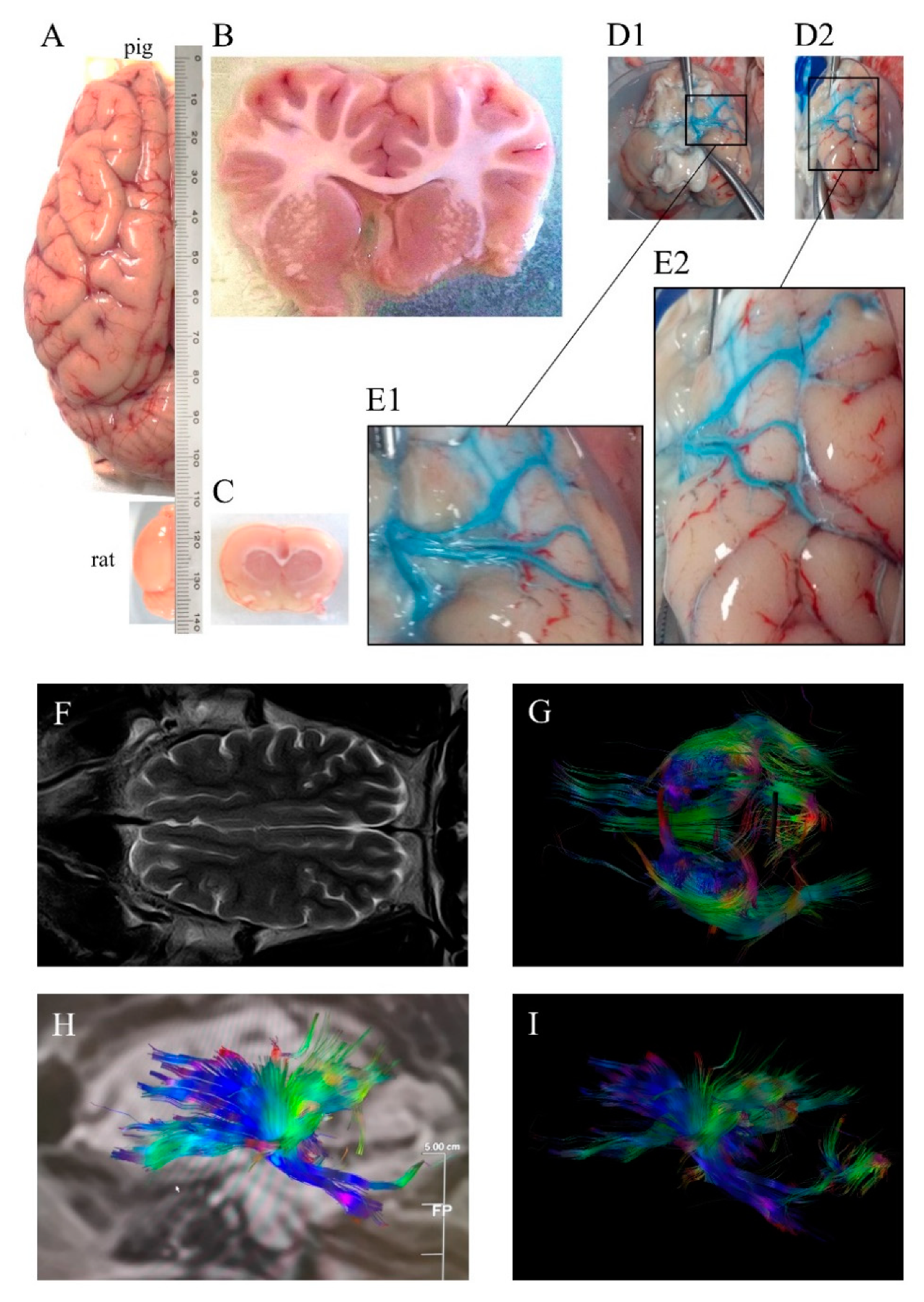
2. Pig Brain to Model Human Stroke Pathophysiology
3. Importance of White Matter Injury in Stroke
4. Interventions to Induce Ischemic Strokes in Pigs
4.1. Electrocoagulation
4.2. Microvascular Clip
4.3. Endovascular Embolization
4.4. Photothrombosis
4.5. Endothelin-1 Injection
5. Interventions to Induce Hemorrhagic Strokes in Pigs
5.1. Autologous Blood Injection in Meningeal Spaces
5.2. Intracerebral Autologous Blood Injection
5.3. Intracerebral Collagenase Injection
5.4. Sonographic Blood-Brain Barrier Disruption
6. Neurological Function Assessment in Pigs
7. Evaluation of Stroke Damage in Swine by Neuroimaging
8. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| GM | Gray matter |
| WM | White matter |
| NHP | Non-human primate |
| ICP | Intracranial pressure |
| DTI | Diffusion tensor imaging |
| CMCiB | Comparative Medicine and Bioimage Centre of Catalonia |
| ICH | Intracranial hemorrhage |
| OL | Oligodendrocyte |
| OPC | Oligodendrocyte precursor cell |
| STAIR | Stroke Treatment Academic Industry Roundtable |
| MCA | Middle cerebral artery |
| MCAO | Middle cerebral artery occlusion |
| ICA | Internal carotid artery |
| ET-1 | Endothelin-1 |
| AChA | Anterior choroidal artery |
| APA | Ascending pharyngeal artery |
| CCA | Common carotid artery |
| DMSO | Dimethyl sulfoxide |
| CT | Computer tomography |
| CSD | Cortical spreading depolarizations |
| SAH | Subarachnoid hemorrhage |
| MRgFUS | Magnetic resonance-guided focused ultrasound |
| MRI | Magnetic resonance imaging |
| MR | Magnetic resonance |
| PET | Positron emission tomography |
| CTP | Computed tomography perfusion |
| ADC | Apparent diffusion coefficient |
| DWI | Diffusion-weighted imaging |
| T2WI | T2-weighted imaging |
| FLAIR | Fluid attenuation inversion recovery |
| T1WI | T1-weighted imaging |
| QSM | Quantitative susceptibility mapping |
| FA | Fractional anisotropy |
| GRE | Gradient echo |
| DCE | Dynamic contrast enhanced |
| MTI | Magnetization transfer imaging |
| tPA | Tissue plasminogen activator |
References
- Sorby-adams, A.J.; Vink, R.; Turner, R.J. Large animal models of stroke and traumatic brain injury as translational tools. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2019. [Google Scholar] [CrossRef]
- Gieling, E.T.; Schuurman, T.; Nordquist, R.E.; Van der Staay, F.J. The pig as a model animal for studying cognition and neurobehavioral disorders. In Molecular and Functional Models in Neuropsychiatry; Springer Science & Business Media: Berlin, Germany, 2011; Volume 7, pp. 359–383. ISBN 9783642287190. [Google Scholar]
- Schomberg, D.T.; Tellez, A.; Meudt, J.J.; Brady, D.A.; Dillon, K.N.; Arowolo, F.K.; Wicks, J.; Rousselle, S.D.; Shanmuganayagam, D. Miniature swine for preclinical modeling of complexities of human disease for translational scientific discovery and accelerated development of therapies and medical devices. Toxicol. Pathol. 2016, 44, 299–314. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.; MacLean, J.; Hatsopoulos, N.G. The marmoset as a model system for studying voluntary motor control. Dev. Neurobiol. 2017, 77, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Lind, N.M.; Moustgaard, A.; Jelsing, J.; Vajta, G.; Cumming, P.; Hansen, A.K. The use of pigs in neuroscience: Modeling brain disorders. Neurosci. Biobehav. Rev. 2007, 31, 728–751. [Google Scholar] [CrossRef] [PubMed]
- Sauleau, P.; Lapouble, E.; Val-Laillet, D.; Malbert, C.H. The pig model in brain imaging and neurosurgery. Animal 2009, 3, 1138–1151. [Google Scholar] [CrossRef]
- Kentar, M.; Mann, M.; Sahm, F.; Olivares-Rivera, A.; Sanchez-Porras, R.; Zerelles, R.; Sakowitz, O.W.; Unterberg, A.W.; Santos, E. Detection of spreading depolarizations in a middle cerebral artery occlusion model in swine. Acta Neurochir. (Wien) 2020, 162, 581–592. [Google Scholar] [CrossRef] [PubMed]
- Craner, S.L.; Ray, R.H. Somatosensory cortex of the neonatal pig: I. Topographic organization of the primary somatosensory cortex (SI). J. Comp. Neurol. 1991, 306, 24–38. [Google Scholar] [CrossRef]
- Roth, G.; Dicke, U. Evolution of the brain and intelligence. Trends Cogn. Sci. 2005, 9, 250–257. [Google Scholar] [CrossRef]
- Jelsing, J.; Hay-Schmidt, A.; Dyrby, T.; Hemmingsen, R.; Uylings, H.B.M.; Pakkenberg, B. The prefrontal cortex in the Göttingen minipig brain defined by neural projection criteria and cytoarchitecture. Brain Res. Bull. 2006, 70, 322–336. [Google Scholar] [CrossRef]
- Conrad, M.S.; Dilger, R.N.; Johnson, R.W. Brain growth of the domestic pig (Sus scrofa) from 2 to 24 weeks of age: A longitudinal MRI study. Dev. Neurosci. 2012, 34, 291–298. [Google Scholar] [CrossRef]
- Ryan, M.C.; Sherman, P.; Rowland, L.M.; Wijtenburg, S.A.; Acheson, A.; Fieremans, E.; Veraart, J.; Novikov, D.S.; Hong, L.E.; Sladky, J.; et al. Miniature pig model of human adolescent brain white matter development. J. Neurosci. Methods 2018, 296, 99–108. [Google Scholar] [CrossRef]
- Simchick, G.; Shen, A.; Campbell, B.; Park, H.J.; West, F.D.; Zhao, Q. Pig brains have homologous resting-state networks with human brains. Brain Connect. 2019, 9, 566–579. [Google Scholar] [CrossRef]
- Gabrielian, L.; Willshire, L.W.; Helps, S.C.; Van Den Heuvel, C.; Mathias, J.; Vink, R. Intracranial pressure changes following traumatic brain injury in rats: Lack of significant change in the absence of mass lesions or hypoxia. J. Neurotrauma 2011, 28, 2103–2111. [Google Scholar] [CrossRef] [PubMed]
- Cai, B.; Wang, N. Large animal stroke models vs. Rodent stroke models, pros and cons, and combination? In Brain Edema XVI; Springer: Cham, Switzerland, 2016; Volume 121, pp. 77–81. [Google Scholar]
- Kaiser, E.E.; Waters, E.S.; Fagan, M.M.; Scheulin, K.M.; Platt, S.R.; Jeon, J.H.; Fang, X.; Kinder, H.A.; Shin, S.K.; Duberstein, K.J.; et al. Characterization of tissue and functional deficits in a clinically translational pig model of acute ischemic stroke. Brain Res. 2020, 1736. [Google Scholar] [CrossRef] [PubMed]
- Mendl, M.; Held, S.; Byrne, R.W. Pig cognition. Curr. Biol. 2010, 20, 796–798. [Google Scholar] [CrossRef] [PubMed]
- Kornum, B.R.; Knudsen, G.M. Cognitive testing of pigs (Sus scrofa) in translational biobehavioral research. Neurosci. Biobehav. Rev. 2011, 35, 437–451. [Google Scholar] [CrossRef]
- Swindle, M.M.; Smith, A.C. Comparative anatomy and physiology of the pig. Scand. J. Lab. Anim. Sci. 1998, 25, 11–21. [Google Scholar] [CrossRef]
- Ashwini, C.; Shubha, R.; Jayanthi, K. Comparative anatomy of the circle of Willis in man, cow, sheep, goat, and pig. Neuroanatomy 2008, 7, 54–65. [Google Scholar]
- Zhang, K.; Sejnowski, T.J. A universal scaling law between gray matter and white matter of cerebral cortex. Proc. Natl. Acad. Sci. USA 2000, 97, 5621–5626. [Google Scholar] [CrossRef]
- Baltan, S.; Besancon, E.F.; Mbow, B.; Ye, Z.C.; Hamner, M.A.; Ransom, B.R. White matter vulnerability to ischemic injury increases with age because of enhanced excitotoxicity. J. Neurosci. 2008, 28, 1479–1489. [Google Scholar] [CrossRef]
- Sommer, C.J. Ischemic stroke: Experimental models and reality. Acta Neuropathol. 2017, 133, 245–261. [Google Scholar] [CrossRef] [PubMed]
- Gladstone, D.J.; Black, S.E.; Hakim, A.M. Toward wisdom from failure: Lessons from neuroprotective stroke trials and new therapeutic directions. Stroke 2002, 33, 2123–2136. [Google Scholar] [CrossRef] [PubMed]
- Matute, C.; Ransom, B.R. Roles of white matter in central nervous system pathophysiologies. ASN Neuro 2012, 4, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.; Feuerstein, G.; Howells, D.W.; Hurn, P.D.; Kent, T.A.; Savitz, S.I.; Lo, E.H. Update of the stroke therapy academic industry roundtable preclinical recommendations. Stroke 2009, 40, 2244–2250. [Google Scholar] [CrossRef] [PubMed]
- Hainsworth, A.H.; Allan, S.M.; Boltze, J.; Cunningham, C.; Farris, C.; Head, E.; Ihara, M.; Isaacs, J.D.; Kalaria, R.N.; Lesnik Oberstein, S.A.M.J.; et al. Translational models for vascular cognitive impairment: A review including larger species. BMC Med. 2017, 15. [Google Scholar] [CrossRef]
- Swindle, M.M.; Makin, A.; Herron, A.J.; Clubb, F.J.; Frazier, K.S. Swine as models in biomedical research and toxicology testing. Vet. Pathol. 2012, 49, 344–356. [Google Scholar] [CrossRef]
- Bassols, A.; Costa, C.; Eckersall, P.D.; Osada, J.; Sabrià, J.; Tibau, J. The pig as an animal model for human pathologies: A proteomics perspective. Proteom. Clin. Appl. 2014, 8, 715–731. [Google Scholar] [CrossRef]
- Wernersson, R.; Schierup, M.H.; Jørgensen, F.G.; Gorodkin, J.; Panitz, F.; Stærfeldt, H.H.; Christensen, O.F.; Mailund, T.; Hornshøj, H.; Klein, A.; et al. Pigs in sequence space: A 0.66X coverage pig genome survey based on shotgun sequencing. BMC Genom. 2005, 6, 1–7. [Google Scholar] [CrossRef]
- Jørgensen, F.G.; Hobolth, A.; Hornshøj, H.; Bendixen, C.; Fredholm, M.; Schierup, M.H. Comparative analysis of protein coding sequences from human, mouse and the domesticated pig. BMC Biol. 2005, 3. [Google Scholar] [CrossRef]
- Robert, C.; Kapetanovic, R.; Beraldi, D.; Watson, M.; Archibald, A.L.; Hume, D.A. Identification and annotation of conserved promoters and macrophage-expressed genes in the pig genome. BMC Genom. 2015, 16, 1–17. [Google Scholar] [CrossRef]
- Renner, S.; Blutke, A.; Clauss, S.; Deeg, C.A.; Kemter, E.; Merkus, D.; Wanke, R.; Wolf, E. Porcine models for studying complications and organ crosstalk in diabetes mellitus. Cell Tissue Res. 2020. [Google Scholar] [CrossRef]
- De Leeuw, F.; De Groot, J.; Achten, E.; Oudkerk, M.; Ramos, L.; Heijboer, R.; Hofman, A.; Jolles, J.; Van Gijn, J.; Breteler, M. Prevalence of cerebral white matter lesions in elderly people: A population based magnetic resonance imaging study: The Rotterdam Scan Study. J. Neurol. Neurosurg. Psychiatry 2001, 70, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Das, A.S.; Regenhardt, R.W.; Vernooij, M.W.; Blacker, D.; Charidimou, A.; Viswanathan, A. Asymptomatic cerebral small vessel disease: Insights from population-based studies. J. Stroke 2019, 21, 121–138. [Google Scholar] [CrossRef] [PubMed]
- Wessels, T.; Wessels, C.; Ellsiepen, A.; Reuter, I.; Trittmacher, S.; Stolz, E.; Jauss, M. Contribution of diffusion-weighted imaging in determination of stroke etiology. Am. J. Neuroradiol. 2006, 27, 35–39. [Google Scholar] [PubMed]
- Wang, Y.; Liu, G.; Hong, D.; Chen, F.; Ji, X.; Cao, G. White matter injury in ischemic stroke. Prog. Neurobiol. 2016, 141, 45–60. [Google Scholar] [CrossRef]
- Egorova, N.; Dhollander, T.; Khlif, M.S.; Khan, W.; Werden, E.; Brodtmann, A. Pervasive white matter fiber degeneration in ischemic stroke. Stroke 2020, 1–7. [Google Scholar] [CrossRef]
- Bailey, E.L.; Smith, C.; Sudlow, C.L.M.; Wardlaw, J.M. Pathology of lacunar ischemic stroke in humans—A systematic review. Brain Pathol. 2012, 22, 583–591. [Google Scholar] [CrossRef]
- Wardlaw, J.M.; Valdés Hernández, M.C.; Muñoz-Maniega, S. What are white matter hyperintensities made of? Relevance to vascular cognitive impairment. J. Am. Heart Assoc. 2015, 4, 001140. [Google Scholar] [CrossRef]
- Bamford, J.; Sandercock, P.; Dennis, M.; Warlow, C.; Burn, J. Classification and natural history of clinically identifiable subtypes of cerebral infarction. Lancet 1991, 337, 1521–1526. [Google Scholar] [CrossRef]
- Regenhardt, R.W.; Das, A.S.; Ohtomo, R.; Lo, E.H.; Ayata, C.; Gurol, M.E. Pathophysiology of lacunar stroke: History’s mysteries and modern interpretations. J. Stroke Cerebrovasc. Dis. 2019, 28, 2079–2097. [Google Scholar] [CrossRef]
- Fu, J.H.; Lu, C.Z.; Hong, Z.; Dong, Q.; Luo, Y.; Wong, K.S. Extent of white matter lesions is related to acute subcortical infarcts and predicts further stroke risk in patients with first ever ischaemic stroke. J. Neurol. Neurosurg. Psychiatry 2005, 76, 793–796. [Google Scholar] [CrossRef]
- Schaapsmeerders, P.; Tuladhar, A.M.; Arntz, R.M.; Franssen, S.; Maaijwee, N.A.M.; Rutten-Jacobs, L.C.A.; Schoonderwaldt, H.C.; Dorresteijn, L.D.A.; Van Dijk, E.J.; Kessels, R.P.C.; et al. Remote lower white matter integrity increases the risk of long-term cognitive impairment after ischemic stroke in young adults. Stroke 2016, 47, 2517–2525. [Google Scholar] [CrossRef]
- Lo, E.H.; Dalkara, T.; Moskowitz, M.A. Neurological diseases: Mechanisms, challenges and opportunities in stroke. Nat. Rev. Neurosci. 2003, 4, 399–414. [Google Scholar] [CrossRef]
- Sozmen, E.G.; Hinman, J.D.; Carmichael, S.T. Models that matter: White matter stroke models. Neurotherapeutics 2012, 9, 349–358. [Google Scholar] [CrossRef]
- Ladecola, C. The pathobiology of vascular dementia. Neuron 2013, 80, 844–866. [Google Scholar] [CrossRef]
- Wu, O.; Cloonan, L.; Mocking, S.J.; Bouts, M.J.; Copen, W.A.; Cougo-Pinto, P.; Fitzpatrick, K.; Kanakis, A.; Schaefer, P.W.; Rosand, J.; et al. The role of acute lesion topography in initial ischemic stroke severity and long-term functional outcomes. Stroke 2015, 46, 2438–2444. [Google Scholar] [CrossRef]
- Srikanth, V.; Beare, R.; Blizzard, L.; Phan, T.; Stapleton, J.; Chen, J.; Callisaya, M.; Martin, K.; Reutens, D. Cerebral white matter lesions, gait, and the risk of incident falls: A prospective population-based study. Stroke 2009, 40, 175–180. [Google Scholar] [CrossRef]
- Hedna, V.S.; Jain, S.; Rabbani, O.; Nadeau, S.E. Mechanisms of arm paresis in middle cerebral artery distribution stroke: Pilot study. J. Rehabil. Res. Dev. 2013, 50, 1113–1121. [Google Scholar] [CrossRef]
- Xie, Q.; Gu, Y.; Hua, Y.; Liu, W.; Keep, R.F.; Xi, G. Deferoxamine attenuates white matter injury in a piglet intracerebral hemorrhage model. Stroke 2014, 45, 290–292. [Google Scholar] [CrossRef]
- Tanaka, Y.; Imai, H.; Konno, K.; Miyagishima, T.; Kubota, C.; Puentes, S.; Aoki, T.; Hata, H.; Takata, K.; Yoshimoto, Y.; et al. Experimental model of lacunar infarction in the gyrencephalic brain of the miniature pig: Neurological assessment and histological, immunohistochemical, and physiological evaluation of dynamic corticospinal tract deformation. Stroke 2008, 39, 205–212. [Google Scholar] [CrossRef]
- Baumann, N.; Pham-Dinh, D. Biology of oligodendrocyte and myelin in the mammalian central nervous system. Physiol. Rev. 2001, 81, 871–927. [Google Scholar] [CrossRef]
- Hinman, J.D. The back and forth of axonal injury and repair after stroke. Curr. Opin. Neurol. 2014, 27, 615–623. [Google Scholar] [CrossRef]
- Dawson, M.R.L.; Polito, A.; Levine, J.M.; Reynolds, R. NG2-expressing glial progenitor cells: An abundant and widespread population of cycling cells in the adult rat CNS. Mol. Cell. Neurosci. 2003, 24, 476–488. [Google Scholar] [CrossRef]
- Kang, S.H.; Fukaya, M.; Yang, J.K.; Rothstein, J.D.; Bergles, D.E. NG2+ CNS glial progenitors remain committed to the oligodendrocyte lineage in postnatal life and following neurodegeneration. Neuron 2010, 68, 668–681. [Google Scholar] [CrossRef]
- Baldassarro, V.A.; Marchesini, A.; Giardino, L.; Calzà, L. Differential effects of glucose deprivation on the survival of fetal versus adult neural stem cells-derived oligodendrocyte precursor cells. Glia 2020, 68, 898–917. [Google Scholar] [CrossRef]
- Chapuisat, G.; Dronne, M.A.; Grenier, E.; Hommel, M.; Gilquin, H.; Boissel, J.P. A global phenomenological model of ischemic stroke with stress on spreading depressions. Prog. Biophys. Mol. Biol. 2008, 97, 4–27. [Google Scholar] [CrossRef]
- D’Esterre, C.D.; Aviv, R.I.; Morrison, L.; Fainardi, E.; Lee, T.Y. Acute multi-modal neuroimaging in a porcine model of endothelin-1-induced cerebral ischemia: Defining the acute infarct core. Transl. Stroke Res. 2015, 6, 234–241. [Google Scholar] [CrossRef]
- Pantoni, L.; Garcia, J.H.; Gutierrez, J.A. Cerebral white matter is highly vulnerable to ischemia. Stroke 1996, 27, 1641–1647. [Google Scholar] [CrossRef]
- Bristow, M.S.; Simon, J.E.; Brown, R.A.; Eliasziw, M.; Hill, M.D.; Coutts, S.B.; Frayne, R.; Demchuk, A.M.; Mitchell, J.R. MR perfusion and diffusion in acute ischemic stroke: Human gray and white matter have different thresholds for infarction. J. Cereb. Blood Flow Metab. 2005, 25, 1280–1287. [Google Scholar] [CrossRef]
- Arakawa, S.; Wright, P.M.; Koga, M.; Phan, T.G.; Reutens, D.C.; Lim, I.; Gunawan, M.R.; Ma, H.; Perera, N.; Ly, J.; et al. Ischemic thresholds for gray and white matter: A diffusion and perfusion magnetic resonance study. Stroke 2006, 37, 1211–1216. [Google Scholar] [CrossRef]
- Mcdonald, J.W.; Althomsons, S.P.; Hyrc, K.L.; Choi, D.W.; Goldberg, M.P. Oligodendrocytes from forebrain are highly vulnerable to AMPA/kainate receptor-mediated excitotoxicity. Nat. Med. 1998, 4, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Salter, M.G.; Fern, R. NMDA receptors are expressed in developing oligodendrocyte processes and mediate injury. Nature 2005, 438, 1167–1171. [Google Scholar] [CrossRef] [PubMed]
- Micu, I.; Jiang, Q.; Coderre, E.; Ridsdale, A.; Zhang, L.; Woulfe, J.; Yin, X.; Trapp, B.D.; McRory, J.E.; Rehak, R.; et al. NMDA receptors mediate calcium accumulation in myelin during chemical ischaemia. Nature 2006, 439, 988–992. [Google Scholar] [CrossRef] [PubMed]
- Domercq, M.; Perez-Samartin, A.; Aparicio, D.; Alberdi, E.; Pampliega, O.; Matute, C. P2X7 receptors mediate ischemic damage to oligodendrocytes. Glia 2010, 58, 730–740. [Google Scholar] [CrossRef]
- Doyle, S.; Hansen, D.B.; Vella, J.; Bond, P.; Harper, G.; Zammit, C.; Valentino, M.; Fern, R. Vesicular glutamate release from central axons contributes to myelin damage. Nat. Commun. 2018, 9. [Google Scholar] [CrossRef]
- Aronowski, J.; Zhao, X. Molecular pathophysiology of cerebral hemorrhage: Secondary brain injury. Stroke 2011, 42, 1781–1786. [Google Scholar] [CrossRef]
- Liu, R.; Cao, S.; Hua, Y.; Keep, R.F.; Huang, Y.; Xi, G. CD163 expression in neurons after experimental intracerebral hemorrhage. Stroke 2017, 48, 1369–1375. [Google Scholar] [CrossRef]
- Shao, Z.; Tu, S.; Shao, A. Pathophysiological mechanisms and potential therapeutic targets in intracerebral hemorrhage. Front. Pharmacol. 2019, 10, 1–8. [Google Scholar] [CrossRef]
- Mandai, K.; Matsumoto, M.; Kitagawa, K.; Matsushita, K.; Ohtsuki, T.; Mabuchi, T.; Colman, D.R.; Kamada, T.; Yanagihara, T. Ischemic damage and subsequent proliferation of oligodendrocytes in focal cerebral ischemia. Neuroscience 1997, 77, 849–861. [Google Scholar] [CrossRef]
- Sozmen, E.G.; Kolekar, A.; Havton, L.A.; Carmichael, S.T. A white matter stroke model in the mouse: Axonal damage, progenitor responses and MRI correlates. J. Neurosci. Methods 2009, 180, 261–272. [Google Scholar] [CrossRef]
- Itoh, K.; Maki, T.; Lok, J.; Arai, K. Mechanisms of cell-cell interaction in oligodendrogenesis and remyelination after stroke. Brain Res. 2015, 1623, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Nait-Oumesmar, B.; Picard-Riéra, N.; Kerninon, C.; Baron-Van Evercooren, A. The role of SVZ-derived neural precursors in demyelinating diseases: From animal models to multiple sclerosis. J. Neurol. Sci. 2008, 265, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Jablonska, B. Chordin-induced lineage plasticity of adult SVZ neuroblasts after demyelination. Nat. Neurosci 2010, 13, 541–550. [Google Scholar] [CrossRef]
- Gensert, J.A.M.; Goldman, J.E. Endogenous progenitors remyelinate demyelinated axons in the adult CNS. Neuron 1997, 19, 197–203. [Google Scholar] [CrossRef]
- Sozmen, E.G.; Rosenzweig, S.; Llorente, I.L.; DiTullio, D.J.; Machnicki, M.; Vinters, H.V.; Havton, L.A.; Giger, R.J.; Hinman, J.D.; Carmichael, S.T. Nogo receptor blockade overcomes remyelination failure after white matter stroke and stimulates functional recovery in aged mice. Proc. Natl. Acad. Sci. USA 2016, 113, E8453–E8462. [Google Scholar] [CrossRef]
- Carmichael, S.T.; Kathirvelu, B.; Schweppe, C.A.; Nie, E.H. Molecular, cellular and functional events in axonal sprouting after stroke. Exp. Neurol. 2017, 287, 384–394. [Google Scholar] [CrossRef]
- Xu, M.; Wang, M.M.; Gao, Y.; Keep, R.F.; Shi, Y. The effect of age-related risk factors and comorbidities on white matter injury and repair after ischemic stroke. Neurobiol. Dis. 2019, 126, 13–22. [Google Scholar] [CrossRef]
- Modo, M.M.; Jolkkonen, J.; Zille, M.; Boltze, J. The Future of Animal Modeling for Post-Stroke Tissue Repair. Stroke 2018, 49, 1099–1106. [Google Scholar] [CrossRef]
- Savitz, S.I.; Baron, J.C.; Fisher, M. Stroke treatment academic industry roundtable X: Brain cytoprotection therapies in the reperfusion era. Stroke 2019, 50, 1026–1031. [Google Scholar] [CrossRef]
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. Heart disease and stroke statistics-2019 update: A report from the American Heart Association. Circulation 2019, 139, e56–e528. [Google Scholar] [CrossRef]
- Boodt, N.; Compagne, K.C.J.; Dutra, B.G.; Samuels, N.; Tolhuisen, M.L.; Alves, H.C.B.R.; Kappelhof, M.; Lycklama à Nijeholt, G.J.; Marquering, H.A.; Majoie, C.B.L.M.; et al. Stroke etiology and thrombus computed tomography characteristics in patients with acute ischemic stroke. Stroke 2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, P.; Tang, Z. Animal models of transient ischemic attack: A review. Acta Neurol. Belg. 2020, 120, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Burbridge, B.; Matte, G.; Remedios, A. Complex intracranial arterial anatomy in swine is unsuitable for cerebral infarction projects. Can. Assoc. Radiol. J. 2004, 55, 326–329. [Google Scholar] [PubMed]
- Arikan, F.; Martínez-Valverde, T.; Sánchez-Guerrero, Á.; Campos, M.; Esteves, M.; Gandara, D.; Torné, R.; Castro, L.; Dalmau, A.; Tibau, J.; et al. Malignant infarction of the middle cerebral artery in a porcine model. A pilot study. PLoS ONE 2017, 12, e0172637. [Google Scholar] [CrossRef]
- Zhang, R.; Bertelsen, L.B.; Flø, C.; Wang, Y.; Stødkilde-Jørgensen, H. Establishment and characterization of porcine focal cerebral ischemic model induced by endothelin-1. Neurosci. Lett. 2016, 635, 1–7. [Google Scholar] [CrossRef]
- Kuluz, J.W.; Prado, R.; He, D.; Zhao, W.; Dietrich, W.D.; Watson, B. New pediatric model of ischemic stroke in infant piglets by photothrombosis: Acute changes in cerebral blood flow, microvasculature, and early histopathology. Stroke 2007, 38, 1932–1937. [Google Scholar] [CrossRef]
- Zhang, L.; Cheng, H.; Shi, J.; Chen, J. Focal epidural cooling reduces the infarction volume of permanent middle cerebral artery occlusion in swine. Surg. Neurol. 2007, 67, 117–121. [Google Scholar] [CrossRef]
- Odden, J.P.; Stiris, T.; Hansen, T.W.R.; Bratlid, D. Cerebral blood flow during experimental hypoxaemia and ischaemia in the newborn piglet. Acta Paediatr. Scand. Suppl. 1989, 78, 13–19. [Google Scholar] [CrossRef]
- Laptook, A.R.; Corbett, R.J.T.; Nunnally, R.L. Effect of Plasma Glucose Concentration on Cerebral Metabolism During Partial Ischemia in Neonatal Piglets. Stroke 1990, 21, 435–440. [Google Scholar] [CrossRef]
- Fisk, G.C.; Wright, J.S.; Turner, B.B.; Baker, W.C.; Hicks, R.G.; Lethlean, A.K.; Stacey, R.B.; Lawrence, J.C.; Lawrie, G.M.; Kalnins, I.; et al. Cerebral effects of circulatory arrest at 20°C in the infant pig. Anaesth. Intensive Care 1974, 2, 33–42. [Google Scholar] [CrossRef]
- Maningas, P.A.; DeGuzman, L.R.; Hollenbach, S.J.; Volk, K.A.; Bellamy, R.F. Regional blood flow during hypothermic arrest. Ann. Emerg. Med. 1986, 15, 390–396. [Google Scholar] [CrossRef]
- Armstead, W.M.; Mirro, R.; Busija, D.W.; Leffler, C.W. Postischemic generation of superoxide anion by newborn pig brain. Am. J. Physiol. Hear. Circ. Physiol. 1988, 255, 401–403. [Google Scholar] [CrossRef] [PubMed]
- Fern, R.; Möller, T. Rapid ischemic cell death in immature oligodendrocytes: A fatal glutamate release feedback loop. J. Neurosci. 2000, 20, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Armstead, W.M.; Riley, J.; Kiessling, J.W.; Cines, D.B.; Higazi, A.A.R. Novel plasminogen activator inhibitor-1-derived peptide protects against impairment of cerebrovasodilation after photothrombosis through inhibition of JNK MAPK. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 299, 480–485. [Google Scholar] [CrossRef]
- Armstead, W.M.; Ganguly, K.; Riley, J.; Kiessling, W.J.; Cines, D.B.; Higazi, A.A.R.; Zaitsev, S.; Muzykantov, V.R. RBC-coupled tissue plasminogen activator prevents impairment of cerebral vasodilatory responses through inhibition of JNK and potentiation of p38 MAPK after cerebral photothrombosis in the newborn pig. Pediatr. Crit. Care Med. 2011, 12, 369–375. [Google Scholar] [CrossRef]
- Armstead, W.M.; Ganguly, K.; Riley, J.; Zaitsev, S.; Cines, D.B.; Higazi, A.A.R.; Muzykantov, V.R. RBC-coupled tPA prevents whereas tPA aggravates JNK MAPK-mediated impairment of ATP- and Ca-sensitive K channel-mediated cerebrovasodilation after cerebral photothrombosis. Transl. Stroke Res. 2012, 3, 114–121. [Google Scholar] [CrossRef]
- Armstead, W.M.; Riley, J.; Yarovoi, S.; Higazi, A.A.; Cines, D.B. tPA-A296-299 prevents impairment of cerebral autoregulation after stroke through LRP dependent increase in cAMP and p38. Stroke 2016, 47, 2096–2102. [Google Scholar] [CrossRef]
- Armstead, W.M.; Hekierski, H.; Pastor, P.; Yarovoi, S.; Higazi, A.A.R.; Cines, D.B. Release of IL-6 after stroke contributes to impaired cerebral autoregulation and hippocampal neuronal necrosis through NMDA receptor activation and upregulation of ET-1 and JNK. Transl. Stroke Res. 2019, 10, 104–111. [Google Scholar] [CrossRef]
- Duberstein, K.J.; Platt, S.R.; Holmes, S.P.; Dove, C.R.; Howerth, E.W.; Kent, M.; Stice, S.L.; Hill, W.D.; Hess, D.C.; West, F.D. Gait analysis in a pre- and post-ischemic stroke biomedical pig model. Physiol. Behav. 2014, 125, 8–16. [Google Scholar] [CrossRef]
- Platt, S.R.; Holmes, S.P.; Howerth, E.W.; Duberstein, K.J.J.; Dove, C.R.; Kinder, H.A.; Wyatt, E.L.; Linville, A.V.; Lau, V.W.; Stice, S.L.; et al. Development and characterization of a Yucatan miniature biomedical pig permanent middle cerebral artery occlusion stroke model. Exp. Transl. Stroke Med. 2014, 6, 1–14. [Google Scholar] [CrossRef]
- Baker, E.W.; Platt, S.R.; Lau, V.W.; Grace, H.E.; Holmes, S.P.; Wang, L.; Duberstein, K.J.; Howerth, E.W.; Kinder, H.A.; Stice, S.L.; et al. Induced pluripotent stem cell-derived neural stem cell therapy enhances recovery in an ischemic stroke pig model. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Webb, R.L.; Kaiser, E.E.; Jurgielewicz, B.J.; Spellicy, S.; Scoville, S.L.; Thompson, T.A.; Swetenburg, R.L.; Hess, D.C.; West, F.D.; Stice, S.L. Human neural stem cell extracellular vesicles improve recovery in a porcine model of ischemic stroke. Stroke 2018, 49, 1248–1256. [Google Scholar] [CrossRef] [PubMed]
- Lau, V.W.; Platt, S.R.; Grace, H.E.; Baker, E.W.; West, F.D. Human iNPC therapy leads to improvement in functional neurologic outcomes in a pig ischemic stroke model. Brain Behav. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Spellicy, S.E.; Kaiser, E.E.; Bowler, M.M.; Jurgielewicz, B.J.; Webb, R.L.; West, F.D.; Stice, S.L. Neural stem cell extracellular vesicles disrupt midline shift predictive outcomes in porcine ischemic stroke model. Transl. Stroke Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Imai, H.; Konno, K.; Kubota, C.; Seki, K.; Puentes, S.; Faried, A.; Yokoo, H.; Hata, H.; Yoshimoto, Y.; et al. Experimental investigation of encephalomyosynangiosis using gyrencephalic brain of the miniature pig: Histopathological evaluation of dynamic reconstruction of vessels for functional anastomosis—Laboratory investigation. J. Neurosurg. Pediatr. 2009, 3, 488–495. [Google Scholar] [CrossRef]
- Mattingly, T.K.; Denning, L.M.; Siroen, K.L.; Lehrbass, B.; Lopez-Ojeda, P.; Stitt, L.; Pelz, D.M.; Das, S.; Ang, L.C.; Lee, D.H.; et al. Catheter based selective hypothermia reduces stroke volume during focal cerebral ischemia in swine. J. Neurointerv. Surg. 2016, 8, 418–422. [Google Scholar] [CrossRef]
- Elliott, J.T.; Diop, M.; Morrison, L.B.; D’Esterre, C.D.; Lee, T.Y.; St. Lawrence, K.S. Quantifying cerebral blood flow in an adult pig ischemia model by a depth-resolved dynamic contrast-enhanced optical method. Neuroimage 2014, 94, 303–311. [Google Scholar] [CrossRef]
- Wright, E.A.; D’Esterre, C.D.; Morrison, L.B.; Cockburn, N.; Kovacs, M.; Lee, T.Y. Absolute cerebral blood flow infarction threshold for 3-hour ischemia time determined with CT perfusion and 18F-FFMZ-PET imaging in a porcine model of cerebral ischemia. PLoS ONE 2016, 11, e0158157. [Google Scholar] [CrossRef]
- Sakoh, M.; Røhl, L.; Gyldensted, C.; Gjedde, A.; Østergaard, L. Cerebral blood flow and blood volume measured by magnetic resonance imaging bolus tracking after acute stroke in pigs: Comparison with [15O]H2O positron emission tomography. Stroke 2000, 31, 1958–1964. [Google Scholar] [CrossRef]
- Sakoh, M.; Østergaard, L.; Røhl, L.; Smith, D.F.; Simonsen, C.Z.; Sørensen, J.C.; Poulsen, P.V.; Gyldensted, C.; Sakaki, S.; Gjedde, A. Relationship between residual cerebral blood flow and oxygen metabolism as predictive of ischemic tissue viability: Sequential multitracer positron emission tomography scanning of middle cerebral artery occlusion during the critical first 6 hours after st. J. Neurosurg. 2000, 93, 647–657. [Google Scholar] [CrossRef]
- Sakoh, M.; Østergaard, L.; Gjedde, A.; Røhl, L.; Vestergaard-Poulsen, P.; Smith, D.F.; Le Bihan, D.; Sakaki, S.; Gyldensted, C. Prediction of tissue survival after middle cerebral artery occlusion based on changes in the apparent diffusion of water. J. Neurosurg. 2001, 95, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Røhl, L.; Sakoh, M.; Simonsen, C.Z.; Vestergaard-Poulsen, P.; Sangill, R.; Sørensen, J.C.; Bjarkam, C.R.; Gyldensted, C.; Østergaard, L. Time evolution of cerebral perfusion and apparent diffusion coefficient measured by magnetic resonance imaging in a porcine stroke model. J. Magn. Reson. Imaging 2002, 15, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Sakoh, M.; Andersen, F.; Rodell, A.; Sørensen, J.C.; Østergaard, L.; Mouridsen, K.; Cumming, P. Statistical mapping of effects of middle cerebral artery occlusion (MCAO) on blood flow and oxygen consumption in porcine brain. J. Neurosci. Methods 2007, 160, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Schöll, M.J.; Santos, E.; Sanchez-Porras, R.; Kentar, M.; Gramer, M.; Silos, H.; Zheng, Z.; Gang, Y.; Strong, A.J.; Graf, R.; et al. Large field-of-view movement-compensated intrinsic optical signal imaging for the characterization of the haemodynamic response to spreading depolarizations in large gyrencephalic brains. J. Cereb. Blood Flow Metab. 2017, 37, 1706–1719. [Google Scholar] [CrossRef] [PubMed]
- Mangla, S.; Choi, J.H.; Barone, F.C.; Novotney, C.; Libien, J.; Lin, E.; Pile-Spellman, J. Endovascular external carotid artery occlusion for brain selective targeting: A cerebrovascular swine model. BMC Res. Notes 2015, 8, 4–9. [Google Scholar] [CrossRef]
- Yang, J.K.; Cote, A.M.; Jordan, C.D.; Kondapavulur, S.; Losey, A.D.; McCoy, D.; Chu, A.; Yu, J.F.; Moore, T.; Stillson, C.; et al. Interventional magnetic resonance imaging guided carotid embolectomy using a novel resonant marker catheter: Demonstration of preclinical feasibility. Biomed. Microdevices 2017, 19. [Google Scholar] [CrossRef]
- Srinivasan, V.M.; Chen, S.R.; Camstra, K.M.; Chintalapani, G.; Kan, P. Development of a recalcitrant, large clot burden, bifurcation occlusion model for mechanical thrombectomy. Neurosurg. Focus 2017, 42, 1–6. [Google Scholar] [CrossRef]
- Brekenfeld, C.; Schroth, G.; El-Koussy, M.; Nedeltchev, K.; Reinert, M.; Slotboom, J.; Gralla, J. Mechanical thromboembolectomy for acute ischemic stroke: Comparison of the catch thromboectomy device and the Merci Retriever in vivo. Stroke 2008, 39, 1213–1219. [Google Scholar] [CrossRef]
- Ringer, A.J.; Guterman, L.R.; Hopkins, L.N. Site-specific thromboembolism: A novel animal model for stroke. Am. J. Neuroradiol. 2004, 25, 329–332. [Google Scholar]
- Culp, W.C.; Erdem, E.; Roberson, P.K.; Husain, M.M. Microbubble potentiated ultrasound as a method of stroke therapy in a pig model: Preliminary findings. J. Vasc. Interv. Radiol. 2003, 14, 1433–1436. [Google Scholar] [CrossRef][Green Version]
- Gao, S.; Zhang, Y.; Wu, J.; Shi, W.T.; Lof, J.; Vignon, F.; Drvol, L.; Xie, F.; Muirhead, D.; Powers, J.E.; et al. Improvements in cerebral blood flow and recanalization rates with transcranial diagnostic ultrasound and intravenous microbubbles after acute cerebral Emboli. Investig. Radiol. 2014, 49, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Peschillo, S.; Diana, F.; Berge, J.; Missori, P. A comparison of acute vascular damage caused by ADAPT versus a stent retriever device after thrombectomy in acute ischemic stroke: A histological and ultrastructural study in an animal model. J. Neurointerv. Surg. 2017, 9, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Gory, B.; Bresson, D.; Kessler, I.; Perrin, M.L.; Guillaudeau, A.; Durand, K.; Ponsonnard, S.; Couquet, C.; Yardin, C.; Mounayer, C. Histopathologic evaluation of arterial wall response to 5 neurovascular mechanical thrombectomy devices in a swine model. Am. J. Neuroradiol. 2013, 34, 2192–2198. [Google Scholar] [CrossRef] [PubMed]
- Mordasini, P.; Frabetti, N.; Gralla, J.; Schroth, G.; Fischer, U.; Arnold, M.; Brekenfeld, C. In vivo evaluation of the first dedicated combined flow-restoration and mechanical thrombectomy device in a swine model of acute vessel occlusion. Am. J. Neuroradiol. 2011, 32, 294–300. [Google Scholar] [CrossRef]
- Mordasini, P.; Brekenfeld, C.; Byrne, J.V.; Fischer, U.; Arnold, M.; Jung, S.; Schroth, G.; Gralla, J. Experimental evaluation of immediate recanalization effect and recanalization efficacy of a new thrombus retriever for acute stroke treatment in vivo. Am. J. Neuroradiol. 2013, 34, 153–158. [Google Scholar] [CrossRef]
- Roth, C.; Junk, D.; Papanagiotou, P.; Keuler, A.; Körner, H.; Schumacher, M.; Reith, W. A comparison of 2 stroke devices: The new aperio clot-removal device and the solitaire AB/FR. Am. J. Neuroradiol. 2012, 33, 1317–1320. [Google Scholar] [CrossRef]
- Gralla, J.; Schroth, G.; Remonda, L.; Fleischmann, A.; Fandino, J.; Slotboom, J.; Brekenfeld, C. A dedicated animal model for mechanical thrombectomy in acute stroke. Am. J. Neuroradiol. 2006, 27, 1357–1361. [Google Scholar]
- Gralla, J.; Schroth, G.; Remonda, L.; Nedeltchev, K.; Slotboom, J.; Brekenfeld, C. Mechanical thrombectomy for acute ischemic stroke: Thrombus-device interaction, efficiency, and complications in vivo. Stroke 2006, 37, 3019–3024. [Google Scholar] [CrossRef]
- Gralla, J.; Burkhardt, M.; Schroth, G.; El-Koussy, M.; Reinert, M.; Nedeltchev, K.; Slotboom, J.; Brekenfeld, C. Occlusion length is a crucial determinant of efficiency and complication rate in thrombectomy for acute ischemic stroke. Am. J. Neuroradiol. 2008, 29, 247–252. [Google Scholar] [CrossRef]
- Turjman, F.; Massoud, T.F.; Vinters, H.V.; Ji, C.; Tardy, M.; Guglielmi, G.; Vinuela, F. Collagen microbeads: Experimental evaluation of an embolic agent in the rete mirabile of the swine. Am. J. Neuroradiol. 1995, 16, 1031–1036. [Google Scholar]
- Arakawa, H.; Murayama, Y.; Davis, C.R.; Howard, D.L.; Baumgardner, W.L.; Marks, M.P.; Do, H.M. Endovascular embolization of the swine rete mirabile with Eudragit-E 100 polymer. Am. J. Neuroradiol. 2007, 28, 1191–1196. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.C.; Tian, Y.; Tang, Y.; Jia, L.J.; Wu, A.L.; Peng, P.; Yang, J.Z.; Du, H.; Wang, X.J.; Wu, L.K. Application of sodium alginate microspheres in ischemic stroke modeling in miniature pigs. Neural Regen. Res. 2013, 8, 1473–1480. [Google Scholar] [CrossRef] [PubMed]
- Kozlov, D.B.; Lang, E.V.; Barnhart, W.; Gossler, A.; De Girolami, U. Adverse cerebrovascular effects of intraarterial CO2 injections: Development of an in vitro/in vivo model for assessment of gas-based toxicity. J. Vasc. Interv. Radiol. 2005, 16, 713–726. [Google Scholar] [CrossRef] [PubMed]
- Medby, C.; Rø, H.; Koteng, S.; Juul, R.; Krossnes, B.K.; Brubakk, A.O. Microdialysis in cisterna magna during cerebral air embolism in swine. Undersea Hyperb. Med. 2002, 29, 226–234. [Google Scholar]
- Chaloupka, J.C.; Huddle, D.C.; Alderman, J.; Fink, S.; Hammond, R.; Vinters, H.V. A reexamination of the angiotoxicity of superselective injection of DMSO in the swine rete embolization model. Am. J. Neuroradiol. 1999, 20, 401–410. [Google Scholar]
- Sheski, F.D.; Mathur, P.N. Cryotherapy, electrocautery, and brachytherapy. Clin. Chest Med. 1999, 20, 123–138. [Google Scholar] [CrossRef]
- Imai, H.; Konno, K.; Nakamura, M.; Shimizu, T.; Kubota, C.; Seki, K.; Honda, F.; Tomizawa, S.; Tanaka, Y.; Hata, H.; et al. A new model of focal cerebral ischemia in the miniature pig. J. Neurosurg. 2006, 104, 123–132. [Google Scholar] [CrossRef]
- Crowell, R.M.; Marcoux, F.W.; Degirolami, U. Variability and reversibility of focal cerebral ischemia in unanesthetized monkeys. Neurology 1981, 31, 1295–1302. [Google Scholar] [CrossRef]
- Hudgins, W.R.; Garcia, J.H. The effect of electrocautery, atmospheric exposure, and surgical retraction on the permeability of the blood-brain-barrier. Stroke 1970, 1, 375–380. [Google Scholar] [CrossRef]
- Hu, J.; Albadawi, H.; Chong, B.W.; Deipolyi, A.R.; Sheth, R.A.; Khademhosseini, A.; Oklu, R. Advances in biomaterials and technologies for vascular embolization. Adv. Mater. 2019, 31, 1–52. [Google Scholar] [CrossRef]
- Armstead, W.M.; Hekierski, H.; Yarovoi, S.; Higazi, A.A.R.; Cines, D.B. tPA variant tPA-A296–299 Prevents impairment of cerebral autoregulation and necrosis of hippocampal neurons after stroke by inhibiting upregulation of ET-1. J. Neurosci. Res. 2018, 96, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Li, Y.; Fu, X.; Li, L.; Hao, X.; Li, S. Nonhuman primate models of focal cerebral ischemia. Neural Regen. Res. 2017, 12, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Macrae, I.M.; Robinson, M.J.; Graham, D.I.; Reid, J.L.; McCulloch, J. Endothelin-1-induced reductions in cerebral blood flow: Dose dependency, time course, and neuropathological consequences. J. Cereb. Blood Flow Metab. 1993, 13, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Yakubu, M.A.; Leffler, C.W. Role of endothelin-1 in cerebral hematoma-induced modification of cerebral vascular reactivity in piglets. Brain Res. 1996, 734, 149–156. [Google Scholar] [CrossRef]
- Hatake, K.; Wakabayashi, I.; Kakishita, E.; Hishida, S. Impairment of endothelium-dependent relaxation in human basilar artery after subarachnoid hemorrhage. Stroke 1992, 23, 1111–1117. [Google Scholar] [CrossRef]
- Virley, D.; Hadingham, S.J.; Roberts, J.C.; Farnfield, B.; Elliott, H.; Whelan, G.; Golder, J.; David, C.; Parsons, A.A.; Hunter, A.J. A new primate model of focal stroke: Endothelin-1-induced middle cerebral artery occlusion and reperfusion in the common marmoset. J. Cereb. Blood Flow Metab. 2004, 24, 24–41. [Google Scholar] [CrossRef]
- Naidoo, V.; Naidoo, S.; Mahabeer, R.; Raidoo, D.M. Cellular distribution of the endothelin system in the human brain. J. Chem. Neuroanat. 2004, 27, 87–98. [Google Scholar] [CrossRef]
- Ziv, I.; Fleminger, G.; Djaldetti, R.; Achiron, A.; Melamed, E.; Sokolovsky, M. Increased plasma endothelin-1 in acute ischemic stroke. Stroke 1992, 23, 1014–1016. [Google Scholar] [CrossRef]
- Stenman, E.; Malmsjö, M.; Uddman, E.; Gidö, G.; Wieloch, T.; Edvinsson, L. Cerebral ischemia upregulates vascular endothelin ETB receptors in rat. Stroke 2002, 33, 2311–2316. [Google Scholar] [CrossRef]
- Marin, M.A.; Carmichael, S.T. Mechanisms of demyelination and remyelination in the young and aged brain following white matter stroke. Neurobiol. Dis. 2019, 126, 5–12. [Google Scholar] [CrossRef]
- Ma, Q.; Khatibi, N.H.; Chen, W.; Tang, J.; Zhang, J.H. History of preclinical models of intracerebral hemorrhage. Acta Neurochir. Suppl. 2011, 111, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Vilela, P.; Wiesmann, M. Nontraumatic intracranial hemorrhage. In Diseases of the Brain, Head and Neck, Spine 2020–2023; Springer: Berlin, Germany, 2020; pp. 45–57. ISBN 9783030384906. [Google Scholar]
- Orlin, J.R.; Osen, K.K.; Hovig, T. Subdural compartment in pig: A morphologic study with blood and horseradish peroxidase infused subdurally. Anat. Rec. 1991, 230, 22–37. [Google Scholar] [CrossRef] [PubMed]
- Durham, S.R.; Duhaime, A.C. Maturation-dependent response of the immature brain to experimental subdural hematoma. J. Neurotrauma 2007, 24, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Hartings, J.A.; York, J.; Carroll, C.P.; Hinzman, J.M.; Mahoney, E.; Krueger, B.; Winkler, M.K.L.; Major, S.; Horst, V.; Jahnke, P.; et al. Subarachnoid blood acutely induces spreading depolarizations and early cortical infarction. Brain 2017, 140, 2673–2690. [Google Scholar] [CrossRef]
- Wagner, K.R.; Xi, G.; Hua, Y.; Kleinholz, M.; De Courten-Myers, G.M.; Myers, R.E.; Broderick, J.P.; Brott, T.G. Lobar intracerebral hemorrhage model in pigs: Rapid edema development in perihematomal white matter. Stroke 1996, 27, 490–497. [Google Scholar] [CrossRef]
- Orakcioglu, B.; Uozumi, Y.; Kentar, M.M.; Santos, E.; Unterberg, A.; Sakowitz, O.W. Evidence of spreading depolarizations in a porcine cortical intracerebral hemorrhage model. In Intracranial Pressure and Brain Monitoring XIV; Springer: Vienna, Austria, 2012; Volume 114, pp. 369–372. ISBN 9783709109564. [Google Scholar]
- Xu, Z.; Carlson, C.; Snell, J.; Eames, M.; Hananel, A.; Lopes, M.B.; Raghavan, P.; Lee, C.; Yen, C.; Schlesinger, D.; et al. Intracranial inertial cavitation threshold and thermal ablation lesion creation using MRI-guided 220-kHz focused ultrasound surgery: Preclinical investigation. J. Neurosurg. 2015, 122, 152–161. [Google Scholar] [CrossRef]
- Mun-Bryce, S.; Wilkerson, A.C.; Papuashvili, N.; Okada, Y.C. Recurring episodes of spreading depression are spontaneously elicited by an intracerebral hemorrhage in the swine. Brain Res. 2001, 888, 248–255. [Google Scholar] [CrossRef]
- Mun-Bryce, S.; Wilkerson, A.; Pacheco, B.; Zhang, T.; Rai, S.; Wang, Y.; Okada, Y. Depressed cortical excitability and elevated matrix metalloproteinases in remote brain regions following intracerebral hemorrhage. Brain Res. 2004, 1026, 227–234. [Google Scholar] [CrossRef]
- Mun-Bryce, S.; Roberts, L.J.M.; Curtis Hunt, W.; Bartolo, A.; Okada, Y. Acute changes in cortical excitability in the cortex contralateral to focal intracerebral hemorrhage in the swine. Brain Res. 2004, 1026, 218–226. [Google Scholar] [CrossRef]
- Mun-Bryce, S.; Roberts, L.; Bartolo, A.; Okada, Y. Transhemispheric depolarizations persist in the intracerebral hemorrhage swine brain following corpus callosal transection. Brain Res. 2006, 1073–1074, 481–490. [Google Scholar] [CrossRef]
- Gerhardson, T.; Sukovich, J.R.; Chaudhary, N.; Chenevert, T.L.; Ives, K.; Hall, T.L.; Camelo-Piragua, S.; Xu, Z.; Pandey, A.S. Histotripsy clot liquefaction in a porcine intracerebral hemorrhage model. Clin. Neurosurg. 2020, 86, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Aviv, R.I.; Huynh, T.; Huang, Y.; Ramsay, D.; Van Slyke, P.; Dumont, D.; Asmah, P.; Alkins, R.; Liu, R.; Hynynen, K. An in vivo, MRI-integrated real-time model of active contrast extravasation in acute intracerebral hemorrhage. Am. J. Neuroradiol. 2014, 35, 1693–1699. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Huynh, T.J.; Huang, Y.; Ramsay, D.; Hynynen, K.; Aviv, R.I. Modeling the pattern of contrast extravasation in acute intracerebral hemorrhage using dynamic contrast-enhanced MR. Neurocrit. Care 2015, 22, 320–324. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Li, F.; Zou, M.; Xue, X.; Yuan, J.; Feng, H.; Lin, J. Experimental high-altitude intracerebral hemorrhage in minipigs: Histology, behavior, and intracranial pressure in a double-injection model. Acta Neurochir. (Wien) 2013, 155, 655–661. [Google Scholar] [CrossRef]
- Zhou, X.; Xie, Q.; Xi, G.; Keep, R.F.; Hua, Y. Brain CD47 expression in a swine model of intracerebral hemorrhage. Brain Res. 2014, 1574, 70–76. [Google Scholar] [CrossRef]
- Zhu, H.T.; Bian, C.; Yuan, J.C.; Liao, X.J.; Liu, W.; Zhu, G.; Feng, H.; Lin, J.K. Hyperbaric oxygen therapy ameliorates acute brain injury after porcine intracerebral hemorrhage at high altitude. Crit. Care 2015, 19, 1–10. [Google Scholar] [CrossRef]
- Cao, S.; Zheng, M.; Hua, Y.; Chen, G.; Keep, R.F.; Xi, G. Hematoma changes during clot resolution after experimental intracerebral hemorrhage. Stroke 2016, 47, 1626–1631. [Google Scholar] [CrossRef]
- Rohde, V.; Rohde, I.; Thiex, R.; Küker, W.; Ince, A.; Gilsbach, J.M. The role of intraoperative magnetic resonance imaging for the detection of hemorrhagic complications during surgery for intracerebral lesions an experimental approach. Surg. Neurol. 2001, 56, 266–274. [Google Scholar] [CrossRef]
- Rohde, V.; Rohde, I.; Thiex, R.; Ince, A.; Jung, A.; Dückers, G.; Gröschel, K.; Röttger, C.; Küker, W.; Müller, H.D.; et al. Fibrinolysis therapy achieved with tissue plasminogen activator and aspiration of the liquefied clot after experimental intracerebral hemorrhage: Rapid reduction in hematoma volume but intensification of delayed edema formation. J. Neurosurg. 2002, 97, 954–962. [Google Scholar] [CrossRef]
- Keric, N.; Kantelhardt, S.R.; Neulen, A.; Dechent, P.; Henning, A.; Vollmer, F.C.; Thiemann, I.; Giese, A. Image-guided intracranial endosonography. J. Neurosurg. Anesthesiol. 2013, 25, 317–323. [Google Scholar] [CrossRef]
- Thiex, R.; Küker, W.; Müller, H.D.; Rohde, I.; Schröder, J.M.; Gilsbach, J.M.; Rohde, V. The long-term effect of recombinant tissue-plasminogen-activator (rt-PA) on edema formation in a large-animal model of intracerebral hemorrhage. Neurol. Res. 2003, 25, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Thiex, R.; Küker, W.; Jungbluth, P.; Kayser, C.; Müller, H.D.; Rohde, I.; Gilsbach, J.M.; Rohde, V. Minor inflammation after surgical evacuation compared with fibrinolytic therapy of experimental intracerebral hemorrhages. Neurol. Res. 2005, 27, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Thiex, R.; Weis, J.; Krings, T.; Barreiro, S.; Yakisikli-Alemi, F.; Gilsbach, J.M.; Rohde, V. Addition of intravenous N-methyl-D-aspartate receptor antagonists to local fibrinolytic therapy for the optimal treatment of experimental intracerebral hemorrhages. J. Neurosci. Methods 2007, 106, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Hua, Y.; Keep, R.F.; Morgenstern, L.B.; Xi, G. Deferoxamine reduces intracerebral hematoma-induced iron accumulation and neuronal death in piglets. Stroke 2009, 40, 2241–2243. [Google Scholar] [CrossRef]
- Shi, Y.H.; Li, Z.W.; Zhang, S.M.; Xie, M.J.; Meng, X.W.; Xu, J.Z.; Liu, N.; Tang, Z.P. Establishing a model of supratentorial hemorrhage in the piglet. Tohoku, J. Exp. Med. 2010, 220, 33–40. [Google Scholar] [CrossRef][Green Version]
- Gu, Y.; Hua, Y.; He, Y.; Wang, L.; Hu, H.; Keep, R.F.; Xi, G. Iron accumulation and DNA damage in a pig model of intracerebral hemorrhage. Acta Neurochir. (Wien) 2011, 111, 123–128. [Google Scholar] [CrossRef]
- Tong, J.; Latzman, J.M.; Rauch, J.; Zagzag, D.S.; Huang, J.H.; Samadani, U. Sonic hedgehog agonist fails to induce neural stem cell precursors in a porcine model of experimental intracranial hemorrhage. In Intracerebral Hemorrhage Research; Springer: Vienna, Austria, 2011; Volume 111, pp. 151–154. ISBN 9783709106938. [Google Scholar]
- Keric, N.; Maier, G.S.; Samadani, U.; Kallenberg, K.; Dechent, P.; Brueck, W.; Heuer, J.; Rohde, V. Tissue plasminogen activator induced delayed edema in experimental porcine intracranial hemorrhage: Reduction with plasminogen activator inhibitor-1 administration. Transl. Stroke Res. 2012, 3, 88–93. [Google Scholar] [CrossRef]
- Küker, W.; Thiex, R.; Rohde, I.; Rohde, V.; Thron, A. Experimental acute intracerebral hemorrhage. Value of MR sequences for a safe diagnosis at 1.5 and 0.5 T W. Acta Radiol. 2000, 41, 544–552. [Google Scholar] [CrossRef]
- Bimpis, A.; Papalois, A.; Tsakiris, S.; Zarros, A.; Kalafatakis, K.; Botis, J.; Stolakis, V.; Zissis, K.M.; Liapi, C. Activation of acetylcholinesterase after U-74389G administration in a porcine model of intracerebral hemorrhage. Metab. Brain Dis. 2012, 27, 221–225. [Google Scholar] [CrossRef]
- Bimpis, A.; Papalois, A.; Tsakiris, S.; Kalafatakis, K.; Zarros, A.; Gkanti, V.; Skandali, N.; Al-Humadi, H.; Kouzelis, C.; Liapi, C. Modulation of crucial adenosinetriphosphatase activities due to U-74389G administration in a porcine model of intracerebral hemorrhage. Metab. Brain Dis. 2013, 28, 439–446. [Google Scholar] [CrossRef]
- Bimpis, A.; Papalois, A.; Voumvourakis, K.; Oláh, O.; Tiszlavicz, L.; Liapi, C. Neuronal tumour necrosis factor-α and interleukin-1β expression in a porcine model of intracerebral haemorrhage: Modulation by U-74389G. Brain Res. 2015, 1615, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Loftspring, M.C.; Clark, J.F.; Wagner, K.R. A novel duplex ELISA method for quantitation of plasma proteins in areas of brain edema. Brain Res. 2007, 1162, 130–132. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Arakawa, H.; Marks, M.P.; Do, H.M.; Bouley, D.M.; Strobel, N.; Moore, T.; Fahrig, R. Experimental study of intracranial hematoma detection with flat panel detector C-arm CT. Am. J. Neuroradiol. 2008, 29, 766–772. [Google Scholar] [CrossRef] [PubMed]
- Clark, J.F.; Loftspring, M.; Wurster, W.L.; Beiler, S.; Beiler, C.; Wagner, K.R.; Pyne-Geithman, G.J. Bilirubin oxidation products, oxidative stress, and intracerebral hemorrhage. Acta Neurochir. Suppl. 2008, 105, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.H.; Wang, L.; Shi, X.T.; You, F.S.; Fu, F.; Liu, R.G.; Dai, M.; Zhao, Z.W.; Gao, G.D.; Dong, X.Z. Real-time imaging and detection of intracranial haemorrhage by electrical impedance tomography in a piglet model. J. Int. Med. Res. 2010, 38, 1596–1604. [Google Scholar] [CrossRef] [PubMed]
- Hvas, C.L.; Nielsen, T.K.; Barklin, A.; Sørensen, J.C.H.; Pedersen, M.; Andersen, G.; Tønnesen, E. Brain death induced by cerebral haemorrhage A new porcine model evaluated by CT angiography. Acta Anaesthesiol. Scand. 2012, 56, 995–1005. [Google Scholar] [CrossRef] [PubMed]
- Santos, E.; Orakcioglu, B.; Kentar, M.M.; Diedler, J.; Uozumi, Y.; Schöll, M.; Unterberg, A.; Sakowitz, O.W. Pressure reactivity index correlates with metabolic dysfunction in a porcine model of intracerebral hemorrhage. In Acta Neurochirurgica Supplementum; Springer: Vienna, Austria, 2012; Volume 114, pp. 363–367. ISBN 9783709109564. [Google Scholar]
- Hvas, C.L.; Fenger-Eriksen, C.; Høyer, S.; Sørensen, B.; Tønnesen, E. Hypercoagulation following brain death cannot be reversed by the neutralization of systemic tissue factor. Thromb. Res. 2013, 132, 300–306. [Google Scholar] [CrossRef]
- Monteith, S.J.; Harnof, S.; Medel, R.; Popp, B.; Wintermark, M.; Lopes, M.B.S.; Kassell, N.F.; Elias, W.J.; Snell, J.; Eames, M.; et al. Minimally invasive treatment of intracerebral hemorrhage with magnetic resonance-guided focused ultrasound: Laboratory investigation. J. Neurosurg. 2013, 118, 1035–1045. [Google Scholar] [CrossRef]
- Harnof, S.; Zibly, Z.; Hananel, A.; Monteith, S.; Grinfeld, J.; Schiff, G.; Kulbatski, I.; Kassell, N. Potential of magnetic resonance-guided focused ultrasound for intracranial hemorrhage: An in vivo feasibility study. J. Stroke Cerebrovasc. Dis. 2014, 23, 1585–1591. [Google Scholar] [CrossRef]
- Kandadai, M.A.; Korfhagen, J.J.; Beiler, S.; Beiler, C.; Wagner, K.; Adeoye, O.M.; Shaw, G.J. In vivo testing of a non-invasive prototype device for the continuous monitoring of intracerebral hemorrhage. J. Neurosci. Methods 2014, 235, 117–122. [Google Scholar] [CrossRef][Green Version]
- Wagner, K.R.; Xi, G.; Hua, Y.; Kleinholz, M.; De Courten-Myers, G.M.; Myers, R.E. Early metabolic alterations in edematous perihematomal brain regions following experimental intracerebral hemorrhage. J. Neurosurg. 1998, 88, 1058–1065. [Google Scholar] [CrossRef] [PubMed]
- Orakcioglu, B.; Kentar, M.M.; Schiebel, P.; Uozumi, Y.; Unterberg, A.; Sakowitz, O.W. Perihemorrhagic ischemia occurs in a volume-dependent manner as assessed by multimodal cerebral monitoring in a porcine model of intracerebral hemorrhage. Neurocrit. Care 2015, 22, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Haque, M.E.; Gabr, R.E.; Zhao, X.; Hasan, K.M.; Valenzuela, A.; Narayana, P.A.; Ting, S.M.; Sun, G.; Savitz, S.I.; Aronowski, J. Serial quantitative neuroimaging of iron in the intracerebral hemorrhage pig model. J. Cereb. Blood Flow Metab. 2018, 38, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Sidyakin, A.A.; Kaysheva, A.L.; Kopylov, A.T.; Lobanov, A.V.; Morozov, S.G. Proteomic analysis of cerebral cortex extracts from Sus scrofa with induced hemorrhagic stroke. J. Mol. Neurosci. 2018, 65, 28–34. [Google Scholar] [CrossRef]
- Haque, M.E.; Gabr, R.E.; George, S.D.; Zhao, X.; Boren, S.B.; Zhang, X.; Ting, S.M.; Sun, G.; Hasan, K.M.; Savitz, S.; et al. Serial metabolic evaluation of perihematomal tissues in the intracerebral hemorrhage pig model. Front. Neurosci. 2019, 13, 1–8. [Google Scholar] [CrossRef]
- Walsh, K.B.; Zhang, X.; Zhu, X.; Wohleb, E.; Woo, D.; Lu, L.; Adeoye, O. Intracerebral hemorrhage induces monocyte-related gene expression within six hours: Global transcriptional profiling in swine ICH. Metab. Brain Dis. 2019, 34, 763–774. [Google Scholar] [CrossRef]
- Mayfrank, L.; Kissler, J.; Raoofi, R.; Delsing, P.; Weis, J.; Küker, W.; Gilsbach, J.M. Ventricular dilatation in experimental intraventricular hemorrhage in pigs: Characterization of cerebrospinal fluid dynamics and the effects of fibrinolytic treatment. Stroke 1997, 28, 141–148. [Google Scholar] [CrossRef]
- Mayfrank, L.; Kim, Y.; Kissler, J.; Delsing, P.; Gilsbach, J.M.; Schröder, J.M.; Weis, J. Morphological changes following experimental intraventricular haemorrhage and intraventricular fibrinolytic treatment with recombinant tissue plasminogen activator. Acta Neuropathol. 2000, 100, 561–567. [Google Scholar] [CrossRef]
- Looi, T.; Piorkowska, K.; Mougenot, C.; Waspe, A.; Hynynen, K.; Drake, J. An MR-based quantitative intraventricular hemorrhage porcine model for MR-guided focused ultrasound thrombolysis. Child’s Nerv. Syst. 2018, 34, 1643–1650. [Google Scholar] [CrossRef]
- Xi, G.; Wagner, K.R.; Keep, R.F.; Hua, Y.; De Courten-Myers, G.M.; Broderick, J.P.; Brott, T.G.; Hoff, J.T. Role of blood clot formation on early edema development after experimental intracerebral hemorrhage. Stroke 1998, 29, 2580–2586. [Google Scholar] [CrossRef]
- Wagner, K.R.; Xi, G.; Hua, Y.; Zuccarello, M.; De Courten-Myers, G.M.; Broderick, J.P.; Brott, T.G. Ultra-early clot aspiration after lysis with tissue plasminogen activator in a porcine model of intracerebral hemorrhage: Edema reduction and blood-brain barrier protection. J. Neurosurg. 1999, 90, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Wagner, K.R.; Packard, B.A.; Hall, C.L.; Smulian, A.G.; Linke, M.J.; De Courten-Myers, G.M.; Packard, L.M.; Hall, N.C. Protein oxidation and heme oxygenase-1 induction in porcine white matter following intracerebral infusions of whole blood or plasma. Dev. Neurosci. 2002, 24, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Hemphil, J.C.I.; Morabito, D.; Farrant, M.; Manley, G.T. Brain tissue oxygen monitoring in intracerebral hemorrhage. Neurocrit. Care 2005, 3, 260–270. [Google Scholar] [CrossRef]
- Loftspring, M.C.; Beiler, S.; Beiler, C.; Wagner, K.R. Plasma proteins in edematous white matter after intracerebral hemorrhage confound immunoblots: An ELISA to quantify contamination. J. Neurotrauma 2006, 23, 1904–1911. [Google Scholar] [CrossRef]
- Wagner, K.R.; Beiler, S.; Beiler, C.; Kirkman, J.; Casey, K.; Robinson, T.; Larnard, D.; De Courten-Myers, G.M.; Linke, M.J.; Zuccarello, M. Delayed profound local brain hypothermia markedly reduces interleukin-1β gene expression and vasogenic edema development in a porcine model of intracerebral hemorrhage. Acta Neurochir. Suppl. 2006, 177–182. [Google Scholar] [CrossRef]
- Yin, X.; Zhang, X.; Wang, W.; Chang, L.; Jiang, Y.; Zhang, S. Perihematoma damage at different time points in experimental intracerebral hemorrhage. J. Huazhong Univ. Sci. Technol. 2006, 26, 59–62. [Google Scholar] [CrossRef]
- Backer-Grøndahl, A.; Lindal, S.; Lorentzen, M.A.; Eldevik, P.; Vorren, T.; Kristiansen, B.; Vangberg, T.; Ytrebø, L.M. A new non-craniotomy model of subarachnoid hemorrhage in the pig: A pilot study. Lab. Anim. 2016, 50, 379–389. [Google Scholar] [CrossRef]
- Nyberg, C.; Karlsson, T.; Hillered, L.; Engstrӧm, E.R. Metabolic pattern of the acute phase of subarachnoid hemorrhage in a novel porcine model: Studies with cerebral microdialysis with high temporal resolution. PLoS ONE 2014, 9, e0099904. [Google Scholar] [CrossRef]
- Nyberg, C.; Karlsson, T.; Hillered, L.; Stridsberg, M.; Engström, E.R. The early endocrine stress response in experimental subarachnoid hemorrhage. PLoS ONE 2016, 11, e0151457. [Google Scholar] [CrossRef]
- Mayberg, M.R.; Okada, T.; Bark, D.H. The role of hemoglobin in arterial narrowing after subarachnoid hemorrhage. J. Neurosurg. 1990, 72, 634–640. [Google Scholar] [CrossRef]
- Mayberg, M.R.; Okada, T.; Bark, D.H. The significance of morphological changes in cerebral arteries after subarachnoid hemorrhage. J. Neurosurg. 1990, 72, 626–633. [Google Scholar] [CrossRef] [PubMed]
- Ebel, H.; Semmelmann, G.; Friese, M.; Volz, M.; Lee, J.Y.; Dück, M.; Schomäcker, K.; Varga, J.; Furka, I.; Schröder, R.; et al. Effects of electrical stimulation of the Gasserian ganglion on regional cerebral blood flow after induced subarachnoid hemorrhage in pigs evaluated by 99mTc-HMPAO-SPECT. Minim. Invasive Neurosurg. 2001, 44, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Dueck, M.; Ebel, H.; Volz, M.; Lee, J.Y.; Semmelmann, G. Triple “H” therapy does not increase cerebral blood flow after induced subarachnoid hemorrhage in pigs. Results of a pilot 99mTC-HMPAO-single photon emission computed tomography study. Acta Neurochir. Suppl. 2001, 77, 243–246. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yin, Y.H.; Jia, F.; Jiang, J.Y. Effects of topical administration of nimodipine on cerebral blood flow following subarachnoid hemorrhage in pigs. J. Neurotrauma 2013, 30, 591–596. [Google Scholar] [CrossRef]
- Ganz, J.C.; Zwetnow, N.N. A quantitative study of some factors affecting the outcome of experimental epidural bleeding in swine. Acta Neurochir. (Wien) 1990, 102, 164–172. [Google Scholar] [CrossRef]
- Ganz, J.C.; Hall, C.; Zwetnow, N.N. Cerebral blood flow during experimental epidural bleeding in swine. Acta Neurochir. (Wien) 1990, 103, 148–157. [Google Scholar] [CrossRef]
- Edwards, D.H.; Byrne, J.V.; Griffith, T.M. The effect of chronic subarachnoid hemorrhage on basal endothelium-derived relaxing factor activity in intrathecal cerebral arteries. J. Neurosurg. 1992, 76, 830–837. [Google Scholar] [CrossRef]
- Salar, G.; Ori, C.; Lob, I.; Costella, G.B.; Battaggia, C.; Peserico, L. Cerebral blood flow changes induced by electrical stimulation of the Gasserian ganglion after experimentally induced subarachnoid haemorrhage in pigs. Acta Neurochir. (Wien) 1992, 119, 115–120. [Google Scholar] [CrossRef]
- Dai, M.; Wang, L.; Xu, C.; Li, L.; Gao, G.; Dong, X. Real-time imaging of subarachnoid hemorrhage in piglets with electrical impedance tomography. Physiol. Meas. 2010, 31, 1229–1239. [Google Scholar] [CrossRef]
- Xu, H.; Chen, X.; Qin, Z.; Gu, Y.; Zhou, P. Effect of recombinant streptokinase on the development of chronic cerebral vasospasm after subarachnoid hemorrhage in a swine model. Acta Neurochir. (Wien) 2011, 153, 1333–1338. [Google Scholar] [CrossRef]
- Takemae, T.; Branson, P.J.; Alksne, J.F. Intimal proliferation of cerebral arteries after subarachnoid blood injection in pigs. J. Neurosurg. 1984, 61, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Alksne, J.F.; Branson, P.J.; Bailey, M. Modification of experimental post-subarachnoid hemorrhage vasculopathy with intracisternal plasmin. Neurosurgery 1986, 19, 20–25. [Google Scholar] [CrossRef]
- Alksne, J.F.; Branson, P.J.; Bailey, M. Modification of experimental post-subarachnoid hemorrhage vasculopathy with intracisternal plasmin. Neurosurgery 1988, 23, 335–337. [Google Scholar] [CrossRef] [PubMed]
- Farstad, T.; Odden, J.P.; Bratlid, D. Effect of intraventricular hemorrhage on pulmonary function in newborn piglets. Biol. Neonate 1994, 66, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, G.A.; Mun-Bryce, S.; Wesley, M.; Komfeld, M. Collagenase-induced intracerebral hemorrhage in rats. Stroke 1990, 21, 801–807. [Google Scholar] [CrossRef] [PubMed]
- Ghanouni, P.; Pauly, K.B.; Elias, W.J.; Henderson, J.; Sheehan, J.; Monteith, S.J.; Wintermark, M. Transcranial MR-guided focused ultrasound: A review of the technology and neuro applications. AJR Am. J. Roentgenol 2015, 205, 150–159. [Google Scholar] [CrossRef]
- Taylor-Rowan, M.; Wilson, A.; Dawson, J.; Quinn, T.J. Functional assessment for acute stroke trials: Properties, analysis, and application. Front. Neurol. 2018, 9, 1–10. [Google Scholar] [CrossRef]
- Broderick, J.P.; Adeoye, O.; Elm, J. The evolution of the modified rankin scale and its use in future stroke trials. Stroke 2017, 48, 2007–2012. [Google Scholar] [CrossRef]
- Purdy, P.D.; Devous, M.D.; Batjer, H.H.; White, C.L.; Meyer, Y.; Samson, D.S. Microfibrillar collagen model of canine cerebral infarction. Stroke 1989, 20, 1361–1367. [Google Scholar] [CrossRef]
- Tibbs, R.; Zubkov, A.; Aoki, K.; Meguro, T.; Badr, A.; Parent, A.; Zhang, J. Effects of mitogen-activated protein kinase inhibitors on cerebral vasospasm in a double-hemorrhage model in dogs. J. Neurosurg. 2000, 93, 1041–1047. [Google Scholar] [CrossRef]
- Kinder, H.A.; Baker, E.W.; West, F.D. The pig as a preclinical traumatic brain injury model: Current models, functional outcome measures, and translational detection strategies. Neural Regen. Res. 2019, 14, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Allen, B.S.; Veluz, J.S.; Buckberg, G.D.; Aeberhard, E.; Ignarro, L.J.; Verrier, E.D. Deep hypothermic circulatory arrest and global reperfusion injury: Avoidance by making a pump prime reperfusate—A new concept. J. Thorac. Cardiovasc. Surg. 2003, 125, 625–632. [Google Scholar] [CrossRef]
- Sagnier, S.; Sibon, I. The new insights into human brain imaging after stroke. J. Neurosci. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, E.E.; West, F.D. Large animal ischemic stroke models: Replicating human stroke pathophysiology. Neural Regen. Res. 2020, 15, 1377–1387. [Google Scholar] [CrossRef] [PubMed]
- MacDonell, J.; Patel, N.; Fischer, G.; Burdette, E.C.; Qian, J.; Chumbalkar, V.; Ghoshal, G.; Heffter, T.; Williams, E.; Gounis, M.; et al. Robotic assisted MRI-guided interventional interstitial MR-guided focused ultrasound ablation in a swine model. Neurosurgery 2019, 84, 1138–1147. [Google Scholar] [CrossRef] [PubMed]
| Type of Approach | Approach | Method | Target | Advantages | Disadvantages | References |
|---|---|---|---|---|---|---|
| Surgical | Craniotomy | ET-1 injection | MCA | (1) Temporary ischemia | (1) Invasive approach | [87] |
| (2) Partial ischemia | ||||||
| Photothrombosis | MCA | (1) Temporary (2) Partial ischemia | (1) Invasive approach | [88,96,97,98,99,100] | ||
| (2) Reproducible | ||||||
| Electrocoagulation | MCA | (1) Partial ischemia (2) Reproducible | (1) Invasive approach (2) Not temporary | [16,52,101,102,103,104,105,106,107] | ||
| ICA | ||||||
| AChA | ||||||
| MCA + ICA | ||||||
| Arterial clip | MCA | (1) Temporary (2) Partial ischemia | (1) Invasive approach | [86,108] | ||
| (3) Reproducible | ||||||
| Cranial burr hole | ET-1 injection | MCA | (1) Temporary (2) Partial ischemia | (1) Relatively invasive approach | [59,109,110] | |
| Striatum | ||||||
| Cortex | ||||||
| Transorbital | Electrocoagulation | MCA | (1) Partial ischemia (2) Reproducible | (1) Very invasive approach (2) Not temporary | [89,111,112,113,114,115] | |
| Arterial clip | MCA | (1) Temporary (2) Partial ischemia | (1) Very invasive approach | [7,111,113,116] | ||
| (3) Reproducible | ||||||
| Endovascular | Endovascular | Mechanical embolization | Extracranial arteries | (1) Minimally invasive (2) Temporary | (1) No infarction | [117] |
| Blood clot injection | Extracranial arteries | (1) Minimally invasive (2) Temporary | (1) Difficult infarction | [118,119,120,121,122,123,124,125,126,127,128,129,130,131] | ||
| CCA | ||||||
| APA | ||||||
| Polymer injection | APA-rete mirabile | (1) Minimally invasive | (1) Difficult infarction | [132,133,134] | ||
| (2) Not temporary | ||||||
| CO2 injection | CCA | (1) Minimally invasive | (1) No infarction | [135] | ||
| (2) Temporary | ||||||
| Air injection | ICA | (1) Minimally invasive | (1) No infarction | [136] | ||
| (2) Temporary | ||||||
| DMSO injection | Rete mirabile | (1) Minimally invasive | (1) No infarction | [137] | ||
| (2) Temporary |
| Type of Approach | Approach | Method | Target | Advantages | Disadvantages | References |
|---|---|---|---|---|---|---|
| Intracranial hemorrhage | Craniotomy | MRgFUS | Brain parenchyma | (1) Reproducible | (1) Invasive | [160] |
| Collagenase injection | Brain parenchyma | (1) Reproducible | (1) Invasive | [161,162,163,164] | ||
| (2) Only used in juvenile pigs | ||||||
| Single blood injection | Brain parenchyma | (1) Reproducible | (1) Invasive | [165] | ||
| Transcranial | MRgFUS | Brain parenchyma | (1) Reproducible (2) Minimally invasive | (1) Target limitation | [166,167] | |
| Cranial burr hole | Double blood injection | Brain parenchyma | (1) Reproducible | (1) Relatively invasive | [51,69,168,169,170,171] | |
| Balloon catheter dilation and double blood injection | Brain parenchyma | (1) Reproducible | (1) Relatively invasive | [172,173,174,175,176,177,178,179,180,181,182] | ||
| Balloon catheter dilation and single blood injection | Brain parenchyma | (1) Reproducible | (1) Relatively invasive | [183,184,185,186] | ||
| Single blood injection | Ventricle | (1) Reproducible | (1) Relatively invasive | [158,159,187,188,189,190,191,192,193,194,195,196,197,198,199,200,201,202,203,204,205,206,207,208,209,210,211,212] | ||
| Brain parenchyma | ||||||
| Meningeal hemorrhage | Transorbital | Single blood injection | Subarachnoid space | (1) Reproducible (2) Minimally invasive | (1) Technically difficult | [213] |
| Cranial burr hole | Single blood injection | Subarachnoid space | (1) Reproducible | (1) Relatively invasive | [214,215] | |
| Craniotomy | Single blood injection | Subarachnoid space | (1) Reproducible | (1) Invasive | [157,216,217,218,219,220] | |
| Balloon catheter dilation and single blood injection | Epidural space | (1) Reproducible | (1) Invasive | [221,222] | ||
| Intrathecal | Single blood injection | Cisterna magna (subarachnoid space) | (1) Reproducible | [223,224,225,226] | ||
| (2) Minimally invasive | ||||||
| Laminectomy | Single blood injection | Pontine cistern (subarachnoid space) | (1) Reproducible | (1) Invasive | [227,228,229] |
| Characteristic | Swine | Rodents |
|---|---|---|
| Brain mass | 80–180 g (depending on strain and age), roughly 10x smaller than human [5,9] | 0.3 g (mice), roughly 4500x smaller than human; 2 g (rat), roughly 700x smaller than human [9] |
| Cortex morphology | Gyrencephalic [5,6,7] | Lissencephalic [1] |
| Myelination timeline | Similar to human, from birth to early adulthood [6,12] | Ends a few days after birth [10] |
| White matter proportion | 60%, same as humans [1,22,23], with similar connectivity [13] | 10% [1,22,23] |
| Intracerebral vessel diameter | Large, human-like enough to allow the use of human endovascular devices [15,20] | Much smaller, complicating surgeries [15,20] |
| Cerebral irrigation | High collateralization, complicates stable infarction generation [85,87], with 2–4 MCAs per side [7,88,89]. Rete mirabile to access ICA [85,86] | Lower collateralization, more stable infarction [15], with 1 MCA per side [23], and without a rete [85,86]. |
| Dura matter | Fibrous in swine, due to brain swelling generates a human-like ICP increase [14] | Delicate in rodents, rudimentary, and underdeveloped [14] |
| Neurological behavior | More sophisticated and inferable to human [3] | Simpler and less generalizable to human [3] |
| Genetics | Shorter phylogenetic distance with human [30,31] | Extended phylogenetic distance with human [30,31] |
| Size | Human-like depending on strain and age, allowing multimodal imaging sequences and instruments used in human [15,16], repeated and larger biological sample collection [3,29], and procedures and equipment from humans [2,3,8] | Much smaller, different imaging instruments [15,16], limited sample extraction [3,29], and different procedures and equipment [2,3,8] |
| Costs | Higher [15], but lower than NHP [12] | Lower [15] |
| Care and use | Difficult [15] | Easier [15] |
| Time to sexual maturity | Prolonged, but shorter than NHP [12] | Much shorter [15] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melià-Sorolla, M.; Castaño, C.; DeGregorio-Rocasolano, N.; Rodríguez-Esparragoza, L.; Dávalos, A.; Martí-Sistac, O.; Gasull, T. Relevance of Porcine Stroke Models to Bridge the Gap from Pre-Clinical Findings to Clinical Implementation. Int. J. Mol. Sci. 2020, 21, 6568. https://doi.org/10.3390/ijms21186568
Melià-Sorolla M, Castaño C, DeGregorio-Rocasolano N, Rodríguez-Esparragoza L, Dávalos A, Martí-Sistac O, Gasull T. Relevance of Porcine Stroke Models to Bridge the Gap from Pre-Clinical Findings to Clinical Implementation. International Journal of Molecular Sciences. 2020; 21(18):6568. https://doi.org/10.3390/ijms21186568
Chicago/Turabian StyleMelià-Sorolla, Marc, Carlos Castaño, Núria DeGregorio-Rocasolano, Luis Rodríguez-Esparragoza, Antoni Dávalos, Octavi Martí-Sistac, and Teresa Gasull. 2020. "Relevance of Porcine Stroke Models to Bridge the Gap from Pre-Clinical Findings to Clinical Implementation" International Journal of Molecular Sciences 21, no. 18: 6568. https://doi.org/10.3390/ijms21186568
APA StyleMelià-Sorolla, M., Castaño, C., DeGregorio-Rocasolano, N., Rodríguez-Esparragoza, L., Dávalos, A., Martí-Sistac, O., & Gasull, T. (2020). Relevance of Porcine Stroke Models to Bridge the Gap from Pre-Clinical Findings to Clinical Implementation. International Journal of Molecular Sciences, 21(18), 6568. https://doi.org/10.3390/ijms21186568






