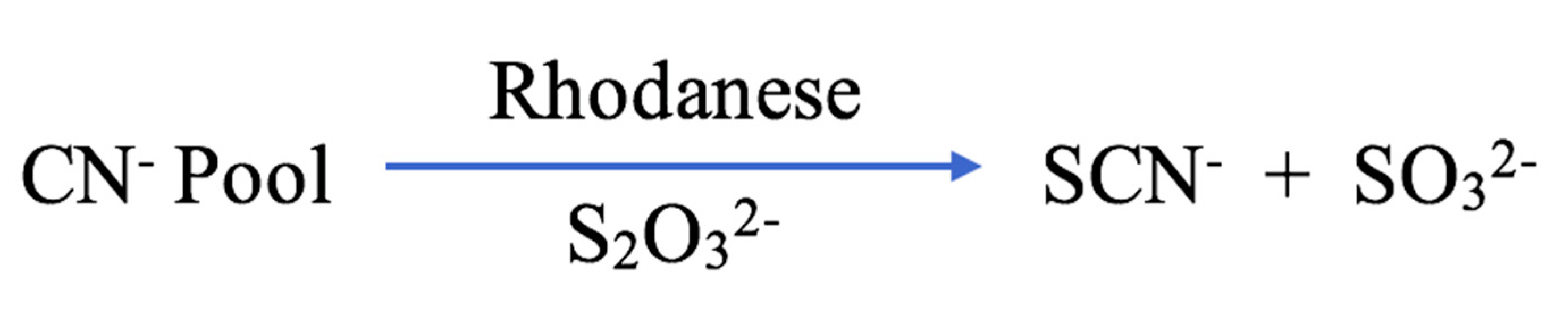Abstract
Thiocyanate (SCN−) is a pseudohalide anion omnipresent across mammals and is particularly concentrated in secretions within the oral cavity, digestive tract and airway. Thiocyanate can outcompete chlorine anions and other halides (F−, Br−, I−) as substrates for myeloperoxidase by undergoing two-electron oxidation with hydrogen peroxide. This forms their respective hypohalous acids (HOX where X− = halides) and in the case of thiocyanate, hypothiocyanous acid (HOSCN), which is also a bactericidal oxidative species involved in the regulation of commensal and pathogenic microflora. Disease may dysregulate redox processes and cause imbalances in the oxidative profile, where typically favoured oxidative species, such as hypochlorous acid (HOCl), result in an overabundance of chlorinated protein residues. As such, the pharmacological capacity of thiocyanate has been recently investigated for its ability to modulate myeloperoxidase activity for HOSCN, a less potent species relative to HOCl, although outcomes vary significantly across different disease models. To date, most studies have focused on therapeutic effects in respiratory and cardiovascular animal models. However, we note other conditions such as rheumatic arthritis where SCN− administration may worsen patient outcomes. Here, we discuss the pathophysiological role of SCN− in diseases where MPO is implicated.
Table of Contents
- Introduction
- 1.1.
- Sources, Secretion and Elimination of SCN−
- 1.1.1.
- Exogenous and Endogenous Sources of SCN−
- 1.1.2.
- Secretion and Elimination of SCN−
- 1.2.
- Role of MPO in SCN− Biochemistry
- 1.3.
- Halides and the Formation of MPO-mediated Oxidants
- SCN− in Diseases
- 2.1.
- Positive Effect of SCN− in Disease Outcome
- 2.1.1.
- Cardiovascular Disease
- 2.1.2.
- Respiratory Disease
- Respiratory Viral Infections
- 2.2.
- Negative Effect of SCN− in Disease Outcome
- 2.2.1.
- Smoking and Respiratory Infections
- 2.2.2.
- Autoimmune Rheumatic Diseases
- 2.2.3.
- Gastrointestinal Disease
- Conclusions
1. Introduction
1.1. Sources, Secretion and Elimination of SCN−
1.1.1. Exogenous and Endogenous Sources of SCN−
Thiocyanate (SCN−) is a 58 Da acidic, anionic thiolate molecule that exists in varying concentrations (0.01–2 mM) in secreted biological fluids, blood and urine [1,2,3]. There is significant variation amongst individuals with regard to SCN− concentrations in physiologic fluids, and this seems due to factors such as diet and smoking habits. Non-smokers have saliva SCN− concentrations of 0.5–2 mM, while concentration in smokers average around 3 mM, and some smokers may have concentrations as high as 6 mM (Table 1) [4,5,6]. One report of the SCN− status of U.S. vegetarians and vegans showed that vegans have average urine SCN− levels of 0.01 mM, almost double the average urine SCN− levels seen in vegetarians, which was 0.006 mM [7].

Table 1.
Ranges of SCN− concentration in various human biological fluids.
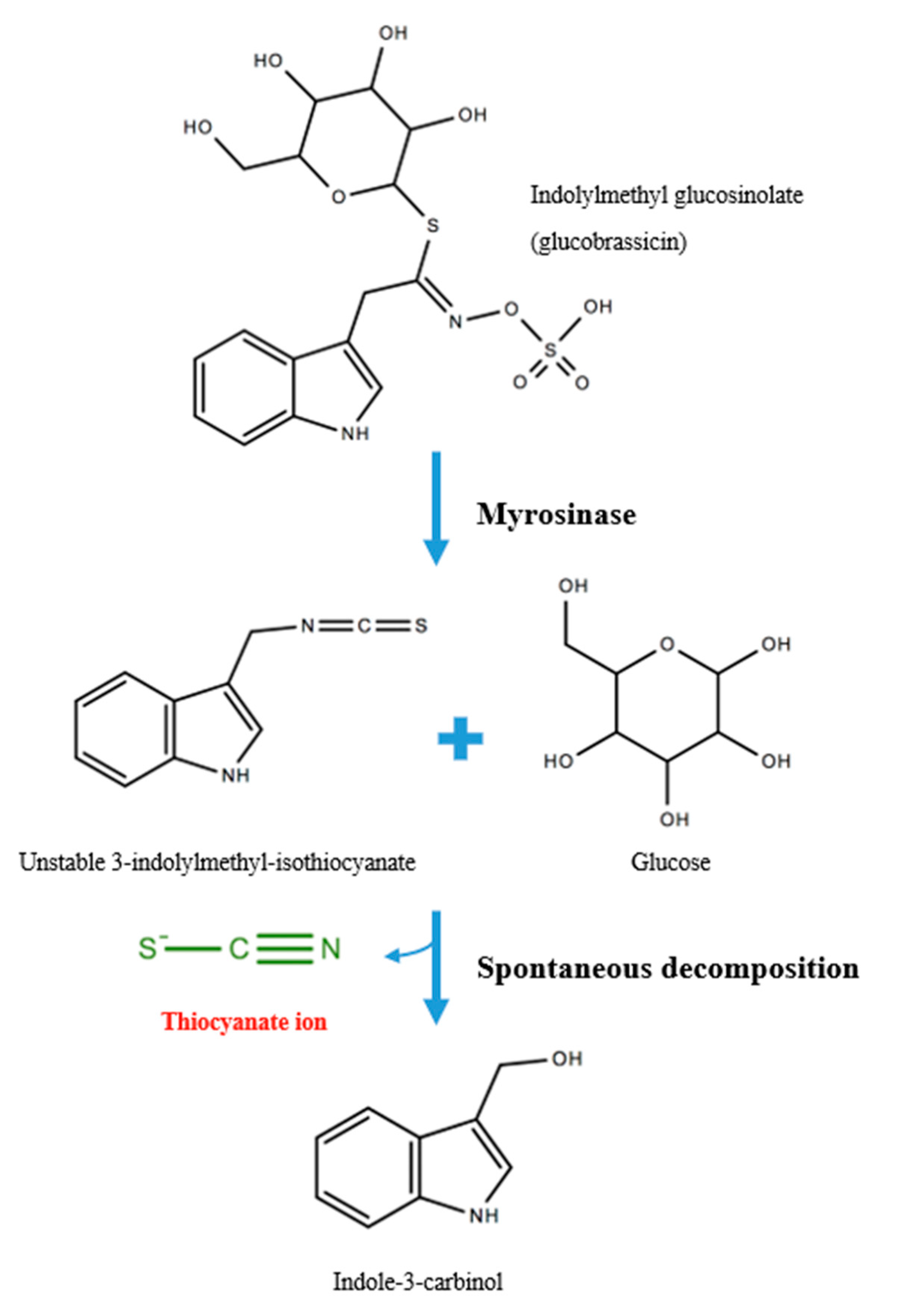
Dietary sources of SCN− include glucosidic cyanogen-rich plants such as almonds, cabbage, kale, broccoli, cassava, yam, maize, sugar cane, sorghum and linseed [7,8]. For example, glucobrassicin is a type of glucosinolate concentrated in cruciferous vegetables whereby plant-derived myrosinase (also known as β-thioglucosidase) mediates the hydrolysis of the glycoside, releasing a glucose and forming the unstable intermediate compound 3-indolylmethyl-isothiocyanate [9]. SCN− is subsequently released at neutral pH to form the stable product, indole-3-carbinol (Figure 1). Tobacco consumption including occupationally-derived smoke intake also contributes significantly to SCN− levels [10]. Further, SCN− is also generated from Pseudomonas aeruginosa cyanogenesis, an opportunistic pathogen that infects wounds and the lungs of immunocompromised individuals [11].
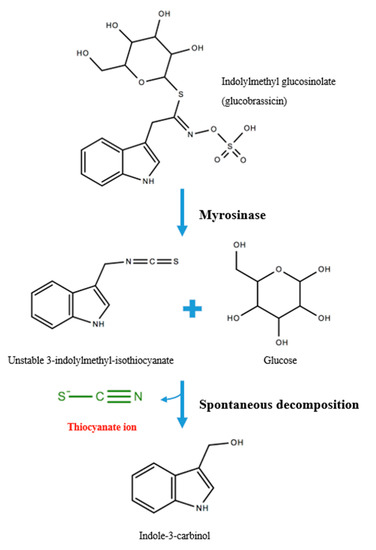
Figure 1.
Exogenous thiocyanate (SCN−) production from glucosinolates.
Although SCN− is mostly acquired from dietary sources, some is produced endogenously as a detoxification product of the reaction between cyanide (CN−) and thiosulfate (S2O32−) in the liver [12,13] (Figure 1). The transfer of a sulfur atom between S2O32− and CN− is catalysed by hepatic enzymes, including mitochondrial thiosulfate sulfurtransferase (or rhodanese) and cytosolic mercaptopyruvate sulfurtransferase (Figure 2) [14]. Sulfurtransferases are responsible for 80% of CN− metabolism [12]. Additionally, oxidation of SCN− into CN− can be mediated by haemoglobin, with the resulting CN− further detoxified by vitamin B12 (cobalamin) and its precursor cobinamide, before being excreted through the renal system [15,16,17].
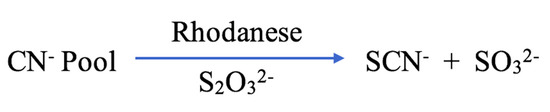
Figure 2.
The cyanide detoxification associated endogenous SCN− production pathway [12].
1.1.2. Secretion and Elimination of SCN−
Levels of SCN− in the secreted fluid of mammals can vary considerably depending on numerous factors, and can reach up to millimolar concentrations for mucous membranes lining the oral cavity, digestive tract and airway [18,19].
SCN− is secreted by airway, salivary, mammary, lacrimal and gastric glands, and is also present in plasma and urine. Mucosal secretions such as parotid, submandibular and whole saliva, as well as gingival crevicular fluid, dental plaque, nasal airway secretions, tears, gastric fluid and lung airway fluid can have up to approximately 2 mM SCN−, with saliva having the highest levels [6,18,20]. Salivary peroxidase and human lactoperoxidase (LPO) are also present in these secretions, and together with other antimicrobial defences in saliva likely account for the high levels of SCN− found [4,5,21]. Airway epithelial and nasal lining secretions, on the other hand, have typically lower levels at approximately 0.5 mM SCN− [18,20]. Blood plasma, breast milk and urine have SCN− levels 2 to 50 orders of magnitude lower compared with the aforementioned mucosal secretions (Table 1) [21].
It was proposed that SCN− is concentrated in certain fluids via energy-dependent active transport, and this was initially demonstrated in 1956 by Fletcher et al. [22]. In 1982, Tenovuo et al. showed that stimulating saliva flow rate via expectoration resulted in increased SCN− concentrations when compared with unstimulated saliva collected by drooling, suggesting that SCN− is actively transported in salivary glands to maintain salivary SCN− levels upon increased secretion [23]. Active transport of SCN− into saliva may also provide a recycling mechanism; as saliva is swallowed continuously, SCN− would be reabsorbed into the blood by gastrointestinal uptake and concentrated again in salivary glands.
In human bronchial epithelium, SCN− appears to be transported and concentrated via the basolaterally located sodium–iodine symporter (SLC5A5) in a Na+-dependent process [24]. Subsequent secretion at the apical membrane is via several separate mechanisms, including: the cAMP-mediated cystic fibrosis transmembrane conductance regulator (CFTR); purinergic agonists; Ca2+ and interleukin-4 (IL-4) sensitive Cl− channels; the IL-4 sensitive SCN−/Cl− exchanged pendrin (SLC26A4) [3,24,25,26].
The half-life of salivary SCN− is reported to be 6–14 days [6,27,28,29,30]. While endogenous SCN− does enter the glomerular filtrate, there is 90% reuptake and this accounts for the low urine SCN− levels found [7,31]. Plasma SCN− half-life in healthy individuals is reported to be from 1 to 5 days, and 9 days in individuals with renal insufficiency [31]. Due to its long biological half-life and the significant differences between smoker and non-smoker saliva, plasma and urinary SCN− concentrations have been used as helpful biomarkers for exposure to tobacco or occupational smoke [6,10].
1.2. Role of MPO in SCN− Biochemistry
Myeloperoxidase (MPO) is a heme homodimeric protein (~146 kDa) with functionally independent monomer units consisting of an iron protophorphyrin IX derivative located within the heavy chain of each monomer [36]. The heme unit is located within a deep cleft, restricting access of the iron atom to hydrogen peroxide (H2O2) [37]. Native MPO contains a heme unit within its active site in its ferric (Fe3+) form that can undergo a 2-electron oxidation reaction with H2O2, generating the highly reactive oxy-ferryl (Fe4+ = O) heme species containing a porphyrin π-cation radical [38], also known as Compound 1. MPO Compound 1 is highly reactive and thus can undergo 2-oxidant reduction by halides (e.g., Cl−, Br−) and pseudohalides (e.g., SCN−) to return to its native Fe3+ form [39]. This pathway is termed the “halogenation cycle”, yielding hypochlorous acid (HOCl), hypobromous acid (HOBr) and hypothiocyanous acid (HOSCN), respectively. Alternatively, Compound 1 can convert back to its native Fe3+ state via an independent pathway involving two sequential one-electron reductions, yielding the intermediate Compound 2 (Fe4+ = O) heme species in a process coined the “peroxidase cycle” [40].
The earliest report on the biological significance of SCN− was in 1814 by German physician Gottfried Reinhold Treviranus, as he discovered a substance in human saliva that produced a blood-red colour when ferric ion was added [21]. In the early to mid-20th century, SCN− had been of particular pharmacologic interest as an oral antihypertensive. Since then, multiple researchers have identified SCN− as a potentially important factor in health and disease.
SCN− plays a vital role as a substrate for human peroxidases, including MPO, LPO, salivary peroxidase, gastric peroxidase, eosinophil peroxidase and thyroid peroxidase [41]. Most of these are present in SCN−-containing extracellular fluids together with H2O2 [41]. These components act in concert, regulating innate immune processes as well as resident and transient flora [33].
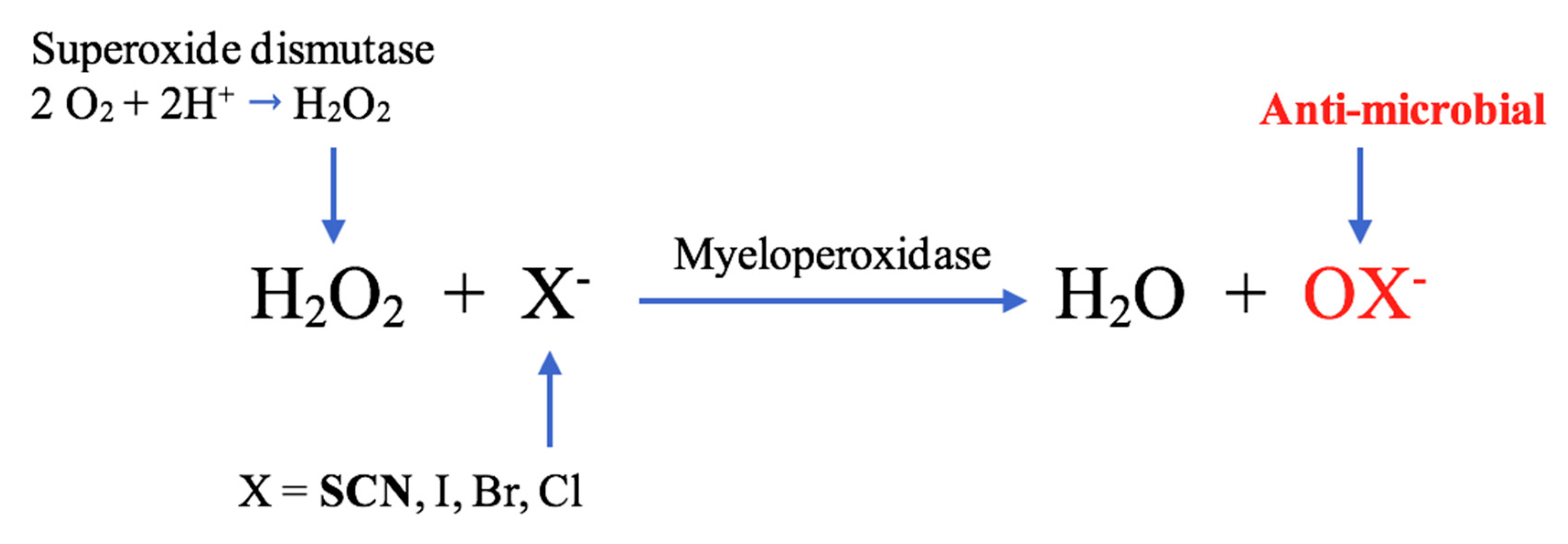
It is important to consider the chain of events in inflammatory lesions that lead SCN− to generate the potent bactericidal radical, HOSCN. Nicotinamide adenine dinucleotide phosphate (NADPH) oxidase is a multi-subunit enzyme present in neutrophils and macrophages. In inflammation, activation of NADPH oxidase catalyses the reaction between oxygen and NADPH, generating superoxide anions, a process that has been coined the “oxidative burst” [42,43]. Superoxide in turn undergoes dismutation, a process through which the anions are simultaneously oxidised and reduced to form H2O2. Dismutation can occur spontaneously or may be catalysed by the enzyme superoxide dismutase [39]. Lactoperoxidase in secretions, as well as MPO from degranulating leukocytes, catalyse the reaction of SCN− with H2O2 to produce HOSCN, which is highly effective in killing microbes in these inflammatory environments through its free radical activity (Figure 3) [39].
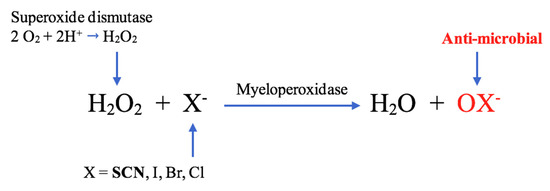
Figure 3.
Myeloperoxidase (MPO)-mediated formation of oxidation products, including hypothiocyanous acid (HOSCN), HOI, hypobromous acid (HOBr) and hypochlorous acid (HOCl) [41].
1.3. Halides and the Formation of MPO-mediated Oxidants
In addition to SCN−, halide ions (negatively charged halogen atoms) such as Cl− and Br− can also be oxidised into hypohalous acids following reaction with MPO and H2O2.
SCN− has a much higher specificity for MPO than Cl−, with specificity constants of 1, 60, and 730 for Cl−, Br−, and SCN−, respectively, so that SCN− is the preferred substrate for MPO [44]. However, plasma SCN− levels are comparatively very low, with normal halide/pseudohalide ion concentrations in healthy human plasma at 100–140 mM (Cl−), 20–100 µM (Br−) and 200–250 µM (SCN−) [44].
SCN− has potential to become a competitive substrate for MPO when its level is elevated beyond normal plasma level, especially in secretions where SCN− levels are much higher. This is in part due to its faster reaction rate compared with Cl− and Br− [45]. The second-order rate constants for SCN−, Cl− and Br− are 9.6 × 106 M−1s−1, 2.5 × 104 M−1s−1 and 1.1 × 106 M−1s−1, respectively [46].
Due to the nature and reactivity of different reactive oxygen species (ROS), HOCl is generally considered as a strong oxidant whilst HOSCN is often classified as a weak oxidant [47]. For example, the second order rate constants against cysteine residues for HOCl and HOSCN are 3.6 × 108 M−1s−1 and 7.8 × 104 M−1s−1, respectively. Similarly, HOSCN oxidises glutathione (GSH) with a second order rate constant considerably lower than HOCl, with values of 2.5 × 104 M−1s−1 and 1.2 × 108 M−1s−1, respectively [48,49].
2. SCN− in Diseases
SCN− is an important part of human host defence and health, with pharmacological interest dating back to 1857, where it was investigated for its clinical use as a hypotensive agent [50]. Use in clinical therapies is now restricted, however, due to potential toxic effects. In the 1970s, it was found that concentrations of SCN− in fasting gastric juice specimens from human patients increased the likelihood of nitrosamine production and this was implicated as a potential contributor to gastric cancer [51]. Moreover, chronic toxicity of sodium thiocyanate (NaSCN) was demonstrated in the 1980s in F344 rats, while high serum concentrations of SCN− were associated with lung cancer [52,53]. A more recent population-based cross-sectional study found that concentrations of urinary SCN− were significantly correlated with several diseases including cancer, chronic bronchitis, emphysema, coughing, wheezing and sleep-related conditions [54]. The positive association of SCN− with cancer and lung diseases in a national, population-based study supported previous investigations that utilised smaller and non-representative human sample sizes and those that used animals [54]. Recent research has focused primarily on respiratory and cardiovascular animal models of disease.
We now discuss the roles of SCN− in modulating MPO in disease activity and outcome.
2.1. Positive Effect of SCN− in Disease Outcome
2.1.1. Cardiovascular Disease
Cardiovascular disease (CVD) is the primary cause of death worldwide, with the majority of CVD deaths related to coronary heart disease (CHD) [55]. CHD is characterised by chronic vascular stenosis and subsequent ischaemic injury/end-organ damage, which is primarily mediated by inflammatory remodelling of the arterial wall [56]. Endothelial dysfunction is the pre-atherosclerotic manifestation associated with invasion of immune cells into the vessel wall and the formation of ROS. It is well documented that MPO is enriched in atherosclerotic plaques and plasma concentration of MPO is a predictive factor in cardiovascular mortality, following angiography in humans [57,58].
The chlorinating activity of MPO is thought to be particularly detrimental during CVD, with high density lipoproteins and low density lipoproteins being vulnerable to oxidation by HOCl and impairing endothelial function via interference with nitric oxide production [59,60,61]. HOCl also induces endothelial apoptosis [62,63,64,65]. It was previously reported that pharmacological inhibition of MPO by 4-aminobenzoic acid hydrazide reduces plaque formation in the mouse apolipoprotein E knockout (ApoE−/−) model of atherosclerosis [66]. Recently, a new generation of small molecule MPO inhibitors significantly reduced the size of atherosclerotic lesion necrotic cores in Ldlr−/− mice fed a western diet [67]. Despite the atherosclerotic plaque area remaining similar, MPO inhibition resulted in atherosclerotic plaque stabilisation in this murine model. Conflicting with this study is that an increase in atherosclerosis was observed in MPO−/− mice [68], and this may suggest that MPO-generated reactive intermediates might be protective in murine atherosclerosis, or alternatively microbial involvement following the complete knockout of MPO, a crucial antimicrobial enzyme. The latter possibility is supported by separate work showing pro-atherogenic effects of Porphyromonas gingivalis in mice, rabbits and pigs [69].
High serum levels of SCN− have been shown to improve long-term survival in patients following an acute myocardial infarction [70]. Ironically, smokers who are typically at risk for the early development of CVD have increased blood levels of SCN− [71]. Unlike HOCl, HOSCN can be specifically degraded via thioredoxin reductase, thereby reducing its oxidative capacity in vivo [72]. In this way, HOSCN may skew the oxidative profile of MPO, thus reducing oxidative injury of the arterial wall in atherosclerosis. This is consistent with observations in ApoE−/− mice fed a western diet, which have reduced atherosclerotic plaque size following 8 weeks of NaSCN treatment [73]. In this study, serum proinflammatory IL-6 levels were decreased, while IL-10 levels increased with NaSCN treatment, though no effect on monocyte or granulocyte infiltration was observed in the atherosclerotic plaque. Similarly, SCN− supplementation in atherosclerosis-prone Ldlr−/− mice transgenic for human MPO decreased the total plaque area with no changes to serum MPO concentrations between SCN− supplemented and control mice [44]. Collectively, these studies highlight the therapeutic potential for modulating MPO oxidative activity towards the production of HOSCN in CHD.
2.1.2. Respiratory Disease
SCN− is extensively involved in modulating the oxidative environments of various respiratory diseases, reducing the cytotoxic effect of more powerfully oxidative HOCl. For example, Xu and colleagues demonstrated the attenuation of MPO cytotoxicity by addition of SCN− to over 100 µM in the Calu-3 human lung epithelial, Neuro2a mouse neuroblastoma, human aortic endothelial cells and Min6 mouse pancreatic β cell lines [74]. In the same study, dose-dependent inhibition of MPO-produced OCl− by SCN− was demonstrated, with slight, partial and near complete inhibition achieved using 10 µM, 50 µM and 100–400 µM SCN−, respectively [74]. MPO activity in lung tissue has been previously linked to the cessation of ciliary beating [75,76] and damage to airway epithelial cells [77]. Physiologically, ciliary beating functions as a pathogen-clearing mechanism and impaired ciliary beating results in ineffective clearance of pathogenic bacterial species that can promote infection [75,78], contributing to various respiratory disorders including cystic fibrosis (CF).
CFTR is a transmembrane receptor that functions as a chloride channel at the apical membrane of epithelial cells and mutations in CFTR results in the clinical presentation of CF [79]. Interestingly, CFTR also conducts SCN−, as the anion permeability of CFTR for SCN− exceeds that of Cl− and the concentration of SCN− in the airway surface liquid is at least ~30 times its concentration in the serum [80,81]. This potentially limits harmful accumulations of Cl− that subsequently form HOCl in the presence of MPO, while also facilitating the formation of the effective antibacterial compound HOSCN [82]. Unsurprisingly, many researchers have reported a deficiency in the secretion of SCN− in both human CF cells [24,82], as well as human patients [20]. While nebulised hypertonic saline therapy in CF is reported to improve both SCN− and GSH airway surface liquid levels, a finding which was reproduced in a CFTR knockout animal model [23,83], there are no current clinical trials evaluating nebulised SCN− to date.
CF patients are particularly vulnerable to chronic P. aeruginosa airway infections, where the subsequent inflammatory response contributes to the major clinical problems associated with CF-lung tissue destruction [84]. Recent studies have investigated the anti-inflammatory and anti-bacterial activity of SCN− through the administration of nebulised SCN− in a cystic fibrosis model using beta epithelial sodium channel (βENaC) mice [85]. Compared with wild-type counterparts, βENaC mice administered SCN− significantly decreased airway neutrophil infiltrate by 68%, and therefore by extrapolation, neutrophil-derived MPO, as well as rebalanced GSH redox ratio in both lung tissue and the lining fluid of airway epithelium [85]. In contrast, there was no significant effect in the reduction in elevated levels of lymphocytes and macrophages in the bronchoalveolar lavage fluid of βENaC mice by administration of nebulised SCN−. Moreover, SCN− treatment had no effect on other cytokines, including C-X-C motif chemokine ligand 1, IL-1β, TNF-α, IFN-γ, IL-2, IL-4, IL-5, IL-6, IL-10, and IL-12 p70. Levels of the granulocyte oxidative activity biomarker glutathione sulfonamide and glutathione disulfide were also decreased in βENaC mice [85]. Interestingly, compared with wild-type mice, mean SCN− levels in the epithelial lining fluid were decreased by 60% in the βENaC mice that were given vehicle treatment. Thus, the administration of SCN− to βENaC mice and subsequent reduction in neutrophil infiltrate may be diverting MPO-modulated oxidative activity into another biological compartment.
Both wild-type and βENaC mice were also infected with P. aeruginosa, where SCN− administration decreased levels of inflammation, bacterial burden, proinflammatory cytokines and 3-nitrotyrosine (only in infected wild-type mice) [85]. Bacterial burden was 70% less in wild-type mice receiving SCN− as compared with wild-type mice given vehicle treatment. Furthermore, βENaC mice had an 80-fold increase in bacteria relative to their wild-type counterparts, with SCN− treatment significantly reducing bacterial burden by 92% [85]. The observed increase in neutrophilic influx after P. aeruginosa infection suggests a consequential increase in secreted MPO, and with these data in consideration, may suggest that bacterial burden is ameliorated potentially through the microbicidal action of MPO/SCN− derived HOSCN.
Taken together, the evidence suggests a protective role for SCN− in respiratory airways, where SCN− supplementation may provide a therapeutic effect in patients with CF. The anti-inflammatory action of SCN− may potentially be attributed to a decrease in bacterial burden, as well as in the mitigation of neutrophilic infiltration, thereby diverting MPO-mediated damage away from pulmonary tissue.
Respiratory Viral Infections
In addition to its antibacterial activity that combats respiratory infections, SCN−, as well as hypothiocyanite (OSCN−), the conjugate base of HOSCN, holds antiviral effects that have been investigated in various in vitro influenza viral systems. OSCN− virucidal activity against the A/H1N1 2009 pandemic influenza virus has been demonstrated in vitro. Specifically, a dose-dependent effect without cytotoxicity was observed where 2 µM OSCN− administered achieved inhibition of viral replication by 50% prior to cell inoculation [86]. Moreover, the LPO/H2O2/SCN− system has been shown to inactivate the A/Swine/02860/2009 influenza A strain virus within both differentiated rat and human tracheobronchial epithelial cells [87], and in a cell-free system [88]. Increased production of mucin and dual oxidase expression was demonstrated in the former model associated with inactivation of the influenza A virus, whilst the latter model displayed inactivation of the influenza A virus strains, including H1N1, H1N2, H3N2, and the influenza B viruses of Yamagata and Victoria lineages, though the extent of this inactivation varied between the influenza strain and LPO substrate (SCN− or I−) [88]. Nevertheless, it appears that the antiviral capabilities of SCN− and its chemical analogues working through the LPO/H2O2/SCN− system are beneficially implicated in inactivating numerous influenza virus strains in vitro. In light of the current global pandemic featuring the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), investigating the effects of SCN− on SARS-CoV-2 in vitro and perhaps in animal models, may be a potential avenue for further research.
2.2. Negative Effect of SCN− in Disease Outcome
2.2.1. Smoking and Respiratory Infections
As tobacco smoking increases SCN− levels considerably in the mucosa, one may assume that tobacco smoke can elicit antimicrobial effects; however, a plethora of evidence exists demonstrating that cigarette smoking exacerbates respiratory infections. For example, both passive and active smoking are significant risks for the development of upper respiratory tract infections, particularly otitis media [89,90,91] and for the colonisation of Streptococcus pneumoniae within the nasopharynx for both children and adults [92]. Furthermore, an examination of several systematic reviews and meta-analyses found that smokers are twice as likely to contract Mycobacterium tuberculosis infections, resulting in the development of, and death from, active tuberculosis, though interpretation may be affected due to differences in data from these studies [93,94,95]. The correlation between increased risk for the development of tuberculosis and cigarette smoke has been reported in various countries including India [96], China [97], South Africa [98] and Mexico [99]. Additionally, cigarette smoking increases the frequency of obtaining community-acquired pneumonia by approximately twofold [100] and in immunocompetent non-elderly adults, smoking is the top independent risk factor for invasive pneumococcal disease [101].
Profound changes in mucous production mechanisms and airway epithelial metaplastic changes can instead explain the increased susceptibility of smokers to respiratory infections. Squamous metaplasia of respiratory epithelium is common in habitual smokers, where specialised bronchial ciliated columnar epithelia is replaced with stratified squamous epithelia [102]. This adaptive mechanism provides a physical barrier against noxious chemicals in cigarette smoke but compromises normal ciliary function and thereby impacts the drainage of secretory products via mucociliary transport. The toxic metabolite of nicotine is also capable of significantly reducing the ciliary beat of epithelial cells as demonstrated in an in vitro model [103]. In addition, cigarette smoke increases the number and size of goblet cells in the respiratory mucosa, resulting in increased airway secretions [104]. Increased respiratory airway secretions coupled with reduced mucociliary transport is thought to increase the risk of respiratory infections, offsetting the benefit increased SCN− may offer.
2.2.2. Autoimmune Rheumatic Diseases
Rheumatoid arthritis (RA) is a T-cell and autoantibody-mediated autoimmune disease which results in joint damage and destruction of cartilage. Neutrophils are prominent in the pathogenesis of RA, as demonstrated in numerous RA mouse models as well as human disease [105]. In two prominent mouse models of RA, namely K/BxN antibody-mediated arthritis and collagen-induced arthritis, a specific role for MPO was indicated by reduced disease severity in MPO−/− mice [106,107,108,109]. Supporting a role for MPO in human disease, the enzyme is found in both intracellular and extracellular locations in the synovium of RA patients [110]. Furthermore, levels of the MPO-mediated specific oxidation product of HOCl, 3-chlorotyrosine (3-Cl-Tyr), are significantly higher in synovial fluids of RA patients compared with those from patients with osteoarthritis [110]. Neutrophils from RA patients spontaneously generate neutrophil extracellular traps ex vivo, which are associated with MPO release and this suggests a role for leukocyte priming [111].
Destruction of cartilage, but not bone, is largely attributed to matrix metalloproteinase (MMP) activity in RA [112]. Several MMPs are implicated in the pathogenesis of RA, including MMP-1, MMP-2, MMP-3, MMP-8, MMP-9, MMP-10, MMP-12 and MMP-13, and these degrade a broad range of matrix components [112]. MMPs are produced as inactive pro-forms which require either serine-protease cleavage of an inhibitory pro-peptide domain, or oxidation of the critical thiol cysteine residue, while both scenarios expose the catalytically active Zn2+ site [113]. The MPO oxidation product HOCl has previously been shown to activate MMP-7 by oxidation of the key cysteine residue to a sulfinic acid form [114]. While HOCl is a relatively promiscuous oxidant, HOSCN is less reactive and highly selective for thiol sites, which represent the major site of reaction [49,115]. Therefore, it is plausible that skewing the oxidative profile of MPO from HOCl to HOSCN can result in increased oxidation of catalytic Zn2+ sites of pro-MMPs, increasing the overall activity of MMPs. Although the respective roles of HOSCN and MMP are not fully elucidated, it is well established that cigarette smoke and the associated increase in serum SCN− is linked with increased severity and incidence of RA, and that this results in increased intensity and duration of disease, so that fewer smoking patients enter full remission [116]. In a rat model of experimental arthritis, sodium/potassium SCN− was supplemented to mimic elevated levels of SCN− in the blood, saliva, and urine of smokers [117]. Rats supplemented with SCN− showed pro-arthritic and proinflammatory changes when subjected to various arthritic-inducing agents.
Carbamylation is a non-enzymatic post-translational modification, whereby amine or thiol groups transform into carbamyl groups via the presence of increased cyanate (OCN−) [118]. OCN− forms a natural homeostatic equilibrium in physiological systems, where OCN− concentration is too low to allow extensive carbamylation of protein. However, several environmental factors, such as smoking, cause a pathophysiological increase in OCN−. Increased SCN− levels from cigarette smoke drive MPO Compound 1-mediated competitive oxidation of SCN− to HOSCN and, to a lesser extent, OCN− [71,119,120]. The conversion of lysine to homocitrulline is the most commonly described carbamylation process, and recently a new autoantibody system has been described in RA. Autoantibodies against proteins that contain homocitrulline residues (anti-carbamylated protein antibodies) are present in a subset of RA patients and can be independent from anti-citrullinated protein antibodies [119,121]. Interestingly, the presence of anti-carbamylated protein antibodies in RA patients is associated with more severe joint damage compared with patients who are negative for anti-citrullinated protein antibodies [122].
From the above, the evidence suggests that SCN− is pro-arthritic via the potential pathway for MPO-derived HOSCN to activate pro-MMP at synovial sites, or the MPO/HOSCN-mediated increase in local OCN− levels, increasing overall carbamylation of local proteins.
2.2.3. Gastrointestinal Disease
Two main conditions drive inflammatory bowel disease (IBD) where there is chronic inflammation in the gut: ulcerative colitis (UC) and Crohn’s disease. A chronic influx of leukocytes into the gut mucosa is a major pathological presentation in IBD, which is thought to result from an abnormal host immune response to otherwise harmless commensal flora [123]. In the context of UC, the extent of neutrophil infiltration correlates with the severity of disease and is incorporated into the clinical UC severity scoring method [124]. Moreover, neutrophil depletion in rodents ameliorates experimental colitis [125,126]. Further, we earlier demonstrated a role for neutrophil-derived MPO in murine experimental colitis, with significant attenuation of disease via pharmacological inhibition of MPO [127].
It is increasingly recognised that ROS generated during inflammation plays a significant role in prolonging gastrointestinal inflammatory cycles and causing gastrointestinal injury. Colonic and faecal MPO, present in polymorphonuclear leukocytes, are significantly increased and correlate with disease severity in UC patients [128,129]. MPO is a primary source of potent ROS, including hypohalous acids HOCl, HOSCN and HOBr in the inflamed colon [130,131]. However, the proportion of MPO-oxidants formed during UC or Crohn’s disease is yet to be assessed. Despite this, 3-Cl-Tyr, a HOCl-specific biomarker, is significantly increased in colonic and serum samples of IBD patients [132].
Interestingly, active smokers who have significantly higher SCN− concentrations in body fluids also exhibit reduced risk (1.7-fold) against UC and protection from the clinical symptoms including reduced flares, less need for steroids and a lower colectomy rate [133]. Thus, we speculated that SCN− supplementation would confer protection during experimental colitis by redirection of MPO halogenation to favour HOSCN production above HOCl.
In our recent study, mice were supplemented with NaSCN to closely match levels in human smokers [134] before being subjected to colitis in a 3% (w/v) dextran sodium sulfate (DSS) colitis model [135]. We observed increased faecal and serum SCN− levels in NaSCN-supplemented mice to above IC50 inhibition levels of MPO/HOCl, as determined by HOCl-mediated oxidation of luminol. Notably, 3-Cl-Tyr was found to be comparatively lower in colonic samples of SCN−-supplemented DSS mice. This indicated a reduction in the production of HOCl, potentially diverging MPO oxidation production to HOSCN by increased presence of free SCN− ions.
However, NaSCN supplementation did not attenuate the course of experimental murine colitis. No data were collected on mouse activity levels, colon lengths, or colonic histopathology in the SCN−-supplemented groups. Interestingly, mice supplemented with DSS/SCN− showed marked upregulation in thiol synthesis markers Nrf2 and GCLC, indicating that thiol synthesis was enhanced in this group of mice and may provide an increase in antioxidant status for the colon during DSS-insult. Overall, increasing SCN− in the gut and circulation provided minimal protection against active experimental colitis.
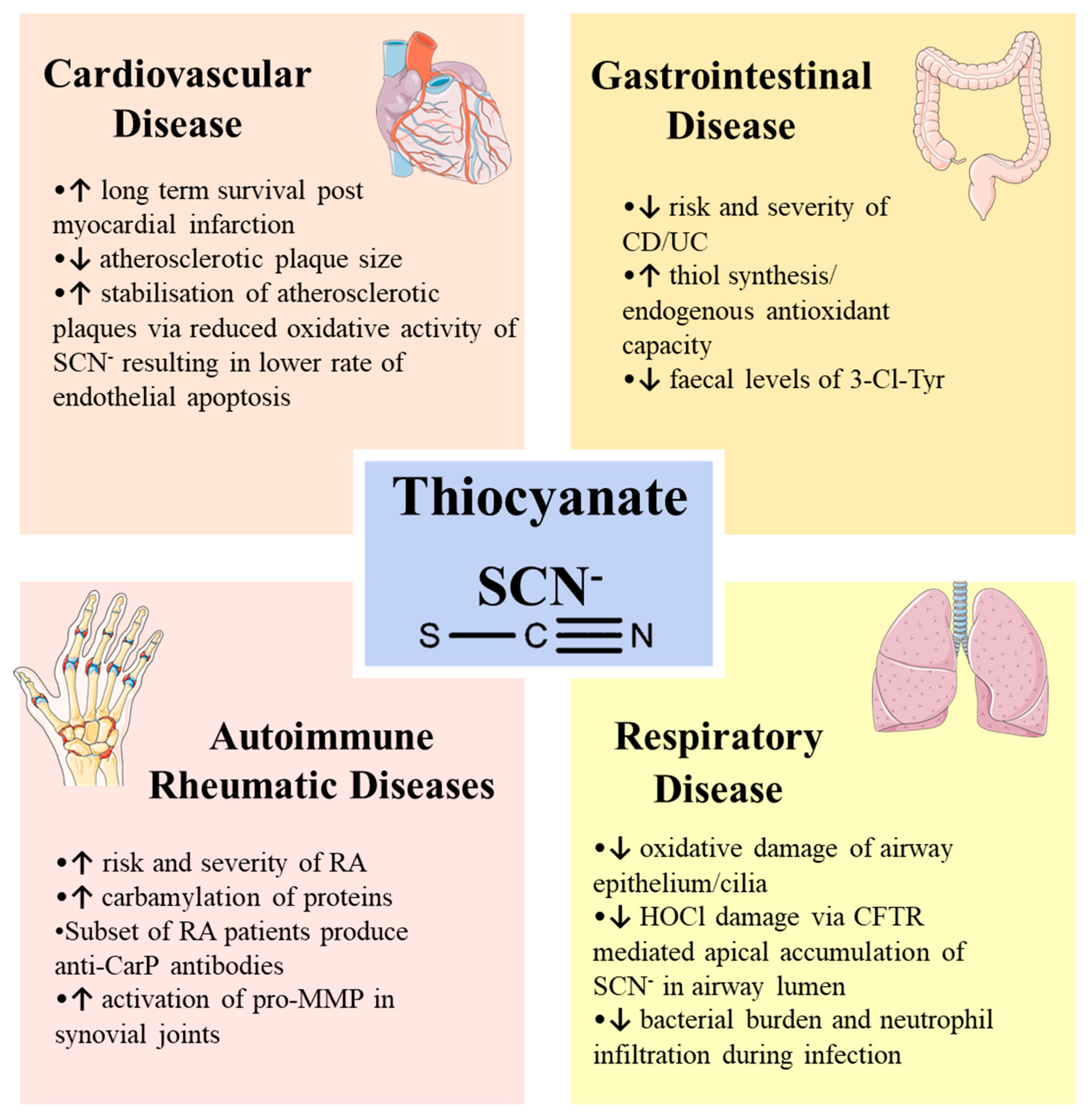
3. Conclusions
SCN− supplementation has been investigated in various disease models (Figure 4). Notably, in respiratory and cardiovascular disease, SCN− appears to be protective against disease via direct modulation of MPO activity, favouring the production of the HOSCN oxidant. On the other hand, SCN− seems to be implicated in the pathogenesis of rheumatic arthritis, while there is limited evidence to support a role in IBD. Irrespective of any possible direct roles for MPO in pathogenic mechanisms, the potential therapeutic value of SCN− must be carefully considered in the context of each specific clinical condition.
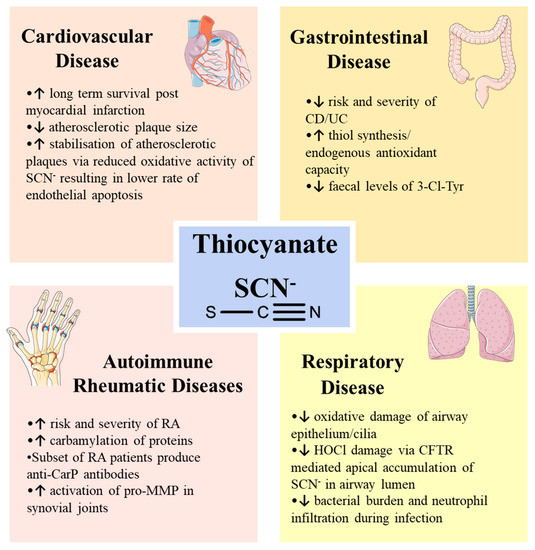
Figure 4.
Schematic overview of SCN− in various human diseases.
Author Contributions
B.C. was responsible for the conceptualization of this review and has contributed significantly to many sections, notably SCN− in disease. P.T.S.G. reviewed the literature and compiled the introduction (along with Y.L.) and diseases section. A.L.S. developed all schematic diagrams and H.Z. provided a substantive review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| 3-Cl-Tyr | 3-chlorotyrosine |
| ApoE | Apolipoprotein E Knockout |
| βENaC | Beta Epithelial Sodium Channel |
| CF | Cystic Fibrosis |
| CFTR | Cystic Fibrosis Transmembrane Conductance Regulator |
| CN− | Cyanide Ion |
| DSS | Dextran Sodium Sulfate |
| GSH | Glutathione |
| H2O2 | Hydrogen Peroxide |
| HOBr | Hypobromous Acid |
| HOCl | Hypochlorous Acid |
| HOSCN | Hypothiocyanous Acid |
| IBD | Inflammatory Bowel Disease |
| IL-4 | Interleukin-4 |
| LPO | Lactoperoxidase |
| MMP | Matrix Metalloproteinase |
| MPO | Myeloperoxidase |
| NADPH | Nicotinamide Adenine Dinucleotide Phosphate |
| NaSCN | Sodium Thiocyanate |
| OCN− | Cyanate Ion |
| OSCN− | Hypothiocyanite Ion |
| RA | Rheumatoid Arthritis |
| ROS | Reactive Oxygen Species |
| S2O32− | Thiosulfate Ion |
| SARS-CoV-2 | Severe Acute Respiratory Syndrome Coronavirus 2 |
| SCN− | Thiocyanate Ion |
| UC | Ulcerative Colitis |
References
- Thomas, E.L. Lactoperoxidase-catalyzed oxidation of thiocyanate: Equilibria between oxidized forms of thiocyanate. Biochemistry 1981, 20, 3273–3280. [Google Scholar] [CrossRef] [PubMed]
- Das, D.; De, P.K.; Banerjee, R.K. Thiocyanate, a plausible physiological electron donor of gastric peroxidase. Biochem. J. 1995, 305 Pt 1, 59–64. [Google Scholar] [CrossRef]
- Fragoso, M.A.; Fernandez, V.; Forteza, R.; Randell, S.H.; Salathe, M.; Conner, G.E. Transcellular thiocyanate transport by human airway epithelia. J. Physiol. 2004, 561, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Schultz, C.P.; Ahmed, M.K.; Dawes, C.; Mantsch, H.H. Thiocyanate levels in human saliva: Quantitation by Fourier transform infrared spectroscopy. Anal. Biochem. 1996, 240, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Minarowski, L.; Sands, D.; Minarowska, A.; Karwowska, A.; Sulewska, A.; Gacko, M.; Chyczewska, E. Thiocyanate concentration in saliva of cystic fibrosis patients. Folia Histochem. Cytobiol. 2008, 46, 245–246. [Google Scholar] [CrossRef][Green Version]
- Madiyal, A.; Ajila, V.; Babu, S.G.; Hegde, S.; Kumari, S.; Madi, M.; Achalli, S.; Alva, P.; Ullal, H. Status of thiocyanate levels in the serum and saliva of non-smokers, ex-smokers and smokers. Afr. Health Sci. 2018, 18, 727–736. [Google Scholar] [CrossRef]
- Leung, A.M.; Lamar, A.; He, X.; Braverman, L.E.; Pearce, E.N. Iodine status and thyroid function of Boston-area vegetarians and vegans. J. Clin. Endocrinol. Metab. 2011, 96, E1303–E1307. [Google Scholar] [CrossRef]
- Han, H.; Kwon, H. Estimated dietary intake of thiocyanate from Brassicaceae family in Korean diet. J. Toxicol. Envrion. Health A 2009, 72, 1380–1387. [Google Scholar] [CrossRef]
- McGregor, D.I. Thiocyanate ion, a hydrolysis product of glucosinolates from rape and mustard seed. Can. J. Plant Sci. 1978, 58, 795–800. [Google Scholar] [CrossRef]
- Buratti, M.; Xaiz, D.; Caravelliand, G.; Colombi, A. Validation of urinary thiocyanate as a biomarker of tobacco smoking. Biomarkers 1997, 2, 81–85. [Google Scholar] [CrossRef]
- Lenney, W.; Gilchrist, F.J. Pseudomonas aeruginosa and cyanide production. Eur. Respir. J. 2011, 37, 482. [Google Scholar] [CrossRef] [PubMed]
- Nagahara, N.; Okazaki, T.; Nishino, T. Cytosolic mercaptopyruvate sulfurtransferase is evolutionarily related to mitochondrial rhodanese. Striking similarity in active site amino acid sequence and the increase in the mercaptopyruvate sulfurtransferase activity of rhodanese by site-directed mutagenesis. J. Biol. Chem. 1995, 270, 16230–16235. [Google Scholar] [CrossRef]
- Wrobel, M.; Jurkowska, H.; Sliwa, L.; Srebro, Z. Sulfurtransferases and cyanide detoxification in mouse liver, kidney, and brain. Toxicol. Mech. Methods 2004, 14, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Weuffen, W.; Franzke, C.; Thurkow, B. The alimentary ingestion, analysis and biological significance of thiocyanate. Nahrung 1984, 28, 341–355. [Google Scholar] [CrossRef]
- Chung, J.; Wood, J.L. Oxidation of Thiocyanate to Cyanide Catalyzed by Hemoglobin. J. Biol. Chem. 1971, 246, 555–560. [Google Scholar] [PubMed]
- Wells, D.G.; Langman, M.J.; Wilson, J. Thiocyanate metabolism in human vitamin B12 deficiency. Br. Med. J. 1972, 4, 588–590. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Broderick, K.E.; Potluri, P.; Zhuang, S.; Scheffler, I.E.; Sharma, V.S.; Pilz, R.B.; Boss, G.R. Cyanide detoxification by the cobalamin precursor cobinamide. Exp. Biol. Med. 2006, 231, 641–649. [Google Scholar] [CrossRef]
- Thomson, E.; Brennan, S.; Senthilmohan, R.; Gangell, C.L.; Chapman, A.L.; Sly, P.D.; Kettle, A.J.; Balding, E.; Berry, L.J.; Carlin, J.B.; et al. Identifying peroxidases and their oxidants in the early pathology of cystic fibrosis. Free Radic. Biol. Med. 2010, 49, 1354–1360. [Google Scholar] [CrossRef]
- Chandler, J.D.; Day, B.J. Biochemical mechanisms and therapeutic potential of pseudohalide thiocyanate in human health. Free Radic. Res. 2015, 49, 695–710. [Google Scholar] [CrossRef]
- Wijkstrom-Frei, C.; El-Chemaly, S.; Ali-Rachedi, R.; Gerson, C.; Cobas, M.A.; Forteza, R.; Salathe, M.; Conner, G.E. Lactoperoxidase and human airway host defense. Am. J. Respir. Cell Mol. Biol. 2003, 29, 206–212. [Google Scholar] [CrossRef]
- Ashby, M. Hypothiocyanite. Adv. Inorg. Chem. 2012, 64, 263–303. [Google Scholar] [CrossRef]
- Fletcher, K.; Honour, A.J.; Rowlands, E.N. Studies on the concentration of radioiodide and thiocyanate by slices of the salivary gland. Biochem. J. 1956, 63, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Tenovuo, J.; Pruitt, K.M.; Thomas, E.L. Peroxidase antimicrobial system of human saliva: Hypothiocyanite levels in resting and stimulated saliva. J. Dent. Res. 1982, 61, 982–985. [Google Scholar] [CrossRef] [PubMed]
- Pedemonte, N.; Caci, E.; Sondo, E.; Caputo, A.; Rhoden, K.; Pfeffer, U.; Di Candia, M.; Bandettini, R.; Ravazzolo, R.; Zegarra-Moran, O.; et al. Thiocyanate transport in resting and IL-4-stimulated human bronchial epithelial cells: Role of pendrin and anion channels. J. Immunol. 2007, 178, 5144–5153. [Google Scholar] [CrossRef] [PubMed]
- Moskwa, P.; Lorentzen, D.; Excoffon, K.J.; Zabner, J.; McCray, P.B., Jr.; Nauseef, W.M.; Dupuy, C.; Banfi, B. A novel host defense system of airways is defective in cystic fibrosis. Am. J. Respir. Crit. Care Med. 2007, 175, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Gould, N.S.; Gauthier, S.; Kariya, C.T.; Min, E.; Huang, J.; Brian, D.J. Hypertonic saline increases lung epithelial lining fluid glutathione and thiocyanate: Two protective CFTR-dependent thiols against oxidative injury. Respir. Res. 2010, 11, 119. [Google Scholar] [CrossRef]
- Pettigrew, A.R.; Fell, G.S. Simplified colorimetric determination of thiocyanate in biological fluids, and its application to investigation of the toxic amblyopias. Clin. Chem. 1972, 18, 996–1000. [Google Scholar] [CrossRef]
- Tenovuo, J.; Makinen, K.K. Concentration of thiocyanate and ionizable iodine in saliva of smokers and nonsmokers. J. Dent. Res. 1976, 55, 661–663. [Google Scholar] [CrossRef]
- Junge, B. Changes in serum thiocyanate concentration on stopping smoking. Br. Med. J. 1985, 291, 22. [Google Scholar] [CrossRef][Green Version]
- Kalburgi, C.V.; Naik, K.L.; Kokatnur, M.V.; Warad, S. Estimation and correlation of salivary thiocyanate levels in healthy and different forms of tobacco users having chronic periodontitis: A cross-sectional biochemical study. Contemp. Clin. Dent. 2014, 5, 182–186. [Google Scholar] [CrossRef]
- Schulz, V.; Bonn, R.; Kindler, J. Kinetics of elimination of thiocyanate in 7 healthy subjects and in 8 subjects with renal failure. Klin. Wochenschr. 1979, 57, 243–247. [Google Scholar] [CrossRef] [PubMed]
- van Haeringen, N.J.; Ensink, F.T.; Glasius, E. The peroxidase-thiocyanate-hydrogenperoxide system in tear fluid and saliva of different species. Exp. Eye Res. 1979, 28, 343–347. [Google Scholar] [CrossRef]
- Lorentzen, D.; Durairaj, L.; Pezzulo, A.A.; Nakano, Y.; Launspach, J.; Stoltz, D.A.; Zamba, G.; McCray, P.B., Jr.; Zabner, J.; Welsh, M.J.; et al. Concentration of the antibacterial precursor thiocyanate in cystic fibrosis airway secretions. Free Radic. Biol. Med. 2011, 50, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
- Wokes, F.; Wedgwood, P.; Wyatt, J. Thiocyanates in milk and other biological fluids. Biochem. J. 1952, 50, xix–xx. [Google Scholar]
- Kirk, A.B.; Dyke, J.V.; Martin, C.F.; Dasgupta, P.K. Temporal patterns in perchlorate, thiocyanate, and iodide excretion in human milk. Environ. Health Perspect. 2007, 115, 182–186. [Google Scholar] [CrossRef]
- Fiedler, T.J.; Davey, C.A.; Fenna, R.E. X-ray crystal structure and characterization of halide-binding sites of human myeloperoxidase at 1.8 A resolution. J. Biol. Chem. 2000, 275, 11964–11971. [Google Scholar] [CrossRef]
- Zeng, J.; Fenna, R.E. X-ray crystal structure of canine myeloperoxidase at 3 Å resolution. J. Mol. Biol. 1992, 226, 185–207. [Google Scholar] [CrossRef]
- Dolphin, D.; Forman, A.; Borg, D.C.; Fajer, J.; Felton, R.H. Compounds I of catalase and horse radish peroxidase: Pi-cation radicals. Proc. Natl. Acad. Sci. USA 1971, 68, 614–618. [Google Scholar] [CrossRef]
- Vlasova, I.I. Peroxidase Activity of Human Hemoproteins: Keeping the Fire under Control. Molecules 2018, 23, 2561. [Google Scholar] [CrossRef]
- Kettle, A.J.; Anderson, R.F.; Hampton, M.B.; Winterbourn, C.C. Reactions of superoxide with myeloperoxidase. Biochemistry 2007, 46, 4888–4897. [Google Scholar] [CrossRef]
- Chandler, J.D.; Day, B.J. Thiocyanate: A potentially useful therapeutic agent with host defense and antioxidant properties. Biochem. Pharmacol. 2012, 84, 1381–1387. [Google Scholar] [CrossRef] [PubMed]
- Baldridge, C.W.; Gerard, R.W. The extra respiration of phagocytosis. Am. J. Physiol. Leg. Content 1932, 103, 235–236. [Google Scholar] [CrossRef]
- Cross, A.R.; Segal, A.W. The NADPH oxidase of professional phagocytes--prototype of the NOX electron transport chain systems. Biochim. Biophys. Acta 2004, 1657, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Morgan, P.E.; Laura, R.P.; Maki, R.A.; Reynolds, W.F.; Davies, M.J. Thiocyanate supplementation decreases atherosclerotic plaque in mice expressing human myeloperoxidase. Free Radic. Res. 2015, 49, 743–749. [Google Scholar] [CrossRef]
- Pattison, D.I.; Davies, M.J.; Hawkins, C.L. Reactions and reactivity of myeloperoxidase-derived oxidants: Differential biological effects of hypochlorous and hypothiocyanous acids. Free Radic. Res. 2012, 46, 975–995. [Google Scholar] [CrossRef]
- Furtmüller, P.G.; Burner, U.; Obinger, C. Reaction of Myeloperoxidase Compound I with Chloride, Bromide, Iodide, and Thiocyanate. Biochemistry 1998, 37, 17923–17930. [Google Scholar] [CrossRef]
- Davies, M.J.; Hawkins, C.L.; Pattison, D.I.; Rees, M.D. Mammalian heme peroxidases: From molecular mechanisms to health implications. Antioxid. Redox Signal. 2008, 10, 1199–1234. [Google Scholar] [CrossRef]
- Pattison, D.I.; Davies, M.J. Absolute rate constants for the reaction of hypochlorous acid with protein side chains and peptide bonds. Chem. Res. Toxicol. 2001, 14, 1453–1464. [Google Scholar] [CrossRef]
- Skaff, O.; Pattison, D.I.; Davies, M.J. Hypothiocyanous acid reactivity with low-molecular-mass and protein thiols: Absolute rate constants and assessment of biological relevance. Biochem. J. 2009, 422, 111–117. [Google Scholar] [CrossRef]
- Gorman, W.F.; Messinger, E.; Herman, M. Toxicity of thiocyanates used in treatment of hypertension. Ann. Intern. Med. 1949, 30, 1054–1059. [Google Scholar] [CrossRef]
- Ruddell, W.S.; Bone, E.S.; Hill, M.J.; Blendis, L.M.; Walters, C.L. Gastric-juice nitrite. A risk factor for cancer in the hypochlorhydric stomach? Lancet 1976, 2, 1037–1039. [Google Scholar] [CrossRef]
- Heliovaara, M.; Karvonen, M.J.; Punsar, S.; Rautanen, Y.; Haapakoski, J. Serum thiocyanate concentration and cigarette smoking in relation to overall mortality and to deaths from coronary heart disease and lung cancer. J. Chronic. Dis. 1981, 34, 305–311. [Google Scholar] [CrossRef]
- Lijinsky, W.; Kovatch, R.M. Chronic toxicity tests of sodium thiocyanate with sodium nitrite in F344 rats. Toxicol. Ind. Health 1989, 5, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Shiue, I. Urinary thiocyanate concentrations are associated with adult cancer and lung problems: US NHANES, 2009–2012. Env. Sci. Pollut. Res. Int. 2015, 22, 5952–5960. [Google Scholar] [CrossRef]
- World Health Organization. Global Health Estimates 2016: Disease Burden by Cause, Age, Sex, by Country and by Region, 2000–2016; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Pasceri, V.; Cheng, J.S.; Willerson, J.T.; Yeh, E.T. Modulation of C-reactive protein-mediated monocyte chemoattractant protein-1 induction in human endothelial cells by anti-atherosclerosis drugs. Circulation 2001, 103, 2531–2534. [Google Scholar] [CrossRef]
- Ronald, J.A.; Chen, J.W.; Chen, Y.; Hamilton, A.M.; Rodriguez, E.; Reynolds, F.; Hegele, R.A.; Rogers, K.A.; Querol, M.; Bogdanov, A.; et al. Enzyme-sensitive magnetic resonance imaging targeting myeloperoxidase identifies active inflammation in experimental rabbit atherosclerotic plaques. Circulation 2009, 120, 592–599. [Google Scholar] [CrossRef]
- Heslop, C.L.; Frohlich, J.J.; Hill, J.S. Myeloperoxidase and C-reactive protein have combined utility for long-term prediction of cardiovascular mortality after coronary angiography. J. Am. Coll. Cardiol. 2010, 55, 1102–1109. [Google Scholar] [CrossRef]
- Exner, M.; Hermann, M.; Hofbauer, R.; Hartmann, B.; Kapiotis, S.; Gmeiner, B. Thiocyanate catalyzes myeloperoxidase-initiated lipid oxidation in LDL. Free Radic. Biol. Med. 2004, 37, 146–155. [Google Scholar] [CrossRef]
- Hadfield, K.A.; Pattison, D.I.; Brown, B.E.; Hou, L.; Rye, K.A.; Davies, M.J.; Hawkins, C.L. Myeloperoxidase-derived oxidants modify apolipoprotein A-I and generate dysfunctional high-density lipoproteins: Comparison of hypothiocyanous acid (HOSCN) with hypochlorous acid (HOCl). Biochem. J. 2013, 449, 531–542. [Google Scholar] [CrossRef]
- Abdo, A.I.; Rayner, B.S.; van Reyk, D.M.; Hawkins, C.L. Low-density lipoprotein modified by myeloperoxidase oxidants induces endothelial dysfunction. Redox. Biol. 2017, 13, 623–632. [Google Scholar] [CrossRef]
- Marsche, G.; Hammer, A.; Oskolkova, O.; Kozarsky, K.F.; Sattler, W.; Malle, E. Hypochlorite-modified high density lipoprotein, a high affinity ligand to scavenger receptor class B, type I, impairs high density lipoprotein-dependent selective lipid uptake and reverse cholesterol transport. J. Biol. Chem. 2002, 277, 32172–32179. [Google Scholar] [CrossRef] [PubMed]
- Marsche, G.; Zimmermann, R.; Horiuchi, S.; Tandon, N.N.; Sattler, W.; Malle, E. Class B scavenger receptors CD36 and SR-BI are receptors for hypochlorite-modified low density lipoprotein. J. Biol. Chem. 2003, 278, 47562–47570. [Google Scholar] [CrossRef] [PubMed]
- Marsche, G.; Heller, R.; Fauler, G.; Kovacevic, A.; Nuszkowski, A.; Graier, W.; Sattler, W.; Malle, E. 2-Chlorohexadecanal Derived From Hypochlorite-Modified High-Density Lipoprotein–Associated Plasmalogen Is a Natural Inhibitor of Endothelial Nitric Oxide Biosynthesis. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 2302–2306. [Google Scholar] [CrossRef] [PubMed]
- Vita, J.A.; Brennan, M.L.; Gokce, N.; Mann, S.A.; Goormastic, M.; Shishehbor, M.H.; Penn, M.S.; Keaney, J.F., Jr.; Hazen, S.L. Serum myeloperoxidase levels independently predict endothelial dysfunction in humans. Circulation 2004, 110, 1134–1139. [Google Scholar] [CrossRef] [PubMed]
- Tiyerili, V.; Camara, B.; Becher, M.U.; Schrickel, J.W.; Lütjohann, D.; Mollenhauer, M.; Baldus, S.; Nickenig, G.; Andrié, R.P. Neutrophil-derived myeloperoxidase promotes atherogenesis and neointima formation in mice. Int. J. Cardiol. 2016, 204, 29–36. [Google Scholar] [CrossRef]
- Roth Flach, R.J.; Su, C.; Bollinger, E.; Cortes, C.; Robertson, A.W.; Opsahl, A.C.; Coskran, T.M.; Maresca, K.P.; Keliher, E.J.; Yates, P.D.; et al. Myeloperoxidase inhibition in mice alters atherosclerotic lesion composition. PLoS ONE 2019, 14, e0214150. [Google Scholar] [CrossRef]
- Brennan, M.L.; Anderson, M.M.; Shih, D.M.; Qu, X.D.; Wang, X.; Mehta, A.C.; Lim, L.L.; Shi, W.; Hazen, S.L.; Jacob, J.S.; et al. Increased atherosclerosis in myeloperoxidase-deficient mice. J. Clin. Investig. 2001, 107, 419–430. [Google Scholar] [CrossRef]
- Zoellner, H. Dental infection and vascular disease. Semin. Thromb. Hemost. 2011, 37, 181–192. [Google Scholar] [CrossRef]
- Nedoboy, P.E.; Morgan, P.E.; Mocatta, T.J.; Richards, A.M.; Winterbourn, C.C.; Davies, M.J. High plasma thiocyanate levels are associated with enhanced myeloperoxidase-induced thiol oxidation and long-term survival in subjects following a first myocardial infarction. Free Radic. Res. 2014, 48, 1256–1266. [Google Scholar] [CrossRef]
- Wang, Z.; Nicholls, S.J.; Rodriguez, E.R.; Kummu, O.; Horkko, S.; Barnard, J.; Reynolds, W.F.; Topol, E.J.; DiDonato, J.A.; Hazen, S.L. Protein carbamylation links inflammation, smoking, uremia and atherogenesis. Nat. Med. 2007, 13, 1176–1184. [Google Scholar] [CrossRef]
- Chandler, J.D.; Nichols, D.P.; Nick, J.A.; Hondal, R.J.; Day, B.J. Selective metabolism of hypothiocyanous acid by mammalian thioredoxin reductase promotes lung innate immunity and antioxidant defense. J. Biol. Chem. 2013, 288, 18421–18428. [Google Scholar] [CrossRef] [PubMed]
- Zietzer, A.; Niepmann, S.T.; Camara, B.; Lenart, M.A.; Jansen, F.; Becher, M.U.; Andrie, R.; Nickenig, G.; Tiyerili, V. Sodium thiocyanate treatment attenuates atherosclerotic plaque formation and improves endothelial regeneration in mice. PLoS ONE 2019, 14, e0214476. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Szep, S.; Lu, Z. The antioxidant role of thiocyanate in the pathogenesis of cystic fibrosis and other inflammation-related diseases. Proc. Natl. Acad. Sci. USA 2009, 106, 20515–20519. [Google Scholar] [CrossRef] [PubMed]
- Kantar, A.; Oggiano, N.; Giorgi, P.L.; Braga, P.C.; Fiorini, R. Polymorphonuclear leukocyte-generated oxygen metabolites decrease beat frequency of human respiratory cilia. Lung 1994, 172, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Worlitzsch, D.; Herberth, G.; Ulrich, M.; Doring, G. Catalase, myeloperoxidase and hydrogen peroxide in cystic fibrosis. Eur. Respir. J. 1998, 11, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Van Der Vliet, A.; Nguyen, M.N.; Shigenaga, M.K.; Eiserich, J.P.; Marelich, G.P.; Cross, C.E. Myeloperoxidase and protein oxidation in cystic fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2000, 279, L537–L546. [Google Scholar] [CrossRef]
- Wanner, A.; Salathe, M.; O’Riordan, T.G. Mucociliary clearance in the airways. Am. J. Respir. Crit. Care Med. 1996, 154, 1868–1902. [Google Scholar] [CrossRef]
- Riordan, J.R.; Rommens, J.M.; Kerem, B.; Alon, N.; Rozmahel, R.; Grzelczak, Z.; Zielenski, J.; Lok, S.; Plavsic, N.; Chou, J.L.; et al. Identification of the cystic fibrosis gene: Cloning and characterization of complementary DNA. Science 1989, 245, 1066. [Google Scholar] [CrossRef]
- Linsdell, P.; Tabcharani, J.A.; Rommens, J.M.; Hou, Y.X.; Chang, X.B.; Tsui, L.C.; Riordan, J.R.; Hanrahan, J.W. Permeability of wild-type and mutant cystic fibrosis transmembrane conductance regulator chloride channels to polyatomic anions. J. Gen. Physiol. 1997, 110, 355–364. [Google Scholar] [CrossRef]
- Frizzell, R.A.; Hanrahan, J.W. Physiology of epithelial chloride and fluid secretion. Cold Spring Harb. Perspect. Med. 2012, 2, a009563. [Google Scholar] [CrossRef]
- Conner, G.E.; Wijkstrom-Frei, C.; Randell, S.H.; Fernandez, V.E.; Salathe, M. The lactoperoxidase system links anion transport to host defense in cystic fibrosis. FEBS Lett. 2007, 581, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Elkins, M.R.; Robinson, M.; Rose, B.R.; Harbour, C.; Moriarty, C.P.; Marks, G.B.; Belousova, E.G.; Xuan, W.; Bye, P.T.P. A Controlled Trial of Long-Term Inhaled Hypertonic Saline in Patients with Cystic Fibrosis. N. Engl. J. Med. 2006, 354, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Konstan, M.W. Therapies aimed at airway inflammation in cystic fibrosis. Clin. Chest Med. 1998, 19, 505–513. [Google Scholar] [CrossRef]
- Chandler, J.D.; Min, E.; Huang, J.; McElroy, C.S.; Dickerhof, N.; Mocatta, T.; Fletcher, A.A.; Evans, C.M.; Liang, L.; Patel, M.; et al. Antiinflammatory and Antimicrobial Effects of Thiocyanate in a Cystic Fibrosis Mouse Model. Am. J. Respir. Cell Mol. Biol. 2015, 53, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Cegolon, L.; Salata, C.; Piccoli, E.; Juarez, V.; Palu, G.; Mastrangelo, G.; Calistri, A. In vitro antiviral activity of hypothiocyanite against A/H1N1/2009 pandemic influenza virus. Int. J. Hyg. Environ. Health 2014, 217, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Gingerich, A.; Pang, L.; Hanson, J.; Dlugolenski, D.; Streich, R.; Lafontaine, E.R.; Nagy, T.; Tripp, R.A.; Rada, B. Hypothiocyanite produced by human and rat respiratory epithelial cells inactivates extracellular H1N2 influenza A virus. Inflamm. Res. 2016, 65, 71–80. [Google Scholar] [CrossRef]
- Patel, U.; Gingerich, A.; Widman, L.; Sarr, D.; Tripp, R.A.; Rada, B. Susceptibility of influenza viruses to hypothiocyanite and hypoiodite produced by lactoperoxidase in a cell-free system. PLoS ONE 2018, 13, e0199167. [Google Scholar] [CrossRef]
- Kitchens, G.G. Relationship of environmental tobacco smoke to otitis media in young children. Laryngoscope 1995, 105, 1–13. [Google Scholar] [CrossRef]
- Jones, L.L.; Hassanien, A.; Cook, D.G.; Britton, J.; Leonardi-Bee, J. Parental Smoking and the Risk of Middle Ear Disease in Children: A Systematic Review and Meta-analysis. Arch. Pediatrics Adolesc. Med. 2012, 166, 18–27. [Google Scholar] [CrossRef]
- Yilmaz, G.; Caylan, N.D.; Karacan, C.D. Effects of Active and Passive Smoking on Ear Infections. Curr. Infect. Dis. Rep. 2012, 14, 166–174. [Google Scholar] [CrossRef]
- Feldman, C.; Anderson, R. New insights into pneumococcal disease. Respirology 2009, 14, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Bates, M.N.; Khalakdina, A.; Pai, M.; Chang, L.; Lessa, F.; Smith, K.R. Risk of tuberculosis from exposure to tobacco smoke: A systematic review and meta-analysis. Arch. Intern. Med. 2007, 167, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.H.; Ezzati, M.; Murray, M. Tobacco smoke, indoor air pollution and tuberculosis: A systematic review and meta-analysis. PLoS Med. 2007, 4, e20. [Google Scholar] [CrossRef] [PubMed]
- Slama, K.; Chiang, C.Y.; Enarson, D.A.; Hassmiller, K.; Fanning, A.; Gupta, P.; Ray, C. Tobacco and tuberculosis: A qualitative systematic review and meta-analysis. Int. J. Tuberc. Lung Dis. 2007, 11, 1049–1061. [Google Scholar] [PubMed]
- Gajalakshmi, V.; Peto, R.; Kanaka, T.S.; Jha, P. Smoking and mortality from tuberculosis and other diseases in India: Retrospective study of 43000 adult male deaths and 35000 controls. Lancet 2003, 362, 507–515. [Google Scholar] [CrossRef]
- Wang, J.; Shen, H. Review of cigarette smoking and tuberculosis in China: Intervention is needed for smoking cessation among tuberculosis patients. BMC Public Health 2009, 9, 292. [Google Scholar] [CrossRef]
- Brunet, L.; Pai, M.; Davids, V.; Ling, D.; Paradis, G.; Lenders, L.; Meldau, R.; van Zyl Smit, R.; Calligaro, G.; Allwood, B.; et al. High prevalence of smoking among patients with suspected tuberculosis in South Africa. Eur. Respir. J. 2011, 38, 139–146. [Google Scholar] [CrossRef]
- Bonacci, R.A.; Cruz-Hervert, L.P.; García-García, L.; Reynales-Shigematsu, L.M.; Ferreyra-Reyes, L.; Bobadilla-del-Valle, M.; Canizales-Quintero, S.; Ferreira-Guerrero, E.; Báez-Saldaña, R.; Téllez-Vázquez, N.; et al. Impact of cigarette smoking on rates and clinical prognosis of pulmonary tuberculosis in Southern Mexico. J. Infect. 2013, 66, 303–312. [Google Scholar] [CrossRef]
- Sanz Herrero, F.; Blanquer Olivas, J. Microbiology and risk factors for community-acquired pneumonia. Semin. Respir. Crit. Care Med. 2012, 33, 220–231. [Google Scholar] [CrossRef]
- Nuorti, J.P.; Butler, J.C.; Farley, M.M.; Harrison, L.H.; McGeer, A.; Kolczak, M.S.; Breiman, R.F. Cigarette Smoking and Invasive Pneumococcal Disease. N. Engl. J. Med. 2000, 342, 681–689. [Google Scholar] [CrossRef]
- Mathe, G.; Gouveia, J.; Hercend, T.; Gros, F.; Dorval, T.; Hazon, J.; Misset, J.L.; Schwarzenberg, L.; Ribaud, P.; Lemaigre, G.; et al. Correlation between precancerous bronchial metaplasia and cigarette consumption, and preliminary results of retinoid treatment. Cancer Detect. Prev. 1982, 5, 461–466. [Google Scholar] [PubMed]
- Agius, A.M.; Wake, M.; Pahor, A.L.; Smallman, L.A. Smoking and middle ear ciliary beat frequency in otitis media with effusion. Acta Oto-Laryngol. 1995, 115, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Mullen, J.B.; Wright, J.L.; Wiggs, B.R.; Paré, P.D.; Hogg, J.C. Structure of central airways in current smokers and ex-smokers with and without mucus hypersecretion: Relationship to lung function. Thorax 1987, 42, 843–848. [Google Scholar] [CrossRef] [PubMed]
- Cecchi, I.; Arias de la Rosa, I.; Menegatti, E.; Roccatello, D.; Collantes-Estevez, E.; Lopez-Pedrera, C.; Barbarroja, N. Neutrophils: Novel key players in Rheumatoid Arthritis. Current and future therapeutic targets. Autoimmun. Rev. 2018, 17, 1138–1149. [Google Scholar] [CrossRef]
- Wipke, B.T.; Allen, P.M. Essential role of neutrophils in the initiation and progression of a murine model of rheumatoid arthritis. J. Immunol. 2001, 167, 1601–1608. [Google Scholar] [CrossRef]
- Tanaka, D.; Kagari, T.; Doi, H.; Shimozato, T. Essential role of neutrophils in anti-type II collagen antibody and lipopolysaccharide-induced arthritis. Immunology 2006, 119, 195–202. [Google Scholar] [CrossRef]
- Eyles, J.L.; Hickey, M.J.; Norman, M.U.; Croker, B.A.; Roberts, A.W.; Drake, S.F.; James, W.G.; Metcalf, D.; Campbell, I.K.; Wicks, I.P. A key role for G-CSF-induced neutrophil production and trafficking during inflammatory arthritis. Blood 2008, 112, 5193–5201. [Google Scholar] [CrossRef]
- Odobasic, D.; Yang, Y.; Muljadi, R.C.; O’Sullivan, K.M.; Kao, W.; Smith, M.; Morand, E.F.; Holdsworth, S.R. Endogenous myeloperoxidase is a mediator of joint inflammation and damage in experimental arthritis. Arthritis Rheumatol 2014, 66, 907–917. [Google Scholar] [CrossRef]
- Nzeusseu Toukap, A.; Delporte, C.; Noyon, C.; Franck, T.; Rousseau, A.; Serteyn, D.; Raes, M.; Vanhaeverbeek, M.; Moguilevsky, N.; Neve, J.; et al. Myeloperoxidase and its products in synovial fluid of patients with treated or untreated rheumatoid arthritis. Free Radic. Res. 2014, 48, 461–465. [Google Scholar] [CrossRef]
- Sur Chowdhury, C.; Giaglis, S.; Walker, U.A.; Buser, A.; Hahn, S.; Hasler, P. Enhanced neutrophil extracellular trap generation in rheumatoid arthritis: Analysis of underlying signal transduction pathways and potential diagnostic utility. Arthritis Res. 2014, 16, R122. [Google Scholar] [CrossRef]
- Itoh, Y. Metalloproteinases in Rheumatoid Arthritis: Potential Therapeutic Targets to Improve Current Therapies. Prog. Mol. Biol. Transl. Sci. 2017, 148, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Kandasamy, A.D.; Chow, A.K.; Ali, M.A.; Schulz, R. Matrix metalloproteinase-2 and myocardial oxidative stress injury: Beyond the matrix. Cardiovasc. Res. 2010, 85, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Kassim, S.Y.; Parks, W.C.; Heinecke, J.W. Hypochlorous acid oxygenates the cysteine switch domain of pro-matrilysin (MMP-7). A mechanism for matrix metalloproteinase activation and atherosclerotic plaque rupture by myeloperoxidase. J. Biol. Chem. 2001, 276, 41279–41287. [Google Scholar] [CrossRef] [PubMed]
- Nagy, P.; Jameson, G.N.; Winterbourn, C.C. Kinetics and mechanisms of the reaction of hypothiocyanous acid with 5-thio-2-nitrobenzoic acid and reduced glutathione. Chem. Res. Toxicol. 2009, 22, 1833–1840. [Google Scholar] [CrossRef] [PubMed]
- Costenbader, K.H.; Feskanich, D.; Mandl, L.A.; Karlson, E.W. Smoking intensity, duration, and cessation, and the risk of rheumatoid arthritis in women. Am. J. Med. 2006, 119, 503.e1–503.e9. [Google Scholar] [CrossRef]
- Whitehouse, M.W.; Jones, M. Pro-inflammatory activity in rats of thiocyanate, a metabolite of the hydrocyanic acid inhaled from tobacco smoke. Inflamm. Res. 2009, 58, 693–704. [Google Scholar] [CrossRef]
- de Brito Rocha, S.; Baldo, D.C.; Andrade, L.E.C. Clinical and pathophysiologic relevance of autoantibodies in rheumatoid arthritis. Adv. Rheumatol. 2019, 59, 2. [Google Scholar] [CrossRef]
- Shi, J.; van Veelen, P.A.; Mahler, M.; Janssen, G.M.; Drijfhout, J.W.; Huizinga, T.W.; Toes, R.E.; Trouw, L.A. Carbamylation and antibodies against carbamylated proteins in autoimmunity and other pathologies. Autoimmun. Rev. 2014, 13, 225–230. [Google Scholar] [CrossRef]
- Delporte, C.; Zouaoui Boudjeltia, K.; Furtmuller, P.G.; Maki, R.A.; Dieu, M.; Noyon, C.; Soudi, M.; Dufour, D.; Coremans, C.; Nuyens, V.; et al. Myeloperoxidase-catalyzed oxidation of cyanide to cyanate: A potential carbamylation route involved in the formation of atherosclerotic plaques? J. Biol. Chem. 2018, 293, 6374–6386. [Google Scholar] [CrossRef]
- Verheul, M.K.; Fearon, U.; Trouw, L.A.; Veale, D.J. Biomarkers for rheumatoid and psoriatic arthritis. Clin. Immunol. 2015, 161, 2–10. [Google Scholar] [CrossRef]
- Shi, J.; Knevel, R.; Suwannalai, P.; van der Linden, M.P.; Janssen, G.M.; van Veelen, P.A.; Levarht, N.E.; van der Helm-van Mil, A.H.; Cerami, A.; Huizinga, T.W.; et al. Autoantibodies recognizing carbamylated proteins are present in sera of patients with rheumatoid arthritis and predict joint damage. Proc. Natl. Acad. Sci. USA 2011, 108, 17372–17377. [Google Scholar] [CrossRef] [PubMed]
- Nishida, A.; Inoue, R.; Inatomi, O.; Bamba, S.; Naito, Y.; Andoh, A. Gut microbiota in the pathogenesis of inflammatory bowel disease. Clin. J. Gastroenterol. 2018, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Marchal-Bressenot, A.; Salleron, J.; Boulagnon-Rombi, C.; Bastien, C.; Cahn, V.; Cadiot, G.; Diebold, M.D.; Danese, S.; Reinisch, W.; Schreiber, S.; et al. Development and validation of the Nancy histological index for UC. Gut 2017, 66, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Buell, M.G.; Berin, M.C. Neutrophil-independence of the initiation of colonic injury. Comparison of results from three models of experimental colitis in the rat. Dig. Dis. Sci. 1994, 39, 2575–2588. [Google Scholar] [CrossRef] [PubMed]
- Natsui, M.; Kawasaki, K.; Takizawa, H.; Hayashi, S.I.; Matsuda, Y.; Sugimura, K.; Seki, K.; Narisawa, R.; Sendo, F.; Asakura, H. Selective depletion of neutrophils by a monoclonal antibody, RP-3, suppresses dextran sulphate sodium-induced colitis in rats. J. Gastroenterol. Hepatol. 1997, 12, 801–808. [Google Scholar] [CrossRef]
- Chami, B.; San Gabriel, P.T.; Kum-Jew, S.; Wang, X.; Dickerhof, N.; Dennis, J.M.; Witting, P.K. The nitroxide 4-methoxy-tempo inhibits the pathogenesis of dextran sodium sulfate-stimulated experimental colitis. Redox. Biol. 2020, 28, 101333. [Google Scholar] [CrossRef]
- Saiki, T.; Mitsuyama, K.; Toyonaga, A.; Ishida, H.; Tanikawa, K. Detection of pro- and anti-inflammatory cytokines in stools of patients with inflammatory bowel disease. Scand. J. Gastroenterol. 1998, 33, 616–622. [Google Scholar] [CrossRef]
- Hansberry, D.R.; Shah, K.; Agarwal, P.; Agarwal, N. Fecal Myeloperoxidase as a Biomarker for Inflammatory Bowel Disease. Cureus 2017, 9, e1004. [Google Scholar] [CrossRef]
- Mancini, S.; Mariani, F.; Sena, P.; Benincasa, M.; Roncucci, L. Myeloperoxidase expression in human colonic mucosa is related to systemic oxidative balance in healthy subjects. Redox Rep. 2017, 22, 399–407. [Google Scholar] [CrossRef]
- Chami, B.; Martin, N.J.J.; Dennis, J.M.; Witting, P.K. Myeloperoxidase in the inflamed colon: A novel target for treating inflammatory bowel disease. Arch. Biochem. Biophys. 2018, 645, 61–71. [Google Scholar] [CrossRef]
- Knutson, C.G.; Mangerich, A.; Zeng, Y.; Raczynski, A.R.; Liberman, R.G.; Kang, P.; Ye, W.; Prestwich, E.G.; Lu, K.; Wishnok, J.S.; et al. Chemical and cytokine features of innate immunity characterize serum and tissue profiles in inflammatory bowel disease. Proc. Natl. Acad. Sci. USA 2013, 110, E2332–E2341. [Google Scholar] [CrossRef] [PubMed]
- Lakatos, P.L.; Lakatos, L. Risk for colorectal cancer in ulcerative colitis: Changes, causes and management strategies. World J. Gastroenterol. 2008, 14, 3937–3947. [Google Scholar] [CrossRef] [PubMed]
- Morgan, P.E.; Pattison, D.I.; Talib, J.; Summers, F.A.; Harmer, J.A.; Celermajer, D.S.; Hawkins, C.L.; Davies, M.J. High plasma thiocyanate levels in smokers are a key determinant of thiol oxidation induced by myeloperoxidase. Free Radic. Biol. Med. 2011, 51, 1815–1822. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Burton, T.; Rayner, B.S.; San Gabriel, P.T.; Shi, H.; El Kazzi, M.; Wang, X.; Dennis, J.M.; Ahmad, G.; Schroder, A.L.; et al. The role of sodium thiocyanate supplementation during dextran sodium sulphate-stimulated experimental colitis. Arch. Biochem. Biophys. 2020, 692, 108490. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).